Abstract
The functions of alkyl benzoates in cosmetics include fragrance ingredients, skin-conditioning agents—emollient, skin-conditioning agents—miscellaneous, preservatives, solvents, and plasticizers. The Cosmetic Ingredient Review Expert Panel reviewed the relevant animal and human data and noted gaps in the available safety data for some of the alkyl benzoates. Similar structure activity relationships, biologic functions, and cosmetic product usage allowed the available data of many of the alkyl benzoates to be extended to the entire group. Carcinogenicity data were not available, but available data indicated that these alkyl benzoate cosmetic ingredients are not genotoxic. Also benzoic acid and tested component alcohols were not reproductive or developmental toxicants, are not genotoxic in almost all assays, and are not carcinogenic. These ingredients were determined to be safe in the present practices of use and concentration.
Keywords
Introduction
Alkyl benzoate esters function in cosmetics as fragrance ingredients, skin-conditioning agents—emollient, skin-conditioning agents—miscellaneous, preservatives, solvents, and plasticizers. The ingredients included in this safety assessment are methyl benzoate, ethyl benzoate, propyl benzoate, butyl benzoate, amyl benzoate, lauryl/myristyl benzoate, C12-15 alkyl benzoate, C16-17 alkyl benzoate, stearyl benzoate, behenyl benzoate, isopropyl benzoate, isobutyl benzoate, isostearyl benzoate, ethylhexyl benzoate, butyloctyl benzoate, hexyldecyl benzoate, and octyldodecyl benzoate. These ingredients are esters of benzoic acid and a corresponding alcohol, with the shorter chain alkyl benzoates (methyl, ethyl, propyl, isopropyl, butyl, isobutyl and amyl benzoate) ranging in molecular weight (MW) from 136 to 192 and the longer chain alkyl acetates (lauryl/myristyl, C12-15 alkyl, C16-17 alkyl, stearyl, isostearyl, behenyl, ethylhexyl, butyloctyl, hexyldecyl, and octyldodecyl benzoate) ranging in MW from 234 to 431.
Because the smaller alkyl benzoates in this report are considered likely to penetrate the skin, it should be expected that they would be metabolized in the skin to benzoic acid and the parent alcohol. Therefore, the safety of these metabolites must be considered when assessing the safety of alkyl benzoates.
Several of the metabolites of the alkyl benzoates in this assessment have been reviewed by the Cosmetic Ingredient Review (CIR) Expert Panel (benzoic acid, sodium benzoate, and the parent alcohols: methyl alcohol, ethyl alcohol, butyl alcohol, myristyl alcohol, behenyl alcohol, isostearyl alcohol). The conclusions are listed below.
Benzyl alcohol, benzoic acid, and sodium benzoate: the conclusion currently states that these ingredients are safe for use in cosmetic formulations at concentrations up to 5%. 1 This safety assessment is currently being reevaluated to possibly remove the limit.
Methyl alcohol: it is safe for use with qualifications as a denaturant in ethyl alcohol for cosmetic products. The panel has not stated that methyl alcohol is safe or unsafe as a solvent. 2
Ethyl alcohol (as “alcohol denaturant”): it is safe in the present practices of use and concentration (up to 99% alcohol denaturant). 3 The CIR Expert Panel considered the safety of cosmetic products containing up to 99% alcohol denaturant and were not concerned regarding dermal exposure to ethyl alcohol from these products. The potential for exposure to ethyl alcohol from ethyl benzoate (maximum use concentration 0.01% reported) is much lower.
Butyl alcohol: it is safe as a cosmetic ingredient in the present practices of use. 4 In 2005, the panel looked at new data and the safety conclusion in the report was confirmed.
Myristyl alcohol: it is safe as a cosmetic ingredient in the present practices of use. 5
Cetyl alcohol: it is safe as a cosmetic ingredient in the present practices of use. 5 In 2005, the Panel reviewed new data and the conclusion in the report was confirmed.
Stearyl alcohol: it is safe as currently used in cosmetics. 6 In 2006, the Panel reviewed new data and the conclusion in the report was confirmed.
Isostearyl alcohol: it is safe for use as a cosmetic ingredient in the present practices of use. 5 In 2005, the Panel reviewed new data and the conclusion in the report was confirmed.
Behenyl alcohol: it is safe as a cosmetic ingredient in the present practices of use. 5 In 2005, the Panel reviewed new data and the conclusion in the current report was confirmed.
Propyl alcohol and isopropyl alcohol: it is safe for use in cosmetic products in the present practices of use and concentration. 7
The probable alcohol metabolites of ethylhexyl benzoate, butyloctyl benzoate, hexyldecyl benzoate, isobutyl benzoate, amyl benzoate, pentadecyl benzoate, heptadecyl benzoate, and octyldodecyl benzoate are not current cosmetic ingredients in the dictionary and thus have not been reviewed by CIR.
Some data from the reports on benzoic acid, sodium benzoate, methyl alcohol, butyl alcohol, myristyl alcohol, cetyl alcohol, stearyl alcohol, isostearyl alcohol, behenyl alcohol, propyl alcohol, and isopropyl alcohol are considered. Data on isononyl benzoate, a related branched alkyl benzoate, and the other alcohols as well as ethylhexyl alcohol (from a Research Institute for Fragrance Materials [RIFMs] Expert Panel review) 8 are included to give a sense of the safety of these likely metabolites.
