Abstract
Synadenium grantii is frequently used for the treatment of various diseases such as allergies, gastric disorders, and especially cancer. The aim of this study was to evaluate the possible antiproliferative potential of the methanol extract, fractions, and pure compounds from the stems of S grantii. Phytochemical analysis was carried out by conventional chromatographic techniques, and the antiproliferative activity was analyzed using the sulforhodamine B assay and an MTT-based assay. Nonpolar fraction and its subfractions from the stems of S grantii exhibited promising cytostatic effect against several human tumor cell lines (glioma, breast, kidney, and lung), with total grown inhibition values ranging from 0.37 to 2.9 μg/mL. One of the active principles of this plant was identified as a rare phorbol diterpene ester, denoted as 3,4,12,13-tetraacetylphorbol-20-phenylacetate. This compound demonstrated antiproliferative activity against glioma, kidney, lung, and triple-negative breast cancer cell lines. These results demonstrate that S grantii stems produce active principles with relevant antiproliferative potential.
Introduction
The Synadenium genus, which belongs to the family Euphorbiaceae, has been linked to some relevant pharmacological properties such as anticancer, 1 anti-inflammatory, 2 fibrinolytic action, 3 and immunoregulation. 4
Synadenium grantii Hook is a shrub from Africa that is commonly found growing as hedges. It is popularly known in Brazil as “Leitosinha” or “Janaúba.” The latex of this plant is widely used in traditional medicine to treat various diseases such as allergies, gastric disorders, and especially cancer. 5 –7
Previous experiments have suggested that S grantii latex may potentially present an antiulcerogenic effect. 7 Moreover, the chloroform extract of the leaves of this plant demonstrated cytotoxicity and antiparasitic activity. 8
In a recent study conducted with the stem bark of S grantii, in vitro and in vivo assays demonstrated significant antioxidant and anti-inflammatory activities, which were associated with the presence of phenolic compounds and terpenes. 9 Other phytochemical investigations indicated the presence of terpenes and phenolic compounds in bark extract of S grantii. 7 Some other compounds have been found in this plant, such as diterpene esters, anthocyanins, and unsaponificable substances. 6 –10
As part of the Network Ribecancer 212RT 0464 (CYTED/CNPq), the aim of this study was to evaluate the antiproliferative activity of the crude methanol extract and fractions/subfractions of the stem of S grantii as well as a rare isolated compound (phorbol diterpene ester) denoted as 3,4,12,13-tetraacetylphorbol-20-phenylacetate (compound 1).
Materials and Methods
Plant Material and Phytochemical Analysis
Synadenium grantii was collected in the town of Itajaí (SC-Brazil) in March and August 2013 and identified by Dr Oscar B. Iza (University of Itajaí Valley). A voucher specimen was deposited at the Barbosa Rodrigues Herbarium (Itajaí-SC) under number VC Filho 108.
Fresh stems (166 g) and leaves (165 g) of S grantii were cut into small pieces and extracted by maceration with methanol at room temperature for a period of 1 week, providing the crude methanol extract of each part after solvent evaporation. These extracts were dissolved in acetone and methanol, and after evaporation, 2 new fractions were obtained from each part of the plant. An antiproliferative screening was conducted with all these fractions. Since only the stem fractions exhibited promising biological activity, they were selected for further studies.
Stems (1 kg) from S grantii were exhaustively extracted by maceration with methanol at room temperature for 7 days. The macerate was filtered and concentrated under reduced pressure in a rotary evaporator, yielding 31 g of crude methanol extract. All the extract was suspended in methanol:water (50:50) mixture and subjected to liquid–liquid partition using solvents of increasing polarity such as chloroform (yield of 2.6%) and ethyl acetate (yield of 5.8%), respectively.
