Abstract
Introduction
However, today it is well known that a great number of phytochemicals alter the activities and expression levels of drug-metabolizing enzymes, particularly cytochrome P450s (CYPs), thus affecting the individual response to a wide array of clinical drug compounds and thousands of chemical carcinogens. There are many scientific evidence that natural preparations taken simultaneously with clinical drugs may cause unexpected life-threatening pharmacological effects like adverse drug reactions, therapeutic failure, and toxicity by affecting drug metabolism. 5 –7 In addition, there are also many reports that phytochemicals may also cause changes in the metabolisms of precarcinogens, thus potentiating the mutagenesis and malignant transformation, or conversely, exerting anticarcinogenic and antimutagenic effects. 6,8 For this reason, identifying the CYP-mediated pharmacokinetic interactions of natural supplements is very important with regard to chemical carcinogeneses, drug metabolisms, and therapeutics.
The CYPs, a superfamily of heme-thiolate proteins, are the terminal enzymes of nicotinamide adenine dinucleotide phosphate (NADPH)-CYP-dependent monooxygenase system. This system plays a critical role in detoxification and bioactivation to yield reactive metabolites that initiate toxic, mutagenic, and carcinogenic events. 9 Multiple isozymes of P450 show different substrate specificities and affinities toward a wide array of xenobiotic substances including environmental contaminants, food additives, phytochemicals, and prescribed drugs. Among CYP isozymes, CYP1, CYP2, and CYP3 families, specifically CYP1A2, CYP2B6, CYP2C9, CYP2E1, and CYP3A4, are involved in the phase I metabolisms of 75% to 80% of clinical drug compounds and thousands of chemicals. 9,10 Within this group, CYP2B6, CYP2C9, and CYP3A4 are mainly involved in drug metabolism; especially, CYP3A4 metabolizes about 50% of all clinical drug compounds thus playing a major role in detoxification of various prescribed drugs. 10 In many studies, the induction and inhibition of this isozyme were described by a number of flavonoids having the potential to change the pharmacokinetic properties of the coadministered drugs. 11 –13 Despite their minor role in drug metabolism, both CYP1A2 and CYP2E1 mainly participate in the metabolic activation of chemical compounds exposed occupationally or ordinarily during routine life, therefore partly contributing to chemical carcinogenesis. It is demonstrated that although some phytochemicals may antagonize the carcinogenic and mutagenic properties of certain precarcinogens through the inhibition of CYP1A2 and/or CYP2E1, some of them increase bioactivation and therefore carcinogenic and mutagenic effects through enzyme induction. 5,8
The SP contains a complex mixture of a variety of phytochemicals, including some vitamins such as ascorbic acid, α-tocopherol, and β-carotenes, some phytopigments like zeaxanthin, chlorophyll-a, xanthophyll, myxoxanthophyll, canthaxanthin, c-phycocyanin, and allophycocyanin, and many other phenolic compounds. 4,14 Taking into account the diverse phytochemical constituents of SP, little is known about its effects on the CYP system. To our knowledge, there is no report available until now describing the effect of SP on the individual change in pharmacologically and toxicologically important CYP isozymes. For this reason, potential interactions are generally not predictable when it is coadministered with other drugs. Infact, although SPs have been claimed to be safe because of its natural origin and long history of use as traditional food, several adverse reactions have been reported with the concomitant drug use. For example, Iwasa et al 15 reported an elevated liver enzyme concentration (alanine aminotransferase and aspartate aminotransferase) in a 52-year-old Japanese patient who used a Spirulina preparation for 5 weeks with concurrent use of simvastatin and amlodipine. Similarly, adverse reactions were reported in a female patient who used a product containing approximately 1400 mg SP for 4 days concurrently with a medication, Synthroid. 16 It is likely that such interactions may be more common. However, they may be underreported, or most probably the observed adverse effects are generally attributed to the disease for which the treatment is taken. Therefore, clearly, studies are needed in order to assess the potential impact of this, one of the most consumed natural preparation—SP, on selective hepatic P450. In the present study, an attempt has been made to evaluate the effects of SP preparation on in vivo activity and expression of pivotal hepatic CYP isozymes including CYP1A2, CYP2B, CYP2C, CYP2E1, and CYP3A. This information may help to infer possible drug/chemical interaction potential of SP and to decrease the incidence of adverse reactions.
