Abstract
The efficacy of stem cell transplantation for promoting recovery of patients with neurological diseases, such as stroke, has been reported in several studies. However, the safety of the intracerebral transplantation of human mesenchymal stem cells (hMSCs) remains unclear. The aim of the study was to evaluate the safety of hMSCs transplanted in cerebrum of Macaca fascicularis and to provide evidence for clinical application. A total of 24 M fascicularis were assigned to 3 groups randomly: low dose (3.0 × 105 cells/kg), high dose (2.5 × 106 cells/kg), and the control (normal saline [NS]). Human mesenchymal stem cells or NS were injected into each monkey for 2 times, with an interval of 3 weeks. The injection point was located outside of the right putamen, according to a stereotactic map and preoperative magnetic resonance imaging of the monkeys. Animal health, behavior, biophysical and biochemical parameters, and brain neurological function were routinely monitored over a 6-month period posttransplantation, and the histopathologic examinations were also performed. The results showed that local pathologic damage including local tissue necrosis and inflammation was induced after the injection. The damage of low-dose and high-dose groups was greater than that of the control group, yet over time, the damage could be repaired gradually. No major hMSCs-associated changes were induced from other indicators, and the transplantation of hMSCs in monkeys did not affect total immunoglobulin (Ig) M, total IgG, CD3, CD4, or CD8 values. We therefore conclude that transplantation of hMSCs to the cerebrum represents a safe alternative for clinical application of neurological disorders.
Introduction
Cerebrovascular disease (CVD) is a common and life-threatening disease that poses a significant public health challenge. He et al 1 reported in 2005 that CVD ranked the third among 5 leading causes of death in men and ranked the second in women. A multicenter research in China indicated that CVD was the most prevalent cause of deaths, accounting for 40% of total deaths. However, there remain a limited number of effective therapies for CVD, and patients still had continued deterioration of neurological function following treatment. 2 –6 Consequently, it is important to develop novel therapies that can limit the extent of neurological damage and improve functional recovery.
Human mesenchymal stem cells (hMSCs), usually obtained from bone marrow, can differentiate into a variety of cell types and can facilitate damage repair and structural and functional repair of aged organ. Related studies have shown that hMSCs-based therapies promote recovery of motor function in stroke mice model 7 and improved neurologic function following induced stroke. 8 These findings have been confirmed and extended by many other animal studies. 9 –13 Stem cell treatment has also shown benefits in our prior studies using experimental intracerebral hemorrhage models in rodents and primates. 11 –13 These properties, together with the fact that MSCs can be readily obtained, rapidly expanded in vitro, and has fewer ethical disputes, make them a promising therapeutic candidates to CVD.
However, the safety of intracerebral hMSCs transplantation is still unclear; thus, it is urgent to explore this approach in a clinically relevant model. 14 The goal of this study was to evaluate the long-term toxicity associated with repeated intracerebral hMSCs transplantation in nonhuman primates (Macaca fascicularis).
Materials and Methods
Animals and Housing
A total of 24 M fascicularis (12 male and 12 female; aged 3 to 6 years; weight 4.1 ± 0.5 kg) were received from the Experimental Animal Center of the Chinese Academy of Military Medical Sciences. All monkeys were kept single in stainless steel cage at 18°C to 28°C and a relative humidity of 40% to 70%. The primate facility is licensed and accredited by the Association for Assessment and Accreditation of Laboratory Animal Care, and all procedures were approved by the Institutional Animal Care and Use Committee of the Chinese Academy of Medical Sciences (MC-07-6004). Animal experiments and housing procedures were carried out in accordance with the laboratory animal administration rules of the Ministry of Science and Technology of the People’s Republic of China.
Preparation of the HMSCs
Bone marrow was collected from healthy adult human volunteers. All participants signed a declaration of informed consent before surgery. The hMSCs were isolated and cultured using a technique that has been detailed in a previous report.
15
Briefly, mononuclear cells were isolated from marrow aspirates with Ficoll-Paque density gradient centrifugation. Cells were counted and cultured in Dulbecco Modified Eagle Medium: Nutrient Mixture F-12 (Gibco Co, Los Angeles) with 2% fetal bovine serum (Hyclone Co, Utah), 2 mmol/L
When reached 80% to 90% confluence, the cells were digested with 0.125% trypsin and replated at a 1:2 dilution under the same culture conditions, then were collected and prepared for transplantation.
Grouping
Following 2 weeks of acclimatization, prestudy examination was done to ensure every monkey was healthy. Then, the monkeys were assigned to 3 groups randomly according to body weight: low dose (3.0 × 105 cells/kg), high dose (2.5 × 106 cells/kg), and the control (phosphate-buffered saline), and the dose is 2.1 and 17.5 times to intended clinical dose (1 × 107 cells/person, the body weight of one person is assumed to be 70 kg), respectively.
Dosing
The animals were fasted overnight prior to surgery. Ketamine (15 mg/kg) and sodium pentobarbital (30 mg/kg) were administered intramuscularly (IM) for the duration of the procedure. Then, the monkeys were placed in a stereotactic frame (Stoelting Co, Illinois) and set the origin coordinates according to a stereotactic map and preoperative magnetic resonance imaging (MRI) scans. The coordinates were recorded and the coordinate origin was set. Generally, the coordinates were AP 16 mm, D 16 mm, and L 14 mm, whereby AP represents the anterior and posterior directions, D represents the depth, and L represents the lateral coordinates. The injection point was located outside of the right putamen, according to a stereotactic map and preoperative MRI of the monkeys. The cells were delivered through 2 injection channels located 1.5 mm apart, and each channel contained 3 injection points spaced 1 mm apart. A total volume of 250 µL was administered at a rate of 40 to 50 μL/min. After a 3-week interval, animals were administered a second round of injections using the same procedure. Penicillin (400 000 units per day, IM) was administered for 3 days after surgery.
