Abstract
People can be exposed to zinc oxide (ZnO) by inhalation of consumer products or during industrial processes. Zinc oxide nanoparticle (NP) exposure can induce acute inhalation toxicity. The toxicological mechanisms underlying the acute effects on the lungs have long focused on the phagolysosomal dissolution of ZnO NPs in macrophages followed by the release of free Zn2+ ions. However, we postulate an alternative mechanism based on the direct interaction of ZnO NPs with the lung surfactant (LS) layer covering the inside of the alveoli. Therefore, we tested the effect of ZnO NPs and Zn2+ ions on the function of LS in vitro using the constrained drop surfactometer. We found that the ZnO NPs inhibited the LS function, whereas Zn2+ ions did not. To examine the role of lung macrophages in the acute toxicity of inhaled ZnO NPs, mice were treated with Clodrosome, a drug that depletes alveolar macrophages, or Encapsome, the empty carrier of the drug. After macrophage depletion, the mice were exposed to an aerosol of ZnO NPs in whole body plethysmographs recording breathing patterns continuously. Mice in both groups developed shallow breathing (reduced tidal volume) shortly after the onset of exposure to ZnO NPs. This suggests a macrophage-independent mechanism of induction. This study shows that acute inhalation toxicity is caused by ZnO NP interaction with LS, independently of NP dissolution in macrophages.
Keywords
Introduction
The amount of zinc in the human body is approximately 1.4 to 2.3 g, 1,2 making zinc the second most abundant transition metal after iron. Most of the zinc is bound to proteins such as metallothioneins, and zinc is a cofactor in more than 300 enzymes. 3 However, the concentration of free Zn2+ ions in the cytosol of healthy cells is very low. 4
Zinc oxide (ZnO) nanoparticles (NPs) are used in several industrial applications, including paint and sunscreen. During production and handling, occupational exposure to inhalable aerosols of ZnO is likely. Also, ultrafine ZnO particles are produced during welding of hot-dip galvanized steel and inhalation of these particles may give rise to metal fume fever, which is characterized by fever, cough, and flu-like symptoms. 5,6
Several studies have been published on ZnO toxicity both in human aerosol challenge experiments as well as in animal studies. 7 -9 The mechanisms underlying the toxicity of ZnO NPs are, however, not yet fully elucidated. 10 It has been shown that toxicity in part is driven by Zn2+ ions, which are released when ZnO NPs are dissolved in the phagolysosome of the macrophages. 10,11 This causes lysosomal damage, Ca2+ influx, mitochondrial perturbation, generation of reactive oxygen species, pro-inflammatory responses, and cell death. 12 In addition to inflammatory effects, inhalation of ZnO NPs was also shown to induce an acute and concentration-dependent decrease in the tidal volume in mice. 13 The response onset occurred within the first 5 minutes of exposure, which makes it unlikely that the effects were driven by a significant amount of ZnO NPs dissolving intracellularly and being released as Zn2+ ions. This suggests that other mechanisms of acute inhalation toxicity may exist in parallel with the macrophage-dependent dissolution of ZnO NPs.
The present study investigates the possible role of lung surfactant (LS) in the acute respiratory effects of ZnO NPs. Lung surfactant is a thin layer of liquid that covers the respiratory parts of the lungs and is composed of phospholipids, neutral lipids, and proteins. 14 Its main function is to regulate the surface tension of the air–liquid interface during breathing and to reduce it to near-zero values at the end of expiration. The inhibition of LS function leads to a high surface tension when the lung volume is at its lowest and can result in the alveoli collapsing, making breathing difficult. The atelectatic alveoli can be forced open by the inhalation of air, but this creates shear forces on the lung epithelium and disruption of the air–liquid barrier. Disruption of LS function has been linked to acute inhalation toxicity, for example, for impregnation products where disruption of LS function correlates with an irreversible decrease in tidal volume of the exposed mice. 15 -18
In addition, to investigate whether the acute respiratory effects of ZnO NPs were driven by macrophages, liposomes containing dichloromethylene-bisphosphonate (clodronate) were administered to mice prior to inhalation exposure of ZnO NPs. This methodology uses the naturally occurring phagocytosis mechanism for liposomes to allow for a selective depletion of the macrophages. 19
Material and Methods
Lung Surfactant Bioassay
The effect of aerosolized ZnO NPs on LS function in vitro was assessed using an optimized version of the constrained drop surfactometer (BioSurface Instruments) which measures surface tension of LS under dynamic conditions. The constrained drop surfactometer setup was altered to allow realistic simulation of the interaction between inhaled particles and LS with continuous aerosol exposure and dose measurement. 20 The minimum surface tension (mN/m) after compression was used as a measure of the functionality of the LS. The mean (with 95% confidence limits) minimum surface tension after compression for each dose was calculated from 5 consecutive compression–expansion cycles in 5 independent experiments. A minimum surface tension higher than 10 mN/m is a critical point in vivo. If the minimum surface tension of the LS reaches this value, atelectasis will start to occur. 21 Therefore, particles increasing the minimum surface tension of LS above 10 mN/m (both mean and lower 95% confidence limit above 10 mN/m) were considered as inhibitory to LS function.
