Abstract
Continued improvements in the ballistic properties of military munitions have led to metal formulations for which little are known about the long-term health effects. Previously we have shown that a military-grade tungsten alloy comprised of tungsten, nickel, and cobalt, when embedded into the leg muscle of F344 rats to simulate a fragment wound, induces highly aggressive metastatic rhabdomyosarcomas. An important follow-up when assessing a compound’s carcinogenic potential is to test it in a second rodent species. In this study, we assessed the health effects of embedded fragments of 2 military-grade tungsten alloys, tungsten/nickel/cobalt and tungsten/nickel/iron, in the B6C3F1 mouse. Implantation of tungsten/nickel/cobalt pellets into the quadriceps muscle resulted in the formation of a rhabdomyosarcoma around the pellet. Conversely, implantation of tungsten/nickel/iron did not result in tumor formation. Unlike what was seen in the rat model, the tumors induced by the tungsten/nickel/cobalt did not exhibit aggressive growth patterns and did not metastasize.
Introduction
The expanded use of novel materials on today’s battlefields, both in military munitions and in improvised explosive devices, is of critical importance when considering the types of wounds sustained. Increasingly, the types of injuries include wounds with embedded metal fragments. 1,2 Since standard surgical guidelines recommend leaving embedded fragments in place, except under certain circumstances, a wounded individual may carry these fragments for decades. In most cases, the toxicological and carcinogenic properties of these metals and metal mixtures have not been investigated. For example, armor-penetrating depleted uranium (DU) munitions were first widely used in the 1991 to 1992 Persian Gulf War and resulted in a number of personnel affected by wounds consisting of embedded fragments of this material. Investigations in rodents initially showed no adverse long-term health effects as a result of implanted DU pellets 3 ; however, subsequent investigation revealed that by greatly increasing the size or surface area of the implanted material, sarcomas could eventually be induced at the implantation site. 4 Extensive medical surveillance of the personnel with the retained DU fragments almost 20 years after being injured has demonstrated no adverse health effects. 5,6 Nonetheless, because of the uncertainty over long-term health effects, many nations, including the United States, have searched for alternatives to DU in armor-penetrating munitions.
Because of its density and high melting point, tungsten is used, in various forms, in a variety of applications. Tungsten has also been used as a replacement for lead in small-caliber ammunition. 7 In the early 1990s, the use of lead shot was banned for the hunting of waterfowl in the United States because of the risk of lead poisoning in waterfowl secondary to ingestion of spent ammunition. 8 In attempting to find an acceptable replacement, the US Fish and Wildlife Service advocated the use of ammunition formulations that were not toxic when ingested by wildlife. Many of the subsequently approved ammunitions contain varying amounts of tungsten in combination with other metals such as nickel, tin, iron, copper, and bismuth. Toxicity testing has shown no adverse health effects of these materials when administered by several routes. 9 -14
As noted earlier, concern over the health and environmental impact of the use of DU has led many countries to search for alternatives and many have begun to replace DU with various tungsten alloys. In many of these formulations, tungsten is combined with 2 or more of the following transition metals: nickel, cobalt, iron, and copper. However, the manufacturing processes for armor-penetrating munitions are quite different than for hunting ammunition. This leads to questions, similar to those originally surrounding DU, as to the health effects of exposure to the tungsten alloys, especially for embedded fragment exposures. In fact, research has shown that one of the military-grade tungsten alloys (tungsten/nickel/cobalt) is a potent carcinogen when implanted in the leg muscles of rats. 15,16 In vitro assessments of the effect of tungsten alloy exposure on cultured cells have demonstrated that tungsten/nickel/cobalt and tungsten/nickel/iron can transform human osteoblast cells to a tumorigenic phenotype. 17 The transformation frequencies observed were greater than those seen following treatment with nickel subsulfide, a known carcinogen. 18 In other in vitro model systems, tungsten/nickel/cobalt treatment was shown to activate gene expression in human liver carcinoma cells (HepG2) affecting signal transduction pathways that could play a role in toxicity and carcinogenicity. 19 In addition, activation appeared to be induced by a synergistic effect of the alloy as activation was far greater for the complete mixture as opposed to the individual metals alone. Treatment of rat muscle cells with tungsten/nickel/cobalt resulted in a decrease in cellular viability 20 and an increase in the production of reactive oxygen species causing significant DNA damage. 21 These studies raise serious concerns as to the long-term health effects of embedded tungsten alloy fragments. The need to confirm the carcinogenicity of such alloys in another rodent species (ie, B6C3F1 mouse, as recommended by the National Toxicology Program) is an important second step required in biological as well as regulatory terms to better assess the cancer risk in humans.
Materials and Methods
Animals
Male B6C3F1 mice were purchased from Harlan (Dublin, Virginia) at 3 to 4 weeks of age and maintained in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International in accordance with the Guide for the Care and Use of Laboratory Animals. 22 The Armed Forces Radiobiology Research Institute’s Animal Care and Use Committee (IACUC) approved all procedures described here. Upon arrival, all mice were screened for common rodent pathogens. Mice were housed in groups (5 mice/group) in plastic microisolator cages with hardwood chips for bedding (Sani-Chips; PJ Murphy Forest Products, Corp, Montville, New Jersey). Mice were fed standard rodent chow (Global Rodent Diet 8604; Harlan Teklad, Madison, Wisconsin) with water provided ad libitum. Animals were on a 12-hour light/dark cycle. Health status was monitored daily, and animals were weighed weekly. In order to identify and monitor the animals in the study, mice were injected subcutaneously (sc) in the mid-dorsal thoracic region with a small transponder (Electronic Lab Animal Monitoring System, BioMedic Data Systems, Seaford, Delaware). The transponder was programmed with a unique animal identification number using a personal computer. The identification number can then be read using a low-power radio frequency scanner.
Metal Pellets
All pellets were cylinders 1 mm in diameter by 2 mm in length. Tantalum (99.95% Ta) and nickel (99.995% Ni) pellets were purchased from Alfa Aesar (Ward Hill, Massachusetts). Tungsten alloy pellets were manufactured by Aerojet Ordnance Tennessee (Jonesborough, Tennessee) using standard kinetic-energy penetrator production methods. Two military-grade tungsten alloys were fabricated: first alloy (WNiCo) consisted of 91.1% tungsten (W), 6% nickel (Ni), and 2.9% cobalt (Co). The second alloy (WNiFe) consisted of 91% tungsten (W), 7% nickel (Ni), and 2% iron (Fe). Prior to implantation, all pellets were cleaned and chemically sterilized using the following procedure. Pellets were placed in 1 mL of 100% ethanol and sonicated for 5 minutes at room temperature. The pellets were then rinsed in 70% ethanol, washed for 3 minutes in 50% nitric acid with gentle agitation, washed with sterile water, rinsed with 70% ethanol, and allowed to air dry. The pellets were placed in tared sterile vials and the weight of the washed pellets obtained. The pellets were covered with 70% ethanol (1-2 mL) until needed. Prior to implantation, the pellets were rinsed with 0.9% saline (USP grade) and blotted dry on sterile filter paper (Whatman #1).
