Abstract
Various metals produced from human activity are ubiquitously detected in ambient air. The metals may lead to induction and/or exacerbation of respiratory diseases, but the significant metals and factors contributing to such diseases have not been identified. To compare the effects of each metal and different oxidation states of metals on human airway, we examined the viability and production of interleukin (IL)-6 and IL-8 using BEAS-2B cell line, derived from human airway epithelial cells. Airway epithelial cells were exposed to Mn2+, V4+, V5+, Cr3+, Cr6+, Zn2+, Ni2+, and Pb2+ at a concentration of 0.5, 5, 50, or 500 μmol/L for 24 hours. Mn and V decreased the cell viability in a concentration-dependent manner, and V5+ tended to have a greater effect than V4+. The Cr decreased the cell viability, and (Cr+6) at concentrations of 50 and 500 μmol/L was more toxic than (Cr+3). Zn at a concentration of 500 μmol/L greatly decreased the cell viability, whereas Ni at the same concentration increased it. Pb produced fewer changes. Mn and Ni at a concentration of 500 μmol/L induced the significant production of IL-6 and IL-8. However, most of the metals including (V+4, V+5), (Cr+3, Cr+6), Zn, and Pb inhibited the production of both IL-6 and IL-8. The present results indicate that various heavy metals have different effects on toxicity and the proinflammatory responses of airway epithelial cells, and those influences also depend on the oxidation states of the metals.
Keywords
Introduction
Particulate matter (PM) in the atmosphere is composed of solid and liquid materials that contain elemental carbon, organic carbon, inorganic salts, and metals. For example, diesel exhaust particles have a carbon core on which organic chemical components including polycyclic aromatic hydrocarbons and semivolatile organic compounds, sulfate and nitrate ions, and heavy metals are adsorbed. 1,2 Human exposure to these constituents of PM occurs through inhalation, which may lead to the induction and/or exacerbation of respiratory diseases such as asthma, chronic obstructive pulmonary disease (COPD), and lung cancer. 3 However, it is not clear which metal compositions contribute to respiratory diseases because PM is an aggregate of a particle and a large number of chemicals and metal materials, and because the compositions of PM can differ by time and place.
Previous experimental studies have indicated that not only the insoluble fraction but also the soluble fraction in PM contributes to respiratory diseases. In an in vitro study, Knaapen et al 4 have suggested that the soluble fraction as well as the insoluble particle fraction induces cellular DNA damage in human alveolar epithelial cells (A549). An in vivo study by Adamson et al 5 has demonstrated that instilling the soluble fraction to mouse lung produces inflammatory changes and lung injury. The water-soluble fraction in PM may contain various metals, and the metals can be one of the risk factors that contribute to the development/exacerbation of respiratory diseases.
Various metals emitted from human activity are ubiquitously detected in Earth’s atmosphere. 6,7 The metal oxide particles are produced from the combustion of fossil fuels and metallurgical activities. They are emitted as fly ash into the atmosphere and may be partly transformed into soluble metals when they coexist with sulfate and nitrate ions. 8,9 The solubility of metals depends on the pH and combustion conditions such as temperature and added reagents. 10,11 Epidemiological studies have shown that increases in the ambient nickel (Ni) and vanadium (V) concentrations are significantly associated with an increased probability of wheezing in young children. 12 Decrements in lung function indices associated with increasing concentrations of zinc (Zn) and iron (Fe) have been observed in patients with COPD. 13 Increases in ambient Zn have been associated with increases in asthma emergency department visits and hospital admissions among children. 14 Hexavalent chromium (Cr+6) has been generally known to cause lung cancer. 15 Laden et al 16 have reported that Ni, lead (Pb), and sulfur in the atmosphere may influence total mortality. However, it has not been fully clarified which metals generated from different sources contribute to respiratory health effects.
In this study, we focused on the effects of relatively soluble metals emitted from human activities on human airway epithelial cells. Specially, we compared the effects of metals including manganese (Mn), V, Cr, Zn, Ni, and Pb and the effects of different oxidation states of metals on cellular viability and proinflammatory responses. The critical point of the study is to compare different metals and different oxidation states under the same condition.
Materials and Methods
Cell Culture
The BEAS-2B cell line, derived from human bronchial epithelial cells transformed by an adenovirus 12-SV40 hybrid virus, was purchased from European Collection of Cell Cultures (Salisbury, Wiltshire, United Kingdom). Airway epithelial cells were seeded in 96-well or 12-well collagen I-coated plates and incubated for 72 hours to reach semiconfluence in the serum-free medium LHC-9 (Life Technologies, Carlsbad, California) at 37°C in a humidified atmosphere of 5% CO2.
Experimental Protocol
Metals (Sigma Chemical, St Louis, Missouri) including Mn, V, Cr, Zn, Ni, and Pb (purity, ≥99.6%) were used in the experiments. Mn was prepared in 1 oxidation state: Mn2+ (MnSO4·nH2O). V was prepared in 2 oxidation states: V4+ (VOSO4·nH2O) and V5+ (V2O5). Cr was prepared in 2 oxidation states: Cr3+ (Cr (NO3)3·9H2O) and Cr6+ (K2Cr2O7). Zn was prepared in 1 oxidation state: Zn2+ (ZnSO4·7H2O). Ni was prepared in 1 oxidation state: Ni2+ (NiSO4·6H2O). Pb was prepared in 1 oxidation state: Pb2+ (Pb (NO3)2). These metals were prepared in sterilized ultrapure water and/or medium.
