Abstract
The present study aimed to investigate the hepatotoxicity of zederone isolated from Curcuma elata in mice. Adult male mice were intraperitoneally injected with a single dose of zederone (50-300 mg/kg body weight [BW]). Twenty-four hours after the injection, zederone induced liver enlargement with scattered white foci over the organ. The medium lethal dose (LD50) value at 24 hours of zederone was approximately 223 mg/kg BW. Hepatic centrilobular necrosis with marked increases in plasma alanine transaminase activity and total bilirubin levels was observed. Zederone at a dose of 200 mg/kg BW markedly decreased the activity of superoxide dismutase and the hepatic glutathione content, whereas the activity of catalase was not altered. The compound at this dose also increased the messenger RNA (mRNA) expression of Cyp2b10 and Cyp3a11, which are the main drug-metabolizing enzymes in the liver. The mRNA expression of proinflammatory cytokine tumor necrosis factor α was increased. The nuclear factor-E2-related factor 2 protein, which is the transcription factor regulating the antioxidant gene expression, was decreased. The histopathology of massive hepatic centrilobular necrosis with an increase in the expression of cytochrome P450 (Cyp) suggests that the possible potentiation of zederone-induced hepatotoxicity implicated the induction of Cyps, which leads to the formation of biological reactive metabolites and that cause the oxidative stress and liver cell injuries.
Introduction
Sesquiterpenes are a group of plant-derived compounds consisting of 3 isoprenoid units. 1 A variety of sesquiterpene skeletons have been found in Curcuma species of Zingiberaceae, which are widely distributed in tropical regions, especially in Southeast Asian countries, and they are widely used as dietary supplements and traditional medicines for the treatment of many diseases. 2 Sesquiterpenes from Curcuma zedoaria Roscoe show a variety of pharmacological properties, including anti-inflammation, antioxidation, and vasodilatation. 2 –5 However, in the course of our recent study on the botanical taxonomy and characterization of phytochemical and pharmacological diversities of Curcuma sp, we found that Curcuma elata Roxb rhizome contained a high proportion of sesquiterpene zederone, and the hexane extract of the rhizome from C elata caused liver enlargement and scattered white foci over the organs at autopsy (unpublished data).
Zederone is a germacrane-type sesquiterpene, which was first reported in Zedoaria rhizome. 6 Most of the plants in Curcuma species contain this compound in trace amount. C zedoaria has been reported to contain zederone at approximately 0.0052% of the air-dried rhizome. 7 The compound was potently cytotoxic against human white blood cancer cells 8 and human prostate cancer cells. 9 However, to date, the available scientific information on the biological effects of zederone in vivo has been limited. In a viewpoint of our finding, plants containing this compound are potently toxic to animals. With the greater use of herbal remedies, the risks of liver injury are also increasing. Therefore, the present study aimed to investigate the potential hepatotoxicity of zederone isolated from C elata in mice. Our results revealed that zederone potently induced hepatotoxicity in mice by causing centrilobular necrosis.
Liver is the primary site for xenobiotic metabolism and biotransformation. 10 In the process of detoxification, many xenobiotics form bioreactive intermediates, which are toxic metabolites and may cause liver injury, particularly in the centrilobular regions. 11 Hepatocytes in different regions of the liver exhibit differential susceptibility to toxins; thus, the site of hepatocyte injury is also an important determinant. 12 The centrilobular zone is the most susceptible to chemical-induced liver injury, particularly chemicals that are directly biotransformed to toxic metabolites in the liver as the mature hepatocytes having high activities of cytochrome P450 (Cyp) enzymes are located in this region. The biotransformation of some chemicals by Cyps may generate bioreactive metabolites with high electrophilic properties, thus leading to oxidative stress and cytotoxicity. 13 The oxidative stress is a condition of an imbalance between pro-oxidant and antioxidant systems. 14,15 In healthy liver, the reactive oxygen species are scavenged by various antioxidant mechanisms, including superoxide dismutase (SOD) and catalase (CAT). The SOD converts superoxide radicals into oxygen and hydrogen peroxide (H2O2), and the CAT converts H2O2 into water and oxygen. Reduced glutathione (GSH) is the main intracellular antioxidant found in liver cells and is able to scavenge electrophilic molecules. 16,17 In the case of chemical-induced liver injury associated with oxidative stress, the centrilobular hepatocytes would encounter a greater degree of oxidative stress and may deplete the antioxidant system. 14 The intracellular antioxidant responses are mainly regulated by a transcription factor nuclear factor-E2-related factor 2 (Nrf2) that acts as a sensor for oxidative or electrophilic stress. 18 Under basal condition, Nrf2 is ubiquitously expressed in the cytoplasm, and when exposed to oxidative stress, Nrf2 is stabilized and translocates into the nucleus to bind to the antioxidant responsive element (ARE). The Nrf2 subsequently coordinates with other transcription factors and initiates the transcription of antioxidant genes. 19
To our knowledge, the available information concerning the toxicity of zederone in vivo is limited. Therefore, the present study aimed to evaluate the potential hepatotoxicity of zederone in mice.
