Abstract
Cocamide diethanolamine (DEA) and some of the other diethanolamides are mainly used as surfactant foam boosters or viscosity increasing agents in cosmetics, although a few are reported to be used as hair and skin conditioning agents, surfactant-cleansing or surfactant-emulsifying agents, or as an opacifying agent. The Cosmetic Ingredient Review (CIR) Expert Panel considered new data and information from previous CIR reports to assess the concerns about the potential for amidases in human skin to convert these diethanolamides into DEA and the corresponding fatty acids. The Expert Panel concluded that these diethanolamides are safe as used when formulated to be nonirritating and when the levels of free DEA in the diethanolamides do not exceed those considered safe by the Panel. The Panel also recommended that these ingredients not be used in cosmetic products in which N-nitroso compounds can be formed.
Keywords
The Cosmetic Ingredient Review (CIR) Expert Panel reviewed the available safety information of Cocamide diethanolamine (DEA) and an additional 32 diethanolamides. Cocamide DEA was previously reviewed in 1996, with the conclusion that this particular diethanolamide is safe when used in rinse-off products and safe at concentrations ≤10% in leave-on cosmetic products, and that cocamide DEA should not be used as an ingredient in cosmetic products in which N-nitroso compounds are formed. 1 Cocamide DEA had been originally reviewed in 1986. 2
Because the data on Cocamide DEA and other available information on tertiary amides reviewed are similar, the Panel determined that the data are sufficient to support the safety of the entire group, and the following 33 diethanolamides are included in this review:
Almondamide DEA Apricotamide DEA Avocadamide DEA Babassuamide DEA Behenamide DEA Capramide DEA Cocamide DEA Cornamide DEA Cornamide/Cocamide DEA Hydrogenated Tallowamide DEA Isostearamide DEA Lanolinamide DEA Lauramide DEA Lauramide/Myristamide DEA Lecithinamide DEA Linoleamide DEA Minkamide DEA Myristamide DEA Oleamide DEA Olivamide DEA Palm Kernelamide DEA Palmamide DEA Palmitamide DEA Ricebranamide DEA Ricinoleamide DEA Sesamide DEA Shea Butteramide/Castoramide DEA Soyamide DEA Stearamide DEA Tallamide DEA Tallowamide DEA Undecylenamide DEA Wheat germamide DEA
The CIR Expert Panel has previously reviewed the related ingredients, lauramide DEA, linoleamide DEA, and oleamide DEA, and concluded that they are safe as used, but not in products containing nitrosating agents. 2 The Expert Panel also concluded in a previous review that isostearamide DEA, myristamide DEA, and stearamide DEA are safe for use in rinse-off products 3 and in leave-on products at concentrations that will limit the release of free ethanolamines to 5%, with a maximum use concentration of 40%. Table 1 provides information on the components included in the review of DEA.
Conclusions of Previously Reviewed Ingredients and Components.

Abbreviation: DEA, diethanolamine.
Chemistry
Definition and Structure
The diethanolamides consist of covalent, tertiary amides, whereby 2 of the nitrogen substituents are ethanol (or at least an ethanol residue) and the third is a carbonyl attached substituent. Figure 1 is an example of behenamide DEA, a tertiary amide wherein 2 of the nitrogen substituents are ethanol and the third is a 22 carbon, carbonyl-attached chain. Although these ingredients are not salts and do not readily dissociate in water, amidases, such as fatty acid amide hydrolase which is known to be present in human skin, could potentially convert these amides to DEA and the corresponding fatty acids. 4 –6
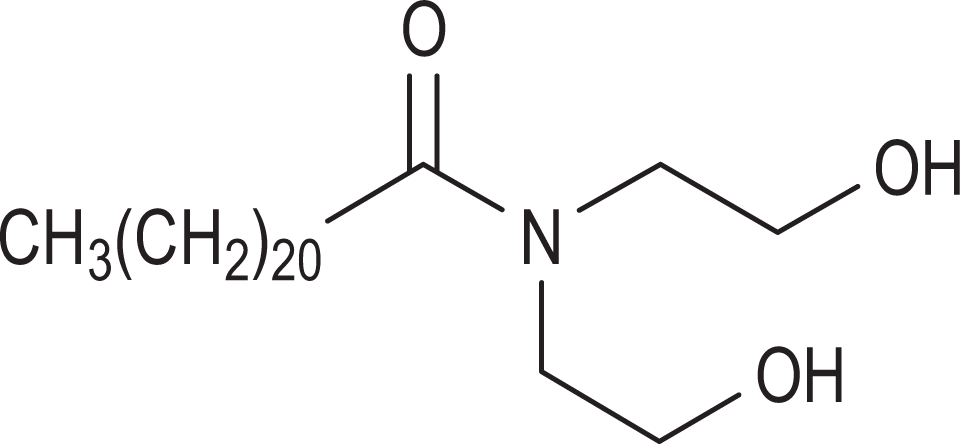
Behenamide diethylamine (DEA).
The CAS registry numbers, definitions, functions, and structures of cocamide DEA and the diethanolamides under consideration are presented in Table 2. The available chemical and physical properties for these ingredients are provided in Table 3.
Definitions and Structures.
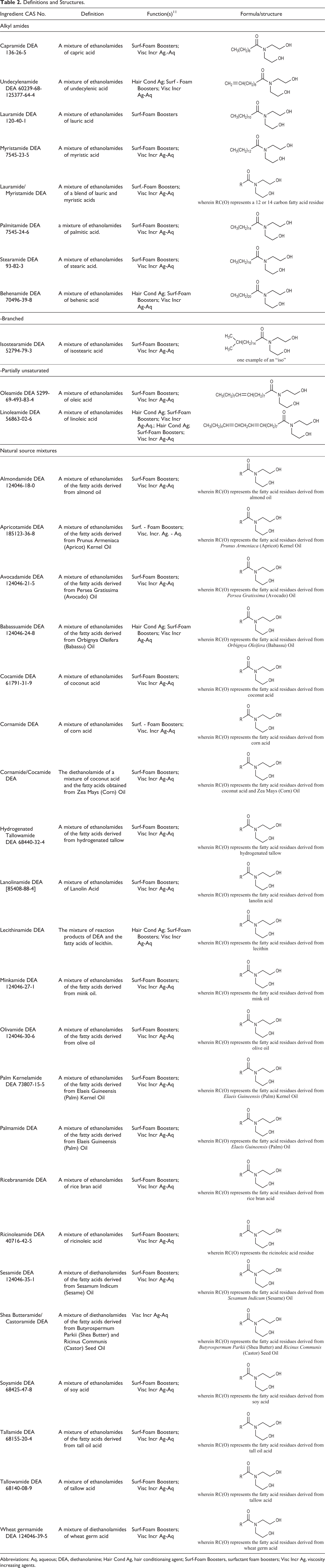
Abbreviations: Aq, aqueous; DEA, diethanolamine; Hair Cond Ag, hair conditionaing agent; Surf-Foam Boosters, surfactant foam boosters; Visc Incr Ag, viscosity increasing agents.
Physical and Chemical Properties.
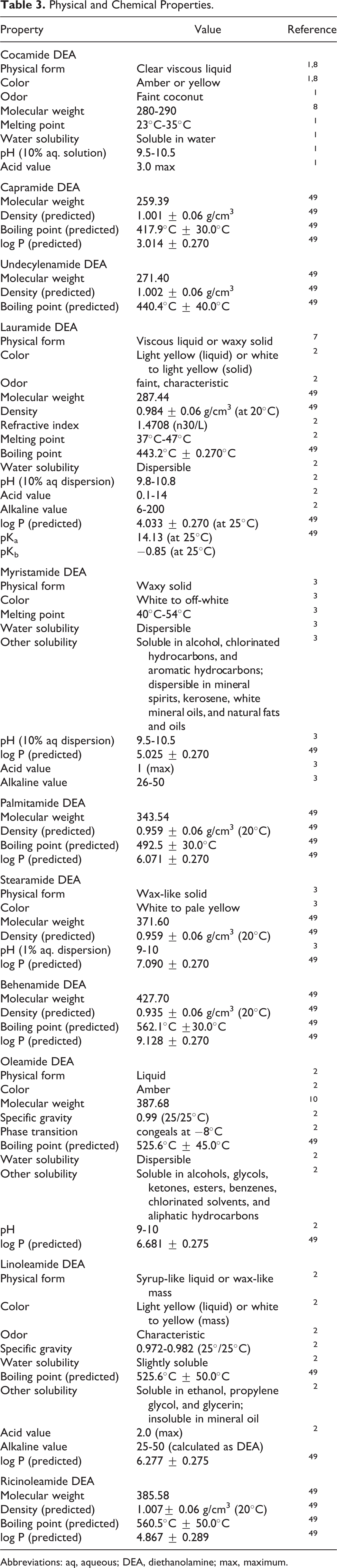
Abbreviations: aq, aqueous; DEA, diethanolamine; max, maximum.
