Abstract
Epidemiologic studies have reported that particulate matter with aerodynamic diameters ≤2.5 μm (PM2.5) affect respiratory diseases, including asthma. The components and/or factors of PM2.5 that contribute to the exacerbation of asthma have not been identified. We investigated the effects of extracts of PM2.5 collected in Japan on the respiratory and immune systems. PM2.5 was collected from an industrial area and an urban area in December 2013. Airway epithelial cells and immune cells were exposed to aqueous or organic extracts of PM2.5. Exposure to extracts from both areas, especially to organic extracts rather than aqueous extracts, caused a pro-inflammatory response via interleukin (IL) 6 production from airway epithelial cells, and it induced the maturation/activation of bone marrow–derived antigen-presenting cells via dendritic and epithelial cell (DEC) 205 and cluster of differentiation (CD) 86 expression and proportional changes in the constitution of the splenocytes. The extracts collected from the industrial area tended to show greater effects than those from the urban area. These results suggest that organic components of PM2.5 affect the respiratory and immune systems. These effects can differ by the collection areas. In addition, IL-6, DEC205, and CD86 can be predictive biomarkers for the respiratory and immune effects of ambient PM2.5.
Introduction
Particulate matter with aerodynamic diameters ≤2.5 μm (PM2.5) is generated by anthropogenic activities such as the combustion of coal and other fossil fuels. Human exposure to components of PM2.5 occurs through inhalation, and the small size of PM2.5 allows it to penetrate deeply into the respiratory tract and reach the alveoli. In addition to the small size, some of the components of PM2.5 are known to contain hazardous substances, which can lead to the exacerbation of respiratory diseases such as asthma via the disruption of the respiratory and immune systems. 1
Particulate matter with aerodynamic diameters ≤2.5 μm is composed of solid and liquid materials that contain elemental carbons, organic carbons (OCs), inorganic salts, and metals. For example, diesel exhaust particles (DEPs) as a representative component of PM2.5 have a carbon core on/in which organic chemical components including polycyclic aromatic hydrocarbons (PAHs) and semi-volatile organic compounds, sulfate and nitrate ions, and metals are adsorbed. 2,3 It is not clear which of the many components and/or factors of PM2.5 play critical roles in the exacerbation of asthma and other respiratory diseases. Moreover, the components of PM2.5 can differ by the sources that generate PM2.5, the geographic area, and the seasons; moreover, secondary changes in PM2.5 can occur.
Airway epithelial cells are the most important target of inhaled PM2.5, because they are the first barriers to xenobiotics and are capable of releasing mediators (including cytokines) that are known to be associated with asthma. Damages such as injury or disruption of cell–cell junctions under nontoxic condition to airway epithelial cells and the pro-inflammatory response are key events in the invasion of inhaled xenobiotics (including allergens). After allergens enter the airways, antigen-presenting cells (APCs) capture the allergens and migrate to secondary lymphoid tissues such as local lymph nodes. Subsequently, maturated or activated APCs present the antigens to T cells, which induces the proliferation/activation of lymphocytes and subsequent immune reactions. 4 The pro-inflammatory responses from airway epithelial cells, the maturation/activation of APCs, and the proliferation/activation of lymphocytes play important roles in the possible mechanisms of the exacerbation of asthma. Accordingly, it is necessary to determine whether the components of PM2.5 affect these biological and immunological responses.
Organic chemicals other than the carbonaceous nuclei of DEPs have been reported to exaggerate allergic airway inflammation, possibly through the enhancement of T helper 2 (Th2) responses in vivo. 5 These results of a prior in vivo study are in good correlation with those of in vitro study using airway epithelial cells and immune cells. 6,7 Totlandsdal et al 6 reported that in airway epithelial cells, both native DEPs and methanol extracts of DEPs, but not residual particles of DEPs, induced marked messenger RNA (mRNA) expressions for interleukin (IL) 6 and IL-8. Koike and Kobayashi 7 showed that in immune cells, DEPs act on immature APCs and enhance the antigen-presenting activity, and they noted that the action contributing to oxidative stress may be mediated chiefly by the organic compounds rather than by the residual carbonaceous particles of DEPs. Alternatively, the use of airway epithelial cells and immune cells is appropriate to screen the effects of components of ambient PM2.5 on allergic airway inflammations such as asthma.
In this study, we investigated the effects of aqueous and organic extracts of PM2.5 on airway epithelial cells. We also examined the effects of extracts of PM2.5 on immune cells—especially bone marrow–derived APCs as the initiating cells of immune responses and splenocytes as the mixture of immune cells—from atopy-prone NC/Nga mice. Our purpose was to identify the components and/or factors of PM2.5 that contribute to respiratory and allergic effects on the basis of pathophysiological mechanisms.
Materials and Methods
Cell Cultures for the Evaluation of the Respiratory System
The BEAS-2B line of airway epithelial cells, derived from human bronchial epithelial cells transformed by an adenovirus 12-SV40 hybrid virus, was purchased from the European Collection of Cell Cultures (Salisbury, Wiltshire, United Kingdom). The airway epithelial cells were seeded in 96-well or 12-well collagen I–coated plates and incubated for 72 hours to reach semi-confluence in the serum-free medium LHC-9 (Life Technologies, Carlsbad, California) at 37°C in a humidified atmosphere of 5% CO2.
Animals and Preparation of Bone Marrow Cells and Splenocytes for the Immune System Evaluation
Ten-week-old male SPF NC/NgaTndCrlj mice were purchased from Charles River (Osaka, Japan). NC/Nga mice are atopy-prone mice that can be used as an animal model for human atopic dermatitis. It was reported that NC/Nga mice are highly susceptible to chemical-induced respiratory allergy. 8 Koike et al 9 noted that NC/Nga mice showed an accelerated differentiation of APCs in response to granulocyte–macrophage colony-stimulating factor (GM-CSF). The mice in the present study were housed in an animal facility that was maintained at 24°C to 26°C with a 12-hour light/dark cycle under conventional conditions. The procedures of all animal studies were approved by the Animal Research Committee at Kyoto University. Mice were sacrificed by cervical dislocation and exsanguinated from the cut abdominal aorta and vein. After the surrounding muscle tissue was removed, the bones were left in 70% ethanol for 3 minutes and washed with RPMI 1640 medium (Life Technologies). Both ends of the bones were cut, and then the marrow was flushed with RPMI 1640 using a syringe with a 24-gauge needle. The marrow suspension was passed through sterile 250-nylon mesh to remove small pieces of bone and debris, and the red blood cells were lysed with BD PharmLyse (Becton Dickinson, Lincoln Park, New York). The spleen was pushed through a sterile 200-mesh stainless steel sheet, and the red blood cells were also lysed with BD PharmLyse. The cells were centrifuged at 400g for 5 minutes at 20°C. After being washed with RPMI 1640, the cells were resuspended in culture medium R10, which is RPMI 1640 supplemented with 10% heat-inactivated fetal bovine serum (MP Biomedicals, Eschwege, Germany), 100 U/mL penicillin, 100 μg/mL streptomycin (Sigma, St Louis, Missouri), and 50 μM 2-mercaptoethanol (Life Technologies). The numbers of viable cells were determined by the trypan blue (Life Technologies) exclusion method.
Differentiation of APCs
Antigen-presenting cells were differentiated using a modification of the protocol provided by Lutz et al. 10 Bone marrow cells (4 × 105/mL) were cultured in R10 medium containing 20 ng/mL GM-CSF. On day 3, the same volume of the medium containing 20 ng/mL GM-CSF was added to the culture. On day 6, half the culture medium was replaced with fresh medium. On day 8, nonadherent and loosely adherent cells were collected. The differentiated APCs were centrifuged at 400g for 5 minutes at 20°C and then resuspended in fresh medium. The numbers of viable cells were determined by the trypan blue exclusion method.
