Abstract
The Cosmetic Ingredient Review Expert Panel assessed the safety of triethanolamine (TEA) and 31 related TEA-containing ingredients as used in cosmetics. The TEA is reported to function as a surfactant or pH adjuster; the related TEA-containing ingredients included in this safety assessment are reported to function as surfactants and hair- or skin-conditioning agents. The exception is TEA-sorbate, which is reported to function as a preservative. The Panel reviewed the available animal and clinical data. Although data were not available for all the ingredients, the panel relied on the information available for TEA in conjunction with previous safety assessments of components of TEA-containing ingredients. These data could be extrapolated to support the safety of all included ingredients. The panel concluded that TEA and related TEA-containing ingredients named in this report are safe as used when formulated to be nonirritating. These ingredients should not be used in cosmetic products in which N-nitroso compounds can be formed.
Keywords
Introduction
In 1983, the Cosmetic Ingredient Review (CIR) Expert Panel issued a report on the safety of triethanolamine (TEA), diethanolamine, and monoethanolamine. In 2010, the panel decided to reopen that safety assessment as 3 separate reports and to include related ingredients in each of the new reviews. This assessment addresses TEA and the following 31 related TEA-containing ingredients:
Inorganic salts
TEA-hydrochloride TEA-sulfate
Organic acid salts
TEA-laurate TEA-laurate/myristate TEA-myristate TEA-palmitate TEA-stearate* TEA-isostearate TEA-undecylenate TEA-sorbate TEA-oleate TEA-canolate TEA-cocoate TEA-hydrogenated cocoate TEA-tallate TEA-glyceryl dimaleate
Hydroxy acid salts
TEA-lactate*
Organo-substituted sulfates
Magnesium/TEA-coco-sulfate Sodium/TEA C12-13 pareth-3 sulfate TEA-lauryl sulfate* TEA-laureth sulfate TEA-oleyl sulfate TEA-C10-15 alkyl sulfate TEA-C11-15 alkyl sulfate TEA-C12-13 alkyl sulfate TEA-C12-14 alkyl sulfate TEA-C12-15 alkyl sulfate TEA-coco-sulfate TEA-C11-15 pareth sulfate TEA-C12-13 pareth-3 sulfate TEA-PEG-3 cocamide sulfate
The ingredients marked with an asterisk have been reviewed previously by the CIR, and the conclusions on safety of these ingredients are provided in Table 1. The safety of many of the “components” of these ingredients has been reviewed by the CIR, and these conclusions are also provided in Table 1.
Conclusions of Previously Reviewed Ingredients and Components.
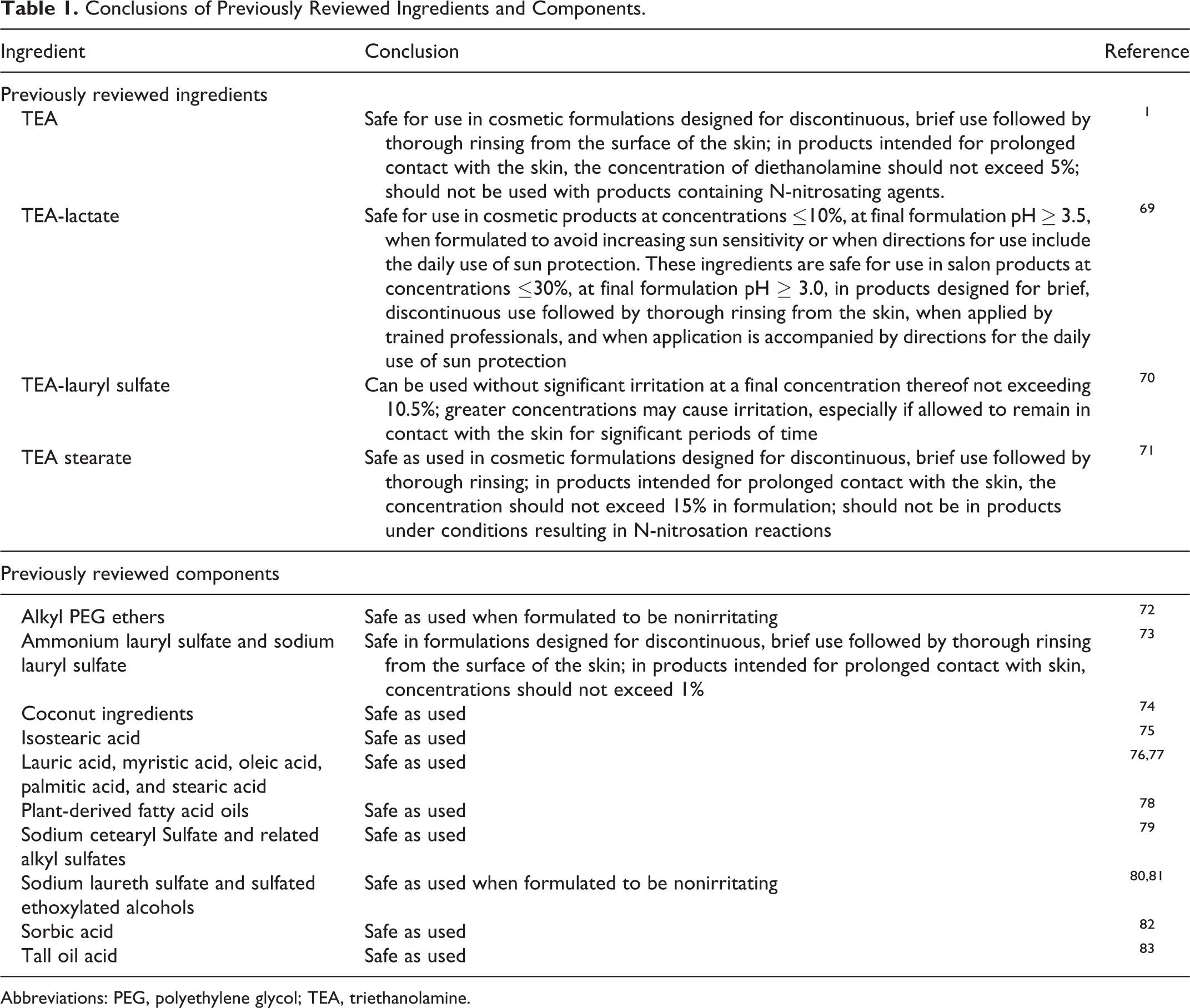
Abbreviations: PEG, polyethylene glycol; TEA, triethanolamine.
The TEA, an ingredient reported to function as a surfactant or pH adjuster in cosmetic products, had been reviewed previously by the CIR Expert Panel. In 1983, the Expert Panel concluded that TEA is safe for use in cosmetic formulations designed for discontinuous, brief use followed by thorough rinsing from the surface of the skin. 1 In products intended for prolonged contact with the skin, the concentration of TEA should not exceed 5%. The TEA should not be used in products containing N-nitrosating agents. In the 1983 assessment, data demonstrated that TEA was a mild skin and eye irritant and that irritation increased with increasing ingredient concentration.
The TEA-containing ingredients are reported to function in cosmetics as surfactants, skin-conditioning agents, or hair-conditioning agents. The TEA-sorbate is reported to function only as a preservative.
Chemistry
As given in CIR’s earlier safety assessment of TEA, 1 this ingredient is an amino alcohol commercially produced by aminating ethylene oxide with ammonia. The replacement of 3 hydrogens of ammonia with ethanol groups produces TEA (Figure 1). The TEA contains small amounts of diethanolamine and ethanolamine. The TEA is reactive and bifunctional, combining the properties of alcohols and amines. The reaction of ethanolamines and sulfuric acid produces sulfates. The TEA can act as an antioxidant against the autoxidation of fats of both animal and vegetable origin.
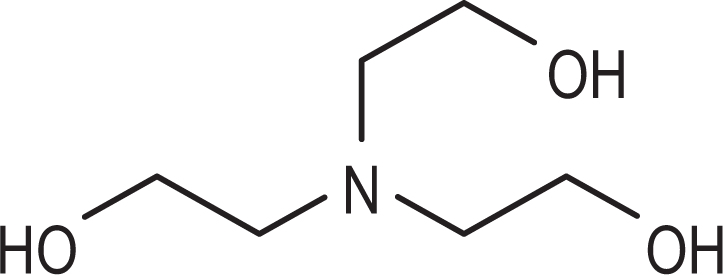
Triethanolamine.
Of concern in cosmetics is the conversion (N-nitrosation) of secondary amines (R 1 -NH-R 2 ), such as diethanolamine (wherein R 1 and R 2 are each ethanol), into N-nitrosamines that may be carcinogenic. Tertiary alkyl amines (NR 1 R 2 R 3 ), such as TEA (wherein R 1 , R 2 , and R 3 are each ethanol), however, do not tend to react with N-nitrosating agents to directly form nitrosamines. However, tertiary amines can act as precursors in nitrosamine formation by undergoing nitrosative cleavage (eg, 1 ethanol functional group can be cleaved off from TEA to generate diethanolamine). 2 The resultant secondary amine (ie, diethanolamine) can then be N-nitrosated (ie, to N-nitrosodiethanolamine [NDELA]). Accordingly, TEA can react, in a formulation or in vivo, with nitrites or oxides of nitrogen to form a nitrosamine. Nitrous anhydride is the oxide of nitrogen that most commonly initiates nitrosation in vivo. 3 –6
Acid Salts
The acid salts (inorganic salts, organic acid salts, and hydroxy acid salts), mentioned previously, are ion pairs that freely dissociate in water (eg, Figure 2). Therefore, these salts are closely related to the corresponding free acids and TEA. In other words, TEA stearate is closely related to stearic acid and TEA.
