Abstract
Kidney diseases have notably increased in the last few years. This is partially explained by the increase in metabolic syndrome, diabetes, and systemic blood hypertension. However, there is a segment of the population that has neither of the previous risk factors, yet suffers kidney damage. Exposure to atmospheric pollutants has been suggested as a possible risk factor. Air-suspended particles carry on their surface a variety of fuel combustion–related residues such as metals, and vanadium is one of these. Vanadium might produce oxidative stress resulting in the damage of some organs such as the kidney. Additionally, in countries like Mexico, the ingestion of sweetened beverages is a major issue; whether these beverages alone are responsible for direct kidney damage or whether their ingestion promotes the progression of an existing renal damage generates controversy. In this study, we report the combined effect of vanadium inhalation and sweetened beverages ingestion in a mouse model. Forty CD-1 male mice were distributed in 4 groups: control, vanadium inhalation, 30% sucrose in drinking water, and vanadium inhalation plus sucrose 30% in drinking water. Our results support that vanadium inhalation and the ingestion of 30% sucrose induce functional and histological kidney damage and an increase in oxidative stress biomarkers, which were higher in the combined effect of vanadium plus 30% sucrose. The results also support that the ingestion of 30% sucrose alone without hyperglycemia also produces kidney damage.
Introduction
In recent years, the incidence of kidney diseases has increased worldwide and often they are associated with diabetes mellitus and metabolic syndrome. 1 However, in some cases, it is difficult to establish the causes that lead to kidney damage. Some risk factors for this damage have been suggested, for example, environmental pollutants and sweetened beverages consumption. 2 –4 Particulate matter (PM) is an atmospheric pollutant linked to several pathologies and is considered the 15th cause of global mortality. 5 In addition, associations between PM and reduced renal function 6 have been found. Some metals are adsorbed into PM such as vanadium. This element is released into the atmosphere as a fossil fuel combustion product, especially in Mexican and Venezuelan oils because it is present in higher concentrations. In the United States, the airborne average concentration of vanadium is 11 ng/m3, but in US Eastern cities, the air concentration is 620 ng/m3. 7 In Mexico City, there are reports of 0.114 μg/m3 of vanadium in PM 10 or 0.093 μg/m3 in PM 2.5, similar to the airborne concentration in European cities. 8 Some foods and dietary supplements have high concentrations of this metal which is poorly absorbed in the gastrointestinal tract. Inhalation is the main route of vanadium absorption. 9 Vanadium has hypoglycemic effects because of its insulin agonist effect. 9 In other studies, it has been associated with the damage of different organs and systems, including the kidney. 10 –12 One of the mechanisms of vanadium toxicity is oxidative stress induction, through the Fenton’s reaction and also by its ability to inhibit some antioxidative enzymes. 13 Vanadium is excreted in urine in a biphasic manner, first in about 20 hours and then after 50 days. 9
The ingestion of sweetened beverages has increased worldwide, 14 and there is controversy as to whether this increase in sugar consumption induces or favors the development of kidney diseases. Bomback et al 15 conducted a study analyzing sweetened drink consumption and found no connection with the induction of kidney diseases. These results contrast with those obtained by Shoham et al 16 because they found evidence that consumption of 2.5 servings of sweetened beverages per day induces kidney damage identified by albuminuria. This finding is supported by experimental models in which morphology and renal function changes are found because of the consumption of sugar-sweetened beverages, and the toxic mechanisms suggested are oxidative stress and the induction of hyperuricemia. 17,18
Studies about sucrose or vanadium inhalation and kidney damage are limited. Both factors, metals and sugar, share oxidative stress as a mechanism of toxicity. 4-Hydroxynonenal (4-HNE) has been used as a good marker of oxidative stress because it is found as a result of lipid peroxidation. Also, 4-HNE interacts with some molecules of the cell to induce damage or to regulate oxidative/antioxidative mechanisms. 19 Therefore, the aim of this study is to analyze the effect of vanadium inhalation and 30% sucrose beverages on kidney histology, renal function, and oxidative stress markers and to test whether the hypothesis that the combination of both factors, vanadium plus 30% sucrose, would result in a greater damage.
Materials and Methods
Animals and Treatments
The experimental protocol was in accordance with the Guide for the Care and Use of Laboratory Animals from the Institute of Laboratory Animal Resources Commission on Life Sciences National Research Council (2010) and the Mexican official norm NOM 06-200-1999 for the production, care, and use of laboratory animals. Forty-eight CD-1 male mice weighing 33 ± 2 g were housed in hanging plastic cages under controlled light conditions (12-hour light/dark regime) and fed with Purina rat chow and water ad libitum. Inhalation was performed as described by Fortoul et al. 20 Mice were obtained from the vivarium at the School of Medicine, Universidad Nacional Autonoma de Mexico. Forty mice were randomly placed in acrylic boxes, 5 mice in each acrylic box, and distributed in 4 groups: group 1 controls (10 mice) inhaled only the vehicle—saline (sodium chloride [NaCl] 0.9%) for 1 hour twice a week; group 2 sucrose, inhaled the vehicle with free access to 30% sucrose-sweetened water for drinking; group 3 vanadium, inhaled [0.02 M] V2O5 (Sigma, St Louis, Missouri) for 1 hour twice a week over 8 weeks; and group 4 vanadium plus sucrose, inhaled vanadium pentoxide and had free access to 30% sucrose-sweetened water for the same period of time. Sucrose drinking water solutions were prepared daily. Water consumption per day was measured in all individuals from each group. This exposure protocol was selected knowing that the half-life of vanadium is about 48 hours. Either the vehicle or the vanadium suspension was introduced in the cage by an ultrasonic nebulizer by Yuehua (model WH-802) that according to the information of the manufacturer indicated that 80% of the nebulized particles reaching the mice have a range of aerodynamic diameters of 0.5 to 5.0 µm.
