Abstract
In the adoption of behavior as a critical end point in safety pharmacology and neurotoxicity screening, federal regulatory agencies have shifted the predominating scientific perspective from pharmacology back to the experimental analysis of behavior (psychology). Nowhere is this more evident than in tier I safety assessment of the central nervous system (CNS). The CNS and peripheral nervous system have multiple behavioral units of general activity. A complete picture of the motor control neural pathways cannot be measured by any one single approach. The CNS safety protocols under International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use S7A are required to be conducted in accordance with Good Laboratory Practices by trained technical staff. The CNS safety assessments necessitate the inclusion of a thorough and detailed behavioral analysis of home cage activity, the response to handling, and transportation to and observations within an open-field apparatus with ancillary measures of basal muscle tone, muscle strength, and tremor in a functional observation battery, as well as quantitative measurements of 3-dimensional activity in an automated photobeam arena. Cost-cutting initiatives or a radical application of the “reduce use” principle of the 3 Rs only jeopardize the spirit, intent, and predictive validity of tier I safety testing assays dictated by current drug safety guidelines.
Keywords
Introduction
All drug and chemical manufacturers are required to conduct an exhaustive and thorough scientific investigation and subsequent review of nonclinical data to determine the acute risks or hazards to human health and well-being prior to the first dose administration in man. The International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) formally adopted guidelines for conducting nonclinical safety assessment studies in 2000. 1 Guidelines have been issued for both acute toxicity and acute pharmacology studies. Information on the acute toxicity testing is used to predict the consequences of human overdose situations and should be available to support phase III clinical trials. These early assessments of acute toxicity are important for therapeutic indications in patient populations that are at higher risk for overdosing (eg, depression, pain, dementia) in outpatient clinical trials; therefore, high-dose administrations are required. In contrast, a subgrouping of the harmonization studies categorized as “safety pharmacology” studies is focused on identifying adverse effects on physiological functions following a single-dose administration in laboratory animals which produces a small (or no) effect to an exposure level that produces marked effects. 2 However, section 2.4.1 of the ICH S7 guideline 1 sets the upper range of the dose–effect function to “doses that exceed the primary pharmacodynamic or therapeutic range.” The highest tested dose in safety study protocols should be a dose that produces only moderate adverse effects. Further, the guidance acknowledges that in practice, some effects in the toxic range (eg, tremors) may confound the interpretation of the results and may also limit or restrict high-dose levels on the study. The core safety pharmacology battery of tests is designed to identify unexpected effects on organ system function and to broaden pharmacological characterization (pharmacological profiling).
Tests in the first tier are designed to determine the presence of neurotoxicity. The functional capacity of the nervous system cannot be determined independent of behavioral analysis. Some of the problems confronting behavioral toxicology are the translation of human subjective complaints (malaise, nausea, illness) into behavioral tests in animals. Determining subtle effects on the nervous system in the face of the well-known functional reserve and adaptability of the nervous system cannot be independently measured, apart from the influence of the subjective or internal milieu of the organism administered drug on the specific measure of interest. Since the international adoption of the harmonized safety screening, there has been a tide of “regulatory creep” within the drug development industry that has the potential to erode the validity of the established core assays of central nervous system (CNS) safety testing which were established as the foundation of the tiered testing system. The purpose of this review is to provide support for the inclusion of the original regulatory agency study designs that included use of 3 independent observational methods designed to assess motor function in rats, including their home cage, open-field, and automated locomotor activity monitoring (LMA) systems, for appropriate characterization of all ICH S7A CNS safety study evaluations.
What Are the Defining Features of the Issue?
Becker et al
3
suggest that any single mandated battery of tests beyond the screening tier (eg, beyond tier I) would not permit flexibility to set rational testing priorities based on anticipated toxicity and exposure potentials. There are a variety of terms used within the industry to define “motor function” testing. The term “spontaneous motor activity” has been used to refer to a behavioral change that can be observed or measured in the absence of any intervention influencing the willingness of the animal to move during the testing period and is usually applied to home-cage observations of activity. For example, spontaneous activity includes behavioral expressions observed when there is no experimenter-presented stimulus applied to motivate the animal to move, such as shock, tapping on the cage, dropping food into the cage, and so on. Observations collected from the home-cage environment can seriously underdocument the changes observed in the free-field situation. Lynch and Mittelstadt
4
have demonstrated that a change in spontaneous locomotor activity is an excellent preclinical marker for CNS effects of test compounds, including effects observed in a modified Irwin test of general behavior. The term “motor activity” (or “general motor activity”) has been used to refer to the summation of movements of any part of the body regardless of the distance traveled in horizontal or vertical direction, in response to the testing condition. This refers to such observed changes in body position when the rat is placed into the center of the open-field arena. Thus, the term “motor activity” includes both locomotor and nonlocomotor movements (rearing counts) that are observed following experimenter manipulation of the environment (capture, hand restraint, and relocation to the open field). The term “locomotor activity” (LMA) has also been used to refer to actual ambulation of the rat. This term refers to a change in the coordinate position of the rat within the testing device (open arena, maze, alley, etc) by walking, circling, running, or rearing. “Nonlocomotor activity” refers to behaviors such as scratching, pawing, grooming, burrowing, head or body shakes, and sniffing that are observed in detailed clinical observations (detailed cage-side observations, home cage, as well as open-field assays).
Making Sense of Observational Data
As just described, the term “motor activity” refers to a multiple component repertoire of behaviors that are assumed to be free of conditioning, such as walking, rearing, scratching, standing, grooming, and so on. Spontaneous locomotor activity in the rat remains an important biomarker for toxicity assessment—it is critical for survival of the individual laboratory rat and the species in the wild. According to Stanton, 5 motor activity is the behavioral criterion for neurotoxicity potential of new molecular entities. 6 –8 The inability to reach or use the water lixit device or to retrieve food from the food bin located in or above the cage can be the first reported signs of test article–induced alterations during the scheduled twice-daily health status checks required by the Good Laboratory Practice (GLP) guidelines. 9 In behavioral testing involving rodents, motor function refers to what is actually a large variety of overt behaviors, 5 with motor activity as only one component. 10,11 Under the federal regulatory guidelines, 12 “motor activity” was originally defined in “activity counts” and the assessment was viewed as “an initial step in determining the potential of a substance to produce neurotoxicity” (ie, tier I assay). Assessment of motor function is a predictive bioassay for studying the effects of drug substances from pharmacology, neurobiology, and toxicology perspectives. 13 Motor activity is only one measure of motor function.
Cory-Slechta and Weiss 10 have suggested that reliance on simple “activity counts” may obscure test article–induced differences in the time course of activity changes. In contrast, test article–induced alterations in motor activity are oftentimes the result of abnormal movements, impaired coordination, slowing of responses, or the interference of normal movements by the increased expression of fixed, repetitive, purposeless behavioral units (drug-induced stereotypies). The simple behavioral unit is thus dependent on many different components, which presents unique opportunities for the test article to elicit toxicity.
Is There a Problem in Translation?
In the development of federal regulatory policies in drug (US Food and Drug Administration [FDA]) and chemical (US Environmental Protection Agency [EPA]) approval pathways to licensure, several expert committees 6,7,14,15 recommended the inclusion of locomotor activity testing in the evaluation of safety and toxicity of all new molecular entities. Rafales 16 emphasized the fact that a change in any aspect of the internal milieu of an animal represents a change in what may be relevant information used and operated on by the CNS in establishing the rat’s needs and directing appropriate behavior. 17 –19
Since safety pharmacology screening assays do not require postmortem histopathology verification of structural damage related to any key findings in the tier I assays, the characteristics of the dependent measure of motor function must be reliable, sensitive, and distributed in a way that conforms to national and international risk assessment plans and data interpretation. Tier I assays are designed to give a simple “yes, danger exists” or “no, it is safe to administer to humans” answer. The scale of measurement plays an important part in the certainty of that judgment. Ross 20 has differentiated observational scores into subjective observations and quantitative procedures. For subjective observations such as “alertness,” “gait,” and “demeanor,” the observational data are based solely on the opinion of the technician, much as a neurologist evaluates a human patient.
A change in activity counts provides little demonstrative evidence of specific neural structure pathology. 21,22 The current risk assessment strategies of US federal regulatory agencies have categorized acceptable standard batteries of measurement as either “investigator administered” or “computer administered” (FR50 [157, §3.3.1.1], p. 42373). Under the current ICH S7A guidelines, none of the functional measurements of test article–related changes in CNS-mediated behavioral units are confirmed or verified by histopathology assessments to identify structural or architectural changes in the CNS, but the opposite also holds true. Sophisticated functional tests in animals have revealed significant effects of drugs or brain lesions in animals, which had no gross motor or sensory impairment. 23 –25
Controversy and debate still exist concerning the definition of adversity and safety. 26 –28 The determination of neurotoxicity is two-pronged: (1) the CNS must be affected by dose administration 29 and (2) some adverse event must be documented. Under current regulatory language, “adverse effects” are always deleterious, but the true impact of test article–induced “adverse effects” depends on the duration of recovery of function from these adverse effects when compared to vehicle-treated controls. Recoverable adversity is not necessarily a threat to human safety—anyone experiencing alcohol-induced hangover or the “sluggishness” or “malaise” experienced the day after taking a therapeutic dose of legally prescribed Ambien (Zolpidem) can attest to this. Mattsson 30 has defined “adverse effect” as a diminished ability to perform a function. For most adults, “hangover” from alcohol or Ambien certainly fits this description. Ethyl alcohol is not a legend or controlled drug, and Ambien, which requires a prescription and is a schedule IV–controlled substance, is still considered to be a safe and effective drug to treat short-term insomnia. The observed data should provide the most predictive index of CNS safety with the assurance by statistical power analyses of institutionally based data that have been derived from known drugs that induce reversible and irreversible CNS disruptions of function.