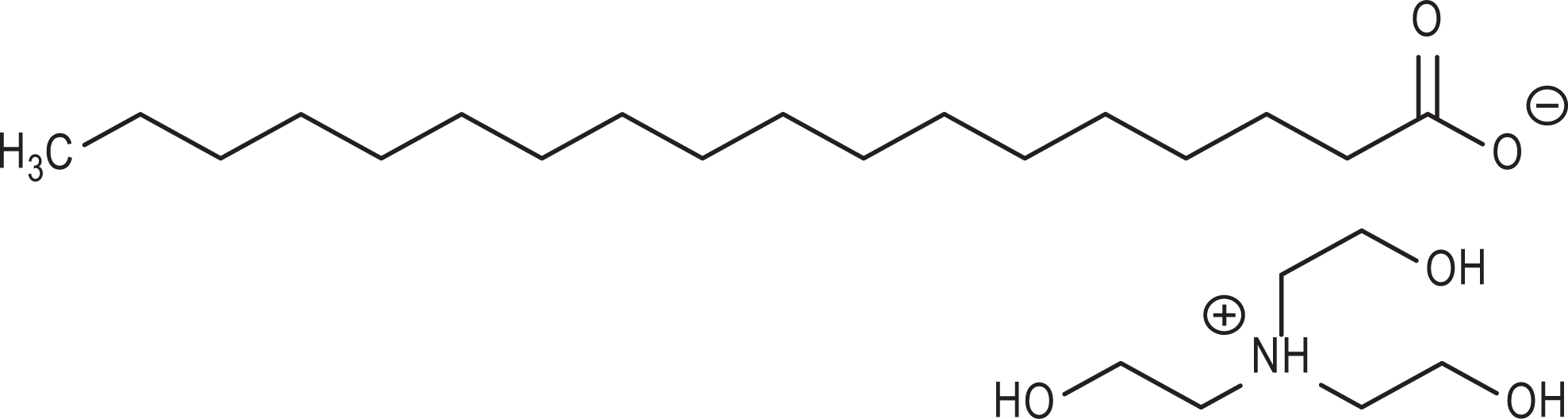
Triethanolamine (TEA) stearate.
Organo-Substituted Sulfates
The sulfates consist of organic acid salts that have the additional functional group of sulfate. For example, TEA lauryl sulfate is a 12-carbon alkyl chain (ie, lauryl) bonded to a sulfate anion, balanced with a triethanolammonium cation (Figure 3).
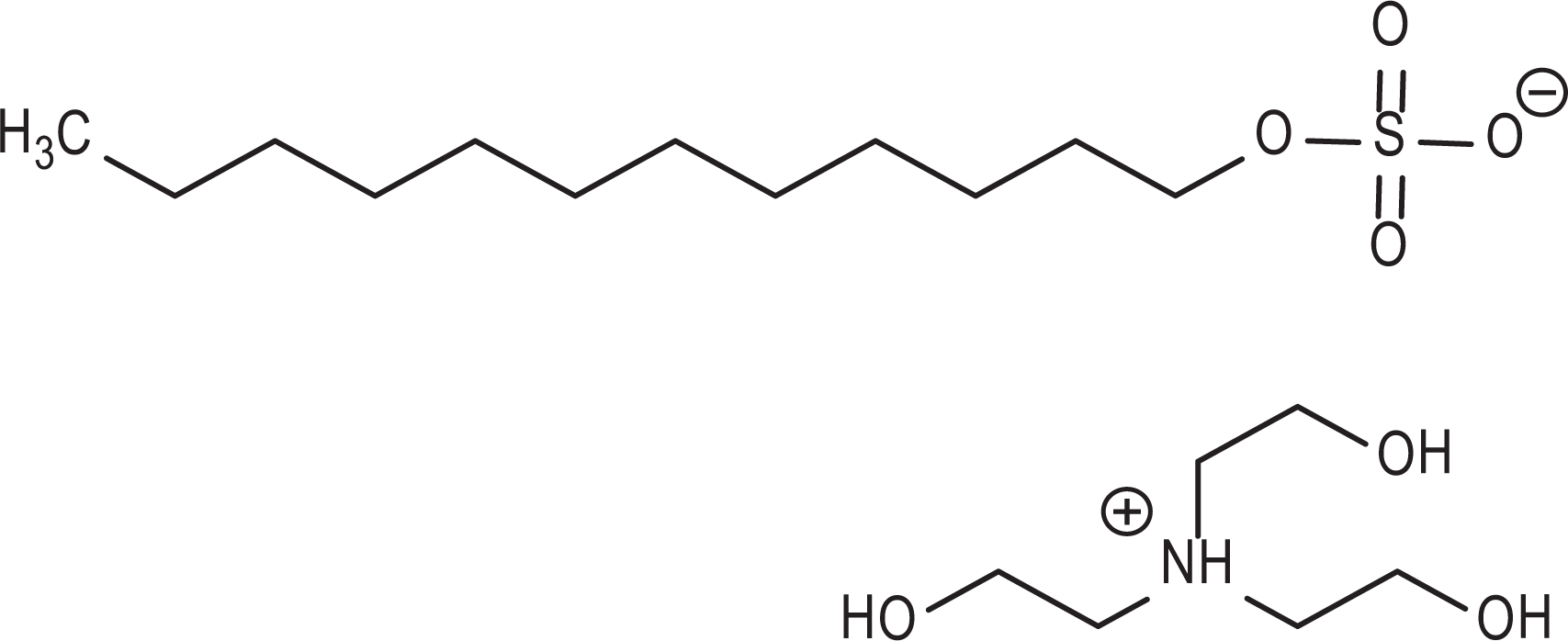
Triethanolamine (TEA)-lauryl sulfate.
Definition and Structure
The definitions and structures of TEA and TEA-containing ingredients are provided in Table 2. Chemical and physical properties are described in Table 3.
Definitions and Structures.
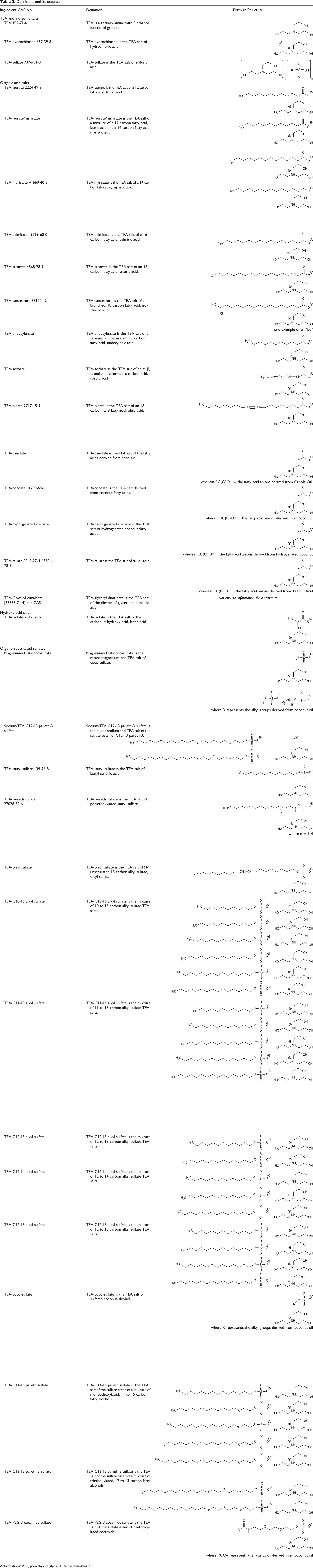
Abbreviations: PEG, polyethylene glycol; TEA, triethanolamine.
Physical and Chemical Properties.

Abbreviation: TEA, triethanolamine.
Method of Manufacture
Triethanolamine
The TEA is produced by reacting 3 moles of ethylene oxide with 1 mole of ammonia; additional ethylene oxide will continue to react to produce higher ethylene oxide adducts of TEA. 7 Typically, ethylene oxide is reacted with ammonia in a batch process to produce a crude mixture of approximately one-third of each ethanolamine, diethanolamine, and TEA. The crude mixture is later separated by distillation.
TEA-stearate
TEA-stearate was produced by mixing partially neutralized stearic acid and TEA at temperatures above 80°C and then cooling. 8 It was determined that the acid–soap complex at a 2:1 fixed stoichiometric ratio was formed between TEA stearate and stearic acid.
TEA-lauryl sulfate
As given in the original CIR safety assessment of TEA-lauryl sulfate, 9 this ingredient is manufactured by neutralizing lauryl sulfuric acid with aqueous (aq) TEA.
Commercial alkyl sulfates are produced by the sulfation of primary alcohols using sulfur trioxide or chlorosulfonic acid followed by neutralization with a base to produce the corresponding salt. 9
Stability
TEA-stearate
TEA stearate was produced as described previously. On cooling, a lamellar gel phase formed. 10 Solid crystals were observed after 5 hours. Upon storage, the sample gradually separated into 2 phases; after a few weeks, a separate liquid phase and a solid-pearly crystalline phase were observed.
Impurities
Triethanolamine
Based on the unpublished survey data collected by the Food and Drug Administration (FDA), a diethanolamine impurity level of 0.3% was found in TEA samples. 11 (Additional details were not provided.)
TEA-lauryl sulfate
As previously reported by the CIR, 9 impurities in TEA-lauryl sulfate may include TEA, TEA-sulfate, unsulfated alcohol, TEA chloride, and formaldehyde (some grades).
Sodium sulfate and residual alcohols may be present as impurities in commercial alkyl sulfate products. 9 Typically, industrial alkyl sulfates contain 1% to 4% sodium sulfate and 0.5% to 18% residual alcohol.
N-Nitrosodiethanolamine Formation
Nitrosamines are compounds containing the R 1 R 2 N-NO functional group. 2 N-nitrosation is the process of converting organic compounds (eg, alkyl amines) into N-nitroso derivatives (eg, nitrosamines) by reaction with nitrosating agents. These agents include nitrous acid (HNO2), oxides of nitrogen (eg, nitrous anhydride or nitrite), and other compounds capable of generating a nitrosonium ion, NO2+.