Method of Manufacture
Although specific methods of manufacture for most of the ingredients included in this assessment were not available, in general these diethanolamides can be produced via condensation reaction with an acid. Cocamide DEA, for example, is produced by a condensation reaction at a 1:1 or 1:2 molar ratio of a mixture of methyl cocoate, coconut oil, whole coconut acids, or stripped coconut fatty acids to DEA. 2 Cocamide DEA has also been produced by the reaction of refined coconut oil with DEA in the presence of sodium methoxide (catalyst), yielding cocamide DEA, 10% glycerine, and 5% coconut fatty acid amide. 1 Lauramide DEA is produced by a condensation reaction at a 1:1 or 1:2 molar ratio of a mixture of lauric and myristic acid to DEA 2 and lauramide DEA is produced by the condensation of lauric acid methyl ester with DEA at elevated temperature and in the presence of a catalyst. 7 Oleamide DEA is produced by a condensation reaction at a 1:1 or 1:2 molar ratio of a mixture of oleic acid to DEA, 2 and linoleamide DEA is produced by a condensation reaction at a 1:1 or 1:2 molar ratio of a mixture of linoleic acid or its methyl ester to DEA. 2
Impurities
The manufacturing process of a 1:2 mixture of fatty acid to DEA produces ethylene glycol and free DEA residues; however, the manufacture of a 1:1 mixture contains much less free amine. Alkanolamides are manufactured by base-catalyzed condensation of DEA, and the methyl ester of long-chain fatty acids are susceptible to nitrosamine formation. 2
Cocamide DEA
Although manufacturing data available for various grades of cocamide DEA suggest free DEA at 4.0% to 8.5%, 1 a National Toxicology Program (NTP) study revealed cocamide DEA at approximately 18.2% free DEA by weight, alkanolamides of unsaturated acids, and amine salts of the acids, and N-Nitrosodiethanolamine (NDELA) detected at a concentration of 219 parts per billion (ppb). 8 In 9 commercial samples of cocamide DEA analyzed for DEA, 9 the amount of DEA ranged from 3.2% to 14.0%. The NDELA was not found in any of the samples.
Lauramide DEA
Various grades of lauramide DEA available for cosmetic use have a free amine value of 10 to 35. 2 Results of an NTP study show the purity of lauramide DEA was approximately 90% for lauric acid DEA condensate, with approximately 5% amine (probably DEA) and 5% other organic impurities. 7 The NDELA was detected at a concentration of 3600 ppb. The report also stated that, based on data provided by the manufacturer the lauramide DEA contained 0.83% free DEA by weight and approximately 9% other organic impurities. The DEA in 9 commercial samples of lauramide DEA ranged from 1.2% to 12.4%. The NDELA was not found. 9
Stearamide DEA
Stearamide DEA is characterized by 9% to 12% free fatty acids (as oleic acid) and 2% to 6% free amines (as DEA). 3
Oleamide DEA
Oleamide DEA contains 6.0% to 7.5% free fatty acids (as oleic acid). 2 In an NTP study, the oleic acid DEA condensate content was 47.5%. 10 Impurities were identified as other fatty acid alkanolamides (approximately 30%), other fatty acids, and unidentified impurities. Free DEA was estimated at 0.19%; NDELA was detected at a concentration of 68 ppb.
Linoleamide DEA
In the analysis of commercial sample of linoleamide DEA, DEA was detected at 4.3% to 5.0%. 9 The NDELA was not found in any of the samples.
Use
Cosmetic
Cocamide DEA is reported to function in cosmetics as a surfactant foam booster or a viscosity increasing agent. 11 Most of the other diethanolamides are reported to have these same functions, although a few are reported to function as a hair and conditioning agent, surfactant-cleansing or surfactant-emulsifying agent, or as an opacifying agent.
According to information supplied to the Food and Drug Administration by industry as part of the Voluntary Cosmetic Registration Program (VCRP), cocamide DEA is used in 710 cosmetic formulations, the majority (596) of which are rinse-off formulations. 12 A use concentration survey conducted by the Personal Care Products Council (Council) showed cocamide DEA use at concentrations of 0.5% to 7%. 13,14 The highest concentration of cocamide DEA is reported to be used in leave-on products is 2%. Lauramide DEA is reported to be used in 281 cosmetic formulations at 0.2% to 9%; the use of lauramide DEA at 9% is the highest concentration of use in a leave-on product reported for any of the diethanolamides. Linoleamide DEA has the highest concentration of use reported, 12% in rinse-off formulations. The remaining diethanolamides have less than 35 reported uses. Concentration and frequency of use data for in-use diethanolamides are provided in Table 4. Ingredients not reported to be in use, according to VCRP data and the Council survey, are listed in Table 5.
Frequency and Concentration of Use According to Duration and Type of Exposure.a
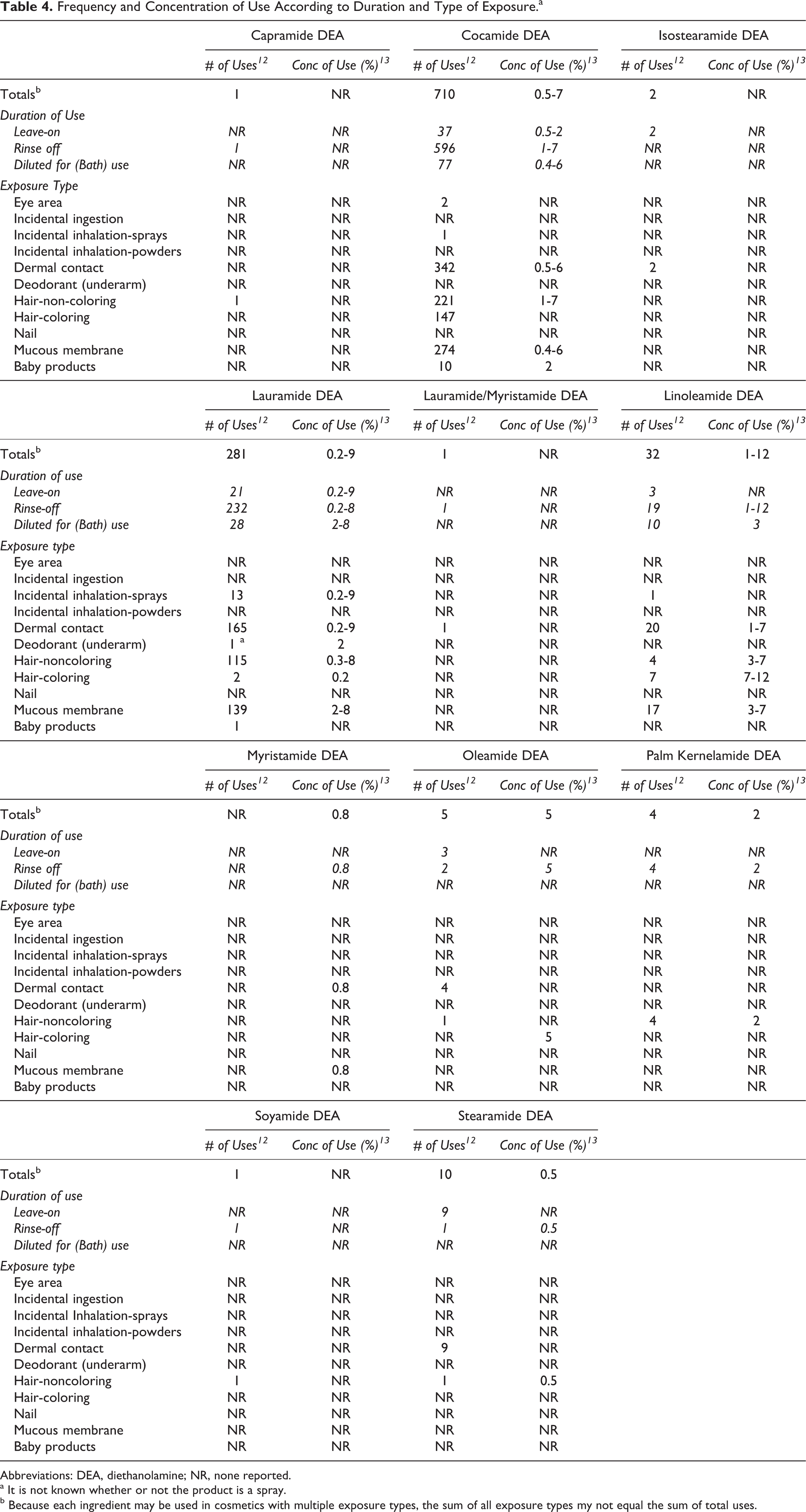
Abbreviations: DEA, diethanolamine; NR, none reported.
a It is not known whether or not the product is a spray.
b Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types my not equal the sum of total uses.
Ingredients not Reported to be in Use.

Abbreviation: DEA, diethanolamine.
Cocamide and lauramide DEA are reported to be used in baby products, and some of the dialkanolamides are used in products that come in contact with the mucous membranes. Additionally, some of the dialkanolamides are reported to be present in hair sprays or fragrance formulations. In practice, 95% to 99% of the aerosols released from cosmetic sprays have aerodynamic equivalent diameters in the range of 10 to 110 µm. 15,16 Therefore, most aerosols incidentally inhaled from these sprays are deposited in the nasopharyngeal region and are not respirable. 17,18 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic diameters in the range considered to be respirable. 18 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays compared to other cosmetic sprays.
Fatty acid dialkanolamides are listed in Annex III of the European Cosmetics Directive, which is a list of substances cosmetic products must not contain except when subject to restrictions, 19 which state a maximum secondary amine content of 0.5% in the finished product; these amides are not use with nitrosating systems; maximum secondary amine content of 5% for raw materials; maximum nitrosamine content of 50 µg/kg; and that these substances are kept in nitrite-free containers. The ingredients listed in Annex III with these restrictions, as well as additional EC information, 20 are provided in Table 6.
Status for use in Europe According to the EC CosIng Database.

