Abstract
Since our earlier publication (Gad et al, 2013), BioMimetix has advanced BMX-010 (Manganese (III) meso-tetrakis(N-ethylpyridinium-2-yl)porphyrin or MnTE020PyP; CASRN 219818-60-7) into clinical development as a topical agent for the treatment of psoriasis, atopic dermatitis, and pruritus (idiopathic nonspecific itch). A multiple dose phase I study has been completed in 64 patients without any serious adverse effects. During the course of development, the formulation was initially a gel but has been modified to a cream formulation. The nonclinical safety program has been carried onward to assess preclinical risk to patients. Additional studies completed and reported here include dermal sensitization in a Guinea Pig maximization test study, 2 rabbit phototoxicity studies, a 28-day oral toxicity study in juvenile mice, a 28-day topical systemic toxicity study in Gottingen minipigs, range-finding studies, and complete embryo–fetal developmental toxicity (Segment II) studies in mice and rabbits, an ICH M7 compliant qualification of impurities using 2 (Q)SAR in silico methods, and a 14-day subcutaneous toxicity study of mice to qualify an impurity. All studies (except the (Q)SAR evaluations) were performed in accordance with Good Laboratory Practices (GLP) using Good Manufacturing Practices (GMP) drug substance. The systemic toxicity studies, with the exception of the juvenile toxicity study, included toxicokinetic evaluations, which are reported here. The phase I clinical study had 67 patient participants who received topically applied BMX-010, and there were no notable safety findings and included pharmacokinetic determinations on these patients which are also reported here. Chronic GLP toxicity studies have been initiated in the mouse (6-month oral) and minipig (9-month dermal). To date, the only observed nonclinical toxicity remains a reversible hypertension seen in mice in response to Cmax levels with a no effect threshold, and there have been no drug-related adverse effects.
Introduction
Manganese (III) meso-tetrakis(N-ethylpyridinium-2-yl)porphyrin (known as MnTE-2-PyP or BMX-010; CASRN 219818-60-7) is a manganese porphyrin drug candidate being developed for use in treating psoriasis, atopic dermatitis, and itch by the dermal route. The structure is shown in Figure 1.Topically, the drug has extensive anti-inflammatory actions, including the ability to reduce early nonspecific proinflammatory events (ie, free radical generation and cytokine production).
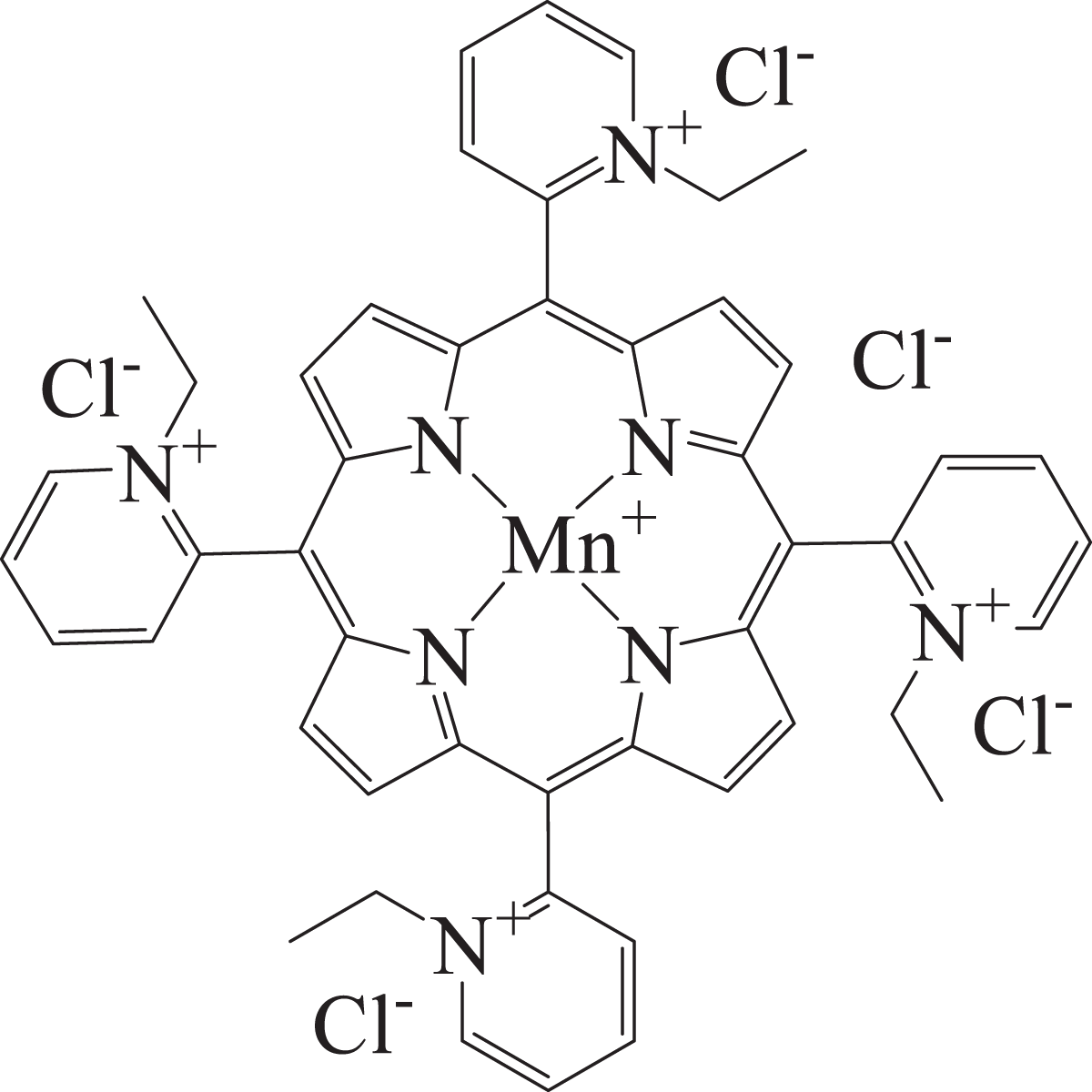
Structure for BMX-001 (Manganese (III) meso-tetrakis(N-n-butoxyethylpyridinium-2-yl)porphyris).
An investigational new drug application (IND) was opened 3 years ago based on safety and efficacy in nonclinical models. Previously described nonclinical studies 1 included 3 genotoxicity studies, an in vitro hERG (human ether-a-go-go-related gene) study, an hemolysis and flocculation study, an intravenous (Iv) irritation study, 3 in vivo safety pharmacology studies (CNS and respiratory in mice and CV safety in telemeterized monkeys) and repeat dose Iv systemic toxicity studies in mice and monkeys (see Table 1). Phase I clinical studies have been completed. To further support ongoing clinical studies, the studies described in Table 2 have been conducted and are reported here.
Prior Reported Nonclinical Safety Assessment Results. 1

Abbreviations: CNS, central nervous system; DRF, dose range finding; IV, intravenous; SC, subcutaneous.
BioMimetix BMX-010 Nonclinical Studiesa Completed Subsequent to Gad et al (2013) Paper.