The formation of a specific nitrosamine, NDELA, from reaction of TEA with nitrite was examined in vitro and in vivo. 12 The TEA used in these studies had an impurity content of 0.4% diethanolamine. In an aq matrix, approximately 3% TEA was converted to NDELA at a pH of 4.0 in the presence of acetic acid. At the same pH, in the presence of sulfuric or hydrochloric acid, only about 1% of the TEA was nitrosated. At pH 7, the greatest nitrosation to NDELA, 0.5%, occurred in the presence of sulfuric acid. No conversion of TEA to NDELA was detected at pH 2 or 10. In nutrient broth cultures (neutral pH), 0.08% and 0.68% of the TEA were nitrosated to NDELA in a diluted (high-cecal inoculum) and full-strength (low-cecal inoculum) media. (The percentage of nitrosation was determined using values that were corrected for diethanolamine impurity-related NDELA formation.)
In vivo, female B6C3F1 mice were dosed dermally or orally with 1000 mg/kg TEA, in conjunction with oral exposure to sodium nitrite. 12 Following 7 days of dermal dosing, no NDELA was detected in the blood, ingesta, or urine of test, vehicle control, or sodium nitrite control mice. (The limits of detection for the blood, ingesta, and urine were 0.001, 0.006, and 0.47 µg/mL, respectively.) With a single oral dose, the concentrations of NDELA found in the blood and ingesta of mice 2 hours postdosing were 0.001 ± 0.0005 and 0.044 ± 0.059 µg/g, respectively.
Use
Cosmetic
The TEA is reported to function in cosmetics as a surfactant or pH adjuster, and it can be used in fragrances. 13 Most of the other TEA ingredients are reported to function in cosmetics as surfactants, skin-conditioning agents, or hair-conditioning agents. TEA-sorbate is reported to function only as a preservative.
Voluntary Cosmetic Registration Program (VCRP) data obtained from the FDA in 2011 indicate that TEA is used in 3756 formulations; 3034 of those products are leave-on formulations, and 3106 formulations involve dermal exposure. 14 According to the data submitted by industry in response to a survey conducted by the Personal Care Products Council (Council), TEA is used at concentrations of 0.0002% to 19%. 15 In leave-on products, the reported use concentrations range from 0.0002% to 6%. With the exception of TEA-lauryl sulfate (302 uses) and TEA-stearate (130 uses), all other in-use TEA-containing ingredients had less than 20 reported uses. 14 TEA-lauryl sulfate had the highest concentrations of use, with ≤40% being reported for rinse-off and ≤8% for leave-on formulations. 16 The available use data on all in-use ingredients are provided in Table 4. Ingredients not reported to be in use, according to the VCRP data and the Council survey, are listed in Table 5.
Frequency and Concentration of Use According to the Duration and Type of Exposure.
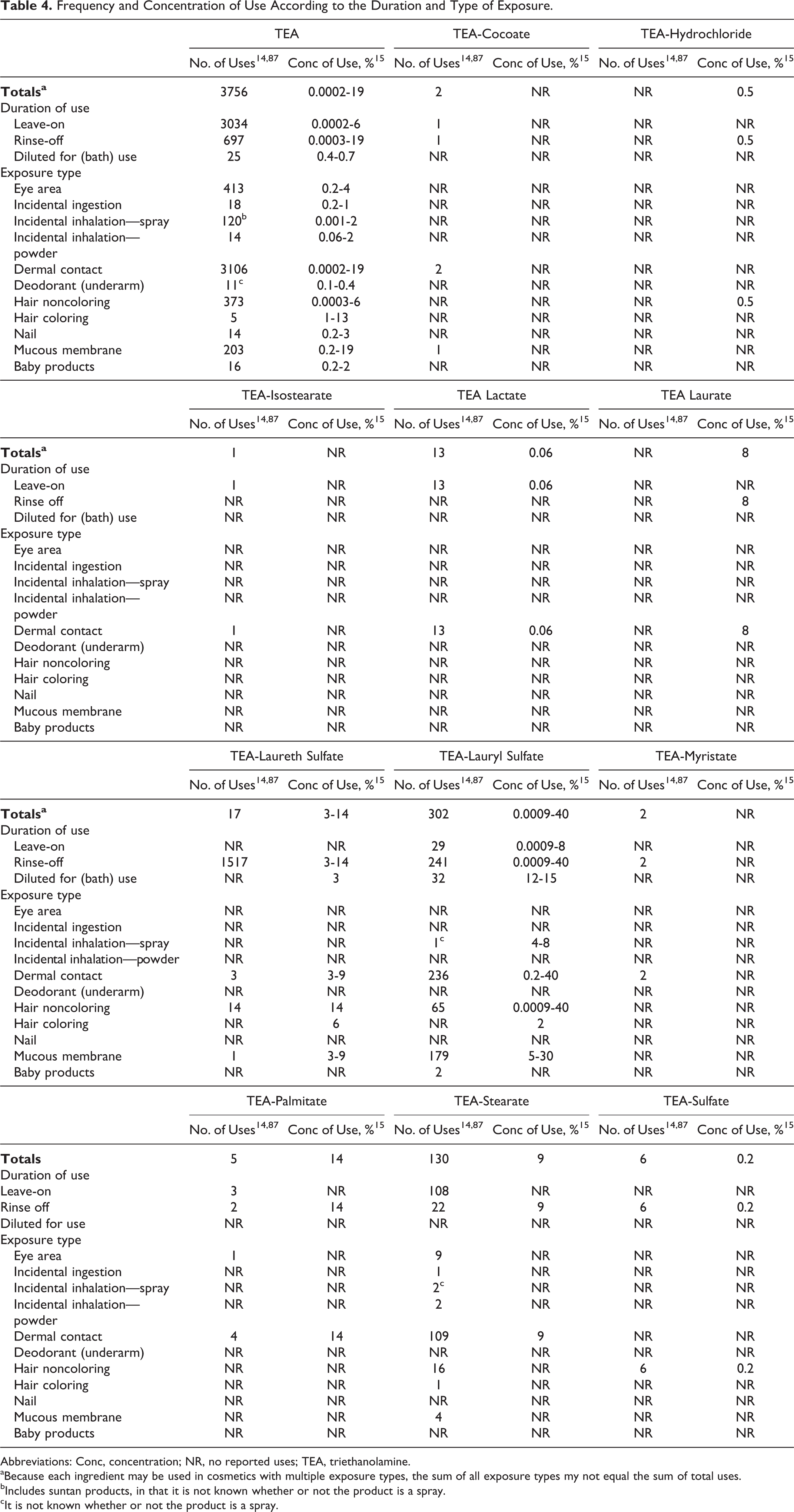
Abbreviations: Conc, concentration; NR, no reported uses; TEA, triethanolamine.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types my not equal the sum of total uses.
bIncludes suntan products, in that it is not known whether or not the product is a spray.
cIt is not known whether or not the product is a spray.
Ingredients Not Reported to be Used.
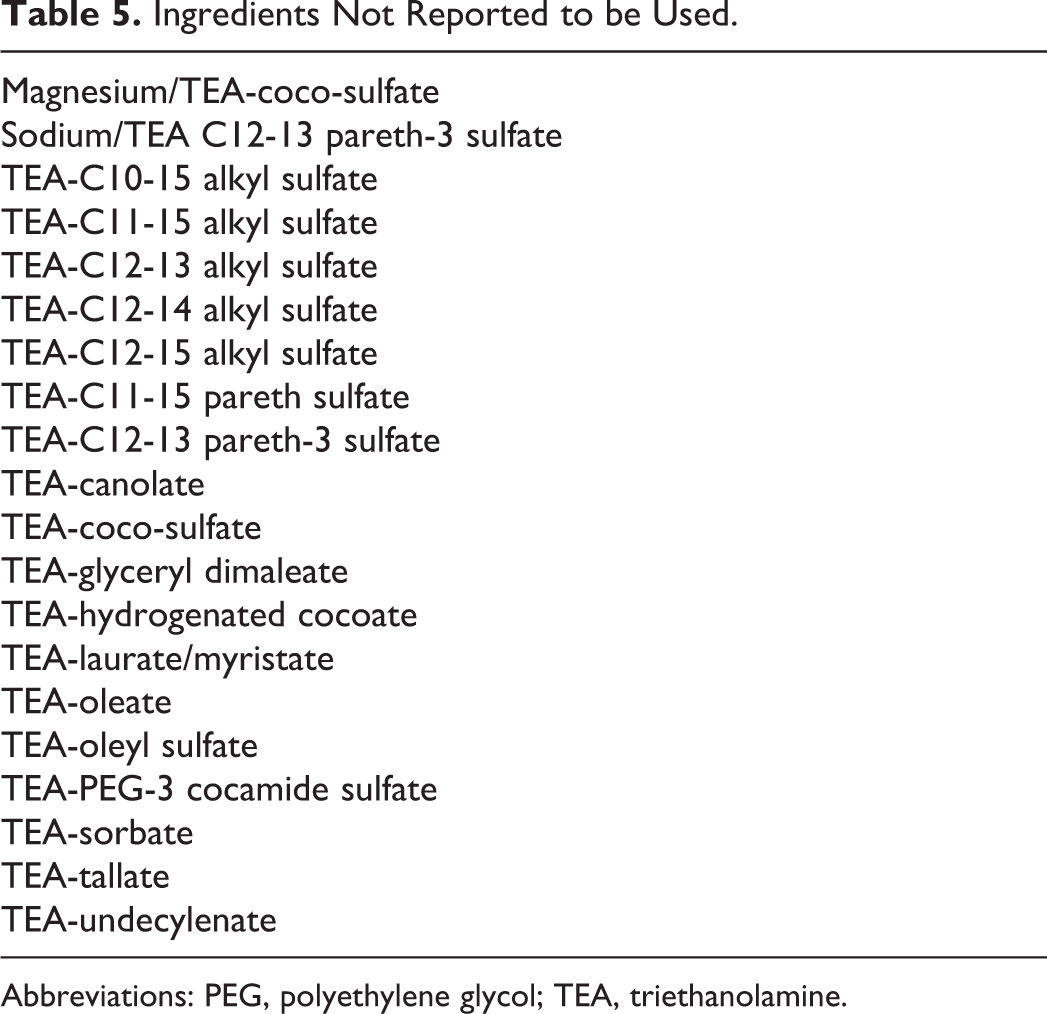
Abbreviations: PEG, polyethylene glycol; TEA, triethanolamine.
