Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel assessed the safety of 19 alkyl glucosides as used in cosmetics and concluded that these ingredients are safe in the present practices of use and concentration when formulated to be nonirritating. Most of these ingredients function as surfactants in cosmetics, but some have additional functions as skin-conditioning agents, hair-conditioning agents, or emulsion stabilizers. The Panel reviewed the available animal and clinical data on these ingredients. Since glucoside hydrolases in human skin are likely to break down these ingredients to release their respective fatty acids and glucose, the Panel also reviewed CIR reports on the safety of fatty alcohols and were able to extrapolate data from those previous reports to support safety.
Keywords
Introduction
This assessment reviews data relevant to the safety of decyl glucoside and 18 other alkyl glucosides as used in cosmetic formulations. Most of these ingredients function in cosmetics as surfactants. Other reported functions of some of these ingredients are skin-conditioning agent, hair-conditioning agent, or emulsion stabilizer. Hexadecyl
The ingredients included in this review are obtained by the condensation of an alcohol with a cyclic form of glucose (
Decyl glucoside
Arachidyl glucoside
Butyl glucoside
C10-16 Alkyl glucoside
C12-18 Alkyl glucoside
C12-20 Alkyl glucoside
C20-22 Alkyl glucoside
Caprylyl/capryl glucoside
Caprylyl glucoside
Cetearyl glucoside
Coco-glucoside
Ethyl glucoside
Hexadecyl
Isostearyl glucoside
Lauryl glucoside
Myristyl glucoside
Octadecyl
Octyldodecyl glucoside
Undecyl glucoside
Although the names of these ingredients imply that they are monoglucosides, these ingredients are not limited to monoglucosides but may involve products that are the result of a number of condensed glucose repeat units.
Glucoside hydrolases in human skin are likely to break down these chemicals to release their respective fatty alcohols and glucose. Therefore, summary information on the appropriate fatty alcohols that have been previously reviewed by the Cosmetic Ingredient Review (CIR) is presentedin Table 1.
Summaries of Information on Fatty Alcohols From Previous CIR Reports.a
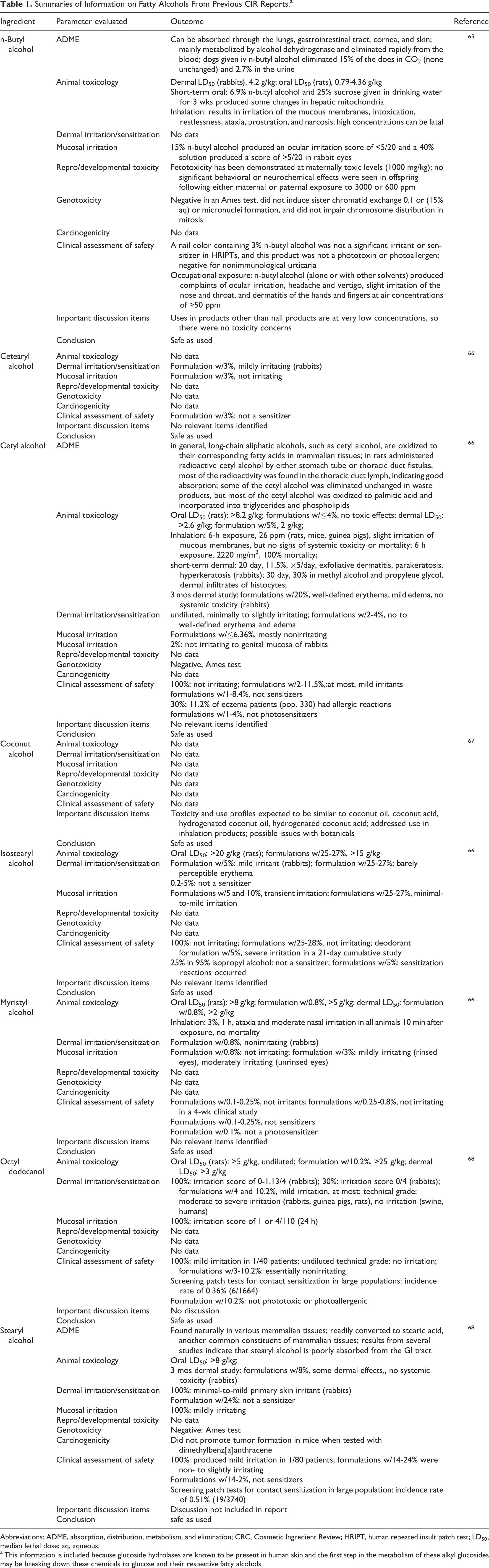
Abbreviations: ADME, absorption, distribution, metabolism, and elimination; CRC, Cosmetic Ingredient Review; HRIPT, human repeated insult patch test; LD50, median lethal dose; aq, aqueous.
a This information is included because glucoside hydrolases are known to be present in human skin and the first step in the metabolism of these alkyl glucosides may be breaking down these chemicals to glucose and their respective fatty alcohols.
Chemistry
Definition and Structure
This group of ingredients consists of anomerically, alkyl-substituted
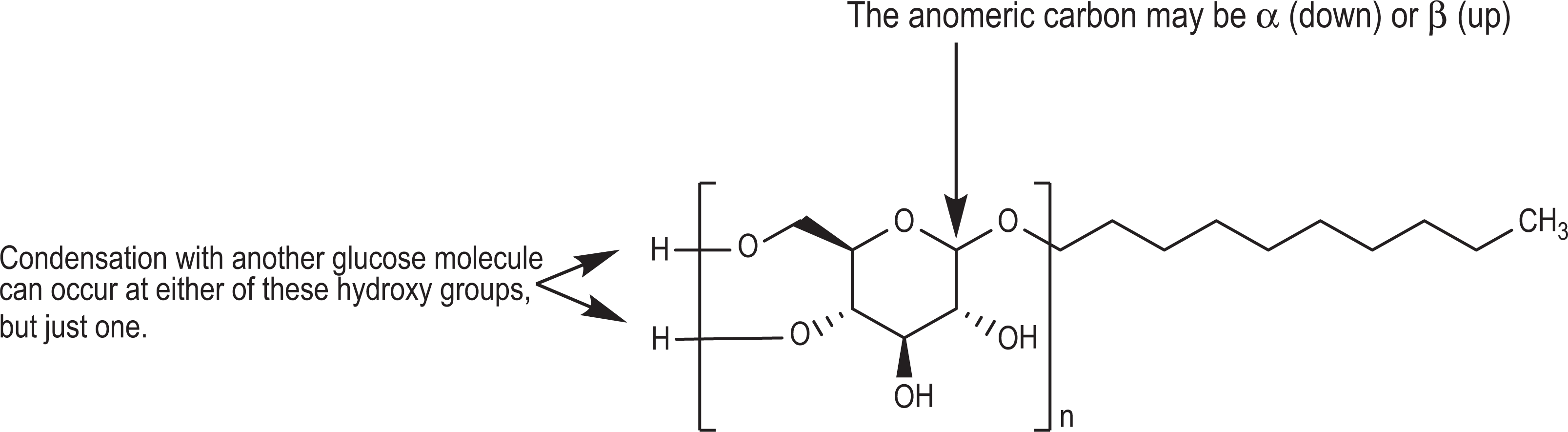
General decyl glucoside structure showing a 10-carbon, alkyl chain-substituted glycopyranoside, wherein n can be 1 (for a monoglucoside) or more (for di-, tri-, oligo-, and polyglucosides).
