Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel assessed the safety of silica silylate, silica dimethyl silylate, trimethylsiloxysilicate, and trifluoropropyldimethyl/trimethylsiloxysilicate as used in cosmetics. These silylates and surface-modified siloxysilicates function in cosmetics as antifoaming agents, anticaking agents, bulking agents, binders, skin-conditioning agents—emollient, skin-conditioning agents—occlusive, slip modifiers, suspension agents—nonsurfactant, and viscosity increasing agents—nonaqueous. The Expert Panel reviewed the available animal and clinical data as well as information from a previous CIR safety assessment of amorphous silica. The CIR Expert Panel concluded that silica silylate, silica dimethyl silylate, trimethylsiloxysilicate, and trifluoropropyldimethyl/trimethylsiloxysilicate are safe as used when formulated and delivered in the final product not to be irritating or sensitizing to the respiratory tract.
Keywords
Introduction
This safety assessment addresses the use of silica silylate, silica dimethyl silylate, trimethylsiloxysilicate, and trifluoropropyldimethyl/trimethylsiloxysilicate in cosmetics. These ingredients function in cosmetics as: antifoaming agents, anticaking agents, bulking agents, binders, skin-conditioning agents—emollient, skin-conditioning agents—occlusive, slip modifiers, suspension agents—nonsurfactant, and viscosity increasing agents—nonaqueous.
Amorphous silica, which is the core of silica silylate and silica dimethyl silylate, has been reviewed by the Cosmetic Ingredient Review (CIR) Expert Panel and was found to be safe as a cosmetic ingredient in the practices of use and concentrations as described in that safety assessment. 1
The ingredients in this safety assessment are also based on amorphous (synthetic amorphous silica and silicates) not crystalline silica. The ingredients in this safety assessment are organosilane hybrid materials, modified to have desired properties for their use in cosmetics.
Data on silane, dichlorodimethyl-, reaction products with silica (CAS No. 68611-44-9) are also included in this literature review, since these chemicals are the same as silica dimethyl silylate. Data from a mixture, siloxanes and silicones, di-Me, and hydroxyl-terminated (as Antifoam M), were also included because they are also considered relevant.
Definition and Structure
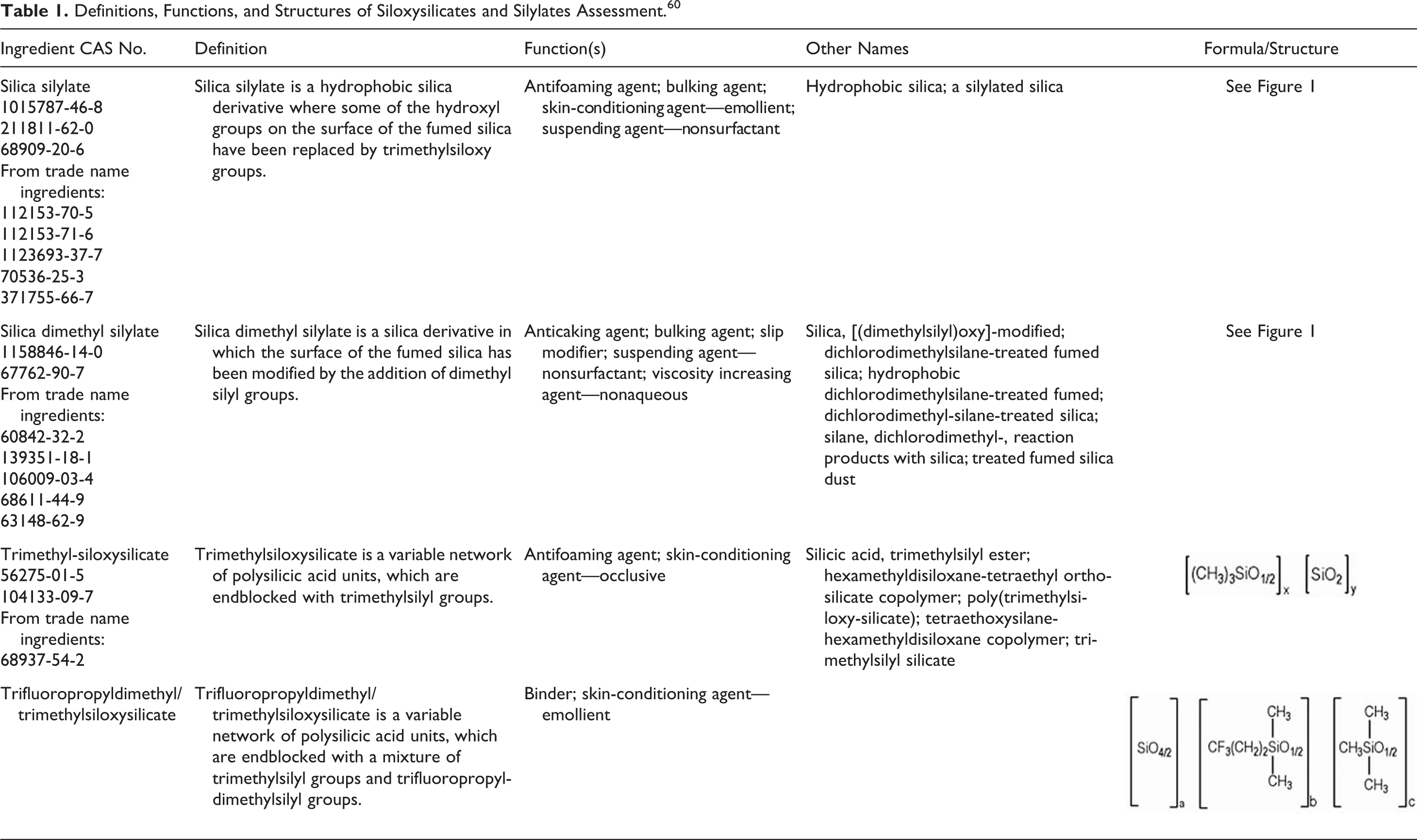
The cosmetic ingredient definitions, functions in cosmetics, and structures of silica silylate, silica dimethyl silylate, trimethylsiloxysilicate, and trifluoropropyldimethyl/trimethylsiloxysilicate are provided in Table 1. These 4 hybrid silica materials can be divided into 2 distinct types, grafted, and cocondensed.
Definitions, Functions, and Structures of Siloxysilicates and Silylates Assessment. 60
Grafted Silica Materials
These materials consist of silica particles that are surface modified by organosilanes. For example, dichlorodimethylsilane is used to produce dimethyl silyl groups on the surface of a particle of fumed silica. 2 Silica silylate and silica dimethyl silylate are grafted silica materials. Silica silylate consists of fumed silica, surface modified with trimethylsilyl groups (Figure 1).