Typical exposure to TEA was calculated. Using an evolutionary computation algorithm method, 17 the dermal exposure of consumers to TEA in an eye make-up powder is 0.0125 mg/kg body weight (bw)/d and to TEA in a body lotion is 6.25 mg/kg bw/d. Using a DERMAL program method, the dermal potential dose rate of a bar soap containing 2.5% TEA is 5.182 mg/d.
Some of the ingredients reviewed in this assessment may be applied to baby skin, used near the eye area or mucous membranes, or could possibly be ingested. The TEA, TEA-lauryl sulfate, and TEA-stearate are reported to be in products that could be inhaled. In practice, 95% to 99% of the aerosols released from cosmetic sprays have aerodynamic equivalent diameters in the 10 to 110 µm range. 18,19 Therefore, most aerosols incidentally inhaled from these sprays are deposited in the nasopharyngeal region and are not respirable. 20,21 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic diameters in the range considered to be respirable. 21 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Internationally, data were available from the European Commission (EC) and Canada. In the EC, Annex III Part 1 of the CosIng database, which is the list of substances that cosmetic products must not contain except subject to the restrictions and conditions laid down, states that trialkylamines, trialkanolamines, and their salts are allowed at concentrations of up to 2.5% in nonrinse-off products; “other” product types do not have a concentration limit for TEA. 22 However, the following additional limitations apply to leave-on and rinse-off products containing TEA: do not use with nitrosating systems; minimum allowable purity is 99%; maximum allowable secondary amine content is 0.5% in raw material; maximum allowable nitrosamine content is 50 μg/kg; and must be kept in nitrite-free containers. Additional relevant information from the EC is that sorbic acid and its salts and undec-10-enoic acid and its salts, including their ethanolamine salts, are listed by the EC for use as preservatives, with maximum concentrations of use of 0.6% and 0.2%, respectively, based on the free acid. 23
According to the data obtained from Health Canada, some leave-on-type products reportedly use TEA as high as 10% and 30%, with some reporting concentration ranges of 30% to 100% (Health Canada, personal communication).
Noncosmetic
The TEA is used in the manufacture of emulsifiers and dispersing agents for textile specialties, agricultural chemicals, waxes, mineral and vegetable oils, paraffin, polishes, cutting oils, petroleum demulsifiers, and cement additives. 1 It is an intermediate for resins, plasticizers, and rubber chemicals. It is used as a lubricant in the textile industry, as a humectant and softening agent for hides, as an alkalizing agent and surfactant in pharmaceuticals, as an absorbent for acid gases, and in organic syntheses.
The TEA, TEA-myristate, TEA-palmitate, TEA-oleate, TEA-cocoate, and TEA-tallate have been used as an indirect food additive. 24 The TEA is used as a rust inhibitor in water-based metalworking fluids. 25
Toxicokinetics
Dermal
In vitro
Triethanolamine and TEA-stearate
The penetration of oil-in-water (o/w) emulsions containing TEA was determined in vitro using human skin samples. 26 Emulsions were prepared using 1% TEA and 5% stearic acid and using 5% TEA and 10.5% stearic acid; the pH values of these emulsions were 8.0 and 8.2, respectively. Because the pH values of commercial lotions containing TEA were reported to be ∼7.0, emulsions were also prepared with the pH adjusted to 7.0. The test samples were applied to the skin for 24 hours at a concentration of 3 mg/cm2, and the area of the exposed skin was 0.64 cm2. Penetration and absorption were measured at 24 hours using the emulsions with a pH of ∼8 and at 24 and 72 hours using the emulsions with a pH of 7.0. The 24-hour skin sample was tape stripped, while the 72 hours sample was not.
Using the emulsions with a pH of ∼8, there was no statistically significant difference in penetration between a 1% and a 5% TEA emulsion. Using the emulsion with a pH of 7 and concentration of 1% TEA, there was no statistically significant difference in penetration observed when comparing the 24 and 72 hours values. There was a statistically significant difference in the total recovery of TEA using the 5% emulsion, pH 7, with the recovery at 24 hours less than that recovered at 72 hours.
The authors suggested that all the TEA in a TEA/stearic acid emulsion existed as the TEA stearate salt, since an excess of stearic acid was used. Radiolabeled TEA stearate was prepared by mixing stearic acid with a radiotracer dose of 0.64 µCi [ 14 C]triethanolamine (5 µg TEA) in an o/w emulsion; the ratio of TEA to stearic acid was 1:4. Using this emulsion, at pH 8.2, 50.1% of the applied dose of TEA-stearate was found in the skin, and 0.51% in the receptor fluid after 24 hours. With pH 7.0, 29.1% of the applied dose was found in the skin and 0.46% in the receptor fluid after 24 hours. The authors speculated that the penetration rate of the TEA molecule was dependent on the penetration of its salts.
Non-human
Triethanolamine
The blood kinetics and absorption, distribution, metabolism, and excretion (ADME) of [ 14 C]triethanolamine were determined following dermal application of 2000 mg/kg neat [ 14 C]triethanolamine without occlusion to 24 male C3H/HeJ mice and with occlusion to 3 male mice. 27 (Nonradiolabeled TEA was 99.6% pure; radiochemical purity was 98.6%.) The TEA was extensively and rapidly absorbed following a single open application of 2000 mg/kg neat [ 14 C]triethanolamine. The majority of the radioactivity, 49% to 62% of the total dose (∼58%-72% of the absorbed dose), was excreted in the urine, primarily as unmetabolized TEA. Diethanolamine and ethanolamine were not detected in the urine. Approximately, 18% to 28% of the total dose (∼20%-32% of the absorbed dose) was excreted in the feces. The amount of radioactivity remaining in the body after 48 hours ranged from 3.3% to 6.1%, and the amount recovered at the application site ranged from 1.2% to 2.1% for the open application and 6% to 11% for the occluded application.
The National Toxicology Program (NTP) examined the ADME of TEA following dermal administration to B6C3F1 mice and F344 rats. 28 Groups of 4 female mice were given a single dose of 79 or 1120 mg/kg [ 14 C]triethanolamine in acetone; the dose contained 12 to 15 µCi, with the appropriate amount of nonlabeled TEA in a volume of 190 µL/dose. (Radiochemical purity of [ 14 C]triethanolamine was 97%; the purity of nonlabeled TEA was confirmed, but the purity was not stated.) The dose was applied to a 1.44 cm2 area of clipped skin, and a nonocclusive cover was used. Approximately 60% to 80% of the dose was absorbed, and absorption increased with increasing dose. In the urine, 22.5% to 27.5% and 48% to 56% of the dose were recovered after 24 and 72 hours, respectively, and TEA was excreted mostly unchanged. Approximately 5% to 9% and 8% to 13% of the dose were recovered in the feces at the same time periods.
Groups of 4 female rats were given a single dermal dose of 68 or 276 mg/kg [ 14 C]triethanolamine in acetone; the dose contained 65 µCi, with the appropriate amount of nonlabeled TEA in a volume of 190 µL/dose. The dose was applied to a 12 cm2 area of clipped skin, and a nonocclusive cover was used. Only 19% to 28% of the dose was absorbed over 72 hours; absorption increased with increasing dose but not significantly. In the urine, 13% to 24% of the dose was recovered in 72 hours as mostly unchanged TEA. The amount recovered in the feces after 72 hours was <0.25%. Very little radioactivity, <1%, was present in the tissues; a number of tissues had elevated concentrations of radiolabel relative to blood.
Oral
Non-human
Triethanolamine
The TEA (purity not specified) was administered orally to male and female rats as a single dose or as a repeated dose for 5 to 6 days. 29 (Dosing details were not described.) At 24 hours after administration of the single dose, the excretion ratio of unchanged TEA in the urine and feces was 53% and 20% of the dose, respectively. With repeated administration, the excretion ratio/day remained constant. Gender did not affect the ratios. TEA glucuronide was detected but in a very small amount (actual concentration not specified). The TEA was rapidly absorbed in the gastrointestinal tract and excreted mostly in the urine in unchanged form.
Other
Non-human
Triethanolamine
A group of 27 male C3H/HeJ mice was given an intravenous (iv) injection of 1 mg/kg [ 14 C]triethanolamine as an aq solution (0.5 mg/mL), and the dose volume was 2 mL/kg. 27 (Nonradiolabeled TEA was 99.6% pure; radiochemical purity was 98.6%.) Radioactivity in the blood declined in a biphasic exponential manner for 24 hours, with a relatively rapid initial phase of [ 14 C] elimination followed by a slower terminal phase. The majority of the radioactivity, approximately 69%, was excreted in the urine, primarily as unmetabolized TEA. Diethanolamine and ethanolamine were not detected in the urine. Some of the radioactivity, ∼17%, was excreted in the feces. The average amount of radioactivity recovered in the tissues after 24 hours was 3.1%.