Pellet Implantation Procedures
Anesthesia was induced with continuous administration of isoflurane using an open circuit system with a scavenger/recapture system. The implantation sites were clipped and cleansed with betadine (Purdue Pharma, LP, Stamford, Connecticut). A small incision was made through the skin to reveal the quadriceps. Two pellets were implanted into each quadriceps. Implantation was accomplished by loading the sterilized pellet into a 16-gauge needle, inserting a specially designed plunger inside the needle, pushing the needle into the quadriceps, then depressing the plunger. This forces the pellet out of the needle and into the muscle. Incision sites were closed with tissue adhesive (VetBond; 3M Corp, St Paul, Minnesota). The mice were closely monitored after pellet implantation until they were ambulatory and an analgesic (buprenorphine, 0.1 mg/kg, sc; Rickitt and Colman, Hull, United Kingdom) administered prior to implantation surgery and then as needed. The pellet implantation sites were examined daily for signs of inflammation and infection for 2 weeks after surgery, then weekly thereafter for the duration of the study.
Experimental Groups
The pellet implantation groups included a surgical sham, tantalum, nickel, and 2 tungsten alloys (WNiCo and WNiFe). Except for the surgical sham, all mice received 4 pellets of test metal (2 pellets per quadriceps). The following 5 experimental time points were run: 1-, 3-, 6-, 12-, and 24-month postimplantation. The 1-, 3-, 6-, and 12-month experimental points had 10 mice/group, while the 24-month time point had 20 mice/group. In addition, for the 24-month experimental points only, there was an additional set of 20 mice each that were implanted with 2 pellets of test metal and 2 pellets of the inert tantalum (to keep metal loads equivalent). These groups were designated as the “low-dose” groups, as opposed to the 4 pellets of test metal, which were designated as the “high-dose” groups. Upon reaching their experimental end point or when indicated by guidelines approved by the IACUC, mice were humanely euthanized by isoflurane overdose followed by exsanguination and confirmatory pneumothorax per the guidelines of the American Veterinary Medical Association. Criteria for euthanasia included tumor size greater than 1 cm in diameter, loss of greater than 10% of baseline body weight, or other indications of approaching moribundity as determined by animal health assessments. After euthanasia, a complete gross pathology examination was conducted, and a variety of tissues isolated for analysis. The wet weights of spleen, testes, liver, and kidney were determined and normalized to body weight. A blood sample for hematological assessments was acquired via cardiac puncture, as was a sample for serum preparation for metal analysis. In addition, a urine sample for metal analysis was removed from the bladder using a syringe and a 25-gauge needle. Urine and serum samples were stored at −80°C until assayed.
Hematology
White and red blood cell counts, hemoglobin, hematocrit, and platelet, lymphocyte, monocytes, and granulocyte counts were obtained on collected blood using a ScilVet Hematology Analyzer (Scil Animal Care Company, Gurnee, Illinois).
Pathology
Tissues for histopathology, including brain, kidney, liver, spleen, testes, and muscle surrounding the implanted pellets, were fixed in buffered zinc formalin (Z-Fix; Anatech Ltd, Battle Creek, Michigan), processed and embedded in paraffin, sectioned at 5 to 6 µm, and stained with hematoxylin and eosin. For immunohistochemical analysis, 5-µm thick sections of formalin-fixed paraffin-embedded tissue were deparaffinized with xylene and rehydrated with graded ethanol washes. After heat-induced antigen retrieval using a commercially available solution (Epitope Retrieval Solution; IHC World, LLC, Woodstock, Maryland), the tissue was reacted with rabbit antidesmin antibody (Abcam, Cambridge, Massachusetts) overnight at 4°C. After washing, color development was achieved using the Rabbit ABC Staining System (Santa Cruz Biotechnology, Inc, Santa Cruz, California) with diaminobenzidine as the peroxidase substrate. The tissue was then counterstained with hematoxylin, dehydrated, and mounted before microscopic examination. Both histopathological and immunohistochemical assessment were conducted by board-certified veterinary pathologists.
Sample Preparation for Metal Analysis
Urine samples were diluted prior to analysis. Serum samples were wet-ashed with 5 mL of 70% nitric acid (Optima Ultrapure Grade; Fisher Scientific, Pittsburgh, Pennsylvania) and 200 µL of 30% hydrogen peroxide (Semiconductor Grade; Sigma-Aldrich Chemical Co, St Louis, Missouri). The samples were heated to just below boiling to avoid sample splashing until completely evaporated. The samples were then dry-ashed for 12 hours at 600°C in a muffle furnace (Fisher Scientific) prior to another cycle of wet-ashing with nitric acid and hydrogen peroxide. After the second run of wet-ashing, the resulting white residue was dissolved in 2% nitric acid and analyzed. Tissue samples were processed by first drying the tissue in a muffle furnace at 100°C for 24 hours. The temperature of the furnace was then increased to 350°C, at 5°C/min, and the samples held there for 24 hours. The furnace temperature was then increased to 600°C, at 5°C/min, and the samples held there for 48 hours. After cooling, the samples were wet- and dry-ashed as described previously.
Metal Analysis
Metal content of the collected samples was determined using an inductively coupled plasma-mass spectrometer (XSeries 2 ICP-MS; ThermoElectron North America, LLC, Madison, Wisconsin) equipped with a Cetac ASX520 Autosampler (Cetac Technologies, Omaha, Nebraska). The plasma gas was high-purity (99.997%) liquid argon. Instrument calibration was accomplished using the appropriate metal standards (SPEX CertiPrep, Metuchen, New Jersey) in 2% HNO3. Metal concentration levels were obtained by reference to the slope of the calibration curve (counts per second/ng/mL) as well as an internal standard. Urine data were normalized to creatinine levels with creatinine content determined with a commercially available kit (Oxford Biomedical Research, Oxford, Michigan).
Statistical Analysis
Survival data were graphed using the Kaplan-Meier procedure 23 and analyzed for statistical significance with the log-rank test. Median survival times were analyzed using the Wilcoxon Mann-Whitney U test. Body weight gain, hematology results, metal concentration determinations, and organ weight data were analyzed by a one-way analysis of variance to assess the effect of metal implantation. If a significant effect was observed, Dunnett’s test was conducted to determine which groups were statistically different from control. In all cases, P < .05 was considered statistically significant.
Results
The pellets used for this project are cylindrical, 1 mm in diameter by 2 mm in length. Although the Ta and Ni pellets are commercially available, the tungsten alloy pellets (WNiCo and WNiFe) were manufactured by Aerojet Ordnance using the same heat-sintering procedures used to produce armor-penetrating munitions for the US military. Nickel was chosen as a positive control since it is a known carcinogen. 18,24 Tantalum functioned as the implantation control as it is considered inert and has been used in other studies 3,15 and implanted prosthetic devices 25 -27 without any adverse effects. The rodent model system used for the investigation of the health effects of embedded metal fragments has been previously validated 28 and used successfully in numerous other rodent metal implantation studies. 3,15,16,29 As with the other studies, no signs of infection, local metal toxicity, or mobility issues were observed as a result of metal implantation. The mice gained weight normally throughout the experiment with the exception of the Ni-implanted animals, which gained weight at a significantly slower rate than the control groups in both the low- and high-dose cohorts (data not shown). The low-dose WNiFe-implanted mice showed statistically slower weight gain than controls from weeks 10 to 14 after pellet implantation, but returned to normal after that. The high-dose WNiCo-implanted mice also gained weight at a slower rate than controls, but this difference did not become statistically significant until week 32 postimplantation. The rate of weight gain in high-dose WNiFe- and Ta-implanted mice was not statistically different from control.
