Abstract
Drug-induced phototoxicity occurs when drugs absorb natural sunlight, leading to chemical reactions causing cellular damage. Distribution to light-exposed tissues is critical and is enhanced by binding to melanin. The International Council on Harmonization S10 guidance document on photosafety evaluation of pharmaceuticals states that although nonpigmented skin tends to be more sensitive than pigmented skin, pigmented skin models should be considered for drugs that bind significantly to melanin. In this study, an in vitro melanin-binding assay was evaluated as prescreening tool for animal model selection. Binding of various structurally diverse phototoxic drugs to synthetic melanin was investigated in vitro and the high-affinity binder sparfloxacin (SPX), moderate-affinity binder 8-methoxypsoralen (8-MOP), and low-affinity binder pirfenidone (PIF) were selected for in vivo studies. Pigmented Brown Norway (BN) rats were compared with nonpigmented Wistar Albino rats to evaluate their sensitivity for the assessment of phototoxicity and skin concentrations of the drugs were measured. For SPX, the onset of phototoxic symptoms was faster for BN rats and drug concentrations were significantly higher in skin of BN rats. For 8-MOP, both models showed comparable sensitivity and skin concentrations did not differ. For the low-affinity binder PIF, no phototoxic effects were observed and skin concentrations in both models were similar. A combined in vitro/in vivo approach was developed that can be applied for accurate photosafety evaluation of pharmaceuticals based on the assessment of possible melanin-binding effects. In view of the presented data, the pigmented model could be considered for compounds showing a high-affinity binding capacity in vitro.
Introduction
Drug-induced phototoxicity is an undesirable reaction caused by the effect of an interaction of a photoactivated drug with biomolecules present in the skin and eyes. Drug-induced phototoxicity is initiated after a topically or systemically administered drug is exposed to natural sunlight and is excited by absorption of photon energy. 1 The absorbed energy may subsequently lead to a chemical change of the drug itself that can result in the formation of photoproducts and/or photoadducts after covalent binding to DNA. It may also lead to generation of reactive oxygen species (ROS) that cause toxicity to target tissues, such as lipids, proteins, or DNA. 2 Repeated drug-induced phototoxicity may result in fragility, blistering, milia formation, or even formation of actinic keratoses and skin cancers. 3 It has been reported that several classes of drugs cause phototoxic reactions in the skin and eyes at clinical doses while drug-induced phototoxicity has also hampered the development of new drugs. 4,5 It is therefore essential to evaluate the phototoxic potential of drugs for accurate risk assessment.
To evaluate the phototoxic potential of compounds, various methodologies, such as the 3T3 neutral red uptake phototoxicity test, have been developed, and European and American regulatory agencies have published guidelines for predicting and avoiding drug-induced phototoxicity in an early phase of drug discovery. 5 For evaluation of the photosafety of pharmaceuticals, the International Council on Harmonization (ICH) of Technical Requirements for Registration of Pharmaceuticals for Human Use has issued a guidance document to recommend international standards. As stated in this S10 guidance document, 1 requirement for the assessment of the photoreactive potential of a drug is to investigate whether light is absorbed by the drug within the range of natural sunlight (290-700 nm). 6 Since ROS are typically generated during phototoxicity, a second characteristic that needs to be assessed is whether the drug generates ROS following absorption of ultraviolet (UV)-visible light. The third critical characteristic for a drug to demonstrate phototoxicity is whether it distributes sufficiently to light-exposed tissues, such as the skin and the eyes. The concentration of a drug depends not only on factors such as plasma concentration, tissue perfusion, partitioning from vascular to interstitial, and/or cellular compartments but also on binding, retention, and accumulation of the drug in various tissues. Although binding, retention, or accumulation of a photoreactive drug in a tissue is not essential for a reaction to occur, longer mean residence times in sun-exposed tissues do increase the likelihood for the photoreactive drug to produce a phototoxic reaction.
One mechanism by which a photoreactive drug can have longer residence times or accumulate in tissues is by binding to the naturally occurring biopolymer melanin. In humans, melanin is present in many tissues, including the eye, skin, and hair. A major biological function of melanin is to attenuate UV light penetration of the eye and skin to protect for UV-induced DNA damage. 7 In addition, it is believed that melanin is a photoprotective agent since it scavenges free radicals and ROS. 8 Various drugs, including antimalarials, antibiotics, β-blockers, central nervous system drugs, and anticholinergics, have been reported to bind to melanin both in vitro and in vivo. Since turnover of melanin in the body is low, a long-term retention in melanin-containing tissues of drugs with high melanin affinity may occur. 9,10 As melanin-rich areas of the body are extensively exposed to light, binding of phototoxic drugs to melanin potentially increases their phototoxicity. For in vivo photosafety studies, models with both the pigmented and nonpigmented animals are available. Although nonpigmented skin tends to be more sensitive than pigmented skin for detecting phototoxicity, pigmented skin models should be considered for chemicals that bind significantly to melanin. 6 Therefore, in order to obtain an accurate risk assessment, it is important to determine the melanin-binding affinity of a photoreactive drug.
In the current study, we have applied the melanin-binding screening assay to study the binding of various structurally diverse phototoxic and/or melanin-binding drugs to synthetic melanin in vitro. From this screening experiment, we have selected the high-affinity binder sparfloxacin (SPX), moderate-affinity binder 8-methoxypsoralen (8-MOP), and low-affinity binder pirfenidone (PIF) for which we assessed the phototoxic effects in different in vivo rat models. It was evaluated if the in vitro melanin-binding assay could be used as a prescreening tool for rat model selection for in vivo photosafety studies. A pigmented rat model, the Brown Norway (BN) rat, was compared with a nonpigmented rat model, the Wistar Albino (WA) rat, in order to assess their sensitivity to detect phototoxic effects of drugs with varying melanin affinities. The goal of this study was to develop a testing strategy which included the assessment of possible melanin-binding effects for accurate photosafety evaluation.
Materials and Methods
Chemicals
Acetonitrile (ACN), ammonium acetate, formic acid, and methanol were all of ultraperformance liquid chromatography/mass spectrometry (UPLC/MS) grade and were obtained from Biosolve B.V. (Valkenswaard, the Netherlands). Acetic acid (glacial), dimethyl sulfoxide (DMSO), dipotassium hydrogen phosphate (K2HPO4), ethanol, formaldehyde (37%-40%), potassium hydrogen phosphate (KH2PO4), and polyethylene glycol 400 (PEG 400) were obtained from Merck KGaA (Darmstadt, Germany). 3,3′,4′,5-Tetrachlorosalicylanilide (TCSA) was obtained from Acros Organics (Morris Plains, New Jersey). Carboxymethylcellulose was obtained from Fagron (Capelle aan de IJssel, the Netherlands). Zoletil 50 was obtained from Virbac Nederland (Barneveld, the Netherlands). Duratears Z was obtained from SA Alcon-Couvreur NV (Puurs, Belgium). Pirfenidone was obtained from Apotheek Sanatheek (Houten, the Netherlands). All other chemicals were obtained from Sigma-Aldrich (Zwijndrecht, the Netherlands) unless stated otherwise.
