Abstract
Several pharmaceutical agents are known to produce ethanol intolerance, which is often depicted as disulfiram-like reaction. As in the case with disulfiram, the underlying mechanism is believed to be the accumulation of acetaldehyde in the blood, due to inhibition of the hepatic aldehyde dehydrogenases, albeit this has not been confirmed in all cases by blood acetaldehyde measurements. Herein, cefamandole, cotrimoxazole, griseofulvin, procarbazine, and propranolol, which are reported to produce a disulfiram-like reaction, as well as disulfiram, were administered to Wistar rats and the hepatic activities of ethanol metabolizing enzymes along with the levels of brain monoamines were determined. Blood acetaldehyde was also evaluated after ethanol administration in rats pretreated with the abovementioned pharmaceutical products. Disulfiram, cefamandole, and procarbazine significantly increased blood acetaldehyde levels after ethanol administration, while on the contrary, cotrimoxazole, griseofulvin, and propranolol had no effect on blood acetaldehyde. Interestingly, all substances used, except disulfiram, increased the levels of brain serotonin. According to our findings, cotrimoxazole, griseofulvin, and propranolol do not produce a typical disulfiram-like reaction, because they do not increase blood acetaldehyde when given together with ethanol. On the other hand, all tested agents share the common property to enhance brain serotonin, whereas a respective effect of ethanol is well established. Hence, the ethanol intolerance produced by these agents, whether blood acetaldehyde concentration is elevated or not, could be the result of a “toxic serotonin syndrome,” as in the case of the concomitant use of serotonin-active medications that provoke clinical manifestations similar to those of a disulfiram reaction.
Keywords
Introduction
The disulfiram-ethanol reaction (DER) refers to the unpleasant symptoms experienced upon drinking alcoholic beverages after having previously ingested disulfiram. Blurred vision, nausea, vertigo, anxiety, and cardiovascular effects, such as hypotension, palpitations, tachycardia, and flushing of the face and neck are some of the symptoms observed in the course of a DER, 1 whereas some isolated cases of fatal reactions have been reported. 2 Classically, the DER symptoms have been attributed to the inhibition of the aldehyde dehydrogenases (ALDHs) and the subsequent accumulation of acetaldehyde in the blood. 3 -6
Acetaldehyde is normally produced as a result of the initial oxidation of ethanol by the hepatic alcohol dehydrogenase (ADH), whereafter it is further oxidized, almost as soon as it is formed, mainly by the hepatic ALDHs. So far, the nicotinamide adenine dinucleotide (NAD)-dependent ALDH systems, located in mitochondria and cytosol, are the principal metabolic pathways, through which acetaldehyde is converted to acetate. 7 -9 As a matter of fact, it has been shown that a low-Km mitochondrial ALDH (ALDH2) is responsible for 60% of the metabolism of acetaldehyde, whereas high-Km cytosolic ALDH (ALDH1A1) metabolizes an additional 20%, and the remaining 20% is metabolized by the microsomal cytochrome P-450 (CYP) 2E1 (CYP2E1). 10,11
Various therapeutic agents have been found to produce untoward effects when combined with ethanol, which often are reported as disulfiram-like reactions. In accordance with the DER, disulfiram-like reactions have been also traditionally attributed to the decrease of acetaldehyde elimination from the body, due to inhibition of the hepatic ALDH. However, in a previous report from our laboratory, we tested several pharmaceutical agents known to produce a disulfiram-like reaction, and we have shown that metronidazole and quinacrine neither inhibit ALDH nor increase blood acetaldehyde levels in the Wistar rat. 12 Interestingly, all tested agents shared the common property to enhance brain serotonin, whereas a respective effect of ethanol is well established. 13 -16 Based on these findings, we concluded that the ethanol intolerance produced by the tested agents, whether ALDH is inhibited or not, could be the result of a “toxic serotonin syndrome,” as in the case of the concomitant use of serotonin-active medications.
Following our previous work, in the present study a number of additional pharmaceutical substances with known disulfiram-like reactions were scrutinized in order to clarify the underlying mechanism. For this reason, cefamandole, cotrimoxazole, griseofulvin, procarbazine, and propranolol which are reported to produce a disulfiram-like reaction 17 -23 were administered to Wistar rats and the activities of the hepatic ethanol-metabolizing enzymes were measured. A group of animals was treated with disulfiram, in order to serve as a positive control. Moreover, in 4 brain subregions (striatum, midbrain, hypothalamus, and frontal cortex), the levels of biogenic monoamines and their metabolites were determined. Finally, after coadministration of ethanol with each one of the abovementioned pharmaceutical agents, the concentration of acetaldehyde was measured in whole blood samples.
Materials and Methods
Animals
Male albino rats (Wistar/Af/Han/Mol/Io/RR), 4 months old and weighing 300 to 350 g, were used in this study. All animals were housed in groups of 2 to 3 in plastic cages (Makrolon) with a wood-chip bedding (Populus sp.) and had free access to tap water and pellet chow (Biozoe, Ioannina, Greece). The animals were maintained on a 12 hour/12 hour light/dark cycle with lights on at 07:00 hour and a constant temperature at 22°C ± 2°C.
Experiments on animals were handled with human care in accordance with the National Institutes of Health guidelines and the European Union directive for the care and the use of laboratory animals (Greek presidential decree No. 160 1991).
Drugs
Disulfiram (Sigma Chemicals, Taufkirchen, Germany) and griseofulvin (Sigma Chemicals, Taufkirchen, Germany) were dissolved in olive oil. Cefamandole (Mandocef, amp 1 g, Lilly, Athens, Greece) and propranolol (Inderal, ampule 1 mg) were dissolved in 0.9% NaCl (saline). Cotrimoxazole (Bactrim, tab. 800 mg sulfamethoxazole + 160 mg trimethoprim, Roche, Basel, Switzerland) and procarbazine (Natulan, tab. 50 mg, Roche, Basel, Switzerland) were suspended in saline and olive oil, respectively.
