Abstract
The activities of different xenobiotic-metabolizing enzymes in liver subcellular fractions from Wistar rats exposed to a glyphosate (GLP)-based herbicide (Roundup full II) were evaluated in this work. Exposure to the herbicide triggered protective mechanisms against oxidative stress (increased glutathione peroxidase activity and total glutathione levels). Liver microsomes from both male and female rats exposed to the herbicide had lower (45%-54%, P < 0.01) hepatic cytochrome P450 (CYP) levels compared to their respective control animals. In female rats, the hepatic 7-ethoxycoumarin O-deethylase (a general CYP-dependent enzyme activity) was 57% higher (P < 0.05) in herbicide-exposed compared to control animals. Conversely, this enzyme activity was 58% lower (P < 0.05) in male rats receiving the herbicide. Lower (P < 0.05) 7-ethoxyresorufin O-deethlyase (EROD, CYP1A1/2 dependent) and oleandomycin triacetate (TAO) N-demethylase (CYP3A dependent) enzyme activities were observed in liver microsomes from exposed male rats. Conversely, in females receiving the herbicide, EROD increased (123%-168%, P < 0.05), whereas TAO N-demethylase did not change. A higher (158%-179%, P < 0.01) benzyloxyresorufin O-debenzylase (a CYP2B-dependent enzyme activity) activity was only observed in herbicide-exposed female rats. In herbicide-exposed rats, the hepatic S-oxidation of methimazole (flavin monooxygenase dependent) was 49% to 62% lower (P < 0.001), whereas the carbonyl reduction of menadione (a cytosolic carbonyl reductase-dependent activity) was higher (P < 0.05). Exposure to the herbicide had no effects on enzymatic activities dependent on carboxylesterases, glutathione transferases, and uridinediphospho-glucuronosyltransferases. This research demonstrated certain biochemical modifications after exposure to a GLP-based herbicide. Such modifications may affect the metabolic fate of different endobiotic and xenobiotic substances. The pharmacotoxicological significance of these findings remains to be clarified.
Introduction
Glyphosate [N-(phosphonomethyl) glycine] (GLP) is the active ingredient of many commercial formulations marketed as broad-spectrum herbicides and used worldwide for weed control. Its mode of action is related to the inhibition of an enzyme involved in the shikimate metabolic pathway, only present in plants and certain bacteria. Because this enzyme pathway does not occur in humans and animals, GLP is considered as a safe compound. Moreover, it has been shown that GLP does not bioaccumulate, biomagnify, or persist in a biologically available form in the environment. 1 –3 For these reasons, GLP is considered harmless for nontarget organisms living in the natural environment.
The use of GLP has globally expanded, associated with the production of genetically modified GLP-resistant crops and no-tillage practices. 4 Furthermore, GLP is employed to synchronize/accelerate the ripening of forage cereals, to control invasive species in the context of conservation practices, and as a defoliant in forestry and recreational areas. 3 Clearly, the extensive use of GLP-containing formulations increases agriculture efficiency and decreases the use of other herbicides more noxious to nontarget species or to the environment. However, the widespread and intensive use (or misuse in certain circumstances) of GLP-based herbicides might increase their environmental impact. In this regard, the presence of GLP, aminomethylphosphonic acid (AMPA, the major GLP metabolic product), and adjuvants of GLP formulations (ie, nonionic surfactants such as alcohol ethoxylates and alkylamine ethoxylates) in surface and groundwater appear to be of major concern. Surface water can be directly contaminated by GLP as a result of aquatic weed control or indirectly when the herbicide bound to soil particles is washed into rivers or water streams. On the other hand, GLP may reach aquatic habitats through inadvertent/unavoidable aerial overspray. 5 In this regard, several studies monitored the occurrence of GLP and AMPA in surface and groundwater of sowed areas. For instance, agricultural basins located in the southeast of Buenos Aires Province (Argentina) contained both GLP and AMPA in the surface water (from traces up to 4 µg/L) and its suspended and nonsuspended particulate matter (between 0.5 and 563 µg/kg). 6 Other investigation carried out within the nucleus area of soybean sowing in Argentina revealed concentrations of GLP in surface water between 100 and 700 µg/L 7 ; the latter is equal to the highest level of GLP allowed in water for human consumption. 8 Interestingly, traces of the most frequently used nonionic surfactants were also detected in ground and soil interstitial water from agricultural sprayed areas. 9 Consequently, the presence of GLP, AMPA, and other components of GLP formulations may impose some risk to nontarget species living in aquatic habitats, whereas human beings and other mammals could be exposed to these substances through the drinking water. In this respect, it has been shown that low concentrations of GLP may cause oxidative stress in fish when the herbicide is used for the control of emergent aquatic weeds in wetlands and margins of water bodies. 10 As far as mammalian species are considered, for example, increased production of hepatic thiobarbituric acid reactive substances (TBARSs) was observed in pregnant rats and their fetuses when the animals received a GLP formulation dissolved in the drinking water during the gestational period. 11 Similarly, in a previous work conducted in our laboratory, rats exposed to sublethal concentrations of GLP through the drinking water had increased levels of reduced glutathione (GSH) in the liver and enhanced glutathione peroxidase (GPx) activity in cytosolic fractions from liver, kidney, and gut mucosa. 12 Besides, long-term sublethal exposure to a GLP-based herbicide increased the release of hepatic aspartate aminotransferase and alanine aminotransferase and induced changes in hepatic liver tissue characterized by increased deposition of connective tissue and Kupffer cell proliferation. 13
The great majority of agrochemicals discharged into the environment may be metabolized by different xenobiotic-metabolizing enzymes (XMEs) located in mammalian tissues. Xenobiotic metabolism (or biotransformation) is a biphasic process, comprising phase 1 (oxidative, reductive, and hydrolytic) and phase 2 (conjugative) enzyme reactions taking place predominantly in the liver. Generally, the biotransformation of insecticides and herbicides yields less lipophilic, more polar, and hence less active metabolites readily eliminated from the body via normal excretion routes. In addition, long-term exposure to agrochemical compounds like organophosphates and fipronil affected the expression and functions of certain XMEs. 14,15 However, there is scant information concerning both the metabolic fate of GLP and its effects upon mammalian XMEs. For instance, rats intragastrically treated with this herbicide during 2 weeks had lower levels and reduced monooxygenase activities of enzymes belonging to the cytochrome P450 (CYP) system and a reduced activity of the intestinal aryl hydrocarbon hydroxylase. 16 More recently, it has been shown that GLP caused a potent inhibition of CYP2C9 activity in human liver microsomes. 15 Notwithstanding this previously published information, the effects of GLP on the expression and functions of different hepatic XMEs require further study.
