Abstract
Phototoxicity has a strong impact on drug development. Although several animal models have been developed to quantitatively assess human risks, none have been validated for standardized use. In this study, we validated an in vivo phototoxicity model using Long-Evans (LE) rats treated with 4 well-known phototoxic drugs, namely 8-methoxypsoralen, lomefloxacin, sparfloxacin, and pirfenidone. Daily macroscopic observations of skin and eyes, ophthalmological examinations 4 days after dosing, and blood sampling for toxicokinetics (TKs) were performed after exposure of treated animals to ultraviolet, and dose-dependent eye and/or skin reactions were noted for all compounds. Margins of safety were calculated when possible and correlated well with known relative phototoxicity of the 4 compounds. We conclude that the present in vivo phototoxicity assay using LE rats with TK analysis can be used to quantitatively predict the risk of pharmaceutical phototoxicity in humans.
Introduction
All new active pharmaceutical ingredients (APIs) and cosmetics that absorb ultraviolet (UV) B, UVA, or visible light in the range of 290 to 700 nm and can reach the skin or eyes need to be tested for potential phototoxicity, as required by regulatory agencies (FDA Guidance for Industry, 1 EMEA Note for Guidance on Photosafety Testing, 2 and International Conference on Harmonisation (ICH) Harmonised Tripartite Guideline Photosafety Evaluation of Pharmaceuticals, S10 3 ).
Various in vitro and in vivo assays are used to assess safety and phototoxicity, and in vitro phototoxicity guidelines have been assembled for cosmetics and external drugs in Europe and are included in the Food and Drug Administration (FDA) guidance. 1 The 3T3 Neutral Red Uptake Phototoxicity Test (3T3 NRU PT) is the most widely used in vitro assay for phototoxicity and is included in the Organization for Economic Cooperation and Development guideline. 4
For phototoxicity assays, experimental animals such as guinea pigs, mice, albino rats, and pigmented rats (Long-Evans [LE] rats) are used widely. However, no formally validated nonclinical in vivo phototoxicity assays have been validated or included in regulatory agency guidelines. Therefore, industries select nonclinical in vivo phototoxicity assays at their discretion. 4
Active pharmaceutical ingredients that show phototoxic potential in the 3T3 NRU PT require additional evaluations using nonclinical in vivo phototoxicity assays. When positive results are anticipated from in vivo phototoxicity studies, additional dose groups can support a no-observed-adverse-effect level (NOAEL)-based risk assessment, typically considering Cmax comparisons in the ICH S10. When systemic exposure at NOAEL in animals is similar, pharmaceutical companies need to consider aborting further development of the API or conducting phototoxicity trials in humans. Hence, the results of nonclinical in vivo phototoxicity assays are crucial for pharmaceutical companies. However, few published reports demonstrate the use of nonclinical in vivo phototoxicity assays to dose-dependently evaluate systemic exposure at the NOAEL in animals or to estimate margins of safety (MoS) in comparison with human systemic exposure levels.
In this study, we assessed human risk using a quantitative NOAEL-based approach with nonclinical in vivo phototoxicity assays. Four well-known phototoxic compounds with various phototoxic potentials were evaluated in pigmented rats (LE rats). Phototoxic potential and dose–responses of LE rats were evaluated using the strongly phototoxic compound 8-methosypsoralen (8-MOP) and the moderately phototoxic compounds lomefloxacin (LMFX), sparfloxacin (SPFX), and pirfenidone (PFD). The phototoxicity of 8-MOP is well known and is labeled with a “contraindication” for light exposure. Drug package inserts for LMFX, SPFX, and PFD are similarly labeled with a “warning” for phototoxicity. All 4 compounds elicited positive reactions in the 3T3 NRU PT (Table 1). The LE rats were orally administrated 1 of the 4 compounds and were exposed to UV at 10 J/cm2, around the Tmax of each compound. To evaluate systemic exposure, toxicokinetic (TK) experiments were performed for each compound in additional satellite groups. Cmax values for each compound at NOAEL in rats were then compared with Cmax values of clinical doses in humans to calculate MoAs and to validate the in vivo phototoxicity assays in LE rats.
Compound Information From the 3T3 NRU PT.
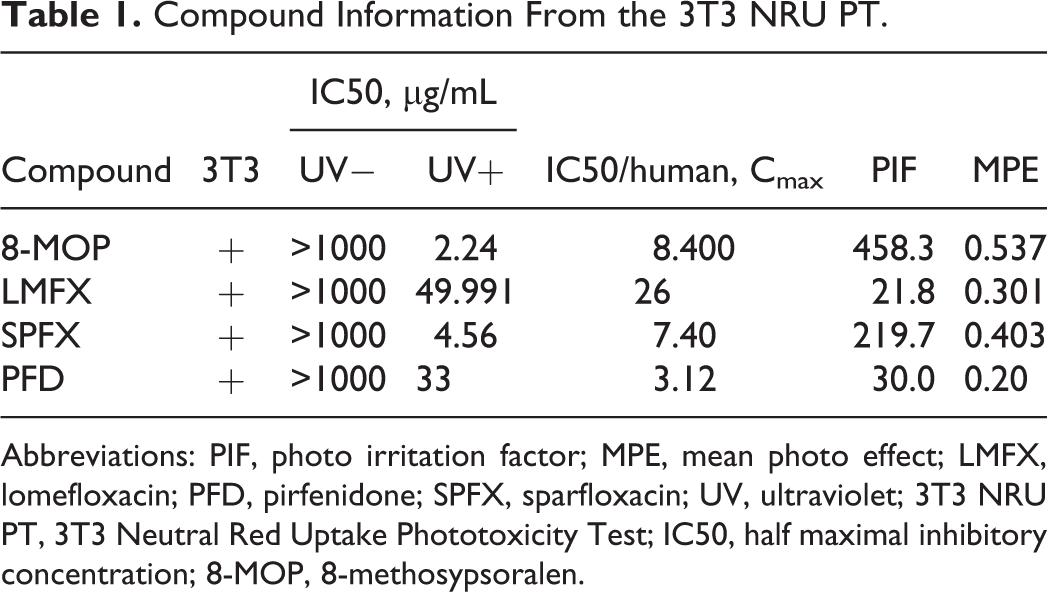
Abbreviations: PIF, photo irritation factor; MPE, mean photo effect; LMFX, lomefloxacin; PFD, pirfenidone; SPFX, sparfloxacin; UV, ultraviolet; 3T3 NRU PT, 3T3 Neutral Red Uptake Phototoxicity Test; IC50, half maximal inhibitory concentration; 8-MOP, 8-methosypsoralen.
