Abstract
Alkyl glyceryl ethers function mostly as skin-conditioning agents in cosmetic products applied to the skin and hair. The Cosmetic Ingredient Review expert panel reviewed the available animal toxicity and clinical data, including the low dermal absorption, and concluded that the alkyl glyceryl ethers are safe in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
The safety of the following ingredients in cosmetics is reviewed in this report:
Ethylhexylglycerin
Caprylyl Glyceryl Ether
Glyceryl Capryl Ether
Cetyl Glyceryl Ether/Chimyl Alcohol
Batyl Alcohol
Glyceryl Allyl Ether
Glyceryl Lauryl Ether
Isodecyl Glyceryl Ether
Isostearyl Glyceryl Ether
Oleyl Glyceryl Ether
These ingredients function mostly as surfactants or skin-conditioning agents in cosmetic products. Cetyl glyceryl ether and chimyl alcohol are listed in The International Cosmetic Ingredient Dictionary and Handbook 1 as discrete chemicals. However, these 2 ingredients are identical chemicals and represent the same entity. Accordingly, 10 ingredients were actually reviewed although there are 11 ingredients listed in this review. Since chimyl alcohol was the ingredient name first assigned in the International Cosmetic Ingredient Dictionary and Handbook, cetyl glyceryl ether will be referred to as chimyl alcohol throughout the report text.
Chemistry
Definition and Structure
These ingredients are alkyl glyceryl ethers. Structurally, this means that an alkyl or alkenyl chain is terminated with glycerin at one end via an ether linkage. For example, caprylyl glyceryl ether is an 8-carbon, alkyl chain terminated with glycerin (Figure 1). Alkoxylipids, which have a similar structure, are widely distributed in human and animal tissues and occur as neutral alkoxylipids and ionic alkoxylipids. 2,3
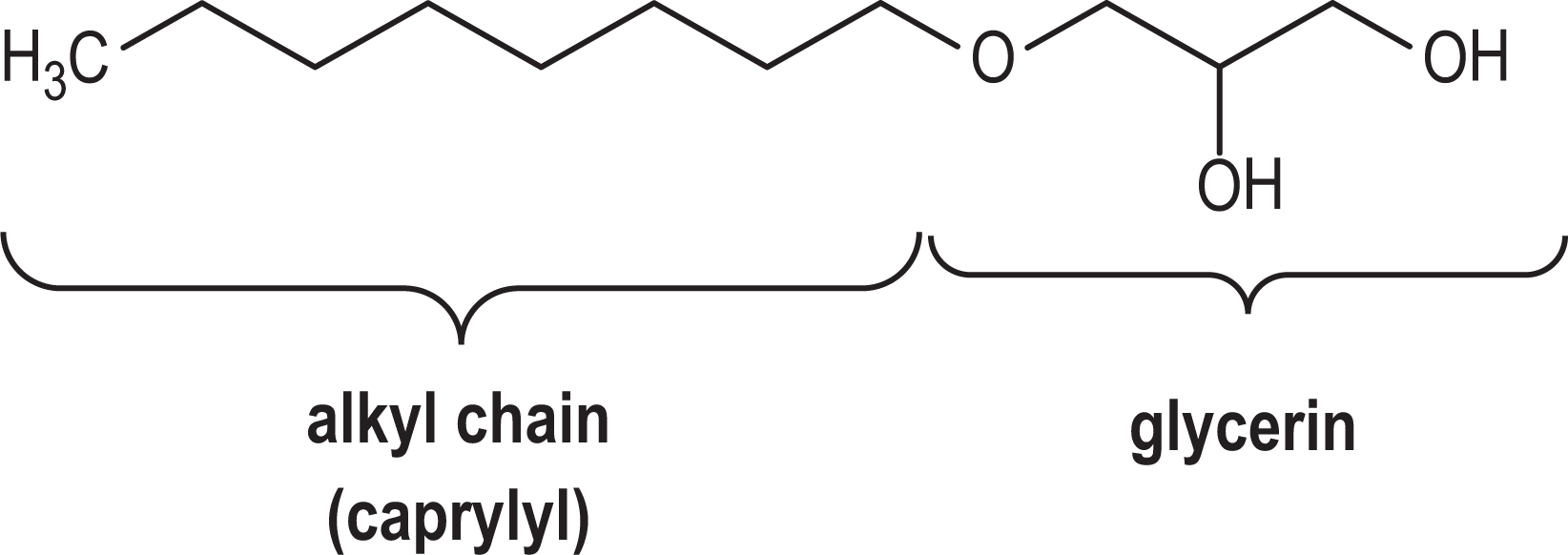
Caprylyl glyceryl ether.
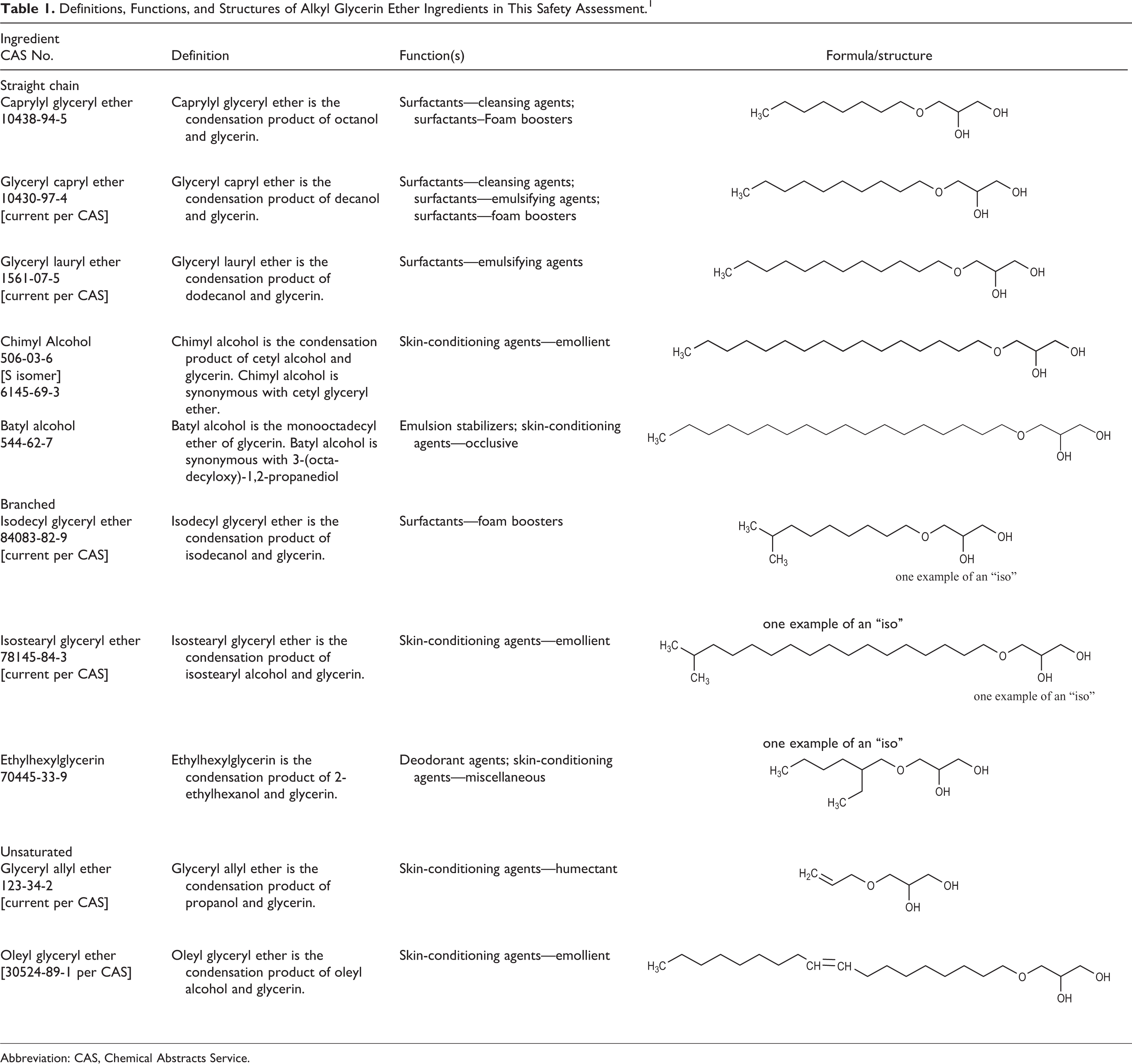
The ingredients reviewed in this safety assessment vary only by alkyl chain length, degree of chain branching, or the degree of unsaturation. Their gross structural characteristics are typical of a surfactant, that is, they contain a hydrophilic head group (ie, glycerin) and a hydrophobic tail (ie, the alkyl chain). The definitions, structures, and functions of these alkyl glyceryl ethers are included in Table 1. 1
Definitions, Functions, and Structures of Alkyl Glycerin Ether Ingredients in This Safety Assessment. 1
Abbreviation: CAS, Chemical Abstracts Service.