Survival of the metal-implanted mice is shown in Figure 1 (panel A—low-dose groups; panel B—high-dose groups). Of the low-dose groups, only Ni-implanted mice showed significantly lower survival as determined by the log-rank test, with a median survival time of 83.5 weeks (Table 1). Mice in both the high-dose Ni- and WNiCo-implanted groups exhibited significantly decreased survival times (Figure 1) with median survival times of 61.5 and 72.5 weeks, respectively (Table 1). Survival and median survival times of the low- and high-dose WNiFe-implanted mice, as well as the Ta-implanted mice, were not significantly different than the surgical-sham control groups. As part of the weekly health assessment, the pellet-implantation sites were palpated to determine any abnormal growth around the implanted metal pellets. Abnormal tissue growth was only found in Ni- and WNiCo-implanted mice; although, as seen in Table 2, the time to tumor development varied widely. However, in all cases, the latency period for tumor formation was significantly shorter in the Ni-implanted mice and the tumor incidence higher than for the WNiCo-implanted mice.
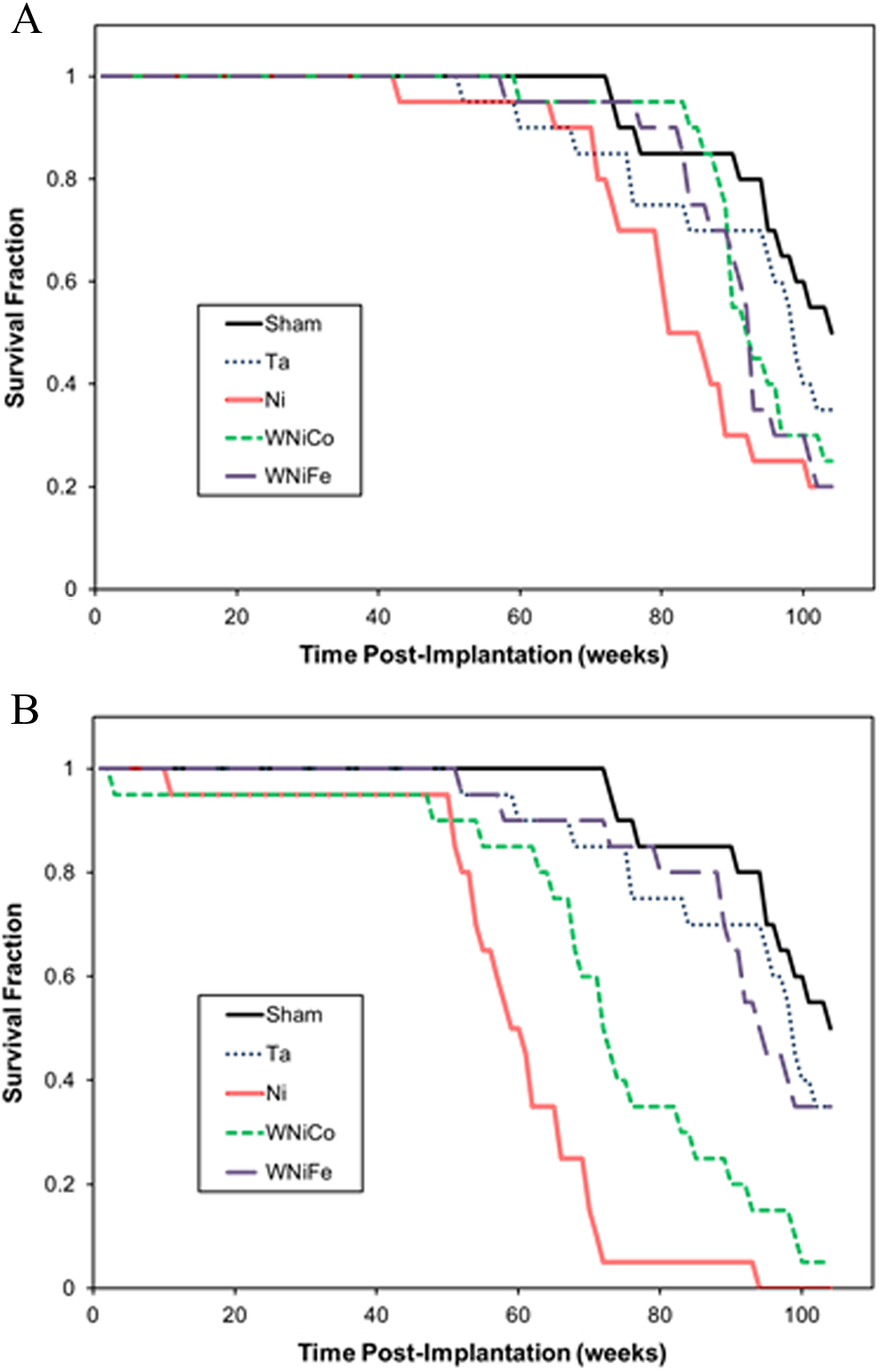
Kaplan-Meier survival curves for metal-implanted mice. Panel A, low-dose groups. Panel B, high-dose groups.