Assessments of phototoxicity apply to both skin and eyes, and ambient radiation from the sun can contain varying amounts of UVB (280-320 nm), UVA (320-400 nm), and visible light (400-760 nm), and substantial amount of UVB is transmitted through the atmosphere to reach the surface of the earth. The human cornea transmits radiant energy at 295 nm and above. The lens absorbs almost all incident energy to wavelengths of nearly 400 nm. The intraocular filters allow only the order of 1% or less to actually reach the retina. 5 The ICH S10 guideline states that phototoxic drugs that absorb light above 400 nm must be assessed for phototoxicity to the retina using detailed histopathological methods. The large doses of UVA reportedly cause cataracts in animals, and this effect is enhanced by treatments with 8-MOP. 6 Thus, ophthalmological and histopathological examinations were performed in rats treated with 8-MOP, LMFX, SPFX, or PFD. Due to the well-known sensitivity of albino rat eyes, 7 ophthalmological and histopathological evaluations of the effects of treatments on eyes were performed in LE rats with pigmented eyes.
The present data show no MoS for 8-MOP, with a lower Cmax value compared to that in humans at therapeutic doses. Although the other 3 compounds produced severe phototoxicity, the corresponding Cmax values were higher than those in humans at therapeutic doses (MoS, 5.0, 3.7, and 6.7 times, respectively). The present data correlate with known phototoxic potential of the test compounds, indicating that the present in vivo phototoxicity assay quantitatively predicts phototoxic risks of pharmaceuticals.
Materials and Methods
Chemicals
8-Methoxypsoralen, LMFX, SPFX, and PFD were obtained from Sigma-Aldrich Corporations (St Louis, Michigan). 8-Methoxypsoralen was administered orally using corn oil as the vehicle, whereas LMFX, SPFX, and PFD were administered orally using 0.5% methylcellulose aqueous solution as the vehicle.
Female LE rats were purchased from Japan SLC, Inc (Shizuoka, Japan) at 9 weeks of age. Breeding was performed at the Institute for Animal Reproduction (Ibaragi, Japan).
During the acclimation period, rats were selected for study on the basis of ophthalmological examinations, physical appearance, pigmentation patterns, and body weights. The rats were assigned to study groups using computer-generated (weight-ordered) randomization procedures.
Administration
Rats (3 animals/group) were administered formulations once by oral gavage. The day of administration was designated day 1 for this phase. Doses were administered on the basis of the most recently recorded body weights.
Dose Levels and Rationale for Dose Selection
8-Methosypsoralen: 1.5, 5, 15, and 50 mg/kg
The highest dose produced positive reactions in phototoxicity studies using LE rats (preliminary data, not shown), and the other 3 doses (15, 5, and 1.5 mg/kg) were set to determine the NOAEL.
Lomefloxacin: 100 and 200 mg/kg
The highest dose reportedly produced positive reactions in single-administration oral phototoxicity studies using Balb/c mice, 8 and the half dose was set to determine the NOAEL.
Sparfloxacin: 50, 100, and 200 mg/kg
The highest dose reportedly produced positive reactions in single-administration oral phototoxicity studies using Balb/c mice. 8 The other 2 doses were set to determine the NOAEL.
Pirfenidone: 320 and 750 mg/kg
The highest dose was designed to be equivalent to the reported Cmax value (50 µg/mL), which produced a positive reaction in a phototoxicity study using guinea pigs. 9 The other dose (320 mg/kg) was set to determine the NOAEL.
Anesthesia Before UV Exposure
On day 1, animals were lightly anesthetized using pentobarbital (intraperitoneal, 24 mg/kg) before UV exposure.
UV Exposure
Before UV exposure, hair of the dorsal region was removed using electrical clippers. After anesthesia, light and dark pigmented regions of the dorsal skin were marked with oil-based ink. Each rat was then placed in a prone position on a plastic tray. A UV-block sheet with 2 holes of 1.3 cm in diameter and a UV-block cloth were used as skin sites. The eyes were lubricated with eye drops immediately before and after light exposure.
Rats were exposed to UVA (from 300 to 430 nm, 10 J/cm2) and UVB (under 310 nm, 0.06 J/cm2) for 30 minutes around the Tmax of each compound. The UV exposure was performed 1 hour after 8-MOP and SPFX treatments, 0.5 hour after LMFX treatment, and 10 minutes after PFD treatment.
Ultraviolet Source
DERMALEY-200, Type-AB (ultraviolet radiation equipment; Toshiba Medical Supply Co, Ltd, Tochigi, Japan).
Skin Reaction Observations
Skin reaction observations were performed 6 times (before dosing, immediately after dosing, 0.5 and 24 hours after UV exposure before dosing on day 1, and on days 2, 3, and 4 after UV exposure). Hair of the dorsal region was removed using electrical clippers. The following are grades of skin appearance: Erythema: grade 1, barely perceptible light redness; grade 2, distinct redness. Edema: grade 1, mild, raised <1 mm; grade 2, moderate, raised 1 to 2 mm.
Ophthalmological Examinations
Ophthalmological examinations were performed on days 1 and 4. The anterior portion and optic media were examined using a slit lamp. Appearances of cornea edema were graded as follows: Corneal edema area; 1+, <50%; 2+, >50%; 3+, 100%.
In the case where indirect evaluations could not be performed, the posterior compartment was evaluated by histopathological examination.
Necropsy
All animals were injected intraperitoneally with sodium thiopental and were killed by exsanguination by cutting the caudal vena cava following final observations on day 4. One eye was placed in 10% neutral buffered formalin after fixation in Davidson solution and was examined using a microscope.
Microscopic Examination
Microscopic examinations were performed in the cornea, chamber, iris, ciliary body, lens, vitreous humor, lumen, conjunctiva, retina, choroid, sclera, optic nerve, and in the eyeball.
Blood Sampling for TK Analysis
Blood for TK analysis was collected at Tmax from additional satellite groups for each compound.
