Abstract

Introduction
Benign prostatic hyperplasia (BPH), a common progressive disease among older men, frequently produces undesirable lower urinary tract symptoms (LUTS; weak urinary stream, frequency, and urgency). 1 Pharmacological therapy for BPH mainly includes prostate 5-α-reductase inhibitors, α1-adrenoreceptor (ADR) blockers, and the combination therapy with drugs of both therapeutic classes. 2 –4
Complementary medicines, like the lipid extracts of saw palmetto (SP; Serenoa repens) fruits, have been widely used to treat BPH/LUTS. 5,6 Lipid extracts of SP contain a mixture of fatty acids, mainly oleic, lauric, and myristic acids. 7 Although some clinical studies have not demonstrated its clinical efficacy, 8,9 the use of SP for treating BPH is still documented. 6,10,11 Intake of SP seems to provide benefits on BPH in terms of a multiple mechanism that includes the inhibition of prostate 5α-reductase, the antagonism of α1-ADR, anti-inflammatory, and antioxidant effects. 12 –15 Clinical studies, including the recent Complementary and Alternative Medicine for Urological Symptoms (CAMUS) trial, have demonstrated that SP is safe and well tolerated. 10,11,16 –18 Despite its widespread public use, published animal toxicity studies on SP are scarce. 19 In particular, published studies on the long-term toxicity or potential carcinogenicity of SP are not available (Entrez PubMed, up to September 30, 2014).
D-004, a lipid extract of the royal palm (Roystonea regia) fruits, also contains a mixture of fatty acids, wherein oleic, lauric, palmitic, and myristic acids are the most abundant. In vitro studies have demonstrated that D-004 dose dependently inhibits 5α-reductase activity (half maximal inhibitory concentration = 2.25 µg/mL) in soluble fractions of rat prostate through a noncompetitive mechanism. 20 This result is consistent with the fact that oral treatment with D-004 (100, 200, and 400 mg/kg/d) for 14 days prevented prostatic enlargement induced with testosterone, but not with dihydrotestosterone, in rats. 21
In other studies, D-004 inhibited in vitro the contractile responses to noradrenaline and acetylcholine in rat vas deferens and was more effective in inhibiting noradrenaline than acetylcholine contractions. The in vivo effect of D-004 on the hypertensive response induced by noradrenaline was also significant. 22 Oral administration of D-004 has been shown to ameliorate testosterone- and phenylephrine-induced prostate hyperplasia 21,23 –25 and to produce antioxidant effects in rodents. 26 D-004 (5.3 mg/kg/d) has been effective to reduce plasma oxidative variables in healthy and BPH men 27,28 and to decrease LUTS in BPH men in accordance with the significant reduction of the International Prostate Symptom Score. 28,29
Experimental toxicology studies have demonstrated that single or repeat oral administration (up to 2000 mg/kg/d) of D-004 in mice and rats to be relatively nontoxic. 30 –32 In particular, long-term (12 months) oral treatment with D-004 (800-2000 mg/kg/d) did not show evidence of D-004-related toxicity in rats, and the highest dose (2000 mg/kg/d) was a no observable adverse effect level (NOAEL). 32 Genotoxicity studies (Ames test, comet assay, bone marrow micronucleus, and chromosomal aberrations tests) did not reveal D-004-related cytotoxic or genotoxic potential 33 –36 and administered up to 1000 mg/kg/d did not induce fetal or reproductive toxicity. 37 Likewise, clinical studies have demonstrated that D-004 (5.3 mg/kg/d) is safe and well tolerated. 27 –29
In light of these grounds, D-004 seems to be a promising substance. Nevertheless, since BPH/LUTS requires chronic treatment and the scarce published information on the experimental toxicity of SP, a substance with similar composition and effects to that of D-004, the assessment of the long-term toxicity/carcinogenicity of D-004 was evaluated. This study was undertaken to investigate the long-term carcinogenic/toxic potential of D-004 administered for 24 months to Sprague Dawley (SD) rats.
Materials and Methods
Animals and Housing Conditions
Young adult (4-6 weeks) male and female SD rats (75-110 g) were purchased at the National Centre for Laboratory Animals Production (CENPALAB; Cuba). Rats were quarantined for 15 days prior to the commencement of the study, maintained in a room with barrier conditions, in filter-protected air-conditioned rooms, with controlled temperature (23°C ± 2°C) and relative humidity (60% ± 5%), 16 air changes/h, and 12-h light/dark cycles.
Rats were housed 3 per cage in plastic shoeboxes, and bedding (processed hardwood chips) was changed and sterilized in autoclave. Animals were fed rat standard chow from CENPALAB and formulated to contain approximately 14.5% protein, 8.5% fat, and 9.5% fiber. According to long-term studies criteria, food was restricted to 70% of the recommended intake, after concluding the growing period (13th week), 38,39 while water was allowed ad libitum.
At treatment completion, rats were fasted for 12 hours prior to sacrifice. 39,40 Rats were handled in accordance to the Cuban Ethical Regulations for Animal Care and the Cuban Code of Good Laboratory Practices. Study protocol was approved by the Institutional Board of Animal Use. The study was periodically audited by the Quality Assurance Unit, adhered to the Good Laboratory Practices. Also, this study was inspecting by a board-certified panel of experts from national regulatory agency (Center for State Control of Drugs, Equipment and Medical Devices).
Test Substance
D-004, obtained from Centre of Natural Products (National Centre for Scientific Research, Havana, Cuba), was used and met control quality specifications. D-004 was obtained through the saponification of royal palm fruits and further extraction and purification in organic solvents. Three batches were used in the study. The average free fatty acid composition (%, w/w) of such batches, assessed by a validated gas chromatography method, was as follows: caprylic (0.2-0.4), capric (0.5-0.6), lauric (22.5-25), palmitic (10-12), myristic (10.5-11), palmitoleic (0.1-0.3), stearic (2.6-2.9), and oleic (42-44) acids. Purity ranged from 91% to 94%. Batches used in this study were within the 2-year expiration date validated by stability studies in climatic zone IV (temperature—30°C and relative humidity—70%). 41,42 D-004 emulsified in Tween-65/water (2%) vehicle 1 hour prior to daily administration confirm that D-004 was stable for a week, in such conditions. The concentrations of the emulsions were adjusted weekly according to bodyweight gain.
Rats were randomized, based on body weight, into 4 groups of 50 rats per sex: a control group treated with the vehicle (Tween-65/water) only and 3 groups treated with D-004 (500, 1000, and 1500 mg/kg/d, respectively). Treatments (vehicle or D-004) were given by oral gavage (2 mL/kg), once daily (8:30-11:30
The lowest dose of 500 mg/kg/d (3000 mg/m2/d) used in the study is within the effective pharmacological range in the rat since 200 and 400 mg/kg/d had been shown to prevent testosterone-induced prostatic hyperplasia in this species. 24 The highest dose of 1500 mg/kg/d (9000 mg/m2/d) is that recommended as upper limit in long-term carcinogenicity studies. 43 Also, the lowest and highest doses of D-004 selected were 94 and 283 times higher, respectively, based on body weights than the proposed daily human dose of 320 mg/d ≈ 5.3 mg/kg/d for a 60-kg individual or 197 mg/m2/d yielding margins of safety of 15 (referred to the lowest dose) and 46 (highest dose), based on BSA. 27 –29
Clinical Symptoms, Hematology, and Blood Biochemistry
Animals were observed twice daily: in the morning (8:00-12:00
Body weights were determined at the end of the quarantine (a day prior to starting the treatment), weekly during the first 13 weeks and monthly thereafter, until sacrifice. Food consumption was assessed similarly. 40,43
Euthanasia under ether anesthesia was performed for moribund animals and to those with appreciable body reduction (≥10%) or with clinical symptoms indicating risk of death. The cause of death was determined for all animals that died on test or were moribund sacrificed (in particular, death related to tumors vs other lesions). All animals were subjected to complete necropsy, in which all cavities and organs were carefully observed for detecting neoplastic and nonneoplastic lesions. The date of sacrifice was recorded and taken for further survival analysis.
