Abstract
Spontaneous unexpected events occasionally develop during the course of rodent preclinical toxicology studies. The presentation of serious adverse events on animal studies may require notification of these events to the Food and Drug Administration if the events are most likely the direct result of test article administration. Classical conditioning of emotional responses may occur over the course of a repeat-dose study and clinical observation calls of “convulsions” are reported to the study director and/or staff veterinarians. In the current heightened environment of most research laboratories related to general animal welfare issues, it is imperative to have an action plan that will help to elucidate the potential origins of these motor events. We provide 10 factors that should be considered to help the study director determine the most likely cause of these motor attacks as being organic or psychogenic in origin.
Introduction
Repetitive and rhythmic motor disturbances are not uncommon in laboratory rodents in standard preclinical toxicology studies conducted during drug development programs. In humans, psychogenic nonepileptic seizures (PNES) superficially resemble epileptic seizures but are not caused by the abnormal electrical discharges (paroxysms) in the brain which are associated with epileptic seizures. Both the motor disturbance and overt seizure represent a dramatic motor disturbance that are considered an experiential and behavioral manifestation of distress and should have some common neuropathways in both rodents and humans. Psychogenic motor disorders have a psychological etiology rather than organic 1 ; in the field of neurology, the term “functional” is synonymous with psychogenic and the current unresolved issue is not specifically related to the use of the term “psychogenic” but rather if these events should be referred to as “seizures” or “attacks.” 2 –4
For the research environment of a general toxicology laboratory conducting preclinical research under the Food and Drug Administration’s (FDA) Good Laboratory Practice guidelines (21 Code of Federal Regulations [CFR] §58.1; GLPs), it is essential that all personnel are trained and achieve proficiency in every aspect of the research protocol they are participating, such as clinical observations. In 2017, the International League Against Epilepsy published its latest operational classification scheme of seizure types in human epileptics 5 ; however, the American Veterinary Medical Association has not adopted this classification scheme for animal use.
There is a frequent lack of correspondence between the electrical activity recorded from the scalp and the explosive behavioral change observed as a convulsion. There is no obligatory relationship between the type of electroencephalogram (EEG) activity encountered and the clinical symptomatology including auras, subjective distortions, or hallucinations, types of automatisms (stereotypies), and levels of awareness. For the sake of simplicity, we have adopted an early definition of an ictal paroxysm first proposed by Klemm 6 as it relates to electrophysiological recording of drug-induced convulsions in animals. Klemm 6 (p228) referred to seizure as: “any convulsive motor disorder associated with bizarre electric activity”. Klemm clearly makes a distinction between the behavioral expression of a “convulsion” and the electrical activity that may be recorded during that behavioral change. Inspection of published EEG records that designate the recording as “epileptiform” or simply “seizures” can occur without a behavioral or convulsive episode. Therefore, as standard language, we have differentiated between the terms “seizure” and “convulsions.”
The term “seizure” has been defined as: A transient occurrence of signs and/or symptoms due to abnormal excessive or synchronous neuronal activity in the brain.
7
A paroxysmal transient, stereotypic, electrical discharge of neurons within the brain, associated with either lack of voluntary control of movement or dyscognition.
8
The idea that the brain can create neurologic deficits, such as paralysis, blindness, sensory loss, seizures, and movement disorders, from factors associated with learning or conditioning has fueled many psychology laboratories over the years. Hallett 19 proposed that physiologically we are not yet capable of differentiating between voluntary and involuntary movements. For an event to be considered psychogenic in animals, the veterinarian must rule out all other physiological possibilities or show that placebo administration ameliorates or diminishes the symptoms. When a psychogenic movement disorder is identified in laboratory animals during preclinical toxicology studies, it is critical to determine a process by which to differentiate between test article–related effects and those that develop as a result of classical conditioning (learning) phenomenon. Test article–related convulsions may require reporting to the FDA and threaten the viability of the test article to advance along the investigational new drug (IND) pathway. If not reported, the research laboratory may lose its credibility with the FDA or trigger a directed inspection by the agency. Under Organization for Economic Cooperation and Development guidelines, the laboratory may lose its regulatory license to conduct future research.
Convulsions Expressed During Preclinical Toxicology Studies
On September 29, 2010, FDA published a final rule amending the IND safety reporting requirements under 21 CFR part 312 to extend the safety reporting requirements for persons conducting bioavailability and bioequivalence studies under 21 CFR part 320, which were previously exempt from such reporting (https://www.fda.gov/downloads/Drugs/Guidances/UCM227351.pdf). Under previous administrative regulations (21 CFR 312.32c[1][i][A] and [B]), sponsors or their subcontracted laboratories (ie, Contract Research Organizations) investigating a drug under an IND were required to notify FDA and all participating investigators, in a written IND safety report, of any adverse experience associated with the use of the drug that was both serious and unexpected and any finding from tests in laboratory animals that suggested a significant risk for humans. The administrative policy was revised to more clearly distinguish between the terms “adverse events” and “adverse reactions.” The term “adverse event” is restricted to human clinical trial data and does not apply to preclinical animal research. However, the term “suspected adverse reaction” does apply to animal studies and means any adverse event for which there is a reasonable possibility that the drug caused the adverse event (21 CFR 312.32[a]). For the purposes of IND safety reporting, “reasonable possibility” means there is evidence to suggest a causal relationship between the test article and the adverse event. A suspected adverse reaction implies a lesser degree of certainty about causality than adverse reaction, which means any adverse event caused by a drug.
Under the revised FDA administrative policy, suspected adverse reactions in preclinical toxicology studies are the subset of all adverse events for which there is a reasonable possibility that the drug caused the event. Inherent in this definition, and in the requirement to report suspected adverse reactions, is the need for the sponsor to evaluate the available evidence and make a judgment about the likelihood that the drug actually caused the adverse event. The application of the “reasonable possibility causality standard” is consistent with the discussion about causality in the International Conference on Harmonization (ICH) E2A guideline (“ICH E2A guidance”). The term “adverse reaction” is defined to mean an undesirable effect, reasonably associated with use of a drug that may occur as part of the pharmacological action of the drug or may be unpredictable in its occurrence. A suspected adverse reaction is considered “serious” under the following stated conditions: Examples of such medical events include allergic bronchospasm requiring intensive treatment in an emergency department or at home, blood dyscrasias or convulsions that do not result in inpatient hospitalization, or the development of drug dependency or drug abuse.
Under the FDA’s GLP regulations (21 CFR §58.1), the determination of test article etiology and contacting the sponsor of the suspected origin of the convulsions rest solely on the study director (SD)—the single point of control on a study. If the SD believes that the event is serious, the event must be considered serious and evaluated by the sponsor for expedited reporting (21 CFR 312.32(a) and 312.32(c)(1)). A report to the sponsor of any convulsions on a study can have significant time and financial ramifications since the presentation of some adverse events identified as most likely “test article-induced” during nonclinical safety assessments is required by law to be transmitted to the FDA (21 CFR §312.32[a]). These requirements are not for just clinical trial incidents (humans); examples given by the FDA specifically list similar events in “animal studies or in vitro studies” (Guidance for Industry and Investigators: Safety Reporting Requirements for INDs and BA/BE Studies; p. 7, Section IV; https://www.fda.gov/downloads/Drugs/Guidances/UCM227351.pdf).