General Observations and Examinations
Animals were observed daily following surgery. Skin and fur appearance, vital signs, secretions, upper and lower limb movements, consciousness, and injection region reactions were assessed at 1, 2, and 3 days after cell administration.
Neurological Function Evaluation
Each monkey was subjected to neurological function tests at regular time intervals (see Table 1). Scores for neurological function were refined and modified from the Kito score scale and also referenced the “Neurological Scale for Middle Cerebral Artery Infarction” (Table 2). The scale includes a total of 100 scores, including 28 scores for awareness, 22 scores for the sensory system, 32 scores for the motor system, and 18 scores for the coordination of skeletal muscles. 11,16,17
Examinations After hMSCs Transplantation.

Abbreviations: Ig, immunoglobulin; hMSCs, human mesenchymal stem cells.
Neurological Deficit Score for Monkeys After Stroke.

Animal Health Parameters Evaluation
To assess the adverse effects produced by hMSCs, the health parameters of each recipient were monitored on a regular basis (see Table 1).
Immunology Evaluation
At regular intervals, blood was taken from brachial vein of the front limb to examine the immunology parameters. The IgG and IgM of the serum were determined with fully automatic biochemical analyzer (HITACHI 7020, Japan). The CD3, CD4, and CD8 of the K2EDTA anticoagulated blood were determined with flow cytometry (FACSCalibur, U.S.A BD Co; see Table 1).
Cerebrospinal Fluid Evaluation
At regular intervals, the cerebrospinal fluid was extracted to examine the related parameters. The color and transparency of the cerebrospinal fluid were observed with naked eyes. The cells of the cerebrospinal fluid were counted with microscope. The content of protein, glucose, and chloride of the cerebrospinal fluid was determined with fully automatic biochemical analyzer (HITACHI 7020, Japan) and potassium sodium and chloride analyzer (Roche 9180, Switzerland Roche co; see Table 1).
Pathological Evaluation
At the fifth week after hMSCs transplantation, 4 monkeys of each group were euthanized and at the 15th and 27th week after hMSCs transplantation, 2 monkeys of each group were euthanized to understand the recovery of possible adverse effect and possible delayed effect. Schematic diagram (Figure 1) of brain dissection illustrated how the brain from individual transplant recipients was segregated into 3- (gray) or 6-mm (white) coronal slices, which are numbered 1 to 13. 18 The slices were embedded in paraffin for slicing, and the slice thickness was 5 μm. The slices were stained with hematoxylin and eosin and observed under a light microscope. Necropsy and histological examinations including 33 organs of the monkeys were performed. Major organs and glands were weighed.
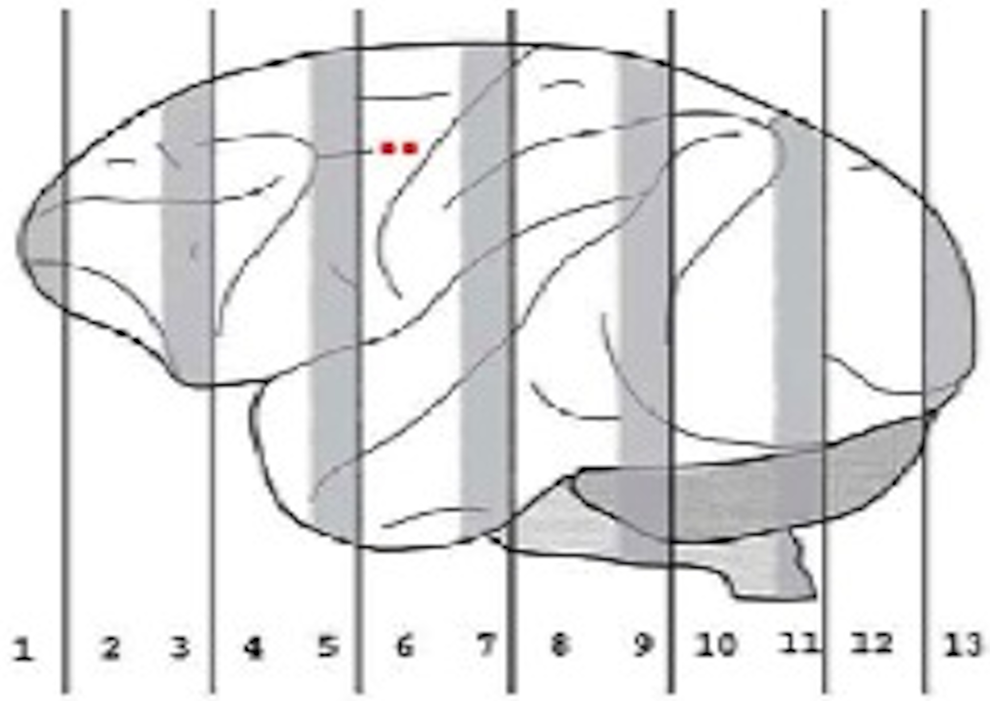
Schematic illustrating how the brain from individual transplant recipients was segregated into 3- (gray) or 6-mm (white) coronal slices, which are numbered 1to13.