Breaking Down the Problem Into Component Parts
Moser 31 reminds us that histopathological changes alone were the “gold standard” to define the field of neurotoxicology, until the demonstration that toxic substances can also alter nervous system functions in profound and varied ways that required regulatory agencies to acknowledge that other types of evaluations are required to ensure the health and safety of the general public. Under the current administrative policies of the FDA, behavioral observations represent the integration and integrity of the CNS, and therefore, observational data are considered as the most sensitive indicator and perhaps the “industry best” assay of neuronal function. 18,31 –36 The current “industry best practice” for tier I safety assessments is to “cast a wide net to detect any potential nervous system effects,” especially with new molecular entities for which little or no information is available. 31
Detailed Cage-Side Observations
The term “naturalistic behaviors” refers to the expression of behavioral units that animals exhibit “in their natural habitat.” Drug regulators have sought naturalistic behavior as a convenience, hoping that unlearned behaviors could reduce the time and expertise needed to prepare animals for nonclinical safety studies. Eating, drinking, sleeping, and ambulating are certainly “natural” to all animals, but these functions are also subject to the same mechanisms that govern all behaviors, whether in the laboratory or in the field. Evans 37 has argued that behaviors of freely moving animals in standard laboratory caging represents one of the more “natural” preparations available to toxicologists, compared to in vitro preparations of cells or tissue slices; invasive physiological preparations requiring sedation, restraint, electrodes, cannula, and so on, and histopathological preparations are frozen at one instant in time. Many improvements in housing and care of lab animals have minimized concerns with stressors associated with confinement and experimental manipulations that may render the behavior of laboratory animals as “unnatural.” 38 The cage-side observational method provides information about specific activities that are not readily detected by more quantitative photocell equipment (LMA), such as orofacial behaviors, for example, grooming and licking. Under the ICH S7A guidelines CNS, safety is based on a single-dose administration in rats (the preferred species); therefore, there is minimal likelihood that any subset of observations on the standard CNS safety study protocol will provide supportive evidence of true neuropathology. The results of CNS safety must be based on the interpretation of very subtle and mild behavioral changes observed in a less sentient animal whose expression of vulnerability by administration of a toxic agent is limited by a long-standing genetically controlled behavioral repertoire to avoid signs of weakness or vulnerability to predators.
Under the neurotoxicity assessment guidance (Fed Reg. 59(158), p. 42387-42388), much of what is of interest to the regulatory agencies are similar clinical neurological end points of interest to neurologists, in general (veterinary or human patient populations). Table 1 summarizes the observational measures and the type of data engendered by each behavioral unit expected from the federal review agency of interest (the US EPA or US FDA).
Summary of Measures in the Functional Observational Battery, Type of Data Produced By Each, and the Relative Reliability of the Measure in Home Cage of Paired- or Group-Housed Rats.
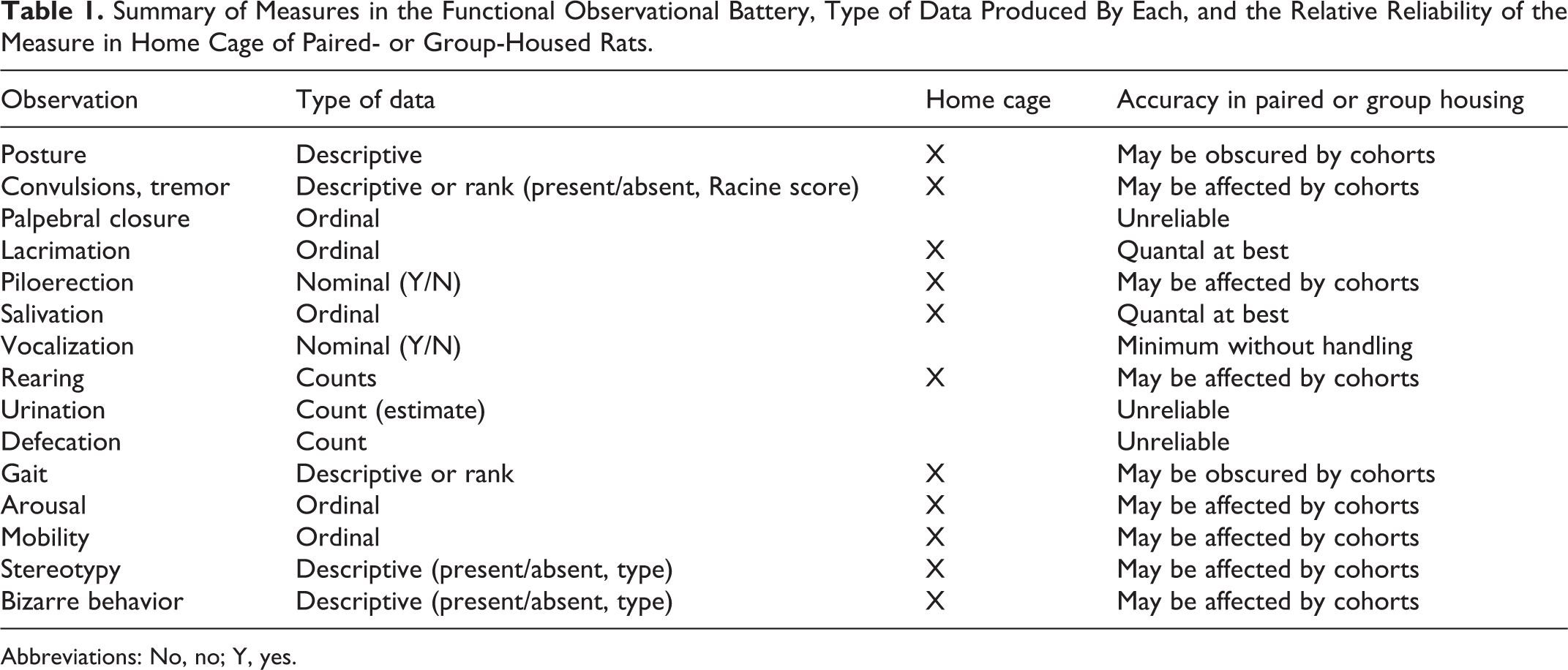
Abbreviations: No, no; Y, yes.
Although some of behavioral units seem to be oversimplistic, the underlying neural mechanisms involved in their expression can provide a valuable tool in the early identification of compromised functions. For example, male laboratory rats spend over 40% of their waking hours grooming. 39 Grooming is an important activity for rodents to maintain their body surface. Maintenance grooming usually occurs in a state of low arousal 40 while mild stress such as exposure to a novel environment also elicits enhanced grooming. 41,42 This novelty-induced grooming is often described as “displacement activity” for lowering a high arousal state. 43,44 van Erp et al 45 have reported that environmental factors induce a physiological stress response in rats within the time frame of standard cage-side observations. Some of the stressors applied are evidently more severe than others, for example, defeat by a male resident versus being placed in a group of conspecifics. van Erp et al 45 demonstrated that repeated handling by the same person reduces the adrenocorticotropic hormone and corticosterone response, which then can be reinstated by a different stressor such as handling by a different person. These data confirm the regulatory-based suggestion that the same observer should be used for each rat for all timed functional observational batteries (FOBs) in the CNS safety assessment protocols.
Evans et al 38 have described several disadvantages of cage-side observation for screening of signs of CNS toxicity:
Observations must be made in the light when rodents are normally inactive.
The observer’s presence can alter ongoing behavior.
Most observations yield ordinal rather than continuous measures, limiting statistical power.
Interobserver reliability is difficult to establish.
Additionally,
5. The standard use of paired or social housing of rats minimizes the opportunity to accurately assess changes in defecation, urination, and food and water consumption.
6. The presence of cage cohorts threatens an accurate observational score of generalized, test article–induced “weaknesses” that would threaten the ethologically based dominance/submissive hierarchy within the dyad or group dynamics of the cage. 46 –49
Signs of lethargy, malaise, illness, disorientation, and so on, will most likely be disguised, masked, or hidden by rats contained within paired or socially housed laboratory rats under cage-side observations. Cage-side observations are valuable, but not truly sensitive to the genetic constraints on behavior of rats administered the test article. In answering the tier I question, “Do we know enough about CNS safety?,” from cage-side observations, the answer is “No, we need more data.”
