Abstract
Estimating cancer risk from environmental mixtures containing polycyclic aromatic hydrocarbons (PAHs) is challenging. Ideally, each mixture would undergo toxicity testing to derive a cancer slope factor (CSF) for use in site-specific cancer risk assessments. However, this whole mixture approach is extremely costly in terms of finances, time, and animal usage. Alternatively, if an untested mixture is “sufficiently similar” to a well-characterized mixture with a CSF, the “surrogate” CSF can be used in risk assessments. We propose that similarity between 2 mixtures could be established using an in vitro battery of genotoxic and nongenotoxic tests. An observed association between carcinogenicity and immunosuppression of PAHs suggests that the addition of immune suppression assays may improve this battery. First, using published studies of benzo[a]pyrene (BaP) and other PAHs, we demonstrated a correlation between the derived immune suppression relative potency factors (RPFs) for 9 PAHs and their respective cancer RPFs, confirming observations published previously. Second, we constructed an integrated knowledge map for immune suppression by BaP based on the available mechanistic information. The map illustrates the mechanistic complexities involved in BaP immunosuppression, suggesting that multiple in vitro tests of immune suppression involving different processes, cell types, and tissues will have greater predictive value for immune suppression in vivo than a single test. Based on these observations, research strategies are recommended to validate a battery of in vitro immune suppression tests that, along with tests for genotoxic and other nongenotoxic modes of cancer action, could be used to establish “sufficient similarity” of 2 mixtures for site-specific cancer risk assessments.
Introduction
Polycyclic aromatic hydrocarbons (PAHs) are a class of chemicals containing 2 or more fused aromatic rings made up of only carbon and hydrogen that are formed during the incomplete combustion or pyrolysis of organic materials. 1 The PAHs belong to the wider chemical class of polycyclic aromatic compounds (PACs) that include N-, S-, and O-substituted PAHs and heterocyclic aromatic compounds with N, S, or O. The PAHs exist in complex mixtures in the environment and are of particular concern at sites contaminated with residues from the burning or heating of coal or petroleum products. Complex PAH-containing mixtures from the burning or heating of these source materials (and other organic materials including wood and vegetable- and animal-based foods) contain nonsubstituted and alkylated PAHs, other PACs, and other organic and inorganic chemicals, including metals. The composition of these mixtures is expected to vary with different source materials and heating conditions. Certain PAHs, and other PACs, are thought to contribute to the increased risk of cancer in humans exposed to complex mixtures such as coal tars, tobacco smoke, coke oven emissions, coal gasification residues, and indoor emissions from household combustion of coal. 1 –3
To inform decisions about cleaning up or mitigating Superfund sites contaminated with complex mixtures, including PAH-containing mixtures, the US Environmental Protection Agency (EPA) has frequently used a component-based approach to estimate cancer risk of exposed populations living in close proximity to the contaminated site (see Figure 1, panel B for overview). In this approach, samples of the environmental media of concern (eg, soil, water, or air) are collected and chemically analyzed to determine the concentrations of target chemicals selected based on the known history of the site and preliminary monitoring investigations. These concentrations are used in complex exposure models to estimate oral, inhaled, or dermal lifetime intakes of each chemical (ie, mg/kg body weight/d) by the exposed population. 6 Once route-specific intakes for each chemical are estimated, chemical-specific cancer risks are calculated by multiplying the intake value by EPA-derived route-specific cancer slope factors (CSFs) that are estimated probabilities for cancer (more commonly termed, cancer risk) per unit lifetime intake of a particular chemical (ie, risk per mg/kg/d). The CSFs are derived only for chemical carcinogens with adequate dose–response data from human epidemiology studies or chronic-exposure animal bioassays. 7 For each exposure route, the resultant cancer risks from target chemicals with CSFs are added to arrive at cumulative cancer risk estimates for the exposed population from lifetime exposure to detected chemical carcinogens in the mixture. These cancer risk estimates are used to help make clean-up decisions, and individual or cumulative cancer risk estimates above 10−6 to 10−4 can lead to clean-up actions.
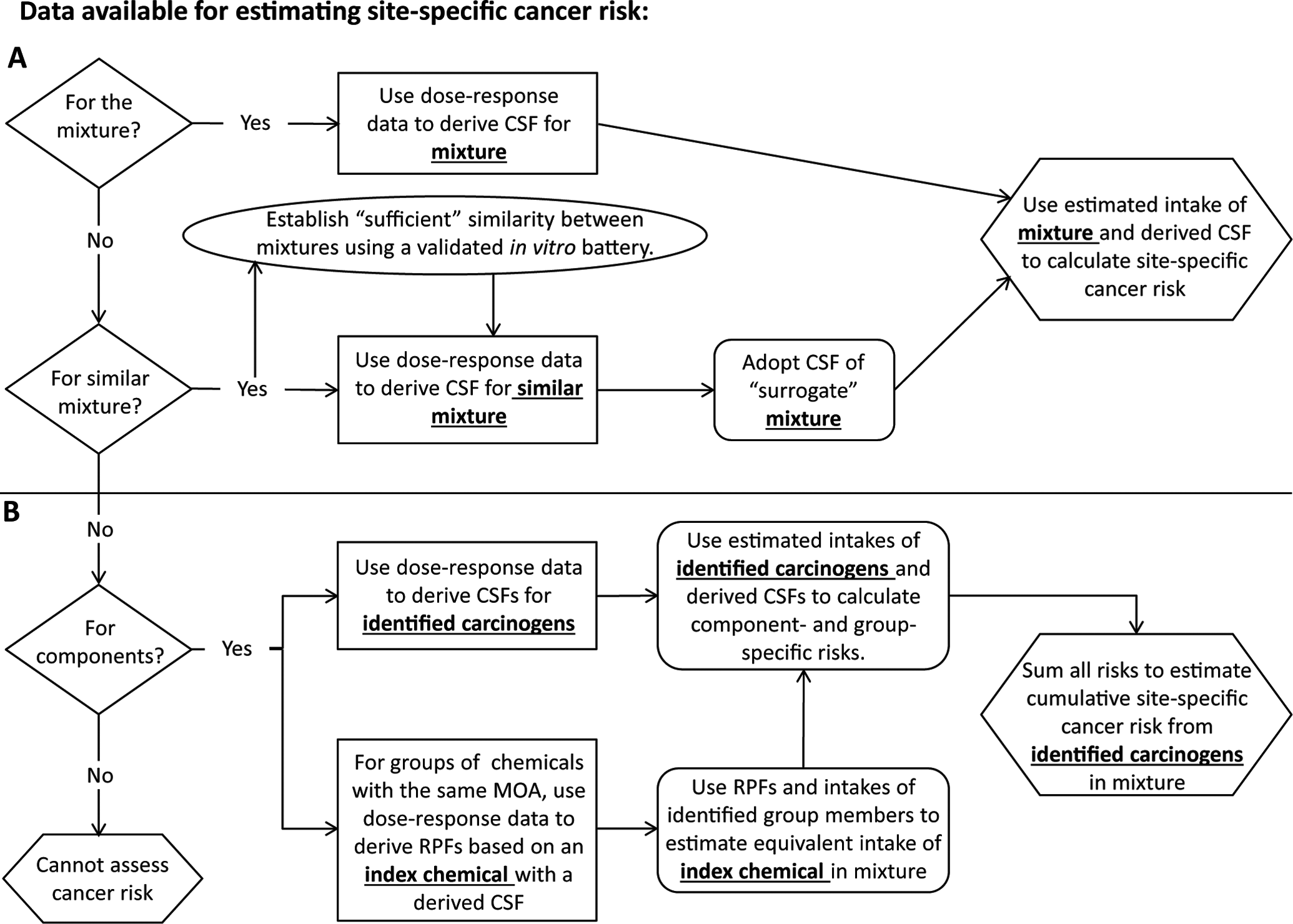
Flowchart for estimating site-specific cancer risk. The flowchart reflects US Environmental Protection Agency (EPA) guidance for estimating human health risks from mixtures encountered at hazardous waste sites4–6, as well as the proposed use of an in vitro test battery to establish similarity between the mixture of concern and a similar mixture with a cancer slope factor (see oval). Panel A illustrates whole mixture approaches resulting in an estimate of cancer risk from the whole mixture. Panel B illustrates component-based approaches resulting in an estimate of cancer risk from carcinogens identified in the mixture of concern. Abbreviations: CSF = cancer slope factor; RPF = relative potency factor; MOA = mode of action.
