Abstract
Forty male and 40 female Crl:SD® CD® IGS rats were fed diets containing 0, 40 000, 80 000, or 120 000 ppm tamarind seed polysaccharide (equivalent to 3450.8, 6738.9, or 10 597.1 mg/kg bw/day and 3602.1, 7190.1, or 10 690.7 mg/kg bw/day for males and females, respectively) for 28 days. Animals were observed for adverse clinical signs, body weight, feed consumption, hematology and clinical chemistry parameters, urinalysis values were recorded, and at the end of the study the rats underwent a full necropsy. Functional Observational Battery (FOB) and Motor Activity (MA) tests were performed on all animals. There were no mortalities, no clinical or ophthalmologic signs, body weight, body weight gain, food consumption and food efficiency, FOB or MA findings associated with the administration of tamarind seed polysaccharide. Initial statistically significant decreases in body weight gain and food consumption resolved after the first week and were considered the result of reduced palatability. There were no adverse changes in hematology, coagulation, clinical chemistry or urinalysis parameters in male or female rats considered the result of test substance administration. At necropsy, there were no macroscopic, histopathological findings, estrus cycle, or organ weight changes deemed related to administration of the test substance. Under the conditions of this study and based on the toxicological endpoints evaluated, the no-observed-adverse-effect level (NOAEL) for tamarind seed polysaccharide in the diet was the highest concentration tested of 120 000 ppm (equivalent to 10 597 mg/kg bw/day and 10 691 mg/kg bw/day for male and female rats, respectively).
Introduction
Tamarind seed polysaccharide, obtained from the tamarind tree (
According to the
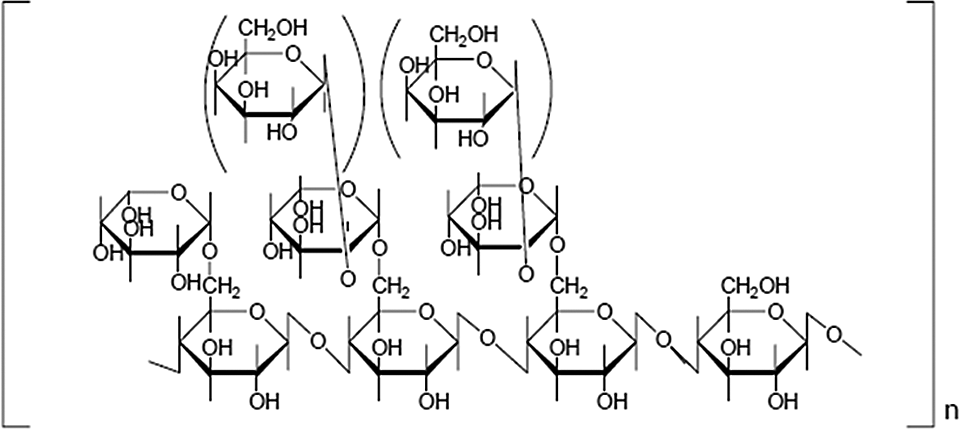
Generalized structural formula of tamarind seed polysaccharide.
The safety of Glyloid brand tamarind seed polysaccharide (DSP Gokyo Food & Chemical Co, Ltd, Osaka, Japan) has been previously evaluated in acute and subchronic toxicity studies, mutagenicity/genotoxicity studies, and a carcinogenicity study. Oral median lethal dose (LD50) values were reported to exceed 5000 mg/kg body weight (bw) in mice and rats.7,8 Tamarind seed polysaccharide fed in the diet for 13 weeks (dose-range finding study) and 78 weeks (carcinogenicity study) produced no toxicologically significant differences from control animals up to the highest concentration tested of 5%, approximately 8200 and 10 600 mg/kg bw/day for males and females, respectively, over 13 weeks, and 6658 and 8575 mg/kg bw/day for males and females, respectively, over 78 weeks.
9
A significant weight-gain decrement reported in both low- and high-dose females in the carcinogenicity study from week 34 to termination was determined to be an effect of the test substance displacing some of the caloric content of the feed. The body weight of the low- and high-dose females slightly deviated from the controls in the outset and diverged further with time to statistical significance. It was not dose dependent; the decrement in weight gain was greater for mice in the low-dose group than for those in the high-dose group. The reduction in body weight of these animals was reported to be the cause of a statistically significant elevation of relative brain, heart, liver, spleen, and kidney weights. Overall, it was concluded that tamarind seed polysaccharide is “not carcinogenic in either male or female B6C3F1 mice with long-term dietary exposure.” In an earlier 2-year study by Iida et al
10
on Sprague-Dawley rats fed with tamarind seed polysaccharide in the diet, no adverse effects attributable to the test substance were reported at up to the highest concentration of 12% (approximately 8300 and 9400 mg/kg bw/day for males and females, respectively). Tamarind seed polysaccharide was not clastogenic when tested in chromosomal aberration tests with Chinese hamster lung-derived fibroblast cells,
11
not mutagenic in an unpublished abbreviated Ames assay using
Due to a minor recent change in the method of extraction of tamarind seed polysaccharide, it appeared appropriate to reassess the product’s genotoxicity and subacute oral toxicity to confirm that no novel potentially toxic impurities may have been introduced. An Ames assay conducted under Good Laboratory Practice (GLP) and following Organization of Economic Cooperation and Development (OECD) Guideline 471 (Bacterial Reverse Mutation Test) using
Based on a dietary assessment, the intended use of tamarind seed polysaccharide as a thickener and stabilizer in foods (eg, ice cream, sauces and condiments, dressings and mayonnaise, fruit preserves, desserts, beverages, pickles, spreads and fillings, flour products, and soup) is estimated to produce intakes of the polysaccharide as high as 91 mg/kg bw/day among heavy consumers of these foods. The doses tested in the current study were chosen to confirm the safety of this level of exposure to tamarind seed polysaccharide.