Abbreviation: DEA, diethanolamine.
Noncosmetic
Many of the diethanolamides included in this safety assessment are used as indirect food additives. 21 Cocamide, soyamide, and tallamide DEA are used in manufacturing as surface active agents. 22 Cocamide DEA is used as a corrosion inhibitor in metalworking fluids and in polishing agents. 1
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
In Vitro
Lauramide DEA
Human liver slices and liver slices from diethylhexyl phthalate (DEHP)-induced and untreated male F344 rats were incubated with [14C]lauramide DEA. 23 Lauramide DEA “partitioned well” into the human liver slices and the liver slices from DEHP-induced and untreated rats. Approximately 70% of the radioactivity absorbed into the slices in 4 hours. The absorbed radioactivity was present mostly as lauramide DEA. In the media from the human, rat, and DEHP-induced rat liver slice incubations, 32%, 18%, and 43% of the radioactivity, respectively, was present in the form of metabolites. The analytes present in the incubation media included half-acid amides, parent lauramide DEA, and 3 other metabolites that are products of ω- and ω-1 to 4 hydroxylation.
The in vitro metabolism of [14C]lauramide DEA, randomly labeled on the DEA moiety, was examined in liver and kidney microsomes from rats and humans to determine the extent of hydroxylation and to determine the products formed. 24 Incubation of lauramide DEA with liver microsomes from control and DEHP-treated rats produced 2 major high performance liquid chromatography peaks that were identified as 11-hydroxy- and 12-hydroxy-lauramide DEA. Treatment with DEHP increased the 12-hydroxylation rate 5-fold, while the 11-hydroxylase activity was unchanged. Upon comparison of lauramide DEA hydroxylation rates using human liver microsomes with the rates measured using rat liver and kidney microsomes, the lauramide DEA 12-hydroxylase activity in human liver microsomes was similar to the activity in liver microsomes from control rats. The 12-hydroxylase activity in liver microsomes was 3 times greater than that observed in rat kidney microsomes.
Dermal
Non-Human
Lauramide DEA
Groups of 4 male B6C3F1 mice and 4 F344 rats were dosed dermally with [14C]lauramide DEA that was randomly labeled on the DEA moiety. 23 The vehicle was ethanol. A nonocclusive application was made to a 0.5 in 2 . area of mouse skin and to a 1 in 2 . area of rat skin. At the end of the study, the excised skin was rinsed with ethanol. Absorption was calculated from the total disposition of radioactivity in the tissues, urine, feces, and dose site. In mice dosed with 5 to 800 mg/kg [14C]lauramide DEA, 50% to 70% of the applied radioactivity was absorbed at 72 hours, and absorption was similar for all the doses. Approximately 32% to 55% of the radioactivity was excreted in the urine. In rats dosed with 25 or 400 mg/kg lauramide DEA, 21 % to 26% of the radioactivity penetrated the skin in 72 hours, and 3% to 5% was recovered at the application site. Approximately 20% to 24% of the radioactivity was recovered in the urine. The tissue/blood ratio was greatest in the liver and kidney. Lauramide DEA and the half-acid amide metabolites were detected in the plasma, with maximum levels found 24 hours after dosing.
The researchers also examined the effects of repeated administration lauramide DEA on absorption and excretion. Lauramide DEA, 25 mg/kg/d, was applied to 5 rats, 5 times/wk, for 3 weeks. The rate of absorption of lauramide DEA did not vary much at the different collection time points, and the amounts excreted were similar at each collection period.
Oral
Non-Human
Lauramide DEA
Three male F344 rats were dosed orally with [14C]lauramide DEA that was randomly labeled on the DEA moiety, 16 to 18 µCi/dose, and that was formulated with an appropriate amount of unlabeled lauramide DEA and water to give delivery of the target dose in a volume of 5 mL/kg body weight (bw). 23 After oral dosing with 1000 mg/kg [14C]lauramide DEA, approximately 10%, 60%, and 79% of the dose was recovered in the urine after 6, 24, and 72 hours, respectively. Approximately 4% of the dose was recovered in the tissues after 72 hours, with almost 3% found in adipose tissue and 1.3% in the liver. At 6 hours, no DEA, DEA metabolites, or unchanged lauramide DEA were present in the urine; only very polar metabolites were found. The researchers postulated that the metabolites were carboxylic acids, and that the acid function was formed from the lauryl chain.
Intravenous
Non-Human
Lauramide DEA
In all, 3 male B6C3F1 mice and 4 F344 rats were dosed intravenously (iv) with [14C]lauramide DEA that was randomly labeled on the DEA moiety, 3 to 5 µCi and 16 to 17 µCi, respectively, and that was formulated to deliver a target dose in a volume of 4 mL/kg in mice and 1 mL/kg in rats. 23 The dose for mice was 50 mg/kg, and the dose for rats was 25 mg/kg. In B6C3F1 mice, lauramide DEA was quickly metabolized and eliminated. At 24 hours after dosing, approximately 95% of the dose was excreted, with 90% found in the urine; the highest concentrations and total amounts of the lauramide DEA were in adipose tissue. In F344 rats, 50% of the dose was excreted in the urine within the first 6 hours, and more than 80% was excreted in the urine by 24 hours. The rats were killed at 72 hours after dosing, and only 3% of the dose was recovered in the tissues; 1% of the dose was in the adipose tissue and 0.67% was found in the liver.
Toxicological Studies
Single-Dose (Acute) Toxicity
Dermal
Cocamide DEA
The acute dermal toxicity of cocamide DEA was evaluated using 3 male and 3 female albino rabbits. 22,25 Cocamide DEA 2 g/kg were applied to intact and abraded skin for 24 hours using occlusive patches. None of the animals died, and the lethal dose, 50% (LD50) was >2 g/kg.
Lauramide DEA
In an acute dermal toxicity study using guinea pigs, 50% lauramide DEA in corn oil was nontoxic. 2 In a study to evaluate the acute dermal toxicity of lauramide DEA in 3 male and 3 female albino rabbits, 22,25 2 g/kg lauramide DEA were applied to intact and abraded skin for 24 hours using occlusive patches. None of the animals died, and the LD50 was >2 g/kg.
Linoleamide DEA
Linoleamide DEA, tested as 10% aqueous (aq) and undiluted, was nontoxic in acute studies with guinea pigs. 2
Oral
Cocamide DEA
In an acute oral toxicity test in male and female Sprague-Dawley rats, undiluted cocamide DEA had an LD50 of 12.2 g/kg. 2 In an acute oral toxicity study of cocamide DEA using groups of 3 male and 3 female Wistar rats, 3 or more animals per group died with doses of ≥6.3 g/kg. 26 The LD50 of cocamide DEA in several other studies using rats was >5 g/kg or 5 mL/kg, which was the highest dose tested. 22,25
Lauramide DEA
In rats, the oral LD50 of 25% lauramide DEA in corn oil was >5 g/kg, of 10% aq was 2.7 g/kg, of a shampoo formulation containing 8% lauramide DEA was 9.63 g/kg, and of a bubble bath containing 6% lauramide DEA was >15 g/kg. 2 The acute oral toxicity of lauramide DEA, 70% pure (composition included 25% water and 5% DEA), was evaluated using groups of 5 male and 5 female Wistar rats. 25 The animals were gavaged with a single aq dose of 5.0 g/kg bw; 1 male and 2 females rats died by day 4. The LD50 was >3.5 g/kg active ingredients. (The LD50 of the 70% solution was 0.5 g/kg). In another study using male and female Wistar rats, the oral LD50 of lauramide DEA, purity not specified, was > 5 mL/kg, which was the highest dose tested. 25
Stearamide DEA
The oral LD50 of a mixture containing 35% to 40% stearamide DEA was >20 g/kg in CFW mice. 3
Oleamide DEA
In rats, the oral LD50 of undiluted oleamide DEA was 12.4 mL/kg. 2
Linoleamide DEA
In rats, the oral LD50 of undiluted and 10% aq linoleamide DEA was >5 g/kg, and the LD50 of a product containing 1.5% linoleamide DEA was 3.16 g/kg. 2
Inhalation
Tallamide DEA
In an inhalation study, groups of 4 male Swiss-Webster mice were exposed to 86 to 219 mg/m3 tallamide DEA for 3 hours. 22,25 Tallamide DEA produced sensory and pulmonary irritation at low concentrations. The lethal concentration, 50% (LC50) value was >219 mg/m3 (additional details were not provided).
Repeated Dose Toxicity
Dermal
Cocamide DEA
Eight New Zealand White (NZW) rabbits received 1.92% cocamide DEA on the intact or abraded skin of the back. Applications of 500 mg/kg of the test product were made 5×/wk for 4 weeks. Dermal irritation was observed at both intact and abraded application sites. No systemic effects attributed to dosing were observed. 2
The repeated dose dermal toxicity of cocamide DEA (containing 18.2% free DEA by weight) was evaluated using mice and rats. Groups of 10 male and 10 female B6C3F1 mice were dosed with 50, 100, 200, 400, or 800 mg/kg bw cocamide DEA in ethanol (20-320 mg/mL), 5 exposures/wk, for 14 weeks. 8 Dermal irritation was observed at the application sites of males and females of the 800 mg/kg dose group. Epidermal and sebaceous gland hyperplasia, parakeratosis, chronic active inflammation, and ulceration were observed; severity generally increased with increased dose. Final mean bws and mean bw gains were similar for test and control animals. The absolute liver and kidney weights and relative liver and kidney weights to bws of males and females of the 800 mg/kg group, relative liver weights to bws of females of the 400 mg/kg group, and absolute lung weights and relative lung weights to bws of females of the 800 mg/kg group were significantly greater than that for those of the controls. The epididymal spermatozoal concentration was significantly greater in males of the 800 mg/kg dose group.