Statistical Analysis
All computations were carried out using SPSS 13.0 (SPSS Inc., Chicago, Illinois, USA). The data were analyzed by analysis of variance with post hoc analysis. Differences were determined to be significant with P < 0.05.
Results
Clinical Observation, Body Temperature, Body Weight, and Food Intake
No adverse effects on animal behavior were observed throughout the study. All animals appeared to be healthy and exhibited normal respiration and gross motor coordination; no changes on body secretions (urine and feces) were noted. The body temperature of all animals fluctuated between 37.5°C and 39.5°C over the course of the study which is normal temperature range of M fascicularis.
A decrease in food and water intake during the first 3 days after surgery was observed in all of the groups. This change likely attributed to the altered feeding regimens following surgical procedures to ensure adequate nutrition intake. The food and water intake subsequently returned to baseline level. Body weight increased steadily in all monkeys over the study, and no significant differences were found in the 3 groups (P > 0.05). Electrocardiogram functions, including heart rate, PR interval, QRS interval, QT interval, and QTc, were all within the normal range. No significant differences were observed in all groups (P > 0.05).
There were no neurological deficits observed in any of the groups, with neurological deficit scores of zero pre- and postoperation (data not shown).
Hematology and Blood Biochemistry
No hMSCs related adverse effects were observed in hematology and blood biochemistry parameters at the study. Compared to the control group, the mean corpuscular hemoglobin concentration of the low-dose group and high-dose group increased significantly at the third week after hMSCs transplantation (P < 0.05 and P < .01, respectively); the prothrombin time of the high-dose group increased significantly at the second week after hMSCs transplantation (P < .05); serum chloride concentration of the high-dose group increased significantly at the eighth week after hMSC transplantation (P < .05). Yet they were all within the normal reference levels and were not considered biologically significant.
Cellular Immunity, Humoral Immunity, and Cerebrospinal Fluid Examination
Compared with the control group, no significant differences were observed for the IgG and IgM content in the treated groups after the hMSCs transplantation (P > 0.05). No significant changes were observed for the cellular immunity parameters (CD3+, CD8+, CD4+, and CD4+/CD8+) at all time point (P > 0.05) except the CD4 content of the high-dose group increased significantly compared to the control group at the second week after the hMSCs transplantation (P < 0.05, Figures 2 –5).
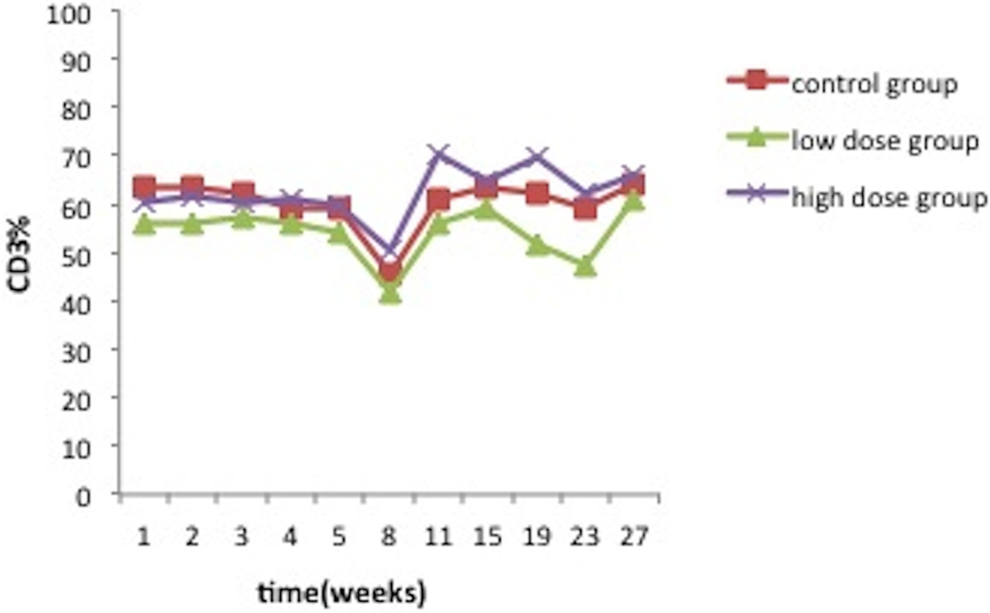
No significant changes were observed for the cellular immunity parameters (CD3, CD8, CD4, CD4/CD8) at all time point (P > 0.05) except the CD4 content of the high-dose group increased significantly compared to the control group at the second week after the hMSCs transplantation (P < .05).