Atmospheric Conditions
Daily various gaseous pollutants, including sulfur dioxide (SO2), nitric oxide (NO), nitrogen dioxide (NO2), nitrogen oxides (NOX), and photochemical oxidant (Ox), were obtained from the Japan Ministry of the Environment (http://www.env.go.jp/en/). Hourly data were recorded near locations where PM2.5 were collected. On the other hand, hourly data on the temperature were provided by the Japan Meteorological Agency (http://www.jma.go.jp/jma/indexe.html) and air pollution monitoring station. We calculated the daily average for each variable and then calculated the average of the collection period for use in this study.
Preparation of Components of PM2.5
Particulate matter with aerodynamic diameters ≤2.5 μm samples were continuously collected at an industrial area in Kawasaki City (Japan, 35° 31′ 44″ N, 139° 44′ 11″ E) and at an urban area in Fukuoka City (Japan, 33° 31′ 52″ N, 130° 28′ 59″ E; Figure 1). The collection was conducted using a high-volume air sampler (HV-700F; Sibata Scientific Technology, Saitama, Japan) equipped with a PM2.5 impactor (HVI-2.5; Tokyo Dylec, Tokyo) and one quartz fiber filter (Pallflex2500QAT-UP; Tokyo Dylec), for 4 to 5 days at a flow rate of 740 L/min, during December 2013 (Kawasaki: from December 5 to 9, Fukuoka: from December 6 to 9). The total volume of air sampled was determined from the measured volumetric flow rate and the sampling time. The mass concentration of PM2.5 in the ambient air was computed as the total mass of collected PM2.5 divided by the total volume of air sampled. The PM2.5 concentration is thus expressed as µg/m3 of air. 11
Map of the study area in Japan. Kawasaki city is located in an industrialized area on the largest, main island of Japan. Fukuoka city is located in an urban area in the southernmost region of the 4 main islands of Japan. The figure was edited from Google Map data 2015.
Water-soluble fractions were extracted from half-cut PM2.5-collected quartz fiber filters using sonication and distilled water (deionized and RNase free; Wako Pure Chemical Industries, Osaka, Japan) at 65°C. The aqueous crude extracts were passed through a laminated filter that consisted of 1- and 0.5-μm glass fiber filters and a 0.2-μm membrane filter.
Lipid-soluble fractions were extracted from the rest of the half-cut filters using Soxhlet and 300 mL of dichloromethane (dioxin analysis grade; Kanto Chemicals, Tokyo) for 16 hours. Both fractions were evaporated at 65°C and then set under a gentle stream of nitrogen gas flow until they were dry. Blank filters were also handled in the same manner.
The dried extracts were resuspended in water/dimethylsulfoxide (1:1, molecular biology grade; Wako) to make the aqueous extracts and the organic extracts at a final concentration of 75 mg/mL using the weight of PM2.5 collected on the filter and stored at 4°C in darkness until the bioassay.
The components of PM2.5 extracts were measured according to the manual of the Japan Ministry of the Environment. 12 Anion species (Cl−, NO3 −, and SO4 2−) and cation species (Na+, NH4+, K+, Mg2+, and Ca2+) were analyzed by ion chromatography. Elemental carbon and OC were measured using the interagency monitor of protected visual environments (IMPROVE) method. Metal was analyzed by microwave digestion and inductively coupled plasma mass spectrometry.
The sample collection, preparation, and measurement were performed by the Japan Environmental Sanitation Center (Kanagawa, Japan), and the extracts were sent via refrigerated cargo to Kyoto University for the assay.
Experimental Protocol
After the airway epithelial cells grew to semi-confluence in LHC-9 medium, they were exposed to aqueous or organic extracts of PM2.5 at the concentration of 0, 7.5, 22.5, or 75 μg/mL for 24 hours. We evaluated the cell viability and release of IL-6 by performing a water-soluble tetrazolium-1 (WST-1) assay and an enzyme-linked immunosorbent assay (ELISA), respectively.
Antigen-presenting cells from NC/Nga mice were exposed to aqueous or organic extracts of PM2.5 at the concentration of 0, 7.5, 22.5, or 75 μg/mL for 24 hours. The dendritic and epithelial cell (DEC) 205 and cluster of differentiation (CD) 86 protein expressions on the cell surface were evaluated by a fluorescence-activated cell sorter (FACS).
Splenocytes from the NC/Nga mice were exposed to aqueous or organic extracts of PM2.5 at the concentration of 0, 7.5, 22.5, or 75 μg/mL for 72 hours. We evaluated the cell proliferation by performing an ELISA for 5-bromo-2′-deoxyuridine (BrdU). The T cell receptor (TCR) and CD19 protein expressions on the cell surface were evaluated by the FACS. All control cells were treated by the blank filter extracts diluted with LHC-9.
Cell Viability
We measured the cell viability by WST-1 assay using the Premix WST-1 Cell Proliferation Assay System (TaKaRa Bio, Shiga, Japan). Water-soluble tetrazolium-1 reagent was added to each well of a 96-well plate and mixed well by gently rocking the plate. Airway epithelial cells were incubated with WST-1 reagent at 37°C for 3 hours. After the incubation, absorbance was measured on an iMark Microplate Absorbance Reader (Bio-Rad Laboratories, Hercules, California) with the wavelength set at 450 nm and a reference wavelength at 630 nm. The results are expressed as the percentage of viable cells compared to untreated cells (0 μg/mL).
Quantitation of Pro-Inflammatory Proteins in the Culture Supernatants
After exposure to extracts of PM2.5, the medium was harvested and centrifuged at 300g for 5 minutes to remove floating cells. The final supernatants were stored at −80°C until analysis. The levels of IL-6 (Thermo Scientific, Waltham, Massachusetts) in the supernatants were measured by ELISA, according to the manufacturer
Fluorescence-Activated Cell Sorter Analysis
For the FACS analysis, the following monoclonal antibodies were used: Mouse BD Fc Block purified anti-mouse CD16/CD32 (Becton Dickinson), DEC205 (NLDC-145, PE-conjugated; BioLegend, San Diego, California), Rat IgG2a, κ Isotype Control (RTK2758, PE-conjugated; BioLegend), CD86 (GL-1, PE-conjugated; Becton Dickinson), Rat IgG2a, κ Isotype Control (R35-95, PE-conjugated; Becton Dickinson), Hamster Anti-Mouse TCR-βChain (H57-597, FITC-conjugated; Becton Dickinson), Hamster IgG2, λ1 Isotype Control (Ha4/8, FITC-conjugated; Becton Dickinson), Rat Anti-Mouse CD19 (1D3, PE-conjugated; Becton Dickinson), and Rat IgG2a, κ Isotype Control (R35-95, PE-conjugated; Becton Dickinson).
After the exposure of the PM2.5 extracts, the cells were resuspended in 50 μL phosphate-buffered saline with 0.3% bovine serum albumin and 0.05% sodium azide (Wako) and then incubated with 0.05 to 1 μg of each antibody for 45 minutes at 4°C. After incubation, the cells were washed, and the fluorescence was measured by a FACSCalibur (Becton Dickinson). For each sample, fluorescence data from 10,000 cells were collected, and positive cells expressed as the percentage events or mean fluorescent intensity (MFI) were calculated.
Cell Proliferation
Cell proliferation was measured with a Cell-Proliferation-ELISA Kit (Roche Molecular Biochemicals, Mannheim, Germany), according to the manufacturer’s instructions. This technique is based on the incorporation of the pyrimidine analogue BrdU instead of thymidine into the DNA of proliferating cells. 5-Bromo-2′-deoxyuridine incorporated into DNA is measured by a sandwich-type enzyme immunoassay using monoclonal anti-BrdU antibodies. Splenocytes were exposed to extracts of PM2.5 for 72 hours, and cell proliferation was measured by adding BrdU to each well 20 hours before the measurement. Absorbance was measured on the iMark Microplate Absorbance Reader with the wavelength set at 450 nm and a reference wavelength at 630 nm.