The general decyl glucoside structure is shown in Figure 1. Decyl glucoside may be monomeric or polymeric, but the International Nomenclature Cosmetic Ingredient name will still be decyl glucoside. “Poly” will be generically used throughout the rest of this report to refer to di-, tri-, oligo-, polyglucosides and mixtures thereof. Decyl glucoside, for example, may consist of one or more of the polyglucosides (in this case isomaltopyranosides) shown in Figure 2.
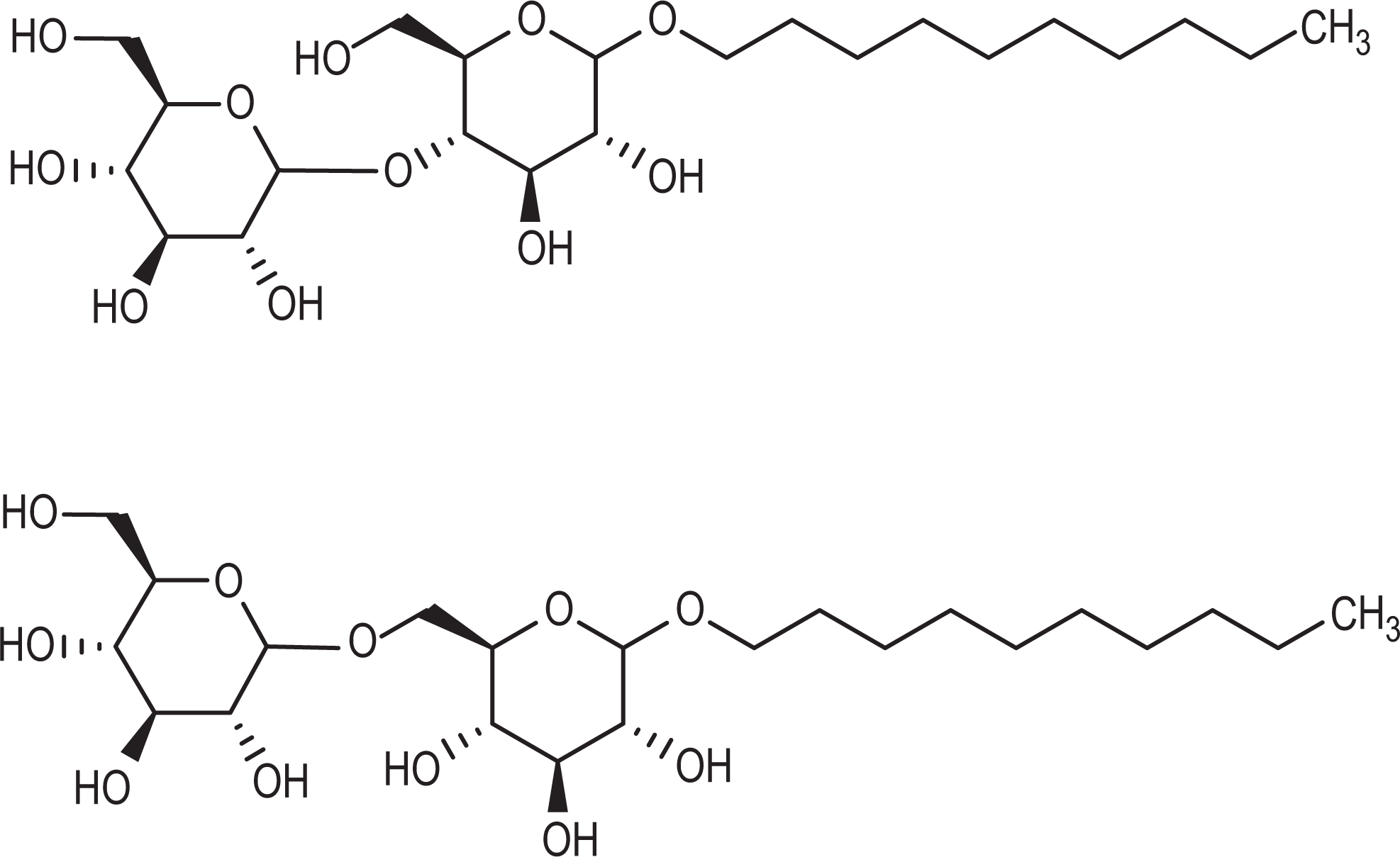
Examples of decyl glucoside forms.
A decyl glucoside with a degree of polymerization of 1.6, for example, would then be a mixture comprising decyl glucopyranoside and one of the decyl maltopyranosides (with a slightly higher percentage of the maltose derivative) shown in Figure 2. Because many of these fatty alcohols are supplied from natural feed stocks, the designated length may be the average (eg, median) length (eg, decyl glucoside may actually be a mixture of C6, C8, C10, C12, C14, and C16 chain lengths, each anomerically attached to a glucopyranose). 1
The definitions and structures of the ingredients included in this review are provided in Table 2.
Definitions, Functions, and Structures of the Alkyl Glucosides in this Safety Assessment.
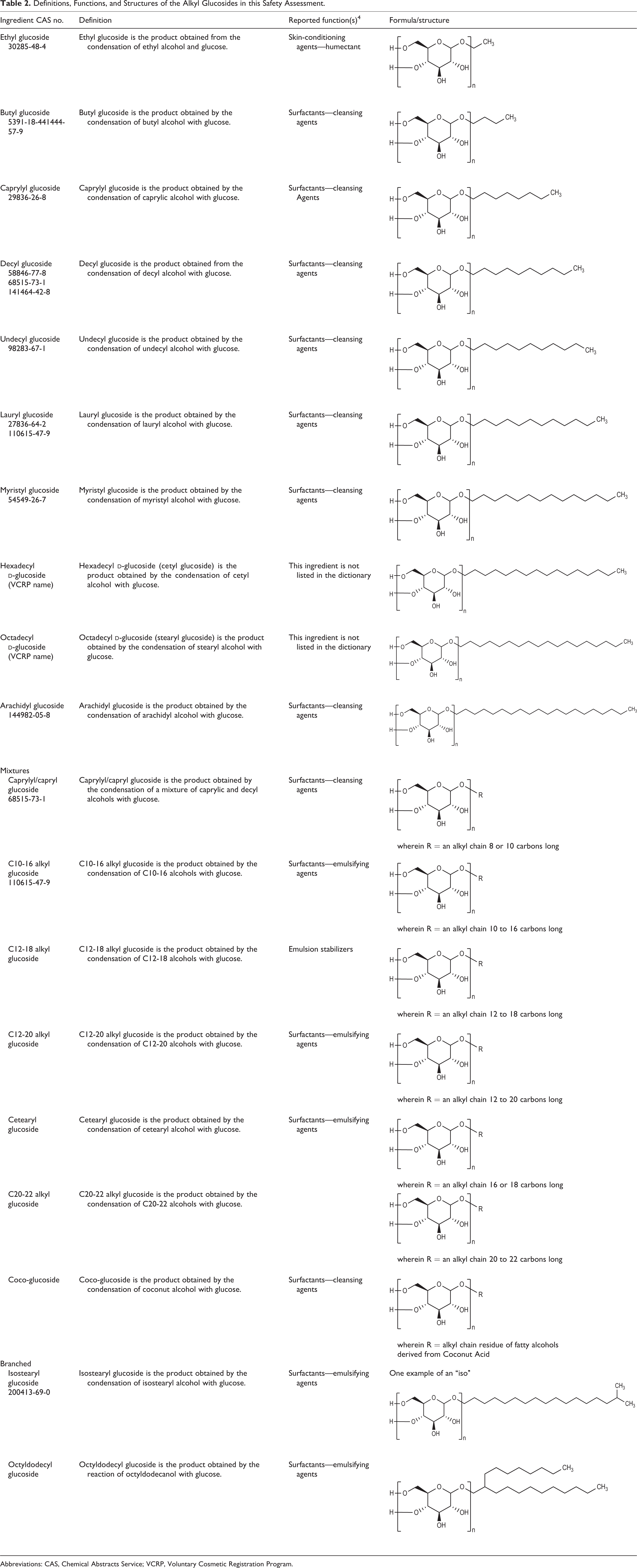
Abbreviations: CAS, Chemical Abstracts Service; VCRP, Voluntary Cosmetic Registration Program.