Median Survival Age for Metal-Implanted Mice.a
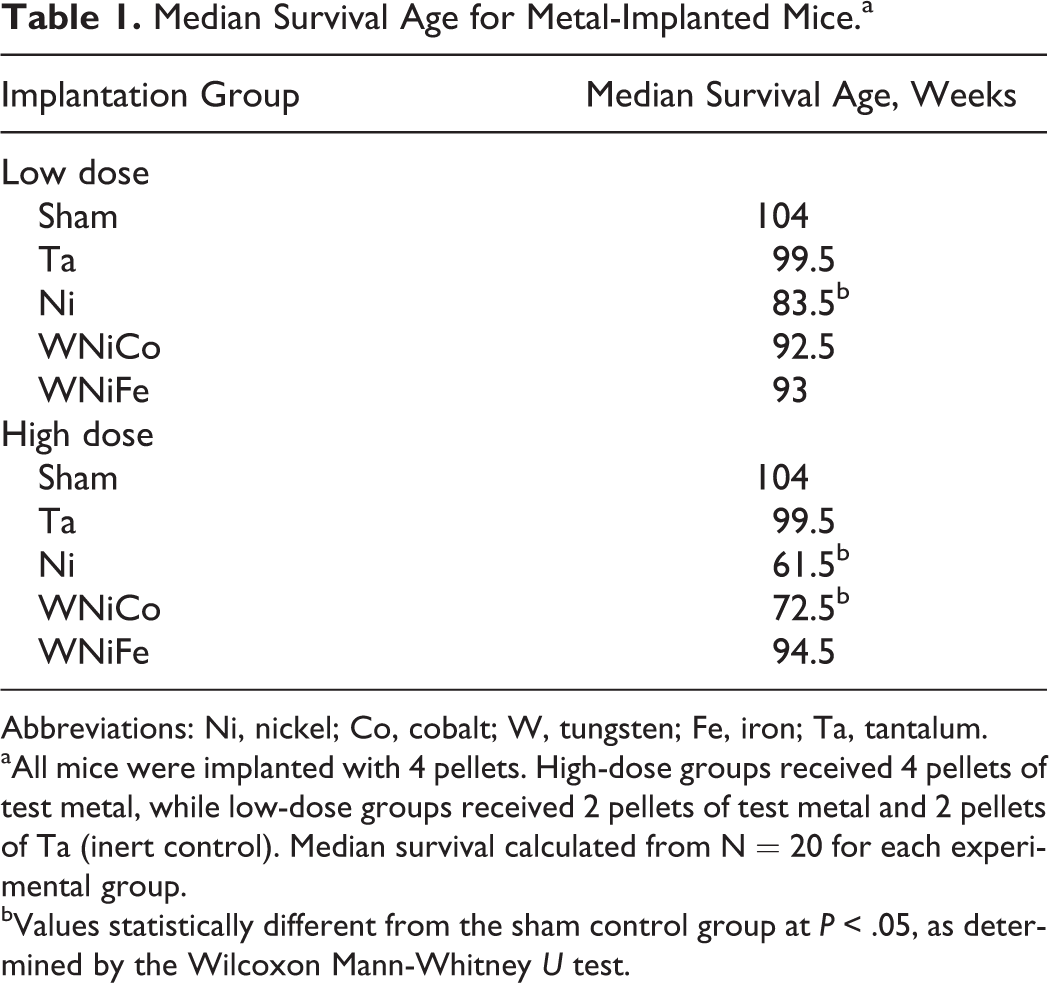
Abbreviations: Ni, nickel; Co, cobalt; W, tungsten; Fe, iron; Ta, tantalum.
aAll mice were implanted with 4 pellets. High-dose groups received 4 pellets of test metal, while low-dose groups received 2 pellets of test metal and 2 pellets of Ta (inert control). Median survival calculated from N = 20 for each experimental group.
bValues statistically different from the sham control group at P < .05, as determined by the Wilcoxon Mann-Whitney U test.
Metal-Induced Tumor Incidence for Ni- and WNiCo-Implanted Mice.a
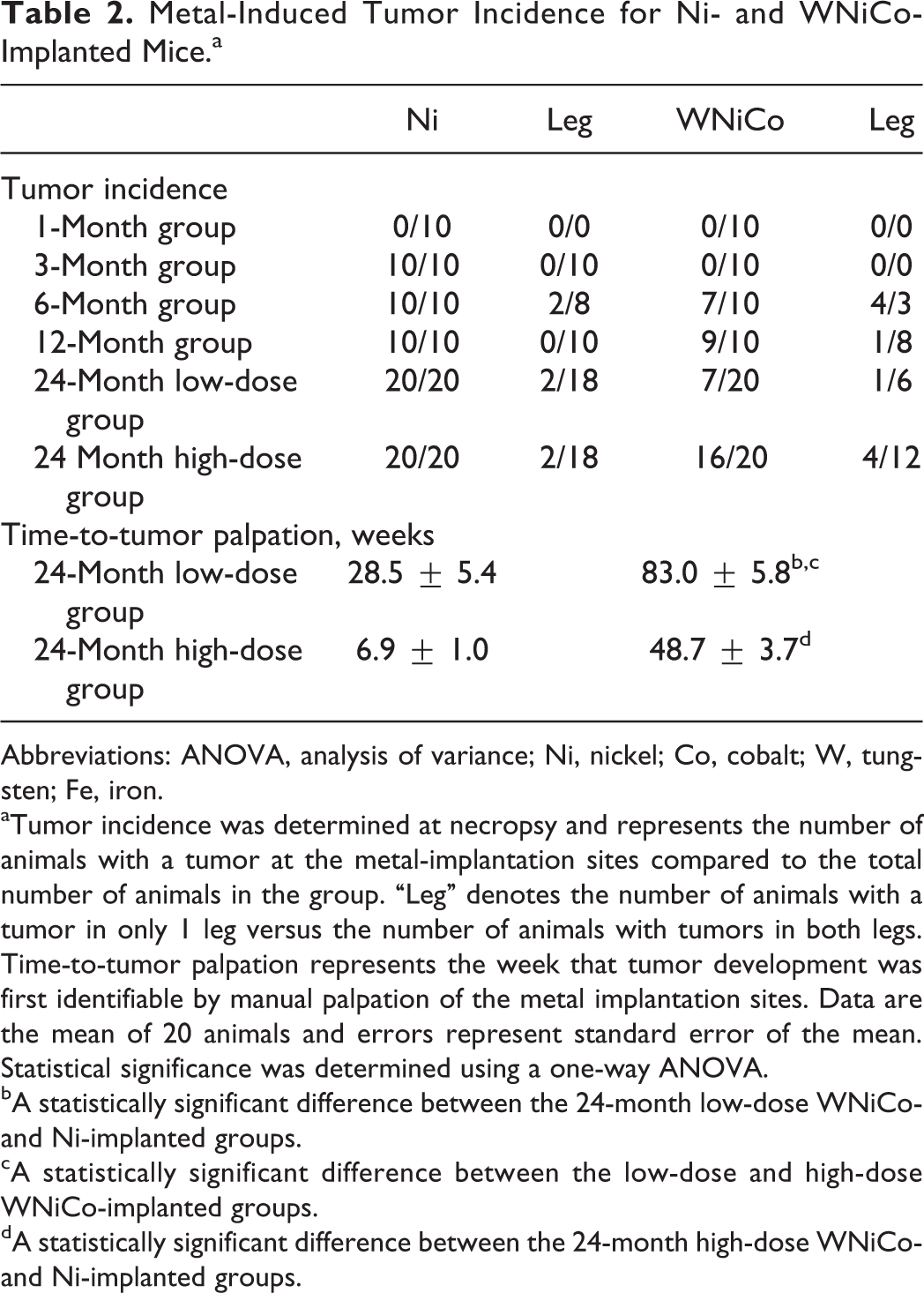
Abbreviations: ANOVA, analysis of variance; Ni, nickel; Co, cobalt; W, tungsten; Fe, iron.
aTumor incidence was determined at necropsy and represents the number of animals with a tumor at the metal-implantation sites compared to the total number of animals in the group. “Leg” denotes the number of animals with a tumor in only 1 leg versus the number of animals with tumors in both legs. Time-to-tumor palpation represents the week that tumor development was first identifiable by manual palpation of the metal implantation sites. Data are the mean of 20 animals and errors represent standard error of the mean. Statistical significance was determined using a one-way ANOVA.
bA statistically significant difference between the 24-month low-dose WNiCo- and Ni-implanted groups.
cA statistically significant difference between the low-dose and high-dose WNiCo-implanted groups.
dA statistically significant difference between the 24-month high-dose WNiCo- and Ni-implanted groups.