Statistical Analysis
The data are presented as the mean ± standard error of the mean (SEM) for each experimental group (n = 4). The significance of variation among different groups was determined by 2-way analysis of variance. Differences among groups were analyzed using Tukey multiple comparison test (Excel Statistics 2012; Social Survey Research Information, Tokyo). A P-value <0.05 was considered to indicate a significant difference.
Results
The Characteristics of PM2.5 in Kawasaki and Fukuoka
The concentrations of gaseous pollutants and temperature in ambient air of Kawasaki tended to be higher than those in Fukuoka (Table 1). The mean concentration of PM2.5 in Kawasaki was 29.5 μg/m3 and that in Fukuoka was 30.5 μg/m3. The concentrations of PM2.5 at these industrial and urban areas were thus quite similar.
The Concentrations of Gaseous Pollutants and Temperature in Ambient Air.

Abbreviations: NO, nitric oxide; NO2, nitrogen dioxide; NOx, nitrogen oxides; Ox, photochemical oxidant; SO2, sulfur dioxide.
The percentages of mass concentration of organic extracts and aqueous extracts in that of PM2.5 from Kawasaki were 23.6% and 44.2%, respectively. The percentages of mass concentration of organic extracts and aqueous extracts in that of PM2.5 from Fukuoka were 16.1% and 68.8%, respectively.
The concentration of components of PM2.5 extracts showed that anion species such as NO3 − and SO4 2− were abundant components in aqueous extracts from Kawasaki and Fukuoka (Table 2). Organic carbon was the most abundant components in organic extracts from Kawasaki and Fukuoka. The concentrations of anion species and OC in extracts from Kawasaki were lower than those from Fukuoka.
The Concentration of Components of PM2.5 Extracts (ng/m3).a,b
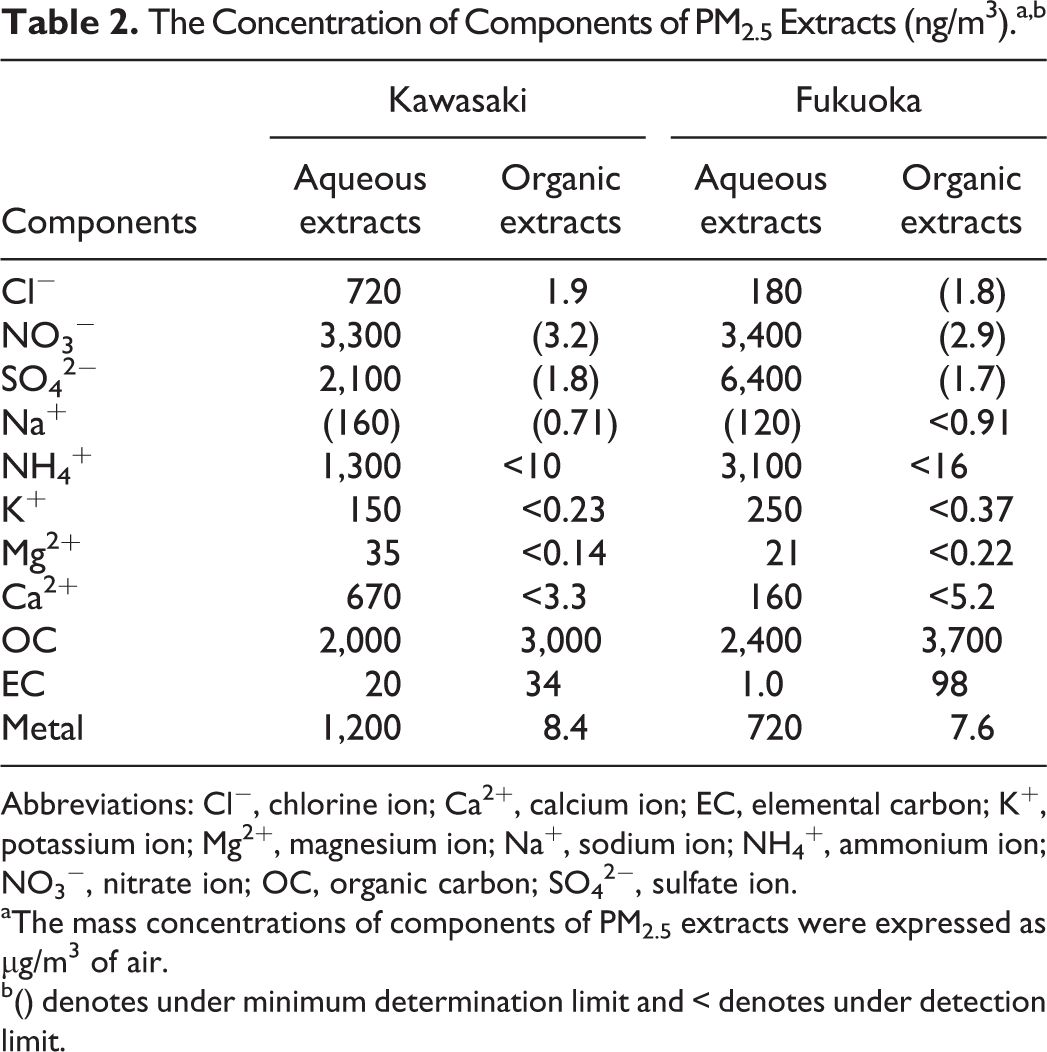
Abbreviations: Cl−, chlorine ion; Ca2+, calcium ion; EC, elemental carbon; K+, potassium ion; Mg2+, magnesium ion; Na+, sodium ion; NH4 +, ammonium ion; NO3 −, nitrate ion; OC, organic carbon; SO4 2−, sulfate ion.
aThe mass concentrations of components of PM2.5 extracts were expressed as µg/m3 of air.
b() denotes under minimum determination limit and < denotes under detection limit.
Effects of Aqueous and Organic Extracts of PM2.5 on the Viability of Airway Epithelial Cells
We investigated the effects of aqueous and organic extracts of PM2.5 on the viability of airway epithelial cells after exposure for 24 hours. Neither the aqueous extracts nor the organic extracts damaged the airway epithelial cells, but they induced an approximately 10% to 15% increase in cellular viability (Figure 2A and B).