Materials and Methods
This study was conducted according to OECD Guideline for the Testing of Chemicals and Food Ingredients No. 407 (Repeated Dose 28-Day Oral Toxicity Study in Rodents) and the US Food and Drug Administration (FDA) Toxicological Principles for the Safety Assessment of Food Ingredients, Redbook 2000 (IV.C.4.a. Subchronic Toxicity Studies with Rodents) and met the requirements of the OECD Principles of GLP and 21 CFR 58: US FDA GLP Standards (1987), with the exception of the analytical testing of the test substance and diet.
Test Substance
The test substance, a water-soluble polysaccharide thickener, is a white to grayish white powder composed of 100% tamarind seed gum sold under the trade name Glyloid and was supplied by DSP Gokyo Food & Chemical Co, Ltd. The stability of the test substance was verified using neat samples taken at the initial, middle, and final diet preparations, which were frozen until the analysis of total dietary fiber using the method of AOAC 991.43. Analysis of the neat test substance and as mixed in the diet was found to be stable over the course of the study. Similarly, sampling from the top, middle, and bottom of the diet preparations showed the test substance to be homogeneously distributed throughout the study. Overall, concentration verification results of the study averaged 87.7%, 86.4%, and 82.7% of corrected nominal concentrations for 40 000, 80 000, and 120 000 ppm total dietary fiber of Glyloid, respectively. The values were considered consistent with this matrix based upon accepted criteria for this analytic method. 14
Vehicle and Diet
The test substance was added to 2016CM Harlan Teklad Global Rodent Diet and mixed thoroughly. Control diet was mixed under the same conditions but without the test substance. All diets were prepared weekly and kept refrigerated. Stability, homogeneity, and concentration were verified by collecting samples at regular intervals, which were kept frozen until the analysis of total dietary fiber using the method of AOAC 991.43. Test substance and dietary analysis were within 20% variation and consistent with laboratory outcomes for this matrix.
Diets were fed ad libitum for 28 days at dietary tamarind seed polysaccharide concentrations of 0 (control), 40 000, 80 000, or 120 000 ppm to target approximate exposures of 0, 3333, 6667, or 10 000 mg/kg bw/day, assuming that a 300-g rat eats 25 g food/day.
Animals and Environment
Eighty 7- to 8-week-old Crl Sprague-Dawley CD IGS rats (40 rats/sex) were used in the study. Body weights ranged from 173 to 198 g (mean = 185.5 ± 6.6 g) for females and 210 to 246 g (mean = 228.6 ± 10.4 g) for males at treatment initiation, with weight variation not exceeding ±20% of the mean weight for each sex. The rats were acclimatized for 6 days prior to study initiation and then were randomly assigned, stratified by body weight, to 4 test groups consisting of 10 rats/sex. Rats were individually housed in suspended stainless steel cages with mesh floor in rooms maintained at 20°C to 23°C, with a relative humidity of 54% to 69% and a 12-hour light/12-hour dark cycle. Test diet and tap water were available ad libitum except when animals were fasted as required in the protocol.
Observations
Clinical Signs
Animals were observed at least twice daily for viability and daily for cage-side observations. Detailed clinical examinations were conducted prior to first treatment and weekly thereafter until study termination.
Ophthalmologic Evaluation
During acclimation and at study termination, mydriatic eye drops were placed into the eyes of the rats, which were then examined in subdued light by focal illumination, indirect ophthalmoscopy, and, when indicated, slit-lamp microscopy.
Body Weight and Body Weight Gain
Body weight was recorded twice during acclimation, on day 0 (1st day of treatment), weekly thereafter, and just prior to necropsy. Body weight gain was calculated for selected intervals and for the overall study.
Feed Consumption and Feed Efficiency
Feed consumption was measured weekly to coincide with body weight measurements. Feed efficiency and mean daily dietary intakes were calculated.
Functional Observational Battery
A functional observational battery (FOB) was performed on all animals at week 4. Each rat was evaluated during handling and while in an open field for excitability, autonomic function, gait and sensorimotor coordination (open field and manipulative evaluations), reactivity and sensitivity (elicited behavior), and other abnormal clinical signs including but not limited to convulsions, tremors, unusual or bizarre behavior, emaciation, dehydration, and general appearance. The rats were observed in random order and without the observer having knowledge of the treatment group. In addition, forelimb and hindlimb grip strength (measured with a digital force gauge in triplicate) and foot splay measurements (duplicate measurements) were taken.
Motor Activity
During the same time period that the FOB was performed, motor activity (MA) was also monitored in all animals using an automated Photobeam Activity System (San Diego Instruments, Inc, San Diego, California). Each animal was placed into a polycarbonate solid bottom cage and immediately evaluated for a single 1-hour phase with photobeam counts accumulated over six 10-minute intervals.
Clinical Pathology
During weeks 2 and 4, blood samples were collected via sublingual bleeding under isoflurane anesthesia from all animals that were fasted overnight prior to each collection. Approximately 500 µL of blood was collected for hematology assessments in precalibrated tubes containing K3EDTA, and approximately 1000 µL of blood was collected in tubes containing no preservative for clinical chemistry assessment and was centrifuged, and the serum was transferred to labeled tubes.
Hematology
The hematology parameters evaluated included erythrocyte count, hematocrit, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, absolute reticulocyte count, total white blood cell and differential leukocyte count, hemoglobin concentration, mean corpuscular volume, red cell distribution width, and platelet count. In addition, separate blood smears stained with new methylene blue of Wright-Giemsa stain were prepared for each animal and examined, if necessary, to substantiate or clarify the results of the hematology findings.
For the determination of prothrombin time and partial thromboplastin time, blood samples (∼1.8 mL aliquots) were collected via the interior vena cava under isoflurane anesthesia at termination and were placed in tubes containing 3.2% sodium citrate. Samples were centrifuged, and the plasma was transferred to labeled tubes.
Clinical Chemistry
The clinical chemistry parameters evaluated included serum aspartate aminotransferase, serum alanine aminotransferase, sorbitol dehydrogenase, alkaline phosphatase, total bilirubin, urea nitrogen, blood creatinine, total cholesterol, triglycerides, fasting glucose, total serum protein, albumin, globulin, calcium, inorganic phosphorus, sodium, potassium, and chloride.