The NTP examined the ADME of TEA following iv administration to B6C3F1 mice and F344 rats. 28 Groups of 4 female mice and 4 female rats were given a single iv dose of 3 mg/kg [ 14 C]triethanolamine in isotonic saline. For mice, the dose contained 6 µCi, with the appropriate amount of nonlabeled TEA, for a dosing volume of 2 mL/kg. (Radiochemical purity of [ 14 C]triethanolamine was 97%; the purity of nonlabeled TEA was confirmed, but the purity was not stated.) At 24 hours, 26% and 14% of the dose were excreted in the urine and feces, respectively, while at 72 hours, these values were 62% and 28%, respectively. The TEA was excreted mostly unchanged. Little, <0.5%, was detected in expired carbon dioxide. For rats, the dose contained 47 µCi, with the appropriate amount of nonlabeled TEA, for a dosing volume of 1 mL/kg. Much more of the radioactivity was excreted in the urine of rats compared to mice, and excretion was more rapid. Approximately 90% of the dose was recovered in the urine in 24 hours, and 98% in 72 hours, mostly as unchanged TEA. Similar to mice, <0.5% was detected in expired carbon dioxide. Only 0.9% of the radioactivity was detected in the tissues after 72 hours.
Toxicological Studies
Acute (Single) Dose Toxicity
Dermal
Triethanolamine
As reported previously, 1 the acute dermal toxicity of TEA was examined using groups of 6 rabbits. Undiluted TEA, 91.8% and 88.1% active, was applied to the intact and abraded skin of 3 rabbits under a 24 hours occlusive patch. The exposure to actual TEA was 2 g/kg. None of the animals died. Mild erythema and edema were reported at 24 hours.
Oral
Triethanolamine
As reported previously, 1 the acute oral toxicity of TEA was determined using guinea pigs and rats. In guinea pigs, undiluted TEA has an median lethal dose (LD50) of 8 g/kg, and the LD50 of TEA in a gum arabic solution was between 1.4 and 7.0 g/kg. Using rats, the oral LD50 of undiluted TEA ranged from 4.19 to 11.26 g/kg. The purity ranged from 78.6% TEA (with 8.6% diethanolamine and 1.7% ethanolamine) to unspecified high purity.
TEA-lauryl sulfate
As reported previously, 9 TEA-lauryl sulfate was moderately to slightly toxic in acute oral studies with rats, LD50 values ranged from 0.27 to >1.95 g/kg. In a more recent study, the oral LD50 of TEA-lauryl sulfate in male and female Wistar rats was >2 g/kg. 30
Other
Triethanolamine
As reported previously, 1 the intraperitoneal LD50 of TEA was 1.45 g/kg for mice.
Repeated Dose Toxicity
Dermal
Triethanolamine
As given in the original CIR safety assessment of TEA, 1 toxicity was reported in a closed-patch continuous exposure test using 10 guinea pigs in which commercial and high-purity TEA, 8 g/kg, was applied daily 5 d/wk. All guinea pigs died by the 17th application; adrenal, pulmonary, hepatic, and renal damage were observed. In a 13-week study, 1 mg/kg of a hair dye formulation containing 0.1% to 0.15% or 1.5% TEA was applied to the backs of 12 rabbits for 1 hour, twice weekly. The test site skin was abraded for half of the animals. No systemic toxicity was observed, and there was no histomorphologic evidence of toxicity. In a 6-month study in which TEA was applied caudally to rats for 1 h/d, 5 days/wk, no toxic effects were observed with a 6.5% solution. However, using a 13% solution, changes (not specified) were seen in liver and central nervous system function. The addition of 1.4 mg/L TEA to the drinking water of the rats dosed dermally with 13% TEA did not increase the toxic effects.
In a 2-week study, undiluted TEA (purity not specified) was applied dermally to B6C3F1 mice and F344 rats, 5 days/wk. 31 Dose levels of TEA were 0.21, 0.43, 0.84, 1.69, and 3.37 g/kg for the mice and 0.14, 0.28, 0.56, 1.13, and 2.25 g/kg for rats. Chronic active necrotizing inflammation of the skin at the application site occurred at a greater frequency and severity in rats than in mice. No renal or hepatic lesions were detected with either species.
In a preliminary study, 50 µL of 1% to 100% TEA was applied to the backs of male C3H mice, 5 days/wk for 2 weeks, and mild epidermal hyperplasia was observed in mice treated with 25%, 50%, or 100% TEA. 32 Based on these results, a 13-week study was completed in which 50 µL of 0%, 10%, 33%, or 100% TEA (99.3% pure) in acetone was applied 3×/wk to a clipped site on the backs of 15 male and female C3H/HeJ mice. (The approximate daily doses were 0, 0.14, 0.46, or 2.0 g/kg for males, respectively, and 0, 0.16, 0.54, or 2.3 g/kg for females, respectively.) No treatment-related clinical signs of toxicity, skin irritation, or mortality were observed during the study. A mild epidermal hyperplasia was observed in treated male and female mice at all 3 concentrations tested, and this reaction was considered to be evidence of mild irritation associated with repeated application of TEA.
In a 13-week NTP dermal study using male and female B6C3F1 mice, application of 250, 500, 1000, or 2000 mg/kg bw TEA in acetone or 4000 mg/kg neat resulted in decreased mean body weights and body weight gains for some male mice. 33 (Purity of TEA was 99%. Functional group titration indicated <0.4% ethanolamine or diethanolamine.) Irritation was observed for the highest dose group. Microscopically, inflammation was observed for this dose group and acanthosis was noted for all dose groups, with severity increasing with dose. Absolute kidney and liver weights of males and females of the 4000 mg/kg group and relative kidney to body weights of males dosed with ≥1000 mg/kg were increased compared to controls. Absolute and relative spleen weights were also significantly increased in high dose female mice compared to controls.
In a 13-week dermal study using male and female F344/N rats, application of 125, 250, 200, or 1000 mg/kg body weight TEA (99% pure) in acetone or 2000 mg/kg neat resulted in significant decrease in mean body weights and body weight gains in the high-dose animals. 33 (Functional group titration indicated <0.4% ethanolamine or diethanolamine present.) Irritation was observed at the application site. Microscopic lesions included acanthosis and inflammation. Kidney weights of males and females dosed with ≥500 mg/kg were increased compared to controls, and dosed females, but not males, had greater incidences of nephropathy, when compared to controls.
TEA-lauryl sulfate
As reported previously, 9 in a 28-day dermal study, application of a diluted shampoo containing 1% TEA-lauryl sulfate to rabbit skin caused erythema, edema, wrinkling, eschar formation, and severe desquamation. In a 13-week dermal study in rabbits with a diluted shampoo containing 2.4% TEA-lauryl sulfate, mild erythema and dryness were observed.
Oral
Triethanolamine
As reported previously, 1 oral studies were conducted in which groups of 8 to 20 rats were dosed with 0.2 to 2.61 g/kg/d TEA for 60 days to 6 months, and groups of 8 guinea pigs were dosed with 0.2 to 1.6 g/kg/d TEA for 60 or 120 doses. Repeated oral ingestion of TEA produced evidence of hepatic and renal damage in both the species. Some deaths occurred in groups of rats fed with ≥0.3 g/kg/d TEA.
More recently, male and female B6C3F1 mice and F344 rats were given drinking water containing 2% to 8% TEA (purity not specified) for 14 days. 34,35 Male and female high-dose mice and male and female rats given ≥4% TEA had decreased body weights. All but 1 of the high-dose rats were euthanized early due to severe dehydration. No treatment-related changes were observed in mice given 4% TEA or rats given 2% TEA in the drinking water.
Inhalation
Triethanolamine
In a dose-range finding inhalation study, 5 male and 5 female Wistar rats were exposed, nose only, for 5 consecutive days to 100, 200, or 400 mg/m3 TEA (target concentrations; 98.9% pure) for 6 hours. 36 Concentration-dependent laryngeal inflammation and edema were observed at microscopic examination, and the no-observed adverse effect concentration (NOAEC) was 100 mg/m3. The full, 28-day/20-day exposure study used target concentrations of 0, 20, 100, and 500 mg/m3, and the mass median aerodynamic diameter was 0.7 to 1.1 µm. A functional observational battery was conducted using 7 rats/sex/group. Minimal-to-moderate focal inflammation in the submucosa of the larynx was observed; effects were concentration dependent. No systemic toxicity was observed, and there were no effects on organ weights. There were no indications of neurotoxicological effects. Based on the results of this study, the 90-day NOAEC for local irritation was calculated to be 4.7 mg/m3. (The extrapolation of the 28-day laryngeal irritation used the calculation of benchmark concentrations for a 5% incidence of mucosal inflammation, without consideration of severity, using a multistage model.)
In 14-day inhalation studies, B6C3F1 mice and F344 rats were exposed to 125, 250, 500, 1000, or 2000 mg/m3 TEA (purity not specified) 6 h/d, 5 d/wk, for 2 weeks. 37,38 Female mice and male and female rats of the high-dose group had decreased body weights, and male mice of the high-dose group had increased kidney weights. Increased kidney weights in rats dosed with ≥500 mg/m3 and decreased thymus and heart weights in mice at all doses were not clearly associated with TEA. The only histopathologic observation was a minimal acute inflammation of the laryngeal submucosa in both mice and rats; however, this occurred sporadically, and there was no dose response associated with this lesion.
Reproductive and Developmental Studies
Dermal
Triethanolamine
As reported previously, 1 hair dyes containing 0.1% to 0.15% or 1.5% TEA were applied topically to the shaved skin of groups of 20 gravid rats on days 1, 4, 7, 10, 13, 16, and 19 of gestation, and the rats were killed on day 20 of gestation. No developmental effects were observed.
The TEA, 0.5 g/kg in acetone (purity not stated), was applied dermally to clipped skin on the back of male and female F344 rats. 39 A volume of 1.8 mL/kg was applied daily for 10 weeks prior to mating, during mating, and through gestation and lactation. No effect on mating or fertility or offspring growth or survival was observed. A similar study was performed in which Swiss CD-1 mice were given daily applications of 2 g/kg TEA at a volume of 3.6 mL/kg. 40 No adverse developmental effects were observed.
Oral
Triethanolamine
A Chernoff-Kavlock teratogenicity screening test was performed using mated female CD-1 mice, in which the animals were dosed by gavage with 1125 mg/kg/d TEA on days 6 to 15 of gestation. 41 (It was stated that the TEA was the “purest grade commercially available.”) No adverse developmental effects were observed.
