Abstract
Thallium (Tl+) is a toxic heavy metal capable of increasing oxidative damage and disrupting antioxidant defense systems. Thallium invades the brain cells through potassium channels, increasing neuronal excitability, although until now the possible role of glutamatergic transmission in this event has not been investigated. Here, we explored the possible involvement of a glutamatergic component in the Tl+-induced toxicity through the N-methyl-
Introduction
Thallium (Tl+) is toxic heavy metal acting at different levels of cell metabolism. Its ability to interfere with a variety of potassium-dependent processes is thought to play a significant role in the generation of its toxic effects. For instance, Tl+ affinity for Na+K+-ATPase is 10 times higher than that of potassium. 1,2 Thallium also accumulates in mitochondria and acts as an oxidative phosphorylation uncoupler. 3 Mitochondria are considered the major source of reactive oxygen species (ROS) production and key contributors to neurodegenerative diseases. 4 Although the reduced form of the tripeptide glutathione (GSH) is probably the most important endogenous antioxidant involved in the cell defense against oxidative damage to mitochondria and other organelles by ROS, this and other antioxidant systems might be affected by metal-induced toxicity. Glutathione modulates the disposal of exogenous peroxides by acting as a cosubstrate in a reaction catalyzed by GSH peroxidase (GPx). 5 In addition, GSH has been shown to play a crucial role in rescuing cells from apoptosis due to fluctuations in the redox environment of cells 6 ; therefore, the evaluation of its role in neurotoxic models produced by metals is of major relevance.
In particular, oxidative stress mediated by heavy metals can cause neurodegeneration. In this regard, excessive ROS formation by toxicants is known to cause a Bcl-2-related Bax activation, which “makes holes” in the outer mitochondrial membrane, releasing cytochrome c. Using the energy provided by adenosine triphosphate (ATP), the released cytochrome c binds to a proapoptotic protease activating factor 1, followed by the aggregation of these complexes to form apoptosomes, complexes that bind to the active proteases, such as caspase 9 and others, which activate other “executive” caspases (3 and 7), ultimately leading to digestion of structural proteins in the cytoplasm and DNA degradation. 7 This mechanism could account for Tl+ toxicity, as evidenced in hepatocytes and isolated rat liver mitochondria, where Tl+ decreases the levels of GSH and alters the mitochondrial membrane potential, while it increases ROS formation and caspase 3 activation, also releasing cytochrome c from mitochondria to induce apoptosis. 8 –11 In PC12 cells, this metal causes high mitochondrial H2O2 generation and depolarization, as well as Bax oligomerization and activation of caspases 9 and 3, while it increases the Fas content and caspase 8 activity. 12,13 In vivo studies have shown that Tl+ accumulates in different brain regions, producing lipid peroxidation (LPx) and changes in behavioral tasks. 14 However, the specific mechanisms for the toxic pattern exerted by Tl+ in the brain remain unsolved and merit additional approaches. In this regard, no previous studies have explored the participation of the glutamatergic transmission on the brain damage as a nonexcluding alternative toxic mechanism for this metal in the central nervous system (CNS). Since Tl+ affects the mitochondrial function, it cannot be discarded that secondary excitotoxic events due to ATP depletion and further voltage-gated activation of glutamatergic receptors might contribute to the toxic pattern exerted by this metal, as it occurs for other mitochondrial neurotoxins such as 3-nitropropionic acid (3-NP). 15 –18
Among the different routes for Ca2+ influx in neuronal cells, the N-methyl-
The aim of the present study was to characterize the effects of MK-801, as a pretreatment, on the Tl+-induced neurotoxicity in rats, including behavioral and biochemical gross end points such as the altered motor activity, the regional changes in LPx, the levels of GSH, and the GPx activity, all in order to provide preliminary in vivo evidence on a possible involvement of the glutamatergic transmission in the toxic pattern elicited by Tl+ in the brain.
Materials and Methods
Reagents
All chemicals used in this work were obtained from Sigma-Aldrich (Mexico) and other well-known commercial sources.
Animals and Treatments
Experiments were carried out using male Wistar rats (250-280 g) provided by the closed breeding colony at the Instituto Nacional de Neurología y Neurocirugía (INNN). The animals were housed 5 per cage in acrylic cages and provided with Rodent Chow (Purina, St Louis, Missouri) and water ad libitum. All rats were maintained under conditions of constant temperature (25 ± 3°C), humidity (50 ± 10%), and lighting (12:12 light–dark cycle). All experimental manipulations were performed according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals, the Principles of the Mexican Official Ethics Standard 062-ZOO-1999, and in compliance with the guidelines and principles stated by The American College of Toxicology Policy on the Use of Animals in Toxicology. A total of 40 animals were used throughout the study and randomly assigned to one of the following groups (n = 10 rats per group): (1) control (sterile saline [SS] + SS) group: rats were administered twice with SS as vehicle via ip with a 30-minute interval; (2) Tl+ (SS + Tl+) group: rats received a first administration of SS, followed 30 minutes later by a single Tl+ acetate administration (32 mg/kg, ip, LD50) 25 ; (3) MK-801 (MK-801 + SS) group: rats were administered with MK-801 (1 mg/kg, ip), 26 followed by a second administration of SS 30 minutes later; and (4) MK-801 + Tl+ group: animals received MK-801 30 minutes before the injection of Tl+ acetate. The rationale for the selection of the doses for both drugs was based on observations of their effects in previous studies. 25,26
The motor activity of animals from all groups was recorded 24 hours after the last drug administration, and immediately thereafter, all rats were sacrificed by decapitation for biochemical purposes. Brains were dissected into 5 different regions: frontal cortex (Cx), cerebellum (Ce), hypothalamus (Ht), hippocampus (Hp), and striatum (S). These regions were dissected according to the method described by Glowinski and Iversen. 27
Locomotor Activity Assessment
All animals from the 4 different experimental groups were subjected to the assessment of spontaneous motor activity in an open-field device. An automated VersaMax Open Field Activity Monitor (AccuScan Instruments Inc, Columbus, Ohio) was used. All experiments were carried out between 6:00
From this point on, each group was split into 2 subgroups: the first one (n = 5) was used for Tl+ content analysis (brain regions from the left side) and LPx assay (brain regions from the right side). The second half group (n = 5) was used to estimate the GSH levels (regions from the left side) and the GPx activity (regions from the right side).