Materials and Methods
Chemicals
Dinitrophenylhydrazine, α-ketoglutamic acid,
Isolation of Zederone
Zederone was isolated from rhizomes of C elata, which were collected from Sawang Daen Din district, Sakon Nakhon province, Thailand. The rhizome of C elata was subjected to taxonomic identification by Soontornchainaksaeng and Jenjittiku. 20 Curcuma elata contains chromosome number of 2n = 63. The plant has been characterized by a long peduncle and a long cylindrical spike branching out from the main rhizome with wide angle. Voucher specimen (SCMU No. 305) was deposited at the Department of Plant Science, Faculty of Science, Mahidol University, Bangkok. For extraction and isolation of the compound, the rhizomes of C elata were cut into small pieces, dried, and grounded to powder, and a portion (8.5 kg) was extracted successively by maceration with n-hexane to obtain, after evaporation of the solvents, the hexane extract (brownish viscous oil, 129.7 g). The chemical constituents and qualitative phytochemical screening of C elata hexane extract were conducted. The hexane extract (129.0 g) was fractionated by silica gel column chromatography (Merck silica gel 60 PF254, 250 g, Merck, Darmstadt, Germany) using n-hexane–ethyl acetate as eluent. Recrystallization of the eluate with ethyl acetate in n-hexane brought about zederone (Figure 1) as colorless needles (11.85 g; 10% of the hexane extract), melting point 153 to 154°C. Thin-layer chromatography (TLC) investigation of the hexane extract revealed 1 distinct spot and several minor components. In addition, the chromatogram of C elata hexane extract obtained using high-performance liquid chromatography revealed the major peak of zederone using ultraviolet detector at wave length 280 nm. The isolated zederone was assessed by TLC and nuclear magnetic resonance spectroscopy and was found to be approximately 99% pure.
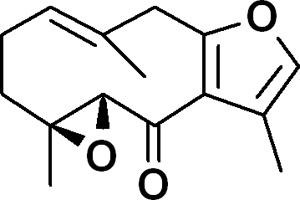
Chemical structure of zederone, a major sesquiterpene compound isolated from Curcuma elata Roxb hexane extract.
Animal Treatment
Adult male ICR mice weighing 30 to 35 g (approximately 6-8 weeks of age) were obtained from the National Laboratory Animal Centre of Thailand (Salaya, Nakhon Pathom, Thailand). Animals were maintained under standard conditions of temperature (25°C ± 2°C), relative humidity (50%-60%), and light (12-hour light/dark cycle). All mice had free access to standard pellets (C.P. Mice feed; Pokphand Animal Fed Co Ltd, Bangkok, Thailand) and water. They were allowed to acclimatize for 1 week before the experiment. The experimental animal protocol was approved by the Animal Care and Use Committee of the Faculty of Science, Mahidol University.
Acute toxicity of intraperitoneal injection of zederone at 24 hours was investigated. Mice were randomly assigned into 6 groups of 10 animals each. They received a single intraperitoneal injection of zederone at 50, 100, 150, 200, and 300 mg/kg body weight (BW). Animals were closely observed for signs of toxicity during the first hour after dosing and at 2, 4, and 24 hours afterward. The rate of mortality was recorded. All animals were euthanized at 24 hours after the injection. At autopsy, visceral organs were collected and weighed for possible toxic effect. Medium lethal dose (LD50) value was calculated using Probit method. Blood samples were collected from posterior abdominal vein, and the liver was rapidly removed, weighed, and kept for histopathological examinations and biochemical analyses. The histopathological lesions were graded by estimating the percentage of affected area from the entire section area. Severities of the lesions were graded according to Lobenhofer et al 21 (+ = minimal [<5%], ++ = mild [5%-25%], +++ = moderate [26%-50%], and ++++ = marked [>50%]). For biochemical analysis, a portion of liver was rapidly rinsed with cold 0.9% NaCl and was frozen in liquid nitrogen and stored at −80°C for RNA extraction. Cytosolic fractions were prepared by centrifugation at 100 000g for 60 minutes.
Biochemical Analysis
Activity of plasma alanine transaminase (ALT) was determined according to Reitman and Frankel. 22 The activities of SOD and CAT in the cytosol were assessed according to the method previously described by Winterbourne et al and Lück, respectively. 23,24 The hepatic GSH content was determined according to the method of Theodorus et al. 25 Total amount of proteins was measured by bicinchoninic acid (BCA) protein assay. 26 Plasma total bilirubin (TBIL) was also analyzed using commercial diagnostic kit (BM Lab, Bangkok, Thailand).
Isolation of Total RNA and Quantitative RT-PCR Analysis
Mouse liver RNA was extracted using TRIzol Reagent according to the manufacturer’s instruction and stored at −80°C. For real-time PCR, analysis was performed in 2 steps by initial reaction with reverse transcription of total RNA using iScript Select cDNA Synthesis Kit. Then, cDNA was mixed with SYBR Green PCR Master Mix. Quantitative real-time PCR was carried out with the ABI PRISM 7500 Sequence Detection System (Applied Biosystem, Bedford, Massachusetts). The PCR amplification was performed for mouse Cyp2b10, Cyp3a11, Cyp2e1, and tumor necrosis factor α (TNF-α). Expression ratios, relative to a calibrator (vehicle control), were determined by quantification of cDNA according to the mathematical model described by Pfaffl, 27 in which the ratio = 2−(ΔCtSample − ΔCtcontrol). For the RT-PCR, an equivalent amount (400 ng) of RNA from mouse liver was subjected to RT-PCR. OneStep RT-PCR system (QIAGEN, Düsseldorf, Germany) was used following the manufacturer’s protocol. The PCR products were visualized on a 1.5% agarose gel stained with ethidium bromide. Gel images were scanned using an image analysis system (Syngene chemi Geius, Cambridge, United Kingdom). The intensity of specific PCR band was quantified in relation to β-actin bands, which were amplified from the same mouse template cDNA. The following sequences of primers were used:
β-actin: Fw: 5′-GAGGGAAATCGTGCGTGACAT-3′
and Rv: 5′-ACATCTGCTGGAAGGTGGACA-3′;
TNF-α: Fw: 5′-AACTTCGGGGTGATCGGTCC-3′
and Rv: 5′-TGGGGGCTGGGTAGAGAATG-3′;
GAPDH: Fw: 5′-CGAGACCCCACTAACATCAAA-3′
and Rv: 5′-TTTGGCTCCACCCTTCAA G-3′;
Cyp3a11: Fw: 5′-GGAAATTCGACATGGAGTGC-3′
and Rv: 5′-ATCGTCTCTGGGTCTGTGA-3′;
Cyp2b10: Fw: 5′-TTCCTGCTCATGCTCAAGTAC-3′
and Rv: 5′-CTCTGAATCTCGTGGATGACTG-3′;
Cyp2e1: Fw: 5′-TTCCTGCTCATGCTCAAGTAC-3′
and Rv: 5′-CTCTGAATCTCGTGGATGACTG-3′.