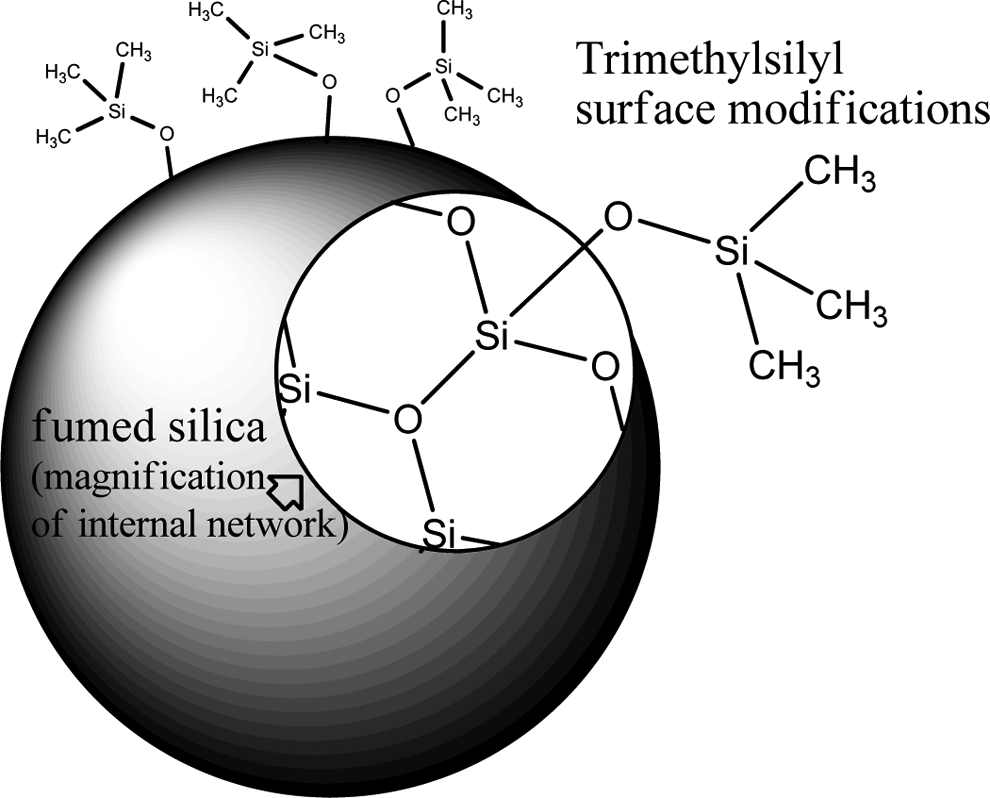
Trimethylsilyl surface modifications.
Cocondensed Silica Materials
In contrast to grafted materials, cocondensed silica materials are not surface-modified silica particles. Instead, cocondensed materials are prepared by the simultaneous reaction of condensable inorganic silica and silylated organic compounds. 2 This process is similar to random copolymer synthesis but is nonlinear. Trimethylsiloxysilicate and trifluoropropyldimethyl/trimethylsiloxysilicate are cocondensed silica materials. Trimethylsiloxysilicate is the cohydrolysis product of a tetraalkoxysilane and a trimethylalkoxysilane. 3 The chemical structure of trimethylsiloxy silicate can be visualized as a 3-dimensional network of polysilicic acid units (resultant from the tetraalkoxysilane), which are endblocked with trimethylsilyl groups (Figure 2).
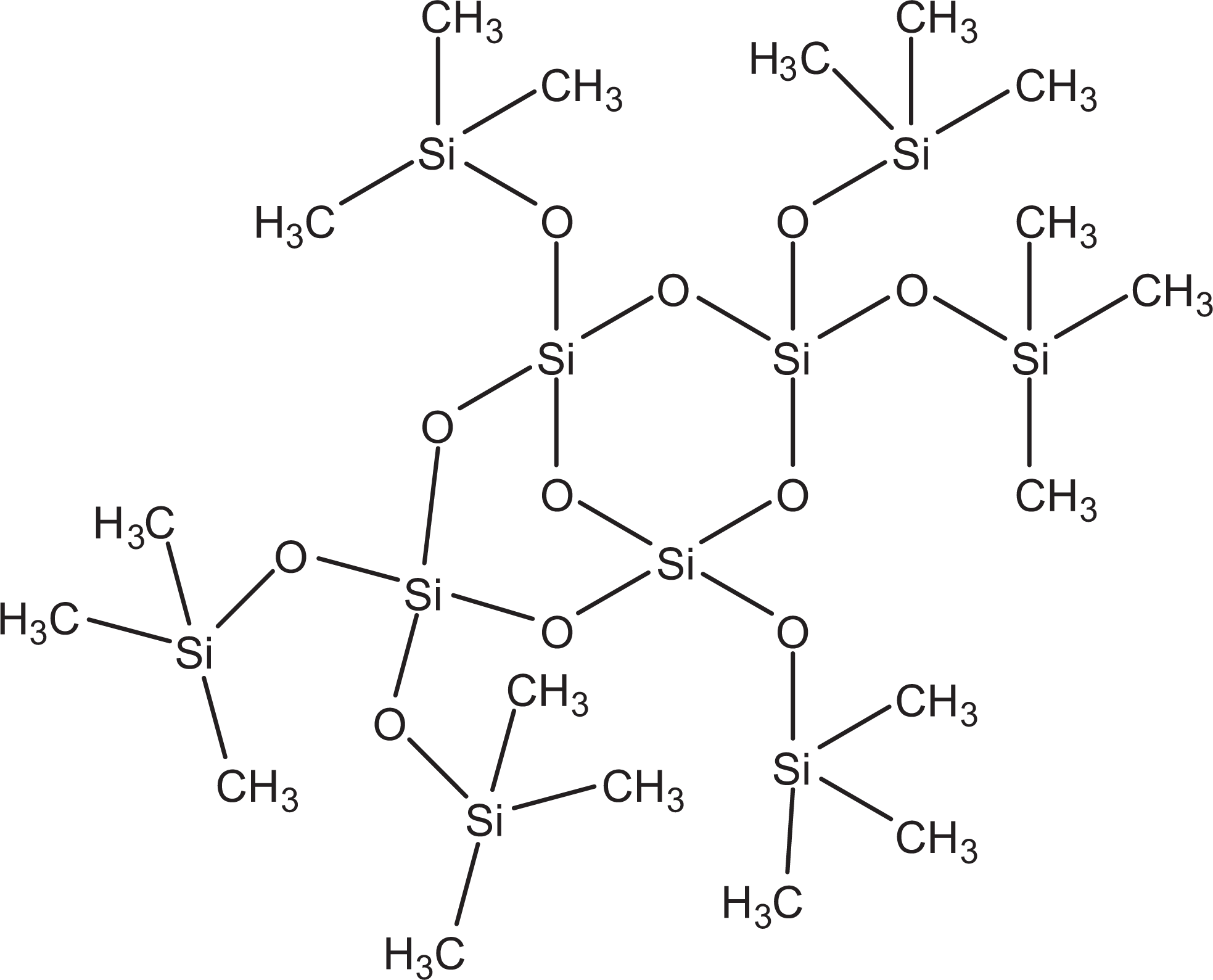
Cocondensed silica.
Trifluoropropyldimethyl/trimethylsiloxysilicate differs from trimethylsiloxy silicate only by the replacement of some of the methyl groups with trifluoropropyl groups.
Physical and Chemical Properties
These ingredients are amorphous solids, with virtually no water solubility. The water solubility of grafted silica materials is below 10e−6 g/L. Chemical and physical properties of silica dimethyl silylate and trimethylsiloxysilicate are provided in Table 2. Silica silylate was stable for 24 months when stored at or below 40°C. 4
Physical and Chemical Properties of Silica Dimethyl Silylate and Trimethylsiloxysilicate.