Vanadium Concentration in Chamber
A filter was positioned at the outlet of the ultra-nebulizer during the inhalation at a constant flow of 10 L/min. The filters were removed and weighed after each inhalation; vanadium was quantified evaluating 6 filters for each inhalation in a graphite furnace atomic absorption spectrometer (model 2380; Perkin Elmer, Shelton, Connecticut). The light source came from a hollow cathode lamp. Accuracy was assured by 3 random determinations of 7 different standard solutions, prepared with the same chemical reactive used during the metal analysis. For vanadium, the wavelength was 318.4 nm; the detection limit was 0.37 ppm, and the slit was 0.7 nm. Each sample was analyzed in triplicate. 21
Glycemic Reports and Sacrifice
With a previous 12-hour fasting period, a OneTouch Ultra glucometer was used for glycemic quantification. The blood sample was obtained from the tail vein at weeks 4 and 8 after the exposure. After the quantification, mice were anesthetized by intraperitoneal injection of an aqueous solution of 0.3 mg/mL pentobarbital sodium (PiSa, Agropecuaria, S.A. de C.V, México; 35 mg/kg body weight), then perfused via aorta with saline, followed by 4% paraformaldehyde (pH 7.4) in phosphate buffer. Kidneys were obtained and processed for light microscopy. Paraffin-embedded tissue sections were stained with hematoxylin and eosin. Two blind observers evaluated the slides with a light source microscope.
After the anesthesia but before the perfusion, urine samples were obtained by bladder puncture. The urine parameters evaluated were hematuria (erythrocyte count), glycosuria, and proteinuria. Dipstick Multistix (Siemens, Erlangen, Germany) were used for this purpose.
Immunohistochemistry of Anti 4-HNE
Tissue sections were dewaxed in xylene and dehydrated in graded ethanol series. Antigen retrieval was obtained by incubation with 2% Diva Decloaker (Diva Decloaker 20x; BioCare Medical, Concord, California), followed by 3% hydrogen peroxide (15 minutes; JT Baker, Phillipsburg, New Jersey). The specimens were then incubated with the primary antibody (anti 4-HNE antibody, diluted 1:200; R&D Systems, Minneapolis, Minnesota) for 30 minutes, followed by incubation with a drop of biotin-labeled universal link antibody for 30 minutes (Dako, Glostrup, Denmark), and later, were incubated with a drop of streptavidin–horseradish peroxidase (Dako) for 30 minutes. Immunoreactivity was identified by the incubation in 0.05% of 3,3′-diaminobenzidine tetrahydrochloride (Invitrogen, Camarillo, California). Finally, sections were counterstained with hematoxylin. Some samples only received the secondary antibody and served as negative controls.
Image Processing and Densitometry Analysis
Olympus microscope (model BH2-RFCA) and a camera of the same brand were used. The images were processed with Adobe Photoshop CC software. The processing included brightness and contrast adjustments and turning the background completely white. Also, in the immunohistochemistry microphotography, they were separated in 3-color channels, but used only the yellow channel to make the densitometry analyzed with DensiFe program developed in MathLab. Five fields (0.059 µm2 each) per mice randomly selected at 400× objective were analyzed, and 5 mice per group.
Statistical Analysis
To evaluate the differences in glycemia and density mean for 4-HNE, 1-way analysis of variance (P < 0.05) followed by Tukey post hoc test was conducted. Student t test was conducted to evaluate statistical differences between controls, fourth week–, and eighth week–exposed mice (P < 0.05). The statistical analysis was performed using Prism Software, V 6.0 GraphPad (San Diego, California).
Results
Vanadium Concentration in the Chamber
The concentration of vanadium in the chamber was 1,436 μg/m3 during the exposure.
Water Consumption
The average of water consumption of control mice was 14 mL per mouse per day, vanadium group consumed 10 mL per mouse per day, sucrose group consumed 5.4 mL per mouse per day, and vanadium plus sucrose group consumed 6.4 mL per mouse per day. The average of sucrose consumed in drinking water was none in control and vanadium groups, 46.28 g/kg body weight/day in sucrose group, and 54.85 g/kg body weight/day in vanadium plus sucrose group.
Glycemia and Urinary Parameters
Vanadium plus sucrose group showed significant hypoglycemia in comparison with the control group in the fourth week, and the vanadium group had significant hypoglycemia in the eighth week. Also in the eighth week, the sucrose group presented a significant hyperglycemia in comparison with control (Figure 1).