Ethanol (99.99%; Riedel de Haen, Seelze, Germany) was administered as a 20% (vol/vol) solution in saline. All drugs and ethanol were administered in a volume of 1 mL/100 g body weight.
Treatment Protocol
Forty-two animals were divided into 7 groups of 6. Six groups of animals were treated for 4 consecutive days with disulfiram (75 mg/kg body weight), cefamandole (200 mg/kg body weight), cotrimoxazole (240 mg/kg body weight), griseofulvin (100 mg/kg body weight), procarbazine (100 mg/kg body weight), or propranolol (50 mg/kg body weight), respectively, with an additional injection on the fifth day, about 3 hour before killing. The last group of animals served as control and received only olive oil (1 mL/100 g body weight) following the abovementioned treatment protocol.
In a parallel series of experiments, 42 animals were divided into 7 groups of 6. Six groups received disulfiram (75 mg/kg body weight), cefamandole (200 mg/kg body weight), cotrimoxazole (240 mg/kg body weight), griseofulvin (100 mg/kg body weight), procarbazine (100 mg/kg body weight), or propranolol (50 mg/kg body weight), respectively, for 4 consecutive days, whereas the remaining group was used as respective control and received only olive oil (1 mL/100 g body weight). On the fifth day, 1 hour after the last injection of the drug or vehicle and 2 hours before sacrifice, all animals received intragastrically 2 g/kg body weight ethanol.
The fact that control animals received olive oil, while some experimental groups (cefamandole, propranolol, and cotrimoxazole) received the drugs in saline is due to solubility reasons. We believe that this discrepancy does not affect our findings, since we have previously concluded that the effects of olive oil and saline on the neurobiochemical parameters examined in this study are identical (unpublished data). Moreover, it has been shown that when used as vehicles neither olive oil nor saline affect drug action because of their own inherent properties. 24
All drugs were administered intraperitoneally in a single dose each day. Injection of substances into the peritoneal cavity is the most common and reliable route in laboratory rodents being technically simple and easy while allowing quite long periods of absorption from the repository site.
Ιn the cases of cotrimoxazole, griseofulvin, and propranolol, the doses were calculated based on the modified Food and Drug Administration (FDA) guidelines which means that they are (per kg body weight) about 6.2 times higher than the doses given to humans in therapeutics. 25 On the other hand, the doses used for disulfiram, cefamandole, and procarbazine were based on previous literature reports. 12,21,26
Tissue Preparation
Rats were sacrificed by decapitation and the hypothalamus, striatum, midbrain, and frontal cortex were microdissected immediately after killing, according to a conventional and well-established technique. 27 Samples were kept at −80°C until neurotransmitter analyses. The frozen tissues were thawed, weighed, and homogenized for 20 seconds with a sonicator in ice-cold 0.2 N perchloric acid. The homogenate was centrifuged at 10 000×g for 15 minutes at 4°C, and then the supernatant was divided into 2 portions. The first aliquot was used for high-performance liquid chromatographic analyses (HPLC) of serotonin (5-HT), 5-hydroxyindole-3-acetic acid (5-HIAA), and homovanillic acid (HVA), whereas the second 0.2 mL aliquot was transferred to an Eppendorf tube containing 20 mg activated alumina in order to extract noradrenaline (NA), dopamine (DA), and 3,4-dihydroxyphenylacetic acid (DOPAC) from the homogenate prior to HPLC detection.
The liver of each animal was also dissected and a small amount (3 g) was homogenized in a mechanical homogenizer, in 3 volumes (wt/vol) ice-cold 0.25 M sucrose solution. The homogenate was then centrifuged for 10 minutes at 700×g. The pellet, which represented the nuclear fraction, was discarded and the mitochondria were isolated by centrifugation at 10,000×g for 15 minutes. The pellet was resuspended in 3 mL sucrose medium and washed at 10,000×g for 15 minutes. The final pellet was resuspended in 1 mL sucrose medium containing 1% sodium deoxycholate and designated the mitochondrial fraction. The supernatant fraction of the first 10,000×g centrifugation was subjected to a new 60-minute centrifugation at 105,000×g to obtain the cytoplasmic fraction. The resultant pellet of the 105,000×g centrifugation was washed once in 0.25 M sucrose solution (at 105,000×g for 30 minutes), and the final pellet was resuspended in 3 mL of the same sucrose solution and designated the microsomal fraction All steps were carried out from 0°C to 4°C and all fractions were stored in small aliquots at −80°C.
In the experiments with ethanol, heart blood samples were collected via cardiac puncture immediately after decapitation and 500 µL were added in 2 mL of ice-cold 0.6 M perchloric acid prepared in saline. After centrifugation at 4000×g for 10 minutes at 4°C, supernatants (500 μL) were collected and acetaldehyde levels were determined within 3 hours by head-space gas chromatography. No corrections have been made for the nonenzymatic formation of acetaldehyde from ethanol during the analytical procedures, because the levels of artifactual acetaldehyde were under the limit of detection.
