Abstract
Regional therapies for metastatic liver disease have garnered interest in recent years due to technological advances in drug delivery. A percutaneous hepatic perfusion (PHP) using a newly developed generation 2 (GEN2) filtration system was designed to mitigate systemic toxicity and cardiovascular risk associated with hepatic blood filtration during hepatic artery infusion of the chemotherapy drug melphalan. The GEN2 system was evaluated in healthy swine, and plasma samples were assessed for clinical chemistry, melphalan toxicokinetics (TK), inflammatory cytokines, catecholamines, hematological, and cardiac biomarkers. Cardiovascular safety was assessed by echocardiography, electrocardiogram, and telemetry. Toxicology parameters included clinical signs, body weight, gross pathology, and histopathology. There were no treatment-related deaths associated with the PHP procedure with GEN2 filtration, and all animals survived to scheduled necropsy. Assessment of the pharmacokinetic/TK plasma concentrations of melphalan demonstrated that the GEN2 filter was able to extract melphalan from blood with high efficiency and reduce melphalan exposure in the systemic circulation. The hemodynamic, immunosuppressive, immunotoxic, cardiotoxic, and histopathologic effects of melphalan were limited. The significant hemodynamic challenge imposed by filtration resulted in a compensatory tachycardia with supranormal left ventricular function, although no wall motion abnormalities were detected and left ventricular function remained normal. Catecholamines decreased and then quickly rebounded during washout. Transient and reversible effects of treatment on cardiac enzymes, catecholamines, and cytokines and reversible hemodynamic effects without cardiac damage indicated that PHP with melphalan was not cardiotoxic or immunotoxic under the conditions tested, due to high efficiency of the filtration system limiting exposure of melphalan to the systemic circulation.
Keywords
Introduction
Unresectable hepatic metastases from solid organ malignancies represent a significant therapeutic challenge in oncology. For patients with many types of cancer such as colorectal adenocarcinoma, 1,2 ocular melanoma, 3,4 intrahepatic (IHP) cholangiocarcinoma, 5 and neuroendocrine tumors, 2 liver metastases frequently represent the sole or predominant site of disease progression. Some tumor types, such as metastatic uveal melanoma, are refractory to therapies such as cisplatin and interstrand cross-linking agents such as mitomycin C, 6 and many systemically delivered therapeutics are dose limiting due to unacceptable thrombocytopenia and off-target systemic and organ toxicities. 2 For diseases where the primary metastases is confined to a single organ, such as the liver, a targeted regional therapy that has the potential to reduce metastases but isolate the target chemotherapeutic effects to the tumor-bearing tissue while simultaneously avoiding systemic toxicity, is desired. Liver-directed, high-dose chemotherapy has evolved from a highly invasive open surgical procedure to a minimally invasive procedure. Initially, IHP diffusion devices garnered clinical interest for more than 50 years but have not gained widespread application because of poorly defined treatment strategies that have limited clinical efficacy outcomes, resulting in significant systemic toxicity and lack of overall safety. Intrahepatic is a complex therapy to administer, and initial studies using IHP devices resulted in high morbidity and limited efficacy associated with the procedure. 3 The best results with hepatic-directed therapy have been with chemoembolization to inhibit leakage of the chemotherapeutic agent into the bloodstream but have been associated with hepatic leakage of agents such as melphalan and tumor necrosis factor α (TNF-α) into the systemic circulation and the combination dose administered which may be associated, in part, with the production of pro-inflammatory cytokines and catecholamines that resulted in a transient hyperdynamic cardiovascular profile. 4 Percutaneous hepatic perfusion (PHP) avoids typical laparotomy procedures and allows for careful positioning and repositioning of a hepatic arterial catheter within the hepatic artery to deliver chemotherapeutic agents locally within the liver. 2,7 –9 Nonclinical dose range-finding studies were originally conducted in healthy swine with a first-generation filtration device (GEN1) and melphalan to establish a safe human starting dose for clinical studies. Subsequent second-generation device development (GEN2) was conducted to enhance filtration efficiency. Melphalan is a small alkylating agent and is a common drug of choice because of its known antitumor activity in an IHP setting. 3,4 A double balloon catheter placed in the retrohepatic vena cava is used to shunt venous blood containing melphalan through an extracorporeal (EC) loop containing a high-efficiency filtration system to remove the melphalan prior to returning blood to the systemic circulation so that only the liver is exposed to the test article therapy. In the present study, a liver-directed, PHP approach using a higher efficiency (GEN2) filtration device and EC loop was used in a healthy swine to prevent off-target organ and systemic toxicity while at the same time circulating high-dose melphalan (220 mg/500 mL; human equivalent dose 3.0 mg/kg tested in clinical trials) within the liver for a shorter duration than was used in IHP and first GEN1 studies and with a high degree of safety, efficacy, and limited cardiovascular risk. Yorkshire swine were considered the most appropriate preclinical model due to anatomical and vascular similarities to the human hepatic system and due to feasibility of catheterization. To enhance treatment outcomes, hepatic mapping and/or coil embolization were performed, and animals were maintained on Neupogen®(Amgen Inc, USA) to mitigate neutropenia postoperatively.
Materials and Methods
Procedure
Fifteen healthy female fully acclimated Yorkshire swine (81-103 kg) were randomly assigned by computer-generated random number selection (Microsoft Excel) to 6 groups (3 different treatments) for PHP with saline or melphalan administration as shown in Table 1. At least 7 days prior to the interventional procedure, animals underwent hepatic mapping and/or coil embolization of the hepatic artery branches to the gastrointestinal branches. On day 0, the animals in groups 1 and 2 were subjected to a sham PHP EC circuit without filter, saline infusion procedure; groups 3 and 4 underwent a PHP procedure during which saline was administered as a continuous 30-minute infusion into the hepatic artery through a hepatic delivery system (HDS) utilizing a GEN2 filter. Groups 5 and 6 underwent a PHP procedure during which melphalan HCl (220 mg/500 mL) was administered as a continuous 30-minute infusion into the hepatic artery through the HDS utilizing a GEN2 filter for survival up to 3 or 28 days. The efficacious dose was determined from prior dose range-finding studies in swine (unpublished) and from human clinical studies using 3.0 mg/kg in a 70-kg human. Blood was returned to the animal through a jugular venous return sheath. Following the 30-minute infusion, the EC blood filtration washout phase was conducted for 30 minutes. Activated clotting times, blood gases, and blood pH were monitored during the procedures. Animals were maintained on Neupogen for up to prenecropsy or 5 days postprocedure.
Treatment Groups.
Abbreviations: GEN2, generation 2 filtration; PHP, percutaneous hepatic perfusion; EC, extracorporeal loop.
Test System Justification
A large animal species was required to mimic the size and hepatic anatomy of humans and for cross-comparison with previous studies; all previous work has been performed in swine. In addition, the swine model is required for testing by the Food and Drug Administration. The swine model was chosen for this study because the size of the liver and vessels is similar to humans. and pigs have previously been used for similar chemosaturation/blood filtration studies.
Toxicology Parameters
Morbidity, mortality, animal health, clinical signs, body weights, body condition scores, clinical pathology, hematology, gross pathology, and histopathology were conducted. Necropsies were performed on days 3 and 28 posttreatment.
Blood Sampling
Pre- and postfilter blood/plasma samples were collected at multiple time points for the assessment of clinical chemistry, hematology, melphalan toxicokinetics (TK), cytokines, catecholamines, and cardiac biomarkers at baseline on day 0 up to 300 minutes postinfusion on interim days 2 to 4 and at prenecropsy on days 3 or 28 posttreatment.
Toxicokinetics
Blood samples were collected from pre- and postfilter for TK analysis in prefilter and postfilter samples at baseline (T0) and during infusion of saline or melphalan (T0, 10, 20, and 30) and in systemic samples at the end of infusion (T5, 10, 15, 30, 60, 120, 210, and 300 minutes postinfusion).