The reporting threshold does not include all adverse events observed during use of a test article, only those adverse events for which “there is some basis to believe there is a causal relationship between the test article and the occurrence of the adverse event” (see 21 CFR 201.57(c)(7) and 201.80(g)). Therefore, when convulsions are reported by the technical staff, it is imperative for the SD to conduct due diligence in the review of all available data, directly observe the incidence, and document the investigation and determinative factors as they relate to seizure liability in preparation for a discussion with the sponsor. The purpose of this review is to delineate characteristic features of PNES in animals that may establish a valid, reliable, and legally defensible position on the reasonable possibility causality standard that does not require such observations to be reported to the FDA.
Differentiating Between Psychogenic and Test Article–Related Convulsions
When a spontaneous movement disorder presents itself during preclinical toxicology studies that is inconsistent over time (the features of the motoric response are different when the animal is observed at subsequent clinical examinations) or is inconsistent with a “classic” convulsive episode or is not consistent with the basic principle of dose dependency (occurs in vehicle controls and high-dose treated animals), it becomes suspicious that the convulsions are psychogenic in origin. It is the responsibility of the SD to engage in a conversation with the veterinary staff and/or Institutional Animal Care and Use Committee (IACUC) to develop a scientifically sound analysis to conclude that clinically a psychogenic movement disorder is involved.
Demonstration of a clear test article–induced dose-dependent response in studies reporting significant perturbations in movement disorders is a challenge even with a known proconvulsant-positive control article, such as the cardiostimulant and pentylenetetrazole (PTZ; cardiazol, Leptazol). Pentylenetetrazole can induce 4 major behavioral phenomena of explosive motor repetitive stereotypies: (1) freezing, (2) myoclonic twitches, (3) clonic convulsions, and (4) tonic–clonic convulsions. 20 While “twitches” and tonic–clonic convulsions are expressed throughout development, there is limited occurrence of freezing and clonic convulsions during the first 2 postnatal weeks in the rat. Stages of convulsion severity are not universal; animals may express freezing right before a full tonic–clonic convulsion with no signs of intervening myoclonic twitches or jumping, and so on. Dose dependency of qualitative and quantitative observable changes in behavior is temporally linked to the dose administration in the acute dosing study, but in cases of psychogenic convulsions, the key observable events may have no ordered temporal relationship to dose administration.
Psychogenic movement disorders are a challenge for the SD. A mistake in its recognition may have important consequences for the sponsor and the subsequent IND and new drug application (NDA) regulatory reviewers. As a result, the diagnosis must be considered very carefully in preclinical toxicology studies conducted in rats, dogs, nonhuman primates, and so on. However, psychogenic movement disorders are not unusual and may be more similar to human absence seizures. Psychogenic seems to be an unspecific term, but unlike clinical neurology veterinary medicine is not amenable to diagnosing a comparable human mental disorder, such as somatoform disorders, factitious disorders, malingering, depression, anxiety, and histrionic personality disorder, that are typically applied to the human patient. Reich 21 has reported that approximately 3% of clinical patients seen in specialty clinics have a psychogenic movement disorder. Benbadis and Hauser 22 estimate the human incidence rate of psychogenic seizures at 2 to 33 per 100,000, making it a “clinically significant neurologic condition.” To the best of our knowledge, the incident rates in veterinary medicine are not known at the present time. Within our own animal research facility, a “best estimate” of incidence rate is less than 0.05% of our total year-to-year rodent population.
The diagnosis of a conditioned or learned anxiety or autonomic compensatory response in animals depends on not just ruling out an organic movement disorder but moreover recognizing features from the detailed observations provided by trained technicians who first witnessed the incident, along with the history and veterinary examinations that are inconsistent or incongruous with an organic movement disorder. The absence of a veterinary psychiatric diagnosis does not preclude a psychogenic cause. 23 The PNES in animals is not only a diagnosis of exclusion, but it can be diagnosed positively in conjunction with the proper documentation of observed events by trained technicians within current standards of GLP compliant observations (detailed clinical observations, functional observational battery, etc). The diagnosis and medical treatment plan requires close cooperation between the veterinary/clinical medicine team and the SD. The problem must never be minimized, and early identification, diagnosis, and treatment plans must be attempted.
Fahn and Williams
24
have categorized human patients into 4 levels of certainty as to the likelihood of their having psychogenic dystonia. In the intervening years, these clinical categories have been generalized to all psychogenic movement disorders, including convulsions.
1,25
–28
Under the Fahn and Williams’s
24
classification scheme, there are 4 degrees of certainty of psychogenic etiologies. As an initial starting point, we propose that these 4 levels of confidence be applied to preclinical toxicology study convulsions, as well: documented psychogenic disorder, clinically established psychogenic disorder, probable psychogenic disorder, and possible psychogenic movement disorder.
Obvious diagnostic identifiers for a preexisting (genetic) or conditioned motor disorder in rodents are the presence of convulsions during the initial acclimation period to the testing environment that is required by federal guidelines. Prior to any randomization and subsequent study assignment or prior to any IACUC-approved protocol-driven function, some measures of health status, such as body weight measurements, clinical observations, or physical examinations, may take place that are used for standard randomization procedures for placement of the rodents into study groups. A spontaneous expression of convulsions at this point may exclude the animals from participation in the study.
An example of an incidence table for convulsions expressed on one of our previous studies is shown in Table 1. The incidence table summarizes the number of observed convulsions on a 104-week dietary study conducted in mice. The data tables may be helpful in the discussion as to the process of data review that the SD and veterinary staff conduct to identify characteristics of a conditioned emotional response (psychogenic). In this case, it was best characterized as a “clinically established psychogenic disorder” by the SD.
Incidence Table for Convulsion Calls on Study.
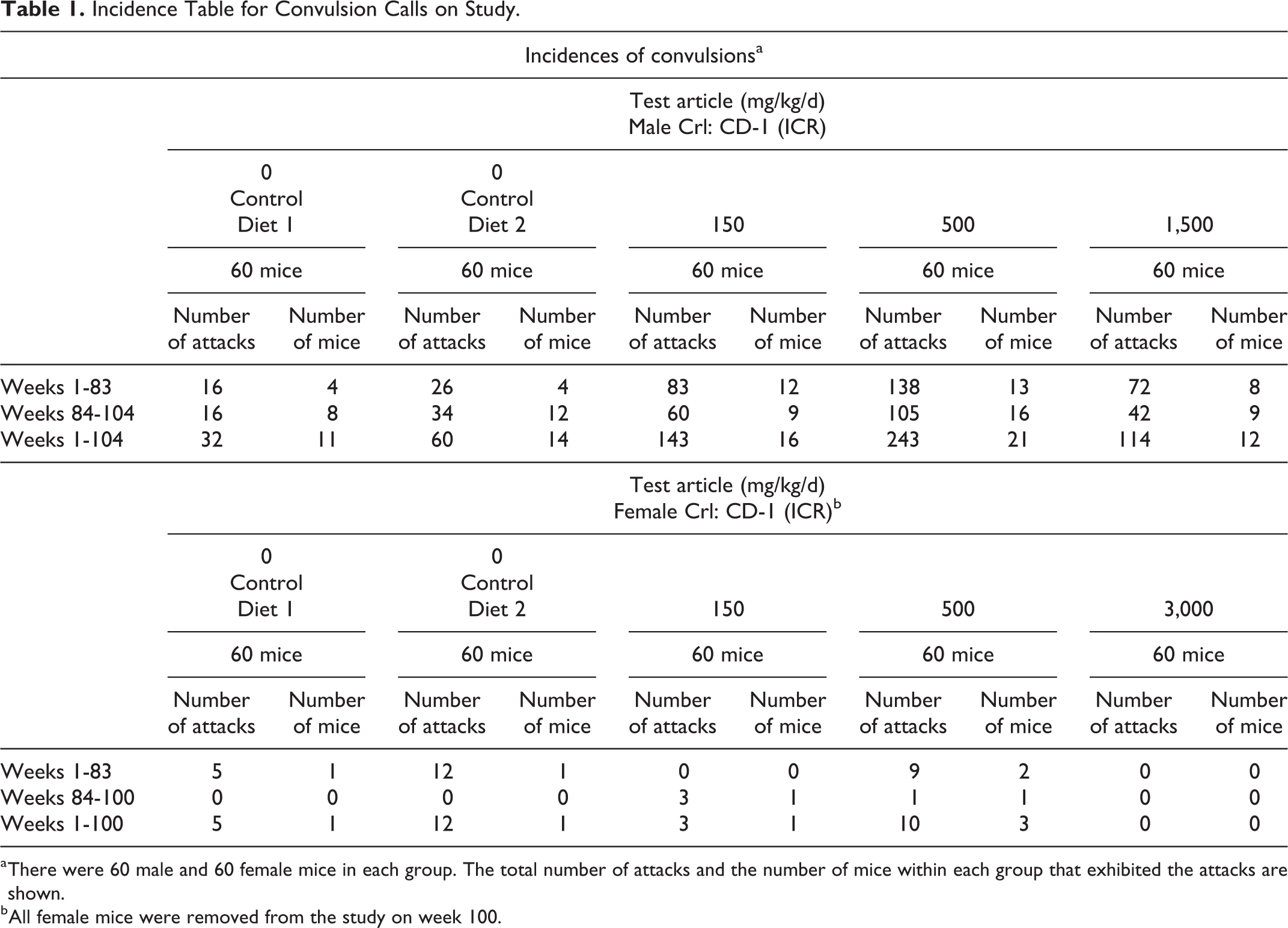
a There were 60 male and 60 female mice in each group. The total number of attacks and the number of mice within each group that exhibited the attacks are shown.