After airway epithelial cells grew to semiconfluence in LHC-9, the cells were exposed to metals at a concentration of 0, 0.5, 5, 50, or 500 μmol/L for 24 hours. The cell viability and the release of interleukin (IL)-6 and IL-8 into the culture supernatants were evaluated by water soluble tetrazolium-1 (WST-1) assay and enzyme-linked immunosorbent assay (ELISA), respectively.
The critical point of this study was to compare the effects of different metals under the same experimental condition. Pb2+ and Ni2+ are known to cause low toxicity, whereas Cr6+ has high toxicity according to the previous reports. 17 –19 The exposure time and doses selected for this study were based on the variety of toxicities previously reported for the determination of cell viability and proinflammatory responses. Previous studies have also used similar doses and time points as those of the present study to investigate the effects of each metal including Mn2+, V4+, V5+, Cr6+, Zn2+, and Ni2+ on airway epithelial cells (Tables 1 and 2).
Literature Evaluation of IL-6 Responses Against Metals Under Various Experimental Conditions Using Airway Epithelial Cells.
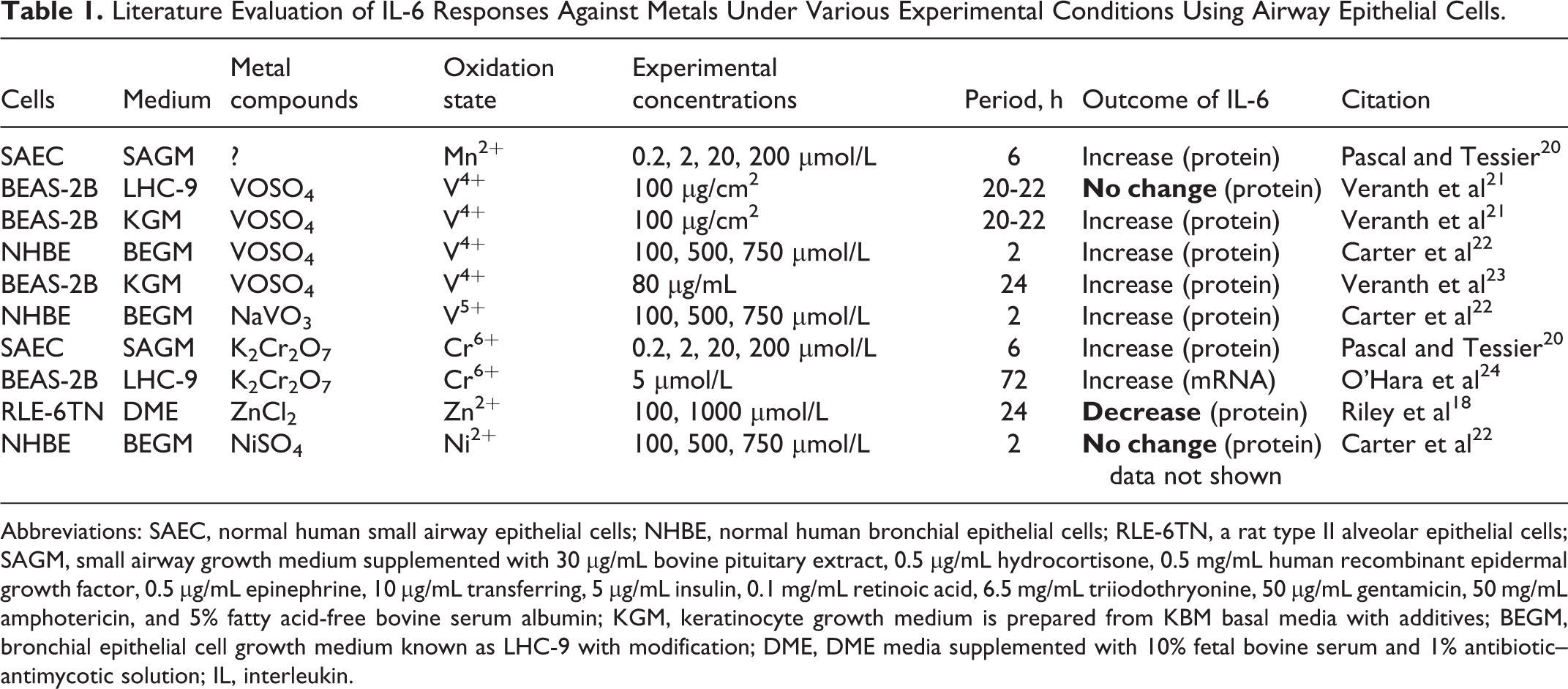
Abbreviations: SAEC, normal human small airway epithelial cells; NHBE, normal human bronchial epithelial cells; RLE-6TN, a rat type II alveolar epithelial cells; SAGM, small airway growth medium supplemented with 30 μg/mL bovine pituitary extract, 0.5 μg/mL hydrocortisone, 0.5 mg/mL human recombinant epidermal growth factor, 0.5 μg/mL epinephrine, 10 μg/mL transferring, 5 μg/mL insulin, 0.1 mg/mL retinoic acid, 6.5 mg/mL triiodothryonine, 50 μg/mL gentamicin, 50 mg/mL amphotericin, and 5% fatty acid-free bovine serum albumin; KGM, keratinocyte growth medium is prepared from KBM basal media with additives; BEGM, bronchial epithelial cell growth medium known as LHC-9 with modification; DME, DME media supplemented with 10% fetal bovine serum and 1% antibiotic–antimycotic solution; IL, interleukin.
Literature Evaluation of IL-8 Responses Against Metals Under Various Experimental Conditions Using Airway Epithelial Cells.