Physical and Chemical Properties
The chemical and physical properties of 6 of the 10 alkyl glyceryl ethers are included in Table 2. These ingredients are typically solids at room temperature and have molecular weights <400 g/mol. In general, an increased chain length translates into greater hydrophobicity. Ethylhexylglycerin, specifically, has “limited solubility in water (≈0.1%) and is highly soluble in organic solvents,” and has an estimated octanol/water partition coefficient of Log Pow = 2.4.
Properties of Alkyl Glyceryl Ethers.

Method of Manufacture
The manufacture of alkyl glyceryl ethers can be accomplished by traditional etherification techniques. For example, one method of synthesis of chimyl alcohol involves a reaction of the sodium salt of acetone glycerol (a form of glycerin wherein 2 of the hydroxyl groups are protected and the third is activated as the sodium salt) with hexadecyl iodide in boiling glycol dimethyl ether to yield the acetone compound α-hexadecyl glycerol. Hydrolysis of the protecting group with acetic acid results in the formation of chimyl alcohol. 4
Alternatively, these ingredients can be produced by the reduction of triglycerides. For example, long-chain alkyl glyceryl ethers can be synthesized via the treatment of a triglyceride with lithium aluminum hydride (LAH) + boron trifluoride etherate, followed by LAH hydrogenolysis. 5 The major reaction products are the glyceryl monoether, glyceryl triether, and free alkanol.
Another method for synthesis of ethylhexylglycerin is via the catalytic splitting of ethylhexylglycidyl ether followed by the addition of water. 6 The product can then be purified by vacuum distillation.
Impurities
Sensiva SC50 is one of the trade names under which ethylhexylglycerin is marketed. According to Schülke and Mayr GmbH, Sensiva SC 50 contains >99% of ethylhexylglycerin. 6 Sensiva SC 50 also contains a small amount of α-tocopherol added to stabilize the molecule, and the impurities 2-ethylhexyl-glycidylether and water. 7
Chimyl alcohol is approximately 98% pure and contains approximately 2% of batyl alcohol as an impurity. It has an acid value of approximately 0.001, which means that fatty acids are not present. 8
Use
Cosmetic
These ingredients function mostly as surfactants or skin-conditioning agents in cosmetic products. 1 These and additional functions are included in Table 1. Ethylhexylglycerin (Sensiva SC 50) reportedly inhibits the growth and multiplication of odor-causing bacteria and enhances the efficacy of cosmetic preservatives, such as phenoxyethanol, methylisothiazolinone, or methylparaben. Sensiva SC 50 + phenoxyethanol and Sensiva SC 50 + methylisothiazolinone are marketed as preservatives. 9
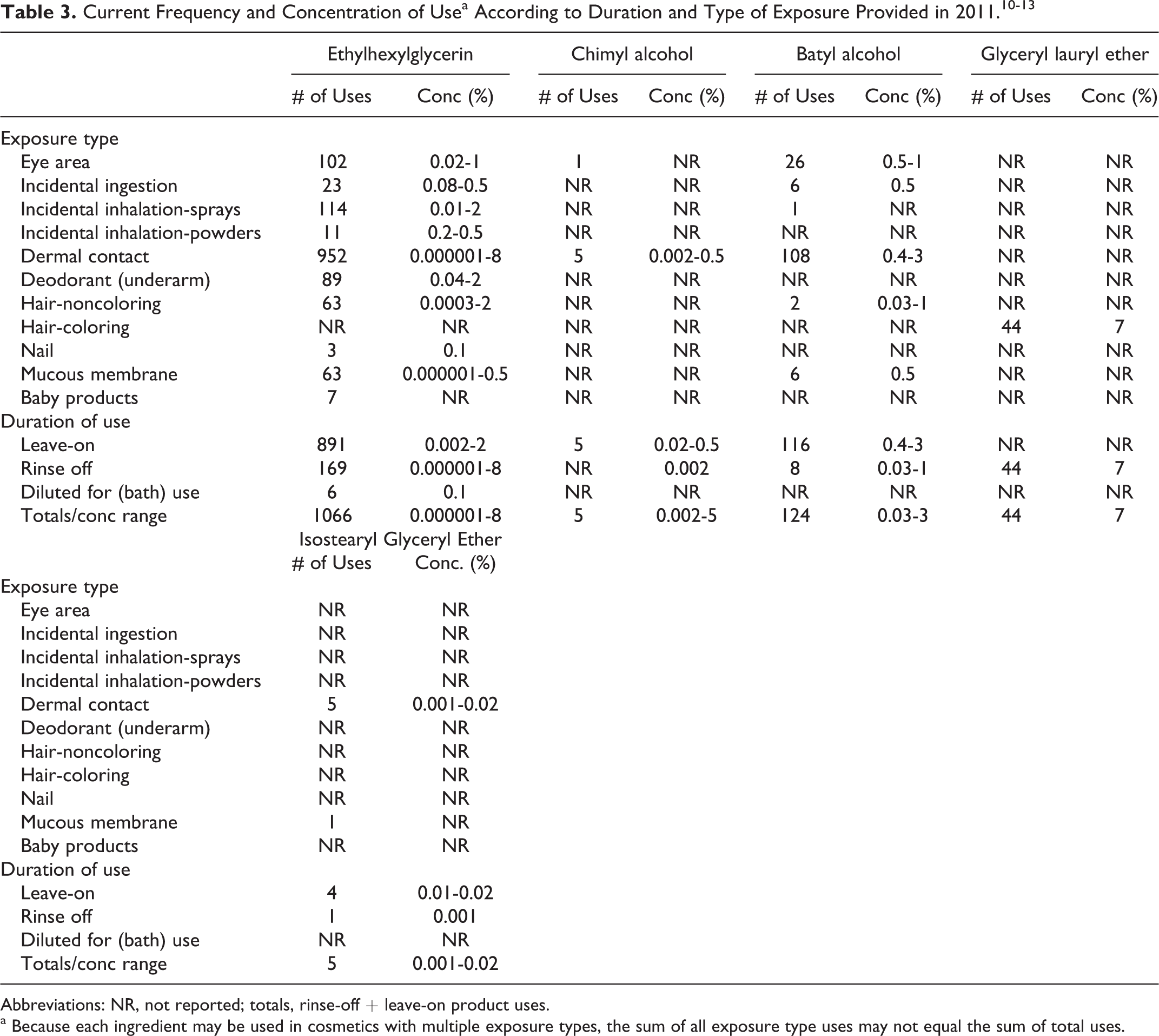
According to reports in the 2011, database of information supplied to the US Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP), the following 4 ingredients were being used in personal care products: ethylhexylglycerin, glyceryl lauryl ether, chimyl alcohol, and isostearyl glyceryl ether. 10 These data are summarized in Table 3. Surveys of ingredient use concentrations conducted by the Personal Care Products Council (also included in Table 3) provided use concentrations for these 5 ingredients: ethylhexylglycerin (up to 8% [maximum in rinse-off products]), chimyl alcohol (up to 0.5% [maximum in leave-on products]), batyl alcohol (up to 3% [maximum in leave-on products]), glyceryl lauryl ether (7% [in rinse-off products]), and isostearyl glyceryl ether (up to 0.02% [maximum in leave-on products]). 11–13 Except for the use concentration data on glyceryl lauryl ether and batyl alcohol (collected in 2011), the data on these ingredients were collected in 2010. Ethylhexylglycerin was reported as being used in 3 baby products in the VCRP, but use concentration data were not reported in the Council survey. 11
Abbreviations: NR, not reported; totals, rinse-off + leave-on product uses.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum of total uses.