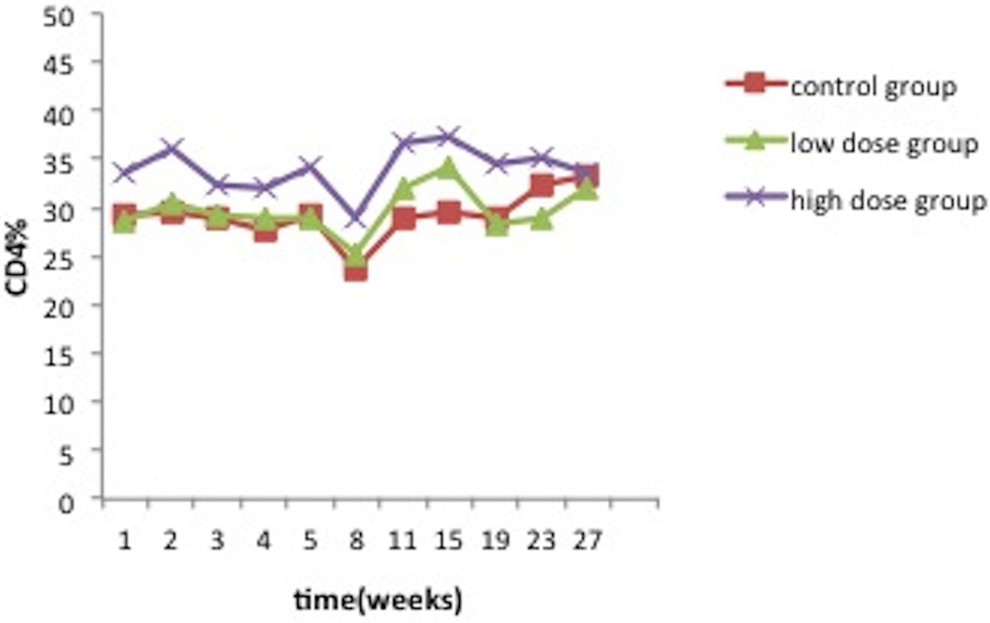
No significant changes were observed for the cellular immunity parameters (CD3, CD8, CD4, CD4/CD8) at all time point (P > 0.05) except the CD4 content of the high-dose group increased significantly compared to the control group at the second week after the hMSCs transplantation (P < .05).
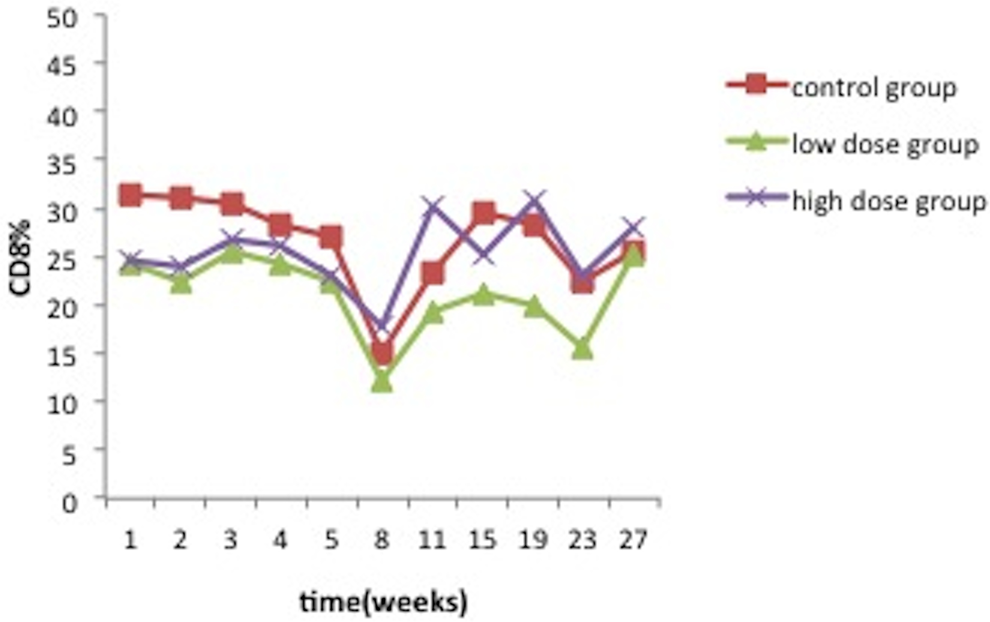
No significant changes were observed for the cellular immunity parameters (CD3, CD8, CD4, CD4/CD8) at all time point (P > 0.05) except the CD4 content of the high-dose group increased significantly compared to the control group at the second week after the hMSCs transplantation (P < .05).
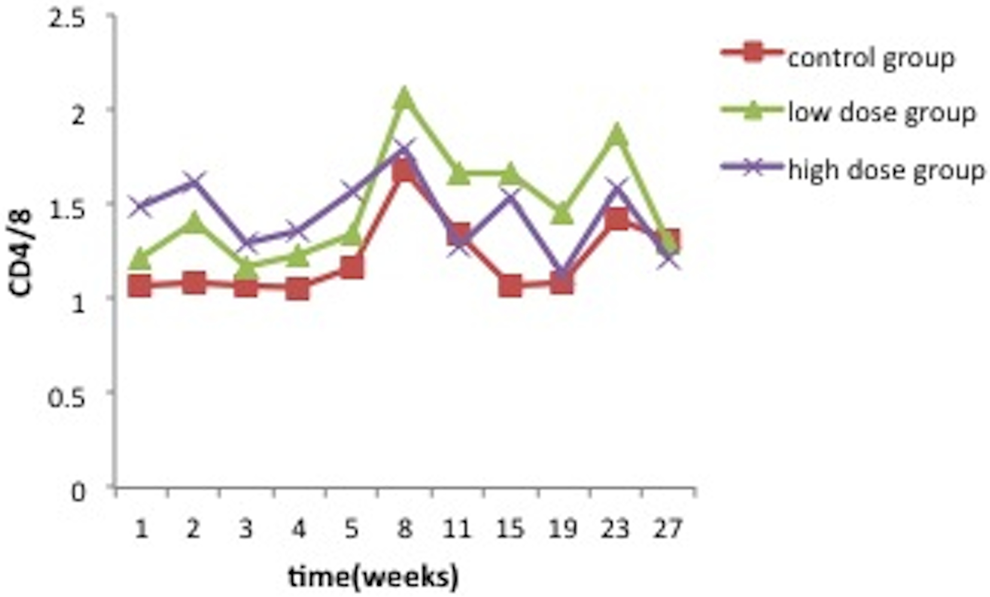
No significant changes were observed for the cellular immunity parameters (CD3, CD8, CD4, CD4/CD8) at all time point (P > 0.05) except the CD4 content of the high-dose group increased significantly compared to the control group at the second week after the hMSCs transplantation (P < .05).