Upon euthanasia and prior to a full necropsy and tissue collection, a blood sample was obtained and analyzed for a variety of hematological parameters including white blood cells, red blood cells, hemoglobin, hematocrit, platelet, lymphocyte, monocyte, and granulocyte numbers. There were no consistent deviations from control values observed for the implanted animals. Similarly, no prolonged and significant changes were observed in organ/body weight ratios among the different groups for spleen, kidney, liver, and testes.
In both the WNiCo- and Ni-induced tumors, the metal pellet could be found in the interior of the tumor. No metal-induced tumors were found in any of the other experimental groups. Histopathological examination of hematoxylin and eosin-stained sections of the tumors revealed invasive sarcomas with areas of necrosis, hemorrhage, infiltration, and effacement or destruction of pre-existing tissues (Figure 2). Some had a progression of lesions surrounding the pellet site, with a gray or amphophilic deposition of material (corroded alloy). The tumors had high mitotic rates (sometimes as high as 10 mitotic figures/high-power field) and occasional bizarre mitoses. Morphology varied from spindle cells forming streams and bundles and occasional strap-like cells (characteristic of rhabdomyosarcomas) to a pattern with many pleomorphic, some giant cells, and even a few multinucleated giant tumor cells (pleomorphic rhabdomyosarcoma). Immunohistochemical analysis using antidesmin antibody demonstrated that the tumors were most likely derived from skeletal muscle myoblasts (Figure 2, panel D). Neither the Ni- nor WNiCo-induced tumors metastasized as has been reported for studies using these compounds in a rat model system. In addition, no other metal group-specific tumors were identified.
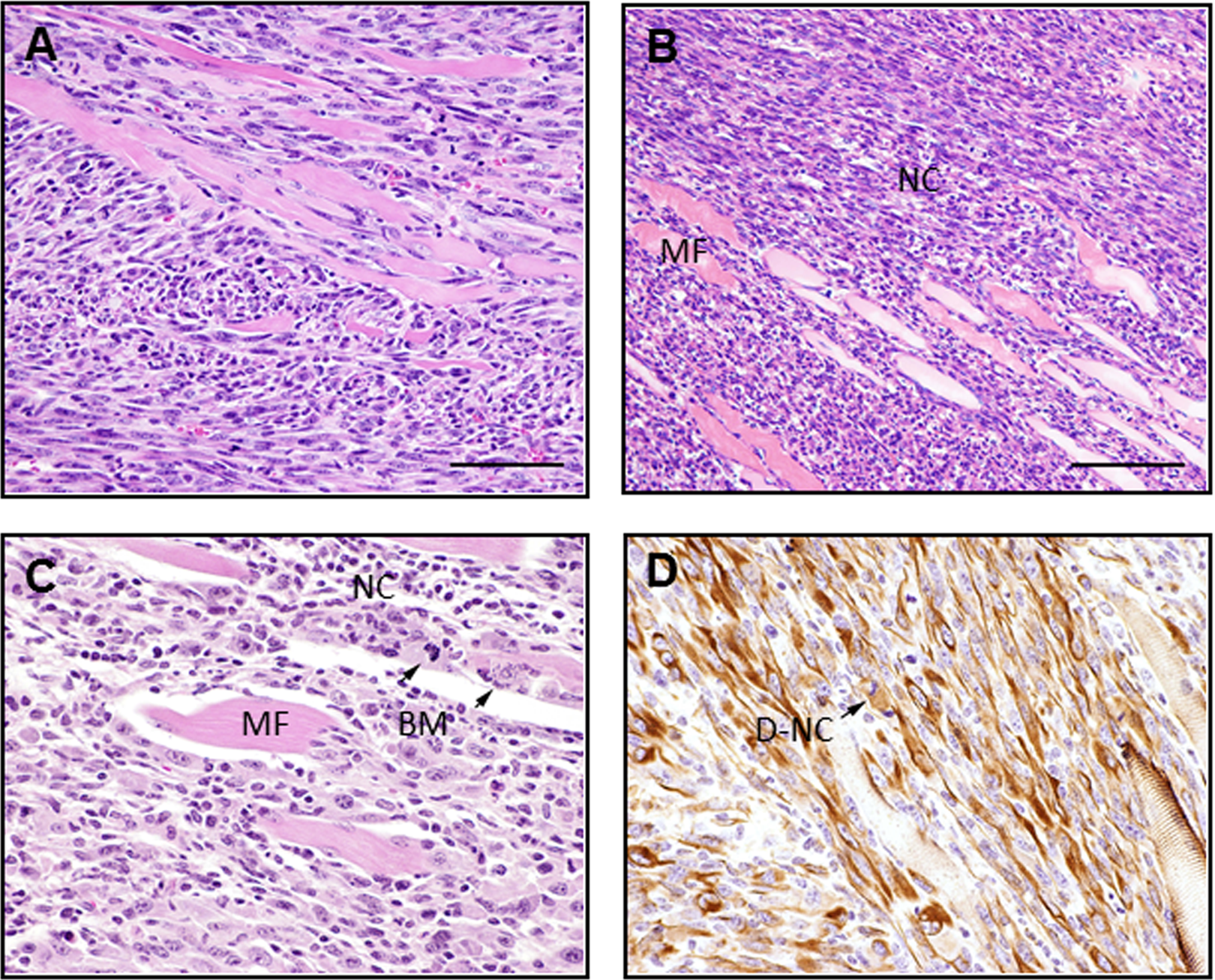
Histopathologic examination of leg tumor surrounding WNiCo pellet (high-dose group, 67 weeks). Panels A and B, H&E-stained sections of tumor surrounding WNiCo pellet showing neoplastic cells (NCs) infiltrating normal muscle fibers (MFs). Magnification ×400. Panel C, H&E-stained WNiCo-induced tumor section showing normal muscle (MF) surrounded by neoplastic cells (NC) with bizarre mitoses (BM). Magnification ×600. Panel D, desmin staining of neoplastic cells (D-NC). Magnification ×600. H&E indicates hematoxylin and eosin; Ni, nickel; Co, cobalt; W, tungsten.