Thallium Tissue Content Analysis by Atomic Absorption
Tissue samples from different brain regions of rats receiving vehicle or Tl+ were obtained by dissection and digested in 0.5 to 1.0 mL of concentrated HNO3 Suprapur, handled and analyzed as described previously, 28 using a Graphite Furnace Atomic Absorption Spectrophotometer AA 600 Perkin-Elmer (Perkin-Elmer Life and Analytical Sciences, Shelton, CT) with an autosampler (AS 800, Waltham, Massachusetts) and a Tl+ hollow cathode lamp (276 nm of wavelength and slit of 0.7 nm). Continuous argon flow (10 mL/min) served as purge gas for the analysis. Results were expressed as µg of Tl+ per g of wet tissue.
Lipid Peroxidation Assay
Tissue samples of different brain regions from all rat groups were used for fluorometric detection of lipid fluorescent products at 370 nm excitation and 430 nm emission wavelengths, using a Perkin-Elmer Luminescence Spectrometer LS50B (Waltham, Massachusetts), according to our previous report. 14 This is a simple and accurate method to obtain relative levels of semistable peroxidized products. Tissue samples were collected 24 hours after the last Tl+ administration (immediately after subjected to the motor activity test) and homogenized in 3 mL of saline solution. Aliquots of 1 mL were added to a 4 mL chloroform–methanol mixture (2:1 vol/vol). After vortexing (10 seconds), the mixture was ice-cooled for 30 minutes to allow phase separation, and the chloroform layer fluorescence was then measured. The equipment sensitivity was adjusted to 300 arbitrary units of fluorescence with a quinine standard solution (0.1 µg/mL). Results were expressed as relative fluorescence intensity units per mg of protein.
Detection of Total GSH by Fluorometric Assay
The levels of GSH in brain regions were determined fluorometrically, as previously described, 29 with some discrete modifications. Briefly, tissue samples were homogenized in 3.75 mL EDTA-phosphate buffer (pH 8.0) plus 1 mL HPO3 (25%). Homogenates were centrifuged at 3000×g for 15 minutes, and supernatants were separated to assay GSH. Five hundred microliters of supernatant aliquots were added to 4.5 mL phosphate-EDTA buffer. An aliquot of 100 µL was added to 1.8 mL of phosphate-EDTA buffer plus 100 µL o-phthalaldehyde (1 mg/mL). Mixtures were incubated at room temperature for 15 minutes. Each sample was measured at 350 nm excitation and 420 nm emission wavelengths in a Perkin-Elmer LS50B luminescence spectrophotometer. Final results were expressed as µg of GSH per mg of protein.
Glutathione Peroxidase Activity
Glutathione peroxidase activity was determined using a method based on the GSH nonenzymatic oxidation, as reported previously, 30 and modified by us. 31 Briefly, tissue samples were homogenized in phosphate buffer (pH 7.4). Aliquots (30 µL) were incubated at 37ºC in 0.4 mol/L sodium phosphate buffer (pH 7.0) also containing 0.4 mmol/L EDTA, 2.0 mmol/L GSH, and 0.01 mol/L sodium azide (NaN3; final volume 2.0 mL). Five minutes later, 1 mL of 1.25 mmol/L H2O2 was added to the incubation medium, and 3 minutes later, 1 mL of the mixture was removed and added to 4.0 mL of metaphosphoric acid. Samples were centrifuged at 1500×g for 15 minutes, and supernatants (1 mL) were added to a phosphate buffer (pH 7.0) plus EDTA solution also containing 0.5 mL 5,5-dithiobis-2-nitrobenzoic acid (DTNB). Optical density was determined at 512 nm in a Perkin-Elmer Lambda 20 spectrophotometer 2 minutes after the addition of DTNB. A standard curve was constructed using increasing concentrations of GSH + phosphate buffer/EDTA + DTNB. An enzyme unit of activity was defined as a 50% decrease of GSH per 60 minutes after the decrease in GSH from nonenzymatic reaction had been subtracted. Results were expressed as units of activity per mg of protein.