Urinalysis
The rats were fasted for at least 15 hours the day prior to collection of blood samples and then placed in metabolism cages for collection of urine. Urine samples were analyzed for quality, pH, ketone, color, glucose, bilirubin, clarity, specific gravity, blood, volume, protein, urobilinogen, and microscopic urine sediment examination.
Gross Necropsy and Organ Weights
At the end of the study, all surviving animals were euthanized by exsanguination from the abdominal aorta under isoflurane anesthesia and underwent a full necropsy including examination of the external surface of the body, all orifices, and the thoracic, abdominal, and cranial cavities and their contents. Vaginal smears were taken from all females prior to necropsy to microscopically determine estrus stage. Wet organ weights were recorded for adrenals, kidneys, spleen, brain, liver, thymus, epididymides, ovaries, uterus with oviducts, heart, and testes. Epididymides, eyes with optic nerve, and testes were preserved in modified Davidson fixative and stored in ethanol. All other organs and tissues (prostate, seminal vesicles, ileum with Peyer patches, salivary glands, adrenals, jejunum, kidneys, all gross lesions, larynx, skeletal muscle, aorta, liver, skin, femur, bone marrow, lungs, skin, spinal cord, lymph nodes, brain, mammary gland, spleen, ovaries, stomach cecum, pancreas, thymus, cervix, sciatic nerve, thyroid/parathyroid, colon, pharynx, trachea duodenum, pituitary, urinary bladder, esophagus, rectum, uterus, vagina, heart, and Harderian gland) were preserved in 10% neutral-buffered formalin.
Histopathological Examination
Histopathological examinations were performed under light microscopy by a board-certified veterinary pathologist on the preserved organs and tissues of rats from the control and high-dose groups and on any gross lesions of potential toxicological significance noted in any test group. Prior to examination, the tissues were fixed, trimmed, processed, embedded in paraffin, sectioned with a microtome, placed on glass microscope slides, and stained with hematoxylin and eosin.
Statistical Analyses
Mean and standard deviations were calculated for all quantitative data. Significance was judged at a probability value of
In-Life and Organ Weight Data
If warranted by sufficient group sizes, data within groups were evaluated for homogeneity of variances and normality by Bartlett test. 15 Where Bartlett test indicated homogeneous variances, treated and control groups were compared using a 1-way analysis of variance (ANOVA). When 1-way ANOVA was significant, a comparison of the treated groups to control was performed by Dunnett test16,17 for multiple comparisons. Where variances were considered significantly different by Bartlett test, groups were compared using a nonparametric method. 18 When nonparametric ANOVA was significant, comparison of treated groups to control was performed using Dunn test. 19 The MA data were further analyzed using 2-way repeated measures of ANOVA (SigmaStat version 2.03). Other statistical methods were used, when appropriate, at the time of analysis.
Clinical Pathology
Initially, Levene test 20 was used for homogeneity, and Shapiro-Wilk test 21 was used for normality. If the preliminary test was not significant, a 1-way ANOVA followed with Dunnett test was performed. If the preliminary test showed significant results, transforms of the data to achieve normality and variance homogeneity were used; the order of the transforms attempted were log, square root, and rank order. Other statistical methods were used, when appropriate, at the time of analysis.
Results
The use of the words “significant” or “significantly” indicates a statistically significant difference between the control and the experimental groups.
Observations
Survival
All animals survived till study termination.
Clinical Signs
Incidental clinical signs noted in males included slight-to-moderate alopecia (both treated and control groups), unilateral red ocular discharge (1 of 10 mid-dose rats), lacrimation (1 of 10 mid-dose rats), and hair loss (both treated and control groups). Signs noted in females included slight-to-moderate alopecia and hair loss (both treated and control groups).
Ophthalmologic Evaluation
All animals were normal upon final ophthalmic examination.
Body Weight and Body Weight Gain
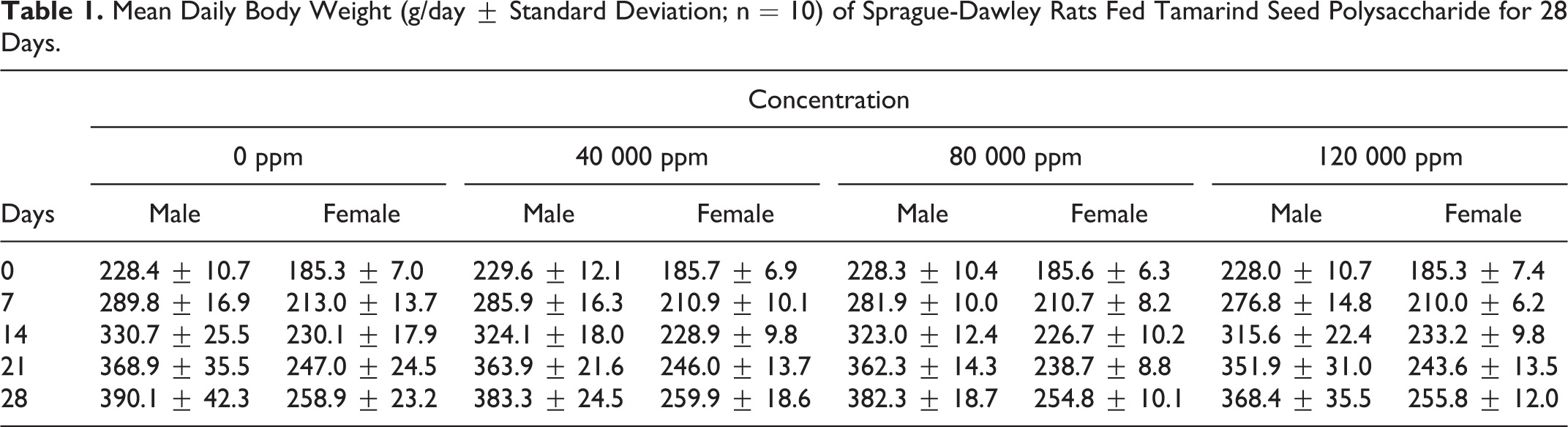
There were no changes in the body weight or body weight gain attributable to the administration of tamarind seed polysaccharide. Slight yet significant decreases in male body weight gain (7.66 ± 0.91 and 6.97 ± 1.04 g/day for mid- and high-dose groups, respectively, for days 0 to 7 versus 8.77 ± 1.07 g/day for the corresponding control group) did not persist and were considered incidental due to the palatability of the test substance and therefore of no toxicological significance (Tables 1 and 2).