The appearance of the cerebrospinal fluid is transparent and colorless, and no differences were observed with respect to the content of protein, glucose, and chloride of the cerebrospinal fluid in all groups at various time intervals of evaluation.
Organ Weight and Weight Ratio
No significant differences for the organ weight and weight ratio were measured in the 3 groups (P > 0.05). The testis weight and weight ratio of 1 animal of the high-dose group decreased at the fifth week after hMSCs transplantation. The testis weight and weight ratio of 1 animal of high-dose group increased at the 27th week after hMSCs transplantation.
Necropsy and Histopathology
Necropsy observation conducted at 5th, 15th, and 27th week posttransplantation demonstrated a few findings that were not hMSCs related. The following are the detailed individual macroscopic observations. At the fifth week posttransplantation, the color of a small area of the edge of the right liver lobe of animal No. 9 (♀, low-dose group) turns yellow. At the 15th week posttransplantation, a few white cyst appeared outside the intestinal wall of animal No. 7 (♂, control group) and the biggest of which is 0.8 × 0.6 × 0.3 cm3, and a few red vesicular complexes whose surface are smooth appeared on the ileocecal mucosal surface of animal No. 17 (♀, high-dose group). At the 27th week posttransplantation, a notch appeared at the lower pole of the left kidney of animal No. 4 (♀, low-dose group), and a few gray white dots whose diameters are 0.1 to 0.3 cm appeared on the liver surface of animal No.15 (♂, low-dose group). No abnormality was discovered for the other animals.
The histopathology changes that attributed to the hMSCs were the pathological injury of the injection area of the cerebrum.
The cannula injection channel was verified by histological examination for each animal for proper placement within the intended region. At the fifth week posttransplantation, necrosis was observed around the injection site in the brains of the control animals, and a proliferation of hemoglobin-containing foam cells was noted. In the low-dose group, spindle-shaped tissue necrosis and infiltration of inflammatory cells including foam cells and eosinophils of the injection sites were observed, and vessel congestion and vascular cuff around the necrotic site was