This research is based on the possible occurrence of a GLP-based herbicide in the drinking water. As mentioned previously, herbicide formulations containing GLP consist of a mixture of the active principle plus adjuvants. These adjuvants include nonionic surfactants that enhance GLP stability and penetration into cells. Herbicide formulations containing ethoxylated surfactants were found more toxic than GLP itself in different mammalian cell lines. 17,18 Moreover, these authors highlighted that investigations on the toxic and/or biochemical effects of pesticides should be carried out by exposure to their mixtures rather than to the active principle alone. Thus, we hypothesized that in nontarget species, exposure to a GLP-based herbicide may induce changes in the activity of several hepatic XMEs. Thus, the experimental aim of this work was to evaluate the activities of different phase 1 and phase 2 XMEs in liver subcellular fractions obtained from rat exposed to a GLP-based herbicide formulation.
Materials and Methods
Chemicals
Glucose-6-phosphate and glucose-6-phosphate dehydrogenase were purchased from Roche Applied Science (Buenos Aires, Argentina). Methimazole (MTZ) was a generous gift from Gador S.A. (Argentina). Buffer salts (KCl, NaHCO3, Na2HPO4, NaH2PO4, K2HPO4, KH2PO4, and CH3COONH4) were purchased from Baker Inc (Phillipsburg, New Jersey), while all other chemicals were from Sigma-Aldrich Chemical Company (St Louis, Missouri).
Animals and Treatments
Healthy adult Wistar male (n = 13) and female (n = 13) rats weighing 328 ± 30 g were maintained under temperature controlled conditions (25°C) and a normal photoperiod of 12 hours of darkness and 12 hours of light. Animals of both sexes were randomly allocated into 3 groups. The first group (6 males and 6 females) remained untreated throughout all the experimental period and received water ad libitum. The second (3 females and 4 males) and third (3 males and 4 females) groups received a commercial formulation containing 662 mg/mL of GLP potassium salt (Round up full II) dissolved in the drinking water (ad libitum). Animals were exposed to the herbicide during 90 days. Based on the concentration of the active principle in the commercially available product, 2 sublethal concentrations of GLP were provided: (1) the highest GLP level (0.7 mg/L) allowed in water for human consumption 8 and (2) a 10-fold higher concentration (7 mg/L). Water consumption was monitored in each experimental group every day.
Animal procedures and management protocols were carried out according to internationally accepted animal welfare guidelines 19 and approved by the Animal Welfare Committee of the Faculty of Veterinary Medicine, Universidad Nacional del Centro de la Provincia de Buenos Aires, Tandil, Argentina (Internal Protocol: 06/11; approval date: November 10, 2011). At the end of the experimental period, rats were fasted overnight and then killed in agreement with these institutional and internationally accepted animal welfare guidelines.
Sample Collection and Preparation of Subcellular Fractions
After killing, the abdomen was opened and the liver removed. Liver was rinsed with ice-cold KCl 1.15%, covered with aluminum foils and immediately frozen, and stored in liquid N2 during 1 or 2 days until used for preparation of subcellular fractions. Liver tissue samples were weighed and placed into vessels filled with 2 volumes of ice cold homogenization buffer (20 mmol/L Tris, 150 mmol/L KCl, 1 mmol/L EDTA, pH 7.5). Tissue homogenization was performed with a Potter-Elvenjem tool (4-6 passes; Glas-Col, Indiana, USA). Homogenates were filtered through hydrophilic gauze, centrifuged at 10 000g for 20 minutes, and the resulting supernatant at 100 000g for 65 minutes. Aliquots of supernatants (cytosolic fractions) were frozen in liquid N2 and stored at −70°C. Pellets (microsomal preparations) were suspended in buffer Tris 10 mmol/L (containing 1 mmol/L EDTA and 20% glycerol), frozen in liquid N2, and stored at −70°C until used for incubation assays. An aliquot of each subcellular fraction was used to determine protein content using bovine serum albumin as a control standard. 20
Determination of Reduced and Oxidized Glutathione Content
Reduced (GSH) and oxidized (GSSG) glutathione levels were estimated in hepatic cytosolic fractions by the method of Hissin and Hilf. 21 Briefly, cytosolic fractions were diluted in a 0.1-mol/L phosphate buffer (pH = 8) containing 5 mmol/L EDTA. Then, 100 µL of 0.1% o-phthalaldehyde were added and GSH was fluorometrically measured at an excitation wavelength (λ) of 350 nm and a maximum of emission λ of 412 nm in a spectrofluorophotometer (Shimadzu RF-5301PC; Shimadzu Corporation, Kyoto, Japan). An aliquot of the diluted cytosolic preparation was treated with 40 mmol/L N-ethylmaleimide and then mixed with 0.1 mol/L NaOH. Then, 100 µL of a 0.1% o-phthalaldehyde solution were added to the mixture and GSSG was fluorometrically measured at an excitation λ of 350 nm and an emission λ of 584 nm. Known amounts of GSH or GSSG were added to the phosphate buffer to obtain calibration standards (0.156-2.5 µg/mL) by triplicate. These samples were treated and measured as explained previously. Then, calibration curves were constructed using the least squares linear regression analysis (Instat 3.00; Graph Pad Software, Inc, San Diego) of the intensity versus the nominal concentration of each calibration standard. The concentrations in the experimental samples were determined following interpolation of the intensity measured into the standard curves.