Materials and Methods
Chemicals
The following chemicals were purchased from Sigma-Aldrich Chemical Company (St Louis, Missouri): acrylamide, aniline, bovine serum albumin (BSA), caffeine, formaldehyde (HCOH), folin phenol reagent,
Animals and Treatments
The
Healthy male Wistar albino rats (12 weeks old) were purchased from a certificated laboratory for the production and care of experimental animals. After 7 days of adaptation period, SP suspension was given to the animals for 5 consecutive weeks by oral gavage. The SP suspension was prepared in water everyday freshly. For the SP application, animals were randomly divided into 4 groups. (1) Control-water, (2) SP 200 mg/kg/body weight (bw), (3) 500 mg/kg/bw, and (4) 1000 mg/kg/bw. The dose range was consistent with doses used in other studies found in the literature. All animals appeared healthy and active throughout the experiment. However, 1 animal from 500 mg/kg/bw SP group died 5 days after the dosing was started despite the fact that no apparent toxic effects could be evidenced at the autopsy. Each group contained 8 animals, except the 500 mg/kg/bw SP dose group, which contained 7 animals. Rats were housed in small cages at an ambient temperature of 22 ± 1°C on a 12-hour light/dark cycle and were fed commercial rat food with water ad libitum. At the end of the experimental period, rats were fasted for 16 hours and anesthetized subcutaneously with alfamine. Then, animals were euthanized by cervical dislocation. Liver samples were removed, weighed, and rinsed with cold physiological saline and stored at −80°C until analyses. The study design was approved by the Institutional Ethical Committee for Experimental Animal Care and Use at the Çanakkale Onsekiz Mart University (Canakkale, Turkey).
Preparation of Tissue Microsomes
Tissues were homogenized in 4-part homogenization solution (1.15% KCl containing 3 mmol/L EDTA, 0.5 mmol/L PMSF, 0.3 mmol/L e-aminocaproic acid, 0.15 mmol/L butylated hydroxytoluene, and 0.025% Triton X-100) using a tissue homogenizer with a teflon pestle at 4°C. Subcellular fractions of rat tissues were prepared by standard differential centrifugation with calcium aggregation as described by Sen and Kirikbakan. 17 The amount of protein in individual fractions was measured using the method of Lowry et al, 18 with BSA as the standard.
Microsomal CYP Enzyme Assays
The CYP1A2-dependent caffeine N-demethylase (CND), CYP2C6-dependent aminopyrene N-demethylase (APND), and CYP3A1-dependent erythromycin N-demethylase (END) activities of rat liver microsomes were determined by measuring the quantity of HCOH formed, according to the method of Nash 19 and modified by Cochin and Axelrod. 20 The CYP2B1-dependent 7-benzyloxyresorufin O-deethylase (BROD) activity of rat liver microsomes was measured according to the method of Burke and Mayer 21 optimized by Arinc and Sen. 22 Microsomal CYP2E1-dependent aniline 4-hydroxylase (A4H) activity of rat liver microsomes was determined by measuring the quantity of p-aminophenol formed, as described by Imai et al. 23
Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis and Western Blot Analysis
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was performed on 4% stacking gel and 8.5% separating gel in a discontinuous buffer system as described by Laemmli. 24 Rat liver microsomes from control and SP-treated groups were analyzed by Western blot analysis, essentially as described by Towbin et al. 25 Details of the procedure were given by Arinc and Sen. 26 Accordingly, microsomal proteins, each 30 µg, were subjected to SDS-PAGE as described earlier, and separated proteins were transferred to nitrocellulose membrane using the trans-blot membrane transfer cell (Thermo, VEP2) containing Tris-glycine buffer, pH 8.3, and methanol at 90% (v/v), for 90 minutes. Following transfer, the membranes were blocked using 5% nonfat dry milk in 20 mmol/L Tris-HCl, pH 7.4, 400 mmol/L NaCl, and 0.1%, v/v Tween 20 (TBST) for 60 minutes and incubated with rabbit polyclonal anti-rat CYP1A2, CYP2E1, CYP2B1, CYP2C6, or CYP3A1 antibodies (diluted 1:1000 in blocking solution) for 120 minutes at room temperature. The membranes were then washed with TBST (3 × 5 minutes), incubated with secondary antibody (ALP-conjugated anti-rabbit immunoglobulin G at a 1:5000 or 1:10,000 dilution) for 60 minutes, and again washed with TBST (3 × 5 minutes). Visualization of the bands was carried out using the NBT/BCIP substrate system. The final images were photographed using computer–based, gel-imaging instrument and analyzed using the Scion Image Version Beta 4.0.2 software.