At euthanasia, a variety of tissues, including brain, kidney, liver, spleen, testes, and femur, as well as serum and urine, were collected and analyzed for metal content using inductively coupled-plasma mass spectrometry. To assess the in vivo degradation of the tungsten alloy pellets, a comparison of tungsten levels in tissues from WNiFe- and WNiCo-implanted mice is shown in Figure 3. For brevity, only the tungsten levels are shown. Because of the high levels of endogenous iron in most tissues, as well as in the food and water consumed by the mice, coupled with the low levels of iron in the WNiFe pellets, measurement of iron levels in tissue provided no useful information as to the in vivo degradation of the WNiFe pellets. On the other hand, endogenous levels of nickel and cobalt in tissues are generally very low, and tungsten levels in normal mouse tissue are often below the level of detection. Therefore, tungsten levels are excellent indicators of metal solubilization from implanted tungsten alloy pellets. In all cases, tungsten levels in tissues from WNiFe-implanted mice were 10- to 20-times lower than those found in tissues from WNiCo-implanted mice (Figure 3). In addition, tissue tungsten levels in the WNiFe-implanted groups generally peaked early then returned to near background values, while tissue tungsten levels in the WNiCo-implanted mice increased throughout the life of the animal. Tungsten also deposited in the femur in both the WNiFe- and WNiCo-implanted groups, although to a much greater extent in the WNiCo groups (Figure 4). As the embedded metals solubilized, they were also excreted in the urine as seen in Figure 5. Again, urinary metal levels in the WNiFe-implanted groups were much lower than those of the WNiCo-implanted groups. Tungsten, solubilized from the implanted pellets, was able to cross the blood–brain barrier and deposit in the brain. Tungsten levels in the brain of the control and Ni-implanted groups were below the level of detection. For the WNiFe-implanted animals, tungsten was only measurable at the 1-month time point, but the level was not statistically significant. After this time, brain tungsten levels were below the level of detection. Conversely, in the WNiCo-implanted groups, brain tungsten levels rose from a mean ± standard error of 2.2 ± 0.4 ng/g of tissue for the 1-month implantation group to a high level of 35.9 ± 3.5 ng/g of tissue for the 24-month implantation group, indicating a continued release of metal from the implanted pellets.
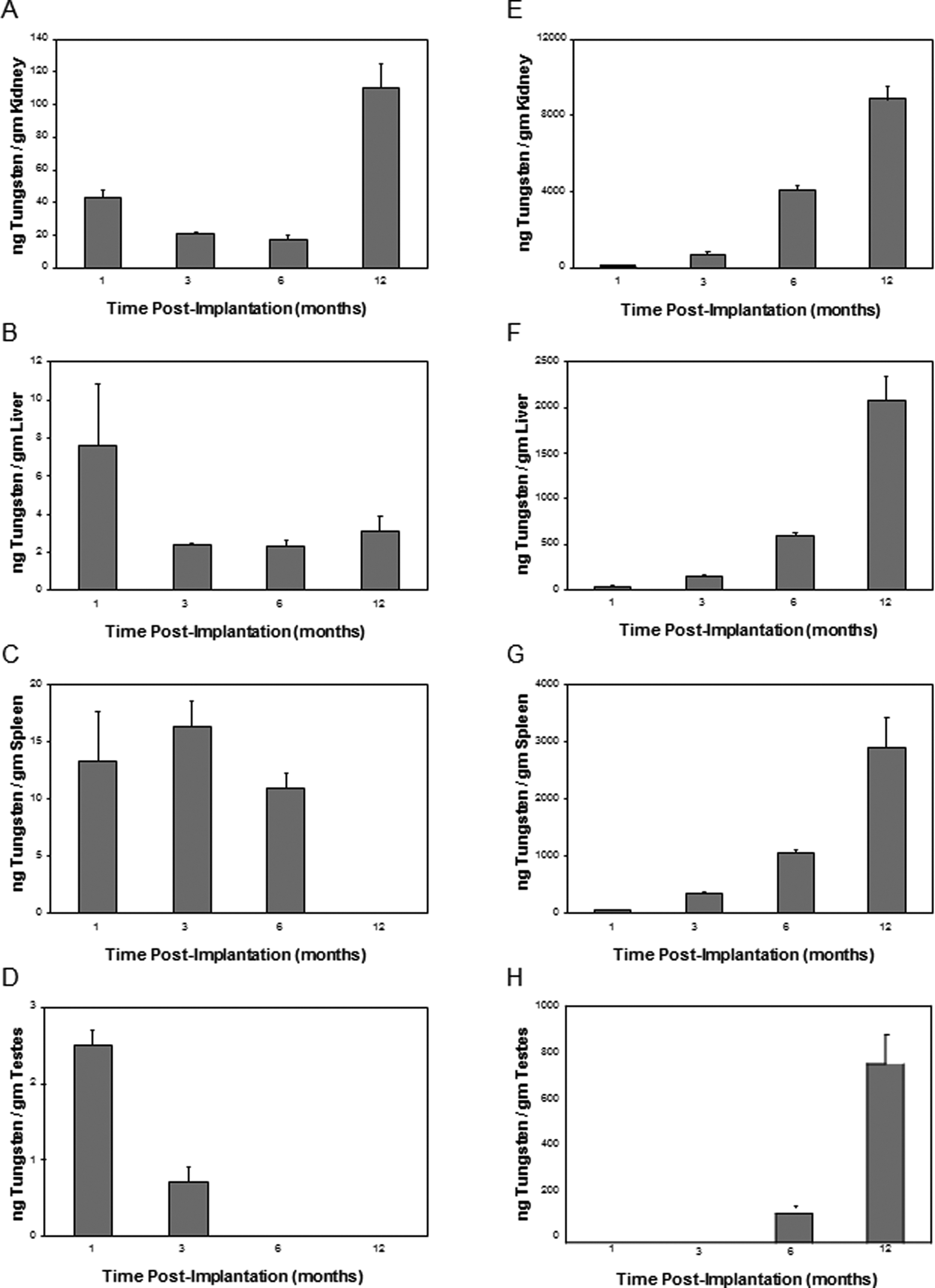
Tissue tungsten levels. Graphs show the mean and standard error of the mean for 10 independent tungsten measurements of tissues from 1-, 3-, 6-, and 12-month implantation groups. Data are expressed as ng of tungsten per gram of tissue. Panel A, WNiFe kidney; panel B, WNiFe liver; panel C, WNiFe spleen; panel D, WNiFe testes; panel E, WNiCo kidney; panel F, WNiCo liver; panel G, WNiCo spleen; and panel H, WNiCo testes. In all cases, the tungsten levels were significantly higher (P < .05, one-way analysis of variance [ANOVA]) than control, except for the following low-dose groups: 12-month spleen and 6- and 12-month testes. Note that the y-axis values are not identical between the graphs. Ni indicates nickel; Co, cobalt; W, tungsten; Fe, iron.