Open-Field Observations
An assessment of “learning and memory” is a core function of CNS integrity that can be better assessed in the more complex open-field environment. Learning can be defined as a measure of change in motor activity as a result of genetic constraints on behavior and experience. 50 In 1934, Hall 51 described a simple behavioral test for purpose-bred laboratory rats in which the rat was simply placed into a novel environment—a short-walled box, open on the top, to allow for the simple visual monitoring of activity by an externally positioned observer. Many mammals, when exposed to strange or noxious stimuli, will initially “freeze” or remain still. Denenberg 52 has described the freezing or immobility response to have an adaptive significance in that it is more difficult for a predator to observe a nonmoving animal (the response is innate). A second consequence of exposure to such novel stimuli is that the novelty will often initiate a physiological response of the autonomic nervous system (ANS). 53,54 One of the results of such activity is that the animal will defecate and urinate. 55,56 Drugs that impair the rat’s capacity to process environmental stimuli will diminish the magnitude or prevent this initial response to being placed in the open field. As part of the “fight or flight response,” the ANS prepares the rat for attack by diverting valuable blood flow and resources away from the gut and alimentary tract to be ready for the muscular responses required for the impending fight or flight response that is expected to come. 57 Thus, an emotional animal may be defined as one which, when exposed to noxious or novel stimuli, initially does not move about and will defecate and urinate. 58
The open-field assay is often used as an animal model of anxiety-like behavior and permits the evaluation of drug-related effects on different aspects of animal behavior. 59,60 In behavioral terms, if an animal cannot adapt to a changing environment as rapidly as vehicle control cohorts, the increased time required to achieve baseline performance can be considered a measure of an adverse effect. The motor function in open-field activity measures in the rat is influenced by CNS-based mood, affect, and other cognitive functions. If a given screening assay does not adequately or accurately address what is meant as “motor function,” then behavioral changes should not be interpreted as defining the observation as “adverse.” Under the World Health Organization criteria, 15 if a change in motor activity is observed, additional tests are needed to determine the cause. Analysis of general motor activity data, as defined above and collected during the nonclinical safety screening mode, provides little to no information about the origin of the problem with respect to “motor function.” 22,23
The first exposure to the open field has been used to assess the effects of various drugs on the levels of anxiety as well as on nonspecific effects of drugs on locomotor activity. Upon repeated exposure to the open field, animals quickly habituate to the exposure of the arena and their ambulatory movement tends to decrease. 61 Ambulation into the center of the arena and the total number of “rearing” counts are also used as measures of “exploration” and anxiety. Following the initial response to unfamiliar territory, the external stimuli evoke a subjective state of arousal, fear, or anxiety, which then elicit a pattern of behavior characterized by exploration (ambulation and rearing) and by the displacement of other expected “normal” behaviors, such as grooming. 62 –64 Special note should be taken that changes in simple “behavioral units” are not always suggestive of CNS toxicity. As highlighted by Evans, 9 a significant decline in open-field “rearing counts” was not a CNS-mediated toxicity, but rather was correlated with test article–induced increases in the threshold for sciatic nerve stimulation in rats exposed to methylmercury. 9
According to Denenberg, 52 the rationale underlying the use of the open-field test is roughly as follows: Rats have been categorized as “novelty seekers” in that all members of the species display an innate programmed behavioral response when placed in a new environment. The presence of unfamiliar stimuli elicits an “emergent process” of exploration. 65 Price and Huck 66 have confirmed only minimal differences in the open-field behavior of wild and domesticated Norway rats, 66,67 suggesting that the assay is sensitive to innate, genetically based motor engrams that provide for within-laboratory and between-laboratory reproducibility of the data. 68 –72
An assessment of “learning and memory” is a core function of CNS integrity. Learning is defined by a measure of change in motor activity as a result of genetic constraints on behavior and experience. 50 The documentation of learning represents an integral part of assessing psychological maturity, demeanor, sensory processing, and motivation in a living organism. The placement of the rat into the center of the open field is aversive to the rat—it soon escapes the perceived vulnerability to detection of a predator by first moving to the wall or corners to minimize detection (threat). 51 –53 The delay in “taking cover” provides valuable data on CNS integration of sensory/motor functions. 57,58 The “escape” reaction also provides a valuable piece of information with respect to an assessment of the rat’s processing of environment stimuli; its subjective or internal experience of “threat,” “anxiety,” or “fear”; and the motoric effort to diminish the threat (approach/avoidance learning). Berlyne 73,74 highlighted a pattern of “learning” or habituation to this novelty. The animal’s exploratory behavior (wandering, searching, rearing, and sniffing around, as well as approaching a particular feature of the environment) declines with continued exposures. That is, for activity counts and/or the rate of ambulatory or investigative behaviors observed within a scheduled session or between the 3 scheduled days of ICH S7A open-field observations, significant decrements should be expected to occur. 75 The presence or absence of within-session or between-session changes is a function of the schedule of exposure, a critical measure of basic learning processes, even in control group animals. Russell and Williams 76 have reported that activity observed in the first trial of the open field (eg, the predose FOBs, day −1) is influenced by a hyperactivity phenomenon unrelated to exploration that stems from differential habituation of approach and avoidance tendencies (mediated by fear or anxiety), so that the initial decline in activity observed reflects the relatively rapid habituation of approach tendencies to a stable level and the subsequent increase in activity may then be the result of the continued habituation of fear resulting in a decrease in incompatible avoidance effects.
A failure to document these decrements within and between sessions in vehicle or handling control rats should initiate an investigation as to the training and skills of the technicians conducting the open field. If the decrements are not documented following test article administration, the test article is presumed to be a reportable sign of simple learning impairments. This should initiate tier II testing in more complex forms of conditioning paradigms.
Rats innately prefer staying in the periphery to activities in the open center of the arena. Rats are thigmotactic—that is, they prefer walking against the wall to investigate the local area using their vibrissae to explore their intimate life space. Ataxia, catalepsy, limb coordination, and exploratory strategies of rearing, stretch-attend postures, and sniffing bouts provide demonstrative evidence of an intact CNS in open-field tests. Broadhurst 77 demonstrated that critical situational factors of ambient illumination, size of the open arena, and the exploratory time allowed for each rat within a session are critical independent variables in this experimental condition. Grooming behavior is a “displacement response” and is naturally expressed in any novel environment. 78 Grooming serves to allay anxiety in stressful situations—it is self-comforting. 79
Table 2 lists the required behavioral units to be observed and recorded for submission to regulatory agencies for licensure review. There is a dynamic opponent process of motivation competing for the animal’s attention, cognition, and processing capacities within and between scheduled FOBs. As described above, following the initial genetic predisposition to express “freezing” behavior when placed in the center of the open field, the intact rat is expected to process incoming sensory information from olfactory, visual, auditory, and haptic senses. Suddenly, the animal will express elementary patterns of orientation and position itself to ambulate. Once the rat interprets the environment as “safe” and/or cannot identify the presence of a predator, exploratory behaviors will be initiated. Coordinated muscle patterns do not need to be learned in the open field. Hardwired, fixed action patterns of rearing, stretch-attend postures, sniffing, and walking will be initiated, only if the observational period of monitoring is sufficiently long enough to capture these events. In our review of the literature, the standard duration of observation in the open field reported in the peer-reviewed scientific literature is 2 to 10 minutes.
Summary of Measures in the Functional Observational Battery, Type of Data Produced by Each, and the Relative Reliability of the Measure in the Open-Field Arena.
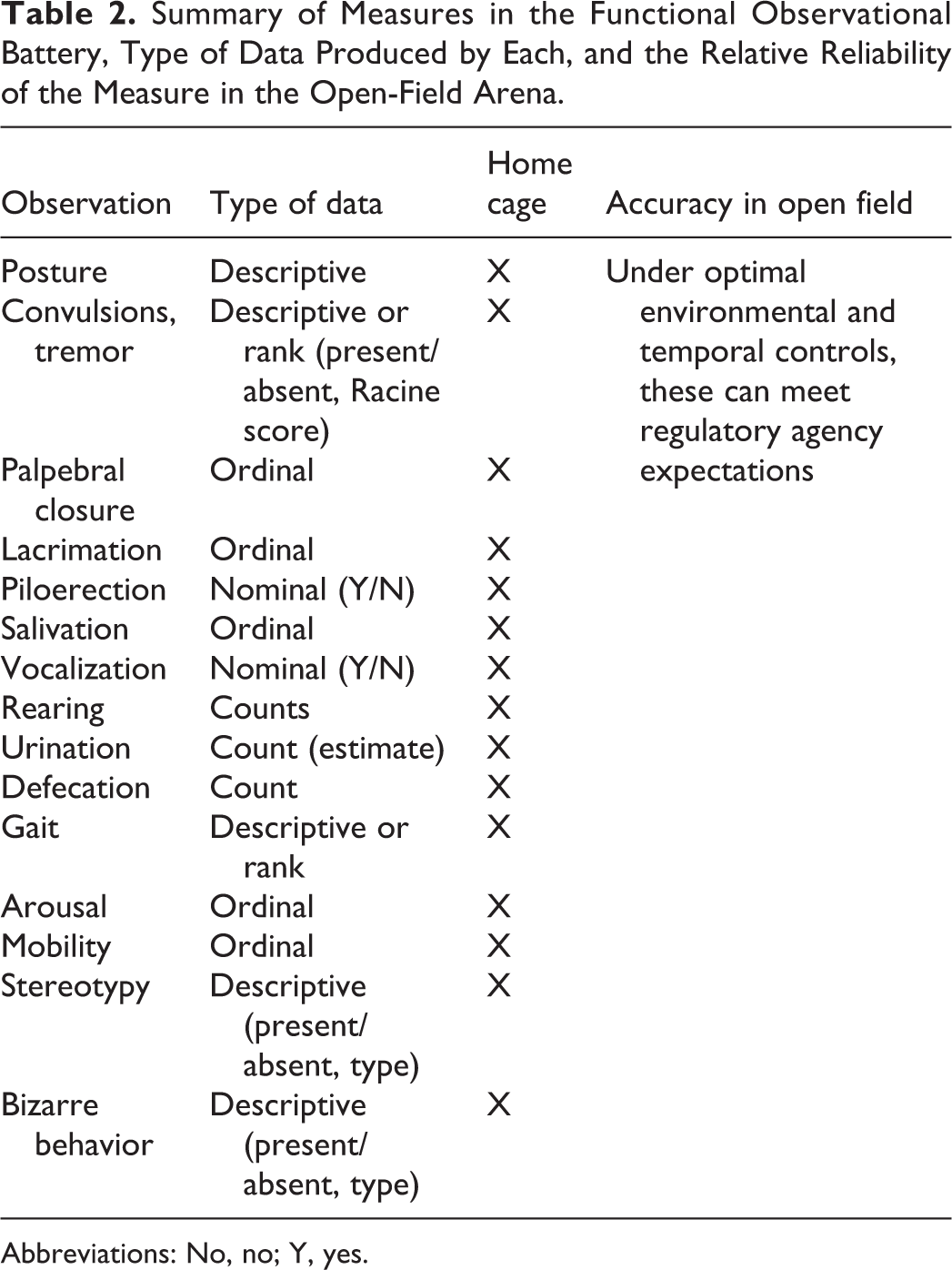
Abbreviations: No, no; Y, yes.
