Abstract
Diethyl and diphenyl disulfides, naphtha sweetening (Chemical Abstracts Service [CAS] # 68955-96-4), are primarily composed of low-molecular-weight dialkyl disulfides extracted from C4 to C5 light hydrocarbon streams during the refining of crude oil. The substance, commonly known as disulfide oil (DSO), can be composed of up to 17 different disulfides and trisulfides with monoalkyl chain lengths no greater than C4. The disulfides in DSO constitute a homologous series of chemical constituents that are perfectly suited for a hazard evaluation using a read-across/worst-case approach. The DSO constituents exhibit a common mode of action that is operable at all trophic levels. The observed oxidative stress response is mediated by reactive oxygen species and free radical intermediates generated after disulfide bond cleavage and subsequent redox cycling of the resulting mercaptan. Evidence indicates that the lowest series member, dimethyl disulfide (DMDS), can operate as a worst-case surrogate for other members of the series, since it displays the highest toxicity. Increasing the alkyl chain length or degree of substitution has been shown to serially reduce disulfide toxicity through resonance stabilization of the radical intermediate or steric inhibition of the initial enzymatic step. The following case study examines the mode of action for dialkyl disulfide toxicity and documents the use of read-across information from DMDS to assess the hazards of DSO. The results indicate that DSO possesses high aquatic toxicity, moderate environmental persistence, low to moderate acute toxicity, high repeated dose toxicity, and a low potential for genotoxicity, carcinogenicity, and reproductive/developmental effects.
Introduction
The creation of chemical categories is an accepted method of grouping chemicals with similar structural, physiochemical, toxicological, or environmental properties for the purpose of hazard assessment. 1 The benefits include a reduction in animal usage, testing costs, and the time needed to conduct a hazard assessment. As such, categorical analysis has become an efficient and effective method of examining potential hazards of a host of interrelated chemicals. Guidance on the construction and use of chemical categories has been developed by several organizations including the Organisation for Economic Co-operation and Development (OECD), European Centre for Ecotoxicology and Toxicology of Chemicals (ECETOC), European Chemicals Agency (ECHA), and the United States Environmental Protection Agency (USEPA) under the High Production Volume Challenge Program (HPVCP). 2 –5 Establishment of a chemical category is predicated on the assumption that each member of the category is related in some systematic fashion and that shared molecular or physical properties play a definitive role in eliciting an adverse effect. Once created, based on similarities in chemical structure, mechanism of action and/or toxicological profile, obvious data gaps can be identified and filled using qualitative or quantitative structure activity analysis. 6
Inherent to the category approach is the understanding that a reference substance exists within the chemical category whose test results may be applied to those members lacking the requisite information needed for a complete characterization of the physical properties or hazards in a particular domain. 7 This is the so-called “read-across” provision that provides the basis for extrapolating the results from 1 member of the category to another when sufficient justification exists. This justification necessarily includes evidence that members of the chemical category operate through a common initiating event or mode of action and that response differences occur in a regular and predictive fashion. In this article, we discuss an analogous situation in which the hazards of a complex substance containing similar constituents can be assessed using a read-across/worst-case approach for the most toxic member of the series.
The HPVCP identified diethyl and diphenyl disulfides, naphtha sweetening (Chemical Abstracts Service [CAS] # 68955-96-4), as candidate materials based on production volume estimates exceeding 1 million pounds per year. Commonly known as disulfide oil (DSO), this substance is a combination of dialkyl disulfides and trisulfides which are a by-product of petroleum refining. The substance is neither sold commercially nor used directly in any downstream products, so exposure is generally limited to workplace operations associated with its disposal as a waste product. The status of DSO under the HPVCP has evolved considerably since first proposed in 1990.
After sponsorship by the American Petroleum Institute’s Petroleum HPV Testing Group (PHPVTG) in 2003, DSO was grouped together with 4 other disulfide waste streams that ultimately lost sponsorship in 2006 due to production termination, errors in inventory reporting, or in the identification of enclosed recycling and reuse practices. The DSO was then merged into a new product category termed Reclaimed Substances, where it represented 1 of the 5 subgroups. Companies belonging to the PHPVTG sponsored approximately 400 petroleum substances for review under the HPVCP. These substances were organized into 13 chemical categories to facilitate data sharing and to avoid redundant testing. A data review and assessment for DSO were originally submitted under the HPVCP in 2008 together with the required robust summaries. Following a revision and update that reflected comments received from the USEPA, the data package was resubmitted in 2010. Final acceptance and approval of the resubmission was issued in March 2012 with the conclusion that data gaps did not exist in the hazard analysis. 8
Polysulfides such as those in DSO recently came under close scrutiny as inducers of oxidative stress in biological systems. 9 Their ability to form reactive sulfur species that disrupt the overall redox status within a cell is well documented for the various classes. 10 The nature of the sulfide bridge and hydrocarbon side chain can, however, dramatically impact the overall reactivity and capacity to induce oxidant damage. In general, the reactivity increases in following order with tetrasulfides > trisulfides > disulfides. Similarly, hydrocarbon functional groups display differences in free radical production with alkenes > alkanes > phenyl substituents. 11,12 Important exceptions have been shown to exist, however, particularly with the olefins where the location of the double bond can have a decided impact. Since DSO does not contain any aryl- or alkenyl-substituted disulfides or trisulfides, the primary concern lays with the relative toxicity of the alkyl-substituted polysulfides.
Dimethyl disulfide (DMDS) in particular has undergone extensive toxicological testing with the results showing a pattern of behavior that is consistent with an oxidative stress response. 13 The general applicability of this specific mode of action provides a basis for evaluating the hazards of all disulfide and trisulfide constituents of DSO. There have been numerous examples of the successful application of the category approach for assessing a wide array hazards ranging from skin sensitization to teratogenicity. 14 Although the category approach was successfully used to describe the hazards for individual members of a chemical class, there were few examples of its use with a homologous series of chemicals. The following case study examines the evidence that was used to satisfy the HPVCP hazard evaluation requirements for DSO and highlights the use of a categorical approach with a mode of action determination to justify a read-across hazard assessment. 15 The analysis takes advantage of recently released DMDS study summaries prepared by the pesticide registration divisions at USEPA and California Environmental Protection Agency. 16,17
Formation and Composition of DSO
Disulfide oil is generated during mercaptan removal from selected hydrocarbon streams by a process known as sweetening, whereby sour-smelling sulfides are removed from the stream by an extraction process. 18 The mercaptans can be extracted from this feedstock in an entirely closed system, such as a Merox unit, which can be designed to operate with any of a variety of petroleum streams including liquefied petroleum gas, naphtha, or other light distillates such as jet fuel and kerosene. Merox units use a basic solution of caustic soda as the extracting solvent, which is recycled and reused in a continuous loop following each cycle. Once removed, the mercaptans are oxidized to disulfides, which are separated from the caustic soda solution. The final DSO is then either handled as waste on site or processed as: (1) an internal fuel, (2) a feedstock for sulfuric acid production, or (3) an agent for conditioning refinery catalysts.
The DSO is primarily composed of low-molecular-dialkyl disulfides and trisulfides with a total carbon content of C2 to C8. The DSO does not contain measurable amounts of aryl mercaptans or aryldisulfides, despite their suggested presence from the formal CAS nomenclature. Although the composition of DSO has historically relied upon retention time comparisons following gas chromatographic separation, more sensitive and specific methods are now employed. Using high-resolution gas chromatography together with mass spectrometry (GC-MS, American Society for Testing and Materials [ASTM] D 3670), a representative sample of DSO was submitted for qualitative and quantitative analyses by a certified laboratory. 19 The results reveal the presence of dialkyl disulfides and trisulfides as the major constituents with small amounts of monosulfides, heterocyclic disulfides, mercaptans, miscellaneous sulfur-bearing substances, and a limited number of residual hydrocarbons. Taken together, the secondary compounds account for approximately 9% of the overall mass by weight. As shown in Table 1, the composition of this sample was characterized by the presence of 12 disulfides, with 10 possessing individual concentrations greater than 0.5%. The 10 disulfides comprise approximately 87% of the total weight and were representative of a homologous series with DMDS beginning the series. The molecular weight of the disulfides ranged from 94 to 150 amu and showed a strong similarity in chemical structure, with each possessing a characteristic disulfide linkage attached to a C1 to C4 alkyl group. In addition, 4 dialkyl trisulfides were present at levels ranging from 0.4% to 1.6%. By comparison, previous analyses using less accurate gas chromatographic techniques showed the presence of methyl butyl, ethyl butyl, and propyl butyl disulfides in various isomeric forms at levels that ranged up to 3.5%. Likewise, remarkably similar distribution ratios were observed in these previous assays with the methyl, ethyl, and propyl homologs predominating at individual concentrations generally ranging from 5% to 20%. A full description of the analytic results using GC-MS is presented in Supplemental Table 1. It shows that less than 0.5% of DSO is composed of hydrocarbons and that the balance is composed of low-molecular-weight monosulfides and trisulfides that individually comprise less than 2% of the total weight percentage.
Identity and Concentration of the Individual Disulfides in a Sample of Disulfide Oil.

Abbreviations: Conc, concentration; CAS, Chemical Abstracts Service; DSO, disulfide oil; Mol Wt, molecular weight.
aConcentration may vary depending on the characteristics of the unsweetened crude oil.
At room temperature, DSO exists as a yellow liquid with an extremely foul and obnoxious odor. 20 Measurement of key chemical and physical properties (see Table 2) using standardized methods reveals that the substance is highly flammable with a moderately high-vapor pressure and low-water solubility. 21 The chemical and physical properties for the individual DSO disulfides were estimated using EPA’s Estimation Program Interface (EPI) Suite software package and are provided in Supplemental Table 2 along with actual measurements when available. 22 Except for melting points, the estimated and measured values agree well. The estimates also show a regular incremental change as the molecular weight increases for each disulfide. Boiling points and octanol/water partition coefficients show a direct relationship with molecular weight; whereas, the melting points, vapor pressures, and water solubilities are inversely related.