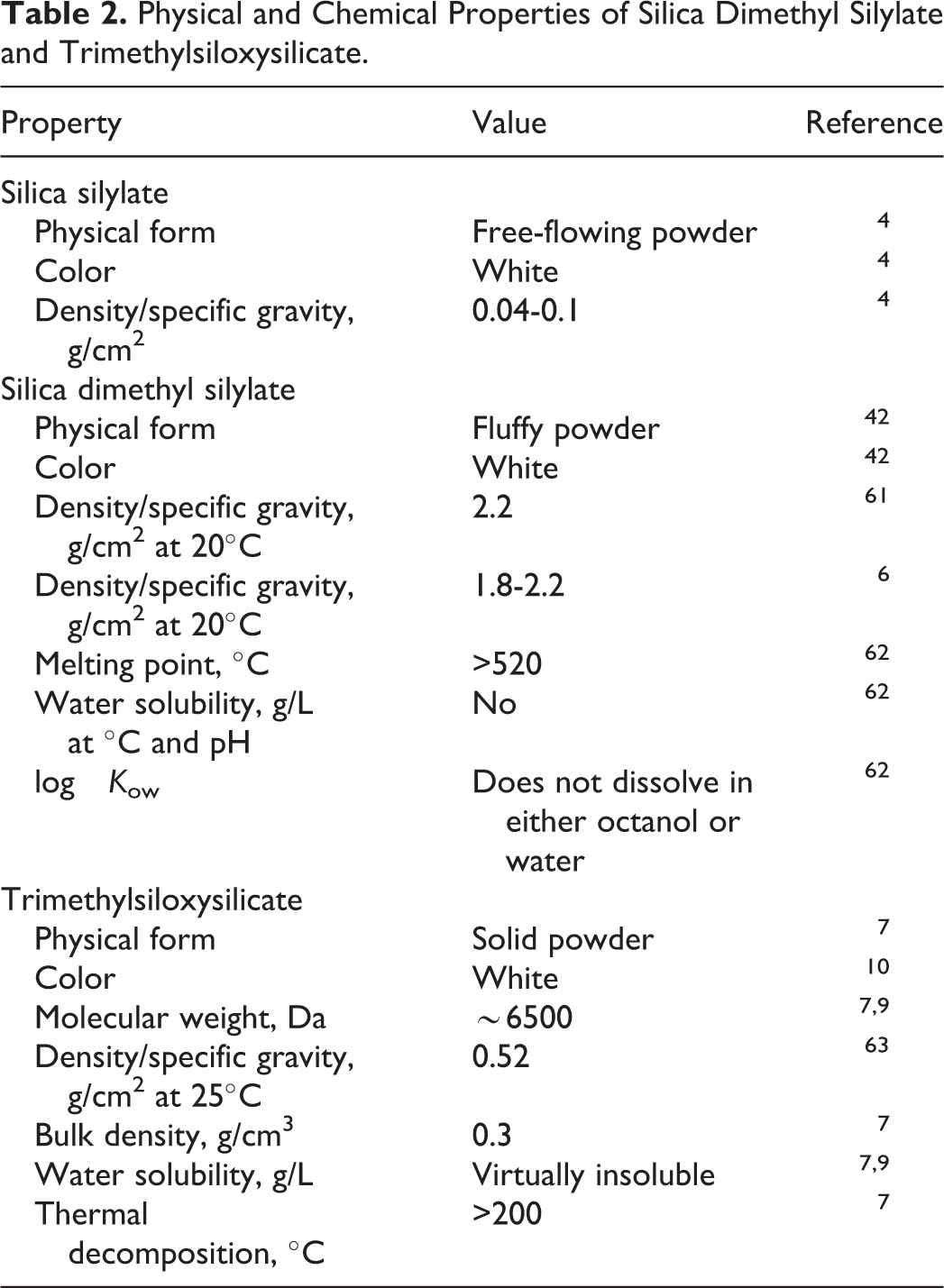
One manufacturer reported that less than 0.83% of silica dimethyl silylate had a particle size of <125 µm, and none were <90 µm. 5 The Synthetic Amorphous Silica and Silicate Industry Association has suggested that this is true industry wide. 6
It was reported in a material safety data sheet that trimethylsiloxysilicate dissolves in organic and silicone oils up to the concentrations of 50%. 7 No special storage measures are required for trimethylsiloxysilicate when stored at or below 32°C in an unopened container for 24 months; thus, it is considered stable. This manufacturer reported that trimethylsiloxysilicate has a bulk density of ∼0.3 g/cm 3 and goes through thermal decomposition at >200°C. 7 The particle size is ∼10 µm. Two other manufacturers reported that the average particle size is 10 µm or ranged from 20 to 100 µm. 8,9
Trimethylsiloxysilicate releases formaldehyde vapors when heated above 150°C in the presence of air. 10 Trifluoropropyldimethyl/trimethylsiloxysilicate was stable after five 2-day cycles of −10°C and 45°C for 24 hours at each temperature. 11 This ingredient was also stable after 3 months storage at 45°C.
Physical and chemical properties were not discovered for silica silylate.
Analytical Methods
The presence of silylate particles may be quantified and counted by a scanning mobility particle sizer. 12 Gas chromatography (GC) was used to identify fluorine compounds in trifluoropropyldimethyl/trimethylsiloxysilicate. 13 Samples of trifluoropropyldimethyl/trimethylsiloxysilicate were analyzed for stability using infrared and nuclear magnetic resonance. 11
Impurities
A manufacturer reported that trimethylsiloxysilicate was >99% pure. 7 Benzene and toluene may be present at <0.0001% after an extensive drying step. Another manufacturer reported the only impurity to be alkanes (C7-10-iso) at a maximum of 0.35%, present as a residual solvent from the production process. 9
Analysis of trifluoropropyldimethyl/trimethylsiloxysilicate by GC showed that the product does not contain trifluoropropene or initial manufacturing materials. 13
Ultraviolet Absorption
Although no data were available, the ingredients included in this review would not be expected to have any significant ultraviolet (UV) absorption, because these materials do not contain any of the functional groups commonly associated with UV absorption.
Method of Manufacture
Grafted materials such as silica silylate and silica dimethyl silylate can be manufactured via reaction of a fumed silica particle with one of an alkoxysilane (eg, (CH3O)3SiCH3), a halosilane (eg, ClSi(CH3)3), or an alkylsilazane (eg, NH[Si(CH3)3]2). 2 For example, amorphous silica can be modified by reaction with hexamethyldisilazane (HMDS), in hexanes at 275°C and 30 atm, to manufacture silica silylate. 14 Solvents are removed by heated evaporation. The degree of surface modification can be adjusted by varying the concentration of the silylating agent (eg, increasing the amount of HMDS).
Cocondensed silica materials can be manufactured via the cohydrolysis of a tetraalkoxysilane (eg, tetraethoxysilane, which result in the inorganic silane groups in the reaction product) and a trialkylalkoxysilane (eg, trimethylethoxysilane, which will result in the organosilane groups in the reaction product). 2 Solvents are removed by heated evaporation. Some residual alkoxyl (ie, leaving groups that did not leave; Si-OR) and hydroxyl (Si-OH) functional groups are likely to be present. 15 The average molecular weight (MW) can be adjusted by varying the ratio of the silanes.