Measurement of CYP Content
The CYP content was determined as the carbon monoxide difference spectrum (450-490 nm) of sodium dithionite-reduced microsomal suspensions. 22
Enzyme Assays
All enzyme activities were measured under conditions yielding 0 order rates with respect to cofactor and substrate concentrations and ensuring linearity with respect to time and protein concentrations. Selenium-independent GPx activity, also referred to as glutathione S-transferase α (GSTα), was measured using cumene hydroperoxide as substrate following the method described by Nebbia et al. 23 Catalase (CAT) activity was assayed using the decomposition of hydrogen peroxide by the method of Aebi. 24 Carboxylesterase activity was assayed in microsomal fractions using 0.33 mmol/L p-nitrophenyl acetate as substrate, 0.015 mg of protein, and measuring the amount of indophenol released. 25
Metabolic rates of the in vitro biotransformation of the following CYP substrates were fluorometrically measured: 7-ethoxycoumarin (as a general substrate), 7-ethoxyresorufin and methoxyresorufin (for monitoring the activities of CYP1A1/2), benzyloxyresorufin (CYP2B), and oleandomycin triacetate (TAO; CYP3A). All incubations were performed at 37°C in the presence of a reduced nicotinamide adenine dinucleotide phosphate (NADPH) generating system (0.32 mmol/L NADP+, 6.4 mmol/L glucose-6-phosphate, 2.5 mmol/L MgCl2, 0.8 mmol/L EDTA, and 1.25 U of glucose-6-phosphate dehydrogenase in 0.1 mol/L Tris-HCl, pH 7.4). The enzyme activity 7-ethoxycoumarin O-deethylase (ECOD) was determined using 1 mmol/L substrate concentration and 0.2 mg/mL of microsomal protein following the method reported by Dent et al., 26 adapted to measure the amount of hydroxycoumarin formed using a continuous method (excitation λ: 385 and emission λ: 460). The O-dealkylation rates of 1 µmol/L 7-ethoxyresorufin O-deethlyase (EROD), 5 µmol/L methoxyresorufin O-debenzylase (MROD), and 2 µmol/L benzyloxyresorufin O-debenzylase (BROD) were assayed fluorometrically (excitation λ: 510; emission λ: 586) by measuring the amount of resorufin formed using a continuous method 27 in the presence of 0.25 mg/mL of microsomal protein. The oxidative CYP-dependent N-demethylation activity toward TAO was assayed following the incubation of this substrate (0.3 mmol/L) in the presence of the NADPH producing system and 1 mg/mL of microsomal protein. After a 15-minute incubation, the reaction was stopped with trichloroacetic acid (10%, w/v) and, after centrifugation, the amount of the released formaldehyde was determined fluorometrically (excitation λ: 410 and emission λ: 510) on an aliquot of the clear supernatant with Nash reagent as detailed by Werringloer. 28
Methimazole S-oxidation was selected as a flavin monooxygenase (FMO)-dependent pathway and assayed following the method described by Dixit and Roche 29 using 0.2 mg of microsomal protein. Briefly, the enzyme activity was measured at 37°C in the presence of 0.1 mmol/L NADPH, 0.06 mmol/L 5,5′-dithiobis nitrobenzoic acid, and 0.02 mmol/L dithiothreitol dissolved in 0.1 mol/L tricine buffer (pH 8.4) containing 1 mmol/L EDTA. The difference in absorbance between identical assay mixtures with and without MTZ (1 mmol/L) was monitored at 412 nm for 5 minutes in an UV–VIS spectrophotometer (T80+; PG Instruments Limited, Leicestershire, England). The cytosolic carbonyl reduction of menadione (MEN) was measured at 37°C, monitoring the NADPH oxidation at 340 nm for 5 minutes (continuous method) following a previously described method. 30
Glutathione S-transferase activities were monitored in cytosolic fractions by a continuous spectrophotometric method 31 using the following substrates: 1-chloro-2,4-dinitrobenzene (CDNB; nonspecific substrate), 1,2-dichloro-4-nitrobenzene (DCNB) for the isoenzyme GSTμ, and ethacrinic acid for GSTπ. Microsomal uridinediphospho-glucuronosyltransferase (UDPGT) activity was determined using 1-naphthol (50 µmol/L) as substrate and 0.4 mg of microsomal protein essentially as described by Bock et al. 32
Statistical Analysis
Data are expressed as mean ± standard deviation. A normality test was performed for testing whether the data were sampled from populations that follow Gaussian distributions. This assumption was tested using the Kolmogorov and Smirnov method. Initially, all biochemical parameters from male and female rats within control and herbicide-exposed groups were statistically compared by means of Student t tests with Welch correction (Instat 3.00; Graph Pad Software, Inc). Further statistical analysis among experimental groups was performed by means of parametric (1-way) analysis of variance followed by Tukey multiple-range test. For all statistical comparisons, P values less than 0.05 were considered significant.
Results
Except for CYP-dependent enzyme activities, there were no significant sex differences for all other parameters measured in both control and herbicide-exposed rats. Therefore, data from these biochemical determinations were handled and analyzed without gender discrimination.
The presence of a GLP-based herbicide formulation dissolved in the drinking water did not modify the pattern of water intake. Thus, control rats consumed 37.8 ± 6.22 mL of water/animal/d. Similarly, water intake in rats exposed to the herbicide at both GLP concentrations tested (0.7 and 7 mg/L) was 41.8 ± 6.58 and 42.0 ± 7.14 mL/animal/d, respectively. Based on animal’s body weight and the total amount of water ingested, the estimated daily doses of GLP were 0.09 ± 0.01 mg/kg/d (GLP 0.7 mg/L) and 0.90 ± 0.12 mg/kg/d (GLP 7 mg/L).
The measurement of different parameters indicative of oxidative stress in liver cytosolic fractions is summarized in Table 1. Total glutathione levels (GSH + GSSG) were 1.5-fold higher (P < 0.05) in the liver of rats exposed to the GLP-based herbicide compared to those observed in unexposed control animals. This observation was a consequence of an increased GSH content at both exposure levels (GLP 0.7 mg/L: 6.4-fold higher, P < 0.05; GLP 7 mg/L: 7.85-fold higher, P < 0.001), whereas GSSG levels remained unchanged (data not shown). Therefore, higher GSH/GSSG ratios were observed in rats exposed to the herbicide. Hepatic cytosolic fractions obtained from rats receiving the herbicide showed higher GPx metabolic rates (GLP 0.7 mg/L: 39% higher, P < 0.01; GLP 7 mg/L: 61% higher, P < 0.001) with respect to control animals. Oppositely, the CAT activity decreased in hepatic cytosolic fractions of exposed animals at both GLP exposure levels: 0.7 mg/L (46% lower, P < 0.01) and 7 mg/L (37% lower, P < 0.05).
Effect of Exposure to a Glyphosate-Based Herbicide Formulation on Parameters Indicative of Oxidative Stress Measured in Rat Liver Cytosolic Fractions.a
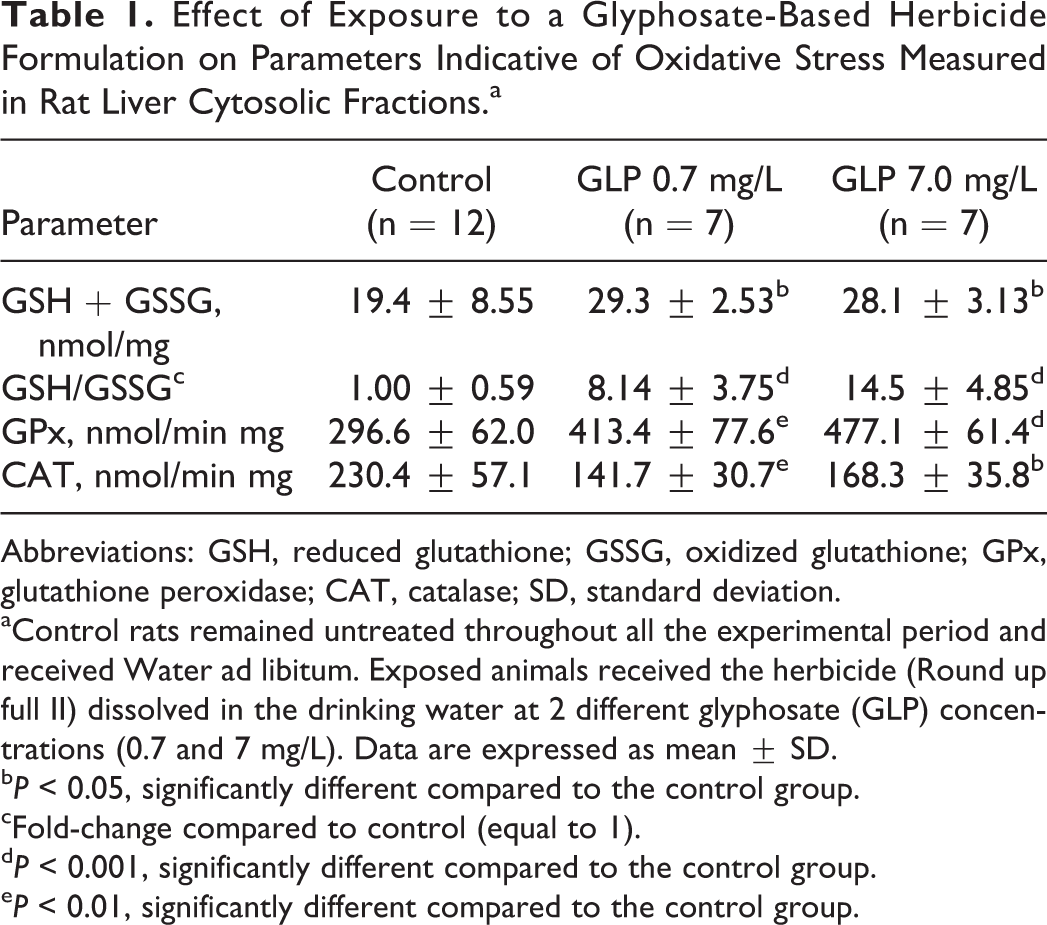
Abbreviations: GSH, reduced glutathione; GSSG, oxidized glutathione; GPx, glutathione peroxidase; CAT, catalase; SD, standard deviation.