Groups of 20 male and 20 female F344/N rats were dosed dermally with 25, 50, 100, 200, or 400 mg/kg/bw cocamide DEA in ethanol (30-485 mg/mL), at 5 exposures/wk, for 14 weeks; 10 rats per group were used for clinical chemistry and hematology evaluation. 8 Vehicle only was applied to the negative control group. All animals survived until study termination. Dermal irritation was observed at the application sites of 2 males and 1 female of the 100 mg/kg group and in nearly all males and females of the 200 and 400 mg/kg dose groups. Lesions included epidermal and sebaceous gland hyperplasia, parakeratosis, chronic active inflammation, and ulceration; incidence and severity generally increased with increasing dose. Final mean bws and mean bw gains of males and females of the 200 and 400 mg groups were significantly less than those of the controls. Kidney weights of females of the 50 mg/kg group were significantly greater than those of the controls. Decreases in epididymal weights in 200 and 400 mg/kg males were attributed to decreased bws. Changes in some hematology and clinical chemistry parameters were noted, and the researchers stated there was an indication of altered lipid metabolism, as evidenced by decreased cholesterol and triglyceride concentrations. The incidences of renal tubule regeneration were greater in females of the 100 dose group, and the incidences and severities were greater in females of the 200 and 400 mg/kg dose groups, when compared to controls.
Lauramide DEA
The dermal toxicity of lauramide DEA was evaluated in two 13-week studies using Sprague-Dawley rats. No systemic toxic effects were observed for a 0.45% aq solution containing 4.0% lauramide DEA, tested in 15 females, and a solution containing 5.0% lauramide DEA, tested in 10 males and 10 females. 2
Groups of 10 male and 10 female B6C3F1 mice were dosed with 50, 100, 200, 400, or 800 mg/kg bw lauramide DEA in ethanol (90% purity; 0.83% free DEA by weight), 5 exposures/wk, for 14 weeks. 7 All animals survived until study termination. Dermal irritation was observed at the application sites of males and females dosed with 400 or 800 mg/kg lauramide DEA. Final mean bws and mean bw gains were similar for test and control animals. The absolute kidney weights of males of the 100, 400, and 800 mg/kg bw groups, the relative kidney to bws of all dosed males, and the liver weights of females of the 200, 400, and 800 mg/kg bw groups, were statistically significantly greater than those of the control mice. The absolute thymus weights of males of the 400 and 800 mg/kg groups were significantly less than those of the controls. There were no statistically significant differences in reproductive tissue evaluation or estrous cycle between the treated and the control groups. At the application site, incidences of nonneoplastic lesions of the skin, including hyperplasia of the epidermis and sebaceous gland, chronic inflammation, parakeratosis, and ulceration, were increased in males and females dosed with ≥200 mg/kg lauramide DEA.
Groups of 20 male and 20 female F344/N rats were dosed dermally with 25, 50, 100, 200, or 400 mg/kg bw lauramide DEA in ethanol, 5 exposures/wk for 14 weeks; 10 rats per group were used for clinical pathology. 7 All animals survived until study termination. Dermal irritation was observed at the application site of males dosed with ≥100 mg/kg and in females dosed with 200 or 400 mg/kg lauramide DEA. Final mean bws and mean bw gains of males of the 200 and 400 mg/kg bw group were statistically significantly less than those of the control group. Kidney weights of females dosed with 200 or 400 mg/kg bw were statistically significantly greater, and absolute liver weights of males dosed 400 mg/kg lauramide DEA were statistically significantly less, than those of the control groups. There were no statistically significant differences in reproductive tissue evaluation or estrous cycle between the treated and the control groups. At the application site, incidences of nonneoplastic lesions of the skin, including hyperplasia of the epidermis and sebaceous gland, chronic inflammation, parakeratosis, and ulceration, were statistically significantly increased with increasing dose.
Oleamide DEA
The repeated dose dermal toxicity of oleamide DEA (47.5% oleic acid DEA condensate content; 0.19% free DEA) was evaluated using mice and rats. Groups of 10 male and 10 female B6C3F1 mice were dosed with 50, 100, 200, 400, or 800 mg/kg bw oleamide DEA in ethanol (20-320 mg/mL), 5 exposures/wk, for 13 weeks. 10 All animals, except 1 high dose male, survived until study termination. Final mean bws and bw gains of males of the 800 mg/kg group and females of the 400 mg/kg group were statistically significantly less than those of controls. Dermal irritation was observed at the application site of all treated males and for most females dosed with ≥100 mg/kg oleamide DEA. Lesions included epidermal hyperplasia, parakeratosis, suppurative epidermal and chronic active dermal inflammation, sebaceous gland hypertrophy, and ulceration; severity generally increased with increased dose. Heart weights of females of the 200 mg/kg and males and females of the 400 and 800 mg/kg groups, kidney weights of males of the 50, 100, and 400 mg/kg groups, and liver weights of all dose groups were statistically significantly greater than those of controls. The incidences of hematopoietic cell proliferation of the spleen of males of the 800 mg/kg group and females of the 400 and 800 mg/kg groups were statistically significantly greater than the controls. Sperm motility and vaginal cytology parameters of dosed mice were similar to those of the controls.
Groups of 20 male and 20 female F344/N rats were dosed dermally with 25, 50, 100, 200, or 400 mg/kg bw oleamide DEA in ethanol (30-485 mg/mL), 5 exposures/wk for 13 weeks; 10 rats per group were used for clinical chemistry and hematology evaluation. 10 All animals survived until study termination. Dermal irritation was observed at the application site of most males dosed with ≥100 mg/kg and all females dosed with ≥50 mg/kg oleamide DEA. Lesions included epidermal hyperplasia, parakeratosis, suppurative epidermal and chronic active dermal inflammation, and sebaceous gland hypertrophy; severity generally increased with increased dose. The final mean bws and mean bw gains of males of the 200 and 400 mg/kg groups and mean bw gains of females of the 400 mg/kg group were statistically significantly less than controls; some associated lower organ weights were observed. Kidney weights were statistically significantly greater for females of the 200 and 400 mg/kg groups when compared to controls. Some increases in segmented neutrophil counts and alkaline phosphatase concentrations were reported. There were no biologically significant differences in sperm motility or vaginal cytology parameters between treated and control rats.
Linoleamide DEA
In a 13-week study using a formulation containing 3.0% linoleamide DEA, solutions were applied at 2.5%, 25% solution, or at a 25% solution that was rinsed after 15 minutes, to groups of 10 male and 10 female Sprague-Dawley rats. Dermal irritation was observed, and the formulation containing 3% linoleamide DEA was not a cumulative systemic toxicant. 2
Oral
Lauramide DEA
In the first of two 13-week dietary studies, groups of 15 male and 15 female SPF rats were fed 0% to 2% lauramide DEA. A reduction in growth was associated with reduced feed intake at doses of ≥0.5% lauramide DEA. There were no treatment-related gross or microscopic lesions. The no-effect dose was 0.1% lauramide DEA. In the second study, groups of 20 male and 20 female Wistar rats were fed 0 to 250 mg/kg/d. No adverse effects were reported, and the no-effect dose for rats was 250 mg/kg/d. Groups of 4 male and 5 female Beagle dogs were fed 0 to 5000 parts per million (ppm) lauramide DEA for 12 weeks. No adverse effects were reported, and the no-effect dose for dogs was 5000 ppm lauramide DEA. 2
Reproductive and Developmental Toxicity
Cocamide DEA
Groups of gravid female Sprague-Dawley rats (number per group not specified) were gavaged with 5 mL/kg bw of 0, 100, 300, or 1000 mg/kg/d cocamide DEA, 90% to 95% pure, on days 6 to 15 of gestation. 25 Controls were dosed with arachis oil. The dams were killed on day 20 of gestation. No deaths occurred in any of the groups. Salivation and propulsion of the head was observed in all test groups; salivation was “severe” in the 1000 mg/kg group. The bws and weight gains were comparable for all groups, as were fetal bws. Postimplantation loss and total embryonic deaths were statistically significantly increased in all treated groups compared to the controls; these findings were considered incidental by the researcher because 1 single female accounted for these findings in each group. Although retardation of ossification was statistically significantly increased in the 300 and 1000 mg/kg groups, these values were within the normal range of variation for this strain. The incidence of ossification of the skull bones was statistically significantly increased in 2 dams (accounting for 10 of the 17 findings) in the 1000 mg/kg group. The NOAELs for maternal toxicity and developmental toxicity were both reported as 1000 mg/kg/d.
No other reproductive and developmental toxicity studies of the diethanolamides were found. Because DEA may be an impurity in the diethanolamides, and amidases in the skin might convert some of the diethanolamide to DEA and the corresponding fatty acid, data on DEA and other dialkanolamide components was reviewed.