Mean Daily Body Weight (g/day ± Standard Deviation; n = 10) of Sprague-Dawley Rats Fed Tamarind Seed Polysaccharide for 28 Days.
Mean Daily Body Weight Gain (g/day ± Standard Deviation; n = 10) of Sprague-Dawley Rats Fed Tamarind Seed Polysaccharide for 28 Days.
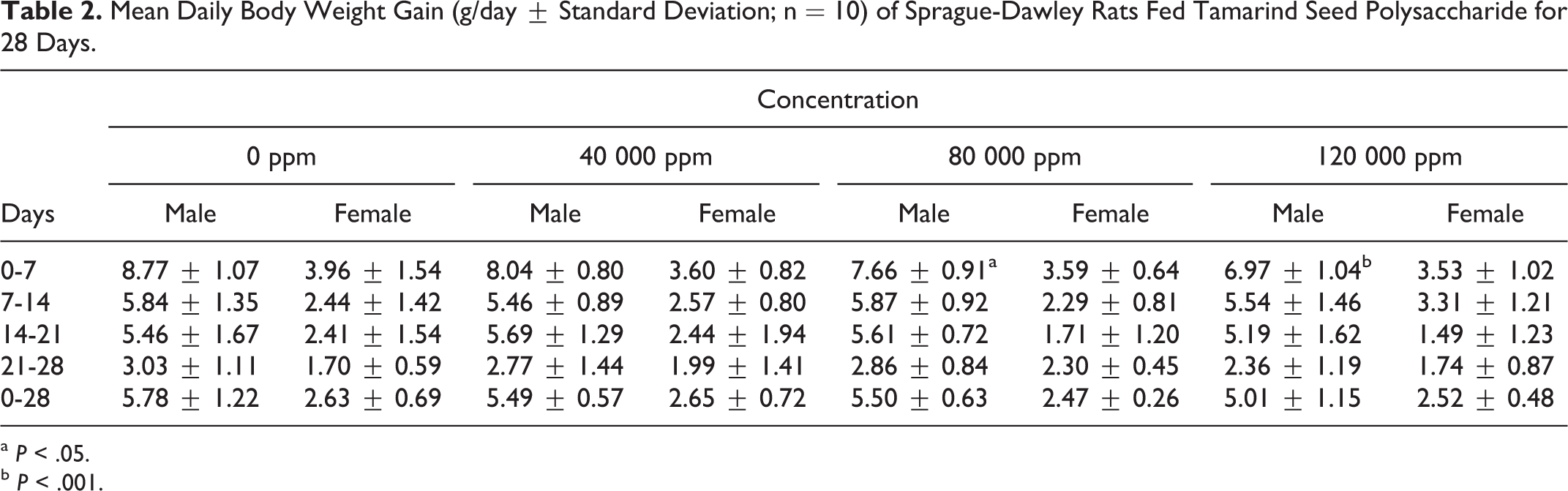
a
b
Feed Consumption and Feed Efficiency
The dietary concentrations of 0, 40 000, 80 000, and 120 000 ppm used in this study provided overall (days 0-28) mean daily tamarind seed polysaccharide intakes (ranges shown in parenthesis) of 0, 3450.8 (2844.0-4299.3), 6738.9 (5561.8-8350.1), and 10 597.1 (8748.8-12714.6) mg/kg bw/day for male rats and 0, 3602.1 (3322.4-3925.2), 7190.1 (6607.5-7807.7), and 10 690.7 (9808.5-11433.3) mg/kg bw/day for female rats, respectively. Mean daily feed consumption and feed efficiency were similar to controls with the exception of a significant decrease in mean daily feed consumption of high-dose females at the start of the study compared to the corresponding controls (18.81 ± 1.20 g/day vs 20.96 ± 1.71 g/day; days 0-7) and a significant decrease in feed efficiency of high-dose males at the start of the study compared to corresponding controls (0.259 ± 0.023 versus 0.305 ± 0.029; days 0-7; Tables 3 and 4).
Mean Daily Feed Consumption (g/day ± Standard Deviation; n = 10) of Sprague-Dawley Rats Fed Tamarind Seed Polysaccharide for 28 Days.
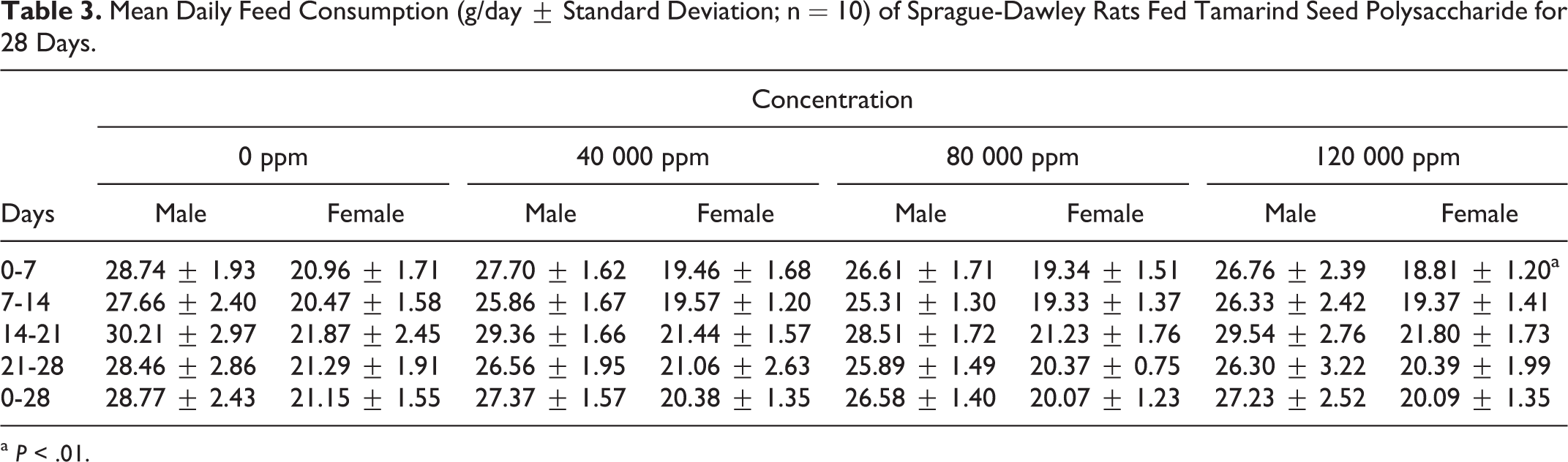
a
Feed Efficiencya (Mean ± Standard Deviation; n = 10) in Sprague-Dawley Rats Fed Tamarind Seed Polysaccharide for 28 Days.
