Abstract
Rebaudioside D (Reb D) is one of the several glycosides found in the leaves of Stevia rebaudiana (Bertoni) Bertoni (Compositae) which has been identified as a potential sweetener. The metabolism of Reb A and Reb D was evaluated in various in vitro matrices (simulated gastrointestinal fluids, rat liver microsomes, and rat cecal contents) and through analysis of plasma collected from rats in a dietary toxicity study. Reb A and Reb D showed similar stability when exposed to simulated stomach and small intestine fluids, with susceptibility to hydrolytic degradation by enteric bacteria collected from the cecum. Incubations with rat liver microsomes indicated that neither compound is expected to be metabolized by the liver enzymes. Plasma concentrations of Reb D, Reb A, and/or the final hydrolysis product of each compound, free/conjugated steviol, were consistent between animals administered either Reb D or Reb A in the diet. A repeated exposure dietary toxicity study was conducted to compare the safety of Reb D, when administered at target exposure levels of 500, 1000, and 2000 mg/kg body weight (bw)/d to Sprague-Dawley rats for 28 days, to that of Reb A administered at a target exposure level of 2000 mg/kg bw/d. There were no treatment-related effects on the general condition and behavior of the animals and no toxicologically relevant, treatment-related effects on hematology, serum chemistry, or urinalysis. Macroscopic and microscopic findings revealed no treatment-related effects on any organ evaluated. Results were comparable between the group administered 2000 mg/kg/d Reb D and the group administered 2000 mg/kg/d Reb A.
Introduction
Rebaudioside D (Reb D) is one of the several glycosides of the diterpene derivative, steviol (ent-13-hydroxykaur-16-en-18-oic acid) naturally occurring in the plant, Stevia rebaudiana (Bertoni) Bertoni (Compositae). 1 Reb D is being explored for use as a high-potency sweetener. The chemical structure of Reb D is shown in Figure 1.
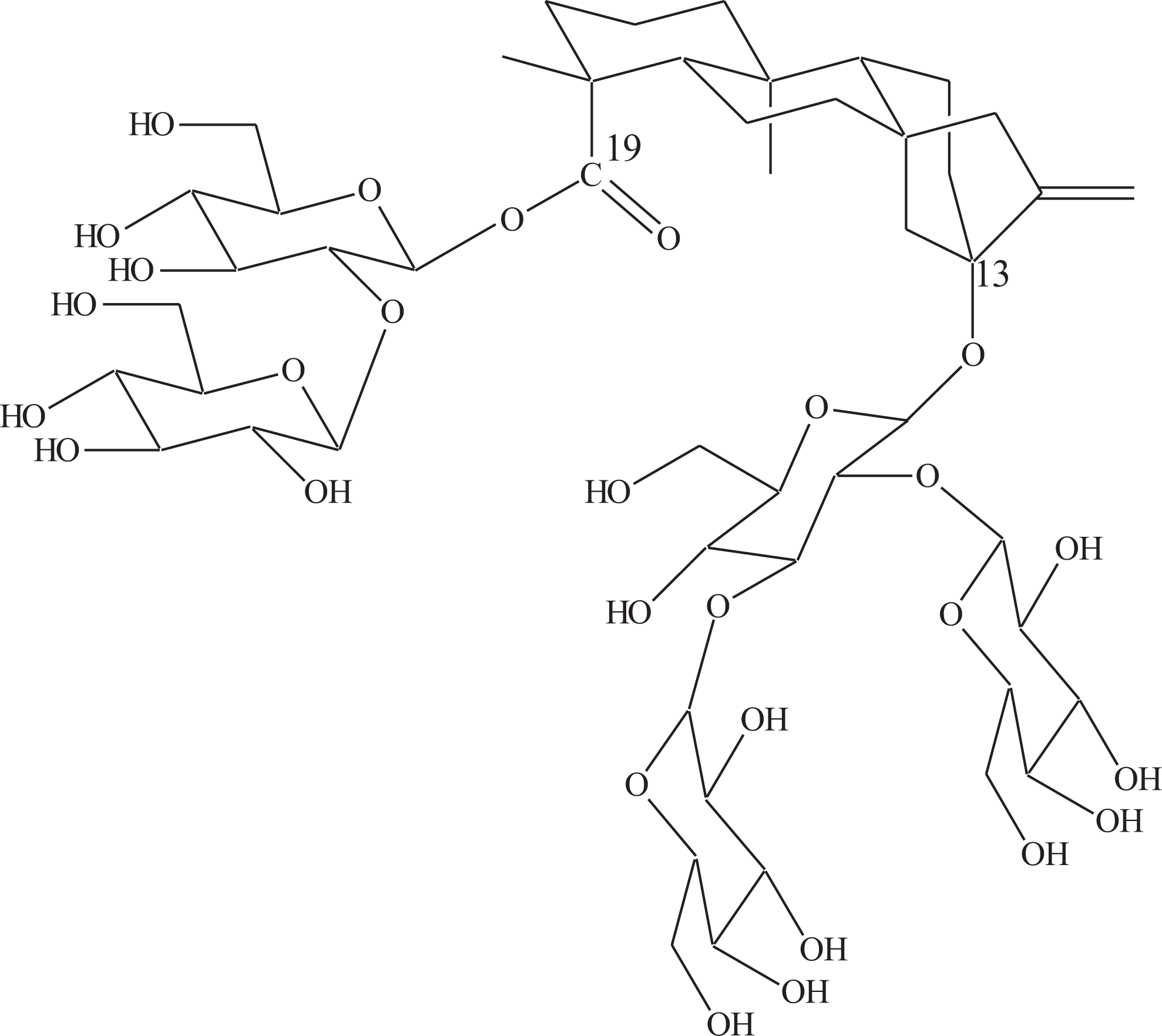
Chemical structure of Rebaudioside D.
The Joint Food and Agricultural Organization of the United Nations/World Health Organization Expert Committee on Food Additives (JECFA), 2 during its 69th meeting, evaluated steviol glycosides and assigned an acceptable daily intake of 0 to 4 mg/kg body weight (bw; expressed as steviol) based on a no observed effect level of 970 mg/kg bw/d from a long-term experimental study with stevioside (383 mg/kg bw/d expressed as steviol) and a safety factor of 100. The final specifications for steviol glycosides were prepared at the 73rd meeting of JECFA, 3 identifying stevioside and Reb A as the primary component glycosides, with associated steviol glycosides including Reb D as secondary component glycosides.
Reb A achieved Generally Recognized As Safe status in 2008 based on Notifications submitted to the Food and Drug Administration (FDA) by Whole Earth Sweetener Company (GRN 252) 4 and Cargill Inc (GRN 253). 5
Because Reb D is closely structurally related to Reb A, having just 1 additional glucose unit (refer to Figure 2), it was hypothesized that the toxicological profile of each compound would be similar. The studies presented herein were conducted in order to demonstrate that the metabolism and toxicity profiles of Reb D are similar to those of Reb A. With a demonstration of a similar toxicological profile, the existing safety database of Reb A and steviol glycosides may be used in the safety evaluation of Reb D. In addition to experiments performed to evaluate the metabolism of Reb A and Reb D in various in vitro matrices, a 28-day dietary bridging toxicity study was conducted. To corroborate the in vitro metabolism study results, blood samples were collected and plasma analyzed (for parent Reb A, Reb D, and major metabolites) from subgroups of animals included in the 28-day dietary bridging toxicity study. Genetic toxicity studies with Reb D were not conducted due to the extensive number of in vitro and in vivo mutagenicity studies available for Reb A, all of which were negative (GRN 252 4 and GRN 253 5 ).