In Vitro Melanin-Binding Screening Assay
Melanin-binding experiments were performed with 16 compounds: chlorpromazine (CPZ), ciprofloxacin (CPX), diclofenac (DIC), dorzolamide (DOR), doxycycline (DOX), hydrochlorothiazide (HTZ), levofloxacin (LVX), 8-MOP, nalidixic acid (NA), olaquindox (OLA), PIF, piroxicam (PIX), procaine (PRO), SPX, TCSA, and timolol (TIM) at final concentrations of 1 and 100 µM. Melanin was mixed with 100 mM potassium phosphate buffer pH 7.4 to form a 2 mg/mL suspension, which was sonicated for 5 minutes. Test compound incubation solutions were prepared by diluting 10 mM and 100 µM test compound solutions prepared in DMSO 50-fold in 100 mM potassium phosphate buffer pH 7.4 to obtain solutions containing 200 µM and 2 µM (2% DMSO), respectively. Melanin suspensions and test compound solutions were prepared less than 4 hours before starting the incubations. Incubation mixtures were prepared in triplicate by mixing 200 µL of melanin suspension with 200 µL of the test compound incubation solution. Corresponding control samples were prepared in triplicate by mixing 200 µL of the test compound incubation solution with 200 µL of 100 mM potassium phosphate buffer pH 7.4. Incubation mixtures and the corresponding control samples were incubated at room temperature under gentle shaking for 24 hours. After incubation, incubation mixtures and the corresponding control samples were centrifuged for 15 minutes at 16,000
Ultraperformance Liquid Chromatography Analysis
The UPLC–MS system consisted of an Acquity UPLC I-class system (Waters, Milford, Massachusetts) equipped with a Xevo TQ-S MS (Waters). Chromatographic separations were achieved by gradient elution using a Waters Acquity UPLC BEH C18 (2.1 mm × 50 mm, 1.7 µm particle size) column which was operated at 40°C. The column effluent was analyzed in the multiple reaction monitoring mode. The system was controlled by the MassLynx version 4.1 software (Waters). For the in vitro experiments, the concentrations of the test compounds DOR, DOX, HTZ, NA, OLA, PIF, PIX, PRO, SPX, and TCSA were measured using UPLC. For the in vivo studies, concentrations of SPX, 8-MOP, and PIF in skin samples were measured by UPLC. Standard calibration curves of the test compounds were always included during measurements. More details about the analytical conditions that were applied and the calibration range of the analytical methods can be found in supporting information.
High-Performance Liquid Chromatography Analysis
The high-performance liquid chromatography (HPLC)–photodiode array (PDA)-MS system consisted of a surveyor MS HPLC pump (Thermo Fisher Scientific Inc, Waltham, Massachusetts), HTC PAL autosampler with cooled stack (CTC Analytics AG, Zwingen, Switzerland) and a surveyor PDA detector (Thermo Fisher Scientific Inc) equipped with a linear trap quadrupole linear ion-trap LC-MS (Thermo Fisher Scientific Inc). Chromatographic separations were achieved by gradient elution using a Waters symmetry C18 (2.1 mm × 50 mm, 3.5 µm particle size) column which was operated at room temperature. Data were acquired and interpreted with Xcalibur software version 2.0 (Thermo Fisher Scientific Inc). For the in vitro experiments, the concentrations of the test compounds CPX, CPZ, DIC, LVX, 8-MOP, and TIM were measured using HPLC. Standard calibration curves of the test compounds were always included during measurements. More details about the analytical conditions that were applied and the calibration range of the analytical methods can be found in supporting information.
Animals, Housing, and Husbandry
Nonpregnant, nulliparous females (approximately 9 weeks old) of 2 rat strains with varying levels of melanin in the skin were used: the nonpigmented WA strain (Crl: WI(Han) outbred SPF-Quality, Charles River, Germany) and the pigmented BN strain (Crl: BN inbred SPF-Quality, Charles River, Germany). Animals were group housed in Makrolon type IV (MIV type, height 18 cm) plastic cages with a maximum of 5 animals per cage. Environmental controls for the animal room were set to maintain 18°C to 24°C, a relative humidity of 40% to 70%, at least 10 air changes/hour, and a 12-hour/12-hour light/dark cycle. Animals had free access to pelleted rodent diet (SM R/M-Z from SNIFF Spezialdiäten GmbH, Soest, Germany) and tap water. The animals were further supplied with sterilized sawdust as bedding material (Lignocel S 8-15; J. Rettenmaier & Söhne GmbH + CO KG, Rosenberg, Germany) and paper as cage enrichment (Enviro-dri; Wm Lillico & Son [Wonham Mill Ltd], Surrey, United Kingdom). The acclimatization period was at least 5 days before the start of treatment under laboratory conditions. All in vivo studies were approved by the Animal Welfare Officer and the ethical committee at Charles River Den Bosch as required by the Dutch Act on Animal Experimentation (February 1997).
Minimal Erythema Dose and Minimum Tolerated Dose Determination
An experiment was performed for the different strains in order to investigate their sensitivity to irradiation. Irradiation was performed using the Oriel solar simulator (ASTM E892 and IEC 904-3, Newport Corporation, Irvine, California) equipped with the AM 1.5 global filter (Newport Corporation) matching the total spectrum when the sun is at a zenith angle of 48.2°. The ultraviolet light B (UVB) part of the solar spectrum was attenuated by using a WG320 filter (Newport Corporation), significantly blocking the wavelengths below 320 nm. The ultraviolet light A (UVA) dose was measured using a UVA meter (Honle Technologies, Germany).
The minimal erythema dose (MED), the dose of UVA adequate to elicit a barely perceptible response in skin, and the minimum tolerated dose (MTD) for the eyes, the highest dose which showed no severe adverse effects, were determined as follows. Three animals per strain were anesthetized by intraperitoneal injection of 40 mg/kg Zoletil 50. Subsequently, all animals had the hair removed from their backs with electric clippers. The animals were positioned in a plastic tray and in order to irradiate the eyes, the head of each animal was elevated so that the eyes were on a plane equivalent to the skin site. The eyes remained open during anesthesia and were lubricated with Duratears Z immediately before exposure and, if necessary, during or after irradiation. The trays were placed under the solar simulator. In order to irradiate a specific skin spot (approximately 3 cm × 3 cm), the remaining skin of the animal was protected against irradiation using cardboard on top of the tray. Per animal, 4 skin spots were irradiated. The irradiation of the eyes could be interrupted independently from the skin irradiation by covering with a piece of cardboard. Animals were exposed with increasing UVA doses with attenuation of the UVB part of the solar spectrum. The lower and upper limit of the UVA doses applied were 5 J/cm2 and 20 J/cm2, respectively, which were selected based on the ICH S10 guidance document. 6 The dose of 10 J/cm2 was included in the range as this irradiation dose is frequently used in photosafety evaluation studies. 11,12 Skin reactions were visibly assessed immediately and 0.5, 1, 4, 24, 48, and 72 hours after irradiation. Ophthalmoscopy was performed on the eyes prior to irradiation and on the day of the last skin reaction observations.
Main Study
A study with phototoxic compounds with varying melanin affinity was performed. Ranging from high to low affinities, SPX (100 mg/kg) with 95% melanin-binding affinity, 8-MOP (15 mg/kg) with 69% binding affinity, and PIF (300 mg/kg) with 2% binding affinity were formulated in PEG 400 as vehicle and dosed orally once daily for 3 consecutive days to WA and BN rats. The study design and group allocation for the study with different melanin binders are described in Table 1. After the final formulation administration, animals were anesthetized and the hair removed from their backs with electric clippers. After positioning in the plastic tray, specific skin spots were irradiated as described above. The irradiation UVA dose was 12 J/cm2 and the UVB part of the spectrum was attenuated using the WG320 filter. For each test compound, irradiation was started in such a way that
Scheme of Study Design for the Study With Different Melanin Binders.