Chemistry
Definition and Structure
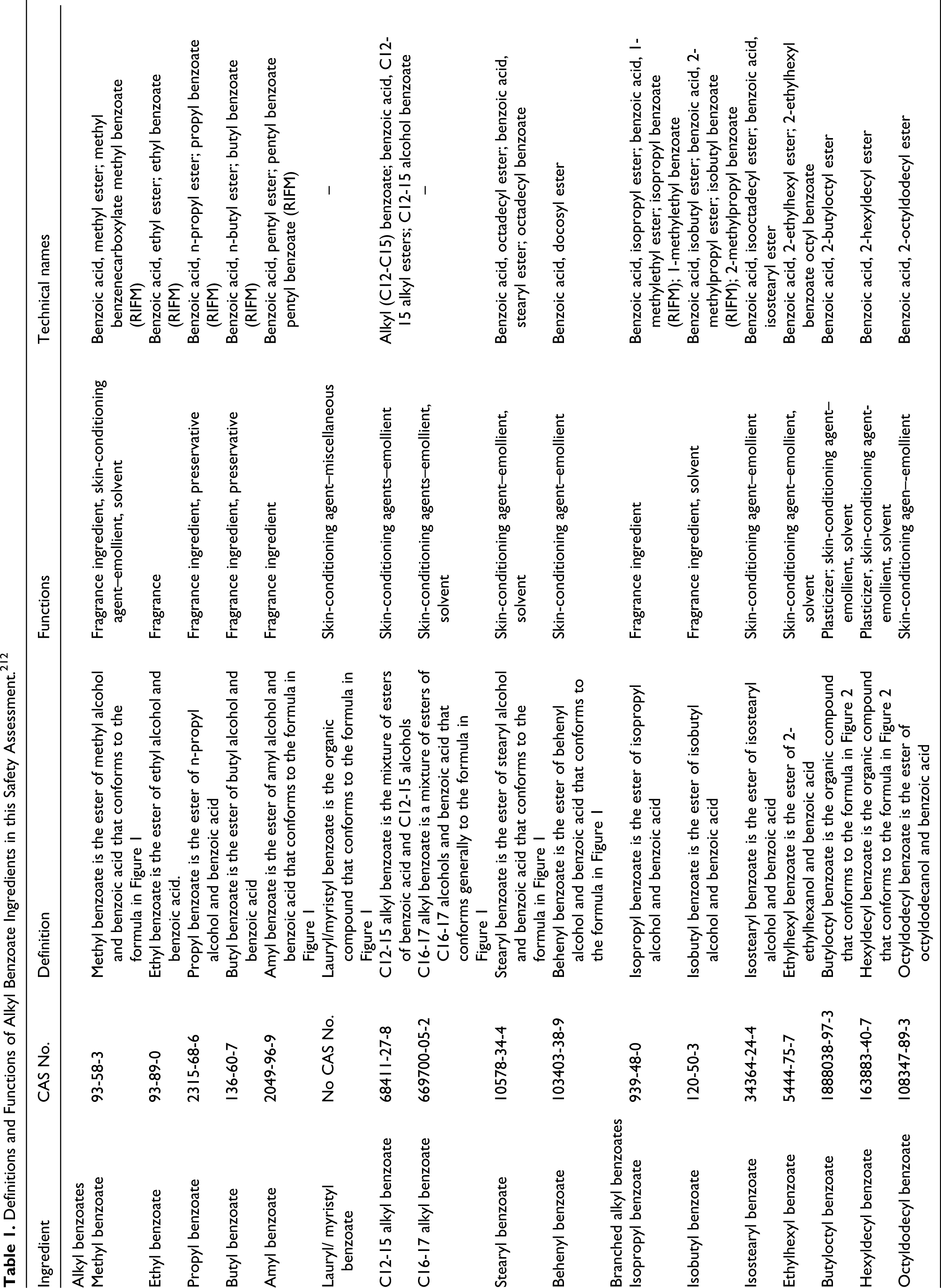
Alkyl benzoates mostly function as skin-conditioning agents, preservatives, solvents, and plasticizers. The CAS numbers, definitions, functions, and technical and trade names of the ingredients under review are presented in Table 1. Structures and potential metabolic pathways of these ingredients are presented in Figures 1 and 2.
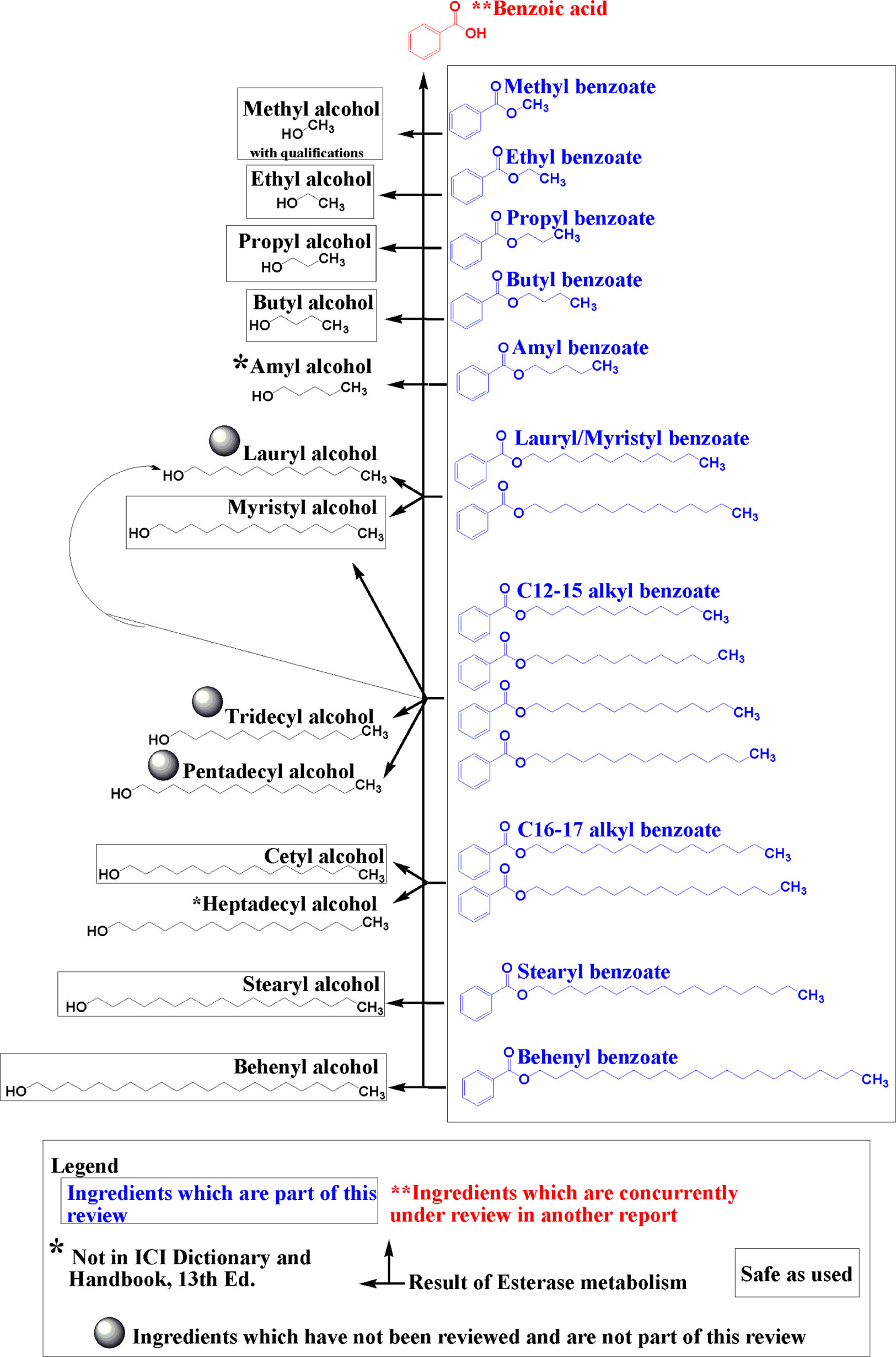
Straight chain alkyl benzoates: structures, esterase metabolism, and metabolites.