Chemical and Physical Properties of a Sample of Disulfide Oil.

Abbreviations: ASTM, American Society for Testing and Materials; DIN, Deutsches Institut für Normung.
aRequired information per the High Production Volume Challenge Program.
Environmental Fate
The environmental fate of DSO was not specifically evaluated, because it is a UVCB (unknown or variable composition, complex reaction products, or biological material substance). Insight into likely fate processes, compartment preferences, and steady-state distributions can be gained, however, by examining individual disulfides using available structure activity models and by examining available categorical test data for key homologs in the disulfide series. Many of the disulfides in DSO are naturally found in the environment either as ingredients in vegetables, especially Allium species such as garlic and onions, or as products of the microbial oxidation of assimilated mercaptans. 23 Similarly, natural background emission rates of DMDS were reported for salt marshes, rice paddy fields, food waste decomposition, and some soil types. 24 –26 The DMDS emissions were estimated to contribute 3% to the atmospheric load of sulfur on a global scale, but the contribution can be as high as 10% on a regional scale. 27 Interestingly, DMDS was shown to be absorbed from air into moist and dry soils at a rate that was influenced by the presence of soil microbes, which facilitated the uptake into moist, but not dry, soil. 28 These findings are consistent with the observation that DMDS failed to show a consistent air exchange rate in 3 subtropical forests with both positive and negative fluxes observed. 29
The DMDS was also found to rapidly photo-oxidize under tropospheric conditions to form sulfur dioxide and methane sulfonic acid. 30 The rate constant for reaction with hydroxyl radicals in the atmosphere was determined to be 2.3 × 10−10 cm3/mol sec, which is equivalent to a daytime half-life of 0.84 hours (lifetime 1.2 hours) assuming a radical concentration of 1 × 106 radicals/cm3. 31 Reaction with nitrate radicals during the overnight hours occurs in a similarly rapid fashion with a rate constant of 0.7 × 10−12 cm3/mol sec and a half-life of 11 hours (lifetime 16 hours). The atmospheric photodegradation of the remaining disulfides can be estimated using the AopWIN (v 1.92) subroutine in the EPI Suite of quantitative structure activity relationship (QSAR) programs. As shown in Table 3, the rate of tropospheric photo-oxidation by reaction with hydroxyl radicals is nearly identical for the 10 disulfides in DSO with a hydroxyl radical half-life of 0.51 to 0.56 hour. The atmospheric half-life of each disulfide is therefore approximately 30 minutes, which meets the definition of a rapidly removed volatile organic compound. The estimated rates of DMDS hydroxyl radical reactivity also compared well with the actual value (0.56 vs 0.84 hour). A small portion of the discrepancy between the measured and the estimated values is associated with the assumed hydroxyl radical concentrations used in AopWIN program (1.5 × 106 radicals/cm3). When the measured values are corrected for this difference, the daytime half-life decreases to 0.80 hours.
Estimated Environmental Fate Parameters from EPI Suite.
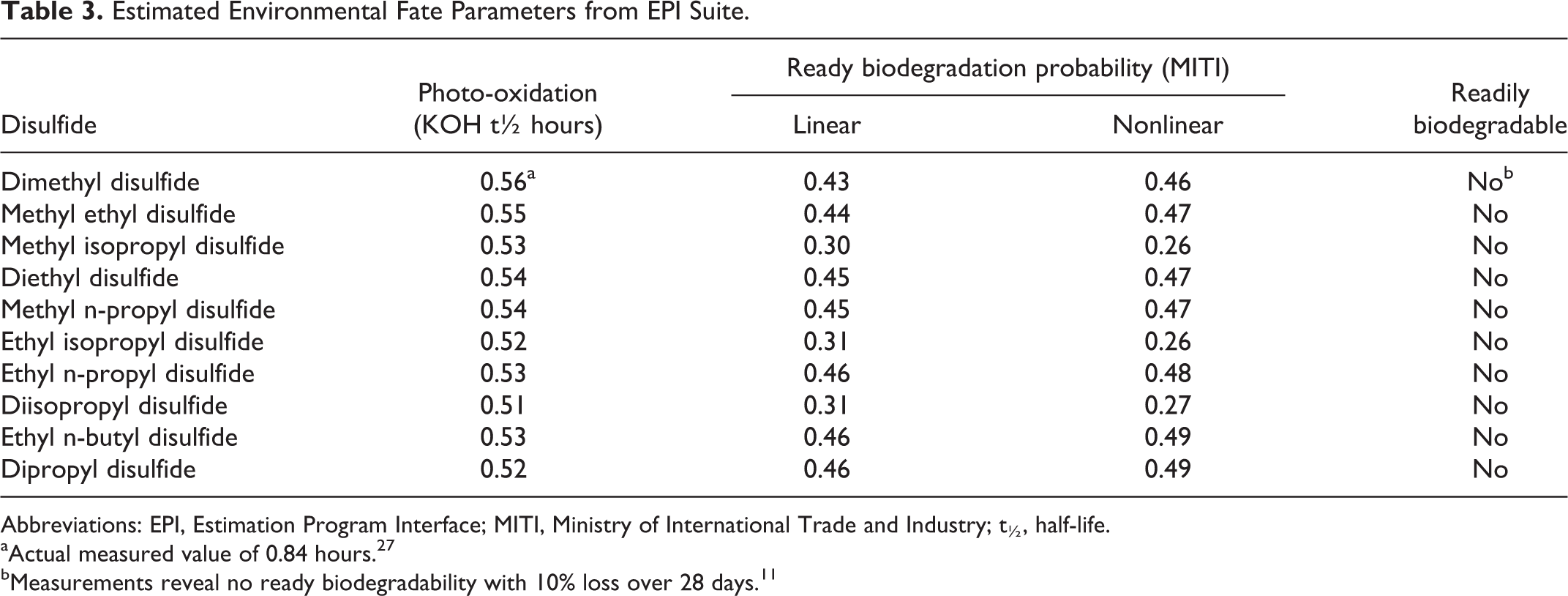
Abbreviations: EPI, Estimation Program Interface; MITI, Ministry of International Trade and Industry; t½, half-life.
aActual measured value of 0.84 hours. 27
bMeasurements reveal no ready biodegradability with 10% loss over 28 days. 11
Although no information is available on the aqueous stability of DSO, pertinent facts can be derived from an examination of test data for DMDS. Available information for DMDS indicates that aqueous hydrolysis at ambient temperature is too slow to be an important environmental fate process when the pH is less than 12. 32 The study suggests that DMDS may hydrolyze slowly to nonvolatile methane sulfonic acid in water at pH 11 to 12. An evaluation of the aqueous hydrolysis of 14 C-radiolabeled DMDS at pH 4, pH 7, and pH 9 in a buffered solution maintained at 50 °C did not reveal any decay and in most cases a slight increase in concentration occurred over the 5-day measurement period. 33 This conclusion is consistent with the relative stability of the disulfide bridge to acid–base hydrolysis and the absence of any water-sensitive functional groups such as an ester or epoxide linkage. None of the remaining disulfides in DSO could be evaluated for aqueous stability, because the HYDROWIN algorithm for estimating aqueous stability has only been validated with a limited number of chemical classes. Using the results for DMDS in a read-across fashion, the aqueous hydrolysis of DSO is not expected to be an important environmental fate process.
The biodegradability of DSO can be reasonably estimated by examining the activity of its key constituents. Although studies have revealed that low concentrations of DMDS (up to 1 mmol/L) were completely degraded within 38 days using adapted, sulfate-reducing sludges, higher concentrations (≥ 2 mmol/L) were not fully degraded to methanethiol after 50 days of incubation. 34 These results contrast with estimates from the Biodegradation Estimation subroutine (BIOWIN, v 4.00) within the EPI Suite of QSAR programs. The BIOWIN routine evaluates the outcomes from 7 different evaluations to determine the biological degradation of a target chemical under either aerobic or anaerobic conditions. Each evaluation considers the activity associated with individual molecular fragments within the target chemical. The fragments are examined using coefficient-based multiple linear and nonlinear regression routines and the resulting values summed up to arrive at a final probability estimate. Although several of the BIOWIN routines suggested the probability of disulfide biodegradation is relatively high, it is believed that the most reliable results are achieved with the Japanese Ministry of International Trade and Industry (MITI) model, which predicts biodegradability in accordance with OECD301C test conditions using a training set containing 900 discrete substances. 35 As shown in Table 3, the MITI probability estimates for the linear and nonlinear MITI models are less than 0.5, which is the cutoff for assigning a “readily biodegradable” result. These findings are partially substantiated by a closed bottle ready biodegradability test performed with DMDS, which indicated that less than 10% of the test chemical was degraded over a 28-day period. 36 Ready biodegradability, as defined in accordance with OECD guidelines, only occurs when at least 60% of a chemical is biologically removed from the environment within a 28-day period (as measured by oxygen consumption or CO2 evolution). Accordingly, DSO is expected to fail the biodegradability test and degrade slowly under aerobic and anaerobic conditions.
