Abstract

Conclusion
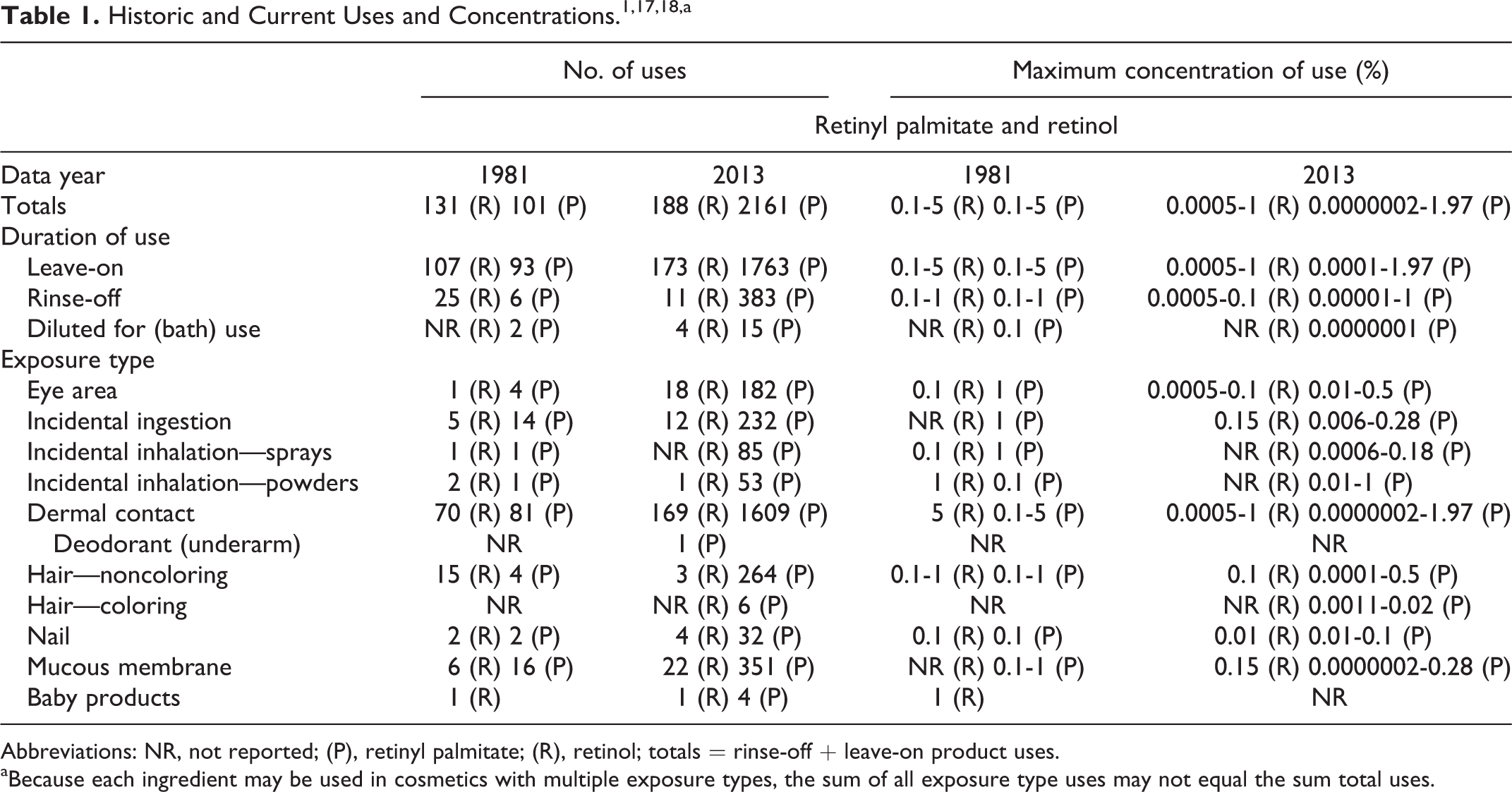
In a 1987 safety assessment of retinyl palmitate and retinol, the Cosmetic Ingredient Review Expert Panel stated that these ingredients are safe as cosmetic ingredients in the present practices of use and concentration. 1 The Panel reviewed newly available studies since that assessment along with updated information regarding types and concentration of use 2 –152 and did not reopen this safety assessment (Table 1). The Expert Panel confirmed that retinyl palmitate and retinol are safe as cosmetic ingredients in the present practices of use and concentration and recommended monitoring the progress of a new, ongoing National Toxicology Program (NTP) photococarcinogenesis study on retinyl palmitate and retinoic acid.
Abbreviations: NR, not reported; (P), retinyl palmitate; (R), retinol; totals = rinse-off + leave-on product uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Discussion
The Panel thoroughly reviewed a 2012 NTP photococarcinogenicity study on retinyl palmitate and retinoic acid, including an expert panel’s review of the study findings. The Panel noted the methodological flaws and, on that basis, determined that the findings could not be properly interpreted to suggest additional risks associated with these ingredients. A second NTP photococarcinogenesis study to address flaws in the original study may be considered when the new study is completed.
New toxicity data on retinol and retinyl palmitate and data on retinoic acid, retinyl acetate, and retinyl propionate that became available since the final safety assessment was issued were reviewed. The Panel recommended that data on residual levels of retinyl palmitate and retinol that remain in the epidermis following ingredient application in the presence of UV light be included when the second NTP study data are reviewed and that retinoic acid be removed from the report because it is a US Food and Drug Administration–approved drug.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.