Abstract
To evaluate the clinical safety profile for the use of gold nanoshells in patients with human prostate cancer. This follows on the nonclinical safety assessment of the AuroShell particles reported previously. Twenty-two patients, with biopsy diagnosed prostate cancer, underwent nanoshell infusion and subsequent radical prostatectomy (RRP). Fifteen of these patients had prostates that were additionally irradiated by a single-fiber laser ablation in each prostate hemisphere prior to RRP. Patients in the study were assessed at 9 time points through 6 months postinfusion. Adverse events were recorded as reported by the patients and from clinical observation. Blood and urine samples were collected at each patient visit and subjected to chemical (16 tests), hematological (23 tests), immunological (3 tests, including total PSA), and urinalysis (8 tests) evaluation. Temperature of the anterior rectal wall at the level of the prostate was measured. The study, recorded 2 adverse events that were judged attributable to the nanoparticle infusion: (1) an allergic reaction resulting in itching, which resolved with intravenous antihistamines, and (2) in a separate patient, a transient burning sensation in the epigastrium. blood/hematology/urinalysis assays indicated no device-related changes. No change in temperature of the anterior rectal wall was recorded in any of the patients. The clinical safety profile of AuroShell particles is excellent, matching nonclinical findings. A recent consensus statement suggested that the published literature does not support a preference for any ablation technique over another.1 Now that clinical safety has been confirmed, treatment efficacy of the combined infusion plus laser ablation in prostate will be evaluated in future studies using imaging modalities directing the laser against identified prostate tumors.
Introduction
Prostate cancer is the most frequent cancer type among US men, with an estimated 233 000 new cases in 2014, and is the second most common cause of cancer death in men, with approximately 29 500 deaths expected for 2014. 2 The treatment of localized disease has historically been restricted to treatment of the whole gland, either by surgical prostatectomy or by external beam radiation. 3 Currently, men diagnosed with early-stage and low-grade disease face a challenging array of treatment options including active surveillance, focal ablation, or whole gland treatment.
Although whole gland treatment with prostatectomy and radiation is still considered the gold standard for the treatment of prostate cancer, both cause significant side effects in a substantial number of patients. Principal among these are incontinence and erectile dysfunction. In one study of 1288 men undergoing radical prostatectomy, it was found that the percentage of men reporting “no incontinence” went from 87% pretreatment to 32% at 12 months posttreatment to 35% at 60 months. Within this group, “total urinary control” was reported by 87% of men pretreatment, 35% at 12 months posttreatment, and 35% at 60 months posttreatment. 4 The same study also found that the percentage of men reporting “no erectile dysfunction” went from 51% pretreatment to 5% at 12 months posttreatment to 9% at 60 months. Similarly, a recent review of 18 randomized controlled studies and 473 observational studies found similar results for brachytherapy, radical prostatectomy, external beam radiation, conformal external-beam radiation, and nerve sparing radical prostatectomy. 5 The percentage of patients reporting impotence was 30% to 90% for radical prostatectomy, 65% to 85% for external-beam radiation, and 35% to 60% for nerve sparing radical prostatectomy.
An effort to reduce such side effects has led to advances in magnetic resonance imaging technology that have proven capable of identifying an index lesion with a sensitivity and specificity of 89% and 100%, respectively. 6 Such increased imaging precision has permitted a more refined diagnosis of an individual’s disease with an opportunity to localize the lesion within the prostate. 7 And, such anatomical identification of tumor has ushered in an era of focal ablation that may provide an opportunity to avoid the unwanted side effects of whole gland treatment. Early investigations into focal ablation across treatment platforms have seen a significant reduction in side effects historically thought unavoidable. 8
Simultaneously, over the past decade, nanoparticles have been developed for cancer therapy as a means of delivering drugs, 9,10 contrast agents, 11 –13 and specifically to ablate tumor tissue by a variety of targeting strategies. 14 AuroLase therapy is a particle-directed photothermal therapy that uses the tumor-specific accumulation of infrared-absorbing nanoparticles within tumor tissue to generate lethal doses of heat when illuminated by near-infrared (NIR) light. These particles are injected intravenously and accumulate passively in tumor tissue via the enhanced permeability and retention effect. 15,16 With the appropriate placement of a light-delivery optical fiber and with an appropriate choice of NIR power, it is possible to create lethal photothermal lesions that are confined to the tumor tissue and a small surrounding margin while sparing adjacent tissue in which nanoparticles have not accumulated. These particles are activated by near-infrared (NIR) radiation where their tuneable plasmon resonance 17 transduces the incident NIR radiation into optical wavelength phonons 18 yielding heat where the particles have accumulated. Proof of concept studies have been carried out using particles systemically directed against inoculated tumors in the brain, 19 directly injected into canine prostate 20 and in human prostate tumors grown orthotopically in mice. 21,22
AuroShell particles passively accumulate in the perivascular space provided by the fenestrated vasculature of solid tumors and sites of inflammation. Their biochemical and physiological inertness class them as a medical device by the Food and Drug Administration (FDA). After intravenous infusion, the particles are cleared from the bloodstream by the reticuloendothelial system and end up predominately in the liver and spleen. Preclinical AuroShell particle safety was established previously in vitro and in vivo in both acute (28 days) and chronic (10 months) animal studies. 23
Patients, Materials, and Methods
Patients
Patients who presented with evidence of prostate cancer based on the Gleason score of a standard 12-needle biopsy, and for whom a radical prostatectomy was indicated, were afforded the opportunity to enroll in the study after reviewing and granting informed consent.
