Abstract
SYN-004 is a first in class, recombinant β-lactamase that degrades β-lactam antibiotics and has been formulated to be administered orally to patients receiving intravenous β-lactam antibiotics including cephalosporins. SYN-004 is intended to degrade unmetabolized antibiotics excreted into the intestines and thus has the potential to protect the gut microbiome from disruption by these antibiotics. Protection of the gut microbiome is expected to protect against opportunistic enteric infections such as Clostridium difficile infection as well as antibiotic-associated diarrhea. In order to demonstrate that oral SYN-004 is safe for human clinical trials, 2 Good Laboratory Practice-compliant toxicity studies were conducted in Beagle dogs. In both studies, SYN-004 was administered orally 3 times per day up to the maximum tolerated dose of the formulation. In the first study, doses of SYN-004 administered over 28 days were safe and well tolerated in dogs with the no-observed-adverse-effect level at the high dose of 57 mg/kg/day. Systemic absorption of SYN-004 was minimal and sporadic and showed no accumulation during the study. In the second study, doses up to 57 mg/kg/day were administered to dogs in combination with an intravenous dose of ceftriaxone (300 mg/kg) given once per day for 14 days. Coadministration of oral SYN-004 with intravenous ceftriaxone was safe and well tolerated, with SYN-004 having no noticeable effect on the plasma pharmacokinetics of ceftriaxone. These preclinical studies demonstrate that SYN-004 is well tolerated and, when coadministered with ceftriaxone, does not interfere with its systemic pharmacokinetics. These data supported advancing SYN-004 into human clinical trials.
Introduction
Antibiotic use is a primary risk factor for the development of Clostridium difficile infection (CDI) and antibiotic-associated diarrhea, 1 –4 and this holds true for intravenously (IV) administered antibiotics such as ceftriaxone. When certain IV antibiotics are administered, a substantial portion of the dose is excreted in the bile and enters the intestine. 5 –7 The biliary concentration of unchanged ceftriaxone can actually exceed the plasma concentration of the drug. 5 These excreted antibiotics can disrupt the gut microbiome leading to dysbiosis, an imbalance in the gut microflora, 8,9 and sets the stage for outgrowth of C difficile and other opportunistic enteric pathogens. 10
SYN-004 is a recombinant type A β-lactamase that is being developed for oral delivery with the objective of degrading excess IV β-lactam antibiotics excreted into the intestine through the bile, thus protecting the gut microbiome. SYN-004 is a modified, extended spectrum version of the P1A β-lactamase, which had previously been under development for a similar indication. 11 –14 SYN-004 degrades most β-lactam-containing antibiotics and was engineered from the Bacillus licheniformis PenP enzyme 12 to expand the hydrolysis of β-lactams to cephalosporins, including ceftriaxone, while maintaining its penicillin-degrading activity. P1A was originally under development by Ipsat Therapies Oy (Finland), 13 and much of the preclinical development work and safety studies which support SYN-004 development are based on these earlier P1A studies. 12,14
SYN-004 is initially being developed to be orally coadministered with IV β-lactam antibiotics and has been formulated as enteric pellets to be released in the proximal small intestine at pH >5.5. 12 It is anticipated that when the IV β-lactam antibiotic is excreted into the small intestine through the bile, active SYN-004 will be present to degrade the antibiotic before it reaches the colon and disrupts the gut microbiome. The dosage of SYN-004 has to be of sufficient quantity and frequency to ensure that active enzyme is present to degrade the biliary excreted antibiotics between antibiotic doses. It is expected that SYN-004 may be dosed 3 or 4 times daily to ensure that sufficient enzyme is present in the small intestine when the antibiotics are excreted in the bile, but the final dosing regimen will be determined in future clinical trials.
Prior to clinical testing of SYN-004 in humans, it was necessary to demonstrate that this modified enzyme was safe and well tolerated in animals. It was also important to ensure that SYN-004 was safe when administered in conjunction with an IV β-lactam antibiotic, especially cephalosporins. Further, for SYN-004 to be a useful and effective therapy for prevention of disruption of the gut microbiome, it should not affect the pharmacokinetics (PK) of the circulating antibiotics that have been administered to treat the primary infection (eg, pneumonia, urinary tract infection, etc).
The Beagle dog was chosen for these studies due to the similarity of its gastrointestinal (GI) tract with that of humans, its frequent use in pharmacology and toxicology studies, and the significant historical control data available. 15 –17 Dogs were also used for efficacy testing and demonstrated intestinal bioavailability of P1A and SYN-004, along with concomitant intestinal degradation of IV β-lactam antibiotics, in a jejunum-fistula dog model (manuscript in preparation). 12,14 Here we report on 2 repeat-dose studies designed to evaluate the toxicity potential of SYN-004 in enteric-coated pellets when administered orally, either alone or in combination with IV ceftriaxone. These studies also assessed the plasma levels of SYN-004 and its effect on the PK of IV ceftriaxone.
Materials and Methods
Test Article
SYN-004, an enzyme of ∼29 kDa, was expressed in a recombinant Escherichia coli system and then purified by a contract manufacturing organization. The purified SYN-004 bulk protein was formulated as enteric pellets designed to release active enzyme when the pH rises above 5.5 in the intestine. 12 Each enteric-coated pellet contained ∼14.2% SYN-004 (active enzyme). Placebo pellets contained formulation buffer and the other excipients of the active pellets but no active drug. The appropriate amounts of SYN-004 or placebo pellets (pellets weighing approximately 1 mg each) were weighed and placed in size 12 gelatin capsules for oral dosing. The SYN-004 enteric pellets and placebo pellets were shown to be stable over the course of the 2 studies when stored at 2°C to 8°C (data not shown).