The US EPA guidelines acknowledge limitations and areas of uncertainty in using component-based approaches for estimating cancer risks of exposed populations, but for practical reasons, the approach is widely used for complex mixtures at hazardous waste sites. 6,4 Major uncertainties associated with using the component-based approach include
The possible presence of undetected and unquantified chemical carcinogens in the mixture (as compositional complexity increases, the possibility of undetected carcinogens being present is expected to increase, and the approach would underestimate cancer risk).
Chemicals detected in the mixture may have inadequate data to determine their potential carcinogenicity and derive a CSF (For example, many nonsubstituted, substituted, and heterocyclic PAHs that have been identified in PAH-containing mixtures have inadequate data to derive CSFs. If they contribute to the carcinogenicity of the mixture, the approach would underestimate cancer risk).
The possible occurrence of nonadditive interactions among chemicals in the mixture. (The approach assumes that components of the mixture act in an additive manner. The occurrence of greater-than-additive interactions [syngergism or potentiation] could lead to underestimation by the approach. Less-than-additive interactions [eg, antagonism] could lead to overestimation).
When PAHs are suspected to occur in a mixture at a site, the US EPA Superfund program decreases the uncertainty described in item 2 above by supplementing the component-based approach with a component-based relative potency factor (RPF) approach for estimating cancer risk from nonsubstituted PAHs (see Figure 1, panel B for overview). 5 Benzo[a]pyrene (BaP) is the only nonsubstituted PAH with adequate data to derive a CSF for environmentally relevant routes of exposure (oral, inhalation, and dermal). However, although numerous other nonsubstituted PAHs have tumor bioassay data that are inadequate for CSF derivation, many have adequate data to derive RPFs based on tumorigenic potency relative to BaP. In the RPF approach, estimated intakes of carcinogenic PAHs with RPFs are added together, after scaling the intakes relative to the potency of BaP, the index chemical. 4 The summed BaP-equivalent intakes are then multiplied by the CSF for BaP to provide an estimate of cancer risk from the nonsubstituted PAHs. The estimated risk from nonsubstituted PAHs is then added to the estimated cancer risks from other chemical carcinogens detected and quantified in the mixture to estimate site-specific cumulative cancer risk estimates. Although the RPF approach for nonsubstituted PAHs allows for inclusion of a greater number of nonsubstituted carcinogenic PAHs in the cancer risk estimate, only 7 of the 16 PAHs routinely assessed at PAH-containing Superfund sites have RPFs that were recommended by EPA in 1993. 5 Efforts are ongoing at EPA to expand the number of carcinogenic nonsubstituted PAHs with recommended RPFs. 8 Additionally, the RPF approach for nonsubstituted PAHs will not decrease uncertainties associated with the possible presence of other potential chemical carcinogens in the mixture without CSFs (eg, PAHs with alkylated, N-, S-, or O-containing side groups) or the possible occurrence of nonadditive interactions among components of the mixture (items 1 and 3 above). These limitations contributed to the conclusion by an EPA-convened committee of experts that the component-based RPF approach for PAH mixtures should only be used “as a last resort.” 9
The US EPA guidelines specify that whole-mixture approaches to estimating cancer risks from environmental mixtures are preferable to component-based approaches, but adequate data to use these approaches are most often not available. 4 In a whole-mixture approach, cancer risks are estimated by multiplying estimates of lifetime intakes of the whole mixture (eg, mg of the whole mixture/kg/d) by a CSF derived specifically for the mixture (see Figure 1, panel A for overview). The CSF would be based on the exposure–tumor response data for humans or animals exposed to the whole mixture. This approach eliminates all of the uncertainties associated with the component-based approach, if the tested mixture is identical in composition to the site-specific mixture. However, this is not expected to occur with environmental PAH-containing mixtures, as the composition is complex and influenced by source materials, conditions during use, and conditions in the environment. 1 –3 Further changes in composition are expected after deposition in soil or sediments, due to physical and biological weathering processes. Because of this compositional complexity and variability, coupled with the high cost and animal usage of 2-year bioassays and the difficulties in obtaining sufficient material to conduct a 2-year bioassay, cancer testing of site-specific, environmentally encountered whole mixtures, although desirable, is not practical.