Determination of Brain Biogenic Monoamines
Ion-pair reverse-phase chromatography was performed with an HPLC system (LC-9A; Shimadzu, Co. Kyoto, Japan) equipped with an analytical column of 250 mm × 4.6 mm (ODS, C-18; Jones-Apex, Llanbradach, UK), 5-µm particle size, coupled to an electrochemical detector (L-ECD-6A; Shimadzu, Co. Kyoto, Japan) maintained at 0.75 V. The mobile phase was a mixture of 0.1 M citric acid, 0.1 M sodium acetate, and 0.27 mM octyl sulfate with 25% methanol (vol/vol). The separations were performed isocratically at a flow rate of 0.6 mL/min. 28,29 Samples were injected manually (20 μL) and the compounds under investigation were identified by comparison with the retention times of the authentic standards. The order of elution of the standard solutions was NA (5.6 minutes), DA (7.1 minutes), DOPAC (8.4 minutes), 5-HIAA (10.5 minutes), 5-HT (12.1 minutes), and HVA (14.2 minutes).
Enzyme Assays
All the assays of dehydrogenase enzymes were performed spectrophotometrically by following the formation of reduced nicotinamide adenine dinucleotide at 340 nm in a Shimadzu UV1601 spectrophotometer. Alcohol dehydrogenase as well as ALDH2 was measured at 25°C, whereas the cytoplasmic isozyme of ALDH (ALDH1A1) at 37°C.
Alcohol dehydrogenase was assayed according to Koivula et al 30 in a mixture containing 70 mM NaOH-glycine buffer (pH 9.6), 0.67 mM NAD, and 10 mM ethanol, whereas for the determination of ALDH1A1, the assay mixture contained 75 mM sodium pyrophosphate buffer (pH 8.0), 1 mM pyrazole (for the inhibition of ADH), 1 mM NAD, and 5 mM propionaldehyde as substrate. 31
The mitochondrial enzyme ALDH2 was assayed using 50 μM acetaldehyde as substrate. The assay mixture contained 75 mM sodium pyrophosphate buffer (pH 8.0), 1 mM NAD, 1 mM pyrazole (for the inhibition of ADH), and 2 μM rotenone (for the inhibition of NAD oxidase). 21
Cytochrome P-450 (CYP) 2E1 activity was also determined spectrophotometrically by measuring the hydroxylation of p-nitrophenol (p-NP) to 4-nitrocatechol (4-NC) in a Shimadzu UV1601 spectrophotometer. The assay mixture (1 mL) consisted of 100 mM potassium phosphate buffer (pH 6.8), 1 mM reduced nicotinamide adenine dinucleotide phosphate, 100 μM p-NP, and 200 µL of the microsomal sample. After incubation for 10 minutes at 37°C, the reaction was terminated by the addition of 0.5 mL of 0.6 N perchloric acid. After centrifugation at 1,300×g for 5 minutes, 1 mL of the supernatant was mixed with 100 µL of 10 N NaOH, and absorbance was read at 546 nm. Cytochrome P-450 (CYP) 2E1 activity was calculated from a 4-NC standard curve, and the incubation mixture without substrate was used as the blank. 32 Protein determination was carried out by the method of Lowry and coworkers. 33
Statistical Analysis
The SPSS software package (SPSS Statistics, version 25 for Windows, SPSS, Inc, IBM Company, New York, New York) was applied for statistical analysis. Data were explored for normality using Shapiro-Wilk test. One-way analysis of variance (ANOVA) was used to analyze data showing normal distribution, while Kruskal-Wallis test was used for the data showing non-normal distribution. Dunnett post hoc test was used for pair-wise comparisons between the groups when ANOVA test was significant, while Mann-Whitney U post hoc test was used for pair-wise comparisons between the groups when Kruskal-Wallis test was significant.
Data are expressed as the group mean ± standard deviation. The limit for statistical significance was at P values less than 0.05.
Results
Enzyme Activities
Disulfiram (P < 0.001), cefamandole (P < 0.001), procarbazine (P < 0.001), and propranolol (P < 0.05) inhibited ALDH2 activity, which on the contrary was not significantly affected by the administration of cotrimoxazole or griseofulvin (Table 1). Aldehyde dehydrogenase 1A1 activity was also inhibited by disulfiram and cefamandole (P < 0.01), whereas the application of griseofulvin induced the activity of the enzyme (P < 0.01; Table 1). Alcohol dehydrogenase activity was not significantly influenced by any of the drugs tested, with the exception of cefamandole, which induced its activity (P < 0.001; Table 1). Finally, disulfiram and cotrimoxazole inhibited (P < 0.01), while propranolol induced (P < 0.01) CYP2E1 activity (Table 1).