Physical and Chemical Properties and Impurities
These compounds are often available as viscous liquids, but they can also be solids. The available impurity and physical and chemical property information is presented in Table 3.
Chemical and Physical Properties.

Abbreviations: APG, alkyl polyglucoside; aq, aqueous.
Method of Manufacture
The first report of the synthesis of alkyl glucosides in 1893 involved reacting glucose with anhydrous ethanol under acidic conditions to produce ethyl glucoside. 2 Alcoholysis of glucose and polysaccharides under acidic conditions is still the method of choice. It is considered to be a “green” process that can involve the use of natural and renewable sources (eg, the alcohols can be obtained from coconut oil or palm oil and the glucose or polysaccharide can be obtained from corn, potato, or wheat starch). 3 Of note, the reaction conditions that produce an ether linkage between a fatty alcohol and the anomeric hydroxyl group of glucose are known to cause condensation of one molecule of glucose with another molecule of glucose, thereby producing alkyl polyglucosides (APGs) even when an alkyl monoglucoside may be the intended product.
Use
Cosmetic
The alkyl glucosides named in this safety assessment are reported to function primarily as surfactants. 4 A few are reported to function as skin-conditioning agents, hair-conditioning agents, or emulsion stabilizers.
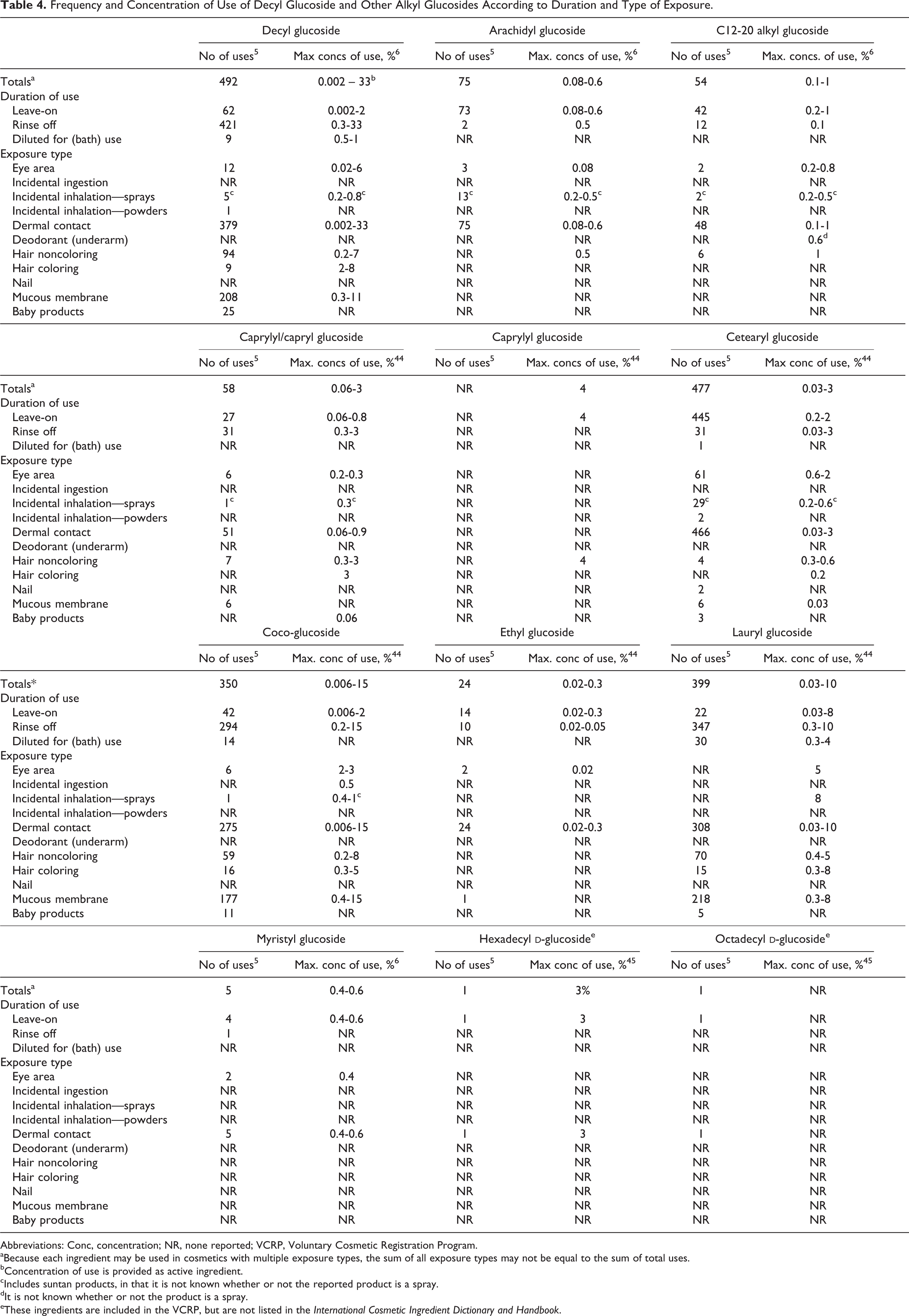
The VCRP data obtained in 2011 for this ingredient group indicate that decyl glucoside has the highest frequency of uses reported, 492; the majority of these uses, 421, are in rinse-off formulations.
5
Cetearyl glucoside, lauryl glucoside, and coco-glucoside have 477, 399, and 350 reported uses, respectively. Cetearyl glucoside is reported mostly to be used in leave-on products. The remaining ingredients that are reported to be used have ≤75 uses. Based on the data from a survey conducted by the Personal Care Products Council (Council), lauryl glucoside has the highest leave-on concentration of use at 8%; this leave-on use is in a hair color spray; it is also reported to have the highest leave-on concentration of use that involves dermal contact, and that concentration is 5%. Decyl glucoside has the highest rinse-off concentration of use at 33%.
6
Two ingredients, hexadecyl
Frequency and concentration of use data are provided in Table 4. In some cases, reports of uses were received in the VCRP, but no concentration of use is available. For example, decyl glucoside is reported to be used in 25 baby products, but no use concentration was available. In other cases, no reported uses were received in the VCRP, but a use concentration was provided in the industry survey. For example, caprylyl glucoside was not reported in the VCRP to be used in noncoloring hair products, but the industry survey indicated that it was used in such products at 4%. It should be presumed that caprylyl glucoside is used in at least 1 hair care product.