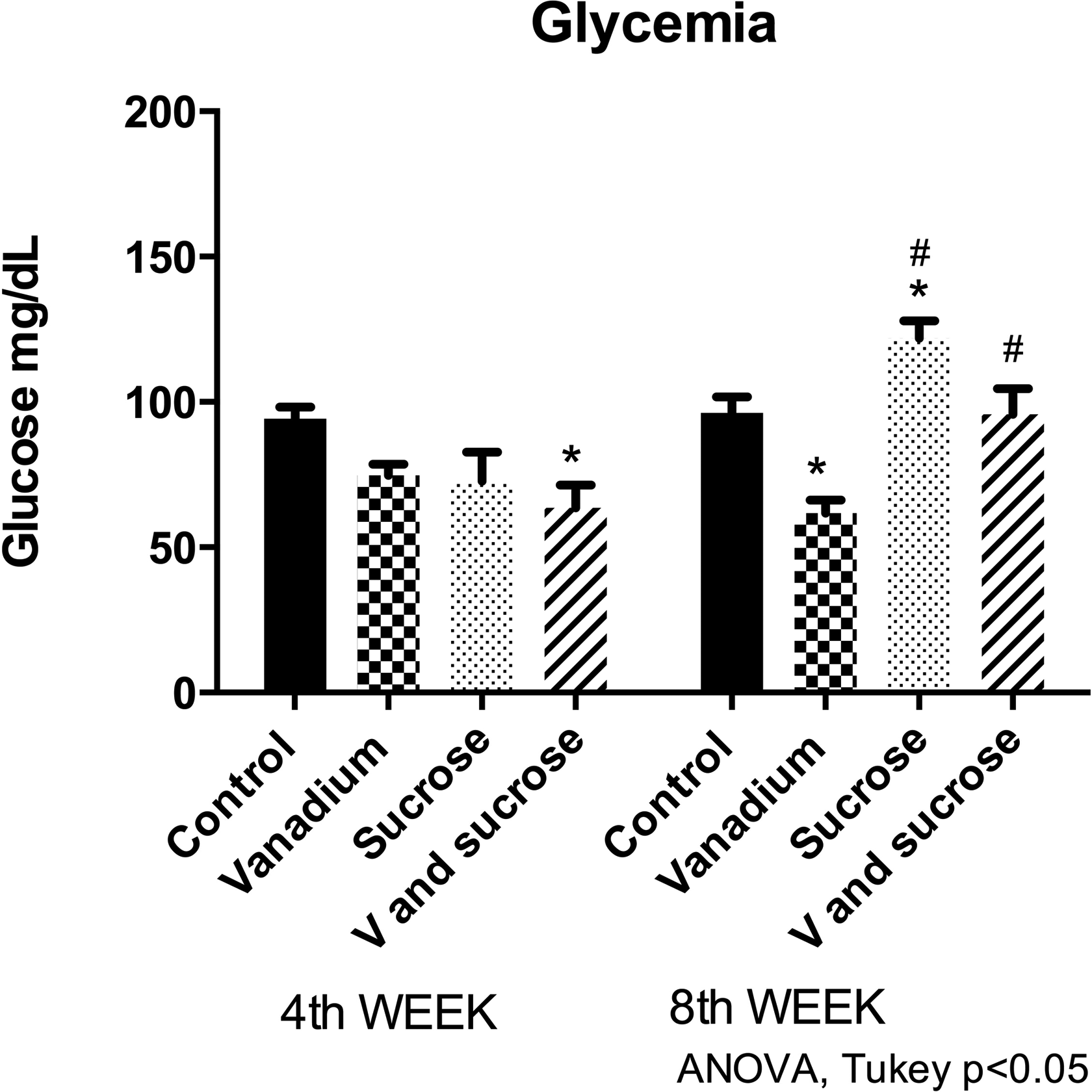
Vanadium + sucrose group had hypoglycemia at fourth week, as well as vanadium at eighth week compared to control. In contrast, sucrose group was hyperglycemic compared to control at eighth week. Mice n = 10 per group at fourth week and n = 5 per group at eighth week. *Differences between control. #Differences compared with vanadium group at eighth week.
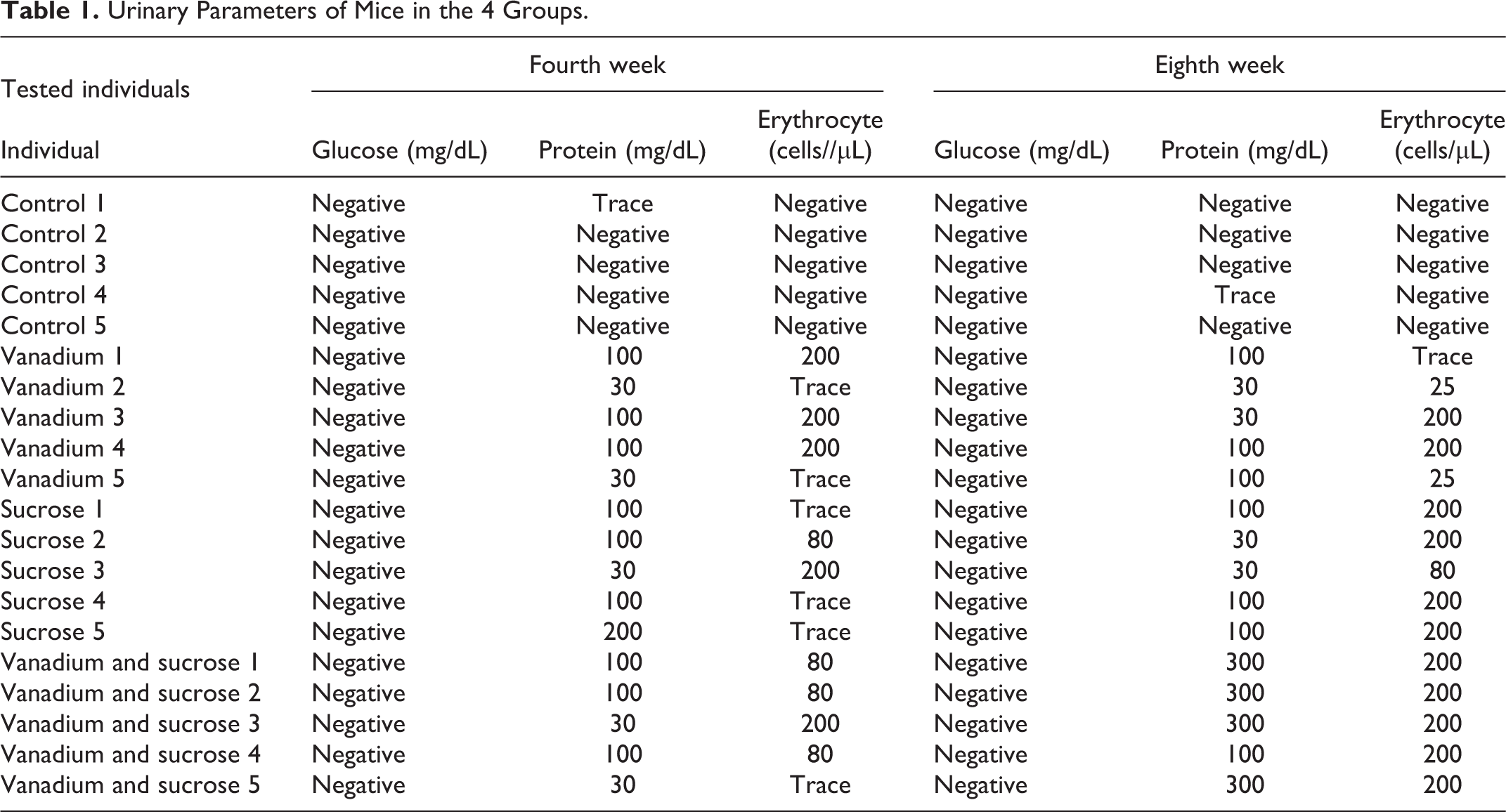
The urinary parameters in the control group were among normal values. Glycosuria was not found in any group in the fourth or eighth weeks. Vanadium, vanadium plus sucrose, and sucrose groups showed hematuria and proteinuria in the fourth and eighth weeks. Both parameters were more severe on the eighth week in vanadium plus sucrose group (Table 1).
Urinary Parameters of Mice in the 4 Groups.