also observed. In the high-dose group, a larger area of tissue necrosis with granulosis formation was observed around the injection site. Vascular cuff made up of perivascular lymphocytes and eosinophils formed (Figure 6). At the 15th week posttransplantation, in the control group, a needle-like glial scar with hemoglobin-containing foam cell formed in the injection site of the cerebrum. In the low-dose group, local bands of scar tissue was observed with a proliferation of hemoglobin-containing foam cells. In the high-dose group (No. 17), a larger area of tissue necrosis and granulation tissue formed in the injection site. In the necrosis area, inflammatory cells such as foam cells and eosinophils were observed yet the surrounding tissue remained intact and without edema. The cerebrum of the other monkey (No. 24) in the high-dose group revealed bands of scar tissue in the injection site, with a proliferation of hemoglobin-containing foam cells (Figure 7). At the 27th week posttransplantation, the injection sites in all 3 groups exhibited only a needle-like scar formation, with a hemoglobin-containing foam cell proliferation (Figure 8).
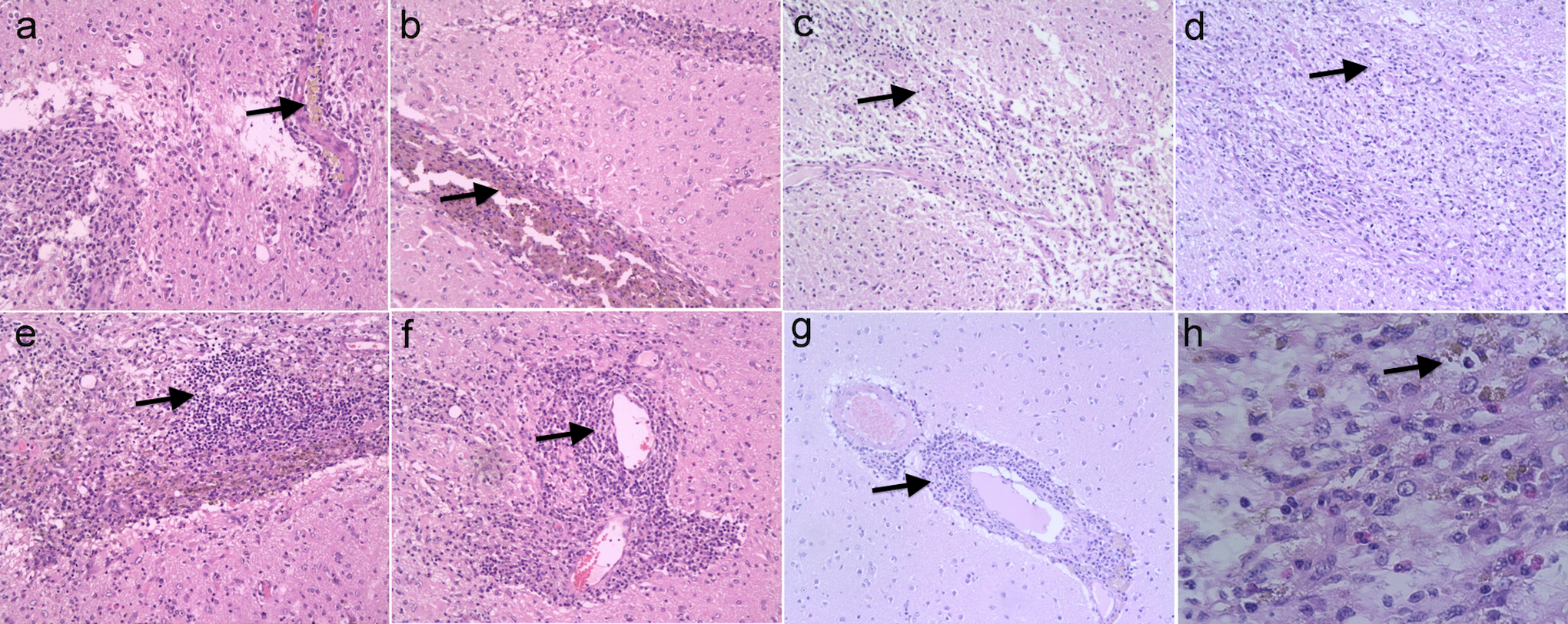
At 5 weeks posttransplant, necrosis was observed around the injection site of the control, and a proliferation of hemoglobin-containing foam cells was noted (A). In the low-dose group, the neural injection sites exhibited tissue necrosis, with infiltration of inflammatory cells (B), including foam cells and eosinophils and vessel congestion around the necrotic site and the blood vessel (C and D). In the high-dose group, a larger area of tissue necrosis with granulosis formation was observed around the injection site (E, F, and H). Perivascular lymphocytes and eosinophils formed around the vascular cuff (G). Control group (A ×100); low-dose group (B, C, and D ×100); high-dose group (E, F, and G ×100; h ×400).
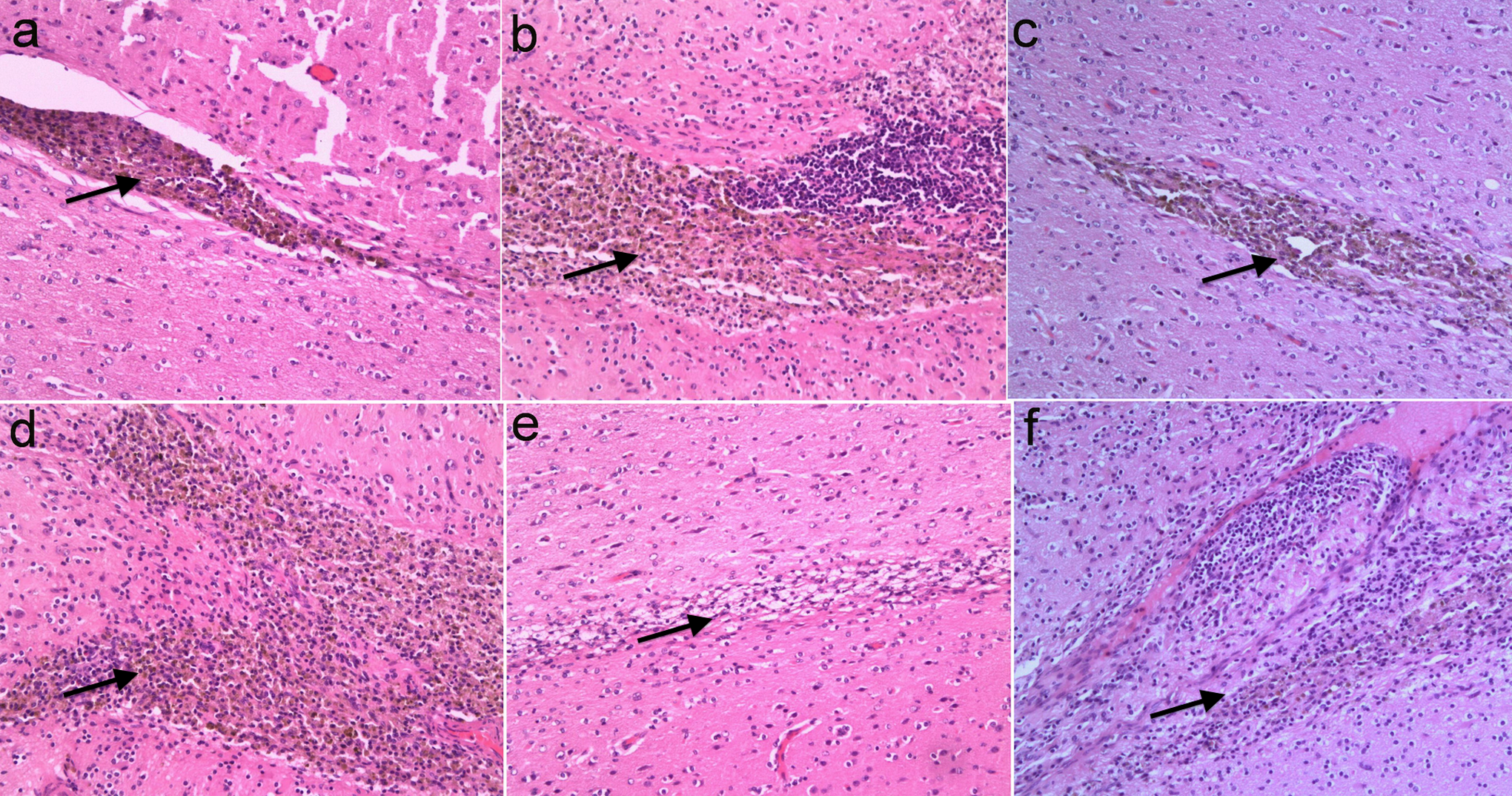
At 15 weeks posttransplant, the injection site of the control animal exhibited a needle-like glial scar with hemoglobin-containing foam cell formation (A). In the high-dose group (E and F) and low-dose group (B, C, and D), hemoglobin-containing foam cells and lymphocytes were observed. Control group (A ×100); low-dose group (B, C, and D ×100); high-dose group (E and F ×100).