RNA Isolation and Reverse Transcription-Polymerase Chain Reaction of CYP Messenger RNAs
Total RNA was extracted from 100 mg rat livers using Trizol reagent. Extracted RNA was quantified spectrophotometrically at 260/280 nm, and the integrity was checked using 1% agarose gel. For complementary DNA (cDNA) synthesis, EasyScript Plus, cDNA Synthesis Kit was used. Semiquantitative reverse transcriptase-polymerase chain reaction (RT-PCR) assay was performed using gene-specific primers. The oligo sequences used as forward and reverse primers for rat CYP450 isozymes were based on those reported in Minn et al. 27 β-Actin was used as housekeeping gene. Preliminary control experiments (data not shown) were carried out to verify the RT-PCR conditions allowed for linear amplification of the PCR products. The PCR products were analyzed by electrophoresis on 1.5% agarose gels containing ethidium bromide. The intensity of the bands was measured using Scion Image Version Beta 4.0.2 software. Levels of messenger RNA (mRNA) for CYP genes were determined by measuring the band intensity of the RT-PCR product on each agarose gel and were reported relative to β-actin expression.
Statistical Analysis
Each experimental procedure was performed at least 3 times. Statistical analyses were performed using the SPSS Windows Version release 16.0. Data were expressed as means ± standard error of the means. Group means were compared by a Student
Results
In order to evaluate the effects of SP application for 5 consecutive weeks on the expression and activity level of rat liver CYP-dependent, drug-metabolizing enzymes, we first determined the enzymatic activities. Isozyme-specific enzyme activities and percentages of inhibition are presented in Table 1. Accordingly, as given in Table 1, CYP1A2-associated CND activities were significantly decreased about 35%, 22%, and 26% for 200, 500, and 1000 mg/kg/bw SP doses, respectively, compared to the control group (