b All female mice were removed from the study on week 100.
Table 1 shows the incidence of observed convulsions over the 100- (female) and 104- (male) week study. Three test article treatment groups were compared to 2 placebo and control diet groups composed of 60 male and 60 female mice per group. The clinical observations over the course of the study highlighting both scheduled and unscheduled observations for calls of “convulsions” and “tremors” for each group are presented in Tables 1 and 2. Table 1 shows the total number of convulsions documented during week 1 through week 83, week 84 to the end of study, and the overall totals for week 1 through the end of the study. Female mice were euthanized in week 100 and male mice euthanized in week 104. Under the GLPs, quality assurance audits must be conducted during each “phase of the study” with the intent to comply with the guidance in conducting multiple or repeated inspections of critical study events throughout the full study cycle §58.35(b)(3). Long-term studies such as this carcinogenicity study are typically divided into at least 2 distinct phases based on ad hoc survival trend tests for mice as described by the FDA’s Draft Guidance document, Guidance for Industry Statistical Aspects of the Design, Analysis, and Interpretation of Chronic Rodent Carcinogenicity Studies of Pharmaceuticals (https://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM079272.pdf).
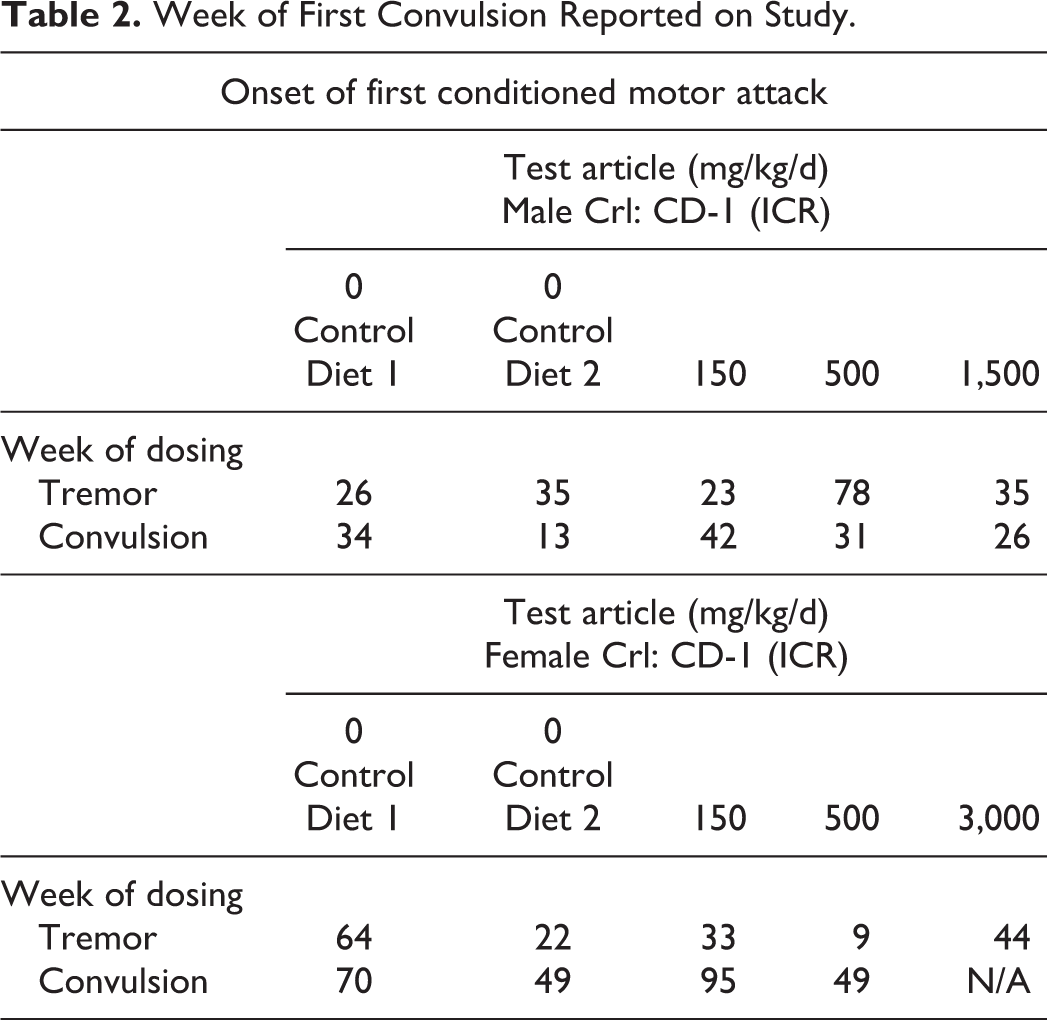
Table 2 simply summarizes the weeks of study in which the first documented report of witnessed convulsion and tremor for each group was documented in the 104-week dietary study. It has been shown that when electrical or chemical stimulation is repeatedly applied to rodents, explosive motor attacks may gradually develop in response to the stimulation, as in this study. Once seizures have been developed, they appear reliably thereafter whenever stimulation is applied. Using the typical rank ordered scoring system for clinical features of seizure propagation in rodents, “tremor” is positioned in lower ranked scores (stage 2 or 3) when compared to full tonic–clonic seizure (stage 4 or 5). In this study, observations of tremor were first reported weeks following the first documented incidence of convulsions in 3 of the 5 groups of male mice (control 2: 22 weeks following convulsions; 500 mg/kg test article group: 47 weeks following convulsions; and the high-dose 1,500 mg/kg test article group: 9 weeks following the first convulsion).
Week of First Convulsion Reported on Study.
