Abstract
Empagliflozin, a selective inhibitor of the renal tubular sodium-glucose cotransporter 2, was developed for treatment of type 2 diabetes mellitus. Nonclinical safety of empagliflozin was studied in a battery of tests to support global market authorization. Safety pharmacology studies indicated no effect of empagliflozin on measures of respiratory or central nervous system function in rats or cardiovascular safety in telemeterized dogs. In CD-1 mouse, Wistar Han rat, or beagle dogs up to 13, 26, or 52 weeks of treatment, respectively, empagliflozin exhibited a toxicity profile consistent with secondary supratherapeutic pharmacology related to glucose loss and included decreased body weight and body fat, increased food consumption, diarrhea, dehydration, decreased serum glucose and increases in other serum parameters reflective of increased protein catabolism, gluconeogenesis, and electrolyte imbalances, and urinary changes such as polyuria and glucosuria. Microscopic changes were consistently observed in kidney and included tubular nephropathy and interstitial nephritis (dog), renal mineralization (rat) and tubular epithelial cell karyomegaly, single cell necrosis, cystic hyperplasia, and hypertrophy (mouse). Empagliflozin was not genotoxic. Empagliflozin was not carcinogenic in female mice or female rats. Renal adenoma and carcinoma were induced in male mice only at exposures 45 times the maximum clinical dose. These tumors were associated with a spectrum of nonneoplastic changes suggestive of a nongenotoxic, cytotoxic, and cellular proliferation-driven mechanism. In male rats, testicular interstitial cell tumors and hemangiomas of the mesenteric lymph node were observed; both tumors are common in rats and are unlikely to be relevant to humans. These studies demonstrate the nonclinical safety of empagliflozin.
Introduction
Sodium-glucose cotransporters (SGLTs) are members of the soluble linked carrier gene superfamily. These membrane-bound proteins cotransport glucose or sugar across the cell membrane via a Na+ gradient. The principal members are SGLT-1 and SGLT-2, which are expressed predominantly in the intestinal epithelial cells and brush border of the renal proximal convoluted tubule, respectively. Approximately 90% of glucose filtered through the glomerulus is reabsorbed by SGLT-2 in the proximal convoluted tubule. 1 Inhibition of renal SGLT-2 has the effect of blocking glucose reuptake from the glomerular filtrate leading to increased excretion of glucose in the urine, a concept that has been exploited to reduce hyperglycemia in patients with type 2 diabetes mellitus. Phlorizin, a natural inhibitor of SGLTs isolated from the root bark of the apple tree, was first identified in 1835, and modern SGLT-2 inhibitors have been derived to increase potency and specificity for the SGLT-2 isoform. 2
Empagliflozin is a potent and selective SGLT-2 inhibitor having an 50% inhibitory concentration (IC50) of 3.1 nmol/L toward human SGLT-2 and a high degree of selectivity over other SGLT isoforms. 3 Empagliflozin is >2500-fold selective for human SGLT-2 over SGLT-1. Similar to other SGLT-2 inhibitors, empagliflozin has reduced selectivity (approximately 60-fold lower) for rat SGLT-2 versus SGLT-1. 3,4
In clinical studies, empagliflozin improved glycemic control in patients with type 2 diabetes mellitus and has demonstrated a favorable safety profile. 5 Reported here are the pivotal safety pharmacology, genetic toxicology, general toxicology, and carcinogenicity evaluations of empagliflozin supporting global market authorization.
Materials and Methods
Empagliflozin, (1S)-1,5-anhydro-1-(4-chloro-3-{4-[(3S)-tetrahydrofuran-3-yloxy]benzyl}phenyl)-
Pivotal studies were conducted according to Good Laboratory Practice (except in vitro studies of cardiovascular safety) and are listed together with duration of treatment, dose levels tested, and identified no observed adverse effect levels in Table 1.
Pivotal Toxicology Studies of Empagliflozin.
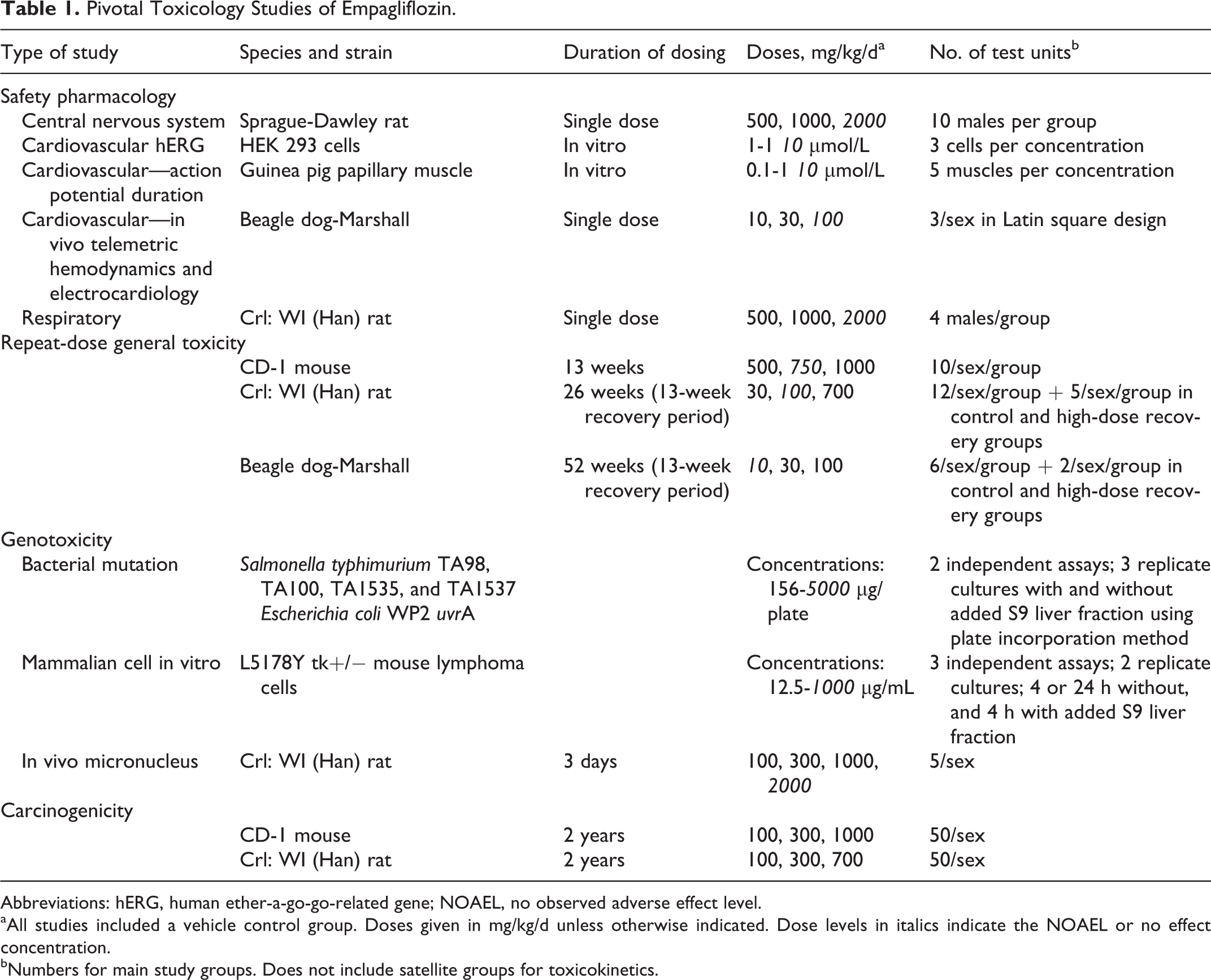
Abbreviations: hERG, human ether-a-go-go-related gene; NOAEL, no observed adverse effect level.
aAll studies included a vehicle control group. Doses given in mg/kg/d unless otherwise indicated. Dose levels in italics indicate the NOAEL or no effect concentration.
bNumbers for main study groups. Does not include satellite groups for toxicokinetics.