Use
Cosmetic
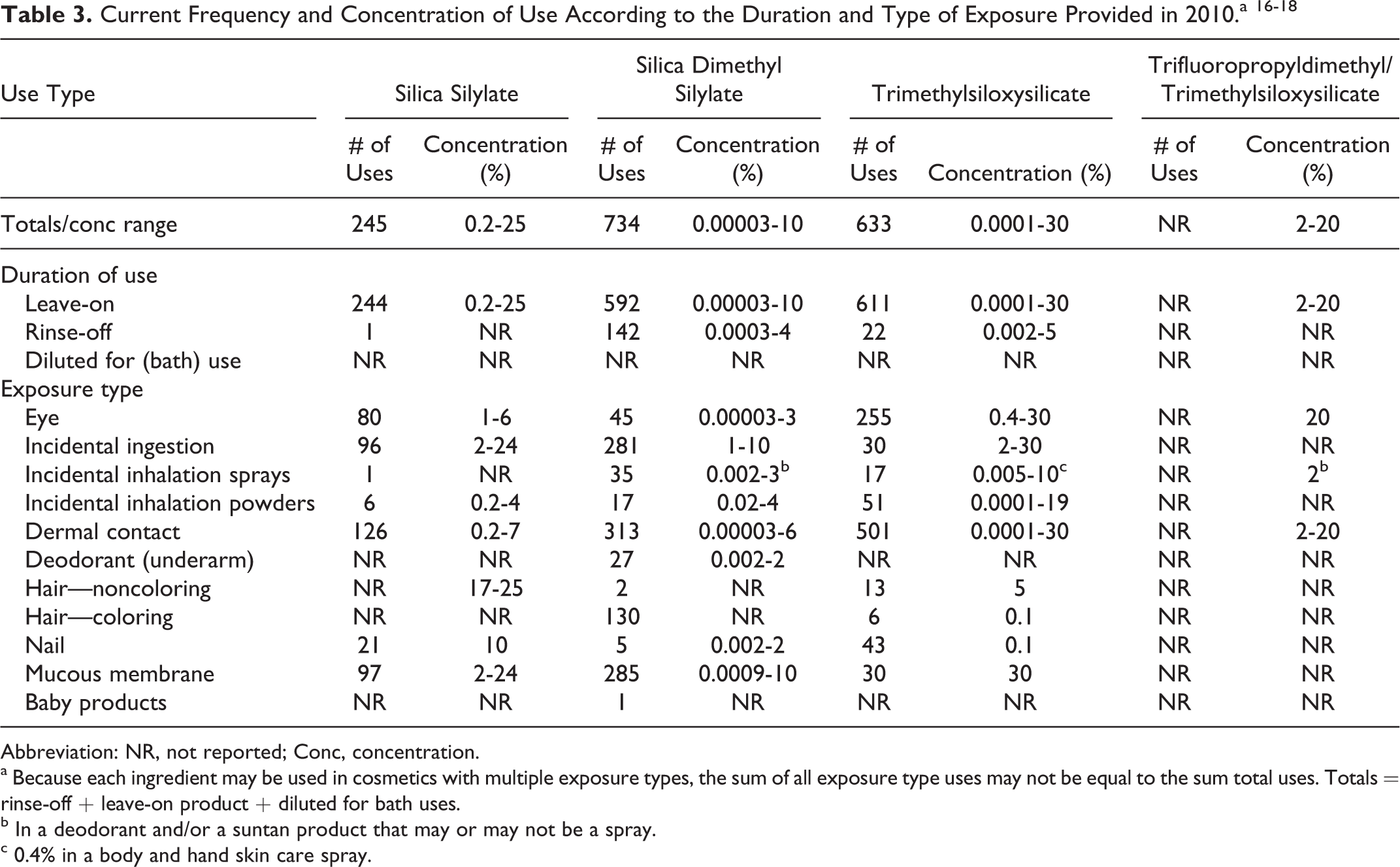
According to the Voluntary Cosmetic Registration Program administered by the Food and Drug Administration (FDA), the total number of reported uses of silica dimethyl silylate was 734 (592 leave-on and 142 rinse-off products). 16 A survey conducted by the Personal Care Products Council (Council) found that silica dimethyl silylate was used at 0.00003% to 10% in leave-on products (highest concentration in lipsticks) and 0.0003% to 4% in rinse-off products (highest concentration in personal cleanliness products; Table 3). 17 There were 633 reported uses of trimethylsiloxysilicate at 0.0001% to 30% in leave-on products (highest in eyeliner and lipsticks) and 0.002% to 5% in rinse-off products (highest in hair straighteners). There were 245 reported uses of silica silylate (244 in leave-on and 1 rinse-off product) at 0.2% to 25% in leave-on products. 18 There were no reported uses of trifluoropropyldimethyl/trimethylsiloxysilicate to FDA, but the Council reported use at 2% to 20% in leave-on products (highest use concentration was in eyeliners).
Abbreviation: NR, not reported; Conc, concentration.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not be equal to the sum total uses. Totals = rinse-off + leave-on product + diluted for bath uses.
b In a deodorant and/or a suntan product that may or may not be a spray.
c 0.4% in a body and hand skin care spray.
Silica dimethyl silylate is reportedly used in perfumes. This product category may include products that are aerosolized or used as powders. In practice, 95% to 99% of the aerosols released from cosmetic sprays have aerodynamic equivalent diameters in the 10 to 110 µm range. 19,20 Therefore, most aerosols incidentally inhaled from these sprays are deposited in the nasopharyngeal region and are not respirable. 21,22 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic diameters in the range considered to be respirable. 23 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Noncosmetic Use
The FDA has approved silicon dioxide (a compound similar to silica dimethyl silylate) to be used as a direct food additive as an anticaking agent up to 2% and in the manufacture of materials that come in direct contact with food in various production, manufacturing, packaging, preparing, transporting, and holding operations. 24
Silica aerogel (an amorphous silica gel) is generally recognized as safe (GRAS) in dietary supplements. 25
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Oral
Silica Dimethyl Silylate
Antifoam M (siloxanes and silicones, di-me, and hydroxyl-terminated; 0.5 mg/kg) or silica (6 mg/kg) was orally administered in sesame oil to male Buckberg mice (n = 12) after fasting. 26 Controls were administered sesame oil (0.5 mL). There was no increase in urinary and biliary silicon in both the groups. The authors suggested that the source was organosoluble silicon rather than inorganic silica.
Antifoam M (21.8 or 41.8 mg/kg) was labeled using randomly radiolabeled [ 14 C] polydimethylsiloxane and orally administered to rhesus monkeys (n = 5) that were then observed for 7 days. 26 Antifoam M was expired in the breath (0.1%-0.2%) and in the urine (0.22%) with a half-life of 24 hours. There was 0.1% to 0.9% in the bile in the first 24 hours after dosing. Over 92 hours, 93% to 97% of the dose was recovered in the feces. There was < 0.01% detected in ∼40 tissues examined in one monkey necropsied after 7 days. A range of 93% to 98% was recovered in the feces.
Human patients were orally administered Antifoam M (100 mg/kg; n = 6) after 5 days of a consistent diet (that continued through the rest of the experiment) on day 6 and data collected through day 7. 26 Increased silicon levels were not detected in urine, feces, or expelled air after oral administration of Antifoam M.
Inhalation
Silica Dimethyl Silylate
Rats (n = 40) were exposed to aerosolized silica dimethyl silylate (200 mg/m3; particle size not provided) for 5 h/d for 3 days. 27 At 24 hours after the last exposure, there was 0.91 mg test substance in the lung and none at 1 month post-exposure. There was 0.383, 0.239, and 0.173 mg in the mediastinal lymph nodes at 1, 2, and 3 months, respectively. At 3 months, 81% of the test substance had been eliminated.
Female Sprague Dawley rats (n = 50) were exposed to aerosolized silica dimethyl silylate (50 mg/m3; <7 µm) for 5 hours. 15 At necropsy, silica was deposited in the lungs (0.156, 0.034, and 0.034 mg at 20 hours, 1, and 3 months, respectively) and mediastinal lymph nodes (0, 0.003, and 0.004 mg). The test substance was eliminated at 78% and 85% at 1 and 3 months, respectively.
Female Sprague Dawley rats (n = 30) were exposed to aerosolized silica dimethyl silylate (50 mg/m3; <7 µm) for 5 hours for 3 days. 15 At necropsy, silica deposited in the lungs (0.34, 0.085, and 0.30 mg at 20 hours, 1, and 3 months, respectively) and mediastinal lymph nodes (0.34, 0.085, and 0.30 mg). Test substance was eliminated at 75% and 92% at 1 and 3 months, respectively.
Other
Silica particles (4 nm) placed in simulated physiological conditions dissolve completely in ∼32 hours. 28
Toxicological Studies
Acute Toxicity
Dermal—Non-Human
Silica Dimethyl Silylate
Silica dimethyl silylate (2000 mg/kg in propylene glycol) applied in a single dose to the skin of Wistar rats (n = 5/sex) for 24 hours caused no mortality. 29 No clinical signs were observed, and necropsies were unremarkable.