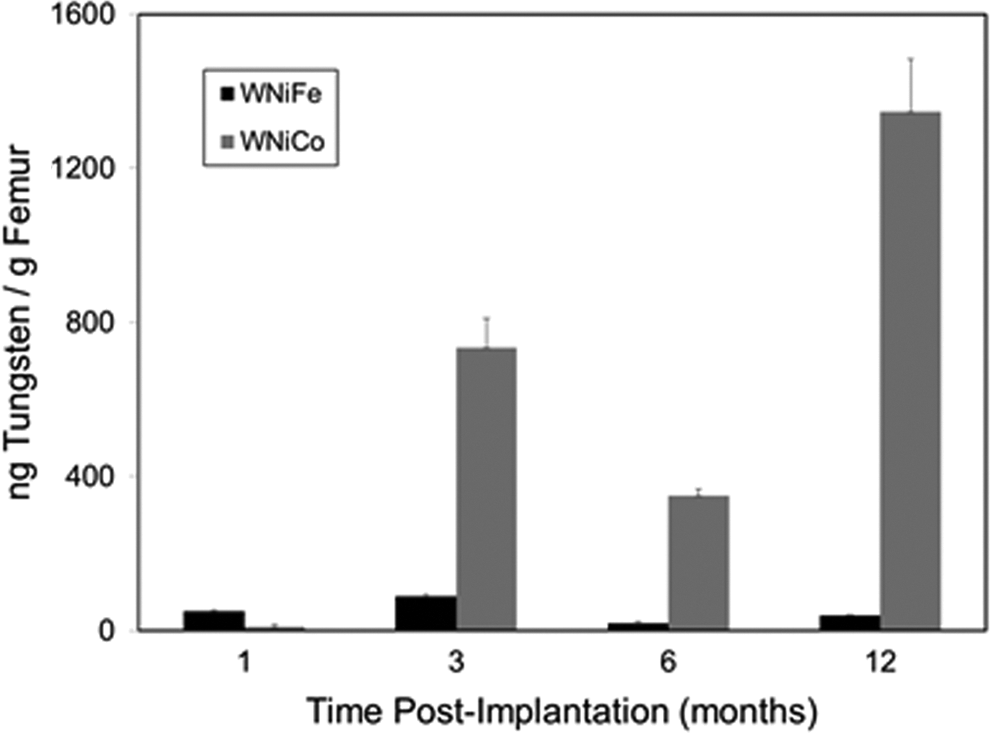
Femur tungsten levels. Graph shows the mean and standard error of the mean for 10 independent tungsten measurements of femurs from 1-, 3-, 6-, and 12-month implantation groups. Data are expressed as ng of tungsten per gram of tissue. Black bars: WNiFe groups; gray bars: WNiCo groups. In all cases, the femur tungsten levels were significantly higher (P < .05, one-way analysis of variance [ANOVA]) than control. Ni indicates nickel; Co, cobalt; W, tungsten; Fe, iron.
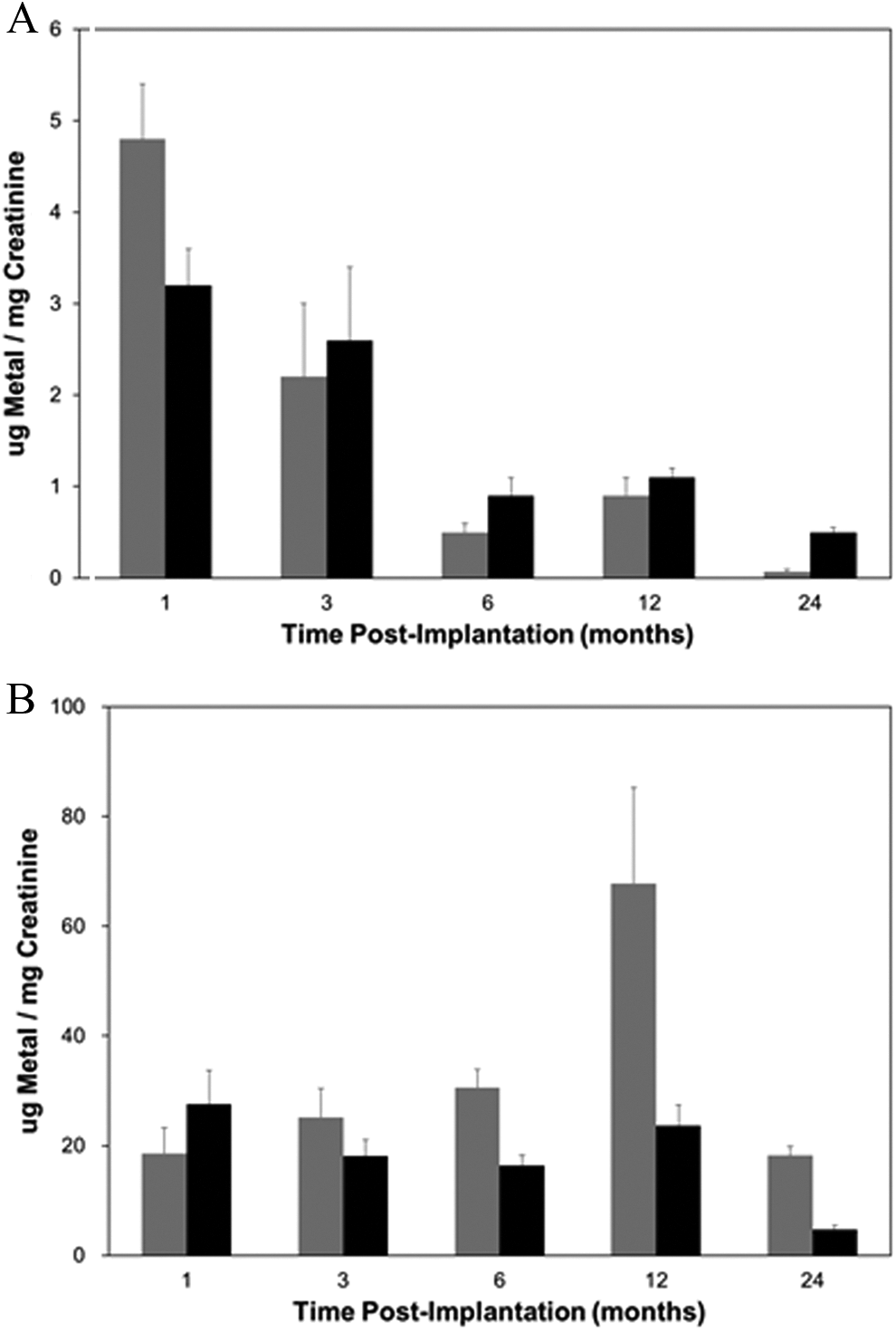
Urinary metal levels. Graph shows the mean and standard error of the mean for 10 independent measurements of urinary tungsten (gray bars) and nickel (black bars) from 1-, 3-, 6-, 12-, and 24-month implantation groups. Data are expressed as µg of metal per milligram of creatinine. Panel A, WNiFe groups; panel B, WNiCo groups. Note that the y-axis scales are not identical. Ni indicates nickel; Co, cobalt; W, tungsten; Fe, iron.
Discussion
The search for munition composites that are less toxic and more environmentally friendly has led many countries to investigate the use of tungsten-based materials as replacements for lead in small-caliber ammunition and DU in armor-penetrating rounds. One tungsten-based composition, WNiCo, proposed as a replacement for DU, has been shown to induce metastatic rhabdomyosarcomas when implanted in the leg muscles of laboratory rats. 15,16 Regulatory guidance recommends testing in a second rodent species before a definitive determination of carcinogenicity is made. Previous studies on tungsten alloy were conducted in the F344 rat; therefore, in order to more thoroughly assess the carcinogenic potential of these materials, this study utilized the B6C3F1 mouse. Two military-grade tungsten alloys (WNiCo and WNiFe) were tested for adverse health effects when implanted in the leg muscles of mice in order to simulate an embedded fragment wound.