A drop of LS (Curosurf, 10 µL of 2.5 mg/mL, mean surface area of 0.254 cm2) was placed on a sharp-edged pedestal and cycled continuously (30 cycles/min, maximum compression of 47.0% ± 2.8%) throughout the experiment by introducing and removing liquid through the base of the pedestal connected to a computer-controlled syringe. Due to evaporation, the volume of the drop decreases over time. Short breaks during cycling allowed refilling the drop to maintain its volume. Before exposure, the ZnO NPs were ground manually to reduce the number of large aggregates and filled into a commercially available dry-powder inhaler (Turbuhaler) that was emptied and carefully cleaned with pressurized air prior to use. The air was heated to 37 °C before reaching the aerosol device. The aerosol of ZnO NPs was led into the chamber by a Venturi tube connected to the aerosol device and sucked out of the chamber through the hollow bottom with an airflow of 5 L/min. This allowed continuous air exchange in the chamber. To monitor the dose delivered to the LS in real-time, a quartz crystal microbalance (QCM, Vitrocell) custom-made by Vitrocell to fit into the exposure chamber was placed close to the LS drop. Micronized lactose (negative control, Lactohale 300, a gift from DFE Pharma) was aerosolized under the same conditions. The aerodynamic equivalent diameter of the ZnO particles in the aerosol was measured using an Electrical Low Pressure Impactor (Dekati Ltd) in 14 size bins in the size range 6 nm to 10 µm.
In addition to the study of ZnO NPs, the effect of Zn2+ ions was studied in a separate experiment. A solution of zinc sulfate monohydrate (ZnSO4 × H2O at 1.5 mol/L, Sigma-Aldrich) was led from a glass syringe into a Pitt no.1 jet nebulizer 22 by means of an infusion pump (New England Medical Instruments Inc).
The effects of ZnO NPs and Zn2+ ions were measured at predetermined doses (ng/cm2) throughout the dose–response experiment. The dosing rate was kept constant between experiments with ZnO NPs and lactose (166 ng/cm2/min). The profile of the LS drop was recorded throughout the exposure with a camera and the images were analyzed with the axisymmetric drop shape analysis software (ADSA 23 ) to quantify the surface tension of LS after compression.
Animals and Liposome Treatment
Thirty inbred female BALB/cJ mice aged 6 to 7 weeks were purchased from Taconic M&B, Ry, Denmark. Upon arrival, the animals were randomly divided into 6 cages each housing 5 animals. Blinding was not performed. The cages were made of polypropylene (380 × 220 × 150 mm) furnished with bedding material (Lignocel S8), small aspen gnaw sticks (Tapvei), plastic nesting houses (mouse house [80-ACRE011, Techniplast]), and nesting material (Enviro Dri, Lillico Biotechnology). The photoperiod was from 6:00 to 18:00, and the temperature and mean relative humidity in the animal room were 19 °C to 22 °C and 43% ± 8%, respectively (mean ± standard deviation [SD]). The experiments were performed in the light period. Food (Altromin no. 1324, Altromin) and tap water was available ad libitum. At the time of exposure, the mice had a bodyweight of 20.8 ± 2.4 g (mean ± SD). Treatment of the animals followed procedures approved by The Animal Experiment Inspectorate, Denmark (2011/561-1990) and by the local animal ethics committee prior to the project.
Dichloromethylene diphosphonate (clodronate) was administered in the form of liposomes (Clodrosome) purchased from Encapsula Nanosciences LLC. Clodronate has previously been used to deplete the number of macrophages in the lungs. 24,25 Clodrosome was suspended in phosphate-buffered saline (PBS), 1 mL of suspension contained 5 mg clodronate, 18.8 mg phosphatidylcholine, and 4.2 mg cholesterol. The mice were anesthetized by inhalation of 4% isoflurane (Piramal), then each mouse in the Clodrosome group (n = 10) received 100 µL of Clodrosome suspension intratracheally and 150 µL intravenously through the tail vein. The mice in the Encapsome (Encapsula Nanoscience LLC) group (n = 10) received an equivalent dose of the liposomes without clodronate intratracheally (100 µL) and intravenously (150 µL). Furthermore, 10 mice were instilled intratracheally with 100 µL saline and intravenously with 150 µL saline (control group).