As shown in Figure 1A and B, the rate of ongoing ambulatory (horizontal) movements and the physical location of the rat vary over the 10-minute observation window. Once placed into the center of an open field, the rat moves to the close proximity of the walls of the box (fear or anxiety driven, within the first 20 seconds; 16% of 120 seconds). The rats remain actively moving in the chamber exploring the novel environment. The rate of movement and the distance of mobility from the wall are inversely proportional to the interval of observation up to midway through the 10-minute window. Experience within the box results in increased time spent away from the walls and toward the center of the arena (less anxiety). At approximately 6 minutes, the activity level of the rats decrease to a nadir (Figure 1A and B, right panel) and the rat portions the movements equally to wall area and open area of the field (approximately 50%-50%). Interestingly, during the last half of the 10-minute open-field observation period, the rats seem to show an ethologically based balance between the “novelty-seeking” and “predator/prey” mobility and migrate back to predominant wall walking for the last 4 minutes of the 10-minute observations. While Figure 1A suggests that there is a point of diminishing returns using open-field assessment durations of greater than 3 to 4 minutes, the “U-shaped” functions of Figure 1B provides for the temporal constraints of “simple learning,” also referred to as “habituation.” One important feature of toxicity testing is the assessment of test article–induced changes in measures of “learning and memory.” Although this is not a critical feature of tier I safety pharmacology testing required prior to the first delivered dose in man, extending the observation period of the open field to 10 minutes in subsequent toxicology study designs may be helpful.
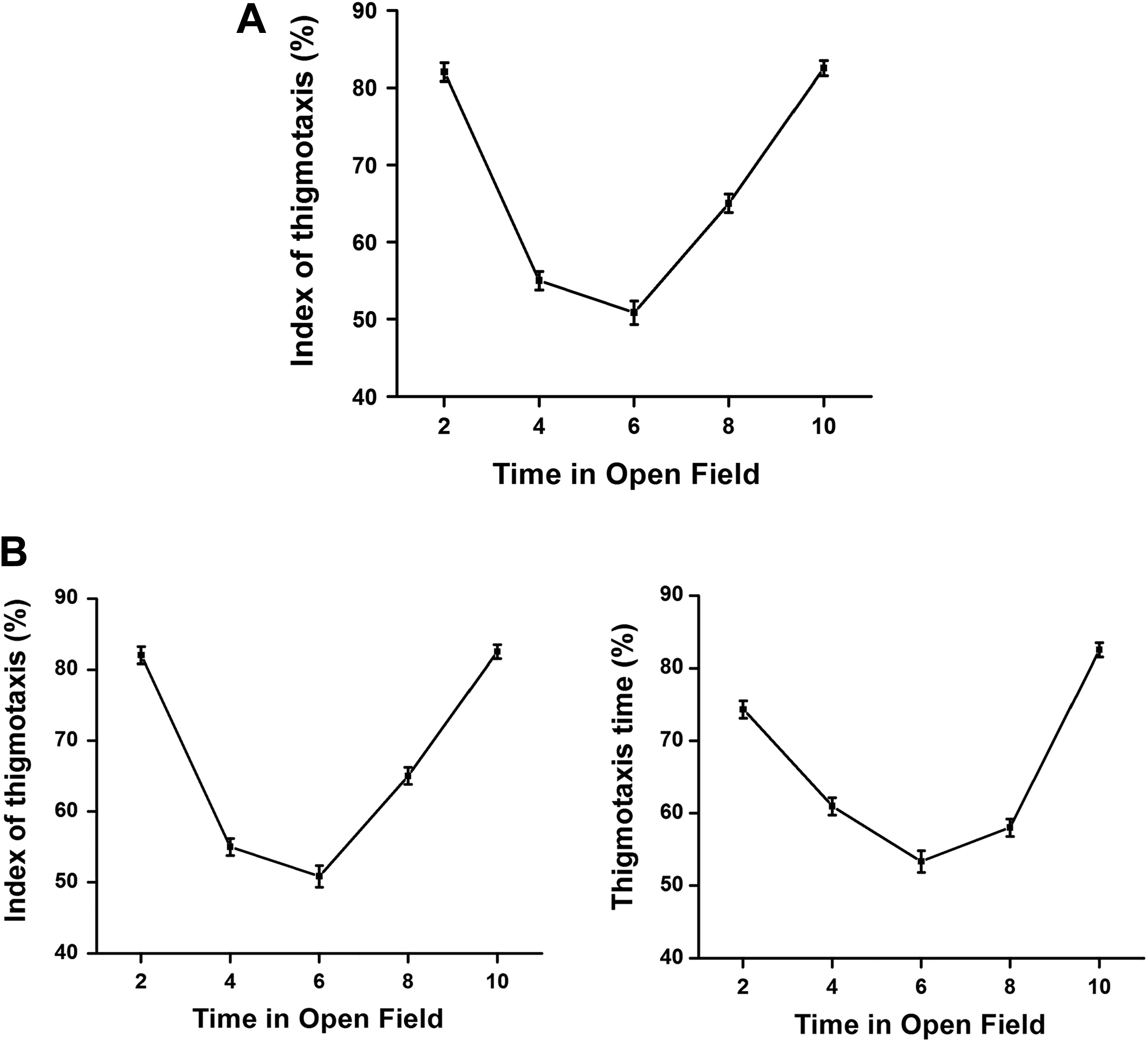
A, The amount of activity during the first few minutes averaged over 10 different daily trials of a standard open-field assessment in rats. Activity in the open field is a negatively accelerated function of time. There is a point of diminishing returns over the first 5 to 6 minutes of the open-field assessment. 80,81 The most valuable data from the open-field measure of the FOB are achieved within the first 2 to 3 minutes of the observation period. B, Assessment of the behavioral changes in the open-field test of adult rats was previously reported by Ouhaz et al 82 and adapted for use. The assessment of thigmotaxis patterns over 10-minute in the open field by 14 control rats is shown. An “index of thigmotaxis” was calculated as the ratio of ambulatory traveled distance (in centimeters) within a restricted area of less than 2.5 cm away from the walls. The total distance traveled in 2-minute intervals was expressed as a percentage of total session distance (in centimeters) for the full 10-minute observation period (eg, 2, 4, 6, 8, and 10 minutes; left panel). The “thigmotaxis time” was calculated as the ratio of time spent near the walls (less than 2.5 cm from the wall) during each 2-minute interval of the 10-minute open-field observation period (right panel). Each point represents the group mean (1 standard deviation) for each 2-minute monitoring period. FOB indicates functional observational battery.
The simplicity and cost-effectiveness of the open-field test is a main reason for its inclusion in the standard CNS evaluations; however, the assay is not free of “contamination”: The opportunity for direct observations of rats in the open field must be of sufficient duration to allow for exploration and the documentation or proof or learning (habituation). The time frame for scoring of rats in an open field must be sufficient to allow for the full assessment of “natural behaviors” of a rat under the self-perceived threat and idiosyncratic motivation to explore the open space. As part of the FOB, the open-field test is conducted in the same time frame as the cage-side observations. Retrieving rats from their home cage and transporting to the open-field arena may stimulate defecation and urination during retrieval or transport that is not quantified in either home-cage or open-field data sets. The reason there is no fecal boli in the open field may be the result of the rat purging the bowel and bladder at the time of removal from its home cage or in transit to the open field. The documentation of behavioral change must be conducted by a human observer in close proximity to the arena; the physical locality of the open-field arenas is generally within the same testing room as other rats in the study which exposes the animals to the smells and sounds of multiple sources. Distracting noises of conversations between technicians (regardless of the intended loudness of the vocal communications within the room) can interfere with free expression of normal rodent behavior. Interactions between observers remain vital to ensure smooth unfettered access within the room without bumping into cage carts, open cages, or established transportation routes (pathways) to and from cages and open fields within the room with many technicians—these still have the potential to distract the rat; other interfering events (eg, blood draws in the observation room) should be avoided. The repetitive and random presentation of “auditory clickers” used for startle testing is not restricted to the close environment of the open field. To serve as a functional auditory startle stimulus, the clicker must be of a minimum sound level that cannot be muted between testing stations. During short pretreatment intervals on day 1 testing, there will be animals being dosed at the same time as their cohorts are being observed in the open field, along with analgesia testing, grip strength testing, and so on. Vocalizations of stressed cohorts cannot be fully masked by ambient white noise.