The Rat Liver Microsomal Activities of CYP Isozymes.
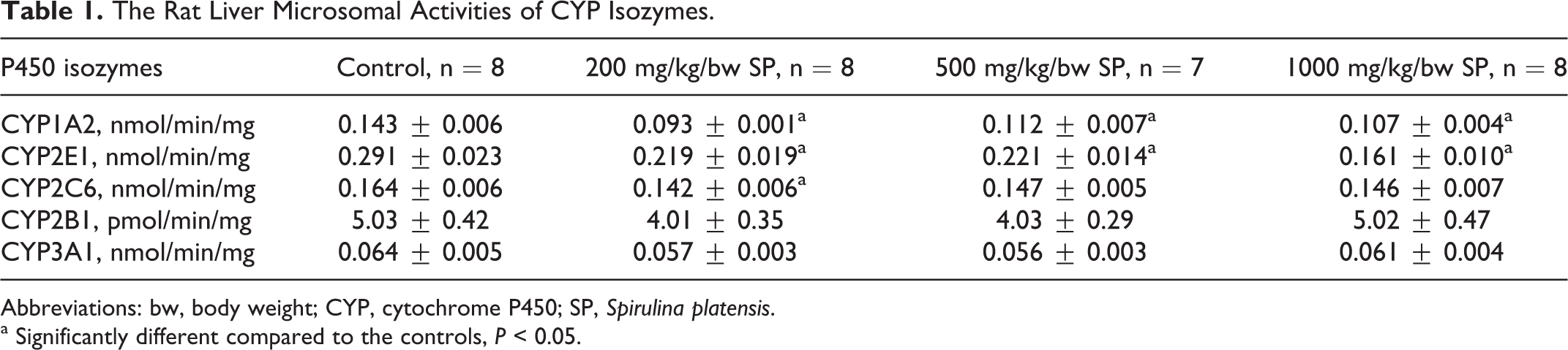
Abbreviations: bw, body weight; CYP, cytochrome P450; SP,
a Significantly different compared to the controls,
The densitometric analyses obtained from Western blot and RT-PCR are demonstrated in Figures 1 to 3. The mean intensity of the bands obtained from Western blot and RT-PCR obtained from the control animals was taken to be 100%, and the value obtained from SP-treated group was expressed as percentage of control. As represented in Figure 1A and C, the densitometric analyses of Western blot for CYP1A2 isozyme showed that the protein levels decreased about 67%, 30%, and 44% for 200, 500, and 1000 mg/kg/bw SP doses, respectively, when compared to controls (
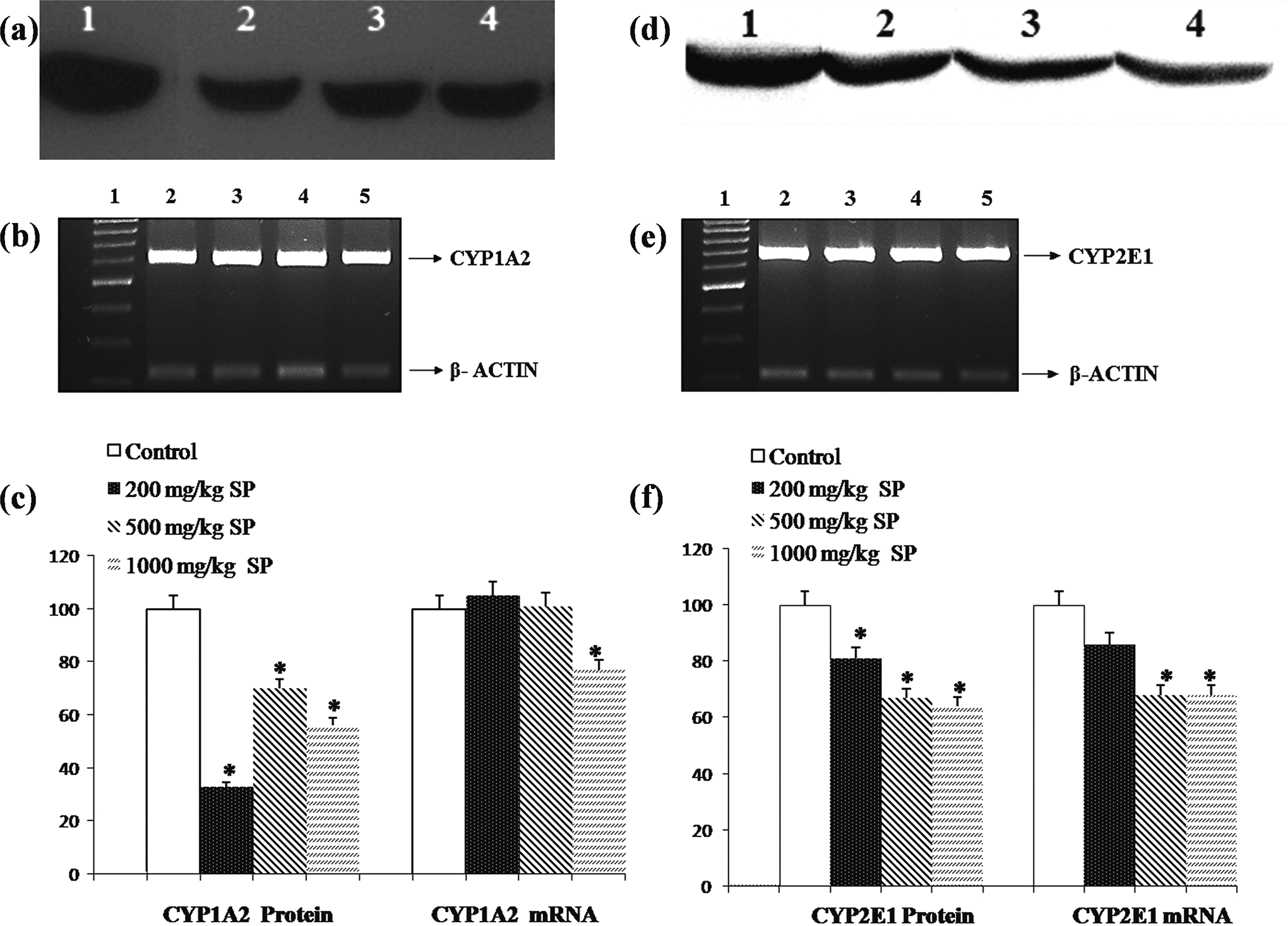
The expression level of CYP1A2 and CYP2E1 protein and mRNA in control and SP-treated rats. A Representative immunoblot analysis of liver microsomal CYP1A2 and (D) CYP2E1 proteins in experimental groups. For both (A) and (D), lane 1: control, lane 2: 200 mg/kg/bw SP treatment, lane 3: 500 mg/kg/bw SP treatment, and lane 4: 1000 mg/kg/bw SP treatment. B, Representative agarose gel showing the effect of SP treatments on regulation of CYP1A2 and (E) CYP2E1 mRNA expressions in liver, analyzed by reverse transcriptase-polymerase chain reaction (RT-PCR). For both (B) and (E), lane 1: 1 kDa DNA ladder, lane 2: control, lane 3: 200 mg/kg/bw SP treatment, lane 4: 500 mg/kg/bw SP treatment, and lane 5: 1000 mg/kg/bw SP treatment. C, Comparison of CYP1A2 and (F) CYP2E1 protein and mRNA levels among experimental groups. Experiments were repeated at least 3 times. Results are presented as mean ± standard deviation. *
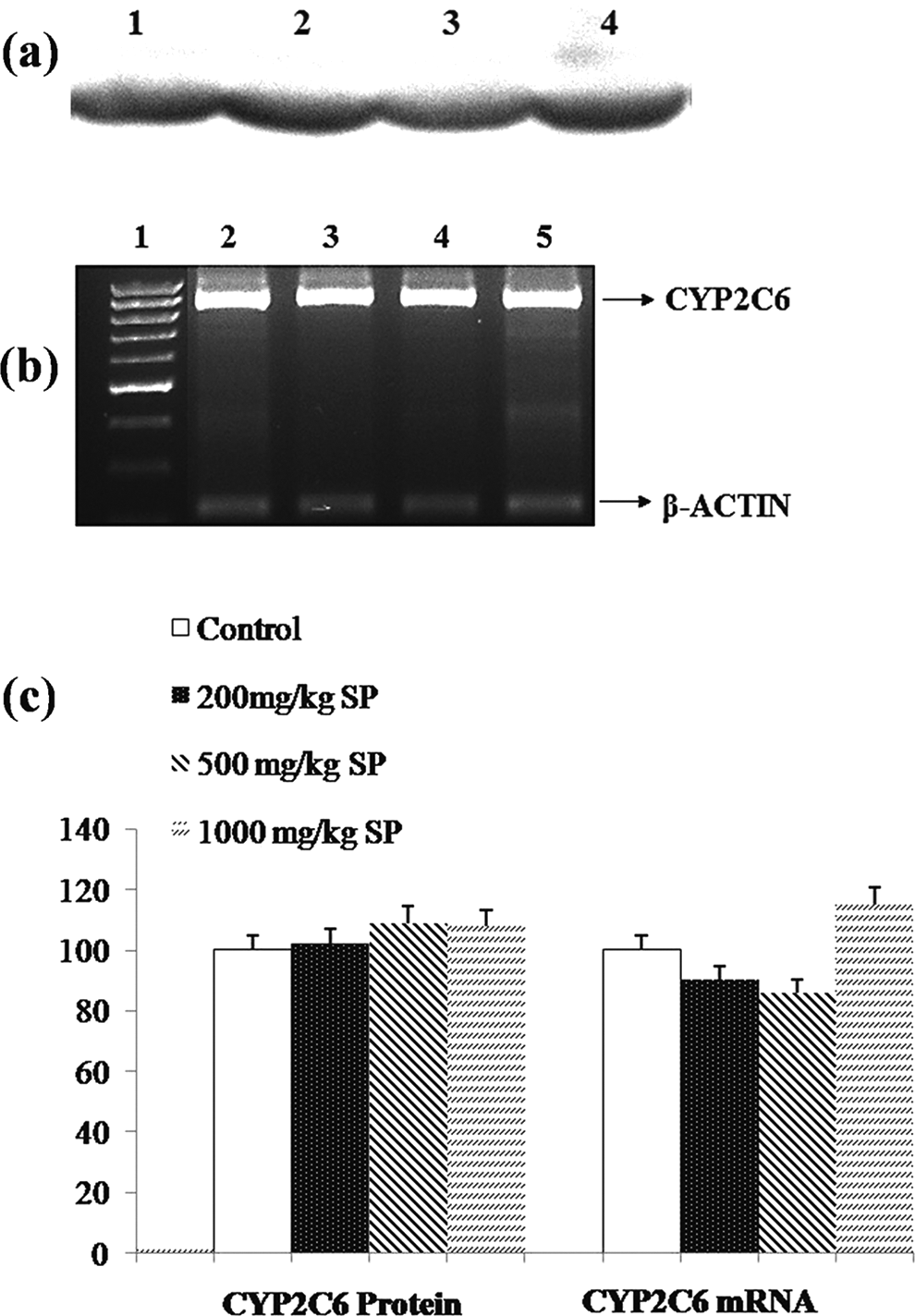
