Abstract
Our recent studies have shown that trichloroethylene (TCE) was able to induce multisystem injuries in the form of occupational medicamentosa-like dermatitis, including skin, kidney, and liver damages. However, the role of complement activation in the immune-mediated liver injury is not known. This study examined the role of complement activation in the liver injury in a mouse model of TCE-induced sensitization. Treatment of female BALB/c mice with TCE under specific dosing protocols resulted in skin inflammation and sensitization. Skin edema and erythema occurred in TCE-sensitized groups. Trichloroethylene sensitization produced liver histopathological lesions, increased serum alanine aminotransferase, aspartate transaminase activities, and the relative liver weight. The concentrations of serum complement components C3a-desArg, C5a-desArg, and C5b-9 were significantly increased in 24-hour, 48-hour, and 72-hour sensitization-positive groups treated with TCE and peaked in the 72-hour sensitization-positive group. Depositions of C3a, C5a, and C5b-9 into the liver tissue were also revealed by immunohistochemistry. Immunofluorescence further verified high C5b-9 expression in 24-hour, 48-hour, and 72-hour sensitization-positive groups in response to TCE treatment. Reverse transcription–polymerase chain reaction detected C3 messenger RNA expression in the liver, and this was significantly increased in 24-hour and 48-hour sensitization-positive groups with a transient reduction at 72 hours. These results provide the first experimental evidence that complement activation may play a key role in the generation and progression of immune-mediated hepatic injury by exposure to TCE.
Introduction
1,1,2-Trichloroethylene (TCE) is the most widely used chlorinated organic solvent. Trichloroethylene is mainly used for degreasing metal products before painting and exposure by humans often occurs in workplace and/or ambient environment via inhalation and/or direct dermal contact. 1 –3 Trichloroethylene constituted the most commonly identified binary mixture in groundwater at 1700 National Priority List hazardous waste sites surveyed in the United States, and it has been estimated that TCE is present in one-third of municipal water supplies. 4 The number of cases of TCE-induced occupational skin disorders has been rising since mid-1990s in Asia, especially in China. 5 In China, every year the use of TCE is increasing, mostly in coastal industrialized regions. In Guangdong province, according to the diagnosis of various medical and health institutions, we have found 394 patients since the first medicamentosa-like dermatitis induced by TCE was diagnosed in 1988. 6 Trichloroethylene exposure has also been associated with mouse liver injury 7 and allergic hepatitis in the guinea pig maximization test (GPMT). 8 More importantly, occupational exposure to TCE was associated with impaired hepatic function. 9
The liver is a vital organ for the synthesis of complement components. The complement system is composed of approximately 35 proteins that are present either as soluble factors in the blood or as membrane-associated proteins. 10 The function of complement system is to directly destroy invading microbes or to assist in phagocytosis of various foreign targets. 11 Complement can be activated by 3 pathways: the antibody-dependent classical pathway, the alternative pathway, or the lectin-associated (mannose-binding lectin [MBL]/MBL-associated serine protease 2) pathway. Each pathway results in C3 activation and leads to the generation of anaphylatoxins C3a and C5a, which activate innate immune cells, such as mast cells, monocytes, basophils, and neutrophils. Anaphylatoxins C3a and C5a bind to their receptors (C3aR/C5aR) to exert a biological effect. Kupffer cells (KCs), hepatic stellate cells (HSCs), and sinusoidal endothelial cells (SECs) in the liver from mice all express C3a and C5a receptors (C3aR/C5aR). The C5b-9 complex, also known as membrane attack complex (MAC), formed from the activation of terminal complement components C5b, C6, C7, C8, and C9. The C5b-9 can kill microbial pathogens and damage host tissue. 12,13 Deposition of MAC not only directly induces cellular injury but also amplifies the inflammatory response by promoting the expression of proinflammatory mediators. 14
Complement activation has been implicated in several hepatic disease models, including ischemia reperfusion, nonalcoholic steatohepatitis, and hemorrhagic shock. 10 In a previous study, we found that the levels of serum complements were significantly decreased in a guinea pig toxicity model induced by TCE and severe liver damage occurred. 15 We therefore hypothesize that complement activation may play a role in the pathogenesis of TCE-induced liver injury as a novel mode of immunotoxicity in addition to type IV allergic reaction as many studies have already shown. The critical issue is the evidence of complement activation in the liver tissue and its role in liver damage. This study therefore examined changes to the level of complements in serum and liver following TCE sensitization. Deposition of C5b-9 in the liver was determined to indicate the participation of complements in liver damage by TCE.
Materials and Methods
Animals
BALB/c female mice aged 6 to 8 weeks, weighing 28.80 ± 2.57 g, were obtained from Hunan Slac Jingda Laboratory Animal Co, Ltd (Hunan, China). All animals were healthy, housed 6 animals per cage at 20°C, and maintained in a 12:12-hour light–dark cycle (light: 07.00-19.00). Animals received tap water and a standard diet ad libitum. After a week of adaptive feeding, the healthy BALB/c mice were randomly divided into blank control group, solvent (olive oil and acetone) control group, and TCE-treatment group. The use of animals and the experimental protocol were approved by the Animal Care and Use Committee of Anhui Medicine University.
Experimental Protocols
We established a BALB/c mice TCE-sensitization model by reference to GPMT and Naoki Inagaki’s Mouse Allergic Dermatitis Models. 16 The procedures are briefly described as follows: (1) preparation of the solutions: the solvent, 50% TCE (TCE:olive oil:acetone = 5:2:3) and 30% TCE (TCE:olive oil:acetone = 3:2:5) were prepared; (2) treatment of BALB/c mice with TCE: 100 μL of 50% TCE and Freund complete adjuvant (1:1) mixed fluid was used for the first sensitization on first day by intradermal injection into the abdomen, and 100 μL of 50% TCE was then painted on the abdominal skin on 4th, 7th, and 10th day; a week later, 30% TCE was painted twice challenging the back skin on 17th and 19th day. The solvent control group followed the same procedure without TCE. The blank control group received no treatment. Cutaneous reaction scores were judged by “The technical specification of toxicity identification for chemicals” 17 after 24 hours. If the cutaneous reaction score of any animal in the treatment group was equal to 1 or greater, it would be judged as a positive reaction and classified as sensitization positive and otherwise as sensitization negative. According to the sensitization outcome on the final challenge, the TCE treatment group was subdivided into 24-hour group, 48-hour group, 72-hour group, and 7-day group. In each group, samples of the liver and blood were collected for analysis; blood was obtained by retro-orbital puncture.
Body Weight and Relative Organ Weight
The animals were sacrificed after blood was collected from ocular venous plexus and the liver removed. The absolute weight of the removed liver was measured, and the relative organ weight (liver to body weight ratio) was calculated as follows:
Relative liver weight = (weight of liver/body weight of the mouse on the day of sacrifice) × 100.
Liver Enzyme Determinations
Activities of the serum enzymes alanine aminotransferase (ALT) and aspartate transaminase (AST) were determined using an automatic biochemical analyzer in Clinical Laboratory of The First Affiliated Hospital of Anhui Medical University to assess the TCE-induced damage to liver function.
Liver Histopathology
The liver fragments were fixed in 10% buffered formaldehyde solution for at least 48 hours, embedded into paraffin blocks, and processed according to routine protocols. Sections of 5-μm thickness were cut from each sample and stained with hematoxylin and eosin for histopathological examination.