Part of chloroform fraction (700 mg) was subjected to column chromatography (0.063-0.20 mm, 105.4 g, 3.5 × 50 cm; Merck, Darmstadt, Germany) over silica gel and eluted with hexane:ethyl acetate (100:0 → 0:100) in increasing order of polarity to afford 33 fractions that were combined based on the thin-layer chromatography (TLC) profiles, using hexane:ethyl acetate (80:20) as the mobile phase and reaction with sulfuric anisaldehyde heated at 100°C. The fractions 5 to 13 (118 mg) were rechromatographed as before, using a solvent system hexane:ethyl acetate, yielding new 19 subfractions, which were combined according to their TLC profiles. Two of them, which presented better TLC profiles, 11 to 15 (37 mg) and 16 to 19 (26 mg) were evaluated in antiproliferative assay.
A part of subfraction 11 to 15 (30 mg) was analyzed by high-performance liquid chromatography (HPLC) and then submitted to a flash chromatography eluted with hexane:acetone 8:2, yielding 12 mg of the phorbol diterpene ester 3,4,12,13-tetraacetylphorbol-20-phenylacetate (compound 1), which was identified by spectroscopic data (nuclear magnetic resonance [NMR] 1H and 13C) and compared with those recently published in the literature. 8
High-performance liquid chromatography analysis
A Shimadzu LC-20AT LC system (Shimadzu, Tokyo, Japan) was used, consisting of a SPD-M20A photodiode array detector, SIL-20AHT autosampler, and software LC-Solution (Shimadzu). The subfractions 11 to 15 from S grantii stems and compound 1 were diluted in methanol at 1 mg/mL and filtered through a 0.45-mm cellulose regenerated membrane filter. Samples (20 µL) were injected on a C18 column (Luna Phenomenex, 250 × 4.6 mm; 0.5-μm film thickness) conditioned at 35°C. The mobile phase consisted of acetonitrile (A) and water (pH 2.5, phosphoric acid; B) eluted in a gradient system, starting with 2% A and after 98 minutes 100% A. This was followed by a 10-minute equilibrium period prior to the injection of next sample. The analyses were monitored at 200 nm. All solvents used were HPLC grade and were degassed in an ultrasonic bath.
In Vitro Anticancer Activity Assay
Human tumor cell lines of U-251 (glioma), MCF-7 (breast), NCI/ADR-RES (ovary expressing multidrug resistance phenotype), 786-0 (kidney), NCI-H460 (lung, nonsmall cells), HT-29 (colon), and K562 (leukemia) were kindly provided by the NCI. Nontumor cell line HaCat (human keratinocytes) was donated by Professor Dr Ricardo Della Coletta, FOP/UNICAMP. Stock cultures were grown in medium containing 5 mL RPMI 1640 (GIBCO BRL, Sao Paulo, SP, Brasil) supplemented with 5% fetal bovine serum. Penicillin:streptomycin (1,000 microg/L:1,000 U/L, 1 mL/L) was added to the experimental cultures. Cells in 96-well plates (100 μL cells/well) were exposed to sample concentrations in dimethyl sulfoxide (DMSO)/RPMI (0.25, 2.5, 25, and 250 μg/mL) at 37°C, 5% of CO2 in air for 48 hours. The final DMSO concentration did not affect cell viability. Afterward, cells were fixed with 50% trichloroacetic acid, and cell growth was determined by spectrophotometry (540 nm) of cellular protein content using the sulforhodamine B assay. 11 Doxorubicin was used as standard (positive control). The concentration–response curve for each cell line and the total growth inhibition (TGI) values were determined through nonlinear regression analysis using the software ORIGIN 8.0 (OriginLab Corporation, Wellesley Hills, MA). 12
For the cell proliferation assays, using the triple-negative breast cancer (TNBC) cell lines MDA-MB231 and HBL100, kindly provided by the ATCC, the cells were cultured in Dulbecco modified Eagle medium (DMEM) supplemented with 10% fetal bovine serum and antibiotics (penicillin at 100 mU/mL and streptomycin at 100 μg/mL) at 37°C in a humidified atmosphere in the presence of 5% CO2 and 95% air.