Macrophage Depletion Characterization
Five mice from each of the Clodrosome, Encapsome, and saline control group were killed by subcutaneous injection of Hypnorm (fentanyl/fluanisone, Janssen Pharma) and Dormicum (midazolam, Roche) 48 hours after liposome treatment. Bronchoalveolar lavage fluid was recovered by flushing the lungs of each mouse 4 times with 0.8 mL of physiological saline using a tracheal cannula. Here the saline-treated mice were used as a control for how the liposome exposure affected the lungs. The bronchoalveolar lavage fluids from each animal were pooled and centrifuged (500 g, 10 minutes, 4 °C). Total protein concentration was measured in the supernatant using the BCA Protein Assay Kit (Pierce) according to the manufacturer’s instructions. The cell pellet was resuspended in 100 µL PBS buffer containing heparin (20 IE/mL) and bovine serum albumin (0.003%, Sigma Aldrich). The total number of cells was determined using a NucleoCounter NC-200 haemocytometer with Via1-Cassette (Chemometec). For differential cell counts, cytospin preparations were made (Cytofuge 2, StatSpin Inc; 1,000 g, 4 minutes, room temperature). Slides were stained with May-Grünwald/Giemsa and the same technician counted all slides. Two hundred cells were counted and differentiated into neutrophils, eosinophils, epithelial cells, lymphocytes, and macrophages by morphology.
Inhalation Bioassay and Aerosol Characterization
Anesthesia was not used during the inhalation experiments because the bioassay depends on the animals being fully awake with uncompromised breathing. The breathing and general well-being of the animals were continuously monitored throughout the experiment.
Forty-eight hours after the administration of the liposomes, 5 mice from both groups were simultaneously exposed for 60 minutes to an aerosol of ZnO NPs generated by a dry powder aerosol generator (Microdosing system; Fraunhofer ITEM). The generator was operated at a pressure of 1.0 bar, which resulted in an airflow of 14.7 L/min. The ZnO NPs (synthesized by PlasmaChem) were spherical, monodisperse, had a primary particle size of 13.2 ± 5.4 nm, and a specific surface area of 26.2 m2/g. 26,27 Five mice from the control group were exposed to clean air for 60 minutes. During aerosol exposure, mice were placed in body plethysmographs, and the animals were exposed head-only. The control mice were exposed to air, to determine the effect on respiration coming from the strain of sitting in the plethysmographs. Control mice were not exposed to ZnO NP as untreated mice have previously been exposed to this particle. 13 The NOTOCORD-hem data acquisition software (Notocord Systems SA) was used to collect respiratory parameters as described previously 28 and tidal volume was used to assess the effect of ZnO NP exposure. Before exposure, the mice were allowed to breathe clean air for 15 minutes in order to obtain a baseline for each mouse. The baseline period was followed by a 60-minute exposure period to allow the study of time-dependent respiratory changes. After the exposure, the mice were allowed to breathe clean air for 15 minutes, termed the recovery period.
The aerosol was continuously sampled on a filter from the breathing zone of the mice for gravimetrical analysis. Aerosol sampling was conducted in accordance to the Danish Standard DS/EN 481. 29 The flow rate through the inlet of the filter cassette was 1.9 L/min corresponding to an air velocity of 1.25 m/s. Sampling was performed throughout the exposure period. The aerodynamic equivalent diameter of the ZnO particles in the aerosol was measured using an Electrical Low Pressure Impactor as for in vitro experiments.
Results
Effects of ZnO NPs and Zn2+ Ions on LS Function In Vitro
The minimum surface tension of LS increased to above 10 mN/m (both mean and lower 95% confidence limit) from the first dose of ZnO NPs exposure (200 ng ZnO/cm2). On the other hand, exposure to micronized lactose (negative control) did not result in the increase of the minimum surface tension, even at the highest exposure dose (1,200 ng/cm2; Figure 1A). The ZnO NPs aerosol generated in the constrained drop surfactometer chamber had a peak in the number size distribution around 250 to 600 nm, which was consistent with the aerosol generated in the mouse bio assay (Figure 2).