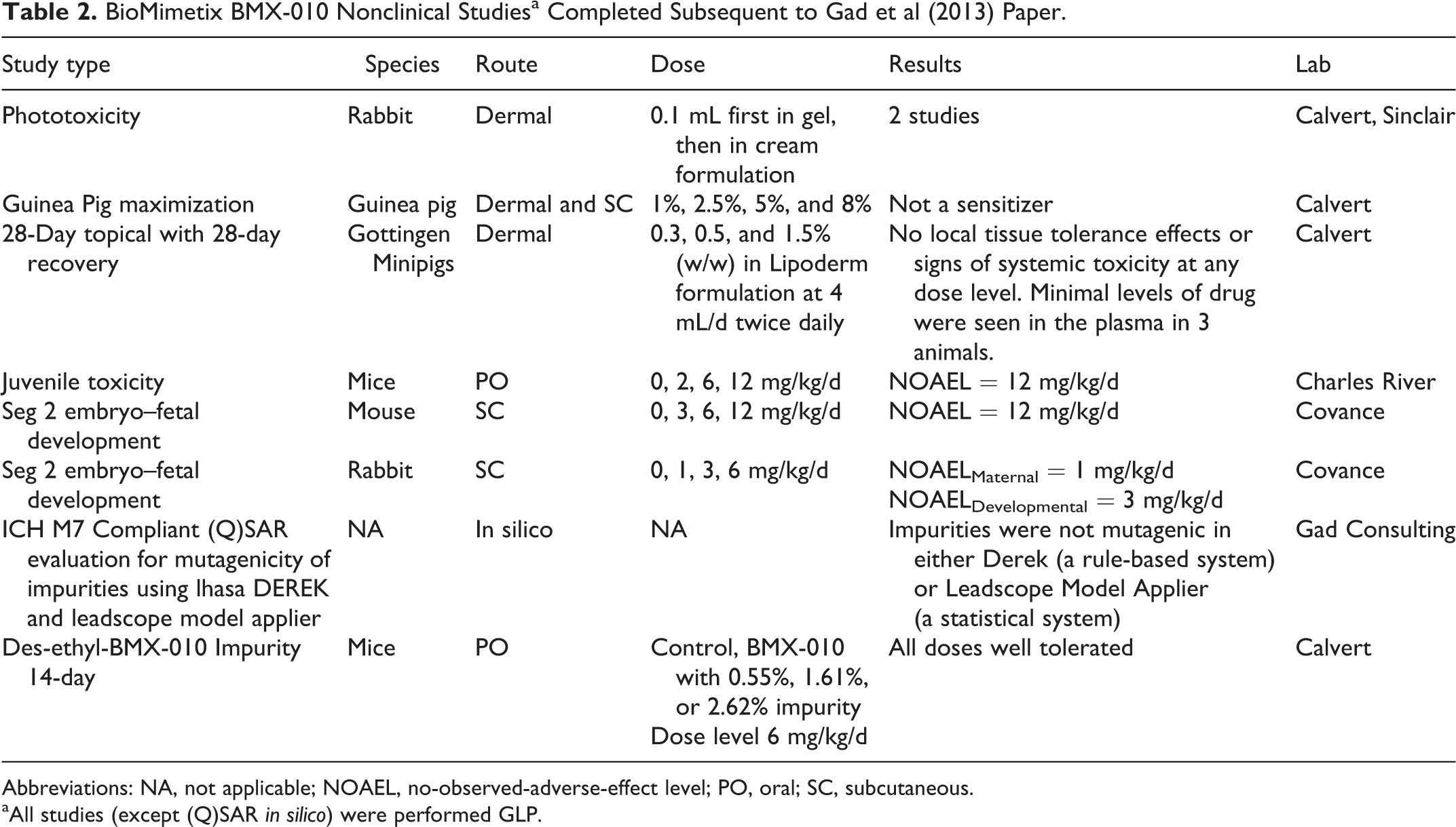
Abbreviations: NA, not applicable; NOAEL, no-observed-adverse-effect level; PO, oral; SC, subcutaneous.
aAll studies (except (Q)SAR in silico) were performed GLP.
In phase 1 studies, a total of 137 individuals have been treated to date (62 in phase I and 75 in phase II). No serious or unexpected adverse events have been observed, although 1 participant was removed from the trial due to skin irritation. Formulation evolved to meet patient acceptance expectations (feelings and suggestions from potential partners).
Materials and Methods
Drug Substance
Cyclic guanosine monophosphate BMX-010 (98% pure) was produced by Albany Molecular Research (Rensselaer, NY) and appears as a dark brown to black powder. The drug substance is very water-soluble and is not extensively metabolized, and therefore when administered orally or parenterally frequently “wine-colored” urine is observed.
Phototoxicity
Two separate phototoxicity studies were conducted in rabbits (at 2 different facilities) with BMX-010. In the first, a gel formulation was tested at a concentration that was minimally irritating. The study was subsequently repeated using a cream formulation with a prescreen study beforehand, used to identify the highest nonirritating concentration. In both studies, exposure of control and test areas was to ultraviolet (UV)-A (∼5 J/cm2) and to UV-B (∼0.05 J/cm2) for 2 hours. Concurrent positive controls were included and gave positive results.
In the first study, 2 BMX-010 (0.1%), vehicle (Placebo for BMX-010 Gel, Vehicle), the positive control (1% 8-Methoxypsoralen), and negative control (100% ethanol) were placed on intact skin sites (to the left of the midline with at least 1 inch distance separating adjacent treatment sites, sites arranged in a straight line from head to tail) on 3 male and 3 female New Zealand white rabbits. An additional site remained untreated with test article, vehicle, positive, and negative control but still received UV exposure to serve as an untreated control (sham). After allowing 15 minutes for drying, the animals were exposed for 1 hour (±15 minutes) to nonerythemogenic (ie, UV-A greater than 320 nm wavelength) UV light (FL-40) at a target distance of approximately 10 inches. The right side was simultaneously irradiated but without test materials present. After removal of the UV light source, the same materials were applied to skin treatment sites to the right of midline in the same sequence as previous applications on the left. After allowing 15 minutes for drying, the animals were returned to their cages. Observations for dermal irritation were recorded at 24, 48, and 72 hours after UV exposure. Grading of irritation was according to the method of Draize were performed for all dose sites at 1, 24, 48, and 72 hours after the second-dose application.
In the second study, 3 6 healthy naive young adult female New Zealand White (NZW) rabbits were used. Each animal in the study was topically administered (0.2 g/site) with placebo cream, test article formulations in 2 (0.03% and 0.1%) concentrations with 2 sites for each formulation in each animal. On study day 1, the control and test articles were applied on the right side of the trunk first and exposed to UV-A (∼5 J/cm2) and UV-B (∼0.05 J/cm2) along with visible light for 2 hours. Following the UV radiation exposure, the control and test articles were applied on the other side of the trunk. Draize scores were then taken for all sites.
Guinea Pig Maximization Test
A guinea pig sensitization (Magnusson-Kligman maximization) study was conducted with BMX-010 to determine whether the test article concentration and/or vehicle administered intradermally and topically elicits a delayed dermal contact hypersensitivity response. 4
Based upon the intradermal dose range-finding study, the intradermal dose utilized was 2.5% of BMX-010 0.03%. Based upon the topical dose range-finding study, the topical induction was performed at 25%. A secondary dose range-finding study was performed to obtain a nonirritating dose for topical challenge. The challenge dose utilized was 1%. Due to equivocal results observed with the test article and vehicle at challenge, a rechallenge was performed utilizing a dose of 0.1% of BMX-010 0.03%.
The main study was conducted with 10 animals/sex for the test article group, 5 animals/sex for the vehicle control group, and 3 animals/sex for the positive control (1-chloro-2,4-dinitrobenzene [DNCB]) group.
For the intradermal induction phase (day 1), each guinea pig received intradermal injections (0.1 mL each) at 6 sites between the shoulders according to Table 3.
Test Materials at Guinea Pig Sensitization Test Sites

Abbreviation: FCA, Freund’s Complete Adjuvant.
On day 7, the test sites were clipped to be free of hair. On day 8, the test article (25%) was spread over a 2 × 4 cm filter paper (0.3 mL) and applied to the injection site area and occluded with Blenderm tape (3M). The Blenderm tape was held in place with an appropriate bandage. A minimum of 48 hours later the dressings were removed. The sites were then unwrapped, and residual material removed with water and a piece of surgical gauze. This same procedure was employed with the vehicle control and the positive control in the vehicle and positive control animals, respectively.
Thirteen days after the topical induction, the fur was removed from right and left flanks. On the following day, BMX-010, vehicle (distilled water), and positive control animals were challenged with occluded patches for a minimum of 24 hours (no longer than 26 hours) on the left and right flanks.
Approximately 24 to 26 hours later, the wrapping was removed, and the sites wiped clean with surgical gauze and water. Approximately 22 hours after the sites were depilated with Nair Lotion Hair Remover. The depilatory was thoroughly washed off with water and animals patted dry with surgical gauze and returned to their cages. Three to 5 hours later, the sites were graded for elicited skin reactions (24-hour grade). Approximately 24 to 26 hours, later the sites were graded a second time (48-hour grade).
Since equivocal responses were observed in the initial challenge application, a rechallenge was performed. Groups of animals exhibiting possible sensitization being rechallenged 10 days after challenge. Naive animals were utilized for the rechallenge, and the naive animals were dosed with test article (at 0.1%) and vehicle (as received). The test article–treated animals and animals in the vehicle group were rechallenged per the challenge methods. The test article group was rechallenged with only test article at 0.1%, and the vehicle animals were rechallenged with only vehicle (as received). Materials applied in the rechallenge were placed at naive sites (test article group received test article on the right mid dorsal, vehicle group received vehicle on the right mid dorsal, and naïve animals received test article on the right mid dorsal and vehicle on the left mid dorsal).