Trimethylsiloxysilicate
Trimethylsiloxysilicate (100%; 0.5 g) was administered to the intact skin of New Zealand White rabbits (n = 6) under occlusion for 4 hours. 30 All rabbits survived and gained weight during the study. There were no signs of toxicity.
Trimethylsiloxysilicate (2 g/kg) was administered to the shaved skin of New Zealand White rabbits (n = 10) under occlusion for 24 hours. 30 The patch was then removed, and the skin was rinsed in corn oil. The rabbits were observed for 14 days. All rabbits gained weight. There were no signs of toxicity. There were no abnormalities observed at necropsy.
Oral—Non-Human
Silica Dimethyl Silylate
The oral median lethal dose (LD50) of silica dimethyl silylate was >5000 mg/kg for Sprague Dawley rats. 31,32 Another study in rats found the oral LD50 to be >7900 mg/kg. 33
Trimethylsiloxysilicate
Trimethylsiloxysilicate (5 g/kg in corn oil) was orally administered to Sprague Dawley rats (n = 5/sex). 30 There were no clinical signs. All rats gained weight. There were no lesions at necropsy. The observation time was not provided.
The oral LD50 of trimethylsiloxysilicate was reported to be >1 g/kg in mice. 34
Trifluoropropyldimethyl/trimethylsiloxysilicate
The oral LD50 of trifluoropropyldimethyl/trimethylsiloxysilicate was reported to be >2 g/kg in mice. 35
Inhalation—Non-Human
Silica Dimethyl Silylate
Inhalation toxicity studies using rats are presented in Table 4. There was no mortality up to 520 mg/m3.
Inhalation Studies of Silica Dimethyl Silylate and Silica Silylate.

Intraperitoneal—Non-Human
Silica Dimethyl Silylate
Silica dimethyl silylate (up to 30 mg in water with Tween 80) was administered intraperitoneally (ip) to mice (n = 120; strain and sex not provided). 36 All mice survived treatment. The observation period was not provided, but the report stated that at necropsy fibrosis was not observed, although thickening of the liver and spleen capsules was observed. Histopathology showed that the test substance was found in the abdominal cavity in a tight network of reticulin and collagen. Slight phagocyte accumulations and necrosis were observed. Histopathology of the liver showed some evidence of the test substance there, also in a tight network of reticulin and collagen.
Silica dimethyl silylate (up to 200 mg in water with 0.5% Tween) was administered ip to female rats (n = 100; strain not provided) as described above. 36 All rats survived treatment. At necropsy, there was no fibrosis observed. Histopathology showed that the test substance was found in the abdominal cavity in a tight network of reticulin and collagen. Slight phagocyte accumulations and necrosis were observed. Histopathology of the liver showed some evidence of the test substance there, also in a tight network of reticulin and collagen.
Ocular—Non-Human
Silica dimethyl silylate (0.1-0.2 g; 0.1 mL) was applied to one eye of New Zealand white rabbits (n = 3/sex). 37 Three of the treated eyes were not rinsed, and 3 were rinsed with saline after 20 to 30 seconds. Two females had decreased feed consumption as well as soft stool, anogenital staining, and reduced fecal volume.
Repeated Dose Toxicity
Oral—Non-Human
Silica Dimethyl Silylate
Silica dimethyl silylate (500 or 1000 mg/kg) was orally administered to Wistar rats (n = 40/sex) by gavage for every other day for 19 or 39 days. 38 Rats were killed and necropsied at the end of the treatment period or after 4 weeks of recovery. There were no clinical signs or treatment effects observed. The no-observed adverse effect level (NOAEL) was 1000 mg/kg.
Silica dimethyl silylate (0, 500, 1000, and 2000 mg/kg; the high-dose groups was gradually increased to 4000, 8000, and 16 000 mg/kg) was orally administered to Wistar rats (n = 40/sex) in feed daily for 5 or 8 weeks for the high dose. 39 Rats were killed and necropsied at the end of the treatment period. In all, 2 males and 2 females in the high-dose group died after 9 and 13 days of exposure to 16 000 mg/kg. Clinical signs in the high-dose group after increasing the dose to 16 000 mg/kg were apathy and decreased grooming activity. Cachexia and hemorrhagic mucosa of the nose and eyes were observed prior to death. There was severe body weight decrease in males and females following 1 week exposure to 8000 mg/kg and exposure to 16 000 mg/kg. Feed consumption severely decreased following exposure to 16 000 mg/kg in males and females. There was hemorrhage in the mucous membranes of the eyes and nose in animals exposed to 16 000 mg/kg. In 2 females of the mid-dose group and 8 animals of the high-dose group, atrophic hepatocytes with decreased appearance [sic] and decreased glycogen contents of the cytoplasm were observed. Under the conditions of this study, the NOAEL was 500 mg/kg and the lowest observed adverse effect level (LOAEL) was 1000 mg/kg.
Silica dimethyl silylate (0 and 500 mg/kg) was orally administered to Wistar rats (n = 40/sex) in feed daily for 6 months. 40 Rats were killed and necropsied at the end of the treatment period or after 3 weeks recovery. There were no clinical signs or treatment effects observed. The NOAEL was 500 mg/kg.
Silica dimethyl silylate (100 mg/kg) was orally administered to Wistar rats (n = 20/sex) in feed daily for 24 months. 41 Rats were killed and necropsied at the end of the treatment period or after 3 weeks recovery. There were no clinical signs or treatment effects observed. The NOAEL was 100 mg/kg.
Inhalation—Non-Human
Silica Dimethyl Silylate
Repeat-dose inhalation studies from 1 week to 1 year are presented in Table 4. In rats, clinical signs included crusty eyes, muzzle, and nose; crust around ear tags; closed eyes; irregular breathing; irritable disposition; lacrimation and salivation; scabs; and red- and yellow-/brown-stained fur. At 2 weeks, there was an increase in lymphocytes and neutrophils. Reduced body weights were observed. Silica was deposited in the lungs and lymph nodes, but the deposits cleared over time. At necropsy, focal bronchiolar mucus proliferation, intraluminal mucus deposition, granulomata, focal increased septal cellularity, and accumulation of alveolar macrophages were observed in the lungs. One study noted slight necrosis or atrophy of the olfactory epithelium after a year at 35 mg/m3. Shorter exposure times did not generate remarks on the nasal area. A LOAEL of 31 mg/m3 was concluded in one study.
Reproductive and Developmental Toxicity
Silica Dimethyl Silylate
Silica dimethyl silylate (0, 500 mg/kg/d) was administered in feed to male (n = 2) and female (n = 10) Wistar rats for 6 months, during which the rats were mated twice, followed by a 3-week recovery period. 42 The offspring were observed through the 4-week lactation period then killed and necropsied. There were no mortalities attributable to treatment. There were no effects observed during treatment or at necropsy in the adults or the offspring. The NOAEL was 500 mg/kg.
Silica dimethyl silylate (0, 497, and 509 mg/kg/d) was administered to Wistar rats (n = 40/sex) in feed for 6 months, after which the rats were mated (1 male to 5 females). 43 The adult rats were killed and necropsied, and the offspring were observed for external appearance and development. No abnormalities were observed in either generation. The NOAEL was 497 mg/kg/d for parental generation.
Silica dimethyl silylate (0 and 100 mg/kg/d) was administered to Wistar rats (n = 20/sex) in feed for 24 months, after which the rats were mated (1 male to 5 females). 41 The offspring were adjusted to 5/sex in each litter and allowed to mature. After 7 months, they were mated, and their litters were also adjusted to 5/sex. Both sets of offspring were killed and necropsied. There were no reproductive toxicity effects observed.