At study completion, survivors were isolated in individual cages, fasted for 12 hours with free access to water, anesthetized under diethyl ether atmosphere, and sacrificed by complete bleeding. Rats were randomized in similar groups for the daily sacrifice.
Blood was drawn from the abdominal aorta and samples were collected for serum biochemical and hematological determinations at study termination, placed at room temperature for 30 minutes, and centrifuged at 3000 rpm for 10 minutes. Samples were processed the same day of blood sampling. Samples were not collected for toxicokinetics.
Serum aliquots were taken to assess the values of glucose, triglycerides, cholesterol, creatinine, albumin, alanine aminotransferase, aspartate aminotransferase, lactate dehydrogenase, creatine phosphokinase, and both alkaline and acid phosphatases. Serum biochemical parameters were determined by using reagent kits (Roche and SPINREACT; Crumlin Co, Antrim, United Kingdom), except blood acetylcholinesterase, determined according to Voss and Sacsse. 44 In turn, hematological parameters (hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, and counts of red blood cells [RBCs], platelets, and white blood cells) were determined in an hematological equipment (model KX-21N; SySMEX, Kobe, Japan).
Pathology
During the necropsy, the abdominal, thoracic, and cranial cavities of all animals were examined. The liver, heart, spleen, lungs, thymus, brain, kidneys, adrenal glands, testis, prostate, epididymis, seminal vesicles, uterus, and ovaries were weighed (Sartorius Universal Scale, Goettingen, Germany). The organ to body weight ratios were determined and expressed as percentage. 45
Samples of the organs mentioned previously and of lymph nodes (mandibular and mesenteric), bone marrow of sternum, pituitary gland, thyroid with parathyroid, larynx/trachea, bronchi, salivary glands, tongue, oesophagus, stomach (glandular and nonglandular), small (duodenum, jejunum, and ileum) and large intestine (cecum, colon, and rectum), pancreas, penis, urinary bladder, vagina, skeletal muscle, skin and subcutis, eyes, Harderian glands, Zymbal gland, sciatic nerve, and spinal cord were preserved in 10% buffered formaldehyde. 46
Samples from all animals with macroscopic lesions, and from the control and highest dose groups, were taken and embedded in paraffin, sectioned with a rotary microtome (Leitz microtome, Wetzlar, Germany), stained with hematoxylin and eosin, and examined by light microscopy. Slides were read by 2 experienced pathologists (MD, PHD, and DVM). The diagnostic criteria for tumors assessment were made by using the International Classification of Rodent Tumours and Histopathology of Preclinical Toxicity Studies. 47,48 A Zeiss Primo Start microscope (Zeiss Optical Co, Ltd, Tokyo, Japan) was used for these observations.
Statistical Analysis
Data were analyzed following the recommendations for toxicological studies. 43,49,50 Mortality data were compared with survival analysis, using the Cox, Cox-Mantel, Log-rank, Gehan Wilcoxon, Peto and Peto Wilcoxon, and Kaplan-Meier tests. Survival time was defined as the time elapsed from the beginning of the study up to the day of the death (spontaneous or provoked). After checking data homogeneity, continuous variables were processed through an analysis of variance. Categorical data were compared with the chi-square test.
Neoplastic lesions were analyzed according to their frequency, type, and affected organ/tissue. Data were interpreted considering the spontaneous frequencies referred by other authors 51 –55 and our data. 56,57 Nonneoplastic lesions were also analyzed.
Statistical significance was a priori established for α = 0.05. All statistical analyses were 2 tailed and performed independently by sex using the STATISTIC data analysis software (StatSoft, Inc 2003; Tulsa, Oklahoma, version 6; www.statsoft.com).
Results
Mortality Data and Survival Analyses
Overall, 122 (30.5%) of the 400 rats died before study completion. Of them, 98 (80.3%) were associated to neoplastic lesions, without differences between treated and control groups. The cause of death was identified for all animals. In all, 80.3% of deaths were associated with neoplastic lesions. Five deaths were related to technical (gavage) errors. The deaths of 1 female treated with the highest dose of D-004 and 2 males treated with the dose of 500 mg/kg/d occurred immediately after dosing, when the animals suddenly had dyspnea and progressive worsening of health status and the 2 others (males treated with the dose of 1500 mg/kg/d) died 1 day later. Presence of D-004 emulsion into the trachea, bronchi, and lungs was found during the necropsy, so that such death was attributed to wrong oral gavage procedure.
The mortality rates in control (19 of 50, 38%) and females treated with D-004 at 500 mg/kg/d (23 of 50, 46%), 1000 mg/kg/d (21 of 50, 42%), and 1500 mg/kg/d (19 of 50, 38%) were similar. The mortality rate in treated males as a whole (24 of 150, 16%) was significantly lower (P < 0.05) than in the control group (16 of 50, 32%; Table 1). The first deaths occurred near to month 12 after starting the treatment. The onset of nodular lesions was similar in treated and control animals. Survival analysis did not show significant differences between control and treated groups as a whole or by sex. Survival times were similar in control and treated groups/sex: 658 and 664 days in control and treated females; and 680 and 697 days in control and treated males, respectively.
Rat Deaths During Treatment with D-004.

Clinical Signs and Safety Indicators
Regarding daily observations, no obvious findings or toxic signs were found. Body weight (Table 2) and food consumption (data not shown) were similar in treated and control groups, in males being greater than in females.
Bodyweight Values (g; mean ± SD) of Rats Treated With D-004 for 24 Months.
