Abstract
Dermal exposure to chemicals may result in allergic or irritant contact dermatitis. In this study, we performed ex vivo local lymph node assay: bromodeoxyuridine-enzyme-linked immunosorbent assay (LLNA: BrdU-ELISA) to compare the differences between irritation and sensitization potency of some chemicals in terms of the 3 end points: lymphocyte proliferation, cytokine profiles (interleukin 2 [IL-2], interferon-γ (IFN-γ), IL-4, IL-5, IL-1, and tumor necrosis factor α [TNF-α]), and ear swelling. Different concentrations of the following well-known sensitizers and irritant chemicals were applied to mice: dinitrochlorobenzene, eugenol, isoeugenol, sodium lauryl sulfate (SLS), and croton oil. According to the lymph node results; the auricular lymph node weights and lymph node cell counts increased after application of both sensitizers and irritants in high concentrations. On the other hand, according to lymph node cell proliferation results, there was a 3-fold increase in proliferation of lymph node cells (stimulation index) for sensitizer chemicals and SLS in the applied concentrations; however, there was not a 3-fold increase for croton oil and negative control. The SLS gave a false-positive response. Cytokine analysis demonstrated that 4 cytokines including IL-2, IFN-γ, IL-4, and IL-5 were released in lymph node cell cultures, with a clear dose trend for sensitizers whereas only TNF-α was released in response to irritants. Taken together, our results suggest that the ex vivo LLNA: BrdU-ELISA method can be useful for discriminating irritants and allergens.
Keywords
Introduction
In recent years, in the industrialized countries, the incidence of skin toxicity has a tendency to increase in parallel to the increase in the number of potential chemicals. The 2 most frequent manifestations of skin toxicity are irritant contact dermatitis and allergic contact dermatitis. 1
The murine local lymph node assay (LLNA) is a validated method for the evaluation of skin sensitization potential of chemicals. The assay is based upon measurement of induced proliferative responses in lymph nodes by in vivo incorporation of [ 3 H] methyl thymidine. Several authors have conducted investigations for the development of an alternative nonradioactive method because such radioisotope-based methods require special facilities and handling procedures. 2 -16 Interagency Coordinating Committee on the Validation of Alternative Methods (ICCVAM) approved the validation of LLNA: bromodeoxyuridine-enzyme-linked immunosorbent assay (LLNA: BrdU-ELISA) for identification of skin sensitization potency of chemicals that was based on the protocol developed by Takeyoshi et al. 10 On the other hand, ICCVAM recommends future studies to characterize the limitations of the LLNA: BrdU-ELISA test method. 17 We developed an ex vivo LLNA: BrdU-ELISA and demonstrated that ex vivo LLNA: BrdU-ELISA has sensitivity that is comparable to LLNA; however, there is a compromised accuracy in LLNA-BrdU-ELISA. Recently, the advantages of ex vivo LLNA-BrdU have also been concluded by Williams et al. 16 Ex vivo LLNA BrdU provided many advantages when compared to LLNA and LLNA: BrdU-ELISA methods. First, intraperitoneal injection of bromodeoxyuridine (BrdU) is not necessary, which may increase reproducibility of the method by eliminating the potential for variability resulting from inconsistent injection technique. Second, lymph node cellularity data provide an early indicator of sensitization potential. Third, because lymph node cells are isolated from the animals before BrdU labeling, these cells can also be used for other experimental end points like cytokine analyses. 16 Many studies have tried to characterize and distinguish the chemical sensitizers from irritants or nonsensitizers according to cytokine profiles. 13,18 -29
In this study, we performed ex vivo LLNA: BrdU-ELISA to compare the differences between irritation and sensitization potency of some chemicals in terms of the 3 end points: lymphocyte proliferation; proinflammatory (tumor necrosis factor α [TNF-α], interleukin 1 [IL-1]), T helper type 1 (Th1; IL-2 and interferon-γ [IFN-γ]), and Th2 (IL-4, IL-5) cytokine profiles; and ear swelling results. 28,29 Different concentrations of the following well-known sensitizers and irritant chemicals were applied to mice: dinitrochlorobenzene (DNCB), eugenol, isoeugenol, sodium lauryl sulfate (SLS), and croton oil.