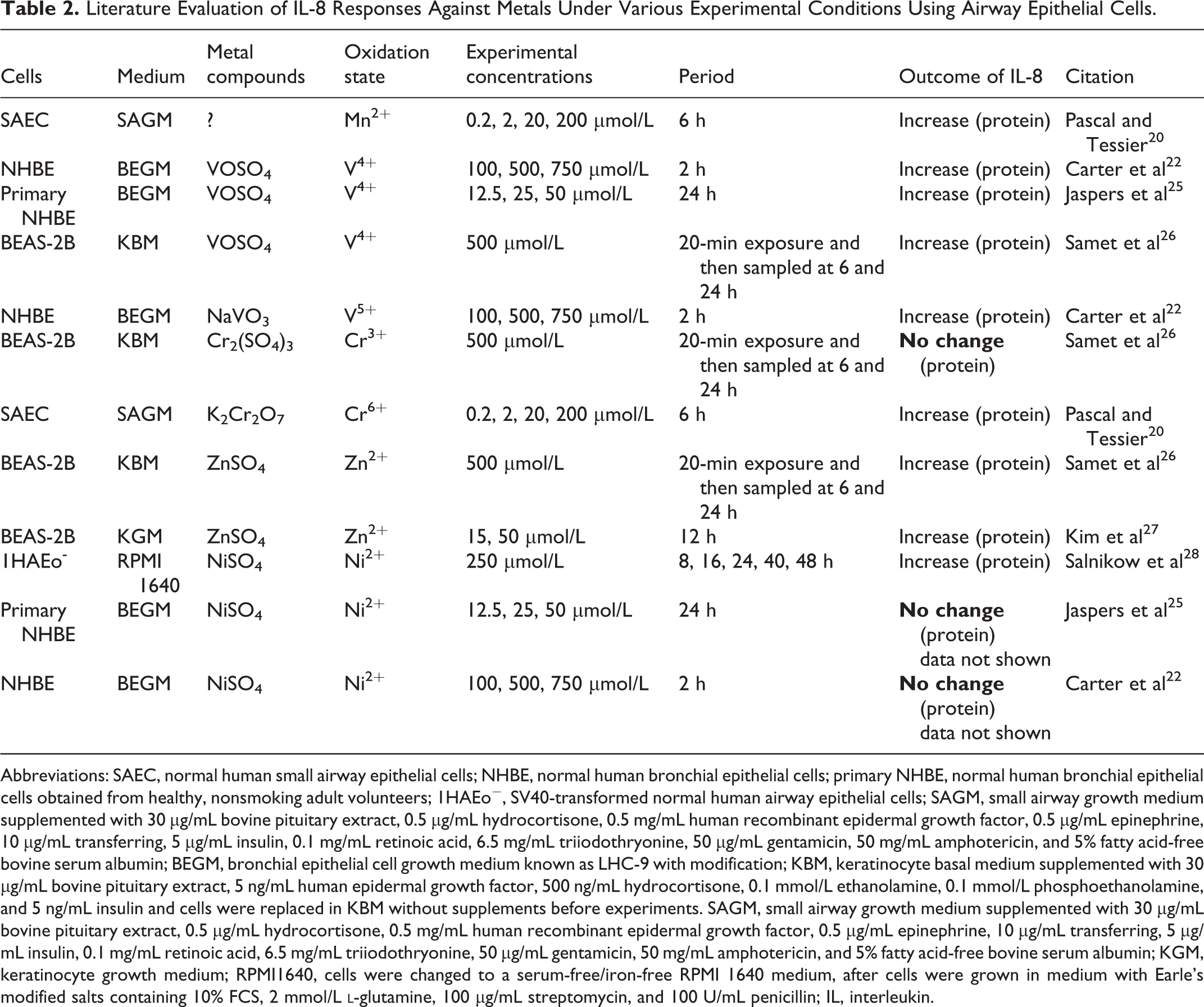
Abbreviations: SAEC, normal human small airway epithelial cells; NHBE, normal human bronchial epithelial cells; primary NHBE, normal human bronchial epithelial cells obtained from healthy, nonsmoking adult volunteers; 1HAEo
Cell Viability
Cell viability was measured by WST-1 assay using the Premix WST-1 Cell Proliferation Assay System (TaKaRa Bio, Shiga, Japan). In brief, WST-1 reagent was added to each well of 96-well plate and mixed by gently rocking the plate. Airway epithelial cells were incubated with WST-1 reagent at 37°C for 3 hours. After incubation, absorbance was measured on an iMarkMicroplate Absorbance Reader (Bio-Rad Laboratories, Hercules, California) with the wavelength at 450 nm and a reference wavelength at 630 nm. Results are expressed as the percentage of viable cells compared to untreated cells (0 μmol/L).
Quantitation of Inflammatory Proteins in the Culture Supernatants
After exposure to metals, the medium was harvested and centrifuged at 300g for 5 minutes to remove floating cells. The final supernatants were stored at −80°C until analysis. The levels of IL-6 and IL-8 (Thermo Scientific, Waltham, Massachusetts) in the culture medium were measured by ELISA according to the manufacturer’s instructions. Absorbance was measured on the iMark Microplate Absorbance Reader with the wavelength at 450 nm and a reference wavelength at 550 nm. The detection limits of the IL-6 and IL-8 assay were less than 0.8 to 1.2 and 0.5 to 2.6 pg/mL, respectively.
Statistical Analysis
Data are represented as mean ± standard error of the mean for each experimental group (n = 3-4). Differences among groups were analyzed using the Dunnett multiple comparison test (Excel Statistics 2010; Social Survey Research Information Co Ltd, Tokyo, Japan). A P value <0.05 was considered to indicate a significant difference.