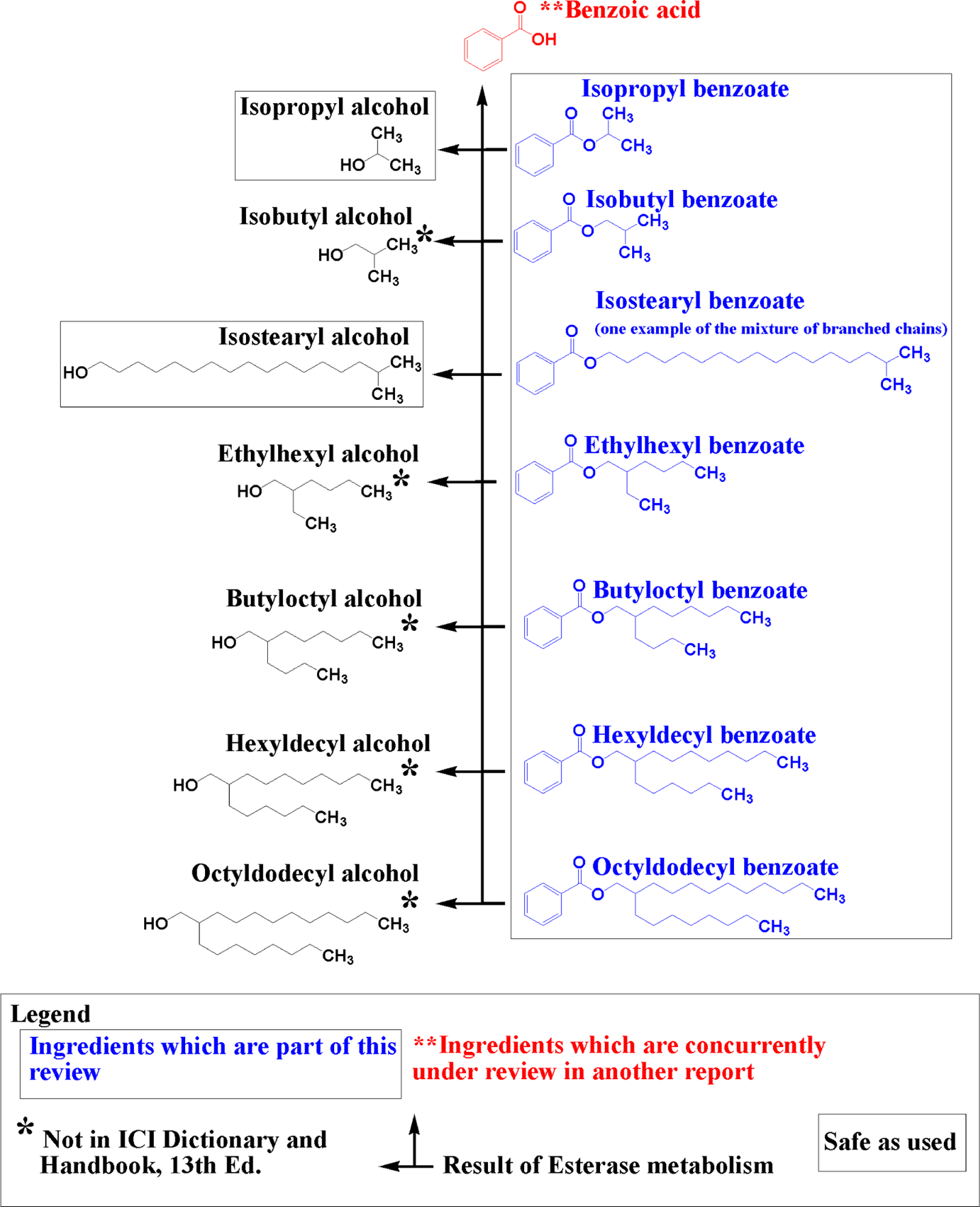
Branched chain alkyl benzoates: structures, esterase metabolism, and metabolites.