Materials and Methods
Mice
Female Balb/c mice (8-12 weeks of age) were obtained from Gulhane Military Medical Academy (Ankara, Turkey) and housed at a temperature of 23°C and relative humidity of 55%, with a 12-hour light/dark cycle. Mice were provided mouse chow (Optima, Kırklareli, Turkey) and water ad libitum. All animal procedures were conducted in an Association for Assessment and Accrediation of Laboratory Animal Care-accredited facility under an animal protocol approved by the Ankara University Animal Experiments Ethics Board. Before the experiment, mice were weighed, randomized, and marked for identification. Ear thickness was measured with digital micrometer.
Chemicals
Sodium lauryl sulfate (SLS; 0.05%, 0.1%, 0.25%, and 0.5%; Prolab, Belgium), croton oil (0.05%, 0.1%, 0.25%, and 0.5%; Sigma-Aldrich, MO, USA), DNCB (0.025%, 0.05%, 0.1%, and 0.25%; Sigma-Aldrich, MO, USA), isoeugenol (0.5%, 1%, 5%, and 10%; Acros Organics, NJ, USA), eugenol (2.5%, 10%, 20%, and 50%; Acros Organics, NJ, USA), and para aminobenzoic acid (PABA; 1%, 2.5%, 5%, and 10%; Sigma-Aldrich, MO, USA) in acetone:olive oil (AOO; 4:1 v/v; Sigma-Aldrich, MO, USA) were applied to the animals in these studies. Para aminobenzoic acid was used as the negative control.
Combined Ex Vivo LLNA-BrdU and Irritancy Assay
A combination of ex vivo LLNA-BrdU and irritancy assay was conducted following a modification of previously published procedures. 13,28,29 Five groups of mice (n = 4/group) were exposed topically on the dorsum of both ears to 25 μL of different concentration of chemicals and vehicle (AOO) alone daily for 3 consecutive days. Before the first chemical application, the thickness of the right and left pinnae of each mouse ear was measured using a digital micrometer (Mitutoyo Co, Japan) for the irritancy assay. Ear thickness measurements were taken again 24 hours following the final exposure. The results of the irritancy assay were expressed as percentage change in ear swelling using the formula. 28,29
% change in ear swelling = ([mean postchallenge ear thickness − mean prechallenge ear thickness]/mean pre challenge ear thickness) × 100. To reduce the number of animals required, these evaluations were conducted in conjunction with the LLNA.
The mice in this study were rested on day 4. On day 5, all mice were killed by cervical dislocation, and their auricular lymph nodes were excised and weighed. Excised right and left lymph nodes were pooled and homogenized, and cells were suspended in 15 mL physiological saline temperature. After counting, cells from the suspension were seeded into 96-well culture plates (at 105 cells/well, in RPMI 1640 medium supplemented with 10% fetal bovine serum [FBS] and 1% penicillin–streptomycin; all from Biochrom, Israel). After culturing for 48 hours at 37°C, BrdU (10 μL of a 10-μmol/L BrdU labeling solution; final concentration = 1 µmol/L BrdU) was then added to the wells for a 24-hour labeling period. The cells in the wells were then recovered by aspiration, and the extent of BrdU incorporation was measured by ELISA (Roche, Germany), according to the manufacturer’s instructions. The absorbance was measured at a wavelength of 450 nm (optical density at 450 nm [OD450]) with a reference wavelength of 620 nm, using an ELISA reader (SpectraMAX; Molecular Devices Inc); these values were used to define the BrdU labeling index. Stimulation index (SI) was calculated as the ratio of the mean ex vivo BrdU incorporation (labeling index) for each treatment group to that of the vehicle control group.
Culture of Lymph Node Cells and the Measurement of Cytokine Levels
Harvested lymph node cells from the ex vivo protocols outlined previously were seeded in a 24-well culture plate (at 1 × 106 cells/well) in 1 mL of RPMI 1640 medium supplemented with 10% FBS and 1% penicillin–streptomycin. Following the seeding steps, the wells were supplemented with 5 μg/mL of phytohemagglutinin-L (PHA-L; Biochrom) immediately. The PHA-L was selected as the mitogen here (as opposed to concanavalin A) in that it has been widely used for mitotic stimulation of T-lymphocytes. After 72 hours of culture in a 37°C incubator containing 5% CO2, supernatants were collected and stored at −80°C until analyzed for levels of IL-2, IFN-γ, IL-4, IL-5, and TNF-α. The levels of each cytokine in the culture supernatants were measured using commercially available ELISA kits (BenderMed Systems), according to the manufacturer’s instructions. The SI was calculated as the ratio of the mean cytokine levels for each treatment group to that of the vehicle control group.