Measurement of Drug Concentration in the Skin
Skin samples were frozen rapidly in liquid nitrogen and stored in a freezer. Frozen skin samples (light pigmented regions of 45-90 mg) were ground using a Multi-Beads Shocker (Yasui Kikai, Osaka, Japan). Samples were then suspended in volumes of purified water that were 9 times the weight of the skin sample. Concentrations of 8-MOP, LMFX, SPFX, and PFD in the lenses and skins were measured using liquid chromatography tandem mass spectrometry.
Animal Welfare
All procedures in this study were performed according to the “Rules for Feeding and Storage of Experimental Animals and Animal Experiments” and approved by the Institutional Animal Care and Use Committee of the test facility from the point of view of animal welfare.
Results
8-Methosypsoralen
Table 3 shows 8-MOP-related changes in rats following UV exposure. Skin reactions, such as erythema, mild edema, and fissure, were observed in darkly and lightly pigmented skin on rats that received ≥5 mg/kg 8-MOP. Corneal opacity was observed in rats that received ≥15 mg/kg 8-MOP, and erythema and swelling of lid margins were observed in rats that received 50 mg/kg 8-MOP. In ophthalmological examinations, diffuse corneal edema was noted in rats that received ≥15 mg/kg doses. In microscopic examinations of rats treated with ≥15 mg/kg 8-MOP, vacuolation of corneal epithelium, intercellular edema, corneal epithelium, infiltration of neutrophil in stroma, edema in stroma, and loss of corneal endothelium were observed. However, no phototoxicity was observed in the skin or eyes of rats treated with 1.5 mg/kg 8-MOP. Thus, the NOAEL was 1.5 mg/kg 8-MOP.
Pharmaceutical Compound Information.
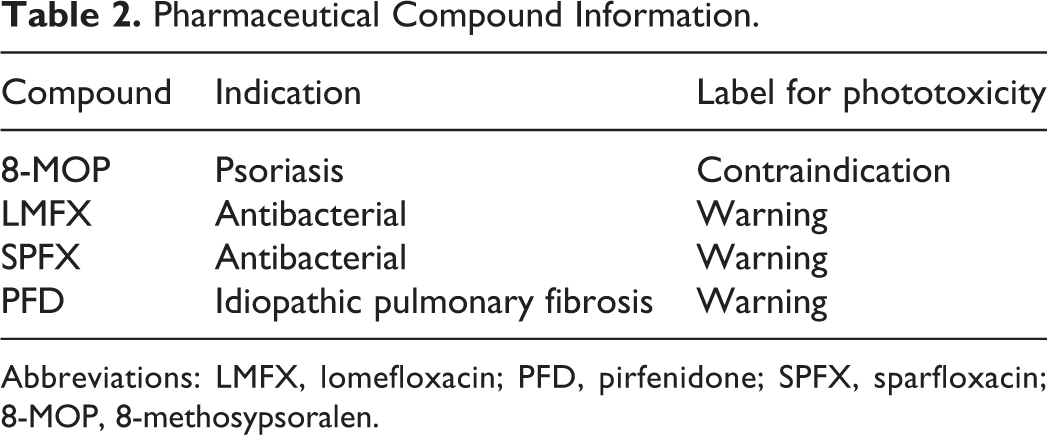
Abbreviations: LMFX, lomefloxacin; PFD, pirfenidone; SPFX, sparfloxacin; 8-MOP, 8-methosypsoralen.
8-Methosypsoralen-Related Changes in Rats Exposed to UV.
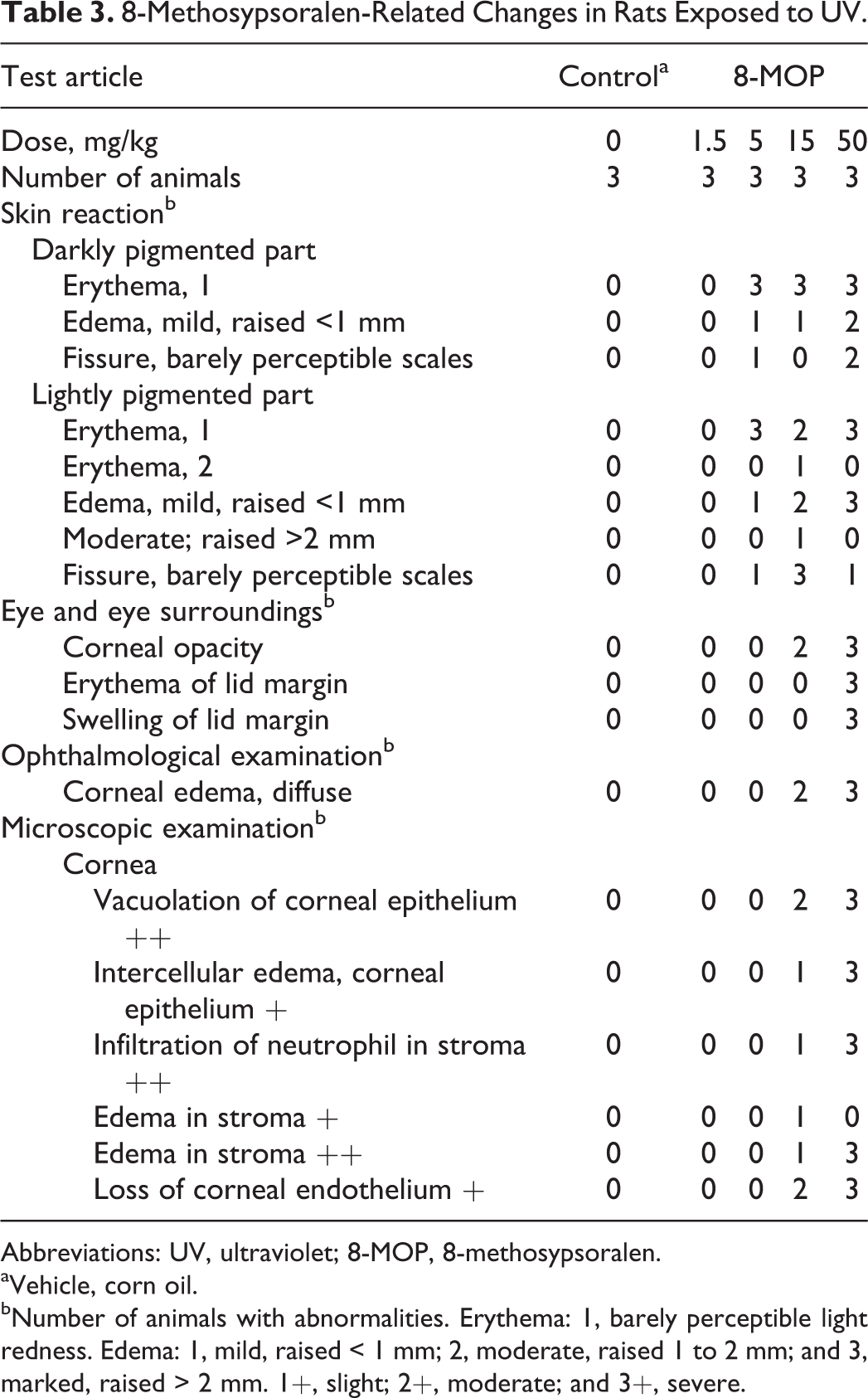
Abbreviations: UV, ultraviolet; 8-MOP, 8-methosypsoralen.