Histopathological Findings
Inflammatory foci were observed in the kidneys of all experimental groups (vanadium, sucrose, vanadium plus sucrose). The foci were characterized by the presence of lymphocytes and plasma cells and were predominantly located around the blood vessels. The inflammatory foci were well defined in vanadium and sucrose groups, but in the vanadium plus sucrose group, the inflammatory cells were spread throughout the kidney. Some inflammatory foci around the blood vessels are marked with arrows in Figure 2.
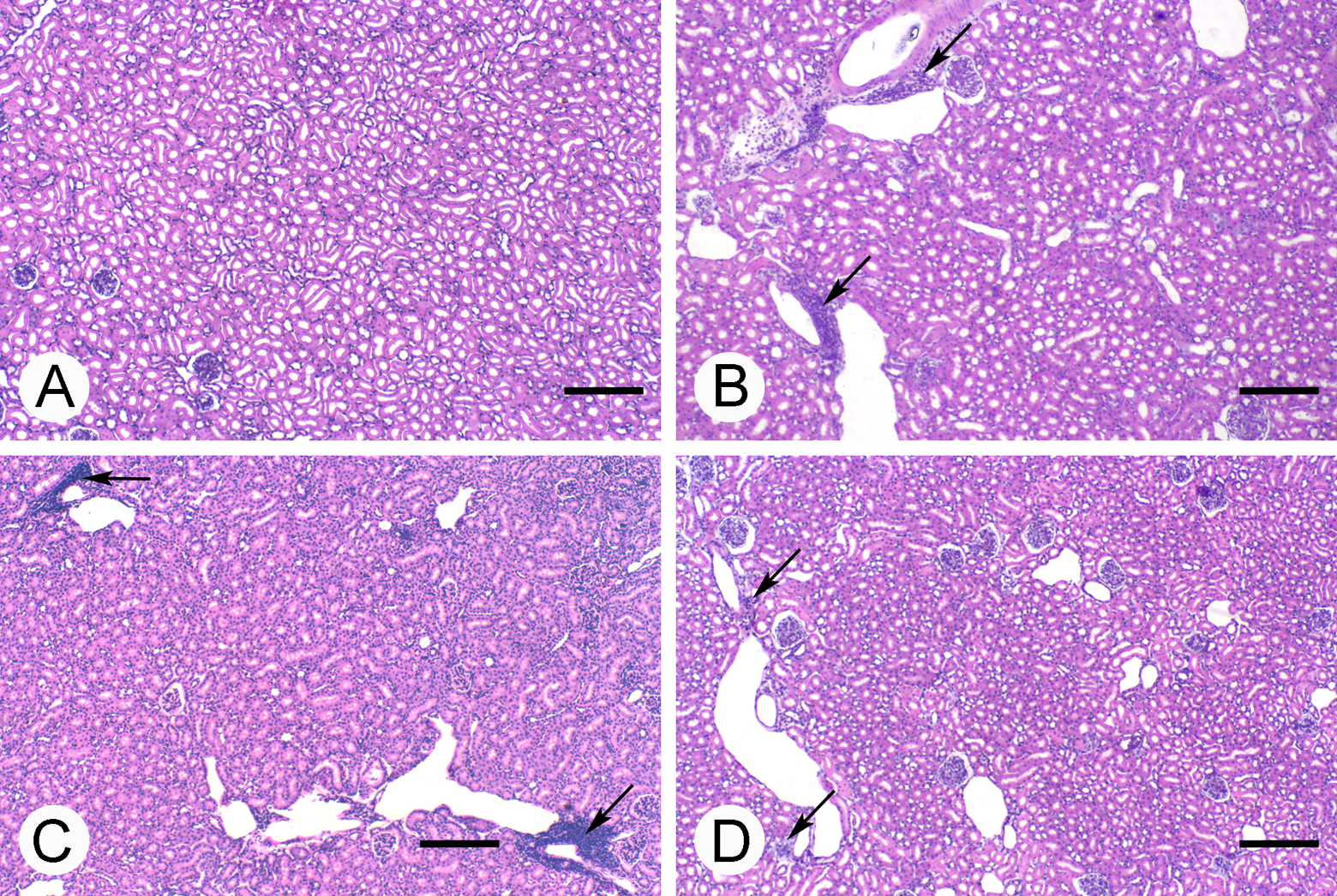
Histopathological changes in renal tissue of mice in the 4 groups. (A) Control without inflammation, (B) vanadium, (C) sucrose, (D) vanadium + sucrose. Arrows show inflammatory foci with lymphocyte infiltration. Scale bar: 200 μm.