Determination of Protein Content
Protein content in tissue samples used in all experiments was measured by the Folin and Ciocalteu phenol reagent. 32
Statistical Analysis
Data were expressed as mean values ± standard error of the mean (SEM). Statistical evaluations were performed using paired Student t test and 1-way analysis of variance (ANOVA) followed by Tukey post hoc test, and normal distribution was assumed. If pertinent, biochemical results were first explored using a 2-way ANOVA, and behavioral tests were analyzed with repeated-measures ANOVA. The minimal level of significance chosen was P < .05. Each data represent the measurements of 10 (for motor activity tests) or 5 rats (for biochemical results) per treatment.
Results
Pretreatment With MK-801 Ameliorated the Tl+-Induced Hyperactivity in Rats Evidenced as Horizontal but Not as Vertical Movements
Two locomotor variables were evaluated in rats from all experimental groups 24 hours after treatment: horizontal and vertical movements. The time sequence of motor events (activity recorded every 5 minutes for 60 minutes total) is shown in Figure 1. Thallium administration (SS + Tl+ group) produced a significant increase in locomotor activity, particularly in horizontal movements, since frequency counts increased between 5 and 40 minutes (Figure 1A) when compared with the SS + SS group. These changes clearly denoted an early pattern of hyperactivity. After 40 minutes, no significant differences were observed among these 2 groups. The Tl+-induced changes in horizontal movements were significantly attenuated by pretreatment with MK-801 (MK-801 + Tl+ group), which reached values close to the SS + SS (control) group in almost all time points evaluated, except for the point at 25 minutes. MK-801 per se (MK-801 + SS group) presented moderate but no significant changes in horizontal activity when compared with the SS + SS group. In regard to vertical activity (Figure 1B), no significant differences were observed among groups (Figure 1B), nor even at 10 minutes of recording.
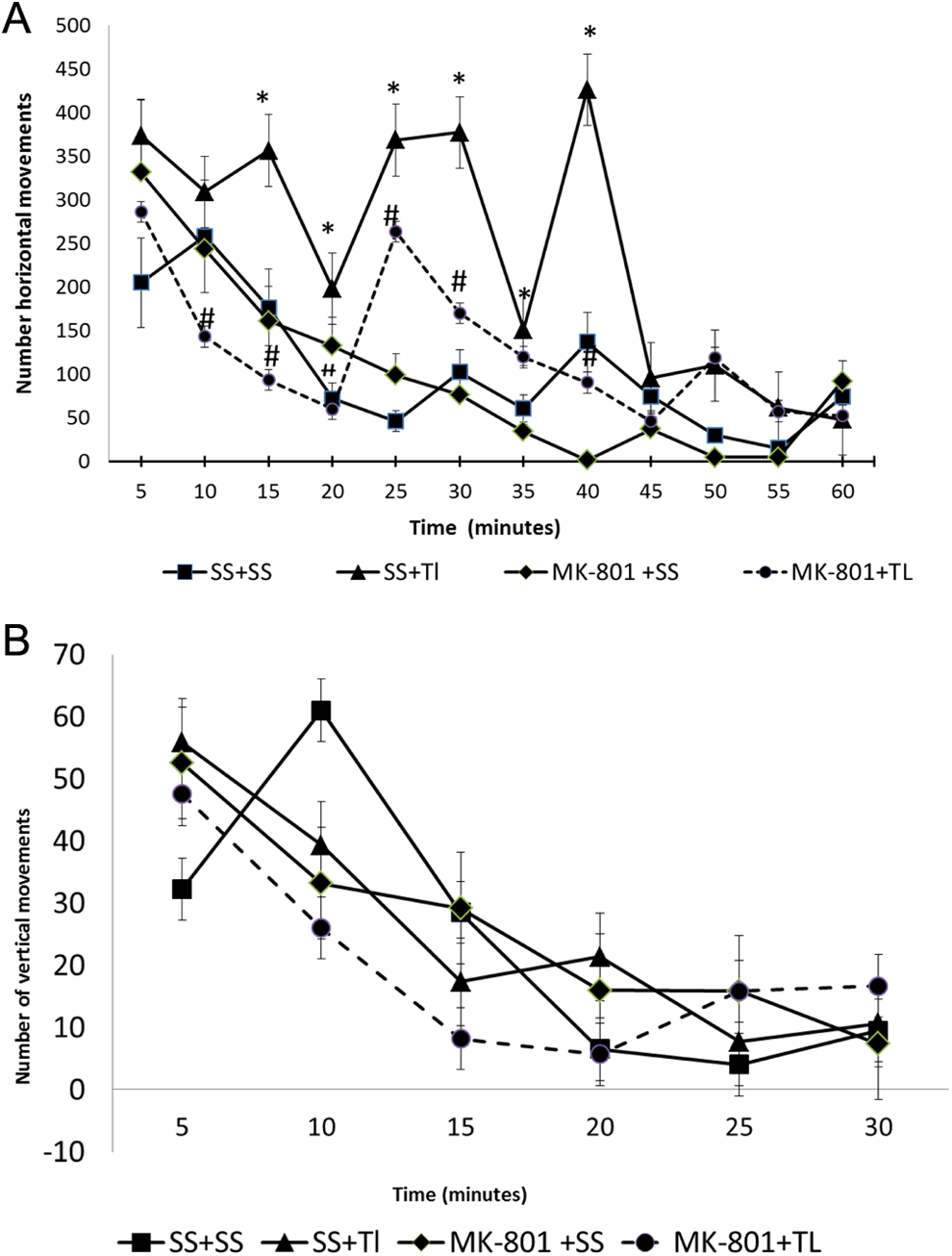
MK-801 attenuates the thallium (Tl+)-induced altered motor activity. Rats were administered with sterile saline (SS) or MK-801 (1 mg/kg, intraperitoneally [ip]) 30 minutes before a single injection of SS or Tl+ (32 mg/kg, ip), and placed on an open-field device 24 hours later. Horizontal (A) and vertical (B) movements were evaluated every 5 minutes for a total of 60 minutes. Data are expressed as mean values ± standard error of the mean (SEM) of 10 rats per group. *P < .05 represents significant differences against the SS + SS group (black squares line); # P < .05 represents significant differences against the SS + Tl+ group (black triangle lines). One-way analysis of variance (ANOVA) followed by Tukey’s test for every single time point.