No uses of the remaining 5 ingredients reviewed in this safety assessment were reported in the VCRP database or in the Council survey. 10,11
Cosmetic products containing these ingredients may be applied to the skin and hair, or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Ethylhexylglycerin is used in hair sprays and body and hand sprays, and effects on the lungs that may be induced by aerosolized products containing this ingredient are of concern. In practice, 95% to 99% of the aerosols released from cosmetic sprays have aerodynamic equivalent diameters in the 10 to 110 µm range. 14,15 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, able to enter the lungs) to any appreciable amount. 16,17 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 17 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Noncosmetic
The use of ethylhexylglycerin as an emollient in an alcohol-based surgical hand disinfectant (Surgicept) has been reported. 18
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Oral Studies
Chimyl Alcohol
The absorption and metabolism of chimyl alcohol were evaluated using adult male rats. 19 (1- 14 C)-Chimyl alcohol (5 mg) in 0.5 mL olive oil was administered via stomach tube. Feces were collected in 24-hour portions. The amount of chimyl alcohol absorbed was calculated as the difference between the amount of radioactivity administered and the amount recovered from the feces. The amount of [ 14 C]-labeled chimyl alcohol absorbed after feeding was 95%, and the remainder was recovered in the unsaponifiable fraction of fecal lipids. The percentage of total absorbed activity (whole body) that was recovered in the lymph lipids was 45% (1 rat) and 59% (1 rat). The distribution of the activity between the different fractions in the lymph lipids was phospholipid fatty acids (3% and 4%) and glyceride fatty acids (45% and 51%). Also, in the lymph lipids, 45% and 51% of the activity were present as chimyl alcohol, of which free chimyl alcohol accounted for more than half. The remainder was esterified.
A similar study on the absorption and metabolism of chimyl alcohol using rats (strain not stated) was performed. α-(1-C 14 )-Chimyl alcohol was fed, by stomach tube, as free alcohol or as chimyl dioleate (in olive oil). 20 In one series of experiments, intestinal contents were analyzed during the absorption phase, 2 to 4 hours after administration. In another series, the distribution of activity in expired CO2 was determined at 24 hours postadministration. In other experiments, the distribution of activity in lymph lipids was monitored after chimyl dioleate (in olive oil) was fed to rats with a thoracic duct fistula (for lymph collection). Results for dosing with free chimyl alcohol indicated the incorporation of free fatty acids into chimyl alcohol, with the formation of alkoxydiglycerides in lymph lipids. The results of analyses of intestinal contents together with lymph analyses clearly indicated the dynamic situation in the intestine. In the intestinal lumen, the ether linkage was intact during the entire absorption process, but, once absorbed into the intestinal mucosa, more than 50% of the compound was rapidly converted into palmitic acid.
Following administration of 14 C-chimyl dioleate (chimyl alcohol esterified with oleic acid) in olive oil to rats by stomach tube, 8% of the total administered radioactivity were excreted as exhaled 14 CO2 in 24 hours. Results also indicated that fatty acids in both the 1- and 2-position in the alkoxydiglyceride molecule were exchanged during hydrolysis by pancreatic enzymes in vivo. Apparently an equilibrium between the synthesis and hydrolysis of mono- and diesterified chimyl alcohol and free chimyl alcohol was reached. Approximately 75% of radioactivity in the lymph were present as glyceride fatty acids and, approximately 2%, phospholipid fatty acids. Approximately 25% of lymph activity was present as chimyl alcohol, of which more than two-thirds was present as esterified chimyl alcohol. More than half the radioactivity was associated with palmitic acid, indicating a splitting of the ether bond in mucosal cells in the small intestines. It was concluded that chimyl alcohol can be absorbed unchanged, but that it is already extensively metabolized in the mucosal cells, whether it is fed as free chimyl alcohol or as an alkoxydiglyceride. 20
Parenteral Studies
Chimyl Alcohol
The 14 C-chimyl alcohol (dose not stated) was injected intravenously (iv) into rats of an unspecified strain resulted in excretion of 24% of the administered radioactivity as 14 CO2 in 24 hours. 20
Male albino Sprague-Dawley (SD) rats (a total of 40 used) were injected intraperitoneally (ip) with 2- 3 H-α-1- 14 C-α-1- 14 C-2- 3 H-chimyl alcohol (5.60 mg) or β1- 14 C-chimyl alcohol (6.30 mg). 21 After dosing, the animals were killed and the entire liver and small intestine removed. Lipids were then extracted. The purpose of these timed experiments (4-hour or 8-hour postinjection) was to determine if these alcohols would be converted directly to alkyl glyceryl ether phospholipids in the liver and small intestines. Both labeled glyceryl ethers were converted to alkaline stable glyceryl ether phospholipids in the intestine. The β-ether was not incorporated into the glyceryl ether phospholipids as rapidly, when compared to the α-ether. This difference in incorporation of the β-glyceryl ethers might be due to the unsaturated fatty acid attached to the secondary carbon of glycerol.
For both the α- and β-chimyl alcohols, phosphatidyl choline was the major labeled intestinal fraction. For any of the liver lipid extracts, <1% of alkaline stable glyceryl ether phospholipids was detected. In each experiment examining the metabolism of labeled glyceryl ethers, <1% of the radioactivity was detected in each of the following: free fatty acids, long-chain alcohols, cholesterol esters, monoglycerides, and long-chain aldehydes. To substantiate that the glyceryl ether was converted to the glyceryl ether phospholipids, the rats were injected ip with double-labeled α-chimyl alcohol. At 4-hour postinjection, very little tritium was detected in the total liver lipids but was present in substantial quantities in the intestine. This finding was attributed to the degree of cleavage of the linkage in the liver and small intestines. 21
The metabolism of chimyl alcohol was evaluated using 6 female rats of the Carworth Farm Nelson strain. 14 C-Chimyl alcohol was injected iv (10 µCi/100 g body weight) as a fat emulsion. 22 Approximately 28% of the 14 C from chimyl alcohol was eliminated as 14 CO2 within 6-hour postinjection and the urine contained 1% of the 14 C from chimyl alcohol. Approximately 6% to 13% of the radioactivity was accounted for as lipid in the liver, and the remainder was associated with adipose tissue or transferred into a nonlipid metabolic pool. These in vivo results were confirmed in the in vitro liver slice system, in that the 14 C from chimyl alcohol was found in triglycerides, free fatty acids, fatty alcohols, glyceryl ether monoesters, lecithin, and cephalin. Less than 2% of the chimyl alcohol was oxidized to 14 CO2 during the 3-hour incubation period. When bone marrow cells and spleen slices were studied in vitro, the bone marrow cells were less active in metabolizing the labeled chimyl alcohol. Less than 6% of the 14 C from 14 C-chimyl alcohol was incorporated into phospholipids by bone marrow cells or spleen slices during the 3-hour incubation period.
The metabolism of chimyl alcohol and phosphatidylethanolamine (PE) in the brain was studied using 3 groups of male SD rats (18 days old). 23 Use of a control group was not mentioned. Each rat was anesthetized and given an intracerebral injection of 0.375 mL of an emulsion consisting of 20 mg of 6 µc chimyl alcohol- 3 H, 7.3 mg of 25 µc PE- 14 C, and Tween 20 (36 mg/mL). The brains from 3 groups of animals were removed, and ethanolamine phosphoglycerides were isolated and analyzed. It was evident that label from both substrates was incorporated into the brain lipids of the rats. A major portion of the 14 C activity (25%-35%) appeared in the front fraction defined as containing cholesterol, ceramide, cerebroside, choline phosphoglycerides, sphingomyelin, and lysophosphatidylcholine. The 14 C and 3 H activities in the dimethyl acetals derived from alkenyl acylethanolamine phosphoglycerides and in the glyceryl ethers derived from the alkyl acylethanolamine phosphoglycerides were measured. The absence of 14 C in the dimethyl acetals indicated that phosphatidylethanolamine was not transformed into phosphatidal ethanolamine. The increase, with time, of the 3 H content of the glyceryl ethers and dimethyl acetals indicated that chimyl alcohol was a precursor of both types of phospholipids. 23
Batyl Alcohol
Male albino SD rats (a total of 40 used) were injected ip with α-1- 14 C-batyl alcohol (7.24 mg) or β1- 14 C-batyl alcohol (10.37 mg). 21 After dosing, the animals were killed and the entire liver and small intestine removed. Lipids were then extracted. After 8 hours, 24.5% of the injected dose was present in the liver lipids. Approximately 15% of the α-1- 14 C-batyl alcohol liver lipid label was identified as intact free glyceryl ether. All the liver lipid label were identified as esterified fatty acid, with the exception of the remaining free glyceryl ether that had been injected. No significant free fatty acid label was observed in any of the liver lipid fractions. A different labeling pattern was found for the intestinal lipids. At 8-hour postinjection of α-1- 14 C-batyl alcohol, 65% of the intestinal lipid label were present as free glyceryl ether, and 8% of the injected dose were found in the small intestine. Only 7% of the dose were identified as esterified fatty acids. The major portion of the radioactive label in the intestine derived from α-1- 14 C-batyl alcohol was in the phospholipids. After 8 hours, approximately 18% of the total lipid label was associated with the phospholipids. β1- 14 C-batyl alcohol was not incorporated into the glyceryl ether phospholipids as rapidly as the α-ether.