Frequency and Concentration of Use of Decyl Glucoside and Other Alkyl Glucosides According to Duration and Type of Exposure.
Abbreviations: Conc, concentration; NR, none reported; VCRP, Voluntary Cosmetic Registration Program.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not be equal to the sum of total uses.
bConcentration of use is provided as active ingredient.
cIncludes suntan products, in that it is not known whether or not the reported product is a spray.
dIt is not known whether or not the product is a spray.
eThese ingredients are included in the VCRP, but are not listed in the International Cosmetic Ingredient Dictionary and Handbook.
Six ingredients were not reported to be used in the VCRP database nor were use concentration data provided, 5,6 including butyl glucoside, C10-16 alkyl glucoside, C12-18 alkyl glucoside, C20-22 alkyl glucoside, isostearyl glucoside, octyldodecyl glucoside, and undecyl glucoside. These ingredients would appear to be not in current use in cosmetics.
Products containing alkyl glucosides are reported to be used on baby skin or applied to the eye area, and mucous membranes may be exposed to these products. Coco-glucoside is reported to be used in a product that could be ingested. Some of the alkyl glucosides are used in cosmetic sprays, including hair and body and hand sprays, and could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared to pump sprays.7,8 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, able to enter the lungs) to any appreciable amount.9,10 However, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. Inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions may cause toxic effects depending on their chemical and other properties. There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 10 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
All of the glucosides named in the report, with the exception of C20-22 alkyl glucoside, hexadecyl
Noncosmetic
Caprylyl glucoside and similar alkyl glucosides are effective solubilizers of lipids and proteins below their critical micelle concentrations and are used in various biochemical techniques and membrane research. These ingredients can also be used to reconstitute enzymes or other proteins from crude biological preparations. 12
The use of decyl glucoside as a stabilizer in nanosuspensions for dermal delivery has been investigated; decyl glucoside was effective as a stabilizer with resveratrol 13 and hesperetin ((S)-2,3-dihydro-5,7-dihydroxy-2-(3-hydroxy-4-methoxy-phenyl)-4H-1-benzopyran-4-one) 14 nanosuspensions.
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Dermal
Glucoside hydrolases are known to be present in human skin. Therefore, the first step in the metabolism of these ingredients may be breaking down these chemicals to glucose and their respective fatty alcohols. 15
In Vitro
Caprylyl/Capryl Glucoside
The dermal penetration of caprylyl/capryl glucoside, diluted to 10% in Hanks buffered salt solution, pH 6.5, was evaluated in vitro using human skin. 16 Two skin samples from each of the 3 donors were used in the study (n = 6). The receptor fluid was Krebs-ringer’s bicarbonate buffer without hydroxyethyl piperazineethanesulfonic acid and glucose. After 24 hours, the mean recovery was 0.52% of caprylyl/capryl glucoside from 2 tape strips and 0.30% of caprylyl/capryl glucoside in the further 18 tape strips. The mean amount of caprylyl/capryl glucoside removed from the skin (by washing) ranged from 109% to 145% of the dose applied. The mean absorbed dose of caprylyl/capryl glucoside, as the sum of the amounts found in the viable epidermis, dermis, and receptor medium, was 0.01%.
Oral
Nonhuman Caprylyl Glucoside
Three female NMRI mice were given a single oral dose, by gavage, of 37 MBq/mmol caprylyl [U- 14 C]glucoside in 0.05 mL of a 5% aqueous (aq) solution of phophatidylcholine. 17 The animals were killed 2 hours after dosing. The highest levels of radioactivity were found in the stomach, intestines, liver, and kidneys, with most of the radioactivity (81%-98%) distributed in the aq phase. High levels of radioactivity which were not extractable with chloroform were found in the urine, which, according to the researchers, indicated a high rate of degradation to water-soluble metabolites.
In the stomach, 75% of the radioactivity was associated with unchanged substrate. In the kidneys and intestines, 50% of the total radioactivity was unchanged substrate, while only a trace amount found in the liver was associated with unchanged substrate. Labeled glucose was detected in all 4 of these organs. In the stomach, intestines, and kidneys, 13% to 19% of the radioactivity was contained in the chloroform extract, and most of it was derived from caprylyl [U- 14 C]glucoside. In this extract in the stomach, approximately 2% acylated-labeled substrate was detected.
Ethyl Glucoside
Groups of 6 male Wistar ST rats were fed a diet for 39 days in which sucrose was replaced with 10% or 20% ethyl glucoside, and a control group was fed unaltered (ie, sucrose-containing) feed. 18 A 24-hour urine collection was made once weekly to check the volume. Approximately 60% to 90% of the ethyl glucoside ingested by treated animals was recovered in the urine.
Absorption Enhancement
Caprylyl glucoside has been shown to increase the absorption of poorly absorbed drugs (eg, insulin) both in vitro across human carcinoma monolayers and in vivo through mucosal membranes. In an in vitro study, the enhancement of the permeability of insulin across T84 and Caco-2 cell monolayers by caprylyl glucoside was concentration dependent; permeability of insulin was not significantly enhanced at concentrations of 0.2% and 0.3%, while it was enhanced with 0.4% and 0.5% caprylyl glucoside. 19
In a transmucosal absorption study, the effect of caprylyl glucoside on the nasal, buccal, and rectal absorption of insulin was examined using male Lewis rats. 12 A 5% solution of caprylyl glucoside had an enhancing effect on buccal absorption. The effect of other alkyl glycosides, including decyl and lauryl glucoside, on mucosal penetration was also evaluated. A 5% solution of decyl glucoside also enhanced the buccal absorption of insulin, but 5% lauryl glucoside did not have a significant effect. The researchers stated that there was no consistent relationship between alkyl chain length and penetration enhancement.
Toxicological studies
Single-Dose (Acute) Toxicity
Dermal
Caprylyl/Capryl Glucoside
Groups of 5 male and 5 female New Zealand White (NZW) rabbits were given a single dermal dose of 2 g/kg body weight (bw) caprylyl/capryl glucoside, 50% active ingredient (ai; as C8/C10 APG); the degree of polymerization, n, was 1.6. 20 (Whether occlusion was used was not stated.) Mild to moderate irritant effects, fecal staining, yellowing around the application site, emaciation, nasal discharge, and lacrimation were observed. One animal died of an unrelated infection, and at necropsy, 5 had spotty areas of hemorrhage on the lungs.
C10-16 Alkyl Glucoside
Groups of 5 male and 5 female NZW rabbits were given a single dermal dose of 2 g/kg bw C10-16 alkyl glucoside, 50% ai (as C10-16 APG; n:1.6). 20 (Whether occlusion was used was not stated.) Slight depression, hunched posture, mild to marked erythema, and marked desquamation were observed. None of the animals died during the study.
Oral
Caprylyl Glucoside
Female NMRI mice were given a single oral dose of 0.040 g (2 g/kg bw) caprylyl glucoside as a suspension in 0.2 mL of a 5% aq solution of phosphatidylcholine. 17 No toxic effects were observed during a 2-week postdose observation period. Growth and behavior were not affected.
Caprylyl/Capryl Glucoside
Groups of 5 male and 5 female Sprague-Dawley rats were given a single oral dose of 5 g/kg bw caprylyl/capryl glucoside (as C8/10 APG; n:1.6, 50% ai). 20 None of the animals died during the study.