The study was carried out in 2 arms: (1) the first arm, group 1, consisted of 7 patients who were infused with AuroShell particles 1 day prior to their scheduled prostatectomies and (2) the second arm, group 2, consisted of 15 patients who were infused 1 day prior to the laser procedure and 5 ± 1.5 days prior to their prostatectomies. Patients of both groups were assessed for the safety of the infused dose of AuroShell particles.
Two patients, #s 7 and 16, originally enrolled for group 2 were allocated to group 1 with the consequence that their prostatectomies occurred 6 days after particle infusion rather than the day following infusion.
Ethics Statement
The study was carried out under the auspices of the Federal Commission for Protection against Health Risks (Comisión Federal para la Protección contra Riesgos Sanitarios, COFEPRIS), under COFEPRIS authorization #CAS/OR/01/CMN/103300410C0010-1968/2010. Global IQ Mexico S.A. de C.V. (Ciudad México, México), an international Clinical Research Organization coordinated and verified data collection and recording.
Study Design
The study was an open-label, multi-center, single-dose pilot study of AuroLase therapy in the treatment of patients with primary resectable prostate cancer. Only patients that were scheduled for a radical prostatectomy were enrolled. The trial was divided into 2 arms: (1) Group 1 consisted of up to 10 patients who were given an infusion of AuroShell particles 1 day prior to a scheduled radical prostatectomy and (2) group 2 consisted of up to 15 patients who were given an infusion of AuroShell particles 1 day prior to a laser treatment, and 3 to 7 days prior to a scheduled radical prostatectomy. Patients were followed for 6 months following particle infusion with regular checks of vital signs, hematology, blood chemistry, and urinalysis. In total, 7 patients completed the study in group 1 and 15 patients completed the study in group 2.
Nanoparticle fabrication
As previously reported, 23 Nanoshells were fabricated under clean conditions in a Class 1000 clean room based on the method of Oldenburg et al. 24 Briefly, gold colloids 1 to 3 nm in diameter were prepared using the method of Duff and Baiker. 25 Aminated spherical silica particles 120 ± 12 nm in diameter (Precision Colloids, LLC, Cartersville, California) were exposed to the gold colloid solution, forming gold colloid nucleation sites on the silica core. The gold seed sites were then further reacted with hydrogen tetrachloroaurate (HAuCl4) in the presence of formaldehyde. This caused the surface colloid to grow and coalesce, ultimately forming a complete metal shell. Finished particles possessed a 12- to 15 nm-thick shell that resulted in an optical absorption peak between 780 and 800 nm. Thiolated polyethylene glycol (SH-PEG; Laysan Bio, Arab, Alabama) was then assembled onto nanoshell surfaces by combining 1 mmol/L SH-PEG and nanoshells in a ratio of 1.5 mL/mL in deionized H2O for 12 hours followed by diafiltration to remove the excess SH-PEG. Particles were then suspended in 10% trehalose to create an iso-osmotic solution for injection and concentrated by tangential flow filtration to an extinction of 100 ± 5 optical density (OD) at 800 nm (approximately 2.77 × 1011 particles/mL). The nanoshell solution was passed through a 0.45 nm filter and filled into IV bags. The nanoshell solution was terminally sterilized by e-beam irradiation. The concentration of nanoshells in the 100 OD solution was measured using neutron activation analysis to assess the amount of gold in the solution. Particle size and size distribution were measured using transmission electron microscopy and dynamic light scattering using a Malvern ZetaSizer (Malvern Instruments Ltd, Worcestershire, United Kingdom). The FDA classifies AuroShell particles as a device owing to their lack of chemical or metabolic interaction with tissues and organs. The method of action is to transduce incident NIR light into heat resulting in the thermal ablation of tumor tissues in which they passively accumulate. AuroShell -particles are colloquially referred to as “Nanoshells” even though the FDA does not classify them as such owing to their 150 nm diameter.