Diethanolamine
Hair dyes containing up to 2% DEA were applied topically to the shaved skin of groups of 20 gravid rats on days, 1, 4, 7, 10, 13, 16, and 19 of gestation, and the rats were killed on day 20 of gestation. No developmental or reproductive effects were observed. 27
Gravid mice dosed dermally with 20 to 320 mg/kg DEA from day 6 of gestation through PND 21 showed no effects on skeletal formation, but dose-dependent effects on some growth and developmental parameters were observed. In a study in which parental mice were treated dermally with 20 to 320 mg/kg DEA for 4 weeks prior to mating, sperm motility was decreased in a dose-dependent manner. In rats and rabbits, dermal dosing with up to 1500 mg/kg/d and 350 mg/kg/d DEA, respectively, during gestation, did not have any fetotoxic or teratogenic effects. The NOEL for embryonal/fetal toxicity was 380 mg/kg/d for rats and 350 mg/kg/d for rabbits. 28
In an oral developmental study in which rats were dosed with up to 1200 mg/kg/d DEA on days 6 to 15 of gestation, maternal mortality was observed at doses of ≥50 mg/kg; the NOEL for embryonal/fetal toxicity was 200 mg/kg/d. In a study in which gravid rats were dosed orally with up to 300 mg/kg/d DEA, the dams of the 300 mg/kg group were killed due to excessive toxicity; the LD50 was calculated to be 218 mg/kg. The LOAEL for both maternal toxicity and teratogenicity was 125 mg/kg/d. 28
In a developmental study in which rats were exposed by inhalation to DEA on days 6 to 15 of gestation, the NOAEC for both maternal and developmental toxicity was 0.05 mg/L, and the NOAEC for teratogenicity was >0.2 mg/L. 28
Lecithin
In oral studies, ≤1600 mg/kg lecithin was not a reproductive toxicant in mice or rats and ≤47 mg/kg was not a reproductive toxicant in rabbits. In an iv reproductive study, the lowest toxic daily iv dose for rats was >1000 mg/kg. Lecithin, ≤3.0 mmol/L, had no significant effect on human sperm motility. 29
Palm Oil
Crude palm oil (10%) was not a reproductive toxicant in a study in which male and female Wistar/NIN inbred weanling rats were fed prior to mating. Mean litter sizes were comparable between test and control groups. No significant changes were found in liver or kidney weight in adult animals. Neither untreated palm oil (15%) nor 15% heated palm oil in the diet induced anomalies with respect to fertility and in utero growth when fed to male and female Sprague-Dawley SPF rats prior to mating. In a study investigating the effects of palm oil on sexual maturation and endocrine function, vaginal opening was observed significantly earlier (compared to 5% corn oil control) in weanling rats fed 20% palm oil in the diet. No significant differences were observed in endocrine function. 30
Palm Kernel Oil
Offspring from the mated adult Mongolian gerbils fed a diet containing 8.75% w/w palm kernel oil showed no statistically significant differences in frequency of litters, mean litter size, total of newborns, and suckling death. Animals receiving a basal diet served as the control. 30
Ricinus Communis (Castor) Seed Oil
Groups of mice and rats fed diets containing 0.62%, 1.25%, 2.5%, 5.0%, and 10% castor oil continuously for 13 weeks had a slight decrease in epididymal weight (6% to 7%) in mid- and high-dose groups of male rats; however, this finding was not dose related. No effects on any other male reproductive end point (testes weight and epididymal sperm motility, density, or testicular spermatid head count) or female reproductive end point (estrous cycle length, or time spent in each phase of the cycle) were noted. Castor oil served as the vehicle control in a study evaluating the effect of long-term treatment with ICI 182,780 (an antiestrogen) on the rat testis. In the control group, 4 male Sprague-Dawley rats were injected subcutaneously (sc) with castor oil (0.2 mL) once per week and then killed 100 days after the first injection. Spermatogenesis appeared normal in each of the 4 control rats. 31
Sesamum Indicum (Sesame) Seed Oil
Although not teratogenic, oral dosing with sesame oil (4 mL doses) increased the incidence of resorptions in rats when compared to controls. In a 42-week, 2-generation reproduction study involving rats, sesame oil (vehicle control, dose volume not stated) did not induce any adverse effects on reproductive performance, fertility, or reproductive organ weights of male or female rats through 2 consecutive generations. Oral dosing with sesame oil (vehicle control, single intragastric dose [not stated]) on day 9 of gestation also had no adverse effect on the fetal survival rate or crown-rump length in mice. Dosing with sesame oil sc did not adversely affect the development of mice receiving doses (0.05 mL injections) beginning at 3 to 5 days of age or induce teratogenic effects in their offspring. In a study involving rats, dosing with sesame oil sc (0.05 mL injections) did not have an adverse effect on the following when compared to untreated controls: uterine and ovarian weight (female rats) and weight of the testes, prostate, and seminal vesicles (male rats). Dosing with sesame oil intraperitoneally (0.4 mL) was associated with a marked increase in the incidence of deciduomas in mice. 32
Tall Oil Acid
No treatment-related effects were observed in rats fed diets containing 5% and 10% tall oil acid in a 2-generation study. 33
Genotoxicity
Cocamide DEA
Cocamide DEA was not mutagenic in an Ames assay (0.1-200 µg/plate), did not induce mutations in L5178Y mouse lymphoma cells (1.25-50 nL/mL), nor SCEs (0.5-30 µg/mL) or chromosomal aberrations (16-50 µg/mL) in Chinese hamster ovary (CHO) cells; all tests were performed with and without metabolic activation. 8 Significant increases in the frequencies of micronucleated normochromatic erythrocytes were found in peripheral blood of male and female mice at the end of a 14-week repeated dose study (described earlier).
Lauramide DEA
Lauramide DEA was not mutagenic or genotoxic in multiple Ames assays, a DNA damage assay using Bacillus subtilis, an in vitro transformation assay using Syrian golden hamster embryo cells, or an in vivo transformation assay using hamster embryo cells. Lauramide DEA was mutagenic in the spot test with 2 strains of Salmonella typhimurium (quantitative results were not provided). 2
Lauramide DEA (0.3-1000 µg/plate) was not mutagenic in the Ames test with or without metabolic activation, was negative in a L5178Y mouse lymphoma assay (2.5-60 µg/mL), did not increase the number of chromosomal aberrations in CHO cells (1.5-100 µg/mL), with or without metabolic activation, and was not clastogenic in a mouse micronucleus test (50-800 mg/kg). 7 Lauramide DEA (2.49-49.7 µg/mL) induced SCEs in CHO cells, in the presence and the absence of metabolic activation.
Oleamide DEA
Oleamide DEA was not mutagenic in an Ames test (0.1-200 µg/plate) and did not induce mutations in L5178Y mouse lymphoma cells (1.25-20 nL/mL), with or without metabolic activation. 10
Carcinogenicity
Dermal
Conclusions of NTP dermal carcinogenicity studies on lauramide DEA, oleamide DEA, cocamide DEA and DEA are summarized in Table 7.
Conclusions of NTP Dermal Carcinogenicity Studies.
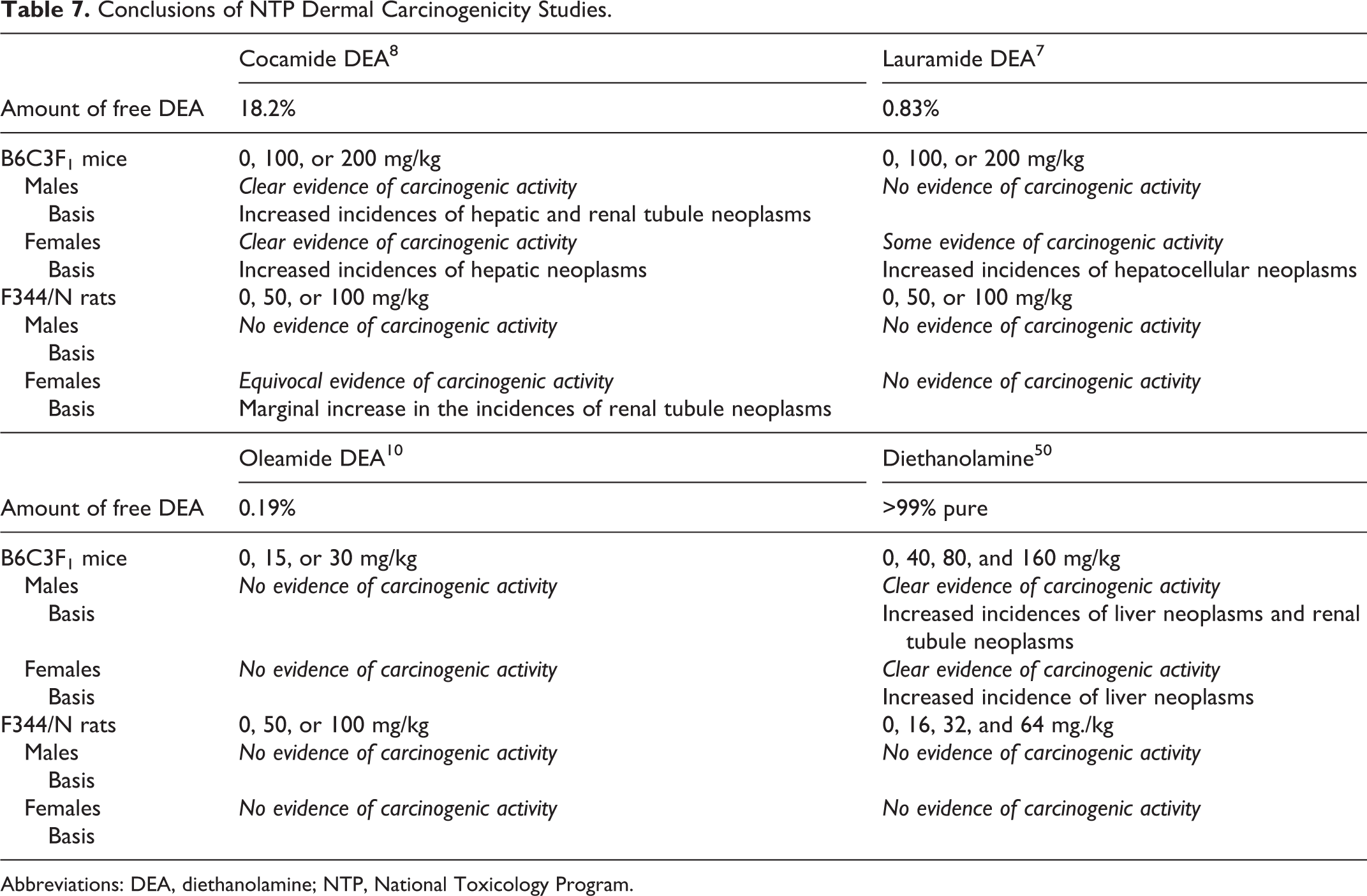
Abbreviations: DEA, diethanolamine; NTP, National Toxicology Program.