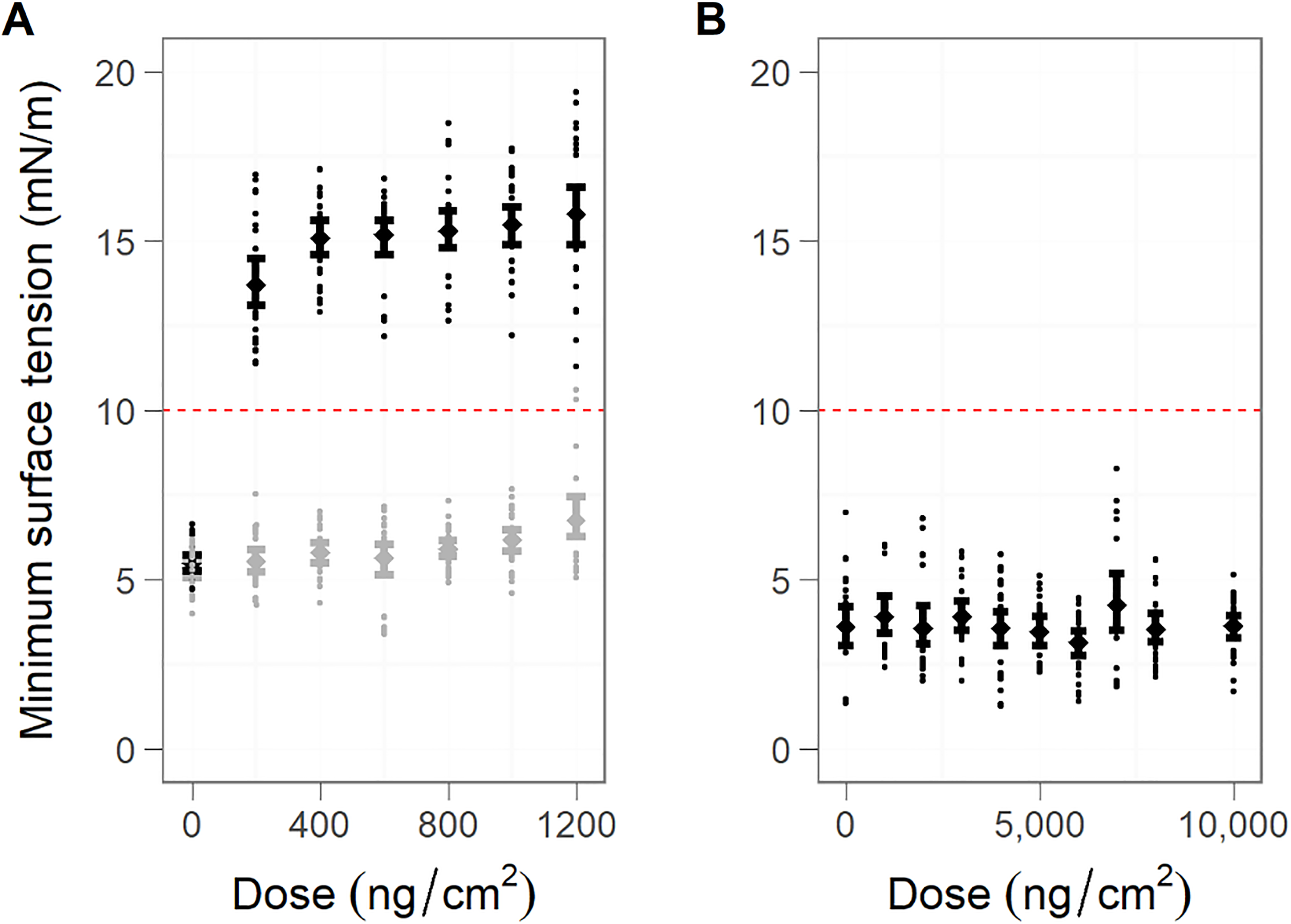
Minimum surface tension (mN/m) as a function of the deposited dose (ng/cm2). A drop of Curosurf (10 μL, 2.5 mg/mL) was exposed to an increasing dose of aerosolized ZnO NPs. The LS drop was cycled throughout the experiment, and the mean minimum surface tension was calculated at set deposited doses measured by a QCM placed adjacent to the LS drop. Five independent experiments were performed. Each data point corresponds to the average of the minimum surface tension of 5 consecutive cycles for 5 independent experiments. The 95% confidence limits are represented. The red dashed line represents the threshold at 10 mN/m above which alveolar collapse would be observed in vivo. A, Zinc oxide (ZnO) NPs (black) and lactose (negative control, grey). B, Zn2+ ions in the form of ZnSO4 solution. NP indicates nanoparticle; LS, lung surfactant; QCM, quartz crystal microbalance.