Statistical Analyses
The differences in cytokine levels between the vehicle group and each treatment group were statistically analyzed with Mann-Whitney U test, and P < 0.05 was taken as the level of significance.
Results
Lymph Node-Related Parameters
The auricular lymph node weights, lymph node cell counts, and percentage change in ear swelling associated with experimental groups are shown in Table 1 (mean ± standard deviation). Treatment of Balb/c mice with sensitizer (DNCB, eugenol, and isoeugenol) and irritant (croton oil and SLS) chemicals resulted in increased lymph node weight and lymph node cell count. The responses exhibited a concentration–response. Treatment of Balb/c mice with negative control (PABA) did not show changes in these parameters.
Lymph Node Weight, Cell Count, and Percentage Change in Ear Swelling Values After Treatment of Mice With Different Concentrations of Chemicals in Acetone:Olive Oil (AOO; 4:1 v/v).
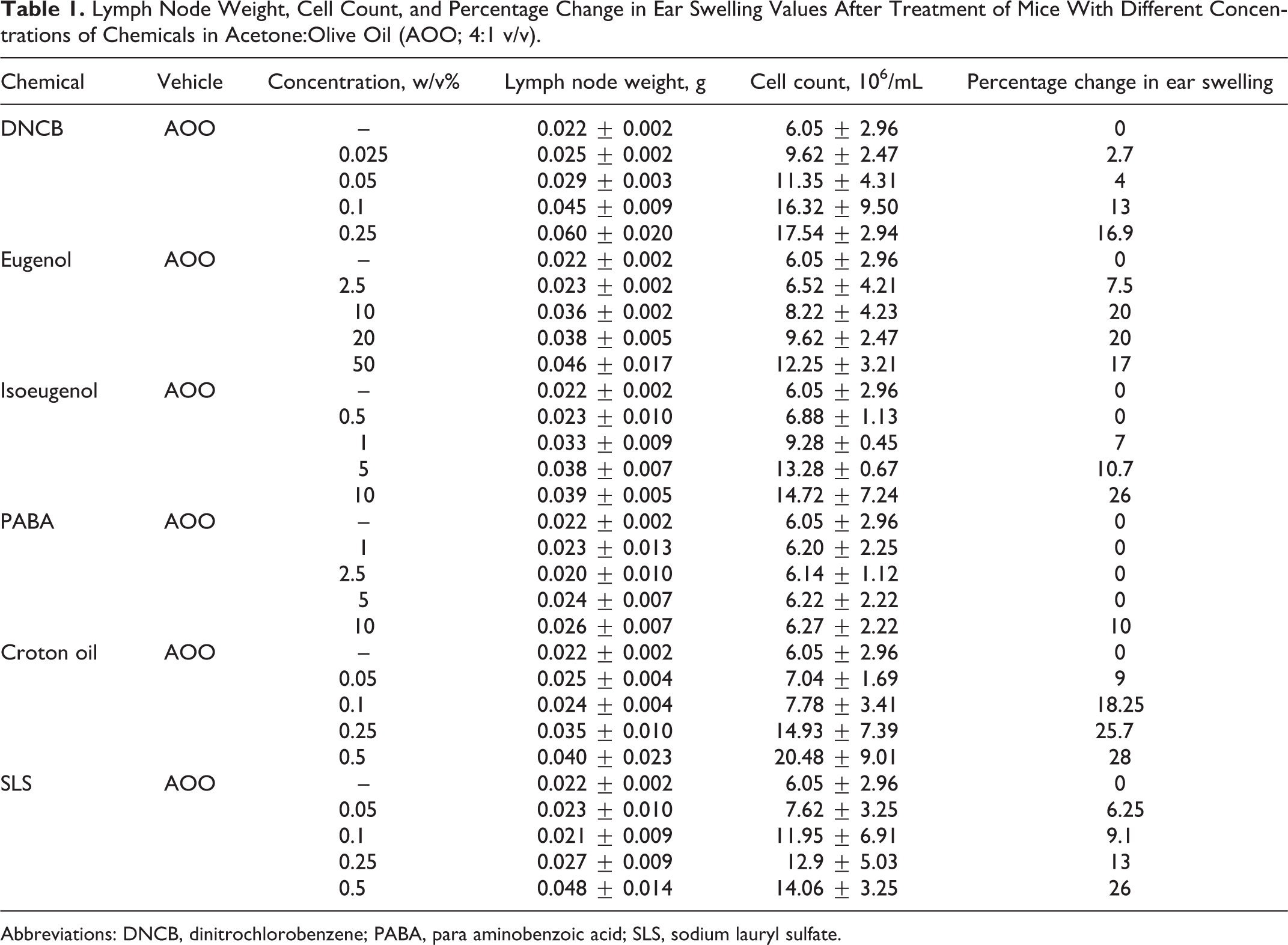
Abbreviations: DNCB, dinitrochlorobenzene; PABA, para aminobenzoic acid; SLS, sodium lauryl sulfate.
