Abstract
Earlier reports have suggested that exposure to radiation at workplace may induce cytogenetic abnormalities. However, the association between plasma antioxidants and the cytogenetic abnormalities in these patients has not been elucidated till now. Hence, the present study was undertaken to determine the relationship between the cytogenetic abnormalities, plasma antioxidant system, and the radiation exposure levels in men who were occupationally exposed to ionizing radiation. The study included 134 male volunteers, among whom 83 were occupationally exposed to ionizing radiation. Incidence of micronuclei and chromosomal aberration was assessed in lymphocytes. Total and reduced glutathione (GSH), total antioxidant capacity (TAC), superoxide dismutase (SOD), and lipid peroxidation were assessed in the plasma. The micronuclei frequency and chromosomal aberrations were significantly higher in the exposed group in comparison to the nonexposed group (
Keywords
Introduction
The effect of exposure to low levels of diagnostic and therapeutic radiation at their workplaces is a serious concern to a large number of health care workers. 1 Although health workers are not exposed to radiation directly, the risk of receiving scattered radiation is extremely variable. 2,3 It is estimated that approximately 7.25 million medical radiation workers worldwide 4 are being exposed to low levels of ionizing radiation at their workplaces. Studies have demonstrated that exposure to even low levels of chronic radiation can potentially increase the frequency of chromosomal aberrations, micronucleus incidence, 5 -8 and aneuploidy 9 in the blood lymphocytes.
The biological effects of ionizing radiation are induced directly by damaging the DNA or indirectly by generating reactive oxygen species (ROS). The indirect process mediated by ROS is a cause of 70% of all biological effects. 10,11 One of the ROS-mediated cellular damage is membrane lipid peroxidation. This results in the formation of malondialdehyde (MDA) which has been widely used as a marker to monitor the degree of peroxidative damage. In order to protect against ROS-mediated damage, the body has evolved a variety of defense mechanisms involving antioxidant molecules and radioprotective enzymes. 12,13 Previous studies have reported enhanced antioxidant levels in the blood plasma of chronically exposed radiation health workers. 14,15 Although our earlier study has reported the association between sperm chromatin integrity and seminal plasma antioxidant system, 16 there has been no data elucidating the relationship between the blood plasma antioxidants and cytogenetic aberrations in the occupationally exposed participants. In view of these considerations, this study was designed to quantify the blood plasma antioxidant levels to determine its association with cytogenetic abnormalities and radiation exposure level.
Methods
Study Participants
The present study included 134 male volunteers in the age range of 21 to 50 years, among whom 83 were handling radiation sources (X rays, γ rays, and β emitters) at various hospitals. In this study, the radiation-exposed participants were radiographers, radiotherapy technicians, or nuclear medicine technologists involved in various diagnostic or therapeutic procedures using X rays, γ rays, and β particles. The nonexposed participants (N = 51) were employees of the same hospitals but were not exposed to any of the mentioned radiation sources in their occupational settings. The nonexposed participants had been working in various departments of the hospitals as laboratory technicians, nurses, and administrative supporting staff. All the participants were considered as chronically exposed to low-dose radiation. Volunteers who fulfilled the criteria were given a questionnaire to obtain information about the duration of stay at their workplace, type of radiation source they were exposed to, and their lifestyle factors such as smoking, alcohol intake, and diet pattern. Information collected on lifestyle factors was limited to only 5 years history of the study participants. Approval from the Institutional Ethical Committee was obtained before the commencement of the study.
Calculating Radiation Absorbed Dose
Thermoluminescent dosimeter (TLD) device-based exposure monitoring was performed for each participant. Health workers handling radiation source (exposed group) wore TLD device. Participants in the nonexposed group did not wear TLD device. The cumulative exposure level of each participant was collected from the radiation safety officer of the respective hospital where the participant was enrolled.
Peripheral Blood Sample Collection and Lymphocyte Culture
Peripheral blood was obtained from each participant by venipuncture. Freshly collected 0.5 mL of whole blood was added to 4.5 mL of Rosewell Park Memorial Institute 1640 (Cat. No. R6504; Sigma Aldrich Inc St. Louis, MO, USA) medium supplemented with 10% (v/v) fetal bovine serum, antibiotics (50 µg/mL penicillin), and 5 µg/mL phytohemagglutinin (Cat. No. L8902; Sigma Aldrich Inc St. Louis, MO, USA) and mixed well. The cultures were maintained at 37°C in a 5% CO2 incubator for 72 hours. Blood plasma was separated by centrifugation of the whole blood at 3000 rpm for 20 minutes. The separated plasma was aliquoted in multiple sterile vials and stored at −80°C until the biochemical estimation (maximum storage period was 2 years). An analysis was performed once the samples were collected from all the study participants.