Cells were plated in 24-well plates at 8,000 cells/well (MDA-MB231) and 15,000 cells/well (HBL100) and cultured overnight in DMEM + 10% fetal bovine serum. The next day, medium was replaced with DMEM containing compound 1 in the concentrations 1, 5, 10, 25, 50, and 100 μmol/L. Cell proliferation was analyzed by an MTT-based assay as described previously. 13
Results
Crude methanol extracts of both S grantii stems and leaves and their respective acetone (apolar to medium polar compounds) and methanol (polar compounds) fractions were evaluated in a panel of 4 human cancer cell lines (U-251 [glioma], MCF-7 [breast], 786–0 [kidney], and NCI-H460 [lung, nonsmall cells]) in order to select the more promisor extract and/fraction for further evaluations.
This way, stems extract (GI50 between 8.1 and 30.3 µg/mL) showed better antiproliferative results than leaves extract (GI50 between 35.6 and >250 µg/mL). Moreover, the acetone fraction of S grantii stems demonstrated higher potency (50% growth inhibition, GI50, between 0.37 and 2.9 µg/mL) than that observed for crude stems extract, suggesting that active compound was concentrated in acetone fraction (Table 1). A similar profile was observed between acetone and methanol leaf fraction being acetone leaf fraction more active than methanol fraction and methanol crude extract (Table 1). Based on these results, S grantii stems methanol extract was selected for phytochemical analysis.
Antiproliferative Activity of Doxorubicin (Positive Control), Acetone (ac), and Methanolic (me) Fractions From the Stems and Leaves of Synadenium grantii Against Human Cancer Cell Lines.a
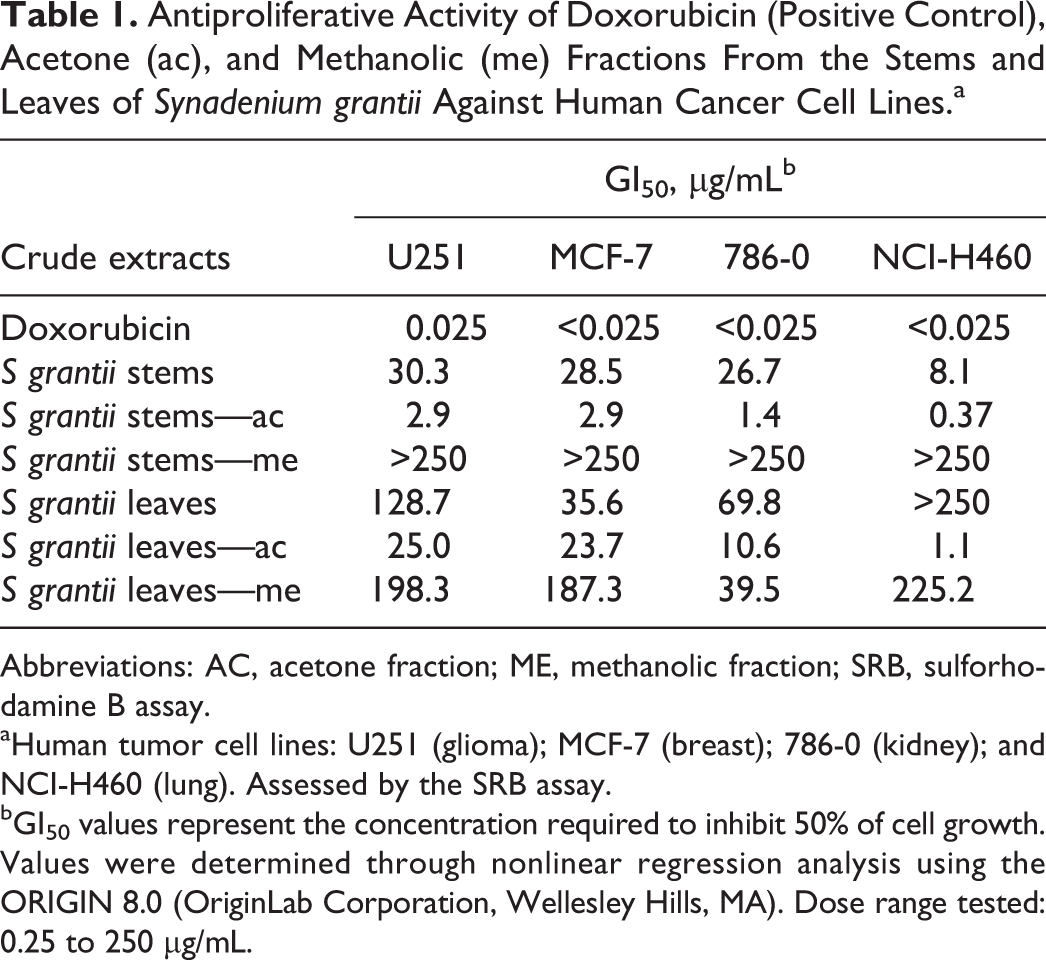
Abbreviations: AC, acetone fraction; ME, methanolic fraction; SRB, sulforhodamine B assay.