Genotoxicity
In Vitro
Triethanolamine
As reported previously, 1 TEA, at the concentrations of ≤100 mg/plate, was not mutagenic in Salmonella typhimurium with or without metabolic activation. The TEA with sodium nitrite, but not TEA alone, was mutagenic in Bacillus subtilis without metabolic activation. The NDELA, which is not mutagenic in B subtilis without metabolic activation, was found in the mixture. In an unscheduled DNA synthesis test in which primary rat hepatocyte cultures were exposed to 10−8 to 10−1 mol/L TEA and [ 3 H]thymidine, simultaneously, TEA did not appear to cause DNA-damage-inducible repair.
In more recent reports, TEA, in distilled water or dimethylsulfoxide, was not mutagenic to Escherichia coli 42 or S typhimurium, with or without metabolic activation, at doses of 0 to 20 000 µg/plate. 42 –44 The TEA (88.2% purity) did not cause gene conversion in Saccharomyces cerevisiae. 43 The TEA was negative in a rec assay at doses of 0 to 4000 µg/disk. No induction of sister chromatid exchanges occurred in Chinese hamster ovary cells at 0 to 1010 µg/mL without metabolic activation or 0 to 10 100 µg/mL with metabolic activation, 45 and chromosomal aberrations were not induced in cultured rat liver cells 43 or at doses of 5 to 100 µg/mL in cultured Chinese hamster cells. 42,45 The TEA, 25 to 500 µg/mL, was negative in a cell transformation assay using hamster embryo cells. 42
In Vivo
Triethanolamine
A mouse peripheral blood micronucleus test was performed using samples collected from mice, which were dosed dermally for 90 days with 0 to 4 g/kg TEA in an NTP study. 46 Results were negative in both male and female mice.
Carcinogenicity
Dermal
Triethanolamine
As reported previously, 1 in a series of 3 experiments using a total of 560 CBA × C57Bl6 male mice, the carcinogenic effects of 99%+ pure TEA and 80%+ industrial grade TEA and the cocarcinogenic effects of TEA and syntanol DC-10 (alcohols C10-18 ethoxylated) were examined over a 14- to 18-month time frame. The TEA was not carcinogenic or cocarcinogenic.
More recently, a 2-year study was performed in which TEA in acetone was applied dermally at doses of 200, 630, or 2000 mg/kg/d to male B6C3F1 mice and at doses of 100, 300, or 1000 mg/kg/d to female B6C3F1 mice. 47 Purity of TEA was 99+%. Using high-performance liquid chromatography/mass spectrometry, 0.491% diethanolamine was detected as impure. A slight increase in diethanolamine was seen in acetone and ethanol solutions after 11 days of storage; the dose formulations were prepared approximately every 2 weeks. The body weights of high-dose males were decreased compared to controls during weeks 17 to 37 and at the end of the study. Dermal irritation increased with increasing dose and was more severe in males than in females. At necropsy, treatment-related epidermal hyperplasia, suppurative inflammation, and ulceration and dermal chronic inflammation occurred at the application site in most test groups, and the incidence and severity increased with increasing dose. Lesions were found, and it was concluded that there was equivocal evidence of carcinogenic activity of TEA in male mice, based on the occurrence of liver hemangiosarcoma, and some evidence of carcinogenic activity in female mice, based on increased incidences of hepatocellular adenoma.
The carcinogenic potential of TEA was evaluated using a Tg·AC transgenic mouse model. 48 Groups of 10 to 15 female homozygous mice were dosed dermally with 3 to 30 mg TEA/mouse in acetone, 5×/wk for 20 weeks. The TEA was inactive in Tg.AC mice.
Oral
Triethanolamine
The oral carcinogenic potential of TEA was examined by administering 1% or 2% TEA in drinking water to groups of male and female B6C3F1 mice for 82 weeks. 49 Diethanolamine was present as an impurity at 1.9%. Body weights of male mice of the 2% group were decreased during weeks 1 to 20 when compared to controls. No significant changes in organ weights were observed. No dose-related increase in the incidence of any tumors was observed in the treated groups, and there was no evidence of carcinogenic potential of TEA upon oral administration.
Drinking water containing 1% or 2% TEA was given to groups of male and female F344 rats for 2 years. 50 Diethanolamine was present as an impurity at 1.9%. From week 69 onward, the dose concentrations for females were reduced by half because of associated nephrotoxicity. A dose-related decrease in body weight gain was reported for male and female test group rats, and a dose-dependent increase in mortality, starting at week 60, was observed. Absolute and relative kidney to body weights were significantly increased in males and females, and the increase was dose related. Severe chronic nephropathy was statistically significantly increased in males of the high-dose group and females of both the dose groups. No treatment-related effects were found in the liver. There was no increase in the incidence of any tumors in the treated groups compared to controls when using the chi-square test. Since increased nephrotoxicity appeared to affect the life span of the treated animals, especially the females, an age-adjusted statistical analysis was performed on the incidences of main tumors or tumor groups for males and females, and a positive trend was noted in the occurrence of hepatic tumors (neoplastic nodule/hepatocellular carcinoma) in males and of uterine endometrial sarcomas and renal-cell adenomas in females. The researchers stated that because these tumors have been observed spontaneously in F344 rats, and since their incidences in the control group was lower than that of historical controls, the occurrence of the tumors may not be attributable to TEA. Instead, increased incidence of renal tumors in the high-dose group may have been associated with renal damage. The researchers concluded that TEA was toxic to the kidneys, especially in females, but it was not carcinogenic to F344 rats.
Possible Mode of Action for Carcinogenic Effects of TEA
It has been reported that choline deficiency induces liver cancer in rodents 51,52 ; therefore, the potential of TEA to cause choline deficiency in the liver of female B6C3F1 mice was investigated as a mode of tumorigenesis. 53 Female mice were dosed dermally with unoccluded applications of 10, 100, 300, or 1000 mg/kg/d TEA in acetone, 5 d/wk for 3 weeks, and female CDF rats were dosed in a similar manner with 250 mg/kg/d TEA. (Purity of TEA was 99+%; diethanolamine impurity levels were 0.04% and 0.45%.) No clinical signs of toxicity were noted for mice or rats. Phosphocholine and betaine levels were statistically significantly decreased in the high-dose mice, and the levels were decreased in these mice. The decrease in phosphocholine levels was variable but dose related. (More pronounced effects were observed when the TEA having 0.45% diethanolamine impurity was used.) In rats, no changes in choline or its metabolites were noted. The potential of TEA to inhibit the uptake of [ 3 H]choline by Chinese hamster ovary cells was also investigated, and a dose-related decrease was observed. The researchers concluded that TEA may cause liver tumor in mice via a choline depletion mode of action, and this effect is likely caused by the inhibition of choline uptake by the cells. The researchers stated that although diethanolamine impurity may contribute to choline depletion, a choline-deficiency mode of tumorigenesis appears to be a property of TEA, exclusive of any diethanolamine impurity.
Carcinogenic Potential in Humans
Triethanolamine
According to an evaluation of TEA by the International Agency for Research on Cancer (IARC) Working Group, there is inadequate evidence in humans, as well as in animals, for the carcinogenicity of TEA. 54 The overall evaluation of the IARC is that TEA is not classifiable as to its carcinogenicity to humans (group 3).
Irritation and Sensitization
Dermal Irritation
In vitro
Triethanolamine
The dermal irritation potential of TEA was determined in 2 in vitro assays, and these results were compared to results obtained in the in vivo Draize test and human patch test. 55 The tissues used in the in vitro tests were fully differentiated 3-dimensional reconstituted human epidermal cultures. Each in vitro test was performed in triplicate. In the first test, the in vitro patch test, TEA was applied to the skin samples for 4 hours using a 0.95-cm2 polypropylene chamber. In the second in vitro test, the direct topical application test, 100 µL TEA was applied directly to a 0.63-cm2 area of the epidermal surface for 4 hours. Histology, cell viability was determined via 3-[4,5-dimethyl-thiazol-2-yl]-2,5-diphenyl tetrazolium bromide reduction, and interleukin 1α (IL-1α) release end points were measured in both the tests, and a combination of the outcomes of these end points was used to determine the potential for irritation. The TEA, which was described as “nonclassified” in the Draize and human patch tests, was classified as a nonirritant in the in vitro patch test, and as an irritant in the direct topical application test. The irritant classification was based on tissue necrosis and a higher amount of released IL-1α compared to the negative control (water). The concentration of TEA tested for each of the studies was not specified.
Non-human
Triethanolamine
As reported previously, 1 the primary skin irritation potential of undiluted TEA was determined using rabbits. After 10 open applications of 0.1 mL to rabbit ears and 10 unoccluded applications to the intact skin of the abdomen, and 3 semioccluded 24-hour applications to abraded skin, it was concluded that TEA was slightly-to-moderately irritating, and prolonged or repeated exposure may be irritating. Twenty-four hours occluded patch tests using groups of 8 male rabbits were performed in 22 laboratories; the primary irritation score ranged from 0 to 5.5 of 24, and the total score for all 22 laboratories was 27.3 of 400. In a preliminary study, occlusive dermal applications of 50% to 100% aq TEA to pairs of guinea pigs resulted in 1 erythematic reaction to undiluted TEA, and in another preliminary study, no irritation was observed when 5%, 10%, or 25% TEA was applied to the backs of guinea pigs.
In a more recent study, the irritancy potential of TEA (purity not specified) was evaluated in an ear-swelling test using female BALB/c mice. 56 A significant increase in irritancy was observed with 25% and 50% TEA compared to the vehicle (4:1 acetone–olive oil).
TEA-lauryl sulfate
As reported previously, 1 the dermal irritation of TEA-lauryl sulfate ranged from not irritating to moderately irritating in rabbit skin at concentrations up to 46%. Concentrations of up to 10% produced slight-to-mild irritation, while concentrations of 25% and 39% produced moderate irritation. Mild irritation was reported with 40% TEA-lauryl sulfate, while testing with 46% TEA-lauryl sulfate produced nonirritant results in some laboratories and irritant results in others.