Animals and Husbandry
Mice were of the CD-1 strain from Charles River Laboratories (Raleigh, North Carolina or Portage, Michigan) and approximately 6 to 8 weeks of age and 19 to 35 g at start of dosing. Rats were of the Wistar (Han) strain from Charles River Laboratories (Raleigh, North Carolina, or Sherbrooke, Quebec, Canada) and were approximately 7 to 8 weeks of age and 190 to 250 g (males) or 165 g (females) at start of dosing. Dogs were beagles from Marshall BioResources (North Rose, New York) and were approximately 6 months of age and from 5.9 (females) to 7.5 kg (males) at start of dosing. All animals were maintained in temperature-, light-, and humidity-controlled rooms in housing and under husbandry conditions consistent with the National Research Council Guide for the Care and Use of Laboratory Animals.
Experimental Design
The design features of the pivotal toxicology studies are given in Table 1.
Safety Pharmacology
The assessment of central nervous system effects following a single oral dose of empagliflozin was conducted in 4 groups of 10 male rats. Rats were observed for apparent neuropharmacological or toxicological signs at 15, 30, and 45 minutes, 1, 2, 3, 4, and 24 hours following the single dose. Body temperature was measured at 60 minutes following dose administration. The assessment of respiratory system effects following a single dose of empagliflozin was conducted in 4 groups of 4 male rats using the head-out plethysmography technique. Baseline respiratory parameter data were obtained for 5 minutes, rats were removed from the chambers, dosed, and returned to the chamber. Respiratory parameters were recorded 15 minutes and 1, 2, 3, 4, and 6 hours after administration. In vivo cardiovascular safety was assessed in 2 male and 2 female conscious dogs using a crossover randomized design so that each dog served as its own control and received single doses of empagliflozin or vehicle on separate study days. Dogs were surgically implanted with telemetry transducers including an aortic pressure sensor and 6-lead electrocardiogram (ECG). Hemodynamic and electrocardiographic parameters were evaluated for 24 hours postadministration. The effect of dimethyl sulfoxide solutions of empagliflozin on human ether-a-go-go-related gene (hERG)-mediated potassium current and on the action potential configuration was studied in vitro in HEK293 cells and isolated guinea pig papillary muscle, respectively. Membrane currents from HEK293 cells were recorded using whole-cell patch-clamp technique. Isolated guinea pig papillary muscles were evaluated using a tension-loading transducer. Muscles were stimulated over 3 ms with a frequency of 1 Hz for an equilibrium period of 1 hour and 0.33 Hz during experiments. Action potentials were recorded by microelectrode.
General Toxicology
General toxicology was assessed in mice, rats, and dogs via daily gavage administration of empagliflozin for 13, 26, or 52 weeks, respectively. Recovery groups were included in the rat and dog studies. Dose levels were selected based on results from shorter duration studies (not presented). In-life parameters collected on all studies included daily observations for mortality and morbidity and general clinical examinations for signs of toxicity prior to daily dosing. Detailed physical examinations and measures of food consumption and body weight were conducted weekly. Ophthalmic examinations were performed on rats and dogs during the pretest period and near the end of drug phase. Electrocardiology evaluations were conducted in dogs during the pretest period, week 39, and the last week of dosing. Clinical pathology evaluations in rats (weeks 5, 13, 26, and recovery week 13) and dogs (pretest, week 52, and recovery week 13) included hematology, coagulation, clinical chemistry, urinalysis, and urine chemistry panels. Only clinical chemistry was conducted in the mouse study. At the end of drug phase, main study animals were humanely euthanized using CO2 anesthesia (mice and rats) or sodium pentobarbital injection (dogs) and exsanguination. Tissues and organs were collected, selected tissues were weighed, and all were fixed in 10% neutral-buffered formalin or other fixative, routinely dehydrated and embedded into paraffin, sectioned, and stained with hematoxylin and eosin. Special methods such as Oil Red O for visualization of lipid in rat liver and kidney or immunohistochemical staining for Ki-67 as a marker of cell proliferation in mouse kidney were also employed. All microscopic evaluations were conducted and peer reviewed by veterinary pathologists.
Toxicokinetic evaluations were conducted in satellite groups of mice and rats and on all dogs during drug week 1 and during the last week of drug phase for all studies and in the middle of drug phase for rat and dog general toxicology studies (drug weeks 4 and 13 for rats and drug week 14 for dogs). During the carcinogenicity studies, toxicokinetic evaluations were conducted on drug day 1 and during drug weeks 13 and 26. Following their last toxicokinetic blood collection, satellite groups of mice and rats were humanely euthanized. Plasma extracts were separated chromatographically using a reversed phase column, and empagliflozin was quantified using liquid chromatography–mass spectrometry multiple reaction monitoring transitions with [ 13 C6]-empagliflozin as the internal standard.
Genetic Toxicology
The mutagenic potential of empagliflozin was investigated in a study with Salmonella typhimurium strains TA1535, TA1537, TA98, and TA100 and Escherichia coli WP2 uvrA (pKM101) using the plate incorporation method in the presence and absence of an Aroclor 1254-induced rat liver preparation (S9 mix). Concentrations ranged from 0 to 5000 µg/plate. The mutagenic and clastogenic potential of empagliflozin was also evaluated in the L5178Y tk +/− mouse lymphoma cell assay in the presence and absence of S9 mix. Concentrations ranged from 0 to 350 µg/mL. An in vivo micronucleus test for chromosomal aberrations was conducted following administration of empagliflozin to rats for 3 days at doses up to 2000 mg/kg/d. Approximately 24 hours after the last dose, the rats were euthanized and bone marrow smears were prepared for micronucleus evaluation. Bone marrow smears were stained with acridine orange and evaluated for alteration in the ratio of polychromatic to normochromatic erythrocytes and the frequency of micronucleated polychromatic erythrocytes. All genetic toxicology studies followed International Conference on Harmonisation and Organization for Economic Cooperation and Development guidelines.
Carcinogenicity
Carcinogenicity studies in mouse and rat were performed at Covance Laboratories, Inc (Madison, Wisconsin) by daily oral dosing for up to approximately 2 years. Parameters evaluated include mortality, clinical signs of toxicity, body weights, food consumption, ophthalmic examinations, and anatomic pathology. Plasma samples were collected for toxicokinetic evaluations (see General Toxicology).
Results
Dose Formulation Analyses
All analyses of formulations and control/vehicle solutions were within acceptable ranges of nominal concentration, were homogeneous, and stable during the period of dosing.
Safety Pharmacology
In the study of effects on the central nervous system, there were no effects on behavioral end points or body temperature. In the study of respiratory system effects, there were no effects on tidal volume, minute volume, or respiratory rate. The IC50 for inhibition of the hERG tail current was greater than 30 µmol/L. There was no effect on the action potential configuration or contractile function of the isolated guinea pig papillary muscle at concentrations up to 10 µmol/L. In the study of cardiovascular function in telemeterized dogs, there was no effect on systolic or diastolic arterial blood pressure, maximal LVP, LVP dP/dt-max, heart rate, QRS complex, or QT interval duration. Cardiovascular function (heart rate, blood pressure, and ECG parameters) was also assessed during the repeat-dose studies in dog (including studies of 2-, 4-, 26-, and 52-week duration). There were no physiologically meaningful changes observed in any of these studies.
General toxicology
Exposure to empagliflozin generally increased proportionally with dose. Exposure in rodents was typically lower in males than females and therefore exposure multiples are expressed relative to males (Table 2). Exposure was similar between male and female dogs and therefore exposure multiples are expressed relative to the sex-averaged exposure.
Summarized Key Findings and Toxicokinetic Data from Pivotal General Toxicology Studies of Empagliflozin.