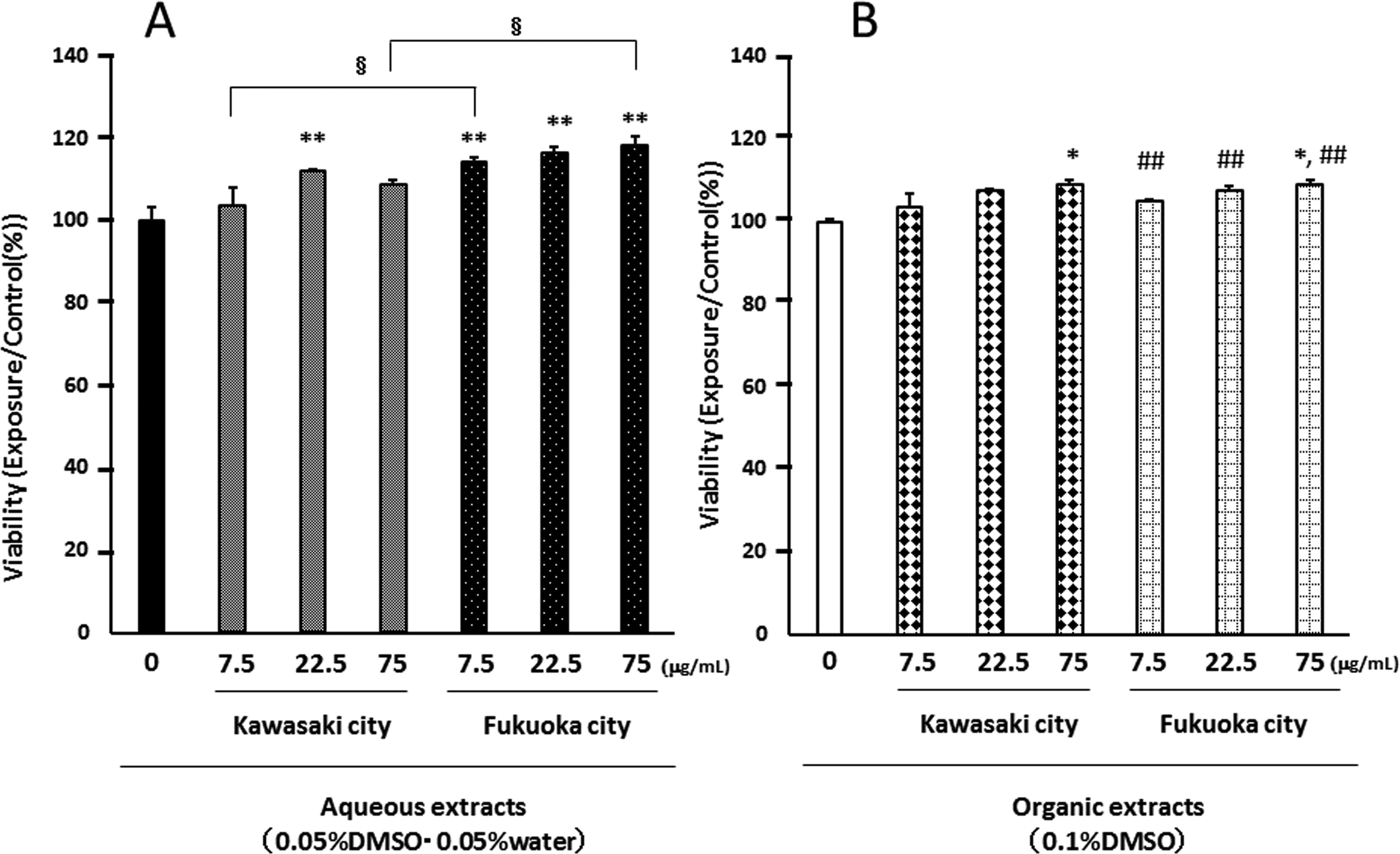
Effects of aqueous (A) and organic extracts (B) of PM2.5 on the viability of airway epithelial cells. Cell viability was assessed by WST-1 assays. Cells were treated with the indicated concentrations of PM2.5 for 24 hours. The data, presented as the percentage of the viability of the control, are the mean ± SEM of 4 individual cultures. *P < 0.05, **P < 0.01 versus 0 μg/mL, ## P < 0.01 versus aqueous extract-exposed group at the same concentration in the same city, § P < 0.05 versus each other. PM2.5 indicates particulate matter with aerodynamic diameters ≤2.5 μm; SEM, standard error of the mean; WST-1, water-soluble tetrazolium-1.
Effects of Aqueous and Organic Extracts of PM2.5 on the Production of Pro-Inflammatory Proteins From Airway Epithelial Cells
To evaluate whether the aqueous and organic extracts of PM2.5 had pro-inflammatory effects on airway epithelial cells, we examined the production of IL-6 after 24-hour exposure (Figure 3A and B). The aqueous extracts of PM2.5 from Kawasaki at the concentrations of 22.5 and 75 μg/mL and those from Fukuoka at the concentration of 7.5 μg/mL decreased the levels of IL-6 (Figure 3A). On the other hand, the organic extracts of PM2.5 from Kawasaki and Fukuoka concentration-dependently increased the protein release of IL-6 (Figure 3B). The levels of IL-6 released by the organic extracts from the Kawasaki (industrial) site were higher than those from the Fukuoka (urban) site.
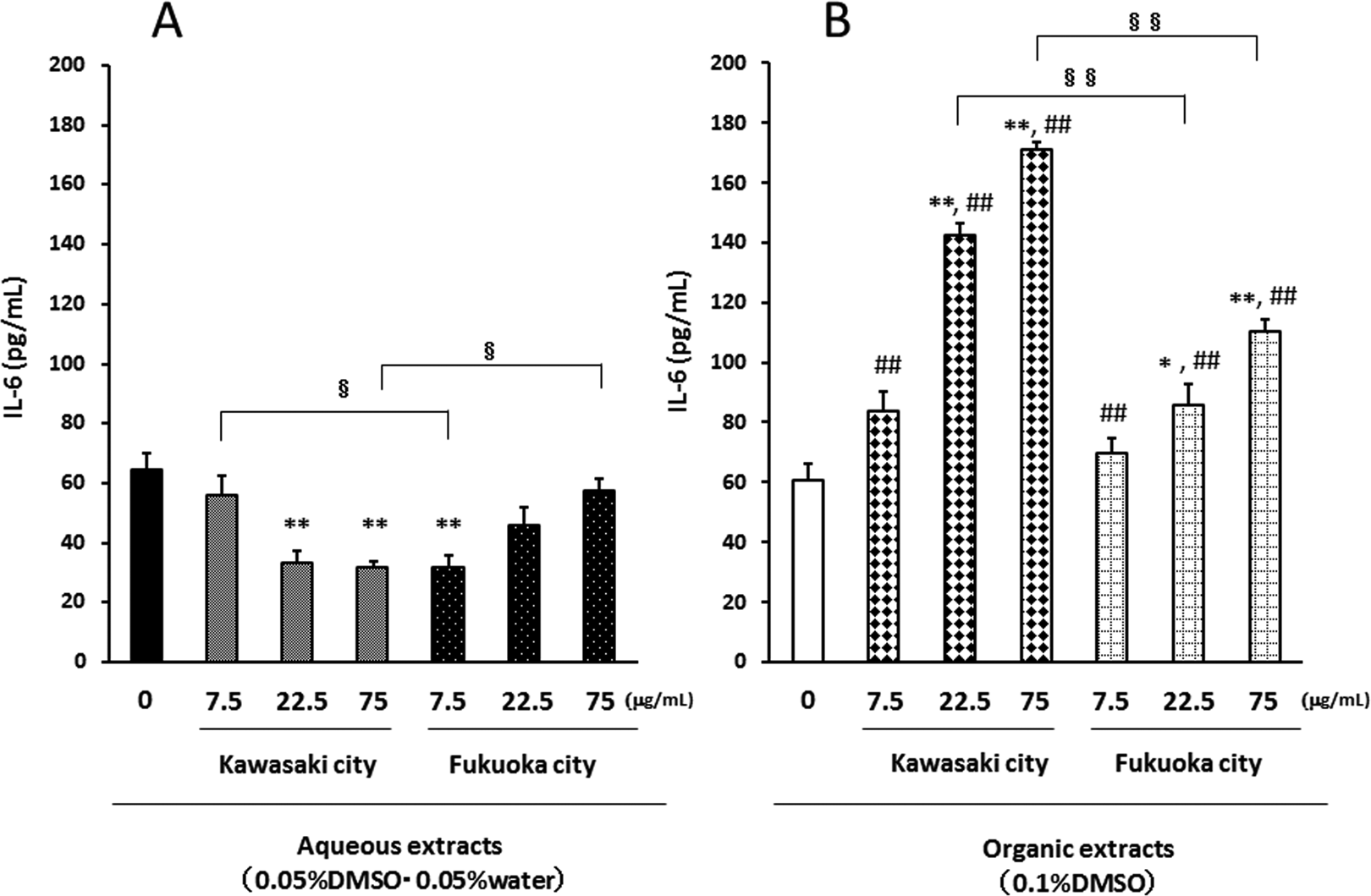
Interleukin 6 production from airway epithelial cells in response to aqueous extracts (A) and organic extracts (B). The levels in the culture supernatant were measured by ELISA. Data are mean ± SEM of 4 individual cultures. *P < 0.05, **P < 0.01 versus 0 μg/mL, ## P < 0.01 versus aqueous extract-exposed group at the same concentration in the same city, § P < 0.05, §§ P < 0.01 versus each other. ELISA indicates enzyme-linked immunosorbent assay; SEM, standard error of the mean.