Hepatic Enzyme Activities of Alcohol Dehydrogenase (ADH), Aldehyde Dehydrogenase 1A1 (ALDH1A1), Aldehyde Dehydrogenase 2 (ALDH2), and Cytochrome P-450 (CYP) 2E1 (CYP2E1), After Treatment of Male Wistar Rats With DIS (75 mg/kg), CEF (200 mg/kg), COT (240 mg/kg), GRIS (100 mg/kg), PROC (100 mg/kg), or PROP (50 mg/kg).a
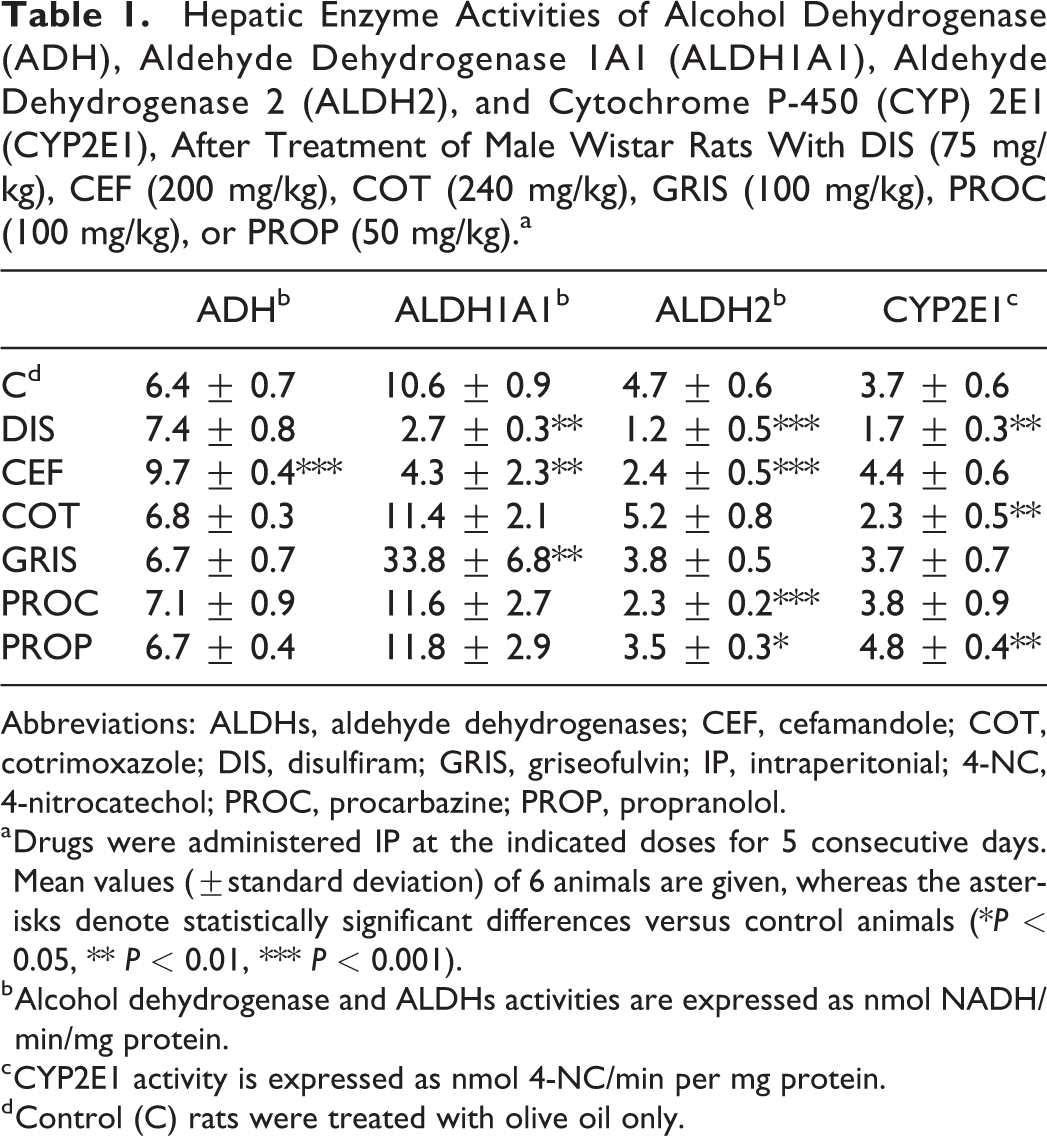
Abbreviations: ALDHs, aldehyde dehydrogenases; CEF, cefamandole; COT, cotrimoxazole; DIS, disulfiram; GRIS, griseofulvin; IP, intraperitonial; 4-NC, 4-nitrocatechol; PROC, procarbazine; PROP, propranolol.
a Drugs were administered IP at the indicated doses for 5 consecutive days. Mean values (±standard deviation) of 6 animals are given, whereas the asterisks denote statistically significant differences versus control animals (*P < 0.05, ** P < 0.01, *** P < 0.001).
b Alcohol dehydrogenase and ALDHs activities are expressed as nmol NADH/min/mg protein.
c CYP2E1 activity is expressed as nmol 4-NC/min per mg protein.
d Control (C) rats were treated with olive oil only.
Blood Acetaldehyde Levels
Disulfiram, cefamandole, and procarbazine resulted in elevated blood acetaldehyde levels after ethanol administration (P < 0.01, P < 0.05, and P < 0.01, respectively; Figure 1). On the contrary, coadministration of cotrimoxazole, griseofulvin, or propranolol with ethanol had no effect on blood acetaldehyde levels (Figure 1).
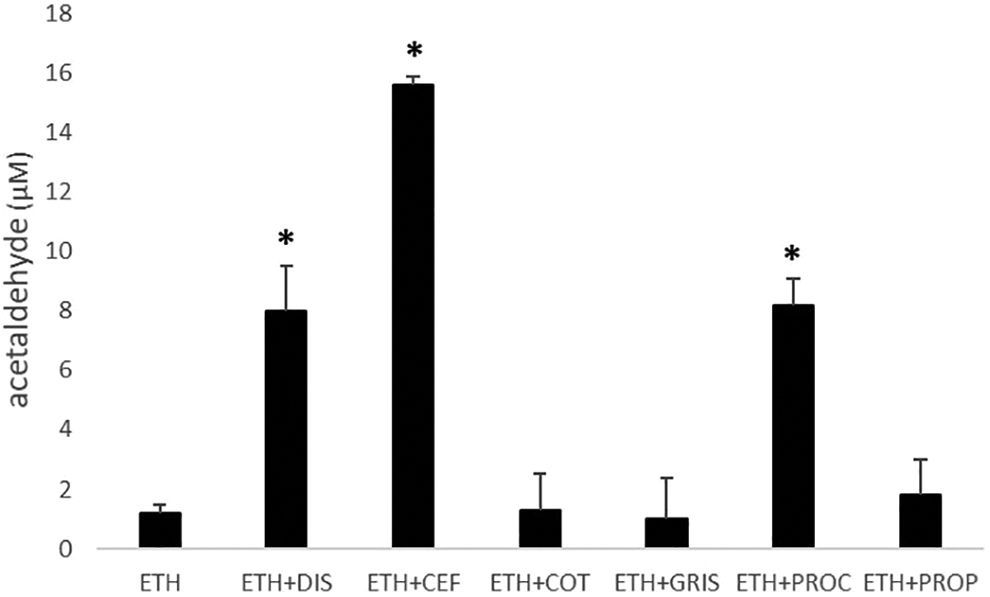
Effects of intraperitoneally administered disulfiram (DIS, 75 mg/kg), cefamandole (CEF, 200 mg/kg), cotrimoxazole (COT, 240 mg/kg), griseofulvin (GRIS, 100 mg/kg), procarbazine (PROC, 100 mg/kg), or propranolol (PROP, 50 mg/kg) on the levels of blood acetaldehyde (µM) of Wistar rats after an intragastric dose of ethanol (ETH, 2 g/kg) 2 hours before sacrifice. The control group was treated with olive oil, followed by an ethanol dose according the abovementioned treatment protocol. The results are the mean values ± standard deviation of 6 animals. Asterisks denote statistically significant differences versus control animals (*P <0.05).