aControl rats remained untreated throughout all the experimental period and received Water ad libitum. Exposed animals received the herbicide (Round up full II) dissolved in the drinking water at 2 different glyphosate (GLP) concentrations (0.7 and 7 mg/L). Data are expressed as mean ± SD.
b P < 0.05, significantly different compared to the control group.
cFold-change compared to control (equal to 1).
d P < 0.001, significantly different compared to the control group.
e P < 0.01, significantly different compared to the control group.
Table 2 shows CYP content and metabolic rates of different CYP-dependent enzyme activities in liver microsomes from unexposed (control) male and female rats. No statistically significant differences between male and female animals were observed for the hepatic CYP content and the CYP2B-dependent MROD activity. However, higher metabolic rates for ECOD (11-fold higher, P < 0.001), EROD (2-fold higher, P < 0.001), BROD (3.7-fold higher, P < 0.001), and TAO N-demethylase (1.7-fold higher, P < 0.01) were observed in male compared to female rats. Figure 1 shows the effect of exposure to the GLP-based herbicide on hepatic CYP content and CYP-dependent enzyme activities in male and female rats. In radial diagrams, data are expressed as fold change with respect to their respective control values (equal to 1). The hepatic CYP content was lower in herbicide-exposed rats (males: 45%-49% lower, P < 0.01; females: 52%-54% lower, P < 0.001) compared to male and female control animals. Sex-related differences were observed for several CYP-dependent enzyme activities in rats exposed to the herbicide. Thus, in female rats, the hepatic ECOD activity was 57% higher (P < 0.05) in herbicide-exposed (at both GLP 0.7 and 7 mg/L) compared to control animals. Conversely, this enzyme activity was lower in male rats receiving the herbicide (GLP 0.7 mg/L: 59% lower, P < 0.05; GLP 7 mg/L: 58% lower, P < 0.05) compared to control male animals. Lower (P < 0.05) EROD (36%) and TAO N-demethylase (43%) enzyme activities were observed in liver microsomes from male rats exposed to the herbicide at GLP 7 mg/L. Conversely, in females receiving the herbicide, EROD increased (GLP 0.7 mg/L: 168%, P < 0.05; GLP 7 mg/L: 123%, P < 0.05), whereas TAO N-demethylase did not change at both exposure levels. A higher BROD enzyme activity was observed in herbicide-exposed female rats (GLP 0.7 mg/L: 158% higher, P < 0.01; GLP 7 mg/L: 179% higher, P < 0.01) compared to control female animals. Conversely, exposure to the herbicide did not cause any change in this enzyme activity in male rats.
Cytochrome P450 (CYP) Content and CYP-Dependent Enzyme Activities in Liver Microsomes From Unexposed (Control) Male and Female Rats.a
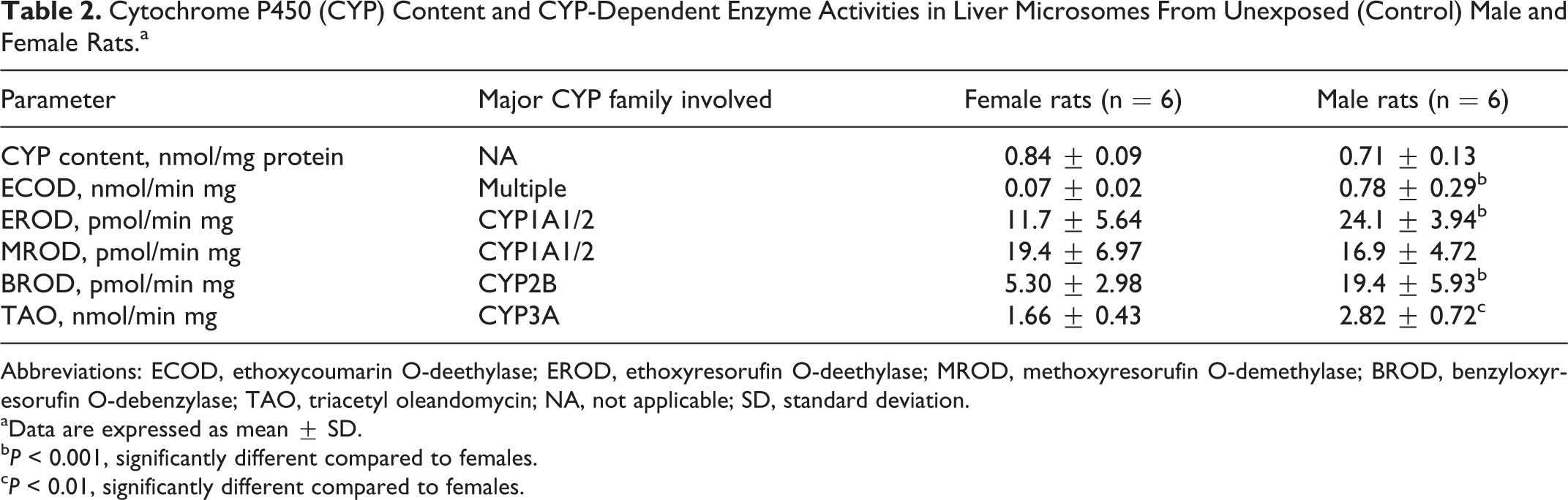
Abbreviations: ECOD, ethoxycoumarin O-deethylase; EROD, ethoxyresorufin O-deethylase; MROD, methoxyresorufin O-demethylase; BROD, benzyloxyresorufin O-debenzylase; TAO, triacetyl oleandomycin; NA, not applicable; SD, standard deviation.
aData are expressed as mean ± SD.
b P < 0.001, significantly different compared to females.
c P < 0.01, significantly different compared to females.