MK-801 Reduced the Brain Regional Content of Tl+ in Tl+-Exposed Rats
Thallium distribution among the different brain regions of rats from all experimental groups is shown in Figure 2. As expected, Tl+ was not detected in brain regions from the SS + SS (control) group nor in the MK-801–treated (MK-801 + SS) group. Interestingly, at this time point of evaluation and at the dose used, Tl+ was differentially concentrated along the brain regions analyzed in the Tl+-treated group (SS + Tl+). In increasing order, Tl+ content was found as follows: Hp > Ce = S > Cx = Ht. More interesting was the fact that when MK-801 was preadministered to the Tl+-treated rats (MK-801 + Tl+ group), the brain content of Tl+ was significantly decreased in all regions in comparison to the Tl+ group (SS + Tl+), with the exception of Cx. In decreasing order, Tl+ content diminished in magnitude as follows: Ht (82%) > Ce (53%) > Hp (36%) > S (35%).
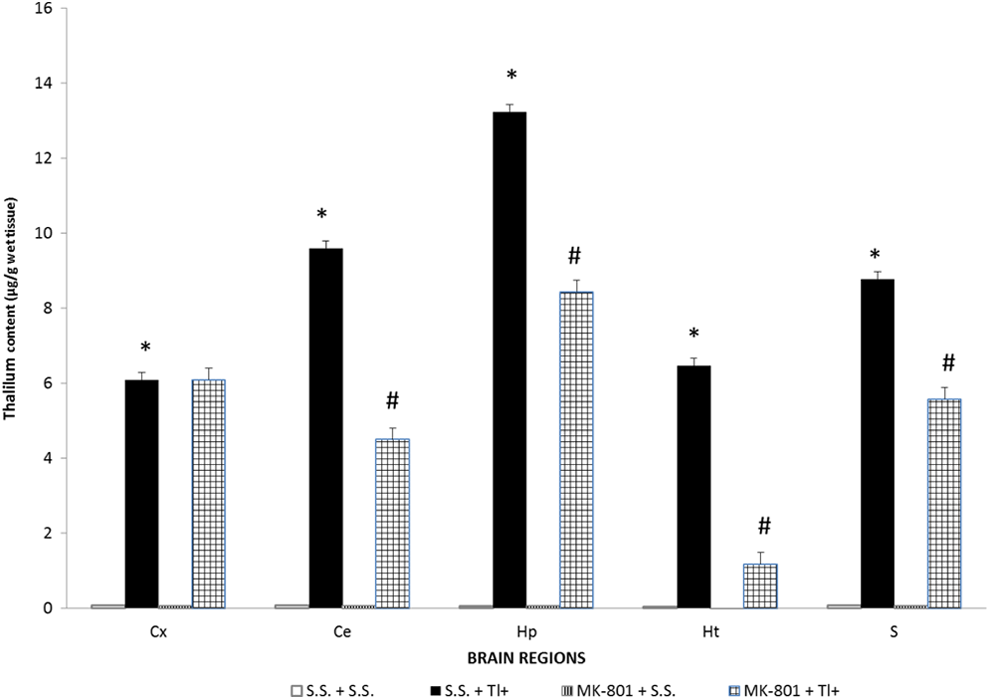
Thallium (Tl+) concentration in different brain regions. Thallium injection was given as a single ip administration. Twenty-four hours after Tl+ injection, rats were sacrificed and rat brain regions were obtained by dissection. Levels of Tl+ in hippocampus (Hp), striatum (S), hypothalamus (Ht), cerebellum (Ce) and frontal cortex (Cx) were assessed by atomic absorption spectrometry. Data are expressed as mean ± standard error of the mean (SEM) of measurements of 5 rats per group. *P < .05 represents significant differences against the sterile saline (SS + SS group). Student t test.
MK-801 Mitigated the Tl+-Induced Brain Regional LPx in Rats
Lipid peroxidation was estimated in rat brain regions as an index of oxidative damage to lipids. Basal levels of LPx were calculated in first place as units of fluorescence per mg of protein; however, they were further normalized in order to make more objective comparisons among regions and pharmacological effects (Figure 3). Under the experimental conditions used in this study, Tl+ (SS + Tl+ group) significantly increased LPx in Ce (190% above the SS + SS group), followed in magnitude by Hp (130% above) and S (50% above). Noteworthy, these 3 regions are the same that accumulated more Tl+. In contrast, no changes in LPx were induced by Tl+ in Cx and Ht, the same brain regions exhibiting the lowest Tl+ accumulation. In turn, the pretreatment of Tl+-exposed rats with MK-801 (MK-801 + Tl+ group) significantly decreased LPx in most of the regions evaluated (except for Ht) not only when compared with the SS + Tl+ group (95% decrease in HP, 91 in Ce, 67% in Cx, and 61% in S) but also even far below the SS + SS group (60% decrease in Hp, 52% in Cx, 41% in Ce, and 17% in S). In Ht, MK-801 per se slightly increased LPx by 25% when compared with the SS + SS group, whereas when administered to Tl+-exposed rats (MK-801 + Tl+ group), MK-801 enhanced LPx by 39% above baseline levels.