Micronucleus Assay
Micronucleus (MN) assay in lymphocytes was performed according to the method previously described 17 with minor modifications. Cytokinesis was blocked using 5 µg/mL of cytochalasin B (Cat. No. C6762; Sigma Aldrich Inc St. Louis, MO, USA) after 44 hours of culture. Cells were processed at the end of 72 hours by treating with 10 mL of 0.56% potassium chloride (Cat. No. P5405; Sigma Aldrich Inc St. Louis, MO, USA) followed by methanol–acetic acid fixation (Carnoy’s fixative). The washed cells were transferred onto prechilled glass slides, air-dried, and then stained with propidium iodide (4 µg/mL). The cells were evaluated under a fluorescence microscope using 40× objective, and the number of MN in binucleated cells (BNCs) was identified. Cells having 2 distinct nuclei of approximately equal size, which may be attached by a clear nucleoplasmic bridge or touching each other at edges, were considered as BNC. Micronuclei are morphologically identical to the main nuclei but are smaller and not linked to the main nuclei via a nucleoplasmic bridge. A total of 2000 BNCs in each group were scored, and the frequency of cells with 1 (MN1), 2 (MN2), and 3 (MN3) micronuclei was calculated. The mononucleated and multinucleated cells were counted separately to calculate the nuclear proliferation index.
Chromosome Aberration Assay
The chromosomal aberration assay was performed as described by Preston et al. 18 The metaphase plates were obtained in the conventional manner from the culture of peripheral blood lymphocytes after 72 hours of incubation. Colchicine (5 µg/mL; Cat. No. C9754; Sigma Aldrich Inc St. Louis, MO, USA) was added to the culture 3 hours prior to the metaphase preparation, and the cells were treated with 0.56% potassium chloride (Cat. No. P5405; Sigma Aldrich Inc St. Louis, MO, USA) for 30 minutes followed by fixation in 3:1 methanol–acetic acid (Carnoy’s fixative). Cells were washed and dropped gently on clean, prechilled microscope slides, air-dried, and stained with Giemsa. From each participant, a minimum of 50 metaphase plates were analyzed for both structural and numerical chromosomal abnormality (aneuploidy). Chromosome aberrations were classified according to an International System for Human Cytogenetic Nomenclature. To evaluate the mitotic index, cytogenetic preparations were analyzed using a light microscope at 100× magnification and expressed as number of metaphase plates per 1000 nuclei analyzed.
For all the microscopic analysis such as MN and chromosome assessment, the slides were coded to avoid observer’s bias. Slides from all the study participants were scored by a single person, and a second evaluation was performed independently to confirm the primary observation.
Biochemical Estimation of Blood Plasma for Antioxidants and ROS
Assessment of Total and Reduced Glutathione
The total glutathione (GSH) concentration was determined according to the method described by Anderson 19 with minor modifications. Briefly, 50 μL of blood plasma was added to 100 μL of reaction buffer, 1.5 mmol/L phosphate buffer, 2.4 mmol/L NADPH, 10 mmol/L 5,5’-dithiobis(2-nitrobenzoic acid; DTNB), and 0.5 U GSH reductase, pH 7.5, and the optical density (OD) was recorded after 1 minute using a Biophotometer (Eppendorf, Germany) at a wavelength of 405 nm. A standard graph was prepared by using various concentrations of GSH. The total GSH level was determined by plotting the OD values of the blood plasma against the standard graph and dropping a perpendicular line to the x-axis.
The estimation of reduced GSH was done according to Beutler et al 20 with minor modifications. Briefly, 50 µL of blood plasma was mixed with 50 µL phosphate buffer, 25 µL of precipitating reagent, and 25 µL of fresh DTNB. The OD of the test solution was determined at 405 nm exactly after 1 minute. A standard graph was prepared by using various known concentrations of reduced GSH. The reduced GSH level in blood plasma was determined by plotting the OD values of the blood plasma against the standard graph and dropping a perpendicular line to the x-axis.
Assessment of TAC in Plasma
The ability of the blood plasma to scavenge ABTS+ [2, 2’-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid)] radical cation was compared to the Trolox standard. 21 Briefly, the ABTS+ radical cation was pregenerated by mixing 7 mmol/L ABTS (Cat. No. A1888; Sigma Aldrich Inc St. Louis, MO, USA) stock solution with 2.45 mmol/L potassium persulfate and incubated for 12 to 16 hours in dark at room temperature. The absorbance of the ABTS+ solution was equilibrated to 0.70 (± 0.02) OD value by diluting with phosphate-buffered saline at room temperature, then 1 mL of the equilibrated ABTS+ solution was mixed with 10 μL of the blood plasma followed by measurement of absorbance at 650 nm after 1 minute. The inhibition of absorbance (%) was calculated and plotted as a function of the concentration of standard and sample to determine the trolox equivalent antioxidant concentration.