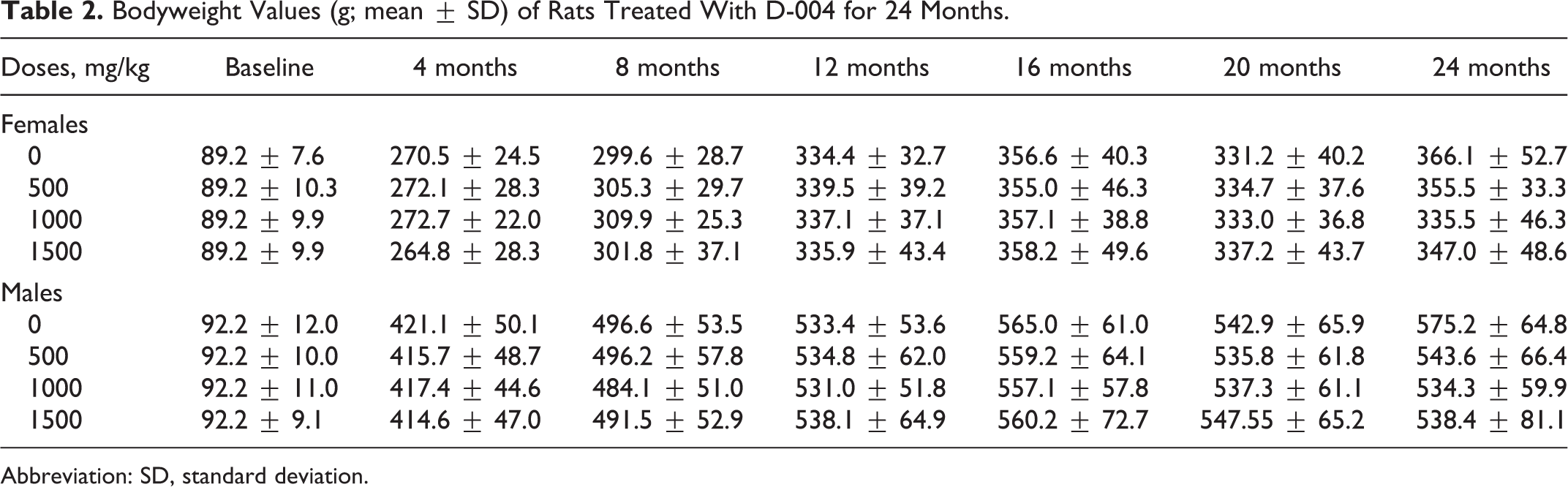
Abbreviation: SD, standard deviation.
No significant differences or trends with the doses were found in terminal blood biochemistry and hematological indicators (Tables 3 and 4, respectively), with the exception of the hematocrit and RBC counts, which were significantly higher in females treated with 1000 mg/kg/d than in the controls. But the data show that there were no differences in the 1500 mg/kg/d dose, so these changes were not considered toxicologically relevant.
No significant differences of absolute and relative organ weights among the groups were found (Table 5).
Hematological Parameters (Mean ± SD) of Rats Treated With D-004 for 24 Months.
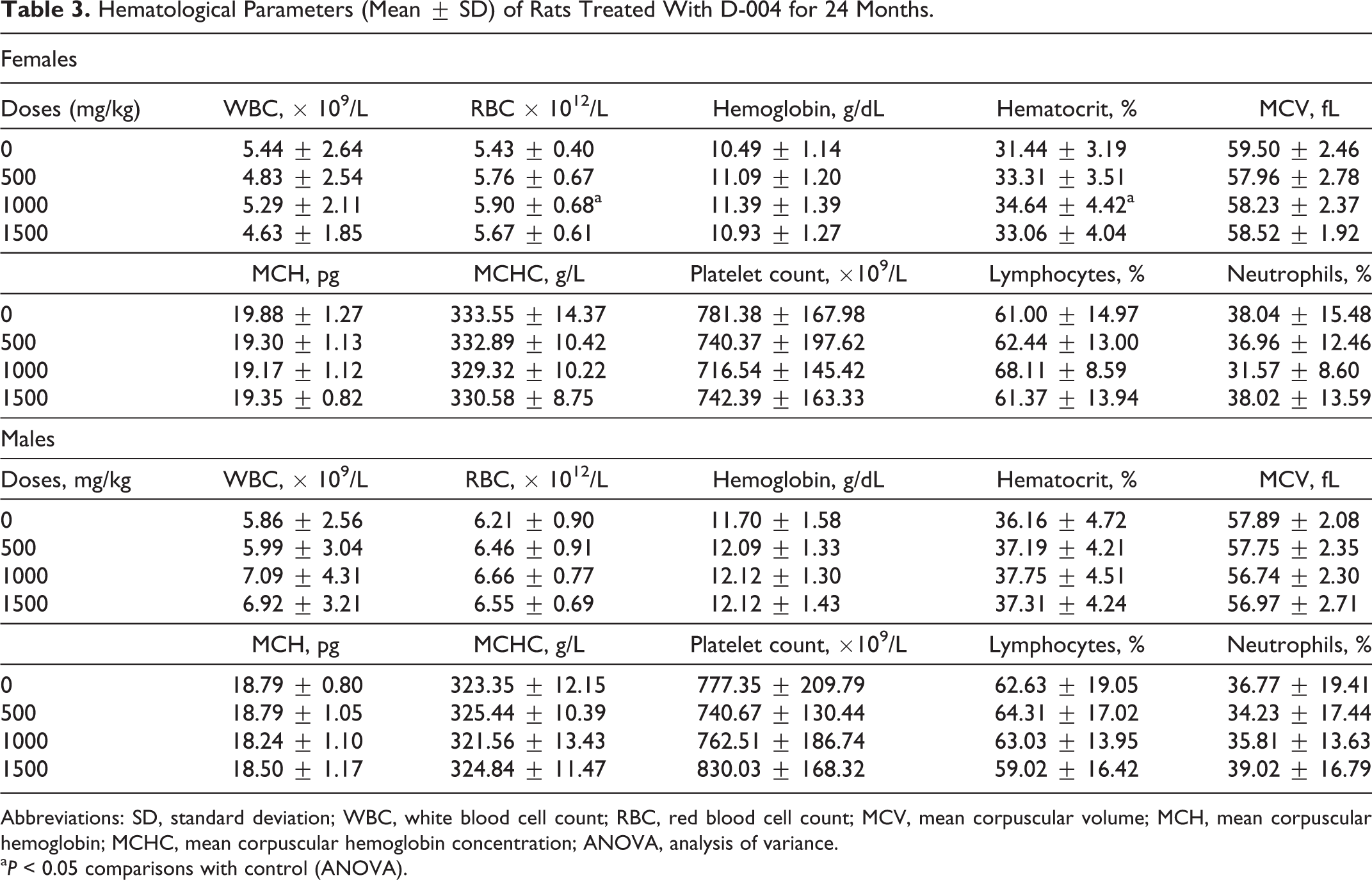

Abbreviations: SD, standard deviation; WBC, white blood cell count; RBC, red blood cell count; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; ANOVA, analysis of variance.
a P < 0.05 comparisons with control (ANOVA).