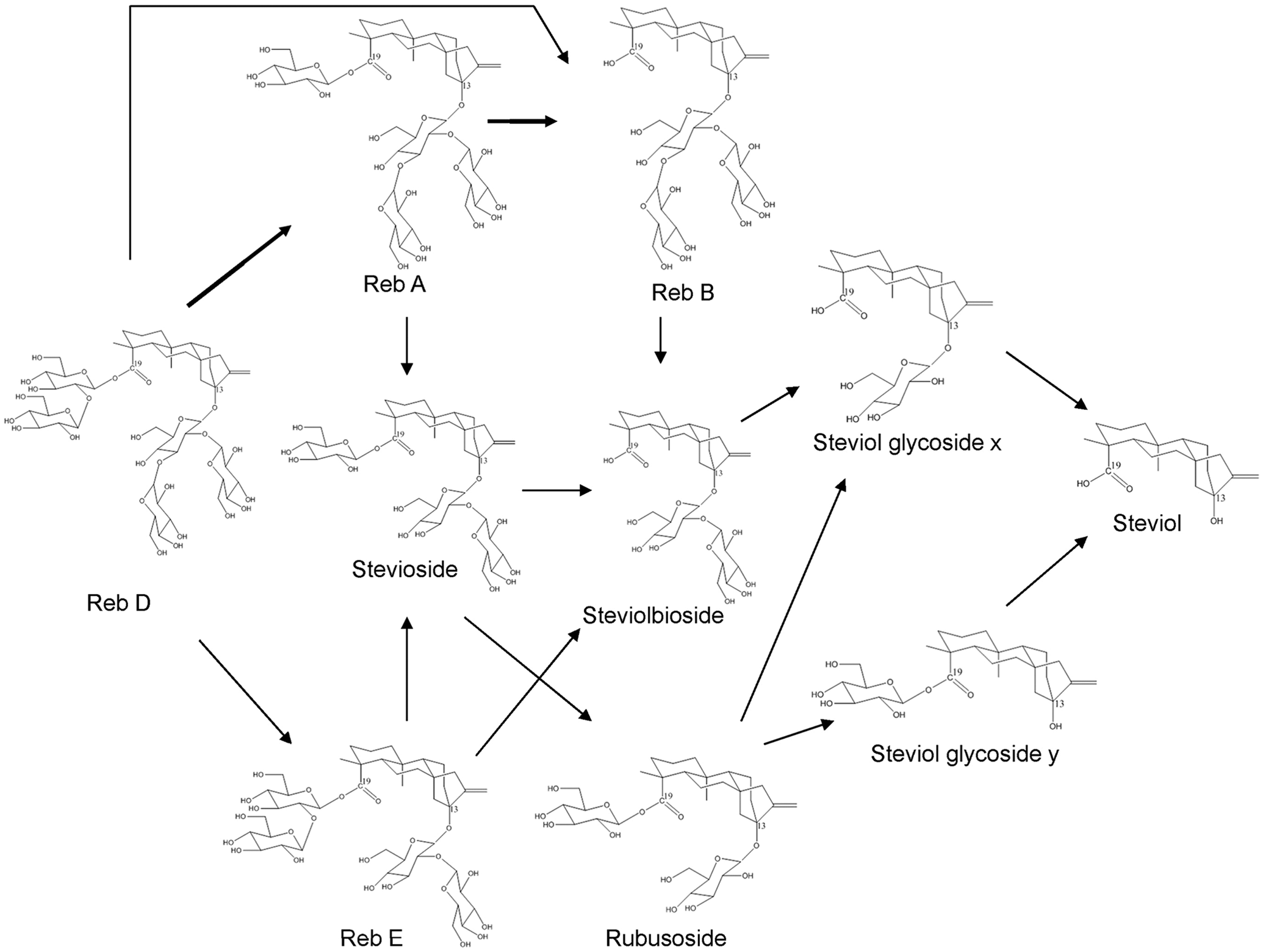
Hydrolysis pathways of Rebaudioside D to the aglycone, steviol.
Materials and Methods
Test and Reference Articles
The test articles, Reb D (CAS RN 63279-13-0) and Reb A (CAS RN 58543-16-1), were received from PureCircle USA Inc (Oak Brook, Illinois) on behalf of the Sponsor. The purity of Reb D was 93.5% (with the remaining 6.5% comprising predominantly steviol glycosides) and the purity of Reb A was 98.9%. The test articles were stored at room temperature, protected from light, with controlled humidity, and were considered stable under these conditions.
Reference standards for potential metabolites (stevioside, Reb B, steviolbioside, and steviol) were obtained from ChromaDex, Inc (Irvine, California) and stored according to the manufacturer instructions.
Animal Receipt, Acclimation, and Husbandry
For the 28-day dietary toxicity study, Crl:CD(SD) rats (100 males and 100 females) were received in good health from Charles River Laboratories, Inc (Raleigh, North Carolina) on February 16, 2010. Male Crl:CD(SD) rats utilized in the in vitro metabolism study were originally received in good health from Charles River Laboratories and obtained from the WIL Research stock colony (Ashland, Ohio) prior to collection of cecal contents and intestines. Each animal was uniquely identified with a subcutaneous microchip (Bio Medic Data System) implanted in the scapular area. Upon arrival, all the animals were housed 2 to 3 per cage by sex for approximately 3 days. Thereafter, all animals were housed individually in clean, stainless steel, wire mesh cages suspended above cage board.
All animals were housed for a minimum 15-day acclimation/pretest period prior to initiation of test diet administration in the 28-day toxicity study. During this period, each animal was observed twice daily for mortality and changes in general appearance or behavior. Individual bws and food consumption were recorded, and detailed physical examinations were performed periodically during the pretest period. Ophthalmic examination data were also recorded for pretest animals prior to randomization.
Animals were maintained in accordance with the Guide for the Care and Use of Laboratory Animals (National Research Council, 1996). 6 The animal facilities at WIL Research are accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. Enrichment devices were provided to all animals as appropriate throughout the study for environmental enrichment and to aid in maintaining the animals’ oral health and were sanitized weekly.
Assignment of Animals to Treatment Groups (28-Day Dietary Toxicity Study)
The animals judged suitable for assignment to the study were selected for use in a computerized randomization procedure based on bw stratification in a block design. Individual bws at randomization were within ±20% of the mean for each sex. Animals not assigned to the study were transferred to the WIL Research stock colony.
Each group consisted of 10 males and 10 females. These animals were then randomized into 5 study replicates to allow for the reasonable conduct of the functional observational battery (FOB) and motor activity (MA) assessments. Each dose group and sex were approximately equally represented within each study replicate. The animals were approximately 6 weeks old at the initiation of the test diet administration. Individual bws ranged from 166 to 209 g for males and from 129 to 171 g for females at the time of randomization (study day 2/ 3 [males/females]).
In Vitro Experiments
Screening experiments (not subject to Good Laboratory Practice Regulations) were performed at WIL Research to evaluate the metabolism of Reb A and Reb D in various in vitro matrices (freshly collected rat cecal contents, cryopreserved rat liver microsomes, and simulated gastric and intestinal fluids).
Simulated Gastrointestinal Fluid Incubations
Simulated gastrointestinal (GI) fluids were prepared with and without digestive enzymes using the instructions in the United States Pharmacopeia and National Formulary. 7 The activity of the enzymes in the simulated GI fluids was evaluated prior to formulation of the simulated fluids. Simulated gastric fluid with enzymes (SGF+) and simulated gastric fluid without enzymes (SGF−) test solutions had a final pH of approximately 1.2; SGF+ was prepared with pepsin. Simulated intestinal fluid with enzymes (SIF+) and simulated intestinal fluid without enzymes (SIF−) test solutions had a final pH of approximately 7.5; SIF+ was prepared with pancreatin. Test solutions containing approximately 0.2 mmol/L Reb A or Reb D were incubated for up to 240 minutes at 38°C. Reactions were quenched with methanol at various time points for sample collection.