The expression level of CYP2C6 protein and mRNA in control and SP-treated rats. A, Representative immunoblot analysis of liver microsomal CYP2C6 proteins in experimental groups. Lane 1: control, lane 2: 200 mg/kg/bw SP treatment, lane 3: 500 mg/kg/bw SP treatment, and lane 4: 1000 mg/kg/bw SP treatment. B, Representative agarose gel showing the effect of SP treatments on regulation of CYP2C6 mRNA expressions in liver, analyzed by reverse transcription-polymerase chain reaction (RT-PCR). Lane 1: 1 kDa DNA ladder, lane 2: control, lane 3: 200 mg/kg/bw SP treatment, lane 4: 500 mg/kg/bw SP treatment, and lane 5: 1000 mg/kg/bw SP treatment. C, Comparison of CYP2C6 protein and mRNA levels among experimental groups. Experiments were repeated at least 3 times. Results are presented as mean ± standard deviation. *
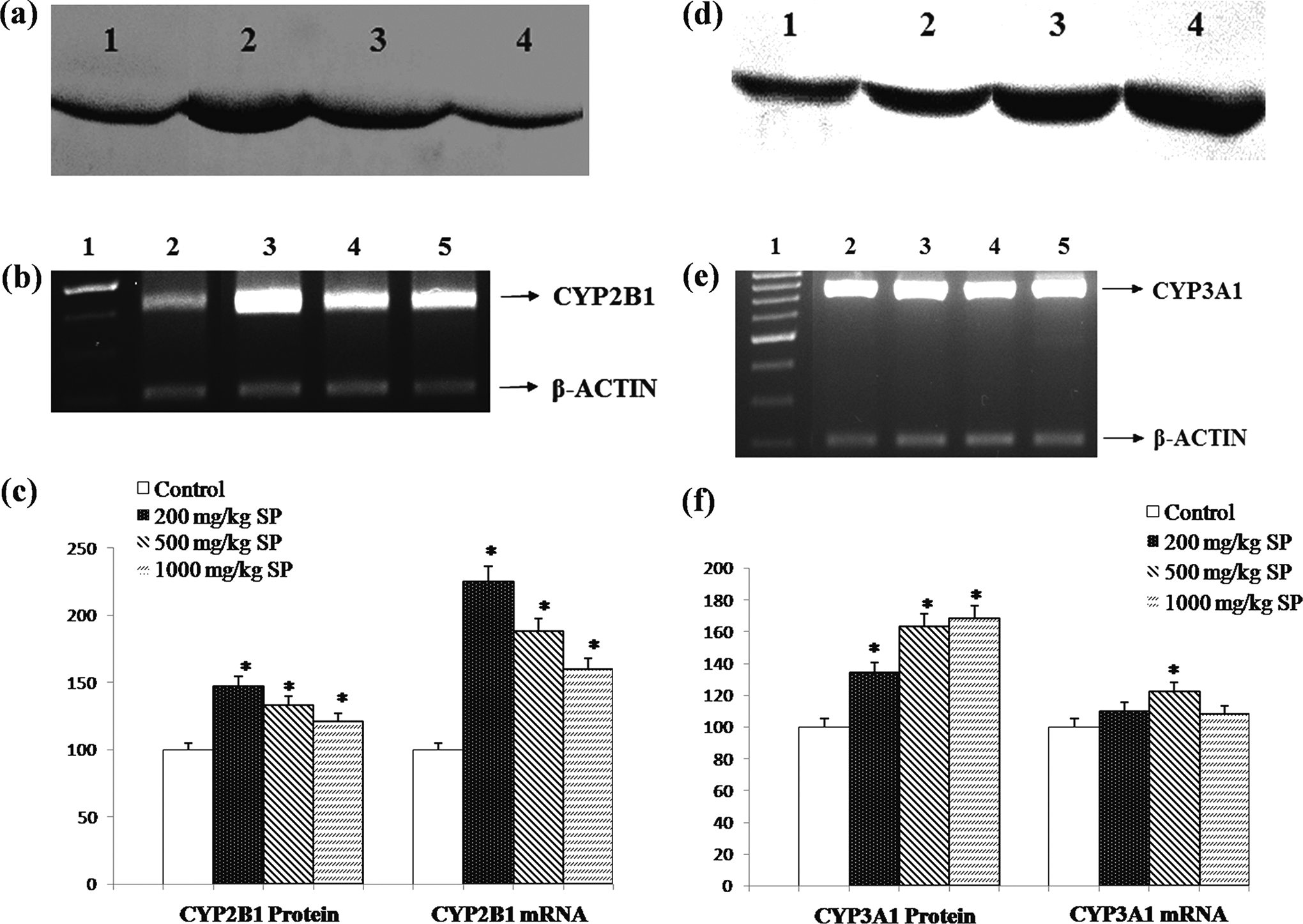
The expression level of CYP2B1 and CYP3A1 protein and mRNA in control and SP-treated rats. A, Representative immunoblot analysis of liver microsomal CYP2B1 and (D) CYP3A1 proteins in experimental groups. For both (A) and (D), lane 1: control, lane 2: 200 mg/kg/bw SP treatment, lane 3: 500 mg/kg/bw SP treatment, and lane 4: 1000 mg/kg/bw SP treatment. B, Representative agarose gel showing the effect of SP treatments on regulation of CYP2B1 and (E) CYP3A1 mRNA expressions in liver, analyzed by reverse transcriptase-polymerase chain reaction (RT-PCR). For both (B) and (E), lane 1: 1 kDa DNA ladder, lane 2: control, lane 3: 200 mg/kg/bw SP treatment, lane 4: 500 mg/kg/bw SP treatment, and lane 5: 1000 mg/kg/bw SP treatment. C, Comparison of CYP2B1 and (F) CYP3A1 protein and mRNA levels among experimental groups. Experiments were repeated at least 3 times. Results are presented as mean ± standard deviation. *