Blood Biochemistry Data (Mean ± SD) in Rats Treated With D-004 for 24 Months.a
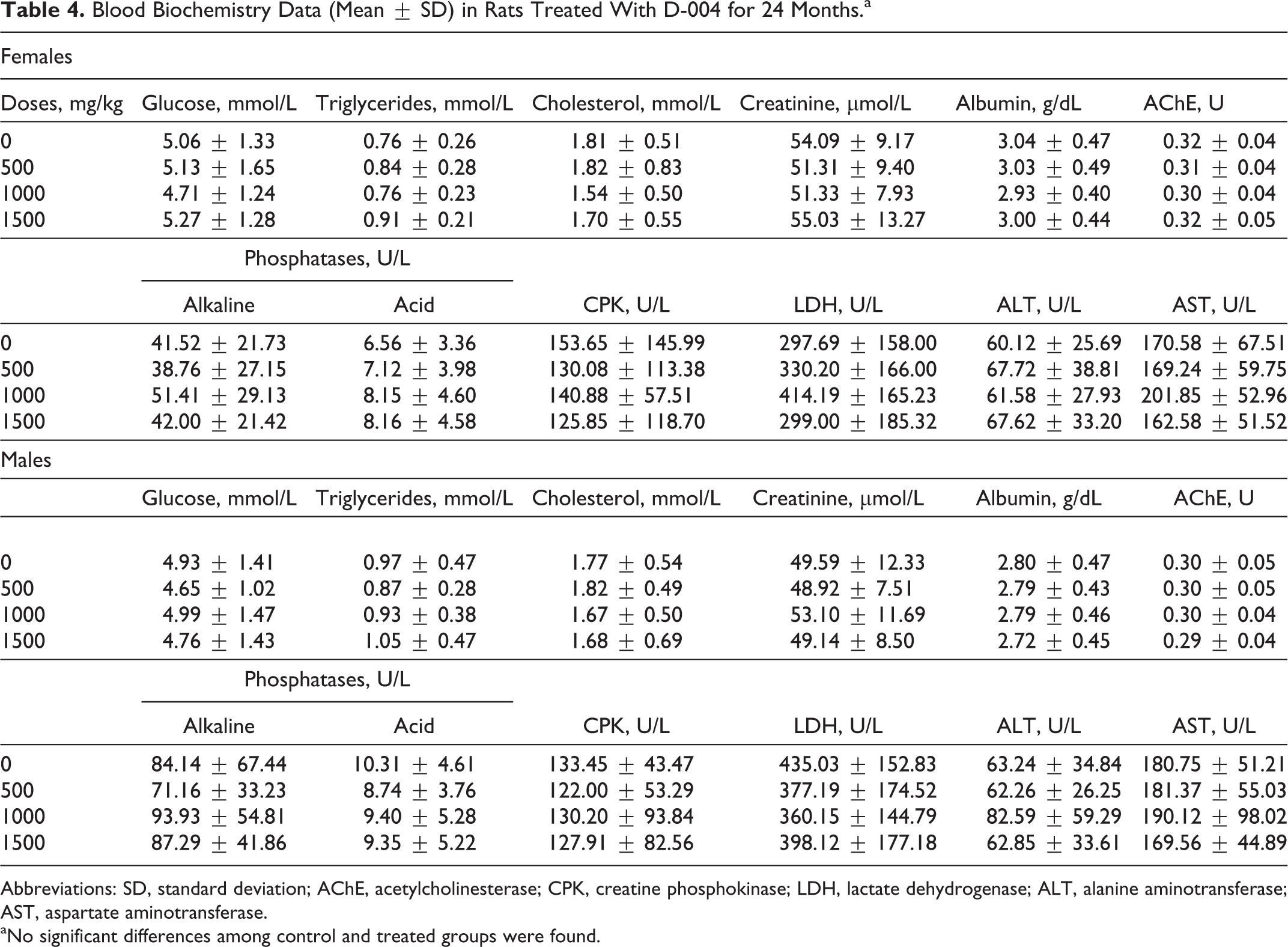

Abbreviations: SD, standard deviation; AChE, acetylcholinesterase; CPK, creatine phosphokinase; LDH, lactate dehydrogenase; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
aNo significant differences among control and treated groups were found.
Relative Organ Weight Data a (Mean ± SD) in Rats Treated With D-004 for 24 Months.
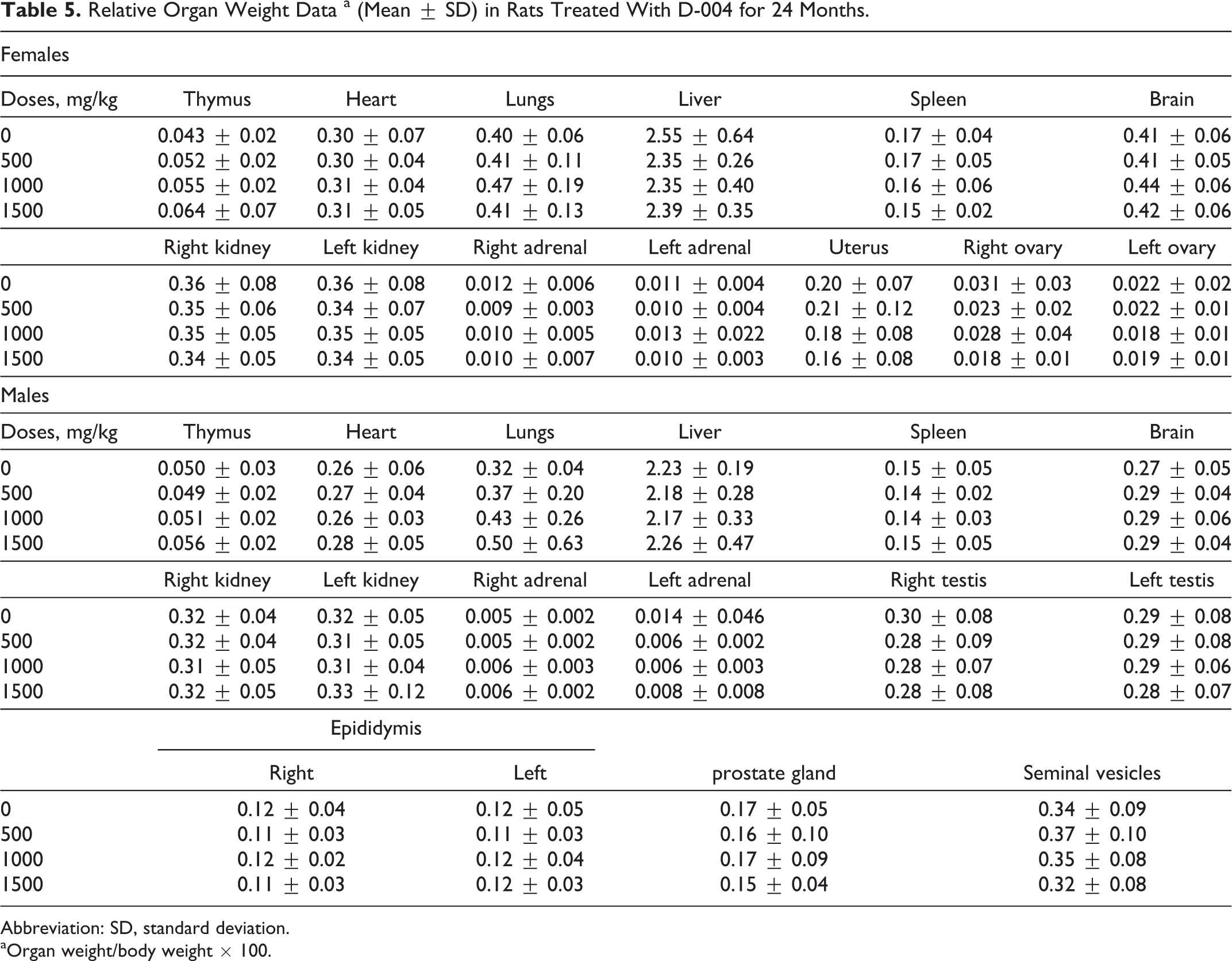

Abbreviation: SD, standard deviation.
aOrgan weight/body weight × 100.