aVehicle, corn oil.
bNumber of animals with abnormalities. Erythema: 1, barely perceptible light redness. Edema: 1, mild, raised < 1 mm; 2, moderate, raised 1 to 2 mm; and 3, marked, raised > 2 mm. 1+, slight; 2+, moderate; and 3+, severe.
Lomefloxacin
Table 4 changes in rats following treatment with LMFX and exposure to UV. Skin reactions, such as erythema, were noted in darkly and lightly pigmented skin on rats treated with 200 mg/kg LMFX. No phototoxic changes in the skin were observed in rats treated with 100 mg/kg or less LMFX. No changes in the eyes were observed at any dose. Thus, the NOAEL was 100 mg/kg LMFX.
Sparfloxacin
Table 5 shows SPFX treatment-related changes in rats exposed to UV. Skin reactions, such as erythema, were noted in darkly and lightly pigmented skin on rats treated with ≥100 mg/kg and with 200 mg/kg, respectively. No phototoxicity was observed in the skin of rats treated with 50 mg/kg, and no changes were observed in the eyes of SPFX-treated rats. Thus, the NOAEL was 50 mg/kg.
Pirfenidone
Table 6 shows PFD treatment-related changes in rats exposed to UV. Skin reactions, such as erythema, were noted in darkly and lightly pigmented skin on rats treated with 750 mg/kg. No phototoxic changes were observed in rats treated with 320 and 160 mg/kg. No changes were noted in the eyes of rats from any PFD treatment groups. Thus, the NOAEL was 320 mg/kg.
The LMFX-Related Changes in Rats Exposed to UV.
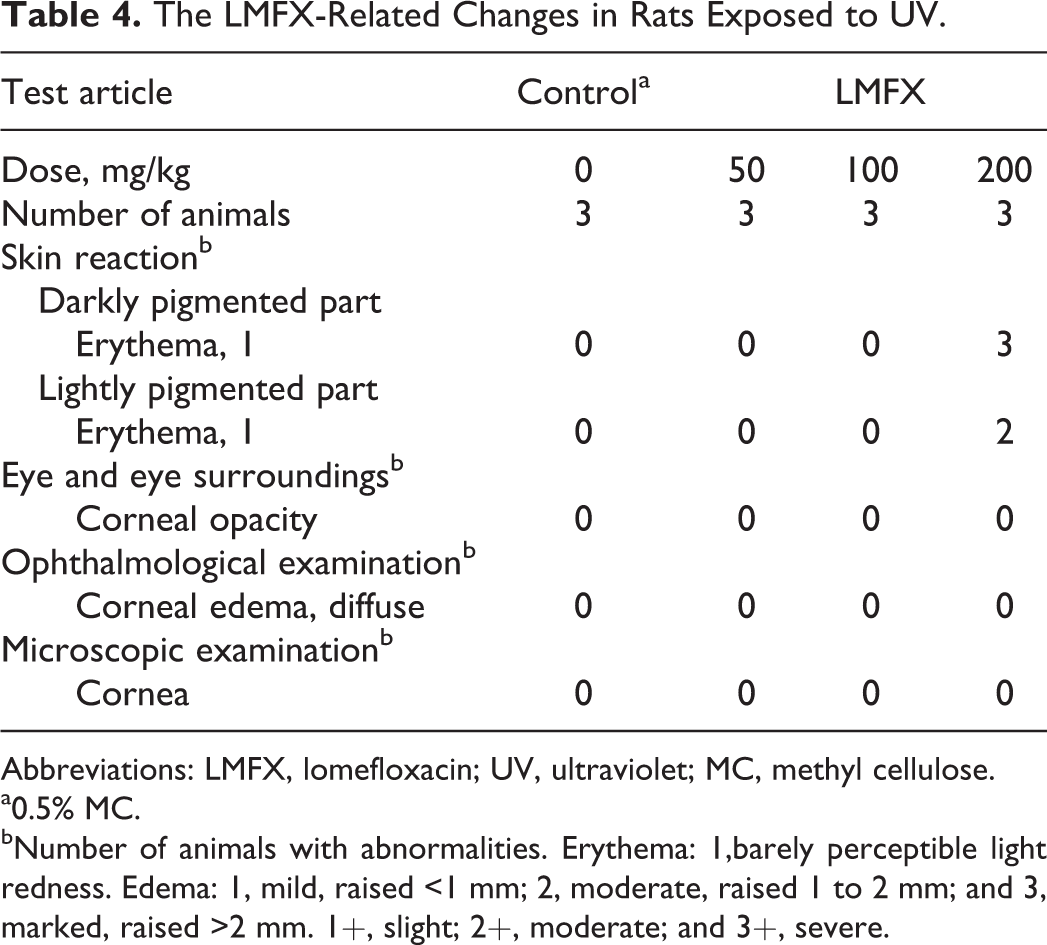
Abbreviations: LMFX, lomefloxacin; UV, ultraviolet; MC, methyl cellulose.
a0.5% MC.
bNumber of animals with abnormalities. Erythema: 1,barely perceptible light redness. Edema: 1, mild, raised <1 mm; 2, moderate, raised 1 to 2 mm; and 3, marked, raised >2 mm. 1+, slight; 2+, moderate; and 3+, severe.