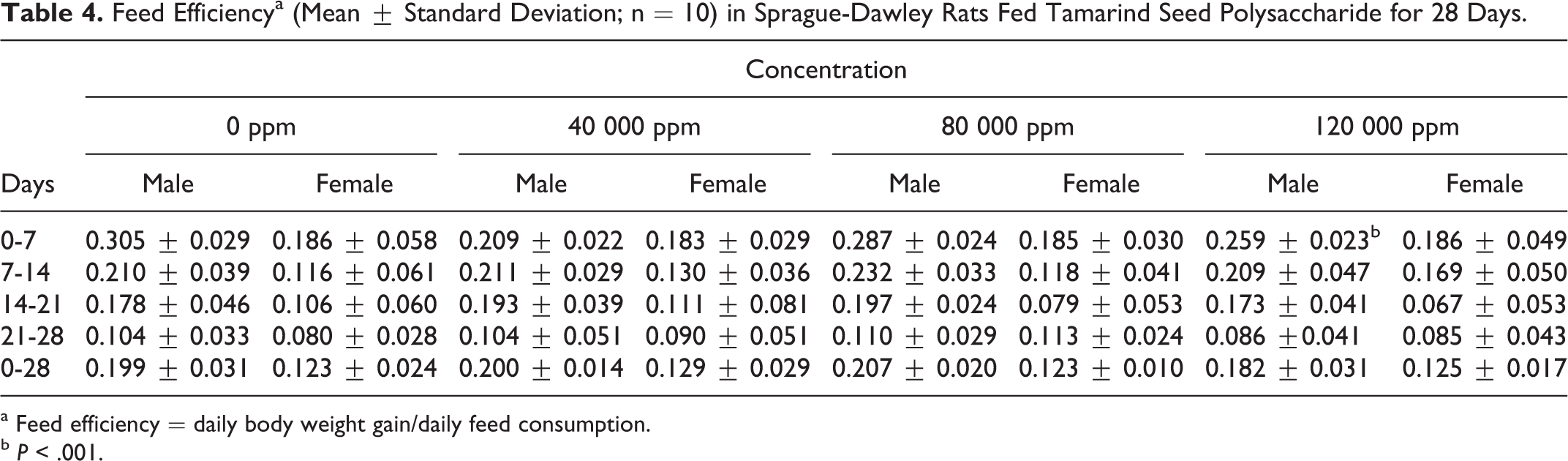
a Feed efficiency = daily body weight gain/daily feed consumption.
b
Functional Observational Battery
Incidental findings reported in males included a normal/sleeping posture (both treated and control groups), lacrimation (1 of 10 mid-dose rats), hair loss (1 of 10 mid-dose and 1 of 10 high-dose rats), no tactile response (both treated and control groups), and no tail pinch response (1 of 10 mid-dose rats) and in females included hair loss (both treated and control groups), no tactile response (1 of 10 low-dose rats), and no tail pinch response (1 of 10 control rats). For both sexes, mean quantitative measurements of forelimb/hindlimb grip strength and hindlimb foot splay were comparable to controls.
Motor Activity
The MA results of the treated rats were comparable to that of the corresponding controls. In general, all groups of animals (including controls) exhibited a similar level of movement over all intervals. No statistically significant differences were noted in any male or female test groups compared to their corresponding control groups.
Hematology and Clinical Chemistry
Summaries of mean hematology and clinical chemistry parameters are presented in Tables 5 and 6. The only significant changes were decreased absolute reticulocyte count in mid- and high-dose males (day 9), decreased red blood cell count, hemoglobin concentration, and hematocrit in mid-dose females (day 9), decreased platelet count in high-dose females (day 24), increased prothrombin time in low-dose females (day 30), increased triglycerides in mid-dose males (days 9 and 24), decreased total protein in high-dose males and females (day 9), decreased albumin in high-dose males (day 9), decreased inorganic phosphate in mid- and high-dose males (day 9), increased alkaline phosphatase in high-dose females (day 24), increased glucose in mid-dose females (day 9), and decreased globulin in all treated females (day 9) and in high-dose females (day 24). These differences were not considered toxicologically significant, since they were not accompanied by any other corresponding clinical or histopathological changes and were very small, occurred in only 1 sex, were not dose dependent, and/or were not significant at study termination.
Hematology Values (Mean ± Standard Deviation; n = 10) for Sprague-Dawley Rats Fed Tamarind Seed Polysaccharide for 28 Days.