Twenty-Eight–Day Dermal Toxicity Study in Minipigs
Thirty-two experimentally naive Göttingen Mini-pigs (16M/16F), approximately 4 months old and weighing 7.4-9.8 kg at the outset of the study, were assigned to treatment groups as shown in Table 4. 5 BMX-010 was prepared and used in twice-daily dermal administrations with Lipoderm base as a vehicle for topical dermal administration.
Study Design for 28-day Minipig Dermal Study.
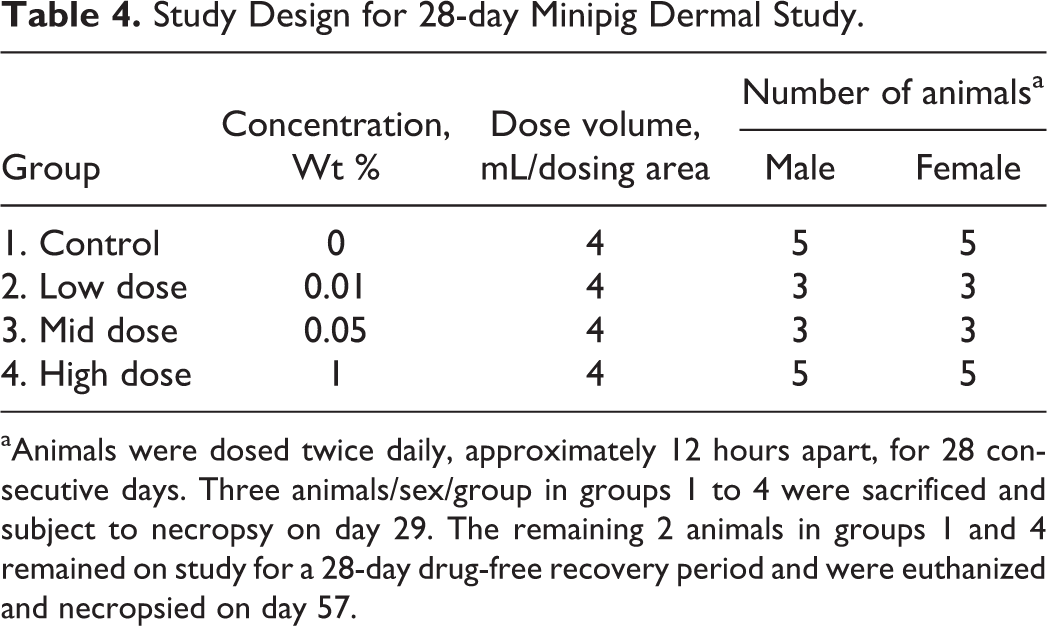
aAnimals were dosed twice daily, approximately 12 hours apart, for 28 consecutive days. Three animals/sex/group in groups 1 to 4 were sacrificed and subject to necropsy on day 29. The remaining 2 animals in groups 1 and 4 remained on study for a 28-day drug-free recovery period and were euthanized and necropsied on day 57.
All animals were observed once daily during the study prior to scheduled necropsies on day 29 and day 57 (recovery animals). Irritation at the treatment sites was scored daily for days 1 to 7 and weekly thereafter including the recovery period using the Draize evaluation scoring system. All animals were weighed at the time of randomization, weekly, and prior to termination. Animals were weighed (fasted) prior to scheduled sacrifice on day 29 and day 57 (recovery animals). Food consumption values recorded daily during the dosing and recovery phases of the study. Ophthalmology examinations were performed prior to treatment initiation and during the final week of the dosing period. Electrocardiograms were obtained from all animals prior to treatment initiation and prior to terminal sacrifices. Blood for evaluation of hematology, coagulation, and clinical chemistry was collected from all animals prior to treatment initiation and prior to terminal sacrifice on day 29 and day 57 (recovery animals). Urine samples for urinalysis were collected prior to treatment initiation and termination. Blood for toxicokinetic (TK) evaluation was collected from all animals at selected time points after the first daily dose on day 1 and day 19 and after the last dose on day 28. Blood samples were collected from surviving recovery animals in groups 1 and 4 on days 42 and 56. All surviving animals were sacrificed on days 29 and 57. A full range of tissues were harvested at necropsy, selected organs were weighed, and selected tissues from all animals were evaluated microscopically (by a board-certified veterinary pathologist) or for bioanalysis. A validated liquid chromatography/mass spectrometry method was used for bioanalysis. Blood samples were also evaluated for full clinical chemistry and clinical pathology panels.
Juvenile Toxicity
The objective of this study was to determine the potential toxicity of BMX-010 when administered to juvenile Crl: CD1(ICR) mice by oral gavage for 4 weeks, with a recovery period in order to evaluate the toxicity and the potential reversibility of any findings. 6 Performance of this GLP study was equivalent to that of the other repeat-dose studies. The study design was as depicted in Table 5.
Experimental Design.
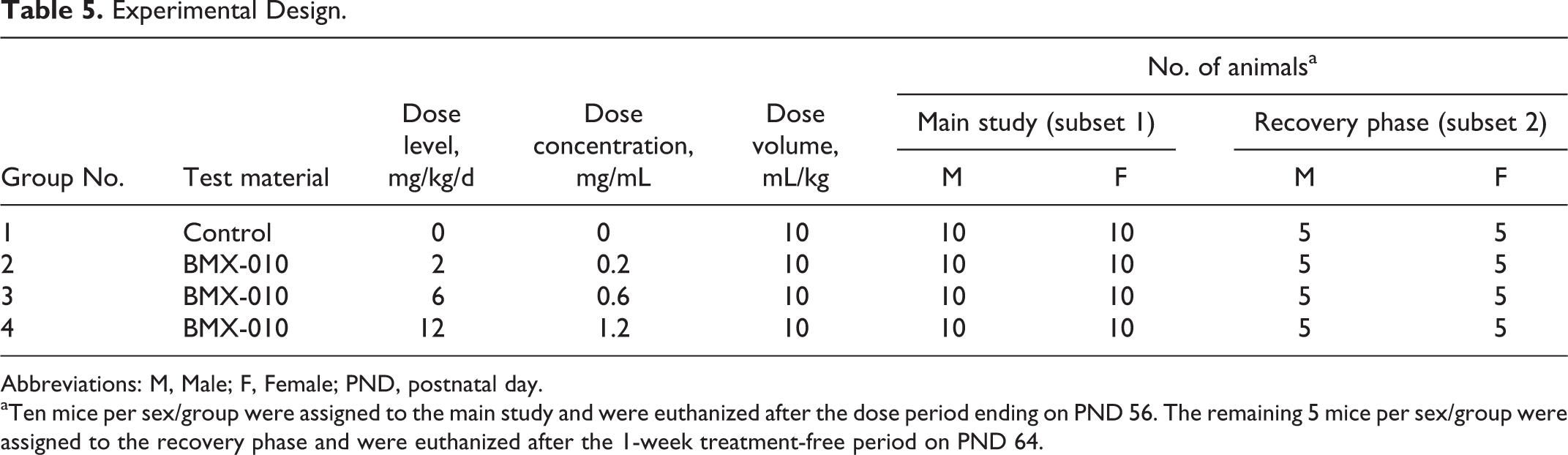
Abbreviations: M, Male; F, Female; PND, postnatal day.
aTen mice per sex/group were assigned to the main study and were euthanized after the dose period ending on PND 56. The remaining 5 mice per sex/group were assigned to the recovery phase and were euthanized after the 1-week treatment-free period on PND 64.
Eighty juvenile Crl: CD1(ICR) male and female mice were assigned to 4 main study (subset 1) dose groups (groups 1 through 4), 10 mice/sex/group. Additionally, 40 mice were assigned to 4 recovery phase (subset 2) dose groups (groups 1 through 4), 5 mice/sex/group. Mice were 14 to 16 days of age and weighed 6.4 to 11.6 g at the start of study.
Mice were given formulations of the control (phosphate buffered saline) or test article (BMX-010) by oral gavage once every 3 days beginning on postnatal day (PND) 28 and continuing for 4 weeks (specifically on PNDs 28, 31, 34, 37, 40, 43, 46, 49, 52, and 55). A total of 10 doses were administered.
Following the completion of dose administration, subset 2 mice (5 mice/sex/group in groups 1 through 4) assigned to the recovery phase had a test article–free period beginning on PND 56 through PND 64.
The following parameters and end points were evaluated in this study: viability, clinical signs, body weight values, body weight gain values, food consumption values, clinical pathology parameters (hematology and clinical chemistry), complete necropsy findings, organ weights, femur measurements, and histopathologic examinations (standard tissue and clinical panels).