Isolation of Total Protein and Western Blot Analysis for Nrf2
Total protein was prepared from 100 mg of liver tissue. Briefly, the tissues were immersed in Pierce radio immunoprecipitation assay buffer (Thermo Scientific, Rockford, Illinois) containing protease inhibitor cocktail (AMRESCO, Ohio) and were homogenized with TissueLyser LT (QIAGEN, Hilden, Germany) at 50 Hz for 3 minutes. Samples were incubated on ice for 20 minutes before being centrifuged at 12 000 rpm for 20 minutes. The supernatant was then collected and stored at −80°C until use. Protein concentrations of samples were determined using BCA protein assay kit (Thermo Scientific). Western blot analysis was conducted using 100 µg protein of each sample fractionated by sodium dodecyl sulfate polyacrylamide gel electrophoresis on 5% to 10% gradient gel before transfer to a nitrocellulose membrane (Bio-Rad Laboratories). The membrane was blocked with 5% nonfat dry milk in Tris-buffered saline solution with Tween-20 (TBS-Tween), probed with the Nrf2 primary antibody (C-20; Santa Cruz Biotechnology, California) for overnight, at 4°C and was subsequently washed with TBS-Tween. After washing, membrane was incubated with goat polyclonal secondary antibody to rabbit immunoglobulin G (HRP; Abcam, Massachusetts) for 1 hour at room temperature. The membrane was washed with TBS-Tween, and protein bands were visualized using a SuperSignal West Pico Chemiluminescent detection system (Thermo Scientific) and captured by Hyperfilm (Amersham, Rockford, Illinois). Bands were quantitated by densitometric analysis using ImageJ densitometry software.
Statistical Analysis
Data are expressed as means ± standard error of means. The statistical difference was analyzed using 1-way analysis of variance, followed by Dunnett test and, where appropriate, Student t test. Values of P < 0.05 and P < 0.01 were considered to be significantly different.
Results
Toxicity of Zederone in Mice
Toxicity of zederone was evaluated at 24 hours after a single intraperitoneal injection. Mice treated with zederone at a dose of 100 mg/kg BW or above showed several toxic signs that progressed from a decrease in locomotor activity, tremor, weakness of hind limbs (paraplegia), diarrhea, and drowsiness to, eventually, death. Mortality was increased proportionally with the dose. Using Probit method, 28,29 the linear dose–effect relationship was determined, and the LD50 value of zederone was estimated to be 223 mg/kg BW (Table 1). On the day of euthanasia, the liver of mice treated with zederone at doses of 100 mg/kg BW or above showed scattered white foci with a marked increase in liver weights, indicating that liver is a target of the compound toxicity. As shown in Table 2 and Figure 2, the histopathological features of the mouse liver were graded. In control mice, the well-defined architecture with hepatic central vein, portal tracts, and hepatic cords separated by blood sinusoid was observed. However, 24 hours after zederone treatment at a dose of 50 mg/kg BW, small foci of hydropic degeneration of hepatocytes encircling the central vein were seen. More extensive centrilobular necrosis with inflammation and hemorrhage was noticeable by increasing the dose of zederone. At a dose of 200 mg/kg BW, hepatocytes necrosis extended toward the portal area and was accompanied by prominent hemorrhage. Only 3 of the 10 mice survived at the dose of 300 mg/kg BW, but the livers of these mice showed similar histopathological features to those treated at a dose of 100 to 200 mg/kg BW; their biochemical data were not explored.