In a review paper by Walsh and Cummins 83 on the use of the open-field assay, they highlight the fact that there is little or no consensus on the interpretation of the individual behavioral units measured in the 2- to 10-minute observation period. The use of the open-field assay has also been criticized on the significant variability of the data recorded, the variability between laboratories in the duration of the observation period, and the opinion that the observations are not sensitive or show specificity in CNS-mediated control. 21,22,83 –85 Foss 84 has concluded that although the economic and efficiency of the open field is a major reason for its continued use in CNS safety protocols by drug developers, the assay retains limitations as the means to identify critical factors that determine the CNS effects. For Foss, 84 it is unlikely that an understanding of the true CNS safety factors would ever come from a single tier I study prior to the allowance of the first dose administration to humans in phase I clinical trials. Tier I safety testing does not require postmortem histology. With the lack of professional pathologists’ review and interpretation of neurohistologic data, the open-field neurotoxicity screen under ICH S7A does not provide much opportunity to identify the links between the observation in the open field (FOB) and motor activity test (LMA) and data from other levels of analysis that might lead to an explanation of mechanisms of toxicity, such as acute and repeat-dose toxicology studies. 84 The reliability of the tier I safety pharmacology test could be improved by expanding the range of the “shot-gun approach” to include the quantitatively and objectively measured third battery of locomotor function—the automated 3-dimensional activity count assay. 86 –90
Tier I Testing: Do We Know Enough Without LMA?
Figure 2A through F shows observational scores of behavioral units required for regulatory submissions related to both cage-side and open-field assays. The standardized subjectively rated scores for nonmanipulative behavioral units from an FOB were conducted during institutional positive control article tests (for details refer to Gauvin et al 23,84,85 ). All observations were conducted by trained technicians who were blind to the treatment doses administered. In an independent group, repeated-measures design cage-side and open-field observations were conducted as part of Institutional Animal Care and Use Committee–approved CNS safety pharmacology protocols, to assess baseline values (day −1), acute behavioral effects (day 1, 30 minutes postdose), and any residual behavioral effects (day 2, 24 hours postdose). Data were generated to serve as “positive controls” in the Safety Pharmacology Department at CRL Laboratories (Mattawan Campus). For illustration purposes, only nonmanipulative observational scores are shown from independent groups of 10 male Sprague Dawley rats (CD-1; Charles River Laboratories, Inc, Wilmginton, MA) that were administered saline (vehicle, subcutaneously [SC]), amphetamine (3 mg/kg, SC), haloperidol (2 mg/kg, SC), caffeine (60 mg/kg, SC), or pentylenetetrazole (PTZ; 60 mg/kg, SC).
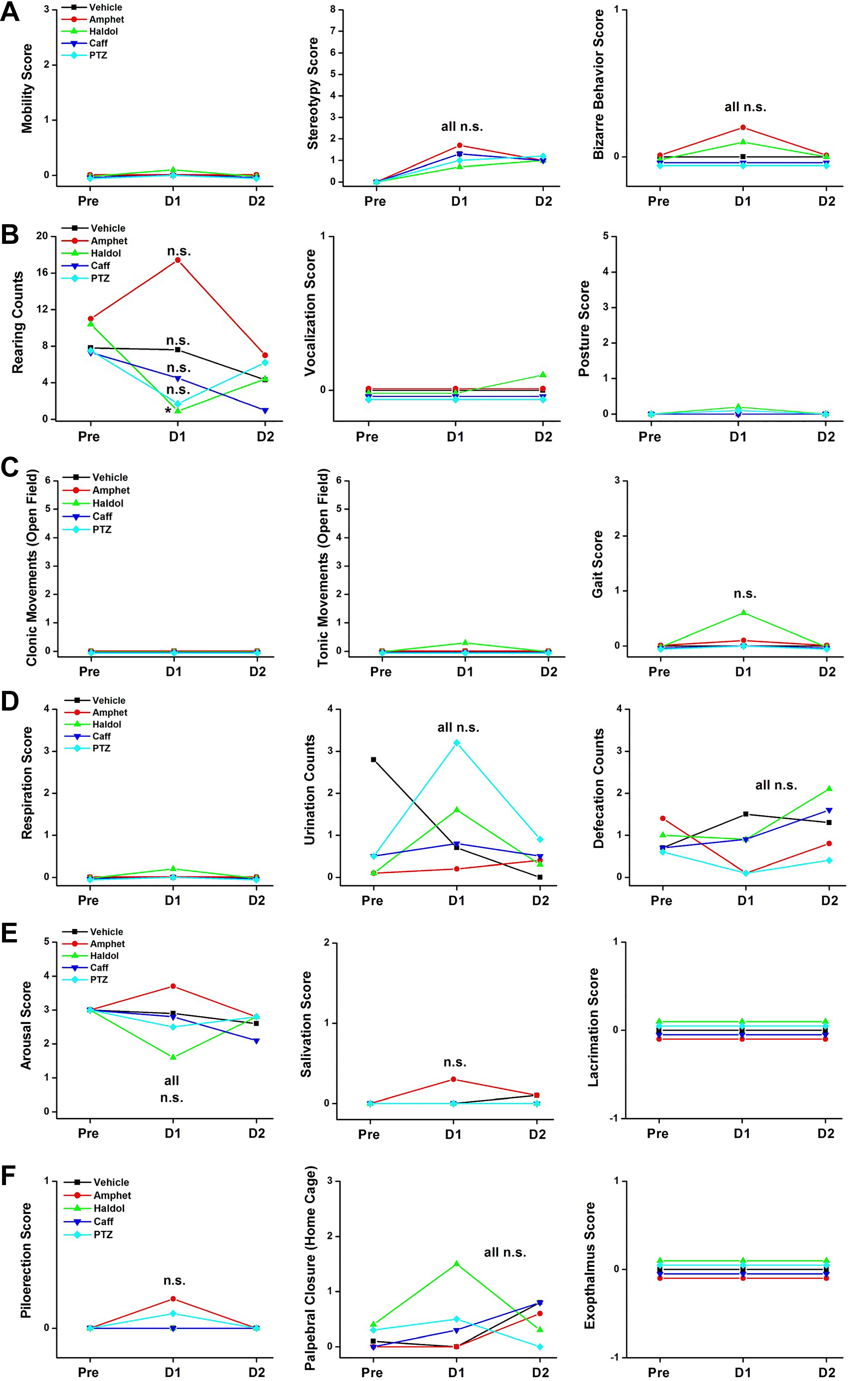
A-F, Eighteen behavioral parameters rated by trained technicians during cage-side and open-field assays of a standard ICH S7A CNS safety assessment FOB. These 18 behavioral observations are documented while the rats are unrestrained. Of total, 16 observational scores are documented based on subjective ratings by trained technicians. Two of the observations (defecation and rearing scores) are quantitative counts during the open-field assay. CNS indicates central nervous system; FOB, functional observational battery; ICH, International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use.
The basis for dose selection of these common positive comparator drugs for S7A CNS safety evaluations is listed in Table 3.
Drugs Selected for Historical Positive Control Data and Justifications for Dose Selections.
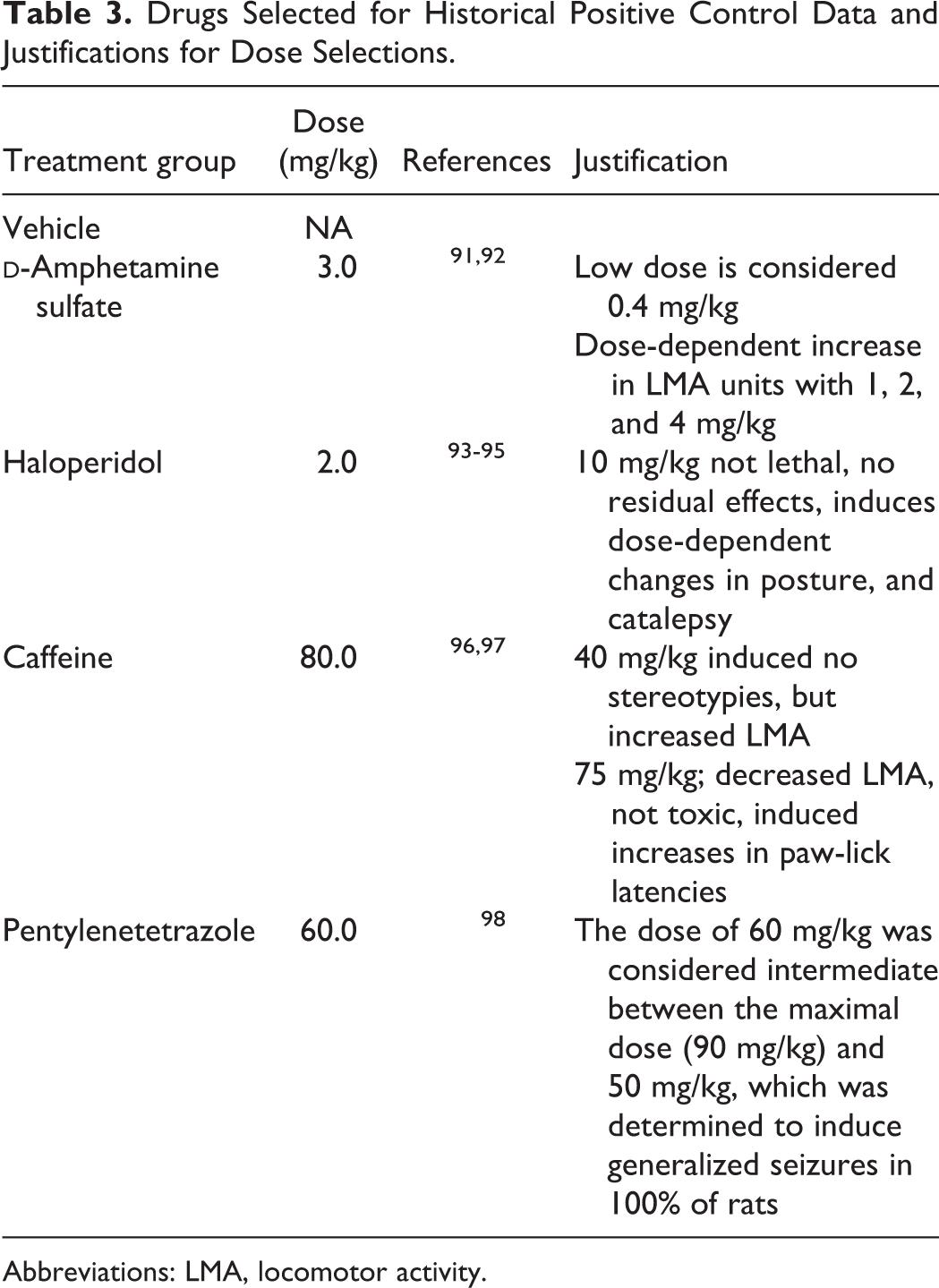
Abbreviations: LMA, locomotor activity.