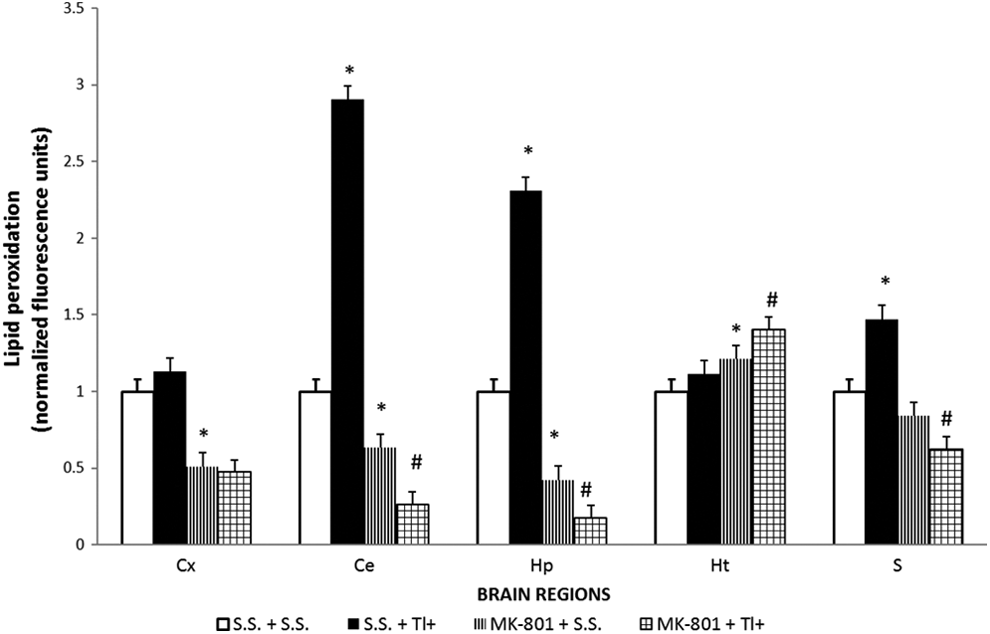
MK-801 decreases the thallium (Tl+)-induced lipid peroxidation in different brain regions. Rats were administered with sterile saline (SS) or MK-801 (1 mg/kg, intraperitoneally [ip]) 30 minutes before a single dose of Tl+ (32 mg/kg, ip) or SS as vehicle. Twenty-four hours later, the following rat brain regions were obtained: hippocampus (Hp), striatum (S), hypothalamus (Ht), cerebellum (Ce), and frontal cortex (Cx). Lipid peroxidation was measured by a fluorometric method as described in the methodological section. Data are expressed as mean ± standard error of the mean (SEM) of 5 rats per group. *P < .05 represents significant differences against the SS + SS group; # P < .05 represents significant differences against the SS + Tl+ group. One-way analysis of variance (ANOVA) followed by Tukey’s test.
The Brain Regional GSH Levels Were Unaltered by Tl+ but Differentially Modified by MK-801
Figure 4 depicts the GSH levels along the brain regions studied from Tl+-treated rats and/or MK-801-treated rats. Rats receiving Tl+ (SS + Tl+ group) showed no significant changes in GSH levels along the evaluated regions when compared with the control rats (SS + SS group), except for Ht, where there was a 43% increase in GSH in the Tl+-treated rats. The preadministration of MK-801 to the Tl+-treated rats did not modify the GSH regional levels observed in the Tl+-treated animals (SS + Tl+), except for Ce, where there was a slight but not significant increase in GSH among these 2 groups (16%). Surprisingly, the GSH contents in HP, S, and Cx were considerably reduced by MK-801 pretreatment (MK-801 + SS group) when compared with the control (SS + SS group; 21%, 34%, and 40% below, respectively), whereas increases in GSH levels produced by MK-801 (MK-801 + SS group) were observed when compared with the control (SS + SS group) in Ce and Ht (above 28% and 77%, respectively). These effects suggest the early emergence of compensatory and adaptive mechanisms to preserve regional GSH content at homeostatic levels via redox regulation.