Results
Effects of Metals on the Viability of Airway Epithelial Cells
We investigated the effects of metals on the cellular viability of airway epithelial cells after exposure to each metal for 24 hours (Figure 1). Mn2+, V4+, and V5+ decreased the cell viability in a concentration-dependent manner, and V5+ tended to have a greater effect on cell viability than V4+. Cr6+ and Cr3+ decreased the cell viability, and Cr6+ at concentrations of 50 and 500 μmol/L was more toxic than Cr3+. Zn2+ at a concentration of 500 μmol/L produced a dramatic decrease in cell viability. In contrast, Ni2+ at a concentration of 500 μmol/L increased cell viability, whereas Pb2+ showed less change. The order of toxicity based on toxic concentration 50% (TC50) values (concentration that reduces cell viability to 50%) was Mn2+ (3.0 μmol/L) > Cr6+ (7.5 μmol/L) > V5+ (36.3 μmol/L) > V4+ (86.8 μmol/L) > Zn2+ (427.6 μmol/L). Ni2+, Pb2+, and Cr3+ showed low or no inhibition of cellular viability, and therefore, their TC50 values could not be calculated.
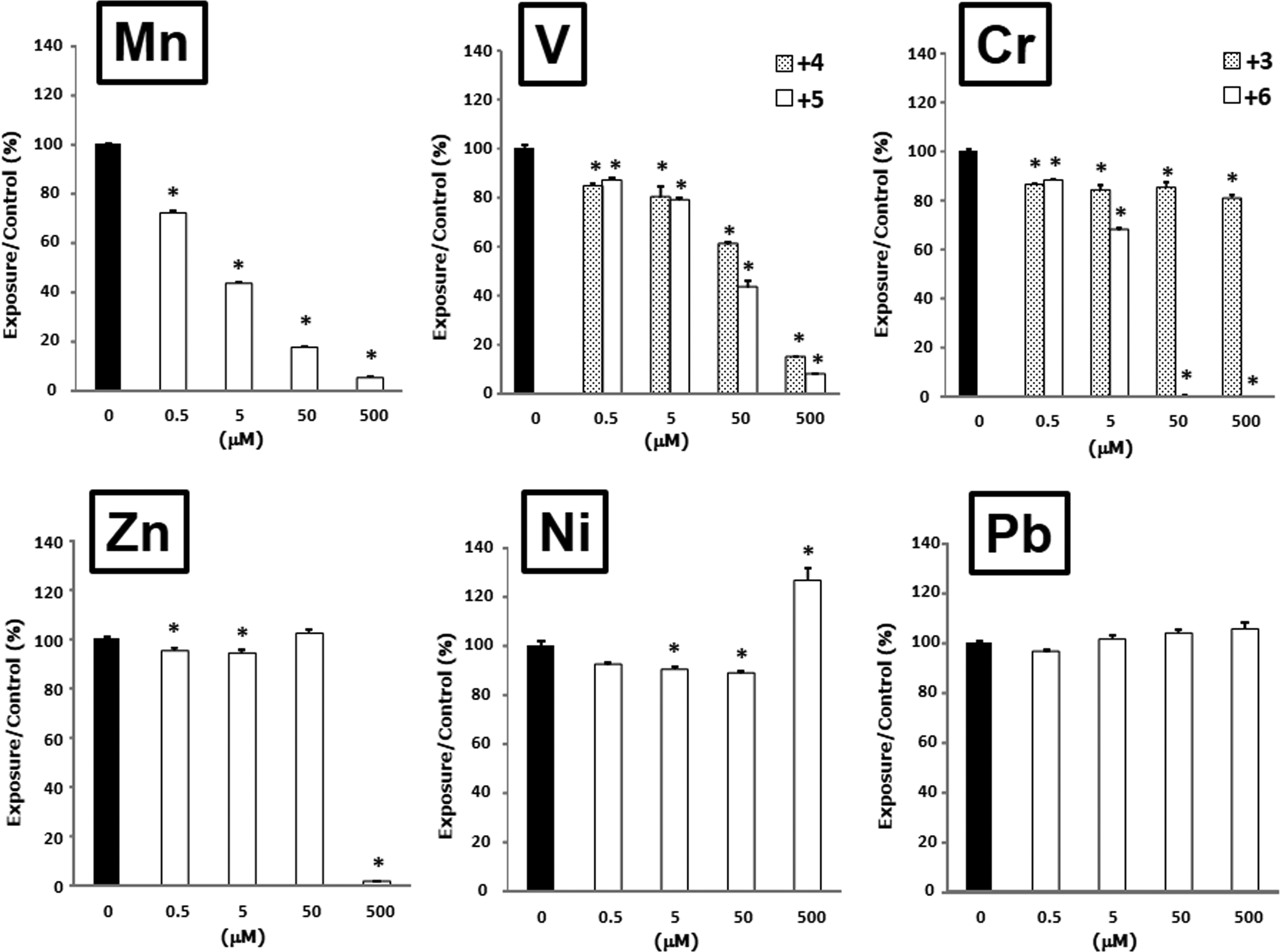
Effects of metals on the viability of human airway epithelial cells. Cells were treated with the indicated concentrations of metals for 24 hours. Cell viability was assessed by WST-1 assays. Data are presented as the percentage of the viability of the control (0 μmol/L). Data are mean ± standard error of the mean (SEM) of 3 to 4 individual cultures. *P < 0.05 versus 0 μmol/L.