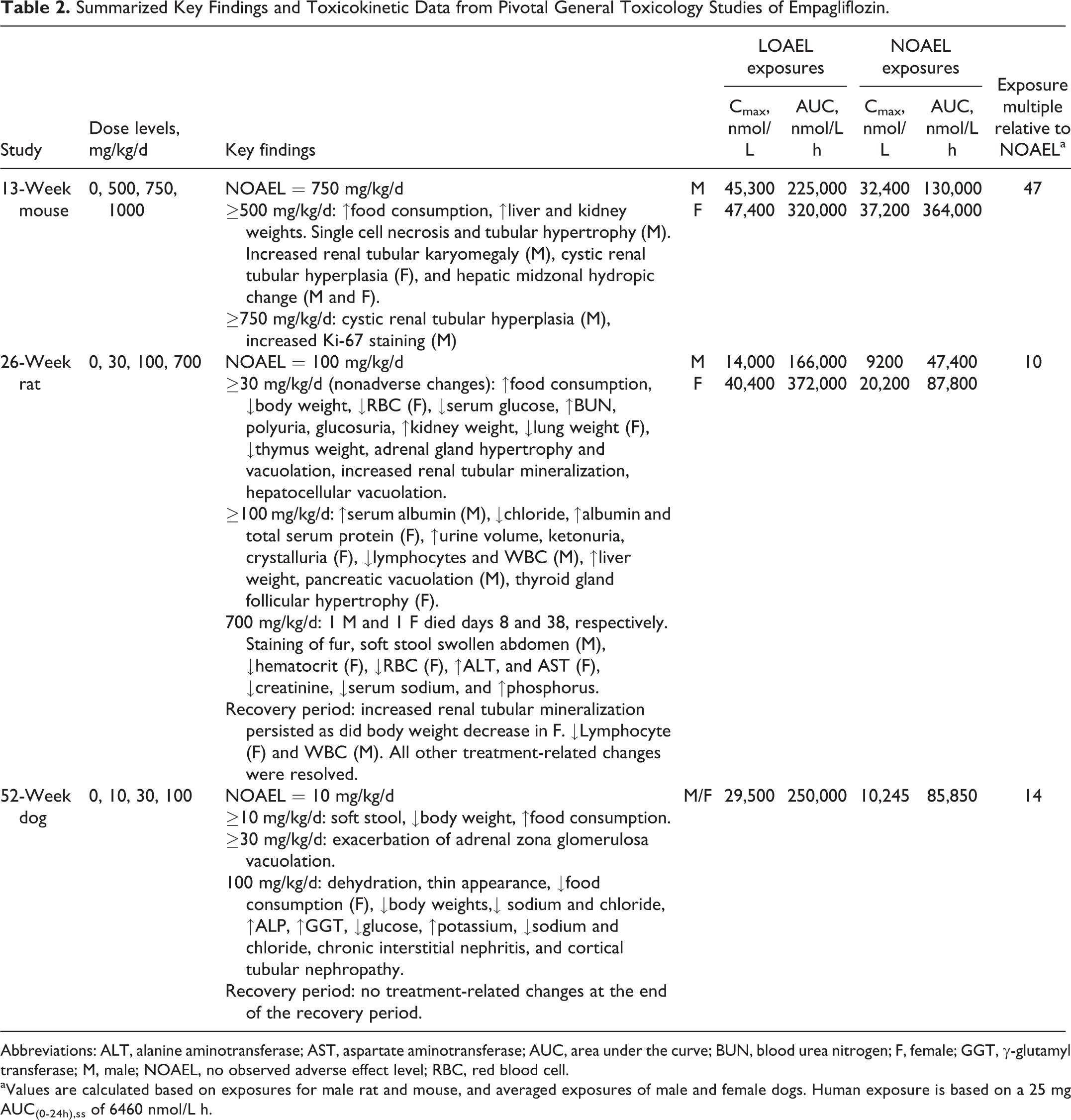
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; AUC, area under the curve; BUN, blood urea nitrogen; F, female; GGT, γ-glutamyl transferase; M, male; NOAEL, no observed adverse effect level; RBC, red blood cell.
aValues are calculated based on exposures for male rat and mouse, and averaged exposures of male and female dogs. Human exposure is based on a 25 mg AUC(0-24h),ss of 6460 nmol/L h.
Mouse
Repeat-dose toxicity in mice was evaluated in a study of 13-week duration (Table 2). Body weight and body weight gain were not adversely affected by treatment (Figure 1) but mean food consumption was increased in all dose groups. Clinical signs of toxicity and hematology parameters were unaffected. Liver weight increases (24%) were attributed to midzonal hydropic change (clearing of the cytoplasm). Histopathological changes in kidneys included cystic tubular hyperplasia, increased tubular karyomegaly, increased single cell necrosis, and cell proliferation (Figure 2). Tubular cell proliferation was identified by increased Ki67 staining in the superficial cortex that was accompanied by increased mitoses. The location of these changes correlated with the location of the nonproliferative and proliferative changes in the kidneys of males in the carcinogenicity study in mice. For the purposes of establishing dose levels for the carcinogenicity study, the maximum tolerated dose was determined to be greater than 750 mg/kg/d. This dose produced systemic exposures approximately 47-fold greater than that associated with administration of 25 mg/d to humans.
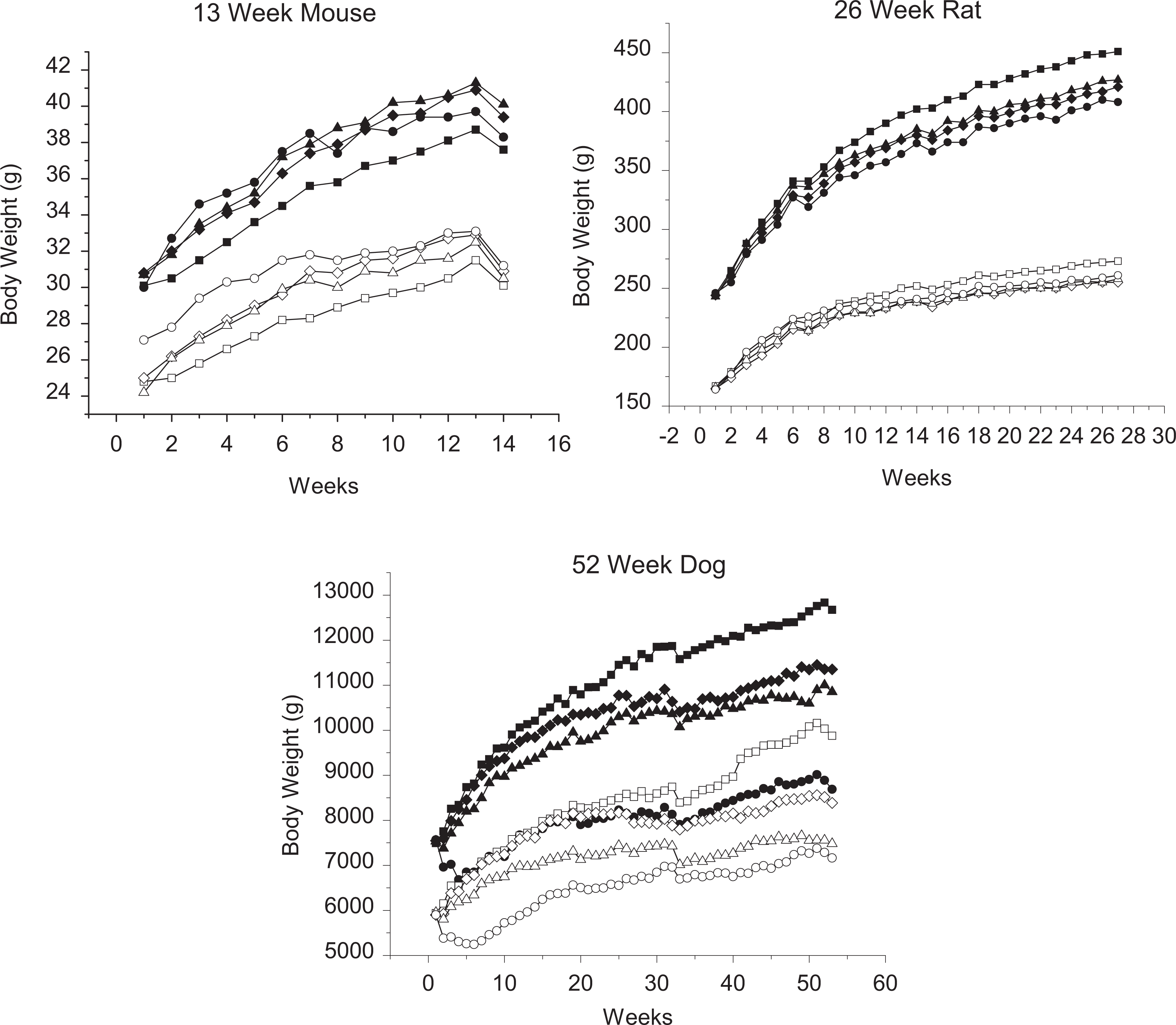
Growth curves for 13-week mouse, 26-week rat, and 52-week dog studies. Males, filled symbols and females, open symbols. Control, squares; low dose, diamond; mid dose, triangle; and high dose, circles. Dose levels are mouse: 0, 500, 750, and 1000 mg/kg/d; rat: 0, 30, 100, and 700 mg/kg/d; and dog: 0, 10, 30, 100 mg/kg/d.