Definitions and Functions of Alkyl Benzoate Ingredients in this Safety Assessment. 212
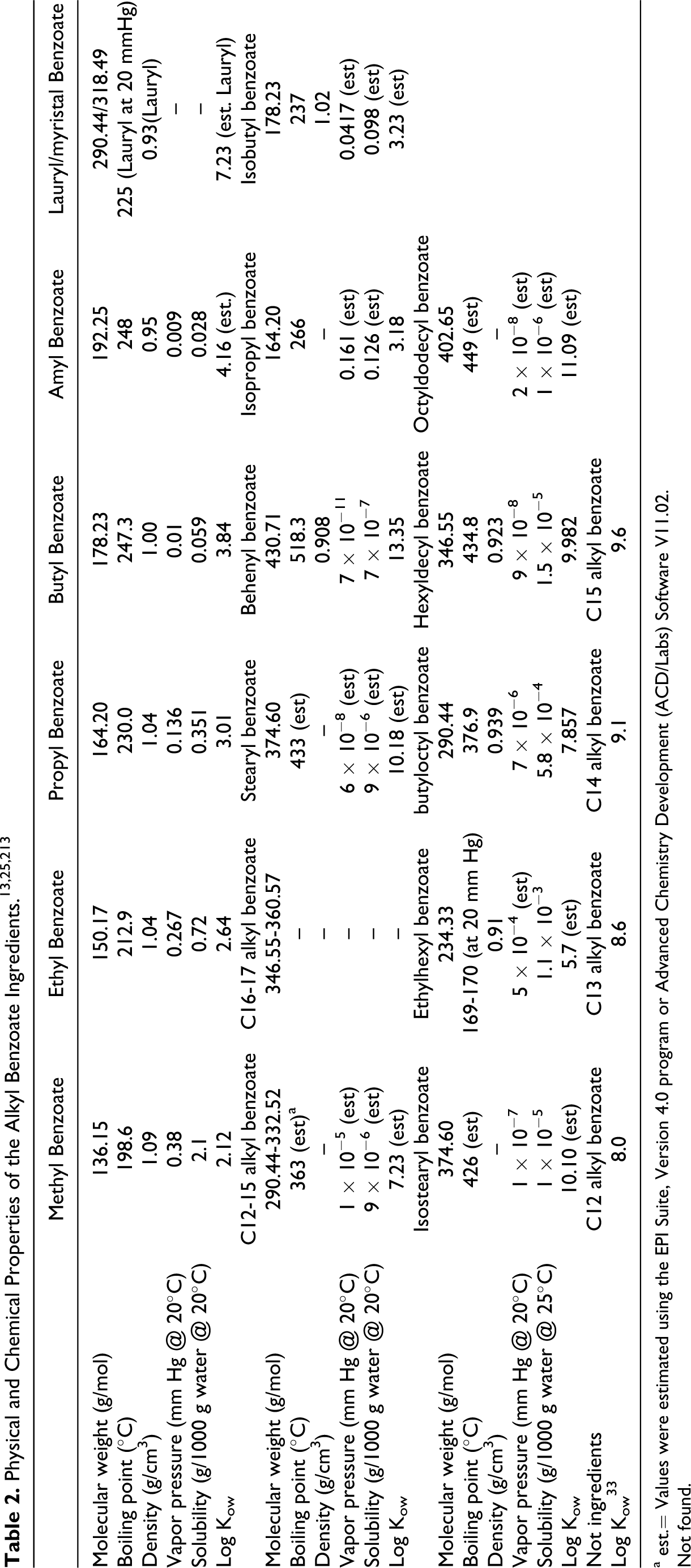
Physical and Chemical Properties
The shorter chain alkyl benzoate esters are colorless liquids. Viscosity generally increases as the molecular mass (chain length) increases. 9 The physical and chemical properties of the benzoates are shown in Table 2. At room temperature and pressure, methyl benzoate, ethyl benzoate, butyl benzoate, and isobutyl benzoate are fragrant, colorless oils, and are insoluble in water. 10 A ultraviolet (UV) absorption spectrum of C12-15 alkyl benzoates was maximum at ∼200 and 235 nm. 11
a est.= Values were estimated using the EPI Suite, Version 4.0 program or Advanced Chemistry Development (ACD/Labs) Software V11.02.
Not found.
Manufacture and Production
In general, the alkyl benzoates can be produced industrially via esterification of benzoic acid. 9 The manufacture of butyl benzoate, for example, is traditionally accomplished via an acid-catalyzed (eg, sulfuric acid) reactive distillation process between benzoic acid and butyl alcohol (Figure 3). 12
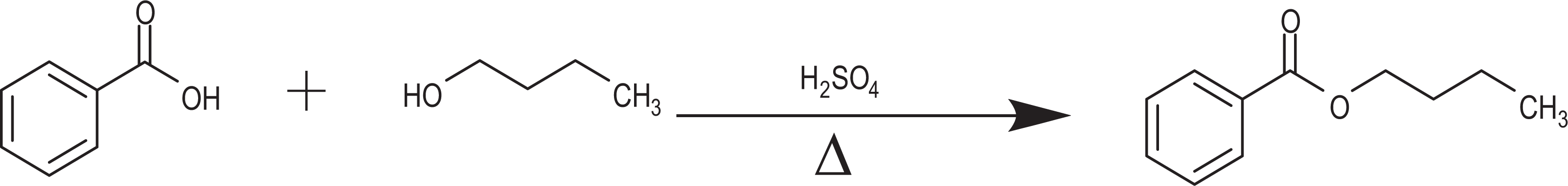
The synthesis of butyl benzoate.
Methanol and ethanol are normally obtained via fermentation of natural sources. However, some alcohols with chains longer than ethanol are often produced synthetically. An important process for producing C3- C22 industrial alcohols involves a process known as oxosynthesis (a process for the production of aldehydes which occurs by the reaction of olefins (which can be natural or petroleum sourced) with carbon monoxide, hydrogen, and a catalyst (typically cobalt based), followed by hydrogenation of the aldehyde products, to form the alcohols. 13 A biocatalytic process specifically for the manufacture of esters for use in the formulation of cosmetic and personal care ingredients (ie, for producing cosmetic grade esters) was developed in 2004. 14
Impurities
The manufacturing processes of the benzoic esters are typically high yielding (>90%) and easily purified (eg, by distillation). Therefore, the starting materials and water, at least, may be expected to be present in preparations of these esters as the major impurities.