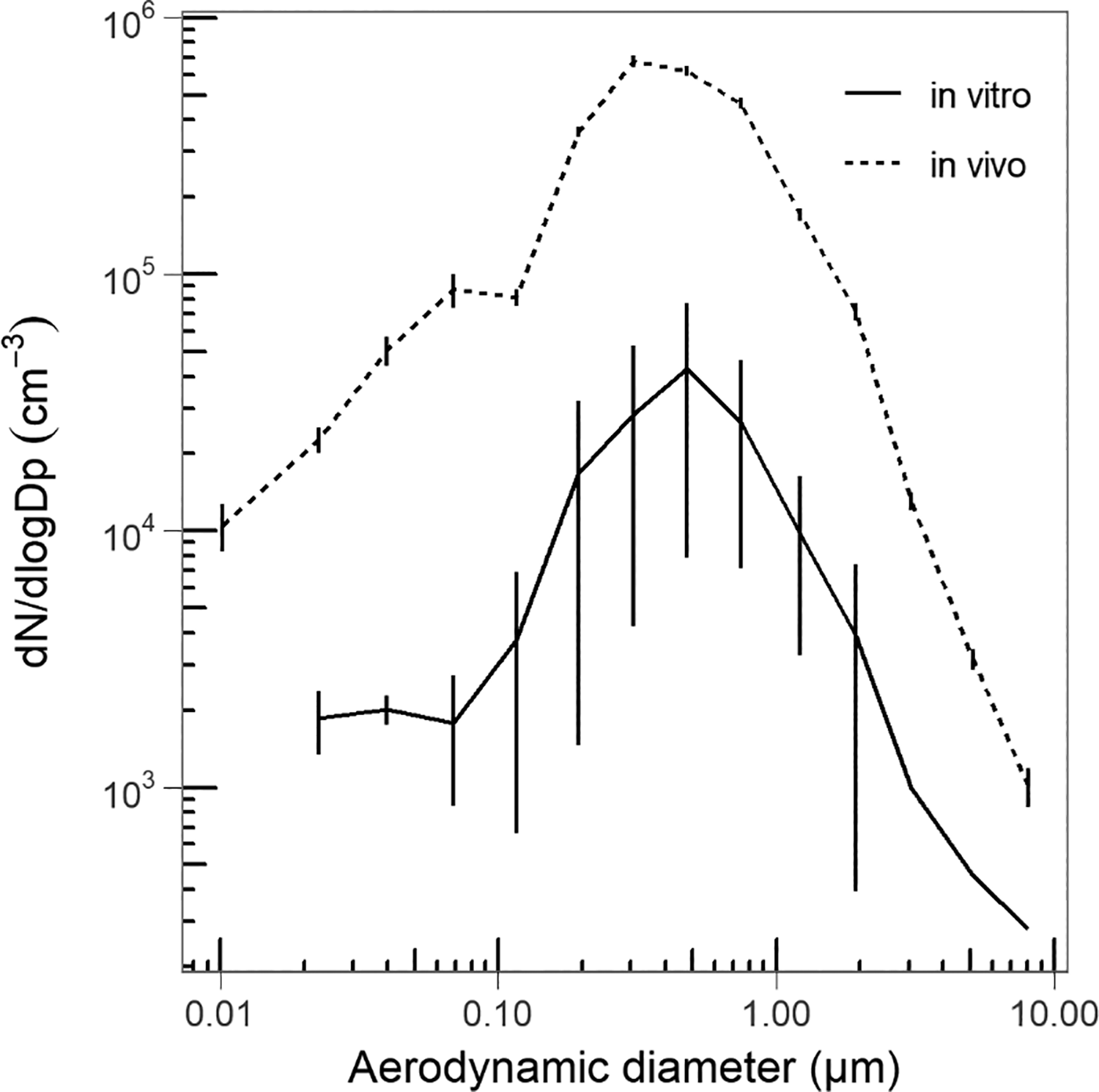
Averaged zinc oxide (ZnO) NPs aerosol size distributions for the in vivo and in vitro experiments measured with the electrical low pressure impactor. NP indicates nanoparticle.