Chemicals
Ceftriaxone sodium (ROCEPHIN) 18 was purchased from a commercial supplier. The ceftriaxone was resuspended in sterile water for injection, United States Pharmacopeial Convention (USP), and then diluted in 0.9% saline prepared with sterile water for injection, USP, and sodium chloride, granular, USP. This saline was also used as the vehicle control in the second study.
Animals
Two studies to assess the toxicity of SYN-004 alone and when coadministered with ceftriaxone were performed in groups of healthy male and female Beagle dogs (Marshall BioResources, North Rose, New York). The dogs were approximately 5 to 6 months old at receipt (males 6.4-9.0 kg; females 5.3-7.6 kg), deemed appropriate for use by a laboratory veterinarian, and acclimated for 2 weeks prior to placing on study.
Both studies were conducted in compliance with the U.S. Food and Drug Administration (FDA) Good Laboratory Practice (GLP) Regulations (21 CFR Part 58) at WIL Research (Ashland, Ohio). All dogs were maintained in accordance with the Guide for the Care and Use of Laboratory dogs. 19 The facilities at WIL Research are fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International.
Test Article Administration
In both studies, the prepared SYN-004 capsules or placebo capsules were administered 3 times daily (approximately 4 hours ± 15 minutes apart) to all dogs for 14 or 28 consecutive days. To decrease potential variability due to stomach and gallbladder emptying, approximately 100 g of feed was offered approximately 30 minutes prior to the first dose of the day (dose 1), approximately 100 g of feed was offered approximately 30 minutes prior to the second dose of the day (dose 2), and approximately 200 g of feed was offered approximately 30 minutes prior to the third dose of the day (dose 3). The dogs were allowed access to the remaining food for a period of 1 hour following dosing. Following each dose administration, a 10-mL flush of deionized water was administered orally to each dog.
Parameters Evaluated
In both studies, the dogs were observed twice daily for mortality and moribundity. Observations were performed daily, and detailed physical examinations were performed once a week. Individual body weights were recorded weekly (± 2 days), and food consumption was reported daily. Clinical pathology parameters (hematology, coagulation, serum chemistry, and urinalysis) were analyzed prior to the initiation of dose administration, at the end of the dosing period, and, for the 28-day study, at the end of the recovery period. In addition, during the 28-day study, ophthalmic examinations and electrocardiograms (ECG), heart rate, and waveform intervals (PR, QRS, RR, QT, and QTcV) were recorded predose and during the last week of the study. For both studies, a complete set of necropsies was conducted on all dogs, including a detailed assessment of the GI tract. In both studies, a complete set of organs were weighed and tissues collected, routinely processed, and examined microscopically. Final examinations/assessments were performed by veterinarians with board certification in their responsible areas.
Experimental Design: 28-Day Oral Repeat Dose (3 times per day)
A 28-day study with SYN-004 administered orally 3 times per day, with a 14-day recovery period, was conducted to evaluate the toxicity and toxicokinetics (TK) of SYN-004. All dogs were dosed for 28 days and then euthanized either the day after dosing or after a 14-day recovery period. The dogs received either 6.6 mg/kg/day, 18 mg/kg/day, or 57 mg/kg/day of SYN-004 split into 3 equal doses per day (Table 1). Dogs in the control group (group 1) received placebo pellets in capsules containing the same number of pellets administered to the high-dose group (1 capsule/dog, 3 times daily). The high dose of 57 mg/kg/day was based on the highest no-observed-adverse-effect level (NOAEL) of the major excipient (Eudragit L30 D-55, Evonik Industries AG, Essen Germany) which has been documented to cause intestinal lesions in rats and dogs at high doses in repeat-dose toxicity studies. 20 In dogs, the Eudragit NOAEL of 100 mg/kg/day was based on diarrhea and submucosal intestinal fibrosis. 20
Groups for 28-Day SYN-004 Oral Administration Study.
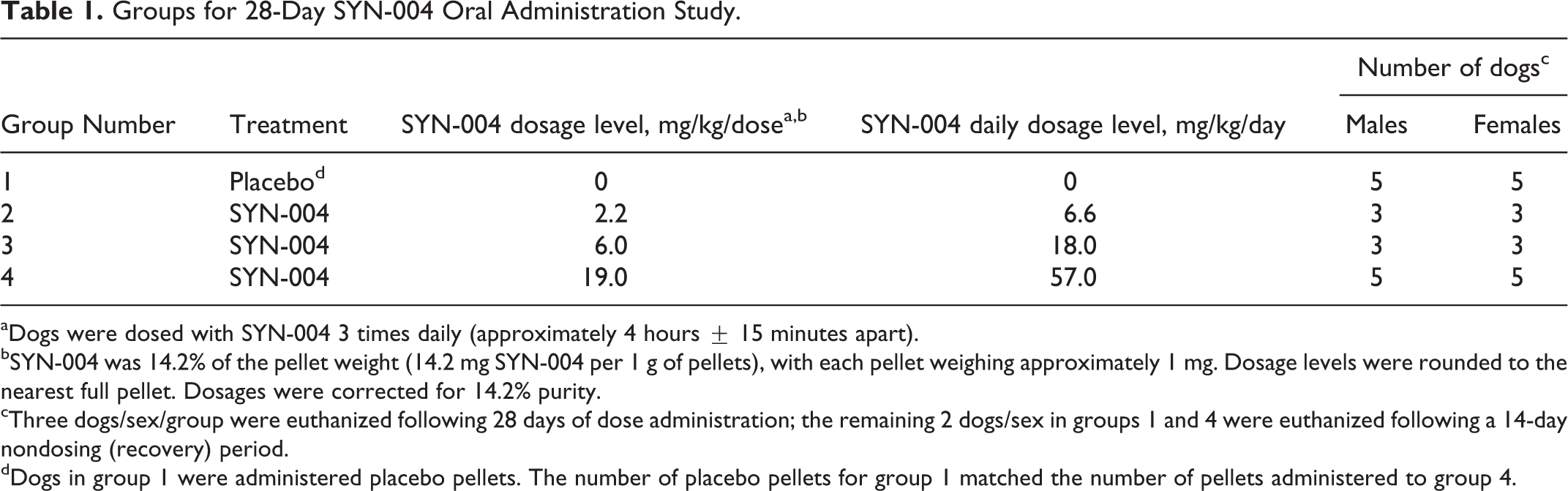
aDogs were dosed with SYN-004 3 times daily (approximately 4 hours ± 15 minutes apart).