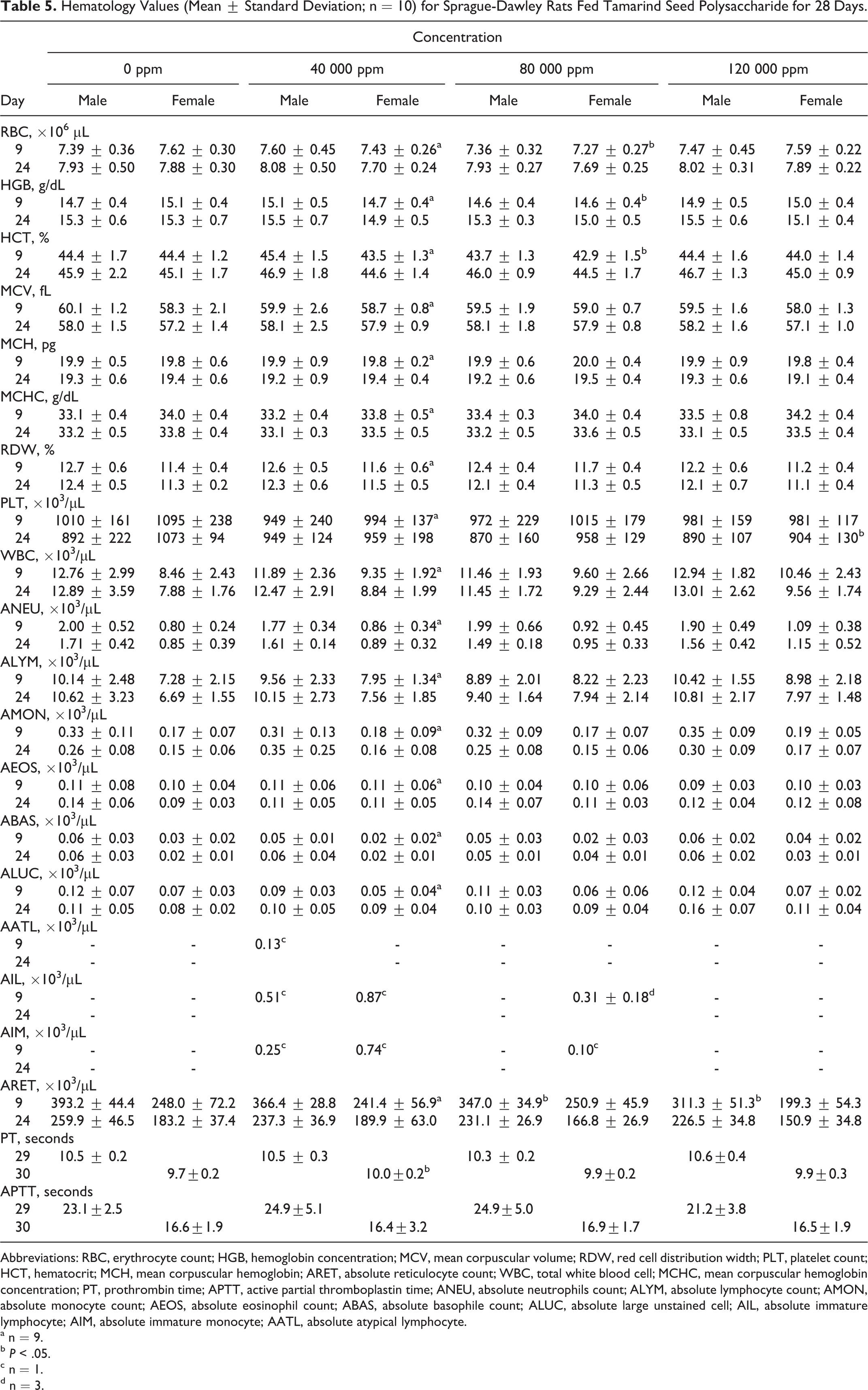
Abbreviations: RBC, erythrocyte count; HGB, hemoglobin concentration; MCV, mean corpuscular volume; RDW, red cell distribution width; PLT, platelet count; HCT, hematocrit; MCH, mean corpuscular hemoglobin; ARET, absolute reticulocyte count; WBC, total white blood cell; MCHC, mean corpuscular hemoglobin concentration; PT, prothrombin time; APTT, active partial thromboplastin time; ANEU, absolute neutrophils count; ALYM, absolute lymphocyte count; AMON, absolute monocyte count; AEOS, absolute eosinophil count; ABAS, absolute basophile count; ALUC, absolute large unstained cell; AIL, absolute immature lymphocyte; AIM, absolute immature monocyte; AATL, absolute atypical lymphocyte.
a n = 9.
b
c n = 1.
d n = 3.
Clinical Chemistry Values (Mean ± Standard Deviation; n = 10) for Sprague-Dawley Rats Fed With Tamarind Seed Polysaccharide for 28 Days.
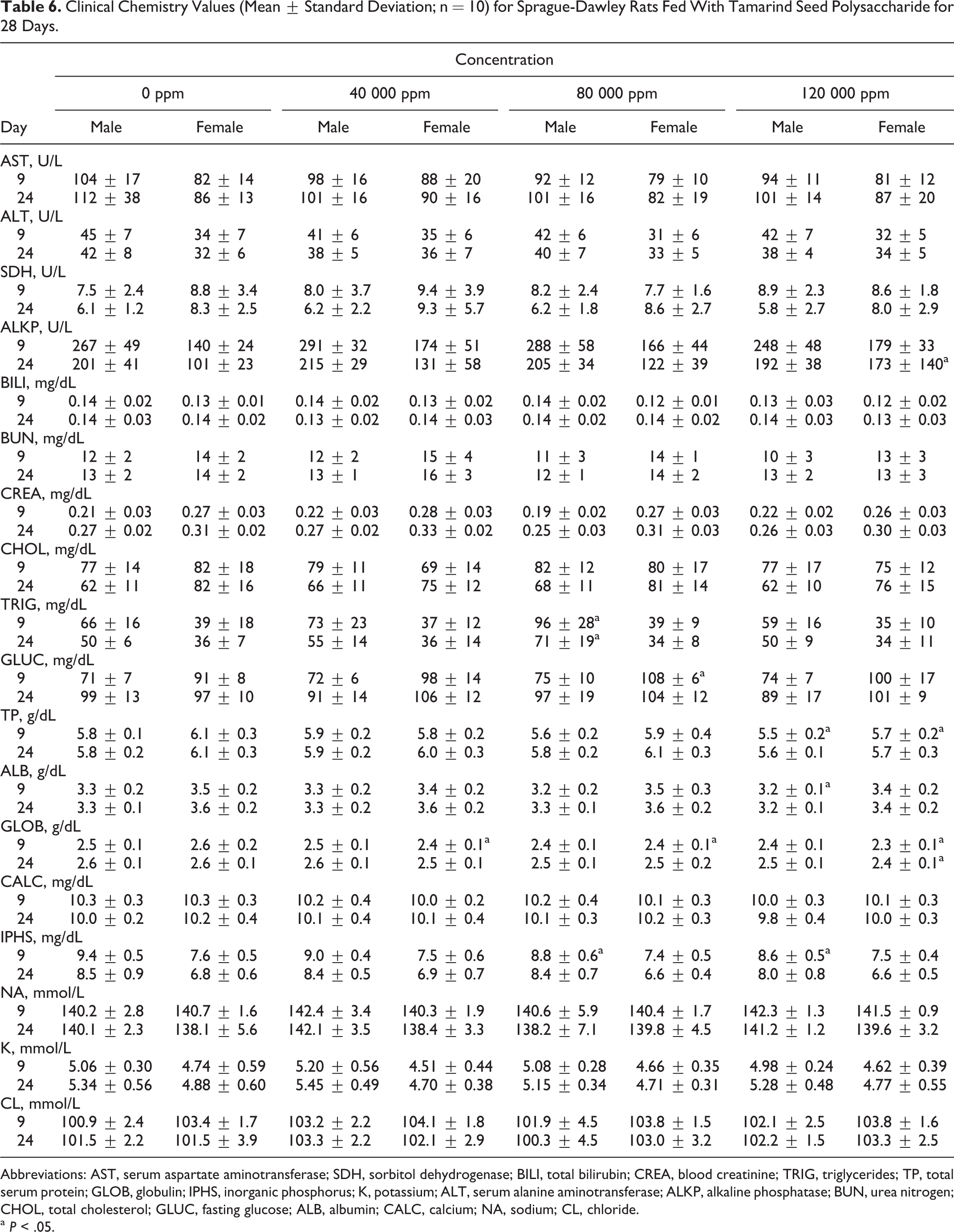
Abbreviations: AST, serum aspartate aminotransferase; SDH, sorbitol dehydrogenase; BILI, total bilirubin; CREA, blood creatinine; TRIG, triglycerides; TP, total serum protein; GLOB, globulin; IPHS, inorganic phosphorus; K, potassium; ALT, serum alanine aminotransferase; ALKP, alkaline phosphatase; BUN, urea nitrogen; CHOL, total cholesterol; GLUC, fasting glucose; ALB, albumin; CALC, calcium; NA, sodium; CL, chloride.
a
Urinalysis
There were no significant changes in mean urinalysis results on days 9 and 24 in male or female rats.
Gross Necropsy
Incidental macroscopic findings reported in males included diverticulum of the jejunum (1 of 10 high-dose rats) and in females included paraovarian cyst (1 of 10 control rats), spleen stricture (1 of 10 high-dose rats), and fluid-filled uterus attributable to variation in the estrus cycle in individual animals (both controls and treated rats). These findings were of sporadic incidence and showed no trends or patterns to suggest a relationship to exposure to the test substance.
Organ Weights
There were no absolute or relative organ weight changes attributable to the administration of tamarind seed polysaccharide. The only significant changes reported were as follows: decreased absolute adrenal weight in high-dose males, decreased absolute, relative-to-body weight, and relative-to-brain weight adrenal weight in low-dose females, and decreased absolute, relative-to-body weight, and relative-to-brain weight uterus-oviduct weight in low- and high-dose females (see Table 7). All of these differences were within the acceptable range, and variance for animals of this age, sex, and strain of rat 22 were not dose-dependent, were not accompanied by clinical or histologic signs, and were considered incidentally and toxicologically insignificant.
Statistically Significant Organ Weight Changes (Mean ± Standard Deviation; n = 10) for Sprague-Dawley Rats Fed With Tamarind Seed Polysaccharide for 28 Days.
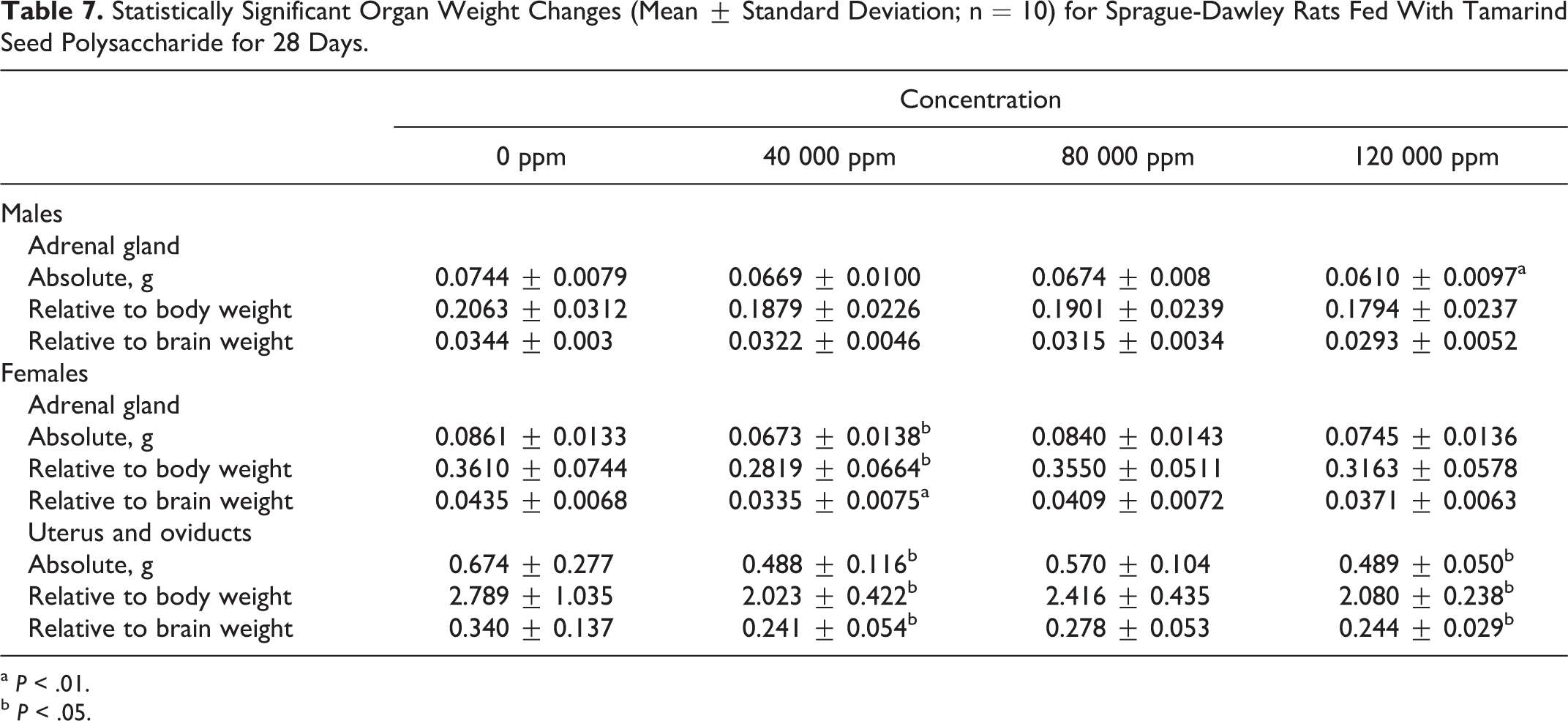
a
b
Histopathological Examination