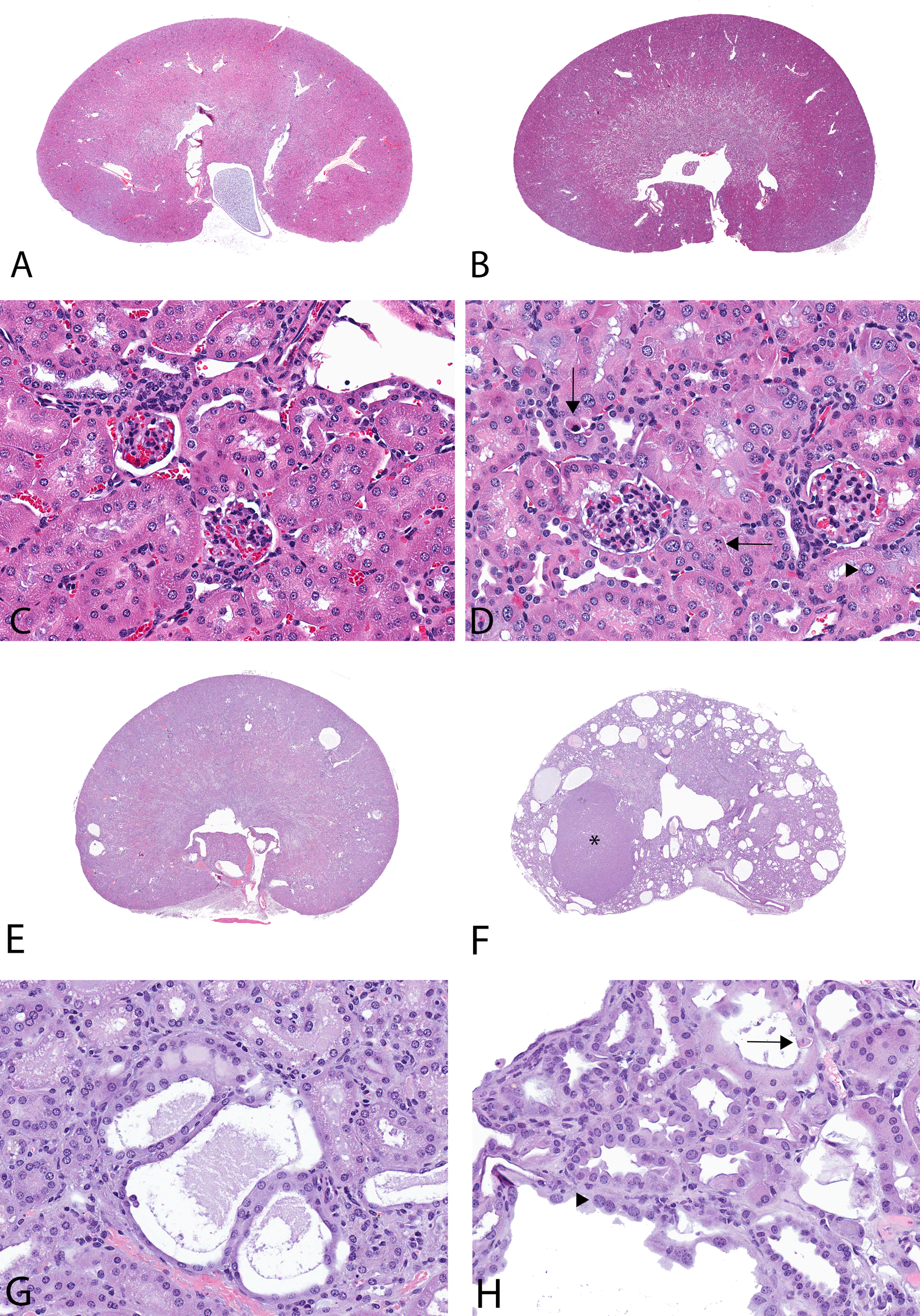
A 13-week toxicity study with empagliflozin in the CD-1 mouse (images A-D). A, Kidney of control male (10×). B, Kidney of a 1000-mg/kg/d male (10×). C, Higher magnification of renal cortex of the control male with normal tubules (400×). D, Higher magnification of renal cortex of the 1000 mg/kg/d male with single cell necrosis (arrows) and karyomegaly of tubular epithelial cells (eg, arrowhead; 400×). Two-year carcinogenicity study with empagliflozin in the CD-1 mouse (images E-H). E, Kidney of control male with scattered background tubular cysts (10×). F, Kidney of 1000 mg/kg/d male mouse with renal tubular adenoma (*), moderate cysts, and cystic hyperplasia (10×). G, Higher magnification of renal cortex of the control animal with tubular cysts/cystic hyperplasia (400×). H, Higher magnification of renal cortex of the 1000 mg/kg/d male mouse with tubular cysts/cystic hyperplasia, single cell necrosis (arrow), and karyomegaly of tubular epithelial cells (eg, arrowhead; 400×).
Rat
Repeat-dose oral toxicity evaluations in rat included studies of 2, 4, 13, and 26 weeks in duration. In all studies, there was a largely consistent pattern of observations including increases in food consumption, decreases in body weight gain, and clinical chemistry. Changes in urinalysis parameters were also similar and included decreases in plasma glucose, glucosuria, and polyuria, all of which are related to the primary mode of pharmacological action (Table 3). Decreased serum chloride and sodium were attributed to polyuria while increases in phosphorus were attributed to secondary compensatory responses to altered calcium homeostasis. At higher doses and/or longer term exposures, additional observations included increases in blood urea nitrogen, plasma alanine aminotransferase (ALT), aspartate aminotransferase (AST), triglycerides, and ketonuria. Alanine aminotransferase and AST were increased less than 2× compared to controls and were not associated with liver toxicity or other histopathological correlate. These effects are consistent with secondary responses to high-dose pharmacology and are consistent with an adaptive metabolic shift toward gluconeogenesis and fatty acid metabolism. All clinical chemistry changes returned to control values during recovery periods.
Notable Changes in End-of-Dosing Phase Clinical Chemistry and Urinalysis Parameters in Pivotal General Toxicology Studies of Empagliflozinin Rat and Dog.a
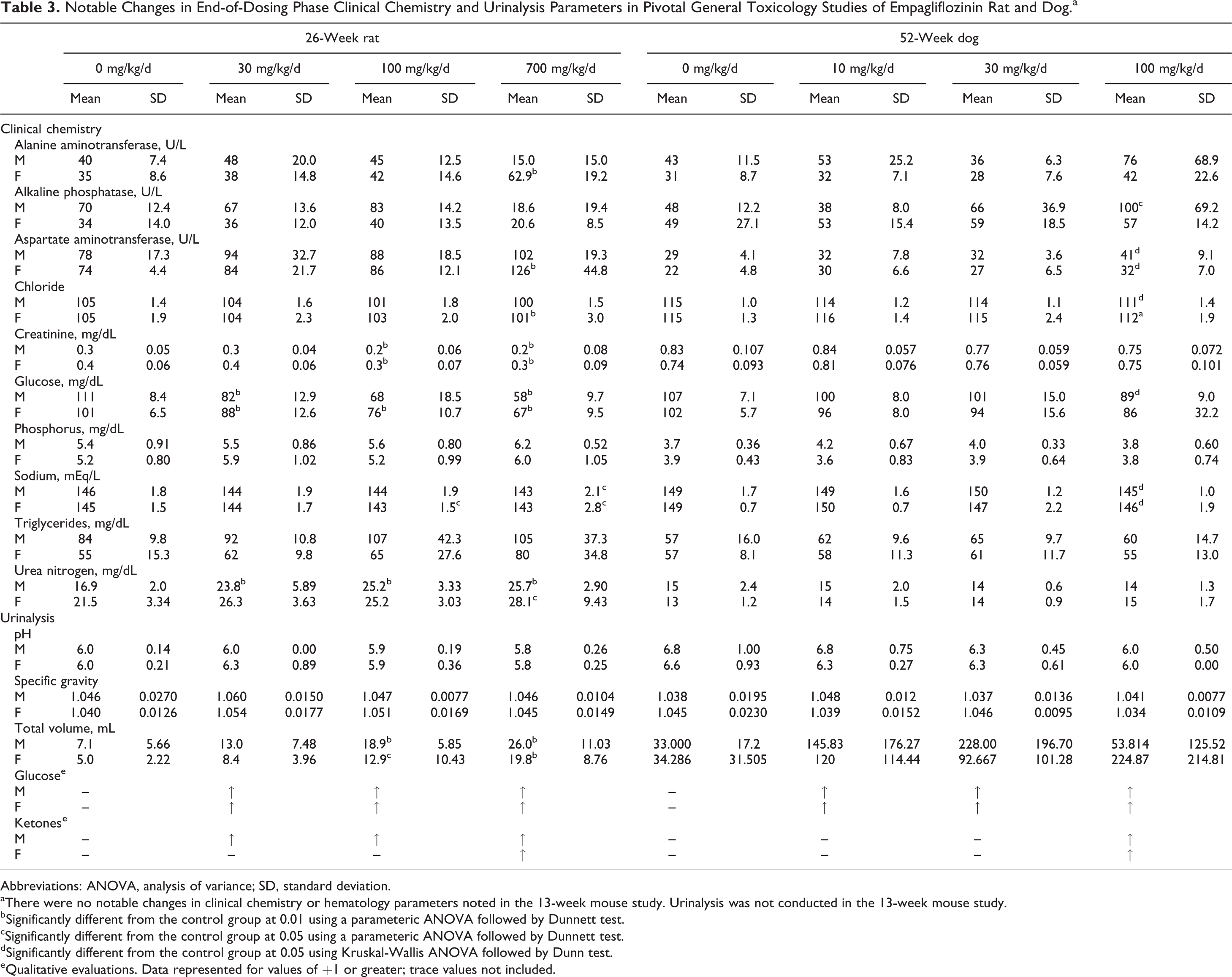
Abbreviations: ANOVA, analysis of variance; SD, standard deviation.