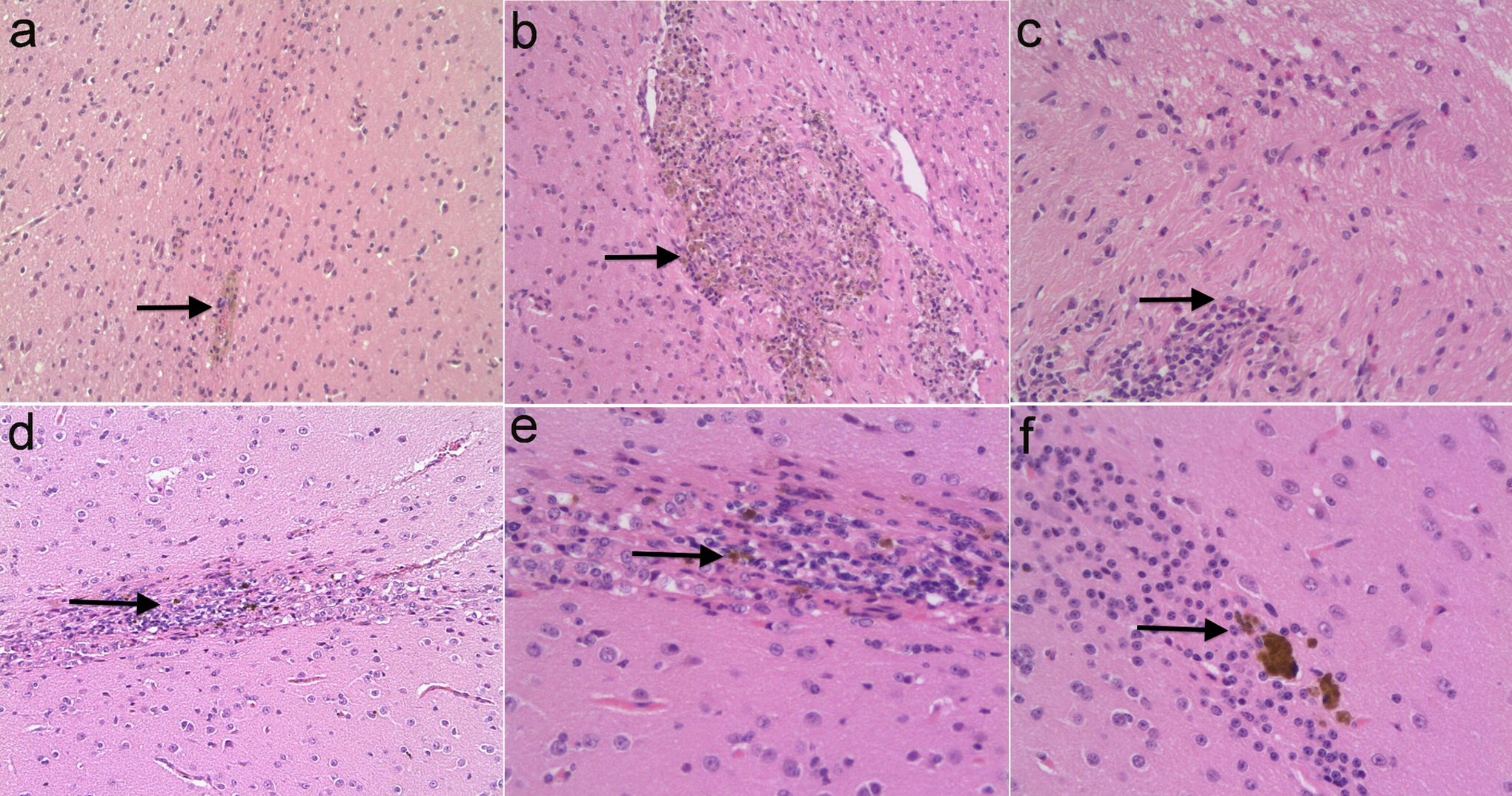
At 27 weeks posttransplant, the neural injection sites in all 3 treatment groups exhibited only a needle-like scar formation, with a hemoglobin-containing foam cell proliferation. In the low-dose group, the neural injection sites exhibited with inflammatory cells (C), including eosinophils. In the high-dose group, a proliferation of glial cells and pigmentum deposition were observed around the injection site (E and F). Control group (A and B ×100); low-dose group (C ×100); high-dose group (D ×100; E ×200; f ×200).