9
For example, methyl benzoate is available with a minimum of 99.2% purity, wherein the major contaminants are water (
Analytical Methods
The benzoic esters can be analyzed using gas chromatography/mass spectrometry (GC/MS), nuclear magnetic resonance (NMR) spectroscopy, UV spectroscopy, and infrared (IR) spectroscopy.9,13,16 High-performance liquid chromotography (HPLC) was used to detect C12-15 alkyl benzoates. 17
Use
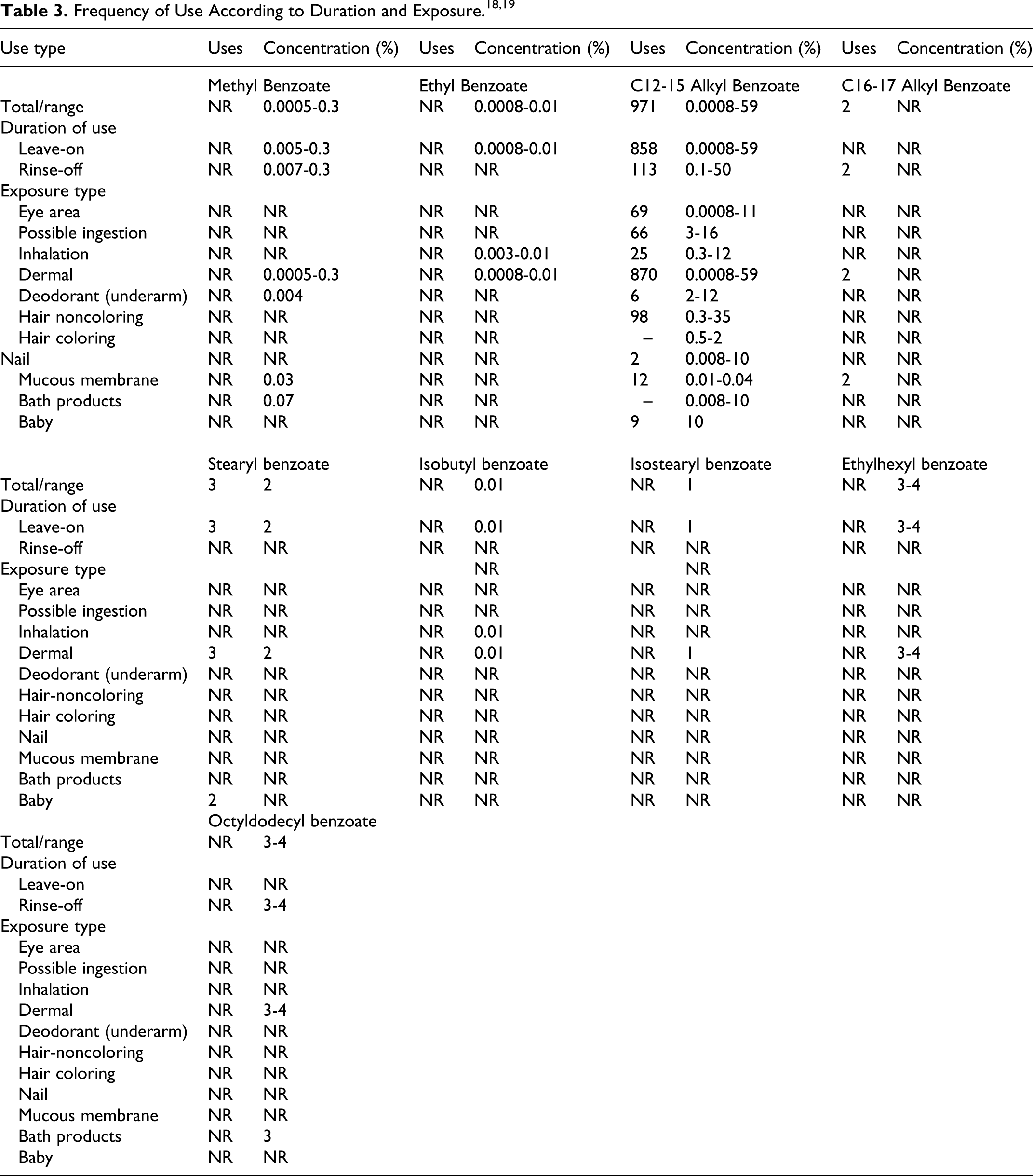
Cosmetic
According to the Voluntary Cosmetic Registration Program (VCRP) administered by the Food and Drug Administration (FDA), the total number of uses of C12-15 alkyl benzoate was 971 (858 leave-on and 113 rinse-off products). 18 A survey conducted by the Personal Care Products Council (Council) found that C12-15 alkyl benzoate was used at 0.0008% to 59% (highest concentration in tonics, dressings, and other hair grooming aids) in leave-on products and 0.0008% to 50% (highest concentration in paste masks [mud packs]) in rinse-off products (Table 3). 19 There were 2 uses reported of C16-17 alkyl benzoates (bath soaps and detergents). Stearyl benzoate was reported to have 3 uses (including face and neck creams, lotions, and powders) and used at 2%. While there were no uses reported by VCRP, the Council reported methyl benzoate use at 0.0005% to 0.3% (highest concentration in perfumes), ethyl benzoate use at 0.0008% to 0.01% (highest concentration in foot powders and sprays), isobutyl benzoate use at 0.01% (perfumes), isostearyl benzoate use at 1% (body and hand creams, lotions, and powders), and octyldodecyl benzoate at 3% to 4% (highest concentration in shaving cream). No uses or concentrations of use were reported for propyl benzoate, butyl benzoate, amyl benzoate, lauryl/myristyl benzoate, behenyl benzoate, isopropyl benzoate, ethylhexyl benzoate, butyloctyl benzoate, and hexyldecyl benzoate.
C12-15 Alkyl benzoate and other benzoates are used in hair sprays and perfumes, and effects on the lungs that may be induced by aerosolized products containing these ingredients are of concern. The aerosol properties that determine deposition in the respiratory system are particle size and density. The parameter most closely associated with deposition is the aerodynamic diameter, da, defined as the diameter of a sphere of unit density possessing the same terminal settling velocity as the particle in question. In humans, particles with an aerodynamic diameter of ≤10 µm are respirable. Particles with a da from 0.1 to 10 µm settle in the upper respiratory tract and particles with a da < 0.1 µm settle in the lower respiratory tract.20,21
Particle diameters of 60 to 80 µm and ≥80 µm have been reported for anhydrous hair sprays and pump hairsprays, respectively. 22 In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range, and the mean particle diameter in a typical aerosol spray has been reported as ∼38 µm. 23 Therefore, most aerosol particles are deposited in the nasopharyngeal region and are not respirable.