aThere were no notable changes in clinical chemistry or hematology parameters noted in the 13-week mouse study. Urinalysis was not conducted in the 13-week mouse study.
bSignificantly different from the control group at 0.01 using a parameteric ANOVA followed by Dunnett test.
cSignificantly different from the control group at 0.05 using a parameteric ANOVA followed by Dunnett test.
dSignificantly different from the control group at 0.05 using Kruskal-Wallis ANOVA followed by Dunn test.
eQualitative evaluations. Data represented for values of +1 or greater; trace values not included.
Histologically, an increase in renal tubular mineralization, characterized as tubular, papillary, and pelvic mineralization, was observed in the 13- and 26-week studies. In the 26-week study, the increase in renal tubular mineralization was observed at doses ≥30 mg/kg/d and persisted through the 3-month recovery period. Other organs having nonadverse secondary responses to empagliflozin include pancreas, thyroid, liver, and adrenals and were typically only observed in the 26-week study. Pancreatic changes in rats featured occasionally observed vacuolation of the basilar portion of cells, which were depleted of zymogen, an effect considered secondary to the increased food consumption and decreased body weight. 6 In thyroid glands, minimal follicular cell hypertrophy was observed at a low incidence only in males and was suggestive of increased activity. Liver changes were characterized as diffuse microvesicular hepatocellular vacuolation, which is consistent with mobilization of lipids for energy, secondary to chronic glucose loss. Finally, in adrenal glands of rats of the 26-week study, test article-related nonadverse vacuolation of the zona glomerulosa and hypertrophy of the zona fasciculata were observed in both sexes in all dosed groups and correlated with dose.
Dog
Repeat-dose oral toxicity evaluations in dog included studies of 2, 4, 13, 26, and 52 weeks in duration. Similar to studies in the rat, there was a largely consistent pattern of observations including soft stool/diarrhea, decreased body weight gain, and clinical chemistry and urinalysis changes including decreased plasma glucose, glucosuria, and polyuria (Table 3). Unlike the rat, however, secondary responses to glucose loss indicative of shifts in intermediary metabolism were not typically observed. Only in the 52-week study, changes in serum electrolytes were apparent, which included increases in serum potassium and decreases in serum sodium and chloride, all of which can be related to diarrhea and polyuria. Increases in bone-specific serum alkaline phosphatase were observed but these were within published limits, there were no changes in serum osteocalcin, and there were no microscopic findings in bone. Histologically, the kidney was the single consistent target organ of toxicity of empagliflozin. Interstitial nephritis and tubular nephropathy were observed in the 4-, 13-, and 52-week studies. Although not fully recoverable in 1 dog of the 4-week study given a 4-week recovery period, complete recovery of this lesion was observed after a 13-week recovery period in the 13- and 52-week studies.
Atrophy of the prostate was observed in males of all repeated dose studies. This change may have been related to weight loss and/or emaciation. It was considered nonadverse as it was readily reversible during treatment-free recovery periods.
Genetic toxicology
Empagliflozin did not cause mutations in all strains of S typhimurium in the presence or absence of metabolic activation, when tested up to bacteriotoxic concentrations of 5000 μg/plate. Empagliflozin did not induce gene or chromosomal damage at the tk locus of mouse lymphoma L5178 tk +/ − cells, when tested up to cytotoxic concentrations both in the presence or absence of metabolic activation. Empagliflozin did not induce an increase in the frequency of micronuclei in polychromatic erythrocytes at any dose level up to the highest dose of 2000 mg/kg in 3-day repeat-dose studies. Thus, the results of the in vitro and in vivo studies demonstrate that empagliflozin is not genotoxic.
Carcinogenicity
Mouse
There were no treatment-related tumors observed in female mice. Survival among male mice given 1000 mg/kg/d decreased to 16/50 by week 97 resulting in early termination of that group. The only empagliflozin-related tumors observed in male mice were renal tumors (tubular adenomas and carcinomas) in the 1000 mg/kg/d group, with a total incidence of 10% (Table 4) occurring at an exposure multiple of 45 times the exposure associated with a 25 mg therapeutic dose. Renal neoplasms in males given 1000 mg/kg/d were generally associated with chronic empagliflozin-related degenerative tubular changes that resulted in an increased incidence of proximal tubule single cell necrosis (Figure 2), tubular epithelial hypertrophy, karyomegaly (Figure 2), cystic hyperplasia, and atypical hyperplasia (Table 5). These degenerative tubular changes were present at a much lower incidence or absent in all other groups, including controls. Therefore, renal neoplasms in males given 1000 mg/kg/d were considered secondary to chronic and persistent tubular degeneration, necrosis, and regeneration. Because the incidence of single cell necrosis and karyomegaly was not increased for males given 300 mg/kg/d, the single renal tubular adenoma in a male given 300 mg/kg/d was considered not likely related to empagliflozin. In comparison to the exposure associated with the 25 mg/d dose in humans, the drug-related renal tumors were absent at exposures of 11-fold.
Incidence of Empagliflozin-Related Renal Proliferative Microscopic Findings in the Male Mouse Carcinogenicity Study.