The metabolism of batyl alcohol was evaluated using 6 female rats of the Carworth Farm Nelson strain. 14 C-Batyl alcohol was injected iv (10 µc/100 g body weight) as a fat emulsion. 22 Approximately 13% of the 14 C from batyl alcohol was eliminated as 14 CO2 within 6 hours postinjection, whereas the urine contained 6.5% of the 14 C from batyl alcohol. Approximately 6% to 13% of the radioactivity were accounted for as lipid in the liver, and the remainder was associated with adipose tissue or transferred into a nonlipid metabolic pool. These in vivo results were confirmed in the in vitro liver slice system, in that the 14 C from batyl alcohol was found in triglycerides, free fatty acids, fatty alcohols, glyceryl ether monoesters, lecithin, and cephalin. Less than 0.2% of the batyl alcohol was oxidized to 14 CO2 during the 3-hour incubation period.
Human Study
Chimyl Alcohol
Two adult patients (1 male and 1 female) were used to evaluate the absorption of chimyl alcohol. 24 The female patient with chyluria was maintained on a liquid formula enriched with corn oil containing 25 mg of 14 C-labeled chimyl alcohol. The male patient (with bilateral chylothorax secondary to carcinoma) was maintained on a mixed hospital diet, and, on day 1 the chest was emptied by thoracentesis. This process was followed by feeding with bread containing 14 C-labeled chimyl alcohol (18 mg). A second thoracentesis was performed 48 hours later, prior to the removal of chylous fluid. In the female patient, 95% of administered labeled chimyl alcohol was absorbed and approximately 40% of the dose was recovered in the urine within 12-hour postadministration. This indicated that this patient shunted approximately 40% of her intestinal lymph into the urinary tract. Approximately half of the radioactivity in the lymph was identified as chimyl alcohol, and the remaining 50% had been converted to palmitic acid, found in triglycerides, phospholipids, and free fatty acids. Essentially the same results were reported for the male patient. Study results also indicated that rupture of the ether linkage of chimyl alcohol can occur in the intestinal mucosa of man and that the palmitoyl moiety is readily oxidized to palmitic acid.
Percutaneous Absorption
In Vivo Study
Ethylhexylglycerin
An acute toxicokinetic study on ethylhexylglycerin (Sensiva SC 50) was performed using 2 groups of 5 rabbits of an unspecified strain. 25 The test substance was applied (single application) to the 2 groups at concentrations of 1% and 5% in corn oil, respectively. Each concentration was applied at a dose of 2 g/kg body weight, spread evenly over surgical gauze (25 × 25 mm). The occlusive dressing remained in place for 4 hours. There were no signs of irritation at the application site as late as 60-minute postapplication of either concentration. Changes in body weight were unremarkable. Ethylhexylglycerin was detected in the plasma of 3 of 5 rabbits tested with the 5% concentration. The mean absorption through skin in this group was 0.02% at approximately 2 hours after the beginning of exposure. Plasma concentrations in animals treated with 1% ethylhexylglycerin were below the limit of detection. In all blood samples collected after treatment ended, the concentrations of ethylhexylglycerin were below the limit of detection (1.03 µg/mL).
In Vitro Study
Ethylhexylglycerin
The skin penetration of 14 C-ethylhexylglycerin (Sensiva SC 50) was evaluated using human skin. 25 The experiments were performed after administration of the test substance at 3 concentrations formulated as solutions in ethanol–water, 10:90 v/v, and concentrations of the test substance that were determined using quantitative radiochemical analysis. The lowest test concentration was based on dermal application of the test substance at a concentration of 2% in cream (at 1.4 mg cream/cm2 of skin). Based on concentrations determined in the receptor fluid, mean absorption values for 14 C-ethylhexylglycerin at the 3 concentrations evaluated were as follows: 44.65% (at 31 µg/cm2), 47.15% (at 60 µg/cm2), and 54.94% (at 151 µg/cm 2 ). These 3 values corresponded to mean penetration rates of 2.38, 8.19, and 20.38 µg/cm2/h, respectively. Results indicated that the lag time was essentially the same for all concentration tested. The rate at which 14 C-ethylhexylglycerin moved from the outer skin surface (dissolved in ethanol–water [10:90 v/v]) to the receptor fluid appeared to have been linear.
Skin Penetration Enhancement
The penetration of indomethacin (from propylene glycol solutions) through rat abdominal skin in vitro was substantially increased in the presence of 0.2% or 1% (w/w) of α-monoisostearyl glyceryl ether. 26
Toxicology
Acute Inhalation Toxicity
Ethylhexylglycerin
The acute inhalation toxicity of ethylhexylglycerin (Sensiva SC 50) was evaluated in a study involving groups of 10 (5 males and 5 females/group) SD rats.25,27 The animals were exposed to the aerosolized ethylhexylglycerin (nose only, mean achieved concentrations of 1.89, 2.96, and 4.98 mg/L) for 4 hours according to the Organization for Economic Cooperation and Development (OECD) TG 403 test method, using a continuous flow system. After exposure, the animals were observed for 14 days. The mean diameter of the aerosol particles was between 1.2 and 1.4 µm. Clinical observations included labored and noisy respiration and occasional sneezing (frequent when noted), ataxia, and gasping respiration. The acute inhalation lethal concentration 50 was 3.07 mg/L (low to moderate toxicity), and the following mortalities were reported: 4.98 (4 males and 5 females), 2.96 (3 males and 2 females), and 1.89 mg/L (1 male). Morphological findings for the lungs were described as dark or abnormally reddened appearance, pale patches, enlargement, and an isolated incidence of hemorrhage. Thus, an irritant effect on the respiratory tract was evident. An abnormally dark liver (accentuated lobular pattern) was also reported for the male in the lowest dose group that died. The lung was described as a target organ, based on rapid deaths, severe respiratory changes, and abnormal coloration and enlargement of the lungs.
Acute Oral Toxicity
Ethylhexylglycerin
In an acute toxicity study on undiluted ethylhexylglycerin (Sensiva SC 50), 5 male and 5 female Wistar rats each received a single oral dose (2000 mg/kg) by gavage according to the OECD TG 401 test method.25,27 An lethal dosage (LD50) of >2000 mg/kg (low toxicity, no deaths) was reported, and clinical observations included irregular respiration and hemorrhagic nasal discharge for up to 24-hour postadministration. No test-substance-related morphological findings were reported.
Chimyl Alcohol
The acute oral toxicity of chimyl alcohol was evaluated using 6-week-old SD rats of the Crl: CD (SD) strain (5 males and 5 females). 28 Animals received a single oral dose of chimyl alcohol in a 1 w/v% Tween 80 solution (concentration = 200 mg/mL; dose volume = 10 mL/kg). None of the animals died. Diarrhea was observed after dosing; however, body weight gain was described as normal. Necropsy findings did not reveal any abnormalities. It was concluded that the acute toxicity induced by chimyl alcohol was extremely low (LDLO > 2000 mg/kg).