Microsomal Incubations
Cryopreserved rat liver microsomes were obtained from XenoTech, LLC (Lenexa, Kansas). Test solutions containing approximately 0.2 mmol/L Reb A or Reb D and 0.2 mg/mL liver microsomal protein were incubated at 37°C for up to 90 minutes following the addition of β-nicotinamide adenine dinucleotide 2′-phosphate (NADPH) to initiate the enzymatic reaction. Reactions were quenched with methanol at various time points for sample collection. Control experiments were also conducted in the absence of NADPH. Enzymatic activity in rat liver microsomes was confirmed in parallel incubations by the transformation of 7-ethoxycoumarin to 7-hydroxycoumarin.
Cecal Content Incubations
The cecum was ligated and removed from 5 male Crl:CD(SD) rats at the time of euthanasia by carbon dioxide (CO2) inhalation. The cecums were then placed in an anaerobic chamber for collection of cecal contents and brief centrifugation to separate solids. Test solutions containing approximately 0.2 mmol/L Reb A or Reb D and dilute cecal contents’ supernatant or bicarbonate buffer were incubated at 37°C for up to 90 minutes. Reactions were quenched with methanol at various time points for sample collection. The viability of the rat cecal contents was confirmed by the degradation of bovine insulin in parallel incubations.
Analysis of Test Solutions
Following incubation in the respective matrix, the concentration of Reb A, Reb D, and potential metabolites (stevioside, Reb B, steviolbioside, and steviol) was determined by liquid chromatography/tandem mass spectrometry (LC/MS/MS) or LC/ultraviolet (LC/UV) absorption spectroscopy.
28-Day Dietary Bridging Toxicity Study
Experimental Design Overview
The toxicity of Reb D was compared to that of a structurally related and previously studied compound, Reb A (Nikiforov and Eapen), 8 in a 28-day feeding study in Sprague-Dawley rats conducted at WIL Research. This study was conducted in accordance with the U.S. FDA Redbook 2000 testing guidelines 9 and in compliance with the U.S. FDA Good Laboratory Practice Regulations 10 and the OECD Principles of Good Laboratory Practice. 11
Reb D was administered for a minimum of 28 consecutive days on a continuous basis in the diet to 3 groups (groups 2 to 4) of Crl:CD(SD) rats. Target dosage levels were 500, 1000, and 2000 mg/kg/d. Dietary concentrations were adjusted weekly based on expected average weight and current food consumption. A concurrent control group (group 1) received the basal diet, PMI Nutrition International, LLC Certified Rodent LabDiet 5002 (meal), on a comparable regimen, and a concurrent comparator group (group 5) received Reb A at a target dosage level of 2000 mg/kg/d on a comparable regimen. Each group consisted of 10 animals/sex. Following at least 28 days of dietary exposure, all animals were euthanized. Table 1 provides the animal allocation to groups 1 to 5 and the mean average Reb D or Reb A consumption for the 28-day period.
Experimental Design and Mean Average Reb D and Reb A Consumption.
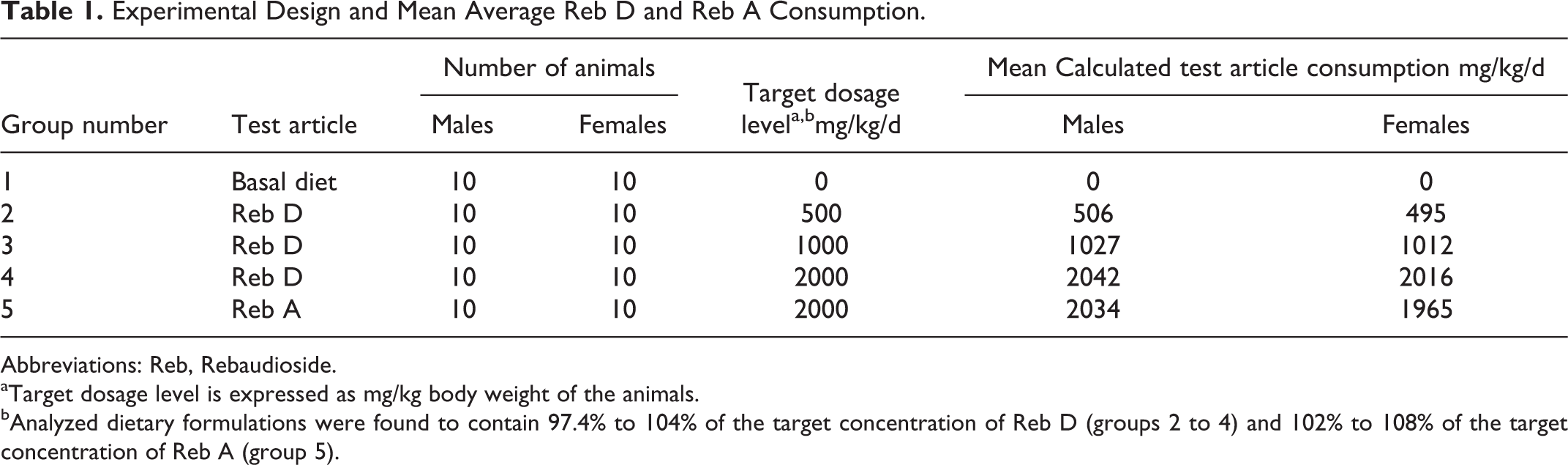
Abbreviations: Reb, Rebaudioside.
aTarget dosage level is expressed as mg/kg body weight of the animals.
bAnalyzed dietary formulations were found to contain 97.4% to 104% of the target concentration of Reb D (groups 2 to 4) and 102% to 108% of the target concentration of Reb A (group 5).
All animals, which were individually housed for the duration of the study, were observed twice daily for mortality and moribundity. Clinical examinations were performed daily, and detailed physical examinations were performed weekly. Individual bws and food consumption were recorded weekly. The FOB and locomotor activity data were recorded for all animals during study week 3. Ophthalmic examinations were performed during pretest (study day 6 for males and study day 7 for females) and near the end of the treatment period (study day 28 for males and study day 27 for females). Blood samples were collected for hematology and serum chemistry evaluations from all animals on the day of the scheduled necropsy (study day 28). Urine was collected overnight from animals housed in metabolism cages. Complete necropsies were conducted on all animals, and selected organs were weighed at the scheduled necropsy. Selected tissues and organs were collected from all animals and placed in the appropriate fixative. Selected tissues were examined microscopically from all animals in the control group (group 1), the 2000 mg/kg/d Reb D group (group 4), and the 2000 mg/kg/d Reb A group (group 5). As no treatment-related or toxicologically relevant findings were observed in tissues from animals at the high-dose levels, tissues from animals in the remaining dose groups were not examined microscopically.
In addition to the main toxicology study animals, subgroups of 9 animals/sex/dose (groups 2A to 5A) were maintained as toxicokinetic phase animals and administered the test articles (Reb D or Reb A) for 28 consecutive days. An untreated toxicokinetic phase group of 3 animals/sex (group 1A) received the basal diet on a comparable regimen. All toxicokinetic phase animals, which were individually housed for the duration of the study, were observed twice daily for mortality and moribundity. Individual bws and food consumption were recorded weekly.
Preparation of Test and Control Diets
For the control group, based on the laboratory’s experience, an appropriate amount of PMI Nutrition International, LLC, Certified Rodent LabDiet 5002 (meal) was weighed in a properly labeled storage bag on a weekly basis.
Diets containing the test articles were prepared approximately weekly on a weight/weight basis and placed in properly labeled storage bags. The initial diet concentrations were based on average food consumption and bws during the pretest period. Test article concentration in the diet was adjusted as necessary based on the mean bw and food consumption for each group (by sex) to maintain the appropriate target dosage.