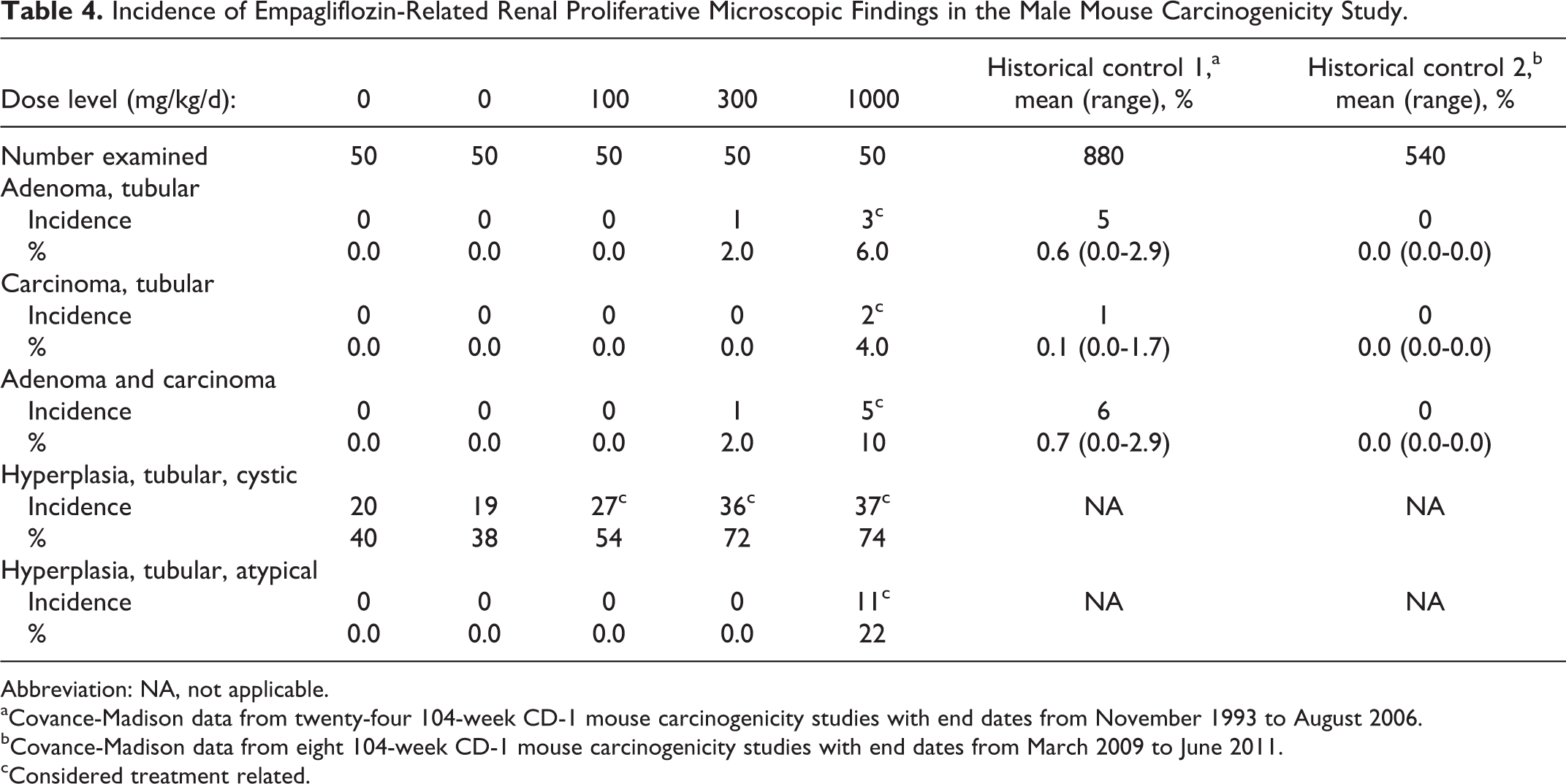
Abbreviation: NA, not applicable.
aCovance-Madison data from twenty-four 104-week CD-1 mouse carcinogenicity studies with end dates from November 1993 to August 2006.
bCovance-Madison data from eight 104-week CD-1 mouse carcinogenicity studies with end dates from March 2009 to June 2011.
cConsidered treatment related.
Incidence of Empagliflozin-Related Renal Nonproliferative Microscopic Findings in Mouse Carcinogenicity Study.
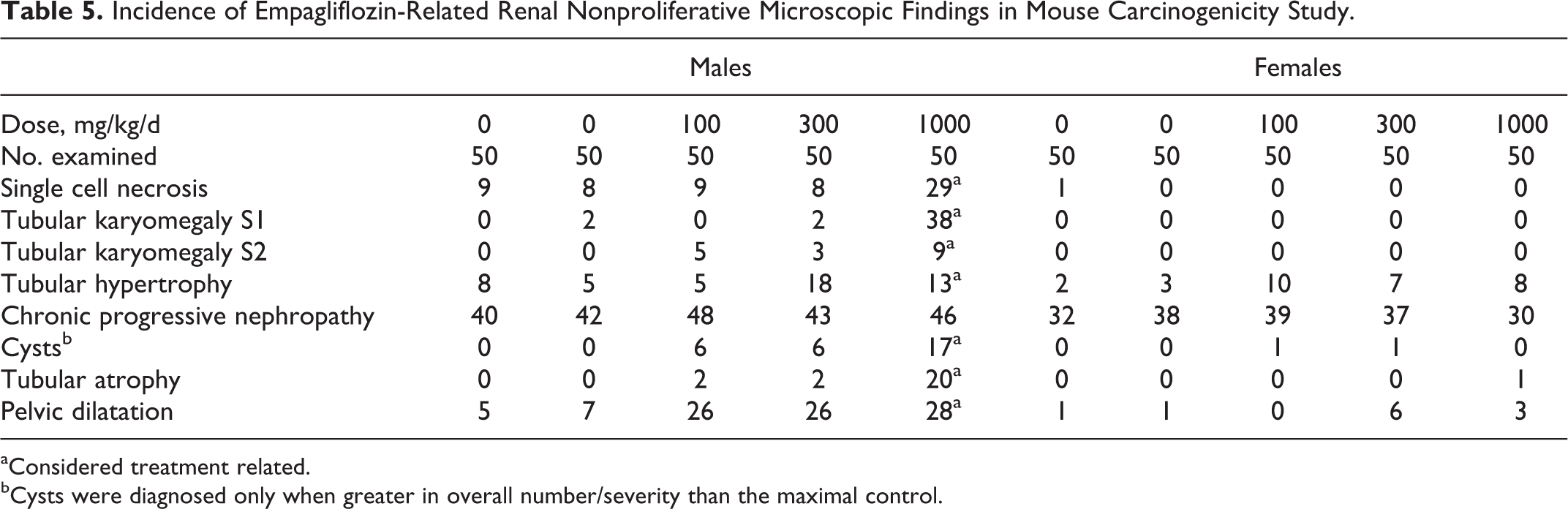
aConsidered treatment related.
bCysts were diagnosed only when greater in overall number/severity than the maximal control.
Incidence of Empagliflozin-Related Renal Nonproliferative Microscopic Findings in Rat Carcinogenicity Study.
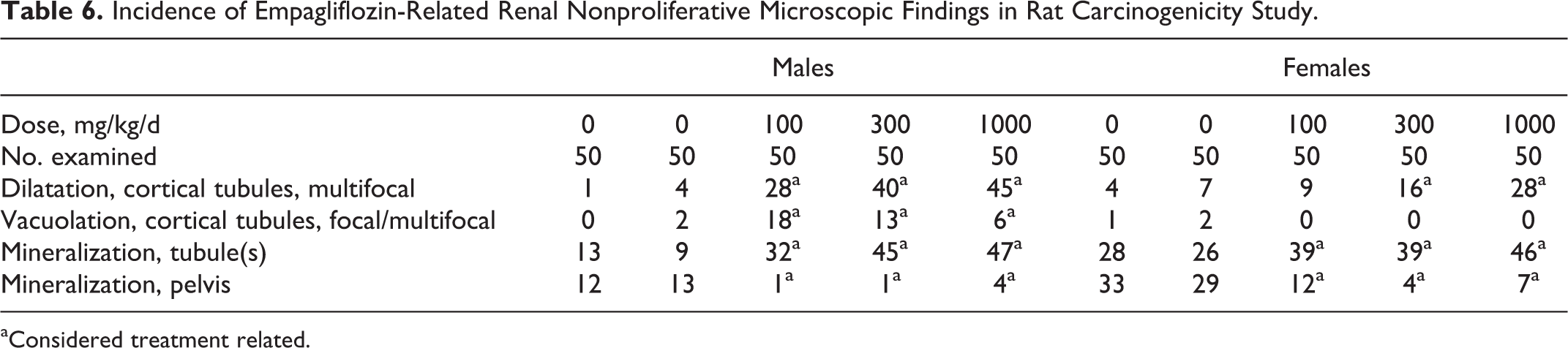
aConsidered treatment related.
Rat
There was no effect of empagliflozin on overall survival rates in males or females. There were no treatment-related tumors observed in female rats. In male rats, hemangioma of the mesenteric lymph node was observed in 6%, 0%, 2%, 8%, and 18% in the 0, 0, 100, 300, and 700 mg/kg/d groups, respectively. Also in males, a statistically increased incidence of benign interstitial (Leydig) cell tumors of the testes of 14% and 12% was observed in males given 300 or 700 mg/kg/d, respectively. However, this finding did not have a clear dose response, was not associated with an increased incidence of interstitial cell hyperplasia, and was considered likely not to be directly related to administration of empagliflozin. The incidence of interstitial cell tumors in males is within or only slightly above the published historical control ranges (0%-12%). 7 Relevant to this finding was a dose-dependent decrease in the incidence of benign and malignant mammary gland tumors in female rats (total mammary tumor incidence of 24%, 28%, 24%, 10.2%, and 6.0%, respectively).