Laser System
The laser system consisted of a Diomed/Angiodynamics D15+ laser (810 ± 10 nm). Laser energy was delivered to the prostate using The Visualase Cooled Laser Applicator System (Visualase, Inc, Houston, Texas), which consisted of a 400 μm core optical fiber terminated with a 1-cm long isotropic diffuser. The optical fiber fitted within an 28-cm long, 16G Cooled Catheter System (CCS) that provided a water-cooled lumen for transferring heat away from tissue adjacent to the fiber. Room temperature saline was circulated through the cooling jacket by a clinical peristaltic pump. Both the laser and the cooling pump are FDA approved for clinical use.
The laser output power was confirmed with a factory-calibrated integrating sphere optometer (P-9710-2, Gigahertz-Optik, GmbH, Puchheim, Germany), which provided a calibrated measurement of the total emitted flux from the isotropic diffuser. The optometer used a sterile, single-use test tube in its sample port to preserve sterility of the laser catheter during measurement. Laser outputs systematically spanning 3.0 to 5.0 W delivered for 3 to 4 minutes for each treatment site were employed.
Although the laser was only used interstitially, all participants in the OR wore laser safety eyewear (LG5, Thorlabs, Inc., Newton, New Jersey), rated at OD6+ @ 800 to 820 nm.
Nanoparticle Infusion
Each patient in groups 1 and 2 received an infusion of ∼100 OD AuroShell particles at a target dose level of 7.5 mL/kg of body weight. Particles were administered intravenously via peripheral arm vein under control of an infusion pump (Infusomat Space Pump IV, B. Braun Medical, Inc. Bethlehem, PA) at an initial rate of 120 mL/h for 15 minutes, and then at 600 mL/h until the full amount was infused. The 7 patients in group 1 weighed 78.2 ± 6.9 kg and were infused at 7.6 ± 0.5 mL/kg, and the 15 patients in group 2 weighed 70.2 ± 9.3 kg and were infused at 7.5 ± 0.1 mL/kg. Patients were monitored for 2 hours following the end of infusion for signs of adverse reactions.
Surgical Procedure
The laser procedure was carried out on the day following particle infusion of the group 2 patients. The patients were placed in lithotomy position. The optical fiber catheter was introduced into the prostate via a transperineal approach using a 14GIV catheter/cannula, (Abbocath-T 14G × 140 mm, Butler-Schein, Dublin OH) as an introducer for the flexible fiber catheter. The introducer was advanced under ultrasound guidance (MicroMaxx, SonoSite, Bothell, Washington), the needle removed and replaced by the CCS, and the cannula retracted to permit direct irradiation of the surrounding tissue. A single insertion was made into each lobe. A thermocouple (T-RA4, Exacon Scientific A/S, Roskilde, Denmark) was secured to the transrectal ultrasound probe and used to monitor the temperature of the anterior wall of the rectum adjacent to the prostate. Depending upon the caudal-cranial length of the prostate, an additional laser treatment was carried out after retracting the Cooled Catheter by 8 mm, in order to create slightly overlapping treatment zones along the optical fiber axis. Patients were recovered from anesthesia, monitored until fully recovered, and released to go home until their scheduled prostatectomies.
Prostatectomy
Radical prostatectomies were performed the day following particle infusion for patients in group 1 and 5 ± 1 days following infusion for patients in group 2 (and thus 4 ± 1 days following laser ablation). Prostates were fixed in formalin, transversely sectioned at ∼5 mm intervals, and embedded in paraffin. The bulk of the tissue was retained for clinical histopathological analysis, while thin sections were made available to the sponsor for analysis of regions of photothermal ablation.
Results
Laser Treatment
Fifteen patients in group 2 received a laser treatment on the day following particle infusion. A transrectal ultrasound probe was used to direct the placement of the optical fiber system within the cortex of the left and right hemispheres of the prostate. Adjustment of the position of the optical fiber catheter within each hemisphere was based on the location of the highest Gleason score indicated by the needle biopsy. The safety aspect of the laser treatment demonstrated that the rectal wall would be spared of any heat effect. The thermocouple, attached to the ultrasound probe and positioned against the anterior wall of the rectum at the level of the prostate, recorded temperatures with a ±1°C resolution and 1 Hz rate. No temperature >37°C was recorded for any treatment.