aHuman tumor cell lines: U251 (glioma); MCF-7 (breast); 786-0 (kidney); and NCI-H460 (lung). Assessed by the SRB assay.
bGI50 values represent the concentration required to inhibit 50% of cell growth. Values were determined through nonlinear regression analysis using the ORIGIN 8.0 (OriginLab Corporation, Wellesley Hills, MA). Dose range tested: 0.25 to 250 μg/mL.
Thus, a bioguided phytochemical study of S grantii stems methanol extract was performed seeking to determine the active principles. First, methanol crude extract was partitioned by liquid–liquid partition into chloroform (nonpolar compounds) and ethyl acetate (polar compounds) fractions.
Based on the initial results (Table 1), in which the nonpolar fraction showed greater antiproliferative activity, as well as the recent study conducted by other researcher group, 14 part of chloroform fraction was subjected to column chromatography, yielding 2 subfractions, 11 to 15 and 16 to 19. Both fractions presented suitable but complex chromatographic profiles and pronounced and promising biological results, but 11 to 15 exhibited the best chemical profile. Figure 1 indicates the HPLC profile of this fraction (11-15). It furnished a rare phorbol diterpene ester identified as 3,4,12,13-tetraacetylphorbol-20-phenylacetate (compound 1; Figure 2).
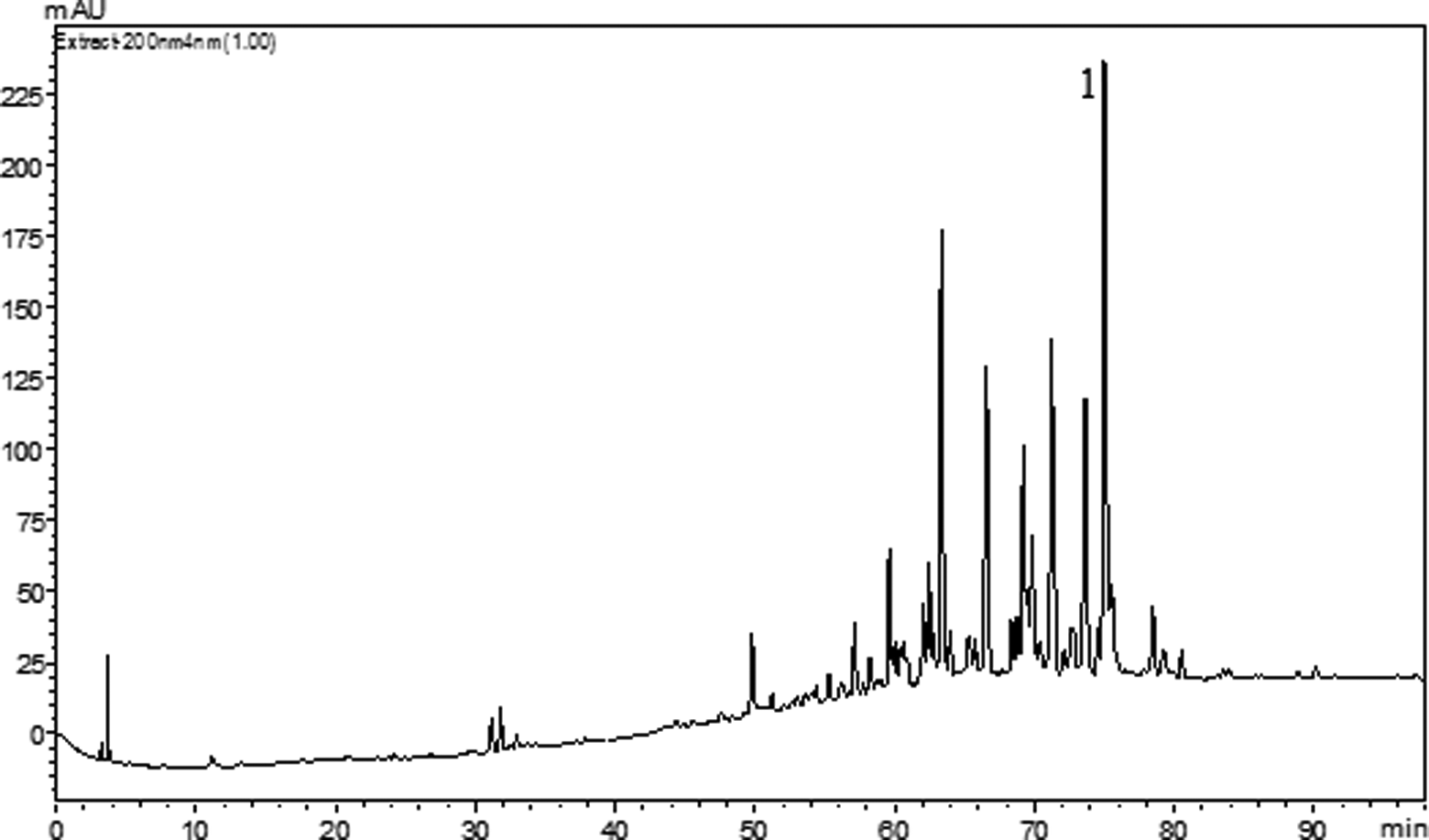
High-performance liquid chromatography (HPLC) analysis of subfractions 11 to 15 from S grantii stems and compound 1.
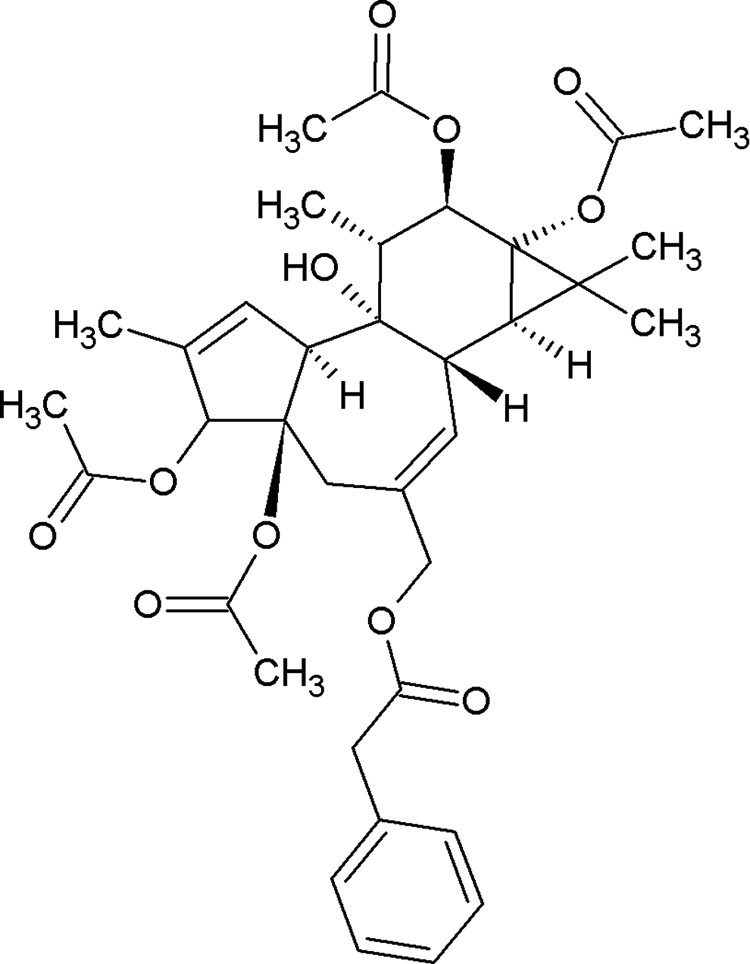
Molecular structure of 3,4,12,13-tetraacetylphorbol-20-phenylacetate (compound 1) isolated from S grantii stems.