Intraperitoneal LD50 Values of Zederone in Mice.a
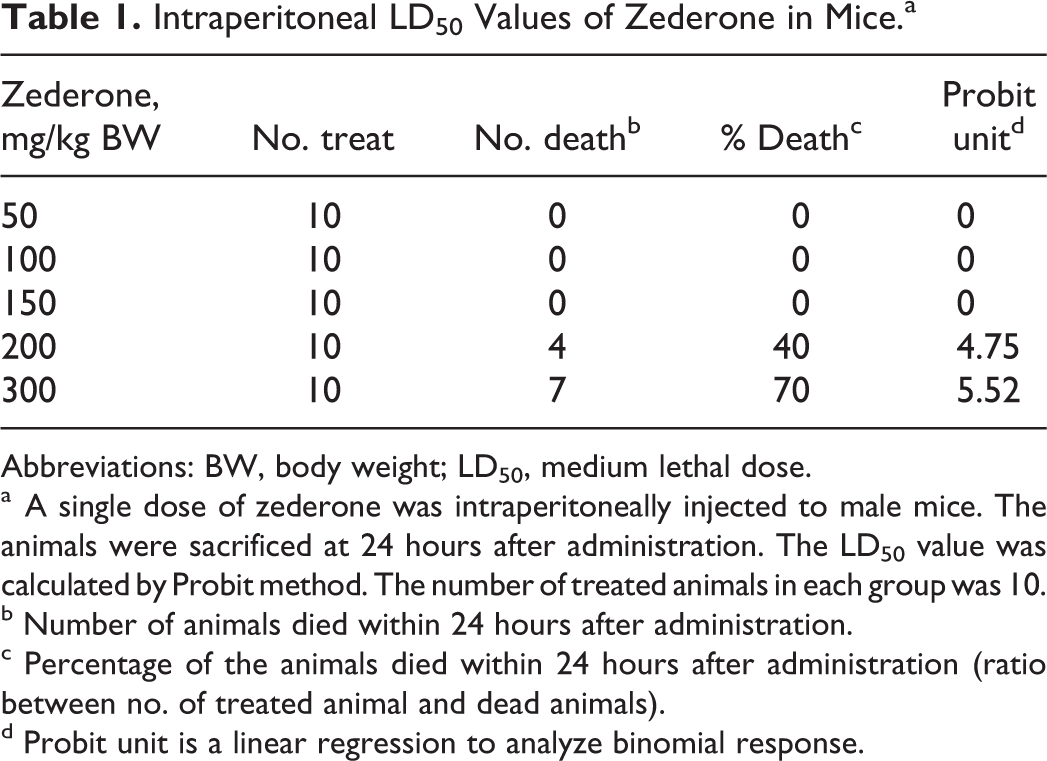
Abbreviations: BW, body weight; LD50, medium lethal dose.
a A single dose of zederone was intraperitoneally injected to male mice. The animals were sacrificed at 24 hours after administration. The LD50 value was calculated by Probit method. The number of treated animals in each group was 10.
b Number of animals died within 24 hours after administration.
c Percentage of the animals died within 24 hours after administration (ratio between no. of treated animal and dead animals).
d Probit unit is a linear regression to analyze binomial response.
Histologic Grading of Liver Lesions.

Abbreviations: BW, body weight; +, minimal (<5%); ++, mild (5%-25%); +++, moderate (26%-50%); ++++, marked (>50%).
a The lesions were graded by estimating the percentage of affected area from the entire section area.
b Severities of lesions were graded according to Lobenhofer et al. 21
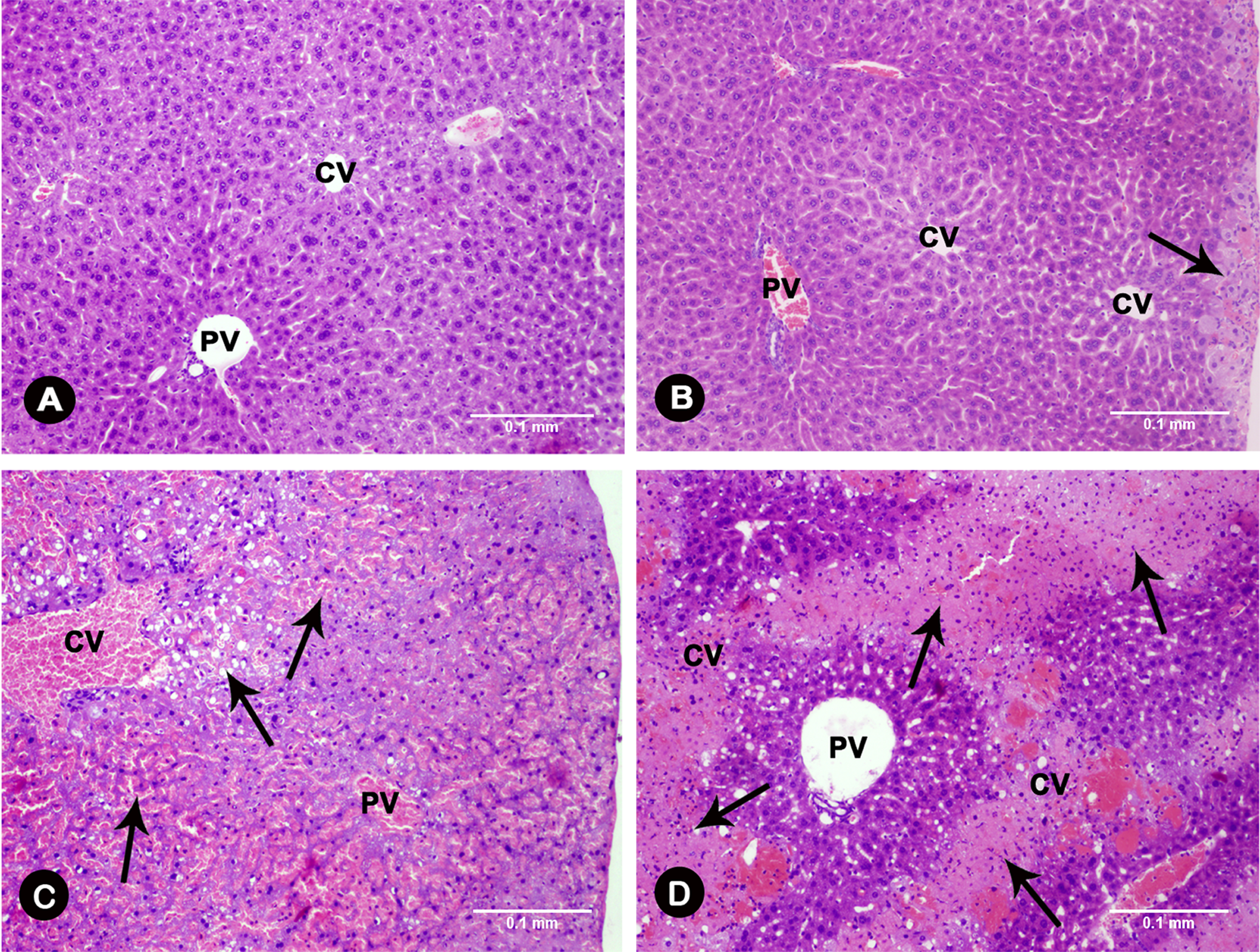
Effect of zederone on liver histopathology. Mice were intraperitoneally injected with a single dose of zederone and euthanized at 24 hours after administration. A, Control group; (B) zederone treatment at a dose of 50 mg/kg BW; (C) 100 mg/kg BW; and (D) 200 mg/kg BW. Hematoxylin and eosin staining, magnification ×100. Arrows indicate hepatocyte injuries showing the small foci of hydropic degeneration (B), extensive centrilobular necrosis with inflammation and hemorrhage (C), and the massive hepatocytes necrosis with prominent hemorrhage (D). The bar length of 0.1 mm was applied to all panels. BW indicates body weight; CV, central vein; PV, portal vein.