Effects of the Metals on Cytokine Production From Airway Epithelial Cells
Airway epithelial cells, which have important roles in the physical barrier and immunological responses against xenobiotics, are a source of cytokines. We investigated the effects of the metals on the proinflammatory responses of human airway epithelial cells, and we examined the productions of IL-6 (Figure 2) and IL-8 (Figure 3) after exposure to each metal for 24 hours.
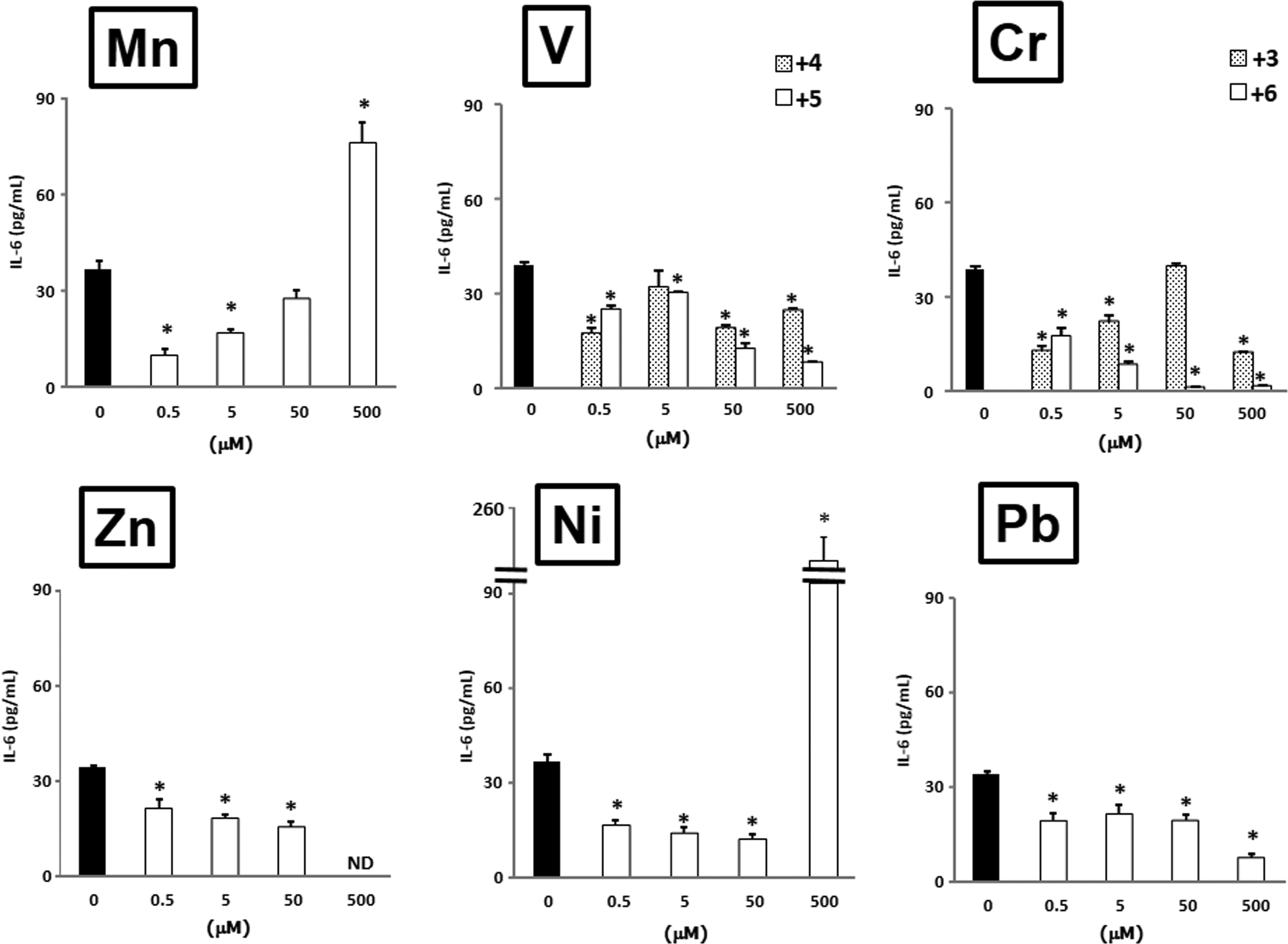
Interleukin 6 (IL-6) production from airway epithelial cells in response to metals. The protein levels in the culture supernatant after exposure to metals for 24 hours were measured by enzyme-linked immunosorbent assay (ELISA). Data are mean ± standard error of the mean (SEM) of 3 to 4 individual cultures. *P < 0.05 versus 0 μmol/L. ND indicates not detected.