Environmental Distribution
The environmental distribution of DSO is inextricably linked with the behavior of its component disulfides. Preliminary studies with DMDS and dipropyl disulfide (DPDS) showed that these 2 disulfides were relatively stable in soil and water. 37 DMDS, in particular, was found in many environmental compartments and is considered to have an integral role in the global sulfur cycle. 38 Natural background concentrations of DMDS were measured in a variety of media including air, surface waters, sediment, wastewater effluent, vegetation, and expired human air. 39 Canadian saltwater lakes, wetland ponds, and freshwater lakes all exhibited a positive net flux of DMDS to the atmosphere with average rates as high as 35 μmol S/m2-day for saltwater lakes, which releases an appreciably higher mass of DMDS than freshwater lakes.40,41 These atmospheric release levels are consistent with the moderately high Henry law constant of 0.0012 atm m3/mol for DMDS. Laboratory determination of soil organic carbon/water partition coefficients for DMDS added to 5 soil types yielded values ranging from 10 to 83 mL/g, which indicate high mobility for DMDS in the soil compartment.42,43 Aerobic and anaerobic soil biodegradation studies with DMDS indicate that bacterial biodegradation to methanesulfonic acid and methanethiol (anaerobic only) can take place in clay and loam soils; however, the calculation of a biodegradation half-life was hampered by the rapid volatilization of DMDS from the test system.44,45 Observed half-lives for dissipation and biotransformation ranged from approximately 6 to 10 days under aerobic and anaerobic conditions. The structural similarity between DMDS and the remaining disulfides in DSO suggested that analogous processes and fates apply.
Evaluation of each disulfide in a multimedia transport model provides the steady state distribution for a 1000 kg/h emission load to each of 4 compartments, water, air, soil, and sediment. The modeling can be performed on any of the 4 levels depending on the number of processes considered and the amount of detail required in the results. A Level 1 fugacity analysis performed using the Equilibrium Criterion Model, v2.02 indicated that virtually 100% of each disulfide is distributed to the air compartment, which is inconsistent with the known partitioning behavior of the lowest homolog, DMDS. Consequently, a Level III multimedia fugacity model was selected, since it considers the intercompartmental transfer rates between the individual phases. 46 The results presented in Table 4 show that all 10 disulfides have a preference for the water and soil compartments with an increasing percentage in soil as the dialkyl carbon number increases from C2 to C6. This systematic shift in steady state distribution with increasing molecular weight is consistent with changes in the physical properties of the individual homologs that are depicted in Table 5. The increase in soil organic carbon/water partition coefficient (Koc) and decrease in water solubility with increasing molecular weight explains, to a large degree, the phase shift observed when moving from the lowest to the highest homolog in the series.
Multimedia Environmental Distribution Estimates for DSO Disulfides.
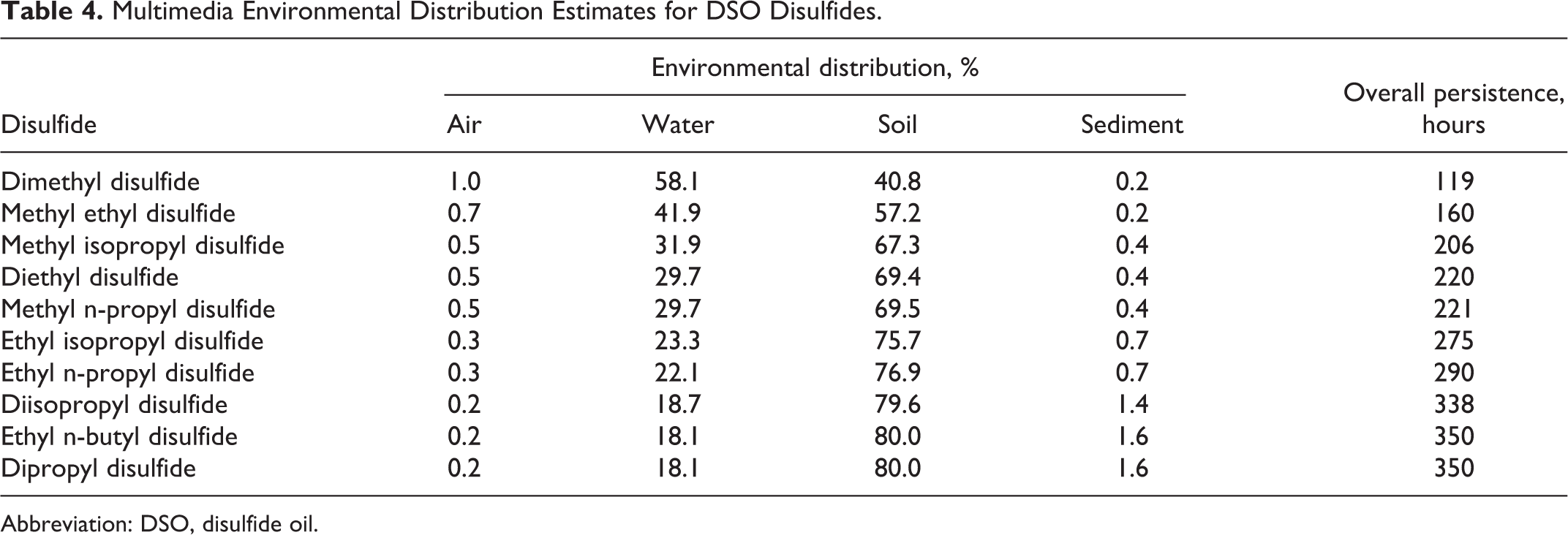
Abbreviation: DSO, disulfide oil.
Estimated Physiochemical Constants for DSO.
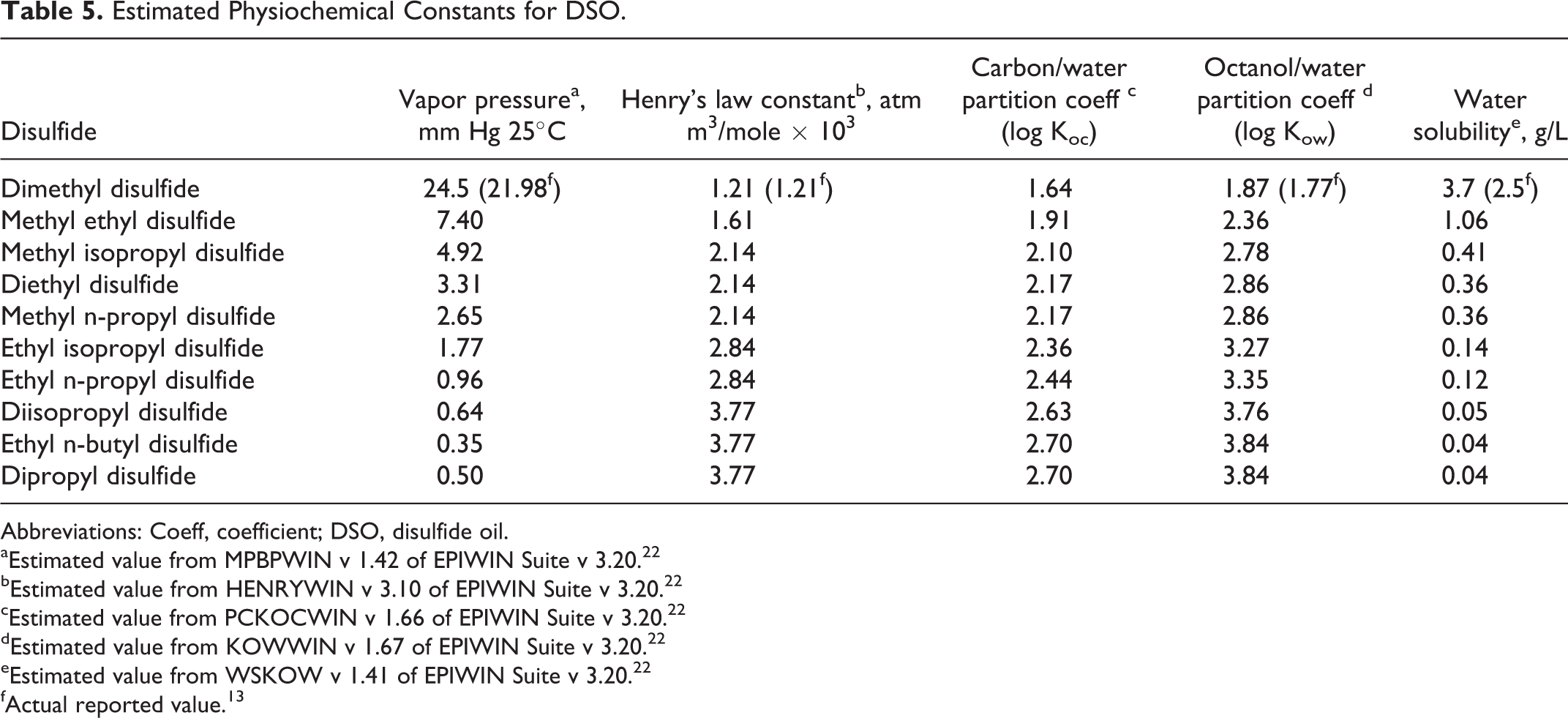
Abbreviations: Coeff, coefficient; DSO, disulfide oil.
aEstimated value from MPBPWIN v 1.42 of EPIWIN Suite v 3.20. 22
bEstimated value from HENRYWIN v 3.10 of EPIWIN Suite v 3.20. 22
cEstimated value from PCKOCWIN v 1.66 of EPIWIN Suite v 3.20. 22
dEstimated value from KOWWIN v 1.67 of EPIWIN Suite v 3.20. 22
eEstimated value from WSKOW v 1.41 of EPIWIN Suite v 3.20. 22
fActual reported value. 13
The ultimate goal of determining the environmental distribution of a chemical is a determination of its environmental persistence. Persistence is calculated from an assessment of the environmental rate constant and associated half-life for each individual compartment, which is then used to compute a weighted mean rate constant that considers the mass fraction in each compartment. For the DSO disulfides, the estimated half-lives varied only slightly with values of 1.1 hours, 360 hours, 720 hours, and 135 days for air, water, soil, and sediment, respectively. The mass concentrations in each compartment did change as a function of molecular weight with greater redistribution to the sediment compartment resulting in a change in the overall persistence. As shown in Table 4, the persistence in the environment ranged from 119 to 350 hours (5-15 days) for the individual disulfides, which would rank these chemicals as slightly persistent in the environment. By comparison, the environmental behavior of DSO would be dictated by the homolog with the longest residence time, so it is anticipated that DSO would have a persistence of at least 15 days.