Acute Dermal Toxicity
Ethylhexylglycerin
The acute dermal toxicity of undiluted ethylhexylglycerin (Sensiva SC 50) was evaluated in a study involving 5 male and 5 female Wistar rats.27,25 The undiluted test substance was applied (2.17 mL on porous gauze/elastic dressing; dose = 2000 mg/kg) to shaved, intact skin according to the OECD TG 402 test method. The application site was defined as a 5 × 10 cm area on the back, and the occlusive dressing remained in place for 24 hours. No abnormal clinical signs or signs of erythema or edema were observed during the study. None of the animals died, and an acute dermal LD50 of >2000 mg/kg (nontoxic) was reported. Necropsy findings were described as normal.
Acute Subcutaneous Toxicity
Batyl Alcohol
Single subcutaneous (sc) doses of batyl alcohol were administered to adult white mice (10-15, age not stated), and deaths were reported at 24-hour postdosing. 29 A mean LD50 of >8.8 mmol/L/kg was reported. Details relating to the test protocol and study results were not included.
Repeated Dose Toxicity
Ethylhexylglycerin
The oral toxicity of ethylhexylglycerin (Sensiva SC 50, in polyethylene glycol 400) at doses of 100, 500, and 1500 mg/kg was evaluated in a 28-day study using groups of 10 rats of an unspecified strain. The test substance was administered at a dose volume of 5 mL/kg.25 There were no treatment-related mortalities. Body weight development was retarded in mid- and high-dose groups, and this finding was accompanied by a nonstatistically significant reduction in food intake when compared to the control (polyethylene glycol 400) group. Both eye and ear functions remained unchanged during the study and increased salivation (dose not stated) was the only clinical symptom observed. Hematological parameters were unaffected by treatment; however, serum analyses indicated changes in aminotransferase (above normal) in high-dose rats. Urinalysis and necropsy findings were unremarkable.
Increased liver and adrenal weights were reported for high-dose rats, especially females. Microscopic findings in the liver were described as follows: slight to moderate hypertrophy (rats from all dose groups), fatty infiltration of hepatocytes (high-dose males), and necrosis of hepatocytes (1 rat each from mid- and high-dose groups). A dose of 100 mg/kg was defined as the no-observed-adverse effect-level (NOAEL), assuming that the slight hepatocyte hypertrophy observed in 5 of 10 rats in this dose group was not toxicologically significant.25
In a 13-week study, ethylhexylglycerin (Sensiva SC 50, in polyethylene glycol [PEG] 400) was administered at oral doses of 0, 50, 200, and 800 mg/kg/d to groups of SD rats (10 males and 10 females/group).25,30 A fourth group (control) was dosed with PEG 400 only. Testing was done in accordance with the OECD TG 407 test method. There was no evidence of treatment-related deaths or statistically significant changes in body weight gain. Furthermore, neurotoxicology testing and ophthalmoscopic examinations revealed no test-substance-related changes. Compared to controls, statistically significant changes in some clinical parameters (eg, statistically significant decrease in lymphocytes) were observed but were considered toxicologically insignificant. Abnormally noisy respiration and an increased incidence of subcutaneous masses were reported for the highest dose group. Noisy respiration was also observed in some animals in the mid-dose group, and the incidence increased with time in mid- and high-dose groups. Treatment-related macroscopic effects were not found in animals killed at the end of dosing or the 4-week recovery period or in animals that died during the study.
In males of all dose groups and high-dose females, a statistically significant increase in absolute and relative-to-body weight liver weights was observed during dosing. Relative liver weights remained increased in males at the end of the recovery period. Increased liver weight was the only toxicologically significant finding that was also observed in the low-dose group. However, this finding was not accompanied by changes in clinical parameters associated with functional impairment of this organ. Increased absolute and relative kidney weights (statistically significant) were observed in females of low- and high-dose groups, but this finding was not observed at the end of the recovery period. Microscopic examination revealed generalized hepatocytic hypertrophy in the high-dose group. When compared to controls, this finding was statistically significant in high-dose male rats. An increased incidence of renal mineralization was observed in high-dose females, but this finding was not apparent at the end of the recovery period. According to the National Industrial Chemicals Notification and Assessment Scheme (NICNAS, Australia) summary of data from this 13-week study, it was not possible to establish an NOAEL because increased liver weight was observed at all doses. The study authors considered 50 mg/kg/d to be an NOAEL, because this dose was associated with increased liver weights without any other effects. 30 In comparison, the NICNAS identified 50 mg/kg/d as an lowest observed adverse effect level (LOAEL), based on the increased liver weights reported in this study. 27
Batyl Alcohol
The hematopoietic activity of batyl alcohol was evaluated using Dunkin-Hartley male guinea pigs (ages not stated). 31 One group of 12 guinea pigs was dosed sc with batyl alcohol (4 mg) in 1 mL of arachis oil on each of 5 consecutive days. The control group (10 guinea pigs) received sc injections of arachis oil. On day 6, the animals were killed and erythrocyte and leukocyte counts were performed on blood obtained from a small ear incision. Changes in bone marrow were also evaluated. Results suggested that batyl alcohol may be an erythropoietic stimulant. The authors suggested that batyl alcohol exerted an influence on the lymphocytic, but not the granulocytic, population of the bone marrow. Observed changes in the bone marrow included increased population of small lymphocytes (P < .05); increased bone marrow reticulocyte count (P < .05); increase in the number of cells intermediate between small lymphocytes and blast-like cells (P ≈ .05); and an increase in the mean numbers of nucleated cells at each stage of the red cell series (.10 > P > .05). Significant reticulocytosis (P < .05) in the blood was also observed. Stimulation of hematopoiesis was also observed in Wistar rats given 15 daily 50 mg doses of a 10% suspension of racemic batyl alcohol in distilled water, 32 and in Wistar rats that received 20 sc injections of peanut oil containing 12.5 mg or 25.0 mg of racemic batyl alcohol 5 days per week over a period of 4 weeks. 33 In the latter study, the erythrocytosis and reticulocytosis were more marked and appeared earlier in rats that received the higher dose.
Ocular Irritation
Ethylhexylglycerin
Undiluted ethylhexylglycerin (Sensiva SC 50, 0.1 mL) was instilled into the left conjunctival sac of each of 3 New Zealand white rabbits (sex not specified), and animals were observed up to day 21 postinstillation.25,27 Right eyes served as untreated controls. Testing was in accordance with the OECD TG 405 test protocol. Conjunctival redness and chemosis were observed in all animals and irritation scores of 2 or 3 predominated. Conjunctival irritation persisted up to day 14, and, in 1 animal, corneal opacity persisted beyond the 21-day observation period. Thus, ethylhexylglycerin (Sensiva SC 50) caused severe damage to the eyes of rabbits. A 5% aqueous solution of the test substance instilled into the eyes of 3 additional rabbits using the same procedure resulted in conjunctival erythema (score = 1) in each animal at 1-hour postinstillation. Conjunctival irritation resolved completely within 24 hours. There were no signs of irritation to the iris or cornea. Thus, the 5% aqueous solution of ethylhexylglycerin (Sensiva SC 50) was mildly irritating to the eyes of rabbits.25,27
Chimyl Alcohol
In the Draize test, the ocular irritation potential of chimyl alcohol was assessed using 6 male Japanese White rabbits of the Kbl: JW, SPF strain. 34 The test substance (0.1 g) was instilled into the right eye of each animal. The eyes of 3 rabbits were rinsed after instillation. In all unrinsed eyes, conjunctival redness and chemosis (score = 1) and discharge (score = 2 or 3) were observed at 1 hour postinstillation. Reactions had cleared by 48-hour postinstillation. Chimyl alcohol was classified as minimally irritating in unrinsed eyes (maximum mean Draize score = 9.3). Ocular reactions were not observed in rinsed eyes. Chimyl alcohol was classified as having minimal ocular irritation potential in this study.
Skin Irritation and Sensitization
Nonhuman Studies
Ethylhexylglycerin
The skin irritation potential of ethylhexylglycerin (Sensiva SC 50) was evaluated using 3 New Zealand White rabbits (sex not specified) according to the OECD TG 404 test protocol. 27 Undiluted test substance (0.5 mL) was applied to intact skin (shaved) and the test site was covered with a semiocclusive dressing secured with elastic tape. Erythema (score = 1) was observed in 2 rabbits, but edema was not observed in any of the animals. Signs of erythema had cleared by day 4 postapplication. Ethylhexylglycerin (Sensiva SC 50) was classified as a mild skin irritant.