The EPA has developed only 2 toxicity values for PAH-containing whole mixtures to date; an inhalation CSF for coke oven emissions based on the epidemiologic data and an inhalation reference concentration for chronic noncancer toxicity of diesel engine exhaust based on a 2-year rat inhalation study. 10,11 The availability of tumor dose–response data from 2-year dietary administration studies of mice exposed to 2 samples of complex coal tar mixtures from coal gasification plant waste sites presents another opportunity from which a CSF for coal tars from gasification plant waste sites could be developed and applied to estimate cancer risks from populations living close to other coal gasification waste sites. 12,13
Since whole-mixture toxicity values are limited, the US EPA guidelines 4 recommend the use of data for “surrogate” mixtures to estimate health risk for cases when appropriate toxicity data are available for a group of similar mixtures that are known to be generated by the same process (eg, a commercial process or an emission source) and expected to have similar toxicological activity. In these cases, a CSF for the group could be derived and used to estimate cancer risks from an environmentally encountered mixture that is “sufficiently similar” to the tested, or surrogate, mixtures (see Figure 1, panel A for overview). The guidelines provide a broad definition that sufficiently similar mixtures have components that “are not very different and are contained in about the same proportion” and do not have “significant and systematic differences” in environmental fate, pharmacokinetics, bioavailability, or toxicologic effect. However, although using a mixture of sufficient similarity to assess the risks of an untested PAH mixture is appealing and practical, there is no clear scientific agreement on how to assess “sufficient similarity” of mixtures. Therefore, the utility of the whole mixture approach would be increased with a standardized approach for testing the similarities between 2 complex mixtures that is both efficient and scientifically defensible. Although evaluation of chemical composition and generation of “chemical fingerprints” have been proposed to assess mixture similarity, 13,14 it has been demonstrated that in vitro biological testing is more predictive of similar in vivo toxicological responses between 2 mixtures than chemical analyses alone. 16 Therefore, the development of a battery of inexpensive, short-term tests of carcinogenicity based on different modes or mechanisms of action may be useful in determining whether an untested PAH-containing mixture is sufficiently similar to a tested mixture with a derived CSF (see Figure 1, panel A). The battery should include short-term tests of genotoxicity (eg, gene mutation, chromosomal aberration, and aneuploidy), as they are widely recognized to be valuable for identifying carcinogenic chemicals that act through genotoxic mechanisms in rodents. 17 –20 Assays of nongenotoxic mechanisms (eg, deregulation of cell cycle progression, altered cell signaling pathways, and impaired intercellular communication) would be a beneficial supplement to genotoxicity assays. 21,22 This type of battery has been proposed to screen chemicals for carcinogenic potential in order to prioritize toxicity testing under the US EPA’s ToxCast program 23 –25 ; however, to our knowledge, it has not been considered to assess the similarities of mixtures for the purposes of site-specific cancer risk assessment.
Additionally, we propose that in vitro tests of immune suppression, once developed and validated, may be useful supplements to the in vitro battery described previously. This proposal is based on the evidence from clinical and animal studies that “evading immune destruction” is an emerging hallmark of cancer21,26 and preliminary associations between the carcinogenic and the immunosuppressive potentials of PAHs and other chemicals in animal studies. 27,28
Objectives and Focus
The premise of this analysis is that a better mechanistic understanding of the immunotoxicity of BaP, the best-studied PAH, could identify key mechanistic events or cell signaling networks that, when induced or perturbed, increase the risk of immune suppression. Identification of such key events or cell signaling networks may be useful in developing in vitro tests of immune suppression. 22,29,30 We propose that such tests would be a useful addition to a battery of in vitro tests designed to assess mixture similarity in the cancer risk assessment process. First, we evaluated the proposed association between the carcinogenic and the immunotoxic potentials of PAHs by comparing the calculated immune relative potency factors (I-RPFs) for multiple PAHs with available cancer relative potency factors (C-RPFs). Next, we reviewed the available literature on immune suppression in animals exposed to BaP, including mechanistic events and cell signaling pathways that may be involved in BaP-induced immune suppression. The outcome is an integrated knowledge map illustrating immunosuppressive effects and known mechanistic events. From this, we derive recommendations to develop and validate a battery of in vitro tests of immune suppression. This immunosuppression battery, along with validated assays of genotoxic and other nongenotoxic modes of action, could potentially provide a standard screening tool for evaluating the similarity of carcinogenic potential between untested and tested mixtures in the risk assessment process.
Methods