Administration of Diets
The test article and control diets were offered ad libitum for 28 consecutive days, until the day prior to the scheduled necropsy. The dosage levels selected for this study were based on the dosage levels used in a previous 90-day dietary toxicity study evaluating Reb A (Nikiforov and Eapen), 8 since this study was conducted for the purpose of comparing Reb D with Reb A. These dosage levels represent feasible dietary exposures that are palatable and do not replace a significant amount of nutrients in the basal rodent diet. The selected route of administration for this study was oral (dietary) as the test article is a food ingredient and intended for human consumption.
Analysis of Reb D and A in Rat Feed
From the first admix prepared for dosing, samples for homogeneity, concentration, and stability determination were collected from the top, middle, and bottom strata of the bulk groups 2 and 4 preparations (5110 and 21 650 ppm Reb D, respectively). In addition, samples for stability determinations were collected from the middle stratum of an extra feed jar placed in the study room following room temperature storage for up to 10 days. Samples for concentration analysis were collected from the middle stratum during study week 0 from the control group, the remaining Reb D group, and the Reb A group and study week 3 from all the groups. All analyses were conducted using a validated high-performance LC (HPLC) method using UV absorbance detection (internal laboratory method meeting acceptance criteria for specificity/selectivity, calibration reproducibility, precision, accuracy, and ruggedness; calibration range was 50.0 to 200 μg/mL).
Clinical Observations and Survival
All animals were observed twice daily, once in the morning and once in the afternoon, for mortality and moribundity. Detailed physical examinations were conducted on all animals weekly, beginning at least 1 week prior to test article administration and prior to the scheduled necropsy. Daily clinical observations were not performed on days when detailed physical examinations were conducted.
Body Weights
Individual bws were recorded approximately weekly, beginning during the pretest period. Mean bws and mean bw changes were calculated for the corresponding intervals. A final nonfasted bw was collected on the day prior to the scheduled necropsy. A final bw (fasted) was recorded on the day of the scheduled necropsy.
Food Consumption
Individual food consumption was recorded weekly, beginning approximately 1 week prior to test article administration (study week 1 to 0). Food intake was calculated as g/animal/d for the corresponding bw intervals. The mean amounts of Reb D and Reb A consumed (mg/kg/d) by each sex per dose group were calculated from the mean food consumed (g/kg/d) and the appropriate target concentration of test article in the food (mg/kg).
Functional Observational Battery FOB and Locomotor Activity
The FOB and locomotor activity assessments were recorded for all the animals during study week 3. Testing was performed by trained technicians without knowledge of the animals’ group assignments in a sound-attenuated room equipped with a white noise generator set to operate at 70 ± 10 dB. Animals were observed for the FOB parameters defined in the previously developed protocols (Gad 12 ; Haggerty 13 ; Irwin 14 ; Moser et al.; 15 Moser et al.; 16 O’Donoghue 17 ). Total locomotor activity was defined as a combination of fine locomotor skills (ie, grooming; interruption of a single photobeam) and ambulatory locomotor activity (eg, interruption of 2 or more consecutive photobeams).
Clinical Pathology
Blood and urine samples for clinical pathology evaluations (hematology, coagulation, serum chemistry, and urinalysis) were collected from all animals at the scheduled necropsy (study day 28). Analysis was conducted on those parameters reported in the previous 90-day dietary toxicity study with Reb A (Nikiforov and Eapen 8 ) in accordance with the FDA Redbook 2000 guidance. 9 The animals were fasted overnight prior to blood collection, while in metabolism cages for urine collection. Blood was collected for hematology and serum chemistry evaluation via the retro-orbital sinus of animals anesthetized by inhalation of isoflurane. Blood was collected for coagulation parameters at the time of euthanasia via the vena cava of animals euthanized by inhalation of CO2. Blood was collected into tubes containing potassium EDTA (hematology), sodium citrate (coagulation), or no anticoagulant (serum chemistry).
Macroscopic Examination
A complete necropsy was conducted on all animals. Animals were euthanized by CO2 inhalation followed by exsanguination. The necropsies included examination of the external surface, all orifices, and the cranial, thoracic, abdominal, and pelvic cavities including viscera. At the time of necropsy, select tissues and organs were collected and preserved in the appropriate fixative. The listing of tissues and organs was as reported in the previous 90-day dietary toxicity study with Reb A (Nikiforov and Eapen 8 ) in accordance with the FDA Redbook 2000 guidance. 9
Histopathologic Procedures and Microscopic Examination
Fixed tissues were trimmed, processed into paraffin blocks, and sectioned followed by staining with hematoxylin and eosin (Luna). 18 Microscopic examinations were performed on collected tissues from all animals in the control (group 1), 2000 mg/kg/d Reb D (group 4), and 2000 mg/kg/d Reb A (group 5) groups at the scheduled necropsy.
Toxicokinetic Evaluation
To demonstrate the nature of absorbed doses, blood samples were collected from 3 nonfasted animals/sex/group at approximately 0, 4, 8, 12, 16, and 24 hours following the removal of food on study days 1 and 22/21 (males/females) for groups 2A to 5A. In addition, blood samples were collected from 3 animals/sex in group 1A on study days 1 and 22/21 (males/females). All toxicokinetic group animals were euthanized and discarded following 27/26 (males/females) days of test diet administration. Plasma was isolated after centrifugation under refrigerated conditions and stored frozen at approximately 70°C until analysis.
Plasma collected from each blood sample on days 1 and 22/21 (males/females) from toxicokinetic group animals was analyzed for concentrations of Reb D, Reb A, and their potential major hydrolysis or conjugation product (Reb B, stevioside, steviolbioside, steviol, and steviol glucuronide) concentrations using a HPLC/MS/MS method in the negative electrospray ionization mode. The lower limit of quantitation (LLOQ) was 10 ng/mL for all analytes (qualification of internal laboratory method conducted at WIL Research). Quantification of an additional hydrolysis product, Reb E, was also attempted; however, a reference standard for Reb E was not available at the time of study conduct. Pharmacokinetic parameters were not calculated, since dietary exposures were available ad libitum.
Statistical Methods
Analyses were conducted using 2-tailed tests for significance levels of 5% and 1%, comparing each test article-treated group to the control group by sex. The bw, bw change, food consumption, locomotor activity data, clinical pathology parameters, and absolute and relative organ weight values were subjected to a parametric 1-way analysis of variance (ANOVA; Snedecor and Cochran) 19 followed by Dunnett’s Test (Dunnett). 20 The FOB parameters that yielded scalar or descriptive data were analyzed using Fisher Exact Test (Steel and Torrie). 21
Repeated measures analysis of variance (RANOVA) were conducted for total and ambulatory locomotor activity counts recorded during pretest and after dosing.
Results: In Vitro Metabolism Study
Results obtained in the various matrices are summarized below and demonstrate that while the test articles were not susceptible to substantial degradation by the simulated GI fluids or liver enzymes, incubation in the cecal contents matrix did result in extensive degradation of the test articles; therefore, the cecum is expected to be the primary site of hydrolysis in both Reb A and Reb D.