In contrast to ZnO NPs, the exposure of the LS film to Zn2+ ions did not induce any increase in the minimum surface tension, even with 10,000 ng ZnSO4/cm2 (Figure 1B). Thus, the inhibition of LS exposed to zinc in the form of NPs mediated by the particles and not by Zn2+ ions.
Characterization of Macrophage Depletion
A decrease of almost 80% in the number of macrophages was observed 48 hours after administration of Clodrosome, compared to the Encapsome group (Figure 1, Supplemental material). Liposome treatment (both Clodrosome and Encapsome groups) resulted in a significant elevation of total protein concentration in bronchoalveolar lavage fluid (data not shown) and of neutrophils and eosinophils compared to the control group treated with saline (Figure 1, Supplemental material). This indicates that the liposomes in themselves caused inflammation in the lungs.
Effect of Exposure to ZnO NPs on Tidal Volume
Mice treated with either Clodrosome or Encapsome were simultaneously exposed to an aerosol of ZnO NPs at a concentration of 212 mg/m3. The aerosol contained agglomerates of particles with a peak in the number size distribution around 250 to 600 nm (Figure 2). Thus, the particles were able to reach all regions of the airways. 13 Inhalation of ZnO NPs gave rise to a clear reduction in the tidal volume in both groups of mice (Figure 3). The onset of the response occurred within the first 5 minutes of the exposure and the response developed gradually over the 60 minutes of exposure. At the end of the ZnO NPs aerosol exposure, the tidal volume was reduced to approximately 40% of the baseline level, and no recovery was seen during the 15-minute exposure to clean air. No difference was observed between the Clodrosome and the Encapsome groups. The control mice (treated with saline instead of liposomes) showed a slight reduction of tidal volume during the 90-minute exposure to clean air (5% lower at the end of exposure compared to baseline; Figure 3).
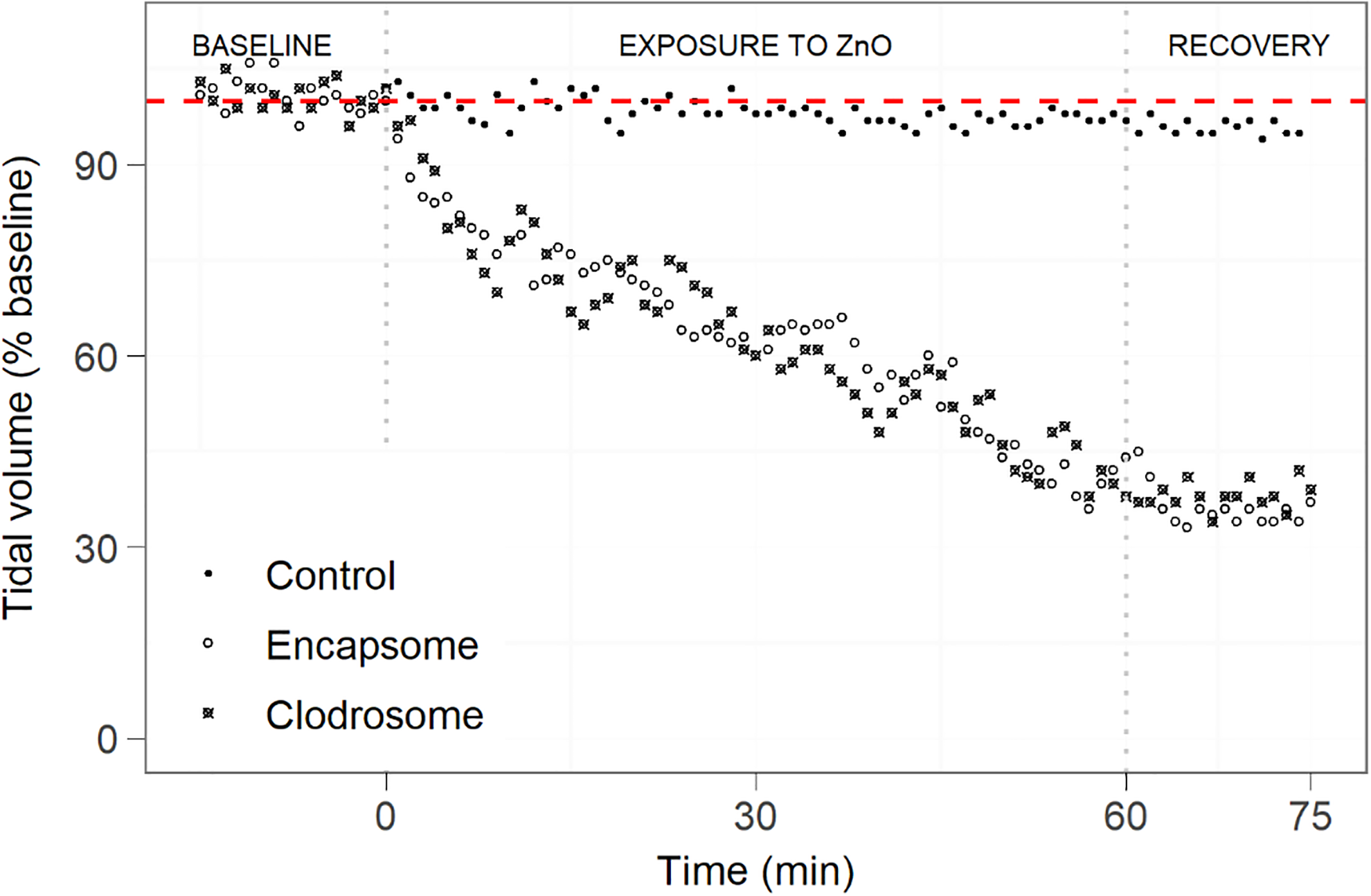
Tidal volume (percentage of the baseline) in mice exposed for 15 minutes to clean air, followed by a 60-minute exposure to zinc oxide (ZnO) NPs, and finally a 15-minute exposure to clean air. Mice in the air control group were exposed 90 minutes to laboratory air. Each group contains 5 animals. The red-dashed line represents 100% of the baseline value.
Discussion
The aim of the study was to investigate the mechanism of ZnO NPs-induced acute inhalation toxicity. Several studies have proposed that the acute toxicity of inhaled ZnO NPs is driven mainly by Zn2+ ions arising from macrophage lysosomal dissolution of ZnO. 10,11 However, we showed that the ZnO NPs but not the free Zn2+ ions were a potent inhibitor of LS function in vitro. Using the mean surface area of the cycling drop (0.254 cm2) and the deposited