Cardiovascular Assessments
A comprehensive cardiovascular risk assessment was conducted to include continuous blood pressure monitoring, electrocardiogram (ECG, MacLab; GE Health Care, Chicago, Illinois), and echocardiogram (ECHO, Sequoia 512; Siemens, Malvern, Pennsylvania) obtained at baseline, throughout the procedure, and postoperatively. Additional pre- and postfilter blood samples were collected from baseline and at regular intervals throughout the procedure to measure catecholamines (dopamine, adrenaline, noradrenaline, inflammatory cytokines [interleukin, IL-4, IL-6, IL-8, interferon, IFN-γ, and TNF-α], and cardiac enzymes [creatine kinase, CK, CKMB, and troponin 1]).
Necropsy/Histopathology
On days 3 and 28, the animals were euthanized, subjected to a comprehensive necropsy, and the catheterization sites in the hepatic artery and inferior vena cava with other designated tissues were collected for histological evaluation.
Light microscopy of representative samples of the liver, heart, kidneys, lungs, brain, spleen, bone marrow, and tissues with macroscopic changes was assessed for any toxicologic or other histopathologic responses. Evaluated histopathologic parameters included necrosis, fibrosis, and inflammation (with characterization of cellular constituents), and additional parameters were scored at the discretion of the pathologist. Representative organ sections were assessed according to Table 2.
Histomorphometry Scoring Matrix.a

a Data were analyzed by t test and analysis of variance. Data are expressed as mean (±standard deviation [SD]) or standard error of the mean (SEM) value.
Results
There were no intraprocedural complications and all animals survived to their scheduled necropsy. The animals maintained good body condition and the 28-day groups maintained or gained weight as expected for the breed/age. Minor clinical observations included decreased appetite in 9 of 15 animals. This lasted for 3 to 9 days in most cases but did not affect overall body weight gain by the time of necropsy.
Toxicokinetics
Melphalan concentrations increased during hepatic drug infusion at all sampling sites, reaching a peak at the end of infusion. Melphalan concentrations at the postfilter site were similar to those at the systemic site, which were both considerably lower than at the prefilter site. Mean melphalan exposures, as reflected by maximum plasma concentration (Cmax) and area under the curve (AUC) values during the drug infusion phase, were 27 to 44 times higher at the prefilter sampling site than at the postfilter and systemic sampling sites. During the washout phase, the mean prefilter AUC value was approximately 14 and 15 times higher than at the postfilter and systemic sites, respectively. After the completion of the washout phase (postfiltration phase), melphalan concentrations at the systemic sampling site increased rapidly to maximum levels comparable to those seen during the drug infusion phase and then declined in a generally biexponential fashion with a terminal half-life (t½) of approximately 70 minutes. The observed spike in systemic melphalan post washout may be a result of residual melphalan release from the liver. Limiting the washout phase to 30 minutes helps to ensure that the effect of treatment on blood cells, enzymes, and hemodynamics is transient and reversible. The dramatically higher melphalan concentrations at the prefilter site relative to those at the postfilter and systemic sites indicates the GEN2 filter was able to extract melphalan from blood with high efficiency, which reduced melphalan exposure in the systemic circulation considerably and shown in Figure 1A to C.
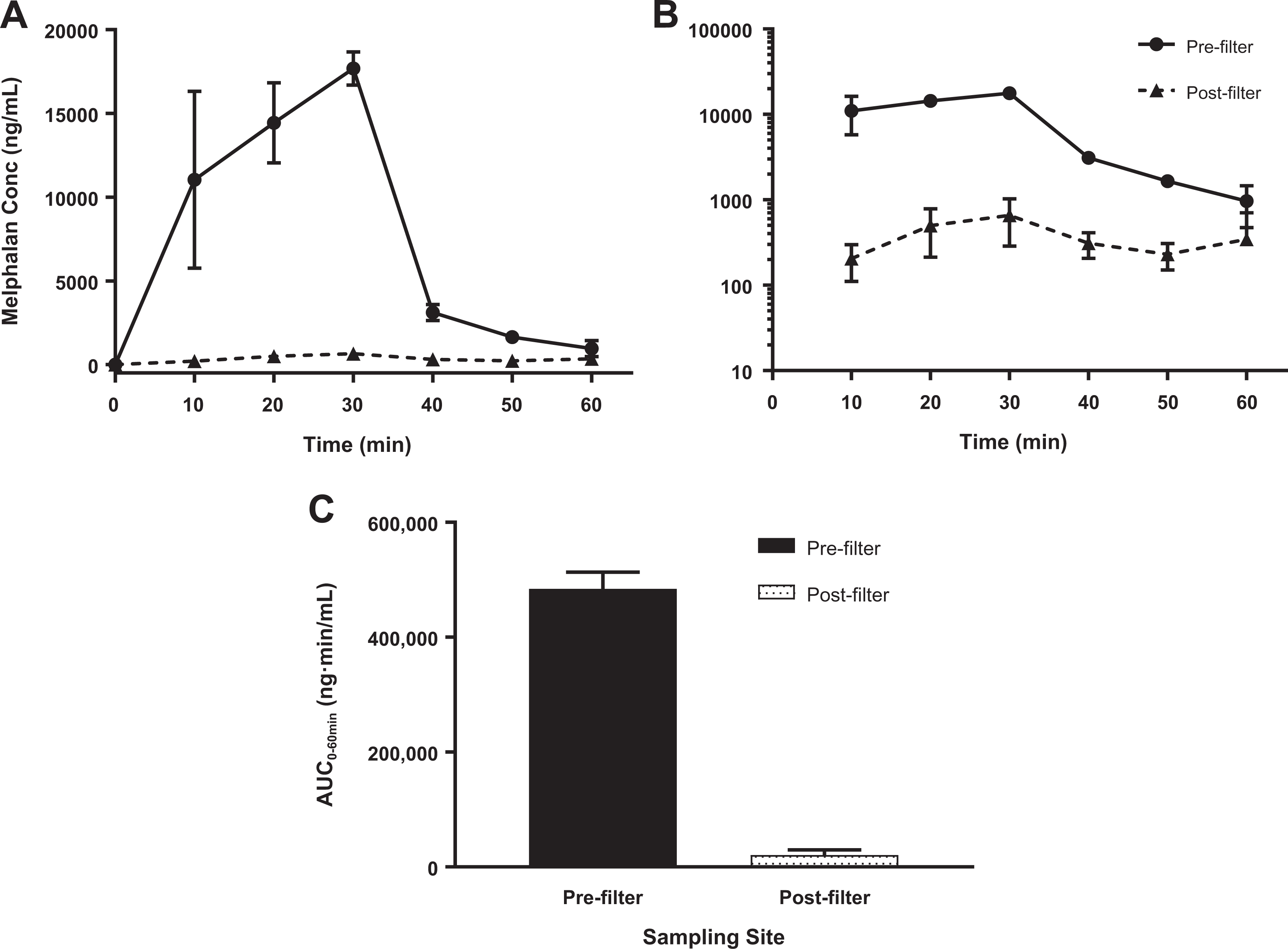
Mean (±standard deviation [SD]) melphalan plasma concentration–time curves. (A) Rectilinear; (B) Semilogarithmic, and (C) area under the curve values from 0 to 60 minutes (AUC0-60min) at the pre- and postfilter sampling sites. Female pigs (n = 5) received 3 mg/kg melphalan HCl by percutaneous hepatic perfusion with the generation 2 (GEN2) filtration system in the EC Loop. Drug was perfused over the first 30 minutes, followed by a washout period from 30 to 60 minutes. Plasma melphalan concentrations reached maximum levels at the end of infusion at both sampling sites and declined rapidly during the washout period. Concentrations at the prefilter site were substantially higher than those at the postfilter site. Similarly, the Mean AUC0-60min value at the prefilter site was 33 times higher during the infusion period and 23 times higher over the 60-minute time course than that at the postfilter sampling site. These data indicate highly efficient extraction of melphalan from plasma by the GEN2 filtration system.