Assessment of Superoxide Dismutase Level
Estimation of superoxide dismutase (SOD) was done according to Marklund and Marklund 22 with minor modifications. One unit of SOD is defined as the amount of enzyme that inhibits auto-oxidation of pyrogallol by 50%. Briefly, 100 μL of blood plasma was added to 2.85 mL of 0.1 mol/L phosphate buffer (pH 8.4) and 50 μL of 100 mmol/L pyrogallol (Cat No. RM170; HiMedia Laboratories Mumbai, India) prepared in 20 mmol/L HCl. The absorbance of the reaction mixture was recorded at 0 and 3 minutes. Similarly, the auto-oxidation was measured by adding 2.95 mL phosphate buffer (pH 8.4) and 50 μL pyrogallol at 0 and 3 minutes using a Biophotometer at a wavelength of 405 nm. The SOD activity was measured using the calibration curve of percentage inhibition and was expressed as enzyme units/μL of blood plasma.
Measurement of Lipid Peroxidation by MDA Assay
The lipid peroxidation levels in the blood plasma was measured using a thiobarbituric acid reactive substances (TBARS) assay, which reflects the production of MDA. 23 Briefly, 100 μL of blood plasma was added to 1.8 mL of 3% phosphoric acid (Cat. No. 215104; Sigma Aldrich Inc St. Louis, MO, USA) and 0.6 mL of 0.6% TBA (Cat. No. T5500; Sigma Aldrich Inc St. Louis, MO, USA). These mixtures were heated in boiling water for 45 minutes. After cooling, the intensity was measured at 550 nm using a Biophotometer, and the level was determined using the molar absorption coefficient at 550 nm.
Statistical Analysis
SPSS (SPSS 16.0) was used for the data analysis. Data have been presented as mean and standard error of mean (Mean ± SEM) for continuous variables, whereas qualitative variables were represented in percentage. Independent Student
Results
Study Participants
The details of the participant characteristics have been provided in the Table 1. The difference between the mean age of the nonexposed and exposed participants was not statistically significant. The average work experience of exposed participants was 6.51 ± 0.66 years. The smoking habits, alcohol intake, and diet did not differ significantly between the 2 groups. The radiation-exposed participants were further subdivided into the low absorbed dose (LAD) and high absorbed dose (HAD) groups based on their cumulative radiation exposure level (Table 1). The participants with absorbed dose of >0.05 mSv (here after referred as high absorbed dose, HAD) and participants with absorbed dose of ≤0.05 mSv (here after referred as low absorbed dose, LAD group) were compared to determine the effect of radiation absorbed dose on the various parameters.
Characteristics of the Participants Included in the Study. 24
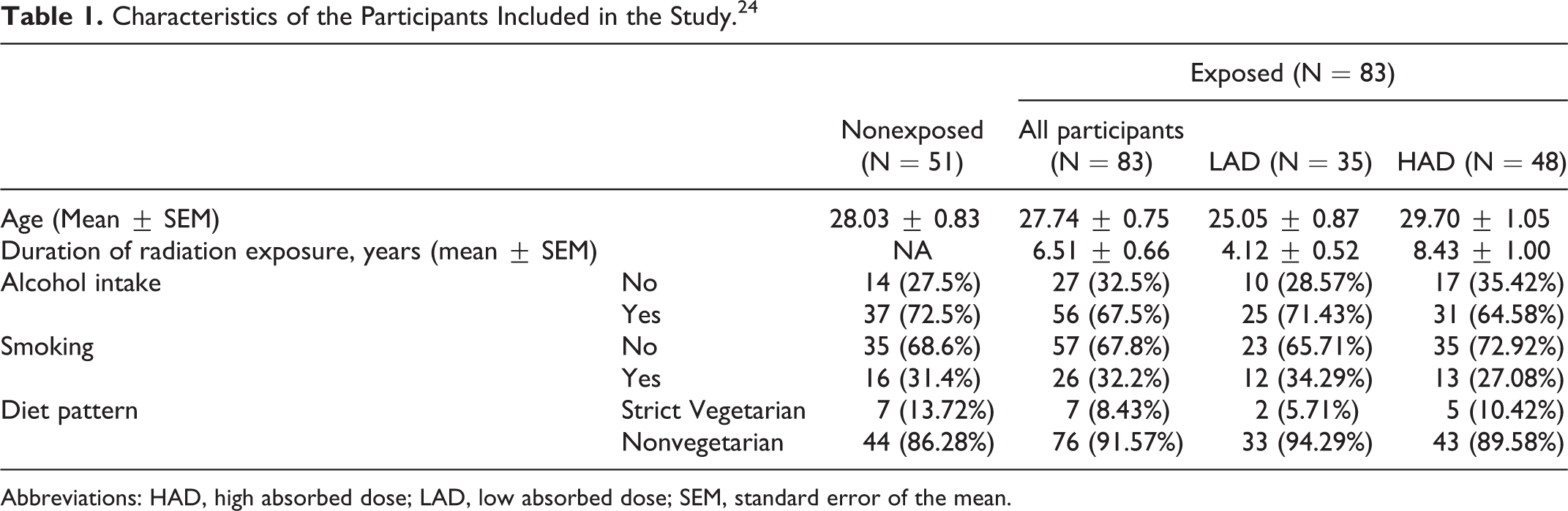
Abbreviations: HAD, high absorbed dose; LAD, low absorbed dose; SEM, standard error of the mean.