Ecotoxicity
The structural similarity of the disulfide components of DSO provides a basis for using QSARs to assess the relative ecotoxicity of the individual homologs. Any number of QSARs exist that would reasonably be anticipated to produce a suitable result. Indeed, initial modeling of DMDS and the remaining disulfide constituents of DSO using EPA’s Ecological Structure Activity Relationships (ECOSAR v 0.99g) software package revealed that the ecotoxicity of the disulfides increased as a function of alkyl chain length. 47 Although this finding is consistent with the observed increase in octanol/water partition coefficients for these disulfides (see Table 5), the results were inconsistent with available test data, which revealed large disparities between the estimated and the actual values for several indicators of toxicity. In particular, Table 6 reveals that the ecotoxicity of DMDS to freshwater and marine organisms was under predicted by a factor of approximately 4 to 96-fold using QSAR routines that assume nonpolar narcosis as the primary mechanism of action. Drawing upon the existing literature on potential mechanisms of action for alkyl disulfides, QSAR estimates were abandoned since the underlying mechanism of action was judged to be incorrect, a condition that occurs frequently when this pathway is invoked indiscriminately. 49 Alternatively, information was compiled on the likely mode of action for disulfides, and a qualitative potency comparison was made for the individual homologs.
Comparison of Actual and Estimated Ecotoxicity Values for Dimethyl Disulfide.
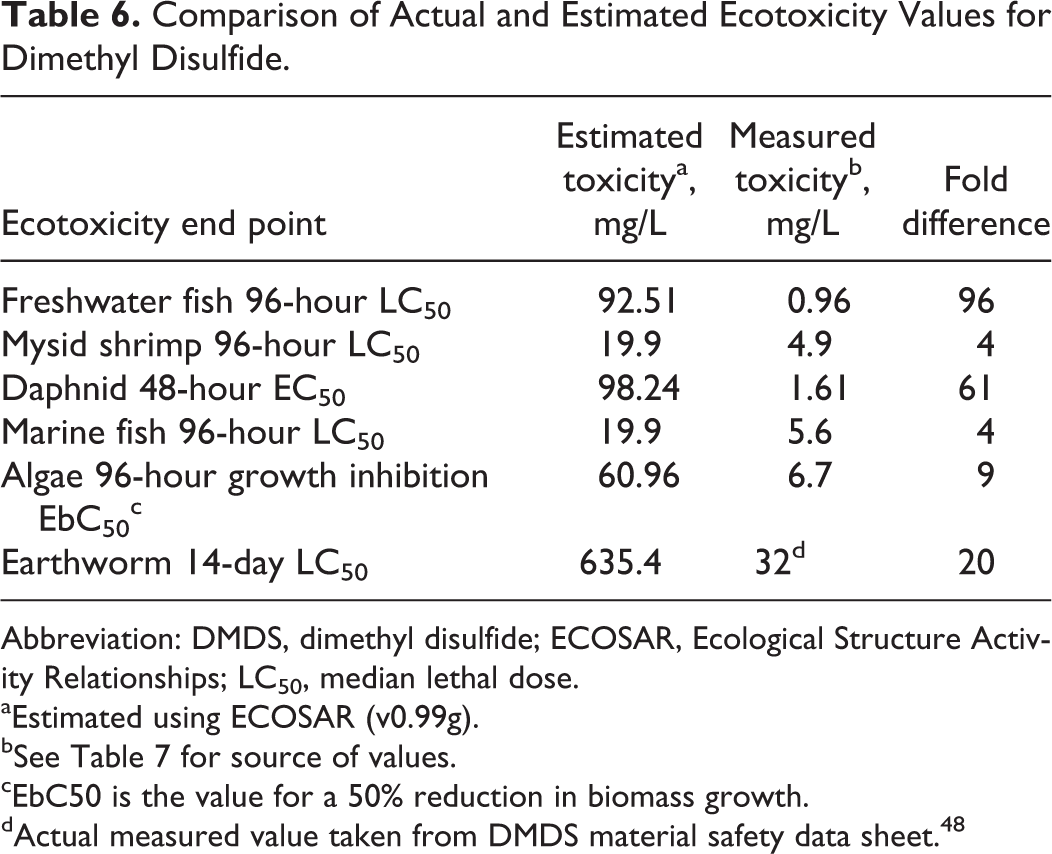
Abbreviation: DMDS, dimethyl disulfide; ECOSAR, Ecological Structure Activity Relationships; LC50, median lethal dose.
aEstimated using ECOSAR (v0.99g).
bSee Table 7 for source of values.
cEbC50 is the value for a 50% reduction in biomass growth.
dActual measured value taken from DMDS material safety data sheet. 48
Once absorbed, many disulfides are readily reduced to unstable thiols that may be reoxidized to the disulfide in a metal-catalyzed reaction that results in the formation of highly reactive oxygen species (ROS) that can react with nearby lipids, proteins, and nucleic acids. 50 Because this reaction pathway is cyclical, it can take place for prolonged periods of time producing large quantities of free radicals through the metabolically generated ROS and the parent disulfide itself. 51 The ROS produced in this reaction can lead to protein interactions and acute toxicities in aquatic species that are typically more severe and less uniform for a homologous series than those elicited by narcotic chemicals. 52 These free radicals result in lipid peroxidation and other adverse effects that are the hallmark of an oxidative stress response. Although studied to a large degree in mammals, redox cycling and oxidative stress can also occur in aquatic species including marine algae, invertebrates, and mollusks. 53,54 In fact, the biochemical events leading to an oxidative stress response are so similar across the plant and animal domains that some researchers proposed using aquatic organisms as a screening tool to investigate the oxidative hazards to humans. Prototypical chemicals capable of causing oxidative stress in freshwater and marine species include transition metal ions, pesticides, and quinones.
Ecotoxicity of Dimethyl Disulfidea.
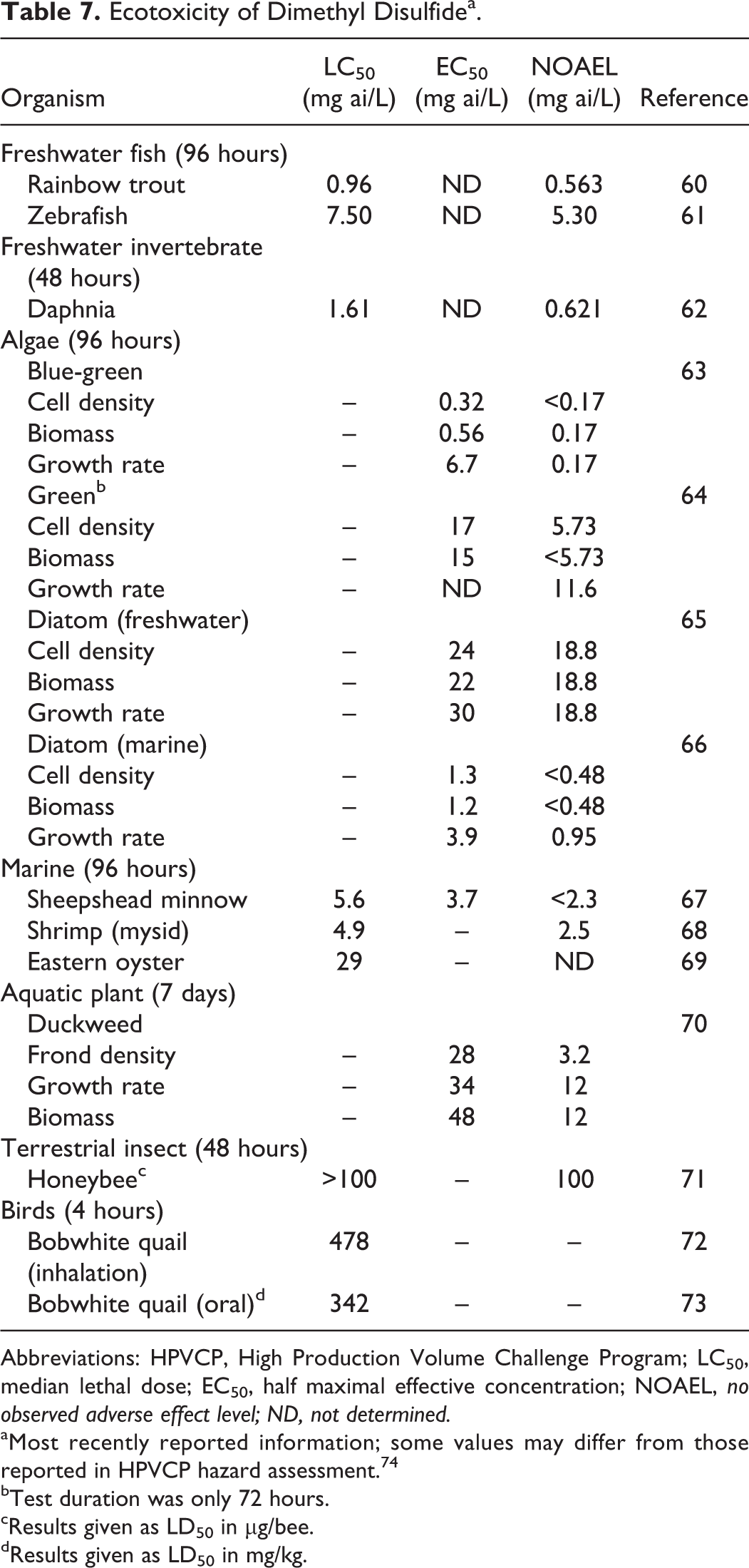
Abbreviations: HPVCP, High Production Volume Challenge Program; LC50, median lethal dose; EC50, half maximal effective concentration; NOAEL, no observed adverse effect level; ND, not determined.
aMost recently reported information; some values may differ from those reported in HPVCP hazard assessment. 74
bTest duration was only 72 hours.
cResults given as LD50 in μg/bee.
dResults given as LD50 in mg/kg.