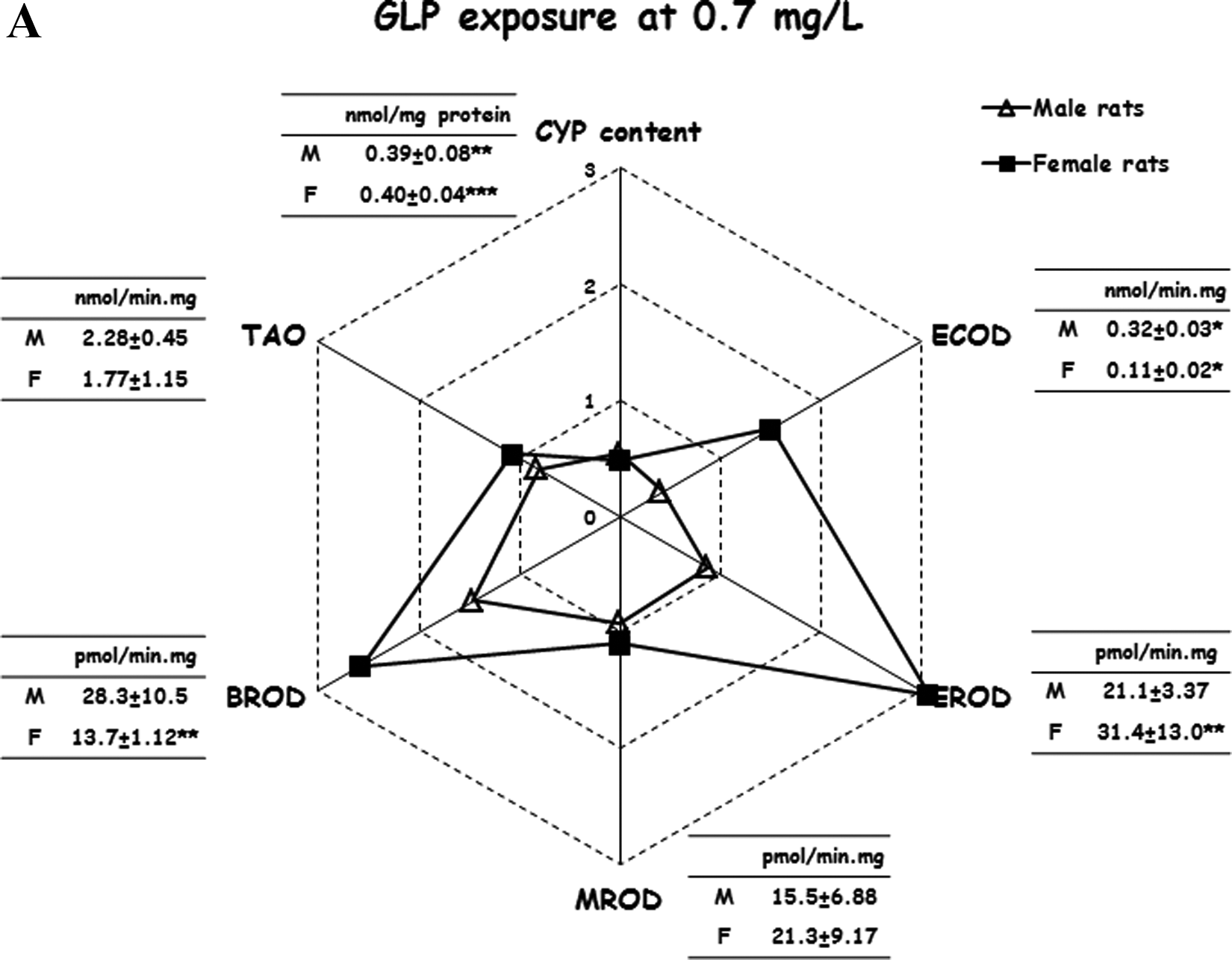
Cytochrome P450 (CYP) content and CYP-dependent enzyme activities in male and female rats exposed to a glyphosate-containing herbicide. Data in radial diagrams are expressed as fold-change with respect to their respective control values (from unexposed animals receiving water ad libitum). Exposed animals received the herbicide (Round up full II) dissolved in the drinking water (during 90 days) at 2 different exposure levels of glyphosate (GLP): 0.7 mg/L (A) and 7 mg/L (B). Inserted tables indicate CYP contents (nmol/mg) and enzyme activities (nmol or pmol/min mg) in herbicide-exposed animals (as mean ± standard deviation [SD]). Values observed in herbicide-exposed rats are significantly different (*: P < 0.05, **: P < 0.01 or ***: P < 0.001) than those shown in Table 2 for their respective male/female control group. ECOD indicates ethoxycoumarin O-deethylase; EROD, ethoxyresorufin O-deethylase; MROD: methoxyresorufin O-demethylase; BROD, benzyloxyresorufin O-debenzylase; TAO, triacetyl oleandomycin.
The hepatic FMO-mediated sulfoxidation of MTZ (Figure 2A) was 49% to 62% lower (P < 0.001) in herbicide-exposed compared to control rats. Conversely, the carbonyl reduction of MEN was higher (P < 0.05) when animals received the herbicide at both exposure levels (Figure 2B), whereas the carboxylesterase enzyme activity (nitrophenyl acetate hydrolysis) did not change (Figure 2C).