Glomerular alterations were not observed, but tubular modifications were observed. Some tubular cells had cytoplasmic protrusions, and vacuoles and others were sloughed. Microvilli loss was widespread in all experimental groups, but in the eighth week, the tubules were almost denuded. An interesting finding was that in the sucrose and vanadium plus sucrose groups, hyperplastic urinary tubules’ epithelia with pyknotic nuclei were observed. In Figure 3, in the representative figure of the control group, tubules with normal microvilli in the epithelium were observed, in comparison to the loss of microvilli in the 3 experimental groups. The vanadium plus sucrose group was the one with the most significant loss of microvilli. The arrows in the vanadium or sucrose groups’ figures indicate vacuoles in tubular cells. The asterisk in vanadium and sucrose group indicates hyperplastic urinary epithelia in the tubules’ epithelia.
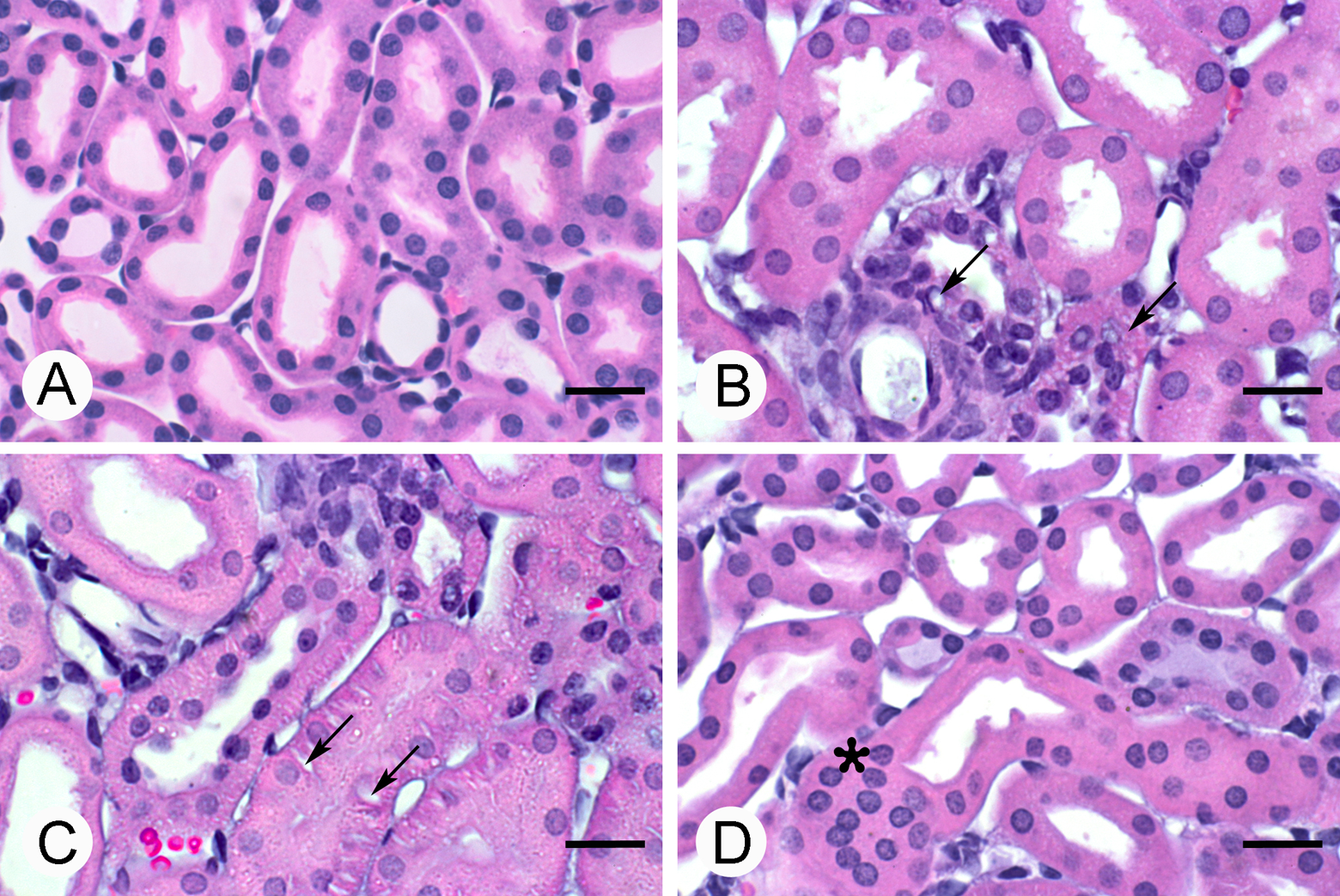
Histopathological changes in renal tubules of mice in the 4 groups. (A) Control. Tubules with normal microvilli are observed. (B) Vanadium. (C) Sucrose. Arrows show vacuolated cells. (D) Vanadium + sucrose. Asterisk shows urinary tubules with hyperplastic epithelia. Note the loss of microvilli in this group. Scale bar: 20 μm.