Complement Quantification by Sandwich Enzyme-Linked Immunosorbent Assays
The concentrations of complement C3a-desArg, C5a-desArg, and C5b-9 were determined by enzyme-linked immunosorbent assay kit (R&D Systems, Minneapolis) using a standard curve. The kit was taken out from the 2°C to 8°C environment and equilibrated for 30 minutes at the room temperature. The prepared samples and antibodies labeled with enzyme were added into the plate and incubated for 60 minutes at 37°C. The plate was then washed 5 times with the washing buffer. Chromogen solution was added followed by a 10-minute incubation at 37°C. The reaction was terminated by adding the stop solution into each well. The optical density (OD) at 450 nm was read in a microplate reader within 10 minutes.
Liver Immunohistochemistry
Liver tissue sections of 5 μm were deparaffinized in xylene and rehydrated through a series of graded alcohols. Slides which dipped in sodiocitrate (pH 6.0) were treated in a microwave oven for antigen retrieval. The sections were first incubated with monoclonal mouse anti-C3a antibody (Santa Cruz, California) diluted 1:400, polyclonal rabbit anti-C5a antibody (Santa Cruz) diluted 1:400, and polyclonal rabbit anti-C5b-9 antibody (Abcam, Cambridge, United kingdom) diluted 1:1000 with 0.1 mmol/L phosphate- buffered saline (PBS; pH 7.4) overnight at 4°C. On the next day, the sections were incubated with antimouse or antirabbit immunoglobulin (Ig) G (MaxVisonTM Maixin, Fujian, China) at room temperature and then stained by avidin–biotin peroxidase reaction.
The semiquantitative scoring of immunoreactions took into account both the area and the intensity of staining in the tissue. The grading of immunoreactant deposition in the liver was based on a semiquantitative assessment of deposition area with the 5 standard assigned grades as follows: 0, the area of positive reaction was less than 5%; 1, less than 25%; 2, less than 50%; 3, less than 75%; and 4, more than 75%. The grading of immunoreaction intensity was estimated with the 4 standard grades: 0, no positive reaction; 1, light brown; 2, moderate brown; and 3, dark brown. The scores of immunoreactions were calculated by the following formula: mean scores = score of positive reaction area + score of reaction intensity. Five tissue sections were selected in each group to assess the deposition intensity. Slides were coded so that the counter was blind to the identity of the slides, and 3 independent assessments were performed for each slide.
Immunofluorescence Study for C5b-9
Liver biopsy specimens were embedded in optimal cutting temperature (JUNG, Leica, Germany). Sections (10 μm) were cut by a cryostat (Leica CM1850, Leica Mycrosystems, Germany). Briefly, sections were incubated for 2 hours in a blocking buffer containing 5% goat serum in 0.1 mmol/L PBS (pH 7.4). Polyclonal rabbit anti-C5b-9 antibody (Abcam, Cambridge, United Kingdom) was applied to the tissue section as the primary antibody diluted 1:1000 with PBS, and the slides were then incubated overnight at 4°C. Phycoerythrin (PE)-conjugated AffiniPure Donkey Anti-Rabbit IgG-PE (Santa Cruz, California) was used as the secondary antibody diluted 1:400 for visualization of antibody binding in a 2-hour incubation. Subsequently, the slides were washed 3 times with PBS and then stained with 4′,6-diamidino-2-phenylindole (4 μg/mL in deionized water, Sigma, St Louis, Missouri) for 15 minutes at room temperature in the dark. Localization of C5b-9 was assessed by an Olympus DP50 fluorescent microscope with appropriate excitation and emission filters at×400.
Reverse Transcription–Polymerase Chain Reaction Study for C3
Total RNA was extracted from liver sample using Trizol reagent source. RNA was resuspended in diethylpyrocarbonate-treated water and quantified by measuring the OD at wavelengths of 260/280 nm. For complementary DNA synthesis, 2 μg of total RNA was reverse transcribed using a Reverse Transcription kit from Thermo Scientific (United States) following the supplier’s instructions. Polymerase chain reaction (PCR) products were normalized against glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Gel image analysis system (Tannon, China) was used to analyze the electrophoresis images, and the ratio of C3/GAPDH gray scale intensity was an index of messenger RNA (mRNA) expression. The following mouse primers were designed with the aid of software (Primer Premier 5.0, Canada) and the use of National Center for Biotechnology Basic Local Alignment Search Tool searches to ensure the specificity of the primers. C3: sense, tacaacgtagaggccacatcc, anti-sense, gcaggtgactttgcttttgag. GAPDH: sense, aggccggtgctgagtatgtc, anti-sense, tgcctgcttcaccaccttct.
Statistical Analysis
All data were expressed as mean ± standard deviation. Statistical analysis was performed using Statistical Package for Social Sciences (SPSS for Windows, version 11.0). The comparison of each intervention group with the appropriate control was analyzed with 1-way analysis of variance followed by post hoc least-significant difference test or Dunnett test. P < .05 was considered statistically significant.
Results
Sensitization Rates of BALB/c Mice in the TCE-Treated Group
The sensitization rate of BALB/c mice challenged by TCE was calculated by the following formula: sensitization rate = (number of redness or swelling)/number of animals in the group. The animals were then divided into sensitization-positive group (dermatitis [+]) and sensitization-negative group (dermatitis [−]). According to the dermal allergic reactions, TCE-treated groups contained 23 dermatitis (+) in 63 animals, and the sensitization rate was 36.51%. The blank control group and the solvent control group contained 0 dermatitis (+) in 12 animals, thus excluding any artifact.
Body Weight and Relative Organ Weight
The initial body weight of BALB/c mice used was 28.80 ± 2.57 g, and at the end of the last challenge, the value was 29.30 ± 2.19. Although the absolute weight of BALB/c mice increased during the experimental period, there was no significant (P > .05) difference in the body weight among blank control group, solvent control group, and TCE-treated groups. However, there was a significant increase in the relative liver weight, which was observed in 24-hour, 48-hour, and 72-hour sensitization-positive groups, compared with blank control group, solvent control group, and the respective sensitization-negative groups (Table 1). This provides evidence for liver enlargement following TCE sensitization.
Liver Enzymes and Relative Organ Weight in BALB/c Mice After Treatment With TCE.
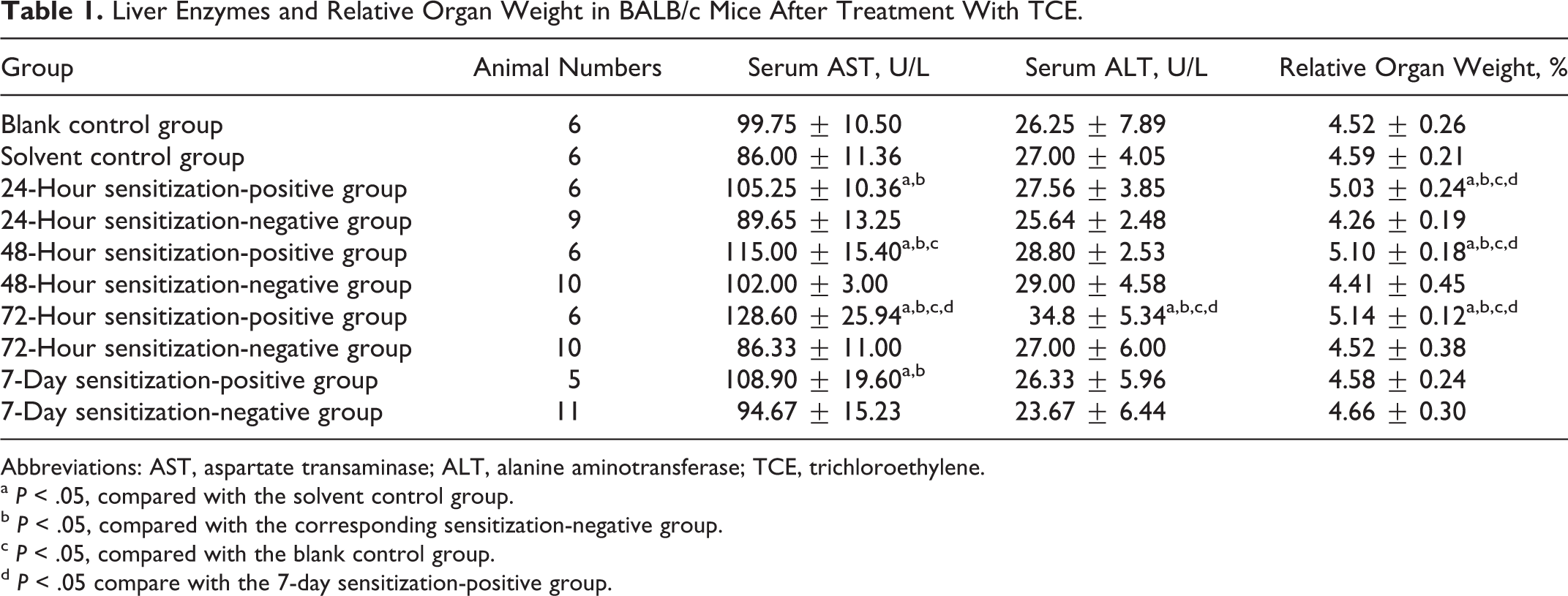
Abbreviations: AST, aspartate transaminase; ALT, alanine aminotransferase; TCE, trichloroethylene.