Micronuclei Frequency
The frequency of the MN cells in the exposed participants was about 4-fold higher than nonexposed participants (
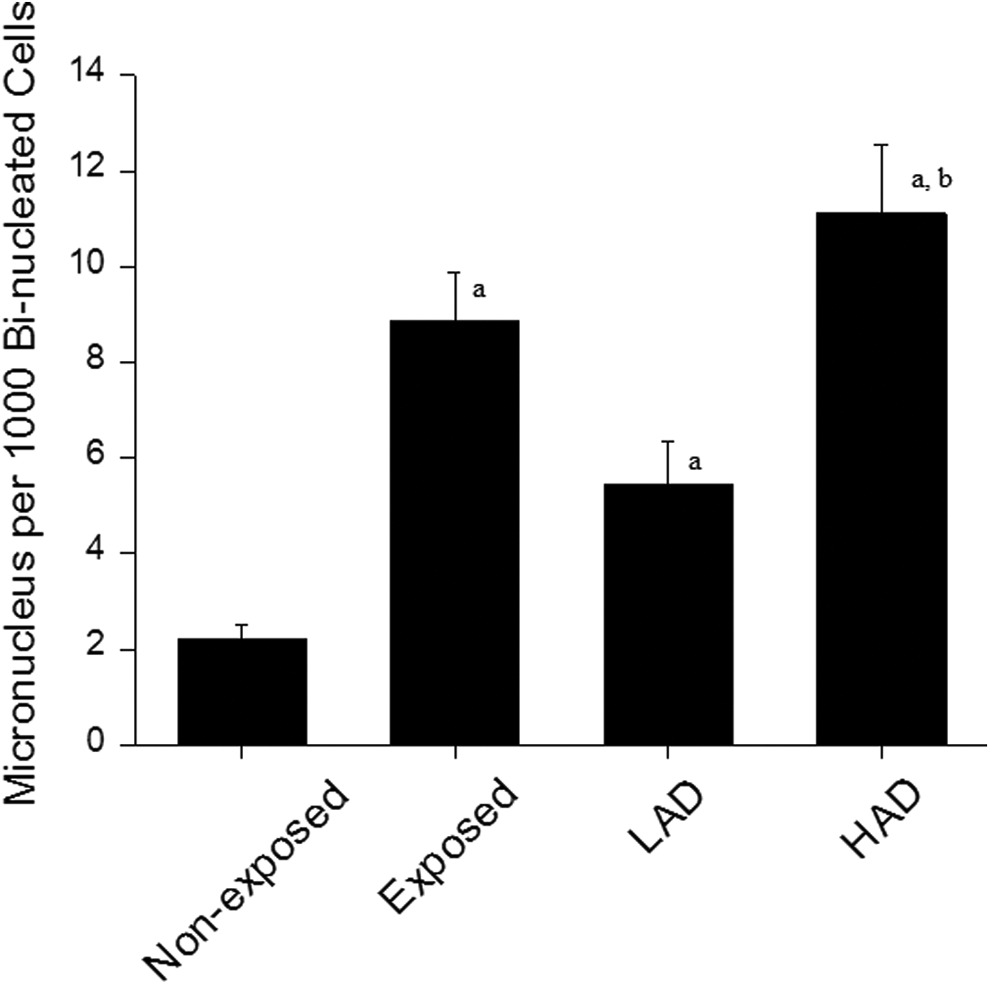
Micronuclei frequency in the blood lymphocyte of participants exposed to ionizing radiation at workplace. LAD indicates low absorbed dose (≤0.05mSV); HAD indicates high absorbed dose (>0.05mSV). a
Chromosomal Aberrations
The frequency of aneuploidy in the exposed participants (4.71 ± 0.37) was significantly higher compared to the nonexposed group (2.42 ± 0.25;
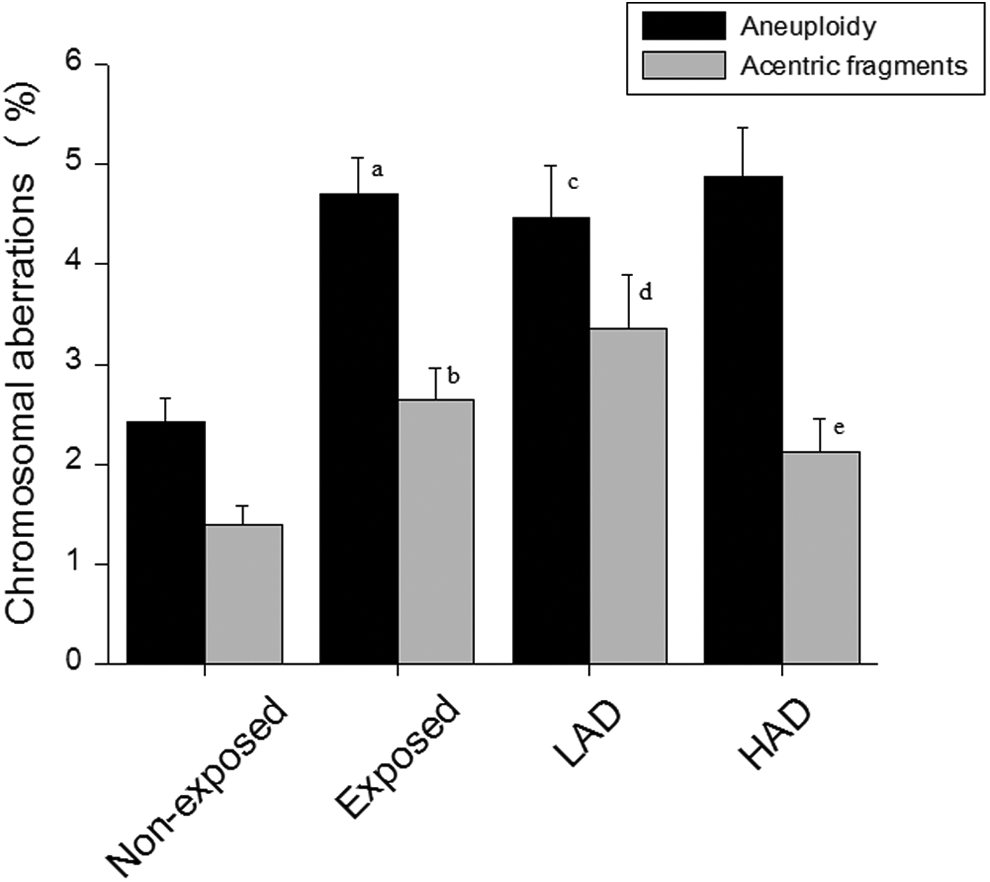