Chemical/Hematology Analysis
Patients in the study were repeatedly assessed from just prior to particle infusion and at 2, 8 (group 2 only), 15 (group 1 only), 30, 60, 90, 120, 150, and 180 days postinfusion. Adverse events (AEs), as reported by the patients and from clinical observation, were recorded and blood was drawn and subjected to chemical (16 tests per patient), hematological analysis (23 tests), immunological analysis (3 tests), and urinalysis (8 tests) at each patient visit. All test results were combined into a single spread sheet, separated by test type, and analyzed using Kaleidagraph (Synergy Software, www.synergy.com). The results of the most pertinent are presented here as box plots, where each box encloses 50% of the data with the median value of the variable displayed as a line. The top and bottom of the box mark the limits of ±25% of the variable population, that is, the interquartile range (IQR). The vertical “whiskers” span the minimum and maximum observations that fall within 1.5× the lower and upper IQRs, and statistical outliers beyond this range are plotted as individual dots. Since we are primarily interested in deviations that might be attributable to the AuroLase therapy, either the infusion alone or the infusion combined with the laser treatment, the vertical axes of Figures 1 and 2 are normalized to the patients’ baseline measurements prior to treatment. The vertical axes of the figures are plotted using the units of measure for each test as indicated in the plot title.
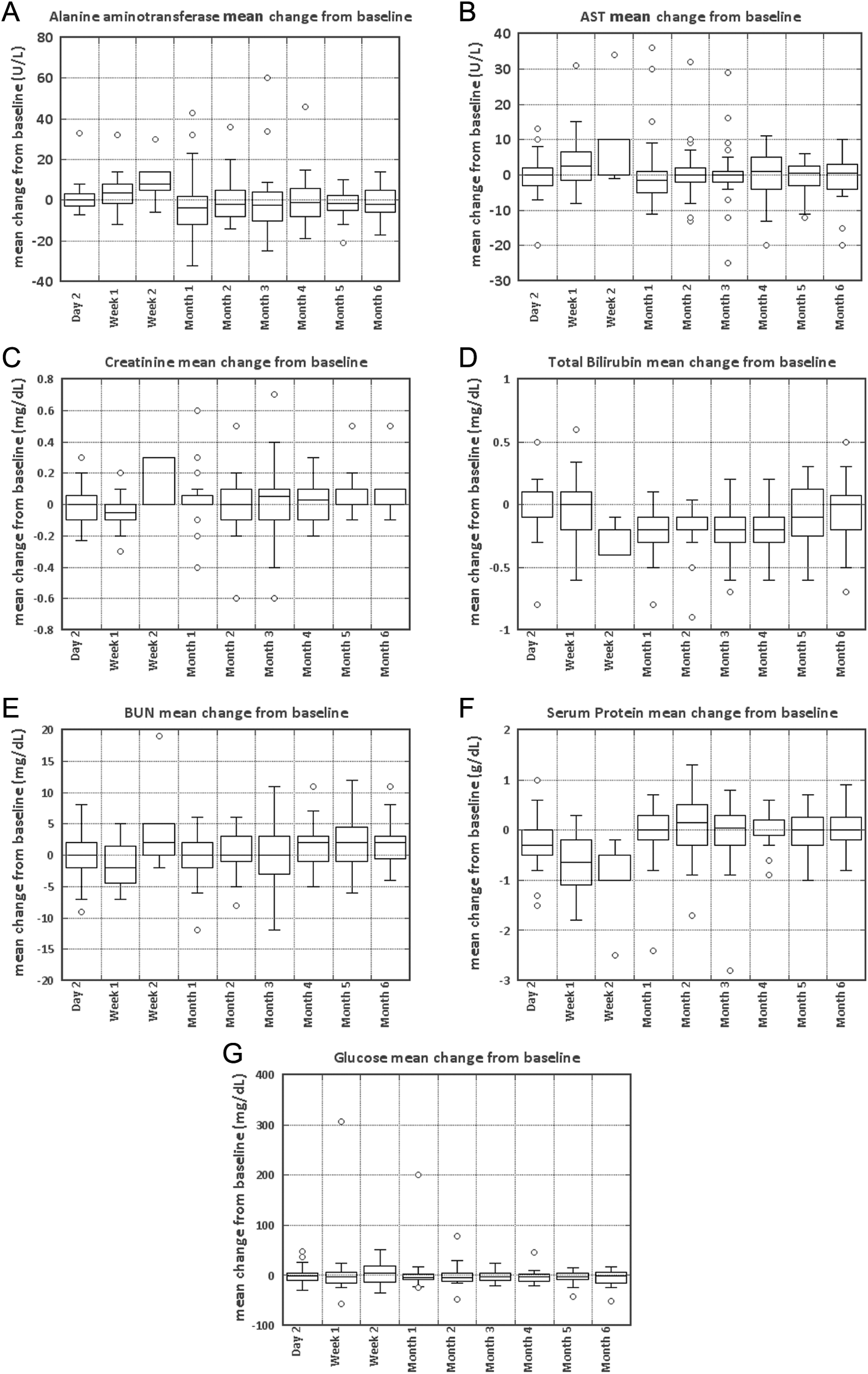
Blood chemistry. Seven of 16 tests are shown.