Despite increased interest in the applicability of this mechanistic pathway, few substances were evaluated, and no QSARs were developed as an aid for a category analysis. 55 One group of researchers found that the oxidative stress caused by a small group of quinones was directly correlated with octanol/water partition coefficient, with the highest activity in those chemicals having the lowest Kow. 56 Likewise, there are no systematic studies of the mode of action of disulfides on aquatic or terrestrial species; however, some general principles are believed to operate based on knowledge gained from studies in laboratory animals. Perhaps the most important factors affecting toxicity are the length of the alkyl groups and the degree of substitution on these side chains. Mammalian studies showed that the reactivity of dialkyl disulfides is inversely related to their molecular weights, with the lowest homolog, DMDS, displaying the greatest propensity to form ROS, and producing an oxidative stress response. 57 This relationship logically applies to marine and freshwater organisms as well, given the ubiquity of the oxidative stress response throughout the animal kingdom. 58,59 As such, the ecotoxicity of DSO in biological systems would be dominated by the DMDS content, which is considerable. These facts justify using the DMDS test data in Table 7 as a logical and reasonable surrogate for DSO and provide a basis for using the test results for DMDS in a read-across fashion for all of the disulfides in DSO.
Additional support for the use of DMDS as a surrogate for the disulfides in DSO comes from available test data for higher homologs in the series. When the acute toxicity of DMDS to fish (0.96 mg/L) is compared to the median lethal dose (LC50) for diethyl disulfide (DEDS; 7.43 mg/L), DPDS (2.62 mg/L), and diisopropyl disulfide (8.31 mg/L), there is no apparent increase in toxicity as a function of chain length. 75 –77 As expected, based on the available mechanistic data for redox cycling, the acute toxicity for freshwater fish declines with increasing molecular weight. A similar case exists when the 24-hour EC50 value for DEDS (14.5 mg/L) in Daphnia magna is compared to the concentration obtained with DMDS. 78 The 48-hour EC50 value of 7 mg/L for DMDS is 2-fold lower than the value obtained with DEDS. 74 Taken together, these data indicate that DMDS is a reliable surrogate for the remaining dialkyl disulfides, and the hazards associated with lower members of the series are less than or equal to the ecotoxicity of DMDS. Additional testing with DSO was therefore deemed to be unnecessary since the results would not be expected to result in effect concentrations less than those observed with DMDS based on mechanistic considerations.
Health Effects
The health effects of DSO were evaluated through the application of the categorical approach for a homologous group of chemicals. Although some published test data are available on DSO itself, the majority of information has been extracted from unpublished company reports, peer-reviewed literature, IUCLID data sets, robust summaries, and/or calculated using accepted computer modeling programs. In many instances, the health effect information were used in a read-across manner to evaluate the toxicity of DSO. The rationale and justification for establishing this surrogate relationship are based on sound scientific principles and a plethora of mechanistic information showing that the dialkyl disulfides in DSO operate through a common toxic mechanism. This mechanism, which was well studied and clearly elucidated in the published literature, focuses on the unique characteristics of the disulfide bridge and the ease with which free radical intermediates can be formed once the bridge is cleaved. Since DSO also contains a small fraction of dialkyl trisulfides, whose potency appears to exceed that of the corresponding disulfide, the relative toxicity of this class of substances was evaluated as well.
The metabolism of many, if not all, disulfides is initiated by a thiol-disulfide exchange reaction that substitutes the sulfhydryl group of glutathione for a mercaptide fragment within the disulfide molecule. This reaction is depicted in Figure 1 for DPDS, whose in vivo metabolism has been studied in far greater detail than DMDS. 79,80 Evidence shows that this same initial glutathione exchange reaction also takes place for a host of alkyl, alkenyl, phenyl, and branched chain disulfides as well as trisulfides. 11,57,81,82 The exchange reaction with glutathione is catalyzed by a thiol transferase, also known as glutaredoxin, which is widely distributed in nature and shows a high level of activity in the tissues and organs primarily affected by dialkyl disulfide toxicity. 83 This reaction is the key step in the toxic mechanism for dialkyl disulfide and trisulfide congeners. The activation mechanism is pertinent, because it has been associated with the initiation of a redox cycle that generates excessive quantities of highly reactive free radical intermediates capable of interacting with tissue macromolecules at or near the site where they formed. In some cases, this site was the hemoglobin in red blood cells and in other cases the liver, depending on the species examined. 57 Some evidence shows that DMDS is metabolized via the same pathways. Mice treated intraperitoneally with 35 to 40 mg/kg DMDS were shown to quickly exhale methyl mercaptan and dimethyl sulfide, which is consistent with the cleavage and methylation reactions observed with DPDS. 84
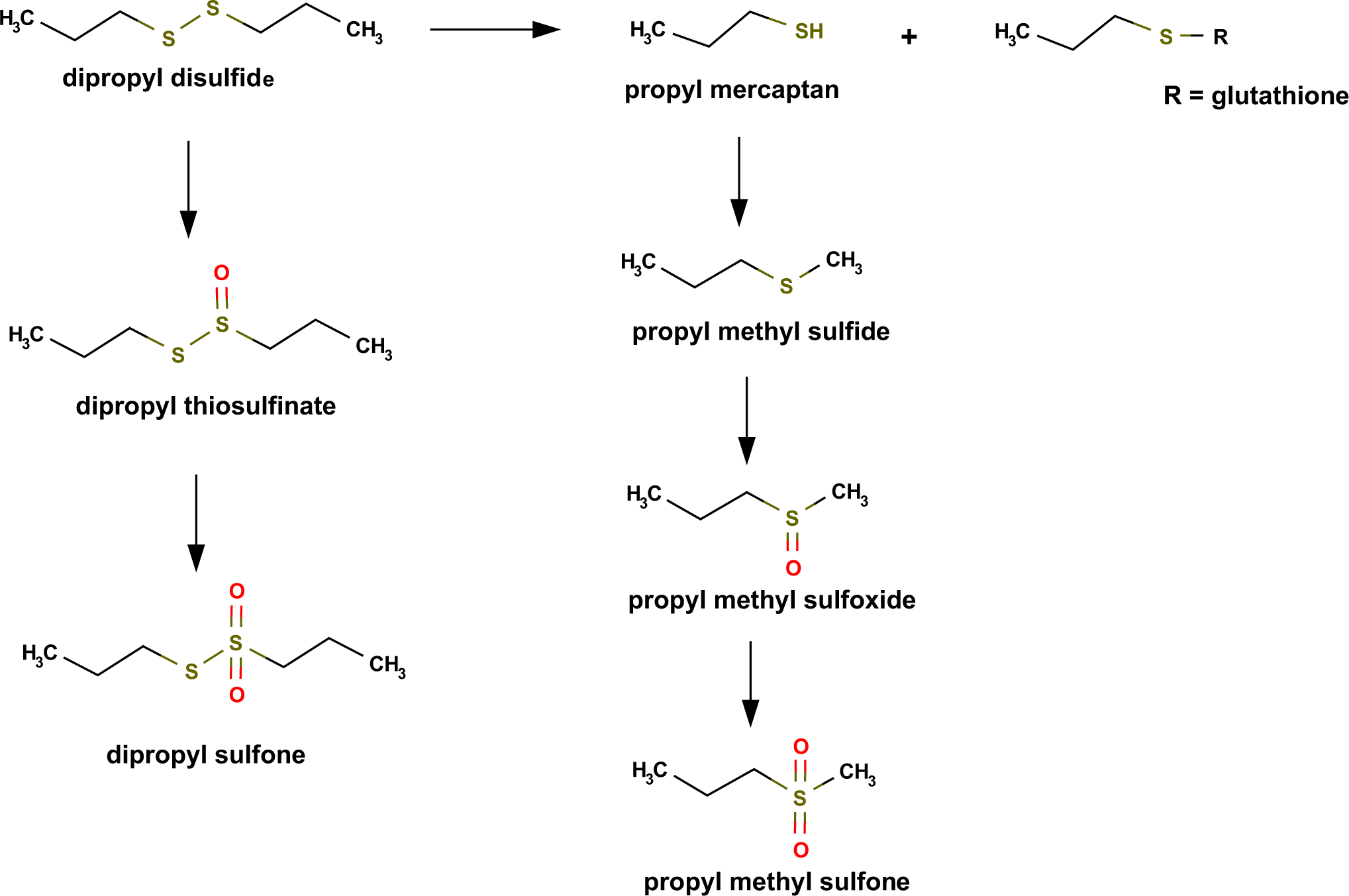
Typical pathways for the in vivo metabolism of dialkyl disulfides.
The sequence of reactions in the redox cycling of dialkyl disulfides is depicted generically in Figure 2. 11 The first product of the initial thiol transferase exchange reaction is an alkyl mercaptan that once ionized, undergoes a 1-electron oxidation to a free radical intermediate. This intermediate is the proximal toxicant responsible for producing a continuous supply of hydroxyl radicals and other ROS that can sustain the redox cycling and cause oxidative stress and tissue injury at sites where they are formed. Importantly, the reactivity of the mercaptans formed in the exchange reaction is directly affected by the length and branching pattern of the attached alkyl substituents, with longer chain lengths leading to reduced radical stabilization and lower oxidation rates. 57 In addition, the reactivity and toxicity of alkyl disulfides have been shown to decrease in the following order n > sec > tert due to the influence of steric factors on thiol transferase activity. These data indicate that DMDS will be the most reactive member of the series with the longer chain lengths and higher branching patterns of the remaining homologs ameliorating the toxicity by affecting the rate of formation and ultimate stabilization of the free radical intermediates.