a P < .05, compared with the solvent control group.
b P < .05, compared with the corresponding sensitization-negative group.
c P < .05, compared with the blank control group.
d P < .05 compare with the 7-day sensitization-positive group.
Histopathology of the Liver and Determination of Serum ALT and AST Activity
The blank and solvent control groups had no obvious abnormalities in the liver; the cells were arranged in neat hepatic cords with uniform cytoplasm and clear nuclear membrane and nucleoli. Trichloroethylene-sensitization-negative group showed a small degree of liver cell edema. The 24-hour, 48-hour, and 72-hour sensitization-positive groups exhibited significant vacuolation, karyopyknosis, single cell necrosis, and inflammatory cell infiltration; 24-hour and 48-hour sensitization-positive groups showed mild edema, while the 72-hour sensitization-positive group displayed massive liver cell edema with nucleus ruptured and lost (Figure 1). These data indicate that TCE sensitization produced structural damage to liver tissue.
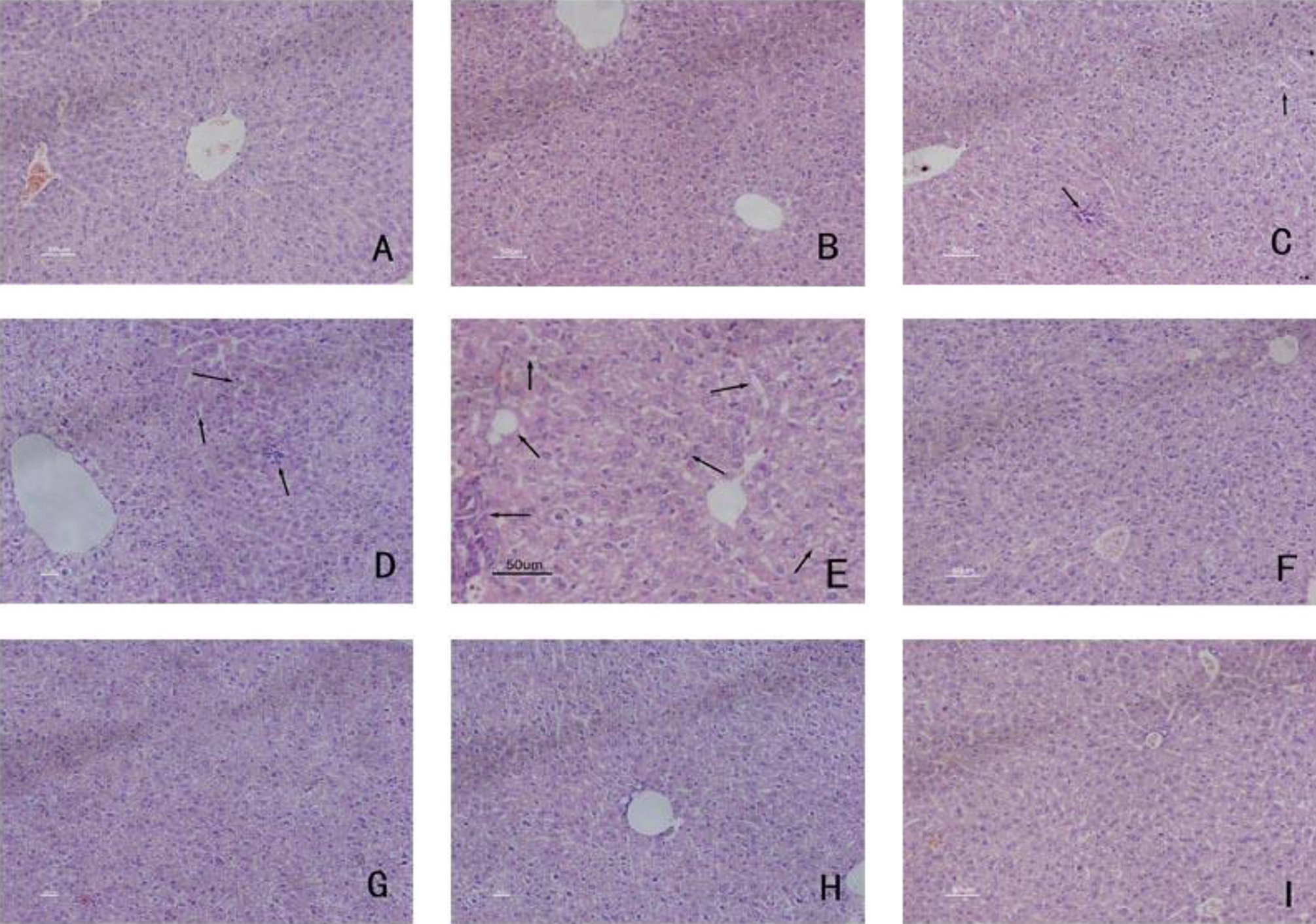
Liver histopathology in control groups and TCE-sensitization-positive groups. (A) Blank control group and (B) solvent control group: the cells neatly arranged in hepatic cords, uniformed cytoplasm, clear nuclear membrane, and nucleoli; (C) 24-hour sensitization-positive group, (D) 48-hour sensitization-positive group, and (E) 72-hour sensitization-positive group: vacuolation, karyopyknosis, single-cell necrosis, and inflammatory cell infiltration (arrows); (F) 7-day sensitization-positive group: no obvious abnormalities, (G) 24-hour sensitization-negative group, (H) 48-hour sensitization-negative group, and (I) 72-hour sensitization-negative group: a small degree of liver cell edema (magnification 200×). TCE indicates trichloroethylene.
Serum AST and ALT levels were determined as a measure of hepatic function. Compared to the blank control group and the solvent control group, 24-hour, 48-hour, 72–hour, and 7-day sensitization-positive groups had significantly higher levels of serum AST (P < .05; Table 1). Sensitization-positive groups (24 hours, 48 hours, 72 hours, and 7 days) also exhibited significantly higher level of AST than their sensitization-negative counterparts. Thus, liver damage was sensitization-specific, different from a direct toxic effect and consistent with an immune response. There was no significant difference in serum ALT between the 2 control groups and 24-hour, 48-hour, and 7-day sensitization-positive groups (P > .05; Table 1). However, the serum ALT level in the 72-hour sensitization-positive group was significantly higher than those from blank and solvent control groups and 72-hour sensitization-negative group (P < .05; Table 1). These changes further demonstrate that the liver function was comprised following TCE sensitization.