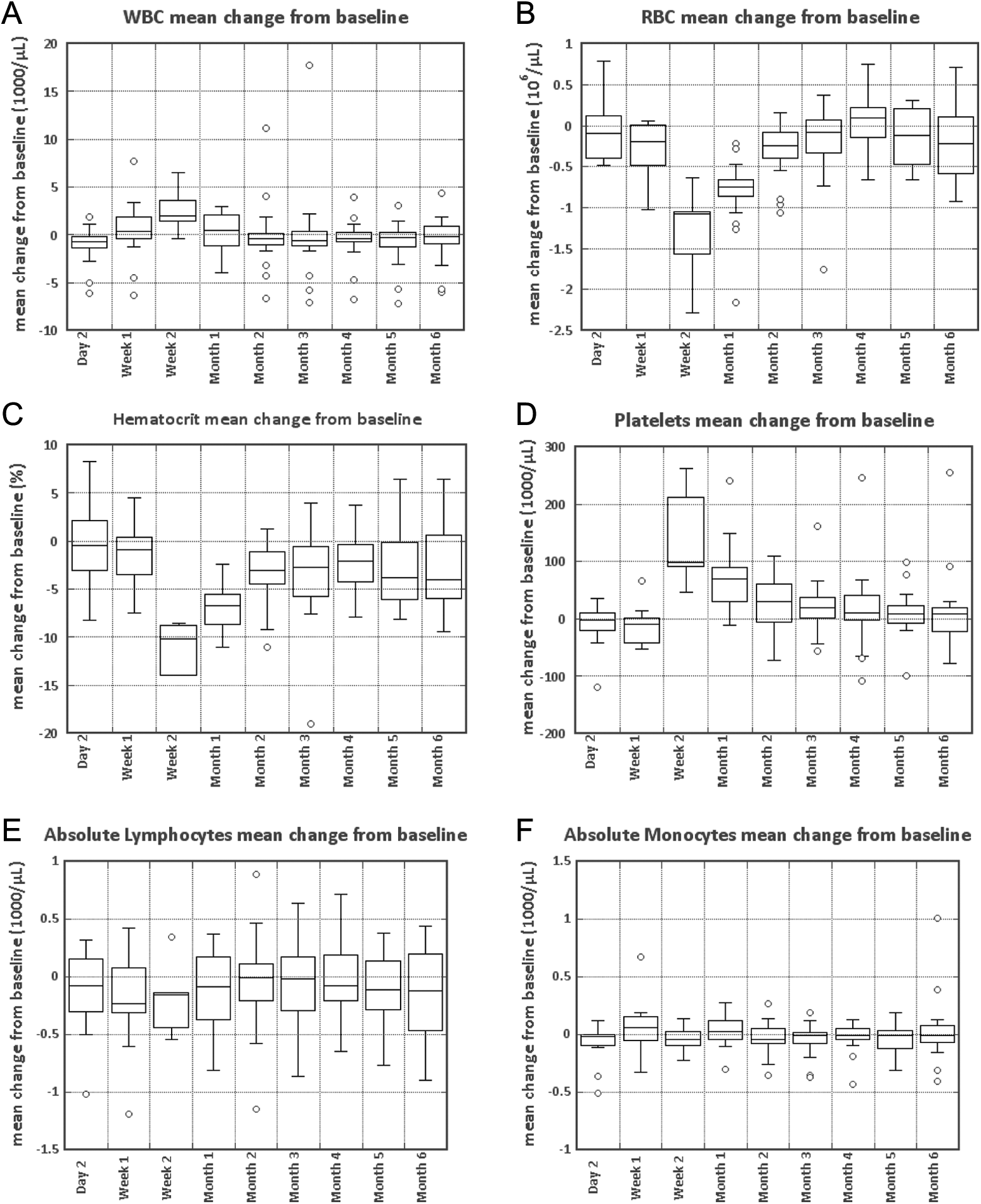
Hematology. Six of 23 assays are shown.
Figures 1 and 2 combine the results of all 22 patients from both groups 1 and 2 in the study. Group 1 had blood chemistry, hematology, and urinalysis at baseline at day 2 (at the time of prostatectomy) and at day 15 (plotted as “week 2” and at months 1-6). Group 2 had blood chemistry, hematology, and urinalysis measured at baseline, day 2 (the date of the laser procedure), at day 4 to 9 (the date of the prostatectomy, plotted as “week 1”), and at months 1 to 6. Hence, in the figures, the week 2 data only apply to the 7 group 1 patients, and the “week 1 data only apply to the 15 group 2 patients. The “baseline” and “months 1 to 6” data apply to both groups.