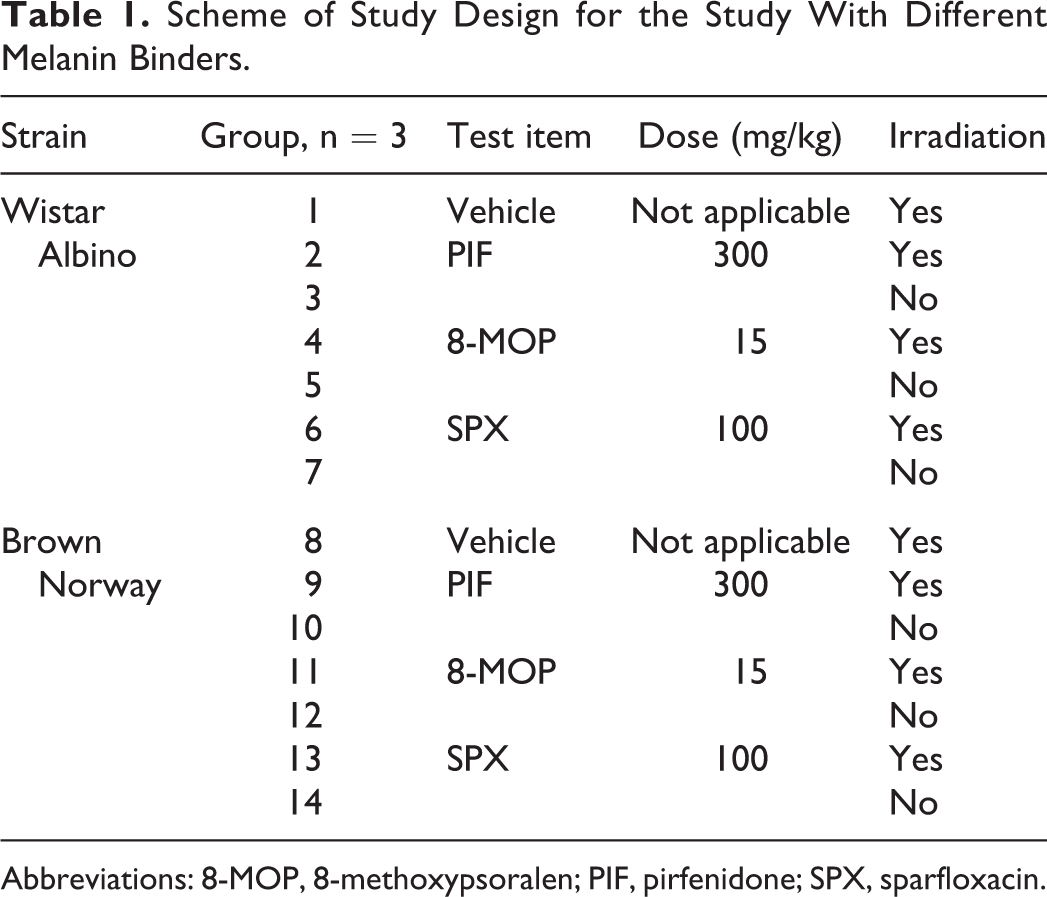
Abbreviations: 8-MOP, 8-methoxypsoralen; PIF, pirfenidone; SPX, sparfloxacin.
Assessment of Skin Reactions
Skin reactions (erythema/eschar and edema formation) were visibly scored using the assessment scheme of Draize. 14 Skin reactions that were less or not present in control animals (either dosed with the test compound but not irradiated or dosed with vehicle and irradiated) were considered indicative for phototoxicity (Table 2).
Evaluation of Skin Reactions.

Ophthalmoscopic Examinations
Ophthalmoscopic examinations were performed by a trained veterinarian. Results were used to evaluate possible reversible effects on the eyes.
Histopathology
All animals were sacrificed by asphyxiation using an oxygen/carbon dioxide procedure. All animals were subjected to necropsy and descriptions of all internal macroscopic abnormalities were recorded. A small skin section of the irradiated area (if available) and a skin section of the nonirradiated area were fixated in formalin. The left and right eye were excised, individually labeled and fixed in modified Davidson solution, and transferred to formalin after fixation for at least 24 hours. The skin samples and the left and right eye were processed, embedded in paraffin wax, cut to slides at a thickness of 2 to 4 µm, and stained with hematoxylin and eosin for histopathological examination.
Measurement of Total Test Compound Concentration in the Skin
From irradiated animals, skin samples of both the irradiated and nonirradiated skin were excised at necropsy, while from animals without irradiation, only nonirradiated skin samples were excised at necropsy. After excision, skin samples were deeply frozen in liquid nitrogen and stored at 80°C. Skin samples of animals dosed with SPX, 8-MOP, and PIF, and corresponding vehicle control samples were minced with scissors and subsequently homogenized in 7.5 mL ACN per gram of skin tissue using an Ultra-Turrax disperser (Boom BV, Meppel, the Netherlands). The resulting suspensions were incubated overnight at room temperature under gentle shaking. Next, 1 mL of the suspension was transferred to an Eppendorf tube followed by centrifugation for 5 minutes at 16 000
Results
In Vitro Melanin-Binding Screen
The melanin-binding affinity of 16 compounds was screened at 2 substrate concentrations (1 and 100 µM) after 24 hours of incubation at room temperature. The results of the melanin-binding screen are given in Table 3. For all tested compounds, the percentage of melanin binding decreased when the test compound concentration in the incubation was increased from 1 to 100 µM. The coefficient of variation was below 11% for all experiments. Spiked reference control samples were analyzed to determine recovery of the test compounds from the control incubation mixtures and recoveries ranged between 90% and 110%, with the exception of CPZ at a 1-µM concentration (77.1%) and PIF at a 100 µM concentration (86.5%). Compounds were considered to be “high-affinity melanin binders” when ≥75% was bound to melanin at a 1 µM test compound concentration, “moderate-affinity melanin binders” when between 25% and 75% was bound to melanin, and “low-affinity melanin binders” when less than 25% was bound to melanin. Based on these criteria, PRO, CPZ, CPX, SPX, LVX, and DOX were considered to be high-affinity melanin binders; 8-MOP, TCSA, TIM, NA, and HTZ were considered to be moderate-affinity melanin binders; and DIC, PIX, PIF, and OLA were considered to be low-affinity melanin binders.
Melanin Library Screen-Binding Data.