Developmental Toxicity Studies
Mouse
Doses were selected based on the results of a range-finding study conducted in the same CRO (Contract Research Organization) using the same source of mice, with 12 mg/kg/d not being well-tolerated in dams. Vehicle or 3, 6, or 9 mg/kg/d BMX-010 were injected subcutaneously (SC) in time-mated female Crl: CD1 (ICR) mice (22 females/dose group) on gestation days (GDd) 6 to 15. 7 Cesarean sections were performed, and females were sacrificed on GD 18. Maternal study parameters included clinical observations, body weight values, food consumption values uterine contents, macroscopic examination, and examination of ovaries for the number of corpora lutea. Each fetus was assigned a sex, weighed, and examined for external abnormalities. One-half of all fetuses from each litter underwent visceral examinations. The remaining fetuses were examined for skeletal malformations. Animals for TK analysis (3 females in control, 18 females/group for BMX-010-treated animals) were bled for plasma. Control and treated animals were bled at 1 hour post dose with treated animals also bled at 0.5, 2, 4, 6, and 24 hours post dose.
Rabbit
For this study, doses were selected based on the results of a range-finding study conducted in the same CRO using the same source rabbits. Time-mated female Hra:(NZW)SPF rabbits (6 to 7 months old at initiation of dosing) were assigned to 4 groups (22 animals/groups) and were administered the vehicle control article (phosphate-buffered saline) or BMX-010 via SC injection, as described in Table 6. 8
Group Designation and Dose Levels.

Abbreviation: GD, gestation day.
aAnimals in group 2 were dosed at a volume of 2.5 mL/kg.
bAnimals in groups 1, 3, and 4 were dosed at a dose volume of 5 mL/kg.
cThree toxicity animals/group were used for collection of toxicokinetic (TK) blood samples.
dGroup 1 was administered vehicle control article only.
Assessment of toxicity for this study was based on mortality, clinical observations, body weight values, food consumption values, and necropsy and cesarean section findings. Fetal toxicity was also assessed via external, visceral, and skeletal examinations. Blood samples were collected and analyzed for later TK evaluation.
Impurity Qualification
(Q)SAR
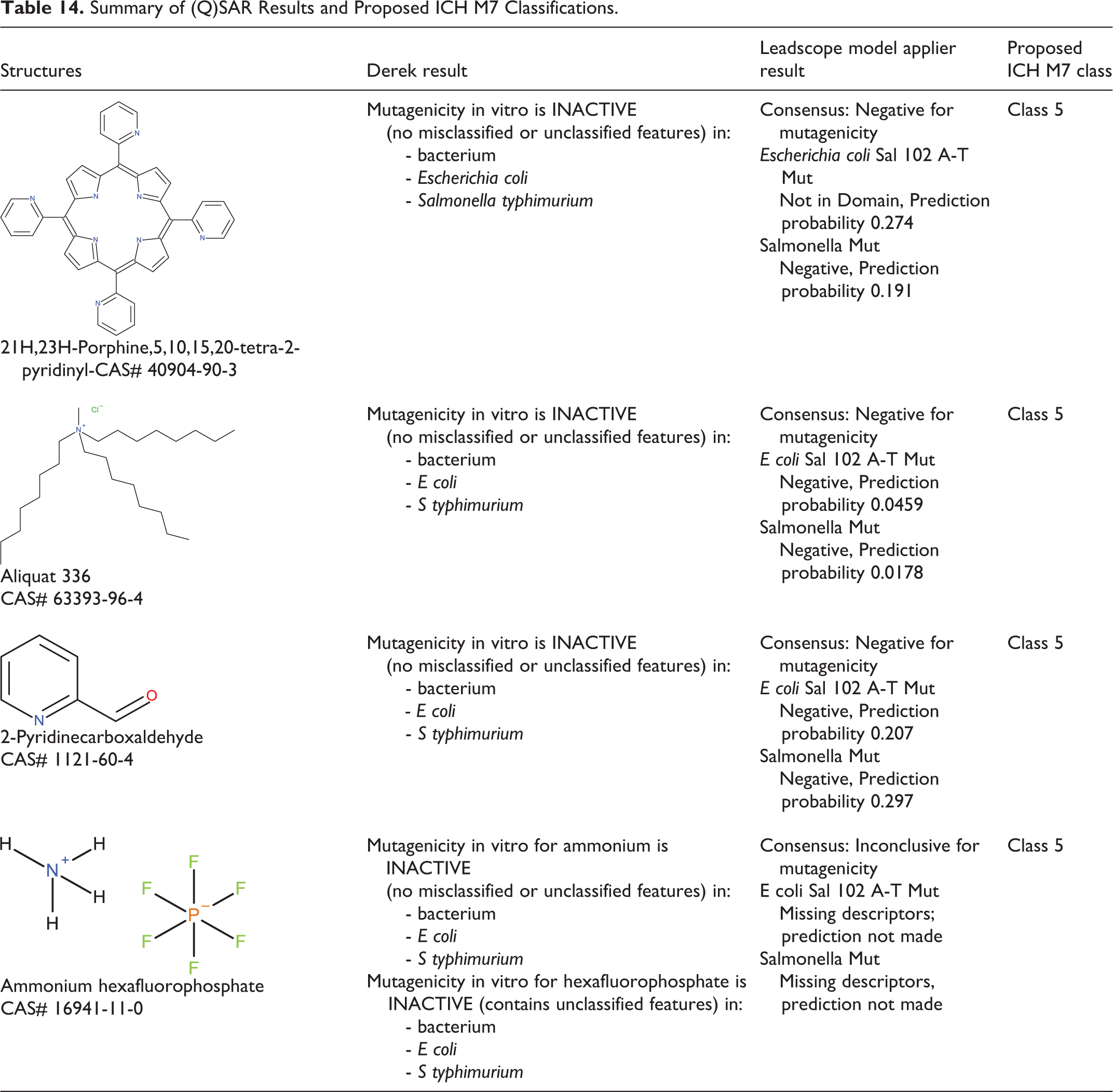
To meet current ICH M7 requirements 9 for assessing the potential mutagenicity of manufacturing impurities, 5 impurities identified in BMX-010 API were evaluated by computational analysis (in silico). These included 21H,23H-porphine,5,10,15,20-tetra-2-pyridinyl (CAS# 40904-90-3), aliquat 336 (CAS# 63393-96-4), 2-pyridinecarboxaldehyde (CAS# 1121-60-4), ammonium hexafluorophosphate (CAS# 16941-11-0), and pyrrole (CAS# 109-97-7). As required by ICH M7, 2 complementary (Q)SAR prediction methodologies were utilized to evaluate potential mutagenicity for each structure. The parent BMX-010 was previously evaluated and found negative in a GLP Ames study. The expert rule-based system was DEREK, 10 and the statistical-based was Leadscope 11 Model Applier. The outcomes were reviewed with the use of expert knowledge to provide additional supportive evidence on relevance of positive, negative, or inconclusive predictions and elucidate underlying reasons in case of conflicting results.
Fourteen-day SC mouse
A 14-Day SC toxicity study in mice with a 14-day recovery period using BMX-010 containing different amounts of Des-ethyl-BMX-010 impurity was conducted to evaluate the toxicity of GLP-prepared BMX-010 containing different amounts of the des-ethyl-BMX-010 impurity when administered by SC injection once daily for a minimum of 14 consecutive days in mice followed by a 14-day recovery period. 12 The SC route was used here to ensure adequate systemic exposure to the parent and impurity.
BMX-010 containing different amounts of a primary impurity that may occur in clinical supplies at levels above those specified in ICH Q3B, des-ethyl-BMX-010, was supplied by Duke University Medical Center as a black (dark brown) powder. The test article was prepared into dosing formulations for SC injection. Ninety-six experimentally naive Crl: CD-1 (ICR) mice (48 males and 48 females), approximately 8 weeks old and weighing 31-36 g for males and 24 to 30 g for females at the outset of the study, were assigned to treatment groups as shown in Table 7.
Experimental Design for 14-Day Impurity Qualification Study.