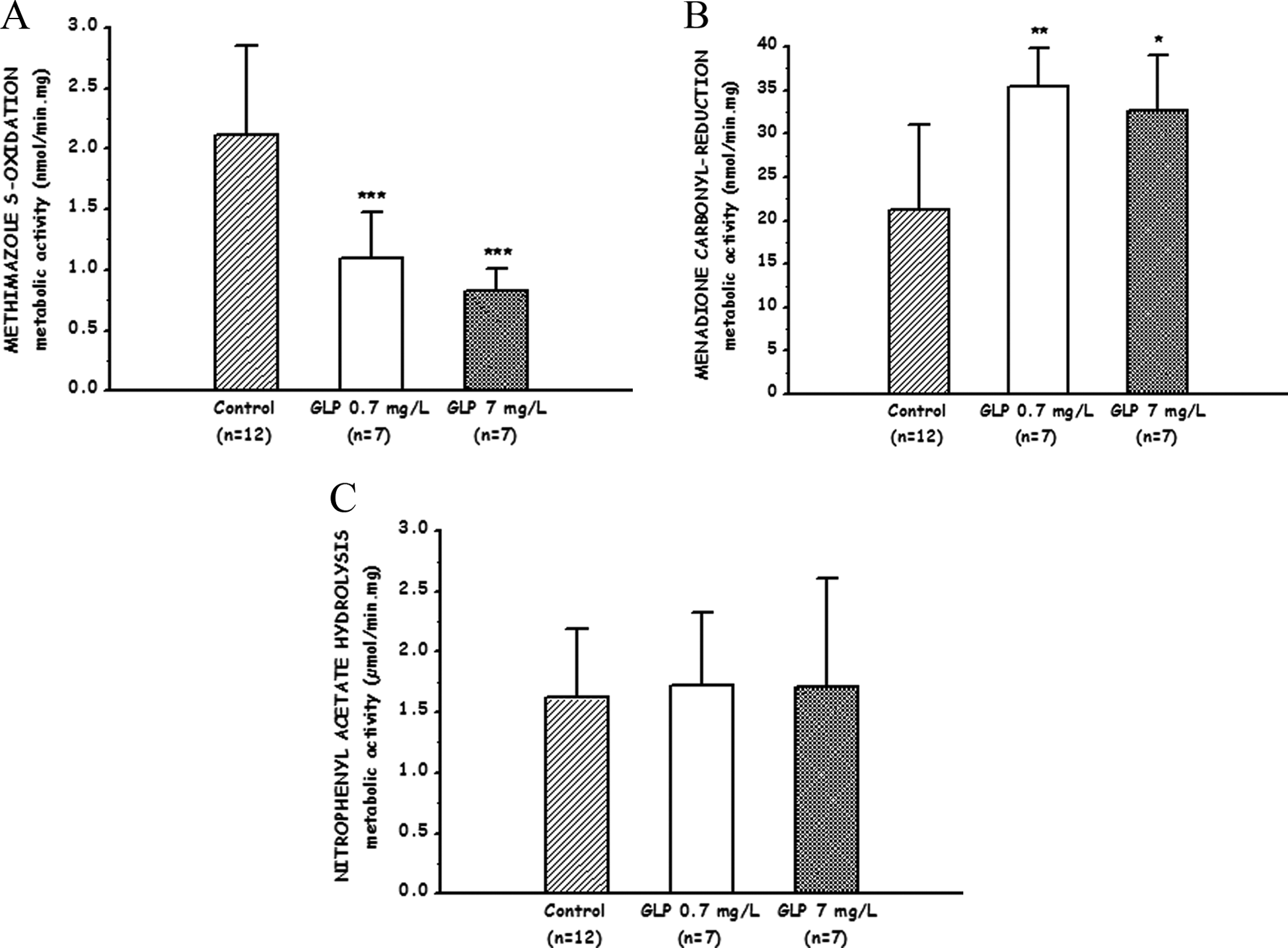
Effects of exposure to a glyphosate-based herbicide on phase 1 enzyme activities in rat liver microsomes: (A) flavin monooxygenase-dependent S-oxidation of methimazole, (B) carbonyl reductase-mediated reduction of menadione, and (C) carboxylesterase activity toward nitrophenyl acetate (C). Control rats remained untreated throughout all the experimental period and received water ad libitum. Exposed animals received the herbicide (Round up full II) dissolved in the drinking water (during 90 days) at 2 different glyphosate (GLP) concentrations (0.7 mg/L and 7 mg/L). Data are expressed as mean ± standard deviation (SD). Values are significantly different (*: P < 0.05, **: P < 0.01 or ***: P < 0.001) than that observed in the control group.
Figure 3 shows metabolic rates of cytosolic GST-dependent conjugations of CDNB, DCNB, and ethacrinic acid and the microsomal UDPGT-mediated conjugation of 1-naphthol in the liver of control and herbicide-exposed rats. A 31% higher (P < 0.01) metabolic rate for the GST-dependent conjugation of DCNB in rats subjected to the highest exposure level of GLP (7 mg/L) was the unique change in the hepatic phase 2 enzyme activities measured.
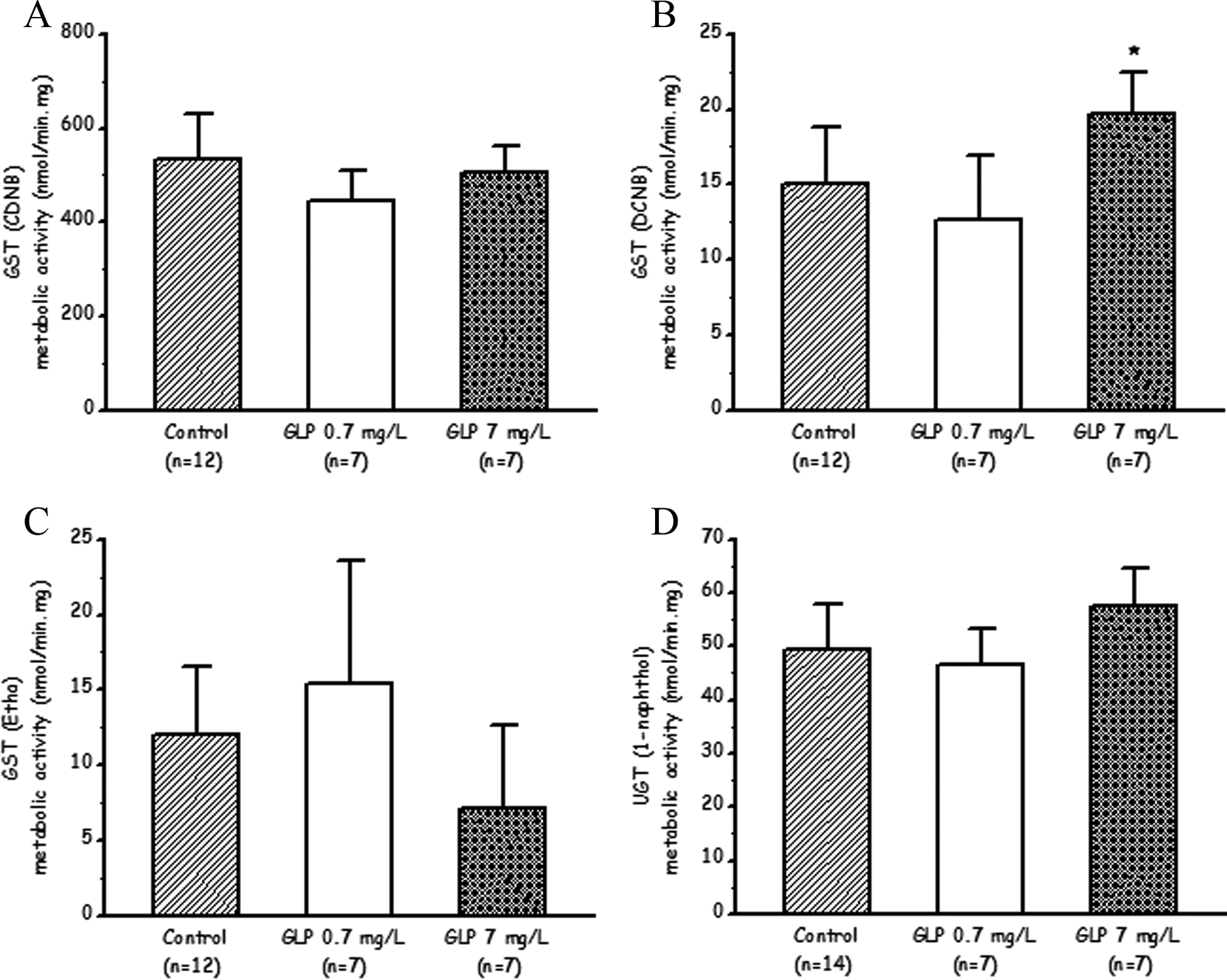
Effects of exposure to a glyphosate-based herbicide on phase 2 enzyme activities in rat liver subcellular fractions: glutathione-S-transferase (GST) activities toward 1-chloro-2,4-dinitrobenzene (CDNB) (A), 1,2-dichloro-4-nitrobenzene (DCNB) (B) and ethacrynic acid (Etha) (C) and uridinediphospho-glucuronosyltransferase (UDPGT) activity toward 1-naphthol (D). Control rats remained untreated throughout all the experimental period and received water ad libitum. Exposed animals received the herbicide (Round up full II) dissolved in the drinking water (during 90 days) at 2 different glyphosate (GLP) concentrations (0.7 mg/L and 7 mg/L). Data are expressed as mean ± standard deviation (SD).