Human
Triethanolamine
As reported previously, 1 clinical studies were performed with formulations containing TEA. In a few studies on formulations containing 0.45% to 2.4% TEA, the researchers concluded that no irritation was observed, while short-lived acute irritation was reported for formulations containing 1.9% to 2.6% TEA. However, according to the Expert Panel’s interpretation of the results of a number of other studies, formulations containing 0.83% to 20.04% TEA were irritating. In clinical provocative testing using 5 to 10 “hyper-reactors,” 100% TEA produced an irritant reaction on nonscarified skin, 10% TEA in ethanol was a marked irritant on scarified skin, whereas 5% in ethanol was a slight irritant on scarified skin.
More recently, a patch test with TEA (purity not specified) was performed on 20 subjects, and erythema and transepidermal water loss (TEWL) were measured, and the contents of suction blister fluids (SBFs) were evaluated for primary proinflammatory mediators. 57 The aq TEA, 50% to 100%, was applied occlusively for 24 hours; 100 to 200 µL, concentrated to 20 µL with drying, were applied. The percentage of nonresponders to 100% TEA was 80%; those that did respond had weak and nonuniform erythema. The incidence was below or about that found with the solvent controls. For the challenge phase, 765 µmol/cm2 TEA was applied occlusively to 12 subjects for 6 to 24 hours. No increase in TEWL or change in eicosanoid profile of the SBF was observed. The TEA was a nonirritant.
TEA-lauryl sulfate
As reported previously, 9 in clinical studies, shampoos containing 10.5% TEA-lauryl sulfate caused no irritation with semiocclusive patches or “use” testing, while diluted shampoos containing 0.15% to 7.5% produced no-to-moderate irritation. In an occlusive patch test, a diluted shampoo containing 4.4% TEA-lauryl sulfate was highly irritating in a 21-day cumulative irritation test.
More recently, the dermal irritation potential of a 10% solution of TEA-lauryl sulfate (vehicle not identified) at neutral pH was determined using 10 subjects. 58 Duhring chambers were applied to the forearm of each subject for 5 days. A 10% solution of TEA-lauryl sulfate caused intense erythema in nearly all subjects by day 4. Testing was terminated (additional details were not provided).
Sensitization
Non-human
Triethanolamine
As reported previously, 1 TEA was not a sensitizer in guinea pigs when 20 guinea pigs were given dermal applications of undiluted TEA 1×/wk for 3 weeks, followed by challenge applications 14 and 21 days after dosing. No sensitization was seen when 4 lots of TEA were evaluated using groups of 20 guinea pigs; induction applications were applied for up to 6 hours, 1×/wk, for 3 weeks, and the challenge was performed after 14 days. One of the studies used undiluted TEA during induction, while the other 3 studies used 50% TEA at induction. All 4 studies used challenge patches with 90% TEA. No sensitization was observed in a similar study in which induction patches contained a 25% active TEA solution, and a challenge patch with the 25% solution was applied after 1 week of nontreatment.
More recently, the sensitization potential of TEA was evaluated in a local lymph node assay (LLNA) performed with groups of 5 BALB/c mice. 56 This study was performed in conjunction with the ear-swelling test described previously. Lymphocyte proliferation increased with dose, but the increases were not statistically significant. The TEA was not identified as a sensitizer in the LLNA.
Also, the hypersensitivity of mice to TEA (99+% pure) was determined. 59 The TEA, in an acetone–olive oil mixture (4:1) at the concentrations of 3%, 10%, or 30%, was applied daily for 5 consecutive days to groups of 8 female B6C3F1 mice, and the animals were challenged 7 days later with a 30% solution. For some animals, dermabrasion, as well as intradermal injections of Freund complete adjuvant (FCA), was used. There were no treatment-related effects on survival or body weights. There were no statistically significant or dose-related hypersensitivity responses to TEA observed with a radioisotopic method or in an ear-swelling test, with or without FCA.
Results were negative in 3 maximization studies examining the sensitization potential of TEA. 25 In the first test, performed using Pirbright-White guinea pigs, induction consisted of intradermal injections of 2% TEA (98.9% pure) in isotonic saline and epicutaneous application of undiluted TEA, and challenge used 10% TEA in isotonic saline. In the second test using 20 Dunkin-Hartley guinea pigs, intradermal and epicutaneous inductions used 1.5% technical grade TEA and 25% technical grade TEA with 10% sodium lauryl sulfate pretreatment, respectively, and challenge doses consisted of 1%, 5%, and 10% technical and analytical grade TEA. In the third study, with 15 animals and the same induction protocol just described (grade of TEA not specified), 2 of the 15 reacted to 10% TEA after 1 day but not 3 days.
Human
Triethanolamine
As reported previously, 1 TEAs studied in patch tests conducted over a number of years had positive reactions for contact dermatitis. The expert panel interpreted these findings as sensitizing.
In 1 patch test with 64 subjects in which 0.5 mL of 1% TEA (containing 88.6% TEA and 6% diethanolamine) was used, the test solution was not sensitizing. Overall, the majority of formulations containing 0.83% to 4.2% TEA were not sensitizing, and a formulation containing 20.04% TEA, tested on 26 subjects, was not considered sensitizing when it produced 2 slight reactions upon challenge. However, according to the interpretation of the CIR Expert Panel, there were a few cosmetic formulations containing 2.1% and 2.4% TEA that the panel determined to be sensitizing. In other studies with cosmetic formulations containing 2.1% TEA, the researchers concluded that reactions observed at challenge were probably due to skin fatigue.
TEA-lauryl sulfate
As reported previously, 9 undiluted shampoo formulations containing 10.5% TEA-lauryl sulfate and dilutions of formulations containing 0.15% to 7.5% TEA-lauryl sulfate were not sensitizers in clinical studies.
Provocative testing
A group of 737 patients was patch tested with 6 different emulsifiers, including 2.5% TEA (purity not specified) in petrolatum (pet). 60 The patch tests were performed according to International Contact Dermatitis Research Group (ICDRG) recommendations. A total of 39 patients had positive reactions to the emulsifiers, and 20 of those patients, 5 males and 15 females, had positive reactions to TEA. There were 106 irritant reactions reported. The results were clinically relevant in 7 patients. Many of the patients allergic to TEA were also allergic to other ingredients.
Over a 4-year period, the incidences of positive patch test reactions to the same TEA-containing cream were 69 of 171 patients in 1 clinic and 49 of 191 in another. 61 It was hypothesized that the difference between the clinics was due to the differences in sampling methods; the first clinic tested only those patients that had recently used the TEA-containing cream or who had suspected reactions. In the follow-up patch testing with a total of 54 patients from the 2 clinics, 15 of which were controls, 19 patients had a positive allergic response to the TEA-containing cream, 40 had a positive irritant response, and 13 had negative responses. With 1.45% to 5% TEA, 6 patients had a positive response. However, with 5% to 20% TEA stearate, 8 of 8 patients and 15 of 15 controls had a positive irritant response. (TEA stearate was tested because it was demonstrated that TEA stearate was formed from the combination of TEA and stearic acid in formulation.) The researchers postulated the reactions were irritant reactions to TEA stearate. (The amount of TEA stearate present in formulation was estimated to be 4.8%, and 6 of 23 patients patch tested with 5% TEA stearate had irritant reactions).
Over a 15-year period, provocative patch testing using TEA was performed on 85 098 dermatological patients. 25 There were 323 positive reactions to TEA, and most of the reactions (289) were weak positives. The researchers stated that occupational exposure was not a risk factor for TEA contact allergy.
Metalworkers that were patients with dermatitis were patch tested with 2.5% TEA in pet. 62 The patches were applied for 1 to 2 days. On day 3, 1 (0.5%) of the 216 patients had a positive reaction.
Patch testing was performed with 2.5% TEA in pet in 184 patients with suspected metalworking fluid contact dermatitis. 63 (All patients were metalworkers.) Patches were applied for 1 or 2 days. On day 3, 4 patients had any type of positive reactions, 2 (?), and 2 (+). The percentage of positive reactions was 1.1.
Phototoxicity/Photoallergenicity
As reported previously, 1,9 TEA at cosmetic use concentrations was not phototoxic or photoallergenic in non-human and human studies, and TEA-lauryl sulfate was not photoallergenic in human studies.
Ocular Irritation
In vitro
Triethanolamine
The ocular irritation potential of TEA was evaluated in several in vitro studies. The ocular irritation potential of TEA was evaluated in the rabbit corneal epithelium model at concentrations of 0.05%, 0.5%, and 1%; TEA was classified as non/mildly irritating at these concentrations. In the luminescent bacterial toxicity test (ie, the Microtox test), TEA had an half maximal effective concentration (EC50) of 110 mg/L, corresponding to non/moderate irritant potential. 64 In the EYTEX assay, undiluted TEA had an EYTEX/Draize equivalent of 42.1, corresponding to a prediction of severe ocular irritant. 65
Non-human
Triethanolamine
As reported previously, 1 the ocular irritation potential of 0.005 to 0.1 mL undiluted TEA was evaluated in a number of studies using rabbits. With high concentrations and long contact time, TEA may be irritating to rabbit eyes. Using rabbits, 10% aq TEA produced essentially no irritation with or without rinsing. A formulation containing 12.6% TEA, 0.1 mL, was evaluated in a study using 6 rhesus monkeys. Slit lamp examination revealed some corneal effects in 2 monkeys at 24 hours and slight positive fluorescein staining in 1 monkey at 72 hours.
TEA-lauryl sulfate
As reported previously, 9 the ocular irritation potential of TEA-lauryl sulfate was evaluated in rabbit eyes; irritation ranged from not irritating to severely irritating at concentrations up to 40%. The severity of irritation observed with each concentration varied among the studies, as illustrated by 5% TEA lauryl sulfate causing no ocular irritation in 1 study, but significant irritation in another.