Clinical Chemistry
Animals in the melphalan groups had notable elevated aspartate aminotransferase (AST; Figure 2A) and alanine aminotransferase (ALT; Figure 2B) during PHP, which reached peak levels at 210 minutes in postfiltered samples and was resolved by day 3 posttreatment for both enzymes. These effects were possibly attributed to residual systemic melphalan which had limited duration of effect. Elevated glucose levels were common in all groups during the bypass/perfusion time points, which may be related to intracellular glucose release or catecholamine-stimulated hyperglycemia. All animals had normal glucose levels by day 3 or 4 as shown in Figure 2C.
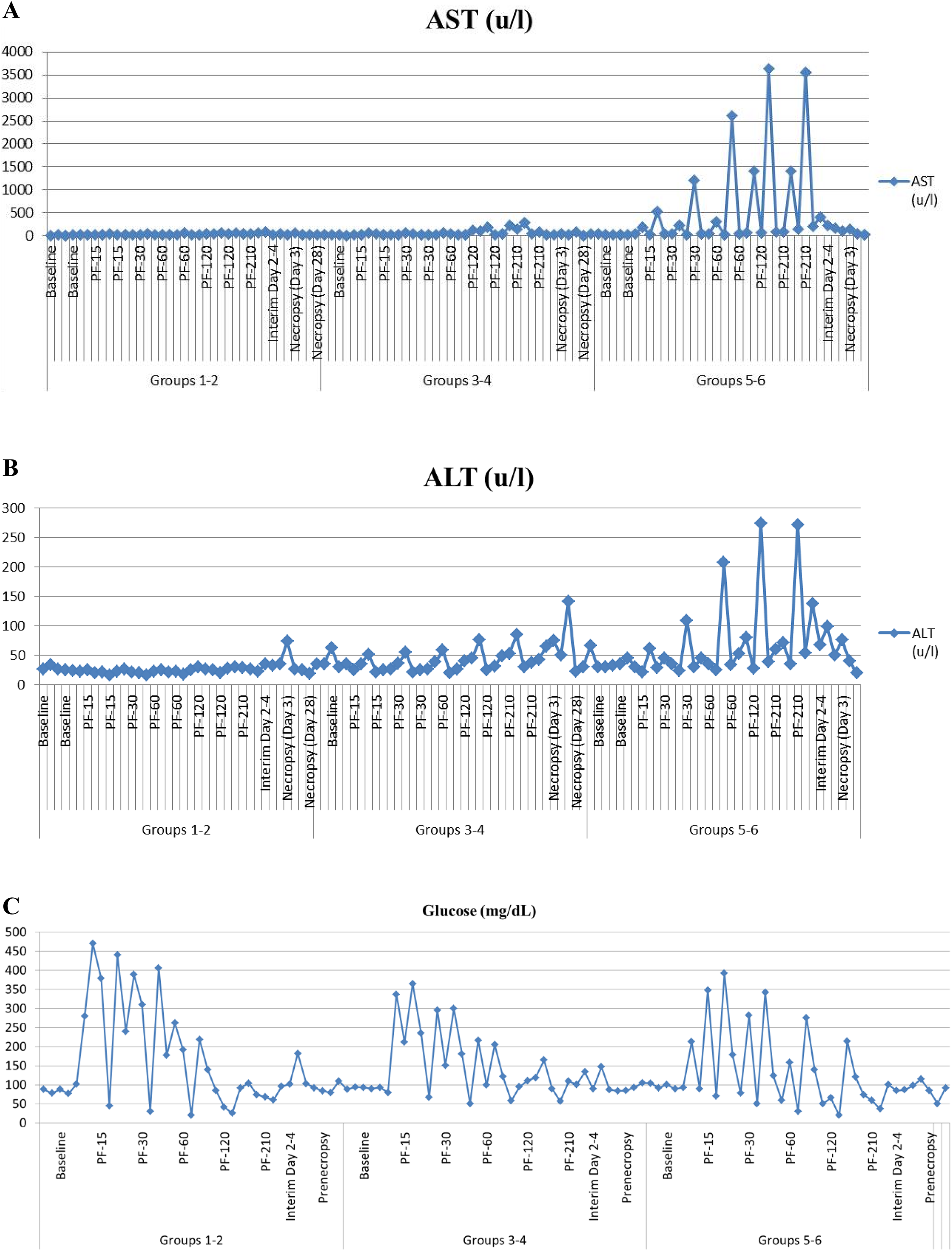
Clinical chemistry: Animals in the melphalan groups had notable elevated aspartate aminotransferase (AST; A) and alanine aminotransferase (ALT; B) during percutaneous hepatic perfusion (PHP), which reached peak levels at 210 minutes in postfiltered samples and was resolved by day 3 posttreatment for both enzymes. These effects were possibly attributed to residual systemic melphalan which had limited duration of effect. Elevated glucose levels were common in all groups during the bypass/perfusion time points which may be related to intracellular glucose release or catecholamine stimulated hyperglycemia. All animals had normal glucose levels by day 3 or 4 as shown in (C). (A) Aspartate aminotransferase, (B) ALT, and (C) glucose.
Hematology
All animals had a baseline prothrombin time of 12.9 to 19.8 seconds (reference range: 12.98-16.9 seconds) and partial prothrombin time of <20 to 22.2 seconds (reference range: 18.1-27.2 seconds). Additional doses of heparin were required throughout the procedure to maintain anticoagulation, and treatment was discontinued after PHP was complete. Animals maintained hematocrit values within the normal range (22.8%-44%) during PHP. There were no treatment- or test-article-related effects on albumin or globulin, and there was no evidence of intravascular or tissue thromboses observed by histopathology during recovery at 3 and 28 days. In melphalan-treated groups, mild neutrophilic or lymphocytic leukocytosis was evident at variable time points, although the animals were maintained on Neupogen daily for a minimum of 5 days postprocedure. The effect may be due to inflammation, stress, or the side effects of Neupogen and not indicative of infection or toxicity. Thrombocytopenia was observed during the perfusion procedure in 6 of 10 animals in the filter groups (saline and melphalan); platelet counts returned to normal within 3 to 4 days as shown in Figure 3. Thrombocytopenia was not observed in any of the nonfilter groups.
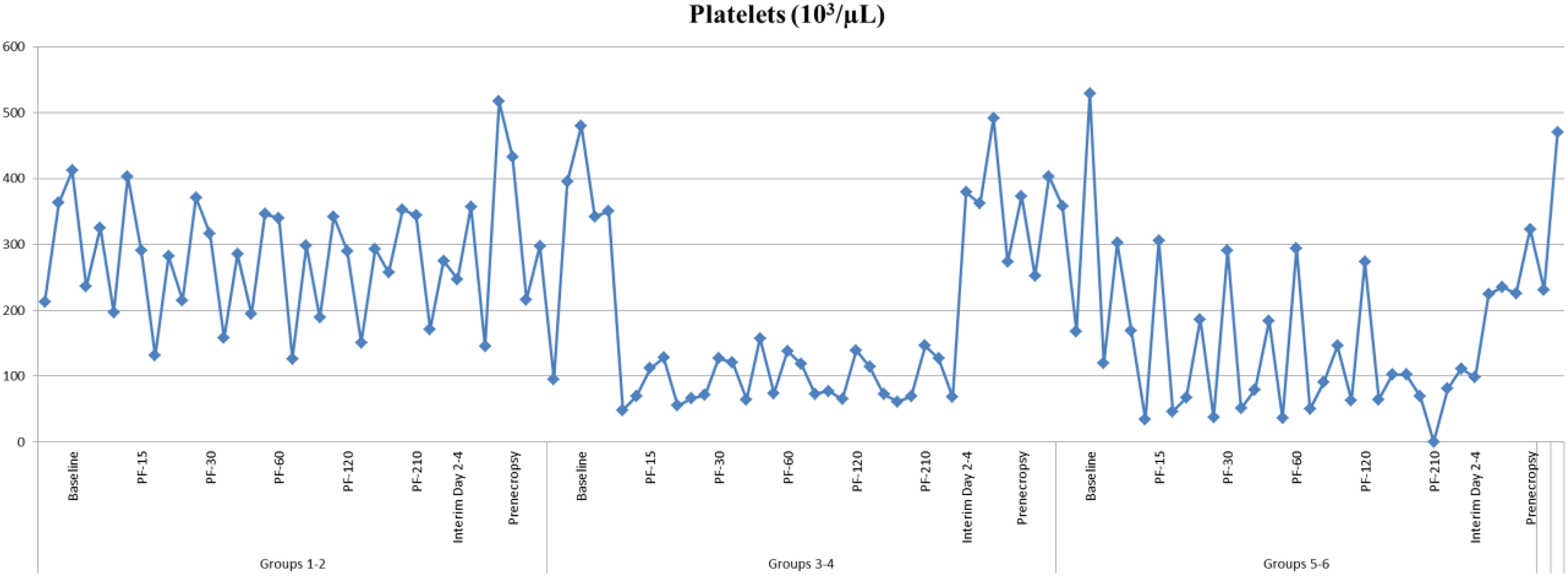
Hematology: Thrombocytopenia of platelets was observed during the perfusion procedure in 6 of 10 animals in the filter groups (saline and melphalan); platelet counts returned to normal within 3 to 4 days as shown in Figure 3. Thrombocytopenia was not observed in any of the nonfilter groups.