Cocamide DEA
The carcinogenic potential of dermally applied cocamide DEA (containing 18.2% free DEA by weight) was assayed by the NTP, using B6C3F1 mice and F344/N rats. 8 Groups of 50 male and 50 female mice were dosed dermally with 0, 100, or 200 mg/kg cocamide DEA in ethanol, 5 days/wk, for 104 to 105 weeks. There were no statistically significant differences in survival between the test animals and the controls. Mean bws of 100 and 200 mg/kg females were less than controls from weeks 93 and 77, respectively. Dermal irritation was observed at the application site of 200 mg/kg males. The incidences of epidermal and sebaceous gland hyperplasia and hyperkeratosis were statistically significantly greater in all dose groups compared to the controls, and in the in 200 mg/kg dose group, the incidences of ulceration in males and inflammation and parakeratosis in females were increased. The incidences of hepatic neoplasms were statistically significantly greater in dosed male and female mice compared to controls. The incidences of eosinophilic foci in dosed groups of males were increased compared to controls, and the incidence of nephropathy was statistically significant. The incidences of renal tubule adenoma and of renal tubule adenoma or carcinoma (combined) in 200 mg/kg males were statistically significantly greater than controls and exceeded the historical control ranges for these neoplasms. In the thyroid gland, the incidences of follicular cell hyperplasia in all dosed groups of males and females were statistically significantly greater than the controls. The researchers concluded that the clear evidence of carcinogenic activity in male and female B6C3F1 mice was associated with the concentration of free DEA present as a contaminant in the DEA test compound.
Groups of 50 males and 50 females rats were dosed dermally with 0, 50, or 100 mg/kg bw cocamide DEA in ethanol (0, 85, or 170 mg/mL, respectively), 5 days/wk for 104 weeks. Survival and mean bws were similar in test and control animals. Dermal irritation was observed at the application site of 100 mg/kg females. The incidences of epidermal and sebaceous gland hyperplasia, parakeratosis, and hyperkeratosis were statistically significantly greater in all dose groups compared to the controls; the severity of the lesions generally increased with increasing dose and ranged from minimal to mild. Incidences of renal tubule hyperplasia in dosed females and of renal tubule adenoma or carcinoma (combined) in females of the 50 mg/kg group were statistically significantly greater than in the controls. Incidences of nephropathy were similar between test and control rats; severity in females increased with increasing dose. In the forestomach, the incidences of chronic, active inflammation, epithelial hyperplasia, and epithelial ulcer were statistically significantly increased in 100 mg/kg females. The incidence of pancreatic acinar atrophy was statistically significantly greater in the 100 mg/kg males than in the controls. The researchers concluded there was no evidence of carcinogenic activity in male F344/N rats dosed dermally with 50 or 100 mg/kg cocamide DEA. There was equivocal evidence of carcinogenic activity in female F344/N rats, based on a marginal increase in the incidences of renal tubule neoplasms.
Lauramide DEA
The NTP evaluated the carcinogenic potential of lauramide DEA (90% purity; 0.83% free DEA by weight) using B6C3F1 mice and F344/N rats. 7 Groups of 50 male and 50 female mice were dosed dermally with 0, 100, or 200 mg/kg/d lauramide DEA in ethanol (0, 50, or 100 mg/mL, respectively), 5 days/wk, for 105 to 106 weeks. No clinical findings were attributable to lauramide DEA. In female mice, the incidence of hepatocellular adenoma was statistically significantly increased in the 100 mg/kg group, and eosinophilic foci were statistically significantly increased in the 200 mg/kg group. The incidences of these lesions in male mice were not statistically significantly different from controls. Incidences of nonneoplastic lesions of the skin at the site of application were statistically significantly increased in treated males and females; the lesions were mostly epidermal and sebaceous gland hyperplasia. The incidence of focal hyperplasia of thyroid gland follicular cells was statistically significantly greater in males of the 200 mg/kg group compared to controls; there were no corresponding increases in the incidences of follicular cell neoplasms. There was no evidence of carcinogenic activity in male mice. Researchers hypothesized that evidence of carcinogenic activity in female B6C3F1 mice based on increased incidences of hepatocellular neoplasms was associated with free DEA that was present as a contaminant.
Groups of 50 male and 50 female rats were dosed dermally with 0, 50, or 100 mg/kg bw lauramide DEA in ethanol (0, 85, or 170 mg/mL, respectively), 5 days/wk, for 104 to 105 wks. Findings showed minimal to moderate irritation at the application site; epidermal and sebaceous gland hyperplasia, hyperkeratosis, and chronic inflammation were statistically significantly increased compared to controls. The incidence of neoplasms was similar for treated and control rats. The incidence of forestomach ulcer in the 100 mg/kg group males, inflammation of the nasal mucosa in all test males, and chronic inflammation of the liver in 100 mg/kg females was statistically significantly lower than that in the controls. There was no evidence of carcinogenic activity of lauramide DEA in male or female F344/rats.
Oleamide DEA
The NTP also examined the carcinogenic potential of dermally applied oleamide DEA (47.5% oleic acid DEA test compound content; 0.19% free DEA) using B6C3F1 mice and F344/N rats. 10 Groups of 55 male and 55 female mice were dosed dermally with 0, 15, or 30 mg/kg oleamide DEA in ethanol (0, 7.5, or 15 mg/mL, respectively), 5 days/wk, for 105 weeks; 5 males and 5 females per group were used for a 3-month interim evaluation. Survival was similar for treated and control mice. Mean bws of females of the 30 mg/kg group were less than controls as of week 76 of the study. Increased incidence of dermal irritation was observed at the application site of males of the 30 mg/kg dose group. The incidences of epidermal and sebaceous gland hyperplasia were statistically significantly increased in all male and female dose groups, when compared to controls, at both the 3-mnth and 2-year evaluation. Additional dermal lesions were observed, but a dose-related increase in neoplasms was not observed. The incidence of malignant lymphoma in female mice increased with increasing dose and was statistically significant in the high-dose group. However, the researchers noted that the incidence in the high-dose group was similar to the incidences observed in other studies that used ethanol as the vehicle. No evidence of carcinogenic activity was found in male or female mice dosed dermally with ≤30 mg/kg oleamide DEA.
Groups of 50 male and 50 female rats were dosed dermally with 0, 50, or 100 mg/kg oleamide DEA in ethanol (0, 85, or 170 mg/mL, respectively), 5 days/wk, for 104 weeks. Mean bws of males of the 100 mg/kg group were slightly less than the controls throughout the study and in the females (100mg/kg group), a decrease in bws was observed from week 24 onward. Mild to moderate irritation was observed, and skin lesions observed at the application site, including statistically significant increases in epidermal and sebaceous hyperplasia, were considered indicative of local irritation, with no neoplastic or preneoplastic changes. Researchers did not consider increased incidences of lesions in the forestomach, testis, and thyroid gland test article related. No evidence of carcinogenic activity in male or female rats dosed dermally with ≤100 mg/kg oleamide DEA was observed.
Irritation and Sensitization
Dermal Irritation
Non-Human
Cocamide DEA
Cocamide DEA, 30% in propylene glycol, was a moderate skin irritant in an irritation study using an occlusive covering. 2
Lauramide DEA
In immersion tests using guinea pigs, a 0.1%-0.5% aq solutions of lauramide DEA was minimally to mildly irritating, a shampoo formulation containing 8% lauramide DEA, tested as a 0.5% solution, was a slight irritant, and a bubble bath containing 6% lauramide DEA, tested as a 0.5% aq solution, was practically nonirritating. In rabbits, a 1.25% to 10% aq solution was practically nonirritaing to slightly irritating, while a 20% aq solution was a severe irritant. In a 14-day cumulative irritation test using rabbits, a 1% aq solution was not an irritant, a 5% solution was a moderate irritant, and a 25% solution was a severe irritant. Liquid soap formulations containing 10% lauramide DEA ranged from mildly to severely irritating in rabbit skin. 2
Stearamide DEA
A mixture containing 35% to 40% stearamide DEA had a primary irritation score of 0 in a dermal study using rabbits. 3
Oleamide DEA
Oleamide DEA in propylene glycol was mildly irritating to rabbit skin when tested at 5% and moderately irritating when tested at 70%. 2
Linoleamide DEA
A 0.1% to 0.5% aq solution of linoleamide DEA was nonirritating to slightly irritating in immersion tests with guinea pigs, and a formulation containing 1.5% linoleamide DEA, tested as a 0.5% aq. solution, was a slight irritant in an immersion test. In primary irritation tests using rabbits, 5% to 10% aq linoleamide DEA was nonirritating to mildly irritating, while an aq solution of 20% linoleamide DEA was a severe dermal irritant in rabbits. A formulation containing 1.5% linoleamide DEA, tested as a 2.5% aq solution, was a minimal dermal irritant in rabbits. 2
Ricinoleamide DEA
Undiluted polyethylene glycol (PEG)-20 glyceryl ricinoleate + ricinoleamide DEA was evaluated for dermal irritation in a Draize test using NZW rabbits. 34 A semiocclusive patch with 0.5 g of the test material was applied to a 6 cm2 shaved site on the dorsal area of the trunk for 4 hours. No signs of irritation were observed, and the surfactant was nonirritating.