Blood Chemistry
The following are the most pertinent of the 16 blood chemistry tests conducted and adverse effect criterion here (Figure 1):
Alanine aminotransferase—levels above 55 enzymatic units/L are suggestive of compromised liver function. Aspartate aminotransferase—levels above 34 enzymatic units/L are suggestive of compromised liver function. Creatinine—levels above 1.3 mg/dL are suggestive of compromised kidney function. Bilirubin—levels above 1.2 mg/dL are suggestive of compromised liver function. Blood urea nitrogen—levels above 20.5 mg/dL are suggestive of compromised kidney function. Serum protein—levels outside the range of 6.4 to 8.3 g/dL are indicative of problems of nutrition, kidney, or liver function. Glucose—levels above 99 mg/dL are suggestive of compromised pancreatic function.
Hematology
The following are the most pertinent of the 23 separate hematology tests conducted (Figure 2).
White blood cells: The normal range is 4.5 to 10 × 103cells/µL. Red blood cells (RBCs): The normal range is 4.7 to 5.8 × 106cells/µL. Red blood cells dropped >1 × 106/µL as a consequence of blood loss at the time of surgery and substantially recovered by month 2. Hematocrit: The normal range is 40.0% to 54.0%. Hematocrit dropped 10% as a consequence of blood loss at time of surgery and substantially recovered by month 2. Platelets: The normal range is 150 to 500 103cells/µL. Platelets by >1 × 105/µL at the time of surgery and substantially returned to normal levels by month 2. Absolute lymphocytes: The normal range is 0.94 to 4.80 103cells/µL. Absolute monocytes: The normal range is 0.09 to 0.80 103cells/µL.
Urinalysis
Urine pH remained substantially unchanged. Four patients in group 2 developed urinary tract infections of moderate severity during the follow-up period. None deemed related to the device were reported in any patients, and all resolved with concomitant treatment (see Table 1).
Group 2 Adverse Events.a
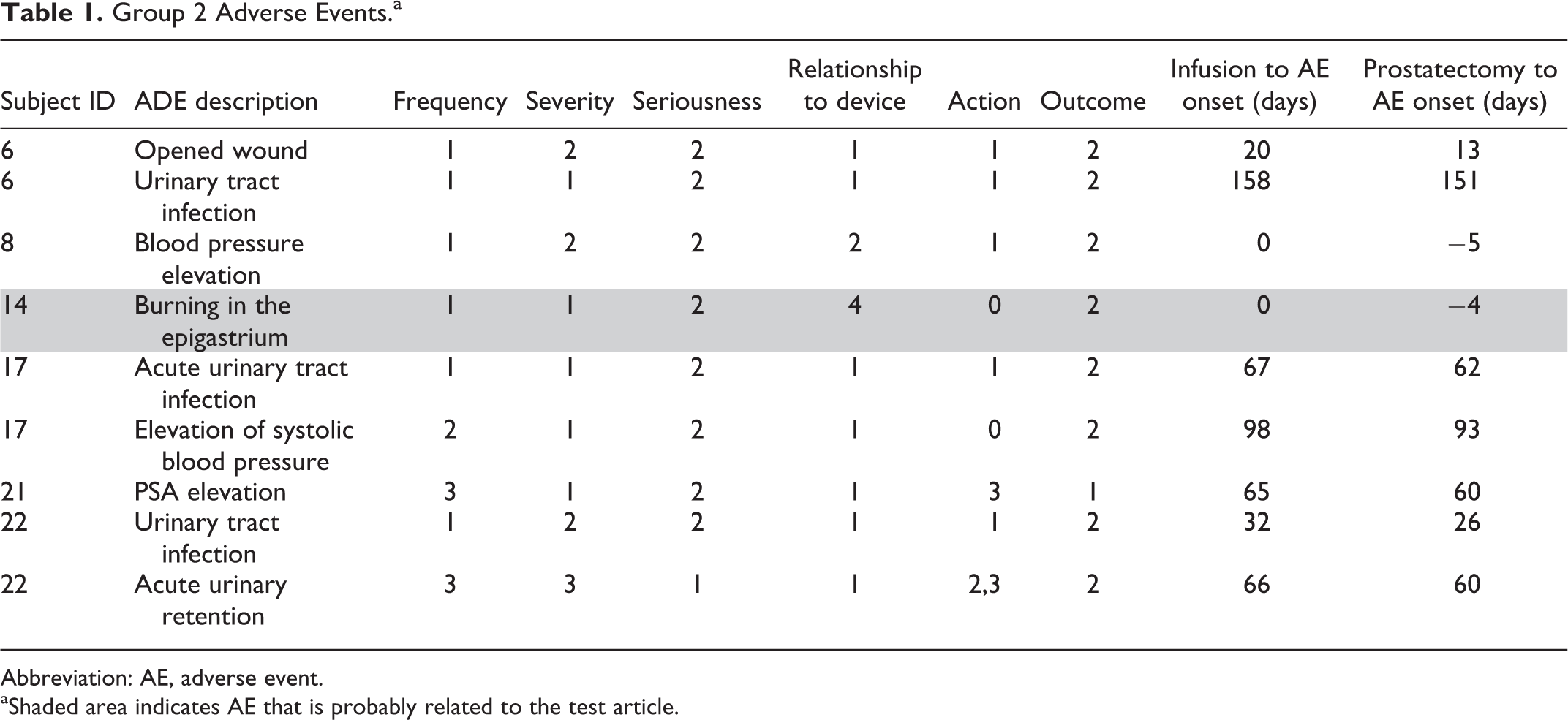
Abbreviation: AE, adverse event.
aShaded area indicates AE that is probably related to the test article.