Cardiovascular Hemodynamics
All animals, regardless of treatment group, experienced hypotension requiring vasopressor support during filtration followed by compensatory and reflex tachycardia. The induction of hypotension and tachycardia was primarily due to the release of approximately 750 mL of blood from the animal into the EC circuit and reduced venous return of blood to the heart. Hypotension resolved immediately after filtration and return of blood volume to the systemic circulation, followed by hypertension due predictably to residual effect of vasopressor (synthetic phenylephrine) administration. Mean arterial blood pressure <40 mm Hg with resultant acidemia and arterial pH <7.2, despite use of intravenous crystalloid fluid and vasopressor support, is consistent with transient shock during the filtration phase. All hemodynamic aberrations rapidly normalized following filtration and administration of standard supportive measurements to facilitate return to euvolemia as shown in Figure 4A to C for all treatment groups.
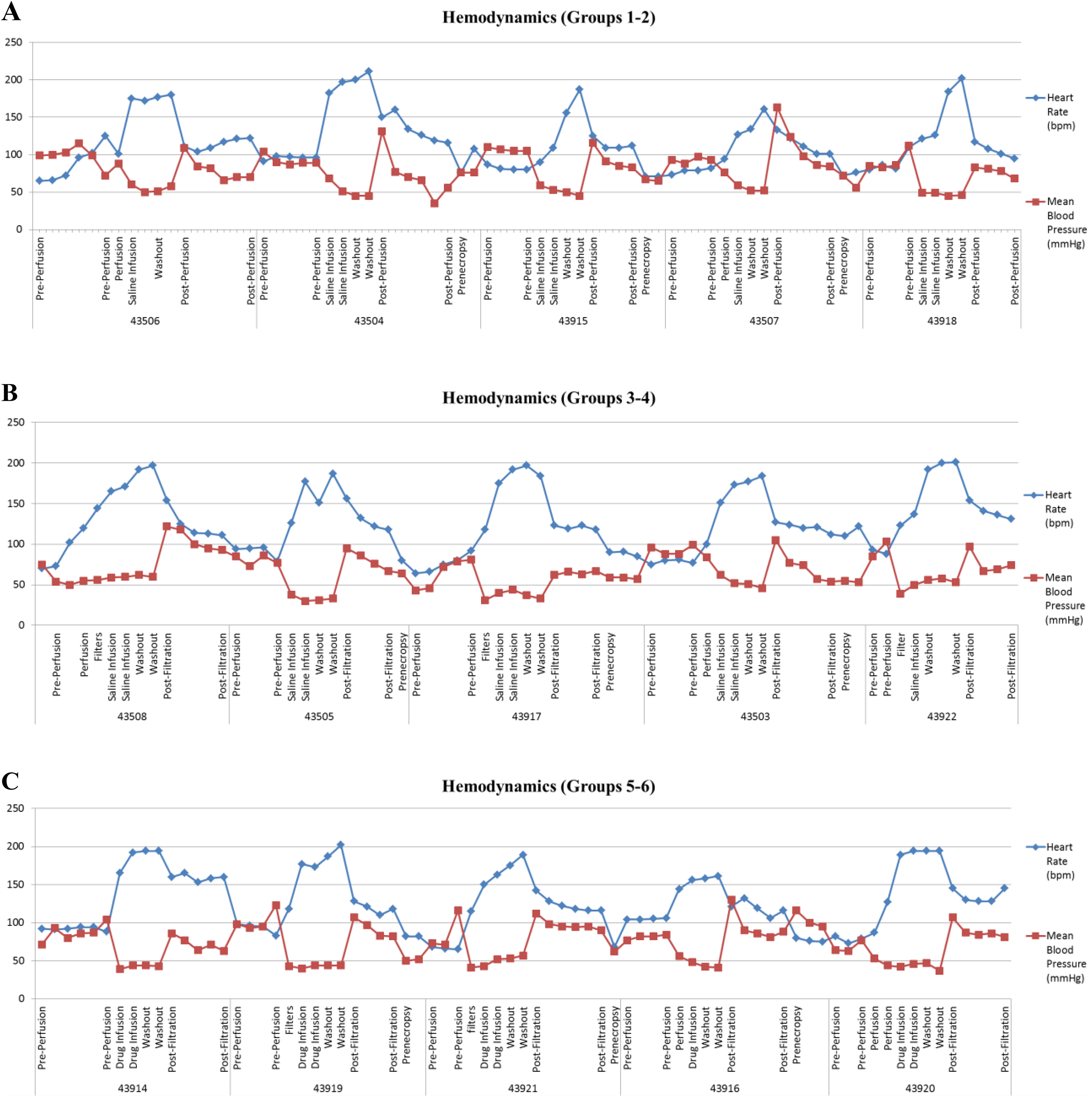
Cardiovascular hemodynamics. Regardless of treatment group, all animals experienced hypotension requiring vasopressor support during filtration followed by compensatory and reflex tachycardia. The induction of hypotension and tachycardia was primarily due to the release of approximately 750 mL of blood from the animal into the EC circuit and reduced venous return of blood to the heart. Hypotension resolved immediately after filtration and return of blood volume to the systemic circulation, followed by hypertension due predictably to residual effect of vasopressor (synthetic phenylephrine) administration. Mean arterial blood pressure <40 mm Hg with resultant acidemia and arterial pH <7.2, despite use of intravenous crystalloid fluid and vasopressor support, is consistent with transient shock during the filtration phase. All hemodynamic aberrations rapidly normalized following filtration and administration of standard supportive measurements to facilitate return to euvolemia as shown in (A) to (C) for all treatment groups. A, Heart rate and blood pressure (groups 1 and 2). B, Heart rate and blood pressure (groups 3 and 4). C, Heart rate and blood pressure (groups 5 and 6).
Electrocardiogram and ECHO
No ECG changes suggestive of myocardial ischemia or infarction and no malignant arrhythmias were noted across all animals and time points. All animals across all treatment cohorts and time points exhibited normal (EF > 45%) or hyperdynamic left ventricular systolic function as assessed by 2-D multiplane transesophageal echocardiography as shown in Figure 5.