The OD450 and SI data from ex vivo BrdU labeling studies are presented in Table 2. Approximately 3-fold induction in OD values was found at concentrations of 0.05% DNCB, 20% eugenol, 1% isoeugenol, and 0.5% SLS. For croton oil and negative control (PABA), induction was less than 3-fold at the applied concentrations.
Optic Density (OD) and Stimulation Index (SI) Values After Treatment of Mice With Different Concentrations of Chemicals in Acetone:Olive Oil (AOO; 4:1 v/v).
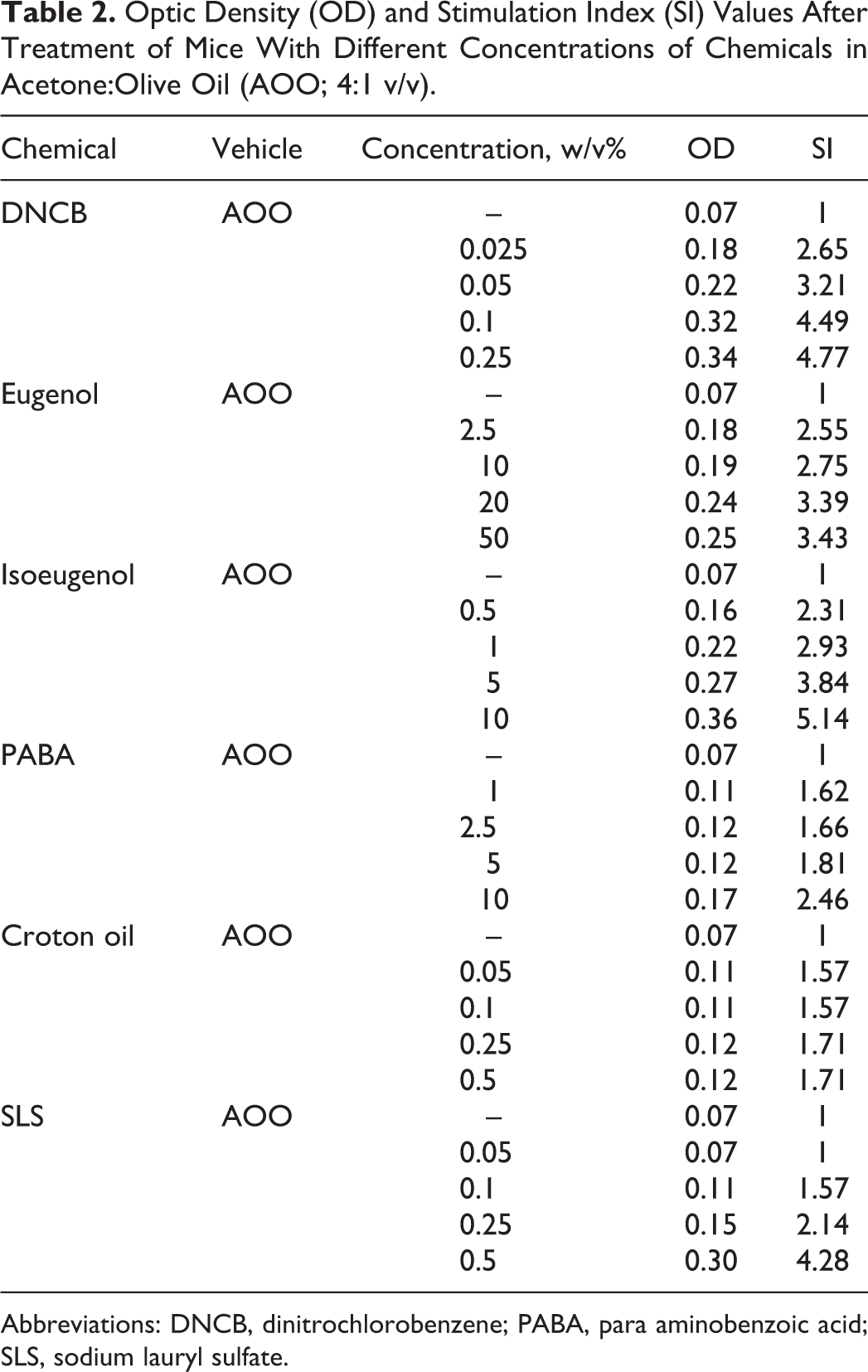
Abbreviations: DNCB, dinitrochlorobenzene; PABA, para aminobenzoic acid; SLS, sodium lauryl sulfate.