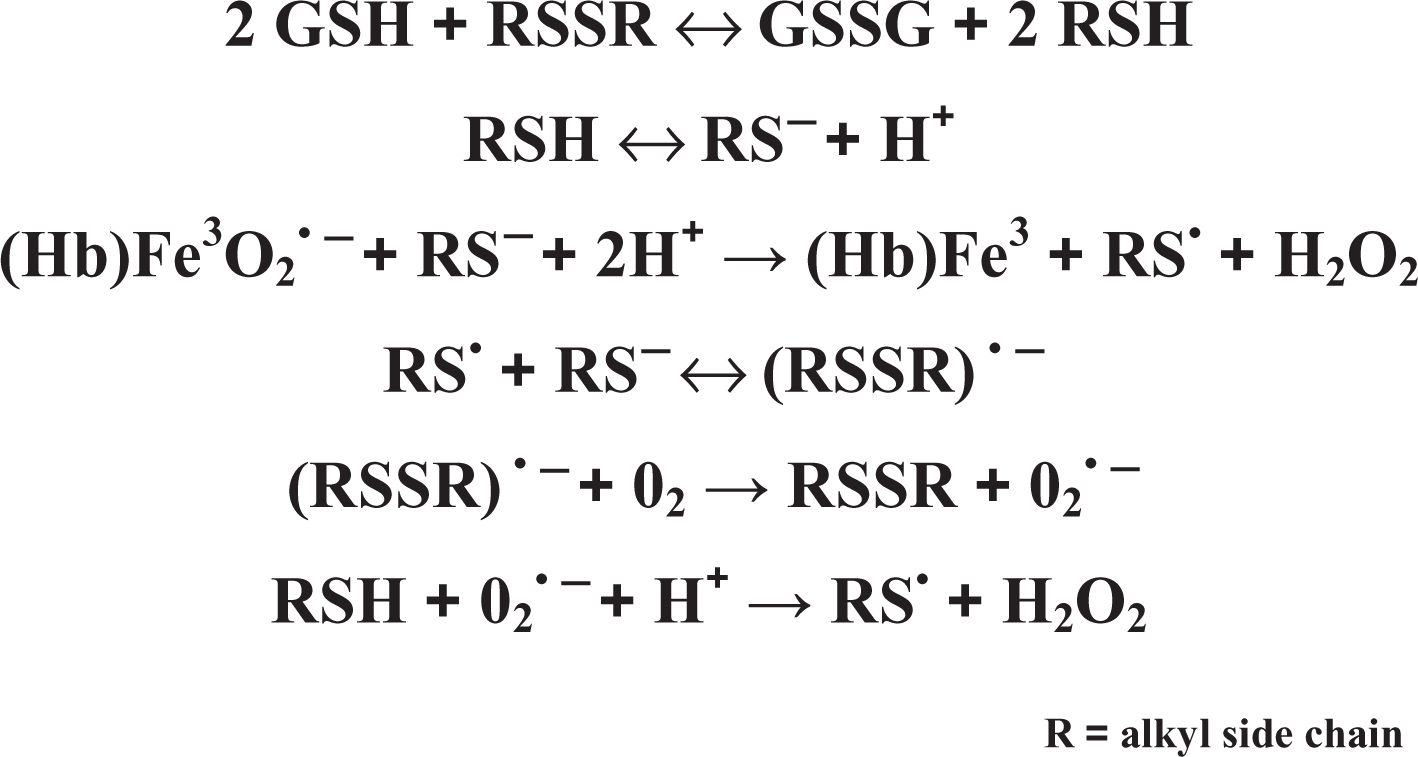
Mechanism of redox cycling and free radical formation from dialkyl disulfide metabolism.
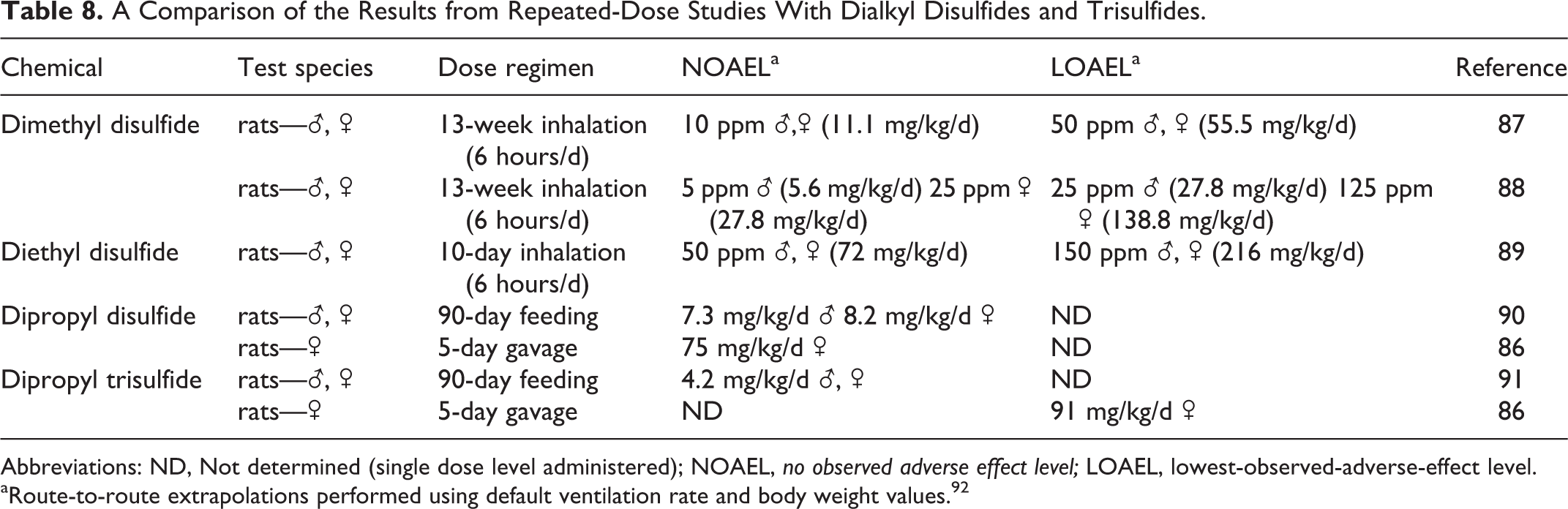
Evidence suggests, however, that dialkyl trisulfides are an exception, with the intensity of radical formation and toxic potency exceeding that of the corresponding disulfide. Although DSO is predominantly composed of dialkyl disulfides, which are believed to be primarily responsible for the toxicological effects of this substance due to their prevalence, the impact of several minor trisulfides may also play some role. Measurable amounts of the following 4 dialkyl trisulfides were present in the DSO sample analyzed: dimethyl trisulfide (1.6%), diethyl trisulfide (0.7%), methyl propyl trisulfide (0.5%), and diisopropyl trisulfide (0.4%). Although the mode of action and routes of metabolism for the dialkyl trisulfides are similar to the disulfides, they were shown to be more active at causing redox cycling, GSH depletion, and hematological anomalies than the corresponding dialkyl disulfides. 85,86 The increase in trisulfide potency is not, however, associated with an increase in the effective toxicity as defined by the threshold concentration capable of eliciting an effect. A comparison of the no observed effect levels (NOELs) from 90-day repeated-dose studies with DMDS, DPDS, and dipropyl trisulfide (DPTS) indicates that all 3 congeners have similar toxic thresholds (see Table 8). Consequently, although the trisulfides may be more potent with a steeper dose–response (DR) relationship, the threshold doses eliciting demonstrable adverse health effects are not substantially different from DMDS.
A Comparison of the Results from Repeated-Dose Studies With Dialkyl Disulfides and Trisulfides.
Abbreviations: ND, Not determined (single dose level administered); NOAEL, no observed adverse effect level; LOAEL, lowest-observed-adverse-effect level.
aRoute-to-route extrapolations performed using default ventilation rate and body weight values. 92
Analysis of all available information provides reasonable support for the use of DMDS as a surrogate for the higher chain length disulfides and trisulfides in DSO and substantiates the use of DMDS data in a “read-across” transfer to the other sulfide-containing substances in the category. The test data for DMDS is therefore offered as a reliable and mechanistically supportable substitute for DSO, since the toxicity of the other constituents is equal to or less than DMDS. This association does not, however, extend to substances other than DSO that may contain specific aryl or allylic disulfides whose rate of redox cycling can be considerably greater than DMDS. 50 Similarly, the hemolytic anemia that can accompany exposure to disulfide or trisulfides-containing substances has been shown to be highly species specific with cats and dogs being more vulnerable to erythrocyte damage than rats, mice, or humans.