In another study, the skin irritation potential of undiluted ethylhexylglycerin (Sensiva SC 50) was evaluated in 3 rabbits (strain not stated) according to the protocol stated in the preceding paragraph.25 Very slight erythema was observed in 2 of 3 rabbits. It was concluded that ethylhexylglycerin could not be classified as a skin irritant.
In the maximization test (OECD TG 406 test method), the skin sensitization potential of ethylhexylglycerin (Sensiva SC 50) was evaluated using 2 groups of 20 guinea pigs (10 males and 10 females/group); one of the 2 groups served as the negative control.25,27 The test substance (0.1 mL) was injected intradermally into the neck region (2 pairs of injection sites) at concentrations of 0.5% in peanut oil and 0.5% in Freund complete adjuvant/saline 1:1, respectively; and at a third pair of sites with Freund’s complete adjuvant/water 1:1. Additionally, an undiluted test substance (applied under occlusive dressing) and a challenge test (50% in peanut oil, under occlusive dressing) were conducted. Sensitization was not observed in any of the animals tested.
The sensitization potential of ethylhexylglycerin (Sensiva SC 50) was evaluated in the local lymph node assay at concentrations up to 50%.25 The test substance did not induce 3-fold increases (EC3 value) in tritiated thymidine (3HTdR) incorporation at any concentration tested, and it was concluded that ethylhexylglycerin was not a sensitizer.
Chimyl Alcohol
The skin irritation potential of chimyl alcohol was evaluated in the Draize test using 3 male Japanese white rabbits of the Kbl: JW, SPF strain (11 weeks old). 35 Chimyl alcohol (0.5 g) moistened with propylene glycol (0.5 mL) was applied to intact and abraded skin sites on each animal. Each application site was covered with an occlusive patch for 24 hours. At 24-hour postapplication, all animals received an erythema score of 1 at intact and abraded skin sites. Reactions had cleared by 72-hour postapplication. Skin irritation was not observed at intact or abraded sites tested with propylene glycol (0.5 mL). Chimyl alcohol was classified as a mild irritant (primary irritation index [PII] = 0.5, intact and abraded skin).
A 14-day cumulative skin irritation study was performed using 3 male Japanese white rabbits of the Kbl: JW, SPF strain. Chimyl alcohol (10 w/v%, suspended in propylene glycol) was applied to the skin at a volume of 0.2 mL, without an occlusive patch. 36 Applications were made once daily for 14 days. Propylene glycol (solvent control) was applied to control sites. Skin irritation was not observed at sites treated with chimyl alcohol or control sites. It was concluded that chimyl alcohol did not have cumulative skin irritation potential.
Human Studies
Ethylhexylglycerin
The skin irritation and sensitization potential of a facial cream containing 0.4975% of ethylhexylglycerin was evaluated in an repeated insult patch test (RIPT) using 600 participants (age not stated). 37 Occlusive patches (0.5” × 0.5”) containing the test substances were applied for a total of 10 induction applications. Following an initial 48-hour challenge application and a second 48-hour challenge 1 week later, there was no evidence of primary irritation, skin fatiguing, or allergic eczematous dermatitis.
A foundation containing 0.4% ethylhexylglycerin was evaluated for skin irritation and sensitization potential in an RIPT involving 108 males and females (18-70 years old). 38 Partially occlusive patches (2 × 2 cm) containing the test substance (∼150 mg) were applied for 24 hours, for a total of 9 induction applications. During the challenge phase (105 participants), test sites were scored at the time of patch removal and 24 hours and 48 hours later. The induction application site and a new test site were challenged. The foundation was neither a skin irritant nor sensitizer.
An RIPT on a liquid eyeliner containing 0.5% ethylhexylglycerin was performed using 115 male and female participants (16-79 years old). 39 A semiocclusive patch test of the substance (0.2 g) applied 3 times per week and a challenge patch test conducted after 3 weeks at a new site adjacent to the original induction site did not result in skin irritation or allergic contact sensitization potential.
The skin irritation and sensitization potential of a makeup preparation containing 0.995% ethylhexylglycerin was studied in 111 male and female participants (18-70 years old) using the same test procedure, except for the application of 100 µL of test material to the partially occlusive patch. 40 The number of participants who participated in the challenge phase was 108. The makeup preparation was neither a skin irritant nor a sensitizer.
An RIPT on a fragranced body lotion containing 0.6965% ethylhexylglycerin was conducted using 108 participants (between 18 and 70 years old). 41 A semiocclusive patch containing the test substance was applied (24 hours) 3 times per week for a total of 9 induction applications. Reactions were scored after patch removal. After a 2-week nontreatment period, a challenge patch was applied to a previously untreated site for 24 hours. Reactions were scored at 24 hours, 48 hours, and 72 hours. The body lotion was not a skin irritant or sensitizer.
Skin Tanning
Chimyl Alcohol
A study was performed with the objective of proposing a new-skin-lightening concept based on a chemical that would control keratinocyte function. 42 Human keratinocytes were cultured for 24 hours in a medium containing chimyl alcohol at concentrations up to 10 μmol/L. The cultures were then exposed to UV-B light (5.0 mJ/cm2). The effect of chimyl alcohol on the skin model was determined by changing the minimum erythema dose; the effect on pigmentation was determined visually. Chimyl alcohol suppressed the production of chemical mediators of melanogenesis from UV-B-irradiated keratinocytes in vitro, possibly via the peroxisome proliferator-activated receptor pathway. Chimyl alcohol also substantially suppressed UV-induced tanning in human skin.
Phototoxicity
Ethylhexylglycerin
The phototoxicity of ethylhexylglycerin (Sensiva SC 50) was evaluated at concentrations ranging from 25% to 100% using albino guinea pigs (number not stated).25 Each solution was applied to the flank, after which sites were irradiated with UV-A light at a dose of 20 J/cm2. The opposite flank (control) was treated with the test substance but not irradiated. The study results indicated no evidence of phototoxic effects.
Photoallergenicity
Ethylhexylglycerin
The photoallergenicity of ethylhexylglycerin (Sensiva SC 50) was evaluated.25 Initially, albino guinea pigs (number not stated) were pretreated with Freund adjuvant. The animals were then treated dermally (5 times in 14 days) with the undiluted test substance and irradiated with UV-A and UV-B light. None of the animals had reactions during the challenge phase that followed, and it was concluded that ethylhexylglycerin was not a photosensitizer.
Case Reports
Ethylhexylglycerin
A 57-year-old female developed perioral and periocular itchy erythematosquamous edema of the eyelids after using a facial cream that contained ethylhexylglycerin. 43 Patch test results for 10% ethylhexylglycerin in petrolatum in this participant were positive (+) on days 2 and 4, but results were negative in 5 control participants.
A 64-year-old female with chronic eczema was patch tested with ingredients provided by a product manufacturer. Reactions were scored on days 3 and 7. 44 A strong positive reaction (++, +) to 5% ethylhexylglycerin in petrolatum was reported. Results of a repeated open application test (ROAT, 7 days) using 5% ethylhexylglycerin were strongly positive on day 2, after only 4 applications. The reaction spread to the entire right arm and to the face. Patch test results were negative for 5% ethylhexylglycerin in 25 consecutive controls.
Recurrent and spreading facial dermatitis was observed in a 68-year-old, nonatopic female after using a cream containing ethylhexylglycerin. 45 Occlusive patches (Finn chambers) were applied for 2 days and reactions were scored on days 3 and 7. Results were positive for 5% ethylhexylglycerin in petrolatum. Patch test results were negative in 25 consecutive controls.
Reproductive and Developmental Toxicity
Ethylhexylglycerin
The effects of ethylhexylglycerin (Sensiva SC 50) on pregnancy and embryo-fetal development were evaluated in a prenatal developmental toxicity study involving groups of female rats of an unspecified strain (no. of animals not stated).25 The test substance was administered by gavage at doses of 50, 200, and 800 mg/kg/d. None of the animals died. Salivation and rales were considered the most relevant signs observed in mid- and high-dose groups. There were no treatment-related effects on terminal body weight, uterine weight, or absolute weight gain. Litter data and the results of macroscopic examinations of females were also unaffected by treatment, and the malformations observed in 3 fetuses at external examination were considered incidental. The results of visceral and skeletal examinations did not reveal any changes between test and control groups that were considered treatment related. Based on these results, the NOAEL was considered to be 800 mg/kg/d, and this was the highest dose tested in the 1-generation developmental toxicity study summarized below.