For the development of the knowledge map, PubMed and Google Scholar were searched for studies on the immunotoxicity of BaP through April 2013. From these references, studies evaluating functional immune assays following in vitro and in vivo exposure to BaP and at least one other PAH were used to qualitatively compare relative immune suppression potential with carcinogenic potential (as defined by the International Agency for Research on Cancer 1 )—see Tables 1 and 2, respectively. After review of the available data for in vitro functional immune assays following PAH exposure, the antibody forming cell (AFC) response assay was selected for quantitative analysis, because it was the only immunoassay with adequate data to derive RPFs for multiple PAHs. It is an appropriate assay for the derivation of I-RPFs, as it involves the coordination of multiple immune cell types and is widely used for regulatory purposes. 28,31,32 Using these data, I-RPF values were derived for 9 PAHs using BaP as the index chemical as described in the Supplementary materials. The I-RPFs were regressed against C-RPFs derived by the California EPA (CalEPA) 33 to determine the strength of the association between immune suppression and carcinogenic potencies (Microsoft Excel, 2010). For chemicals with appropriate data, but without a CalEPA C-RPF, we derived C-RPFs for this analysis (see Supplemental materials for details).
In Vitro Evidence for Immunosuppression in Carcinogenic PAHs, But Not PAHs with Low Carcinogenic Potential.a,b
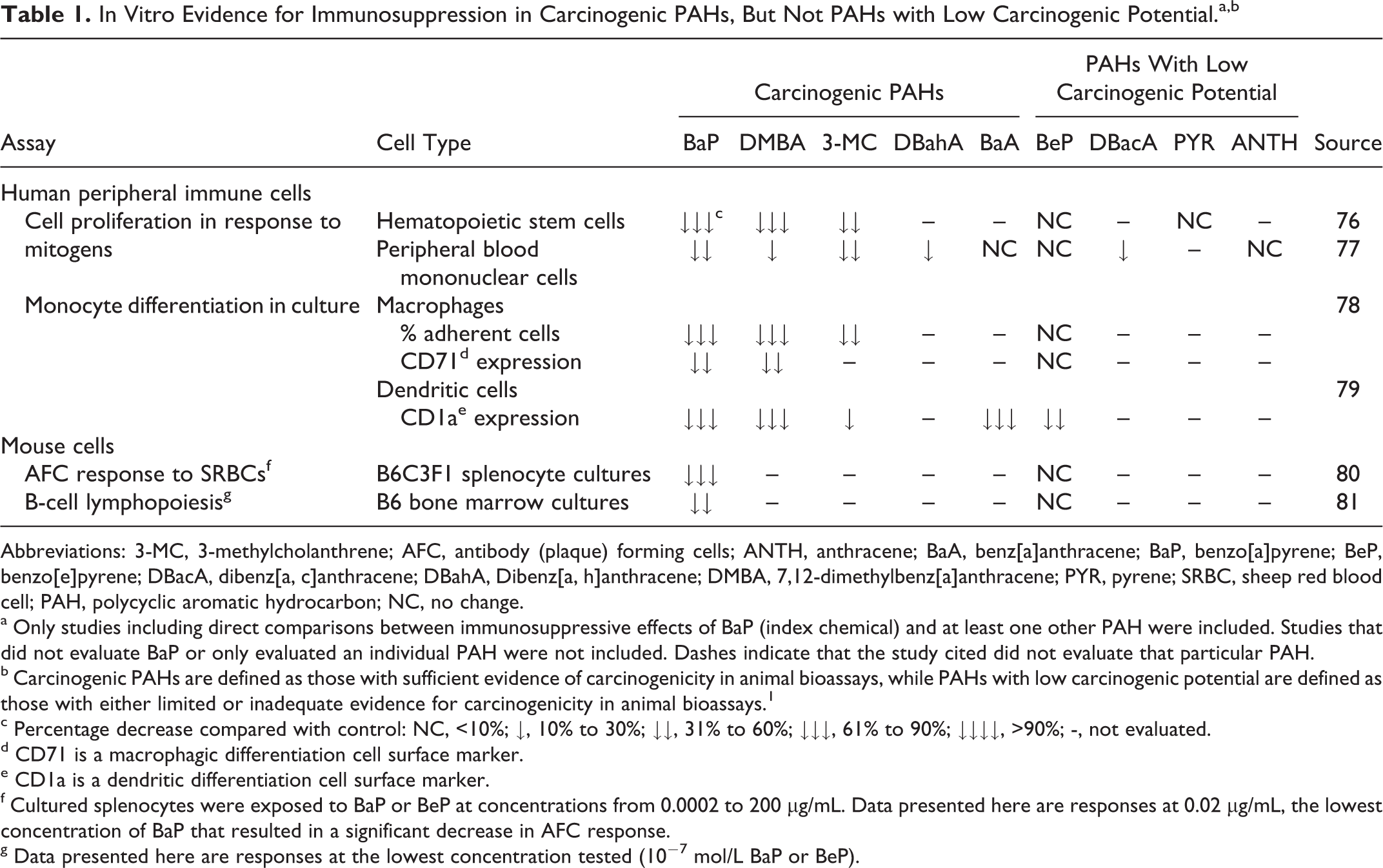
Abbreviations: 3-MC, 3-methylcholanthrene; AFC, antibody (plaque) forming cells; ANTH, anthracene; BaA, benz[a]anthracene; BaP, benzo[a]pyrene; BeP, benzo[e]pyrene; DBacA, dibenz[a, c]anthracene; DBahA, Dibenz[a, h]anthracene; DMBA, 7,12-dimethylbenz[a]anthracene; PYR, pyrene; SRBC, sheep red blood cell; PAH, polycyclic aromatic hydrocarbon; NC, no change.
a Only studies including direct comparisons between immunosuppressive effects of BaP (index chemical) and at least one other PAH were included. Studies that did not evaluate BaP or only evaluated an individual PAH were not included. Dashes indicate that the study cited did not evaluate that particular PAH.
b Carcinogenic PAHs are defined as those with sufficient evidence of carcinogenicity in animal bioassays, while PAHs with low carcinogenic potential are defined as those with either limited or inadequate evidence for carcinogenicity in animal bioassays. 1
c Percentage decrease compared with control: NC, <10%; ↓, 10% to 30%; ↓↓, 31% to 60%; ↓↓↓, 61% to 90%; ↓↓↓↓, >90%; -, not evaluated.
d CD71 is a macrophagic differentiation cell surface marker.
e CD1a is a dendritic differentiation cell surface marker.
f Cultured splenocytes were exposed to BaP or BeP at concentrations from 0.0002 to 200 μg/mL. Data presented here are responses at 0.02 μg/mL, the lowest concentration of BaP that resulted in a significant decrease in AFC response.
g Data presented here are responses at the lowest concentration tested (10−7 mol/L BaP or BeP).
In Vivo Evidence for Immunosuppression in Carcinogencic PAHs, But Not PAHs With Low Carcinogenic Potential.a,b

Abbreviations: 3-MC, 3-methylcholanthrene; ACEN, acenaphthene; ANTH, anthracene; BaA, benz[a]anthracene; BaP, benzo[a]pyrene; BeP, benzo[e]pyrene; BgjiP, benzo[g,h,i]perylene; BbF, benzo[b]fluoranthene; BkF, benzo[k]fluoranthene; CHRY, chrysene; DBacA, dibenz[a,c]anthracene; DBahA, dibenz[a,h]anthracene; DMBA, 7,12-dimethylbenz[a]anthracene; FLU, flourene; FLUO, fluoranthene; I[123cd]P, indeno[1,2,3,c,d]pyrene; PEL, peritoneal exudates lymphocytes; PERY, perylene; PHEN, phenanthrene; PYR, pyrene; PAH, polycyclic aromatic hydrocarbon; AFC, antibody (plaque) forming cell; sc, subcutaneous; NC, no change; ip, intraperitoneal.
a Only studies including direct comparisons between immunosuppressive effects of BaP (index chemical) and at least one other PAH were included. Studies that did not evaluate BaP or only evaluated an individual PAH were not included. Dashes indicate that the study cited did not evaluate that particular PAH. Unless otherwise noted, exposure was 0, 5, 20, or 40 mg/kg/d for 7 to 14 days via sc injection in B6C3F1 mice.
b Carcinogenic PAHs are defined as those with sufficient evidence of carcinogenicity in animal bioassays, whereas PAHs with low carcinogenic potential are defined as those with either limited or inadequate evidence or for carcinogenicity in animal bioassays. 1
cStudies report effect at various pathogen doses. Data reported here reflect the largest percentage change from vehicle control observed.
dPercentage change compared with control at highest dose tested (unless otherwise noted): NC, <10%; ↓, 10% to 30%; ↓↓, 31% to 60%; ↓↓↓, 61% to 90%; ↓↓↓↓, >90%; -, not evaluated.
e Data presented here are average of male and female data from original report.
f Exposure was a single gavage dose of 0.1, 1, 10, or 100 mg/kg in C57BL/6 mice.
g Exposure was a single ip injection of 0, 2.5, 10, or 50 mg/kg in B6, C3H, or D2 mice; data presented here are an average of the findings for all 3 mouse strains tested.
Results
Evidence for an Association Between Immunosuppressive and Carcinogenic Activities of PAHs
Evidence from in vitro studies with animal and human cells (Table 1) and in vivo animal studies (Table 2) demonstrates that BaP consistently impairs performance in immune function assays, indicating that it is a potent immunosuppressant. These studies also support a categorical association between immune suppressing and tumorigenic activities of several PAHs. In support of this, I-RPFs and C-RPFs were clearly correlated for the 9 non-BaP PAHs shown in Table 3 (R 2 =.8976; see Supplemental Figure S1). These results extend findings from previous studies indicating that, in general, the immunosuppressive and carcinogenic activities of chemicals are correlated. 27,28 Confirmation of this association in PAHs supports our proposition that in vitro assays of immune suppression would be a useful addition to an in vitro battery of genotoxic and other nongenotoxic mode of action assays that could be used to establish “sufficient similarity” between 2 PAH-containing mixtures.