Phytochemical analysis using TLC, together with standard compounds already isolated from the plant (friedelin, 3β-friedelinol, eufol, and citrostadienol) 9 –14 indicated that these were absent, suggesting other active principles that could act as antiproliferative agents, like compound 1.
Subfractions 11 to 15 and 16 to 19 and compound 1 were tested for their antiproliferative activity against different human cell lines (Table 2). Both subfractions demonstrated high potency for all human cancer cell lines tested, except leukemia, with TGI values of between 5.3 and 11.7 µg/mL, being subfractions 16 to 19 slightly more active, especially against glioma (U251), breast (MCF-7), and kidney (786-0) cancer cell lines than subfractions 11 to 15. Compared to the control doxorubicin, both subfractions were less cytotoxic against the nontumor cell line (keratinocyte). Compound 1 demonstrated a moderate antiproliferative activity against glioma (U 251, TGI = 25.2 μg/mL), kidney (786-0, TGI = 24.1 µg/mL), and lung (NCI-H460, TGI = 31.1 µg/mL), suggesting that compound 1 could contribute partly to antiproliferative activity observed for subfractions 11 to 15. Furthermore, compound 1 did not show activity against the nontumor cell line (keratinocyte).
Antiproliferative Activity of Doxorubicin (Positive Control), Subfractions From S grantii Stems and Compound 1 Against Human Cancer Cell Lines.a
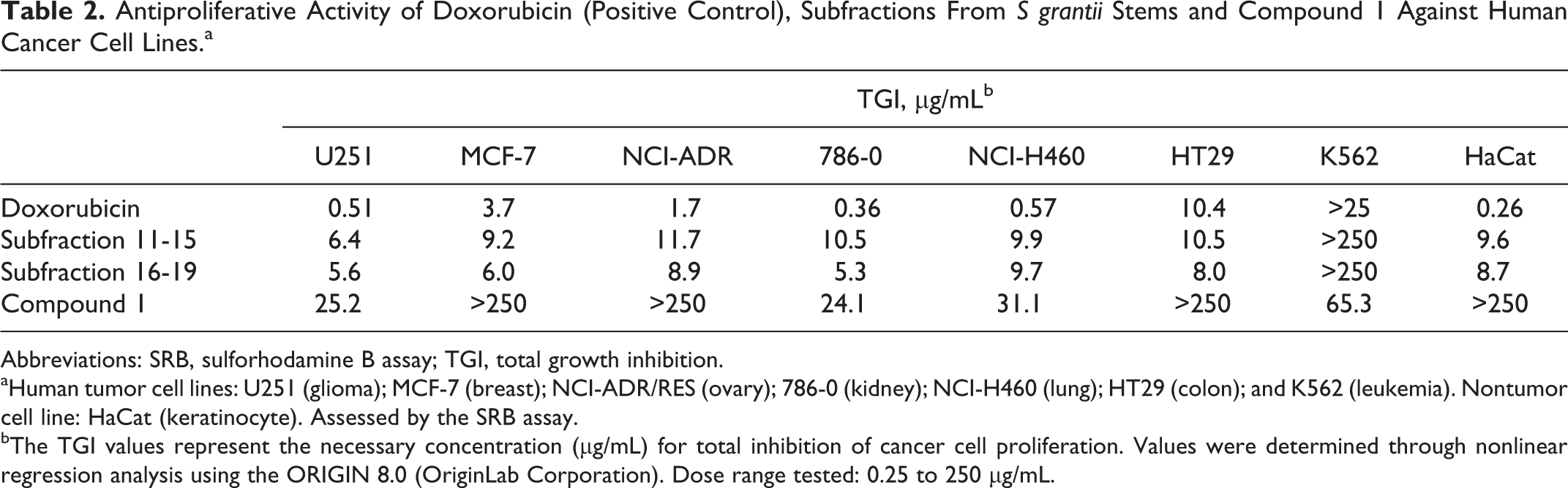
Abbreviations: SRB, sulforhodamine B assay; TGI, total growth inhibition.