bSYN-004 was 14.2% of the pellet weight (14.2 mg SYN-004 per 1 g of pellets), with each pellet weighing approximately 1 mg. Dosage levels were rounded to the nearest full pellet. Dosages were corrected for 14.2% purity.
cThree dogs/sex/group were euthanized following 28 days of dose administration; the remaining 2 dogs/sex in groups 1 and 4 were euthanized following a 14-day nondosing (recovery) period.
dDogs in group 1 were administered placebo pellets. The number of placebo pellets for group 1 matched the number of pellets administered to group 4.
Plasma (K2EDTA) samples for analysis of SYN-004 were collected from all dogs from blood taken at approximately 0.5, 1, 2, and 4 hours (prior to the second daily dose) after the first dose, approximately 1 and 4 hours (prior to the third daily dose) after the second dose, and approximately 1 hour following the third dose. This blood sampling occurred on the first and last day of dosing.
Experimental Design: 14-Day Oral Repeat Dose (3 times per day) of SYN-004 Coadministered With IV Ceftriaxone (once per day)
A 14-day toxicity study was conducted to evaluate the toxicity of orally delivered SYN-004 (3 times per day) when coadministered with IV ceftriaxone (once per day) and to evaluate the PK of ceftriaxone. SYN-004 pellets were administered orally in gelatin capsules 3 times daily for 14 consecutive days to 2 groups of dogs at 6.6 or 57 mg/kg/day (2.2 and 19 mg/kg/dose, respectively) for groups 2 and 3, respectively (Table 2). These SYN-004-treated groups were coadministered with IV ceftriaxone (300 mg/kg/day, 5 mL/kg) given once daily by slow bolus injection, 15 minutes after the first daily dose of SYN-004. Two concurrent control groups were similarly treated with either placebo pellets in gelatin capsules and IV saline (group 1) or with gelatin capsules only and IV 300 mg/kg/day ceftriaxone (group 4). The dosage level of ceftriaxone was at or near the maximum tolerated repeat-dose level in dogs based on nonproprietary information in the public domain 18 and on a preliminary ceftriaxone range-finding study (data not shown).
Groups for 14 Day SYN-004 and Ceftriaxone Co-administration Study.
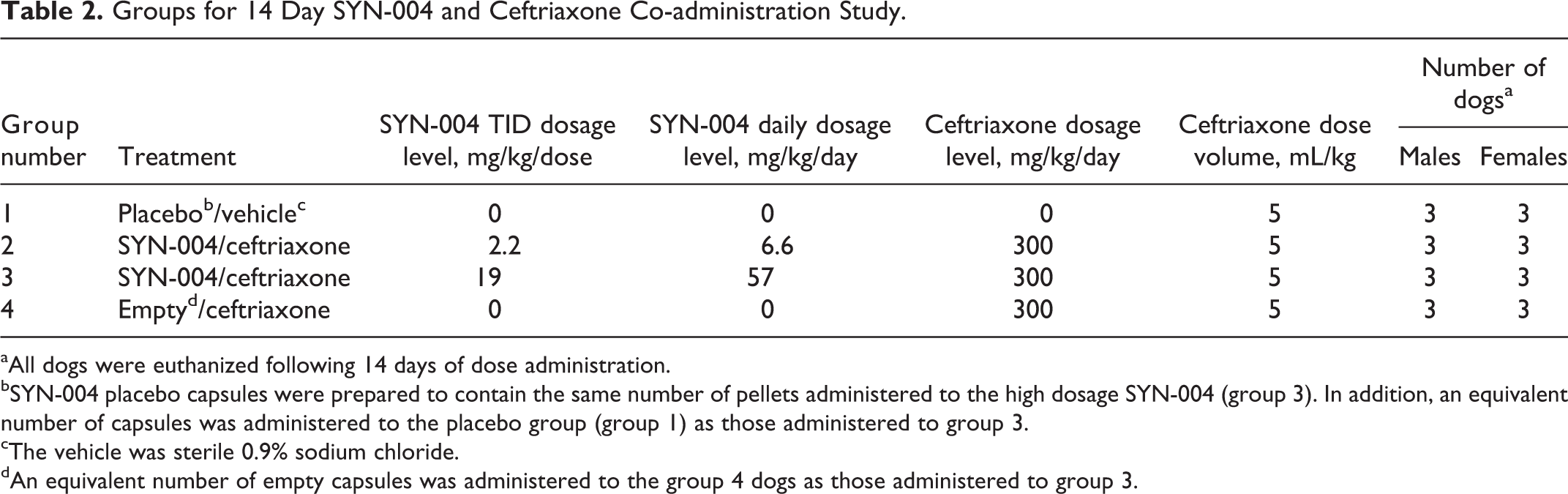
aAll dogs were euthanized following 14 days of dose administration.
bSYN-004 placebo capsules were prepared to contain the same number of pellets administered to the high dosage SYN-004 (group 3). In addition, an equivalent number of capsules was administered to the placebo group (group 1) as those administered to group 3.
cThe vehicle was sterile 0.9% sodium chloride.
dAn equivalent number of empty capsules was administered to the group 4 dogs as those administered to group 3.