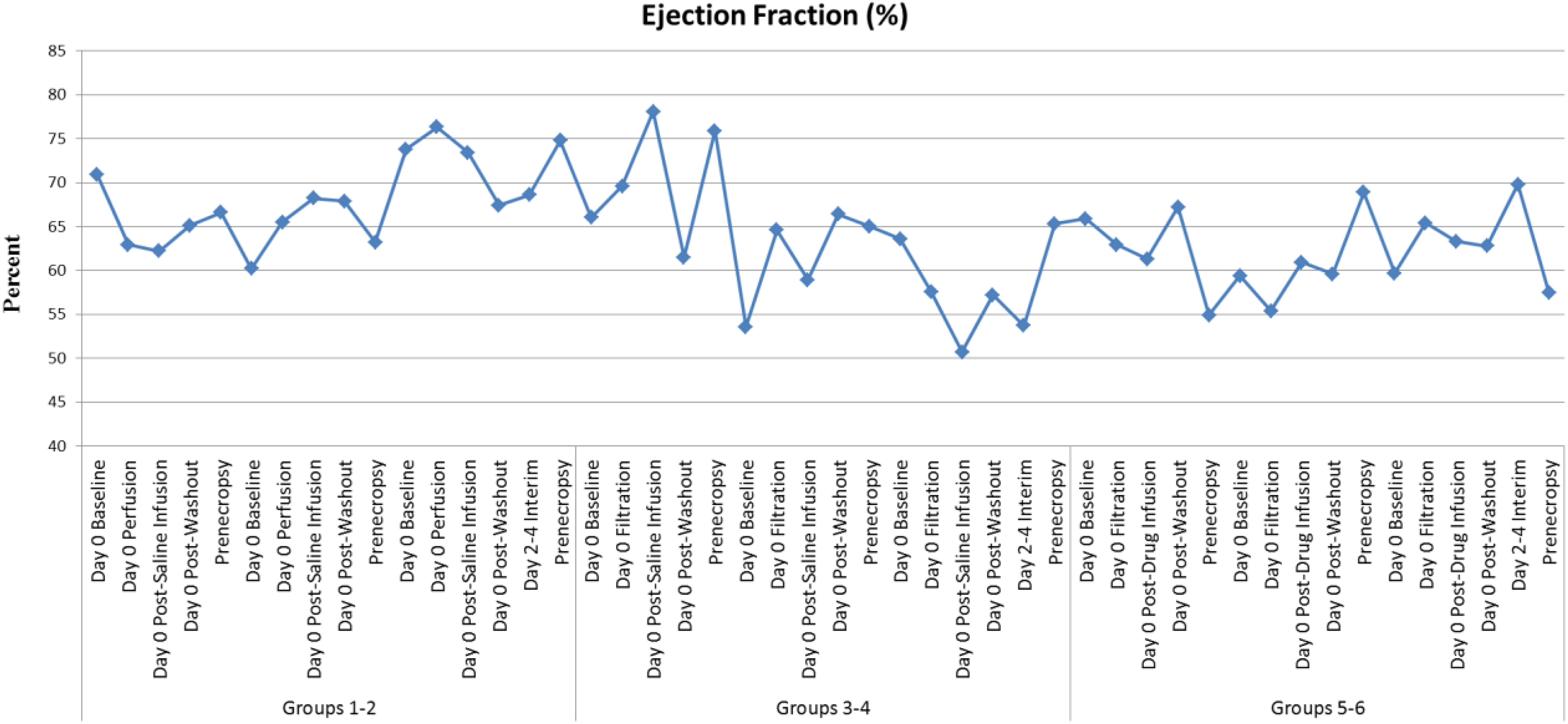
Electrocardiogram (ECG) and echocardiograms (ECHO): Ejection fraction (EF). No ECG changes suggestive of myocardial ischemia or infarction and no malignant arrhythmias were noted across all animals and time points. All animals across all treatment cohorts and time points exhibited normal (EF > 45%) or hyperdynamic left ventricular systolic function as assessed by 2-D multiplane transesophageal echocardiography as shown.
The significant hemodynamic challenge imposed by filtration resulted in a compensatory tachycardia with supranormal left ventricular function. Prenecropsy imaging showed no wall motion abnormalities, and left ventricular function remained normal.
Cardiac Biomarkers
Elevations in cardiac troponin-I (Tn-I) and CK-MB isoenzyme were noted in all treatment arms that required filtration (groups 3-6). Within the sham groups 1 and 2 that did not undergo filtration, there were no significant elevations in cardiac biomarkers. Among the animals that underwent filtration, 3 of 5 animals with saline treatment and 3 of 5 animals with melphalan had significant elevations in Tn-I. These elevations correlated with the degree of aforementioned sustained shock, with mean arterial blood pressure <40 mm Hg and consequent acidemia with arterial pH <7.2. Troponin I and CK-MB normalized at interim day 2 to 4 and prenecropsy, showing filtration impact on cardiac biomarkers was transient and reversible as shown in Figure 6A to C. The transient elevations were not associated with any evidence of cardiac muscle damage observed grossly at necropsy and by histopathology.
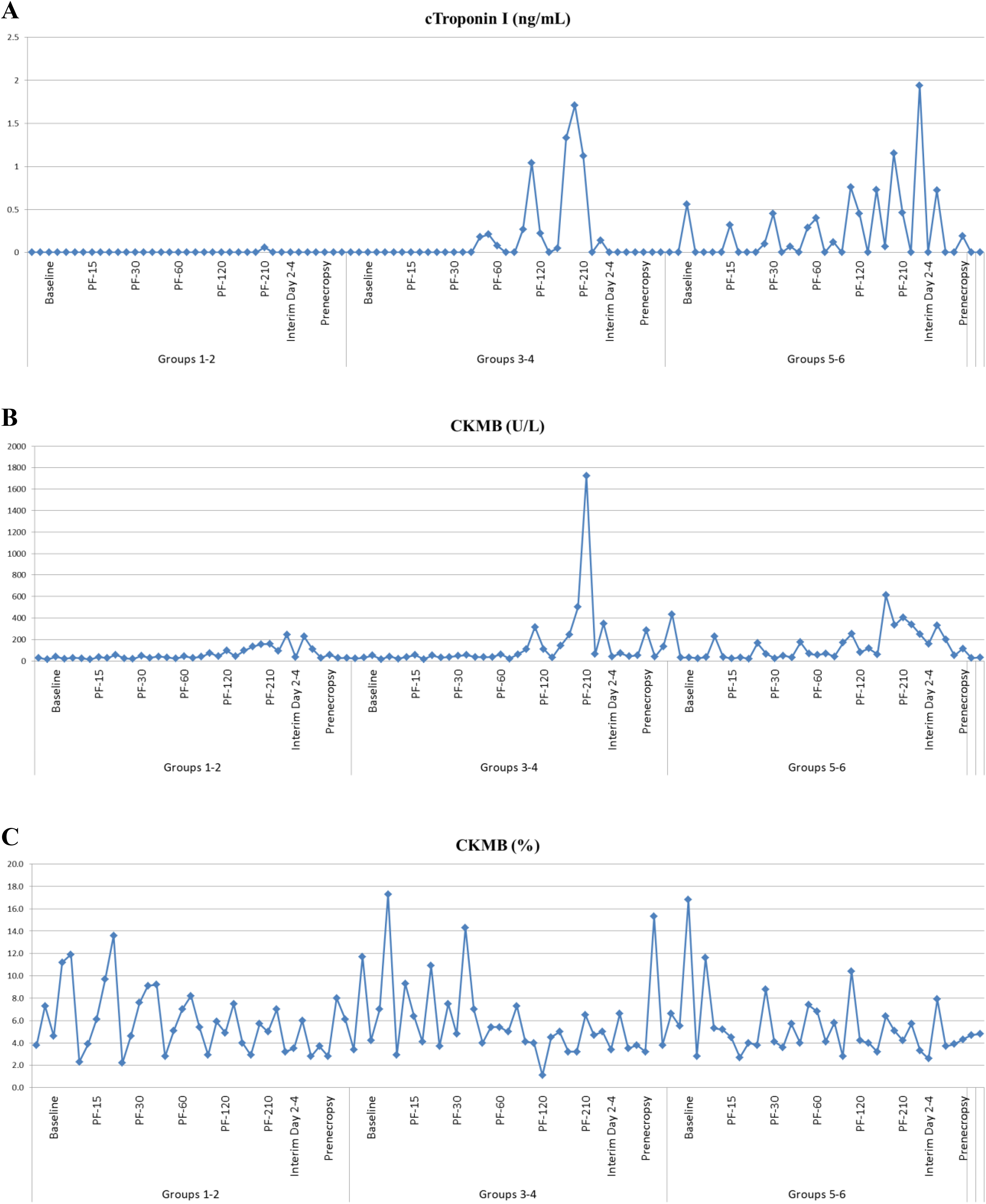
Cardiac biomarkers elevation in troponin-I (cTn-I) and creatine kinase (CK) MB isoenzyme was noted in all treatment arms that required filtration (groups 3-6). Within the sham groups 1 and 2 that did not undergo filtration, there were no significant elevations in cardiac biomarkers. Among the animals that underwent filtration, 3 of 5 animals with saline treatment and 3 of 5 animals with melphalan had significant elevations in troponin-I. These elevations correlated with the degree of aforementioned sustained shock with mean arterial blood pressure <40 mm Hg and consequent acidemia with arterial pH <7.2. Troponin I and CKMB normalized at interim day 2 to 4 and prenecropsy, showing filtration impact on cardiac biomarkers was transient and reversible as shown in (A) to (C). A, cTroponin 1. B, Creatine kinase (CK) MB. C, Creatine kinase (CK) MB%.