The WNiFe formulation, when implanted into the mouse quadriceps muscle, resulted in no adverse health effects throughout the life of the animal. There was no difference in survival between the WNiFe-implanted mice and controls. There were no hematological or organ-specific alterations due to the WNiFe and no signs of neoplastic changes were found at the pellet implantation sites. Tissue and urinary tungsten levels were slightly, yet significantly, elevated early after metal implantation, but returned to near normal levels at the later experimental time points. This is similar to the results previously reported where the amount of tungsten solubilized from an implanted WNiFe pellet in an F344 rat decreased over time due to the formation of an impenetrable layer on the surface of the pellet that was attributed to the presence of iron in the mixture. 16 As in this investigation, that study also found no adverse health effects resulting from implanted WNiFe pellets.
Conversely, mice implanted with WNiCo pellets survived for shorter periods of time and developed tumors at the pellet implantation sites. As in the previously published rat studies, 15,16 these tumors were identified both histopathologically and immunohistochemically as rhabdomyosarcomas. Unlike the tumors in the rat model, the tumors that developed in the B6C3F1 mouse were only locally aggressive and did not metastasize, although the metal loads were higher. Based on the weight of the tungsten alloy pellets, the number of pellets implanted, and the body weight of the animal at implantation, the tungsten alloy loads for the low-dose (4 pellet) and high-dose (20 pellet) groups in rat were approximately 560 and 2800 mg/kg, respectively. For the mouse, the loads are approximately 2800 mg/kg for the low-dose (2 pellet) groups and 5600 mg/kg for the high-dose (4 pellet) groups. Although we expected more rapid tumor development in the mouse, the longer latency period is not unusual in this strain. 30,31 What was surprising was the lack of effect of the WNiCo on hematological parameters and spleen weight in the mouse. In the F344 rat, WNiCo implantation resulted in significant increases in hemoglobin and hematocrit levels, as well as a large increase in spleen/body weight ratio over control groups. 15 These effects, indicative of polycythemia, 32 were not observed in the WNiCo-implanted B6C3F1 mice.
Although the species difference in response to implanted WNiCo was surprising, it is not without precedent. An investigation on the carcinogenicity of potassium bromate administered in drinking water to F344 rats and B6C3F1 mice showed that the rats developed tumors as a result of potassium bromate administration, while the mice did not. 33 The species variations were attributed to differences in bromate metabolism and subsequent lipid peroxidation and oxygen free radical generation. Species and sex differences were also seen in the response of F344 rats and B6C3F1 mice to the mytotoxin fumonisin B1. In this study, the compound was found to be tumorigenic when administered orally to male F344 rats and female B6C3F1 mice but not to female F344 rats and male B6C3F1 mice. 34 A variety of factors can be proposed to explain the varying species responses in carcinogenicity testing including metabolic pathway differences, endogenous antioxidant levels, genetic makeup, and body weight. It has been shown that rodents on a restricted caloric intake diet exhibit a reduced incidence of spontaneous and induced tumors as well as an increased time to tumor development. 35 Although neither the mice in this study nor the rats in the previously published studies on implanted tungsten alloys 15,16 were calorie restricted, the diets differed which could explain the difference in tumor latency. However, a more likely explanation is the difference in genetic makeup. 36 The B6C3F1 mouse is a hybrid, while the F344 rat is inbred. These differences in genetic variability could explain the differences in tumor aggressiveness observed between the 2 species. It would be of interest to determine whether there are differences between inbred, hybrid, and outbred strains of the same species with respect to tumor formation and malignancy for embedded metal fragments.
Tungsten levels were significantly elevated in all tissues analyzed from the WNiCo-implanted mice. Although once thought to be insoluble, pure tungsten implants have been shown to solubilize rapidly 37 and all 3 metals comprising the 2 tungsten alloys tested in this study solubilize over time. As noted earlier, solubilization of implanted WNiFe resulted in the release of low, yet statistically significant, levels of metals that can be measured in all tissues sampled. However, release of metals from the embedded pellets decreased over time and tissue metal concentrations returned to near background levels. As previously shown, this behavior is the result of the formation of an iron-containing layer on the WNiFe pellet that prevents further in vivo degradation. 16 No such layer is formed around the WNiCo pellets and, as such, they continue to degrade and release significantly high levels of metals through the life of the animal. The tungsten released was eventually excreted in the urine, as previously reported, 38 but can also concentrate in the kidney, liver, and spleen. In addition, the released tungsten can cross the blood–brain and blood–testis barriers and concentrate in those organ systems as well. The translocation of metals from the embedded pellets across the blood–brain barrier raises the possibility of neurological perturbations, especially in the case of polytrauma with embedded metal fragments and traumatic brain injury. 39 We have also found significant levels in the femur, which correlated with the proposed biokinetics of tungsten deposition in the body. 40 In addition, the high levels of tungsten in these organs increase concern about potential long-term health effects of the metal, both alone and in combination with other metals. 41,42 Recent reports suggest that tungsten can alter B-cell development and induce DNA damage in bone marrow cells. 43,44 Studies by Miller and colleagues suggest that a synergistic effect between the metals of the alloy is responsible for the deleterious effects of the WNiCo mixture, since damage induced by the mixture is greater than the additive effects of the metals alone. 17,19 Reconstituted tungsten alloy mixtures administered by intratracheal or intraperitoneal routes have also been shown to result in cellular damage to pulmonary cells and changes in the urinary metabolome, respectively. 45,46 Although no human occurrences of embedded fragments of the military-grade tungsten alloys have as yet been reported, 2 cases of granulomas induced by fragments of chainsaw and lawnmower blades have been published. 47,48 In both cases, the embedded fragments consisted primarily of tungsten with smaller amounts of other metals including cobalt and copper.
This study demonstrated that military-grade WNiCo, when embedded into mouse skeletal muscle to simulate a fragment wound, induced the formation of a rhabdomyosarcoma. These results, together with the previously published studies using a rat model, strongly suggest that embedded WNiCo fragments will be carcinogenic in humans. Conversely, implantation of military-grade WNiFe did not result in any tumor formation or apparent adverse health effects. It should be pointed out that the 2 tungsten-based materials tested in this study were military grade and were produced for use in armor-penetrating munitions. They are not the formulations used in small-caliber or hunting ammunition. However, these studies clearly emphasize the critical need to incorporate health effects testing early in the munition development process, as has been previously proposed. 49 The lack of knowledge as to the in vivo degradation, biokinetics, and health effects of the metal compositions that could be found in embedded fragments puts the long-term health of wounded military and civilian populations at risk.
Footnotes
Author Contribution
J. Kalinich contributed to conception and design, acquisition, analysis, and interpretation; drafted the article; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. C. Emond contributed to conception and design, acquisition, analysis, and interpretation; critically revised the article, gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. V. Vergara contributed to design, acquisition, analysis, and interpretation; critically revised the article, gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. E. Lombardini and S. Mog contributed to analysis and interpretation; critically revised the article; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
The views expressed are those of the authors and do not reflect the official policy or position of the Armed Forces Radiobiology Research Institute, the Uniformed Services University, the Department of Defense, or the United States Government.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the US Army Medical Research and Materiel Command Peer-Reviewed Medical Research Program (WX81XWH-06-2-0025).