Irritancy Assay
The results of the irritancy studies for sensitizer (DNCB, eugenol, and isoeugenol) and irritant (croton oil and SLS) chemicals are presented in Table 1. Excessive local irritation is indicated by an ear thickness of ≥25%. The percentage change in ear swelling was found to be higher than 25% at 10% for isoeugenol, 0.25% croton oil, and 0.5% SLS.
Cytokine Determinations
The levels of IL-2, IFN-γ, IL-4, IL-5, TNF-α, and IL-1 present in the lymph node cell culture supernatants and the SI values are provided in Table 3. In the DNCB-, eugenol-, and isoeugenol-exposed groups, the formation/release of IL-2, IFN-γ, IL-4, and IL-5 was strongly elevated in a clear dose-related trend. This strong elevation was reflected in the calculated SI values (Table 3). In the lymph node cell cultures treated with lower concentrations of sensitizers, TNF-α release was increased significantly (P < 0.05). A clear dose-related trend was observed in TNF-α levels in the croton oil (irritant chemical)-exposed group. The levels of TNF-α from the SLS-exposed group were also found to significantly increase at 0.05%, 0.1%, and 0.25% concentrations, compared to the control vehicle group. Both Th1 (IL-2 and IFN-γ) and Th2 (IL-4 and IL-5) cytokine releases were observed in irritant-exposed experimental groups, but these releases did not reflect a dose-related trend. The IL-1 levels were found to be very low in all the experiment groups.
Cytokine Stimulation Index (SI) Values for Lymph Node Cells after Treatment of Mice With Different Concentrations of Chemicals in Acetone:Olive Oil (AOO; 4:1 v/v).a
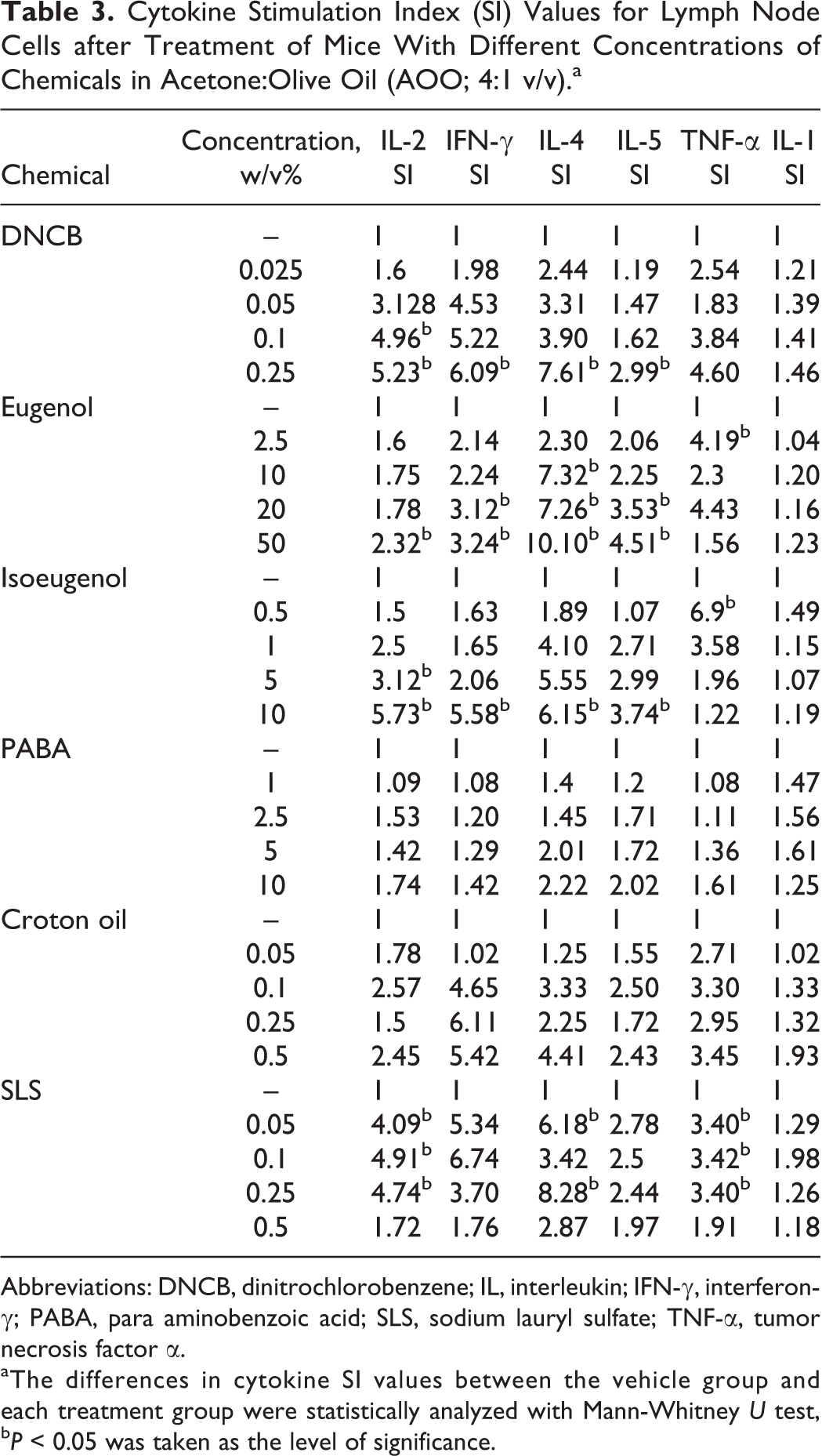
Abbreviations: DNCB, dinitrochlorobenzene; IL, interleukin; IFN-γ, interferon-γ; PABA, para aminobenzoic acid; SLS, sodium lauryl sulfate; TNF-α, tumor necrosis factor α.
aThe differences in cytokine SI values between the vehicle group and each treatment group were statistically analyzed with Mann-Whitney U test, b P < 0.05 was taken as the level of significance.
Discussion