Effects of the Aqueous and Organic Extracts of PM2.5 on the DEC205 and CD86 Expression of APCs
To determine the effects of the exposure to aqueous and organic extracts of PM2.5 for 24 hours on the maturation and activation of APCs, we analyzed the expression patterns of DEC205 and CD86 on APC (Figures 4 and 5). Neither the aqueous extracts of PM2.5 collected from Kawasaki nor those from Fukuoka affected the ratio of DEC205-positive cells. On the other hand, the organic extracts of PM2.5 from the Kawasaki site and those from Fukuoka concentration-dependently increased the ratio of DEC205-positive cells. In a comparison of the 2 collection areas, the organic extracts of PM2.5 from the Kawasaki (industrial) site had a stronger effect than those from the Fukuoka (urban) site.
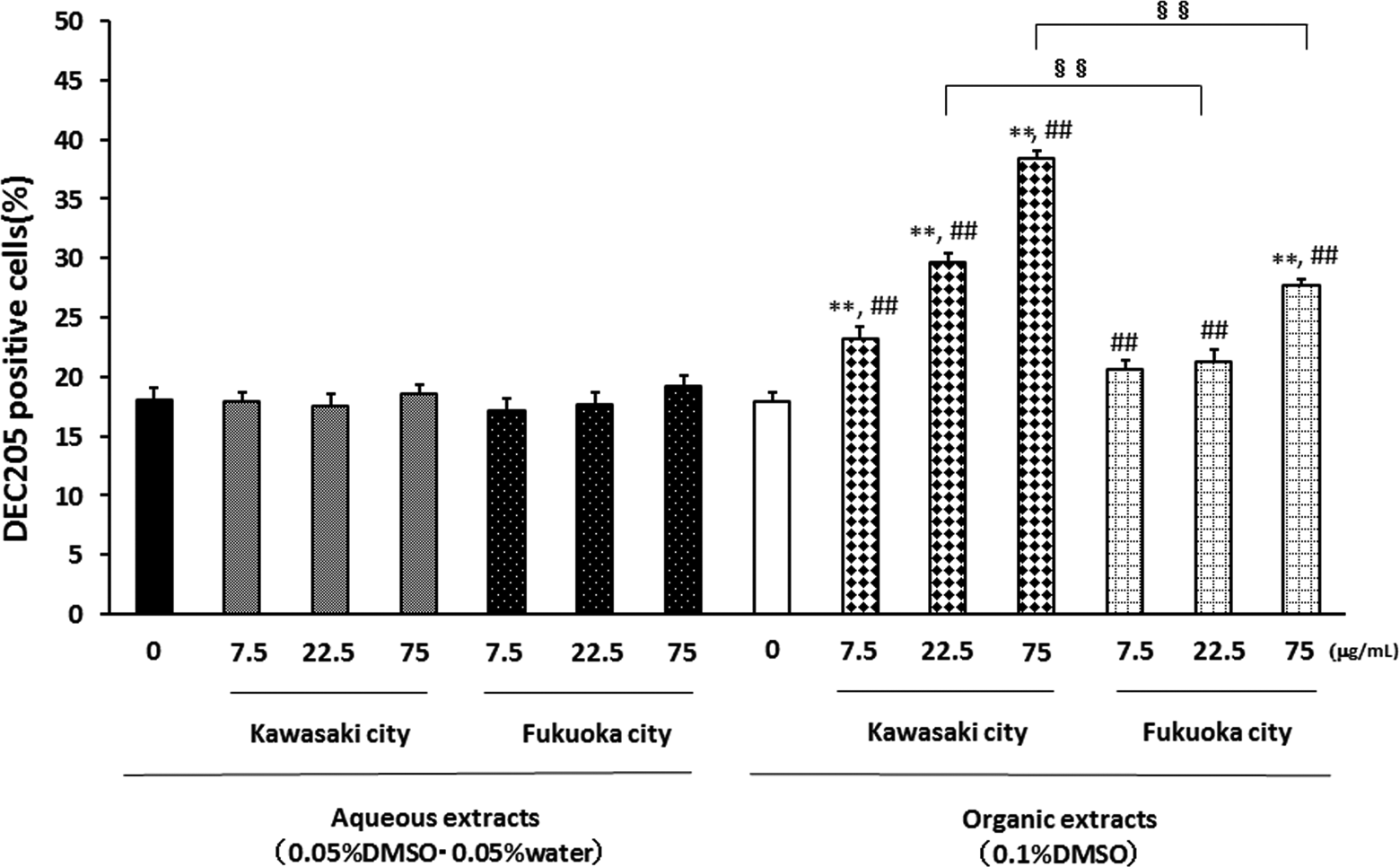
Effects of aqueous and organic extracts of PM2.5 on the DEC205 expression of APCs. Antigen-presenting cells from NC/Nga mice were treated with the indicated concentrations of PM2.5 for 24 hours and then analyzed by FACS. The data, presented as positive cells expressed as the percentage events, are the mean ± SEM of 4 individual cultures. **P < 0.01 versus 0 μg/mL, ## P < 0.01 versus aqueous extract-exposed group at the same concentration in the same city, §§ P < 0.01 versus each other. APC indicates antigen-presenting cells; DEC, dendritic and epithelial cell; FACS, fluorescence-activated cell sorter; PM2.5, particulate matter with aerodynamic diameters ≤2.5 μm; SEM, standard error of the mean.
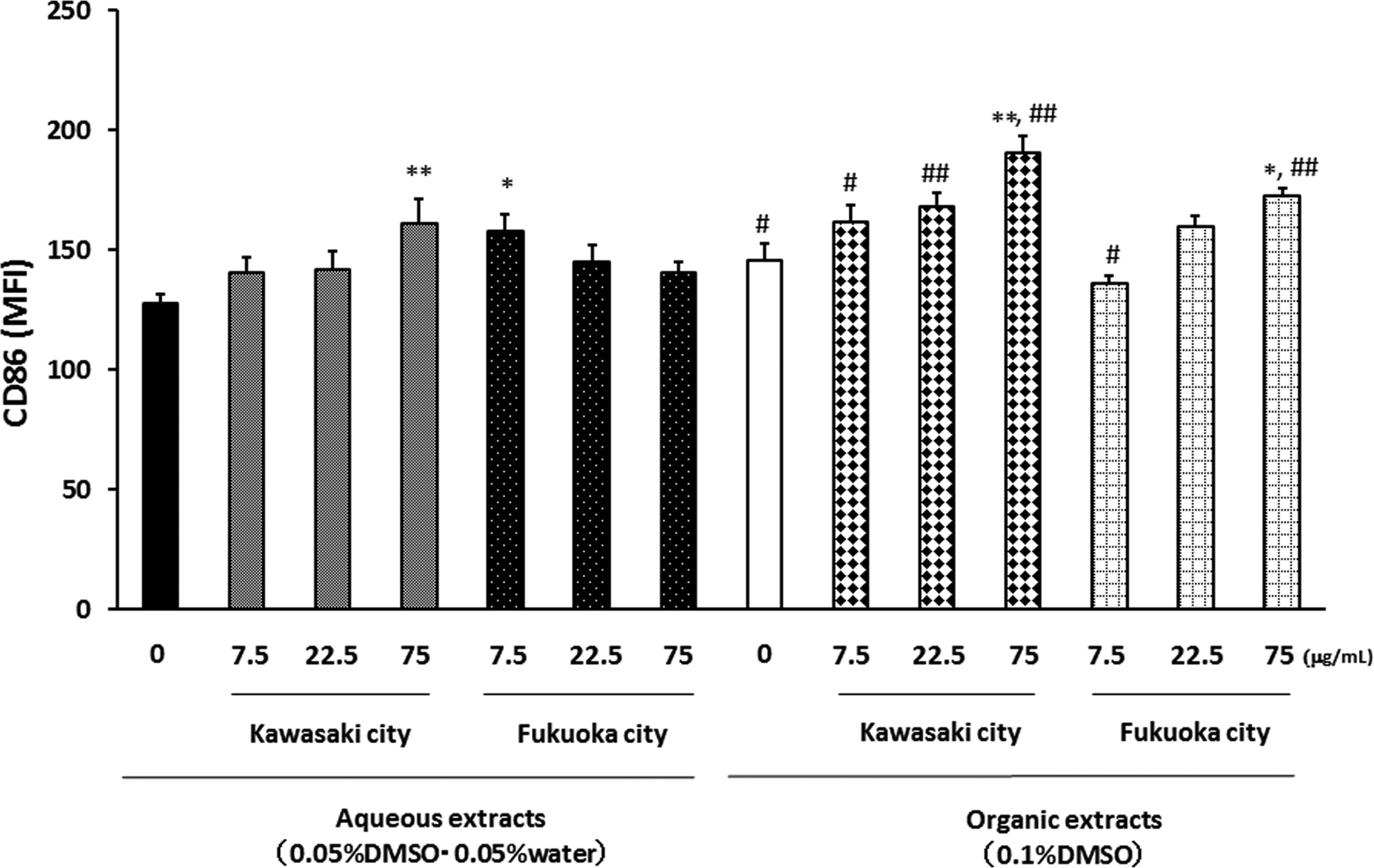
Effects of aqueous and organic extracts of PM2.5 on the CD86 expression of APCs. Antigen-presenting cells from NC/Nga mice were treated with the indicated concentrations of PM2.5 for 24 hours and then analyzed by FACS. The data, presented as MFI values, are the mean ± SEM of 4 individual cultures. *P < 0.05, **P < 0.01 versus 0 μg/mL, # P < 0.05, ## P < 0.01 versus aqueous extract-exposed group at the same concentration in the same city. APC indicates antigen-presenting cells; CD, cluster of differentiation; FACS, fluorescence-activated cell sorter; MFI, mean fluorescent intensity; PM2.5, particulate matter with aerodynamic diameters ≤2.5 μm; SEM, standard error of the mean.