Genotoxicity
In Vitro
Silica Dimethyl Silylate
In an Ames assay of a toluene extract of silica dimethyl silylate (15.8-5000 µg/plate) using Salmonella typhimurium (TA98, TA100, and TA1537) and Escherichia coli (WP2urvA) with and without metabolic activation, no mutagenicity was observed. 44 Controls had the expected results.
An Ames assay of a product (0-5000 µg/plate) containing silica dimethyl silylate (27%) was conducted using S typhilmurium (TAI535, TA1537, TA98, and TA100) and E coli (WP2 trp, WP2 trp uvrA) with and without metabolic activation. 45 The concentration was calculated to be 1250 µg/plate dimethyl silicones and siloxanes and 100 µg/plate dimethyl silicones and siloxanes reaction products with silica. The test substance was not mutagenic.
An Ames assay of silica dimethyl silylate (0-5000 µg/plate) was conducted using S typhimurium (TA98, 100, TA1535, and TA1538) with and without metabolic activation. 46 There was no evidence of mutagenicity. The controls had the expected results.
An Ames assay of silica dimethyl silylate (5-1580 µg/plate) was conducted using S typhlmurium (TA98, 100, TA1537, and TA1538) and E coli (WP2 trp uvrA) with and without metabolic activation. 47 There was no evidence of mutagenicity. The controls had the expected results.
An in vitro mammalian chromosome aberration test of silica dimethyl silylate (63, 125, 250, and 500 µg/mL) using Chinese hamster ovary (CHO) cells with and without metabolic activation was conducted. The frequency of effects without S9 was 0%, 1%, 0%, and 0% at 63, 125, 250, and 500 µg/mL, respectively, and 3%, 1%, 1%, and 3% with S9, respectively. 48 The author concluded that there was no evidence of genotoxicity. The controls had the expected results.
Trimethylsiloxysilicate
An Ames assay (0, 30, 80, 250, 700, 2000, and 5000 µg/plate) using S typhimurium (TA98, TA100, TA1535, and TA1537) and E coli (WP3uvrA) with and without metabolic activation was conducted. 30 There were no toxic effects, and the revertant frequencies were similar to controls. Positive controls had the expected results. The authors concluded that trimethylsiloxysilicate was nonmutagentic in this assay.
In an Ames assay, trimethylsiloxysilicate (156-500 µg/plate; MW: 3000-10 000) was not mutagenic to S typhimurium (TA98 and TA100) with or without metabolic activation. 34 In another Ames assay, trimethylsiloxysilicate (156-500 µg/plate; 3000-5000 in acetone) was not mutagenic to S typhimurium (TA98 and TA100) with or without metabolic activation.
An Ames assay of a mixture of trimethylsiloxysilicate (60%) and isododecane (40%; 156-5000 µg/plate) using S typhimurium (TA98, TA100) was negative. 49
In Vivo
There were no in vivo genotoxicity studies discovered for any of the ingredients in this safety assessment.
Carcinogenicity
Silica dimethyl silylate (100 mg/kg) was administered orally to Wistar rats (n = 20/sex) in feed daily for 24 months. 41 Rats were killed and necropsied at the end of the treatment period or after 3 weeks recovery. There were no carcinogenic effects observed. The nature and incidence of tumors were comparable with the historical control data.
Irritation and Sensitization
Irritation
Dermal—Non-Human
Silica Dimethyl Silylate
Silica dimethyl silylate (0.5 g moistened with tap water) was applied to the shaved skin of New Zealand white rabbits (n = 3; 1 male, 2 female) under occlusion for 4 hours. 50 There was mild erythema in 1 rabbit at 1 hour after removal. The irritation score was 0.2.
Silica dimethyl silylate (0.5 g; 100%) was applied to the intact skin of New Zealand white rabbits (n = 3/sex) for 4 hours under semiocclusion. 51 After removal, the skin was scored at 1, 24, 48, and 72 hours. There were no signs of irritation at any observation period.
Silica dimethyl silylate (6% in aqueous methyl hydroxyethyl cellulose gel) was applied to the intact and abraded skin of New Zealand white rabbits (n = 3/sex) for 24 hours under occlusion. 52 The skin was scored at removal, 48 hours, and daily for 14 days. There were no signs of irritation at any observation period.
Silica dimethyl silylate (50% in olive oil) was applied to the intact and abraded skin of New Zealand white rabbits (n = 3/sex) for 24 hours under occlusion. 52 After removal, the skin was scored at removal, 48 hours, and daily for 14 days. There were no signs of irritation at any observation period.
A product (0.5 mL) containing silica dimethyl silylate (25 wt%) was tested for dermal irritation using male New Zealand white rabbits. 53 The test substance was administered to the clipped skin under semiocclusion for 4 hours and then washed. There was no dermal irritation observed at 1, 24, 48, and 72 hours.
Trimethylsiloxysilicate
Trimethylsiloxysilicate (100%; 0.5 g) was administered to the intact skin of New Zealand White rabbits (n = 6) under occlusion for 4 hours. 30 There were no signs of erythema or edema observed. The authors rated trimethylsiloxysilicate as a dermal nonirritant.
Trimethylsiloxysilicate (2 g/kg) was administered to the shaved skin of New Zealand White rabbits (n = 10) under occlusion for 24 hours. 30 The patch was then removed, and the skin was rinsed in corn oil. The rabbits were observed for 14 days. There were slight signs of grade 1 irritation in 4 rabbits, which was resolved by day 2.
Trimethylsiloxysilicate (30% in olive oil; 0.1 mL; MW: 3000-10 000) was not dermally irritating to white rabbits (n = 3) when applied to clipped skin for 4 consecutive days. 34
Trifluoropropyldimethyl/Trimethylsiloxysilicate
Trifluoropropyldimethyl/trimethylsiloxysilicate (50%) was not a dermal irritant when administered to the clipped skin of Japanese white rabbits (n = 3). 35
In a cumulative dermal irritation test using Japanese white rabbits (n = 3), trifluoropropyldimethyl/trimethylsiloxysilicate (50%) was not irritating to normal, clipped skin when administered for 4 consecutive days. 35
Ocular—Non-Human
Silica Dimethyl Silylate
Silica dimethyl silylate (100%; 0.1 g) administered into the eyes of New Zealand white rabbits (n = 3) caused only slight conjunctivae redness at 1 hour after instillation. 54
In an ocular irritation test using New Zealand white rabbits (n = 6), silica dimethyl silylate (25 wt. %; 0.5 mL) was administered to the eye and examined at 1, 24, 48, and 72 hours. 55 There was a diffuse crimson coloration of the conjunctivae and slight swelling of the eyelids observed in 1 rabbit. Slight redness of the conjunctivae alone was seen in the remaining 5 animals. The mean conjunctival redness was 0.6. Ocular reactions had resolved completely in all animals by 1, 3, or 7 days after instillation. All corneal and iridial scores for all animals at all observation times were zero. The authors concluded that the test substance was a nonirritant.
Silica dimethyl silylate (0.1-0.2 g; 0.1 mL) was applied to 1 eye of New Zealand white rabbits (n = 3/sex). 37 Three of the treated eyes were not rinsed, and 3 rinsed with saline after 20 to 30 seconds. The eyes were scored at 1, 24, 48, and 72 hours. There were no signs of irritation at any observation period.
Silica dimethyl silylate (0.1-0.2 g) was applied to 1 eye of New Zealand white rabbits (n = 8; sex not provided). 56 Five of the rabbits’ eyes were not rinsed, and 3 were rinsed with saline after 20 to 30 seconds. The eyes were scored at 1, 24, 48, and 72 hours, and 7 days. There were no signs of irritation at any observation period.