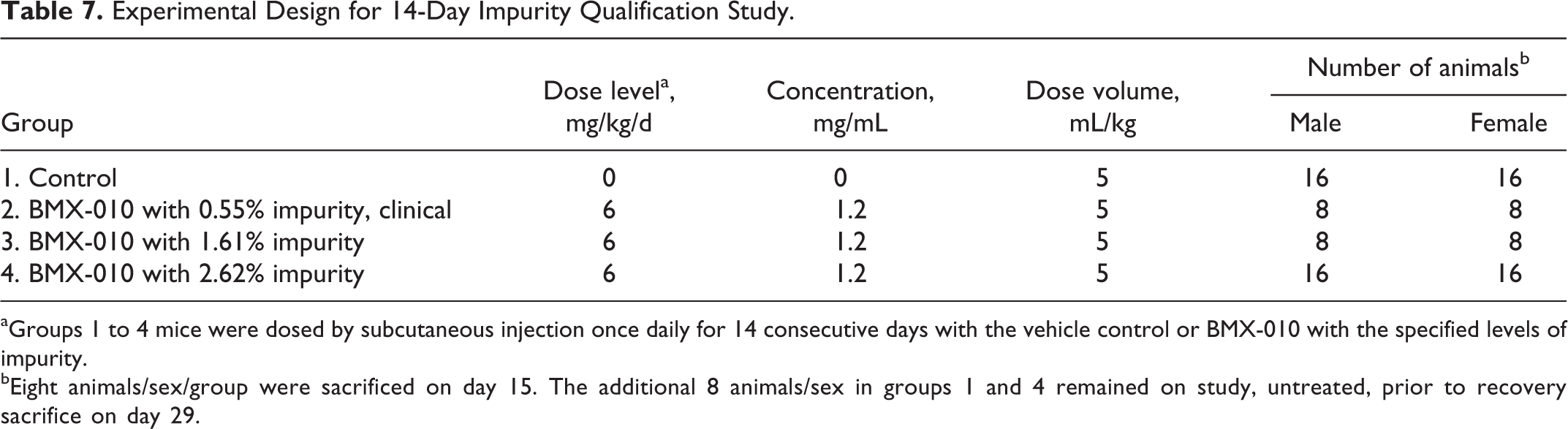
aGroups 1 to 4 mice were dosed by subcutaneous injection once daily for 14 consecutive days with the vehicle control or BMX-010 with the specified levels of impurity.
bEight animals/sex/group were sacrificed on day 15. The additional 8 animals/sex in groups 1 and 4 remained on study, untreated, prior to recovery sacrifice on day 29.
Animals were dosed once daily for 14 consecutive days. Mortality/morbidity was evaluated twice daily and once prior to scheduled sacrifice on days 15 and 29. Clinical observations were recorded daily prior to dosing, immediately post-dose, and approximately 1 to 2 hours post-dose on dosing days. Animals were observed once daily on nondosing days and prior to scheduled sacrifice. Body weights and food consumption values were recorded weekly. Blood for evaluation of hematology and clinical chemistry was collected on days 15 and 29 (recovery animals). All animals in the toxicology groups were sacrificed on day 15 or day 29 (recovery animals). Selected tissues were harvested at necropsy and selected organs weighed. Selected tissues from the control and high-dose groups were evaluated microscopically.
Results/Discussion
Phototoxicity
In the first study, no signs of erythema or edema were observed at any time point at the sites irradiated with negative control or receiving irradiation prior to negative control application. No irritation was noted at any of the sites irradiated with vehicle applied at any time point. Irritation was not observed at any of the sites receiving irradiation prior to vehicle application at any time point.
At the sites irradiated with test article applied, no edema was noted at any time point. At 24 hours following UV-A exposure, 1 animal showed no erythema, 4 animals showed very slight erythema, and 1 animal exhibited well-defined erythema. At 48 hours following UV-A exposure, 4 animals showed no erythema and 2 animals presented with very slight erythema. Very slight erythema persisted in 1 animal by 72 hours with remaining animals showing no scores for erythema.
The test article (0.1% BMX-010 gel) was concluded to have caused a phototoxic reaction in rabbits, since erythema was observed at the sites receiving irradiation with test article applied, and no erythema/edema was observed at the sites receiving the vehicle following irradiation.
In the second study, both the placebo control site and the test sites (both concentrations) on each animal appeared to have similar skin responses post dose application and/or UV radiation exposure. Erythema scores ranged from 0 to 2 and were either a grade 1 or 2 and observed on the majority of the treated sites during the entire study period up to 72 hours. The erythema scores indicated that some mild skin irritations occurred during the study period, but the reactions were not related to any test article treatments. Edema scores mostly were 0, with a few instances of scores of 1 during the entire study period. Compared to the non-UV radiation exposure sites, the UV radiation–exposed sites appeared to have slightly higher scores in frequency and severity. Overall, there were no test article–related remarkable higher scores compared to the placebo control after dose administration.
In conclusion, the test article BMX-010 Cream did not cause any skin irritation after topical application at 0.2 g/site at both 0.03% and 0.1% concentrations for both the UV radiation exposure or nonexposure conditions. The test article did not cause any local skin phototoxicity under the study conditions. The active drug molecule has UV absorption peaks at 363.5, 409, 454, 499, 555, and 782 nm. 13 BMX-010 was found not to be phototoxic in this second study. No signs of phototoxicity have been observed in 137 patients treated topically to date.
Guinea Pig Maximization Test
No mortality was observed during the course of the study. All animals appeared normal throughout the course of the main study with the following exceptions: Red urine was visible in the cage pan below 3 animals in the test article group on day 6. No red discharge was visible on these animals. Red urine was visible in the cage pan below one animal in the test article group on day 8. No discharge was visible on this animal. On day 24, red urine was observed in the pan underneath 2 animals in the test article group. Note that this is attributable to the color or the unmetabolized BMX 010 (verified in earlier studies by analysis of urine—drug present but no blood cells or hemoglobin).
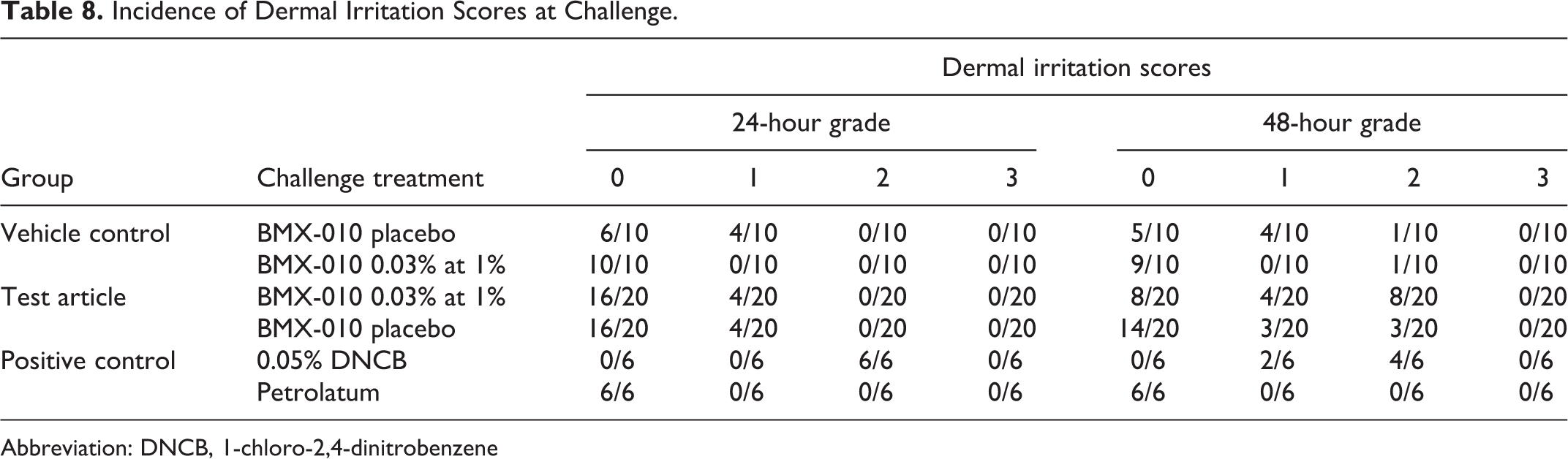
It was decided to proceed to the main study using a test article concentration of 2.5% intradermal induction, 25% topical induction, 1% topical challenge, and 0.1% topical rechallenge. Challenge results are shown in Table 8, and rechallenge results are shown in Table 9.
Incidence of Dermal Irritation Scores at Challenge.
Abbreviation: DNCB, 1-chloro-2,4-dinitrobenzene
Incidence of Dermal Irritation Scores at Rechallenge.