It was observed that the relative mRNA level for CYP1A2 decreased significantly, that is about 23%, at the highest (1000 mg/kg/bw) SP dose. However, other doses did not exhibit a significant change in terms of mRNA level (Figure lB and C). In the case of CYP2E1, the relative mRNA levels were decreased significantly about 30% (
Discussion
During the last 20 years, an explosion has been seen in the consumption of herbal or other natural preparations all over the world, especially among older people and chronic patient groups who are more likely to receive routine clinical medicine and also more sensitive to carcinogenic chemicals found in the environment and diet. In the scope of current work, we aimed to elucidate the effects of SP, one of the most widely used natural preparation all over the world, on pharmacologically and toxicologically important CYPs, namely, CYP1A2, 2E1, 2B1, 2C6, and 3A4, for the first time. In this study, we choose to evaluate the effects of whole SP preparation, which contains 100% pure dried biomass of SP, rather than its active bioconstituents for 2 reasons, (1) people usually use the whole natural preparation and not isolated constituents and (2) each of the active compounds found in the complex mixture acts on the metabolic enzymes additively or synergistically so that the resulting overall effects become very different from the expected one. Such informations are very important to infer possible drug interactions when SP is used concomitantly with clinical drugs and very helpful to decrease the incidence of adverse drug reactions.
In the current study, the doses of SP used for in vivo administration to rats were selected considering the previous reports 28 –30 and daily recommended doses for human use, which vary between the range of 500 mg and 13.5 g as given in different commercial products. This dosage range was also preferred in the clinical studies of SP performed with different patient groups such as patients with human immunodeficiency virus(10 g/d), 31 allergic rhinitis (2 g/d), 32 and hyperlipidemic nephritic syndrome (1 g/d). 