Differentiating if Convulsions Are Psychogenic (Idiopathic) or Organic (Test Article Induced)
We provide 10 clinical features of laboratory rodent behaviors that may assist in determining the clinical significance of a reported “convulsion” during the conduct of a standard rodent repeat-dose toxicology study. Good preclinical laboratory practice requires immediate reporting of each ictal event to the staff veterinarian and SD. The identification of key discriminative features of each event, and all previous events on the study, may help to determine the best and most accurate diagnosis of PNES or test article–induced neurotoxicity.
Factor #1: Variability in the Frequency of Convulsions (Total Counts)
Table 1 shows a clear difference between and within groups of male and female mice with respect to the presentation of convulsions. Twenty-five out of 120 male mice (group 1 [11/60]) and group 2 [14/60]) compared to 2 out of 120 female mice (group 1 [1/60] and group 2 [1/60]) in the placebo control groups expressed convulsions over the full study schedule (21% males vs 1.6% females). Both control groups were given access to standard mouse diet and control diets, only. To avoid unintentional exposure to test article during these long-term rodent studies, standard operating procedures in the laboratory require control groups to be collectively (paired or grouped housed) and physically housed separately from test article–treated cohorts (cage racks located in separate room or strategic spatial location within the same vivarium maintained in a negative air pressure environment). The group of rats receiving exposure to the highest dose of test article (females, 3,000 mg/kg day) did not exhibit any convulsions on this study, with only 1 female control diet female showed 5 spontaneous convulsions during the first 83 weeks of the study and absolutely no convulsions during the subsequent 16 final weeks of the study (weeks 84-100). These inconsistencies are highly suggestive of a diagnosis of PNES in this study.
Factor #2: Dose Dependency
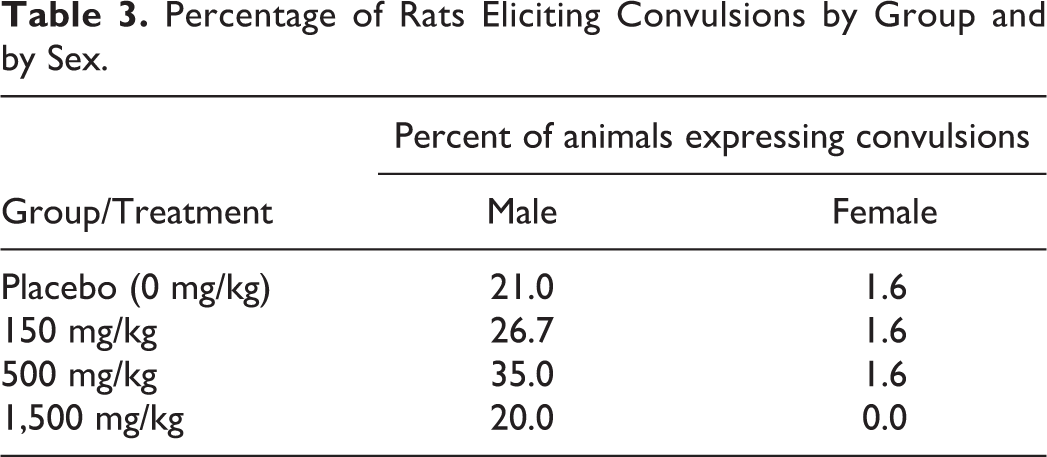
Figure 1 reveals that 25 out of 120 male mice receiving control diet (21% of groups) expressed convulsions over the 104 weeks of the study, while 12 out of 60 high-dose test article–treated male mice (1,500 mg/kg) also expressed seizures (20% of the group) over the same interval (nonsignificant). No group differences were documented in female mice cohorts. Table 3 displays the convulsion incidence rates in male mice. The total number of male mice expressing a convulsion engendered a blunted inverted U-shaped dose–effect function.
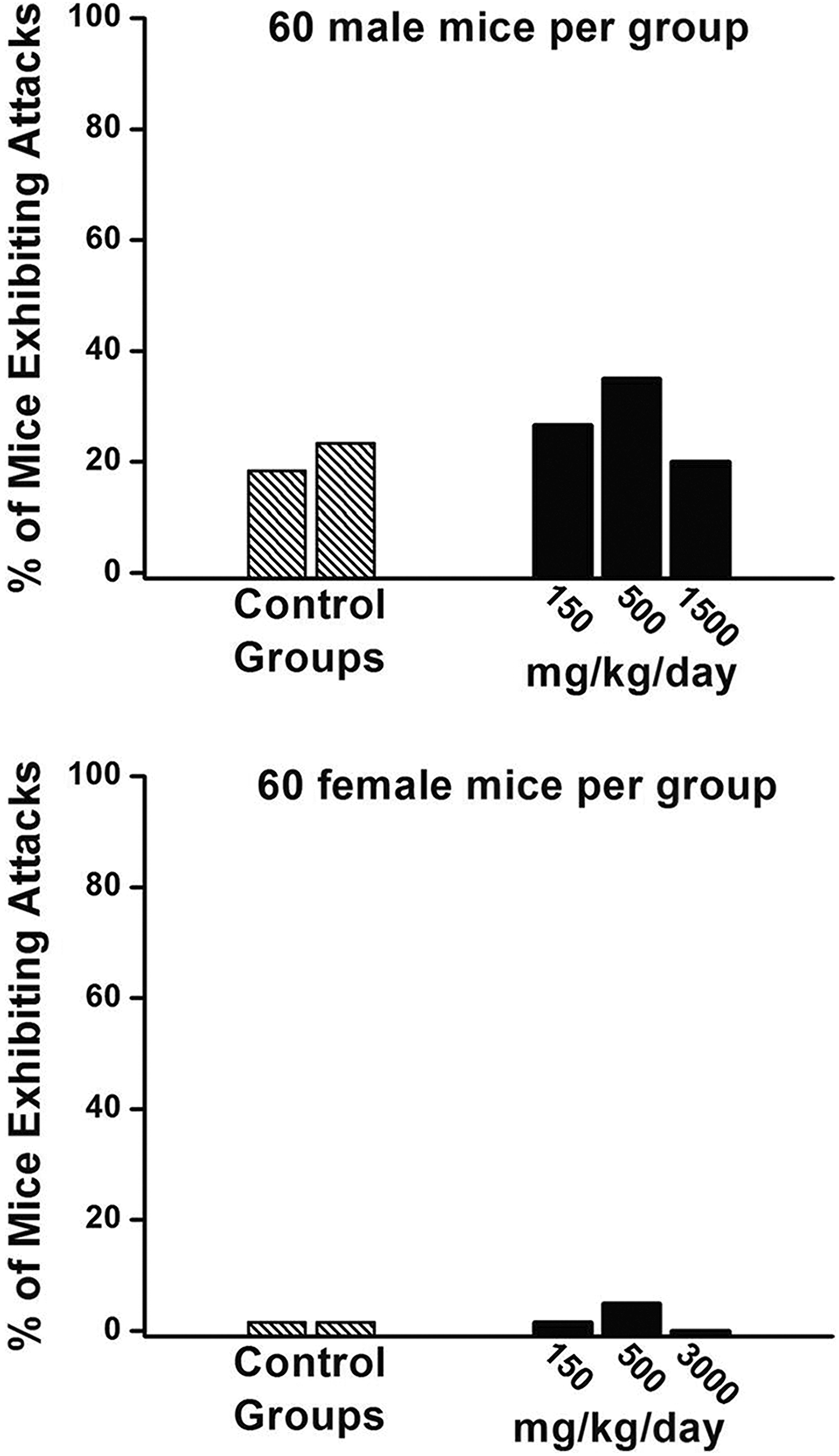
The percentage of male (top panel) and female (bottom panel) mice expressing convulsions on a 104-week dietary toxicology study. There were no statistically significant group differences in the total number of convulsions expressed (control vs test article-treated groups). There was a clear sex-related difference in the clinical presentation of convulsions.