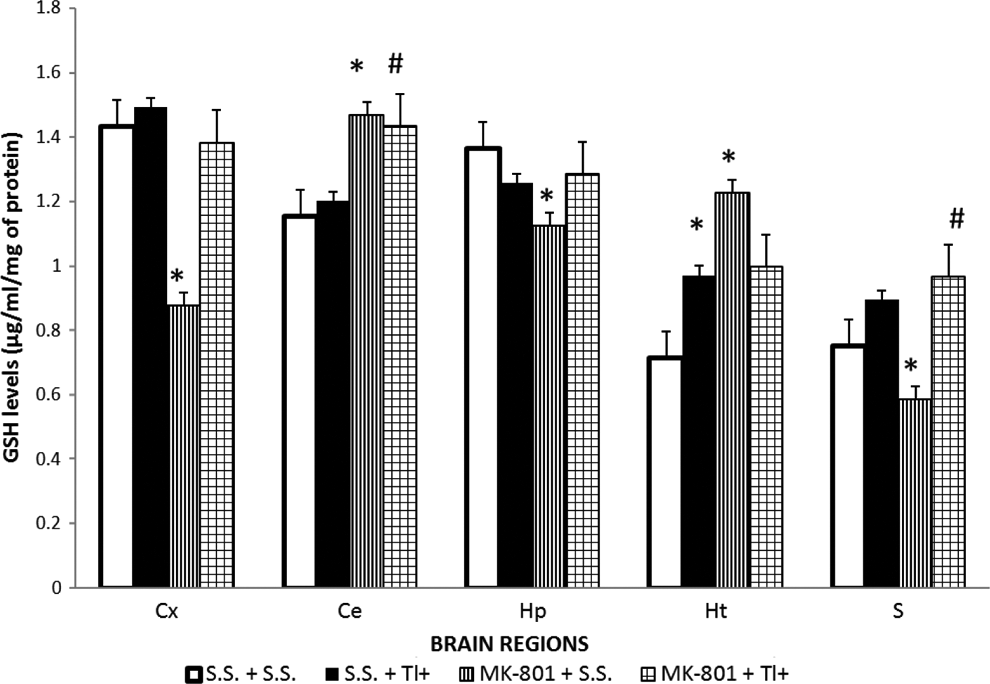
MK-801, but not thallium (Tl+), modifies the glutathione (GSH) levels in rat brain regions. The content of GSH was estimated in rat hippocampus (Hp), striatum (S), hypothalamus (Ht), cerebellum (Ce), and frontal cortex (Cx) by the fluorometric method described in the methodological section. Four groups were designed: (1) Sterile saline (SS + SS), (2) SS plus 32 mg/kg of Tl+ acetate (SS + Tl+), (3) 1 mg/kg intraperitoneally (ip) of MK-801 (MK-801 + SS), and (4) MK-801 + Tl+. The toxic metal was administered 30 minutes after a single MK-801 administration. Twenty-four hours later, rat brain regions were dissected for the corresponding analysis. Data are expressed as mean ± standard error of the mean (SEM) of 5 rats per group. *P < .05 represents significant differences against the SS + SS group; #P < .05 represents significant differences against the SS + Tl+ group. One-way analysis of variance (ANOVA) followed by Tukey’s test.
Brain Glutathione Peroxidase Activity Was Unchanged by Tl+ or MK-801 Treatments to Rats
No changes were found in GPx activity along the different brain regions studied and treatment groups (Figure 5), implying that this enzyme is not participating in the early oxidative changes induced by Tl+ in this model.
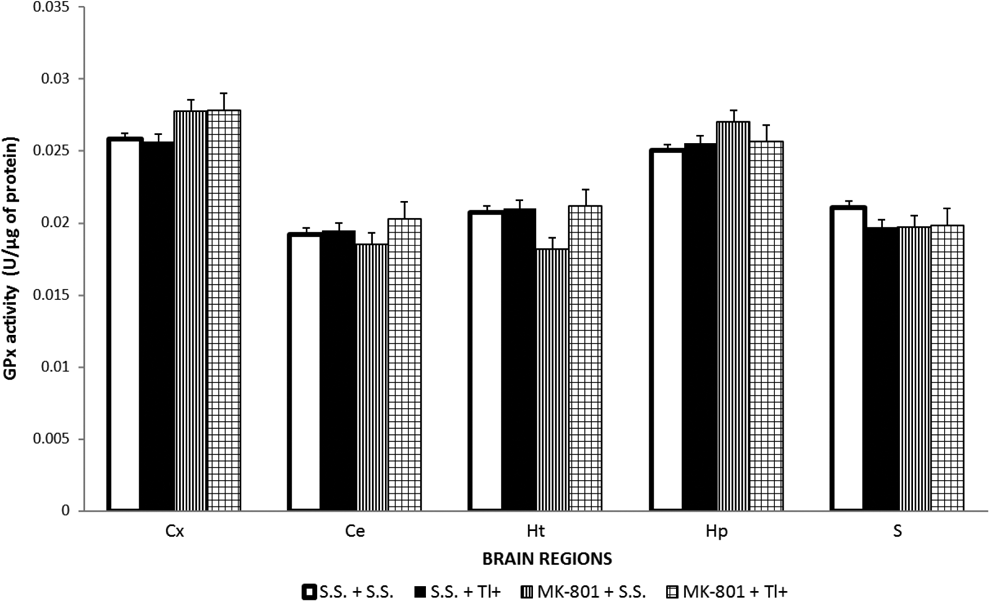
GSH peroxidase (GPx) enzyme activity was not midified by thallium (Tl+) nor by MK-801. Brain regions of 5 rats per group were assayed by a fluorometric method used to estimate the GPx enzyme activity, as described in the methodological section. Four groups were designed: (1) sterile saline (SS + SS), (2) SS plus 32 mg/kg of Tl+ acetate (SS + Tl+), (3) 1 mg/kg intraperitoneally (ip) of MK-801 (MK-801 + SS), and (4) MK-801 + Tl+. The toxic metal was administered 30 minutes after a single MK-801 administration. Twenty-four hours later, rat brain regions were dissected for the corresponding analysis. Data are expressed as mean ± standard error of the mean (SEM) of 5 rats per group.