The SPFX-Related Changes in Rats Exposed to UV.
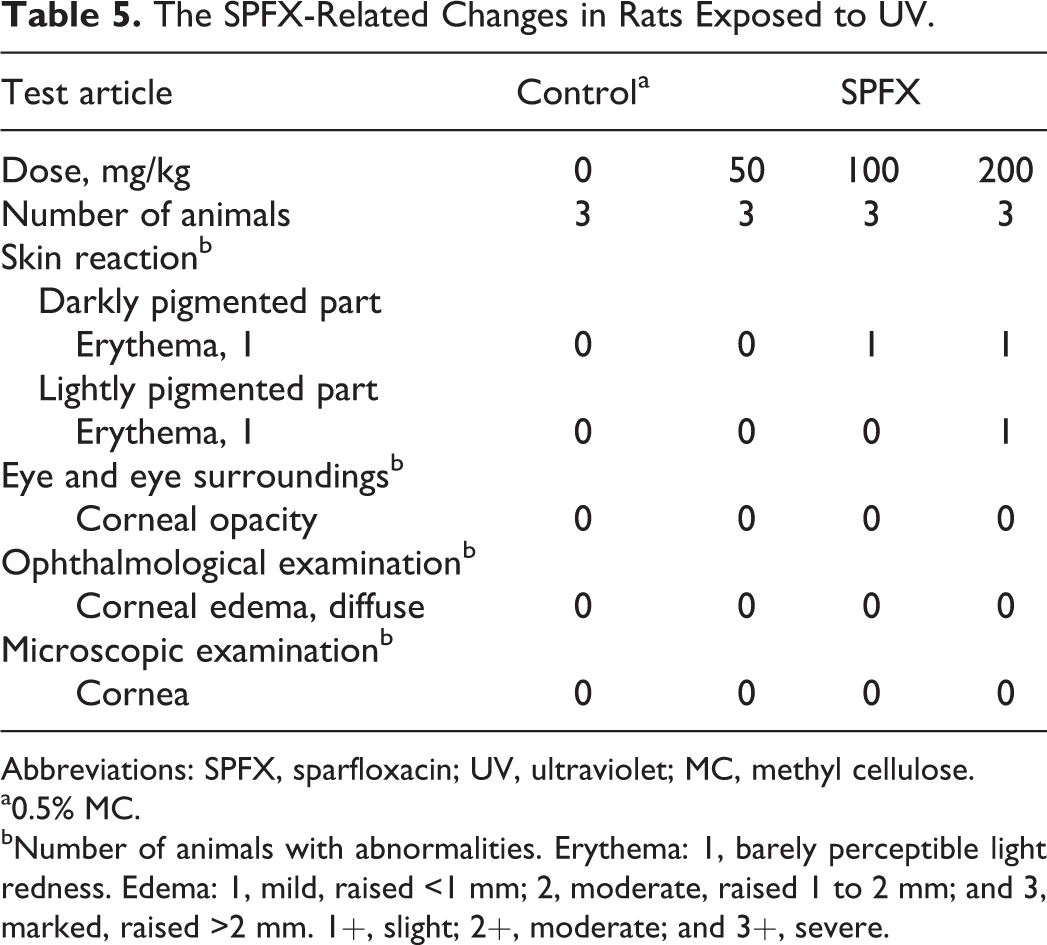
Abbreviations: SPFX, sparfloxacin; UV, ultraviolet; MC, methyl cellulose.
a0.5% MC.
bNumber of animals with abnormalities. Erythema: 1, barely perceptible light redness. Edema: 1, mild, raised <1 mm; 2, moderate, raised 1 to 2 mm; and 3, marked, raised >2 mm. 1+, slight; 2+, moderate; and 3+, severe.
Toxicokinetics Parameters and Concentrations of Each Compound in Skin and Eyes
The TK parameters of each compound are summarized in Table 7. Concentrations of each compound in the skin and eyes are summarized in Table 8.
The PDF-Related Changes in Rats Exposed to UV.
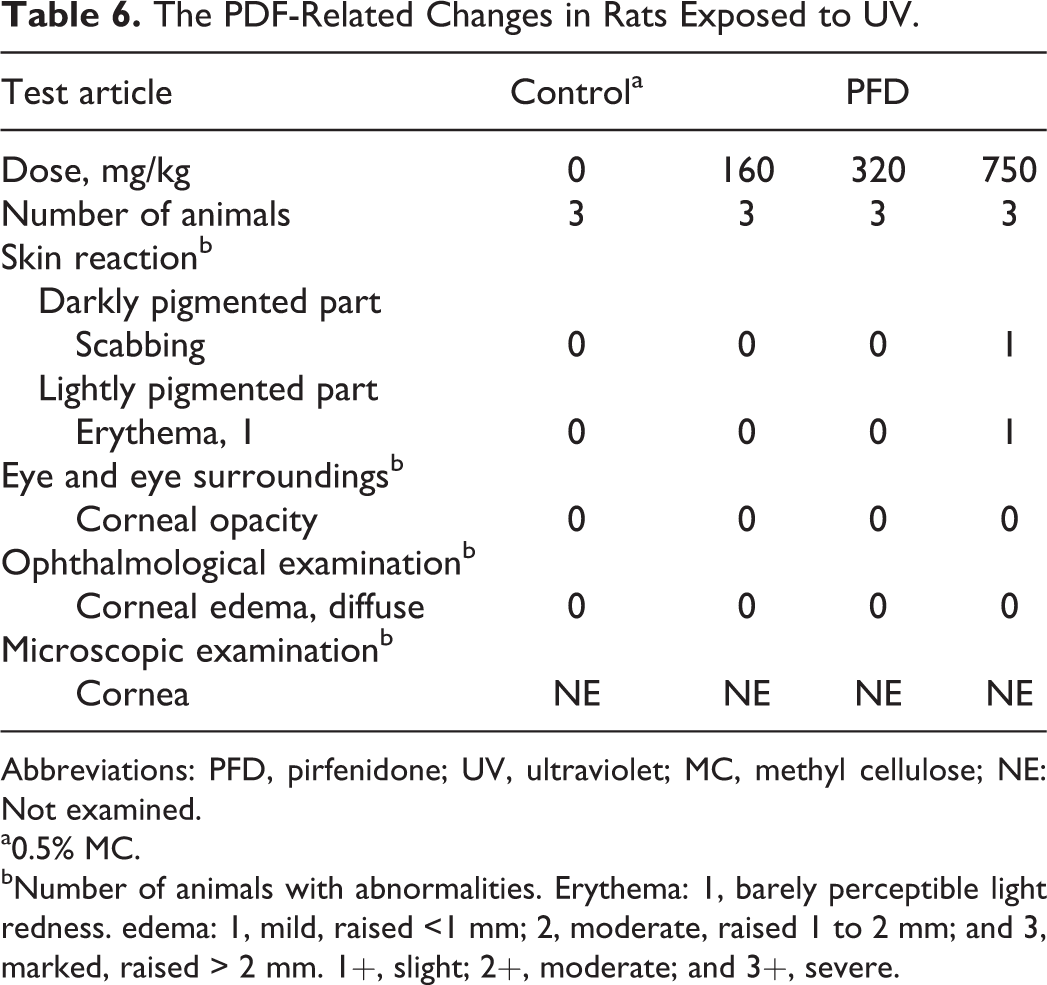
Abbreviations: PFD, pirfenidone; UV, ultraviolet; MC, methyl cellulose; NE: Not examined.