Biochemical analysis of plasma revealed that zederone induced a dose-dependent increase in plasma ALT activity, which was highly elevated from 15.4 ± 2.1 IU/L in control group to 106.8 ± 10.0 IU/L (P < 0.05), 373.6 ± 55.6 IU/L (P < 0.05), 2991.0 ± 105.8 IU/L (P < 0.01), and 4553.0 ± 435.2 IU/L (P < 0.01) in animals treated at doses of 50, 100, 150, and 200 mg/kg BW, respectively. The TBIL was modestly increased by 2-fold from 0.8 ± 0.1 mg/dL in the control group to 1.6 ± 0.1 mg/dL (P < 0.01) at a dose of 200 mg/kg BW (Table 3). As most data obtained at 150 mg/kg BW were similar to those of zederone at 200 mg/kg BW, further investigations on their cellular and molecular mechanism were conducted at doses of 50, 100, and 200 mg/kg BW.
Effects of Zederone on Body and Liver Weights, Plasma ALT Activity, and Total Bilirubin (TBIL) in Mice.a
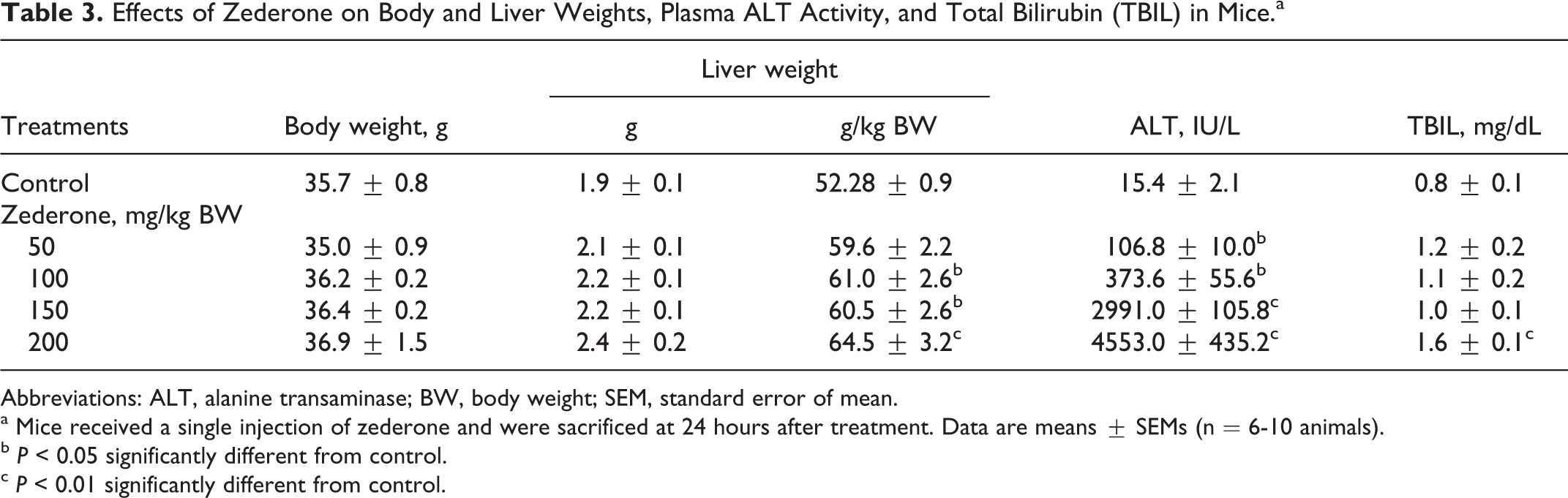
Abbreviations: ALT, alanine transaminase; BW, body weight; SEM, standard error of mean.
a Mice received a single injection of zederone and were sacrificed at 24 hours after treatment. Data are means ± SEMs (n = 6-10 animals).
b P < 0.05 significantly different from control.
c P < 0.01 significantly different from control.