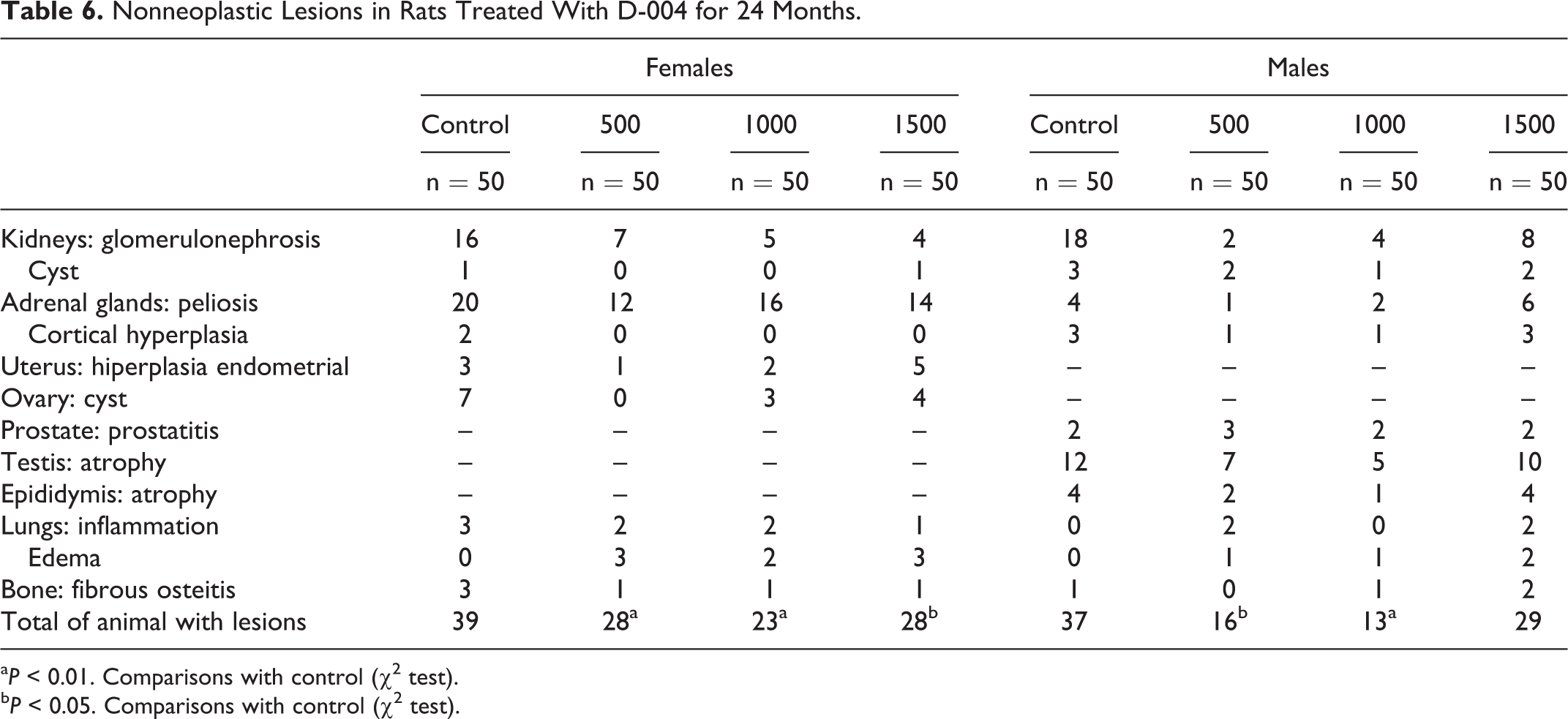
Nonneoplastic Lesions
Nonneoplastic lesions were also seen, as expected (Table 6). Peliosis in the cortex of adrenal gland, the most frequent nonneoplastic lesion, was present in 62 females: 20 controls (40%), 12 of 500 mg/kg/d (24%), 16 of 1000 mg/kg/d (32%) and 14 of 1500 mg/kg/d (28%), and in 13 males: 4 (8%) controls and 1 (2%), 2 (4%), and 6 (12%) males treated with 500, 1000, and 1500 mg/kg/d, respectively. These numbers account for a higher frequency (P < 0.001) of peliosis in females as compared to males. Animals with this lesion showed cystic changes, congestion, hemorrhage, thrombosis, and inflammation.
D-004 treatment did not increase the frequency of such lesion since the differences among treated and control groups were not significant.
Nonneoplastic Lesions in Rats Treated With D-004 for 24 Months.
a P < 0.01. Comparisons with control (χ2 test).
b P < 0.05. Comparisons with control (χ2test).
Glomerulonephrosis, the second commonest nonneoplastic lesion in the study, was present in 32 females and 32 males. Rats with this lesion showed pale kidneys with cystic spaces visible to the naked eye. Microscopically, tubules were dilated and most contained hyaline casts. Tubular epithelial cells were often flattened and atrophic, and sometimes hyperplastic with cytoplasmatic changes (vacuoles and hyaline droplet change). The glomerulus showed focal or global glomerulosclerosis, and the interstitium contained chronic inflammatory cells.
The frequency of glomerulonephrosis in treated females (16 of 150, 10.7%) was lower (P < 0.001) than in matched controls (16 of 50, 32%); and a similar situation occurred in males: 14 (9.3%) of the 150 treated and 18 (36%) control males exhibited such kind of lesion.
In males, 34 (12 controls, 24%; 22 D-004 treated, 14.7%) had testicular atrophy. Despite the frequency of such lesion in treated groups seemed to be lower than in the controls, the differences were not significant. In contrast, the frequency of ovarian cysts in treated rats (7 of 150, 7.4%), was significantly lower (P < 0.001) than in female controls (7 of 50, 14%).
The frequency of other nonneoplastic lesions (renal cyst, endometrial hyperplasia, prostatitis, epididymis atrophy, and lung inflammation) was low and similar in treated and control groups. None of the nonneoplastic lesions found were preneoplastic.
Neoplastic Lesions
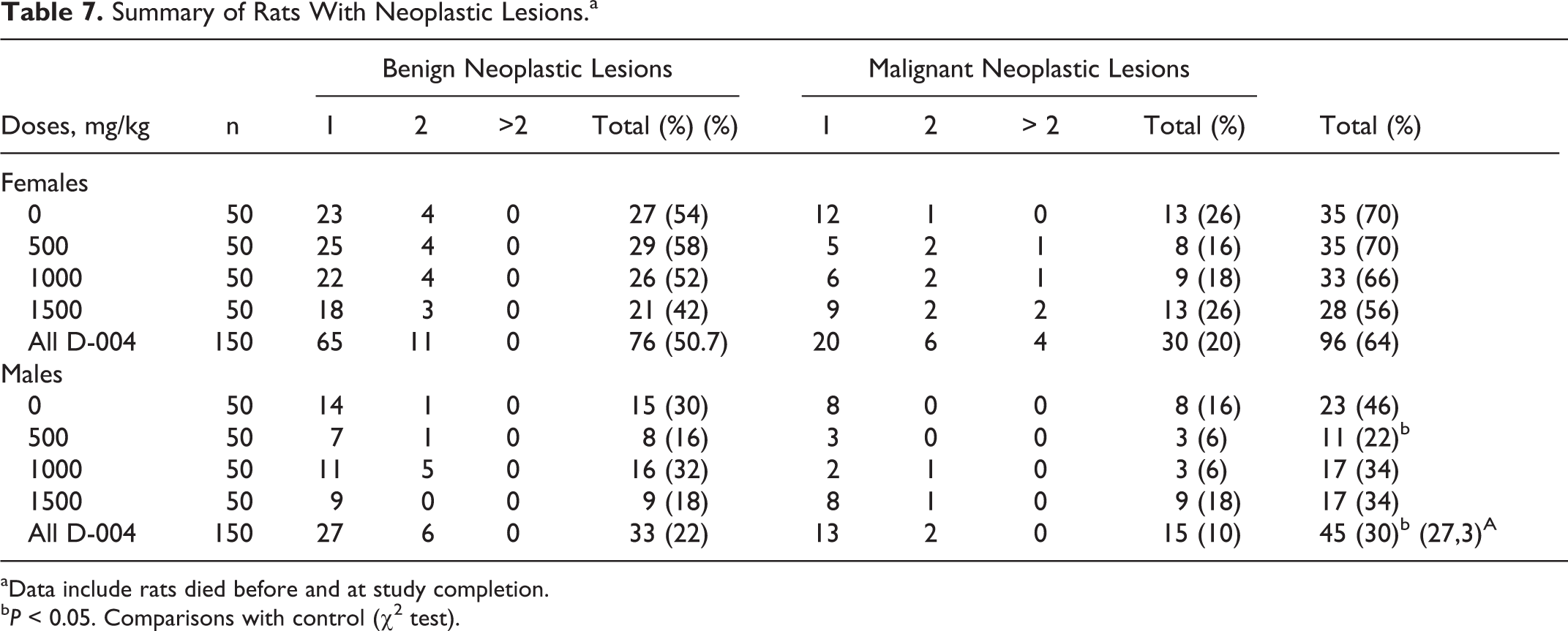
One hundred and ninety-nine rats displayed at least some neoplastic lesion (benign and/or malignant) (Table 7). The frequency of female rats with some neoplasm (131 of 200, 65.5%) was significantly higher (P < 0.001) than that found in males (68 of 200, 34%). The preponderance of neoplastic lesions in females was also evident in the control groups: there were more females (35 of 50, 70.0%) than control males (23 of 50, 54.0%) with some tumor (P < 0.05 difference between groups).