Discussion
In this work, we evaluated different end points of toxicity in brain regions of rats after an acute systemic Tl+ administration (24 hours) at a 32 mg/kg dose. In addition, for the first time, we investigated a novel possible mechanism as part of Tl+ toxicity, excessive excitability (risk of excitotoxicity) of nerve cells as a source of tissue damage. This issue was addressed through a simple experimental approach: the challenging of Tl+-treated animals with a pretreatment using the potent noncompetitive NMDAr antagonist MK-801 (given 30 minutes before Tl+ administration) as a tool to evidence a possible glutamatergic component in Tl-induced toxicity. Although our findings are preliminary, they clearly support this NMDA component involved in Tl+ toxicity. The use of a higher dose (32 mg/kg, ip), when compared to our previous report, 14 is relevant as it constitutes a different toxic model developed to produce a faster and more intense toxic effect of the metal.
Noteworthy, contrasting effects in most of the end points estimated here were found when compared to our previous report, 14 and they will be discussed point-by-point along this section. In the meantime, a general explanation for these discrepancies could be initially addressed when considering that the dose used in this work is twice the maximum dose used in our previous report. In addition, the time at which we are evaluating all end points is 24 hours post-exposure, which obviously differs from our previous report for several days. Therefore, in this study, we are readily describing early acute effects.
Acute Changes in Tl+ Content Were Decreased by MK-801
In our present study, Tl+ was differentially accumulated along brain regions, where Hp, Ce, and S exhibited the higher contents of the metal. In a previous study of our group, a dose of 32 mg/kg was also tested, revealing differences in brain regional TI+ concentrations. 28 In that report, we demonstrated that 24 hours after Tl+ administration, some brain regions reached the highest concentrations of this metal. Noteworthy, the distribution found in that report is in clear contrast to another study of our group, where Tl+ doses of 8 and 16 mg/kg produced a homogeneous distribution among the same brain regions but estimated at 7 days after Tl+ administration. 14 As aforementioned, differences in doses and time of analysis may account for these differential distributions. It is likely that at shorter times, the metal has not yet reached a homogeneous distribution, and a different mechanism might account for selective regional Tl+ accumulation. In support of this consideration, Wanger et al 33 have recently shown that Tl+ tissue distribution depends on the period of time for which the metal is being administered: At short times (5 minutes), Tl+ is preferentially distributed in olfactory bulb and cortical areas II/III and V, while at longer times of exposure (24 hours) the metal accumulates in caudoputamen and Hp. Other metals such as Pb2+, Hg+, and Al3+ have also shown to differentially accumulate in rat brain regions, Hp being once again the most sensitive region. 34,35 In addition, metals such as Zn+, Pb2+, Cu2+, Cd2+, Li3+, Hg+, and methylmercury have shown to modulate NMDAr, AMPA/kainite, and GABA receptors activity 36 ; however, to date, there are no reports about a role of glutamate receptors on Tl+ distribution and toxicity. Although Pb2+ and Zn2+ have shown to modulate the expression of NMDAr in Hp by decreasing NMDAr-2A subunit expression 35,37 and Mn2+ produces a strong voltage-dependent response to NMDA, 38 to our knowledge, there is nothing available in regard to the modulation of metal contents by glutamate receptors. Our results showing that the pretreatment with MK-801 to rats decreased the Tl+ content in most of the evaluated regions suggest a modulatory mechanism of NMDAr on the metal distribution once into the brain. Of course, this is merely speculative and will need further demonstration, but given the wide distribution of NMDAr among different organs and systems, these receptors might somehow modulate the Tl+ absorption in the brain. In the meantime, Tl+ is known to have a high affinity for Na+/K+-ATPases because its atomic radius and valence are both similar to K+. Like other metals, Tl+ might also modulate the NMDAr-channel upon entering nerve cells. If this is the case, the NMDAr antagonist MK-801 might block Tl+ entrance to the cell or minimally attenuate the damage produced by Ca2+ influx. These possibilities shall need experimental demonstration.
Motor Alterations Induced by Tl+ Were Attenuated by MK-801
MK-801 has been shown to provide protection against the neurotoxicity associated with experimental models of Parkinson disease, HD, and epilepsy at different levels. 23,24,39 In this work, we found that horizontal movements were increased in rats treated with Tl+, supporting a hyperactive pattern of locomotion. Thus, Tl+ produced hyperlocomotion that was attenuated by MK-801 administration likely due to an active participation of NMDAr. This early pattern of motor hyperactivity has been suggested to resemble those changes observed during the first phases of HD as well as in HD experimental models. Such hyperactivity is produced by the mitochondrial toxin 3-NP, 40 which recruits voltage-gated NMDAr overactivation through alterations in membrane potential. 41
MK-801 Reduced Tl+-Induced Oxidative Damage
Thallium toxicity has also been related to an increase in ROS production and biomolecular oxidative damage, in particular LPx. 12,14 Alterations in the antioxidant defense system in response to heavy metals—including Tl+—in different brain regions have been widely reported. 11,14,34,42 Here, we observed that the brain is susceptible to the Tl+-induced LPx in a regional manner just 24 hours after its administration, being Ce, Hp, and S the most susceptible regions. Although this effect supports previous observations 11 showing that Ce and S are vulnerable to Tl+-induced oxidative damage, under the experimental conditions used in this study, Hp was more vulnerable to oxidative stress. Whatever the source of these discrepancies (dose, time, etc), it was clear that MK-801 resulted in LPx levels far below Tl+ and control conditions. Although difficult to explain in principle, this effect might be related to the redox modulatory properties (electron transfer and redox metabolites bioactivity) that have been reported for MK-801, 43 so the possibility that this metal might also exert potent redox activity cannot be discarded. This profile, in turn, could also help to explain why the GSH levels were affected by MK-801, since some intense redox activity is taking place during the first hours of MK-801 exposure.