aHuman tumor cell lines: U251 (glioma); MCF-7 (breast); NCI-ADR/RES (ovary); 786-0 (kidney); NCI-H460 (lung); HT29 (colon); and K562 (leukemia). Nontumor cell line: HaCat (keratinocyte). Assessed by the SRB assay.
bThe TGI values represent the necessary concentration (μg/mL) for total inhibition of cancer cell proliferation. Values were determined through nonlinear regression analysis using the ORIGIN 8.0 (OriginLab Corporation). Dose range tested: 0.25 to 250 μg/mL.
The effect of compound 1 was further tested in 2 cellular models of TNBC: MDA-MB231 and HBL100 cells (Figure 3). These analyses, using MTT metabolization as a readout of the cell number, indicated that compound 1 at concentrations between 1 and 100 µmol/L was active against the 2 cell lines, being more potent on HBL100 than on MDA-MB231. In HBL100 cells, compound 1 showed good activity at concentrations of 1 µmol/L and above. Compound 1 was less potent as an antiproliferative drug on MDA-MB231 cells, which is a cell line characterized by a high proliferation rate and resistance to several drugs.
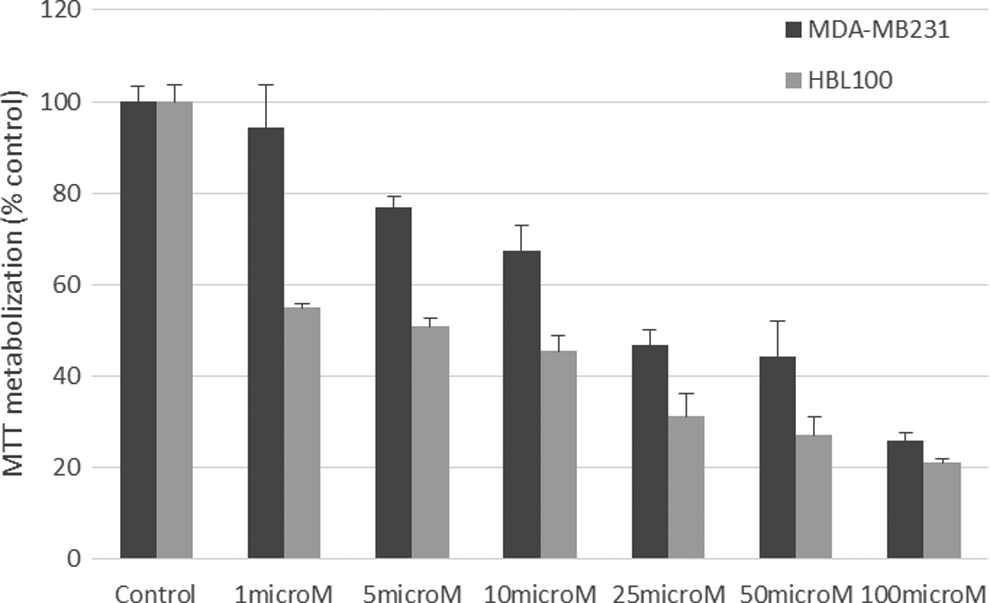
MTT metabolization (percentage control) of compound 1 against 2 triple-negative breast cancer cell lines, MDA-MB231 and HBL100.