Summary of Rats With Neoplastic Lesions.a
aData include rats died before and at study completion.
b P < 0.05. Comparisons with control (χ2test).
In this study, all tumors found are common to this species/strain. In males, no animal showed combined benign and malignant lesions. Meanwhile, in females occurred in 2 controls, 2 treated with 500 mg/kg/d, and 1 treated with the higher dose of D-004.
D-004 treatment did not the frequency of neoplastic lesions in any sex. In females, the frequencies of rats with tumors in treated and control groups were similar. Curiously, the frequency of treated males with some neoplastic lesion (45 of 150, 30%) was lower (P < 0.05) than in that of the controls. This finding results from the significant (P < 0.05) decrease found in the 500 mg/kg/d group (11 of 50, 22%) as compared to the control; together with an apparent, albeit not significant, reduction seen in the 1000 and 1500 mg/kg/d groups.
Malignant tumors were more frequent (P < 0.001) in females than in males. In total, 43 females and 23 males had some malignant lesion, being noted that some rats had more than 1 lesion.
Oral administration of D-004 did not increase the frequency of all malignant lesions in any sex. In all, 43 female rats: 13 controls (26%) and 30 treated with D-004 (20%) had some malignant tumor: 8 (16%), 9 (18%), and 13 (26%) in 500, 1000, and 1500 mg/kg/d groups; respectively. In turn, 8 (16%) controls and 3 (6%), 3 (6%), and 9 (18%) males treated with 500, 1000, and 1500 mg/kg/d, respectively, showed some of these lesions.
The frequency of benign neoplasms among female rats was more frequent (P < 0.001; 76, 50.7%) than in males (33, 22%). The frequencies of such lesions in control females (27, 54%) was similar than in those treated with 500, 1000, and 1500 mg/kg/d: 29 (58%), 26 (52%), and 21 (42%), respectively. Likewise, the frequency of benign tumors in control males displayed (15, 30%) was comparable to that seen in males treated with 500, 1000, and 1500 mg/kg/d: 8 (16%), 16 (32%) and 9 (18%), respectively.
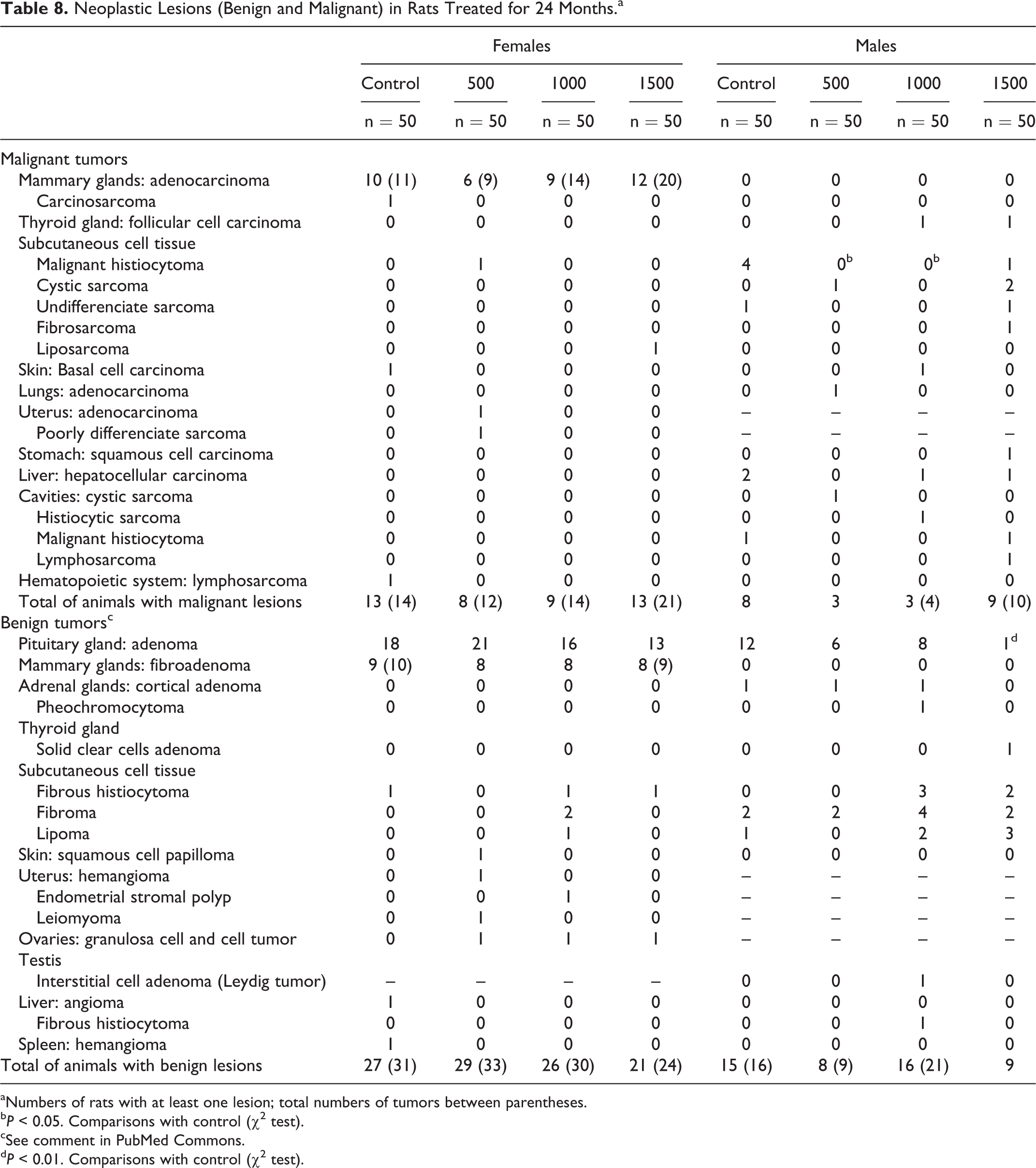
Table 8 displays detailed data on the neoplastic lesions found in the study, the most frequent being hypophyseal and mammary tumors.