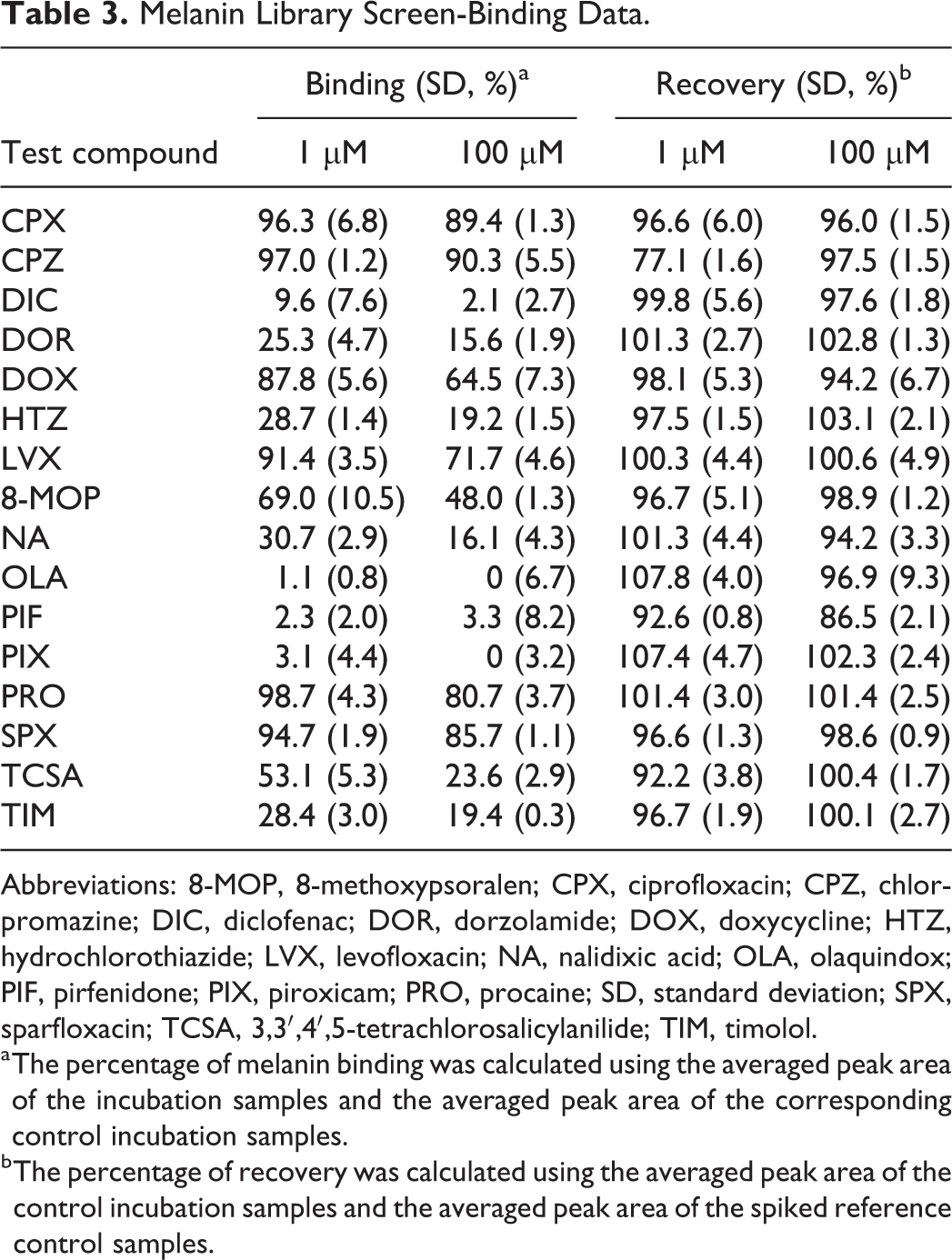
Abbreviations: 8-MOP, 8-methoxypsoralen; CPX, ciprofloxacin; CPZ, chlorpromazine; DIC, diclofenac; DOR, dorzolamide; DOX, doxycycline; HTZ, hydrochlorothiazide; LVX, levofloxacin; NA, nalidixic acid; OLA, olaquindox; PIF, pirfenidone; PIX, piroxicam; PRO, procaine; SD, standard deviation; SPX, sparfloxacin; TCSA, 3,3′,4′,5-tetrachlorosalicylanilide; TIM, timolol.
a The percentage of melanin binding was calculated using the averaged peak area of the incubation samples and the averaged peak area of the corresponding control incubation samples.
b The percentage of recovery was calculated using the averaged peak area of the control incubation samples and the averaged peak area of the spiked reference control samples.
Minimal Erythema Dose and MTD Determination
The MED was determined to exceed a UVA dose of 20 J/cm2 with attenuation of the UVB part based on the absence of any skin reaction at any irradiation dose at any time point up to 72 hours. For the eyes, the MTD was determined to exceed a UVA dose of 10 J/cm2 for the WA rats and a UVA dose of 20 J/cm2 for the BN rats. This was based on the observation of degenerating cells in the outer nuclear layer of 1 eye for the WA rats irradiated at a UVA dose of 20 J/cm2 and the absence of findings in the eyes of the BN rats.
Main Study: Skin Responses
Three phototoxic compounds with varying melanin-binding affinity were tested: SPX (95%), 8-MOP (69%), and PIF (2%). None of the control groups, dosed with vehicle and irradiated or dosed with phototoxic compound and not irradiated, showed any signs of erythema. In addition, none of the animals showed any signs of edema. Detailed results for individual animals can be found in Tables 4 and 5, while histopathological findings can be found in supporting information.
Erythema Grades in the Wistar Albino Rat Strain.
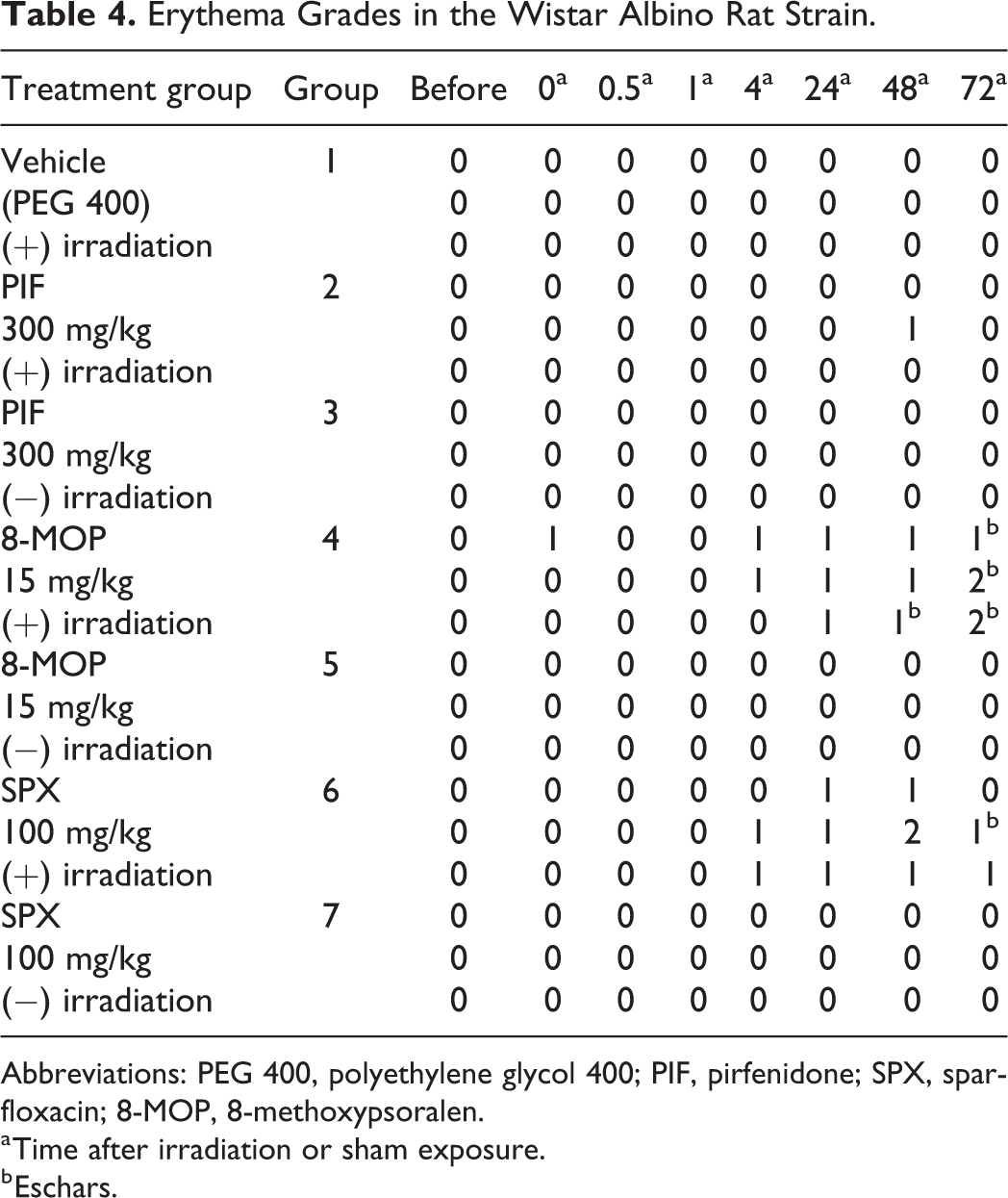
Abbreviations: PEG 400, polyethylene glycol 400; PIF, pirfenidone; SPX, sparfloxacin; 8-MOP, 8-methoxypsoralen.
a Time after irradiation or sham exposure.
b Eschars.