Silica dimethyl silylate (50%; 0.1 mL in olive oil) was applied to 1 eye of New Zealand white rabbits (n = 8; sex not provided). 56 Five of the rabbits’ eyes were rinsed with saline after 5 minutes, and 3 were rinsed after 24 hours. The eyes were scored at 1, 24, 48, and 72 hours. Conjunctiva redness was scored at 1.0 at the first 3 observations and was resolved at 72 hours. There were no other signs of irritation at any observation period.
Silica dimethyl silylate (0.1 mL; 0.1-0.2 g undiluted) was applied to 1 eye of New Zealand white rabbits (n = 5 males, 4 females). There was mild conjunctivae redness at 1 and 24 hours which was resolved at 48 hours.
An EpiOcular Human Cell Construct assay was conducted on a product containing silica dimethyl silylate (2%). 57 There was no irritation predicted.
Trimethylsiloxysilicate
Trimethylsiloxysilicate (100%; 0.1 mL) was administered to the right eye of New Zealand White rabbits (n not provided). 30 The eyes were examined at 0, 1, 24, 48, and 72 hours. There were no clinical signs or signs of irritation at any observation period.
Trimethylsiloxysilicate (50% in olive oil; 0.1 mL; MW: 3000-10 000) was not an ocular irritant to Japanese white rabbits (n = 3). 34
Trimethylsiloxysilicate (50% in olive oil; 0.1 mL; MW 3000-5000) had a Draize score of 2 when administered to the eyes of Japanese white rabbits (n = 3).
A mixture of trimethylsiloxysilicate (60%) and isododecane (50% in olive oil) was reported to be practically nonirritating in rabbits (n = 3). 49
Trifluoropropyldimethyl/Trimethylsiloxysilicate
Trifluoropropyldimethyl/trimethylsiloxysilicate (100%; 0.1 mL) had a Draize score of 0 in Japanese white rabbits (n = 3). 35
Dermal—Human
Silica dimethyl silylate, trimethylsiloxysilicate, and trifluoropropyldimethyl/trimethylsiloxysilicate were not irritating up to 30%, 20%, and 50%, respectively, in multiple human patch tests and use tests of the ingredients and products containing the ingredients (Table 5).
Human Irritation and Sensitization Studies of Silica Dimethyl Silylate, Trimethylsiloxysilicate, and Trifluoropropyldimethyl/Trimethylsiloxysilicate and Products Containing These Ingredients.
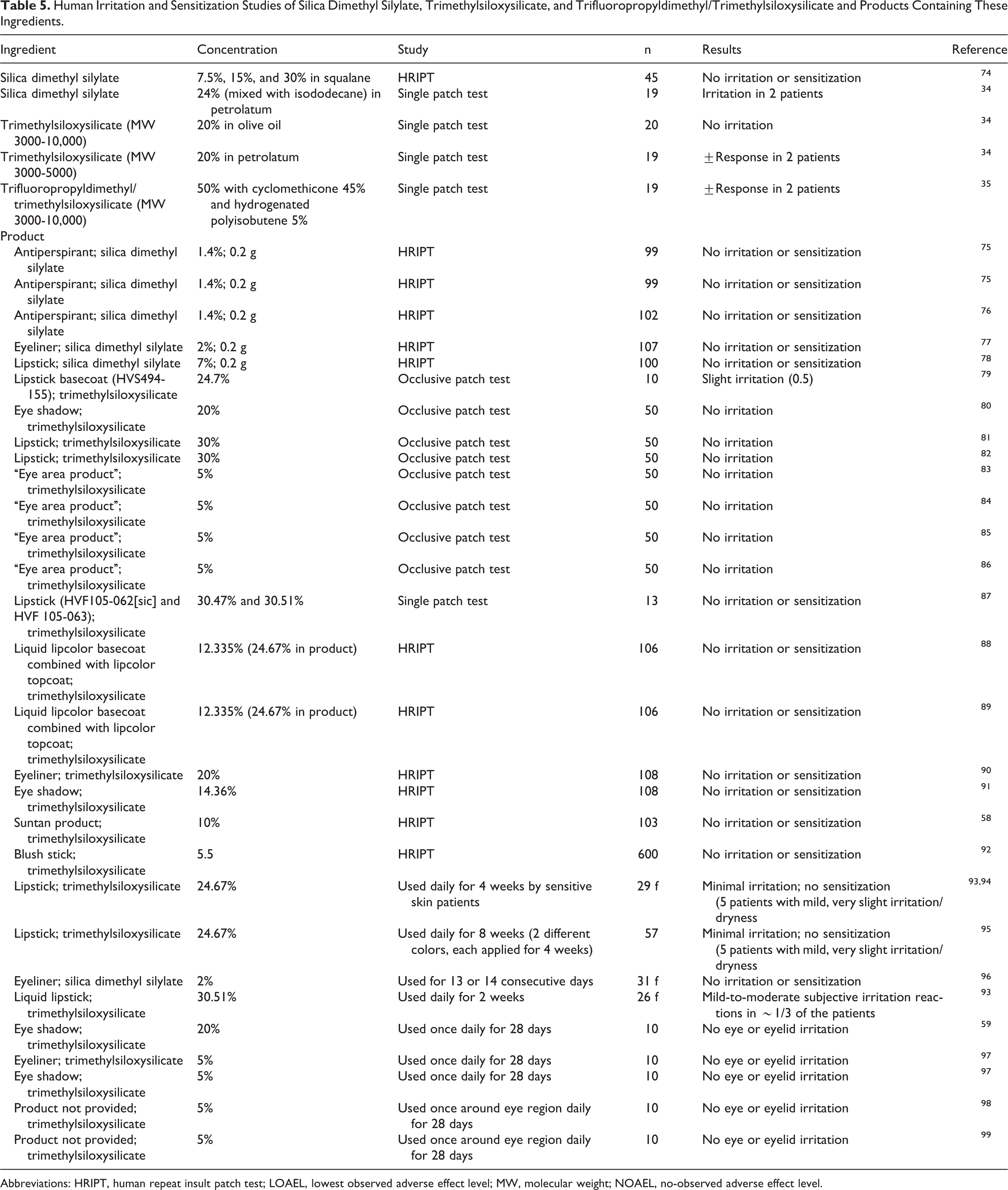
Abbreviations: HRIPT, human repeat insult patch test; LOAEL, lowest observed adverse effect level; MW, molecular weight; NOAEL, no-observed adverse effect level.
Ocular—Human
An eyeliner containing silica dimethyl silylate (2%) and an eye shadow containing trimethylsiloxysilicate (20%) were not irritating in use tests (Table 5). 58,59
Sensitization
Non-Human
Trimethylsiloxysilicate
In a local lymph node assay (LLNA), trimethylsiloxysilicate (15%, 30%, and 60% in acetone/olive oil) was dermally administered to the entire dorsal surface of each ear of mice (strain and n not provided) for 3 consecutive days. 30 The stimulation indexes were 1.0, 1.1, and 0.8 at 15%, 30%, and 60%, respectively. The authors concluded that trimethylsiloxysilicate had no reaction that was identified as sensitization.
Trimethylsiloxysilicate (50% in alcohol) was a weak sensitizer in a guinea pig maximization test (n = 5). 34
Trifluoropropyldimethyl/Trimethylsiloxysilicate
Trifluoropropyldimethyl/trimethylsiloxysilicate (100%) was a weak sensitizer in a guinea pig maximization test (n = 5). 35
Human
Silica dimethyl silylate, trimethylsiloxysilicate, and trifluoropropyldimethyl/trimethylsiloxysilicate were not sensitizing up to 30%, 20%, and 50%, respectively, in multiple human repeat insult patch tests (HRIPTs) and use test of the ingredients and products containing the ingredients (Table 5).