In the European Union (EU), methyl benzoate, ethyl benzoate, propyl benzoate and butyl benzoate may be used as preservatives in cosmetics up to 0.5% (acid). 24
Noncosmetic
Alkyl benzoate esters are typically used as solvents in paints, lacquers and coatings, and as intermediates in various chemistry processes. 9 Methyl benzoate is used in flavoring and perfumery and as a solvent in resins. 25 Ethyl benzoate is used in flavoring and perfumery and as a solvent in lacquers and resins. 25 Butyl benzoate is used as a solvent for cellulose ether, as a plasticizer, as a perfume ingredient, and for dyeing in textiles. 25 Isobutyl benzoate is used in flavoring and perfumery. 25 Methyl benzoate, ethyl benzoate, propyl benzoate, isopropyl benzoate, and isobutyl benzoate have been approved by the FDA as flavors 26 and have no safety concerns when used as flavoring agents. 27
Toxicokinetics
Orally administered benzoate esters are metabolized to benzoic acid and the corresponding alcohols, and the acid is further metabolized to benzyl glucuronide and benzoyl CoA, 28 an intermediate in the formation of hippuric acid, which is the principal metabolite excreted in the urine.
In general, esters can be hydrolyzed to the parent alcohol and acid by enzymes.9,28 Secondary and tertiary esters are hydrolyzed more slowly than primary esters. These enzymes are in essentially all tissues, including the respiratory tract, skin, and gastrointestinal tract, and blood.29,30
Data on benzyl alcohol show that it is converted into benzoic acid by simple oxidation. 1 Orally consumed benzoic acid is absorbed from the gastrointestinal tract and conjugated with glycine in the liver. The resulting hippuric acid is excreted in the urine (75%-100% within 6 hours). Dermally applied benzoic acid is also excreted in the urine within 24 hours.
In general, alcohols can be metabolized by alcohol dehydrogenases to aldehydes (primary alcohols) or ketones (secondary alcohols). Aldehydes are further metabolized by aldehyde dehydrogenases to the corresponding acids.
Benzoic esters are not absorbed through the skin as rapidly as alkyl esters. 31 If alkyl benzoates are absorbed and metabolized, the alcohols resulting from ester hydrolysis can be oxidized via alcohol dehydrogenases to produce the corresponding aldehyde or ketone. As noted above, these aldehydes can be further oxidized via aldehyde dehydrogenases, aldehyde oxidases, or xanthine oxidases to the corresponding acids.
Dermal Penetration
Short chain length
The penetration of methyl benzoate, ethyl benzoate, n-propyl benzoate, and n-butyl benzoate through excised guinea pig dorsal skin was measured using diffusion cells. 32 The permeability coefficients (Kp) were 20.3 ± 5.8 × 10−2 cm/h, 34.08 ±1.2 × 10−2 cm/h, 62.7 ± 13.0 × 10−2 cm/h, and 79.9 ± 10.1 × 10−2 cm/h, respectively, indicating significant steady-state dermal penetration. Permeability was increased by removal of the stratum corneum by tape stripping and delipidization using a chloroform–methanol mixture. Penetration was decreased by the addition of l-methanol plus 15% ethanol.
Long chain length
In another study, C12-15 alkyl benzoate was applied neat at a dose of 4 mg/cm2 to 3 samples of gently shaved skin from the same pig. 33 Total recovery after 24 hours was 82% of the applied dose, and 29% of the dose was recovered in the skin as measured by HPLC (detection limit 1.0 µg/mL). Of the material recovered, 84%, 5%, and 11%, was in the horny layer, epidermis, and dermis, respectively. None was detected in the receptor fluid.
C12-15 Alkyl benzoates applied to frozen and fresh pig skin did not penetrate the skin. 34 None of the test material was detected in the receptor fluid, 50.76% was recovered in the skin, and 34.04% was still on the skin. There were benzoate compounds (<C12) present in the test material at 4.7%.
Octanol/water partition coefficient values (at pH 3.0) of 8.0, 8.6, 9.1, and 9.6 for C12 alkyl benzoate, C13 alkyl benzoate, C14 alkyl benzoate, and C15 alkyl benzoate would suggest that these longer chain length alkyl benzoates would stay in the stratum corneum and would not penetrate beyond the epidermis. 35
The in vitro dermal absorption and percutaneous penetration of C12-15 alkyl benzoate from 3 product formulations in excised pig skin after 24 hours were examined. A sun lotion (with 7.5% test compound), a baby cream 5.4% (with test compound), and a sun protection spray (with 6.6% test compound) were applied at a rate of 4 mg/test/sample/cm2. Total recoveries were 90%, 88%, and 95% for 3 formulations with 21%, 34%, and 26% found in the skin. The amount in the receptor fluid and dermis for all 3 formulations was less than the detection limit. The amount of C12-15 alkyl benzoate from the sun lotion was 93.5% and 6.5% in the horny layer and epidermis, respectively. For the baby cream, the amount of recovered test substance was 91.5% and 8.6%; and for the sun protection spray, the amount of recovered test substance was 92.5% and 7.5%.
Alcohols
The permeability constants (Kp) for methyl alcohol using human cadaver skin were 0.3, <0.1, 12.0, 3.0, and 2.5 × 103 cm/h in saline, polyethylene glycol 600, isopropyl palmitate, olive oil, and mineral oil, respectively. 36
The permeability coefficient of methyl alcohol was 5.6 ± 1.2 × 103 cm/h through the nail plates of cadavers. 37
Male Fischer F344 rats (n = 3) were orally administered [13C]-tert-amyl alcohol (250 mg/kg in corn oil) and urine was collected for 48 hours. 38 The major metabolites were tert-amyl alcohol glucuronide and 2-methyl-2,3-butanediol and its glucuronide. Free tert-amyl alcohol, 2-hydroxy-2-methylbutric acid, and 2-hydroxy-3-methylbutyric acid were minor metabolites.