Brain Biogenic Monoamines
Noradrenaline and DA
As expected, according to the known inhibitory effect of disulfiram on dopamine β-hydroxylase, the concentration of NA was found to be decreased in hypothalamus (P < 0.05; Table 2), whereas DA content was increased in striatum (P < 0.001; Table 2) after disulfiram treatment. Cefamandole or propranolol administration had no effect on the brain levels of NA or DA. Cotrimoxazole increased the levels of NA in striatum (P < 0.05; Table 2) and decreased the levels of DA in hypothalamus (P < 0.025; Table 2). Griseofulvin decreased the levels of NA in midbrain (P < 0.05; Table 2) and increased the levels of DA in striatum (P < 0.001; Table 2). Finally, procarbazine increased the levels of NA and DA in midbrain (P < 0.001; Table 2).
Effects of Disulfiram (DIS), Cefamandole (CEF), Cotrimoxazole (COT), Griseofulvin (GRIS), Procarbazine (PROC) or Propranolol (PROP) on the Levels of Noradrenaline (NA), Dopamine (DA), DA Metabolites (DOPAC and HVA), and DA Turnover (t.o.)a in Brain Parts of Wistar rats.b
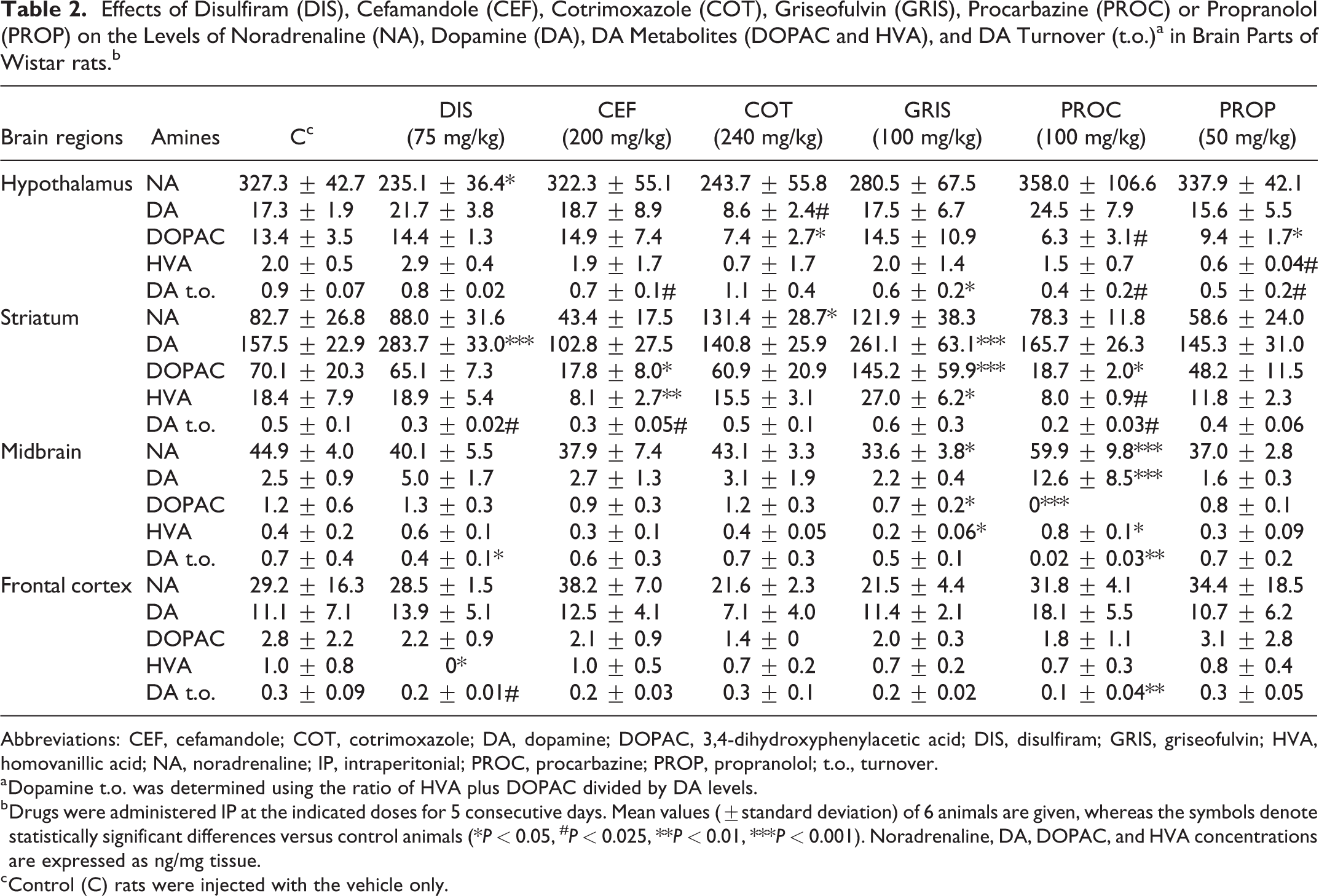
Abbreviations: CEF, cefamandole; COT, cotrimoxazole; DA, dopamine; DOPAC, 3,4-dihydroxyphenylacetic acid; DIS, disulfiram; GRIS, griseofulvin; HVA, homovanillic acid; NA, noradrenaline; IP, intraperitonial; PROC, procarbazine; PROP, propranolol; t.o., turnover.