Oxidative Stress Evaluation (4-HNE Immunohistochemistry)
A densitometric analysis was performed in order to evaluate the metabolite 4-HNE as a marker of lipid peroxidation. The differences within the fourth week sucrose and the vanadium plus sucrose groups were statistically significant involving a higher presence of 4-HNE in comparison with control and vanadium groups. In the eighth week, the vanadium and the vanadium plus sucrose groups presented a significantly higher amount of 4-HNE in comparison with the control group. The vanadium plus sucrose group showed the highest 4-HNE concentration compared with the vanadium or sucrose groups (Figure 4).
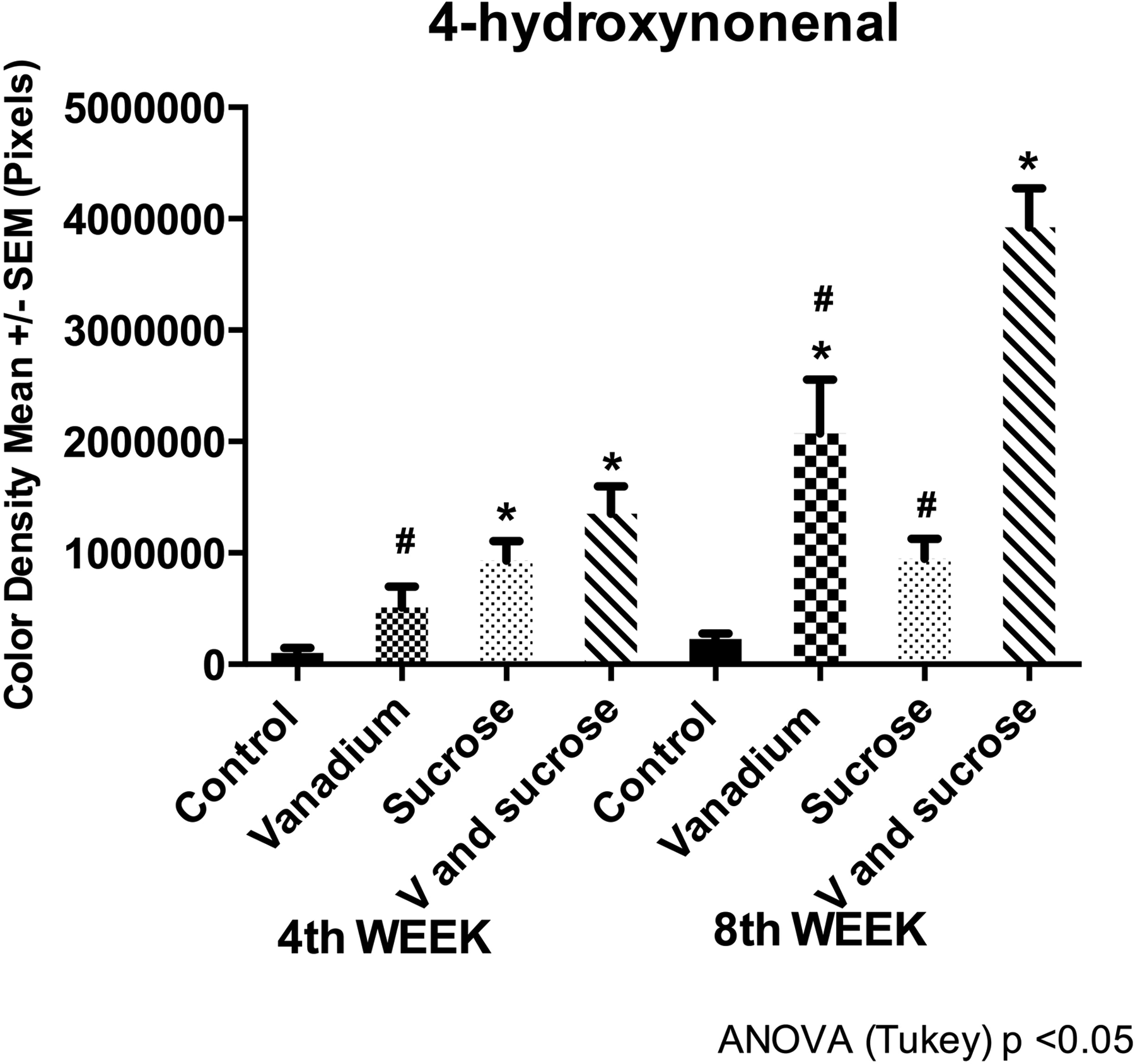
4-Hydroxynonenal densitometry (4-HNE). 4-Hydroxynonenal densitometry increased in sucrose group and vanadium + sucrose group compared to control at the fourth week and in vanadium group and vanadium + sucrose group at the eighth week. Mice n = 10 per group at fourth week and n = 5 per group at eighth week. *Differences compared with control group. #Differences between vanadium compared to vanadium + sucrose Group at the fourth week and differences between vanadium or sucrose group compared to vanadium + sucrose group at the eighth week.