Neoplastic Lesions (Benign and Malignant) in Rats Treated for 24 Months.a
aNumbers of rats with at least one lesion; total numbers of tumors between parentheses.
b P < 0.05. Comparisons with control (χ2 test).
cSee comment in PubMed Commons.
d P < 0.01. Comparisons with control (χ2 test).
Hypophyseal adenoma, the most frequent neoplastic lesion, were present in 95 rats: 68 (34%) females and 27 (13.5%) males, so that its frequency in females was higher (P < 0.001) than in males. This tumor was found in 18 (36%) control females and in 21 (42%), 16 (32%), and 13 (26%) treated with 500, 1000, and 1500 mg/kg/d, respectively. In males, it was present in 12 (24%) controls and 15 (10%) treated rats: 6 with 500 mg/kg/d (12%), 8 with 1000 mg/kg/d (16%), and 1 with 1500 mg/kg/d (2%). No significant differences among control and treated groups were detected, with the exception of the significant reduction (P < 0.01) found in males treated with 1500 mg/kg/d versus the controls.
Mammary tumors (malignant and benign) were found only in females. Overall, D-004 did not increase the frequency of malignant or benign mammary lesions, since the frequency of both tumors in control and treated females was similar.
In all, 37 (18.5%) rats had mammary adenocarcinoma: 10 (20%) controls and 6 (12%), 9 (18%), and 12 (24%) treated with 500, 1000, and 1500 mg/kg/d, respectively. These tumors, the most frequent malignant lesion of the study, displayed glands arranged in a “back-to-back” manner with little interspersed stroma, large and moderately hyperchromatic nuclei, prominent nucleoli, and numerous mitoses. Also, a control female exhibited a carcinosarcoma.
Mammary benign tumors (fibroadenomas) were seen in 33 (16.5%) females (9 controls and 8 in each treated group). These tumors were well-delineated lobules of glandular tissue, separated by dense collagen. Epithelial elements resembled the normal acinus, the cells were arranged in clusters or groups of alveoli of fairly uniform cells with rounded or oval nuclei showing little mitotic activity. The cytoplasm was finely vacuolated or foamy and the ducts contained lightly staining concentric concretions or protein plugs.
Benign tumors of subcutaneous cell tissue, present in more than 5% of males, did not show significant differences in control and treated groups. Nevertheless, the frequency of malignant histiocytoma in males treated with 1000 and 1500 mg/kg/d was significantly lower (P < 0.01) than in the controls (8%).
Other malignant tumors with frequencies ≤4% were hepatocellular carcinoma, observed in 2 control males (4%), basal cell carcinoma, present in 1 control female (2%) and 1 lymphosarcoma, also in control female (2%). All these tumors were similarly present in treated and control groups.
In addition, benign tumors in adrenals, liver, and spleen were detected, but all at frequencies of 2% and not treatment related. Only 1 male of the 1000 mg/kg/d group had a Leydig tumor in the testes.
Oral administration of D-004 did not increase time to tumors, the occurrence of tumors (malignant or benign), and morbidity-related tumors.
Discussion
This study demonstrates that oral administration of D-004 (500-1500 mg/kg/d) for 24 months was not carcinogenic or toxic in SD rats. These results represent the first demonstration of the lack of carcinogenic potential of D-004, which supports its safe long-term use. Since D-004, a lipid extract of the royal palm, contains a mixture of fatty acids, mainly of oleic, lauric, palmitic, and myristic acids, the present study could contribute to the evidence-based knowledge of the long-term safety of similar mixtures of fatty acids, such as SP extracts.
The 5-day/wk dosing schedule is justified in this study because the D-004 remains more than 48 hours in the body according to pharmacokinetic studies, 58 so this dosing schedule did not affect the study results. Also, systemic exposure to D-004 has occurred in the absence of toxicity.
After single-dose administration of oral D-004 radiolabeled with (3H)-oleic acid in rats, the absorption was rapid; mean Cmax was 195.56 (31.12) μgEq/mL, and mean Tmax was 2 hours. Thereafter, a biphasic decay of total radioactivity (TR) was found: a rapid first phase (t1/2α, 1.33 hours), followed by a slower second elimination phase (t1/2β, 36.07 hours). Fifteen minutes after D-004-radiolabeled administration, radioactivity was broadly distributed throughout the tissues, with more accumulating in the prostate than elsewhere. In the first 8 hours, accumulation of TR was greatest in the prostate, followed by the liver, small intestine, and plasma. In contrast, over the periods of 24 and 144 hours after administration, TR increased in the adipose tissue while it decreased in the other tissues and plasma. After 24 and 144 hours, area under the curve (AUC) in the adipose tissue was second only to that in the prostate. In the small intestine and the liver, t1/2λ values were 45.92 and 35.60 hours, respectively, and mean residence time values were 65.10 and 52.44 hours, respectively. Mean peak radioactivity in the prostate (562.41 µgEq/g) was reached at 4 hours and decreased slowly thereafter. The prostate had the highest values of t1/2β and cumulative AUC compared with the other tissues and plasma. Excretion of TR was limited, with similar amounts being excreted in feces and urine. Mean (standard deviation) TR was similar in feces (33.48% [4.90%]) and urine (28.96% [5.32%]), with total excretion being 62.40% (5.90%) of the administered dose. 58
The highest dose evaluated in this study (1500 mg/kg/d) corresponds to the upper limit dose recommended in the ICH guidelines, 59 and the lowest (500 mg/kg/d) was within the effective dose range (400-1000 mg/kg/d) in models of experimentally induced prostate hyperplasia in rats. 21 –25
The control mortality rate (35.0%) is consistent with previous reported studies in the literature 51 –55 and with your own data 57 and therefore supports that this study was conducted in a suitable fashion.
The mortality rates in control (21 of 60, 35.0%) and D-004-treated (62 of 180, 34.4%) females were similar, and the same occurred in control (19 of 60, 31.7%) and treated (48 of 180, 26.7%) males. Overall, 150 (31.3%) of the 480 rats died prematurely and 102 (68%) due to neoplastic lesions, without differences between treated and controls.
The overall mortality rate (122 of 400, 30.5%) found in this study agrees with that reported by other authors 51 –55 and by us. 57
Long-term oral intake of D-004 (500-1500 mg/kg/d) did not increase mortality rates versus the controls. The first deaths happened near to month 12 on treatment, consistent with the spontaneous onset of neoplastic (malignant and benign) lesions and increased mortality in this species and strain. 51 –55
Survival analysis did not show significant differences among treated and control groups. This result supports that oral administration of D-004 over the rat life span did not induce early mortality relative to the control group, which mainly includes tumor-related deaths. In addition, this finding is consistent with the lack of toxicity found in a previous long-term study in rats. 32
Consistently, daily observations, food intake, weight gain, blood biochemical indicators, organ to bodyweight ratios, and histopathology results did not find evidence of D-004-related toxicity. There were no differences in these parameters between treated and control groups, which also agrees with the data of the previous chronic 12-month toxicity study in this species at a higher dose of 2000 mg/kg/d. 32 Hematocrit and RBC counts in females treated with 1000 mg/kg/d were significantly higher than in the control group, but such significance is not biologically meaningful, since both indicators increased rather than decreased, and all individual values were within normal limits, as reported by other groups 46 and by us. 32
The use of moderate dietary restriction (DR) results in a better controlled rodent model with a lower incidence or delayed onset of spontaneous diseases and tumors. Operationally simple, moderate DR significantly improves survival, controls adult body weight, and obesity, reduces age-related renal, endocrine, and cardiac diseases, increases exposure time, and increases the statistical sensitivity of these expensive, chronic bioassays to detect a true treatment effect. A moderate DR regimen of 70% to 75% of the maximum unrestricted ad libitum food intake is recommended as a nutritionally intelligent, well-established method in conducting well-controlled toxicology and carcinogenicity studies. 39 D-004 not adversely food consumption or produce further restriction on protein intake.