Immunosuppression and Cancer Relative Potency Factors for Various PAHs.
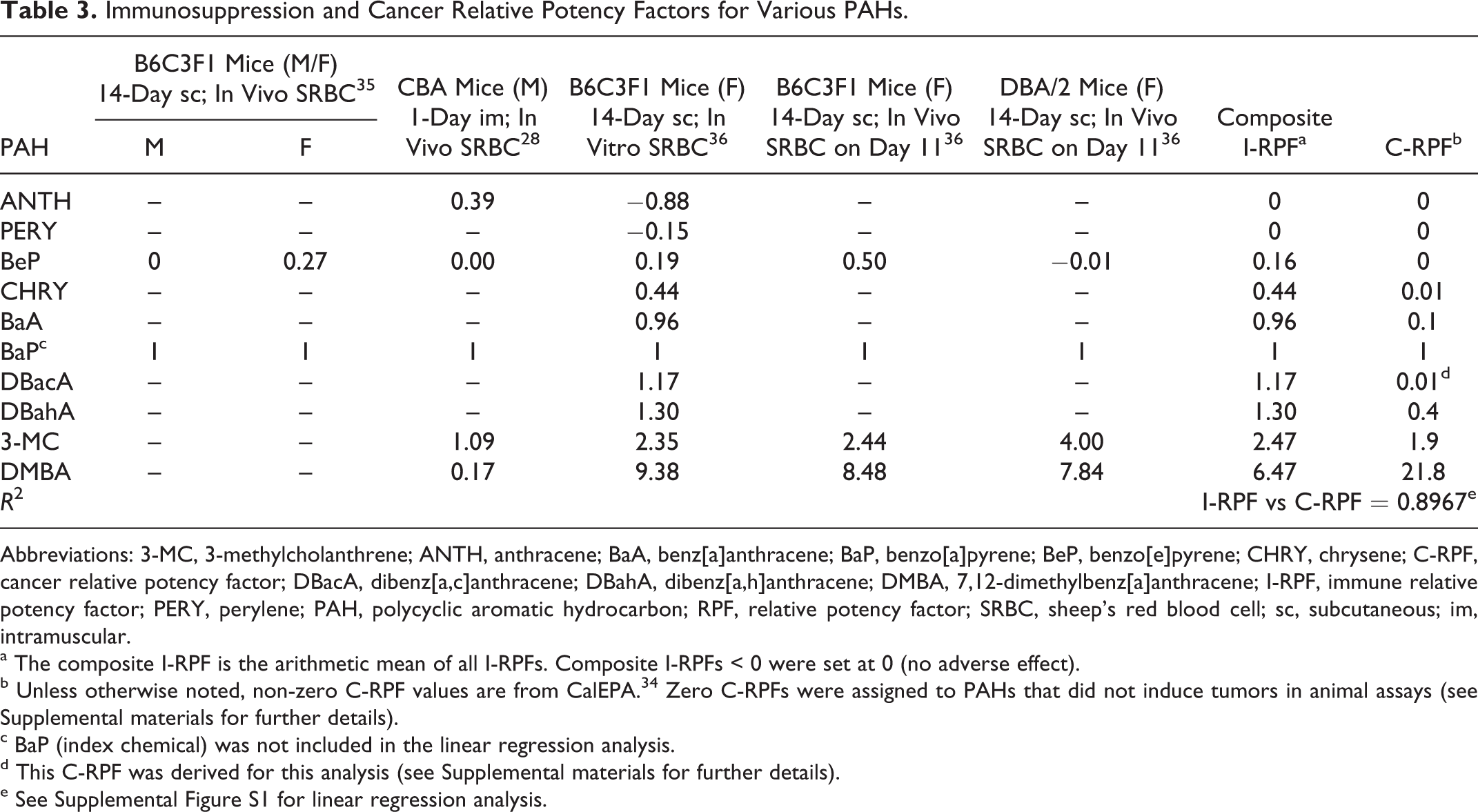
Abbreviations: 3-MC, 3-methylcholanthrene; ANTH, anthracene; BaA, benz[a]anthracene; BaP, benzo[a]pyrene; BeP, benzo[e]pyrene; CHRY, chrysene; C-RPF, cancer relative potency factor; DBacA, dibenz[a,c]anthracene; DBahA, dibenz[a,h]anthracene; DMBA, 7,12-dimethylbenz[a]anthracene; I-RPF, immune relative potency factor; PERY, perylene; PAH, polycyclic aromatic hydrocarbon; RPF, relative potency factor; SRBC, sheep’s red blood cell; sc, subcutaneous; im, intramuscular.
a The composite I-RPF is the arithmetic mean of all I-RPFs. Composite I-RPFs < 0 were set at 0 (no adverse effect).
b Unless otherwise noted, non-zero C-RPF values are from CalEPA. 34 Zero C-RPFs were assigned to PAHs that did not induce tumors in animal assays (see Supplemental materials for further details).
c BaP (index chemical) was not included in the linear regression analysis.
d This C-RPF was derived for this analysis (see Supplemental materials for further details).
e See Supplemental Figure S1 for linear regression analysis.
Integrated Knowledge Map of BaP Immune Suppression
To summarize the immunosuppressive effects of BaP and known tissue-specific mechanistic information, we developed an integrated knowledge map (Figure 2) based on the results of 44 in vivo animal studies, 25 in vitro studies with human cells, and 14 in vitro studies with animal cells (see Table 4). Figure 2 illustrates how BaP adversely affects multiple components of the immune system, demonstrates the complexity of the potential interactions between the various target tissues/cells, and illustrates the mechanisms involved in BaP immunosuppression. Review of this knowledge map identified several key findings that could assist the development of in vitro assays for immune suppression.