Histopathological changes not relevant to the hMSCs transplantation were as follows. At the 15th week posttransplantation, multiple cysts were found on jejunum serosal surface of animal No. 7 (♂, control group), and chronic inflammation was found on the ileocecal mucosal surface of animal No. 17 (♀, high-dose group). At the 27th week posttransplantation, chronic pyelonephritis was found at 2 sides of animal No. 4 (♀, low-dose group), local inflammatory cells infiltration were found at renal interstitium of animal No.18 (♀, high-dose group), and a few eosinophilic abscess formed on the liver of animal No.15 (♂, low-dose group).
Discussion
Human mesenchymal stem cells are nonhematopoietic stem cells in bone marrow. It has multidifferentiation potential, which can differentiate crossing blastoderm into parenchymal cells such as osteocytes, chondrocytes, adipocytes, and so on in proper induction condition. At the same time, it has immunomodulatory property and can modulate the bioactivity of immune cells through various ways. According to the unique properties, more attention should be paid to the effect of hMSCs on the immune system and tumorigenesis induction.
In our study, detailed histopathological examination including 32 organs of the monkeys was done to explore the tumorigenesis possibility of hMSCs; the count of CD3+, CD4+, and CD8+ lymphocytes in peripheral blood and the content of IgG and IgM in serum of the monkeys were examined to explore the immunomodulatory property; the neurological function and cerebrospinal fluid of the monkeys were examined considering the intracerebral injection method.
The results showed no notable changes in common symptoms, body weight, body temperature, hematology, blood biochemistry, electrocardiogram, routine urinalysis, and cerebrospinal fluid were evident in any monkey after transplantation. The transplantation of hMSCs did not induce neurological function injury or tumorigenesis. Fluorescence-activated cell sorting analysis also did not reveal any significant change in the count of CD3+, CD4+, CD8+, and CD4+/CD8+ at any time examined except the CD4+ of high-dose group increased significantly compared with the control group at the second week after hMSCs transplantation (P < 0.05), yet it was within normal limits. Liu 18 also reports the intracerebral injection of allogeneic MSCs had no significant adverse effect on the immune system of monkeys which coincide with our results. we speculate this may be attributed to the low immunity microenvironment of the cerebrum.
The change in the testis weight and weight ratio of particular monkey was discovered, yet the histopathological examination revealed no abnormality. In this study, the monkeys were killed at the age of 3 to 4 years when the testes were in the peak stage of development. Therefore, the change in testis weight and weight ratio was likely due to the development period of the monkey and which have no relationship with hMSCs transplantation.
The histopathologic results showed that local tissue necrosis and inflammation appeared in the injection area of cerebrum. The damage of high-dose and low-dose groups was greater than that of the control group, yet over time, the damage could be repaired gradually. The damage of high-dose group and low-dose group was greater than that of the control group which may be related to these reasons: (1) the rejection reaction of normal brain tissue to hMSCs which induced the local infiltration of inflammatory cells such as foam cells and eosinophils and (2) the death of hMSCs caused by the change in living microenvironment induced greater proliferation of foam cells than that of the control group.
At present, the preclinical safety evaluation of hMSCs is still less. Our study systemically evaluated the safety of hMSCs transplantation in the cerebrum. The results are particularly relevant in that the dosage of administered hMSCs was calculated based on the clinical dose planned for use in treating patients. Moreover, each animal was subjected to 2 rounds of cell transplantation, which may be required clinically to achieve a substantial and sustained benefit. The results showed under the dose of 2. 5 × 106/kg, human BMSC injected in capsula externa region of basal ganglia only induced local pathologic damage of the injection site of cerebrum, and the damage could be repaired gradually which indicates that intracerebral hMSC transplantation in nonhuman primates is safe and produces no observable tumors up to 6 months posttransplantation.
Conclusion
In conclusion, we have demonstrated that the repeated delivery of hMSCs at clinically relevant doses to the basal ganglia region in nonhuman primates is safe. Although some pathological changes were observed in the area of transplantation, these did not result in any neurological deficits or systemic side effects. These data support the use of hMSCs in clinical trials involving treatments for cerebral stroke.
Footnotes
Acknowledgments
The authors acknowledge Professor Zhaohui Zhu, Professor Wanchen Dou, Dr Xiaohai Liu, and Yu Wang for their advice and technical support.
Author Contribution
The first 5 authors contributed equally to this work. Ming Feng contributed to conception and design and gave final approval. Yan Li contributed to conception and gave final approval. Xinjie Bao contributed to design and gave final approval. Qin Han contributed to acquisition, analysis, and interpretation, and gave final approval. Qin Li contributed to analysis and gave final approval. Hua Zhu contributed to analysis and gave final approval. Junji Wei contributed to acquisition and analysis, critically revised manuscript, and gave final approval. Ming Yang contributed to analysis, drafted manuscript, and gave final approval. Wenbin Ma contributed to acquisition and interpretation, critically revised manuscript, and gave final approval. Hong Gao contributed to analysis and gave final approval. Yihua An contributed to acquisition, critically revised manuscript, and gave final approval. Rober Chunhua Zhao contributed to acquisition and interpretation, critically revised manuscript, and gave final approval. Chuan Qin contributed to design, critically revised manuscript, and gave final approval. Renzhi Wang contributed to conception and design, critically revised manuscript, and gave final approval.
Author’s Note
Ming Yang is now in the National University Hospital of Singapore.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by grants from the “863 Project” of the Ministry of Science and Technology of the People’s Republic of China (2013AA020106), the National Natural Science Foundation of China (81100869), and the Youth Foundation of Peking Union Medical College Hospital.