Percentage of Rats Eliciting Convulsions by Group and by Sex.
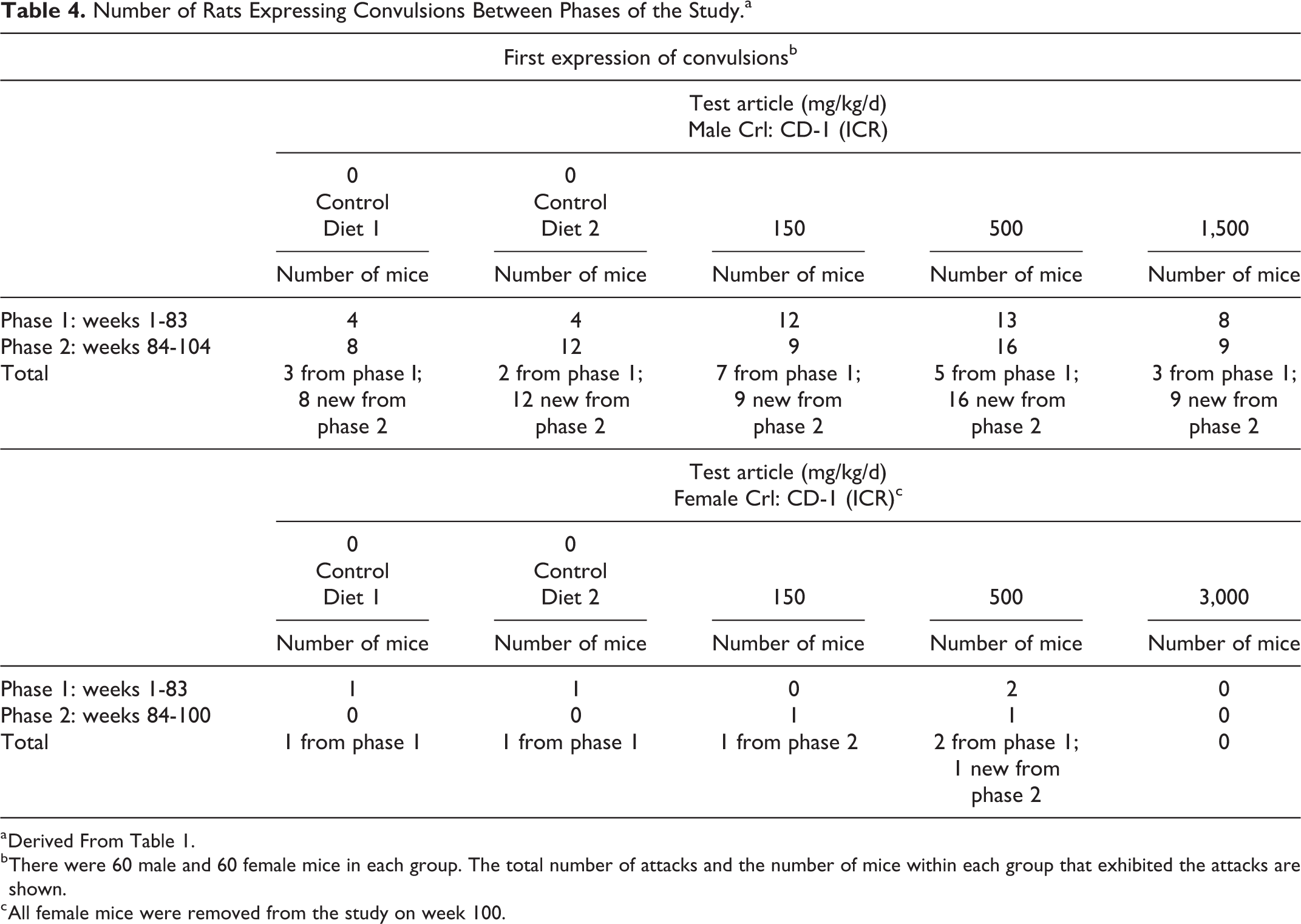
The data were summarized into 2 phases of the study duration: phase 1: weeks 1 to 83, and phase 2: weeks 84 to 104. The incidences of convulsions detailed in Table 1 show the total number of mice for each phase of the study as well as the total duration of the study protocol (week 1 to end of the study). Table 4 breaks down the cumulative counts of seizures to the number of mice from each phase that presented the convulsions:
Number of Rats Expressing Convulsions Between Phases of the Study.a
a Derived From Table 1.
b There were 60 male and 60 female mice in each group. The total number of attacks and the number of mice within each group that exhibited the attacks are shown.
c All female mice were removed from the study on week 100.
Table 4 is derived from data in Table 1. It highlights a number of incidences in which mice engendered convulsions only in phase 1 of the study. It is now known that tolerance does not develop equally to all actions of a drug and reverse tolerance (increased responsiveness) can occur. 29 Systemically administered chemicals have been shown to “kindle” convulsions (eg, strychnine). Once seizures have been developed, they most often appear reliably thereafter whenever stimulation is applied. 30 –33 As previously described by Adler and Geller, 29 repeated dose administrations of a drug are usually required to produce tolerance and dependence. A reduced response to a drug over repeated-dose administration is defined as tolerance and the process may be pharmacokinetic or neuronal. It is now known that tolerance does not develop equally to all actions of a drug and reverse tolerance (sensitization or increased responsiveness) can occur. Tolerance to convulsions induced by a single drug is atypical and most often sensitization to the hyperexcitability (reverse tolerance) occurs to repeated-dose administrations of proconvulsants such as PTZ, bicuculline, and picrotoxin. The behavioral expression of convulsions is generally not a single event during repeated exposures to a proconvulsant—it would be predicted that once a convulsion is expressed, the quantitative and qualitative nature of the convulsion would expand over subsequent repeated-dose administrations. Table 5 shows the number of male mice that expressed a convulsion in only phase 1 of the study. Each mouse may have expressed more than a single motor attack, but the mice did not show progression of convulsion expression over the full study period.
Number of Mice Expressing Convulsions in Only Phase I.a

Abbreviations: grp, group; TA, test article.
a No progression of attack severity over time.
These comparative data are incongruent with classic movement disorders associated with repeated-dose administrations of proconvulsants and are supportive of a reasonable possibility that meets the regulatory “causality standard” for the psychogenic etiology of these motoric attacks.
Factor #3: Temporal Onset of the Motoric Response
As shown in Table 2, the onset of convulsions in this study did not covary with the intensity or duration of the systemic chemical exposure to the test article. The first convulsion in the male mice receiving the high-dose test article (1,500 mg/kg) was documented in week 26 of the study, while nontreated placebo control male mice expressed the first convulsion 13 weeks earlier (control group 2) and 8 weeks later (control group 1). Additionally, the documentation of the first observed convulsion in the test article treatment groups was inversely proportional to the delivered dose: low dose (150 mg/kg): week 42; mid dose (500 mg/kg): week 31; and high dose (1,500 mg/kg): week 26. This inverse relationship with total body load of the test article is not consistent or congruent with a chemically induced (kindled) convulsive diagnosis and lends further support that the convulsions are psychogenic (conditioned) in origin.