The organic extracts from both areas at the concentrations of 75 μg/mL increased the CD86 MFI (Figure 5). The aqueous extracts of PM2.5 from the Kawasaki site at the concentration 75 μg/mL and those from the Fukuoka site at the concentration 7.5 μg/mL slightly increased the CD86 MFI.
Effects of the Aqueous and Organic Extracts of PM2.5 on the Proliferation of Splenocytes
We also examined the proliferation of splenocytes after exposure to aqueous or organic extracts of PM2.5 for 72 hours. The aqueous extracts from Kawasaki and those from Fukuoka had no effect on the proliferation of splenocytes, but both the organic extracts from Kawasaki and those from Fukuoka at the concentration 75 μg/mL decreased the proliferation (Figure 6).
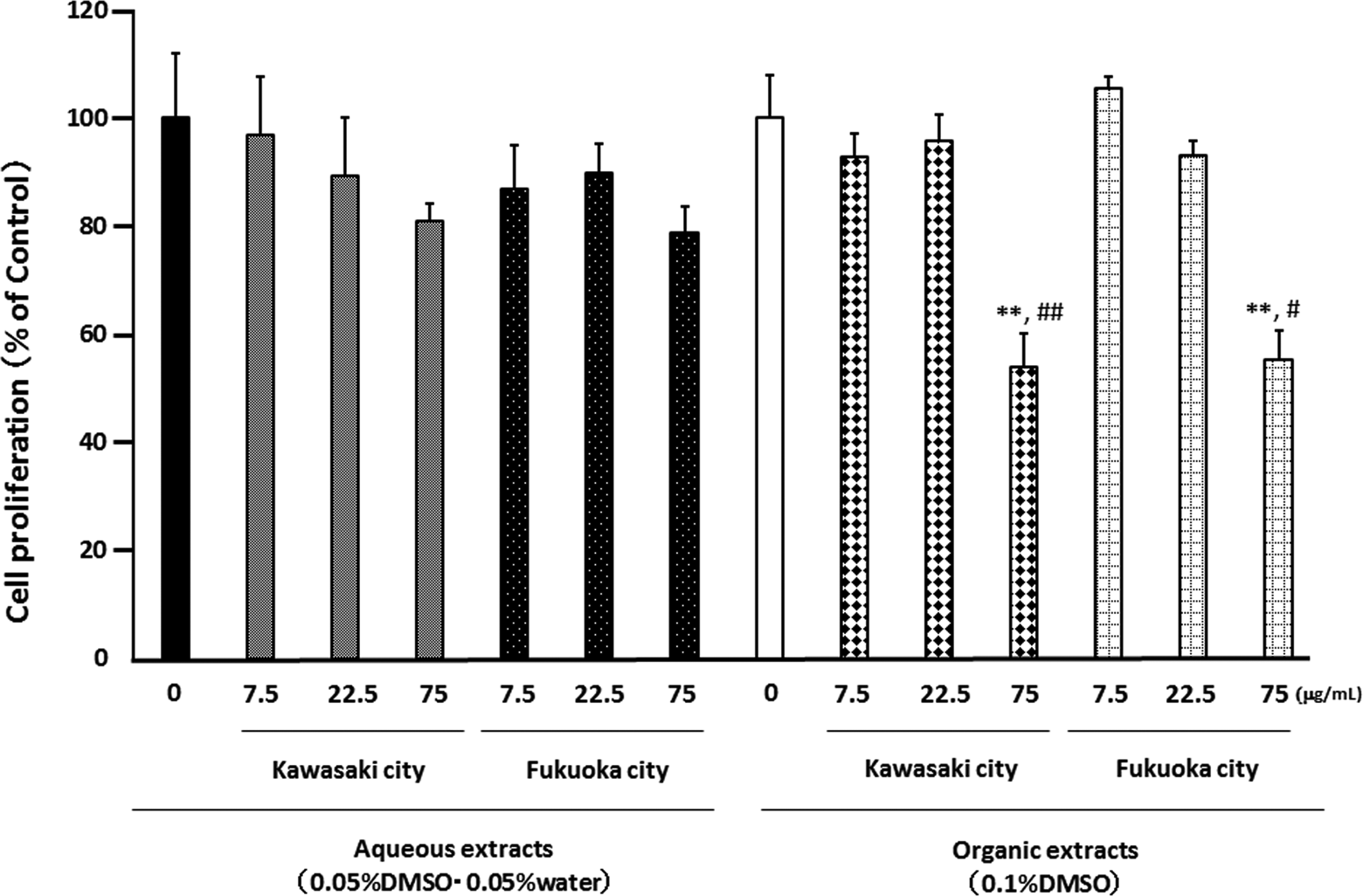
Effect of aqueous and organic extracts of PM2.5 on the proliferation of splenocytes from NC/Nga mice. Cells were treated with the indicated concentrations of PM2.5 for 72 hours. The data, presented as the percentage of the proliferation of the control, are the mean ± SEM of 4 individual cultures. **P < 0.01 versus 0 μg/mL, # P < 0.05, ## P < 0.01 versus aqueous extract-exposed group at the same concentration in the same city. PM2.5 indicates particulate matter with aerodynamic diameters ≤2.5 μm; SEM, standard error of the mean.
Effects of the Aqueous and Organic Extracts of PM2.5 on the TCR and CD19 Expressions of Splenocytes
To determine the effects of exposure to aqueous or organic extracts of PM2.5 for 72 hours on proportional changes in the constitution of the splenocytes, we analyzed the expression patterns of TCR and CD19 on splenocytes (Figures 7 and 8). The aqueous extracts from Kawasaki did not affect the ratio of TCR-positive cells. The aqueous extracts from Fukuoka at the concentration 75 μg/mL slightly elevated the ratio of TCR-positive cells, whereas the organic extracts from both sites concentration-dependently increased the ratio of TCR-positive cells (Figure 7).