In regard to GSH, in our previous report, we showed that this antioxidant was depleted 7 days after 16 mg/kg Tl+ exposure only in S, 14 but in this study, we found that Tl+ did not change GSH regional levels, except for Ht where there was a considerable increase in this marker. This effect is not surprising when considering that the time of analysis (24 hours) is short enough to involve major changes in this endogenous antioxidant. This assumption is reinforced by the lack of changes in the activity levels of GPx, a GSH-related enzyme. Although GSH is known to be a substrate for GPx reaction, 44 GPx activity showed no changes in any brain region tested, suggesting that the redox conversion of GSH in this paradigm did not involve enzymatic activity at the time tested. Nonetheless, it seems evident that GSH could be involved in Tl+ toxicity at longer times of exposure.
In turn, MK-801 induced some regional changes in GSH content. The decreased levels of GSH in Cx, Hp, and S notably contrast with its increased values in Ce and Ht. These oscillations can be explained as adaptive/compensatory changes due to the redox nature of MK-801 itself, 43 but obviously they deserve detailed characterization in order to interpret whether these changes are eventually noxious or merely adaptive.
Thallium, Mitochondria, and Excitotoxicity
Thallium is known to stimulate massive swelling followed by disruption of intracellular and mitochondrial membranes. 45,46 These effects occur simultaneously with an increase in Ca2+ or Na+ concentrations, with a decrease of K+ in TlCl-treated hepatocytes, 46 and an ATP deficiency in cells of rats treated with Tl+ salts. 47 Korotkov 48 reported that Tl+ treatment decreased the State 3- (ADP-stimulated respiration) and 2,4-dinitrophenol–stimulated respiration from mitochondrial swelling rather than from an inhibition of respiratory enzymes. Recently, Tl+ was shown to affect protein sulfhydryl groups, mitochondrial swelling, and respiration. 49 Moreover, Tl+ was found to impair electron transfer chain in liver mitochondria at complexes I, II, and IV, leading to increased ROS production, disruption of oxidative phosphorylation, declined ATP, decreased ATP/ADP ratio, mitochondrial permeability transition collapse, and cytochrome c release. 8 Our results are in agreement with the “secondary excitotoxicity” hypothesis, which proposes that the loss of ATP resulting from energy impairment leads to a disrupted activity of ion pumps, further resulting in membrane depolarization and removal of the voltage-dependent Mg2+ block at the NMDAr. 41 This event occurs for other neurotoxins such as 3-NP, which causes secondary excitotoxicity (primary complex II inhibition) by making neurons more vulnerable to basal levels of glutamate, further resulting in cell death. It can be hypothesized that Tl+ could exert a similar effect on mitochondrial function as that produced by mitochondrial toxins. Therefore, although preliminary, the findings of this study provide evidence of a possible participation of Tl+ on excitatory events in the brain.
Of final consideration is the fact that MK-801 attenuated the neurotoxic effects produced by Tl+ at 24 hours after the metal infusion. This is particularly relevant since Tl+ reaches peak concentrations in brain regions at 24 hours after its infusion, whereas the peak concentration of MK-801 in the brain is achieved between 10 and 30 minutes after its injection (2 mg/kg), with an elimination half-life of 1.9 to 2.05 hours, respectively. 50 In consideration of the fact that Tl+ remains in the brain more than 24 hours after its injection while MK-801 has already been eliminated, the rationale to explain these preventive effects involves, to our consideration, at least 2 possibilities: (1) part but not all the toxic actions induced by Tl+ are prevented during the first minutes/hours of its injection by the presence of MK-801 through NMDAr inhibition (acute protection) and (2) the possible chemical interaction of these 2 molecules during the first minutes/hours in which both are simultaneously present in the brain cannot be discarded, in a possible scenario where Tl+ could be, somehow, inactivated. Both possibilities require more detailed investigation.
Conclusion
The findings of this contribution reveal important clues on acute Tl+ toxicity. This research could advance a new area of investigation and a new approach to the design of therapeutic tools to counteract Tl+ intoxication. As human exposure to Tl+ through environmental, occupational, accidental, and intentional sources constitutes an increasing human health problem, 51 it is important to develop a better understanding on the effects of this metal in the CNS.
Footnotes
Author Contribution
L. Osorio-Rico and J. Villeda-Hernández contributed to design as well as acquisition and analysis. A. Santamaria and M. Konigsberg contributed to conception and design as well as analysis and interpretation. S. Galván-Arzate contributed to conception and design as well as acquisition, analysis, and interpretation. All authors drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Laura Osorio-Rico is to obtain her doctoral degree in the Program Doctorado en Biología Experimental, Universidad Autónoma Metropolitana-Iztapalapa, México City, México.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