As described by many authors (see above), there is much variability in subjectively based observational scores in the rat FOB. There were no statistically significant changes in any of the 18 observational parameters assessed in the standard home-cage and open-field assays in the FOB. With respect to the 4 positive control articles selected for comparison and the regulatory intent of tier I CNS safety pharmacology studies, the most striking point of interest may be the lack of differentiation between the dopamine agonist, amphetamine, and the dopamine antagonist, haloperidol. The first expected interpretation from a reviewer of these data may simply be to conclude that the technicians in this laboratory were not accurately trained, they obviously did not conduct a valid or reliable FOB, and the failure to differentiate these drugs in the open-field arena is “proof positive” of an “observational failure.” We would not agree with this conclusion, as explained below.
In 1960, the research laboratory of Janssen Pharmaceuticals, the manufacturer of R1625 (haloperidol), reported the results of testing the dopamine antagonist on the behavior of rats in an unfamiliar “open-field” situation using a similar 3-minute observation period as was conducted. 99 In that paper, the manufacturer of the drug reported similar observations of “lower scores” in defecation, rearing, and ambulation without producing ataxia in rats administered doses of 0.01 to 2.5 mg/kg haloperidol. However, Bernardi et al 100 reported no statistically significant differences in open-field parameters of locomotion, grooming, rearing, or defecation in rats treated with 2.5 mg/kg Haldol when compared to vehicle control cohorts.
In contrast to these haloperidol data, open-field observations conducted with the dopamine agonist, amphetamine, in Sprague Dawley rats were reported by Jennings.
101
Rats treated with 4 mg/kg amphetamine were observed to have no significant effects on rearing, defecation, or urination when compared to vehicle-treated cohorts, and the drug group, unlike the saline group, showed an absence of feeding, grooming, and freezing; however,
The ICH S7A standard FOBs generally include manipulative parameters that require specialized instrumentation/equipment. 102,103 Figure 3A and B shows 6 dependent measures that must be conducted with the rat with some degree of handheld restraint.
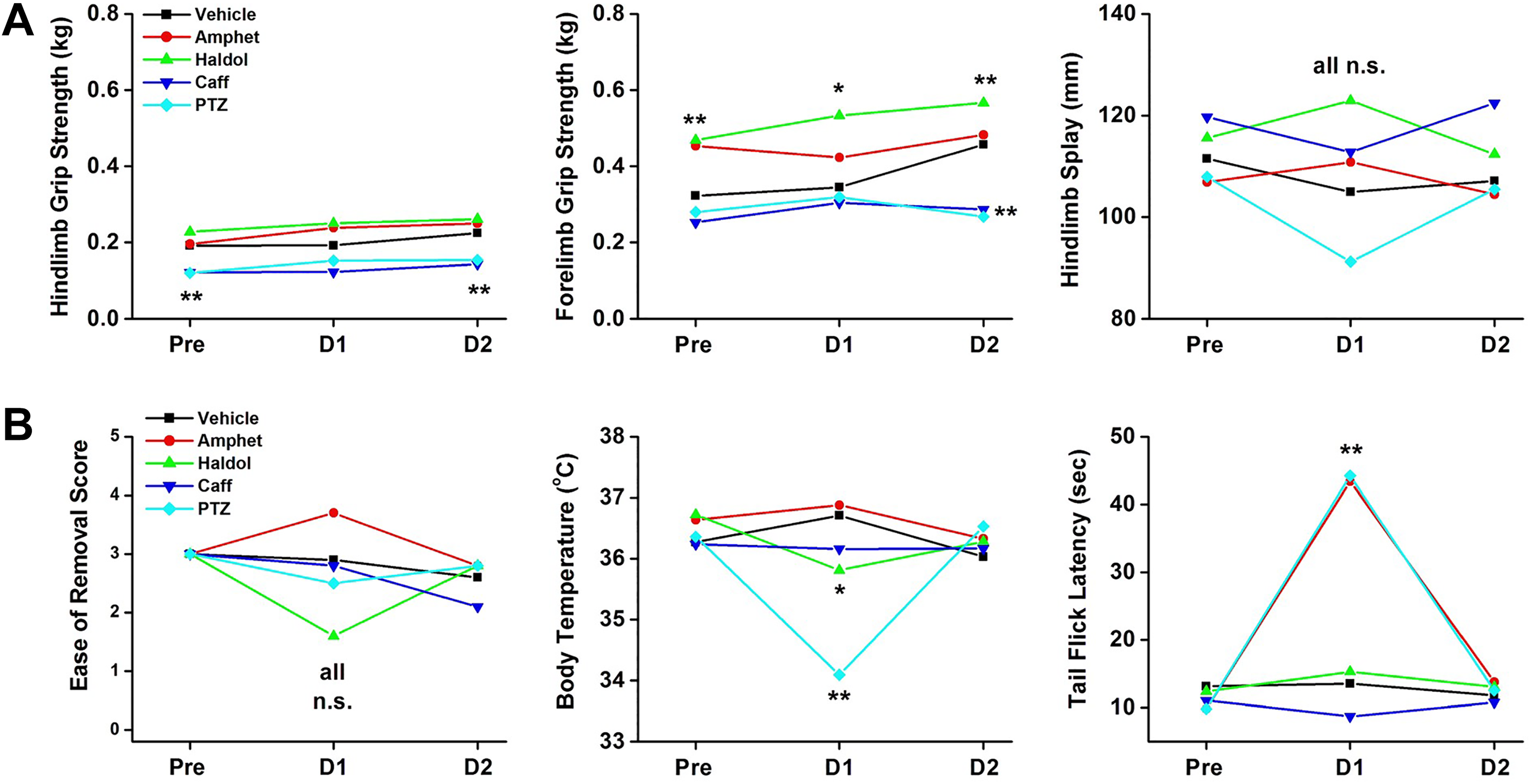
Six additional observational units of measure included in the standard ICH S7A CNS Safety assessment FOBs while the animal is held by the technician. Five measures of neurological function are quantified using calibrated equipment (body temperature, tail-flick latencies, hind limb and forelimb grip strengths, and hind limb splays). The “ease of removal” and handling of the rat is a subjectively rated observation. CNS indicates central nervous system; FOB, functional observational battery; ICH, International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use.
Amphetamine, Haldol, caffeine, and PTZ did not elicit significant changes in subjective scores for “ease of removal” from the home-cage or open-field arenas. Haldol (P < 0.05) and PTZ (P < 0.01) induced decreases in core body temperatures, with no significant changes in core body temperatures recorded in amphetamine-, caffeine-, or vehicle-treated rats over the 3-day monitoring period. Tail-flick latencies (analgesia scores) were significantly increased by stimulants, amphetamine (P < 0.01), and PTZ (P < 0.01), when compared to vehicle cohorts, with no changes in response to thermal stimuli in Haldol-, caffeine- or vehicle-treated cohorts.
Figure 3B highlights the interpretative problems of “between-group” and “within-group” data analyses of behavioral measures that characteristically have high variability. 102 Hindlimb grip strengths did not change from baseline values when measured on day 1 (acute dose effects) or day 2 (residual effects). Time–effect functions were parallel across all groups. However, PTZ and caffeine groups were statistically lower at baseline (day −1) and recovery (day 2) when compared to vehicle control cohorts.
Figure 3B, middle panel, shows the changes in forelimb grip strengths across the 3 days of monitoring. Vehicle control rats showed a day-to-day increase in grip strengths typical of simple practice effects (learning) over 9 trials over 3 days. Haldol-treated rats showed higher baseline grip strengths (P < .01) when compared to vehicle controls that were also increased by practice effects on day 1 (acute effects) and day 2 (residual effects). Caffeine and PTZ induced lower grip strength values on day 2 when compared to vehicle control cohorts. Grip strength readings reflect physiological function (ability to grasp) as well as performance effects (willingness or motivation to grasp). And finally, Figure 3B, right panel, shows the different hindlimb splay values for the 4 treatment groups across the 3 days. The variability in these data prevented the group means to reach statistical significance. Therefore, only one parameter of the “manipulative end points” helped to distinguish between these 4 positive controls.