a0.5% MC.
bNumber of animals with abnormalities. Erythema: 1, barely perceptible light redness. edema: 1, mild, raised <1 mm; 2, moderate, raised 1 to 2 mm; and 3, marked, raised > 2 mm. 1+, slight; 2+, moderate; and 3+, severe.
The TK Parameters in LE Rats.a
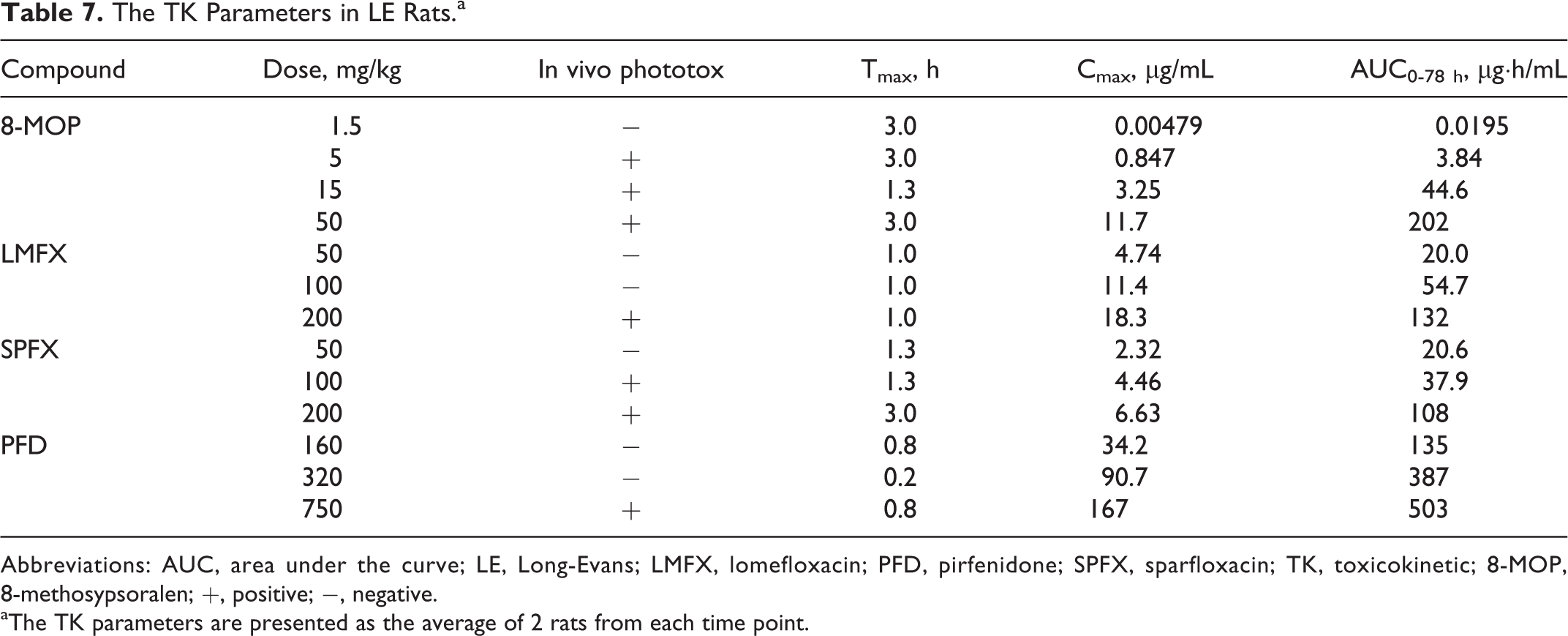
Abbreviations: AUC, area under the curve; LE, Long-Evans; LMFX, lomefloxacin; PFD, pirfenidone; SPFX, sparfloxacin; TK, toxicokinetic; 8-MOP, 8-methosypsoralen; +, positive; −, negative.
aThe TK parameters are presented as the average of 2 rats from each time point.
Concentrations of Compounds in the Skin and Eyes After Administration at Tmax.