The study clearly demonstrates that long-term oral treatment with D-004 in male and female rats on restricted diets did not increase the frequency or the acceleration in the occurrence of neoplastic lesions in any sex. Control and treated groups of both sexes had similar frequencies of tumors. Despite males treated with 500 mg/kg/d and all treated males as a whole had lower tumor frequencies (22% and 30%, respectively) than the controls (46%), we cannot say that long-term treatment with D-004 has antitumor effects in males, since the lack of dose-dependence limits the scope of this finding. These results should be just interpreted as an additional proof of the lack of long-term carcinogenicity of D-004 in SD rats.
Despite a number of common tumors (malignant and benign) being noted in the study, there were no significant differences between control and treated groups either individually or combined. No increase associated with treatment was seen in any case, all lesions being described as spontaneous tumors in this species/strain.
The frequency of pituitary adenoma, the commonest lesion of the study, in control females (36%) outnumbered that of males (24%), consistent with previous reports. 51 –55 No significant differences among control and treated groups were detected, with the exception of the significant reduction (P < 0.01) found in males treated with 1500 mg/kg/d versus the controls. Nevertheless, the total incidence of this lesion was lower than that reported (55%-85%) for aged SD rats. 51 –54 Our current data in males agree with that reported by our group in a previous carcinogenicity study of our group (22%), but values in females are appreciably lower than those found in such study (50%). 57
Mammary tumors (fibroadenomas and adenocarcinomas) were the most frequent tumors, consistent with other reports. 51 –54 The frequency of both tumors in control and treated females was similar. The analysis of neoplastic lesions in untreated SD rats (1340 males and 1329 females) used as controls in 17 carcinogenicity studies demonstrated that mammary tumors were the most common neoplasms in females (31.3%). 51 This result was reinforced in a recent analysis of untreated control SD rats (473 females) used in 8 carcinogenicity studies, but the frequency reported was higher (67.4%). 60 These facts support that although these lesions are the most common in long-term carcinogenicity studies in SD rats, their frequencies are variable. In our study, the frequency of mammary fibroadenomas and adenocarcinomas in control females was 20% and 22%, respectively, values somewhat lower than those reported by other authors, 51 but highly coincident with those of a previous carcinogenicity study conducted by our group: 18.3% of mammary fibroadenomas and 21.7% of adenocarcinomas. 57 These data suggest that it should be the pattern of the mammary tumors in our SD strain.
Several other tumors were detected in the study but not associated to the treatment. No significant differences among control and treated groups were detected. Pheochromocytoma, a frequent benign tumor of the adrenal medulla, was seen in only 1 male treated with 1000 mg/kg/d, a frequency (2%) lower than those referred by other authors (4.0%-12.9% in males and 4.3%-7.2% in females). 51 –53,60
Malignant and benign thyroid tumors, all reported as spontaneous in old SD rats, were also detected. These spontaneous findings included a malignant thyroid follicular cell carcinomas in 1 male treated with 1000 mg/kg/d and in other treated with 1500 mg/kg/d; while a benign thyroid solid clear cells adenoma was found in 1 male of 1500 mg/kg/d (2%) 51 –53 In this study, there was not observed preneoplastic lesions (follicular cell hyperplasia).
Uterine, liver, and subcutaneous tissue tumors in control and treated rats were also unrelated to the treatment and reported as spontaneous for this species/strain. 53,55
Taking into account the antiandrogen effect of D-004, special interest was focused in tumors related to male genital/reproductive system. Nevertheless, only 1 male treated with 1000 mg/kg/d (0.7%) exhibited cell adenoma (Leydig tumor), the most common spontaneous testicular tumor in the rat, whose frequency may reach top values of 16.3%. 51 –53
Overall, the incidences of nonneoplastic findings in the present study are consistent with those reported in the published literature. D-004 treatment did not increase the frequency of these lesions since the differences among treated and control groups were not significant. Taking together the negative results found for all indicators, the absence of treatment-related increase in nonneoplastic lesions corroborates the lack of D-004-related toxicity, consistent with the results of previous toxicology studies. 30 –36
Peliosis, the most frequent nonneoplastic lesion in this study, occurred in control females (40%) and males (8%). This lesion is very frequent in old rats, and the greater incidence in females here found agrees with that reported for other authors. 51,56 Glomerulonephrosis, the second commonest nonneoplastic lesion in the study, was found in control males (32%) and females (32%), consistent with other reports 55 and our previous data. 56 This lesion, probably the renal pathology more common in aged rats, 54 seems to be associated with the dysfunction of the basal membrane. Other inflammatory lesions were also observed. These lesions also agree with those reported for aged rats but not associated with the treatment.
Hitherto, the data here presented, including survival rates and times, tumor incidence and type, support that oral administration with D-004 for long term (24 months) did not produce carcinogenicity in SD rats, as it did not increase the occurrence of tumors (malignant or benign), did not show treatment-related invasive or accelerated tumor growth in the time, and did not induce the occurrence of neoplastic lesions not reported for this species/strain.
The reduction in the frequency of all tumors in treated males compared with the controls, mainly due to a decrease in the group treated with 500 mg/kg/d, should be considered as a random result, devoid of a meaningful sense.
We want to underline that the present results indirectly support the safety of long-term administration of substances with similar composition to D-004, as SP extracts. In light of the wide use of SP for managing BPH/LUTS in humans, 6,10,11 our results support the safety observed in clinical trials and in the empirical use of SP. In such regard, the conduct of long-term carcinogenicity/toxicity studies of natural products should not be minimized as proven by the recent demonstration of the carcinogenic effects of kava and aloe vera extracts for increasing hepatocellular and lower bowel (adenomas and carcinomas) tumors, respectively, in rats. 61 –64 We believe, therefore, that the conduct of long-term carcinogenicity/toxicity studies of natural products, mainly those used for years, should be encouraged.
In conclusion, the present results demonstrate that oral administration of D-004 (500-1500 mg/kg/d) for 24 months was devoid of long-term oral toxicity and/or carcinogenicity in male and female rats on a restricted diet, after concluding the growing period (week 13th) and that the highest dose tested (1500 mg/kg/d) was the NOAEL. In light of the intended use of D-004, results in males appear as particularly relevant.
Footnotes
Author Contributions
Ariadne Gutiérrez contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Rafael Gámez contributed to conception or design; contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Miriam Noa contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Rosa Mas contributed to conception or design; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Maikel Valle contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Nilda Mendoza contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Carlos Nodal contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Yohani Pérez contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Ámbar Oyarzábal contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Isury Bucarano contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Edy Goicochea contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Sonia Jiménez contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Haydee García contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