Acute Toxicity
Oral, dermal, and inhalation studies were performed with DSO. The oral LD50 value was 1590 mg/kg in female rats and 1700 mg/kg in male rats. 93 Gross necropsy on dead and moribund animals revealed intestines filled with red fluid and tan-colored lungs. Darkly colored spleens were noted upon sacrifice of all female rats, with all animals displaying enlarged spleens. In an initial acute oral screening LD50 study on the same material, both female and male rats were administered 5000 mg/kg, after which all the animals died. 94 The 4-hour inhalation LC50 value for DSO vapor was found to be greater than 4.84 mg/L in male and female rats. 95 The dermal LD50 value was greater than 1800 mg/kg in rabbits. 96 Mild to moderate irritation was observed in a Draize rabbit skin test, and the same test material was determined to be minimally irritating in rabbit eyes. 97,98 The DSO was not a dermal sensitizer in a guinea pig sensitization test. 99
Comparable studies with DMDS revealed an oral LD50 value for rats of greater than 190 mg/kg, a dermal LD50 value for rabbits that was greater than 2000 mg/kg, and a 4-hour inhalation LC50 value for rats of 805 ppm (3.10 mg/L). 100 –102 A concentration-related increase in olfactory epithelial degeneration was observed in the nasal turbinates of rats exposed to 8.9, 12.6, and 18.4 ppm of DMDS for 24 hours, but a 5.0 ppm exposure for the same duration was free from this effect. 103 A functional observational battery performed on rats exposed to 100, 200, or 700 ppm of DMDS for 6 hours revealed an increased incidence of partial eyelid closure, grooming behavior, and urination, and a decrease in motor and locomotor activity at the highest exposure concentration. 104 The NOEL for these effects was 200 ppm. The preceding data suggest that DMDS is more acutely toxic than DSO by both the oral and the inhalation routes. By comparison, a single rat oral LD50 value of greater than 2000 mg/kg has been reported for DPDS. 77 In addition, a single 5-hour exposure of male rats to a saturated atmosphere of 4390 ppm (21.95 mg/L) of DEDS resulted in the death of all animals; whereas 5 of 6 animals exposed to 2156 ppm (10.78 mg/L) succumbed. 105 An approximate oral LD50 value ranging between 800 and 1600 mg/kg was found in mice treated with DPTS. 106
Some disulfides, in particular DMDS and DPDS, have been shown to cause mild to severe red blood cell hemolysis in cats, dogs, and a variety of livestock animals following oral ingestion. 57,107,108 Vegetables, particularly onions and onion oil, containing relatively high amounts of these and other disulfides have long been associated with hemolytic anemia following accidental or intentional ingestion by dogs and farm animals.11, 109 Rats, however, are more resistant to dialkyl, but not diaryl, disulfide-induced hemolytic damage. 12
Repeated-Dose Toxicity
No repeated-dose studies were reported for DSO, but subchronic studies are available for DMDS, DPDS, and DPTS. In most cases, these studies have been performed by the inhalation route; however, in some instances, oral treatment has been utilized. In addition, 5- to 10-day repeated dose studies have been performed in rats with DEDS, DPDS, and DPTS. To facilitate comparison, route-to-route extrapolations have been performed for some of the key studies using the default body weight (0.3 kg) and ventilation rate (0.24 L/min) for the rat. 92
DMDS has been examined in 5 separate, well-designed dermal and inhalation studies. In the first study, male and female rats were exposed to 10, 50, 150, or 250 ppm (0.04, 0.19, 0.58, or 0.96 mg/L) DMDS 6 hours/d for 90 days. 110 Findings included decreases in body weight and food consumption, reduced thymus gland weights, and increased liver weights. Possible reductions in hemoglobin, red blood cell count, and packed cell volume were observed at the highest concentration. Histopathological changes were noted in the nose and spleen. Treatment-related changes in alanine aminotransferase, alkaline phosphatase, and total bilirubin indicated some degree of liver involvement. The no observed adverse effect levels (NOAEL) for this study was 10 ppm (11.1 mg/kg/d). In the second inhalation study, rats were exposed for 13 weeks to 5, 25, or 125 ppm (0.02, 0.10, or 0.48 mg/L) DMDS for 6 hours/d. 88 A treatment-related decrease in body weight gain, food consumption, and thymus weight was observed along with an increase in adrenal gland weight. Histopathology did not reveal any increase in the incidence or severity of abnormal tissue alterations relative to controls. Statistically significant decreases were also noted in serum alanine and aspartate aminotransferases and blood urea nitrogen at the 2 highest exposure concentrations; however, they were not considered toxicologically significant since they were not associated with any pathological changes. The NOAEL was 5 ppm (5.6 mg/kg/d) for male rats and 25 ppm (0.10 mg/L) for female rats.
The 2 dermal studies were performed in male and female New Zealand rabbits treated with DMDS for 6 hours/d by applying the neat material under an occlusive bandage. 13 In the first range-finding study, animals treated with DMDS levels of 0.1, 0.5, or 1 mL/kg/d (106, 505, or 1063 mg/kg/d) for 14 days caused dose-related lethargy or unconsciousness in all treatment groups that dissipated by the end of the day. 111 Severe treatment-related skin lesions were also observed in all 3 treatment groups. Although a NOAEL could not be determined, a LOAEL of 106 mg/kg/d was assigned. In the second study, the rabbits were treated dermally at levels of 0.01, 0.1, or 1 mL/kg/d (10.6, 106.3, or 1063 mg/kg/d) for 28 days. 112 Consistent with the range-finding studies, dose-related changes in lethargy and skin irritation were also observed in the more prolonged study. After 13 days, mortality was observed in the rabbits from the high-dose group, so treatment was terminated. The male rabbits from the high-dose group also displayed some abnormal changes in hematology and clinical chemistry measurements that were not observed in the female rabbits. Histopathological examination and organ weight measurements did not reveal any treatment-related changes in the adrenals, brain, heart, kidneys, liver, lungs, ovaries, testis, thyroid, or thymus. The NOAEL for systemic effects was 10.6 mg/kg/d, and the NOAEL for localized dermal irritation was less than 10.6 mg/kg/d.
A subchronic neurotoxicity study performed in rats exposed to 5, 20, or 80 ppm (0.02, 0.08, or 0.31 mg/L) 6 hours/d and 7 days/week for 13 weeks produced only mild effect. 113 The treatments showed no apparent effect in the functional observational battery or on motor activity. Histopathological examination of the nervous system was similarly unremarkable as were measurements of brain weight and length. The NOEL for neurotoxic effects was 80 ppm (88.7 mg/kg/d); however, the observed degeneration of olfactory epithelium in both sexes resulted in an overall subchronic NOEL of 5 ppm (5.6 mg/kg/d).
A 90-day oral feeding study with DPDS did not reveal any toxic effects following the dietary administration of 7.3 mg/kg/d or 8.2 mg/kg/d to male or female rats, respectively. 90 Food consumption and body weights were recorded weekly, and hematological examinations and blood urea nitrogen measurements were performed on half of the animals at 7 weeks and on the remaining animals at 13 weeks. A slight nonstatistical increase in blood urea nitrogen was observed at the end of the study. Organ weight measurements, gross examinations, and tissue histopathology performed at necropsy failed to show any treatment-related effects. Similarly, male and female rats administered a single dose of 4.8 mg/kg DPTS in their feed for 90-days did not exhibit any signs of toxicity. 91 Thorough clinical examinations were performed including clinical chemistry, urinalysis, hematology, and histopathology. Erythrocyte counts, hemoglobin levels, and hematocrit were all within the normal range.
The toxicity of DPDS and DPTS was also evaluated in female rats following a 5-day oral treatment with an equimolar dose level of 75 mg/kg/d or 91 mg/kg/d, respectively. 86 Compared to untreated controls, DPTS but not DPDS produced a statistically significant decrease in packed red cell volume and hemoglobin levels along with an increase in relative spleen weight. In addition, the trisulfide caused an increase in splenic and hepatic erythropoietic activity that was not observed with the disulfide. Glutathione depletion, methemoglobin formation, and hydrogen peroxide formation were also more severe with DPTS than DPDS. These data along with enzyme activity measurements demonstrated that DPTS was a more potent inducer of ROS formation and hematological toxicity than DPDS.
A 10-day inhalation study is available with DEDS in male and female rats at exposure levels of approximately 50, 150, or 450 ppm (0.25, 0.75, or 2.25 mg/L). 89 Gross examination along with histopathology, hematology, and clinical measurements revealed that decreased body weight gain was the only affected end point at the lowest exposure level. Female rats exposed at 50 ppm did, however, exhibit darkened spleens. The 150-ppm exposure group displayed changes in body weight and relative organ weight, whereas the 450-ppm group showed clear evidence of hemolytic anemia with significant decreases in red blood cell counts, hemoglobin, and packed red cell volume. Other gross and histopathological abnormalities observed in the 450 ppm exposure group included a statistically significant decrease in absolute liver and testis weight in male rats, an absolute and relative increase in spleen weight, and evidence of extramedullary hematopoiesis in the liver and spleen. The NOAEL and LOAEL from this study were determined to be 50 ppm (72 mg/kg/d) and 150 ppm (216 mg/kg/d), respectively.
Despite in vivo and in vitro evidence suggesting that trisulfides could be more toxic than the corresponding disulfides, an examination of the findings from repeated dose studies indicates that this difference is limited to potency with no shift of toxic thresholds (see Table 8). This is evident from a comparison of the 90-day results for DPDS and DPTS, which shows no appreciable difference in the NOAELs for these 2 chemicals. Consequently, the observed difference in oxidative stress and redox cycling for disulfides and trisulfides represents a difference in toxic potency rather than a change in the effective threshold dose capable of eliciting a toxic response. Although the disulfides and trisulfides operate through a common mode of action, the difference is strictly a matter of response intensity rather than response threshold. The results displayed in Table 8 are consistent with this supposition and show that of the disulfides and trisulfides examined, all displayed relatively similar no-effect levels. Consequently, from a risk perspective, DMDS can serve as a proxy for the dialkyl disulfides and trisulfides in DSO, since the no-effect levels observed in repeated dose studies are not substantially different.