Clinical Assessment
Case Reports
Triethanolamine
Eczema of the face of 2 female patients was exacerbated by a cream that contained TEA. 66 Patch testing was performed using the ICDRG standard series, a cosmetic battery, the TEA-containing cream, and TEA at 1%, 2%, and 5% in pet. Both the patients reacted to the TEA-containing cream (+reaction) and to 5% TEA pet (++reaction). One patient reacted to 2% TEA (+reaction), and neither reacted to 1% TEA pet. Patch tests were negative for all other compounds. In a control group of 50 patients, patch testing with 5% TEA pet was negative.
Two cases of occupational asthma in metal workers exposed to cutting fluid containing TEA were reported. 67 Exposure to TEA at temperatures higher than that of ambient air was a common feature.
TEA-PEG-3 cocamide sulfate
Two patients with a reaction to a shampoo were patch tested with the ICDRG standard series and a 1% aq solution of the shampoo. 68 Both the patients had positive patch results to the shampoo only. Subsequent patch test with 1% aq TEA-PEG-3 cocamide sulfate (as well as 1% cocamidopropyl betaine) produced positive results in both the patients. A total of 20 patients with eczema patch used as controls had negative patch test results to 10% aq TEA-PEG-3 cocamide sulfate.
Summary
This report is a safety assessment of TEA and TEA-containing ingredients as used in cosmetics. The TEA is reported to function as a surfactant or pH adjuster, and the other TEA-containing ingredients such as surfactants, skin-conditioning agents, or hair-conditioning agents. TEA-sorbate is reported to function only as a preservative. The TEA is reported to be used in 3756 cosmetic ingredients at concentrations of up to 6% in leave-on formulations, 19% in rinse-off formulations, and 0.7% in products that are diluted for (bath) use. All the other ingredients named in this safety assessment that are in use, with the exception of TEA-lauryl sulfate (302 uses) and TEA-stearate (130 uses), are reported to have less than 20 uses. The TEA may contain diethanolamine as an impurity.
Internationally, data were available from the EU and Canada. In the EU, trialkylamines, trialkanolamines, and their salts are allowed for use by the EU at concentrations of up to 2.5% in leave-on products; “other” product types do not have a concentration limit for TEA. There are additional restrictions regarding conditions for the use of TEA in leave-on and rinse-off products, including a maximum allowable secondary amine content of 0.5% in raw material and a maximum allowable nitrosamine content of 50 µg/kg. According to Health Canada, some leave-on products contain TEA at concentrations of 10% to 30%, with some reporting concentrations in the range of 30% to 100%.
Dermal absorption studies of TEA were performed in vitro using human skin samples and in vivo using mice and rats. In vitro, the absorption of TEA through human skin was low under conditions simulating perceived cosmetic use, that is, 1% to 5% at pH 7.0; approximately 5.5% to 9.5% of the dose was recovered in the skin after 24 or 72 hours; however, only ∼0.5% was recovered in the receptor fluid. In mice, [ 14 C]triethanolamine in acetone was rapidly absorbed, and absorption increased with increasing dose. The majority of the radioactivity was excreted in the urine, 48% to 56% in 72 hours, primarily as unchanged TEA. The TEA was absorbed more slowly and less extensively in rats than in mice. In rats, 19% to 28% of the dose was absorbed over a 72-hour period, and 13% to 24% of the dose was recovered in the urine, mostly as unchanged TEA. In an oral dosing study with rats, TEA was rapidly absorbed in the gastrointestinal tract and excreted mostly in the form of unchanged TEA.
In 2- and 13-week repeated dose dermal toxicity studies in mice with 250 to 2000 mg/kg bw TEA in acetone or 4000 mg/kg bw neat, dermal irritation was observed in the highest dose group, and kidney and liver weights were increased with the higher doses. In rats, 125 to 1000 mg/kg bw TEA in acetone or 2000 mg/kg bw neat was applied to rats for 13 weeks, and irritation was observed at the dosing site. Kidney weights were increased in male and female rats dosed with ≥500 mg/kg TEA, and dosed females had higher incidences of nephropathy. In a 14-day drinking water study, animals given 8% TEA were all killed due to severe dehydration before study termination. Treatment-related changes were not observed for animals given 2% or 4% TEA in their water. In inhalation studies with TEA in rats (≤400 mg/m3) and mice (≤2000 mg/m3), no observations were reported that were indicative of a toxic pulmonary effect.
No adverse developmental effects were seen in dermal studies in which rats and mice were dosed with 0.5 and 2 g/kg TEA, respectively, in acetone from before mating through lactation or in an oral teratogenicity screening test in mice with 1125 mg/kg/d TEA on days 6 to 15 of gestation.
The TEA was negative for genotoxic effects in an Ames test with or without metabolic activation, gene conversion assay, rec assay, sister chromatid exchange assay with or without metabolic activation, chromosomal aberration assay, and cell transformation assay.
In 2-year dermal carcinogenicity studies with doses of up to 1000 and 2000 mg/kg/d TEA for male and female mice, respectively, and up to 125 and 250 mg/kg/d TEA for male and female rats, respectively, it was concluded that TEA produced equivocal evidence of carcinogenic activity in male mice based on the occurrence of liver hemangiosarcoma, some evidence of carcinogenic activity in female mice based on increased incidences of hepatocellular adenoma, equivocal evidence of carcinogenic activity in male rats based on a marginal increase in the incidence of renal tubule cell adenoma, and no evidence of carcinogenic activity in female rats. In oral carcinogenicity studies in rats and mice, TEA was not carcinogenic to rats or mice, but it was toxic to the kidneys of rats, especially females. Based on the preliminary data, it has been hypothesized that TEA may cause liver tumors in mice via a choline-depletion mode of action.
The TEA can be a dermal irritant in both animals and humans, but it has not been shown to be a sensitizer. Many of the ingredients in this report are surfactants, which can be irritating to skin and eyes.
Discussion
Although the CIR Expert Panel noted gaps in the available safety data for the 31 TEA-containing ingredients included in this group, the panel relied on the information available for TEA in conjunction with previous safety assessments of the components of TEA-containing ingredients. Those data could be extrapolated to support the safety of these ingredients. For example, myristic acid has been found safe as used, and the panel was able to extrapolate these data to support the safety of TEA-myristate (ie, the TEA salt of myristic acid). Additionally, some of the ingredients reviewed in this assessment, such as TEA-lauryl sulfate, have been previously reviewed by the CIR and found to be safe for use in cosmetics.
The panel was concerned with the levels of free diethanolamine that could be present as an impurity in TEA or TEA-containing ingredients. The panel stated that the amount of free diethanolamine available must be limited to the present practices of use and concentration of diethanolamine itself, as described in the CIR safety assessment of diethanolamine.
Dermal carcinogenicity studies performed by the NTP on TEA reported equivocal evidence of carcinogenic activity in male mice based on the occurrence of liver hemangiosarcoma, some evidence of carcinogenic activity in female mice based on increased incidences of hepatocellular adenoma, and equivocal evidence of carcinogenic activity in male rats based on a marginal increase in the incidence of renal tubule cell adenoma. It has been hypothesized that TEA may cause liver tumors in mice via a choline-depletion mode of action. Humans are much less sensitive to this deficiency, and these hepatic findings are considered to have little relevance to humans regarding the safety of the use of TEA in personal care products.
The panel was concerned that the potential exists for dermal irritation with the use of products formulated using TEA or TEA-related ingredients. The panel specified that products containing these ingredients must be formulated to be nonirritating.
Tertiary alkyl amines such as TEA do not react with N-nitrosating agents to directly form nitrosamines. However, tertiary amines can act as precursors in nitrosamine formation by undergoing nitrosative cleavage. The resultant secondary amine (ie, diethanolamine) can then be N-nitrosated to products that may be carcinogenic. Because of the potential for this process to occur, TEA and TEA-containing ingredients should not be used in cosmetic products in which N-nitroso compounds can be formed.
The TEA, TEA-lauryl sulfate, and TEA-stearate can be used in products that may be sprayed, and so the panel discussed the issue of potential inhalation toxicity. In the absence of sufficient safety test data to evaluate this end point directly, the panel considered other data that were available to characterize the potential for these ingredients to cause systemic toxicity, ocular or dermal irritation or sensitization, and other effects. The panel noted that 95% to 99% of particles produced in cosmetic aerosols are not respirable. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, this information suggested that inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects.
Conclusion
The CIR Expert Panel concluded that TEA and the 31 related TEA-containing ingredients, listed below, are safe in the present practices of use and concentration described in this safety assessment when formulated to be nonirritating and when the levels of free diethanolamine do not exceed the present practices of use and concentration of diethanolamine itself. These ingredients should not be used in cosmetic products in which N-nitroso compounds can be formed.
Triethanolamine
Magnesium/TEA-coco-sulfate*
Sodium/TEA C12-13 pareth-3 sulfate*
TEA-C10-15 alkyl sulfate*
TEA-C11-15 alkyl sulfate*
TEA-C12-13 alkyl sulfate*
TEA-C12-14 alkyl sulfate*
TEA-C12-15 alkyl sulfate*
TEA-C11-15 pareth sulfate*
TEA-C12-13 pareth-3 sulfate*
TEA-canolate*
TEA-cocoate
TEA-coco-sulfate*
TEA-glyceryl dimaleate*
TEA-hydrochloride
TEA-hydrogenated cocoate*
TEA-isostearate
TEA-lactate
TEA-laurate
TEA-laurate/myristate*
TEA-laureth sulfate
TEA-lauryl sulfate
TEA-myristate
TEA-oleate*
TEA-oleyl sulfate*
TEA-palmitate
TEA-PEG-3 cocamide sulfate*
TEA-sorbate*
TEA-stearate
TEA-sulfate
TEA-tallate*
TEA-undecylenate*
*Were the ingredients not in current use (as indicated by *) to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author's Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