Plasma (K2EDTA) samples for ceftriaxone PK evaluation were collected from blood taken from all dogs after the first and last dose at approximately 0.083, 0.25, 0.5, 1, 2, 4, 8, and 24 hours following the start of the IV dose administration and immediately prior to the last dose. To prevent any possible degradation of plasma ceftriaxone by any potentially systemically absorbed SYN-004, 100 µg of tazobactam sodium salt (Sigma, St Louis, Missouri) in 10 µL volume was added to each plasma storage vial and the plasma was stored frozen (−65°C to −85°C) until analyzed. This concentration of tazobactam was shown to inhibit SYN-004 β-lactamase activity at the low plasma levels that might be expected based on the previous study (data not shown).
Analytic Assays
SYN-004 dog plasma method
SYN-004 in dog plasma was measured using an enzyme-linked immunosorbent assay (ELISA) with an anti-SYN-004 polyclonal antibody generated in rabbits for both the capture and the detection steps. Standards and quality control samples were made by spiking SYN-004 into dog plasma. Streptavidin-horseradish peroxidase (S-HRP) was used to detect the biotin bound to the captured SYN-004. Tetramethylbenzidine was used to react with the S-HRP conjugate to produce a colorimetric signal proportional to the amount of SYN-004 captured by the anti-SYN-004 antibody. The plate was read at 450 nm and the absorbance of the quality control and unknown samples converted to and regressed according to a 4 parameter linear regression model using SoftMax Pro GxP 5.0.1. The calibration range of the method was 0.8 ng/mL (lower limit of quantitation, LLOQ) to 25.6 ng/mL (upper limit of quantitation, ULOQ). Intra- and interassay accuracies were between 95% and 116%, and theoretical and precision coefficients of variation were <9% at all concentrations.
Ceftriaxone dog plasma method
Dog plasma was analyzed for ceftriaxone using a liquid chromatography with tandem mass spectrometric detection method. Dog plasma samples containing ceftriaxone were protein precipitated using acetonitrile and then separated on a Phenomenex Synergi Polar-RP, 4 μmol/L, 2 × 50 mm (P/N 00B-4336-B0) using 1% formic acid in water and 1% formic acid in methanol as the mobiles phases. Standards and quality control samples were made by spiking ceftriaxone into dog plasma. Ceftriaxone was detected using a Sciex API 4000, Positive Ionization. Cefotaxime (Sigma) was used as the internal standard. The calibration range of the assay was 10 to 5000 ng/mL. The intra-assay accuracy ranged from 113% to 118%, and intra-assay precision (% relative standard deviation) was <12%.
Data and Statistical Analyses
The PK/TK parameters and related descriptive statistics were calculated using noncompartmental methods as implemented in the Phoenix WinNonlin version 6.3 comprehensive TK/PK analysis software program (Pharsight Corporation; Mountain View, California). For ceftriaxone PK analysis, a 3-minute IV infusion model (the approximate time of the slow bolus injection) was used. Values below the lower limit of quantitation (BLQ) were assigned a value of zero. When all the values at a time point were BLQ, the mean was reported as BLQ. All plasma concentration data from all dogs were included in the analysis.
Body weight, body weight change, food consumption, clinical pathology, and organ weight data were subjected to a parametric 1-way analysis of variance (ANOVA) 21 to determine intergroup differences. Analyses were conducted using 2-tailed tests for minimum significance levels of 1% and 5%, comparing each test article-treated group to the control group by sex. If the ANOVA revealed statistically significant (P < .05) intergroup variance, Dunnett test 22 was used to compare the test article-treated groups. In the second study, for comparison of the antibiotic-treated group (group 4) with the control group (group 1), data were evaluated using the 2-sample t test. 23 These statistical tests were performed using a proprietary system (WTDMS, WIL Research).
Results
Twenty Eight-Day Oral Repeat Dose (3 times per day) of SYN-004
When SYN-004 was delivered orally 3 times per day (∼4 hours between doses) at 3 dosages, all dogs survived to the scheduled necropsies, and there were no test article-related gross toxicology findings observed. SYN-004 administration did not result in test article-related alterations in ophthalmic or ECG parameters, gross pathology findings, organ weights, clinical pathology parameters, or histologic findings, including no test article-related GI lesions.
Food consumption in the 57 mg/kg/day group males was generally lower throughout the study than for the control group with a 15% reduction in the mean food consumption (g/animal/day) across the 4 weeks of the study (Supplemental Figure 1). There was a corresponding transient lower cumulative body weight gain during the study with a 37% reduction in mean cumulative body weight change on day 28 as compared with day 0 in the 57 mg/kg/day group males (Supplemental Figure 2). During the 14-day recovery period, food consumption in the 57 mg/kg/day males returned to the same as food consumption in the control group males, but cumulative body weight gain continued to lag behind in this group through the end of the study with 31% difference in weight gain for the control group males versus the 57 mg/kg/day group males. These findings were not considered to be adverse due to the small magnitude of the changes. No test article-related effects on body weights or food consumption were noted in the 6.6 and 18 mg/kg/day group males and females or the 57 mg/kg/day group females.
Based on the results of this study, oral capsule administration of SYN-004 to dogs 3 times daily for 28 days resulted in a no-observed-effect level of 18 mg/kg/day for males and 57 mg/kg/day for females and an NOAEL of 57 mg/kg/day for the males and females. At this latter dosage level, test article-related effects were limited to transient, slight reductions in food consumption and body weight gain in males.
Plasma Levels of SYN-004
SYN-004 administration resulted in low and sporadic plasma levels of SYN-004 that precluded a rigorous assessment of TK parameters (Table 3). Where measurable, most plasma SYN-004 concentrations were within approximately 1- to 4-fold of the LLOQ (0.80 ng/mL). There were no individual trends, obvious sex-related differences, or differences between day 0 and day 27 values.