a Dopamine t.o. was determined using the ratio of HVA plus DOPAC divided by DA levels.
b Drugs were administered IP at the indicated doses for 5 consecutive days. Mean values (±standard deviation) of 6 animals are given, whereas the symbols denote statistically significant differences versus control animals (*P < 0.05, # P < 0.025, **P < 0.01, ***P < 0.001). Noradrenaline, DA, DOPAC, and HVA concentrations are expressed as ng/mg tissue.
c Control (C) rats were injected with the vehicle only.
3,4-Dihydroxyphenylacetic acid, HVA, and DA turnover
Disulfiram treatment resulted in a reduction of the levels of HVA in frontal cortex (P < 0.05; Table 2), with a parallel reduction of the turnover of DA in striatum (P < 0.025; Table 2), midbrain (P < 0.05; Table 2), and frontal cortex (P < 0.025; Table 2). Dopamine turnover was determined using the ratio of HVA plus DOPAC divided by DA levels.
Cefamandole decreased the levels of DOPAC (P < 0.05; Table 2) and HVA (P < 0.01; Table 2) in striatum, as well as DA turnover in striatum and hypothalamus (P < 0.025; Table 2). Cotrimoxazole decreased DOPAC levels in hypothalamus (P < 0.05; Table 2). Griseofulvin increased the levels of DOPAC (P < 0.001; Table 2) and HVA (P < 0.05; Table 2) in striatum, while it decreased both the levels of DOPAC and HVA (P < 0.05; Table 2) in midbrain, as well as DA turnover in hypothalamus (P < 0.05; Table 2). Procarbazine decreased DOPAC levels in all brain regions but frontal cortex (P < 0.05 for striatum; P < 0.001 for midbrain; P < 0.025 for hypothalamus; Table 2) and HVA levels in striatum (P < 0.025; Table 2), increasing at the same time the levels of HVA in midbrain (P < 0.05; Table 2). In addition, it decreased DA turnover in all brain regions measured (P < 0.025 for striatum and hypothalamus, P < 0.01 for midbrain and frontal cortex; Table 2). Finally, propranolol decreased the levels of DOPAC (P < 0.05; Table 2) and HVA (P < 0.025; Table 2), as well as DA turnover (P < 0.025; Table 2) in hypothalamus.
Serotonin (5-HT), 5-HIAA, and 5-HT turnover
Disulfiram increased the levels of 5-HT in all brain subregions. However, this finding did not reach statistical significance. On the other hand, it increased the levels of 5-HIAA (P < 0.05; Figure 2), as well as of the 5-HT turnover (P < 0.005; Figure 2) in midbrain and frontal cortex. The turnover of 5-HT was determined using the ratio of 5-HIAA/5-HT levels.
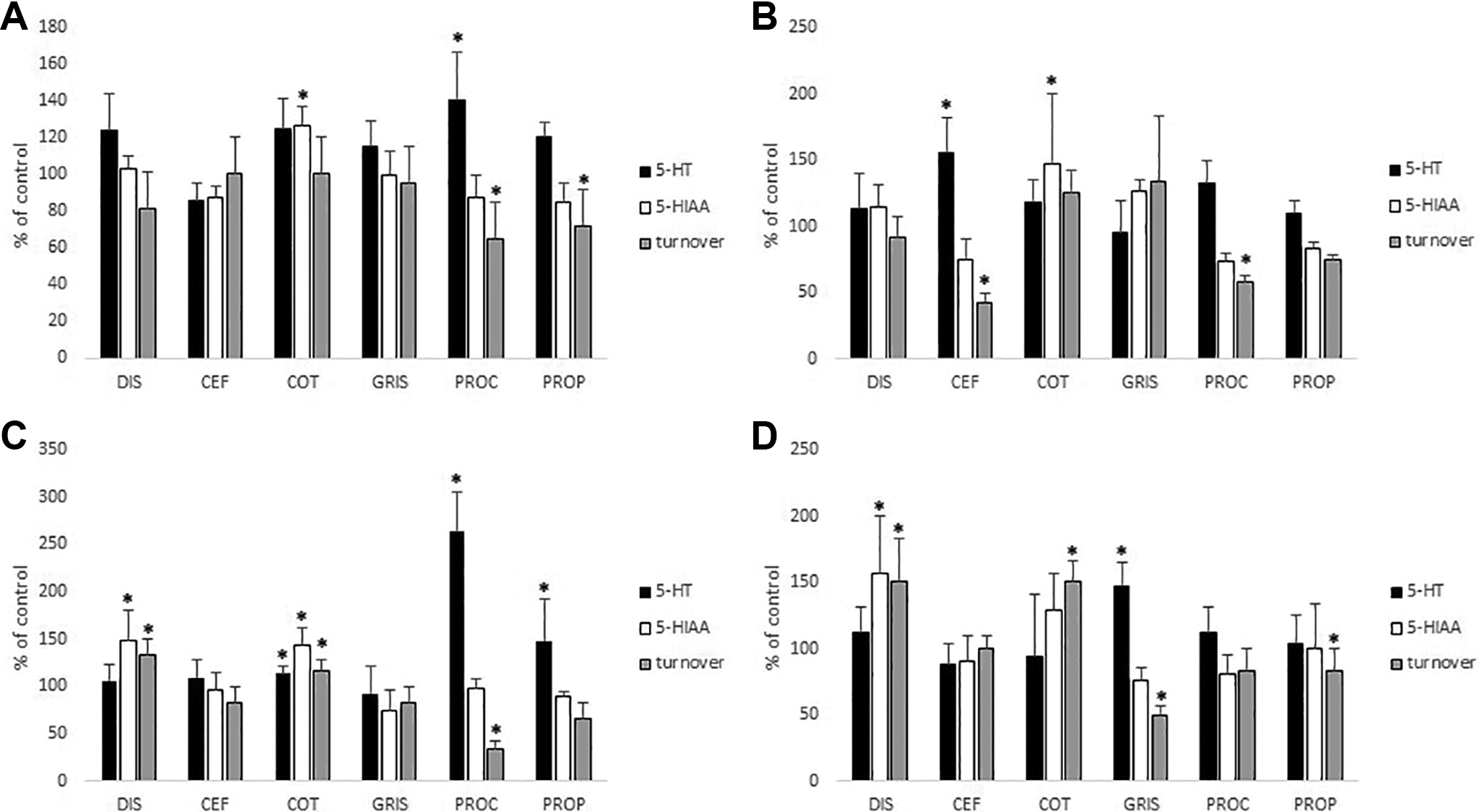
Effects of intraperitoneally administered disulfiram (DIS, 75 mg/kg × 5 days), cefamandole (CEF, 200 mg/kg × 5 days), cotrimoxazole (COT, 240 mg/kg × 5 days), griseofulvin (GRIS, 100 mg/kg × 5 days), procarbazine (PROC, 100 mg/kg × 5 days), or propranolol (PROP, 50 mg/kg × 5 days) on the levels of serotonin (5-HT), 5-hydroxyindole-3-acetic acid (5-HIAA), and on the 5-HT turnover (5-HIAA/5-HT) in the hypothalamus (A), striatum (B), midbrain (C), and frontal cortex (D) of adult male Wistar rats. Control (C) rats were treated with olive oil. Results are expressed as percentage of the control and values are means (±standard deviation) of 6 animals. The asterisks denote statistically significant differences versus control rats (*P < 0.05).