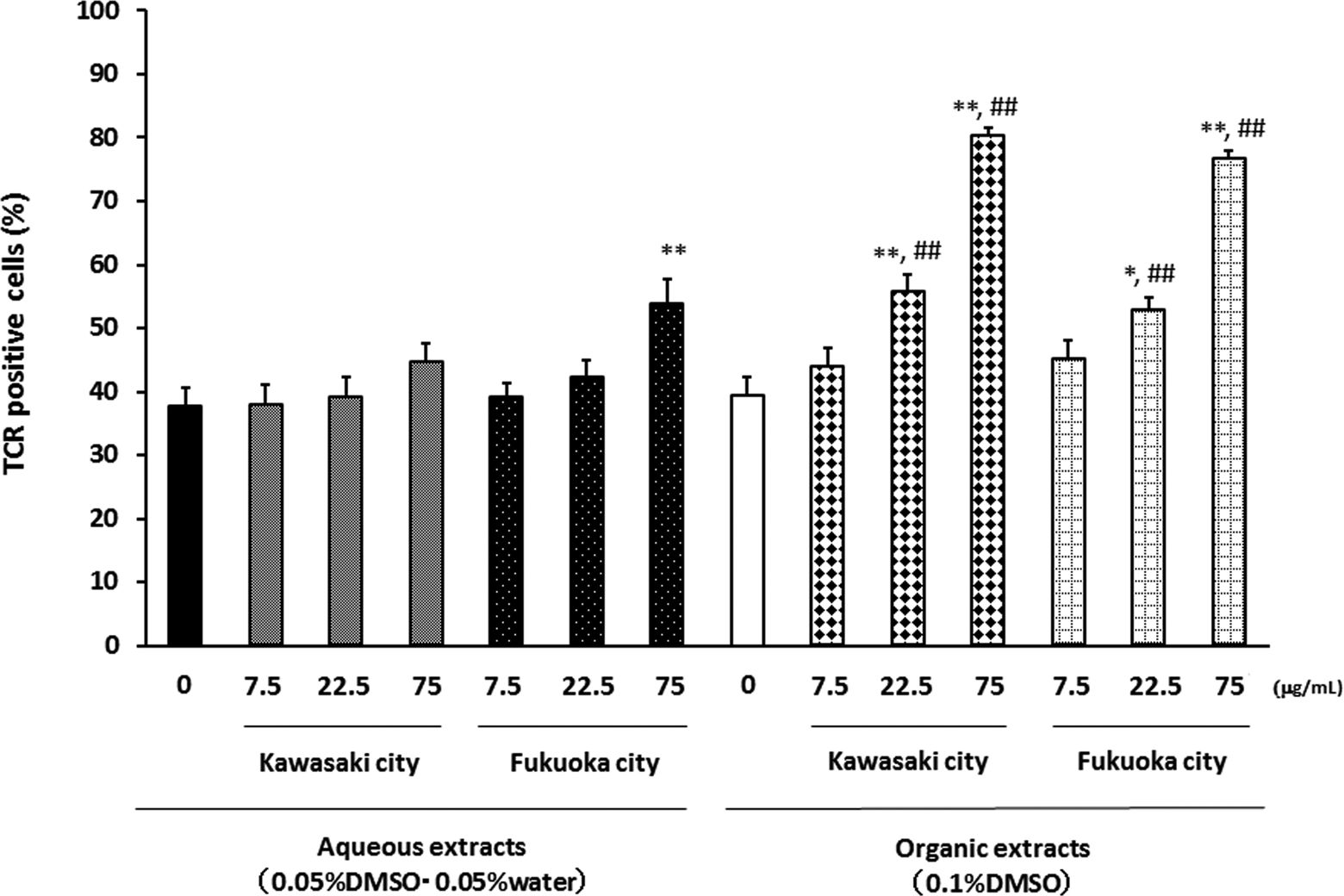
Effects of aqueous and organic extracts of PM2.5 on the TCR expression of splenocytes. Splenocytes from NC/Nga mice were treated with the indicated concentrations of PM2.5 for 72 hours and then analyzed by FACS. The data, presented as positive cells expressed as the percentage events, are the mean ± SEM of 4 individual cultures. *P < 0.05,**P < 0.01 versus 0 μg/mL, ## P < 0.01 versus aqueous extract-exposed group at the same concentration in the same city. FACS indicates fluorescence-activated cell sorter; PM2.5, particulate matter with aerodynamic diameters ≤2.5 μm; SEM, standard error of the mean.
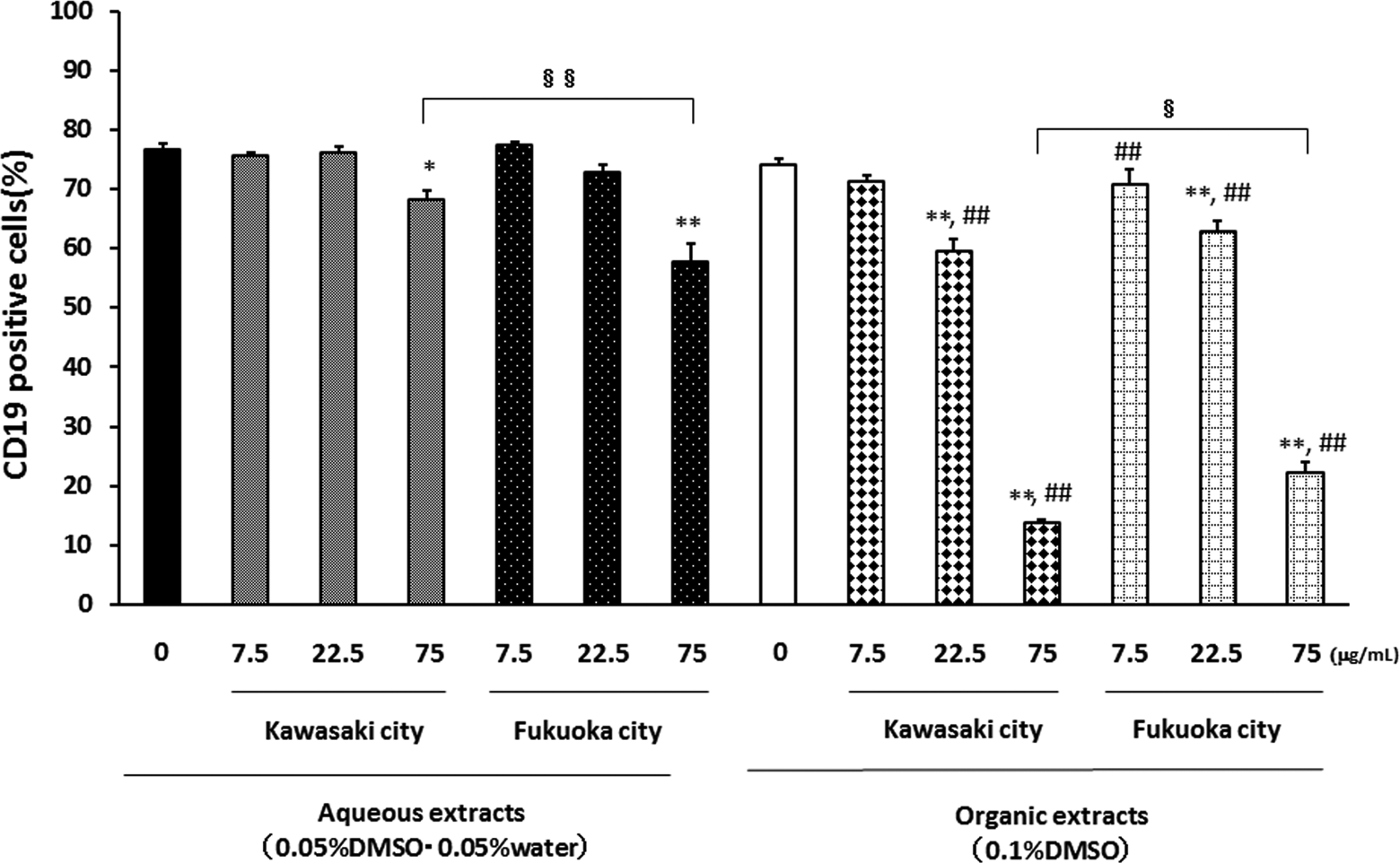
Effects of aqueous and organic extracts of PM2.5 on CD19 expression of splenocytes. Splenocytes from NC/Nga mice were treated with the indicated concentrations of PM2.5 for 72 hours and were analyzed by FACS. Data are presented as positive cells expressed as the percentage events. Data are mean ± SEM of 4 individual cultures. *P < 0.05, **P < 0.01 versus 0 μg/mL, ## P < 0.01 versus aqueous extract-exposed group at the same concentration in the same city, § P < 0.05, §§ P < 0.01 versus each other. CD indicates cluster of differentiation; FACS, fluorescence-activated cell sorter; PM2.5, particulate matter with aerodynamic diameters ≤2.5 μm; SEM, standard error of the mean.