In our study, we used ex vivo LLNA: BrdU-ELISA to distinguish the sensitizing and irritation potential of some chemicals in 3 end points: lymphocyte proliferation, ear swelling, and cytokine profiles. According to the lymph node results, the auricular lymph node weights and lymph node cell counts were increased after application of both sensitizers and irritants in high concentrations. These results indicate that not only sensitizers but also irritants stimulate the lymph nodes after local treatments. Several parameters of draining lymph node activation were measured following topical exposure of mice to contact allergens and to nonsensitizing chemicals. These composed changes in lymph node weight and cellularity and lymphocyte proliferation measured as a function of radiolabeled thymidine incorporation during culture of lymph node cells. 30 -32 The marker that proved to be the most sensitive and selective correlate of sensitizing activity was the induction of lymph node cell proliferation. Our results were consistent with these comments. According to lymph node cell proliferation results, we found that there was 3-fold increase in proliferation of lymph node cells for sensitizer chemicals (DNCB, eugenol, and isoeugenol) and SLS, whereas there was a less than 3-fold increase for croton oil and negative control (PABA). Although a few case reports suggesting contact sensitizing effect of SLS have been published, SLS has been considered an irritant by most researchers and dermatologists. For croton oil, no literature is available on the sensitization potency in humans, and it is considered as an irritant. Both irritants and sensitizers can induce lymphocyte proliferation. Although sensitizers generate antigen-specific lymphocyte proliferation, this response is nonspecific for irritants. In the LLNA, the use of [ 3 H] methyl thymidine incorporation for measurements of lymphocyte proliferation does not allow for differentiation of the 2. For this reason, it has long been debated that the LLNA may give false positives when irritating chemicals are tested. 33,34 For example, topical application of the well-studied surfactant SLS has been shown to test positive in the LLNA with SI values above the threshold limit (3-fold increase). 33 -38 In our study, SLS gave a false-positive result at concentration of 0.5%.