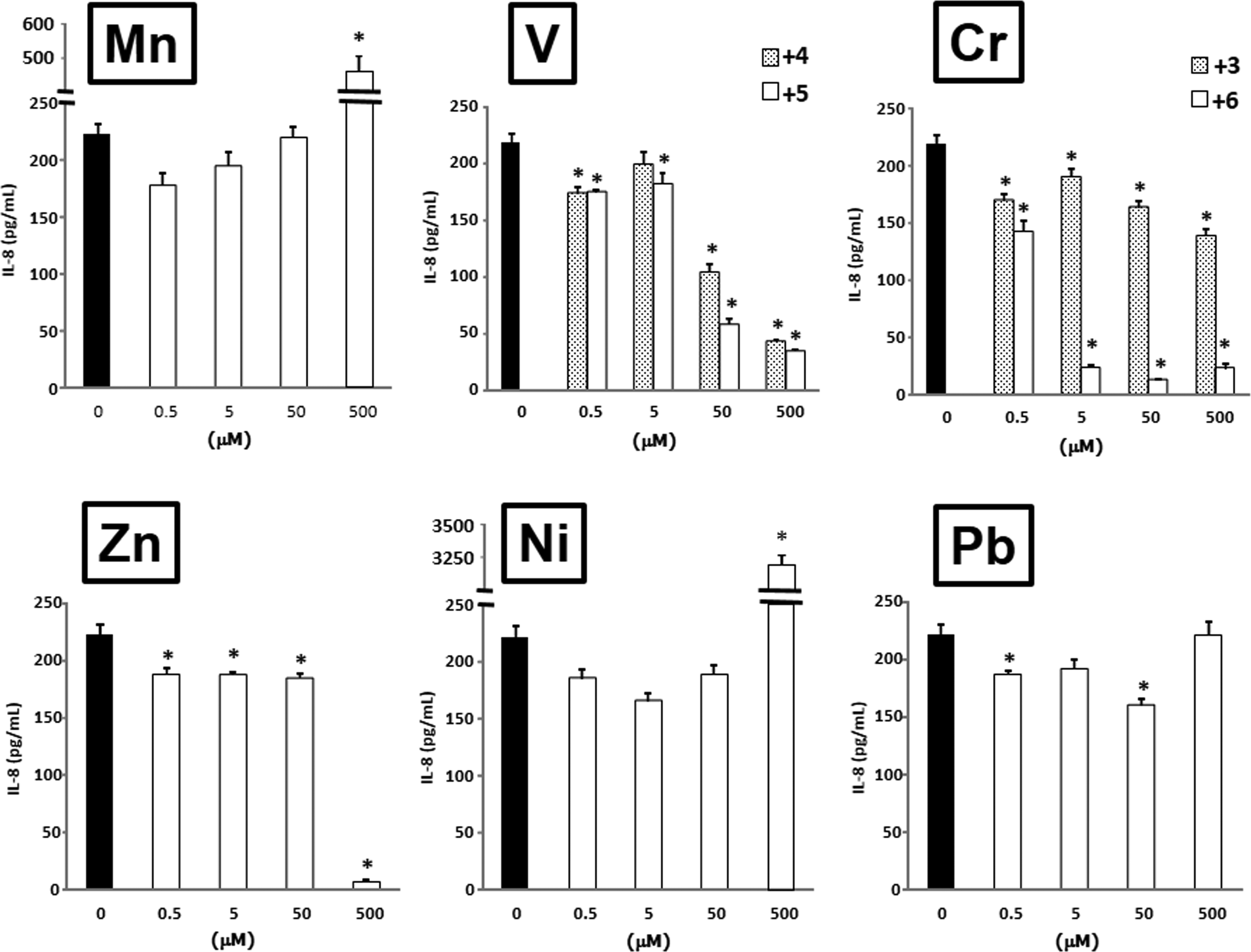
Interleukin 8 (IL-8) production from airway epithelial cells in response to metals. The protein levels in the culture supernatant after exposure to metals for 24 hours were measured by enzyme-linked immunosorbent assay (ELISA). Data are mean ± standard error (SE) of 3 to 4 individual cultures. *P < 0.05 versus 0 μmol/L.
Mn2+ at a concentration of 500 μmol/L elevated the IL-6 release, but the levels of IL-6 at the concentrations of 0.5 and 5 μmol/L were significantly lower than the level at 0 μmol/L. V4+ and V5+ at a concentration of 0.5 μmol/L decreased the release of IL-6 compared to 0 μmol/L, whereas the levels of IL-6 recovered after exposure at 5 μmol/L, and then the levels at concentrations of 50 and 500 μmol/L decreased again. V5+ tended to be more influential than V4+. Cr6+ decreased the release of IL-6 in a concentration-dependent manner. Cr3+ depressed the release of IL-6 except for exposure at a concentration of 50 μmol/L. Zn2+ concentration dependently decreased the IL-6 release, and the IL-6 level at the 500 μmol/L exposure was below the detection limit. In contrast, Ni2+ at a concentration of 500 μmol/L markedly increased the level, whereas Ni2+ at concentrations of 0.5, 5, and 50 μmol/L lowered the level of IL-6. Pb2+ inhibited the IL-6 release at all concentrations.
In the results of IL-8, the IL-8 protein release showed a similar tendency to that of IL-6 release. Mn2+ at a concentration of 500 μmol/L greatly elevated the IL-8 release. V4+ and V5+ decreased the release of IL-8 in a roughly concentration-dependent manner. V5+ tended to be more influential than V4+. Cr3+ and Cr6+ decreased the release of IL-8 at all concentrations. The effects of Cr6+ at concentrations of 5, 50, and 500 μmol/L were stronger than those of Cr3+. Zn2+ at concentrations of 0.5, 5, and 50 μmol/L decreased the levels of IL-8, and the level at a concentration of 500 μmol/L exposure showed a dramatic inhibition. Ni2+ at a concentration of 500 μmol/L produced an extreme elevation of IL-8 release; the levels of IL-8 at a concentration of 500 μmol/L were 14-fold higher than those at 0 μmol/L. Pb2+ at concentrations of 0.5 and 50 μmol/L decreased the IL-8 release.
Mn2+ and Ni2+ elevated the IL-6 and IL-8 protein releases at 500 μmol/L, at which concentration Mn2+ showed inhibition of viability and Ni2+ demonstrated no toxicity. The other metals significantly decreased IL-6 and IL-8 protein release.
Discussion
We found that exposing human airway epithelial cells to some metals affected the cell viability and changed proinflammatory responses via the expression of IL-6 and IL-8. The responses of these biomarkers showed differing profiles when exposed to a variety of metals and oxidation states.