Adverse Events
An AE is defined as any unfavorable and unintended sign (including an abnormal laboratory finding), symptom, or disease temporally associated with the use of a medical treatment or procedure that may or may not be considered related to the medical treatment or procedure.
26
The AEs were scored using the Common Terminology Criteria for AEs v3.0 published by the National Cancer Institute. The AEs were described by scoring 6 different parameters:
Frequency: 1 = single episode, 2 = intermittent, and 3 = continuous; Severity: 1 = mild, 2 = moderate, 3 = severe, 4 = life-threatening or disabling, and 5 = death related to AE; Seriousness: 1 = yes and 2 = no; Relationship to device: 1 = unrelated, 2 = unlikely, 3 = possible, 4 = probable, and 5 = definite; Action taken: 0 = none, 1 = concomitant treatment, 2 = hospitalization, and 3 = other; and Outcome: 1 = continuing, 2 = resolved, residual effects, 3 = lost to follow-up, and 5 = patient died
A serious AE (SAE) was defined as an AE that scored 3 or higher on severity.
Over the course of the 6 months that each patient was followed as part of this study, a total of 28 AEs were recorded in 12 of the 21 patients (10 in patient 1 alone). Three SAEs were reported and shown in Table 2. Of the 28 AEs, it was concluded that 1 AE was possibly related to the test therapy, and 1 AE was probably related to the therapy. These 2 are highlighted in Tables 1 and 2. Neither of the SAEs was judged to be related to the therapy.
Group 1 Adverse Events.a
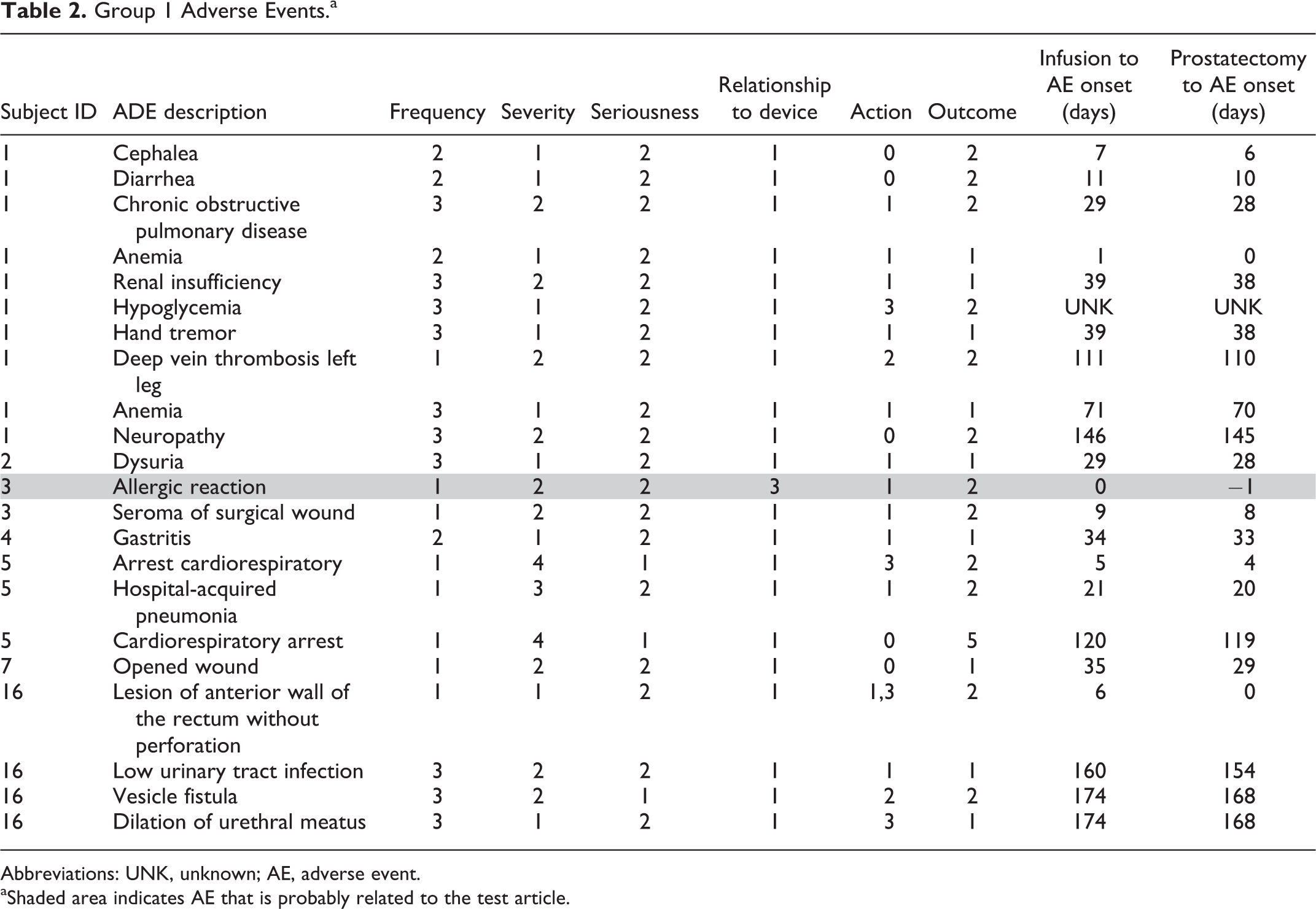
Abbreviations: UNK, unknown; AE, adverse event.