Incidence of chromosomal aberrations in the peripheral blood lymphocyte in participants exposed to ionizing radiation at workplace. LAD indicates low absorbed dose (≤0.05mSV); HAD, high absorbed dose (>0.05mSV). a
Level of GSH
Total GSH level in the exposed group was significantly higher than nonexposed group (
Antioxidant Level and Lipid Peroxidation in the Blood Plasma of the Occupationally Exposed and Nonexposed Participants.
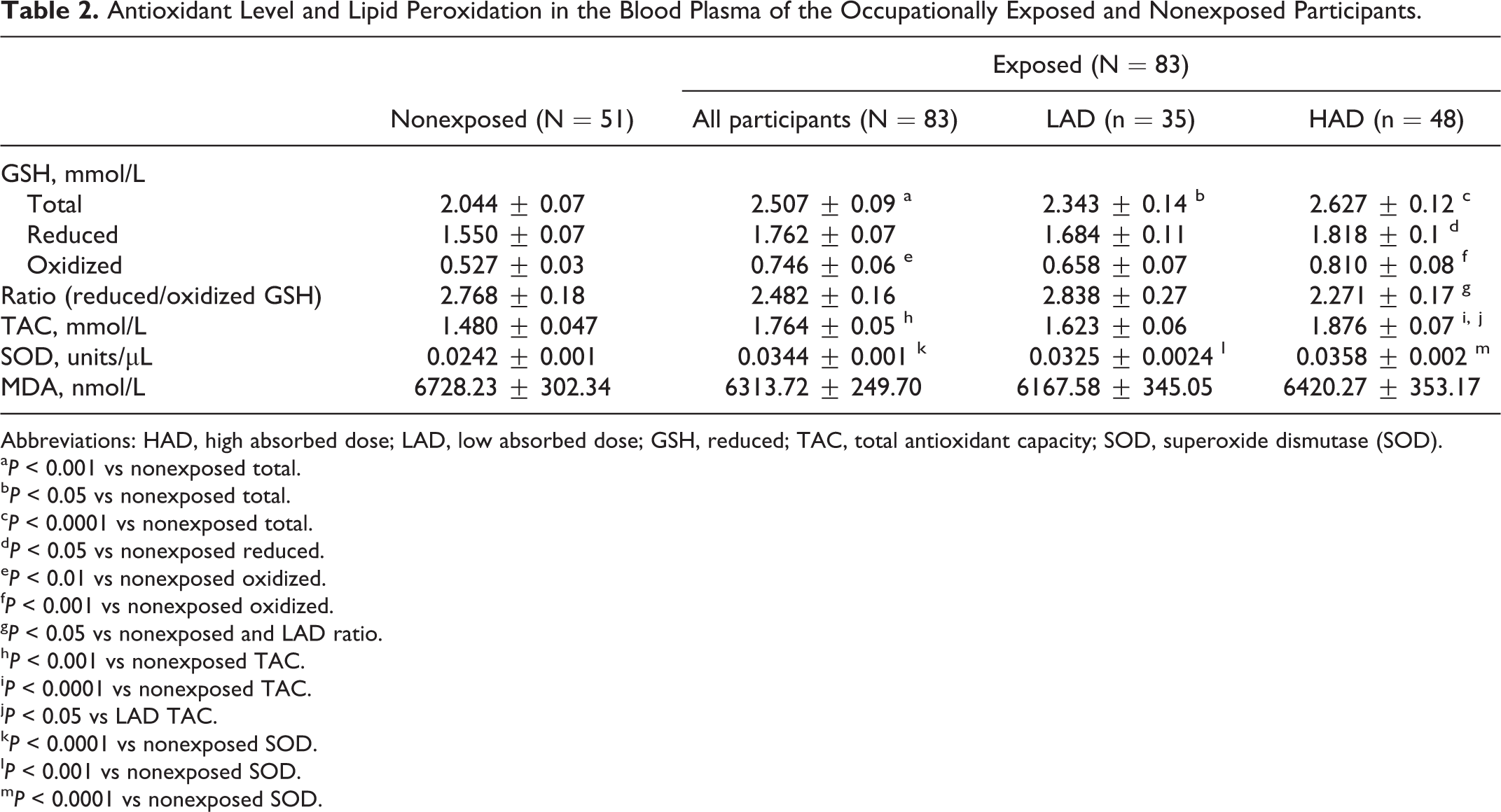
Abbreviations: HAD, high absorbed dose; LAD, low absorbed dose; GSH, reduced; TAC, total antioxidant capacity; SOD, superoxide dismutase (SOD).
a
b
c
d
e
f
g
h
i
j
k
l
m
Total Antioxidant Capacity
The TAC was determined to find out the total enzymatic and nonenzymatic antioxidant activity in relation to exposure level. Similar to GSH, TAC level was also significantly higher in the exposed participants compared to nonexposed participants (
Superoxide Dismutase
The level of SOD in the exposed and nonexposed group was 0.0242 ± 0.001 and 0.0344 ± 0.0013 units/µL, respectively, and the difference was statistically significant (
Lipid Peroxidation
The extent of lipid peroxidation was determined in both nonexposed and exposed participants by estimating the MDA level in blood plasma. The level of MDA between exposed and nonexposed group was not statistically significant. Similarly, no significant difference was observed between HAD and LAD groups (Table 2).