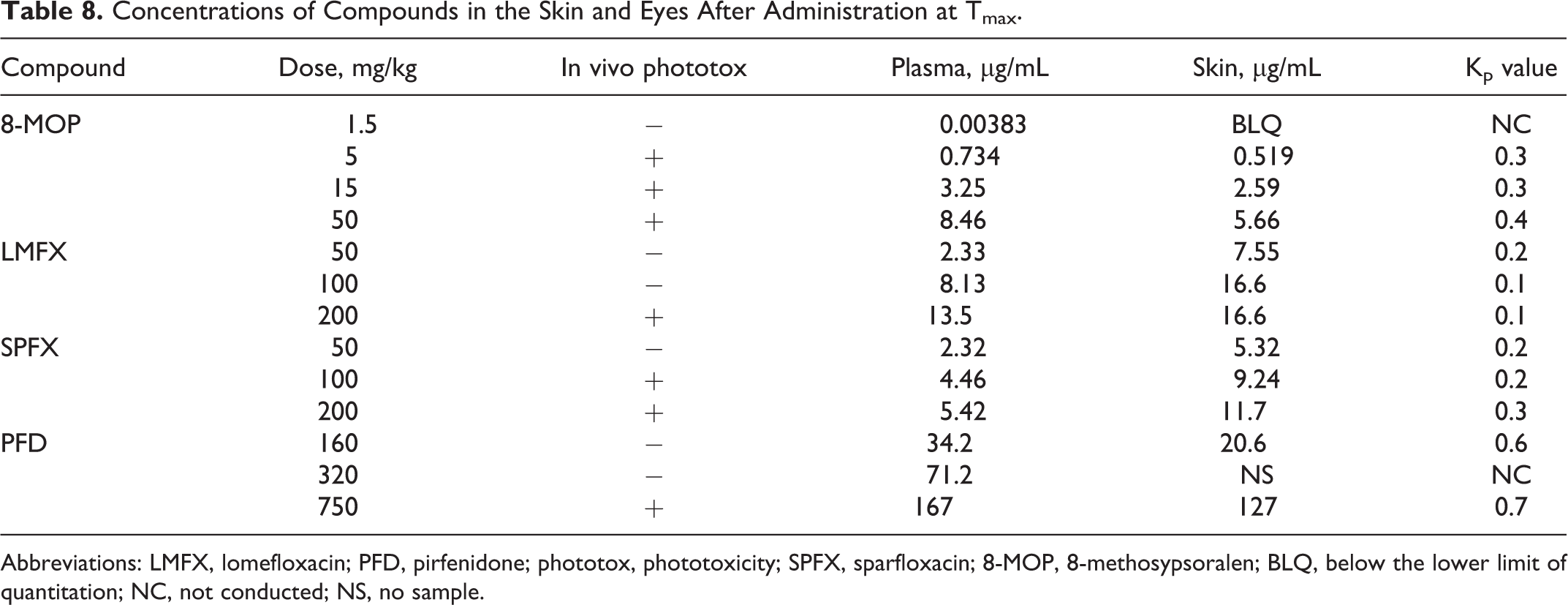
Abbreviations: LMFX, lomefloxacin; PFD, pirfenidone; phototox, phototoxicity; SPFX, sparfloxacin; 8-MOP, 8-methosypsoralen; BLQ, below the lower limit of quantitation; NC, not conducted; NS, no sample.
Calculation of MoS
Margins of safety were calculated for each compound using Cmax values at the NOAEL in LE rats and Cmax values of therapeutic doses in humans (Table 9). Because protein-binding rates of each compound were equivalent in rats and humans (Table 10), calculation of MoS was performed using measurements of total plasma concentrations.
In Vivo Parameters of Each Compound.

Abbreviations: LMFX, lomefloxacin; MoS, margins of safety; NOAEL, no-observed-adverse-effect level; PFD, pirfenidone; SPFX, sparfloxacin; 8-MOP, 8-methosypsoralen.
Plasma Protein Binding Rates of Each Compound.a
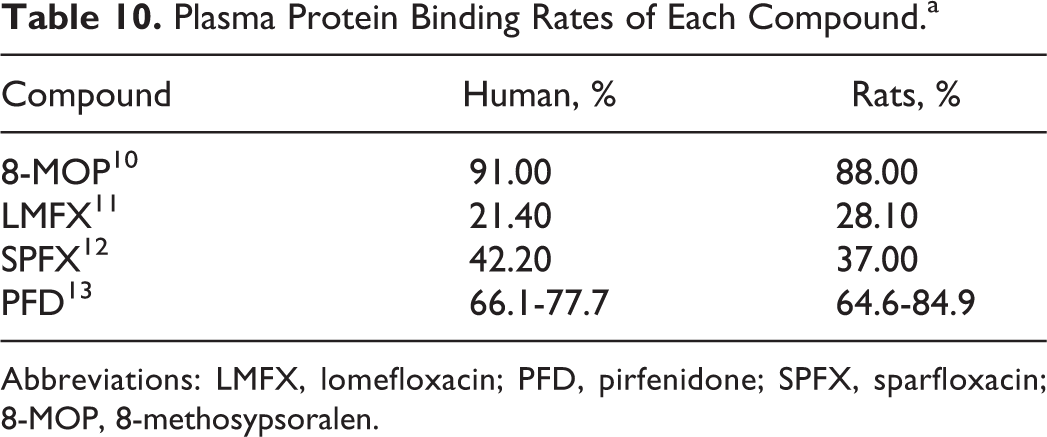
Abbreviations: LMFX, lomefloxacin; PFD, pirfenidone; SPFX, sparfloxacin; 8-MOP, 8-methosypsoralen.
Discussion
This study was designed to assess and validate an in vivo phototoxicity assay using female LE rats for quantitative human risk assessments. As test compounds, the 4 phototoxic drugs 8-MOP, LMFX, SPFX, and PFD varied in their phototoxic effects. In particular, all 4 compounds caused eye and/or skin reactions in LE rats that varied in severity and NOAELs, with much greater effects following treatments with 8-MOP compared to the other compounds.
At therapeutic doses of 8-MOP, skin and eye reactions in LE rats were frequently detected. The plasma Cmax value of 8-MOP at the NOAEL for phototoxicity in rats was lower than that of human clinical exposure levels. Thus, no MoS could be calculated for 8-MOP using this in vivo phototoxicity model, reflecting the “contraindication” for light exposure on the labeling of 8-MOP. In contrast, LMFX, SPFX, and PFD are labeled with a “warning” for light exposure, and MoSs were calculated using the in vivo phototoxicity model, with plasma NOAEL Cmax values of 5-, 3.7-, and 6.7-fold human exposure levels, respectively. In addition, exposures to LMFX, SPFX, and PFD at NOAELs in the present in vivo phototoxicity model were similar to those in clinical phototoxicity studies. These data clearly indicate that this in vivo phototoxicity test and TK analysis can be used to quantitatively predict phototoxic risks of pharmaceuticals in humans.
Compound concentrations in the skin/plasma (Kp value) at Tmax were equivalent for all compounds. Although 8-MOP produced the most severe skin reactions of these compounds, potential dermal transfer of 8-MOP was similar to that of the other compounds, suggesting that plasma concentrations (Cmax) rather than dermal exposures are predictive of human risk.