Mean Plasma Concentrations of SYN-004 after 28 Day Oral Administration in Dogs.a
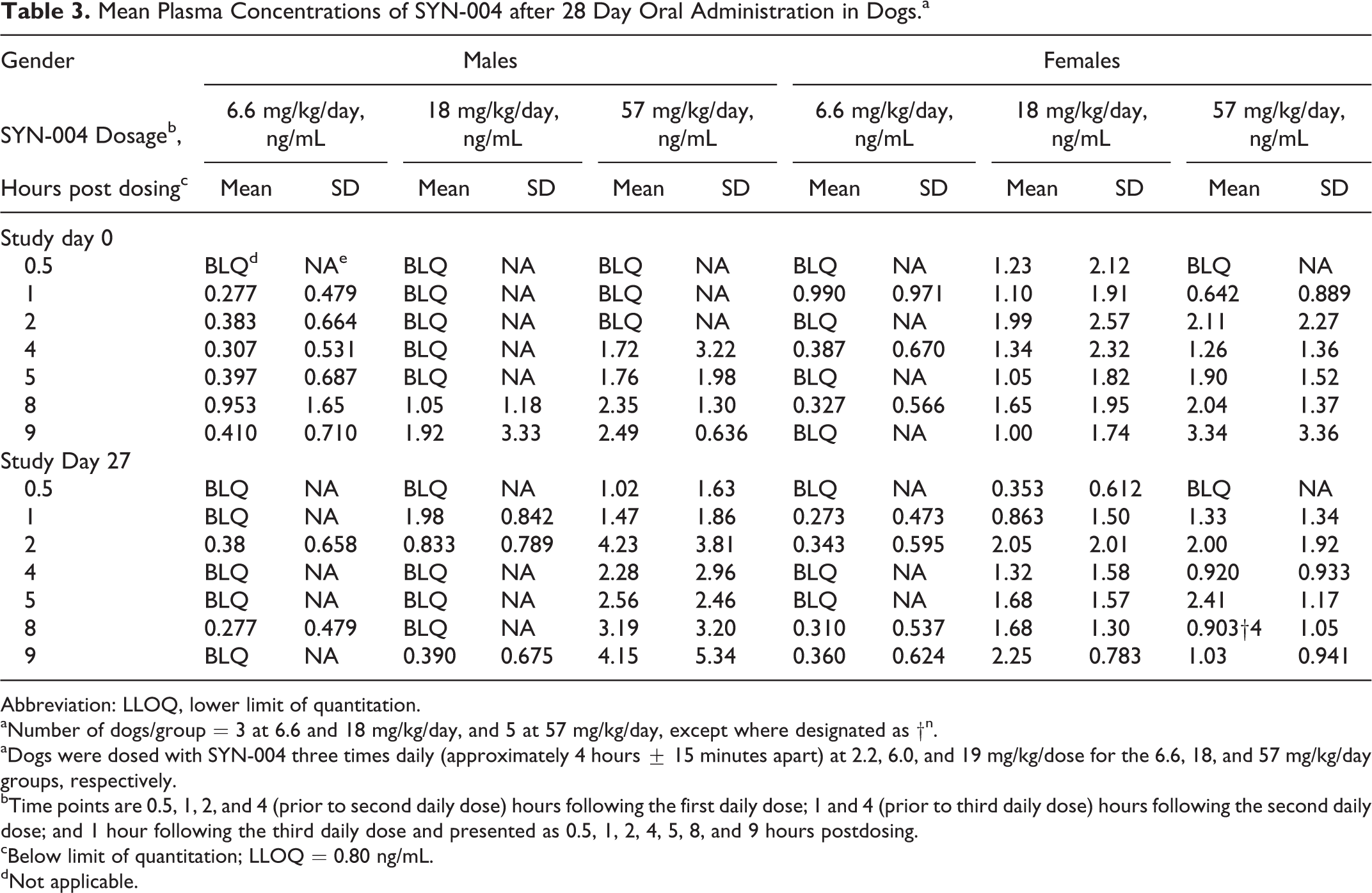
Abbreviation: LLOQ, lower limit of quantitation.
aNumber of dogs/group = 3 at 6.6 and 18 mg/kg/day, and 5 at 57 mg/kg/day, except where designated as †n.
aDogs were dosed with SYN-004 three times daily (approximately 4 hours ± 15 minutes apart) at 2.2, 6.0, and 19 mg/kg/dose for the 6.6, 18, and 57 mg/kg/day groups, respectively.
bTime points are 0.5, 1, 2, and 4 (prior to second daily dose) hours following the first daily dose; 1 and 4 (prior to third daily dose) hours following the second daily dose; and 1 hour following the third daily dose and presented as 0.5, 1, 2, 4, 5, 8, and 9 hours postdosing.
cBelow limit of quantitation; LLOQ = 0.80 ng/mL.
dNot applicable.
Fourteen-Day Oral Repeat Dose (3 times per day) of SYN-004 Coadministered With IV Ceftriaxone (once per day)
SYN-004 is intended to degrade excess IV β-lactam antibiotics that are excreted into the intestine via the bile, thus it was necessary to determine the safety of SYN-004 when coadministered with an IV β-lactam antibiotic. Ceftriaxone was chosen as a representative β-lactam antibiotic for this study because it is widely used and highly associated with CDI 1 which is the intended indication for SYN-004.
When SYN-004 was administered orally 3 times per day (∼4 hours between doses) at 2 dosages with once-daily IV ceftriaxone (300 mg/kg) 15 minutes after the first SYN-004 dose of the day, all dogs survived to the scheduled necropsy. There were no test article-related effects on body weight, food consumption, clinical pathology parameters, or organ weights. There were also no treatment-related macroscopic or microscopic findings, including no treatment-related GI lesions.