Detection of Serum C3a-desArg, C5a-desArg, and C5b-9
The serum levels of C3a-desArg and C5a-degArg were not significantly different between the sensitization-negative groups and the blank and solvent control groups (P > .05) but increased significantly (P < .05) in all 4 sensitization-positive groups. There was a significant difference between each sensitization-positive group and the corresponding sensitization-negative group (P < .05; Figure 2).
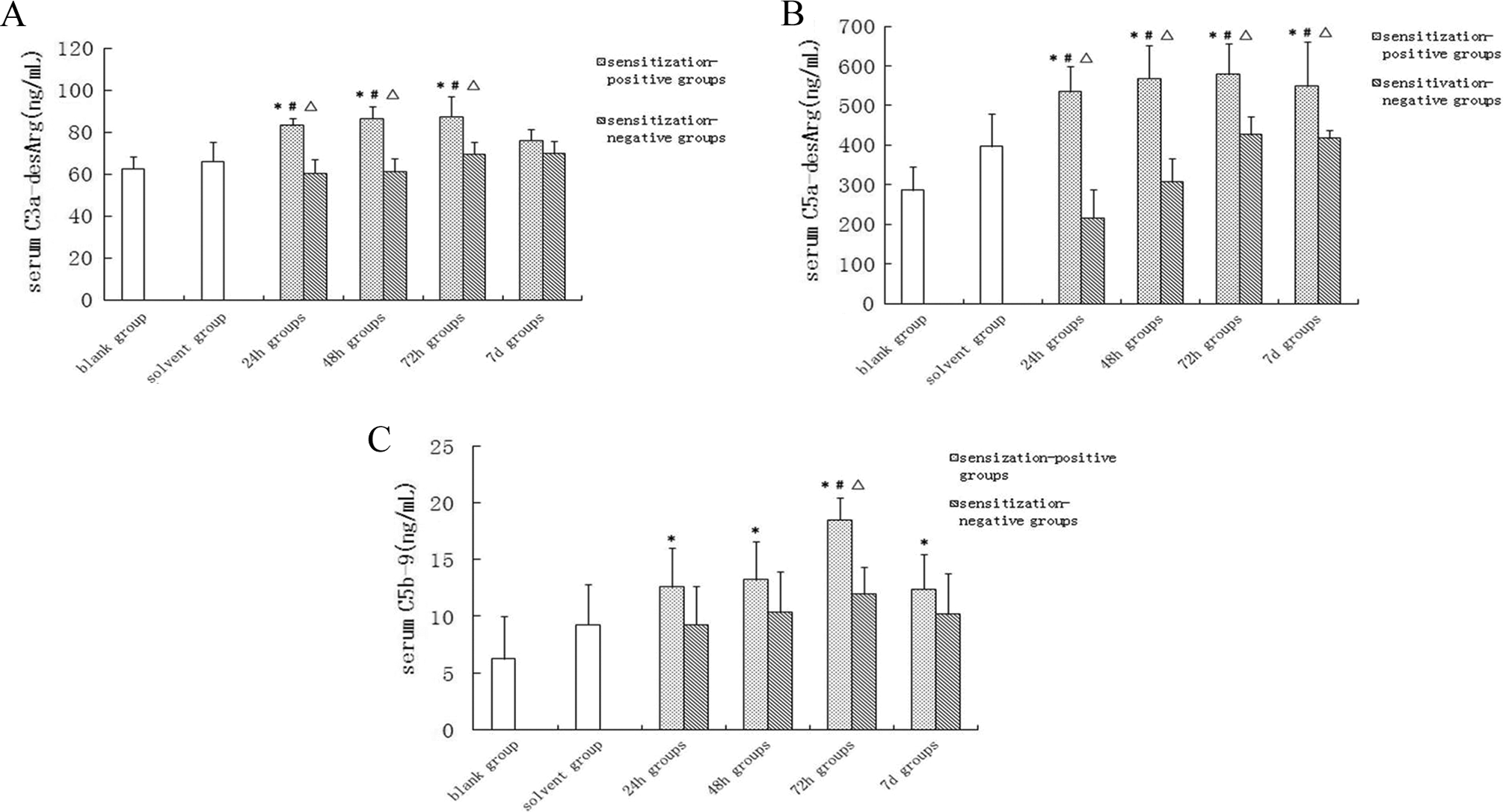
Concentrations of complement activation products C3a-desArg, C5a-desArg, and C5b-9 in serum. The complement activation products C3a-desArg, C5a-desArg, and C5b-9 were measured from serum samples collected from BALB/c mice by retro-orbital puncture. Data are shown as mean ± SD. *P < .05, compared with blank control group. # P < .05 compared with the solvent control group. ▵ P < .05, compared with the corresponding sensitization-negative group. SD indicates standard deviation.
Compared with the blank control group, the C5b-9 levels were significantly higher in 24-hour, 48-hour, 72-hour, and 7-day sensitization-positive groups and peaked at 72 hours. Thus, C5b-9 in the 72-hour sensitization-positive group was higher than those from the blank control group, solvent control group, and 72-hour sensitization-negative group (Figure 2). Therefore, TCE sensitization caused an increase in serum levels of all the above-mentioned complements.
Deposition of Complement C3a, C5a, and C5b-9
Table 2 summarizes the mean scores of the deposition of C3a, C5a on the surface of KCs, HSCs, and SECs, and C5b-9 on the surface of hepatocytes from different experimental groups. Representative micrographs are shown in Figures 3 to 5. There were no immunoreactant depositions in the blank control group and the solvent control group. As shown in Table 2, C3a deposition increased significantly (P < .05) in 24-hour, 48-hour, 72-hour, and 7-day sensitization-positive groups and peaked in the 72-hour sensitization-positive group (mean score: 2.10 ± 1.26). Compared with blank and solvent control groups, the deposition of C5a was significantly greater in 24-hour, 48-hour, 72-hour, and 7-day sensitization-positive groups and peaked in the 72-hour sensitization-positive group (mean score: 2.58 ± 0.46). There was also a significant difference in C5b-9 deposition between the blank/solvent control groups and 24-hour, 48-hour, 72-hour, and 7-day sensitization-positive groups. These changes were absent in the sensitization-negative groups. All the above-mentioned data further suggest that these complements were deposited into the liver tissue following TCE sensitization.
Deposition of C3a, C5a, and C5b-9 in Liver Tissue Sections in BALB/c Mice Following TCE Treatment.