Considering these findings of tier I screening data results, the question at this point must be answered, “Do we know enough”? With not a single differentiating variable among the 18 subjectively scored parameters in the FOB, and the single significant differences in manipulative-related data with inherent within-group and between-group variability, the answer clearly is “No, we need more data.”
Value Added by Automated LMA
The ICH S7A, FDA “Redbook,” and US EPA guidelines do not dictate the assessment of any one particular behavioral category or the use of any one specific type of testing apparatus to assess CNS neurotoxicity and safety. However, the Interagency Committee on Neurotoxicity (ICON) guidelines 8,104 do specify that recordings of locomotor activity be automated, that the behavior of individual animals be observed, and that the automated recording session be long enough for the dependent measure to reach an asymptotic level in vehicle control rats. The ICON guidelines also call for institutional historical control data showing both increases and decreases in the dependent measure produced by positive control articles. From the regulatory agencies’ perspective, the use of automated (infrared photobeam) assays requires the physical transportation of rats to the monitoring chambers and thus quantifiably measures the rats reaction to (1) novelty, (2) a dark environment, (3) ambulatory and voluntary movements free from interference associated with the presence of a human observer (open field), or (4) the presence of another rat(s) in close proximity (home-cage observations). Additionally, the “activity counts” are measured in freely moving rats for a longer duration of time relative to home-cage or open-field arena assessments. While the ICON-adopted neurotoxicity testing “final rule” lists the use of automated equipment for regulatory submissions, in today’s regulatory environment, the federal guidelines are considered as “guidance” only. These administrative guidance documents are not legally binding on pharmaceutical manufacturers nor on the FDA.
Automated recording of animal behavior affords several advantages over human observation: (1) it can be less labor-intensive, (2) it is compatible with the automated data recording demanded by GLP regulations (§58.61 to .63), (3) it reduces human error and differences between individual observers, and (4) it can measure behavior in the dark, when the rodent’s full behavioral repertoire is on display. According to Moser, 105 it is essential to maintain experimental control over all possible testing conditions to minimize variability. Stability of neurobehavioral measures of CNS safety is defined by the variability in the data. As just summarized for 18 different FOB-observed parameters in the cage side and open field of the standard FOB, the variability induced by (1) the differential levels of measurement used (nominal, rank, interval data); (2) the variability between subjectively scored observations versus objectively measured ratings; (3) the variability between rats within the same treatment group; (4) the variability between predose, day 1, and day 2 FOB collection intervals; and (5) the variability between FOB data from tier I safety screens and the subsequent FOB data collected in acute, subchronic, and chronic toxicology studies 21,22,83 –85 can fail to make meaningful distinctions between drugs with varied mechanisms of action.
According to Lynch et al, 106 automated motor monitoring systems may be a better-suited assay for the screening and comparison of the larger number of compounds available during even earlier stages of development. For practical considerations, such as relative cost versus degree of versatility, drug discovery programs should consider these automated systems to use for the testing of target compounds. If data are unstable, true drug-induced changes may not be statistically significant, and regulatory agencies may wrongly conclude that the drug has no CNS risk or that the tier I testing battery is not adequate to move the drug down the pipeline to human exposure testing. 100 In single-dose administration testing in S7A safety pharmacology protocols, the highest degree of experimental control is necessary to achieve low variability (high stability) in all groups of rats, especially when the sample size of most CNS safety studies are most often being reduced from the ICON guidance recommendation of using 10 male and 10 female rats per treatment group. For example, there should be a minimal expectation of significant changes in measures (baseline vs day 2 data) involving muscular strength, coordination, balance, stride, gait, coordination, and body weights in a single-dose administration CNS safety study that is usually conducted over a 3- to 4-day period with the caveat that the dose range being tested is within the expected therapeutic dose range to be delivered to human volunteers in phase I studies. For example, Moser et al 107 have previously reported the results of a 3-month toxicity study in rats in which body weight gains were recorded consistent with “normal” growth curves of the rat strain used (approximately 200 g), with no significant changes in grip strength between predose measures and those quantified on day 91 of the study. Should we expect the same in a 3-day safety pharmacology study?
The automated data collection system provides measures of activity counts (horizontal plane, basic and fine), rearing counts (vertical plane), and the total distance traveled by the rat (Figure 4) for a longer duration of time than the open-field and cage-side observational periods (120 minutes). The LMA system provides a different perspective of motor function when compared to the 18 observational units of the cage-side and open-field assays. A wider array, as well as more complete assessment of motor function in the rat, is provided when all 3 experimental methodologies of behavioral observations are included in tier I CNS safety study protocols (Figure 4).
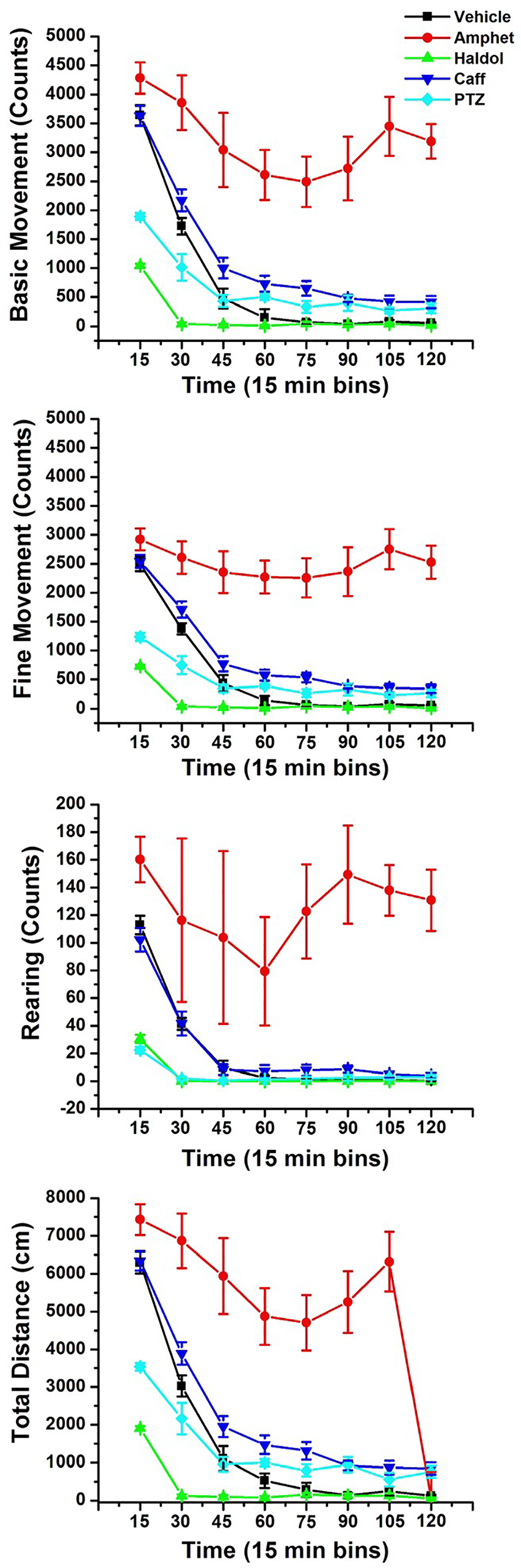
Automated LMA measurements for vehicle control rats and 4 positive control articles, amphetamine, haloperidol (Haldol), caffeine, and pentylenetetrazole (8 male rats per treatment group). Photobeam arrays measured horizontal and vertical beam breaks in each rat for 2 hours following SC dosing. Photobeam chasis contained 16 photobeam paired arrays to assess x- and y-axis movements. Basic movement, fine movement, and rearing counts, as well as total distance traveled in Kinder Scientific open arenas were continuously recorded and stored in 1-minute bins. Data were further summarized into 15-minute bins over the 120-minute observation periods. LMA indicates locomotor activity; SC, subcutaneously.
Using the LMA chambers, amphetamine-induced increases in ambulatory movement counts, rearing counts, and total distance traveled (red closed circles) are clearly dissociable from the haloperidol-induced decreases in all the same measures (green closed diamonds). Additionally, the psychomotor stimulant caffeine can be differentiated from amphetamine. Caffeine-induced increases in basic and fine movement counts as well as the total distance traveled over the 2-hour monitoring period (blue inverted triangles) can be differentiated from vehicle control cohorts (solid black circles) and are relatively lower in magnitude when compared to the pure dopamine agonist, stimulant, amphetamine. It is also clear from these data that caffeine does not induce the magnitude of increases in rearing counts compared to amphetamine-treated cohorts. The dopamine antagonist, haloperidol, induced decreases in both basic and fine movement counts and rearing counts and the total distance of ambulation over the 2 hours of monitoring when compared to vehicle control and stimulant-dosed cohorts (both caffeine and amphetamine). Pentylenetetrazole, a proconvulsant, shifted the time–effect functions for all 4 measures of activity to the left and downward when compared to vehicle control rats.