Discussion
The research work described here was based on the possible occurrence of a GLP-based herbicide in the drinking water. The presence of a commercial formulation containing GLP in the drinking water did not modify the pattern of water intake in rats exposed to the herbicide. The estimated daily dose of GLP in animals receiving 0.7 mg/L (the tolerance limit for the herbicide in water) was 0.09 mg/kg. As expected, the estimated daily dose was 10-fold higher (0.9 mg/kg) in rats receiving GLP at 7 mg/L. In addition, a poor bioavailability of GLP was observed after its oral administration in rats. 33 Therefore, a combination of reduced daily sublethal doses and poor bioavailability emphasize the low exposure level of the experimental animals to the herbicide in the current work.
In healthy aerobic organisms, the production of free radicals, including reactive oxygen and nitrogen species (ROS and RNS, respectively), is continuously regulated by the antioxidant defence system. Several investigations reported on the ability of GLP to induce oxidative stress and/or an impairment of the antioxidant defensive mechanisms. 34,35 Generally, short-term exposure periods to this herbicide lead to increased TBARS production and depleted GSH levels in different tissues. 11,35 However, tissue GSH is often elevated after long-term oxidant exposures. 10 In the work described here, higher hepatic levels of total glutathione (GSH + GSSG) and GSH/GSSG ratios, as consequence of increased GSH levels, were observed in the liver of rats exposed to the herbicide during 90 days. This observation is in agreement with previous findings in rats subchronically exposed to GLP. 12 Increased GSH synthesis, as an adaptive response during oxidative stress, has been reported in aquatic organisms. 10 It has been suggested that oxidative stress may trigger a compensatory mechanism through the induction of hepatic γ-glutamyl-cysteine synthetase, the enzyme that controls the biosynthesis of glutathione. 36 This fact may have accounted for the increment of total glutathione and GSH levels observed in the current work and previously in plasma 34 and liver 12 from rats exposed to GLP.
Glutathione peroxidases constitute a family of isoenzymes that catalyze the reduction in H2O2 or organic hydroperoxides to water or their corresponding alcohols using GSH as an electron donor and/or other reducing equivalents. 37 In the current study, an enhanced metabolic activity of GPx was observed after 90 days of exposure to the herbicide (see Table 1), in agreement with our previous observations. 12 Elevated GPx enzyme activities have been associated with tolerance of nontarget species to deleterious effects caused by different insecticides and herbicides. 38 On the other hand, CAT activity was lower in the liver of rats exposed to the GLP-containing herbicide (see Table 1). Similarly, a significant decrease in CAT activity was shown in the plasma and testis of animals treated with different agrochemicals. 34,39 Catalase inhibition has been related to the effects of free radicals, such as O2 − (superoxide) and ONOO− (peroxynitrite), on the heme group located within the active site of the enzyme. 39 Moreover, CAT activity showed a negative linear correlation with GPx activity, which is consistent with the findings reported in the current work.
Metabolic activities of several XMEs were evaluated in rat hepatic subcellular fractions by means of a biochemical approach based on the assumption that certain molecules are suitable substrates to test their metabolic activities. Microsomal and cytosolic fractions obtained from both unexposed and herbicide-exposed rats were able to metabolize all the assayed substrates. The use of “diagnostic” probes is particularly useful to the overall characterization of the expression and function of XMEs in nontarget species naturally or accidentally exposed to pesticides. 14,15
The CYP system is a superfamily of hemoproteins composed by different families and subfamilies classified on the basis of their amino acid sequence similarities. 40 Family members (designated with numbers) have at least 40% amino acid sequence homology, whereas the structural homology within a subfamily (designed with capital letters) is greater than 55%. More than 15 CYP families have been described in mammals, but only members belonging to families 1 to 3 are known to be mostly involved in xenobiotic biotransformation including 70% to 80% of therapeutically used drugs in humans. 40,41 In the current work, metabolic activities of CYP1A, 2B, and 3A subfamilies were measured in liver microsomes from male and female rats (see Table 2). Sex differences in the expression and function of different members within the CYP system in the liver of rats and humans are well recognized. In humans, such differences are rather small and to some extent controversial or influenced by extrinsic factors. 41 Conversely, male rats exhibit a higher metabolic ability compared to females in most cases. 42,43 Generally, the sex-dependent expression of a given CYP isoenzyme is responsible for the sex difference in the metabolic fate of certain xenobiotics. 43 This fact is in agreement with the observed higher metabolic rates for ECOD (a general CYP reaction), EROD (CYP1A1/2 marker activity), BROD (CYP2B), and TAO N-demethylase (CYP3A) in liver microsomes from male compared to female rats in the current work (see Table 2).
Certain agrochemicals, such as organophosphorus insecticides and fipronil, affected the expression and function of different CYPs in the liver of laboratory animals and humans. 14,15,44 In the assays described here, both male and female liver microsomes had reduced CYP levels after exposure to the herbicide during 90 days, in agreement with previous findings in rats intragastrically treated with GLP during 2 weeks. 16 Notwithstanding the absence of sex differences for this parameter, the exposure to the herbicide caused a differential CYP-dependent metabolic pattern in male and female rat liver (see Figure 1). In fact, in male rats, CYP-dependent enzyme activities tend to be lower in herbicide-exposed animals. On the contrary, the same enzyme activities appear to be induced in exposed compared to unexposed female rats. This observation is consistent with a proposed greater magnitude of CYP induction in female compared to male rat liver. 45 In fact, these authors showed considerably higher responses to model inducers of CYP2B (phenobarbital) and CYP3A (pregnenolone-16α-carbonitrile) in the liver of female rats. Further work should be addressed to investigate the molecular mechanisms involved in these sex-differential effects on CYP-dependent metabolism in nontarget species exposed to GLP.