In contrast, the aqueous extracts from Fukuoka rather than Kawasaki at the concentration 75 μg/mL decreased the ratio of CD19-positive cells. The organic extracts from both the Kawasaki and Fukuoka sites concentration-dependently decreased the ratio of CD19-positive cells. The organic extracts from Kawasaki had greater effects on CD19 than those from Fukuoka. Generally, the effects of the organic extracts of PM2.5 on the expressions of TCR and CD19 were greater than those of the aqueous extracts.
Discussion
We observed that exposure to PM2.5 extracts, especially to organic extracts rather than aqueous extracts, caused a pro-inflammatory response via IL-6 production from airway epithelial cells and induced both the maturation/activation of APCs via DEC205 and CD86 and proportional changes in the constitution of the splenocytes. Moreover, the extracts from the Kawasaki site (an industrial area) tended to show greater effects than those from the Fukuoka site (an urban area).
Airway inflammation is central to the pathogenesis of asthma. We observed that organic components rather than aqueous components from both sites caused a stronger pro-inflammatory response via IL-6 from the airway epithelial cells under nontoxic conditions. Airway epithelial cells—which play important roles in the physical barrier and biological and immunological responses against inhaled xenobiotics—are a source of cytokines such as IL-6. IL-6 is a pro-inflammatory cytokine induced by a response to environmental insults; it plays important roles in acute inflammation in the respiratory system by stimulating lymphocytes. 13 Increased levels of IL-6 in serum have been found in patients with asthma. 14 Various chemicals and allergens, including DEPs, PAHs, metals, and dust, might stimulate IL-6 production from airway epithelial cells. 6,15 -17
In accord with our present findings, it has been reported that PM2.5 induces a marked mRNA expression and protein release of IL-6 from airway epithelial cells under toxic or nontoxic conditions. 18,19 In addition, organic extracts of PM2.5 have stimulated oxidative stress and induced the release of IL-6 and IL-8 from airway epithelial cells. 20 The generation of reactive oxygen species (ROS) is a critical event to induce respiratory inflammation and dysfunction. The organic extracts of PM2.5 contain various organic chemicals such as PAHs. Among the PAHs, benzo[a]pyrene (BaP) and 1-nitropyrene have been reported to generate ROS in airway epithelial cells. 21,22 The quinone derivatives such as naphthoquinone and phenanthrenequinone (PQ) also have the ability to induce a strong generation of ROS. 23,24
As combined effects, Peters et al 25 suggested that coexposure to 9,10-PQ and copper (Cu) resulted in an increase in redox cycling and the production of ROS. Metals can exist in not only aqueous extracts of PM2.5 but also organic extracts. Interestingly, it has been reported that heavy metals in organic extracts of PM2.5 are responsible for redox activity and that the IL-6 and IL-8 release caused by organic extracts was reduced in the presence of a metal chelator in airway epithelial cells. 20 Therefore, PAHs themselves or a combination of PAHs and metals such as Fe and Cu in organic extracts may contribute to pro-inflammatory responses via ROS and thus to the exacerbation of respiratory diseases.
Few experimental studies have examined the effect of ambient PM2.5 on immune cells. Our present findings indicate that the organic components increased the surface expression of molecules related to the maturation and activation of APCs. DEC205 is a member of the macrophage mannose receptor family. This molecule is known to mediate the capture and internalization of ligands for subsequent processing and presentation by APCs. 26 It has been reported that carbon black nanoparticles, Asian sand dust particles, and di-(2-ethylhexyl) phthalate can promote the maturation/activation and function of DEC205 on APCs, which could be related to their enhancing effects on allergic diseases or responses. 27 -29 To our knowledge, the present study is the first to reveal that the components of ambient PM2.5 activated APCs via DEC205 expression.
CD86 on APCs is expressed at high levels by activation, and CD86 is the ligand for CD28 on the cell surface of T cells. The binding of CD86 with CD28 is a costimulatory signal for the activation of T cells. 30 The results of the present investigation using atopy-prone mice also indicated that organic extracts increase the CD86 expression of APCs. It has been reported that ambient PM2.5 exposure upregulates the expression of CD86 on circulating monocytes in diabetic individuals 31 and that organic extracts of DEPs increase the expression of CD86 on rat peripheral blood monocytes. 7 Among the components of PM2.5, BaP in the presence of rat liver S9 homogenate has induced CD86 expression on APCs. 32 Particulate matter with aerodynamic diameters ≤2.5 μm and organic components of PM2.5 can enhance the antigen-presenting ability of APCs. Accordingly, PM2.5 may have the ability to activate the capture, internalization, and presentation of xenobiotics (including allergens) in APCs.
Our study showed that organic extracts of PM2.5 inhibited the proliferation of splenocytes and caused proportional changes in the constitution of the splenocytes. Previous studies indicated that PAHs such as BaP directly affect T cells and B cells and decrease mitogen-induced proliferation. In addition, the growth inhibition may be mediated by an arylhydrocarbon receptor or cytochrome P450. 33,34 In the present study, there was a negative correlation between the TCR- and CD19-positive cells, which indicated that B cells had higher sensitivity against extracts of PM2.5 compared to T cells. This difference in sensitivity might be associated with the metabolism of PAHs. Finally, proportional changes in the constitution cells, that is, increased T cells and decreased B cells, might contribute to the disruption of the immune system.
In the present study, the effects of the extracts from the city of Kawasaki (in an industrial area) tended to show greater effects than those from the city of Fukuoka (in an urban area), although the concentrations of PM2.5 at the 2 sites were almost the same and the concentrations of anion species, especially SO4 2− and OC in extracts from Kawasaki, were lower than those from Fukuoka. Previous studies also examined the effects of PM2.5 and its components collected in various areas on the respiratory and immune systems. In their in vivo study, Wagner et al 19 suggested that exposure to concentrated air particles collected in Detroit, Michigan, enhanced allergic inflammatory end points such as eosinophils in bronchoalveolar lavage fluid and mucus in airways, whereas the inhalation of concentrated air particles collected in Grand Rapids, Michigan, suppressed all the allergic responses.
As for in vitro studies, it has been reported that PM2.5 collected in Benin induced a marked mRNA expression or protein release of IL-6 and IL-8 from airway epithelial cells. 18 PM2.5 collected in Mexico and the organic extracts of PM2.5 from Puerto Rico induced IL-6, whereas they decreased IL-8. 35,36 In contrast, aqueous extracts of PM2.5 collected in Brazil decreased the release of IL-6 and IL-8 from airway epithelial cells. 37 Accordingly, the results of the present and previous studies examining the effects of PM2.5 on the respiratory and immune systems can vary by the sources and collection areas. The difference in the components of PM2.5 can also contribute to the differences in respiratory and immune effects. Further investigations are needed to understand the main components that contribute to the respiratory and immune systems.
Exposure to PM2.5 extracts, especially to organic extracts rather than aqueous extracts, caused a pro-inflammatory response in airway epithelial cells and induced the maturation/activation of APCs and proportional changes in the constitution of the splenocytes. Moreover, the extracts from the industrial area tended to show greater effects than those from the urban area. These results suggest that predominantly organic components of PM2.5 affect the respiratory and immune systems. Those effects can differ by the collection areas. In addition, IL-6 from airway epithelial cells and DEC205 and CD86 expressions on APCs can be predictive biomarkers for respiratory and immune effects of ambient PM2.5.