C10-16 Alkyl Glucoside
Groups of 5 male and 5 female Sprague-Dawley rats were given a single oral dose of 5 g/kg bw C10-16 alkyl glucoside (as C10/16 APG; n:1.6, 50% ai). 20 None of the animals died during the study. Additionally, no mortality was observed upon dosing of 2 male and 2 female Wistar rats with a single oral dose of 2 g/kg bw C12/14 APG, n: 1.6 and 60% ai.
Repeated Dose Toxicity
Dermal
Caprylyl/Capryl Glucoside
Groups of 6 male and 6 female NZW rabbits were dosed dermally with 0, 0.9, and 1.8 g ai/kg (0, 22.5, and 45 w/v%, respectively) caprylyl/capryl glucoside (60% active) in distilled water (4 mL/kg).21,22 Ten 6-hour occlusive applications were made over a 2-week period.
Treatment-related signs of toxicity, such as ataxia, lethargy, and emaciation, were observed in both the test groups. One female of the 1.8 g ai/kg group died after 10 doses, and the death was considered test article related. Slight irritation was observed 1 day after the initial dose, and severe dermal irritation was observed in males and females of both the test groups by days 5 to 6 of the study. Body weights of treated male and female rabbits were significantly less than those of the controls, and mean bw loss was observed for both the groups. Compared to the controls, absolute testes weights were significantly lower in treated males of both the dose groups. No other compound-related changes in organ weights were observed. Small testes were observed in 3 of the 6 treated males of each group; the researchers stated that occurrence of this lesion was rare, and while the occurrence was not statistically significantly different from controls, it was considered biologically significant. Microscopic examination of selected male tissues demonstrated very slight to marked testicular degeneration in all rabbits in the 0.9 g ai/kg group and slight to marked testicular degeneration in 4 rabbits of the 1.8 g ai/kg group. Very slight to moderate atrophy of the prostate and “accessory sex glands” was observed in 3 rabbits of each group. The researchers stated that irritation, inflammation, and stress in these animals were major contributing factors to many, if not all, of the toxicologic effects; however, the researchers also stated that it is possible that caprylyl/capryl glucoside produced some of the effects. (Published findings have reported that degenerative changes occur commonly in the testes of normal rabbits, and these changes may be increased during stress. 23 ) A No-Observed-Effect Level (NOEL) was not obtained.
In another 2-week study, 10 occlusive applications of 0.14, 0.41, and 1.25 g ai/kg (60% active) caprylyl/capryl glucoside in distilled water (0, 3.5, 10.4, and 31.1% ai, respectively) were made to intact skin on the backs of 6 male NZW rabbits per group in order to determine the NOEL for testicular toxicity.24,25 Two of the high-dose animals died during the study, and the 4 surviving animals had signs of treatment-related toxicity. No treatment-related mortality occurred in the low- or mid-dose groups. Dermal irritation, which progressed from slight to severe with time, was observed in all test groups, and slight to moderate irritation was observed in the controls. Changes in some hematology and clinical chemistry values were observed but were attributed to stress of the occlusive procedure, irritation, and bw loss. A decrease in the mean absolute testicular weights in animals of the mid- and high-dose groups was considered treatment related. A treatment-related loss in bw was observed in all the test groups, and the mean terminal bws of rabbits of all test groups were decreased compared to controls. Relatively small testes were observed in 1, 2, 4, and all 6 males of the control, low-, mid-, and high-dose groups, respectively. Treatment-related microscopic changes were observed in the testes, epididymides, prostate, and vesicular glands of the mid- and high-dose group animals; some of the lesions included an increased incidence and severity of diffuse bilateral testicular atrophy with necrotic spermatocytes and atrophy of the prostate and vesicular glands. The NOEL for the microscopic effects in the epididymides, prostate, and vesicular gland was 0.14 g ai/kg. One rabbit of the low-dose group, which had the greatest bw loss, had moderate testicular atrophy and a moderate amount of necrotic spermatocytes/spermatids. The researchers stated that the testes and accessory sex organs of the animals in the control and treatment groups were relatively immature due to age (12 weeks) and low bws, and the immature nature of these organs complicated the evaluation. Changes in the testes and accessory sex glands were attributed to the stress. An NOEL for the study was not established.
In yet another 2-week study, using nonocclusive applications, 2 mL of 0, 0.06, 0.18, or 0.54 g ai/kg caprylyl/capryl glucoside (60% active) in distilled water (corresponding to concentrations of 0, 3, 9, and 27% ai, respectively) were applied to the intact skin of the backs of 6 male rabbits/group. 26 These doses were selected following a 2-week pilot study, in which unoccluded exposure to 0.12, 0.23, and 0.45 ai g/kg caprylyl/capryl glucoside produced slight to moderate erythema and edema. In the main study, treatment-related signs of toxicity were not observed. Slight dermal irritation was observed in all the groups after the initiation of dosing; the irritation became moderate in the high-dose group after 3 days of dosing. Body weights of rabbits of the high-dose group were slightly, but significantly, decreased compared to controls. Absolute testes weights were slightly, but not significantly, decreased in the high-dose group. No treatment-related effects on hematology or clinical chemistry values or organ weights were reported. Microscopically, epithelial hyperplasia, hyperkeratosis, congestion, and eschar formation were observed in the skin of rabbits of the high-dose group; these changes were not observed in rabbits of the other test groups. No test article-related microscopic changes were observed in the testes or accessory sex glands at any dose. The NOEL for systemic toxicity was 0.18 g ai/kg caprylyl/capryl glucoside.
Oral
Ethyl Glucoside
In a study described earlier under “Toxicokinetics,” in which groups of 6 male Wistar ST rats were fed a diet in which sucrose was replaced with 10% or 20% ethyl glucoside for 39 days, bw gains, but not final bws, were statistically significantly decreased in the 20% group when compared to the control values. 18 All animals survived until study termination. Total water intake was increased with increased ethyl glucoside consumption. In animals fed ethyl glucoside, kidney weights were statistically significantly increased and epididymal and abdominal fatty pad weights were statistically significantly decreased. The renal tubules of 2 and 4 control rats were “not dilated” and “slightly dilated,” respectively, and the renal tubules of all the rats in 10% group were “slightly dilated.” In the group fed 20% ethyl glucoside, the renal tubules of 3 rats were “slightly dilated,” while the other 3 had “moderately dilated” renal tubules. No microscopic damage to renal cells was observed.
Alkyl Polyglucosides
Groups of 10 male and 10 female Sprague-Dawley rats were dosed orally, by gavage, with 0, 0.25, 0.5 and 1 g/kg bw C12/16 APG for 13 weeks. 20 An additional 5 male and 5 female control and high-dose rats were used as a recovery group. No treatment-related changes in bws, organ weights, or biochemistry or hematology parameters were observed. Absolute gonad weights were decreased in all the test groups, but the decrease was not considered treatment related by the researchers because of a lack of a dose response. A dose-dependent, slowly reversible, irritation and ulceration of the forestomach mucosa was observed in animals of the 0.5 and 1 g/kg bw groups. Systemic toxicity was not observed in any group. The no-observed adverse effect level (NOAEL) for systemic toxicity was 1 g/kg bw. The no-observed effect concentration for “local compatibility” was deduced as 2.5% ai.