Association Between Frequency of MN, Chromosomal Aberrations, and Radiation Absorbed Dose
An attempt was made to determine the association between the incidence of MN, chromosomal aberrations, and radiation absorbed dose. Frequency of MN in the exposed participants showed a positive correlation with radiation exposure level (
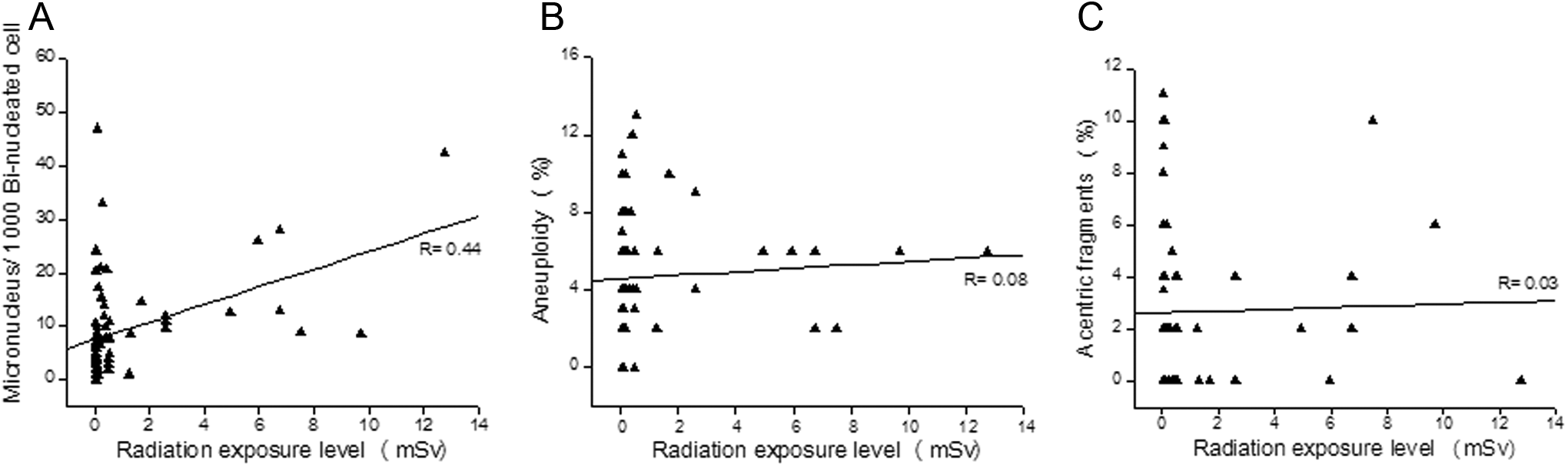
Association between radiation exposure level and micronuclei frequency, aneuploidy, and acentric chromosomal fragments in the exposed participants. A, Correlation analysis of radiation exposure level (mSv) and micronuclei per 1000 binucleated cell in the exposed group (
Influence of Confounding Factors
A cross tabulation of the smoking habits, alcohol intake, and diet pattern of both exposed and nonexposed groups was performed to rule out the effect of confounding factors on the results observed. Information on diet pattern was collected under 5 groups, namely, (1) participants who consumed only vegetables; (2) participants who consumed eggs in addition to vegetables; (3) participants who consumed fish in addition to eggs and vegetables; (4) participants who consumed chicken in addition to fish, eggs, and vegetables; and (5) participants who consumed read meat in addition to chicken, fish, eggs, and vegetables. Similarly, participants were categorized based on the frequency of alcohol intake (everyday, once/week, once/month, and occasional). Participants with smoking habits were segregated based on the frequency of smoking (occasional, <5 times a day, 6-10/ times a day, 11-20 times a day, and >20 times a day). Analysis of individual groups in addition to the broad groups as presented in the Table 1 did not show any significant differences between control and exposed participants. Multivariate analysis was performed using ANCOVA to find out the effect of confounding factors such as alcohol intake, smoking habits, diet pattern, and interaction of these factors with radiation exposure. These factors did not show any effect on all the parameters studied (data not shown).
Discussion
The present study indicates that occupational radiation exposure is associated with alterations in chromosomal integrity and DNA damage in peripheral blood lymphocytes of Indian (noncaucasians) radiation health workers. Earlier, we were the first to provide evidence on the detrimental effects of occupational radiation exposure on functional, genetic, and epigenetic integrity of spermatozoa in health workers, where a significant decline in sperm functional characteristics, increase in sperm DNA damage, and hypermethylation were observed in participants exposed to radiation 24 Further, the present study has demonstrated the increased level of glutathione, TAC, and SOD in the blood plasma of the participants exposed to radiation. The strengths of this study were (1) relatively large sample size than any other study, (2) direct correlations between level of exposure to radiation and MN frequency, chromosomal aberrations in the radiation health workers, and (3) simultaneous evaluation of genetic integrity in peripheral lymphocytes and blood plasma antioxidants level.
Chromosomal aberrations and DNA damage are considered as one of the most important markers of biological dosimetry to assess the genotoxicity caused by ionizing radiation. Earlier studies have clearly demonstrated association of occupational radiation exposure with chromosomal aberrations, DNA damage, 5 -8 and aneuploidy 9 in peripheral blood lymphocytes. The increase in the frequency of aneuploidy, acentric fragments, and MN observed in the lymphocytes of the exposed participants further indicates possible genomic damage induced by occupational radiation exposure. These participants also had increased sperm chromatin abnormalities, hypermethylation, 24 and abnormal seminal plasma antioxidant levels, 16 which suggest that chronic occupational exposure to low levels of ionizing radiation can have detrimental effects on sensitive cell types. Interestingly, structural chromosomal aberration levels in the occupationally exposed participants did not show any significant difference between the HAD and the LAD groups. On the other hand, the HAD group had increased MN frequency in comparison to the LAD.