Erythema Grades in the Brown Norway Rat Strain.
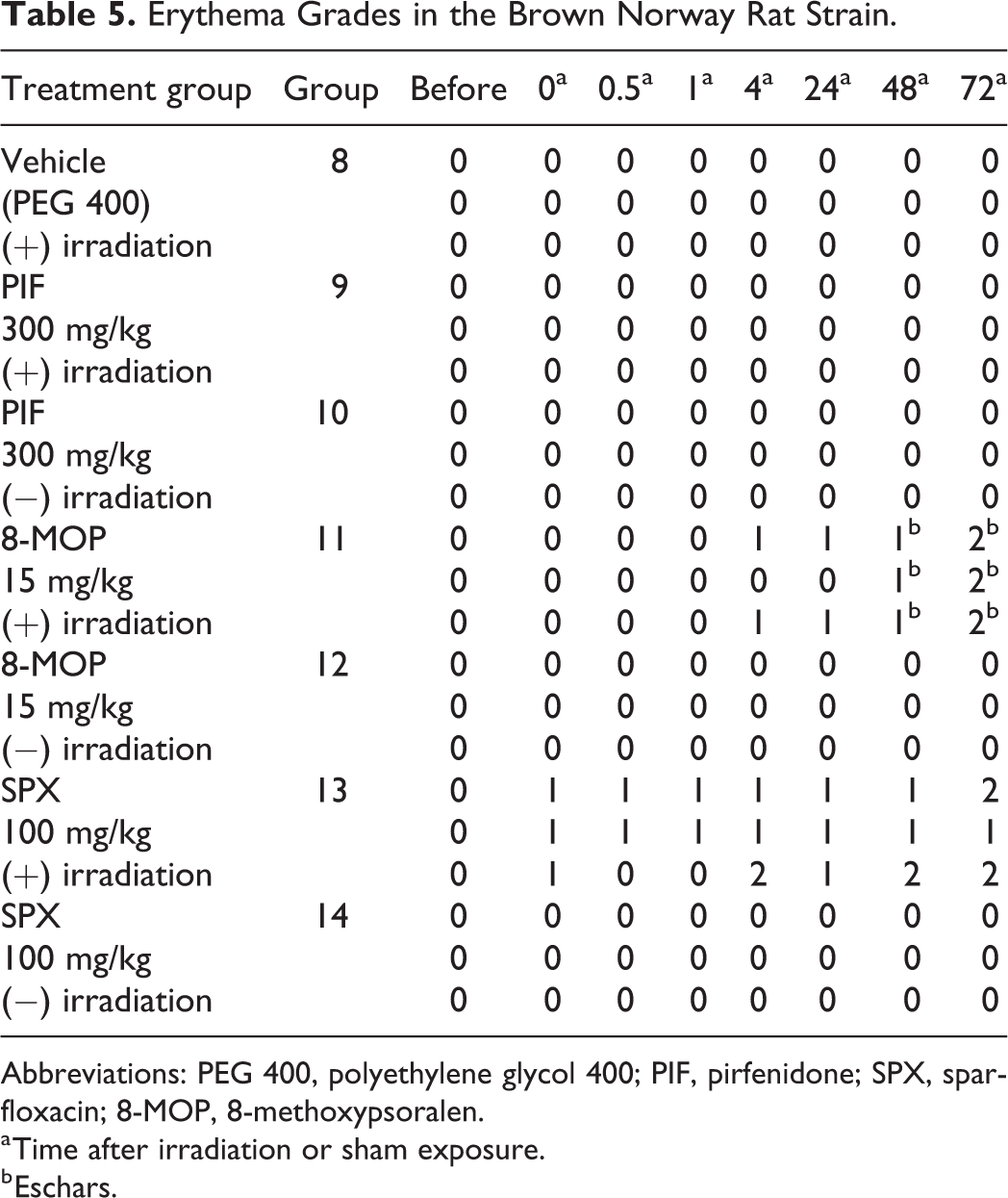
Abbreviations: PEG 400, polyethylene glycol 400; PIF, pirfenidone; SPX, sparfloxacin; 8-MOP, 8-methoxypsoralen.
a Time after irradiation or sham exposure.
b Eschars.
The SPX-dosed and irradiated rats of both the BN and WA strains showed erythema. In the BN rats, erythema was observed at all time points and at the end of the observation period, 2 animals displayed well-defined erythema while 1 animal displayed slight erythema. In the WA rats, the first signs of erythema were observed in 2 animals 4 hours after radiation. All 3 animals displayed slight erythema 24 hours after irradiation while at the end of the observation period, 2 animals displayed slight erythema with 1 of these animals also showing eschar formation. Histopathological evaluation of the skin confirmed the observed skin reaction. The treatment-related erythema scores are shown in Table 6. As can be seen, treatment-related erythema scores for SPX were higher for the animals of the BN strain than for animals of the WA strain.
Treatment-Related Erythema Scores per Skin Type During the Study With Different Melanin Binders.a
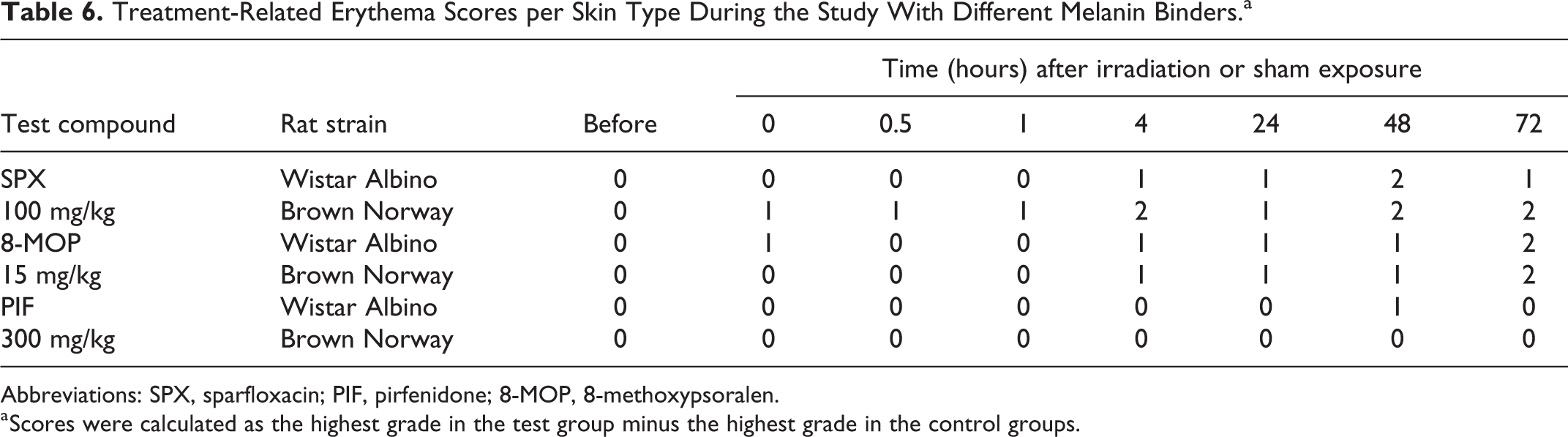
Abbreviations: SPX, sparfloxacin; PIF, pirfenidone; 8-MOP, 8-methoxypsoralen.
a Scores were calculated as the highest grade in the test group minus the highest grade in the control groups.
The 8-MOP-dosed and irradiated rats of both the BN and WA strains showed erythema. For the BN rats, no signs of erythema were seen until 4 hours after irradiation. At the end of the observation period, all 3 animals had well-defined erythema with eschar formation. For the WA rats, 1 animal showed slight erythema immediately after irradiation. The erythema gradually increased for 2 animals displaying slight erythema at 4 hours after irradiation to all 3 animals displaying well-defined (1 animal) or moderate (2 animals) erythema in combination with eschar formation at the end of the observation period. Histopathological evaluation of the skin confirmed the observed skin reaction. As can be seen from Table 6, there were no differences for the treatment-related erythema scores between the WA and BN rats.