Metabolism in Simulated GI Fluids
There was a slight decrease in Reb D concentration over 240 minutes in all matrices, but the decrease was only statistically significant in SGF− and SIF−, decreasing approximately 20% and 8.8%, respectively, from the measured starting concentration (data not shown). There was no statistically significant decrease in Reb A concentration in SGF+ (pH 1.2), SIF+(pH 7.5), or SIF−. In SGF−, there was a small (approximately 10%) and statistically significant decrease in the Reb A concentration over 240 minutes. With the exception of a small concentration (<6 µg/mL) of stevioside in Reb A SGF incubations, no stevioside, Reb B, steviolbioside, or steviol was detected in any SGF or SIF incubations. No Reb D was detected in the Reb A incubations with SGF or SIF. The Reb A was detected in some Reb D incubations (consistent with an expected hydrolysis pathway of Reb D) but was generally below the LLOQ of 2.0 µg/mL. Based on the likelihood that a small amount of Reb A was present in the Reb D test material as characterized by the Supplier (purity of Reb D was 93.5%, with the remaining 6.5% comprising other steviol glycosides), this was not an unexpected result. Purified Stevia extracts generally contain small amounts of other steviol glycosides, the composition of which depends on the composition of the leaves, influenced by soil and climate conditions, and on the manufacturing process. 22
Metabolism by Rat Liver Microsomes
There was no decrease in the concentration of Reb A or Reb D over the time course of the experiment performed with rat liver microsomes in the presence and absence of NADPH (data not shown). There was a low concentration of Reb A (average 3.0 µg/mL) detected in Reb D incubations, which is consistent with an expected hydrolysis pathway of Reb D (see Figure 2) and/or the potential for Reb A to be present as a naturally occurring impurity at low levels. A low concentration of Reb D (average 5.0 µg/mL) was detected in Reb A incubations; this is also attributable to the purity (98.9%) and characterization of the Reb A test material, which probably contained a small amount of Reb D. In addition, a low concentration of steviolbioside (initially 30 µg/mL and decreased to approximately 23 µg/mL after 40 minutes) was detected in microsomal incubations with Reb A and Reb D in the absence of NADPH (data not shown). In the presence of NADPH, no steviolbioside was detected.
Metabolism in Cecal Contents In Vitro
The concentration of both Reb A and Reb D remained unchanged over the time course of the incubation in bicarbonate buffer (data not shown). In the presence of cecal contents, Reb A and Reb D concentrations were decreased extensively by microflora hydrolysis to a level that was not detectable (Table 2). This decrease corresponded with increasing concentrations of stevioside and/or steviol over 90 minutes, which is consistent with expected hydrolysis pathways of Reb A and Reb D (see Figure 2).
Concentration (µg/mL) of Reb D, Reb A, and Major Hydrolysis Products in Rat Cecal Contents Incubations.
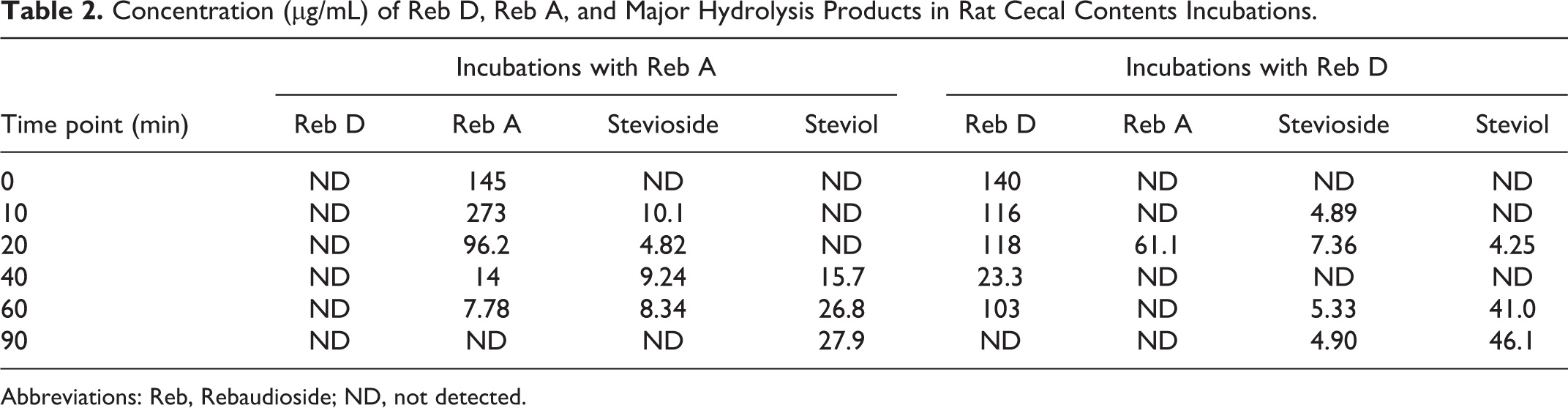
Abbreviations: Reb, Rebaudioside; ND, not detected.
Results: 28-Day Dietary Bridging Toxicity Study
Analyses of Test Diet Formulations
The analyses of the test article dietary formulations were found to contain 97.4% to 104% of the protocol-specified concentration of Reb D and 102% to 108% of Reb A throughout the study and were homogeneous (data not shown).
Survival and Clinical Observations
All animals survived until the scheduled necropsy. There were no test article-related clinical observations. Clinical observations in the test article-treated groups (including hair loss and soft feces) were noted with similar incidence in the control group, were limited to single animals, were not noted in a dose-related manner, and/or were common findings for laboratory rats of this age and strain.
Bws, Food Consumption, and Reb D and A Intake
There were no test article-related effects on bws. A statistically significantly higher mean cumulative bw gain was noted from study day 0 to 21 in the 2000 mg/kg/d Reb D group females (data not shown). However, this difference was not considered toxicologically relevant due to the small magnitude of change.
There were no test article-related effects on food consumption. Although some intervals in the female groups treated with Reb D were statistically significantly increased compared to the control group, these differences were not considered to be toxicologically relevant due to the small magnitude of the changes. There were no other statistically significant differences when the control and test article-treated groups were compared (data not shown).
Average test article consumption (mg/kg/d) was based on the nominal dietary levels of the test article and is shown in Table 1.
Functional Observational Battery and Motor Activity
No test article-related effects on home cage, handling, open field and sensory, neuromuscular, or physiological parameters were observed during the FOB (data not shown). Locomotor activity was unaffected by test article administration. Significantly lower total and ambulatory activity counts were noted in the 500, 1000, and 2000 mg/kg/d Reb D group females. These differences from the control group were not consistently observed in males and are considered to be related to faster habituation, which is not typically regarded as a treatment-related effect. Additionally, there were no remarkable differences in these counts between the 2000 mg/kg/d Reb D group females and the 2000 mg/kg/d Reb A group females.
Hematology, Serum Chemistry, and Urinalysis
There were no test article-related alterations in hematology and coagulation parameters (data not shown). There were no statistically significant differences when the test article-treated group males and females were compared to the control group.
There were no test article-related alterations in serum chemistry or special chemistry parameters (data not shown). There were no statistically significant differences when the test article-treated group males and females were compared to the control group.
There were no test article-related effects on urinalysis parameters (data not shown).
Macroscopic Examination, Microscopic Examination, and Organ Weights
No test article-related alterations were noted. Dilatation of the uterus and clear fluid contents was noted in the 500 mg/kg/d (group 2), and the 2000 mg/kg/d (groups 4 and 5) group females and not in the control group. Histologically, this finding correlated with a dilated uterine lumen and an estrous cycle stage of late proestrus or early estrus. Dilatation of the uterine lumen and accumulation of clear fluid is normal during the estrous cycle, and the change was not considered to be test article related (Li and Davis 23 ; Westwood 24 ).
Remaining histological changes were considered to be incidental findings, manifestations of spontaneous diseases, or related to some aspect of experimental manipulation other than administration of the test article.
There were no test article-related alterations in final bw or organ weights. There were no statistically significant differences when the test article-treated group males and females were compared to the control group.