The localization of 4-HNE in all experimental groups was mainly on the apical membrane of the tubular cells, particularly in the fourth week on the microvilli, except for the vanadium plus sucrose group that presented a mark on the membrane and the cells’ cytoplasm (Figure 5). In the eighth week, 4-HNE was localized in both the membrane and the cytoplasm in all experimental groups.
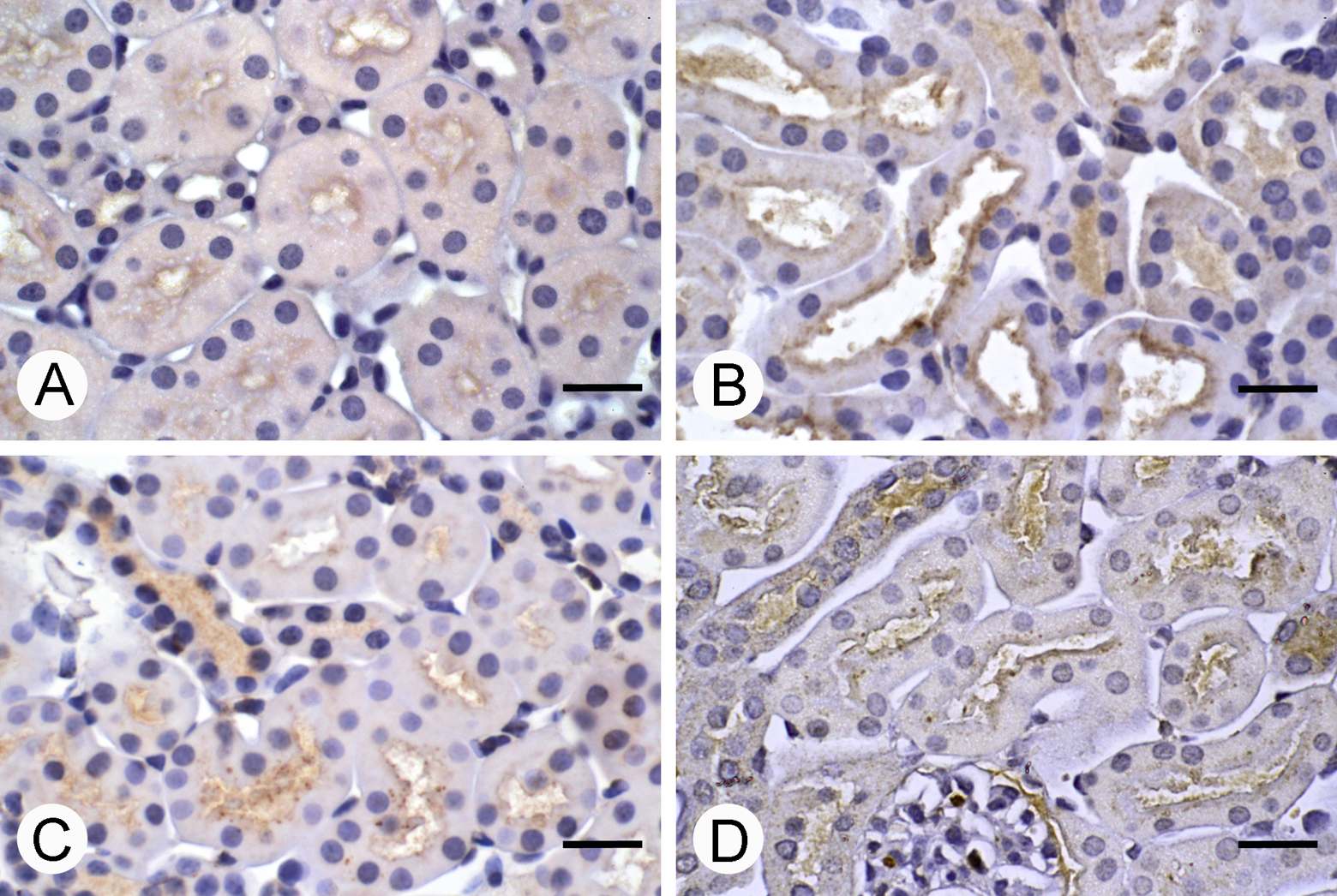
4-Hydroxynonenal (4-HNE) immunoreactivity in mice renal tissue. (A) Control; (B) vanadium; (C) sucrose; (D) vanadium + sucrose. The localization of 4-HNE is in microvilli of the tubular cells in all experimental groups; however, in the vanadium and sucrose group, the immunoreactivity is also observed in cytoplasm of the tubular cells. Scale bar: 20 μm.
Discussion
As a first approach for the understanding of the relationship between sugar-sweetened drinks and vanadium inhalation, we chose to study only male mice, but it is important for further studies to include female mice to explore sex differences. In this study, the vanadium concentration in the chamber is higher than in polluted cities, but smaller than the occupational exposure which reports up to 500 mg/m3 in cleaning boilers. 22
The effects of vanadium inhalation, oral sucrose, and its combination on the kidneys produce histological changes located mainly in the urinary tubules. The vanadium plus sucrose combination seems to potentiate the damage mediated by oxidative stress. Also, vanadium inhalation interferes with glucose regulation.