Reproductive and Developmental Toxicity
Dermal
Caprylyl/Capryl Glucoside
Repeated dose dermal toxicity studies with caprylyl/capryl glucoside (60% active), cited earlier, reported decreased testes weighs, small testes, testicular degeneration, atrophy of the prostate, and microscopic changes in the testes, epididymides, prostate, and vesicular glands.21,22,24–26 These effects were observed in studies using occlusive wraps but not in nonocclusive testing. These effects were attributed to the stress of the study and possible irritation or inflammation.
Oral
Lauryl Glucoside
Groups of 24 gravid female Sprague-Dawley CD rats were dosed orally, by gavage, with 0, 0.1, 0.3, or 1 g/kg bw/day lauryl glucoside (as C10-14 or C10-16, n: 1.4) on days 6 to 15 of gestation. 27 All animals were killed on day 20 of gestation. No maternal toxicity was observed, and no reproductive or developmental effects were indicated. There were also no differences in external, visceral, or skeletal malformations between the groups. The NOAELs for maternal toxicity, embryotoxicity/fetotoxicity, and teratogenicity were all 1 g/kg bw/day.
Lauryl glucoside (as APG C12-C14 fatty alcohol from renewable sources, n: 1.43) was given orally, by gavage, to groups of 10 male and 10 female Sprague-Dawley rats at doses of 0, 0.1, 0.3, or 1 g/kg/day, from 2 weeks prior to mating to 4 days after delivery. 27 No signs of general toxicity were observed in the parental animals. The relative and absolute weights of the testes, epididymides, and seminal vesicles were similar for treated and control animals. There were no test article-related effects on reproductive parameters. The mean litter weights, mean pup weights, sex ratio, and gestation period were similar for all the groups; a slight variation in prebirth loss observed in the high-dose group was not statistically significant. There were no treatment-related effects observed for the neonates.
In Vitro Estrogenicity Assays
Lauryl Glucoside
Lauryl glucoside (as APG C12-C14 fatty alcohol from renewable sources, n: 1.43) was evaluated in the E-Screen assay, in which the induction of cell proliferation in the estrogen-dependent human breast tumor MCF-7 cells is determined, at concentrations of 0.1 to 10 000 nmol/mL. 27 Bisphenol-A and 17-β-estradiol were reference substances, and the medium was the negative control. No effects were reported at concentrations up to 105 higher than the concurrent controls.
The effects of 0.1 to 1000 nmol/mL lauryl glucoside (as APG C12-C14 fatty alcohol from renewable sources, n: 1.43) were determined in the MCF-7 reporter gene assay, in which the induction of luciferase activity in stable transfected MCF-7 cells is determined. 27 No effects were seen with lauryl glucoside alone, and no antiestrogenic or other synergistic effects were observed after incubation with 0.01 to 1000 nmol/mL estradiol–lauryl glucoside (1:1 molar ratio).
Genotoxicity
Alkyl Polyglucosides
The mutagenic potential of APGs (chain length not specified) was determined in 2 Ames tests at concentrations of 8 to 500 µg/L and 11 to 900 µg/plate, with and without metabolic activation. 20 The APGs were not mutagenic. Positive and negative controls gave expected results.
The genotoxic potential of C10/16 APG was evaluated in an assay for chromosomal aberrations using Chinese hamster V79 lung fibroblasts, at concentrations of ≤160 µg/mL with and ≤16 µg/mL without metabolic activation. 20 C10-16 alkyl glucoside was not clastogenic in this assay. Positive and negative controls gave expected results.
Carcinogenicity
Published carcinogenicity studies were not found nor were unpublished data provided.
Irritation and Sensitization
Dermal Irritation and Sensitization
Dermal irritation and sensitization studies are summarized in Table 5. In dermal repeated dose (2-wk) toxicity tests using rabbits, caprylyl/capryl glucoside (60% ai) tested at concentrations ranging from 3.5% to 45% ai in distilled water produced severe irritation over time at all concentrations tested; in a nonocclusive study, slight dermal irritation was seen in similar testing with 3% and 9% (ai) caprylyl/capryl glucoside, and moderate irritation was reported with 27% ai after 3 days of testing. Caprylyl/capryl glucoside, 30% ai, was slightly irritating to rabbit skin in studies for which the details were not provided. The APGs of varying chain length (C8/10 to C12/16; 15%-70% ai) demonstrated a structure–response relationship, with irritation potential decreasing with increasing chain length, and, independent of the degree of polymerization, the irritation was mostly concentration dependent. The primary dermal irritation indices (PDIIs) ranged from 0.0 to 4.6 in rabbits. (A PDII of 2 was considered a positive responder).
Decyl Glucoside and Other Alkyl Glucosides Skin Irritation and Sensitization Studies.a
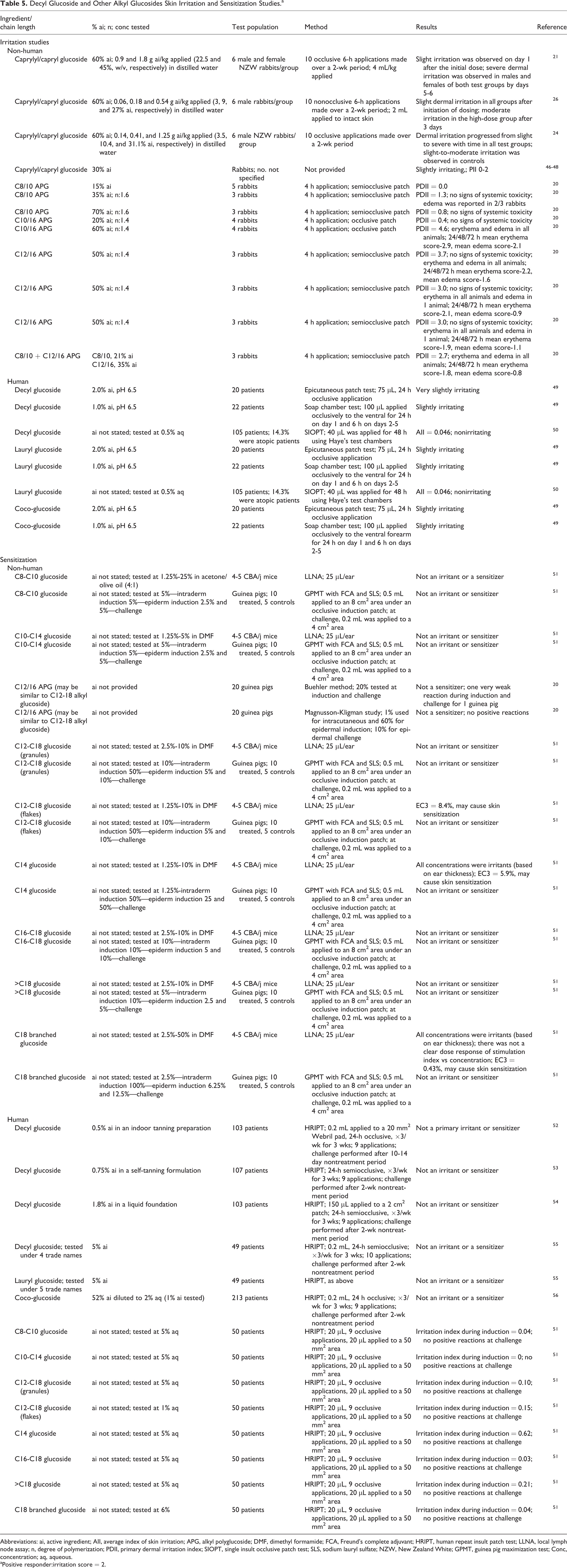
Abbreviations: ai, active ingredient; AII, average index of skin irritation; APG, alkyl polyglucoside; DMF, dimethyl formamide; FCA, Freund’s complete adjuvant; HRIPT, human repeat insult patch test; LLNA, local lymph node assay; n, degree of polymerization; PDII, primary dermal irritation index; SIOPT, single insult occlusive patch test; SLS, sodium lauryl sulfate; NZW, New Zealand White; GPMT, guinea pig maximization test; Conc, concentration; aq, aqueous.
aPositive responder:irritation score = 2.