Effects of Zederone on the Intracellular Antioxidant System
To investigate whether the observed histopathology of hepatocyte necrosis by zederone was associated with the oxidative stress, the effect of zederone on the intracellular antioxidants was determined. As shown in Figure 3, all doses of zederone (50, 100, and 200 mg/kg BW) markedly suppressed the activity of antioxidant enzyme SOD (P < 0.01), but they did not affect the activity of CAT. Low doses of zederone (50 and 100 mg/kg BW) caused an increase in the hepatic GSH content. However, a higher dose of zederone at 200 mg/kg BW markedly decreased the GSH content. Similar to the changes in GSH content, Nrf2 protein, which is a sensor for oxidative stress, was increased by the low dose and was decreased by a high dose of zederone. These findings suggest that liver cells are under oxidative stress.
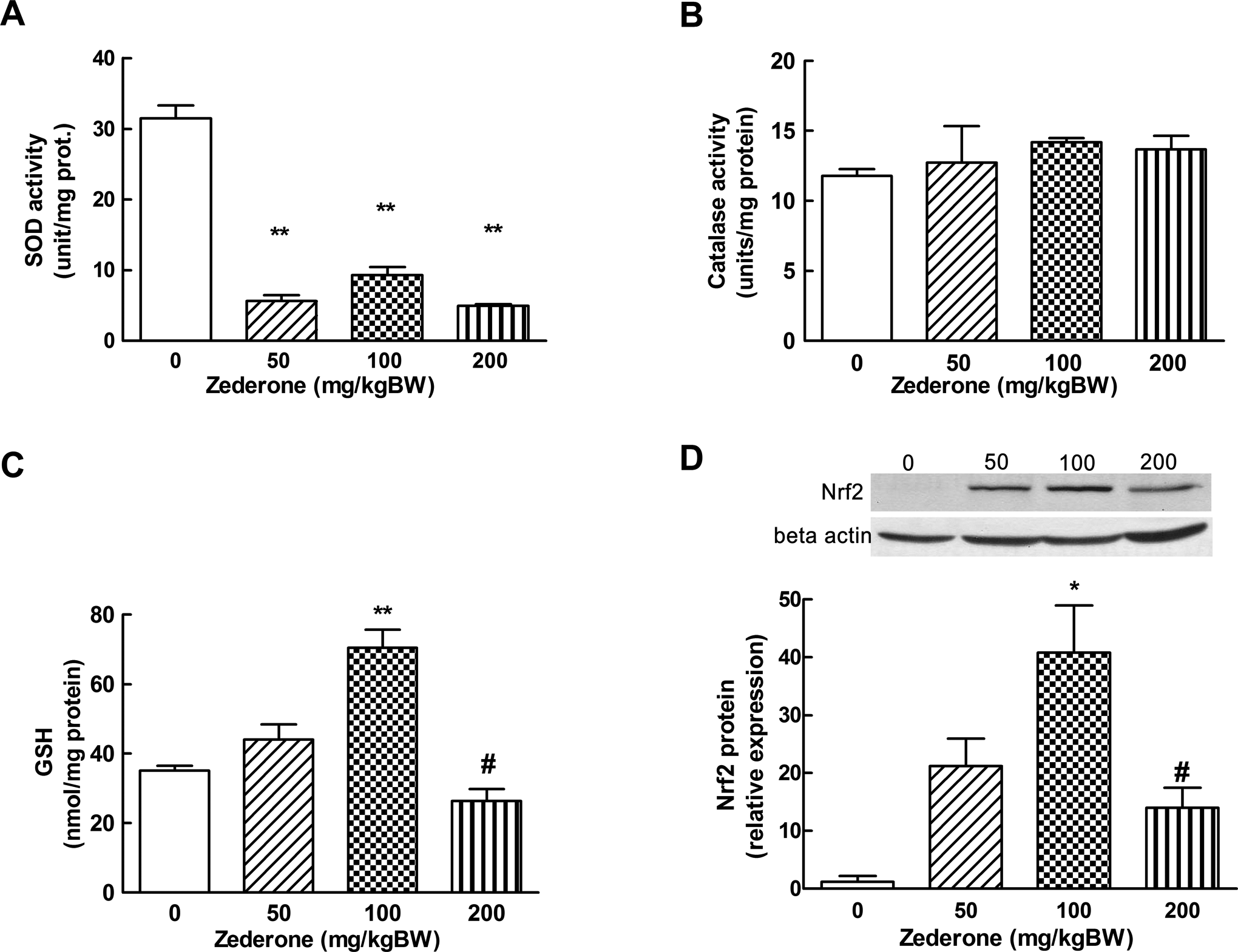
Effects of zederone on the liver antioxidant system. A, Superoxide dismutase (SOD) activity; (B) catalase (CAT) activity; (C) glutathione (GSH) level, and (D) the expression of Nrf2 protein. Mice received intraperitoneal injection of zederone at the doses 50 to 200 mg/kg BW and were euthanized at 24 hours after administration. Cytosolic fraction prepared from the mouse liver was used for enzyme activity analysis. Total liver protein was prepared to investigate the level of Nrf2 expression. Each value is mean ± standard error of mean (SEM) from 4 to 5 animals. *P < 0.05 and **P < 0.01, significantly different from control. # P < 0.05, significantly different from a dose of 100 mg/kg BW. BW indicates body weight; Nrf2, nuclear factor-E2 related factor 2.