For the PIF-treated rats, very slight erythema was only observed in 1 WA rat 48 hours after irradiation. No further skin responses were noted in any of the WA and BN rats. Treatment-related erythema scores are shown in Table 6, and as can be seen, no clear distinction in scores could be made between the WA and BN strains.
Main Study: Effects on the Eyes
Details on ophthalmoscopic results and histopathological evaluation can be found in supporting information. For the vehicle-dosed and irradiated animals, ophthalmoscopy revealed small treatment-related corneal findings for most BN and WA rats. Histopathological evaluation revealed no treatment-related findings. For SPX-dosed and PIF-dosed rats, ophthalmoscopy revealed small treatment-related corneal findings for most irradiated BN and WA rats. Histopathological evaluation revealed no treatment-related findings. For 8-MOP-dosed rats, ophthalmoscopy revealed marked treatment-related corneal findings for both the BN and WA rats with and without irradiation. Histopathological evaluation revealed severe corneal findings for the irradiated groups of both strains with additional findings in the lens, iris, and retina of the WA rats (Figures 1 and 2).
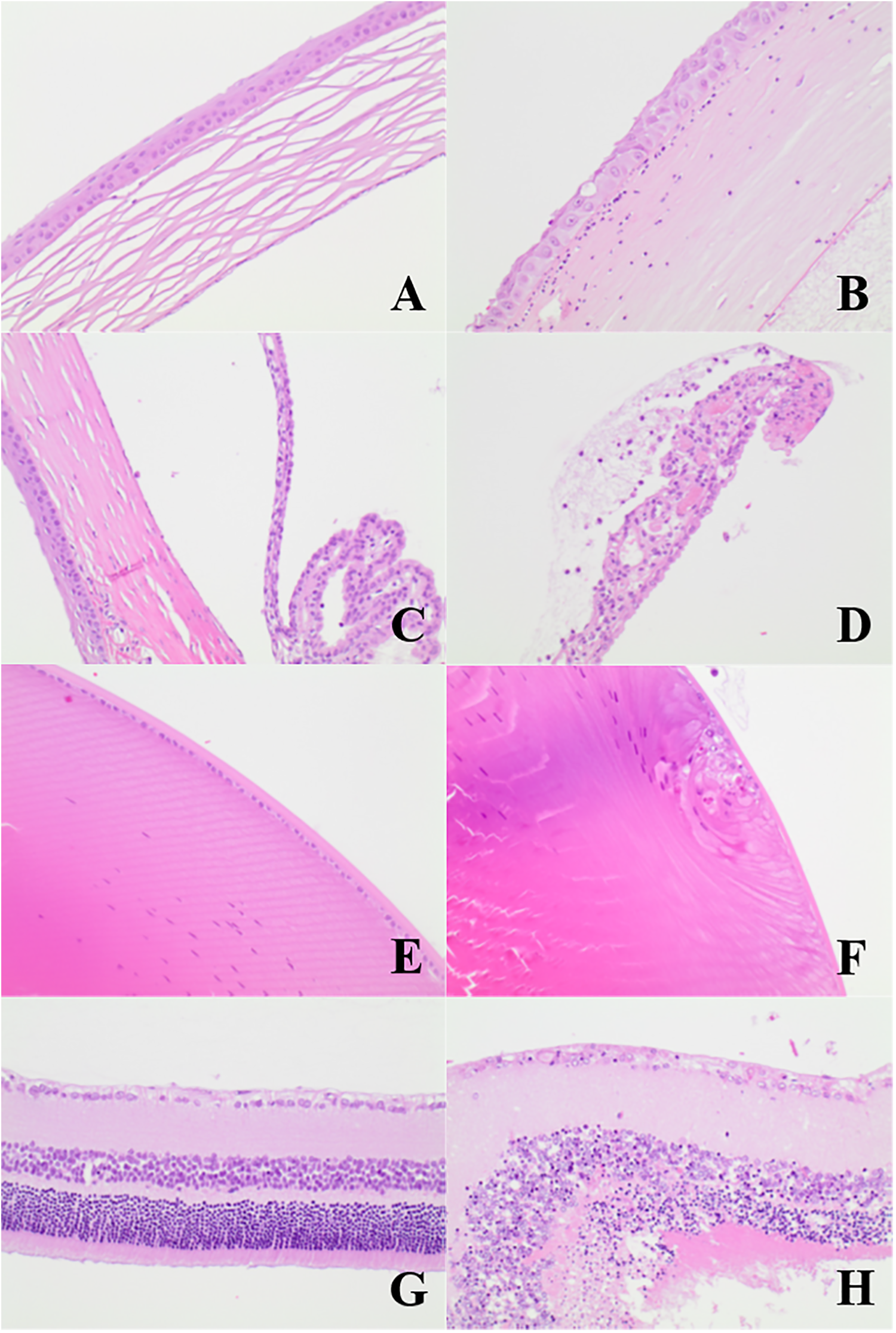
Histopathological staining of the cornea, iris lens, and retina of the Wistar Albino rat. Rats were treated with 15 mg/kg 8-MOP or a vehicle control once daily for 3 consecutive days followed by irradiation with 12 J/cm2 UVA. The unaffected (vehicle control; A) and the affected cornea (8-MOP; B) are shown. The affected cornea showed degeneration of the cornea, including inflammatory infiltrate, vacuolation, apoptosis, and stromal edema. The unaffected (C) and the affected iris (D) are shown. The affected iris showed degeneration of the iris, including inflammatory infiltrate. The unaffected (E) and affected (F) lens are shown. The affected lens showed necrosis. The unaffected (G) and affected (H) retina are shown. The affected retina showed necrosis. 8-MOP indicates 8-methoxypsoralen; UAV, ultraviolet light A.