Cefamandole increased the levels of 5-HT as well as 5-HT turnover in striatum (P < 0.025; Figure 2). Cotrimoxazole increased the levels of 5-HT in midbrain (P < 0.05; Figure 2) along with the levels of 5-HIAA in all brain regions measured with the exception of frontal cortex (P < 0.025 for striatum and midbrain, P < 0.005 for hypothalamus; Figure 2). Additionally, it increased 5-HT turnover in midbrain (P < 0.05; Figure 2) and frontal cortex (P < 0.025; Figure 2). Griseofulvin increased the levels of 5-HT (P < 0.025; Figure 2) and 5-HT turnover (P < 0.01; Figure 2) in frontal cortex. Procarbazine increased 5-HT levels in midbrain (P < 0.01; Figure 2) and hypothalamus (P < 0.05; Figure 2), while it decreased 5-HT turnover in all brain regions but the frontal cortex (P < 0.05 for striatum, P < 0.01 for midbrain and hypothalamus; Figure 2). Finally, propranolol increased 5-HT levels in midbrain and decreased 5-HT turnover in hypothalamus (P < 0.05; Figure 2).
Discussion
The ingestion of alcohol by individuals previously treated with disulfiram brings about marked signs and symptoms, which are attributed to an increase in the concentration of acetaldehyde in the body, due to inhibition of the hepatic ALDH by disulfiram. 3 -6 Besides disulfiram, disulfiram-like reactions may also occur in patients who have ingested alcohol after treatment with a number of pharmaceutical agents. 17 -23,34,35 As in the case with the DER, a disulfiram-like reaction is believed to be basically an “acetaldehyde syndrome.” The inhibition of the metabolism of acetaldehyde by these drugs, which theoretically leads to increased blood acetaldehyde levels, has been suggested as the main explanation for this syndrome, albeit it has not been confirmed in all cases by blood acetaldehyde measurements.
In a previous paper of our laboratory, 12 we aimed to clarify the mechanism behind the alcohol intolerance produced by chloramphenicol, metronidazole, furazolidone, and quinacrine, which has been, somehow arbitrarily, characterized as a disulfiram-like reaction. 17,18,35 Interestingly, we showed that metronidazole and quinacrine do not inhibit the hepatic ALDH nor increase blood acetaldehyde levels and hence they do not produce a typical disulfiram-like reaction. On the other hand, we demonstrated that all pharmaceutical products tested increased the levels of brain 5-HT, while the enhancing effects of ethanol on the brain serotonergic system are well established. 13 -16 Likewise, we suggested that the reaction to ethanol exhibited by these drugs could be the result of a “serotonin syndrome,” that can take place after the concomitant administration of 2 or more serotonergic agents. 36
As a continuation of our previous work, 12 in the present article we tested 5 additional pharmaceutical agents, reported to bring about a disulfiram-like reaction, 17 -23 evaluating their effects on hepatic ethanol metabolism and brain monoamines. As expected, disulfiram, which was used as a positive control, inhibited ALDH1A1, ALDH2, and CYP2E1, while it had no effect on ADH activity. The ALDH2 was also inhibited by cefamandole, procarbazine, and propranolol but not by cotrimoxazole or griseofulvin. Cefamandole also inhibited ALDH1A1, while griseofulvin induced its activity. Interestingly, cotrimoxazole proved to be an inhibitor of CYP2E1 whereas propranolol induced the activity of this enzyme. Finally, ADH was influenced only by cefamandole which was shown to be an inducer of the enzyme.