33 With basic allometric scaling based on body surface area, 34 the experimental doses utilized in the present study (200, 500, and 1000 mg/kg) correspond to approximately 2, 5, and 10 g of daily human equivalent doses for a 60-kg person, respectively. Therefore, chosen doses in the current work represent examples of low, middle, and high levels of recommended daily human doses.
Examining the results, according to Table 1 and Figure 1A and C, it indicated that CYP1A2-associated enzyme activity and protein level in rat liver microsomes significantly decreased at all SP doses compared to controls. The magnitudes of decrease were considerably high, especially at the lowest dose group that was 35% for catalytic activity and 67% for protein level. The CYP1A2 is a kind of toxicologically important CYP isozyme, which accounted for 17% of the P450 activation reactions. 35 It especially plays a role in the bioactivation of polyaromatic hydrocarbons, polyaromatic biphenyls, and dioxin-type environmental contaminants as well as dietary procarcinogens like aromatic amines and aflotoxin. 9,36 In this aspect, the results of the present work indicate that SP has inhibitory effect on metabolic activation of various toxic and carcinogenic chemicals metabolized by CYP1A2, which may in turn decrease the individual risk of organ toxicity, mutagenesis, and carcinognesis induced by chemicals. Actually, the current data may also explain the findings of previous reports demonstrating the protective actions of SP on the treatment by procarcinogenic compounds such as aflatoxin, 37 5-fluorouracil, 38 and 4-nitroquinoline-1-oxide, 30 which are mainly activated by CYP1A2 toxic and carcinogenic to metabolites.