Isopropyl alcohol was absorbed in 4 groups of rabbits (n = 3; strain not specified) exposed to alcohol by gavage (group 1: 2 mL/kg; group 2: 4 mL/kg), whole-body/inhalation combined with dermal application (group 3: 70% isopropyl alcohol soaked towel applied to the chest), and whole-body/inhalation combined with application over a plastic barrier on the chest for 4 hours. 39 Maximum isopropyl alcohol concentrations in blood after oral exposures were 147 mL/dL (2 mL/kg) and 282 mg/dL (4 mL/kg), which were correlated with inebriation and near coma; the blood concentration was 112 mg/dL 4 hours after whole-body/inhalation combined with dermal application. Blood concentrations of acetone (metabolite of isopropyl alcohol) were 74 mg/dL (2 mL/kg by gavage), 73 mg/dL (4 mg/kg by gavage), 19 mg/dL (whole-body/inhalation plus dermal application), and <10 mg/dL (whole-body/inhalation plus application over a plastic barrier). The authors concluded from their results that significant toxicity would require repeated sponging or soaking with isopropyl alcohol for several hours.
In vitro absorption rates for ethylhexyl alcohol in aqueous solution through rat and human skin were 0.22 ± 0.09 and 0.38 ± 0.014 mg/cm2/h, respectively. 40 The corresponding permeability constants were 2.59 ± 1.10 × 10−4 cm/h for rat skin and 4.54 ± 1.66 × 10−5 for human skin.
The absorption rate for ethylhexyl alcohol (1000 mg/kg) applied to the skin of rats for 6 hours was 0.57 mg/cm2/h; 5.2% of the dose was absorbed during exposure. 41
Cytotoxicity
Methyl Benzoate
In a protein count assay (measuring protein synthesis) of methyl benzoate, the EC50 (50% of the concentration of maximum effect) was 1506.58 (CI 1349.27-168.22) mmol/L, the NI50 (the concentration that reduced the uptake of neutral red by 50%) was 683.30 (466.46-1000.91) mmol/L in a neutral red uptake assay, and the ID50 (the concentration that inhibited growth by 50%) was 987.19 (605.15-1610.43) mmol/L in a growth inhibition assay using HeLa cells. 42
Methyl benzoate (2.5 and 5.0 mg/mL) inhibited mycelia growth and aflatoxin release by Aspergillis flavus and A parasiticus. 43
Human diploid embryonic lung fibroblasts (line MRC-5), labeled with [ 3 H]uridine, were incubated in methyl benzoate (25 mmol/L in buffered saline) for 30 minutes. 44 The amount of cell wall damage was measured by the release of the label. Controls released 3% to 6% of the maximum available label. Incubation in methyl benzoate caused a release of 20% of maximum available label. The authors concluded that methyl benzoate not only caused toxic effects to the cells but also promoted membrane penetration by other substances.
Ethyl Benzoate
Human Hep-2 cells (epithelial cell line derived from human carcinoma of the larynx) were exposed to ethyl benzoate. 45 Total inhibition of cell growth was observed at 500 mg/L. This experiment was repeated and samples were taken for protein determination. There were no effects at 289 mg/L. This experiment was repeated and the cells were stained and examined for morphology. At 289 mg/L, the cells lost their typical epithelial shape and became elongated.
The above experiment on human diploid embryonic lung fibroblasts (line MRC-5), labeled with [ 3 H]uridine, was repeated with ethyl benzoate (25 mmol/L). 44 Incubation in ethyl benzoate caused a release of 31% of maximum of available label. The authors concluded that ethyl benzoate not only caused toxic effects to the cells but also promoted membrane penetration by other substances.
Propyl Benzoates
Human Hep-2 cells were exposed to propyl benzoate. 45 Total inhibition of cell growth was observed at 200 mg/L. This experiment was repeated and samples were taken for protein determination. Effects (not defined) were observed at 122 mg/L and the cells seemed to recover by day 7. This experiment was repeated and the cells were stained and examined for morphology. At 122 mg/L, the monolayer of the cells was disturbed within 24 hours. There were no other effects observed.
Butyl Benzoate
Human Hep-2 cells were exposed to butyl benzoate. 45 Total inhibition of cell growth was observed at 100 mg/L. This experiment was repeated and the samples were taken for protein determination. Effects (not defined) were observed at 61 mg/L and the cells seemed to recover by day 7. This experiment was repeated and the cells were stained and examined for morphology. At 61 mg/L, the monolayer of the cells was disturbed within 24 hours. There were no other effects observed.
Human Rhino HeLa cells were incubated in butyl benzoate (in dimethylsulfoxide) for 48 h. The IC50 was 0.5 mmol/L. 46
Alcohols
Methyl alcohol had a 50% production inhibition (PI50) of 1614 mmol/L for Hep G2 cells. 47 Methyl alcohol was toxic to yeast cells (strain ade6-60/rad10-198, h- of Schizosaccharomyces pombe P1 strain) at 0.05% but not V79 Chinese hamster cells up to 10%. 48 Amyl alcohol was toxic to yeast cells (strain ade6-60/rad10-198, h- of S pombe P1 strain) and V79 Chinese hamster cells at 0.5%. 48 Dodecyl alcohol had 50% lysis of human erythrocytes at 15 µmol/L. 49
Toxicology
Single-Dose Toxicity
Methyl benzoate
The reported oral LD50 of methyl benzoate was 2170 mg/kg for rabbits, 4100 mg/kg for guinea pigs, 1350 to 3500 for rats, and 3000 to 3330 mg/kg for mice.50–53
The dermal LD50 of methyl benzoate was >2000 mg/kg for New Zealand white rabbits (n = 5). 54 There was fecal staining for 3 days after treatment. Irritation was observed at the application site. There was weight loss for 1 to 7 days after treatment. There were no gross findings at necropsy. There were no mortalities.