According to the ear swelling results, we found that croton oil and SLS had irritant potency at concentrations of 0.25% and 0.5%, respectively. On the other hand, isoeugenol was found to be a sensitizer and also only at high concentrations (10%) to be an irritant. These results are compatible with the previous reports. 39 -41 The European Food Safety Authority The Panel on Additives and Products or Substances used in Animal Feed considered isoeugenol as an irritant to the respiratory system, skin, and eyes and as a skin and respiratory sensitizer, based on the information available in the literature. 41
Increases in cytokine levels are related to T helper cell proliferation, which plays an important role in the induction and elicitation of contact sensitivity. The regulation of contact sensitivity responses by Th1 and Th2 cytokines has been studied intensively. The findings demonstrate that not only Th1 cytokines such as IL-2 and IFN-γ but also Th2 cytokines such as IL-4 and IL-5 can play essential roles in elicitation of contact sensitivity. 13,41 -43 In the early phases, it is likely that IL-1 and TNF-α driven innate immune responses are involved in both irritant and allergic contact dermatitis. In later phases of irritant contact dermatitis, skin inflammation is still critically dependent on innate responses. However, in allergic contact dermatitis, adaptive immune responses involving antigen-specific T cells take over to amplify skin inflammation. 44 Although the precise cytokine activation cascade in irritant contact dermatitis is still unclear, it is likely that the primary cytokines involved following irritant exposure are IL-1 and TNF-α.
In this study, we measured levels of proinflammatory Th1 and Th2 cytokine releases from lymph node cells. At the lower concentrations in the sensitizer-exposed groups, TNF-α release was found to be increased significantly. This finding is well in line with the suggestion that TNF-α is involved in early phases of allergic contact dermatitis. On the other hand, IL-2, IFN-γ, IL-4, and IL-5 releases in sensitizer-exposed groups were found to be increased in a manner reflecting a clear dose-related trend. The cytokine levels in the higher concentration-exposed groups were found to be significantly higher, compared to the levels of lower concentration-exposed groups. These findings are consistent with the previous reports that cytokines including IL-2, IFN-γ, IL-4, and IL-5 are produced during the elicitation phase in contact allergy. 13 -15,42,43,45,46
Exposure of Balb/c mice to PABA did not stimulate levels of cytokines (IL-2, IFN-γ, IL-4, IL-5, IL-1, and TNF-α). This finding was as expected since PABA is considered as a negative control in LLNA studies. 47
Numerous in vitro studies have shown that various irritants induce the expression of IL-1 and TNF-α in keratinocytes. 48 -53 Kerotinocytes play a key role in production of immune mediators in irritant contact dermatitis. On the other hand, the role of skin infiltrating T lymphocytes in irritant contact dermatitis is also not well defined. In acute reactions of irritant contact dermatitis, cellular infiltrates consisting of mainly T helper lymphocytes were seen with an increased level of IL-2 and IFN-γ. 54 In our study, a clear dose-related trend was observed in TNF-α levels of croton oil-exposed group. The levels of TNF-α of SLS-exposed group were also found to significantly increase at 0.05%, 0.1%, and 0.25% concentration compared to the control vehicle group. These results showed that irritant chemicals induced TNF-α release in lymph node cells. Both Th1 and Th2 cytokines releases were observed in irritant-exposed experimental groups, but these releases did not reflect a dose-related trend. On the other hand, IL-1 levels were found to be very low in all the experiment groups. Lymph node cells were not as critical as keratinocytes for observing IL-1 releasing.
According to our results, proinflammatory Th1 and Th2 cytokines were detected in lymph node cell cultures of both irritant and sensitizer-exposed groups. Cytokine analysis demonstrated that 4 cytokines (IL-2, IFN-γ IL-4, and IL-5) release in lymph node cell cultures showed a clear dose-related trend for sensitizers, whereas only TNF-α release for irritants.
It has been reported that some irritants induce proliferation in the lymph nodes in both radioactive and nonradioactive LLNA. Thus it is important to perform irritancy test to determine the potential of irritancy, if a positive response is obtained. Measuring some cytokine levels can be useful for distinguishing the irritants and sensitizers as an additional end point of the method. These modifications of the nonradioactive LLNA are intended for use as research tools and are not validated for the purpose of hazard identification. The validation studies of ex vivo LLNA: BrdU ELISA method would be needed to be considered as a more data-rich alternative to LLNA without using extra animals.
Footnotes
Author Contribution
S. Arancioglu contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. O. C. Ulker contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, gave final approval, critically revised the article, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. A. Karakaya contributed to conception and design, drafted the article, gave final approval, critically revised the article, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