Genotoxicity
Although there are no results available for DSO, DMDS has been examined in a variety of in vivo and in vitro genetic toxicology screening assays. 13 The test results revealed that DMDS was negative in bacterial mutagenicity assays, negative in mammalian mutagenicity tests, and negative for DNA damage and repair. 114 –116 Except for the DNA damage and repair assay, these tests were performed in the presence and absence of metabolic activation. A more recent mutagenicity study with Salmonella typhimurium strains TA 98, TA 100, TA 135, TA 1537, and Escherichia coli strain WP2 uvrA failed to produce a positive result with or without activation at DMDS concentrations ranging from 1.5 to 5000 μg/plate. 117 Similarly, negative results were obtained when DMDS was evaluated in vivo in a mouse micronucleus assay at inhalation concentrations of 250 or 500 ppm, and DMDS did not cause unscheduled DNA synthesis in the hepatocytes of rats exposed to 500 ppm. 118,119 A second micronucleus assay performed in rats exposed to 212, 425, or 850 ppm for 4 hours yielded similarly negative results. 120 Chromosomal aberrations were detected with and without S9 activation in primary human lymphocyte cultures from male volunteers, but only under maximum in vitro exposure conditions, which proved cytotoxic (300 µg/mL DMDS for 2 or 24 hours). 121 By comparison, DPDS did not cause any reverse mutations in an Ames S. typhimurium assay using strain TA98. 122 None of the disulfides in DSO were judged to be genotoxic by an expert knowledge-based system used to predict the health effects of untested chemical substances (Derek, v 9.0.0). 123
Reproductive and Developmental Toxicity
Although no studies were reported on the reproductive or developmental toxicity of DSO, DMDS has received a great deal of attention. The DMDS was evaluated in rats or rabbits using reproductive, prenatal, lactational, or developmental study designs. These studies were performed using the inhalation route of exposure using established methodologies for assessing the appearance of an adverse effect. Most of these studies were performed in rats, but a single teratology study was performed in a group of 24 pregnant rabbits exposed to 15, 45, or 135 ppm (0.06, 0.18, or 0.53 mg/L) of DMDS for 6 hours/d on day 6 through day 28 of gestation. 124 The DMDS exposures did not result in any treatment-related effects on the fetuses; however, a decrease in food consumption in the does exposed to highest exposure concentration resulted in a maternal NOEL of 45 ppm. The NOEL for teratogenicity was determined to be 135 ppm.
Range-finding and detailed teratology studies were also performed in rats exposed by the inhalation route. The range finding assessment was performed in pregnant dams exposed for 6 hours/d on day 6 through day 15 of gestation to 10, 50, or 250 ppm (0.04, 0.19, or 0.96 mg/L) of DMDS. 125 Treatment-related reductions in body weight gain and food consumption were observed in all-treatment groups, but pregnancy incidence, intrauterine death incidence, preimplantation loss, litter size, sex ratio, and the incidence of malformations were all within the normal range. Mean fetal weights showed an exposure-related reduction in all-treatment groups that was considered to be an equivocal finding. The maternal NOAEL was determined to be less than 10 ppm. In the subsequent detailed study, 3 groups of 30 mated female rats were exposed to DMDS by whole-body exposure at 5, 15, or 50 ppm (0.02, 0.06, or 0.19 mg/L) for 6 hours daily from day 6 to day 15 of gestation. 126 All animals were maintained until day 20 of gestation and then sacrificed. No deaths or unusual lesions were observed, but a higher incidence of rough hair coat was seen at 50 ppm. Clinical conditions at 5 and 15 ppm did not differ from controls. Treatment-related reductions in weight gain were observed at 15 and 50 ppm. Food intake was lower than controls at 50 ppm but comparable at 5 and 15 ppm. There was no effect of the treatment on pre- or postimplantation loss, litter size, or sex ratio. Maternal toxicity was noted at 15 and 50 ppm, but there was no evidence of developmental effects. Litter and fetal weights were reduced at 50 ppm. No malformations were observed in fetuses from the treated groups. A slightly higher incidence of retarded ossification was observed at 50 ppm, which indicated delayed maturation as a result of the lower fetal weight rather than overt teratogenicity. The NOELs for maternal toxicity, fetotoxicity, and teratogenicity were 5, 15, and 50 ppm, respectively.
A range-finding developmental neurotoxicity test was performed in lactating dams with nursing litters. The study was conducted in rats exposed to 2, 20, or 80 ppm (0.008, 0.08, or 0.30 mg/L) for 6 hours/d on postpartum days 5 through 20. 127 The dams showed no treatment-related effects on mortality or mean body weights; however, food consumption was decreased in the high-exposure group. The offspring showed no treatment-related effects on survival, but the mean body weight was affected in the dams exposed to 80 ppm. The maternal and developmental NOEL was found to be 20 ppm. Similar findings were obtained in a lactational study performed in a group of 36 nursing rats exposed to 5, 20, or 80 ppm (0.02, 0.08, or 0.30 mg/L) of DMDS for 6 hours/d on either day 5 through 12 (subset I), day 13 through 20 (subset II), or day 5 through day 20 (subset III) of lactation. 128 The dams in subset I and III showed a decrease in mean body weight gain at the 2 highest exposure concentrations (20 and 80 ppm), whereas the dams in subgroup II only showed a decrease following the 80-ppm exposure. The dams in subset I and II also showed a decrease in food consumption. Offspring survival and body weight gain were unaffected in all exposure concentrations and treatment subgroups. These findings resulted in a maternal NOEL of 5 ppm and a developmental NOEL of 80 ppm.
A prenatal teratogenicity study was performed in a group of 27 mated rats exposed to 5, 20, or 80 ppm (0.02, 0.08, or 0.30 mg/L) of DMDS for 6 hours/d on day 6 through day 19 of gestation. 129 The dams in the 20- and 80-ppm exposure group showed a decrease in weight gain and a decline in food consumption. Fetuses in the high-exposure group displayed a decrease in food consumption and delayed ossification of the sternebra and other bones. Based on these findings, the maternal NOEL was determined to be 5 ppm, and the NOEL for teratogenic effects was 20 ppm. The effects of DMDS on reproductive organs were assessed in male and female rats exposed to 10, 50, 150, or 250 ppm (0.04, 0.19, 0.58, or 0.96 mg/L) DMDS for 6 hours/d for 90 days. 110 Tissue histopathology did not reveal any lesions or damage to the epididymides, prostate, or testes of the male rats nor ovaries or uterus of female rats.
Finally, a 2-generation reproductive study was performed in a group of 30 rats exposed to 5, 20, or 80 ppm (0.02, 0.08, or 0.30 mg/L) of DMDS for 6 hours/d, 7 days/week through the F0 and F1 generation. 130 The F0 generation was exposed for 10 weeks prior to mating up through day 20 of gestation. Exposures were then suspended from day 21 of gestation through day 4 of lactation and resumed on day 5 of lactation to weaning. Mean body weight and food consumption declines were initially noted in male and female rats from both generations exposed to 20 or 80 ppm. The exposures did not adversely affect litter size or pup survival, but mean body weight declines were observed at all treatment levels for males from the F1 generation. This change was more severe for the 5-ppm exposure throughout the lactation period and was not found to be treatment related. No adverse effects were observed for spermatogenesis, reproductive performance, or parturition. The parental NOEL was determined to be 5 ppm based on the body weight declines. The reproductive and developmental NOELs were found to be 80 ppm based on the lack of any notable treatment-related effects in the dams or offspring from either generation.
Carcinogenicity
The carcinogenicity of DSO or its individual disulfide or trisulfide constituents was not specifically determined in long-term animal studies. An examination of ancillary evidence suggests, however, that dialkyl disulfides and trisulfides are not likely to pose an appreciable carcinogenic hazard to humans by any route of exposure. As stated previously, DMDS and DMTS are present in garlic and other Allium species in sizable concentrations ranging from approximately 2 to 20 μg/g, and repeated epidemiological studies have generally shown a decrease in stomach, esophageal, and colorectal cancers in those who regularly consume large amounts of Allium vegetables. 131 Although this effect is generally attributed to the diallyl disulfide and trisulfide contents, the antineoplastic effects of the dialkyl disulfides and trisulfides cannot be completely overlooked. The relative concentration ratio of DMDS, DMTS, diallyl disulfide, and diallyl trisulfide in a typical garlic bulb is in the order 1:7:220:410 with high levels of other diallylic compounds also present.
Genotoxicity studies with DMDS have generally been negative with little evidence of a mutagenic response in either prokaryotic or eukaryotic cell systems. This finding is not surprising, given the absence of any structural alerts that typify a chemical with mutagenic or carcinogenetic potential. 132 Although much has been written on the potential therapeutic effects of disulfides and their ability to inhibit chemically induced DNA damage, these effects need to be tempered with the knowledge that disulfides and trisulfides are potentially capable of causing oxidative DNA damage through ROS generation. 133,134 However, research to date suggests that the beneficial effects of dietary dialkyl disulfide and trisulfide exposure outweigh the risks of any long-term health effects. 50
Conclusions
The preceding examination of the physical properties, health effects, and mode of action of the disulfides in DSO demonstrates that DMDS can be used as a reasonable worst-case surrogate for this substance. Strong and consistent mechanistic evidence shows that DMDS is at least as toxic as other dialkyl disulfides and trisulfides, and that the higher molecular weight sulfur-containing chemicals in DSO do not pose a greater health threat or environmental hazard. A presentation of all available findings to the USEPA under the HPVCP resulted in Agency agreement that all testing requirements were met under this voluntary agreement with no appreciable data gaps requiring further testing. 8 An examination of all available evidence indicates that DSO is highly hazardous to aquatic organisms with moderate environmental persistence. The DSO is also predicted to possess low to moderate acute toxicity, but high subchronic toxicity with a NOAEL less than 25 mg/kg/d. The potential for genotoxic, carcinogenic, and reproductive/developmental effects is not predicted to be a source of concern. This case study provides a good example of how structure activity considerations can be effectively used to identify the health hazards of a substance containing chemically related congeners, thereby saving time and eliminating the need for unnecessary animal testing.
Footnotes
Acknowledgments
The authors appreciate the assistance and sponsorship provided by members of the American Petroleum Institute’s Petroleum HPV Testing Group who offered constructive advice and criticism throughout the preparation of this article. The authors alone are responsible for the content and preparation of this article.
Declaration of Conflicting Interests
David Morgott, Christopher Lewis, and James Bootman work as private toxicology consultants under contract with LyondellBasell, producers of disulfide oil. Marcy Banton is an employee of LyondellBasell.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Several of the authors (DM, CL, and JB) have received financial support from LyondellBasell in conjunction with their contractual responsibilities with the sponsor. Funding was similarly provided for researching, authoring, and submitting this article.
Supplemental Materials
Supplemental Table 1. Chemical Composition of Disulfide Oil. Supplemental Table 2. Estimated Physiochemical Constants from EPI Suite.