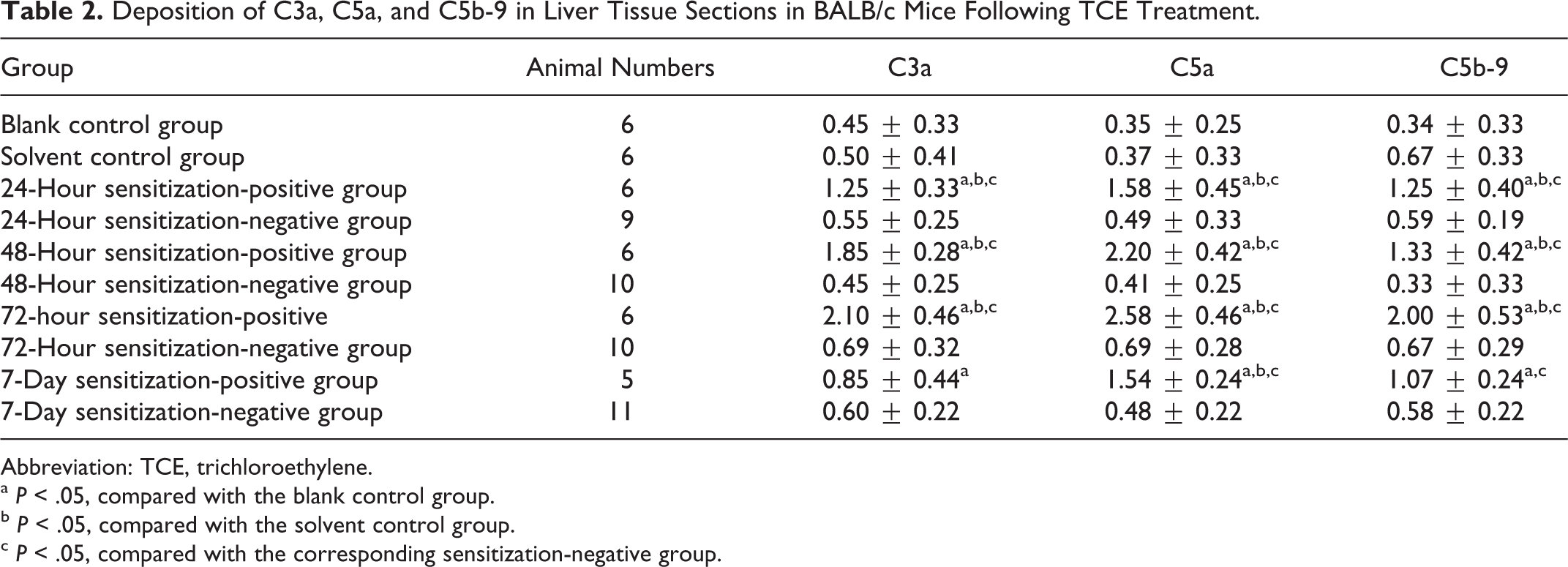
Abbreviation: TCE, trichloroethylene.
a P < .05, compared with the blank control group.
b P < .05, compared with the solvent control group.
c P < .05, compared with the corresponding sensitization-negative group.
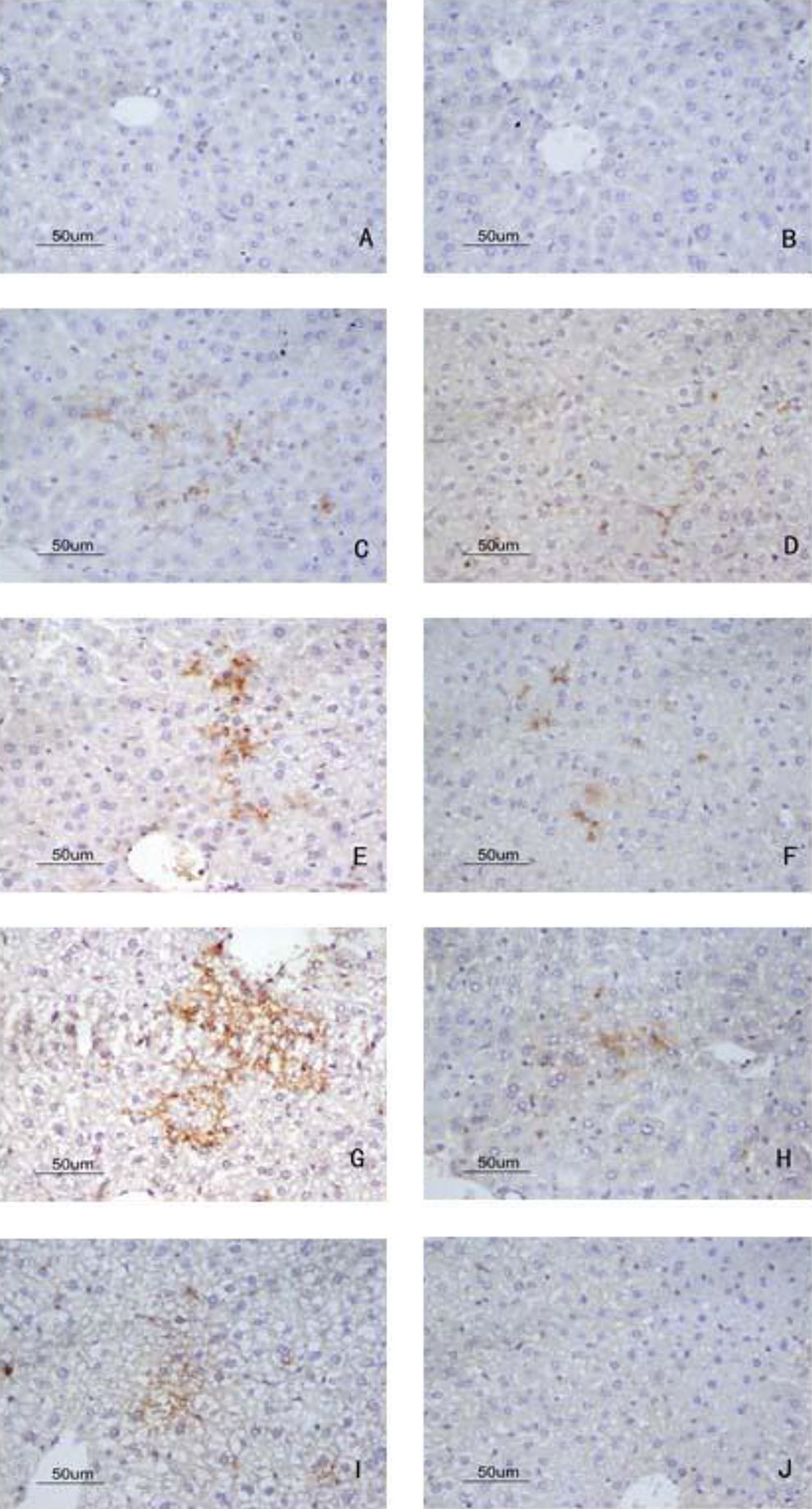
Deposition of C3a in liver by immunohistochemistry detected with DAB kit. (A) Blank control group, no deposition of C3a; (B) solvent control group, no deposition of C3a; (C) 24-hour sensitization-positive group, apparent C3a deposition; (D) 24-hour sensitization-negative group, less C3a deposition; (E) 48-hour sensitization-positive group, more C3a deposition; (F) 48-hour sensitization-negative group, less C3a deposition; (G) 72-hour sensitization-positive group, strong deposition of C3a compared with blank control and solvent control group; (H) 72-hour sensitization-negative group, less deposition of C3a; (I) 7-day sensitization-positive group, less strong deposition of C3a; (J) 7-day sensitization-negative group, no deposition of C3a (magnification 400×). DAB indicates 3, 3′-diaminobenzidine.