Previous phototoxicity assays for 8-MOP, LMFX, SPFX, and PFD were developed using guinea pigs, albino mice, and albino rats by various lamps. Although the differences of lamps used in each assay might affect the results of phototoxicity, the species and/or strain differences were focused in this discussion. Shimoda et al reported 14 the effects of single oral doses of LMFX (100 mg/kg) in BALB/c mice and showed skin photoirritation (erythema and swelling of the auricles) and a Cmax value of 3.90 µg/mL at 100 mg/kg. At a Cmax of 18.3 µg/mL (200 mg/kg in humans), LMFX caused erythema in LE rats but caused no skin reactions at the Cmax value of 11.4 µg/mL (100 mg/kg). These differences indicate sensitivities of BALB/c mice that may obscure the effects of phototoxic compounds or lead to overestimates of phototoxicity.
The phototoxicity of SPFX has been investigated in albino mice, and retinal changes were observed following doses of 50 mg/kg. 15,16 Segments and layers of retina were partially thinned and were completely lost at 96 hours after oral treatments. However, these results were in perspective. In the present study, retinal changes were not noted in LE rats treated with SPFX. The ocular path of mice is shorter than those in rats, some UVR might penetrate to the retina of mice. Therefore, the longer ocular path length might affect the lack of retinal changes in LE rats. In addition, melanin reportedly has photoprotective effects. Thus, the lack of retinal changes in LE rats may indicate the photoprotectivity of melanin pigment. However, no studies have demonstrated SPFX- and photo-related retinal changes in humans to date. Taken together, these data suggest that LE rats are better suited for evaluations of retinal phototoxicity than albino mice.
In a recent study by Matsumoto et al, BALB/c mice were given single oral doses of SPFX (30 mg/kg), and these mice showed skin photoirritation (erythema and swelling of auricles). The Cmax value at 30 mg/kg was 0.93 µg/mL in mice, whereas that associated with erythema in LE rats was 4.46 µg/mL (100 mg/kg). Moreover, the SPFX Cmax value 2.32 µg/mL (50 mg/kg) did not cause skin reactions in LE rats. These data further suggest that BALB/c mice may be too sensitive to UV exposure to offer a clinically accurate model of SPFX or LMFX phototoxicity.
Previous in vivo phototoxicity studies of PFD were performed using hairless mice and guinea pigs. At oral doses of 2.5, 10, 40, and 160 mg/kg once daily for 3 days, concomitant exposure of guinea pigs to UVA (at 10 J/cm2) and UVB (at unknown mJ/cm2) caused no microscopic skin reactions 9 . However, moderate erythema in the auricle was noted in animals treated with 160 mg/kg. The Cmax of PFD at 160 mg/kg was 57.9 µg/mL in guinea pigs, whereas that associated with erythema in LE rats was 167 µg/mL (750 mg/kg). The Cmax value of PFD for skin reactions in LE rats was 90.7 µg/mL (320 mg/kg). Hence, similar to BALB/c mice, the auricles of guinea pigs may be too sensitive for accurate preclinical assessments of PFD phototoxicity.
8-Methosypsoralen is a strong phototoxin and is used as a positive control in in vivo oral and topical phototoxicity assays. 17 However, few studies of 8-MOP phototoxicity include TK data. Thus, species differences in phototoxic reactions to 8-MOP remain unknown.
Taken with data from previous reports, the present observations indicate that LE rats are less sensitive to phototoxicity than albino rats, mice, and guinea pigs, presumably owing to the presence of melanin pigmentation in LE rats. 18 However, phototoxic reactions in the eyes and/or skin of LE rats treated with 8-MOP, LMFX, SPFX, and PFD were detected, and the associated MoSs reflected clinically relevant risks of phototoxicity. Thus, the relative photosensitivity of LE rats may be appropriate for phototoxicity studies of pharmaceuticals for human use. However, further investigations are required to identify optimal animal models for human risk assessments.
According to the ICH S10 guidance, 3 no standardized study methods have been established, and in vivo studies are performed at the discretion of drug developers.
In this study, LE rats were used in preclinical in vivo phototoxicity assays for the following reasons: The in vivo phototoxicity assay using LE rats with TK analysis quantitatively predicted the risk of phototoxicity from the well-known pharmaceuticals 8-MOP, LMFX, SPFX, and PFD. Ophthalmological examinations can be performed in rats using common methods, because LE rats are pigmented and therefore do not adversely respond to light alone. In contrast, the reported sensitivity of albino rat eyes to light exposure may obscure the effects of phototoxic test compounds. Nonetheless, although ocular (cornea) phototoxicity was observed in LE rats treated with 8-MOP, the detection of retinal phototoxicity from systemic drugs that absorb light above 400 nm requires use of a visible light source. Because SPFX has an absorption band in the range of 320 to 450 nm,
19
the lack of retinal phototoxicity after treatments with SPFX in this study may relate to the use of a UV source. 8-Methosypsoralen, LMFX, and PFD do not absorb visible light.
In conclusion, phototoxicities of the well-known human phototoxic compounds 8-MOP, LMFX, SPFX, and PFD were assessed in female LE rats after UV radiation at 10 J/cm2. The TK evaluations were performed, and MoSs were calculated using Cmax values of human therapeutic doses. Critical plasma concentrations of 8-MOP were lower in LE rats than at therapeutic dose levels in humans, and no margin of safety could be calculated. In contrast, LMFX, SPFX, and PFD had MoS of 5, 4, and 7 times the clinically relevant human exposure levels, respectively. These data correlate with the known human phototoxic risks of these compounds, as indicated by the relative severity of 8-MOP, which bears a “contraindication” for light exposure, compared with LMFX, SPFX, and PFD, which bear only a “warning” on their product inserts. In the present in vivo phototoxicity test, MoSs lower than 10 times may indicate the need for phototoxic risk assessments in clinical trials. Hence, this in vivo method offers important information for API development.
Footnotes
Author Contributions
Tamiko Adachi and Yuko Satou contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy. Hiroko Satou contributed to design, acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Hiroshi Shibata and Satoko Miwa contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy. Yumiko Iwase, Toshinobu Yamamoto, Atsuyuki Nishida, and Naoya Masutomi contributed to conception and design, interpretation, drafted the article, critically revised the article, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