The developmental toxicity of ethylhexylglycerin (Sensiva SC 50) was evaluated in a 1-generation developmental toxicity study using groups of male and female rats of an unspecified strain.25 The test substance was administered orally at doses of 50, 200, and 800 mg/kg/d. Male rats were dosed daily for 10 weeks prior to pairing, after which dosing continued through the day before animals were killed. Female rats were dosed for 2 weeks prior to mating and during gestation and postpartum periods; dosing ended on postpartum day 20. Treatment-related findings, rales and salivation, occurred only in males of mid- and high-dose groups. In all, 2 male rats were found dead (1 low-dose and 1 mid-dose; cause not stated) and 3 rats (2 high-dose males, 1 high-dose female) were killed for humane reasons. Necropsy findings in animals found dead or killed did not reveal any treatment-related changes in tissues/organs. On day 21 postpartum, there were 17 to 23 females per group with live pups. Twelve females did not become pregnant. Estrous cycles were comparable between groups, but the fertility index for high-dose animals was lower when compared to controls. Data on prebirth loss and gestation length indicated no treatment-related effects on implantation. The no-observed-effect-level (NOEL) was 50 mg/kg/d for both sexes.
Genotoxicity
Ethylhexylglycerin
The genotoxicity of ethylhexylglycerin (Sensiva SC 50) was evaluated in the Ames test (OECD TG 471 test method) using Salmonella typhimurium strains TA 98, TA 100, TA 1535, and TA 1537 with and without metabolic activation.25,27 The test substance was evaluated at concentrations up to 1.5 µg/plate. Toxicity was observed at the highest test concentration. Ethylhexylglycerin was not genotoxic with or without metabolic activation. Normal reversion rates were observed in negative control plates, and positive controls were genotoxic.
In another in vitro test (mouse lymphoma assay), the genotoxicity of ethylhexylglycerin (Sensiva SC 50) was evaluated with and without metabolic activation using mouse lymphoma L5178Y cells.25 Test concentrations were not stated. Negative results were reported for ethylhexylglycerin.
In the in vivo micronucleus assay, the genotoxicity of ethylhexylglycerin (Sensiva SC 50) in mouse bone marrow cells was evaluated.25,27 According the OECD TG 474 test method, 60 mice of the BOR: NMRI strain (30 males and 30 females) received single oral doses of 1000 and 2000 mg/kg by gavage. The examinations for cell nucleus changes in the bone marrow were performed on animals killed at 24 hours and 48 hours. Reduced activity, alterations in tone, and abnormal gait were among the findings observed up to 1-hour postdosing; however, none of the animals died. A statistically significant decrease in the numbers of polychromatic erythrocyte was observed in bone marrow cells from females of all dose groups. There were no statistically significant increases in micronucleated polychromatic erythrocytes at any time point, and it was concluded that ethylhexylglycerin was nonclastogenic.
Chimyl Alcohol
The genotoxicity of chimyl alcohol (in DMSO) was evaluated in the Ames preincubation test using the following bacterial strains, with and without metabolic activation: 46 S typhimurium strains TA100, TA1535, TA98, and TA1537, and Escherichia colistrain WP2uvrA. Test concentrations up to 5000 µg/plate were used. Chimyl alcohol was not mutagenic to any of the strains tested. The numbers of revertant colonies in the negative (solvent) control and positive control cultures were within the acceptable ranges stipulated at the testing facility.
In the in vitro chromosomal aberrations assay involving CHL/IU cells (derived from female Chinese hamster lungs), the genotoxicity of chimyl alcohol was evaluated with (concentrations up to 1000 µg/mL) and without (concentrations up to 200 µg/mL) metabolic activation in short-term assays and at concentrations up to 100 µg/mL (with metabolic activation) in the 24-hour assay. 47 In all assays, the incidence of cells with chromosomal aberrations was <5% at each concentration tested, with or without metabolic activation. It was concluded that chimyl alcohol did not have the potential to induce chromosomal aberrations under the conditions used in this study.
Batyl Alcohol
Batyl alcohol was evaluated for genotoxicity in the Ames test (reverse mutation assay) using the following S typhimurium strains: TA97A, TA98, TA100, TA102, and TA1535. 48 The test substance was evaluated with and without metabolic activation at doses up to 25 µg/plate. The following chemicals served as positive controls: benzo[a]pyrene (with metabolic activation) and sodium azide; ICR 191; 2,4,7-trinitro-9-fluorene; and mitomycine C (all without metabolic activation). Batyl alcohol was not genotoxic with or without metabolic activation.
Glyceryl Allyl Ether
The genotoxicity of glyceryl allyl ether was evaluated in the following battery of short-term tests: Ames test (S typhimurium strains TA98, TA100, and TA1538 and E coli strains WP2 and WP2 uvrA; test concentrations up to 4000 µg/plate in plate incorporation assay, with and without metabolic activation), yeast assay for mitotic gene conversion (Saccharomyces cerevisiae strain JD1; 0.1 mL test substance added, with and without metabolic activation), and the rat-liver chromosome damage assay (RL4 rat-liver cell line; tested up to 0.125 of the GI50 [50% growth inhibition]). 49 Glyceryl allyl ether did not induce mutations in any of the bacterial strains tested, gene conversion in yeast (S cerevisiae), or chromosome damage in RL4 cells.
Carcinogenicity
Studies on the carcinogenicity of the alkyl glyceryl ethers reviewed in this safety assessment were not found in the published literature nor were unpublished data provided.
Summary
The safety of the following ingredients in used cosmetics is reviewed in this safety assessment: ethylhexylglycerin, caprylyl glyceryl ether, glyceryl capryl ether, chimyl alcohol, batyl alcohol, glyceryl allyl ether, glyceryl lauryl ether, isodecyl glyceryl ether, isostearyl glyceryl ether, and oleyl glyceryl ether. These ingredients function mostly as surfactants or skin-conditioning agents in cosmetic products. Information supplied to FDA as part of the Voluntary Cosmetic Registration Program, reported in 2011 FDA database, and the results of Personal Care Products industry surveys conducted in 2010 and 2011 indicated the use of the following ingredients in cosmetics: ethylhexylglycerin (0.000001%-8%), chimyl alcohol (0.002%-0.5%), glyceryl lauryl ether (7%), and isostearyl glyceryl ether (0.001%-0.02%). The concentration of use survey for batyl alcohol is still underway.
Sensiva SC 50, one of the trade names under which ethylhexylglycerin is marketed contains > 99% ethylhexylglycerin.6 It also contains a small amount of α-tocopherol and the impurities, 2-ethylhexyl-glycidylether and water. Chimyl alcohol is approximately 98% pure and also contains approximately 2% batyl alcohol as an impurity.
Most of the (1-14C)-chimyl alcohol administered orally to rats was absorbed in 24 hours (95% absorption), and the remainder was found in fecal lipids. Other results (rats) indicated that 8% of 14C-chimyl dioleate (esterified chimyl alcohol) administered orally was excreted as expired 14CO2 in 24 hours. Labeled chimyl alcohol was also detected in the lymph of rats dosed orally and was also present in this form as well as in the form of its palmitic acid metabolite in intestinal mucosal cells. After dosing with free chimyl alcohol (rats, via stomach tube), alkoxydiglycerides resulted from the incorporation of free fatty acids into chimyl alcohol. At 8-hours post-ip dosing of rats with α-1-14C-batyl alcohol, 24.5% of the injected dose was present in the liver lipids and 8% of the injected dose was found in the small intestine; 65% of the intestinal lipid label was present as free glyceryl ether. In another study involving rats, approximately 13% of the 14C from batyl alcohol was eliminated as 14CO2 within 6-hour postinjection and the urine contained 6.5% of the 14C from batyl alcohol.
Following oral administration of 14C-chimyl alcohol to human participants, 95% of the labeled chimyl alcohol were absorbed, and approximately 40% of the dose were recovered in the urine within 12-hour postadministration. Approximately half of the radioactivity in the lymph was identified as chimyl alcohol, and the remaining 50% had been converted to palmitic acid, found in triglycerides, phospholipids, and free fatty acids.