In this study, the order of toxicity based on TC50 values was Mn2+ (3.0 μmol/L) > Cr6+ (7.5 μmol/L) > V5+ (36.3 μmol/L) > V4+ (86.8 μmol/L) > Zn2+ (427.6 μmol/L). Ni2+, Pb2+, and Cr3+ showed low or no inhibition of cellular viability. It is especially notable that Mn2+ as well as Cr6+ at low concentrations showed greater toxicity against airway epithelial cells cultured in serum-free LHC-9 medium. Although Mn2+ has been known to induce the lung inflammation in experimental investigation, 29 comparative studies among various metals are little performed. Our results clarified that Mn2+ is a highly toxic metal against airway cells, even among various metals. Riley et al 18 have indicated that the ranking of metal toxicity is V4+ (VCl4) > Zn2+ (ZnCl2) > Cu2+ > Ni2+ (NiCl2) > Fe2+ in a rat lung epithelial cell line (RLE-6TN). In addition, the order of cytotoxicity in BEAS-2B cultured in keratinocyte growth medium has been Cd2+ > Cr6+ (CrO3) > Pt4+ > Pd2+ (PdSO4) = Pt2+ > Ni2+ (NiCl2) > Rh3+. 19 Pascal and Tessier 20 have reported that Cr6+ (K2Cr2O7) and Mn2+, but not Ni2+, are cytotoxic to BEAS-2B cultured in F-12 medium with 10% fetal bovine serum. It is difficult to compare the previous studies with our results, because the culture conditions and metal complexes differ. However, the results on the ranking of metal toxicity based on TC50 values in the present study resembled those of previous studies. 18 –20,29 These results suggest that relatively soluble metals in sulfate, nitrate, oxides, and dichromate form which may exist in the atmosphere 9,30 have different cellular toxicities among a variety of metals and oxidation states of metals.
IL-6 and IL-8 are proinflammatory cytokines induced by environmental insults, and they play important roles in inflammation in the respiratory system by stimulating lymphocytes, inducing neutrophils recruitment, and upregulating mucin secretion. 31 –34 Our study focused on nonspecific inflammatory responses and cell viability in airway epithelial cells rather than specific inflammatory responses by immune cells. Therefore, IL-6 and IL-8 were measured as nonspecific inflammatory response markers. In addition, in our past experiments, IL-6 and IL-8 released from BEAS-2B changed in response to some air pollutants such as Asian sand dust particles. 35 Moreover, the effect of these molecules in vitro correlated well with airway inflammation after in vivo exposure by bronchoalveolar lavage. 36 In this study, Ni2+ and Mn2+ especially elevated the release of IL-6 and IL-8 among metals.
Interestingly, although Ni2+ at the high dose increased viability and release of IL-6/IL-8, Ni2+ at lower doses resulted in reduced viability and release of IL-6/IL-8. It has been reported that exposure to Ni2+ (NiSO4) induces the secretion of IL-8 in airway epithelial cells, 28 and that Ni2+ compounds induce oxidative stress. 37 Ni2+ at the high dose may cause proinflammatory responses via reactive oxygen species (ROS). Ni2+ at the high dose may also induce metallothionein which is an antioxidant and a cytoprotective protein against metal toxicities. 38 It has been also reported that high levels of metallothionein in the nucleus of cells contribute to promoting cell proliferation. 39 In brief, increased proinflammatory responses at the high dose have possibility to occur via ROS. Moreover, increased viability may relate to the effects of metallothionein induced by Ni2+. On the other hand, Ni2+ at lower doses may not produce ROS, although the expression of metallothionein may be slightly induced in response to Ni2+. Therefore, metallothionein as an antioxidative molecule may scavenge ROS under control conditions, and Ni2+ at lower doses mainly inhibit the release of IL-6 as shown in the present study. Accordingly, the cellular events at the high dose can be different from those at the low doses.
Mn2+ at a high dose showed inhibition of viability and elevation of IL-6/IL-8 release. It has been reported that exposure to Mn2+ induces the secretion of IL-6 or IL-8 in airway epithelial cells, 20 and that Mn2+ compounds induce oxidative stress. 40 Mn at a dose of 500 μmol/L showed high toxicity against airway epithelial cells. After Mn induces proinflammatory protein in the early stages, cell death such as necrosis may happen.
The other metals significantly decreased IL-6 and IL-8 protein release. The inhibitory effect on IL-6 and IL-8 caused by V4+, V5+, Cr6+, and Zn2+ at high concentrations may be due to the cytotoxic effect. However, apart from the results obtained with high concentrations, the present findings are inconsistent with those of previous studies. Some studies have reported that Mn2+, V4+, V5+, Cr6+, Zn2+, and Ni2+ (and not Cr3+ and Pb2+) induce IL-6 and IL-8 from airway epithelial cells (Tables 1 and 2). On the other hand, in this study, Cr3+ reduced cell viability to about 80% at all doses and depressed IL-6 release except for a concentration of 50 μmol/L. Pb2+ decreased IL-6/IL-8 in some doses with no cytotoxicity. Although Cr3+ and Pb2+ are known to show low cellular toxicity, 17,41 there are few studies showing changes to proinflammatory cytokines in airway epithelial cells. The discrepancies appear to depend on the differences in cells, media, and metal complex forms. Veranth et al 21 have suggested that the IL-6 response to V4+ (VOSO4) treatments changes when the same cells, BEAS-2B, are grown in KGM or LHC-9 medium. In brief, BEAS-2B in LHC-9 has shown no response to V4 +, whereas BEAS-2B in KGM has produced IL-6. The Veranth group has noted that the method of cell passaging, and the exact growth factors in the media, is likely to affect both the populations of receptors on the cell surface and the intracellular signal transduction. Actually, in addition to the findings reported by Veranth et al, 21 unchanged response of IL-6 and IL-8 to Ni2+ and decreased response of IL-6 to Zn2+ have been also observed. Salnikow et al, 28 Carter et al, 22 and Jaspers et al 25 have shown different IL-8 responses after Ni2+ exposure in different cells and media (Table 2). There have also been reports that the IL-6 release in RLE cultures significantly decreases in response to Zn2+ exposure at a concentration of 100 μmol/L, which causes <20% cell death (Table 1, Riley et al 18 ). Further investigations are needed to understand the meanings and the mechanism by which metals decrease IL-6 and/or IL-8 release.