An increased frequency of fecal changes (soft feces, mucoid feces, and diarrhea) was seen in all IV ceftriaxone-dosed groups as compared with the group that received vehicle alone. An increased incidence of emesis was also noted in IV ceftriaxone-treated groups coadministered either the gelatin capsules alone (group 4) or SYN-004 (group 3). At 1 to 2 hours after ceftriaxone administration, one of the dogs from group 3 that displayed emesis also had a swollen/redden facial area/ears. This observation was transient, only seen after the first dose, and no further incidences occurred for the remainder of the study. Retching, emesis, head shaking, ear scratching, erythema, edema around the eyes and snout, and sporadic panting and licking, all have been observed previously in individual Beagle dogs administered IV ceftriaxone. 18
Plasma Levels of Ceftriaxone with Coadministration of SYN-004
Following IV administration of ceftriaxone with or without administration of SYN-004 capsules, plasma ceftriaxone concentrations were measurable in all ceftriaxone-treated dogs from 5 minutes through 24 hours postdose after the first dose (study day 0) and from predose through 24 hours postdose after the last dose (study day 13; Figure 1). Plasma ceftriaxone concentrations decreased gradually following the end of dose administration in a biphasic manner through 24 hours postdose. Following IV administration of ceftriaxone with or without administration of SYN-004 capsules, exposure to ceftriaxone was similar in male and female dogs on both evaluation days (Table 4). In terms of area under curve (AUC)0-24, exposure to ceftriaxone was similar across all ceftriaxone-treated groups with or without administration of SYN-004. As also shown in Table 4, exposure to ceftriaxone, in terms of AUC0-24, was similar after the first and last dose for both genders across all groups. Accumulation ratios (study day 13 AUCinf/study day 0 AUCinf) ranged from 0.922 to 1.28 at both dose levels. The estimated clearance (Cl = Ceftriaxone Dose/AUCinf) and volume of distribution at steady state (Vss = mean residence time [MRT]inf × Cl) were low and the T½ ranged from 1.59 to 1.79 hours. Clearance ranged from 4.70 to 6.55 mL/min/kg, which is <30% of average hepatic blood flow in dogs (30.9 mL/min/kg 24 ), and Vss ranged from 0.288 to 0.404 L/kg, which is slightly lower than the average total body water volume in dogs (0.604 L/kg 24 ). Thus, there was no obvious relationship between SYN-004 dose and ceftriaxone exposure (Figure 1).
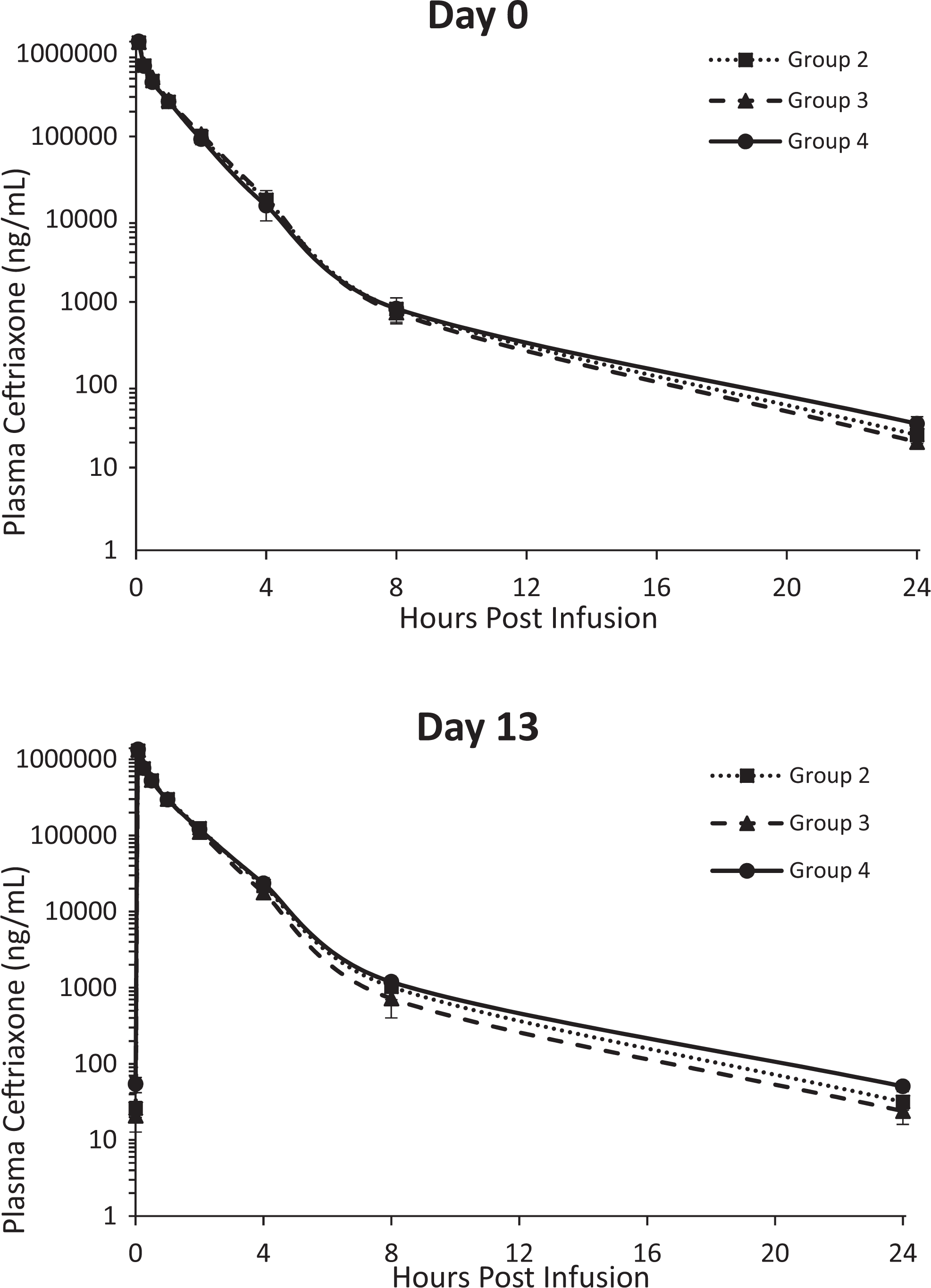
Plasma concentrations of ceftriaxone on days 0 and 13 of the 14-day study. Plasma samples were drawn at various times (hours) postceftriaxone infusion, and the concentrations of the ceftriaxone were determined with a validated liquid chromatography with tandem mass spectrometric detection method. Mean ceftriaxone concentrations are shown for each treatment group ± standard deviation. Data from group 1 are not displayed, as there were no measurable ceftriaxone in any of these plasma samples. Group 2, 6.6 mg/kg/day SYN-004 and 300 mg/kg/day ceftriaxone; Group 3, 57 mg/kg/day SYN-004 and 300 mg/kg/day ceftriaxone; Group 4, 0.0 mg/kg/day SYN-004 and 300 mg/kg/day ceftriaxone.