Hypoglycemia in the vanadium and the vanadium plus sucrose groups may be explained because of the insulin-mimetic effect this metal has, 23 - 26 but in this experiment, hypoglycemia was not a protective factor against kidney damage. Abundant evidence shows that the animal models of sucrose consumption develop hyperglycemia, which later may induce obesity and insulin resistance that lead to hyperglycemia or diabetes and may damage the kidneys. 27 –29
The effect of vanadium in the kidney is controversial. Some vanadium compounds are nephrotoxic, even in therapeutic doses, 11,12,30,31 but also some articles support that some vanadium compounds may improve kidney function because of its hypoglycemic potential. 23 Also, the nephrotoxic effect of sugar-sweetened beverages is controversial. 3,16,17,18,32 This study evaluated both factors alone and combined to provide more evidence about the toxicity of these factors.
In this study, we found histological evidence of kidney inflammation in all experimental groups. In those groups with vanadium exposure, the inflammation may be a consequence of the reactive oxygen species produced by the metal that leads to oxidative stress. 13,31,33,34 Inflammatory cells produce more reactive oxygen species, and these continue the cycle of generating more oxidative stress and inflammation. There is evidence that vanadium can induce cyclooxygenase to generate pro-inflammatory mediators. 35 On the other hand, sucrose also induces the generation of reactive oxygen species and oxidative stress because of glucose autoxidation or by production of advanced glycation products that act in their receptors (RAGE) inducing the signaling cascade that enhances the expression of pro-inflammatory genes that could worsen inflammation and oxidative stress. 36,37 In this study, the combination of vanadium plus sucrose worsened the effects.
Also, we found tubular cell damage with loss of microvilli, cytoplasmic protrusions, and sloughed cells since the fourth week. Tubular necrosis was evidenced by vacuolated cells and pycnotic nuclei, and in some fields, we found tubular cell hyperplasia in the vanadium plus sucrose group on the eighth week. In this study, we demonstrated that oxidative stress is a mechanism involved in this nephrotoxic effect since the biomarker for oxidative stress—4-HNE—increased in all experimental groups, with more severe effects in the vanadium plus sucrose group. Enough evidence exists to confirm that a common link between all forms of renal injury and renal diseases progression is the oxidative stress. 38
Another mechanism that may be involved is the damage of cytoskeleton proteins. Vanadium is an inhibitor of actin polymerization, as Rodríguez-Lara et al previously found in mice testicular cells. 39 This may explain in part the microvilli loss and the cytoplasmic protuberances in some cells. Vanadium and other metals also have a toxic effect on cell junctions, 40 –42 and this could explain why the cells are sloughed.
Histological alterations correlate with the functional changes found in the urine. Glycosuria was not found in any group; hence, hyperglycemia in the sucrose group did not exceed the glucose threshold limit in the tubules. In this experiment, proteinuria and hematuria were found in all experimental groups in comparison with the control group since week 4 and worsened in the eighth week; hematuria was more severe in the vanadium plus sucrose group at the end of the experiment and it correlates with oxidative stress. Tubular damage may explain hematuria and proteinuria. However, we did not find evidence of glomerular histological alteration. The basal membrane may be modified to explain proteinuria; hence, further studies are needed to analyze the integrity of the glomerular basal membrane and to explore another possible mechanism of toxicity involved in these conditions.
This study supports other experiments that have demonstrated the nephrotoxic effect of vanadium. This study used inhalation because it is the best route of vanadium absorption. The concentration of vanadium in the chamber in this study is higher than the concentration in polluted cities, but smaller than the one in occupational exposure, so in a short time we can study the toxic effects in different organs including the kidney. Also, this study provides evidence for sucrose nephrotoxicity, even in the absence of hyperglycemia. There are other studies in rats and mice that evaluate the effects of 30% sucrose solutions ad libitum, but they do not report the sucrose doses in g/kg body weight/day, making it difficult to compare with our study. 17,18,27 –29,43 In addition, we found that the sucrose groups had lower water consumption and dehydration may contribute to kidney damage. On the other hand, it seems that there is a different susceptibility to develop hyperglycemia, metabolic syndrome, and renal damage among strains.
Vanadium as an environmental pollutant is present in the atmosphere of big cities and it is not being considered as a risk factor for kidney damage. Maybe, the environmental pollution must be taken into account to explain the increase in renal diseases of unknown etiology and therefore lead to new measures to decrease the emission of vanadium and other metals. The population should be informed about the toxic effect of sugar-sweetened beverages to make better decisions about what to drink.
Footnotes
Acknowledgments
The authors are grateful to Armando Zepeda-Rodríguez and Francisco Pasos-Najera for the photographic image process, and Raquel Guerrero-Alquicira for their technical assistance. The authors also thank Enrique Pinzón Estrada, Ismael Torres Saldaña, and Victor Manuel Salgado Alfaro from the vivarium of Facultad de Medicina, Universidad Nacional Autónoma de México (UNAM), Mexico City, Mexico. The authors also thank Alejandra Núñez-Fortoul for editing the English presentation of the manuscript.
Author Contribution
M. Espinoza-Zurutuza contributed to conception and design, contributed to acquisition, and drafted the manuscript. A. Gonzalez-Villalva and J. C. Albarrán-Alonso contributed to conception and design, contributed to acquisition, and critically revised the manuscript. L. Colín-Barenque, P. Bizarro-Nevares, M. Rojas-Lemus, and N. López-Valdez contributed to acquisition, drafted the manuscript, and critically revised the manuscript. T. I. Fortoul contributed to conception and design, contributed to acquisition, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by grants PAPIIT-UNAM IN-211315.