The ip dosing of rats with labeled α- and β-chimyl alcohols resulted in conversion to glyceryl ether phospholipids in the intestine. After iv dosing of rats with 14C-chimyl alcohol, approximately 28% of the 14C were eliminated as 14CO2 within 6 hours, and the urine contained 1%. Approximately 6 to 13% of the radioactivity was accounted for as lipid in the liver, and the remainder was associated with adipose tissue or transferred into a nonlipid metabolic pool. Similarly, 24% of 14C-chimyl alcohol injected iv were excreted as expired 14CO2 in 24 hours. After dosing with free chimyl alcohol (rats, via stomach tube), alkoxydiglycerides resulted from the incorporation of free fatty acids into chimyl alcohol. The incorporation of 3H-chimyl alcohol into brain lipids was apparent after intracerebral injection into rats.
In an acute toxicokinetic study in vivo, the mean absorption of ethylhexylglycerin through the skin of rabbits was 0.02% at approximately 2-hour postapplication, and there were no signs of skin irritation. The amount of ethylhexylglycerin in the plasma was below the detection limit at the end of the 4-hour application period. Over a range of 3 concentrations (44.65%, 47.15%, and 54.94%) applied to human skin in vitro, mean penetration rates of 2.38, 8.19, and 20.38 µg/cm2/h were reported.
In an acute inhalation toxicity study using groups of rats exposed to ethylhexylglycerin (nose-only, mean achieved concentrations of 1.89, 2.96, and 4.98 mg/L), a concentration-related increase in mortality was observed. The lung was described as a target organ, based on rapid deaths, severe respiratory changes, and abnormal coloration and enlargement of the lungs.
No mortalities or exposure-related toxicological findings were observed in rats dosed orally with undiluted ethylhexylglycerin or chimyl alcohol. Administration of batyl alcohol to mice sc yielded an LD50 of > 8.8 mmol/L/kg.
No mortalities or signs of skin irritation or abnormal necropsy findings were observed after undiluted ethylhexylglycerin was applied to the skin of rats. Necropsy findings were unremarkable and there were no treatment-related mortalities in rats dosed orally with ethylhexylglycerin at doses up to 1500 mg/kg for 28 days. Increased liver and adrenal weights were observed in the highest dose group, and microscopic findings included slight to moderate liver hypertrophy in rats of all 3 dose groups. The 100 mg/kg dose was defined as the NOAEL. Ethylhexylglycerin administered orally to rats, at doses up to 800 mg/kg/d, in a 13-week study did not result in any treatment-related deaths, macroscopic observations, or neurotoxicity. A statistically significant increase in absolute and relative-to-body weight liver weights was observed in males of all dose groups and females of the highest dose group. Generalized hepatocytic hypertrophy was observed at microscopic examination in the highest dose group, a finding that was statistically significant in males. Two summaries of this 13-week showed that a dose of 50 mg/kg/d (lowest dose) was the LOAEL in one summary and the NOAEL in the other summary. Batyl alcohol stimulated hematopoiesis (both red and white blood cells) in repeated dose studies involving rats and guinea pigs. In these studies, batyl alcohol (4 mg in arachis oil) was injected sc into guinea pigs for 5 days. Rats received 15 daily 50 mg doses of a 10% suspension of batyl alcohol in distilled water or 20 sc injections of 12.5 mg or 25.0 mg of batyl alcohol in peanut oil over a period of 4 weeks (5 days per week).
Undiluted ethylhexylglycerin was severely irritating, but 5% ethylhexylglycerin was mildly irritating, to the eyes of rabbits.
Undiluted ethylhexylglycerin was classified as a mild skin irritant in rabbits in one test and as a nonirritant in another test. Chimyl alcohol was classified as a mild skin irritant in rabbits after a single application but was a nonirritating to the skin of rabbits in a cumulative skin irritation study. Skin sensitization was not observed in guinea pigs tested with 0.5% ethylhexylglycerin during induction and challenged with a higher concentration (50%) in the maximization test. Local lymph node assay results for ethylhexylglycerin at concentrations up to 50% were also negative. Products containing ethylhexylglycerin at concentrations ranging from 0.4% to ∼ 1% were neither skin irritants nor sensitizers in RIPT’s involving human participants. Positive patch test results were reported for dermatitis patients patch tested with ethylhexylglycerin at concentrations up to 10%. However, results were negative for control participants.
Ethylhexylglycerin was not phototoxic or photoallergic in guinea pigs when tested at concentrations up to 100% in the presence of UV-A/UV-B light. Chimyl alcohol suppressed the production of chemical mediators of UV-B-irradiated keratinocytes in vitro and substantially suppressed UV-induced tanning in human skin. Based on these findings, a new concept for skin whitening via controlling keratinocyte function was proposed.
In a preliminary test, the results of visceral and skeletal examinations in litters of female rats given oral doses of ethylhexylglycerin (up to 800 mg/kg/d) were negative. None of the dosed animals died, and the NOAEL for developmental toxicity was considered to be 800 mg/kg/d. In the 1-generation developmental toxicity study (same doses) involving male and female rats, estrous cycles were comparable between groups, but the fertility index for rats of the highest dose group was lower when compared to controls. There were no treatment-related effects on implantation. Necropsy findings in dosed rats found dead or killed did not indicate any treatment-related changes. The NOEL for developmental toxicity in both sexes was 50/mg/kg/d.
Ethylhexylglycerin was nongenotoxic in the Ames test (S typhimurium strains) and in the mouse lymphoma assay in vitro, both with and without metabolic activation. It was also nonclastogenic in the micronucleus assay in vivo. Chimyl alcohol was nongenotoxic (with and without metabolic activation) in the Ames test and in the chromosomal aberrations assay involving Chinese hamster lung cells. Batyl alcohol also was not genotoxic in the Ames test, with and without metabolic activation. Glyceryl allyl ether was nongenotoxic (with and without metabolic activation) in the Ames test (S typhimurium and E coli strains) and yeast assay for mitotic gene conversion (Saccharomyces cerevisiae). Results were also negative in the rat-liver chromosome damage assay (RL4 rat-liver cell line). Studies on the carcinogenicity of the alkyl glyceryl ethers reviewed in this safety assessment were not found in the published literature.
Discussion
The Cosmetic Ingredient Review (CIR) expert panel considered the evidence of minimal percutaneous absorption of ethylhexylglycerin through rabbit skin in vivo and the relevance of such data to the entire group of ingredients. The panel determined that the similar chemical structures of these alkyl glyceryl ethers and the similar ways in which they are used in cosmetics suggested that all of these ingredients would have minimal dermal penetration. Furthermore, a review of the available data on toxicity revealed: an absence of genotoxicity in studies using ethylhexylglycerin, chimyl alcohol, batyl alcohol, and glyceryl allyl ether; an absence of reproductive and developmental toxicity in oral studies using ethylhexylglycerin; negative skin irritation/sensitization data in studies using ethylhexylglycerin and chimyl alcohol; and negative phototoxicity/photoallergenicity data in studies using ethylhexylglycerin. Overall, the available toxicity data, coupled with the limited dermal penetration, suggested that these ingredients could be used safely in the present practices of use and concentration.
However, the panel did note the skin penetration enhancement effect of isostearyl glyceryl ether and emphasized that this should be taken into consideration when formulating cosmetic products that contain alkyl glyceryl ethers.
Because ethylhexylglycerin (up to 2%) can be used in cosmetics that may be sprayed, the panel discussed the issue of incidental inhalation exposure. In the absence of sufficient inhalation data, the panel considered that the available data from other routes of exposure that suggested an absence of genotoxicity, reproductive, and developmental toxicity, dermal irritation and sensitization, and phototoxicity/photoallergenicity for this entire group of ingredients. The panel noted that 95% to 99% of the droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, this information suggested that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects.
Conclusion
The CIR expert panel concluded that the following cosmetic ingredients are safe in the present practices of use and concentration described in this safety assessment:
Ethylhexylglycerin
Caprylyl Glyceryl Ether*
Glyceryl Capryl Ether*
Cetyl Glyceryl Ether/Chimyl Alcohol
Batyl Alcohol
Glyceryl Allyl Ether*
Glyceryl Lauryl Ether
Isodecyl Glyceryl Ether*
Isostearyl Glyceryl Ether
Oleyl Glyceryl Ether*
Were ingredients in this group not in current use to be in the future (indicated by *), the expectation is that they would be used in product categories and at concentrations comparable to others in the group.
Footnotes
Author's Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.