In clinical studies, the dermal irritation of decyl, lauryl, and coco-glucosides was evaluated in epicutaneous patch (2.0% ai) and soap chamber tests (1.0% ai), and decyl glucoside was evaluated in an single insult occlusive patch test (0.5% ai). At most, these ingredients were slightly irritating.
Glucosides with alkyl chain lengths ranging from C8-C10 to >C18, as well as a C18 branched glucoside, were evaluated in both the guinea pig maximization test (GPMT), at concentrations of 1.25% to 10% for intradermal induction, 5% to 100% for epidermal induction, and 2.5% to 50% for challenge and the local lymph node assay (LLNA) at concentrations of 1.25% to 50%. None of the glucosides tested were irritants or sensitizers in the GPMT, but the LLNA indicated that one C12-C18 glucoside, C14 glucoside, and C18 branched glucoside may cause skin sensitization at concentrations of 8.4%, 5.9%, and 0.43%, respectively. In the LLNAs, irritation was observed with in 1 assay with C14 glucoside at all concentrations (1.25%-10%) and C18 branched glucoside at all concentrations (2.5%-50%). The sensitization potential of C12/16 APG was evaluated in studies in guinea pigs using the Buehler method (test concentrations of 20%) and the Magnusson-Kligman protocol (1, 60, and 10% used for intracutaneous induction, epidermal induction, and epidermal challenge respectively). C12/16 APG was not a sensitizer in the Buehler or Magnusson-Kligman studies.
In clinical testing, the sensitization potential of 0.5%, 0.75%, and 1.8% ai decyl glucoside (in formulation), 5% ai aq decyl and lauryl glucoside, and 1% ai aq coco-glucoside was evaluated in a human repeated insult patch test (HRIPT). These ingredients were not irritating or sensitizing.
Case Studies
Decyl Glucoside
Case studies with reactions to antiseptic, hair, and sunscreen products that contain decyl glucoside are described in the published literature.1,28–32 Subsequent patch testing with decyl glucoside at 0.5% to 10% had positive results in these cases. Patch testing with other glucosides also produced positive results in these patients.
Ocular Irritation
Ocular irritation studies are summarized in Table 6. In alternative system studies for ocular irritation, the irritation potential of 0.6% to 3.0% ai decyl, lauryl, and coco-glucosides, and of C10-16 alkyl glucosides (pH 7, 11.5; concentration not stated), were non to slightly irritating. Caprylyl/capryl glucoside (concentration not stated) was highly irritating in a hen’s egg test-chorioallantoic membrane (HET-CAM) assay. In a HET-CAM study with APGs of varying proportions of alkyl chain length, the ocular irritation potential increased with the increased proportion of shorter chain APGs. In studies using rabbits, neutralized lauryl glucoside produced slight ocular reactions. Caprylyl/capryl glucoside was severely irritating to rabbit eyes when tested undiluted; the irritation threshold value was 10% for 30% ai caprylyl/capryl glucoside and 5% for 60% ai caprylyl/capryl glucoside.
Decyl Glucoside and Other Alkyl Glucosides Ocular Irritation Studies.
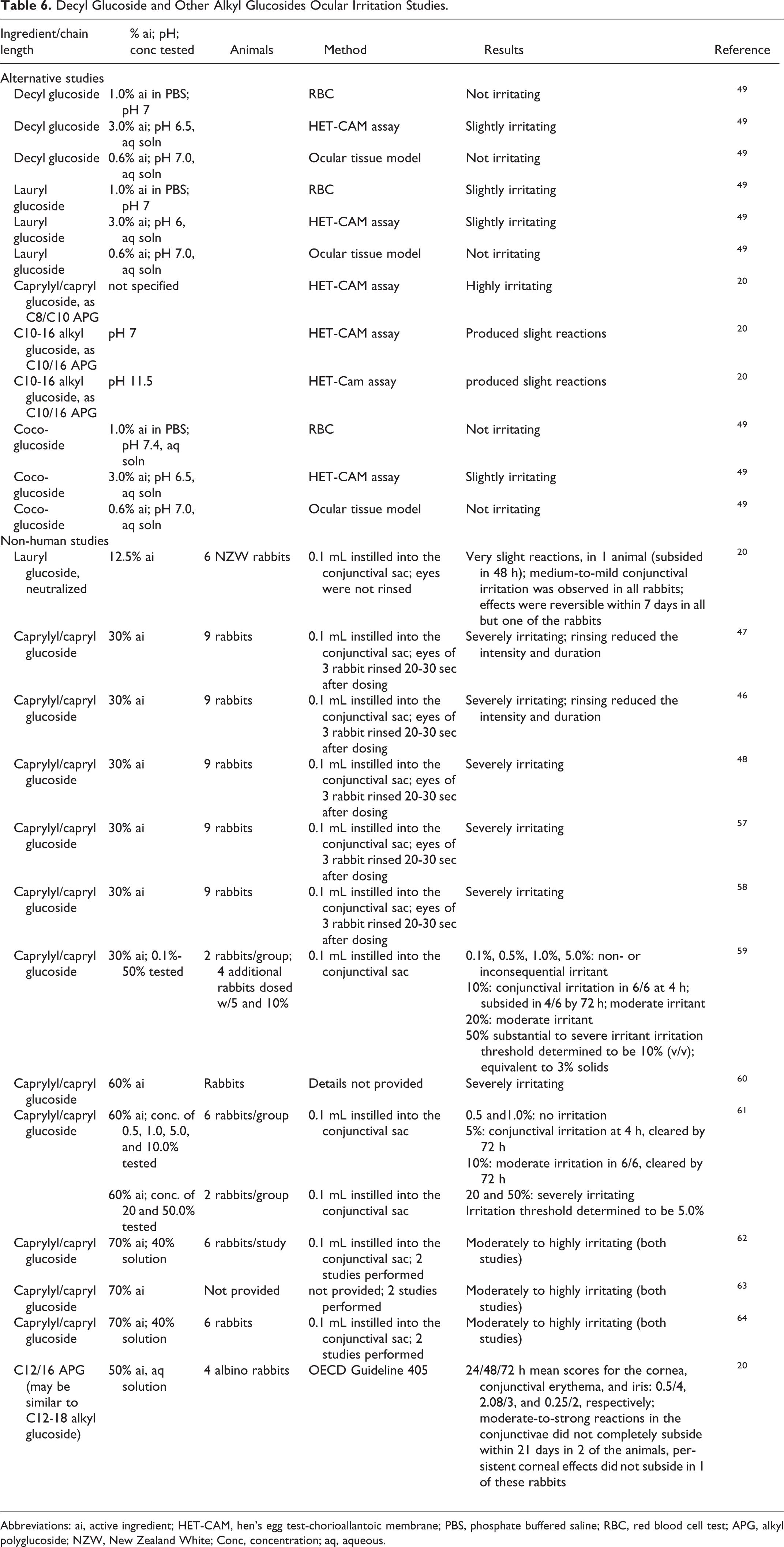
Abbreviations: ai, active ingredient; HET-CAM, hen’s egg test-chorioallantoic membrane; PBS, phosphate buffered saline; RBC, red blood cell test; APG, alkyl polyglucoside; NZW, New Zealand White; Conc, concentration; aq, aqueous.