Summary of TK Parameters for Ceftriaxone.
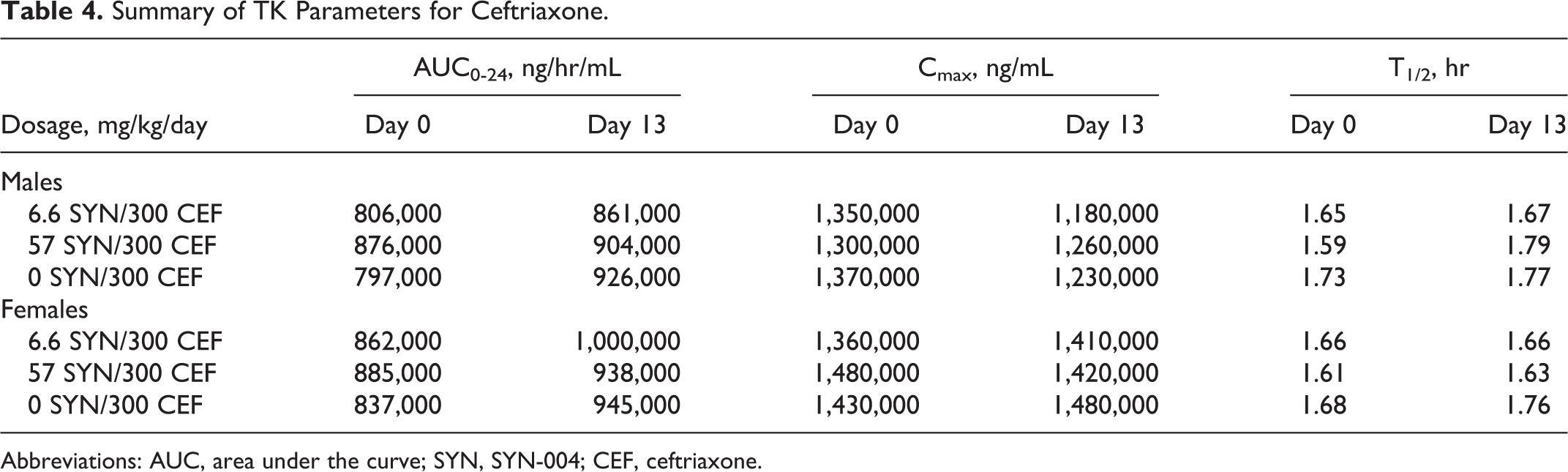
Abbreviations: AUC, area under the curve; SYN, SYN-004; CEF, ceftriaxone.
Discussion
SYN-004 is a modified version of P1A which was under development for a similar indication, and much of the preclinical and early safety work for SYN-004 is based on P1A. 11 –14 Early preclinical work established that oral P1A reduces intestinal levels of IV administered ampicillin in a dose-dependent manner in the intestines of dogs as determined in a jejunum-fistulated dog model. 12 β-lactamase activity is detectable in the dog jejunum for at least 3 hours postadministration of P1A, establishing the stability of the enzyme in the harsh environment of the intestine. 12 Further work in this model demonstrated that while P1A significantly reduces intestinal ampicillin levels and prevents ampicillin-induced changes to the intestinal microbiome of the dogs, serum levels of ampicillin remain similar in P1A-treated and untreated dogs. 14 Furthermore, treatment with P1A appears to reduce the emergence of antibiotic (ampicillin) resistance in the intestinal bacteria. 14 Similarly, excellent intestinal bioavailability of SYN-004 was shown in the jejunum-fistulated dog model after oral administration of a formulated dose of SYN-004, and this bioavailability is associated with intestinal degradation of IV ceftriaxone (manuscript in preparation).
In the present studies, we investigated the potential toxicity of SYN-004 alone and when coadministered with ceftriaxone in dogs, and studied the influence of SYN-004 on plasma levels of ceftriaxone in this species. These GLP studies were intended to be Investigational New Drug enabling to demonstrate the safety of SYN-004 and allow it to progress into clinical trials in humans.