Relevant to the present discussion when the total weight of evidence from (1) cage-side, (2) open-field, and (3) LMA data is reviewed, it should be evident that although haloperidol induced significant decrements in automated locomotor activity in the motor monitoring system, haloperidol is not a “sedative.” The term “sedation” generally refers to CNS depression. Older sedatives were “soporifics”—sleep inducing (like barbiturates). Sedatives induce ataxia, poor coordination, limp body tone, lethargy, sleepiness, hypoactivity, and reduced orienting responses to external stimuli. A decrease in ambulatory movements does not necessarily indicate “sedation.” Behavioral arrest or movement suppression is not “sedation.” There is no single circuit in the brain responsible for the termination of movement, 108 but an immobile rat is not necessarily a sedate rat.
Through evolution of predator/prey, dominance/submission adaptation strategies, “freezing” is activated by the CNS in response to environmental stimuli designating “threat.” “Freezing” or tonic immobility is a universal fear response observed in rats and humans and is a reaction to conditioned (learned) or unconditioned (genetic) threatening stimuli or situations. 109 A strong candidate system for action termination is the basal ganglia. Patients with Parkinson disease, a disorder of dopaminergic control, often show signs of immobility or movement arrest. Haloperidol is a dopamine antagonist.
During “freezing,” 2 counteracting branches of the ANS become activated; the tonic immobility is a sympathetic nervous system activity that increases arousal. Hypervigilant searching for the presence of danger, by sniffing, visual scanning of the area, and echolocation using the pinna and auditory pathways, is met with increases in heart rate and cardiac output, increased arterial pressure, and increased muscle tone and pain suppression—none should be interpreted as “sedation.” In opposition to the sympathetic response to being placed into the open field, the immobility elicits the parasympathetic branch of the ANS to initiate heart rate deceleration or a reduced heart rate acceleration (refer to the study by Roelofs 110 ) The push and pull of 2 competing ANS responses to maintain homeostasis, freezing behavior is observed when parasympathetic drive dominates over sympathetic drive. Freezing is associated with altered respiration rates (which are normally too high to visually count in the rat), which initiates vocalizations in the ultrasonic range. At the moment that the rat shifts to ultrasonic vocalizations, respiration rates drop because the vocalizations require longer periods of exhalation (peak expiratory flow, end-expiratory pauses). Besides a decrease in mobility (ambulation), freezing is characterized by bradycardia. “Freezing” was originally referred to as “crouching” and contrary to sedation is associated with increased muscle tone (ie, as in catalepsy).
In the open-field and cage-side observations data above, haloperidol did not show significant changes in mobility, posture, and gait scores and showed no changes in clonic or tonic movement scores. In the manipulative observations of the FOB (data not shown), haloperidol rats were scored similarly to vehicle controls on (1) ease of removal, (2) handling reactivity, (3) approach response, (4) click response, (5) tail-pinch response, and (6) touch response scores. Using all 3 observation methods, the decreases in basic, fine movement counts as well as the decrease in rearing counts are more demonstrative evidence for a diagnosis of “catalepsy” and not “sedation” (refer to the study by Lorenz et al, Lorenz and Kornegay, Fahn et al, and LeDoux 111 –114 ).
The diagnosis of “catalepsy” is a motor disorder partially based on the findings that the rat is hypoactive. The rat displays normal postures. The cataleptic animal is not asleep, it will react, arouse, or respond to an appropriate stimulus (eg, normative approach, click, and tail-pinch responses). The cataleptic rat is not flaccid (sedated), the muscle tone may be rigid, but the animal can react in the open field. Only by integrating all observational units from cage side, open field and automated activity monitoring can a reliable differentiation between catalepsy and sedation, which can be supported by the data presented in this study. Haloperidol and other typical neuroleptic agents are capable of nearly completely inhibiting ambulation and rearing without producing ataxia, and the rat does not seem to attend to other competing environmental stimuli. A similar effect is observed in the human patient. A loss of slightly motivated, coordinated, motor activity, such as a lack of mimic or monotony of facial and bodily expressions, is often reported in haloperidol-treated patients who are not characteristic of CNS sedatives (refer to study by Janssen et al 99 )
Pentylenetetrazole has been classified as a general, nonspecific, CNS stimulant that is capable of strong excitation of the CNS. The stimulation may be accomplished by one of 2 general mechanisms: by blockade of inhibition or by direct neuronal excitation (which may involve increased neurotransmitter release, more prolonged transmitter action, labilization of the postsynaptic membrane, or decrease in synaptic recovery time). Löscher et al 115 have reported that rats that receive a dose between 70 and 90 mg/kg by SC injections will express generalized clonic seizures. A dose of 60 mg/kg PTZ was administered in the present LMA study data, as described above. Klioueva et al 116 have reported that rats of 6 to 8 weeks of age will express tonic/clonic convulsions within 12 to 18 minutes of dosing 50 mg/kg PTZ (2 doses of 25 mg/kg). A pretreatment interval of 30 minutes was used in the cage-side and open-field observation data collection described in the LMA data, as described above. Based on the reported kinetics of the drug, rats in this group most likely expressed seizures during the pretreatment interval prior to the protocol-required initiation of observations. For all intents and purposes, rats observed at 30 minutes postdose were most likely “postictal” and displayed a hypoactive lethargy typical of “postictal depression”—a period of quiescence following a tonic/clonic (grand mal) seizure. The PTZ-treated rats showed decreases in rearing scores, lower arousal scores, and reduced “click responses” in the open field. When tested on the hotplate test for analgesia, PTZ-treated rats had an escape response latency of over 44 seconds compared to the 13-second response in vehicle controls. Pentylenetetrazole is not an analgesic; the postictal rat most likely can’t evoke an escape response or lift its paw on the hot plate. These observations support a conclusion that rats were in postictal depression and were compromised in CNS processing of sensory stimuli. When rats were dosed with PTZ and placed into the motor monitoring systems, they showed reduced activity counts compared to vehicle controls; however, the beam break counts for basic and fine movements were greater than Haldol-treated cohorts during the first 15 to 30 minutes of recording period. These photobeam breaks may have been initiated by the expression of tonic/clonic seizure-related obstructions of the photobeam arrays.
Overall, when the totality of evidence is reviewed, the FOB and LMA activity data are completely consistent with the known pharmacology of the drugs used in the study and similar to the behavioral effects described in published literature from other laboratories appearing in peer-reviewed scientific journals.
Discussion/Conclusion
Human factor analysis data confirm that human attention span is limited. Tier I CNS safety screening assays must provide the most effective risk assessment of a new drug as possible. The LMA system allows for each rat to be monitored for 2 hours in low ambient light, outside the immediate environment of a human observer and without exceeding the memory storage capacity of the computer software. As we have previously highlighted, Cornish and Dukette 117 reported that adult technicians can only sustain attention for about 20 minutes. Also noteworthy is what happens within the 20 minutes that adults are able to maintain “sustained attention.” Cutting et al 118,119 reported a natural rhythm to human attention that requires the technician to experience a stimulus change every few seconds in order for maximum attention to be maintained. There is a vast and venerable literature on the deleterious effects of work shift duration on both attention and cognition (refer to the study by Gauvin et al 120 for references). “Mind wanderings” can be viewed as lapses of executive control as unrelated stimuli (external and internal) compete for attentional resources. The timing for cage-side and open-field observations of the CNS safety FOBs are generally scheduled for 6 to 8 minutes in duration for each animal—much less time than the 20-minute duration of sustained attention, so for each animals’ observations, there should be no valid argument for attention deficits within a given animal’s observations. However, the concern is how reliable are subjective rating scales scored by technicians conducted in the experimental testing room with the requisite need to minimize communication, loud noises, and other competing stimuli. As CNS safety assessment under ICH S7A, there is no requirement for comparative analysis of structural or anatomical changes in nervous system integrity that may correlate with differential behavioral rating scores in the FOBs.
Overall, the observations of the study technicians are overseen by a supervisory technician and/or study director. As identified by Ross et al, 121 under current regulatory rules, there are no clearly defined characteristics of the background, education, or experience required for adopting the title of study director. Also noted by these authors is the lack of any centralized board of authority, academic nor industry, that sets standards, definitions, or guidances on observational parameters used for regulatory review and licensure for conducting tier I safety protocols. Detailed observation of animals needs the highest level of expertise, since many behavioral changes are transient (eg, only observed during day 1 postdose monitoring), so that it is nearly impossible to obtain post hoc peer review without repeating the full study.
We take the position that in the best interest of compliance with the ICH guidelines and other federal regulatory agency directives, tier I CNS safety studies should be held to the highest industry standards of practice possible. Reducing group size, limiting observations to only a single sex, and excluding parameters of motor function testing using automated LMA chambers are not in the best interest of public health and may provide inadequate safety assessment prior to the first dose administration in phase I clinical trial volunteers. The full panoramic view of the behavioral effects of new drugs that may serve as biomarkers for neurotoxicity is only assured by maintaining the “shot-gun approach” adopted and promulgated by the US Federal Interagency Committee on Neurotoxicity, 8,121,122 which includes the use of automated motor monitoring systems.
Footnotes
Author Contributions
David V. Gauvin contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. Zachary J. Zimmermann contributed to conception and design, contributed to interpretation, drafted the manuscript, and critically revised the manuscript. Joshua Yoder contributed to conception, contributed to interpretation, drafted the manuscript, and critically revised the manuscript. Jill A. Dalton contributed to conception, contributed to interpretation, drafted the manuscript, and critically revised the manuscript. Theodore J. Baird contributed to conception, contributed to analysis and interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