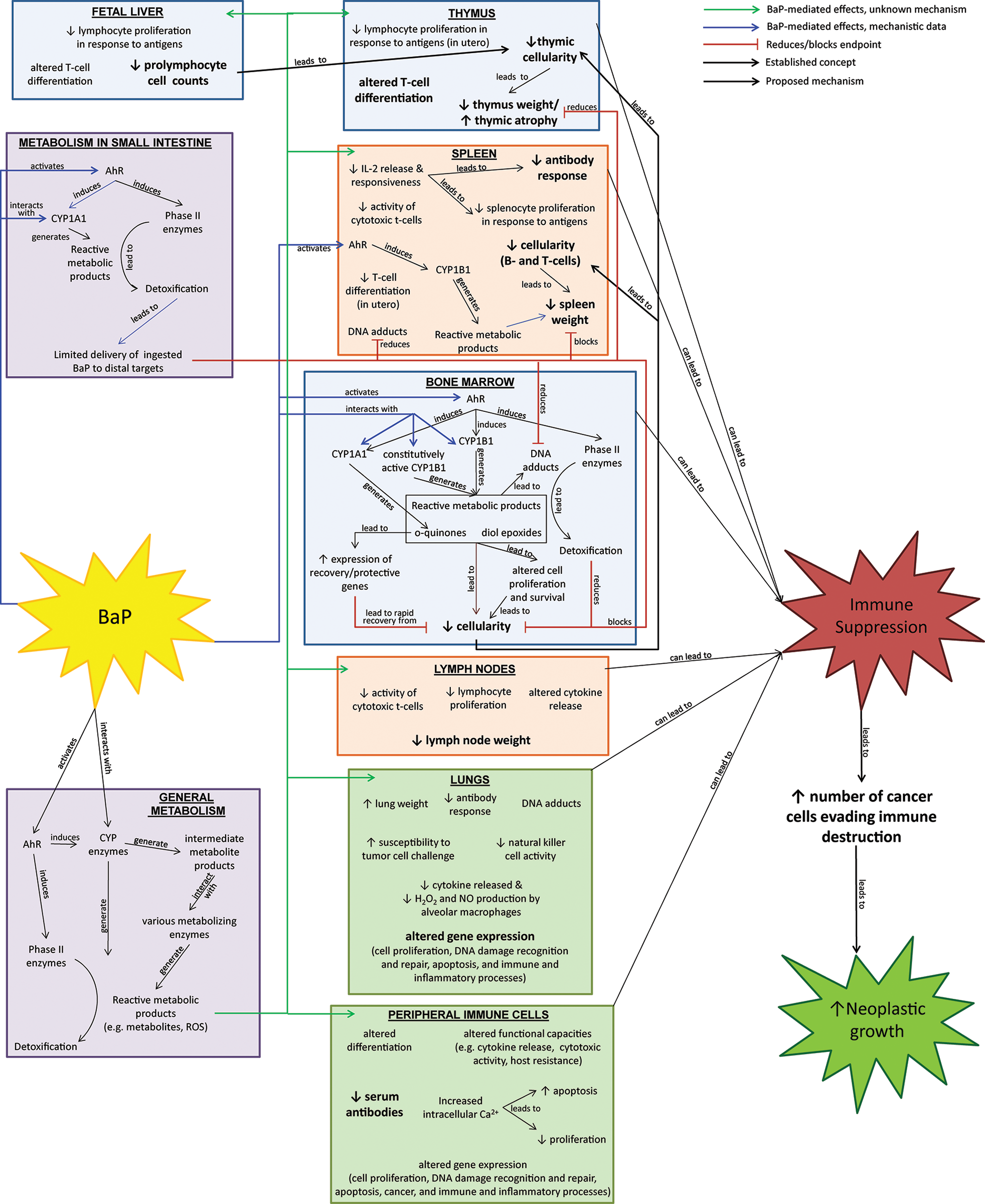
Integrated knowledge map of benzo[a]pyrene (BaP) immunotoxicity. Known effects and mechanisms were organized into tissue-specific target “boxes” (blue = primary immune; orange = secondary immune tissues; and green = peripheral immune cells/tissues). Bolded end points represent adverse effects observed following oral exposure (an environmentally relevant exposure route). When available, tissue-specific metabolic information was placed within the appropriate box. If inadequate information was located regarding tissue-specific metabolic information, general metabolism is represented in the purple box, and may occur prior to delivery to the target tissue, within the tissue, or in both the locations. “Proposed mechanisms” (dotted lines) are possible mechanisms indicated by authors of the primary reports.
Studies Used to Develop the Integrated Knowledge Map (Figure 2).

Key Finding: BaP Exposure Adversely Affects Multiple Immune System Components and Functional Capacities
As illustrated in Figure 2, the literature indicates that BaP exposure affects primary immune tissues (bone marrow, thymus, and fetal liver), secondary immune tissues (spleen and lymph nodes), and peripheral immune cells. Adverse effects include organ weight changes/atrophy; altered cellular proliferation, differentiation, and survival; DNA adduct formation; and various impairments in functional end points. Multiple gene expression changes in immune tissues that may contribute to immune suppression or development of cancer were also observed (see Supplemental Table S7 for details). Observed effects in secondary immune tissues and peripheral elements observed following in vivo exposure to BaP may be due to direct effects from reactive metabolites produced in these organs, indirect effects on progenitor cells or lymphocytes in primary immune tissues, or a combination of both. Review of the knowledge map indicates that BaP exposure can cause a diverse set of effects in various immune tissues which, alone or in concert, have the potential to cause immune suppression and decreased immune surveillance of cancer cells.
Relevance to developing an in vitro battery
The usefulness of a battery of short-term assays of immune suppression will likely be improved if it includes tests of multiple immune tissues and functional end points.
Key Finding: BaP-Induced AhR Activation of In Vivo Detoxification and Toxification Processes
Multiple studies have reported a dual role for activation of the aryl hydrocarbon receptor (AhR) by BaP (reviewed in 110 ). Studies with genetically modified mouse strains indicate that an AhR-regulated CYP1A1 detoxification pathway in the gastrointestinal tract limits the delivery of orally administered BaP to distal targets, including the bone marrow, spleen, and thymus.45,60,61,65,78,81,82 However, upon delivery to distal targets, AhR-regulated CYP1B1 toxification pathways lead to production of metabolites that mediate BaP-induced immunotoxic effects in the bone marrow and spleen, whereas metabolites produced via a non-CYP1B1 pathway (not yet identified) appear to mediate BaP effects in the thymus. 37,82 Nonimmune cells in the bone marrow microenvironment may be required for toxic effects of nearby immune cells, because BaP-mediated effects in bone marrow cultures occurred only when mouse progenitor B cells were cultured with bone marrow stromal cells expressing functional AhR complexes. 89 Additionally, intraperitoneal studies (that bypass gastrointestinal metabolism) indicate that BaP-induced hypocellularity following BaP delivery to the marrow is dependent upon a balance of toxification and detoxification processes, including multiple AhR-regulated metabolic pathways and detoxification/recovery processes within the bone marrow. 37,46,38
Relevance to developing an in vitro battery
Data from oral administration studies demonstrate that delivery of the parent compound, followed by metabolism in bone marrow tissue, is at least partially responsible for the immunotoxic effects of BaP in bone marrow. This indicates that in vitro assays utilizing parent compounds, rather than metabolites, are appropriate as long as test systems are metabolically active and recapitulate in vivo metabolic processes as much as possible. This may require in vitro systems to contain multiple cell types from the target organ to most accurately reflect the in vivo tissue microenvironment.
Key Finding: Mechanisms of Immunotoxicity Are Best Characterized in Bone Marrow
A large portion of current knowledge regarding mechanisms of BaP-induced immunotoxicity comes from in vitro and in vivo studies of BaP toxicity to the bone marrow. As discussed earlier, the mechanisms are complex, including AhR-dependent pathways that are involved in both toxification and detoxification of BaP. The main toxic effect observed in bone marrow is hypocellularity, likely due to cytotoxic activities of BaP metabolites produced in bone marrow tissue.