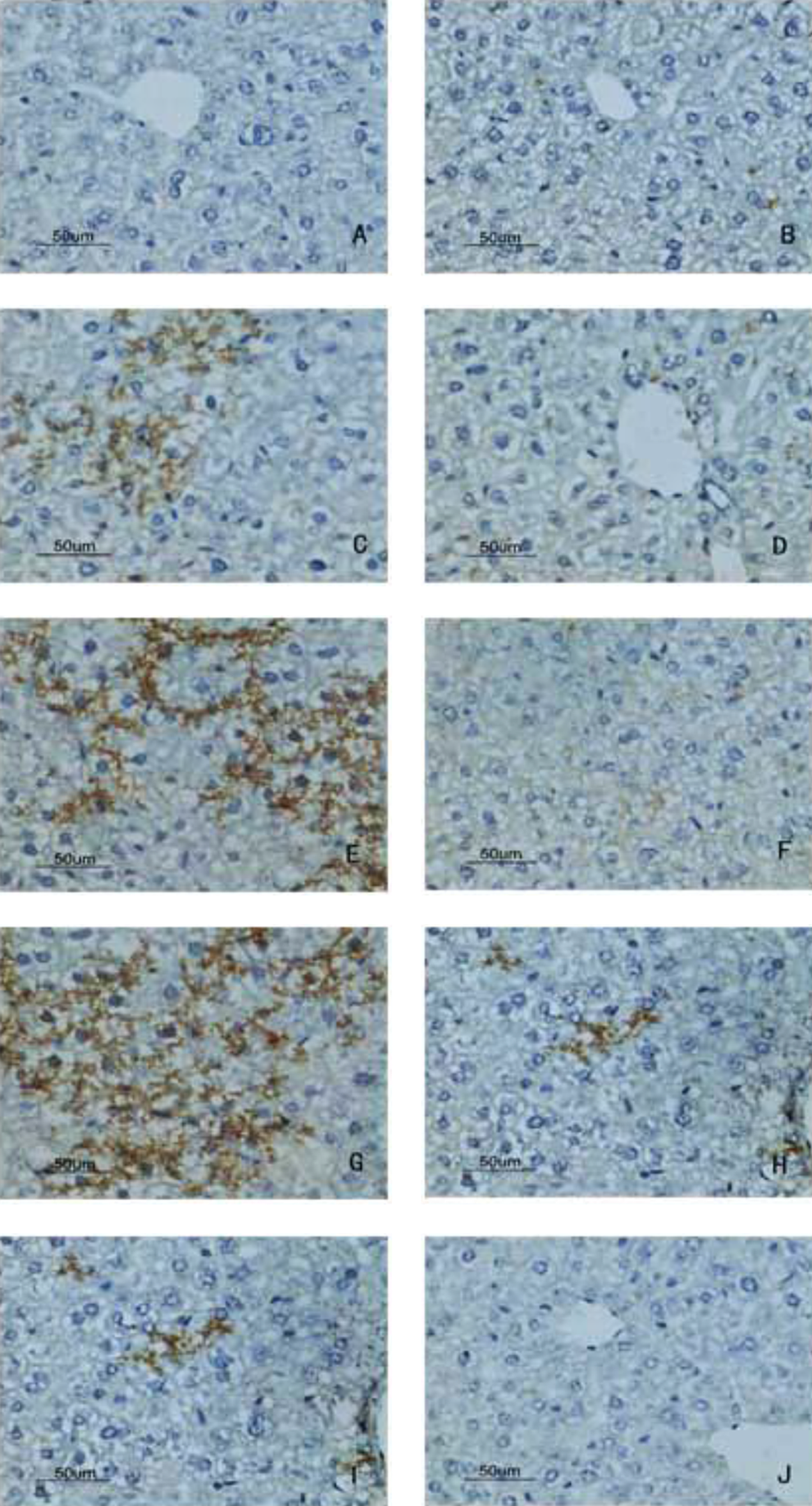
Deposition of C5a in liver by immunohistochemistry detected with DAB kit. A, Blank control group, no deposition of C5a; (B) solvent control group, no deposition of C5a; (C) 24-hour sensitization-positive group, apparent C5a deposition; (D) 24-hour sensitization-negative group, no C5a deposition; (E) 48-hour sensitization-positive group, more C5a deposition; (F) 48-hour sensitization-negative group, less C5a deposition; (G) 72-hour sensitization-positive group, strong deposition of C5a compared with blank control group and solvent control group; (H) 72-hour sensitization-negative group, less deposition of C5a; (I) 7-day sensitization-positive group, less strong deposition of C5a; (J) 7-day sensitization-negative group, no deposition of C5a (magnification ×400). DAB indicates 3, 3′-diaminobenzidine.
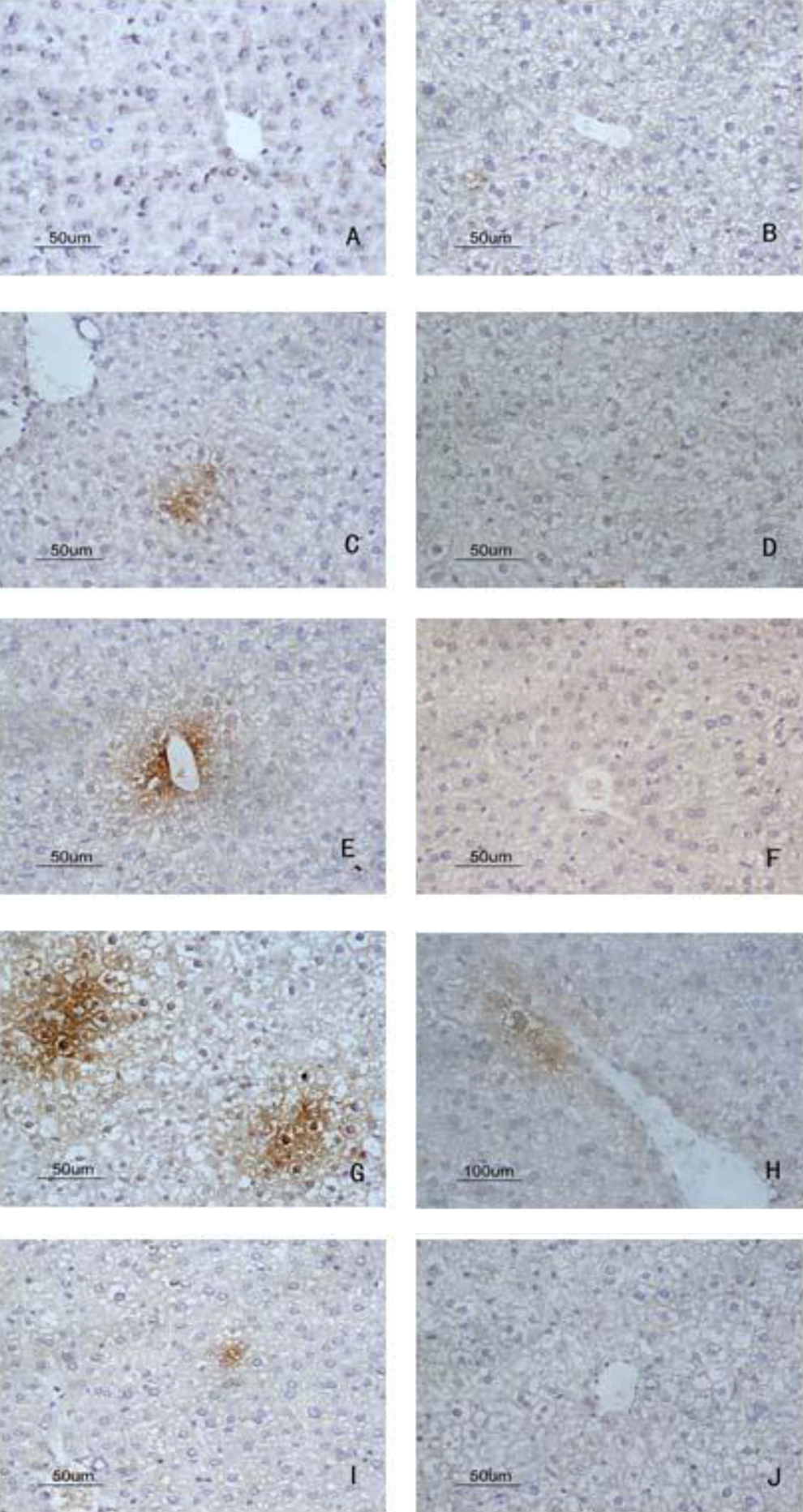
Deposition of C5b-9 in liver by immunohistochemistry detected with DAB kit. A, Blank control group, no deposition of C5b-9; (B) solvent control group, no deposition of C5b-9; (C) 24-hour sensitization-positive group, apparent C5b-9 deposition; (D) 24-hour sensitization-negative group, no C5b-9 deposition; (E) 48-hour sensitization-positive group, more C5b-9 deposition; (F) 48-hour sensitization-negative group, less C5b-9 deposition; (G) 72-hour sensitization-positive group, strong deposition of C5b-9 compared with blank control group and solvent control group; (H) 72-hour sensitization-negative group, less deposition of C5b-9; (I) 7-day sensitization-positive group, less strong deposition of C5b-9; (J) 7-day sensitization-negative group, no deposition of C5b-9 (magnification ×400). DAB indicates 3, 3′-diaminobenzidine.