Human
Cocamide DEA
The irritation potential of 10% cocamide DEA, 20% sodium lauryl sulfate, and 5 other cosmetic-grade surfactant solutions was evaluated in 15 patients. Adverse reactions were not observed. Researchers concluded that skin irritation was not related to the total concentration of the surfactants in contact with the skin but rather the combination of surfactants present. 1
An aq solution of 12.5 mmol/L cocamide DEA was applied to the forearm of 15 volunteers. 35 Using a plastic chamber, a 0.3 mL solution was applied for 45 min/exposure twice a day, 5 days/wk, for a total of 28 applications. The mean transepidermal water loss (TEWL) with cocamide DEA was 7.0 g/m2 l; the TEWL with 12.5 mmol/L sodium lauryl sulfate was 15.2 g/m2 l.
The irritation potential of 0.5% aq cocamide DEA was evaluated in a single insult occlusive patch test using 105 patients, 14.3% of which were atopic patients. 36 Application of 40 µL was using Haye test chambers for 48 hours; the test site was evaluated by erythema and edema. An untreated occlusive patch was used as a negative control. Cocamide DEA had a total average index of skin irritation (AII) of 0.065 and was nonirritating (AII < 0.5) based on an amended Draize scale.
Lauramide DEA
In primary irritation tests (single patch) using 17 to 19 patients, a 1.25% aq solution of a shampoo containing 8%, and a bubble bath containing 6% lauramide DEA, and an unspecified product containing 5% lauramide DEA, tested as a 1% aq solution, minimal to mild irritation was observed. In 3 cumulative irritation soap chamber tests using 12 to 15 patients, liquid soap formulations containing 10% lauramide DEA, tested as 8% aq solutions, were essentially nonirritating to mildly irritating. In a 21-day cumulative irritation study, a medicated liquid soap containing 5% lauramide DEA, tested as a 25% solution, was a moderate skin irritant. A liquid soap containing 10% lauramide DEA, evaluated in 114 patients for 4 weeks, was minimally irritating under normal use and an acne liquid cleanser containing 5% lauramide DEA, evaluated in 50 patients with twice daily use for 6 weeks, was a mild irritant. 2
Linoleamide DEA
In a primary irritation (single patch) study, a product containing 1.5% linoleamide DEA, tested as a 1.25% aq solution in 20 patients, was a mild skin irritant. 2
Sensitization
Human
Cocamide DEA
In 8 occupational exposure studies to evaluate the sensitization potential of cocamide DEA at 0.01% to 10%, positive results were seen; however, it is recognized that while occupational exposure to cocamide DEA can result in sensitization, cosmetic use does not present the same concerns. 1 An in-use study using shampoo containing 2% cocamide DEA on104 female patients patch tested with 2% aq shampoo before and 10 days after 87 days of using the shampoo showed that cocamide DEA was an irritant but not a sensitizer. 2
Lauramide DEA
Six repeat insult patch tests (RIPTs) using 41 to 159 patients were performed on formulations containing 4% to 10% lauramide DEA, as 0.25% to 1.25% solutions. Lauramide DEA was not a sensitizer in any of the studies. 2
Linoleamide DEA
In an RIPT conducted with 100% linoleamide DEA on 100 patients, no irritation or sensitization reactions were observed. A dandruff shampoo containing 1.5% linoleamide DEA, tested as a 1% aq solution in a RIPT using 101 patients, was an irritant but not a sensitizer. 2
Provocative Testing
Cocamide DEA
Metalworkers with dermatitis were patch tested with 0.5% cocamide DEA in pet. 37 The patches were applied for 1 to 2 days. Of the 215 patients, 1 (0.5%) had a positive reaction on day 3.
Coreactivity
Cocamide DEA
Thirty-five patients that had positive patch tests to cocamidopropyl betaine, amidoamine, or both, were tested for coreactivity with cocamide DEA. 38 Two (5.7%) of the patients had positive reactions to cocamide DEA.
Case Studies
Cocamide DEA
In all, 1 patient with dermatitis on the hands and face, and 2 with dermatitis on the hands and forearms, were patch tested using the North American Contact Dermatitis Group standard tray and supplemented with additional chemicals. 39 All the 3 patients had either personal or industrial exposure to cocamide DEA-containing products. All 3 had positive patch test results (2+) to cocamide DEA, and 2 had reactions to several other chemicals. In all patients, the dermatitis cleared with avoidance of cocamide DEA-containing products.
Undecylenamide DEA
One patient with dermatitis of the hands and axillae had positive test reaction to a liquid soap. 40 Subsequent testing with 0.1% and 1% aq undecylenamide DEA, an ingredient in the soap, gave positive reactions. In 10 control patients, testing with 0.1% undecylenamide DEA was negative.
Phototoxicity/Photosensitization
Human
Lauramide DEA
A 10% solution of lauramide DEA, tested in 25 patients, was not phototoxic. In a photosensitivity study of 10% lauramide DEA, tested as a 1% aq solution in 25 patients, slight irritation was seen in 9 patients at induction and 4 at challenge. The test substance was not a photosensitizer. 2
Ocular Irritation
In Vitro
Cocamide DEA
A 10% solution of Cocamide DEA, classified as a nonirritant to minimal ocular irritant, was evaluated in the EpiOcular tissue model. The irritation classification, compared to the results of a Draize test, was similar to a nonirritant score obtained in the Draize test. 41
Myristamide DEA
When Myristamide DEA was evaluated in a neutral red assay, the IC50 values in Chinese hamster fibroblast V79 cells, rabbit corneal cells, and human epidermal keratinocytes were 15.2, 23.9, and 6.2 µg/mL, respectively. The DS20 (concentration predicted to produce a Draize score of 20/110) was 14.4% w/w myristamide DEA. 3
Non-Human
Cocamide DEA
A solution of >64% cocamide DEA and <29% DEA was a severe irritant in rabbit eyes. 1 In another study, a solution of Cocamide DEA, 30% in propylene glycol, was a mild eye irritant in rabbits. 2
Lauramide DEA
Five ocular irritation studies were performed in rabbits with lauramide DEA at concentrations of 1% to 25%. Lauramide DEA 1 % aq was mildly irritating, 5% was slightly to moderately irritating, 10% to 20% was moderately irritating, and 25% was moderately to severely irritating. One bubble bath formulation containing 6% lauramide DEA was practically nonirritating, while another was moderately irritating, and 3 shampoo formulations containing 8% lauramide DEA were nonirritating to moderately irritating. In a mucous membrane irritation test, a soap containing 10% lauramide DEA was significantly more irritating than water to vaginal mucosa of rabbits. 2
Stearamide DEA
A mixture containing 35% to 40% stearamide DEA was not irritating to rabbit eyes. 3
Isostearamide DEA
A formulation containing 8.0% isostearamide DEA was a moderate irritant in rabbit eyes. 3
Oleamide DEA
Undiluted oleamide DEA was practically nonirritating to rabbit eyes. 2
Linoleamide DEA
An aq solution (10 %) administered to rabbit eyes was practically nonirritating, and an undiluted solution was minimally to moderately irritating. A product containing 1.5% linoleamide DEA, applied as a 25% aq solution, and a formulation containing 15% linoleamide DEA were moderate eye irritants in rabbits, while a formulation containing 15% linoleamide DEA, applied as a 25% aq solution, was mildly irritating. 2
Ricinoleamide DEA
Undiluted PEG-20 glyceryl ricinoleate + ricinoleamide DEA (amount present was not stated) was evaluated for ocular irritation using NZW rabbits. 34 No signs of irritation were observed, and the surfactant was a nonirritant.
Summary
This safety assessment includes 33 DEAs as used in cosmetics. Information on some of these ingredients reviewed previously by CIR is included here to fill noted gaps in the available safety data and to create a report on the complete family of ingredients. Cocamide DEA and most of the other diethanolamides are reported to function in cosmetic formulations as a surfactant foam booster or a viscosity increasing agent, although a few are reported to function as a hair and skin conditioning agent, surfactant-cleansing or emulsifying agent, or an opacifying agent.
The DEAs consist of covalent, tertiary amides where 2 of the nitrogen substituents are ethanol (or at least an ethanol residue) and the third is a carbonyl-attached substituent. These ingredients are not salts and do not readily dissociate in water. Amidases, such as fatty acid amide hydrolase which is known to be present in human skin, could potentially convert the diethanolamides to DEA and the corresponding fatty acids. The yield of DEA from metabolism of diethanolamides in human skin is unknown.