Effect of Zederone on Cyps and Inflammatory Mediator
To investigate whether the specific zonal damage in the centrilobular area by zederone affected the activities of Cyps, the expression of some Cyp enzymes, which are implicated in xenobiotic metabolism, was determined. As shown in Figure 4, zederone showed dose-related increases in the messenger RNA (mRNA) expressions of Cyp2b10 and Cyp3a11. However, the statistical significance was detected only at a dose of 200 mg/kg BW, where the Cyp mRNAs were increased about 2.5-fold. Zederone did not significantly alter the mRNA expression of Cyp2e1, although there was a trend toward a decreased at a dose of 200 mg/kg BW. As zederone treatment caused massive hepatocytes necrosis, the involvement of zederone in the induction of inflammatory mediators was also investigated. Zederone at low doses (50-100 mg/kg BW) slightly increased the mRNA expression of proinflammatory mediator, TNF-α (Figure 4D). However, zederone at 200 mg/kg BW markedly increased the expression level of TNF-α up to 4.9 ± 1.4-fold.
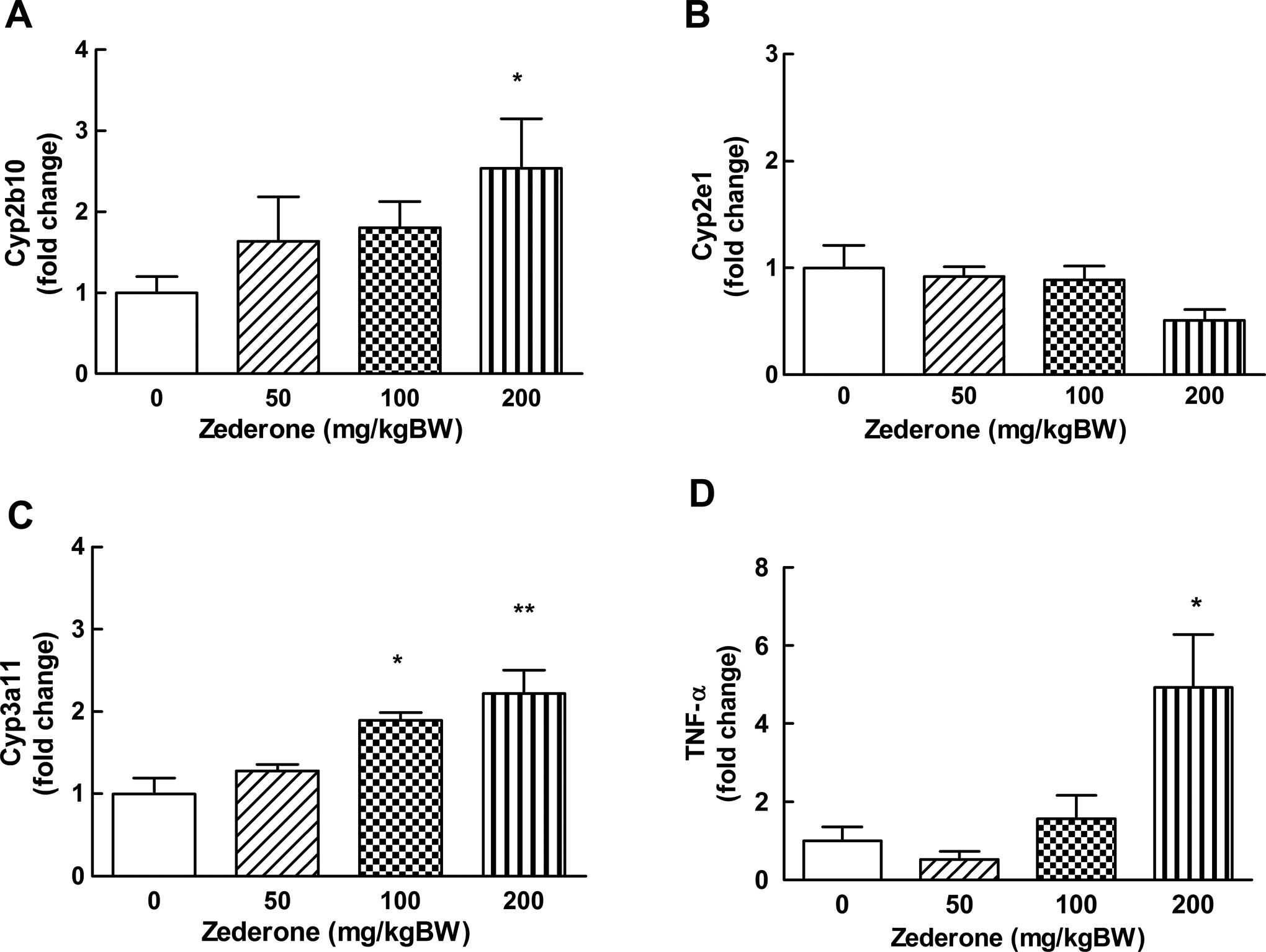
Effects of zederone on the hepatic expression of cytochrome P450; (A) Cyp2b10, (B) Cyp2e1, and (C) Cyp3a11. The mRNA expressions of TNF-α (D), an inflammatory mediator is also presented. Total liver RNA was prepared from mice at 24 hours after intraperitoneal injection with zederone at the indicated doses. The mRNA expression was determined by real-time RT-PCR. Each value is mean ± standard error of mean (SEM) from 4 to 5 animals. *P < 0.05 and **P < 0.01, significantly different from control. mRNA indicates messenger RNA; RT-PCR, reverse transcription-polymerase chain reaction; TNF-α, tumor necrosis factor α.