Nonneoplastic, empagliflozin-related microscopic findings occurred in the kidney and included multifocal dilatation of cortical tubules in males administered ≥100 mg/kg/d and females given ≥300 mg/kg/d, focal to multifocal vacuolation of cortical tubules in males given ≥100 mg/kg/d, and increased mineralization of tubules and decreased mineralization of the renal pelvis in both sexes administered ≥100 mg/kg/d. Nonneoplastic, empagliflozin-related microscopic findings, all considered to be nonadverse, were also observed in the mesenteric lymph node (increased incidence and/or severity of sinus histiocytosis, pigmented macrophages, mast cells, and/or sinus erythrocytes), liver (increased vacuolation of sinusoidal cells), pancreas (granule depletion of acinar cells), and bone of femur (increased residual cartilage in diaphysis) in animals given ≥100 mg/kg/d; bone of sternum (increased trabecular bone) in males given ≥300 mg/kg/d, glandular stomach (erosion/ulceration) in males given 700 mg/kg/d and females given >300 mg/kg/d; and seminal vesicle (decreased secretion) in males given 700 mg/kg/d. Empagliflozin-related vascular mineralization and, less commonly, soft tissue mineralization were observed in multiple tissues in males given ≥100 mg/kg/d and females given ≥300 mg/kg/d.
Discussion
The toxicological potential of orally administered empagliflozin was investigated in a comprehensive battery of tests that included safety pharmacology, repeated dose general toxicology studies, and 2-year carcinogenesis studies in mice and rats. The genotoxic potential of empagliflozin was assessed in vitro in bacterial and mammalian systems and in vivo in rats. All toxicological evaluations support a conclusion that empagliflozin offers a low risk of toxicity when used in adults with type 2 diabetes mellitus. Across species, most toxicity was consistent with secondary pharmacology related to urinary glucose loss and included decreased body weight and body fat, increased food consumption, diarrhea, dehydration, decreased serum glucose and increases in other serum parameters reflective of increased protein catabolism, gluconeogenesis, and electrolyte imbalances, and urinary changes such as polyuria and glucosuria. Microscopic changes were consistently observed in kidney and included tubular epithelial cell karyomegaly, single cell necrosis, cystic hyperplasia and hypertrophy (mouse), renal mineralization (rat), and tubular nephropathy and interstitial nephritis (dog). These findings, including renal tubular cystic dilatation and hyperplasia, appear to be common to the SGLT2 class when tested in high-dose rodent studies. 8,9
Especially in the rat, caloric loss due to urinary glucose elimination resulted in responses consistent with a shift toward fat and protein metabolism. Cumulative mean body weight was 9% to 22% lower than controls, despite food consumption increases of approximately 20% to 30%. In the 26-week study, these responses included elevated serum triglycerides and urinary ketones and reversible microvesicular hepatocellular vacuolation, all evidence of a shift toward mobilization of hepatocellular lipid stores. Alanine aminotransferase was elevated 30% to 40% in the 700 mg/kg/d group, without histological evidence of any hepatotoxicity. Under fasting conditions, hepatic transaminase activities and the expression of hepatic genes involved in glycolysis, gluconeogenesis, and fatty acid oxidation increase. 10 Alanine is an important precursor for hepatic gluconeogenesis, and hepatic cytosolic AST increases the supply of oxaloacetate as a substrate for cytosolic phosphenolpyruvate carboxykinase. 10,11 Similar increases in serum transaminase levels, in the absence of hepatotoxicity, have been observed with other SGLT2 inhibitors. 7,8
The kidney was a target organ of toxicity in all 3 species examined. Tubular dilatation was a common component of the renal pathology in all 3 species following repeat-dose administration of empagliflozin. This finding may be related to the pharmacology of the compound, as the consequence of increased tubular flow rates and intratubular pressure within the early proximal tubule resulting from SGLT2 inhibition. 12 However, there are species and gender differences in the severity and the consequences of this effect. In the rat, dog, and female mouse, the dilatation is generally less severe than in the male mouse and does not lead to cyst formation, hypertrophy, or hyperplasia. Differences in empagliflozin exposure do not explain these differences, as the milder renal effects are detected in rats, dogs, and female mice at equivalent or greater systemic exposure than that at which severe effects are seen in male mice. Osmotic diuresis is an expected consequence of pharmacological renal SGLT2 inhibition. Because the male CD-1 mouse exhibits high background cystic hyperplasia and susceptibility to renal pelvic dilatation, osmotic diuresis can be expected to exacerbate effects related to slower renal flow velocities in relationship to overall output. This mechanical stress is considered tolerable, but otherwise can contribute to secondary renal adaptive effects in the CD-1 male mouse. Based on current understanding of how renal tubular epithelial cells sense fluid flow and its relationship to curbing dedifferentiation proliferation, 13,14 an increased incidence of cysts and cystic hyperplasia at excessively high empagliflozin exposures in the male CD-1 mouse is not unexpected.
Additionally, there are no renal changes in rats, dogs, or female mice indicative of chronic, low-level, renal injury, and compensatory regenerative responses that are hypothesized to contribute to the increased incidence of renal neoplasia in the male mouse. The absence of these changes supports the hypothesis that the pharmacologically mediated effects of empagliflozin on the kidneys (which are seen across the toxicology dose range studied in both genders in all species) are not by themselves adverse, and that the mechanism of chronic renal injury leading to the generation of renal tumors is dependent on a renal metabolite specific to the male mouse and is not directly related to the pharmacology of empagliflozin. 15
Increased renal tubular and pelvic mineralization, other soft tissue mineralization, and the nonadverse effects on bone observed after 2 years of dosing in the rat likely relate to high-dose inhibition of intestinal SGLT1. Although empagliflozin is a potent and selective inhibitor of SGLT2, it is less selective for rat SGLT2 than for the mouse, dog, or human orthologs. 3 The IC50 of empagliflozin for rat SGLT1 (350 nmol/L) is >24× lower than for mouse or human SGLT1. High-dose inhibition of intestinal SGLT1 results in osmotic diarrhea and imbalances in water and electrolyte absorption. 8 Decreased intestinal pH related to gut fermentation of unabsorbed glucose could facilitate intestinal absorption of calcium. Studies in rats of high dietary concentrations of sorbitol, xylitol, or lactose cause enlargement of the cecum, increased absorption of calcium from the gut, and increased urinary excretion of calcium, pelvic, and corticomedullary nephrocalcinosis. 16 -18 Consistent with this observation is that renal or other tissue mineralization was uncommon in the toxicology studies in mouse or dog.