aShaded area indicates AE that is probably related to the test article.
The AEs for group 1 (particle infusion only) and group 2 (particle infusion combined with laser treatment) are shown in Tables 1 and 2, respectively. The 3 SAEs were accounted for by patient 5, 69 years old, who had 2 cardiorespiratory arrests at 5 and 120 days postinfusion, and patient 22, 69 years old, who was hospitalized for acute urinary retention 66 days postinfusion. Patient 5 subsequently died, and patient 22’s condition resolved after 14 days. In neither case were the SAEs determined to be related to either the particle infusion or laser treatment.
Laser Safety
No temperature rise in the anterior rectal wall was recorded in any of the group 2 patients. Nor were any AEs related to the laser procedure reported.
Discussion
As indicated in Figure 1, the results of the blood chemistry analysis indicates that neither the AuroShell particles, nor the laser treatment, nor the prostatectomy had any significant effect on the patients’ chemical balance or metabolism. Further, as indicated in Figure 2, hematology suggests no long-term effects of the particle infusion or laser treatment. Transitory changes in RBCs, hematocrit, and platelets were most reasonably attributed to the known blood loss during prostatectomy.
The 2 AEs that were judged to be related to the AuroLase therapy are highlighted in Tables 1 and 2. Patient 3 had an apparent allergic reaction at the time of infusion, which resulted in itching of the back and extremities and which resolved with intravenous diphenhydramine and dexamethasone. Patient 14 had a single episode of a burning sensation of the epigastrium at the time of infusion, for which no treatment was given, and which resolved spontaneously. The experience of patient 3 suggests that some patients may experience an allergic reaction to the particles, 27 and specifically to the PEG coating. Additionally, patient 8 (Table 1) experienced elevated blood pressure at the time of infusion, though this was judged to be unrelated to the infusion itself. In sum, the AEs recorded seem consistent with the treatment of ill and aging patients.
Conclusion
While there are reports in the literature of special risks associated with nanoparticles, in aggregate these have generally been associated with smaller particles and with higher surface charge densities. Due to the wide variety of nanoparticle compositions, surface chemistries, manufacturing methods, and applications, it is important to treat each nanoparticle configuration individually to determine the potential benefits and hazards to patient health. The evaluation conducted on gold nanoshells in the 22 patients treated in this study has identified no indication of toxicity, lack of tolerance, or immunological effects through 6 months. These data correlate well with previous studies 19,20,23 and demonstrate the safety of systemically infused gold nanoshell particles for use in focal ablations in the prostate. We are in the process of initiating a follow-on clinical study for focal ablation of prostate lesions that will follow patients to 24 months to assess longer term efficacy and safety.
Footnotes
Acknowledgments
The authors gratefully acknowledge the contributions to this study and publication to Dr Laura Garcia de Medina of Global IQ Mexico S.A. de C.V., who coordinated and verified data collection and recording, and Ms Robin Dickson of Nanospectra Biosciences, Inc, who entered and ran the statistical analyses for this study. This study was funded by Nanospectra Biosciences, Inc.
Authors’ Contributions
Stern, J. contributed to design, drafted the manuscript, and critically revised the manuscript; Solomonov, V contributed to acquisition and critically revised the manuscript; Sazykina, E. contributed to acquisition and critically revised the manuscript; Schwartz, J. contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript; Gad, S. contributed to analysis and interpretation and critically revised the manuscript; Goodrich, G. contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Drs. Schwartz and Goodrich are employees of Nanospectra Biosciences, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