Toxicokinetic Evaluation
For animals receiving 2000 mg/kg bw/d Reb D via the diet for 28 days, plasma concentrations of Reb D, Reb A, and its final hydrolysis product, free/conjugated steviol, were measurable in all the samples on both 24-hour sampling days with fluctuations among the time points related to typical nocturnal feeding and sleeping cycles of rats (Figures 3 –6). For animals receiving 2000 mg/kg bw/d Reb A via the diet for 28 days, plasma concentrations of Reb A and its final hydrolysis product, free/conjugated steviol, were measurable in all the samples on both sampling days with similar fluctuations among the time points related to typical nocturnal feeding and sleeping cycles of rats (Figures 3 –6). These results demonstrate low mean systemic exposure to all analytes for both sexes at all time points, particularly for both parent materials (≤1.5 µg/mL), with the majority of the absorbed dose present as free (≤12 µg/mL) or conjugated steviol (≤40 µg/mL). Systemic exposure in plasma to the other Reb D or Reb A hydrolysis products (Reb B, stevioside, steviolbioside) was extremely low or below the levels of quantitation (data not shown). The data shown in Figures 3 –6 illustrate the common hydrolysis pathways of Reb D and Reb A, with hydrolysis occurring slowly so that almost all parent material passes through the gut unchanged, and the small amount of steviol that is formed as the ultimate hydrolysis product is rapidly and efficiently conjugated to form steviol glucuronide, which is excreted in the urine.
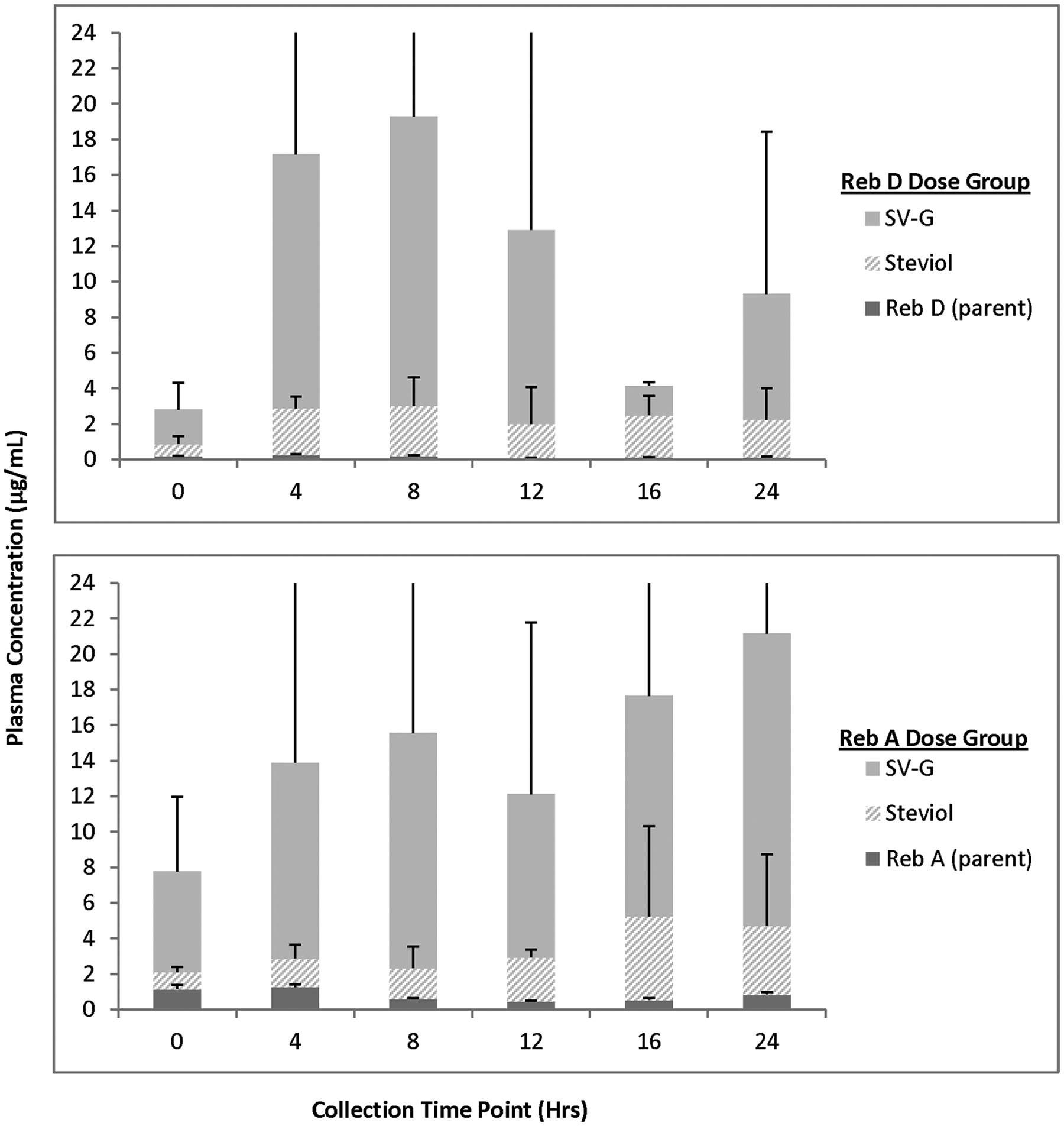
Mean (+standard deviation) plasma concentration of Rebaudioside (Reb) D, Reb A, and final hydrolysis products in male rats fed 2000 mg/kg/d Reb D or Reb A in the diet (day 1). Only positive standard deviation error bars shown for clarity of presentation due to variability in individual animal data at select time points. N = 3 rats/sex/group at each time point (toxicokinetic phase animals); SV-G = steviol glucuronide.
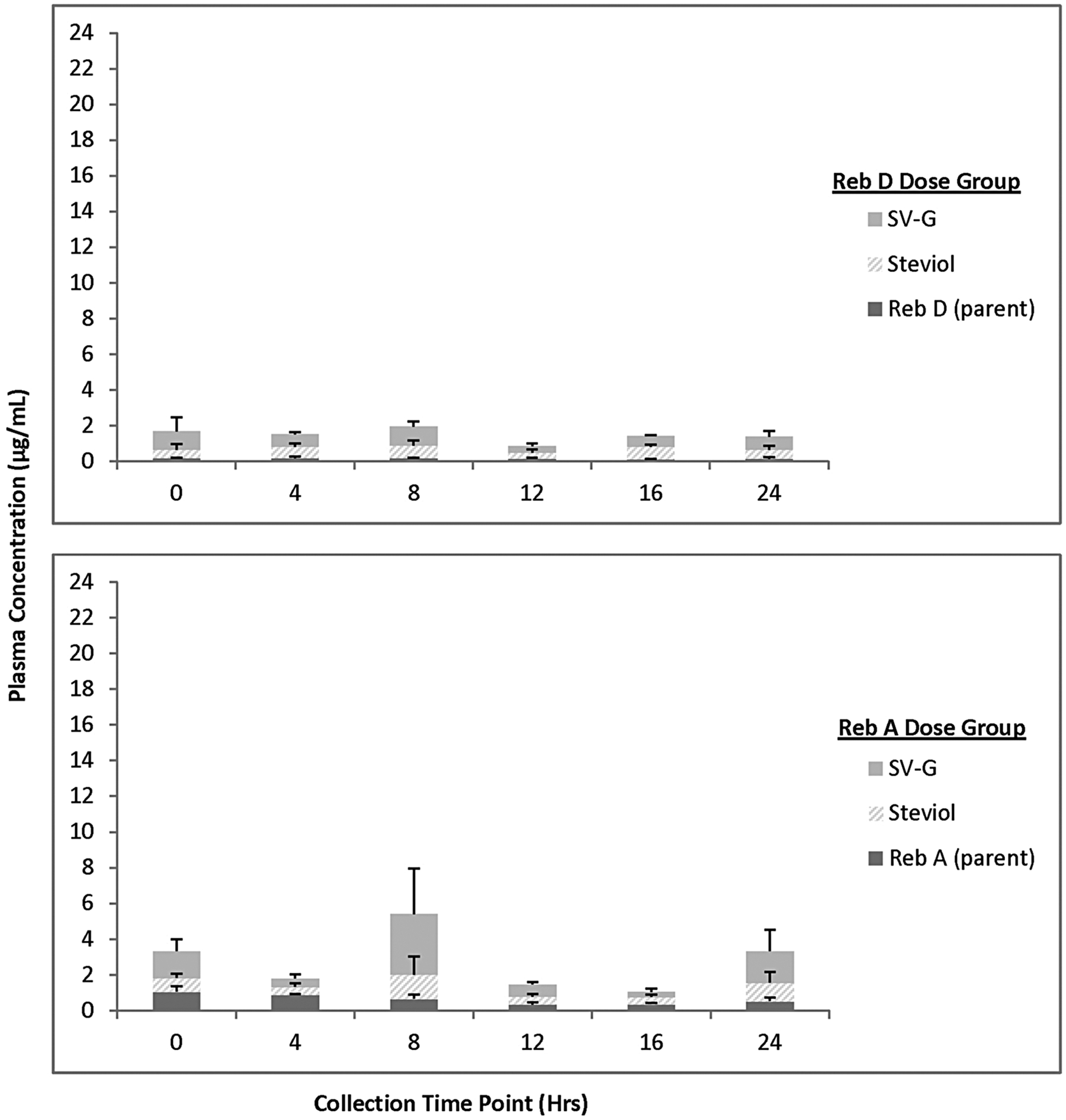
Mean (+standard deviation) plasma concentration of Rebaudioside (Reb) D, Reb A, and final hydrolysis products in male rats fed 2000 mg/kg/d Reb D or Reb A in the diet (day 22). Only positive standard deviation error bars shown for clarity of presentation due to variability in individual animal data at select time points. N = 3 rats/sex/group at each time point (toxicokinetic phase animals); SV-G = steviol glucuronide.