Cytokines
There did not appear to be an effect of filtration with saline or melphalan on systemic cytokine levels. There was a transient elevation in IFN-γ throughout the postfiltration phase that appeared to trend toward baseline at 3 days posttreatment and normalize by 28 days posttreatment (data not shown).
Catecholamine Analysis
Catecholamine filtration was assessed in response to PHP, and adrenaline, noradrenaline, and dopamine levels could not be used to supplement blood pressure changes. In the clinical setting, catecholamine supplementation is used in patients undergoing PHP with the GEN2 filtration device to promote hemodynamic stabilization. Filtration with the GEN2 filter caused markedly decreased catecholamine levels in the postfiltration sampling but had no effect on unfiltered samples and a lesser effect on the systemic levels of catecholamines. In general, with the exception of systemic dopamine, noradrenaline and adrenaline levels decreased toward baseline postprocedure. Systemic dopamine levels remained elevated at 5 hours postprocedure in groups 5 to 6 only, indicating a direct or indirect effect of melphalan on systemic dopamine levels as shown in Figure 7A to C.
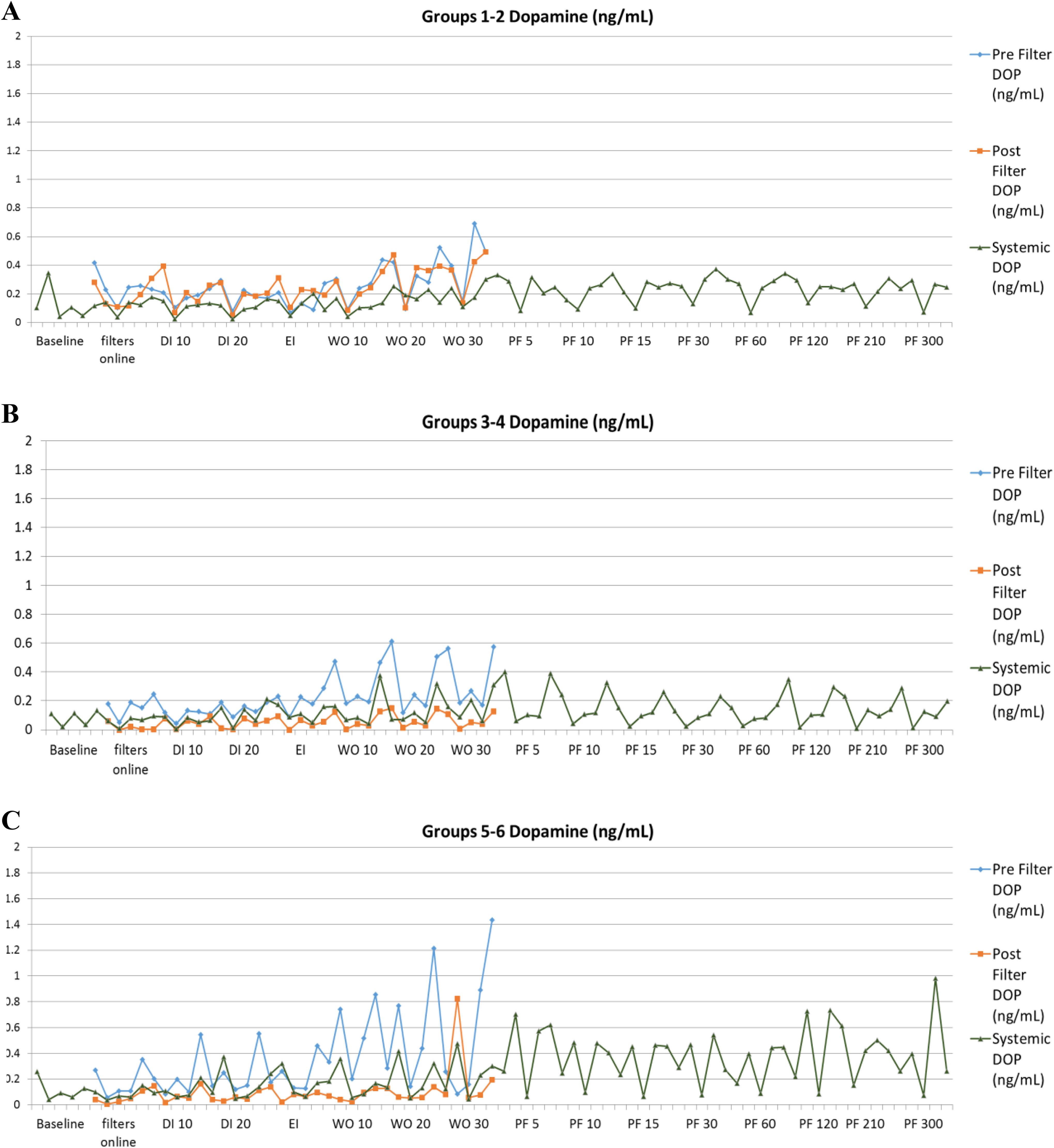
Catecholamines: Dopamine filtration with the generation 2 (GEN2) filtration caused markedly decreased catecholamine levels in the postfiltration sampling but had no effect on unfiltered samples and a lesser effect on the systemic levels of catecholamines. In general, with the exception of systemic dopamine, noradrenaline and adrenaline levels decreased toward baseline during the postprocedure. Systemic dopamine levels remained elevated at 5 hours postprocedure in groups 5 and 6 only, indicating a direct or indirect effect of melphalan on systemic dopamine levels as shown in (A) to (C). A, Dopamine (groups 1-2). B, Dopamine (groups 3-4). C, Dopamine (groups 5-6).
Gross Pathology and Histopathology
Macroscopic observations were present in nearly all animals among day 3 animals, regardless of treatment group, and in both animals from each treatment group at day 28. At both time points, animals in all treatment groups had macroscopic and histologic liver changes that were consistent with postmortem, ex vivo artifact such as autolytic degeneration of hepatic parenchyma (fixation artifact). The results from representative tissues, liver, kidney, heart, and brain are shown in Figure 8A to E.