Discussion
The present study demonstrated the hepatotoxicity of zederone, a sesquiterpene epoxide from C elata. Its hepatotoxicity was characterized by extensive centrilobular injuries with marked increases in plasma ALT activity and TBIL concentration and decreases in hepatic SOD activity. In addition, treatment with a high dose suppressed the intracellular hepatic GSH content and protein expression of Nrf2, a sensor for oxidative stress. The mRNA expression of Cyps, which are mainly involved in xenobiotic metabolism, and the proinflammatory mediator, TNF-α, was also increased. Zederone is a ketoepoxide compound that has been characterized as an unsaturated molecule and is able to generate superoxide radicals, causing toxicity by binding directly to cellular macromolecules. 30 –32 In our recent study in vitro, zederone markedly increased the expressions of CYP2B6 and CYP3A4 genes in human primary hepatocytes. In addition, after incubation of zederone with human liver microsomes and NADPH, 1 highly reactive, electophilic metabolite, a diepoxide sesquiterpene, was formed, which suggested to be resulted from CYP-mediated oxidation. Using GSH trapping, 5 GSH adducts were detected. 33 Thus, hepatotoxicity of zederone in mice in the present study is possibly mediated through the formation of bioactive metabolites that generate highly reactive metabolite and deplete intracellular antioxidant capacity, thereby causing hepatocentrilobular cell injury. To our knowledge, this is the first report on the hepatotoxicity of zederone in an in vivo system.
In the present study, centrilobular necrosis induced by zederone was correlated with the marked increases in plasma ALT activity and TBIL. The increased level of plasma ALT indicated damage to the liver cell membrane, whereas the increased TBIL may reflect impairment of the conjugation function of the liver. 34 (Pratt and Kaplan 2000) However, the extent of the increase in TBIL was less than that of ALT (Table 3). This may be accounted for by the short period of our observation. At 24 hours after treatment, the alteration of liver cell function might not be as great as the cell membrane damage. The discrepancy between the increased ALT and TBIL has also been reported in the malathion-induced hepatotoxicity in which the plasma TBIL was found to be time dependent, gradually increasing with time after exposure to the toxic compound. 35 This might be the case of the present study.
In the present study, zederone selectively induced damage in the centrilobular zone (Figure 2), which is rich in hepatic-metabolizing enzyme Cyps, 10 suggesting that Cyps might be affected following zederone treatment. Indeed, zederone at a high dose markedly induced the mRNA expression of Cyp2b10 and Cyp3a11 in mice, which are the homologs of human CYP2B6 and CYP3A4, respectively, 36 and they are mainly responsible for metabolizing a large number of therapeutic drugs. 37 This finding on the induction of Cyps was consistent with the result in the in vitro study. 33 It is possible that the induction of Cyp enzymes in mice would lead to the formation of biologically reactive metabolites that potentially caused oxidative stress to the cell as reported. 33 During oxidative stress in cells, the unopposed highly reactive radicals could lead to increase in inflammation. An increase in TNF-α, a proinflammatory cytokine, was evident in the present study and has also been reported in other studies. 38,39 The altered GSH level here supports the notion that hepatic cells are under oxidative stress (Figure 3). However, zederone at a low dose increased the level of GSH, which might be attributed to the adaptive response to sublethal doses of oxidative stress. Similar changes in GSH in response to different doses of toxicants have been reported previously, where the sublethal concentration of 2,3-dimethoxy-1,4-naphthoquinone elevated GSH content, but at the higher concentration, GSH content was depleted. 40 The generation of reactive oxygen species (ROS) and oxidative stress are related to the chemical structure of zederone, which contains an epoxide moiety. It is a reactive group that is capable of binding directly to cellular GSH as reported. 33
The Nrf2 is a transcription factor responsible for the expression of several genes in antioxidant defenses and detoxification, including SOD activity and GSH content. 41 Under basal conditions, Nrf2 is ubiquitously sequestered in the cytoplasm. Once the cells are disturbed by reactive oxygen species (ROS), it translocates into the nucleus and then binds to the ARE, thereby coordinating with other transcription factors to initiate the transcription of antioxidant genes. 19 This process contributes to the intracellular protective mechanism against oxidative stress. 42 Interestingly, the pattern of alteration of intracellular GSH content and the expression of Nrf2 protein at both low and high dose of zederone were similar. This finding highlights an important role of GSH and Nrf2 to protect cells against zederone toxicity.
Curcuma elata Roxb, a Zingiberaceae plant, has rhizome morphology similar to that of Curcuma comosa Roxb, which contains phytoestrogens 9,20 and is widely used among menopausal women to relieve unpleasant symptoms. The use of C elata, which contains other compounds, as C comosa may lead to adverse effects and toxic reactions. In considering the amount of zederone to be taken by human, if C elata extract is used at the same dose as that of the C comosa extract (500-1000 mg/kg BW), 43 zederone at approximately 50 to 92 mg would be taken. The hepatotoxicity of zederone at this dose level was also evident in the present study by causing an increase in plasma activity of ALT (Table 3). This finding may indicate the contribution of zederone to the toxicity of the extract.
In conclusion, this is the first study that reveals the acute hepatotoxicity of zederone in animals by causing centrilobular necrosis in mice. The hepatotoxic effects involved the induction of Cyp enzymes and, subsequently, the generation of bioreactive metabolites, causing oxidative stress and leading to the suppression of intracellular oxidative capacity and hepatocellular injury. The present study provides important information on the potential bioactivation of plant compound to highly reactive products in the body, which could cause severe tissue damages.
Footnotes
Acknowledgment
The authors thank Prof Chumpol Pholpramool for critical reading of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by The Thailand Research Fund through the Royal Golden Jubilee PhD Program (grant no. PHD/0032/2550 to PP and PP); the National Research Council of Thailand; Faculty of Science; the Center of Excellence on Environmental Health and Toxicology, Science & Technology (PERDO), Ministry of Education, and Mahidol University under the National Research Universities Initiative (NRU).