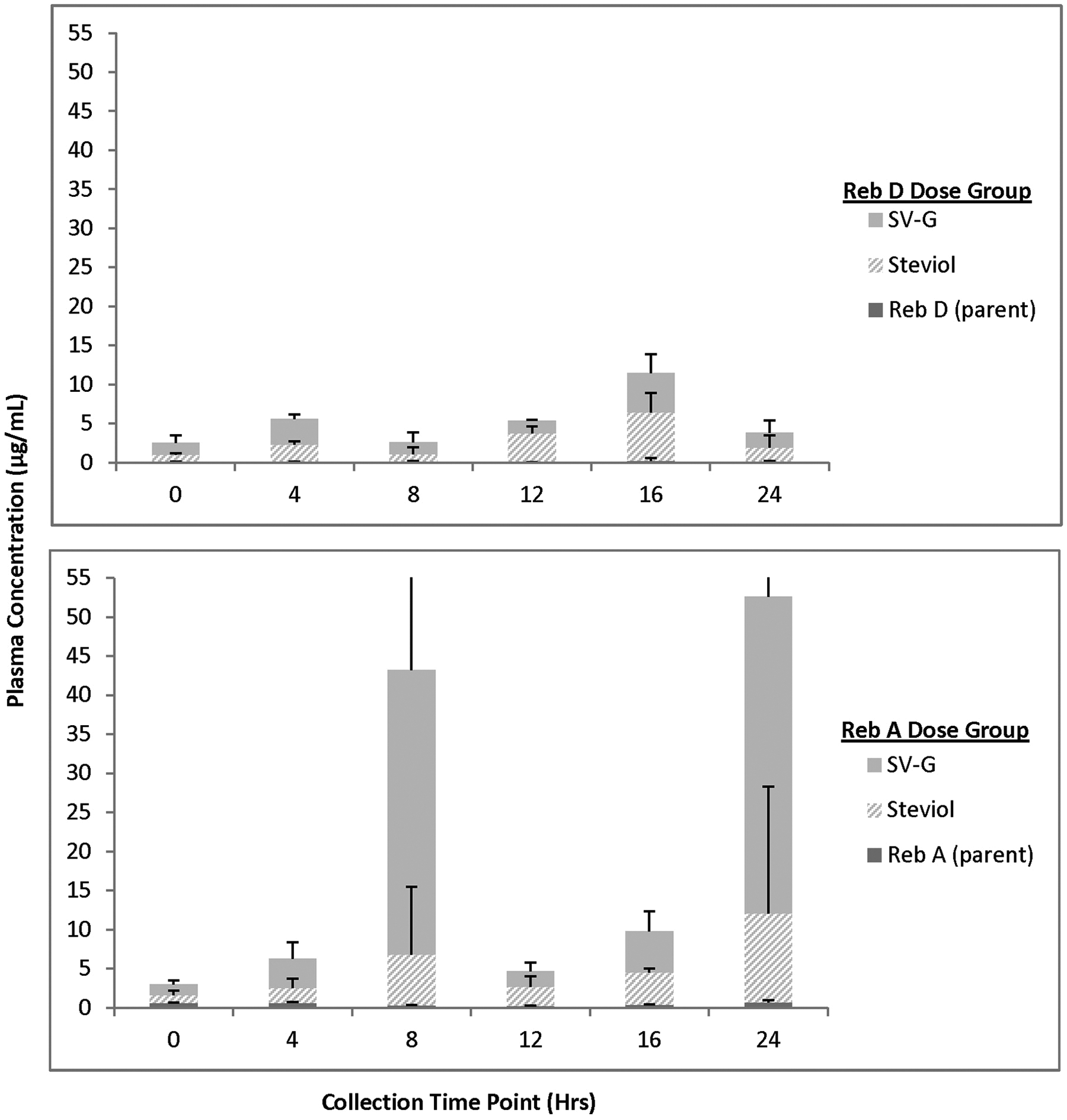
Mean (+standard deviation) plasma concentration of Rebaudioside (Reb) D, Reb A, and final hydrolysis products in female rats fed 2000 mg/kg/d Reb D or Reb A in the diet (day 1). Only positive standard deviation error bars shown for clarity of presentation, due to variability in individual animal data at select time points. N = 3 rats/sex/group at each time point (toxicokinetic phase animals) SV-G = steviol glucuronide.