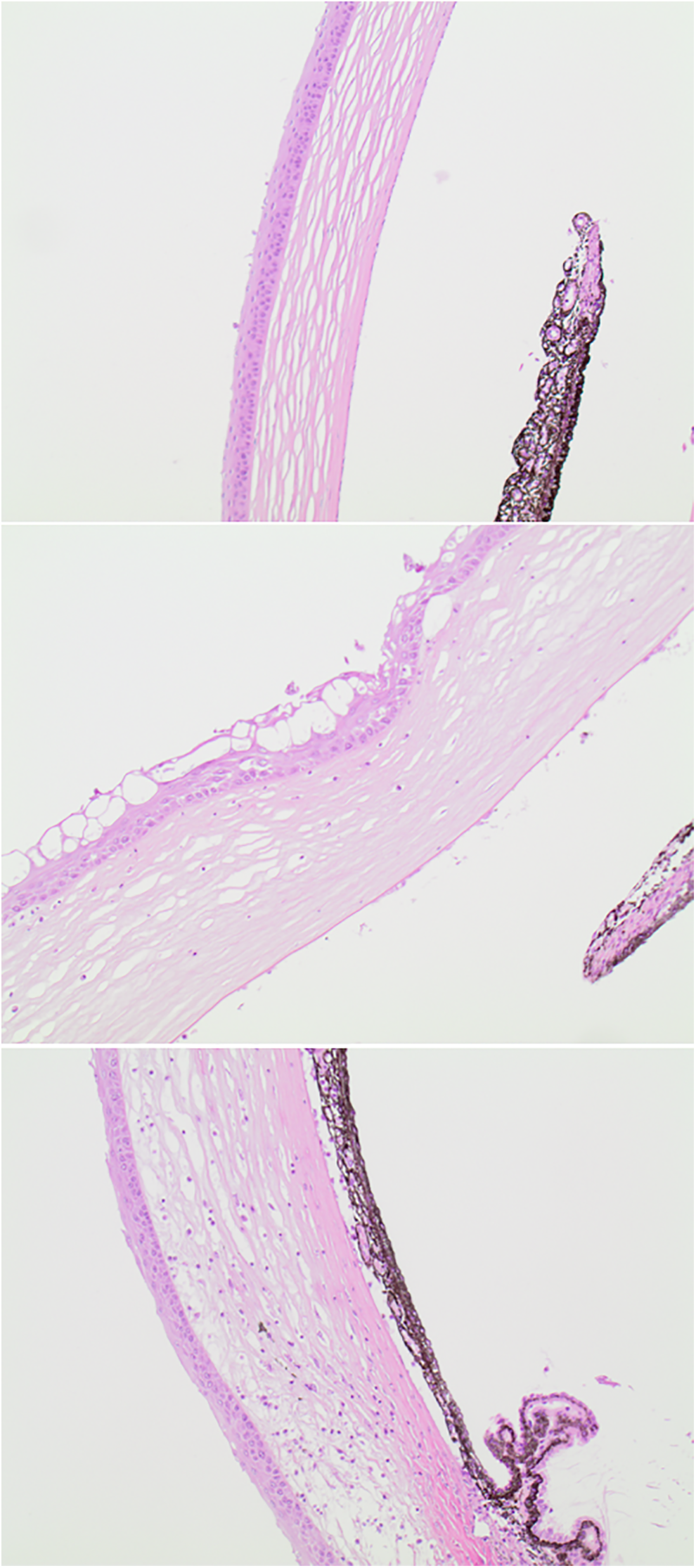
Histopathological staining of the cornea of the Brown Norway (BN) rat. Rats were treated with 15 mg/kg 8-MOP or a vehicle control once daily for 3 consecutive days followed by irradiation with 12 J/cm2 UVA. The unaffected (vehicle control) cornea of the BN rat is shown in the top picture. The middle and bottom pictures show the affected (8-MOP) cornea of the BN. Phototoxic effects on the cornea were shown as degeneration of the cornea, including vacuolation, inflammatory infiltrate, and apoptosis. 8-MOP indicates 8-methoxypsoralen; UAV, ultraviolet light A.
Test Compound Skin Tissue Concentrations
The concentration of the high-affinity melanin binder SPX in skin samples was measured by UPLC-MS, and results are summarized in Table 7. Detailed results for individual animals can be found in supporting information. For all skin samples obtained from the vehicle control animals, SPX concentrations were below the limit of detection (LOD; 5.89 ng SPX/g tissue). In all skin samples of animals that were exposed to SPX, SPX concentrations were above the LOD. The SPX concentrations measured in the irradiated and nonirradiated skin samples obtained from the pigmented BN animals were higher than those measured in the corresponding samples obtained from the nonpigmented WA animals. In the nonirradiated skin, the mean SPX concentration in the BN animals was approximately 10-fold higher than the mean SPX concentration in the WA animals, and the difference between these groups was statistically significant (
Test Compound Concentrations Measured in Skin Samples.
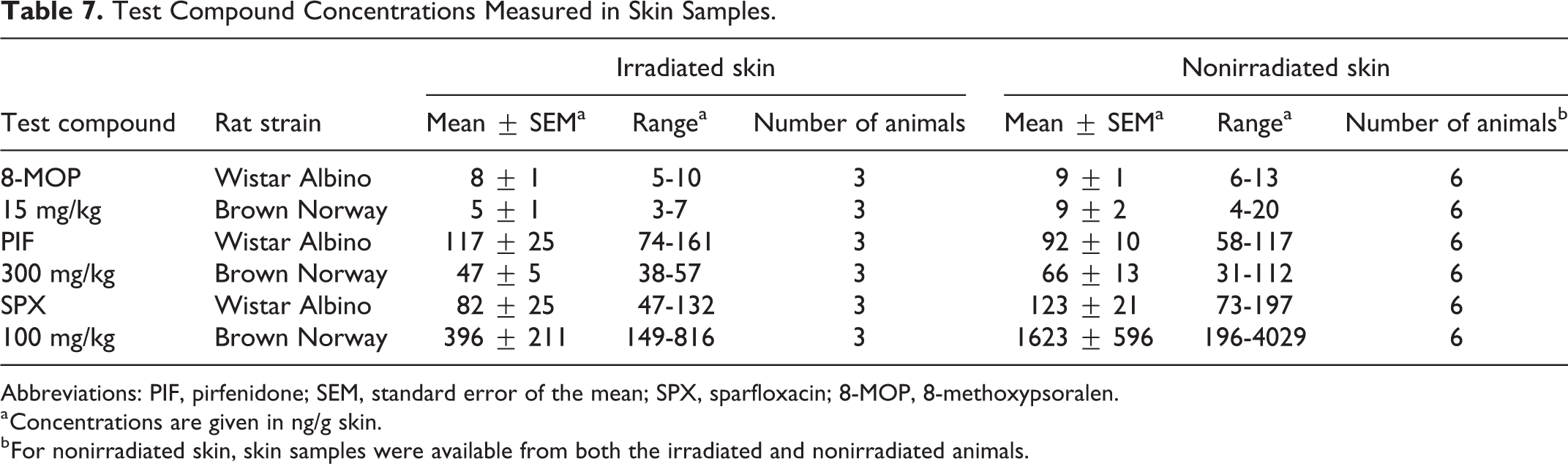
Abbreviations: PIF, pirfenidone; SEM, standard error of the mean; SPX, sparfloxacin; 8-MOP, 8-methoxypsoralen.
a Concentrations are given in ng/g skin.
b For nonirradiated skin, skin samples were available from both the irradiated and nonirradiated animals.
When looking at the results obtained for the moderate-affinity melanin binder 8-MOP, it can be seen from Table 7 that there are no significant differences between the test compound concentrations measured in the WA animals and the BN animals (both for the irradiated and the nonirradiated skin). In the skin tissue samples obtained from the vehicle control animals, concentrations of the test compound were below the LOD (0.32 ng 8-MOP/g tissue). It was furthermore observed that there were no significant differences between the test compound concentrations in the irradiated and the nonirradiated skin in animals from the same strain.
When looking at the results obtained for the low-affinity binder PIF, it can be seen from Table 7 that there are no significant differences between the PIF concentrations measured in the WA animals and the BN animals (both for the irradiated and the nonirradiated skin). In the skin tissue samples obtained from the vehicle control animals, PIF concentrations were below the LOD (2.78 ng PIF/g tissue). No significant differences between the irradiated and nonirradiated skin in animals from the same strain were observed.
Discussion
For humans, one of the most important biological functions of melanin is thought to be photoprotection of the skin. 15 It has been reported that humans with white skin are approximately 70 times more likely to develop skin cancer compared to humans with black skin. 15 -17 In addition, when the photoprotective role of melanin was investigated in both black and Caucasian skin, black epidermis was found to display superior photoprotection. 18 These results suggest that constitutive melanin pigmentation decreases susceptibility to deleterious effects of UV radiation. However, melanin is also thought to retain certain drugs in pigmented cells for long periods, which increases the likelihood for photoreactive drugs to produce a phototoxic reaction. 19 Accumulation of drugs with melanin affinity in ocular tissues and differences in the pharmacological effects of ocular drugs in pigmented and nonpigmented animal models has been reported. 20 However, in a study that compared photodermatoses in African Americans and Caucasians, it was reported that the frequency of photodermatoses in African Americans and Caucasians are similar while porphyrias and phototoxicity occur more commonly in Caucasians. 21
The ICH S10 guidance document states that for drugs that bind significantly to melanin, pigmented skin should be considered for detecting phototoxicity. Based on this, for the current study, it was hypothesized that for photosafety evaluation of drugs with low melanin-binding affinity, a nonpigmented animal model would be most sensitive for detection of phototoxicity as it is not influenced by the possible melanin-related UV protection. For photosafety evaluation of drugs with high melanin-binding affinities, a pigmented animal model would be most sensitive as it takes possible tissue accumulation effects into account. By testing possible in vivo phototoxic effects of drugs with varying melanin-binding affinities in different animal models, it was investigated that if based on these hypotheses, an in vitro melanin affinity threshold for animal model selection could be defined to develop an accurate phototoxicity testing strategy.