The mammalian FMOs are microsomal enzymes that catalyze the NADPH-dependent oxygenation of several endogenous compounds (ie, trimethylamine, methionine, and cysteamine), as well as a large number of structurally diverse xenobiotics containing nucleophilic atoms like nitrogen, phosphorus, and sulfur. 46 Five distinct functional mammalian FMO isoenzymes (FMO 1-5) have been described using different biological methods, being FMO 1-3 the most important enzymes of this system involved in the metabolism of foreign compounds. 47 Pesticides such as organophosphates and carbamates are metabolized by the FMO system 48,49 and, consequently, they may delay the metabolism of other FMO substrates. Although GLP is a phosphorus-containing molecule, there is no evidence supporting a FMO-mediated biotransformation of this herbicide, or about its effects on the expression and function of this enzyme system in mammals. Our results show a reduction in FMO-dependent metabolism in rats exposed to the GLP-based herbicide (see Figure 2A). In mammals, there are few reports on the induction of FMO isoenzymes 50,51 and, generally, those xenobiotics affecting their expression and function cause downregulation or inhibition of the FMO-dependent metabolism. 52 Whether or not the reduced enzyme activity in herbicide-exposed rats is consequence of a reduced gene/protein expression should be the subject for future research. Downregulation of FMO-dependent metabolism may account for a low rate of biotransformation of different endo- and xenobiotics metabolized by this enzyme system.
Phase I enzymes catalyzing the reduction of xenobiotics containing a carbonyl group include short-chain dehydrogenases/reductases (SDR). 53 These enzymes catalyze the NADPH-mediated reduction of a large number of biologically and pharmacologically active substrates, including a variety of endogenous and xenobiotic carbonyl compounds. 53,54 Cytosolic carbonyl reductases (CBRs) and the membrane-bound 11β-hydroxysteroid dehydrogenase are the most relevant enzymes within the SDR superfamily. In humans, there are 2 monomeric cytosolic CBRs named CBR1 and CBR3. 55 In rats, gene/protein sequences of CBR1 and CBR3 were found approximately 85% identical to their respective human counterpart. 55 The rate of NADPH oxidation in the presence of the substrate MEN was shown as a good marker of CBR1 and CBR3 activities. 30,56 In the current work, an increased CBR-dependent reduction of MEN was observed in the liver of herbicide-exposed rats (see Figure 2B). It has been shown that CBRs also have a protective role in oxidative stress through the detoxification of reactive carbonyls formed during lipid peroxidation. 54 Similarly, the observed induction of CBR activity may indicate an adaptive response to the proposed oxidative damage caused by exposure to the herbicide.
Mammalian carboxylesterases (CES) play a relevant role in the biotransformation of a wide array of ester-containing xenobiotics including certain pesticides such as organophosphates, carbamates, and pyrethroids. Substrates such as p-nitrophenyl acetate have been used as biochemical markers for this enzyme system. 57 Inhibition of CES-dependent enzyme activities has been associated with increased toxicity as consequence of exposure to carbamates and organophosphates. Moreover, these enzymes and other esterases, such as butyryl- and acetyl-cholinesterases, have been used as biomarkers of exposure to these pesticides because their activities can be measured in plasma. 57,58 Inhibition of CES and acetylcholinesterase activities has been shown in aquatic species exposed to environmentally relevant levels of GLP or GLP-based herbicides. 59 However, our results show that the CES-dependent hydrolysis of p-nitrophenyl acetate should not be considered as marker of exposure to a GLP-based herbicide in a nontarget mammal species.
The GSTs catalyze nucleophilic attack by GSH on a wide array of nonpolar compounds that contain an electrophilic carbon, nitrogen, or sulfur atom such as plant phenols, mycotoxins, many chemical carcinogens, insecticides, herbicides, quinones, and certain reactive oxygen species. 60 Four main classes of cytosolic GSTs (α, µ, π, and θ) have been identified in rats and humans, each of them containing different isoenzymes. 61 Marker substrates for cytosolic GSTs include CDNB (a general nonspecific substrate), DCNB, and ethacrinic acid (for GSTµ and GSTπ, respectively). 60 On the other hand, mammalian UDPGTs are a superfamily of membrane bound enzymes that catalyze the conjugation of glucuronic acid to a nucleophilic substrate and have been grouped into 2 distinct families, UDPGT1 and UDPGT2. 62 In addition, 1-naphthol is considered a suitable probe to evaluate UDPGT activity in microsomal preparations. A reduction in GST-dependent activities has been shown in rats exposed to GLP and other pesticides, 35,36 whereas increased UDPGT activities were observed after subchronic exposures to organophosphorus insecticides. 63 However, the exposure to a GLP-based herbicide did not show relevant changes in GST- and UDPGT-dependent enzyme activities in rat liver subcellular fractions.
In conclusion, the exposure to a GLP-based herbicide may cause oxidative stress in rats, a nontarget mammal species. Increased total glutathione and GSH levels, as well as enhanced GPx activity, may act as a protective mechanism against possible detrimental effects of the herbicide. The work described here clearly shows that exposure to a GLP commercial formulation modified oxidative (CYP and FMO dependent) and reductive (CBR dependent) enzyme functions in rat liver. These changes may affect the metabolic fate and, hence, the elimination of different endobiotic and xenobiotic substances (including therapeutically used drugs and toxic molecules) metabolized by these XMEs. Overall, the research presented here demonstrated certain biochemical modifications after exposure to a GLP-based herbicide. Such modifications were of the same order of magnitude independently of the exposure level assayed. These changes affecting XMEs may occur even at 3- to 20-fold lower exposure levels compared to the GLP oral reference dose of 2 mg/kg/d. 64 As previously stated, the herbicide formulation under study consists of a mixture of the active principle plus adjuvants (ie, ethoxylated surfactants) and, therefore, the observed effects cannot be attributable to GLP alone. The pharmacotoxicological significance of the findings emerging from this research remains to be clarified in humans.
Footnotes
Authors’ Contribution
Karen Larsen contributed to conception and design, acquisition, analysis, interpretation, drafted manuscript, and gave final approval. Roberto Najle contributed to conception, critically revised manuscript, and gave final approval. Adrián Lifschitz contributed to design, acquisition, critically revised manuscript, and gave final approval. María L. Maté contributed to acquisition and gave final approval. Carlos Lanusse contributed to conception, critically revised manuscript, and gave final approval. Guillermo L. Virkel contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, critically revised manuscript, and gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Comisión de Investigaciones Científicas (CIC), Buenos Aires Province (Resolution 813/13) and Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT) (PICT 1432), all from Argentina. The first author (Karen Larsen) holds a fellowship granted by Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina.