As a consequence of their effects on the hepatic ethanol metabolizing enzymes, disulfiram, cefamandole, and procarbazine significantly increased blood acetaldehyde levels after ethanol administration. On the contrary, cotrimoxazole and griseofulvin had no effect on blood acetaldehyde following ethanol ingestion. Finally, despite the fact that it inhibited ALDH2, propranolol also did not increase blood acetaldehyde concentration when combined with ethanol. As was suggested earlier for the CYP2E1 inducer isoniazid, 37 this observation is probably due to the induction of CYP2E1 by propranolol which theoretically leads to an increased elimination rate of acetaldehyde, preventing its accumulation.
In addition to the effects on the activity of the hepatic ethanol metabolizing enzymes, the drugs tested produced a number of significant changes on the levels of brain biogenic monoamines, among which, those concerning the serotonergic system appear to be more important as a possible explanation behind the ethanol intolerance they provoke. In contrast, the central noradrenergic and dopaminergic systems were affected in an inconsistent way, which can hardly stand as a possible mechanistic explanation of the clinical syndrome arising when these drugs are coingested with alcohol.
Concerning the brain serotonergic system, we demonstrated that all pharmaceutical products tested had an enhancing effect on brain 5-HT levels, with the exception of disulfiram. Alcohol, on the other hand, interacts with serotonergic synaptic transmission in the brain in several ways. Even a single dose of ethanol alters various aspects of the serotonergic system. In humans, for example, the levels of 5-HT metabolites in the urine and blood increase after a single drinking session, indicating increased 5-HT release in the nervous system. 13 In addition, animal studies also have concluded that acute ethanol administration elevates 5-HT levels within the brain. 13 -16
The concomitant administration of two or more agents that elevate synaptic levels of brain 5-HT is implicated in the induction of a toxic syndrome, known as the “serotonin syndrome.” 36 The most serious cases are usually caused by unintended synergism between a selective serotonin reuptake inhibitor (SSRI) and one or more other medications, such as monoamine oxidase (MAO) inhibitors. 38 Moreover, the combination of ethanol with a MAO inhibitor or a SSRI has also been reported to produce a serotonin syndrome. 39,40 The clinical manifestations of this hyperserotonergic condition are a triad of altered conscious state, autonomic dysfunction, and neuromuscular excitability. 41 However, in a retrospective study by Radomski et al, 42 it was shown that the clinical picture of the serotonin syndrome may be highly variable and a wide range of symptoms were reported during its course. Surprisingly, in this study where the full symptom profile of 24 cases of serotonin syndrome was reviewed and analyzed, one can realize that, in fact, all the symptoms observed during a “disulfiram reaction” are included in the detailed list of symptoms provided by the abovementioned study. 42 On the grounds of this observation, and because both ethanol and the group of agents tested in the present study increase brain 5-HT, it is tempting to speculate that the ethanol intolerance produced by these drugs could be due to enhanced 5-HT concentration in the central nervous system, within the scope of a serotonin syndrome. This may equally concern substances with the ability to increase blood acetaldehyde after ethanol ingestion, such as cefamandole and procarbazine, or substances lacking this ability, such as cotrimoxazole, griseofulvin, and propranolol, provided that they can increase the central levels of 5-HT.
In the current study, the doses selected for a number of drugs were based on the modified FDA guidelines, while for the rest of the drugs the doses were dictated by previous publications. Τhis inconsistency constitutes a limitation of our study and the possibility that this might have had an impact on our results cannot be ruled out. In conclusion, despite the aforementioned limitation, the data of the present study gives further support to the findings of our previous work 12 where we suggested that the ethanol intolerance produced by a number of drugs is not always a “disulfiram-like reaction.” Likewise, herein we clearly showed that, at least cotrimoxazole, griseofulvin, and propranolol, do not produce a typical disulfiram-like reaction, as has been reported until now, because they do not increase blood acetaldehyde when given together with ethanol. On the other hand, similarly with our previous findings, we demonstrated that all substances tested share the common property to increase the levels of brain 5-HT, while the enhancing effects of ethanol on the central levels of 5-HT are well established. Accordingly, it could be assumed that the reaction to ethanol exhibited by these drugs, regardless of the inhibition of the hepatic ALDHs or changes in the levels of blood acetaldehyde, may be the result of a serotonin syndrome, as in the case of the concomitant administration of serotonergic agents that provoke clinical manifestations similar to those of a disulfiram reaction.
Finally, in a more general concept, and irrespectively of the ethanol intolerance produced by cefamandole, cotrimoxazole, griseofulvin, procarbazine, and propranolol, we suggest that clinicians should be aware of the serotonergic properties of these pharmaceutical agents, which should be prescribed very cautiously in patients under therapy with MAO inhibitors, SSRIs, or other medications that have a high probability of inducing serotonin syndrome. Due to the potentially serious nature of this condition, it seems prudent that physicians always monitor patients receiving combinations of serotonergic drugs and be alert to the possibility of “serotonergic duplication.”
Footnotes
Author Contributions
Pappas P. contributed to design, contributed to acquisition, and drafted manuscript; Boumba V. contributed to design, contributed to analysis, drafted manuscript, and critically revised manuscript; Marselos M. contributed to conception and design, contributed to interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