Various sources of melanin can be used for in vitro melanin-binding studies. Natural sources include melanin obtained from bovine and porcine eyes and
When looking at the structural properties of the high-affinity binders, it was observed that they all contain strong basic functionalities, such as piperidine or piperazine moieties or other amines, which is in agreement with previous findings. 34 For DOR and TIM, it has been reported in the literature that they bind to melanin with low-to-moderate affinities while it was reported for CPZ and PRO that they bind with high affinities. 28,26,22 In a study by Ono and Tanaka, it was demonstrated that CPX, LVX, and SPX bind to synthetic melanin and that the binding affinities of CPX and SPX are higher than those observed for LVX. 35 All these results agree with the findings of the current study. The high-affinity binder SPX (95% binding at a 1-µM test compound concentration), moderate-affinity binder 8-MOP (69%), and the low-affinity binder PIF (2%) were selected for in vivo experiments based on the fact that in vivo phototoxicity in rats has been reported previously for these drugs. 11 -13,36
For SPX, the treatment-related skin responses were higher in the pigmented rats, compared to nonpigmented rats, which correlates with the higher SPX concentrations found in the skin tissues of the pigmented rats. The finding that lower SPX concentrations were measured in irradiated skin samples compared to nonirradiated skin samples from the same animal can be explained by the fact that SPX may have reacted after exposure to UVA, which resulted in the formation of photoproducts, photoadducts, or ROS and thereby a lower SPX concentration. To confirm these findings, ideally, the study should be repeated with more animals, and irradiated and nonirradiated skin samples should be analyzed for the presence of SPX-related adducts and/or degradation products.
For 8-MOP, there was no clear difference in treatment-related skin responses that correlates with the lack of difference in the 8-MOP skin concentrations. For PIF, no differences in PIF skin tissue concentrations were measured while no skin reactions were observed. Since PIF is a low binder, no difference in skin concentration was to be expected. The absence of skin responses disagrees with previous studies. 12,36 However, in the study of Seto et al, Sprague Dawley rats and a UVA dose of 30 J/cm2 were used while a colorimetrical evaluation on the surface of the skin was used to determine the occurrence of phototoxicity instead of Draize method. 36 In the study of Adachi et al, Long-Evans rats that were exposed to both UVA (10 J/ cm2) and UVB (0.06 J/cm2) were used, and a skin reaction was observed in only 1 animal (out of 3) when PIF was dosed at 750 mg/kg. 12 These differences in study setups could explain why no skin reactions were observed in the current study. Histopathological evaluation of the skin correlated with the skin responses; however, since no additional observation was made based on these evaluations, it was concluded that histopathological evaluations are not essential for phototoxicity studies.
In addition to the assessment of skin reactions, for detection of phototoxicity on the eyes, ocular effects were assessed. Only 8-MOP showed clear ocular phototoxic effects in both the WA and BN rats, which corresponded with previous findings in Sprague Dawley rats. 12 The effects were more pronounced in the WA rats. For SPX, minor phototoxic effects were observed in the WA animals, which were absent in the BN animals. These results indicate that melanin may be protective and that testing for ocular phototoxicity should be performed in the nonpigmented WA model; however, it should be noted that the drugs that were selected for this study were not ideal for investigation of ocular phototoxicity since their absorption spectra mainly lies within the UVA range. For ocular phototoxicity studies, test compounds should be able to absorb visible light. Furthermore, in the current study setup, histopathology of the eyes could only be performed at a single time point, which suggests that any reversible effects occurring shortly after irradiation are most likely missed.
An in vitro melanin-binding screen was successfully applied to assess the affinity of 16 structurally diverse drugs to bind to synthetic melanin. High-affinity, moderate-affinity, and low-affinity melanin binders were identified and 3 phototoxic drugs with varying melanin-binding affinities, being SPX (95% binding affinity), 8-MOP (69%), and PIF (2%), were selected for further in vivo studies. Phototoxic effects for SPX, 8-MOP, and PIF were studied in nonpigmented WA rats and pigmented BN rats in order to establish which animal model would be best for photosafety evaluation in relation to melanin-binding affinity. It was concluded that for the high-affinity binder SPX, the BN model was most sensitive. For 8-MOP, both models showed comparable sensitivity. For the low-affinity binder PIF, no phototoxic effects were observed in any of the animal models. The in vitro melanin-binding affinities of the test compounds were correlated to the skin tissue concentrations measured in the different animal models. For the high-affinity melanin binder SPX, a significantly higher concentration was measured in the skin of the pigmented BN animals while for the other drugs with lower melanin-binding affinities, no significant differences between the concentrations measured in both models could be found. Ocular phototoxicity was found not to be related to melanin-binding affinity in this study.
The current study evaluated the phototoxic effects (using different end points) for 3 different drugs with varying melanin-binding affinities that have been previously associated with phototoxic effects. A pigmented rat model was compared with a nonpigmented rat model in order to assess their sensitivity to detect phototoxic effects to test the hypothesis that for photosafety evaluation of drugs with high melanin-binding affinities, a pigmented animal model would be most sensitive as it takes possible tissue accumulation effects into account. The current results demonstrate that for the high-affinity melanin binder SPX, the onset of phototoxic symptoms was faster and drug concentrations were significantly higher in skin of the pigmented animal model. These data therefore suggest that for photosafety evaluation of drugs with high melanin-binding affinities, a pigmented animal model is most sensitive; however, this should be confirmed by performing follow-up studies with more high-affinity melanin binders. Nevertheless, the current study clearly demonstrates that further research is warranted.
Overall, it can be concluded that a combined in vitro
Supplemental Material
Supplemental Material, DS1_IJT_10.1177_1091581818777998 - Development of a Phototoxicity Testing Strategy for Accurate Photosafety Evaluation of Pharmaceuticals Based on the Assessment of Possible Melanin-Binding Effects
Supplemental Material, DS1_IJT_10.1177_1091581818777998 for Development of a Phototoxicity Testing Strategy for Accurate Photosafety Evaluation of Pharmaceuticals Based on the Assessment of Possible Melanin-Binding Effects by Jelle Reinen, Pieter van Sas, Ton van Huygevoort, Leticia Rubio, Kevin Scase, and Mira Wenker in International Journal of Toxicology
Footnotes
Acknowledgments
The authors would like to thank the (bio)technicians and pathologists for their expertise and technical assistance.
Authors’ Contributions
Jelle Reinen, Pieter van Sas, and Ton van Huygevoort designed the experiments. Jelle Reinen, Leticia Rubio, and Pieter van Sas performed the experiments. Jelle Reinen, Pieter van Sas, and Ton van Huygevoort analyzed the data. Jelle Reinen and Pieter van Sas wrote the manuscript. Ton van Huygevoort, Kevin Scase, and Mira Wenker reviewed the manuscript. Reinen, J. and Van Sas, P. contributed to conception and design; acquisition, analysis, and interpretation; and drafted manuscript. Van Huygevoort, T. contributed to conception and design, interpretation, and critically revised manuscript. Rubio, L. contributed to acquisition and analysis and critically revised manuscript. Scase, K. contributed to design and critically revised manuscript. Wenker, M. contributed to design and critically revised manuscript. All authors gave final approval agree to be accountable for all aspects of work ensuring integrity and accuracy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