We investigated the effect of different oxidation states of V and Cr on human airway epithelial cells. V4+ and Cr3+ are known to be more stable compared with V5+ and Cr6+, respectively. When humans inhale PM, airway epithelial cells may often encounter V4+ and Cr3+. In the aqueous in vitro setting, V5+ and Cr6+ may partially change into chemical forms of V4+ and Cr3+. In this study, even though they are the same element, different oxidation states of V and Cr have demonstrated different behaviors in biological reaction.
V in the atmosphere results from the combustion of residual fuel oil. It has been reported that most of the V spectra in the combustion of residual fuel oil closely resembles those of VOSO4, and oxide, probably V2O5. 8 In present study, V5+ (V2O5) tended to be more influential than V4+ (VOSO4). It has been reported that the toxicity caused by the ingestion of V5+ (V2O5) is higher than the toxicity caused by V4+ (VOSO4). 42 However, Pierce et al 43 have reported that the intratracheal instillation of V4+ (VOSO4) in rat induces a higher neutrophil influx in bronchoalveolar lavage than that of intratracheal instillation of V5+ (V2O5). They have suggested that V5+ (V2O5) would dissolve less quickly in surfactant of the lung. The toxicity of V compounds may differ by exposure routes (oral or intratracheal instillation) in vivo study. However, in an in vitro study using V4+ (VOSO4) and V5+ (Na3VO4), Carter et al 22 have indicated that V4+ (VOSO4) and V5+ (Na3VO4) are equally potent in inducing the production of IL-6, and V4+ (VOSO4) induces slightly higher levels of IL-8 than V5+ (Na3VO4) in normal human bronchial epithelial cells cultured in BEGM media. As mentioned previously, the differences in oxidation state may also depend on the cell type, culture condition, and metal complex. The impact of V compounds on proinflammatory reactions in the human airway has not been clear. Further investigations are needed to understand the different behaviors of V4+ and V5+ in biological reaction.
The oxidation states of Cr which exist in the atmospheric environment are Cr3+ and Cr6+. Chemical reactions between Cr3+ and Cr6+ would occur in the aqueous phase of PM. The unstable species Cr6+ reduce to Cr3+ under typical atmospheric conditions. 44 Previous studies have suggested that Cr6+ is more toxic than Cr3+ in multiple types of cells, and that Cr6+ causes cytotoxicity during the reduction of Cr6+ to Cr3+ in cells. 41 Accordingly, as V4+ and V5+, or Cr3+ and Cr6+ show different responses, it is important to understand the effect of the differences in oxidation state on the airway as well as the differences in metal element.
Humans can be exposed to air pollutants containing various metals. However, in this test system, we have not examined combinations of metals. The synergistic or antagonistic relationships may occur depending on the metal transporter and cell signal transduction. Indeed, it has been reported that the simultaneous addition of iron in either ferric or ferrous form and nickel completely inhibits IL-8 production in the 1HAEo cells. 28 This will be a subject for future analysis.
In addition to the immortalized BEAS-2B cell line used as an in vitro model, we may need studying sensitivity against each metal under conditions that are more close to in vivo such as primary cells. Because BEAS-2B cells have inherent limitations in cell culture studies although BEAS-2B is one of the cell lines used to evaluate biological responses induced by environmental pollutants. For examples, Mn-superoxide dismutase activity has been able to be lower in BEAS-2B cells than in primary cultures. 45 Moreover, BEAS-2B cells in 2-dimensional systems fail to undergo mucociliary differentiation. Recently, polarized human airway epithelial cells in air–liquid interface (ALI) cultures are developing as a respiratory model, they enable mucociliary transport. 46,47 The mucus on the apical side of airway epithelial cells protects from environmental stimuli. These characteristics may have impacts on metal toxicity as involves the production of ROS. Accordingly, in vitro exposures using not only BEAS-2B cell lines but also primary cells and/or ALI cultures may enable us to compare with real human exposures adequately.
Conclusion
The present study obtained comparative data among metals. We have found that exposure to various heavy metals results in differing cell toxicity and proinflammatory responses of airway epithelial cells, and these differences also depend on the oxidation states of the metals. The biological reaction of airway epithelial cells to metals in the atmosphere can lead to airway damage and the development/exacerbation of respiratory diseases.
It has been reported that ambient PM2.5 increases and/or decreases proinflammatory protein in airway epithelial cells. 48 –50 The components of ambient PM2.5 including metals differ in place and time, which changes the effects on respiratory health. In brief, increased and/or decreased proinflammatory protein may depend on quantity of metals. Accordingly, this study contributes to elucidating mechanism by which air pollutants cause/inhibit proinflammatory responses in real exposed situation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partly supported by Japan Automobile Research Institute and Uehara Memorial Foundation.