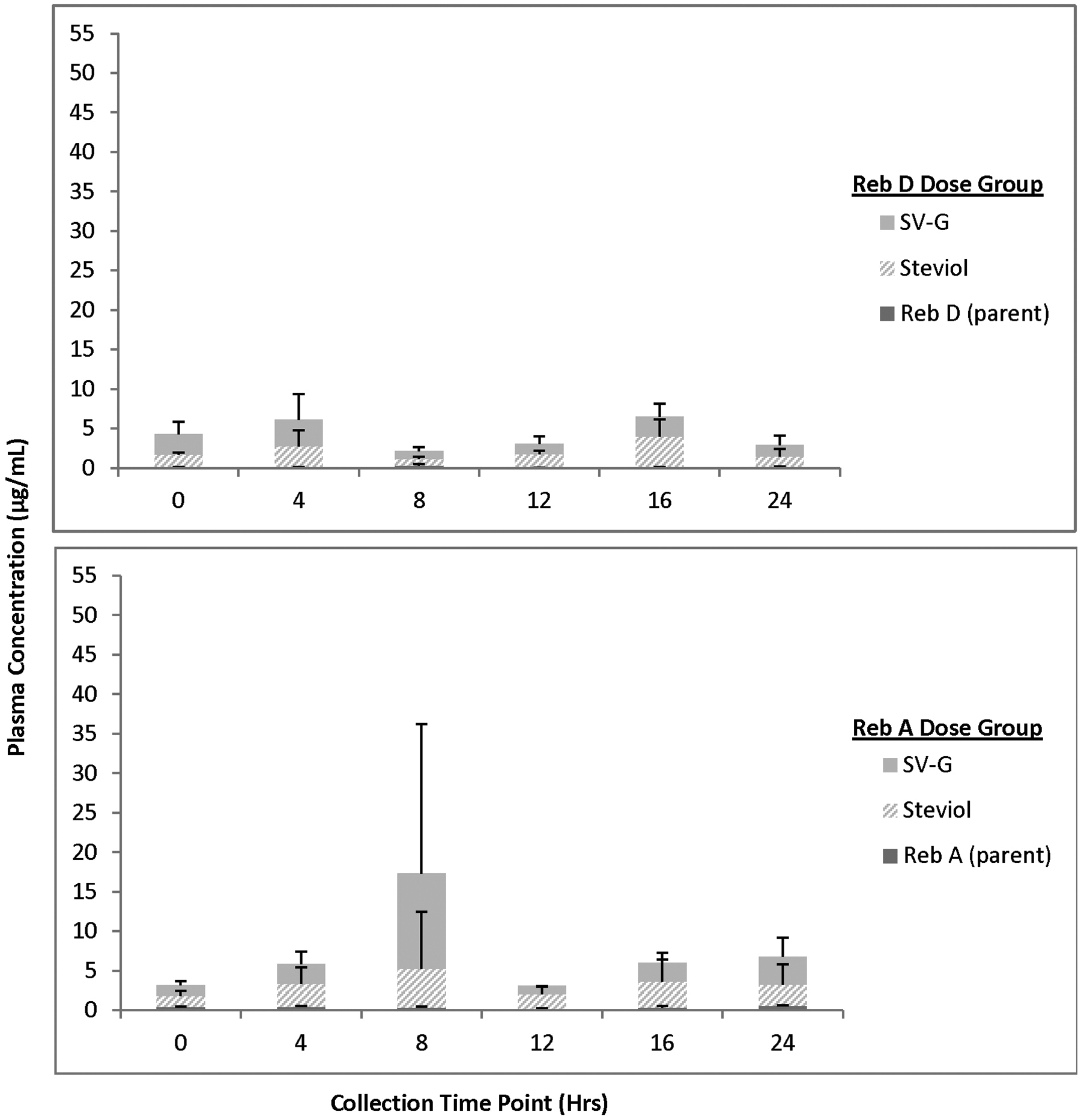
Mean (+standard deviation) plasma concentration of Rebaudioside (Reb) D, Reb A, and final hydrolysis products in female rats fed 2000 mg/kg/d Reb D or Reb A in the diet (day 21). Only positive standard deviation error bars shown for clarity of presentation, due to variability in individual animal data at select time points. N = 3 rats/sex/group at each time point (toxicokinetic phase animals); SV-G = steviol glucuronide.
Discussion
The present studies were conducted to compare the potential metabolism and toxicity profile of Reb D to that of a structurally related compound, Reb A, in order to verify the safety of Reb D for human dietary use in foods, and to confirm acceptability of the safety database for steviol glycosides to support the safety evaluation of Reb D.
In in vitro experiments, Reb A and Reb D solutions showed similar stability when exposed to simulated stomach and small intestine fluids, with susceptibility to hydrolytic degradation by enteric bacteria collected from the cecum. Incubations with rat liver microsomes indicated that neither compound is expected to be metabolized by liver enzymes.
The results of this in vitro study support the conclusion that steviol glycosides are metabolized via the same hydrolysis pathways, and that absorption of Reb D is expected to be minimal and similar to that of Reb A. Results of this in vitro metabolism study were corroborated by the results of plasma analysis from subgroups of toxicokinetic phase animals that were included in the 28-day dietary bridging toxicity study.
For animals receiving Reb D via high dietary concentrations for 28 days, plasma concentrations of Reb D, Reb A, and its final hydrolysis product, free/conjugated steviol, were measurable at low levels in all the samples on both sampling days with fluctuations among the time points related to typical feeding and sleeping cycles of rats. For animals receiving Reb A via high dietary concentrations for 28 days, plasma concentrations of Reb A and its final hydrolysis product, free/conjugated steviol, were measurable at low levels in all the samples on both sampling days, with similar fluctuations among the time points related to typical feeding and sleeping cycles of rats. Systemic exposure in plasma to the other Reb D or Reb A hydrolysis products was extremely low or below the levels of quantitation.
In the 28-day dietary bridging toxicity study, there were no treatment-related, toxicologically relevant effects noted in animals administered Reb D or Reb A. Based on the results of this study, Reb D offered in the diet to Crl:CD[SD] rats over a period of 28 days resulted in no observed adverse effect levels (NOAELs) of 2042 and 2016 mg/kg/d for males and females, respectively, the highest average dosage levels examined.
It has been reported that after oral administration, steviol glycosides are poorly absorbed in experimental animals and humans and excreted primarily in the feces as metabolites or parent material (GRN 252 4 ; GRN 253 5 ; JECFA 22 ). The principal steviol glycosides, Reb A and stevioside, are metabolized in experimental animals and humans by intestinal microflora via successive hydrolysis of glucose sugar moieties (Nikiforov and Eapen 8 ). However, this process does not appear to be efficient, since these substances are essentially noncaloric (Prakash et al. 25 ). Based on its chemical structure, Reb A may be hydrolyzed by intestinal microflora to Reb B or stevioside, and given a long enough residence time in the lower intestine, it could be theoretically hydrolyzed to the aglycone, steviol (Figure 2). As reviewed in Nikiforov and Eapen, 8 there are several published studies that address the metabolism of Reb A and its possible metabolite, stevioside, which are analogous and relevant to the safety assessment of Reb D (Gardana et al 26 ; Geuns et al 27 ; Koyama et al 28 ; Wingard et al 29 ).
The results obtained from toxicokinetic phase animals in the current 28-day dietary bridging toxicity study support the conclusion that Reb D follows the same hydrolysis pathways as Reb A and other steviol glycosides (see Figure 2). Because Reb D has 1 additional glucose unit compared to Reb A, oral administration of this steviol glycoside results in hydrolysis of the ester and ether linkages by bacteria in the cecum (Renwick and Tarka 30 ), converting Reb D preferentially to Reb A or Reb E (or possibly Reb B) and subsequent hydrolysis products, eventually ending with steviol that may be absorbed, conjugated, and excreted in urine (Roberts and Renwick 31 ; Wheeler et al 32 ). As such, the metabolism of Reb D is considered to be identical to that of Reb A but with a more prolonged hydrolysis pathway than Reb A, with low absorption efficiency by the intestinal tract, and excretion primarily in the feces. Therefore, the safety database that exists for Reb A and structurally related steviol glycosides (GRN 252 4 and GRN 253 5 ) is appropriate for toxicological read across to support the safety evaluation of Reb D.
These results demonstrated that a short-term repeated exposure bridging toxicity study is appropriate to permit the use of Reb A data to support the safety evaluation of Reb D. The NOAEL from the current 28-day toxicity study in rats is greater than 2000 mg/kg/d. The results of the present studies, as well as data available for Reb A and other structurally related steviol glycosides, verify the safety of Reb D for human dietary use in foods.
Footnotes
Acknowledgments
The authors thank Beth Chepote for her assistance in preparing this manuscript.
Authors’ Note
The animal studies conducted were selected in consultation with the US Food and Drug Administration (FDA) Toxicological Principles for the Safety Assessment of Food Ingredients as well as other internationally recognized standards and guidelines for determining the safety of a food additive intended for human consumption. The number of animals utilized for this research was the minimum required to achieve scientifically valid results and was consistent with guideline recommendations. Conduct of these studies was intended to permit the use of the existing safety database for Reb A and steviol glycosides in the safety evaluation of Reb D, therefore, eliminating the need for further animal testing with Reb D.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Nikiforov and Rihner were scientific consultants in the design, review, and audit of these studies. They derive no direct financial benefit from the results of this research. Eapen and Thomas are employees of WIL Research, which received funding to conduct these studies. WIL derives no direct financial benefit from the results of this research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. Funding for this research was provided by PepsiCo Inc.