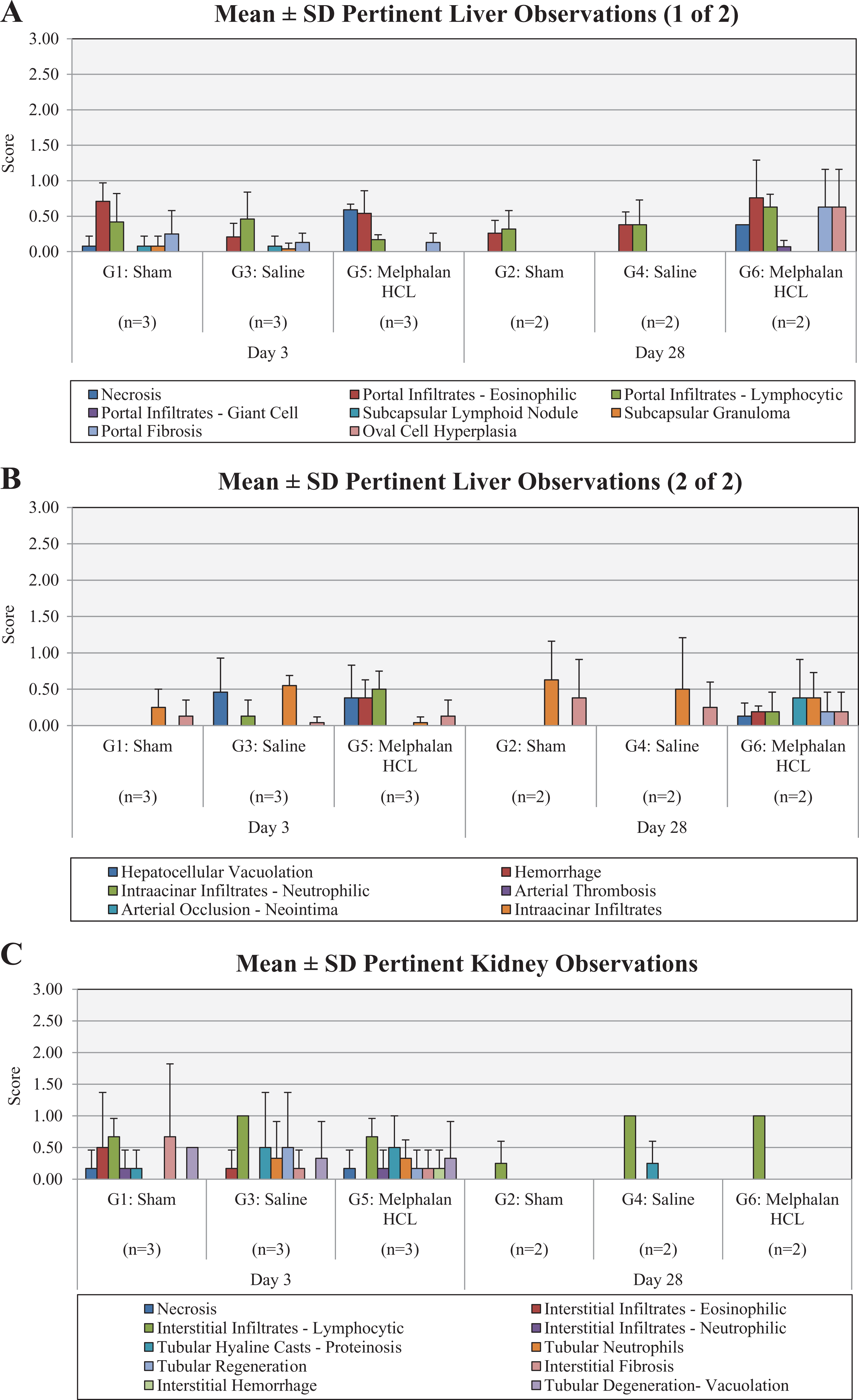
Histopathology liver: Animals in all treatment groups at day 3 and from the sham and saline groups at day 28 exhibited pale foci correlated with typically minimal fibrosis (eg, portal, subcapsular). The presence of such a change, and of portal eosinophilic and/or lymphocytic inflammation, can be due to subclinical or previous infectious, inflammatory, or parasitic process. Areas of hepatic necrosis consistent with melphalan-induced injury were present in all melphalan-treated animals at both time points, but in no other animals. After 3 and 28 days in the porcine model of percutaneous hepatic perfusion, the percutaneous hepatic perfusion (PHP) system with the generation 2 (GEN2) hemofiltration device was, with few exceptions, associated with no device-related adverse tissue responses in local vessels and representative tissues, both with and without concurrent administration of melphalan (A-B). Kidney: Animals from all treatment groups at both time points exhibited minimal renal interstitial inflammatory infiltrates and at day 3 exhibited uncommon and negligible interstitial fibrosis, both of which are considered common and incidental background findings. Renal tubular hyaline casts proteinosis, indicative of slight urinary protein leakage, were minimally present in all day 3 animals and the saline group at day 28. There was rare and negligible (ie, less than minimal) acute tubular necrosis at day 3 in 1 animal each from the sham and melphalan group. One animal from the sham group at day 3 with mild baseline and repeated elevations in serum blood urea nitrogen (BUN) and creatinine exhibited microscopic renal changes consistent with a mild congenital renal dysplasia (C). Heart: Histologic findings in cardiac tissue were limited to a notable focus of ventricular myocardial necrosis at day 3 in saline group, with mineralization and lymphohistiocytic infiltrates. This is consistent with occurrence at the time of interventional procedures (D). Brain: Observations in the brain were uncommon and included minimal vacuolation of the neuropil (brain parenchyma), minimal focal hemorrhage, minimal gliosis (increased cellularity), discrete/localized cerebellar hypoplasia/chronic atrophy in random animals, and did not appear to be associated with melphalan toxicity (E). A, Liver (1). B, Liver (2). C, Kidney. D, Heart. E, Brain.
Liver
Animals in all treatment groups at day 3 and from the sham and saline groups at day 28 exhibited pale foci correlated with typically minimal fibrosis (eg, portal, subcapsular). The presence of such a change and of portal eosinophilic and/or lymphocytic inflammation can be due to subclinical or previous infectious, inflammatory, or parasitic process. Areas of hepatic necrosis consistent with melphalan-induced injury were present in all melphalan-treated animals at both time points, but in no other animals. After 3 and 28 days in the porcine model of PHP, the PHP system with the GEN2 hemofiltration device was, with few exceptions, associated with no device-related adverse tissue responses in local vessels and representative tissues, both with and without concurrent administration of melphalan. Aside from treatment-induced hepatic changes, responses in each were comparable to those seen in the sham treatment group. Vena cava inflammation was negligible at day 3 in all groups and was absent at day 28. In catheterized hepatic artery sections, inflammation was absent or negligible in all groups at day 3 and absent at day 28. Endothelialization in catheterized artery sections was nearly complete at day 3 and complete at day 28, regardless of the treatment group. Multifocal or regional hepatic necrosis of mild–moderate or marked severity was present only in the melphalan group (all animals at both time points). The presence of necrosis was restricted to the area of drug administration with little to no involvement of surrounding tissue. Acute hepatic necrosis was evident at day 3, with chronic changes still visible at day 28 due to ongoing inflammatory and proliferative repair responses in the large affected regions. In the normal, healthy, swine model, repositioning of the catheter to reach the whole of the liver other than the left lateral and medial lobes is limited by the swine hepatic anatomy. Catheter placement situated high within the hepatic artery was determined to prevent leakage of melphalan into lower gastric collateral blood vessels. Therefore, normal, healthy liver tissue was perfused for the duration of the infusion period in only 1 site, potentially resulting in higher, localized exposure to melphalan and more tissue damage than would be observed clinically.
Kidney
Animals from all treatment groups at both time points exhibited minimal renal interstitial inflammatory infiltrates and, at day 3, exhibited uncommon and negligible interstitial fibrosis, both of which are considered common and incidental background findings. Renal tubular hyaline casts proteinosis, indicative of slight urinary protein leakage, were minimally present in all day 3 animals and in the saline group at day 28. There was rare and negligible (ie, less than minimal) acute tubular necrosis at day 3 in 1 animal each from the sham and melphalan groups. One animal from the sham group at day 3 with mild baseline and repeated elevations in serum blood urea nitrogen and creatinine exhibited microscopic renal changes consistent with a mild congenital renal dysplasia.
Heart
Histologic findings in cardiac tissue were limited to a notable focus of ventricular myocardial necrosis at day 3 in the saline group, with mineralization and lymphohistiocytic infiltrates. This is consistent with occurrence at the time of interventional procedures.