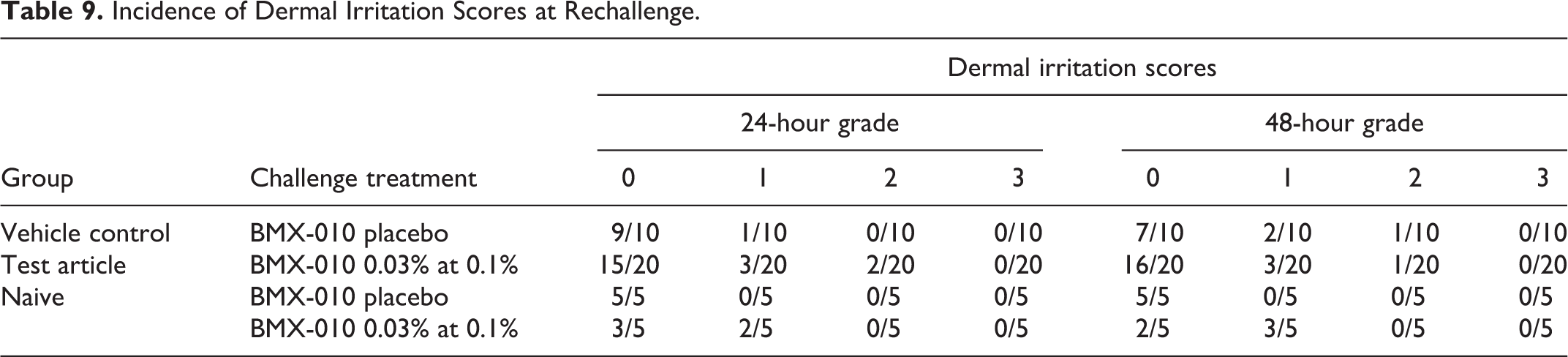
An intradermal induction of BMX-010 0.03% at 2.5%, a topical induction of BMX-010 0.03% at 25%, followed by a topical challenge at 1% to guinea pigs elicited an equivocal response in the test article–treated animals following the challenge. Intradermal induction, topical induction, and topical challenge of BMX-010 placebo as received also elicited an equivocal response in the vehicle control animals following challenge. Based on these results, a rechallenge was performed in the test article group with BMX-010 0.03% at 0.1%. Topical rechallenge of BMX-010 0.03% at 0.1% to guinea pigs showed a weak (grade I) elicitation response at 24 and 48 hours; however, Magnusson and Kligman do not regard sensitization of grade 1 as significant. A rechallenge was also performed in the vehicle group with BMX-010 placebo as received. A topical rechallenge of BMX-010 Placebo to guinea pigs showed a weak (grade I) elicitation response at 24 hours increasing to a mild (Grade II) elicitation response at 48 hours. Signs of grade II were also seen in 2 animals at 24 hours. BMX-010 was not concluded to be a sensitizer.
Twenty-Eight–Day Dermal Minipig
There were no test article-related deaths or signs of local irritation including erythema and/or edema at the application sites. Nor were there any test article–related effects on mean body weight values, mean body weight gain values, food consumption values, or ophthalmological findings during the study.
Dermal dosing did not have any toxicologic effects on cardiac rate or rhythm or electrocardiogram morphology. However, second-degree atrioventricular block and a ventricular couplet were noted in 1 mid-dose mini pig at 0.05 wt% dose prior to terminal sacrifice. This rhythm change is rarely seen in normal mini pigs but may not have been test article related, as it was not seen in any other treated animals. No such finding seen in cardiovascular safety study. There were no test article-related effects on clinical pathology, chemistry, or urinalysis.
Necropsy findings at day 29 terminal sacrifice, in general, included dry flaking skin at the application sites that ranged in color from yellow to dark brown (as was seen during the study). Dark red thyroid/parathyroids and neck musculature were noted across all dose groups. Animals dosed at 0.05 wt% and 1 wt% had dark red thymi. Animals dosed at 0 wt% and 1 wt% had red esophagi, the latter being dark red. At day 57 terminal sacrifice, all animals dosed at 1 wt% had dark brown flaking skin. One male had a dark red thyroid/parathyroid. Degree of coloration declined in recovery animals.
There were no test article–related findings on mean absolute organ weights or their relative ratios with mean body weights and brain weights at days 29 and 57.
There were no test article–associated microscopic changes at the cutaneous treatment site or in any other tissue. All animals had minimal to mild diffuse hyperkeratosis which correlated with the dry, flaking skin observed grossly and was similar at both the terminal and recovery sacrifice. All animals also had granular to globular basophilic foreign material (interpreted as Lipoderm base) superficially on the treated skin. The amounts present on examined slides were generally smaller in the recovery animals, but it was present on all treated skin. The yellow, brown, or dark brown discoloration observed grossly was not evident microscopically and was likely related to the color of the drug substance. Mononuclear cell infiltrates were rarely present and never more than minimal in severity. At the terminal sacrifice, focal cutaneous ulceration and serocellular crusts accompanied by minimal mixed cell inflammation were noted in one group 3 female and one control female which was likely due to self-trauma (rubbing or scratching).
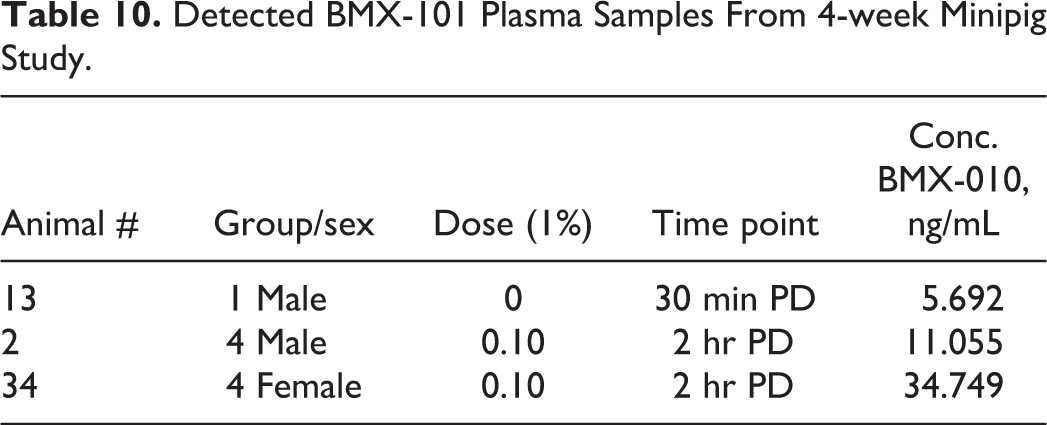
Toxicokinetic evaluation was not possible as all but 2 plasma samples measured below lower limit of quantitation (LLOQ; 71 μg/mL; see Table 10).
Detected BMX-101 Plasma Samples From 4-week Minipig Study.
In conclusion, the levels of BMX-010 in Lipoderm lotion used in this study did not cause any adverse toxicological effects when administered topically twice daily on the skin of Göttingen mini pigs at 0.01 wt%, 0.05 wt%, and 1 wt% for 28 consecutive days followed by a 28-day drug-free recovery period. Although altered mean levels of some hematology and clinical chemistry parameters were noted, the changes were of small magnitude, biologically insignificant, remained within the historical reference ranges, and returned to normal levels at the end of the recovery period. A second-degree atrioventricular block and a ventricular couplet were noted in one mid-dose mini pig at 0.05 wt% dose prior to terminal sacrifice. However, it was not observed in animals dosed at 1 wt%. Based on these results, the no-observed-adverse-effect level (NOAEL) was placed at 1 wt% of BMX-010 in Lipoderm lotion.
Juvenile Toxicity
The administration of BMX-010 by oral gavage every third day from PND 28 to 56 was well tolerated in both male and female mice at levels of 12 mg/kg. One male mouse in the 2 mg/kg dose group was found dead during the study; the death was not associated with BMX-010 administration. All other male mice and all female mice survived to scheduled euthanasia. There were no test article-related clinical observations, and body weights, food consumption values, and femur lengths were unaffected. No target organ effects were observed at any dose levels. Based on these results, the NOAEL was considered to be 12 mg/kg.
Developmental Toxicity Studies
Mouse
No BMX-010-related mortalities or early sacrifices occurred. Clinical observations of brown urine and bedding were noted in all BMX-010-treated groups and were considered related to the color of the test article formulations. From day 1 of dosing to the day following the completion of dosing (GD 6-16), mean maternal body weight gain was increased in the group administered 3 mg/kg/d SC by 17% and remained increased once dosing completed; however, no significant changes in mean body weights and body weight gains were noted at 6 or 12 mg/kg/d SC. Mean food consumption values were slightly increased when compared to controls in the group administered 3 mg/kg/d, which coincided with increased body weight gains in this group. At necropsy, mean corrected body weight values (without uterus) were significantly increased in the group administered 3 mg/kg/d, but there were no differences in the mean numbers of fetuses or fetal weights when compared to controls. No BMX-010-related macroscopic observations, fetal external, or visceral anomalies were noted. No BMX-010-related skeletal malformations were noted. BMX 010-related skeletal variations included increased incidences of unossified calcaneum in all treated groups compared to controls and slightly increased incidences of unossified hindlimb phalanx and unossified hyoid in the group administered 12 mg/kg/d, which were not considered adverse as they were not associated with other developmental effects and are expected to resolve. Table 11 presents a summary of plasma TK data from the study.