Immunofluorescent Study for C5b-9
Deposition of C5b-9, the key complement complex responsible for many downstream inflammatory effects, was further investigated by immunofluorescence staining. Depositions of C5b-9 occurred consistently in the liver from BALB/c mice treated with TCE. As in paraffin-embedded tissue samples using immunohistochemical technique described above, there was extensive C5b-9 deposition in the liver from 24-hour, 48-hour, 72-hour sensitization-positive groups, peaking at 72 hours (Figure 6). In contrast, 24-hour, 48-hour, 72-hour, and 7-day sensitization-negative groups did not exhibit any visible deposition of C5b-9 in the liver (data not shown). Hence, specific deposition of the key complement complex in the liver was confirmed by fluorescence imaging.
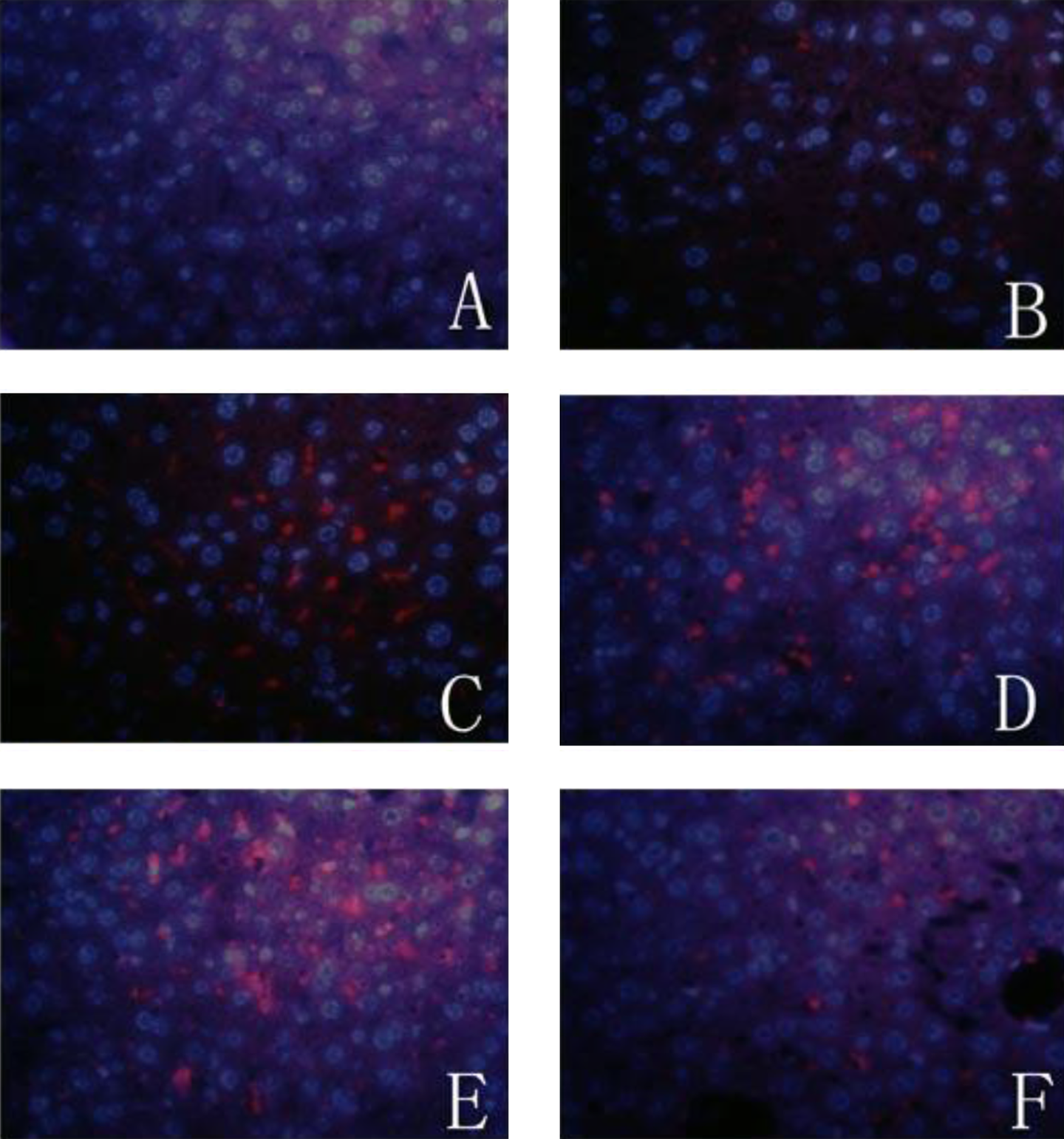
Expression of C5b-9 in liver by immunofluorescence labeled with PE. A, Blank control group, no deposition of C5b-9 in liver; (B) solvent control group, no depositio of C5b-9; (C) 24-hour sensitization-positive group, weak expression of C5b-9 in liver; (D) 48-hour sensitization-positive group, intermediate expression of C5b-9 in liver; (E) 72-hour sensitization-positive group, strong expression of C5b-9 in liver; (F) 7-day sensitization-positive group, weak expression of C5b-9 in liver (magnification 400×). PE indicates phycoerythrin.
Reverse Transcription-PCR Study for C3
To determine whether there was increased production of complements via upregulated transcription, we measured C3 mRNA expression in liver by reverse transcription-PCR. The expression of C3 mRNA was detected in the liver. Gray scale analysis showed that compared to blank control (n = 6), solvent control (n = 6), and all the sensitization-negative groups (n = 23), C3 mRNA expression was significantly increased in the 24-hour sensitization-positive group and the 48-hour group (P < .05; Figure 7). There was a transient reduction in C3 mRNA expression at 72 hours (P < .05), but it was increased again at day 7 (P < .05). The expression of sensitization-negative groups was similar to control groups, thus the data were not shown. These data provide evidence for an upregulation of complement mRNA expression.