The GSH is an abundant tripeptide antioxidant molecule present in the cytoplasm and body fluids. The scavenging properties of GSH are mainly due to its function as cofactor for antioxidant enzyme glutathione peroxidase (GPX) and also its ability to react directly with ROS by donating Hydrogen ion (H+) from its free sulfhydryl group. De novo synthesis of GSH is required for cell activation and proper S and G2 phase transit. 25 When tissues are challenged with oxidative stress, the body reacts by elevating the expression of antioxidant enzymes and antioxidant molecules. 26 These results are in agreement with the observations made in the present study. Furthermore, the level of tGSH, rGSH, and oGSH were significantly higher in the high absorbed dose (HAD) group which further supports the argument that elevated oxidative stress induced by chronic low-dose radiation exposure induces adaptive response. Similar findings have been reported in the literature which has demonstrated high antioxidant levels in the blood of radiation health workers. 14,15 Interestingly, we have observed altered ratio of reduced–oxidized GSH level in HAD group of the radiation-exposed participants. The ratio of reduced GSH to oxidized GSH is an indicator of cellular health, with reduced GSH constituting up to 98% of cellular GSH under normal conditions. 27 The reduced–oxidized GSH ratio is used to evaluate oxidative stress status in biological systems, and alterations in this ratio have been demonstrated in aging, cancer, HIV replication, cardiovascular diseases, 28 -32 and in neurodegenerative diseases, such as Parkinson disease and Alzheimer disease. 33 Thus, measuring the reduced–oxidized GSH ratio is an ideal way to assess potential therapeutics efficacy in maintaining cellular redox potential.
The measure of TAC includes the cumulative action of all the antioxidants present in plasma and body fluids, thus giving an insight into the delicate balance in vivo between oxidants and antioxidants. 34 Hence, measuring the levels of plasma TAC may help in the evaluation of physiological, environmental, and nutritional factors of the redox status in humans. Our data have shown a significant increase in TAC in the blood plasma of the exposed participants, especially between the HAD and the LAD group which suggests an association between antioxidant defense and high radiation absorbed dose in the health workers.
Superoxide dismutase is an enzyme that catalyzes the dismutation of superoxide into either oxygen or hydrogen peroxide. Thus, it is an important antioxidant that acts fundamentally to reduce ROS-mediated diseases such as carcinoma, inflammation, and aging. 35,36 A previous study has shown an increase in the SOD concentration in the blood plasma of the radiation health workers. 14 Our findings are in agreement with this study, where exposed participants have shown an increase in SOD concentration. Similar increase in SOD activity, though lower than HAD group observed in the present study, indicates that even exposure to low-dose radiation can markedly elevate the antioxidant system as a trigger of defense mechanism. Earlier studies have shown that exposure to low-dose radiation can lead to an elevation in antioxidant enzymes such as SOD. 37,38
Our data did not show any change in the ROS level. We hypothesize that increased antioxidant concentration in the blood plasma might have efficiently scavenged the ROS level and thus helped in maintaining cellular homeostasis. Effects of low doses of radiation are considered as research priority in order to better understand the long-term health effects in the health workers occupationally exposed to radiation in their workplace.
The occupational radiation exposure levels in the participants included in the present study were well within the accepted limits. 39 The long-term consequence of low-level radiation exposure is an increase in frequency of chromosomal aberrations and micronuclei frequency in circulating blood lymphocyte. Interestingly, it is also inducing adaptive responses as shown by an elevated antioxidant concentration in the blood plasma and by this way may compensate for the unbalanced ROS and contribute to maintain cellular homeostasis. These findings indicate the functioning of the antioxidant defense mechanism in the blood plasma to prevent genotoxic damage.
In conclusion, the results presented in this study have demonstrated increased genetic instability in lymphocytes and altered antioxidants in the blood plasma of the radiation health workers. This is in agreement with our previous reports where an increase in sperm chromatin abnormality and changes in seminal plasma antioxidant system was observed. 23,16 It is possible that the increased plasma GSH, TAC, and SOD could be an adaptive measure to tackle the oxidative stress to protect genetic integrity in radiation health workers.
Footnotes
Acknowledgments
Statistical help from Dr Asha Kamath, Manipal University, is thankfully acknowledged. Authors thank the administrators and volunteers of all the hospitals for the support.
Author Contribution
D. Kumar contributed to conception and design, acquisition and analysis, drafted manuscript, and critically revised the manuscript. S. Kumari contributed to analysis. S. R. Salian contributed to analysis. S. Uppangala drafted the manuscript. G. Kalthur contributed to interpretation and critically revised the manuscript. S. Challapalli contributed to analysis. G. S. Chandraguthi contributed to analysis. P. Kumar critically revised the manuscript. S. K. Adiga contributed to conception and design, analysis and interpretation, drafted manuscript, and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Indian Council of Medical Research (ICMR; grant Ref: 5/10/11/2008-RHN), Government of India.