Summary of the Mean BMX-010 Toxicokinetic Parameters in Plasma of Pregnant Mice.
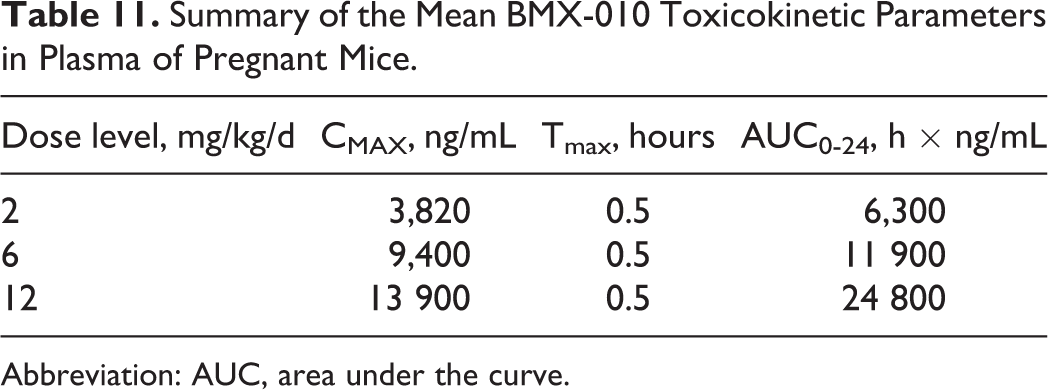
Abbreviation: AUC, area under the curve.
In conclusion, neither maternal toxicity nor embryo–etal toxicity was observed in Crl:/CD1 (ICR) mice administered up to 12 mg/kg/d BMX-010 from GD 6 to 15. Therefore, the NOAEL for maternal and embryo–fetal toxicity is considered 12 mg/kg/d.
Rabbit
Three animals administered BMX-010 6 mg/kg/d were terminated early due to body weight loss and general debilitation. BMX-010-related clinical observations occurred in all test article–treated groups and primarily consisted of fecal findings (few, nonformed, small, or none) (see Table 12). However, the group administered BMX-010 6 mg/kg/d had test article–related thin appearances and incidences of pale ears, pale gingivae, pale noses, pale oral mucosae, and/or warm to touch midline cervical areas. All BMX 010-treated groups had increased incidences of discolored, brown, or red urine which were considered related to the color of the test article. BMX-010-related reduced body weight gains were observed in the groups administered 3 or 6 mg/kg/d throughout the dosing period (GD 7-20); body weight gain was reduced by 36% in the group administered 3 mg/kg/d, and the group administered 6 mg/kg/d did not gain any weight over the dosing period. Once dosing was completed (GD 20-29), body weight gains showed evidence of recovery. Likewise, food consumption values were reduced throughout dosing for groups administered 3 or 6 mg/kg/d (by 10% or 34%, respectively) and showed recovery once dosing completed. Although food consumption values for all test article–treated groups were similar to or increased compared to controls by GD 22 in all treated groups, mean body weights of groups administered 3 or 6 mg/kg/d did not return to control levels. No BMX-010–related macroscopic observations, effects on reproductive performance, or cesarean section findings were noted. No BMX-010–related fetal external malformations or variations were observed. The litter incidence of the visceral malformation of absent gall bladder was slightly increased for those administered 6 mg/kg/d, compared to concurrent and historical controls. It was unclear whether this malformation was BMX-010-related, and the impact of this malformation on fetal survival was unclear. Increased incidence of fluid-filled pericardium was noted in the group administered 6 mg/kg/d, which was not considered adverse. No skeletal malformations or variations were considered BMX-010 related.
Summary of Rabbit Fetal Observations—Fresh Visceral Malformations.
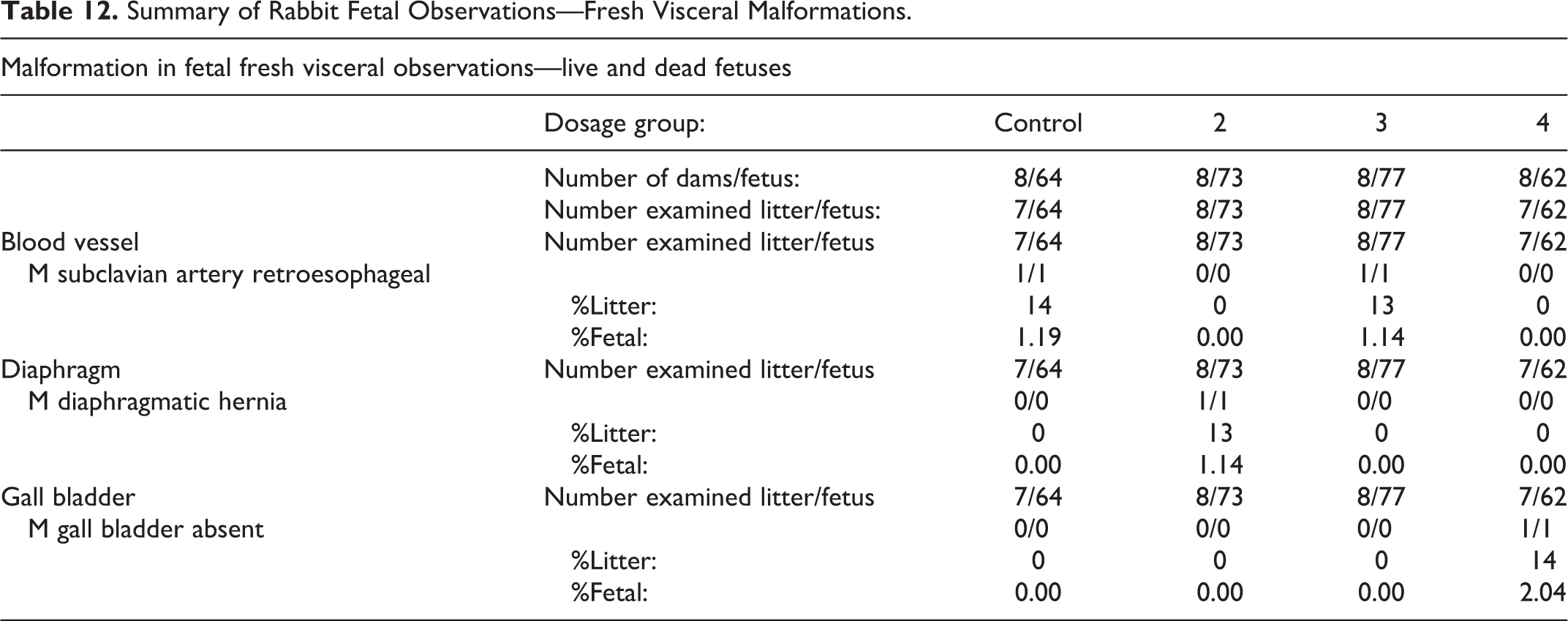
In conclusion, maternal toxicity was observed for those administered 3 mg/kg/d as evidenced by clinical observations that coincided with adverse reduced body weight values and food consumption values. Maternal clinical observations for the group administered 1 mg/kg/d did not correspond to adversely reduced body weight or food consumption values. Therefore, the NOAEL for maternal toxicity is considered 1 mg/kg/d, and the NOAEL for embryo–fetal development is considered 3 mg/kg/d. Table 13 presents the results of TK evaluation.