dose of ZnO NPs on the QCM, we calculated the lowest observed adverse effect dose, defined as a minimum surface tension above 10 mN/m as the adverse outcome, to be 2.03 µg ZnO NPs/mg LS. As NPs often exert their adverse effect because of their large surface area, this can be further converted to 0.53 cm2/mg LS. The surface tension lowering ability of LS was disrupted from the very first exposure dose of ZnO NPs, thus, the inhibition would likely have occurred at even lower exposure doses, but in the current setup, it was not possible to expose to a lower dose of the NPs. The Zn2+ ions did not inhibit LS function, therefore, the lowest observed adverse effect dose could not be calculated.
As opposed to aerosolizing ZnO NPs, others have mixed LS with ZnO NPs to evaluate the effect of LS function 30,31 also using the constrained drop surfactometer. Yang et al 31 studied the interaction of LS with several NPs, among which ZnO (mixed with the natural LS extract Infasurf to a final concentration of 10 µg/mg LS, or 1.2 cm2/mg). The effects observed in the present study are much more severe at a lower exposure dose, where we observe an increase in minimum surface tension increases from 5.5 ± 0.6 to 13.8 ± 1.8 mN/m (n = 5) upon exposure, as opposed to an increase from 2.5 to 4.0 mN/m in the study by Yang et al. 31 Yang et al showed that the rise of the minimum surface tension of Infasurf after exposure to ZnO NPs was directly correlated to alveolar collapse seen in histology and inflammation observed after intratracheal installation in mice. 30
From our in vitro study of the effect of ZnO NPs and Zn2+ ions, we demonstrated that it is the interaction between the NPs and the LS rather than the ions that drive this toxicological mode of action. Therefore, the subsequent animal experiments were performed using ZnO NPs rather than free Zn2+ ions. To determine the role of macrophages on the acute inhalation toxicity of zinc, 2 groups of mice, 1 with lung macrophages and 1 with lungs depleted of macrophages, were exposed to a ZnO NP aerosol. In the present study, mice were exposed to 212 mg ZnO/m3, corresponding to 170 mg Zn/m3. Assuming that 10% of the inhaled ZnO NPs aerosol deposits in the alveolar region of the lungs and that a mouse has 0.15 mg LS, 32 the deposited dose after 5 minutes inhalation (where the changes in respiration pattern became apparent; Figure 2) is estimated to be 37 µg/mg LS, or 9.69 cm2/mg LS. Data for the calculation can be found in Supplementary materials. This is higher than the lowest observed adverse effect dose found in vitro. However, since the mice show an effect on respiration from the first exposure to ZnO NP, we chose to calculate the dose after 5 minutes of exposure to be sure that the changes originated from the exposure and not from stochastic changes in breathing.