Brain
Observations in the brain were uncommon and included minimal vacuolation of the neuropil (brain parenchyma), minimal focal hemorrhage, minimal gliosis (increased cellularity), and discrete/localized cerebellar hypoplasia/chronic atrophy in random animals and did not appear to be associated with melphalan toxicity.
Among other representative tissue, the target organs that are most highly perfused (spleen) or susceptible to the target effects of melphalan (bone marrow) showed minimal histopathological changes. There was minimal bone marrow granulocyte hypoplasia in the melphalan group at day 3; there were no findings at day 28. Minimal neutrophilia in the splenic red pulp at this time point was observed in the melphalan group; there were no findings at day 28 (data not shown).
Discussion
Liver-directed, high-dose chemotherapy evolved from a highly invasive open surgical procedure to a minimally invasive procedure. Initially, IHP diffusion devices garnered clinical interest for more than 50 years but have not gained widespread application because of poorly defined treatment strategies that have limited clinical efficacy outcomes, resulting in significant systemic toxicity and lack of overall safety. Intrahepatic perfusion is a complex therapy to administer, and initial studies using IHP devices resulted in high morbidity and limited efficacy associated with the procedure. 3 The best results with hepatic-directed therapy have been with chemoembolization to inhibit leakage of the chemotherapeutic agent into the bloodstream. 10 Maglivit et al reported results of chemoembolization using cisplatin for patients with metastasized uveal melanoma and observed a 50% response rate and a median survival time of 12 months. Leyvraz et al 11 treated 31 patients with monthly intra-arterial fotemustine (100 mg/m2) over 4 hours via the hepatic artery. The overall partial response rate was 40%, the median duration of response was 11 months, and overall survival was 14 months. Alexander et al 4 described a clinical study in which 29 patients received a 60-minute hyperthermic IHP using 1.5 mg/kg with no evidence of treatment-related mortality, 3 complete responses, and 15 partial responses with median actuarial progression-free and overall survivals of 8 and 12.1 months, respectively. Although advances have been made, the main limitations of IHP appear to be associated with the methods used for shunting blood externally to provide a targeted treatment effect while managing an efficacious chemotherapeutic dose and at the same time mitigating systemic toxicity. In the latter study, 4 a cannula positioned in the gastroduodenal artery, and in an isolated segment of the retrohepatic inferior vena cava following laparotomy, allowed for blood to be shunted externally to the axillary vein using a veno–veno bypass circuit. The dose of melphalan (1.5 mg/kg: mean total dose of 105 mg) was associated with substantial toxicity, presumably due to hepatic leakage of melphalan and TNF-α into the systemic circulation and the combination dose administered which may be associated, in part, with the production of pro-inflammatory cytokines and catecholamines that resulted in a transient hyperdynamic cardiovascular profile.
Percutaneous hepatic perfusion as a technique to deliver high-dose chemotherapy regionally via the hepatic artery involves percutaneously placed catheters to isolate hepatic vascular inflow and outflow and enable simultaneous hepatic venous hemofiltration. 2,9,12 With this technique, the need for surgical procedure is obviated with the exception of establishing ports for arterial and venous blood collection. Throughout the treatment procedure, assessment of the pharmacokinetic (PK)/TK plasma concentrations of melphalan showed dramatically higher melphalan concentrations at the prefilter site relative to those at the postfilter and systemic sites, indicating that the GEN2 filter was able to extract melphalan from blood with high efficiency and reduce melphalan exposure in the systemic circulation. The 27- to 44-fold decrease in Cmax in the systemic circulation versus the prefilter site indicates a substantial improvement over the GEN1 device, where the difference was 10-fold. Accordingly, the hemodynamic, immunosuppressive, immunotoxic, cardiotoxic, and histopathologic effects of melphalan were limited. There were no treatment-related deaths associated with the PHP procedure and GEN2 filtration with and without melphalan, and all animals survived to scheduled necropsy. Transient and reversible effects of GEN2 filtration with and without melphalan were restricted to decreased platelet counts, elevated blood glucose, and transient elevations in liver and cardiac enzymes (ALT, AST, troponin I, and CKMB), all of which were reversed during the postprocedure prior to necropsy. There were no effects of PHP on the hematocrit, albumin, globulin, or evidence of intra- or extravascular thromboses. Overall, at 3 and 28 days in the porcine model of PHP, the PHP system with GEN2 hemofiltration device was associated with no device-related adverse tissue responses in local vessels and representative tissues, both with and without concurrent administration of melphalan. There was no evidence of acute or chronic inflammation associated with melphalan administration or following filtration as evidenced by histopathology and analysis of minor fluctuations in systemic pro-inflammatory cytokines, which normalized to baseline levels prior to necropsy. These results demonstrate that a novel regional treatment designed to treat hepatic metastases, PHP, can be performed safely with predictable and manageable degree of transient toxicity, unlike other hepatic directed therapies.
In terms of cardiovascular risk, the assessments that were conducted showed that all hemodynamic aberrations rapidly normalized following filtration and administration of standard supportive measurements to facilitate return to euvolemia. No ECG changes suggestive of myocardial ischemia or infarction and no malignant arrhythmias were noted across all animals and time points. Melphalan has been associated with supraventricular tachycardia in the clinic when administered systemically in the context of bone marrow transplantation. 13 In the current study, transient and reversible effects on cardiac function were noted by echocardiography of the heart. The significant hemodynamic challenge imposed by filtration resulted in a compensatory tachycardia with supranormal left ventricular function. Prenecropsy imaging showed no wall motion abnormalities, and left ventricular function remained normal. Throughout the infusion period, catecholamines assessed in the study decreased and then quickly rebounded in prefilter and postfilter samples and during washout. Melphalan appeared to exhibit direct or indirect increases in systemic catecholamine levels. However, there is no literature in support of the mechanism related to this effect. With the exception of dopamine, elevated adrenaline and noradrenaline lowered or returned close to baseline during recovery at 3 and 28 days posttreatment. Transient effects of treatment on catecholamine levels did not significantly impact blood pressure or cardiovascular hemodynamics. In the clinical setting, catecholamines are supplemented to promote hemodynamic stabilization. Cardiac enzymes were transiently elevated throughout the PHP procedure but normalized during the recovery phases at 3 and 28 days posttreatment and were not associated with any histopathologic evidence of cardiac muscle damage. Histopathology results revealed evidence of necrotic foci in sections of liver at the treatment site only, with minimal findings in the kidney, brain, and bone marrow.
Conclusions
Transient and reversible effects of treatment on cardiac enzymes, catecholamines, and cytokines and reversible hemodynamic effects without cardiac damage indicated that PHP with melphalan was not cardiotoxic or immunotoxic under the conditions tested due to high efficiency of the filtration system to limit the exposure of melphalan to the systemic circulation. The PHP system with GEN2 hemofiltration effectively limited systemic toxicity and appeared to be safe and well tolerated in normal swine.
Footnotes
Acknowledgments
The authors wish to acknowledge Mr. Peter Markham, CEO, and the staff of CBSET, Lexington, Massachusetts, for their dedication and hard work to enable this study and execute its completion.
Author Contribution
Pauline E. McEwan contributed to conception and design; acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Lynn Bailey contribute to design, acquisition, and analysis; drafted the manuscript; and critically revised the manuscript. Christopher Scull contributed to interpretation; drafted the manuscript; and critically revised the manuscript. David Trost contributed to design and acquisition and critically the revised manuscript. John H. Keating and Misty Williams contributed to design; acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Robert J. Guttendorf contributed to analysis and interpretation; drafted the manuscript; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All of the authors, except Dr Christopher Scull, were paid consultants for the project. Dr. Pauline E. McEwan is a senior consultant employed by Delcath Systems for the duration of the study to participate in the study design, implementation, review of study reports and preparation of this manuscript. Dr Chris Scull is a senior consultant at Biologics Consulting and was involved in the manuscript writing review and reference review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was wholly funded by Delcath Systems.