Relevance to developing an in vitro battery
Because bone marrow plays a central role in supplying progenitor immune cells to peripheral sites of immune action, and immunotoxic mechanisms of BaP are best characterized in the bone marrow, assays of cytotoxicity and apoptosis within an in vitro bone marrow system may be useful assays to consider for the battery. Although less is known about toxic mechanisms in the thymus, this organ also plays a role in supplying progenitor cells to peripheral sites of immune action. Thus, similar assays in thymic cultures should also be considered.
Key Finding: Gene Expression Studies Have Yet to Identify a “Transcriptional Signature” for PAH Immunotoxicity
The most comprehensive gene expression studies following in vivo exposure have revealed several mechanistic aspects of the immunotoxicity of BaP and another carcinogenic and immunosuppressive PAH, 7,12-dimethylbenz[a]anthracene (DMBA; see Supplemental Table S7 for study details). 36,46,48,52 Results from these studies show that (1) multiple cell signaling pathways, including AhR metabolic pathways, oxidative stress and inflammatory pathways, and p53 signaling pathways, are perturbed by in vivo exposure to BaP; (2) perturbations are tissue, dose, and time dependent; (3) perturbations are not yet fully characterized or linked to changes in immune function; and (4) transcriptional changes in the bone marrow following exposure to DMBA differ from BaP-induced changes.
Results from the studies of in vitro exposure of human lung cells and human peripheral macrophages to BaP (or its metabolite benzo[a]pyrene diolepoxide) also show time-dependent alterations in the expression of multiple genes involved in multiple cell signaling pathways. 43,54,76,95,96,103 These in vitro studies indicate that BaP can alter genes involved in (1) multiple signaling pathways, including AhR, p38 MAPK, p53, and c-jun N-terminal kinase pathways; (2) immune inflammatory processes, including cytokine production and release; (3) cell cycle regulation; (4) DNA damage recognition and repair; and (5) control of cell proliferation and apoptosis.
Relevance to developing an in vitro battery
Although progress is being made in this emerging field, a transcriptional signature of immune suppression that could be used to develop targeted in vitro immunotoxicity assays is yet to be identified.
Discussion
Association Between Carcinogenicity and Immunotoxicity of PAHs in Animals
We confirmed that there is an association between carcinogenic and immunosuppressive potentials for various PAHs using both qualitative (Tables 1 and 2) and quantitative analyses (Table 3). Our analysis extends previous reports by Stjernsward 27 and Luster et al, 28 which identified concordances between carcinogenic and immunosuppressive chemicals (including PAHs). However, evaluation of this association could be furthered with the generation of additional immune suppression data for PAHs with varying cancer potencies (eg, benzo[b] fluoranthene, dibenzo[a,l]pyrene, and benzo[c] fluorene) and/or developing I-RPFs based on an immune assay with direct relevance to cancer immunosurveillance (eg, natural killer [NK] cell-mediated cytolysis 21 ). By itself, the association between immunosuppresion and carcinogenic potency factors for PAHs does not demonstrate a causal relationship between the immunotoxic potential of a PAH and its ability to cause tumors, nor does it indicate mechanistic similarities between immunosuppressive and carcinogenic activities. The association may reflect common upstream events (eg, metabolic activation to certain reactive metabolites) that render some PAHs carcinogenic and immunosuppressive, with no further association between the 2 events. However, the immunosuppressive effects of some PAHs may contribute to their carcinogenic potential, because “evading immune destruction” is a potential key event in the development of cancer, 21,26 and BaP can inhibit the immune system’s response to certain types of tumor cells in the laboratory animals. 99,105 Therefore, it is plausible that the addition of in vitro assays of immune suppression would be useful to include in an in vitro battery of genotoxic and other nongenotoxic mode of action assays that could be used to establish “sufficient similarity” between 2 PAH-containing mixtures.
Plausibility of PAH Immune Suppression in Humans
Although the literature review in this analysis focused on animal studies, several epidemiological studies indicate that exposure to PAH mixtures containing BaP may lead to immunosuppresion in humans.111–120 Adverse effects include altered serum immunoglobin and cytokine levels, altered T- and B-cell proliferation and surface antigen expression, increased peripheral blood mononuclear cell apoptosis, altered NK cell numbers, altered phagocytic activity in monocytes, and evidence of DNA damage in peripheral lymphocytes. However, detailed compound-specific and mechanistic data are not available in these human studies due to 1 or more of the following reasons: (1) multiple concurrent chemical exposures, (2) small sample sizes, (3) no measure of exact exposures, and (4) lack of adjustment for potential confounding factors (eg, age). Although these limitations preclude the use of epidemiological studies in this analysis to obtain a better mechanistic understanding of the immunotoxicity of BaP, they do provide evidence that the immunotoxic effects observed in animals might also occur in humans.
Predictive Tests for Immune Suppression
In vivo mouse studies have demonstrated that specific immune end points sensitively and accurately predicted suppression of immune function in vivo (eg, AFC response and quantitation of splenic B and T lymphocytes). 28,121,122 However, even short-term in vivo tests are likely to be too expensive for routine site-specific health risk assessments. Validated in vitro assays are not available for functional immune suppression,123–125 but several prevalidation studies have evaluated the ability of different test systems to distinguish a limited number of immunosuppressants from nonimmunosuppressants. Evaluated test systems included cytotoxic activities and mitogen- and allogeneic cell-stimulated proliferation of cultured rodent splenocytes, and human blood mononuclear cells; cytokine release from mitogen-stimulated human and rodent whole blood cells; cytotoxicity, cytokine release, and antibody- and mitogen-stimulated cell proliferation with cultured rodent and human lymphocytes; and in vitro antibody response to sheep’s red blood cells (SRBCs) and mitogen-stimulated proliferation in cultured rodent spleen cells (reviewed in 125 ). Results from each of these efforts suggest a concordance between in vitro and in vivo immunotoxicity tests; however, these prevalidation studies have numerous limitations. The most critical limitation is that far too few chemicals have been evaluated to fully determine whether any of the “prevalidated” assays are both sensitive and predictive. Additionally, these prevalidation studies have not performed detailed time-course and dose–response analyses.
Based on the multiplicity of tissues and mechanisms involved in BaP immunosuppression illustrated in Figure 2, the complexity of the immune system presents challenges for the development of in vitro tests and suggests that multiple tests involving different processes, cell types, and tissues will likely have greater predictability of in vivo immune suppression than a single test. This was demonstrated for in vivo immune tests, which individually ranged from relatively good-to-poor predictors of host resistance assays (54%-82% concordance) and showed improved predictability when combinations of multiple tests were used (58%-100% concordance). 121 Because of the complexity of the immune system and the possibility of mechanistic variance among immune suppressing chemicals, validation of in vitro immunosuppression tests will be more likely if several tissue systems and immune end points are evaluated individually and in combination. Efforts would require a large number of chemicals with positive and negative effects identified by in vivo immunosuppression tests, 124 preferably host resistance assays. Widespread use of in vitro tests will be enhanced if evaluated tissue systems are easy to obtain and maintain, are metabolically reflective of in vivo metabolism, and represent multiple cell types and processes involved in in vivo immune resistance to challenge from infectious agents and tumors as possible.