Changes in serum organic phosphorus were indicative of altered calcium–phosphate homeostasis. The SGLT2 inhibitors increase phosphorus reabsorption from the renal filtrate. 9 Increased extracellular calcium increases phosphorus reabsorption. 19 Serum inorganic phosphorus was increased in both sexes in the 700 mg/kg/d group of the 13- and 26-week rat studies. Chronically elevated blood levels of phosphate are associated with ectopic mineralization even in the face of normal plasma calcium levels, both by passive deposition as the consequence of an unfavorable calcium–phosphate product, but also by inducing the expression of proteins responsible for active bone mineral deposition in cells of the vasculature and in other soft tissues such as the renal tubules. 20 In an effort to maintain calcium and phosphorus homeostasis, increased urinary excretion of calcium and/or phosphorus ensues, which can then precipitate in renal tubules forming foci of mineralization. Significant changes in trabecular bone accretion were observed in studies of canagliflozin and dapagliflozin that were similarly attributed to off target SGLT1 inhibition. 9,21,22
The tumors observed in the carcinogenicity studies are attributable to nongenotoxic, epigenetic mechanisms having little or no relevance for human safety. The renal neoplasms in male mice given 1000 mg/kg/d were considered secondary to chronic and persistent tubular degeneration, necrosis, and regeneration. The renal tumors were generally localized in the cortical and/or outer medullary tissue and were not identified at necropsy. Only the large fatal carcinoma of 1 animal involved an entire kidney and was detected at necropsy. Renal neoplasms noted in males given 1000 mg/kg/d generally occurred in association with relatively severe chronic degenerative tubular changes evidenced by tubular cysts, atrophy, and cystic and atypical hyperplasia. Consequently, these tumors were interpreted as secondary to chronic and persistent proximal convoluted tubular injury, single cell necrosis, and associated regeneration previously described. The low incidence, limitation to 1 sex, and the late onset/detection, as evidenced by the small size of most neoplasms, are further evidence for the epigenetic development of these neoplasms. 23 In the 13-week mouse study, pathological findings indicating degeneration and regeneration were noted in males at dose levels greater than 500 mg/kg/d, and an immunohistochemical evaluation using Ki-67 antibody revealed enhanced tubular cell proliferation in males at ≥750 mg/kg/d. This profile is largely consistent with that of other renal carcinogens having a cytotoxic mode of action followed by sustained reparative, or compensatory, cell proliferation. 24 Investigative studies supporting this mode of action have been completed and are being prepared for publication. 15
In the rat carcinogenicity study with empagliflozin, the increased incidence of interstitial cell tumors is considered secondary to the profound decreased body weight gain in treated males, a known cause of interstitial cell tumors in this Wistar rat strain. 25,26 The effects can be seen with even modest (10%-15%) decrements in terminal body weight due to caloric restriction and are most pronounced when restrictions are imposed early (within the first 13 weeks of life). 27 In the carcinogenicity study, mean body weight gain was 29%, 34%, and 42% lower for males given empagliflozin doses of 100, 300, or 700 mg/kg/d, respectively, than that of the combined control males. Increase in interstitial cell tumors has also been observed with acarbose and lactitol, both of which cause malabsorption of glucose and a similar relative energy deficit in the presence of an increase in food consumption. 16,26
Although prolactin levels were not measured in the rat toxicology studies, evidence from the published literature supports a primary role for chronically decreased serum prolactin as the primary mechanism for the increase in interstitial cell tumors resulting from empagliflozin-induced caloric restriction. Dopamine signaling mediates gonadotropin-releasing hormone release from the hypothalamus, which results in increased secretion of follicle-stimulating hormone and leutinizing hormone (LH) and decreased secretion of prolactin. 28 Prolactin is necessary for the maintenance of interstitial cell LH receptors in rats, and prolactin receptors are abundantly expressed. Within 7 days of caloric restriction in rats, circulating prolactin levels decrease by altering the level of dopamine-mediated neurotransmission in the hypothalamic nuclei. 29 Lifetime caloric restriction slows the rate of loss of dopaminergic neurons in the hypothalamic nuclei causing sustained depression of prolactin and sustained increases in LH. The differences in dopamine receptor levels become significant at 12 months of age and by 24 months of age animals with lifetime caloric restriction have retained twice the number of dopaminergic receptors as ad libitum fed animals. 30 When prolactin is deficient, interstitial cell LH receptor numbers decline, which results in decreased testosterone synthesis and feedback stimulation of hypothalamic LH secretion. Leutinizing hormone has a mitogenic effect on interstitial cells, and chronically sustained interstitial cell proliferation leads to increased risk of neoplasia. Finally, the incidence of mammary gland tumors, which are known to be prolactin dependent in rats, was decreased in a dose-dependent manner. 31,32 Importantly, although prolactin receptors are expressed abundantly on rat interstitial cells, they are absent on human interstitial cells. 31,33 -35 Thus, the permissive effect of caloric restriction on interstitial cell tumor production observed in rats is not relevant for humans.
Hemangiomas of the mesenteric lymph nodes of rats appear to be a species specific and relatively common spontaneous neoplasm, especially in Wistar Han rats where they appear more commonly in males than females. The background incidence of these tumors is highly variable and tends to drift over time, with reported incidences in males ranging from 0% to as high as 26%. 7,36 -39 Several factors contributing to a male-specific increased incidence in susceptible rat strains have been proposed including genetic drift, congenital/developmental malformation, iron overload, obstruction of vascular outflow, and chronic antigenic stimulation. 40,41 Findings supportive of these mechanisms were detected in the mesenteric lymph nodes in the empagliflozin carcinogenicity study in rats.
Empagliflozin-related changes indicative of vascular obstruction (sinus erythrocytes) and hemolysis, hemosiderosis, and iron overload (pigmented macrophages, indicating erythrocyte breakdown with generation of hemosiderin) were present at an increased incidence in the mesenteric lymph node in males given empagliflozin at ≥100 mg/kg/d, with a trend toward increased severity at ≥300 mg/kg/d. Changes indicative of chronic antigenic stimulation (increased sinus histiocytes and/or mast cells) were also present at an increased incidence in the mesenteric lymph node in males given empagliflozin at ≥100 mg/kg/d, with a trend toward increased severity at ≥300 mg/kg/d. Although these findings were present in the mesenteric lymph nodes in females, empagliflozin-related increases in incidence and/or severity were only apparent at 700 mg/kg/d.
As in other studies where increased incidences of hemangiomas of mesenteric lymph nodes were observed in rats, there was not an increased incidence of vascular tumors either at other lymph node sites or in other tissues nor was there any increased incidence of hemangiosarcomas. In addition, there were no vascular lesions associated with treatment in male or female mice or in male or female dogs. Thus, it is a single species, single gender lesion with a high background in the affected species and gender.
Hemangiomas in human lymph nodes are extremely rare, including in the mesenteric lymph nodes. 41 -43 Considerable evidence is available to suggest that vascular tumors in rodents are distinctly different from those that occur in humans. In rodents, benign and malignant vascular tumors appear to arise from the same precursor endothelial cells, regardless of the tissue in which the lesion arises and regardless of whether the lesion is spontaneous or induced by treatment with a nongenotoxic or genotoxic chemical. 44 In contrast, hemangiomas in humans appear to arise from mature endothelial cells, expressing markers of endothelial cell differentiation but not markers of myeloid or hemangioblast differentiation. 45 Based on such observations and their clinical behavior, many pathologists and clinicians have classified hemangiomas as hamartomas (developmental anomalies) rather than as true neoplasms. 46 They do not evolve into malignancies, regardless of their location. Taken together the increased incidence of hemangioma of the mesenteric lymph node in male rats of the 700 mg/kg/d dose group is a finding that is considered of low relevance to human safety.
Although 3 of the currently marketed SGLT-2 inhibitors induce renal proliferative toxicity at high doses, the species specificity varies (data on other inhibitors, ipragliflozin, tofogliflozin, and luseogliflozin, are not available). In the case of dapagliflozin and canagliflozin, these proliferative effects appear in the rat; these compounds did not induce renal tumors in the CD-1 mouse. An increased incidence and severity of atypical hyperplasia of the renal tubules were observed in male Sprague-Dawley rats treated with dapagliflozin, while renal tubular adenoma/carcinoma was observed in male and female Sprague-Dawley rats treated with canagliflozin. 8,9 No renal proliferative effects of empagliflozin were observed in the Wistar Han rat, suggesting an empagliflozin, male-mouse specific mode of action.
In conclusion, the general pharmacology, general toxicology, genetic toxicology, and carcinogenicity data demonstrate an acceptable safety profile for empagliflozin. The range of high-dose toxicities observed in the general toxicology studies is consistent with the class of SGLT-2 inhibitors. The male mouse-specific renal tumors have not been observed with other SGLT-2 inhibitors, which suggests a mode of action unique to empagliflozin. Studies to investigate this possibility are complete and will be published.
Footnotes
Acknowledgment
Boehringer Ingelheim Pharmaceuticals, Inc funded this research.
Author Contributions
Matthew Bogdanffy contributed to conception and design, analysis and interpretation, drafted the article, critically revised the article, gave final approval, and, agrees to be accountable for all aspects of work ensuring integrity and accuracy. Robert Stachlewitz, Susan van Tongeren Brian Knight, Dale Sharp, and Warren Ku contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy. Susan Emeigh Hart contributed to, interpretation, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Kerry Blanchard contributed to conception and design, interpretation, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. When the data were generated, all authors were employed by Boehringer Ingelheim Pharmaceuticals, Inc, which manufactures and sells empagliflozin.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