For SYN-004 to be a viable drug, it needs to be safe and effective, and it also needs to confine its enzymatic activity to the GI tract so as to not reduce the effectiveness of systemic antibiotics for treating the primary bacterial infection. Thus, the plasma PK of IV antibiotics should not be affected by administration of oral SYN-004. In the 28-day, repeat-dose study (3 doses per day), dogs received 6.6, 18, or 57 mg/kg/day SYN-004 divided into 3 equal oral doses. The maximum dose was limited not by the SYN-004 active ingredient but rather by the maximum tolerated repeat-dose level previously demonstrated by the main excipient of the formulation, the protective enteric coat. 20 All 3 doses of SYN-004 were shown to be safe and well tolerated with all dogs surviving to terminal necropsies. There were no SYN-004-related macroscopic or microscopic findings. The only finding of note was decreased food consumption (Supplemental Figure 1) and weight gain (Supplemental Figure 2) by the male dogs at 57 mg/kg/day. This was not considered to be an adverse reaction due to the transient nature of the body weight effect and small magnitude of the change. There were no other differences noted between male and female dogs, recorded by any measured parameter. A dose of 57 mg/kg/day is equivalent to almost 4 g/day of SYN-004 in a 70-kg adult human and provides an excellent margin of safety for human clinical trials where clinical doses are expected to be below 1 gram per day.
SYN-004 is designed to degrade excreted β-lactam antibiotics in the intestine and, as a protein of ∼29 kDa, 13 would not be expected to be significantly systemically absorbed when given orally. Indeed, systemic absorption of active SYN-004 at concentrations that could degrade systemic antibiotics would be highly undesirable. An ELISA assay for detection of SYN-004 in dog plasma found only sporadic and low-level SYN-004 in the dog plasma samples across all dose levels (Table 3). Plasma SYN-004 concentrations were either BLQ (< 0.8 ng/mL) at all time points or were generally only measurable at intermittent points in each dog. Where measurable, most plasma SYN-004 concentrations were within approximately 1- to 4-fold of the LLOQ of the assay. Overall, exposure in individual dogs was highly variable as indicated by relatively high standard deviations (Table 3). In summary, consistent with what was expected for a protein and with previous reports from P1A, 12,14 systemic absorption of SYN-004 was low and sporadic in dogs.
Since SYN-004 will be orally administered to patients receiving treatment with IV β-lactam antibiotics, it was also important to ensure that these 2 drugs can be administered together safely. Coadministration of oral SYN-004 at 6.6 or 57 mg/kg/day (split into 3 equal doses) with once-daily IV ceftriaxone at 300 mg/kg/day for 14 consecutive days was safe and well tolerated with all dogs surviving to necropsy. Human dosing with ceftriaxone is an IV infusion of 1 g to 2 g of ceftriaxone given once a day for the duration of treatment. 18 Therapy is to be continued for at least 2 days after the signs and symptoms of infection have disappeared, and the usual duration of therapy is 4 to 14 days. 18 There were no test article-related effects on body weight, food consumption, clinical pathology parameters, or organ weights, and there were no test article-related macroscopic or microscopic findings. Clinical observations related to ceftriaxone were observed and included increased frequency of fecal changes (soft feces, mucoid feces, and/or diarrhea). An increased incidence of emesis and fecal changes were noted in dogs receiving ceftriaxone either with or without SYN-004, and thus these changes were considered related to ceftriaxone 18 rather than to SYN-004. One dog only in group 3 exhibited an infusion-type reaction, also a known consequence of ceftriaxone administration. 18
Coadministration of SYN-004 with ceftriaxone also did not alter the plasma PK of ceftriaxone in dogs (Table 4), which was consistent with the sporadic and low levels of SYN-004 found in dog plasma being either nonfunctional enzyme (ie, ELISA was not detecting active, full-length enzyme), or of insufficient concentration to significantly change the PK of ceftriaxone (Figure 4). This finding was consistent with previous observations of P1A and ampicillin in dogs. 14 The PK parameters (Vss and Cl) generated for ceftriaxone were similar to those following an IV dose of 50 mg/kg, 25 while the AUC values corrected for free plasma levels of ceftriaxone were about 5- to 6-fold above the reported human AUC. 18,26 Most importantly, the findings from this study showed that high doses of SYN-004 can be orally administered with IV antibiotics and do not reduce the systemic plasma concentration of the antibiotic, an important attribute of a successful therapy to treat the primary infection.
Results from these 2 toxicity studies have demonstrated that oral administration of high doses of SYN-004 was safe and well tolerated even when coadministered with high doses of ceftriaxone. As expected for a protein, absorption of oral SYN-004 from the intestine appeared to be minimal and sporadic and did not affect the PK of coadministered ceftriaxone. These results support the initiation of human clinical studies with SYN-004 for degradation of biliary-excreted IV β-lactam antibiotics in the intestine, with the intent of protecting the gut microbiome and ultimately preventing CDI, other opportunistic infections, and antibiotic-associated diarrhea.
Footnotes
Acknowledgments
The authors would like to thank Sevan Brodjian and the technical staff at WIL Research (Skokie, IL and Ashland, OH) and Raj Mangaraj, Sara Brady, Kelly Bourdon and the technical staff at Q2 Solutions (formerly Quintiles, Ithaca, New York) for their excellent technical assistance with these studies.
Author Contribution
Kokai-Kun, J. contributed to conception and design and acquisition, analysis, and interpretation, drafted the manuscript; and critically revised the manuscript. Bristol, A. contributed to conception and design and acquisition, analysis, and interpretation and critically revised manuscript. Setser, J. contributed to design and acquisition, analysis, and interpretation and critically revised manuscript. Schlosser, M. contributed to conception and design and acquisition, analysis, and interpretation and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. John Kokai-Kun and Andrew Bristol are employees of Synthetic Biologics, Inc. Michael Schlosser is a paid consultant for Synthetic Biologics, Inc. John Setser is a paid contractor for Synthetic Biologics, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Synthetic Biologics, Inc.