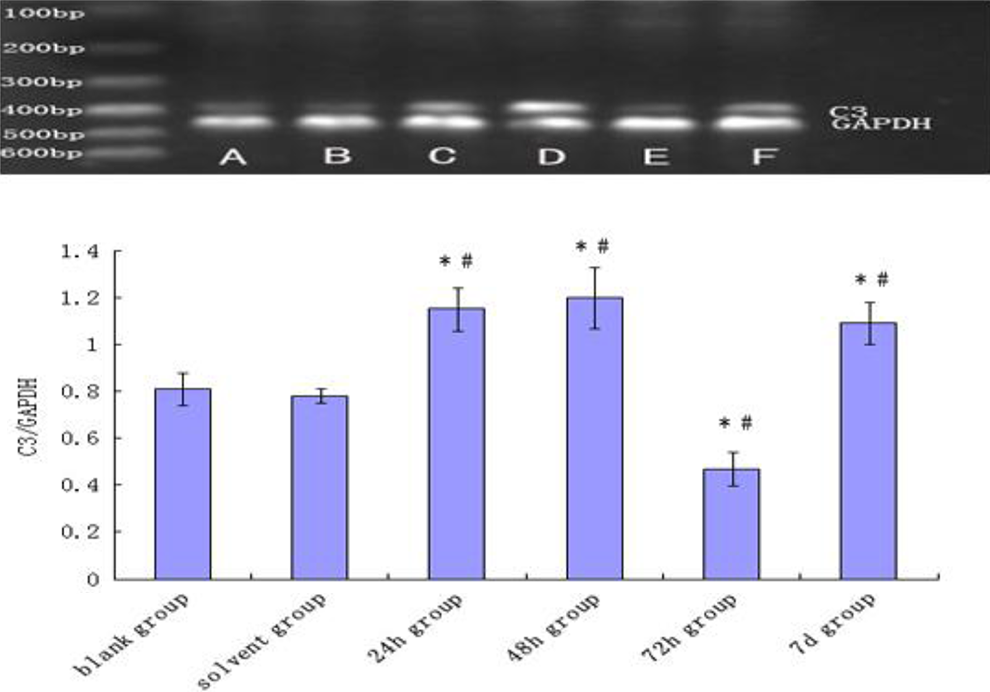
The expression of C3 mRNA in BALB/c mice liver after provocation. RT-PCR products for C3 mRNA, from left to right: Maker (A) blank control group, (B) solvent control group, (C) 24-hour sensitization-positive group, (D) 48-hour sensitization-positive group, (E) 72-hour sensitization-positive group, and (F) 7-day sensitization-positive group. The expression of sensitization-negative groups was similar to control groups, thus the data were not shown. Data in the plot are mean ± SD. *P < .05 compared with blank control group. # P < .05 compared with solvent control group (N = 35). mRNA indicates messenger RNA; SD, standard deviation; RT-PCR, reverse transcription–polymerase chain reaction.
Discussion
The present study demonstrated a significant increase in major complement activation products in the liver tissue in TCE-sensitized mice and provides the first experimental evidence for involvement of the complement system in immune-mediated liver toxicity in response to TCE in vivo.
Occupational exposure to TCE presents a new challenge to public health in emerging industrialized countries, especially in Asia, 18 despite a better control in the established industrialized countries due to technological innovation and legislation. 9,19,20 The toxicity associated with TCE exposure has been increasingly recognized in recent years, including multiorgan impairment, such as the nervous system, skin, and liver. 21 Of particular importance are medicamentosa-like dermatitis and liver damage. 22 In contrast to the classic toxicity mediated by a direct cytotoxic effect where the severity is proportional to the dose of the chemical, TCE-induced dermatitis, and associated hepatic injury are probably mediated by an immune mechanism, such as delayed-type hypersensitivity. 8 The involvement of the immune system was supported by our previous observation that liver dysfunction and ultrastructure damage occurred in guinea pigs sensitized by TCE and the extent of injury increased gradually with time. 15
However, the immunotoxicity in double-mutant heat-labile toxin (DmLT) may not solely be mediated by type IV allergic reaction. Many studies have shown that TCE is capable of eliciting a variety of subcellular actions, including oxidative reactions leading to skin damage and hepatomegaly. 23 –25 An interesting observation is that the serum concentration of complement C3 in patients before medical treatment was significantly lower, suggesting that C3 could have been consumed due to complement activation in DmLT; consistently activated complement induced liver injury. 26
We further demonstrated in a recent study that TCE not only generated skin, liver, and kidney injury via type IV allergic dermatitis but also led to complement activation. The serum levels of Ig (IgA, IgG, and IgM) and complements (C3 and C4) decreased significantly in TCE-sensitized animals, consistent with the consumption of complements following their activation. 27 A high degree of C3 and C5b-9 deposition in the renal tubular epithelial cells in TCE-sensitized groups suggests that TCE may have induced renal tubular cell damage by excessive activation of complements. 28 The complement system is an important element for both innate and adaptive immune systems 29 and responsible for triggering inflammatory responses. 30 –32 Complement activation have diverse biological effects, including oxidative stress, cytokine release, apoptosis, tissue homeostasis, cell proliferation, and differentiation. 33 –35 The multimodal actions of complements may thus underlie hepatocyte apoptosis, necrosis, and differentiation in liver injury. Further support comes from the evidence that complement-depleted animals accumulated significantly fewer polymorphonuclear cells in the postischemic liver and sustained significantly less liver injury. 36 C5-deficient mice exhibited decreased levels of liver myeloperoxidase and serum ALT. 33
In order to ascertain a direct evidence for complement activation in liver tissue in TCE-sensitized BALB/c mice and to assess the role of excessive complement activation in immune-mediated liver injury, the current investigation examined C3a-desArg, C5a-desArg, and C5b-9 in serum and the deposition of C3a, C5a, and C5b-9 in liver tissue. In addition, we investigated serum biochemistry and relative organ weights, that are functional indicators of liver injury, and direct histopathological changes. Trichloroethylene sensitization produced hepatocyte damage and inflammatory infiltration and significantly increased serum ALT and AST and the relative liver weight. These liver functional and structural lesions were accompanied by elevated concentrations of serum C3a-desArg, C5a-desArg, and C5b-9 and direct deposition of C3a, C5a, and C5b-9 in the liver. These changes are specific to TCE sensitization, as they were not observed in sensitization-negative mice, in support of an immune mechanism. At the molecular level, TCE sensitization can stimulate the expression of compliment mRNA—supported by increased C3 mRNA expression at most time points. Thus, increased production via upregulated mRNA expression is an important mechanism for complement deposition. All these data provide compelling evidence that TCE is capable of inducing immune-mediated liver damage as a consequence of complement activation.
Koch et al reported that liver cells were relatively resistant to complement-induced lysis using cultured liver cells as a model. 37 In our model of TEC-induced immunotoxicity and liver damage, we did not observe an apparent resistance as seen in Koch’s experiment. Our earlier study indicated a correlation between complement activation and liver injury, and the present study again observed definitive liver damage—both histologically and functionally. This discrepancy may be due to the difference between in vivo and in vitro models. It may suggest that in TEC-induced complement activation in vivo, the liver resistance may not occur or TEC-mediated immunoresponse can overcome the initial intrinsic resistance of the liver cells to elicit liver damage.
Given all the above evidence, we propose that the complement system is an important factor contributing to TCE-mediated immunotoxicity in addition to type IV reaction. Such immune action contributes to the inflammatory lesions in TCE-induced liver diseases. From occupational health point of view, care should be given to repeated exposure to low doses of TCE, especially in individuals at high risk of developing a hyperactive immune response to drugs and environmental chemicals. These data also warrant further investigation into the subcellular mechanisms of TCE-mediated complement activation and the associated liver injury and other regulatory factors in this process.
In summary, our data suggest that TCE can induce liver injury via immune sensitization, mediated by excessive activation of the complement system. Such a mode of action may represent an independent contributor in TCE-induced immune dysfunction and liver injury, in particular the inflammatory damage in TCE-induced liver diseases.
Footnotes
Acknowledgments
We thank Clinical Laboratory of The First Affiliated Hospital of Anhui Medical University and Central Laboratory of The First Affiliated Hospital of Anhui Medical University for valuable help with part of our experiment. CW gratefully acknowledges support from Biotechnology and Biological Sciences Research Council (BBSRC; BB/G015554/1; BB/I025379/1).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by “National Nature Science Foundation of China (30872147)”.