Factor #4: Progressive or Nonprogressive Nature of the Observable Behavioral Expression
As stated above, in the discussion of Table 2, using the typical rank-ordered scoring system for clinical features of seizure propagation 31,33 in rodents, “tremor” is positioned in lower ranked scores (stage 2 or 3) when compared to full tonic/clonic seizure (stage 4 or 5). In this study, observations of tremor were first reported weeks following the first documented incidence of convulsions in 3 out of the 5 groups of male mice (control 2: 22 weeks following convulsions; 500 mg/kg test article group: 47 weeks following convulsions; and the high-dose 1,500 mg/kg test article group: 9 weeks following the first convulsion). These findings are, again, inconsistent with the “classic” or “normal” progression of chemical-induced sensitization processes of the central nervous system (CNS).
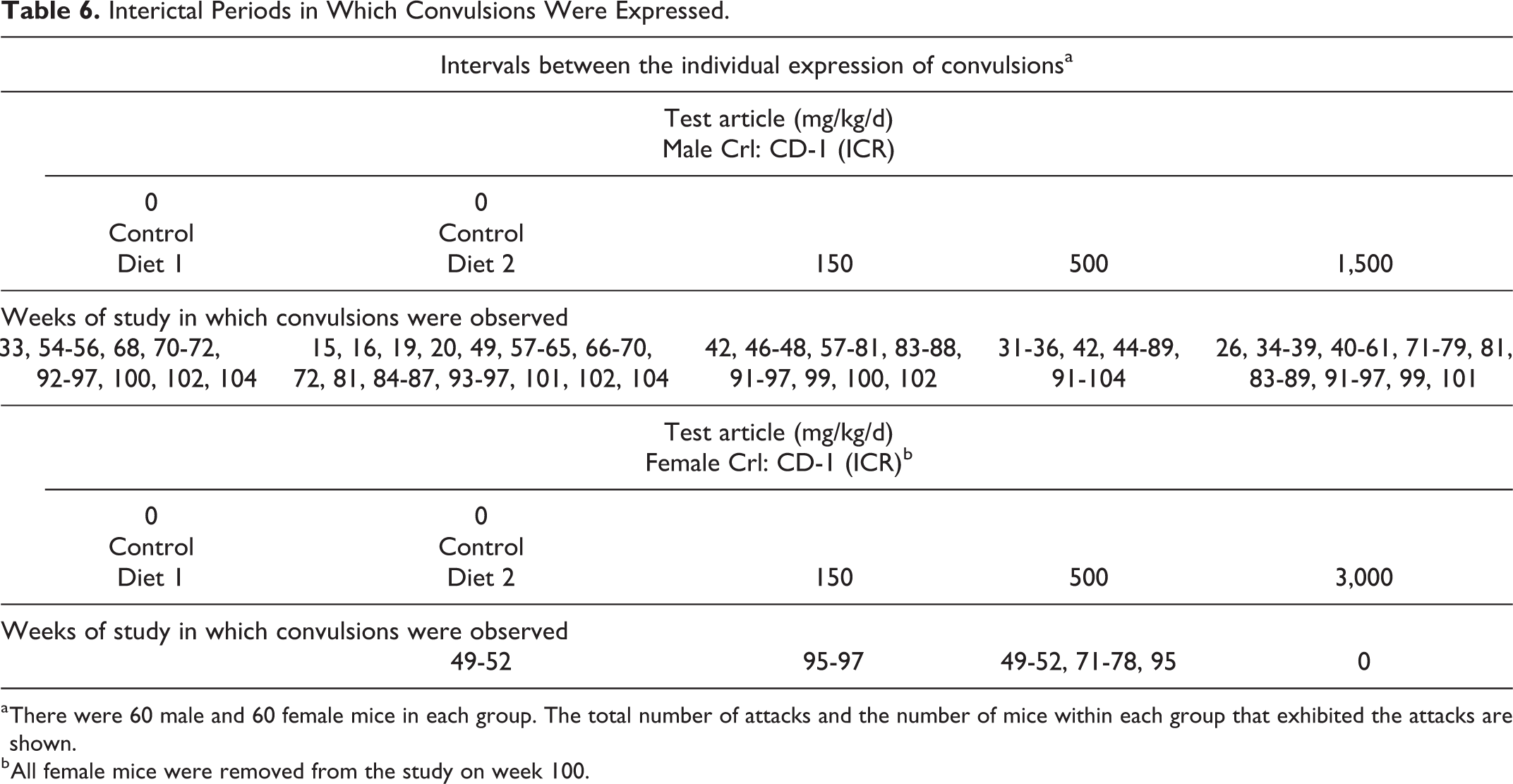
Factor #5: Incidence or Intervals Between Observed Convulsions
In general, when repeat-dose administrations of a test article subsequently induce a convulsion, reexposure to the test article generally produces a similar motor attack or a more qualitatively severe behavioral expression of the convulsion (sensitization 30 –33 ). Table 6 shows the weeks of study that a convulsion was reported for each treatment group. In contrast, Table 7 shows the week of the study that was “convulsion-free.” For example, in male mice, the control group 1 (placebo) had a 23-week convulsion-free period between recorded convulsions; control group 2 (placebo) had a 9-week event-free interval; the low-dose test article group (150 mg/kg) had a similar 9-week event-free interval; the mid-dose test article group (500 mg/kg) was free of convulsions for 6 weeks; and the high-dose treatment group (1,500 mg/kg) had a 9-week convulsion-free period. When documented clinical observations for individual rats were reviewed, even more dramatic incidents of instability were noted, for example.
Interictal Periods in Which Convulsions Were Expressed.
a There were 60 male and 60 female mice in each group. The total number of attacks and the number of mice within each group that exhibited the attacks are shown.
b All female mice were removed from the study on week 100.
Longest Duration of Ictal-to-Ictal Attacks: Pauses in Expression of Convulsions.
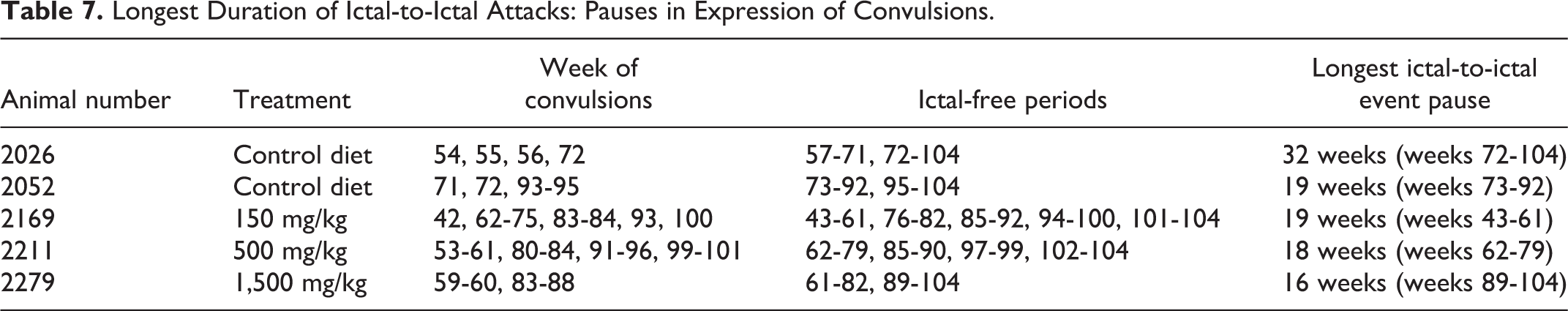
In previous seizure-induction studies conducted in mice and rats, once a full clonic convulsion is induced by systemic administration of a suspected proconvulsant, further exposure to the proconvulsant elicits a more intense convulsion up to point of lethality. Table 7 shows that prolonged convulsion-free periods of 16 to 32 weeks following repeated expressions of clonic convulsions in this study example are not consistent with the regularity of convulsive events reported in these previous chemical kindling studies published in the peer-reviewed scientific journals. The pauses in convulsions once repeated convulsions are noted in the study provide supportive data for a diagnosis of psychogenic etiology of the convulsions in this study.