Summary of the Mean BMX-010 Toxicokinetic Parameters in Plasma of Pregnant Rabbits.a
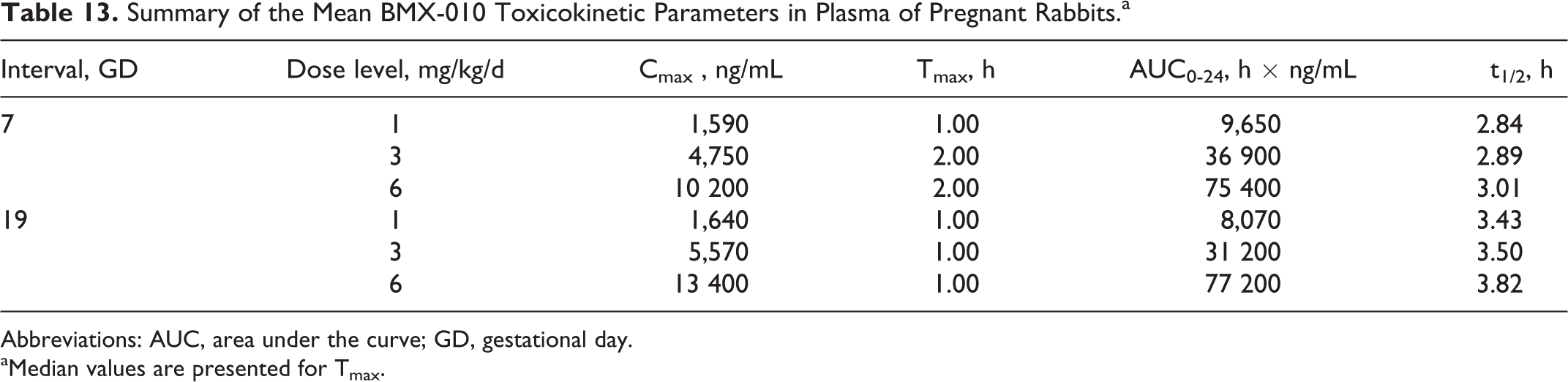
Abbreviations: AUC, area under the curve; GD, gestational day.
aMedian values are presented for Tmax.
Impurity Qualification
(Q)SAR
Table 14 presents the results of assessment of mutagenicity by both the Derek and the Leadscope Modeler system as well as the resulting ICH M7 classification of each impurity. None of the 4 impurities showed structural alerts in either QSAR modeling system for potential mutagenicity, and all were classified as M7 Class 5—that is, as without potential mutagenicity risk.
Summary of (Q)SAR Results and Proposed ICH M7 Classifications.
There was no early death during the study. There were no adverse clinical findings. The only clinical observation was amber-colored urines in all males and females treated with BMX-010 at 3 dose levels of des-ethyl-BMX-010 impurity. All treated animals, irrespective of impurity levels, had amber-colored urines post-dose starting on day 1 until day 15. On a few occasions during the treatment phase, amber-colored urine was not observed in some animals. All animals appeared normal during the recovery period.
There were no test article-related effects on body weights and food consumption. There was no test article–related effects on hematology and clinical chemistry parameters at days 15 and 29 (recovery animals).
Necropsy findings at days 15 and 29 (recovery animals) included a slightly green tinge of discoloration at the injection sites in treated animals and multiple red foci at the injection site in the male mice. There was no test article–related findings on group mean absolute organ weights, relative mean organ-to-body weight ratios, and relative mean organ-to-brain weight ratios at days 15 and 29 (recovery animals).
Microscopically, the only significant finding in the high-dose animals given BMX-010 with 2.62% impurity was the presence of foreign material in the SC region of the injection site. This foreign material had a granular appearance and was brown in color when viewed microscopically. It could be found free in the SC tissue or phagocytosed by local tissue macrophages. In rare cases, there was minimal neutrophilic inflammation associated with the deposited foreign material, considered to be a normal result of a SC injection and not an adverse reaction. Otherwise, there was not a significant tissue reaction to the injected material in the tissue sections examined. In some cases, the foreign material could not be identified, but likely only because there was only a small region of the SC tissue available in the section examined. In summary, while the injected material was visible in most of the injection site sections examined, there was no apparent adverse tissue reaction to it, and no evidence of other findings in any of the other tissues examined.
In conclusion, Crl: CD-1 mice dosed SC for 14 consecutive days with BMX-010 containing 0.55%, 1.61% and 2.62% des-ethyl-BMX-010 impurity did not exhibit any adverse clinical signs post-dose and following a 14-day recovery period. Based on clinical observations, changes in body weight values and food consumption values, clinical pathology and histopathology findings, and dosing with BMX-010 containing ≤ 2.62% des-ethyl BMX-010 impurity were well tolerated in the mice. The high dose was the NOAEL.
Toxicokinetics
To facilitate comparison, previously reported TK results from the 18-day mouse IV study previously performed and reported in the earlier journal article are repeated here. Those results show that substantial levels of BMX-010 were detected in plasma, as shown in Table 15.
Pharmacokinetic Parameters for the Mouse 18-Day Intravenous Study.
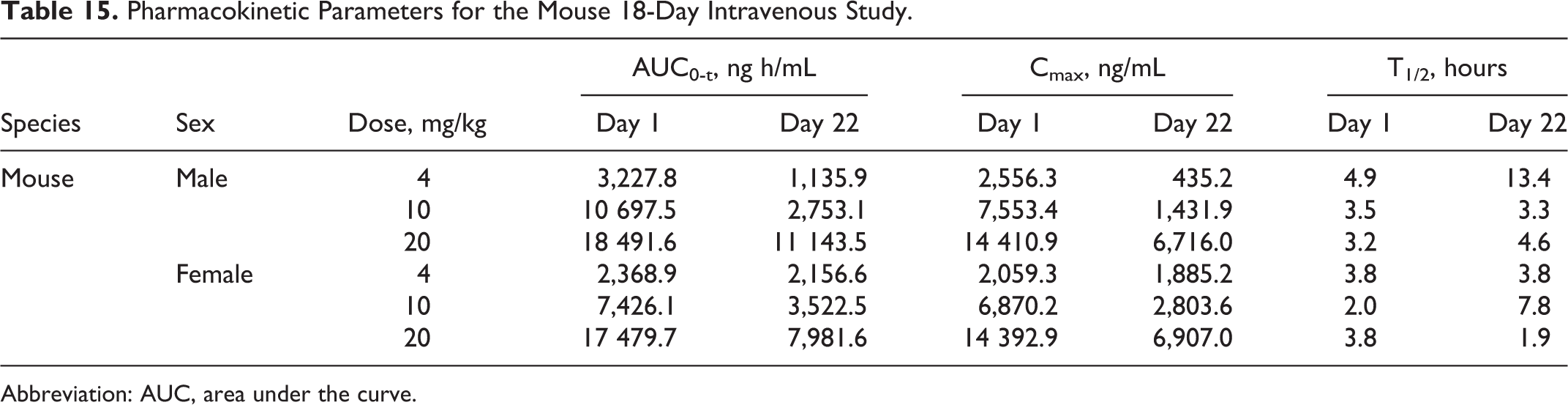
Abbreviation: AUC, area under the curve.
For the BMX-010 in mini pig dermal study where plasma samples were analyzed by high-performance LC-MS/MS detection, there were only 3 of 416 samples containing BMX-010 above the LLOQ level (5 ng/mL). These values are shown in Table 10. 14 Therefore, no repeated bioanalysis was performed, and no pharmacokinetic analysis was conducted. Since all but 3 of 416 mini pig plasma samples measured below the lower limit of quantitation, there was no evidence to support exposure to BMX-010 following topical dermal BMX-010 gel formulation application to the back of male and female Göttingen mini-pigs.
In clinical studies among dermatologic patients, to date a randomized block sample of 12 patients have had plasma samples taken and analyzed. As with the Gottingen pigs, only a few samples had drug modestly above the LLQ.
Conclusions
As previously demonstrated, no signs of manganese toxicity (manganism) have been observed in any animal receiving BMX-010. Any daily animal or patient exposure to free manganese were less than 0.1 ug/kg/d. BMX-010 is metabolically quite stable. Tests performed continue to meet ICH M3(R2) 15 requirements.
In rabbits, maternal body weight reduction was seen at ≥3 mg/kg/d. No juvenile, maternal, embryo–fetal, or adult toxicity has been seen at dose levels up to 12 mg/kg/d in mice. The drug is not a dermal sensitizer or phototoxic, and no sign of either of these effects have been seen in any of the patients participating in clinical trials. The 4 identified impurities in clinical test materials are not potentially mutagenic, and the one metabolite potentially occurring at levels above ICH Q3B did not cause signs of toxicity in mice at levels up to 2.62%. The one impurity occurring at levels requiring qualification (in accordance with ICH Q3B 16 ) caused no detected effect.
In both the Gottingen mini pig and in humans participating in clinical trials, minimal systemic levels of drug have been found in the plasma of only a few individuals.
Footnotes
Author Contribution
Gad, S. contributed to conception and design, acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript; Sullivan, D. contributed to conception and design, acquisition, analysis, and interpretation and critically revised the manuscript; Mujer, C. and Spainhour, C. contributed to conception and design, analysis, and interpretation and critically revised the manuscript; Crapo, J. contributed to conception and design, contributed to acquisition, and critically revised manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by BioMimetix, JV, LLC.