Summary
The functions of silica silylate, silica dimethyl silylate, trimethylsiloxysilicate, and trifluoropropyldimethyl/trimethylsiloxysilicate in cosmetics include: antifoaming agents, anticaking agents, bulking agents, binders, skin-conditioning agents—emollient, skin-conditioning agents—occlusive, slip modifiers, suspension agents—nonsurfactant, and viscosity increasing agents—nonaqueous. These grafted and cocondensed hybrid materials are amorphous and practically insoluble in most common solvents, much like unmodified silica.
Silica dimethyl silylate was reported to be used in 734 cosmetic products (593 leave-on and 142 rinse-off products) at 0.00003% to 10%; up to 10% in leave-on products; and up to 4% in rinse-off products. There were 245 reported uses of silica silylate (244 in leave-on at 0.2%-25%,highest in other hair preparations, and 1 rinse-off product; no concentrations of use were reported for rinse-off products). There were 633 reported uses of trimethylsiloxysilicate at 0.0001% to 30%; up to 30% in leave-on products; and up to 5% in rinse-off products. There were no reported uses of trifluoropropyldimethyl/trimethylsiloxysilicate, but concentration of use was reported to be 2% to 20% in leave-on products.
Silica dimethyl silylate is used in perfumes. The product category may include products that are aerosolized or used as powders. Particles of silica dimethyl silylate average >125 µm, and none were <90 µm, and only particles with an aerodynamic diameter of ≤10 µm are respirable.
Orally administered silica dimethyl silylate was eliminated from the body primarily in the feces in mice, monkeys, and humans. Inhaled silica dimethyl silylate collected in the lungs and lymph nodes of rats.
In acute studies, dermally administered silica dimethyl silylate and trimethylsiloxysilicate up to 2 g/kg were not toxic to rats. The oral LD50 of silica dimethyl silylate was >7900 mg/kg for rats; trimethylsiloxysilicate had no effects at 5 g/kg. The oral LD50 of trifluoropropyldimethyl/trimethylsiloxysilicate was > 2 g/kg in mice. There were no mortalities from the inhalation of silica dimethyl silylate up to 520 mg/m3 in rats. Intraperitoneally administered silica dimethyl silylate caused thickening of the liver and spleen capsules. The test substance was observed in the abdominal cavity.
The oral NOAEL for silica dimethyl silylate in rats was 500 mg/kg for 6 months and 100 mg/kg for 24 months.
Rats that inhaled treated fumed silica dust for up to 4 weeks were observed to have crusty eyes, muzzle, and nose; closed eyes; irregular breathing; irritable disposition; lacrimation and salivation; scabs; and red and yellow/brown stained fur. The inhalation LOAEL was 31 mg/m3 for 2 weeks.
Aerosolized silica dimethyl silylate caused mortality at 209 mg/m3 and respiratory distress at lower doses. There was a dose-dependent reduction in weight gain, reduction in feed consumption, increase in lung weight, decrease in relative liver weights, and decrease in absolute kidney weights. There was an increase in red blood cell counts, packed cell volume, and hemoglobin.
Silica dimethyl silylate did not cause any developmental toxicity to rats up to 3.8 g/kg/d, rabbits up to 1600 mg/kg, hamsters up to 1600 mg/kg, or mice up to 1340 mg/kg. There were no reproductive effects in rats up to 509 mg/kg/d.
Silica dimethyl silylate was not genotoxic in several Ames assays and a mammalian chromosome aberration test. A product containing siloxanes and silicones, di-Me (dimethyl silicones and siloxanes; 18%) and dimethyl silicones and siloxane, and reaction products with silica (2%) was not genotoxic in an Ames assay. Trimethylsiloxysilicate was not genotoxic in Ames assays. There were no in vivo genotoxicity studies discovered.
Orally administered silica dimethyl silylate at 100 mg/kg was not carcinogenic to rats. Silica dimethyl silylate and trimethylsiloxysilicate were not dermally irritating to rabbits up to 100%.
Trifluoropropyldimethyl/trimethylsiloxysilicate was not dermally irritating to rabbits at 100%. In a human patch test, a mixture containing trimethylsiloxysilicate at 24% resulted in irritation in 2 of 19 patients. Silica dimethyl silylate at 100% was slightly or not irritating to the rabbit eye. Trimethylsiloxysilicate at 100% was practically or nonirritating to the rabbit eye. Silica dimethyl silylate and trimethylsiloxysilicate were not dermally irritating to rabbits up to 100%. Trifluoropropyldimethyl/trimethylsiloxysilicate was not dermally irritating to rabbits at 100%. Silica dimethyl silylate and trimethylsiloxysilicate were not irritating or sensitizing up to 30% and 20%, respectively, in multiple human patch tests. Trifluoropropyldimethyl/ trimethylsiloxysilicate was not irritating at 50% in multiple human patch tests. Silica dimethyl silylate was slightly or not irritating to the rabbit eye. Trimethylsiloxysilicate was practically or nonirritating to the rabbit eye. The results were negative in an LLNA of trimethylsiloxysilicate up to 60%. The HRIPTs of products containing silica dimethyl silylate up to 7% were negative.
Discussion
The CIR Expert Panel noted gaps in the available safety data for some of the silylates and surface-modified siloxysilicates in this safety assessment; however, since these ingredients have similar structures and are used in cosmetics in similar ways, the available data can be used to support the safety of the entire group. These ingredients are stable amorphous solids, used mostly in formulations applied to the skin. These ingredients have virtually no water solubility, and it appears that the only impurity would be alkanes (C7-10-iso) at a maximum of 0.35%, a residual solvent from the production process, which raises no safety concerns for dermal use.
The available safety test data demonstrated an absence of dermal irritation and sensitization at the reported concentrations of use. Silica dimethyl silylate did not cause reproductive or developmental toxicity in animal studies. Orally silica dimethyl silylate at 100 mg/kg was not carcinogenic. Although there were no long-term studies that addressed systemic toxicity, these ingredients were not likely to pass through the stratum corneum of the skin because of their large size and their solubility properties.
Because these ingredients can be used in products that may be aerosolized, including sprays and powders, the panel discussed the issue of potential inhalation toxicity. The data available from multiple inhalation studies, including acute and chronic exposure studies, indicate little potential for pulmonary overload or other respiratory effects at relevant doses. Although particles appear to have reached the lungs in these animal studies, the sizes of the particles used were either clearly within the respirable range (ie, ≤10 µm) or were not reported. The panel believes that the sizes of a substantial majority of the particles of these ingredients, as manufactured, are larger than the respirable range and/or aggregate and agglomerate to form much larger particles in formulation. Thus, the adverse effects reported using high doses of respirable particles in the inhalation studies do not indicate risks posed by use in cosmetics.
The panel considered other data available to characterize the potential for silylates and surface-modified siloxysilicates to cause systemic toxicity, irritation, sensitization, or other effects. The panel noted the lack of systemic toxicity at high doses in several acute and subchronic oral exposure studies and 1 chronic oral exposure study, little or no irritation or sensitization in multiple tests of dermal and ocular exposure, the absence of genotoxicity in multiple Ames tests and a CHO test, and lack of carcinogenicity in a lifetime oral exposure study. In addition, these ingredients are macromolecules, insoluble in water, and chemically inert under physiological conditions or conditions of use, which supports the view that they are unlikely to be absorbed or cause local effects in the respiratory tract. Further, these ingredients are reportedly used at concentrations ≤10% in cosmetic products that may be aerosolized. The panel noted that 95% to 99% of the particles produced in cosmetic aerosols are not respirable. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, this information indicates that inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects.
Conclusion
The CIR Expert Panel concluded that silica silylate, silica dimethyl silylate, trimethylsiloxysilicate, and trifluoropropyldimethyl/trimethylsiloxysilicate are safe in the present practices of use and concentration described in this safety assessment safe when formulated and delivered in the final product not to be irritating or sensitizing to the respiratory tract.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.