Factor #6: Presence of Postictal Depression
The term “ictal” is derived from the root term for “storm”; a seizure represents an electrochemical storm within the CNS. Seizures are defined by the recording of synchronized high-amplitude, low-frequency electrical activity in EEGs. These changes reflect similar large fluctuations of intracellular ion concentrations (sodium, potassium, chloride) which persist following the cessation of the observed convulsion. It takes time for the CNS to return to normal homeostasis following true seizures; this is often referred to as postictal depression. The postseizure recovery duration varies, but behaviorally this period is often characterized by lethargy, motor depression, or slowness of the animal. In this study, there were no reports of any postconvulsion lethargy, inactivity, or somnolence. Mice appeared to return to normal gait and posture and usually started grooming or interacting with cage mates following the convulsion. The lack of any signs of postictal recovery strongly supports the exclusion of any significant insult to the brain neurochemistry as the result of the behavioral expression of rhythmic motor alternans.
Factor #7: Absence of “Typical” Clinical Signs of Seizure Onset for the Species
Rodents, in general, show a progression in the qualitative and quantitative expression of CNS excitability with repeated exposures to proconvulsants from “normal” to full tonic/clonic convulsions. Freezing behavior, “popcorn”-like jumping, running fits, tremors, and kangaroo postures most often appear in steps or stages. With stage 1 being “normal,” most rodents suspected of PNES generally do not present signs typically characteristic of mid stages of convulsion development. The first sign of behavioral pathology is the frank presentation of clonic seizures with loss of postural control. Often digit, tongue, or body tremors are not presented during an active convulsion. With little or no warning, the convulsions are almost quantal in character, for example “normal” or “clonic convulsion.” In this 104-week dietary exposure study, these convulsions are generally elicited by technicians entering the room to conduct daily observations, body weight measurements, or cage cleaning. In other oral or parenteral test article administration studies, these convulsions seem to occur even prior to dosing (see below). These types of findings are atypical of true CNS proconvulsant or chemical kindling study findings and may support a diagnosis of “psychogenic” etiology.
Factor #8: Visual Observation of the Activation of Antagonistic Muscle During the Convulsion
Table 2 shows a number of groups of mice that exhibited the onset of “tremors,” a lower stage of convulsion expression, weeks to months after the first full clonic convulsion. Freezing behaviors, tonic extension of the limbs, jumping, running, and body tremors are not witnessed prior to or during the clonic convulsion that initiates the first call to the SD and veterinary staff. Subsequent direct visual or remote video observations of the rodents prior to and following technicians entering the room generally find normal rodent behaviors and social interactions expected in group-housed animals. When a convulsion is expressed, the psychogenic convulsion lacks any opposing or rhythmic alternations during its expression—no tonic–clonic alterations, merely intense muscle clonus, sometimes with ptosis, and rarely with tongue biting injuries.
Factor #9: Distraction Interrupts Convulsion
One of the best diagnostic procedures in our laboratory that we have used to help differentiate psychogenic from organic (test article-induced) convulsions is the use of “distraction” (see Figure 2). Although the rodent is observed having a clonic motor attack, the trained technician, SD, or veterinarian can simply use the clicker to present a novel but alerting auditory stimulus while being held closely over the rodent (see Figure 2). In many cases, the rodent orients to the sound and escapes in the opposite direction. The true organic seizure does not respond to verbal commands or other exteroceptive stimuli. We have also used a “tail pinch” or “forcep pinch” to the side of the animal to test for the escape response. The abatement of a full clonic convulsion by a distractor is strong evidence for the diagnosis of psychogenic origin.
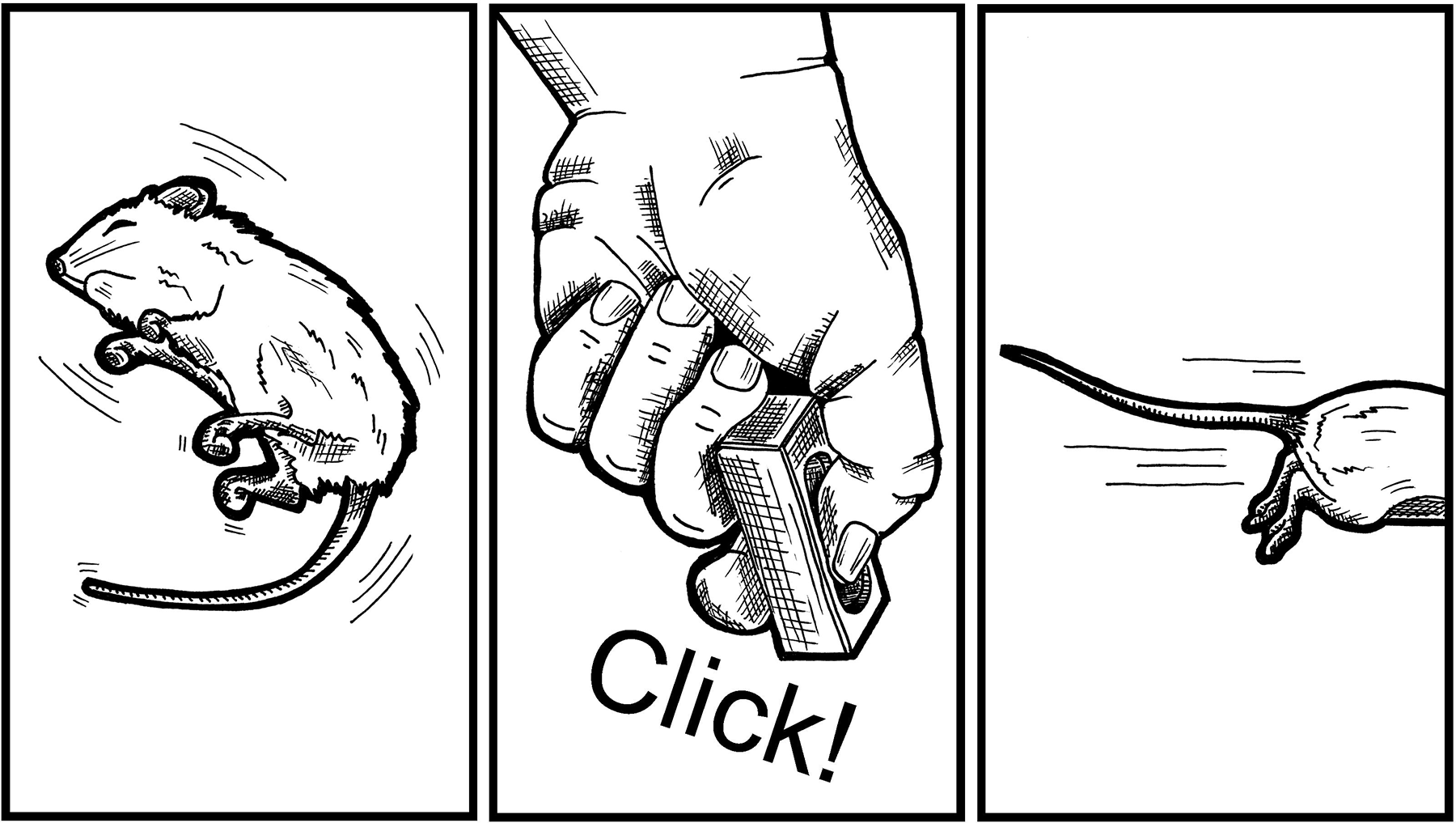
Presentation of a handheld “clicker” standard to most International Conference on Harmonization (ICH) S7A functional observational battery study designs is used to distract the rodent during a witnessed convulsion that is suspected to be psychogenic in origin.