The diethanolamides generally have some amount of free DEA, and that amount can vary greatly by ingredient. For example, in the NTP studies, it was estimated that oleamide DEA contained 0.19% free DEA, while cocamide DEA contained 18.2% free DEA by weight.
The VCRP data obtained in 2011 indicate that cocamide DEA is used in 710 cosmetic formulations, the majority of which are rinse-off formulations. With the exception of lauramide DEA, which is reported to be used in 281 cosmetic formulations, the remaining diethanolamides have less than 35 uses, and most are not reported to be used. The reported concentration of use of the diethanolamides ranges from 0.2% to 12%; the greatest leave-on concentration reported was 9%. Fatty acid dialkanolamides are allowed for use in products in Europe with restrictions; the restrictions address secondary amine content.
[14C]Lauramide DEA partitioned well into rat and human liver slices, and the absorbed radioactivity was mostly unchanged lauramide DEA. In the media, 18% to 42% of the radioactivity was present in the form of metabolites. Using microsomes to compare hydroxylation, lauramide DEA 12-hydroxylase activity in human liver microsomes was similar to that in rat liver microsomes, but 3 times the rate observed in rat kidney microsomes.
Mice and rats were exposed dermally to 5 to 800 mg/kg and 25 or 400 mg/kg [14C]lauramide DEA, respectively. Absorption in rats was similar for each dose when calculated as a percentage of dose, and absorption was greater in mice (50%-70% of the applied dose) than in rats (20%-24%). The parent compound and the half-acid amide metabolites were detected in the plasma of rats. Repeated application of 25 mg/kg/d lauramide DEA did not appear to affect absorption or excretion. In rats dosed orally with 1000 mg/kg [14C]lauramide DEA, 4% of the dose was recovered in the tissues and 79% in the urine after 72 hours; at 6 h, no DEA, DEA metabolites, or unchanged lauramide DEA were found in the urine; only very polar metabolites were found. With iv dosing, a 50 mg/kg dose of lauramide DEA was quickly metabolized and eliminated by mice; approximately 95% of the dose was excreted in the urine in 24 hours. More than 80% of a 25 mg/kg dose was excreted in the urine by rats in 24 hours.
Acute dermal testing with undiluted cocamide and lauramide DEA, 50% lauramide DEA, and undiluted and 10% aq linoleamide DEA and acute oral testing with several fatty acid diethanolamides did not result in notable toxicity. In an acute inhalation toxicity study with 86 to 219 mg/m3 tallamide DEA in rats, low concentration produced sensory and pulmonary irritation. The LC50 value was >219 mg/m3.
In repeated dose dermal studies with cocamide, lauramide, and oleamide DEA in mice and/or rats, irritation was observed at the site of application. Increases in liver and kidney weights were observed in most studies, while decreases in bw were observed sporadically. The incidence of renal tubule regeneration was greater in female rats dosed with 100 to 400 mg/kg cocamide DEA when compared to controls. A formulation containing 3% linoleamide DEA was not a cumulative systemic toxicant in a 13-week dermal study; dermal irritation was observed.
With repeat oral dosing of lauramide DEA, the NOEL was 0.1% in feed in a study with SPF rats and 250 mg/kg/d in a feeding study using Wistar rats. The NOEL for Beagle dogs fed lauramide DEA for 12 weeks was 5000 ppm.
In a developmental toxicity study in Sprague-Dawley rats, the NOAEL for maternal toxicity and developmental toxicity was 1000 mg/kg/d that was the highest dose tested. No other data on the reproductive and developmental toxicity of the diethanolamides were found. Available reproductive and developmental toxicity data on DEA and some of the fatty acids from previous CIR reports show no significant toxic effects noted. For DEA, the NOEL for embryonal/fetal toxicity with dermal application was 380 mg/kg/d for rats and 350 mg/kg/d for rabbits. In one oral study, the NOEL for embryonal/fetal toxicity was 200 mg/kg/d in rats, and in another, the LOAEL for both maternal toxicity and teratogenicity was 125 mg/kg/d in rats. In an inhalation study, in rats, the NOAEC for both maternal and developmental toxicity was 0.05 mg/L, and the NOAEC for teratogenicity was >0.2 mg/L.
Cocamide DEA, lauramide DEA, and oleamide DEA were, generally, nongenotoxic in a number of assays. There was an increase in the frequency of micronucleated erythrocytes in mice by cocamide DEA and the induction of SCEs in CHO cells by lauramide DEA.
The carcinogenic potential of dermally applied cocamide, lauramide, and oleamide DEA was evaluated in B6C3F1 mice and F344/N rats in an NTP study. Cocamide DEA produced carcinogenic activity (hepatic and renal tubule neoplasms) in male and female mice (100-200 mg/kg), equivocal evidence (renal tubule neoplasms) in female rats (50-100 mg/kg), and no evidence in male rats (50-100 mg/kg). Lauramide DEA produced evidence of carcinogenic activity (hepatocellular neoplasms) in female mice (100-200 mg/kg), and no evidence in male mice (100-200 mg/kg) or male and female rats (50-100 mg/kg). Oleamide DEA produced no evidence of carcinogenic activity in male or female mice (15-30 mg/kg) or male or female rats (50-100 mg/kg).
The dermal irritation of fatty acid diethanolamides, in non-human and human testing, varied greatly with formulation and test conditions. Lauramide DEA and linoleamide DEA were not sensitizers in humans. Cocamide DEA, 0.01% to 10%, produced positive results in provocative sensitization studies. Lauramide DEA was not phototoxic in humans. The ocular irritation of fatty acid also varied greatly with formulation and test conditions.
Discussion
The CIR Expert Panel agreed to reopen the review of cocamide DEA, and add 32 similar diethanolamides. Some of the ingredients included in this rereview, specifically isostearamide DEA, lauramide DEA, linoleamide DEA, myristamide DEA, oleamide DEA, and stearamide DEA, have been reviewed by the CIR in the past. Although the Panel noted gaps in the available safety data for many of the diethanolamides included in this group, the Panel was able to extrapolate the existing data, including the data from previous CIR assessments as well as recently published data, to support the safety of all the diethanolamides included in this safety assessment. Similar structure–activity relationships and functions made that extrapolation feasible.
The Panel expressed concern about the lack of reproductive and developmental toxicity data for most of the diethanolamides. Since DEA may be present as an impurity in the diethanolamides, and because amidases in the skin might convert some of the diethanolamides to DEA and the corresponding fatty acid, the Panel determined that data from the CIR safety assessment on DEA as well as from assessments on the other “components” was applicable. The lack of reproductive toxicity for DEA or any of the components alleviated this concern.
The Panel was also concerned with levels of free DEA that could be present as an impurity in diethanolamides. The Panel reasoned that the “clear evidence of carcinogenic activity” of cocamide DEA reported for male and female mice and the “equivocal evidence of carcinogenic activity” of cocamide DEA reported in female rats, as well as “some evidence of carcinogenic activity” of lauramide DEA in female mice, was due to the presence of free DEA. This opinion was supported by carcinogenicity studies showing that the level of carcinogenic activity in cocamide DEA, lauramide DEA, and oleamide DEA corresponded to the amount of free DEA found in the test substance. The Panel stated that the amount of free DEA available in diethanolamides must be limited to the present practices of use and concentration of DEA itself. The Panel was also concerned that free DEA present as an impurity in the diethanolamides could be converted (nitrosated) into N-nitrosamines that may be carcinogenic. Consequently, they recommended that diethanolamides should not be used in cosmetic products in which N-nitroso compounds can be formed.
Studies showed that products formulated using diethanolamides are potential dermal irritants. The Expert Panel specified that products must be formulated to be nonirritating.
Because some of the ingredients named in the assessment can be used in products that may be sprayed, the Panel discussed the issue of potential inhalation toxicity. In the absence of sufficient safety test data to evaluate this end point directly, the Panel considered other data that were available to characterize the potential for the diethanolamides to cause systemic toxicity, ocular or dermal irritation or sensitization, and other effects. The Panel noted that 95% to 99% of particles produced in cosmetic aerosols are not respirable. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, this information suggested that inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects.
Conclusion
The CIR Expert Panel concluded that the following 33 diethanolamides are safe in the present practices of use and concentration described in this safety assessment (ingredients not in current use are identified with *), when formulated to be nonirritating, and when the levels of free DEA in the diethanolamides do not exceed the present practices of use and concentration of DEA itself. The Expert Panel cautions that ingredients should not be used in cosmetic products in which N-nitroso compounds can be formed.
Almondamide DEA*
Apricotamide DEA*
Avocadamide DEA*
Babassuamide DEA*
Behenamide DEA*
Capramide DEA
Cocamide DEA
Cornamide DEA*
Cornamide/Cocamide DEA*
Hydrogenated Tallowamide DEA*
Isostearamide DEA
Lanolinamide DEA*
Lauramide DEA
Lauramide/Myristamide DEA
Lecithinamide DEA*
Linoleamide DEA
Minkamide DEA*
Myristamide DEA
Oleamide DEA
Olivamide DEA*
Palm Kernelamide DEA
Palmamide DEA*
Palmitamide DEA*
Ricebranamide DEA*
Ricinoleamide DEA*
Sesamide DEA*
Shea Butteramide/Castoramide DEA*
Soyamide DEA
Stearamide DEA
Tallamide DEA*
Tallowamide DEA*
Undecylenamide DEA*
Wheat Germamide DEA
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