Discussion
Cancer remains a major public health problem and one of the leading causes of death in the world, particularly in developing countries. 15,16 The identification of new drugs and treatments to solve this problem is therefore urgent.
There are a number of distinctly different types of kidney cancer, each with a different histology and clinical course, which respond differently to therapy. This cancer is responsible for over 115,000 deaths annually worldwide. 17 –19
Malignant gliomas account for 40% to 60% of primary brain tumors and are responsible for a disproportionate level of morbidity and mortality among patients with cancer. Despite treatments of brain tumors, such as surgery, radiotherapy, chemotherapy, and immunotherapy, the median survival time is <15 months. Therefore, the challenges in the treatment of malignant gliomas remain considerable. 20,21
The TNBC accounts for 15% of all breast cancers and tend to exhibit an aggressive metastatic behavior. These tumors respond to conventional chemotherapy but have relapse very frequently and have a worse prognosis. 22,23
The toxicity of the Synadenium genus has already been demonstrated. Valadares et al 24 and Mota et al 25 found that Synadenium umbellatum has potential cytotoxic and mutagenic effects.
In vitro and in vivo assays demonstrated that S grantii latex possesses cytotoxic effect and antitumoral activity against B16F10 melanoma cells. 14 Citrostadienol, which was isolated from this plant, showed cytotoxic activity when tested in vitro against melanoma cells. 14 Terpenes that are present in latex as well as other parts of the S grantii are described as presenting cytotoxic activity. 7,8 –10 Interestingly, the majority of the compounds previously described as active principles for this plant 8,9 –14 are different from those evidenced in our studies with subfractions 11 to 15 and 16 to 19, as evaluated by spectroscopic (NMR) and chromatographic methods (TLC and HPLC).
The phorbol diterpene ester 3,4,12,13-tetraacetylphorbol-20-phenylacetate isolated from the subfractions 11 to 15 is a rare compound and was isolated only recently from the leaves of S grantii. 8 The antiproliferative action of this compound was not as good as the one presented by subfraction that was isolated, suggesting a possible synergy between the compounds, or the presence of other active principles present in S grantii stems.
Phorbol esters are members of the tigliane family of diterpenes. These compounds have been isolated from plants belonging to the family Euphorbiaceae. 26 –28
Phorbols and their different derivatives are reported to be tumor promoters. These compounds do not induce tumors but promote tumor growth following exposure to a subcarcinogenic dose of a carcinogen. Some phorbol esters isolated from Jatropha curcas were found to have carcinogenic and mutagenic properties. However, some naturally occurring phorbols are tumor inhibitors and possess antileukemic and antimycobacterial activity. 28 –30
It will be interesting in the future to elucidate the antiproliferative mechanism of action of the phorbol isolated in this study.
In conclusion, these results demonstrate that the stems of S grantii possess active principles with relevant antiproliferative potential, distinct from those already described for this plant, stimulating the progress of studies to discover the compounds responsible for the indicated effects. Studies are in progress to confirm these effects in other pharmacological models and to isolate the other possible active principle of this promising plant as an anticancer agent.
Footnotes
Acknowledgments
The authors are grateful to the Network Ribecancer 212RT 0464 (CYTED/CNPq), the National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico–CNPq), and ProPPEC/UNIVALI for their financial support
Author Contributions
Adriana Campos substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content. Débora Barbosa Vendramini-Costa and Ana Lúcia Tasca Gois Ruiz contributed to acquisition, analysis, or interpretation of data and critically revised the manuscript for important intellectual content. Giovanna Barbarini Longato and Tailyn Zermiani contributed to acquisition, analysis, or interpretation of data. João Ernesto de Carvalho substantially contributed to conception or design and contributed to acquisition, analysis, or interpretation of data. Atanasio Pandiella substantially contributed to conception or design, acquisition, analysis, or interpretation of data and critically revised the manuscript for important intellectual content. Valdir Cechinel Filho substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