Factor #10: Presentation of Other Psychogenic/Social Dynamics in Group/Paired Housing
Sometimes when a rodent expresses a clonic convulsion during body weight measurements, or returning the rodent following the completion of detailed clinical observations, some of the cage mates will suddenly express similar clonic convulsions. This is most likely attributable to conditioned danger cues, or strain-specific alarm substances that alert the group of impending danger. These social contagion-like effects are diagnostic of psychogenic etiology.
Discussion
There is no reliable information on the incidence or prevalence of PNES in preclinical toxicology studies within or between testing facilities. Psychogenic nonepileptic seizures have been operationally defined as episodes of altered movement, sensation, or experience similar to epilepsy, but caused by a psychological process and not associated with abnormal electrical discharges in the brain. 34,35 Given the definition of PNES, it is not surprising that a medical diagnosis critically depends on the post hoc review of observed clinical observations that may differentiate them from true epileptic seizures. Restraint or the use of seizure induction procedures (photic or electric stimulation) and pin-lead EEG confirmation of an event only leads to further stressing an already “stressed-out” laboratory animal. The overwhelming majority of PNES are considered involuntary conditioned (learned) expressions of impending psychological stress. 35,36 Diagnosing PNES is important because of the FDA administrative policies regarding notification of the agency of serious adverse events that occur during the conduct of preclinical studies for NDA submissions. The failure to recognize the psychological nature of these seizures may initiate professional conflict between management and technical staff or instigate unwarranted animal welfare concerns for IACUC intervention. These events are unintentionally learned or classically conditioned within laboratories that maintain the highest standards of animal care and can inject a sense of frustration when discussing a potential treatment plan for both animals 37 and humans. 38 Diagnosing PNES is important because of the potential iatrogenic hazards related to the potentially serious side effects of antiepileptic or antianxiety drugs when administered alone or in combination with the test article on the study.
Kindling is a remarkably general phenomenon. Administration of a subthreshold stimulation to the CNS will initially have no overt behavioral response. With continued, even periodic, pairings of stimulations, the animal will develop increasingly more generalized clonic motor attacks, commonly referred to as “fully generalized” or stage 5 convulsions. Kindling has been reported in frogs, mice, gerbils, rats, rabbits, cats, dogs, rhesus monkeys, and baboons. 39 While the first stimulus used in the published literature was focal electrical stimulation, subsequent studies have shown periodic exposure to convulsive agents like electroconvulsive shock, intracranially or systemically administered convulsant drugs, or convulsant vapors will kindle convulsions. 39
Drugs are stimuli. As such, a drug stimulus can enter into functional relationships with physiological and behavioral processes, such as Pavlovian conditioning. 40 Adamec 40 first proposed kindling effects on the emotional behavior of animals. Kindling of after discharges (not motor seizures) following electrical stimulation of feline amygdala and ventral hippocampus increased the emotional and behavioral reaction to a rat or mouse and to a threat vocalization by a cat cohort. 41,42 Pinel et al 43 reported kindling in rats engendered greater reactions to tail tap and a greater resistance to capture by technicians when compared to benign controls. In a simple Pavlovian conditioning model, an unconditioned stimulus (such as the presentation of meat powder to the tongue of a dog [or a proconvulsant test article systemically]) is preceded by the presentation of a neutral stimulus selected by the experimenter, for example, a bell (CS+, or in the present case placement of the animal on a body weight scale). The meat powder will elicit salivation in the dog reflexively; without training, this is the unconditioned response. Repeated pairings of the bell and the meat powder will come to elicit salivation even when meat powder administration is withheld. The bell is now a conditioned (learned) stimulus (CS+) that, by its own presentation alone, will elicit the salivation (conditioned response). In the psychogenic convulsion scenario, like the bell in the Pavlovian conditioning model, during the initial days of the study, some environmental cue(s) that predicts handling, restraint, dosing, or the direct effects of the test article may come to elicit predictable strain- and/or species-specific stress responses. Like environmental cues, several examples of temporal or “time cues” are present. Drugs are usually administered at about the same time each day, and clinical observations, body weight measurements, feed jar replacements, and so on, are generally synchronized to the circadian clock as well (lights on/lights off). After repeated pairings of neutral cues and stress or proconvulsant cues, the neutral stimuli will eventually elicit the physiological responses associated with the stressor or test article.
Classic examples of kindling of psychogenic effects in the published preclinical literature are (1) handling-induced convulsions 44,45 and (2) the conditioning of fear/anxiety behaviors. 46 As detailed in Table 1 of this report showing a clear difference between and within groups of male and female mice with respect to the presentation of convulsions, similar sex-dependent differences have been reported in (1) in vitro slice preparation kindling, 47 (2) amygdala stimulation kindling in whole animals, 48 –50 and (3) handling-induced convulsions in rats. 51
The environmental stimuli that can effectively serve as functional conditioning stimuli (CS+) in animals are near limitless. There is no way of knowing what stimuli captured stimulus control over convulsions once kindling is demonstrated. It could be the lead technician in the study who enters the room for the scheduled early morning or the close environmental cues of the weigh scale basket or the removal and replacement of the daily food weighing and/or replacement. The best chance of intervening with the further conditioning of cues to convulsions is at the identification of the first convulsion on the study, for example, changing technicians, changing the order of dosing, body weight measurements, feed jar replacements, and so on. Attempts could be made to change the location of the rat cages relative to the vivarium doors and lights if they are suspected of serving as controlling stimuli.
Conclusions
The presentation of convulsions on any study is troublesome for the SD, veterinary staff, and the sponsor. The notification of the FDA within 15 days of the determination that there has been an unexpected, test article-related serious (to lethal) adverse reaction in animal preclinical toxicology studies has major legal and financial burdens that may, in fact, “kill a compound.”
We have provided 10 factors to consider in determining evidence to support that spontaneous seizures are of psychogenic etiology. By closely examining the data collected on the study and detailed conversations with the technical and veterinary staff, the SD can provide the sponsor with a logical, legally defensible, and reliable basis for study continuation that does not unduly compromise the FDA confidence in the validity of the study data. By conducting due diligence at the first report of motor attacks, the SD may improve the work: product relationship between the animal testing laboratory and the sponsor and allow for a timely completion of the study. If a definitive diagnosis of psychogenic origin cannot be supported by the existing data set, a tier II neurotoxicity EEG study is highly recommended.
Footnotes
Authors’ Note
The focus study of this review article was conducted in full compliance with US FDA’s Good Laboratory Practice Guidelines (21 USC Chapter 13, §58) and the Guide for the Care Use of Laboratory Animal Research and had prior approval of the IACUC. Compliance with the guidelines and principles listed therein and should indicate that animal protocols were reviewed and approved by an institutional panel.
Author Contributions
D. V. Gauvin contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. Z. Zimmermann, J. Yoder, and T. Baird contributed to conception and design, contributed to interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