The CYP2E1, accounting for 11% of P450 activation reactions, is also another important enzyme mainly playing a role in the metabolism of environmental and dietary chemicals. 35 This isoform is especially responsible for the metabolic activation of small molecules like acrylamide, benzene, chloroform, and nitrosamines into their reactive metabolites, forming covalent adduct with cellular macromolecules including DNA, which may finally lead to malignant transformation. 39 –41 As represented in Figure 1D–F, our data demonstrated that SP treatment causes dose-dependent decrease in the amount of both mRNA and protein expression of CYP2E1 isoform when compared to control group. Compatible with this, CYP2E1-associated aniline hydroxylase activity also decreased significantly as a result of SP treatment in a dose-dependent manner, with nearly 45% inhibition at the highest dose (Table 1). These results collectively suggest that both enzyme inhibition and downregulation seem to contribute to diminished activity of CYP2E1. The inhibition of CYP2E1 by SP may possibly suppress the bioactivation of chemical compounds like benzene, acrylamide, and styrene to which we are exposed everyday in our routine life. Incidentally, it decreases the risk of individuals toward the toxic, carcinogenic, and mutagenic effects of these chemicals. Of note, our findings may also partly explain the results of the previous literature reporting the protective effects of SP on genotoxicity, hepatotoxicity, and nephrotoxicity induced by urethane, 42 carbontetrachloride, 43 and cisplatin, 29 respectively, all of which are activated through CYP2E1.
Besides their toxicological importance, both CYP1A2 and CYP2E1 also participate completely or partially in the metabolism of a number of clinically important drug compounds like acetaminophen, chlorzoxazone, isoniazid, warfarin, verapramil, propranolol, and lidocaine. 44 In this aspect, the decrease in the activity of CYP1A2 and CYP2E1 may alter pharmacokinetic of these drugs as a result of SP, leading to adverse drug reactions like unexpected drug response or toxic side effects. This is especially important for drugs with narrow therapeutic index such as warfarin and lidocaine; thus, special attention must be paid when SP is used concomitantly.
The CYP2C6, corresponding to CYP2C9 in humans, is another enzyme playing a very important role in the metabolism of various drug compounds, especially with narrow therapeutic index like acenocoumoral, warfarin, tolbutamide, and losartan. 44 In the present study, as seen in Figure 2, SP treatment did not result in significant changes in both mRNA and protein level of CYP2C6 isozyme. According to Table 1, a significant decrease was observed in CYP2C6-associated enzyme activity at the lowest dose group. Although, the magnitude of decrease, approximately 14%, seems to be very low, it should be considered for clinical significance, especially when SP is coadministered with drug having narrow therapeutic windows.
The CYP3A1, known as CYP3A4 in human, possesses metabolic activity toward a wide spectrum of clinical drug compounds including antibiotics, calcium channel blockers, antiarrhythmias, sedatives, and immune system modulators. 44 The CYP2B has also received great attention clinically due to its important role in the metabolism of steroid hormones such as testerone and androstedione. 44 In the current study, as seen in Figure 3, the mRNA and protein level for both CYP3A1 and CYP2B1 increased as a result of SP treatment compared to control group. However, as indicated in Table 1, for both isozymes these increases were not represented in the enzymatic activities; slight decreases were observed for the lowest and middle SP doses. A dual effect was observed for both CYP3A1 and CYP2B1 in SP-treated rat liver, which induced mRNA/protein level, but the reduced/unchanged enzymatic activities suggest that enzyme inhibition caused by the constituents of SP may draw the catalytic activities to the values near to that observed in controls. Therefore, future studies to explore inhibitory effect of SP on both CYP3A and CYP2B should be performed using pure or recombinant enzymes.
Conclusion
The results obtained in this study demonstrated for the first time that SP significantly decreases the expression and associated enzyme activities for both CYP1A2 and CYP2E1 in rat liver. The inhibitory effects of SP on carcinogen-activating CYP1A2 and CYP2E1 presented in the current work were found to be well correlated with the protective effects of SP observed toward many procarcinogens in the previous reports. This finding also partly explains its action mechanism as potential chemopreventive agent toward various organ toxicities and tumorogenesis. From pharmacological viewpoint, although the present study shows that SP has little or no modulatory effects on the catalytic activities of pharmacotherapeutically important CYP2B, CYP2C, and CYP3A isoforms, further investigations are needed to confirm these findings. Furthermore, it would still be expected that SP has some potential to change the pharmacokinetics of a group of drug compounds, especially metabolized by CYP1A2 and CYP2E1. This information may be helpful to infer possible drug interactions when SP is used in combinations with clinical drug compounds and to decrease the incidence of adverse drug reactions.
Footnotes
Acknowledgments
The technical assistance of S. Hayretdag and M. Gurkan is acknowledged during animal experiments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Scientific and Technological Research Council of Turkey (Project No. 111T582).