An assay of antibody response in cultured rodent spleen cells is 1 candidate assay that presents multiple processes to be targeted by active immunosuppressants, because it involves the interaction of macrophages, T cells, and B cells.126–128 The in vitro antibody response to SRBCs has been shown to demonstrate high sensitivity and specificity and to closely reflect antibody response following in vivo exposure, 31,126 making it an excellent candidate for further evaluation. To achieve metabolic competency, this system may need to be supplemented with an S9 metabolic system.126,128 Currently, it is controversial whether primary antibody responses can be elicited in human peripheral lymphocyte cultures 125 ; however, it has been shown to elicit antibody responses in rat peripheral blood mononuclear cultures. 31 Assays of other immune functions in human peripheral blood or mononuclear cells would complement a rodent antibody response spleen or peripheral blood culture assays. Of particular interest would be development and validation of assays evaluating immune functions involved in cancer surveillance that have been shown to be affected by BaP exposure, such as cytotoxic T-lymphocyte or NK cell-mediated cytolysis. Other assays to consider include mitogen-stimulated cell proliferation and cytokine release. These assays are in agreement with functional assays for immune suppression suggested by Galbiati and colleagues. 125 Given the central role that bone marrow and thymus play in supplying progenitor immune cells to peripheral sites of immune action, and evidence that BaP can lead to direct cellular toxicity in these tissues (see Figure 2), assays of cytotoxicity and apoptosis with cultured bone marrow or thymus cells may be considered to supplement assays of immune functions in other immune tissue systems. This is consistent with the recommendation that in vitro tests of myelotoxicity be considered as a tier 1 screen for immunosuppression by untested compounds by The European Centre for the Validation of Alternative Methods. 124
Transcriptional Profiles in Immune Tissues as Predictive Tests
Increased understanding of the concordance between PAH-induced changes in gene transcription and immune functions is an active area of mechanistic research. The available information indicates carcinogenic PAHs, such as BaP and DMBA and suppresses immune functions through different mechanisms. Changes in gene expression are compound and tissue/cell specific, involve multiple cell signaling networks, and are dose and time dependent. Current studies do not identify a candidate transcriptional signature for immune suppression by PAHs. Research needed to identify a predictive signature requires a more complete characterization of PAH-induced gene expression changes, including their time-, dose- and tissue-dependent linkage to immune functional changes and investigations of a wider variety of PAHs and other compounds with various carcinogenic and immunosuppressant potencies. Once candidate signatures have been identified, additional research may include development and validation of in vitro systems that reflect responses to in vivo exposure.
Conclusions
Because evading the immune system can be important in the development of cancer, supplementing an in vitro battery of short-term genotoxic and nongenotoxic carcinogenic tests with short-term tests for immune suppression may improve the usefulness of such a battery to accurately predict the toxicological similarity of an untested mixture to a mixture with an existing toxicity value. This type of battery, if validated, would be extremely useful for site-specific cancer risk assessments of hazardous waste sites.
The complexities of the immune system and mechanisms involved in BaP immunosuppression suggest that multiple in vitro tests of immune suppression involving different processes, cell types, and tissues will have greater predictive value for immune suppression than a single test. Future efforts to validate in vitro immunosuppression tests will likely require evaluation of a number of tissue systems and immune end points (alone and in combination), using a large number of chemicals known to have positive and negative results from in vivo immunosuppression tests. Future widespread use of in vitro tests will be enhanced if tissue systems are easy to obtain and maintain, are metabolically reflective of in vivo metabolism, and contain several functional cell types and processes involved in immune function in vivo. Appropriate in vitro tissue systems to evaluate include (1) an assay of antibody response by cultured rodent spleen cells supplemented with an S9 metabolic system; (2) assays of immune function by human peripheral blood or mononuclear cells (particularly assays of immune functions involved in cancer immunosurveillance, eg, NK cell-mediated cytolysis); and (3) assays of cytotoxicity and apoptosis using cultured bone marrow or thymus cells. Current understanding of PAH-induced gene expression changes in immune tissues and their link to immune functions are inadequate to identify gene expression changes that may be predictive of chemical immune suppression.
Following validation of a battery of in vitro immune suppression tests, further research could be designed to evaluate a battery of in vitro tests of genotoxic and nongenotoxic modes of carcinogenic action (including the immune suppression battery) to predict the carcinogenicity of individual chemicals and complex mixtures in cancer bioassays. As with the evaluation of an immune suppression test battery, this validation exercise should use as many noncarcinogenic and carcinogenic chemicals of varying structure as resources permit and evaluate the tests individually and in combinations. A validated battery could be potentially used as a standardized method to establish “sufficient similarity” of carcinogenic potential between 2 mixtures. This method would be efficient, cost effective, and scientifically defensible and may lead to an increase in the use of whole-mixture versus component-based toxicity values in cancer risk assessment of complex environmental mixtures containing PAHs and other substances.
Footnotes
Acknowledgments
We thank Dr Stephen Nesnow, Dr Charles Czuprynski, and several colleagues at SRC, Inc, for critical reading of this manuscript and/or expert technical assistance.
Author’s Note
Supplemental Figure S1. Linear regression analysis of relative potency factors.
Supplemental Table S1. The I-RPF calculation from Munson and White (1983)—14-day exposure in B6C3F1 mice, in vivo SRBC exposure (day 11).
Supplemental Table S2. The BMD modeling results for Munson and White (1983)—14-day exposure in male B6C3F1 mice, in vivo SRBC exposure (day 11).
Supplemental Table S3. The I-RPF calculation from Stjernsward (1966)
Supplemental Table S4. The I-RPF calculation from White et al (1985)—14-day exposure in B6C3F1 mice, in vitro SRBC exposure.
Supplemental Table S5. The I-RPF calculation from White et al (1985)
Supplemental Table S6. The C-RPF calculation for DBacA from Slaga et al (1980).
Supplemental Table S7. Summary of the studies of gene transcriptional changes in mouse tissues with immune system components after in vivo exposure to BaP or DMBA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: There are no actual or potential competing interests to declare. Drs Zaccaria and McClure both work as toxicologists on contracts to SRC, Inc to provide technical support in the area of human health risk assessment to US regulatory (eg, EPA and CPSC) and public health (ATSDR) agencies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support to write this manuscript was provided by internal SRC, Inc funds.