Summary
The 19 alkyl glucosides reviewed in this safety assessment are ingredients that consist of anomerically alkyl-substituted
Although most of these glucosides are reported to function in cosmetics as surfactants, a few are reported to function as skin-conditioning agents, hair-conditioning agents, or emulsion stabilizers. In 2011, decyl glucoside was reported to be used in 492 cosmetic formulations, 421 of which are rinse-offs. The most frequently used glucoside in leave-on formulations is cetearyl glucoside, with 445 of 477 uses being in leave-on formulations. Lauryl glucoside has the highest leave-on concentration of use at 8%; this leave-on use is in a hair color spray. It also is reported to have the highest leave-on concentration of use that involves dermal contact, and that concentration is 5%. Decyl glucoside has the highest rinse-off concentration of use, at 33%.
In an in vitro dermal absorption study using human skin samples, the mean absorbed dose of 10% caprylyl/capryl glucoside was 0.01%. In female mice dosed by gavage, caprylyl glucoside was the highest in the stomach (primarily unchanged substrate), intestines, liver (only a trace amount unchanged), and kidneys. In a feeding study in rats, most of the ingested ethyl glucoside was recovered in the urine.
In single-dose dermal studies with caprylyl/capryl glucoside and C10-16 alkyl glucoside using rabbits, the median lethal dose (LD50) was greater than the 2 g/kg dose administered. In oral studies with the same test substances, none of the mice dosed with 2 g/kg caprylyl glucoside and none of the rats dosed with 5 g/kg C10-16 alkyl glucoside died during the study.
In 2-week repeated dose dermal studies using rabbits, occlusive applications of caprylyl/capryl glucoside produced testicular effects while nonocclusive application did not. A NOEL for testicular effects could not be established, but the NOEL for microscopic effects in the epididymides, prostate, and vesicular glands was 0.14 g ai/kg. In the nonocclusive study, the NOEL for systemic toxicity was 0.18 g ai/kg caprylyl/capryl glucoside, but it was not clear whether the effects were test article related. Severe dermal irritation was observed in occlusive studies, while slight to moderate irritation was reported in the nonocclusive study.
In oral repeated dose toxicity studies, moderately dilated renal tubules were observed in 3 of the 6 rats fed 20% ethyl glucoside for 39 days but in none of the rats fed 10% ethyl glucoside. Kidney weights were statistically significantly increased in the test animals. In rats dosed orally with 0.25 to 1 g/kg C12/16 APG for 13 weeks, reversible irritation and ulceration of the stomach mucosa was observed, but there was no systemic toxicity reported for any group.
Lauryl glucoside delivered by gavage did not produce adverse reproductive or developmental effects in female Sprague-Dawley rats. Lauryl glucoside, 0.1 to 10 000 nmol, did not have any activity in in vitro estrogenicity assays.
The APGs (chain length not specified) were not mutagenic in Ames tests with or without metabolic activation. C10-16 APG, with and without metabolic activation, was not clastogenic in Chinese hamster V79 lung fibroblasts.
In dermal irritation studies, these ingredients ranged from severely to slightly irritating.
Glucosides with alkyl chain lengths ranging from C8-C10 to >C18, as well as a C18 branched glucoside, were evaluated in both the GPMT and the LLNA assays. None of the glucosides tested were irritants or sensitizers in the GPMT, but the LLNA indicated that 1 C12-C18 glucoside, C14 glucoside, and C18 branched glucoside may cause skin sensitization. C12/16 APG was not a sensitizer in animal repeated insult patch test (RIPT) studies. In clinical HRIPTs testing, decyl glucoside (in formulation), decyl and lauryl glucoside, and coco-glucoside were not irritating or sensitizing.
In alternative system studies for ocular irritation, the irritation potential of 0.6% to 3.0% ai decyl, lauryl, and coco-glucosides, and C10-16 alkyl glucosides were non to slightly irritating in ocular testing. Caprylyl/capryl glucoside was highly irritating in a HET-CAM assay. In a HET-CAM study with APGs of varying proportions of alkyl chain length, the ocular irritation potential increased with the increased proportion of shorter chain APGs. In studies using rabbits, neutralized lauryl glucoside produced slight ocular reactions. Caprylyl/capryl glucoside was severely irritating to rabbit eyes when tested undiluted.
Discussion
Alkyl glucosides, like many other cosmetic ingredients, are provided to formulators at less than 100% active substance. The CIR Expert Panel confirmed that the use concentrations, as given in an industry survey, were as ai.
The Panel was satisfied that sensitization data are adequate. The highest leave-on concentration of use that involves dermal contact is 5% lauryl glucoside. Irritation and sensitization data on lauryl and decyl glucoside at 5% ai, indicating no sensitization reactions, were reported.
The Panel was concerned, however, that the potential exists for dermal irritation with the use of products formulated using decyl glucoside or other alkyl glucosides. Therefore, the Panel specified that products must be formulated to be nonirritating.
In dermal repeated dose studies of caprylyl/capryl glucoside using an occlusive wrap, effects on the testes and accessory sex organs of rabbits were observed. These effects were not reported with a nonocclusive application. In the experience of the Panel, changes in these organs can be observed during stress, and it was the view of the Panel that these effects were due to the stress of the study and were not indications of toxicity of the test ingredient.
The Panel noted there were gaps in the available safety data for many of the alkyl glucosides included in this group. These ingredients have similar chemical structures and are used in similar ways in cosmetics, which suggested that they would have similar structure–activity relationships. The Panel determined, therefore, that it was appropriate to extrapolate the existing data, including the data from previous CIR assessments on fatty alcohols, to address all the alkyl glucosides included in this safety assessment. The Expert Panel recognized that the alkyl glucosides can enhance the penetration of other ingredients through the skin. The Panel cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients for which safety was based on their lack of dermal absorption data or when dermal absorption was a concern.
Because some of the alkyl glucosides can be used in products that may be sprayed, the Panel discussed the issue of incidental inhalation exposure. In the absence of inhalation data, the Panel considered oral toxicity data which suggested little systemic toxicity for alkyl glucosides. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, this information suggested that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects.
Conclusion
The CIR Expert Panel concluded that the 19 alkyl glucosides listed below are safe in the present practices of use and concentration when formulated to be nonirritating.
Decyl glucoside
Arachidyl glucoside
Butyl glucoside*
C10-16 Alkyl glucoside*
C12-18 Alkyl glucoside*
C12-20 Alkyl glucoside
C20-22 Alkyl glucoside*
Caprylyl/capryl glucoside
Caprylyl glucoside
Cetearyl glucoside
Coco-glucoside
Ethyl glucoside
Hexadecyl
Isostearyl glucoside*
Lauryl glucoside
Myristyl glucoside
Octadecyl
Octyldodecyl glucoside*
Undecyl glucoside*
Were ingredients in this group not in current use (as indicated by *) to be used in the future, the expectation is that they would be used at concentrations comparable to others in this group and be formulated to be nonirritating.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