Combining intravenous and intratracheal administration of clodronate-loaded liposomes to mice reduced the number of alveolar macrophages by almost 80% compared to PBS-loaded control liposomes 48 hours after administration. The subsequent exposure of mice to an aerosol of 212 mg/m3 ZnO NPs resulted in a marked decrease in the tidal volume in both liposome-treated groups and no difference was observed between the group with and without alveolar macrophages. This suggests that the acute effects of ZnO NPs on the breathing pattern of mice were driven by a macrophage-independent mechanism. The duration of ZnO NPs exposure was relatively short and this makes it unlikely that the ZnO NPs were cleared from the lungs, that is, ZnO NPs accumulate in the lungs over the exposure period, which likely explains the gradual decrease in tidal volume. The reduction in tidal volume occurred within a few minutes after the start of the ZnO NPs exposure and was not reversed during the 15-minute post-exposure recovery period. A previous study investigating the respiratory effects of aerosolized metal oxide NPs, including TiO2, CeO2, ZnO, and Al2O3, showed a similar alteration of the breathing parameters, 13 but in contrast to ZnO NPs exposure, a recovery of the altered breathing patterns (tidal volume and expiratory flow rate) was seen following cessation of exposure to TiO2, CeO2, and Al2O3. It was concluded that the respiratory effects of TiO2, CeO2, and Al2O3 were due to nonspecific physical reactions in the airways. 13 The respiratory depressive effect caused by ZnO NPs was more persistent and the underlying mechanisms are likely different from the other tested NPs.
The inhibition of the surface tension lowering properties of the LS is a likely explanation for the persistent depression in tidal volume observed in mice following ZnO NPs exposure. The fast and irreversible response resembles the effects previously observed after exposing mice to impregnation spray products, 33 for which the mechanism of toxicity was shown to be the inhibition of LS function. 15,16,18 However, the acute effects of ZnO NP inhalation may also be triggered by other mechanisms in the lungs, such as stimulation of nerves, disruption of barrier function, and triggering inflammation.
In vivo, an increase in the minimum surface tension after compression initiates the development of atelectasis, a partial collapse of the lung, which can be seen as a decrease in the tidal volume and may lead to physical lung damage because of the shear stress to the lung epithelium. Shear stress also leads to lung edema due to the extravasation of blood proteins, including albumin, into the lung lumen. Albumin is a known inhibitor of LS function and its presence in the alveoli can exacerbate the situation. 20,34,35
In conclusion, the present study demonstrates an acute toxic effect on the lungs following inhalation of high concentrations of ZnO NPs in vivo. The marked decrease in the tidal volume correlated with the inhibition of the LS function in vitro observed following exposure to ZnO NPs but not Zn2+ ions. The fast-acting effect shows that ZnO NPs do not necessarily need to be dissolved to Zn2+ ions in order to exert toxic effects.
Supplemental Material
Supplementary_materials - Acute Inhalation Toxicity After Inhalation of ZnO Nanoparticles: Lung Surfactant Function Inhibition In Vitro Correlates With Reduced Tidal Volume in Mice
Supplementary_materials for Acute Inhalation Toxicity After Inhalation of ZnO Nanoparticles: Lung Surfactant Function Inhibition In Vitro Correlates With Reduced Tidal Volume in Mice by Søren T. Larsen, Emilie Da Silva, Jitka S. Hansen, Alexander C. Ø. Jensen, Ismo K. Koponen and Jorid B. Sørli in International Journal of Toxicology
Footnotes
Authors’ Note
S.T.L. and E.D.S. share first authorship.
Acknowledgment
The authors thank M. Guldbrandsen, M. Hammer, and S. H. Nielsen for technical support.
Author Contributions
Søren Thor Larsen contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Emilie Da Silva contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Jitka S. Hansen contributed to conception and design, contributed to interpretation, and critically revised manuscript; Alexander CØ Jensen contributed to acquisition, analysis, and interpretation and critically revised manuscript; Ismo Koponen contributed to acquisition, analysis, and interpretation and critically revised manuscript; Jorid B. Sørli contributed to design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the FP7 EU project HINAMOX (Grant agreement ID: 228825) and the Danish Centre for Nanosafety. E.D.S. is supported by The National Research Centre for the Working Environment, and the Technical University of Denmark, Department of Environmental Engineering.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