There were no microscopic findings attributable to the administration of the test substance. Any changes were regarded as sporadic in incidence or spontaneous in nature and were considered unrelated to tamarind seed polysaccharide administration. Incidental changes reported in males consisted of inflammatory cell infiltrates primarily of minimal to slight degree noted in the prostate gland (3 of 10 control and 2 of 10 high-dose rats). Chronic inflammation was characterized by minimal to slight mononuclear cell (lymphocytes and macrophages) infiltrates within the interstitium. Incidental changes reported in females consisted of minimal inflammation in the larynx (2 of 10 high-dose rats). The observed inflammation was granulomatous to pyogranulomatous in nature and involved the caudal portions of the epiglottal cartilage in the anterior portion of the ventral floor of the larynx above the thyroid cartilage. Often, there was bilateral symmetry, and inflammation was centered on the epiglottal cartilage with evidence of cartilage necrosis. These changes exhibited morphologic similarities to laryngeal observations documented for Fischer 344 rats as a spontaneous finding. 23 Any other microscopic findings were considered incidental and typical for the age and strain of the rats used in this study.22,24,25
Estrus Cycles
Vaginal smears taken from surviving females (day 30) just prior to necropsy revealed no cyclic changes that might be considered the result of test substance administration.
Discussion
Overall, the animals tolerated dietary exposure to tamarind seed polysaccharide. Based on stability, homogeneity, and the concentration verification results, the rats were anticipated to have received the targeted 40 000, 80 000 and 120 000 ppm dietary concentrations of tamarind seed polysaccharide. There were no test substance-related or other mortalities during this study. There were no clinical or ophthalmologic signs attributable to test substance administration. Any clinical change including instances of alopecia and ocular discharge across all study groups was considered incidental and unassociated with test substance administration.
There were no body weight, body weight gain, food consumption and food efficiency, FOB, or MA findings associated with the administration of tamarind seed polysaccharide. Initial statistically significant decreases in body weight gain in mid- and high-dose males and feed consumption in high-dose females (days 0-7) resulting from reduced palatability were resolved after the first week and were therefore considered nonadverse and toxicologically insignificant.
There were no adverse changes in hematology, coagulation, clinical chemistry, or urinalysis parameters in male or female rats considered the result of test substance administration. Any statistically significant changes reported was considered toxicologically insignificant, since they were not accompanied by any other corresponding clinical or histopathological change, were very small, occurred in only 1 sex, were not dose dependent, and/or were not significant at study termination.
At scheduled killing on day 29 (males) or day 30 (females), there were no macroscopic or microscopic, estrus cycle, or organ weight changes deemed related to administration of the test substance. Inflammatory cell infiltrates of the prostate gland in control and high-dose males and inflammation of the larynx in high-dose females were considered incidental. Organ weight changes noted with adrenal glands and uterus/oviduct were unassociated with any clinical or microscopic change and were also considered incidental.
Since there were no toxicologically significant effects in this study up to the highest dietary concentration tested of 120 000 ppm, the no-observed-adverse-effect level (NOAEL) for tamarind seed polysaccharide in the diet was considered to be 120 000 ppm or approximately 10 597.1 and 10 690.7 mg/kg bw/day for male and female rats, respectively.
This finding is consistent with the results of earlier research into the oral toxicity of tamarind seed polysaccharide—including a 13-week feeding study in the mouse, a 2-year feeding study in the rat, and a 78-week carcinogenicity study in the mouse—which have invariably established NOAELs at the highest dietary concentrations tested. Additionally, tamarind seed polysaccharide has been approved and widely used as a food additive in Japan and other Southeast Asian countries such as China, Taiwan, and South Korea 1 with no reported adverse events.
It is intended to make tamarind seed polysaccharide available for use in foods such as ice cream and frozen yogurt, sauces and condiments including barbecue sauce and ketchup, mayonnaise and salad dressings, jams and jellies, puddings, pulp-containing fruit juices, bread and pastries, soups, processed cheeses, and candies. The addition level of tamarind seed polysaccharide in these categories of foods would range from less than 0.2% to a maximum of 1.5%. Based on food consumption data from the 2003 to 2006 National Health and Nutrition Examination Survey,26,27 the mean daily intake of tamarind seed polysaccharide among those aged 2 years or older—if it were added to all permitted foods at maximum permitted levels—would be 44.9 mg/kg bw/day, while the 90th percentile intake would be 91.0 mg/kg bw/day.
Since the NOAEL, 10 597 mg/kg bw/day in males and 10 691 mg/kg bw/day in females, in the 28-day feeding study was more than 100 times this level for both male and female rats, it is concluded that the results are supportive of the safety of the intended use of tamarind seed polysaccharide as a food stabilizer and thickener.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors Heimbach and Kennepohl received monetary compensation from DSP Gokyo Food & Chemical Co., Ltd., Osaka, Japan.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by DSP Gokyo Food & Chemical Co, Ltd, Osaka, Japan.