Ethyl benzoate
The reported oral LD50 of ethyl benzoate was 2630 mg/kg for rabbits, 2100 mg/kg for rats, and 6480 mg/kg for female rats.50,53
Ethyl benzoate (10% in acetone) was administered to one-third of the body surface of mice (n = 2 – 4). 55 The mice were observed for 24 h and then necropsied. There were no effects at 10%. This experiment was repeated with calves (with no necropsy) except covering the entire body surface and a 15-day observation period. There were no effects at 10%. No further details were provided.
Ethyl benzoate (up to 100% in “various vehicles”; 20 mL) was administered to the clipped backs of cats (n = 2), massaged into the skin with cotton balls. 50 The cats were to be observed for 2 weeks. At 100%, both cats died within 20 hours. Albino rats (n = 6) showed no adverse effects from exposure to aerosolized ethyl benzoate (approaching saturation) for 8 hours. 53
Intramuscular administration of ethyl benzoate (100%; 0.5 or 1.0 mL) administered to guinea pigs (n not provided) caused musculoskeletal [sic], moderate deterioration of leg function and muscle toughness at 1.25 mL/kg. 56
Butyl benzoate.The reported oral LD50 of butyl benzoate was 5.14 g/kg for female rats. 53 Dermally administered butyl benzoate (5 g/kg) caused no mortalities in rabbits (n =10). 57 Diarrhea was observed during the 14-day observation period.
Intramuscular administration of butyl benzoate (100%; 0.5 or 1.0 mL) to guinea pigs (n not provided) caused musculoskeletal [sic], moderate deterioration of leg function, and muscle toughness at 3 mL/kg. 56
Albino rats (n = 6) showed no adverse effects from exposure to aerosolized butyl benzoate (approaching saturation) for 8 hours. 53
C12-15 Alkyl benzoate
Albino rats orally administered C12-15 alkyl benzoate (5.0 g/kg) exhibited no signs of toxicity over a 14-day observation period. 58 At necropsy, enlarged spleens were noted.
In an acute oral study, C12-15 alkyl benzoate (up to 100%; 40 mL/kg) was administered to albino rats. 59 The rats were observed for 14 days and then killed and necropsied. There was no mortality during the observation period. There were no gross internal changes.
At 30.0 g/kg, the females were described as having slight depression up to day 7. At 24 h, loss of ventral body hair and crusty, scabby skin were noted. There were no gross internal changes. At 33.0 g/kg, hair was matted and unkempt, there was a crust-like substance on the skin and hair loss. One female rat died on day 7. At 37.0 g/kg, 5 rats died. Pyloric and intestinal mucosa were reddened; lung tissue was enlarged and consolidated; and spherical lesions were observed in the lungs.
Female MNRI EOPS mice were orally administered 5000 mg/kg C12-15 alkyl benzoate and observed for 6 days. 60 There were no mortalities and no clinical signs or behavior changes were observed. Weight gain was comparable to controls.
In a dermal toxicity study, C12-15 alkyl benzoate (100%; 2 g/kg) was applied to the intact skin of albino rabbits (n = 6; 3/sex) under occlusion. 59 The rabbits were observed for 14 days. One male rabbit died which was considered nontreatment related. The authors concluded that C12-15 alkyl benzoate was not a toxic material.
Albino Wistar rats (n = 10) were exposed to aerosolized C12-15 alkyl benzoates (200 mg/L) for 1 hour and observed for 2 weeks. 61 There were no toxic effects observed.
Isopropyl benzoate.The reported oral LD50 of isopropyl benzoate was 3730 mg/kg and 3.7 mg/kg for rats.62,63 The reported dermal LD50 of isopropyl benzoate was 20 mL/kg for rabbits. 62 Dermally administered isopropyl benzoate (5 mL/kg) had no effects to rabbits (n = 2). 64 There were no observed effects in rats exposed to aerosolized isopropyl benzoate (saturated vapor) for 4 hours. 62
Isobutyl benzoate
The reported oral LD50 of isobutyl benzoate was 3685 mg/kg28 and 3.7 mL/kg for rats (n = 10). 64
Isobutyl benzoate (5 mL/kg; 100%) was applied to the intact and abraded clipped skin of albino rabbits (n = 4) for 24 h. There were no mortalities or clinical signs during the 14-day observation period. 64
Benzoic acid and sodium benzoate
The oral LD50 of benzoic acid was reported to be 1996 mg/kg in mice and 2000 to 2500 mg/kg in rats. The oral LD100 was reported to be 1520 to 2000 mg/kg for rabbits, and 2000 mg/kg for cats and dogs. The oral LD50 for sodium benzoate was 2100 to 4070 mg/kg for rats and 2000 mg/kg for rabbits and dogs. 65
Alcohols
Methyl alcohol has an oral LD50 of 5628 mg/kg for rats and 7300 mg/kg for mice. 66 The dermal LD50 of methyl alcohol was reported to be 15,800 mg/kg in rabbits. 67 Methyl alcohol has an intraperitoneal (ip) LD50 of 336 (CI = 299, 373) mmol/kg for mice, 237 (222, 252) mmol/kg for rats, 267 (235, 304) mmol/kg for hamsters, and 111 mmol/kg for guinea pigs. 68 The intravenous (iv) LD50 for mice is 147 (126, 171) mmol/kg, 66.5 (61.5, 71.2) mmol/kg for rats and 278 (185, 371) mmol/kg for rabbits.
The oral LD50 of amyl alcohol for Sprague-Dawley rats was reported to be 2.69 g/kg. 69 Deaths occurred within 24 hours. Necropsy revealed evidence of gastrointestinal irritation and pooling of blood.
The dermal LD50 of amyl alcohol for albino rabbits was reported to be >3.2 g/kg. 69 There were signs of central nervous system (CNS) depression. Recovery occurred within 4 to 48 hours.
Swiss mice, Wistar rats, and English short hair guinea pigs (n = 10) were exposed to aerosolized amyl alcohol at near saturation for 6 hours and observed for 14 hours. 69 Preconvulsive movements were observed in mice and guinea pigs; the rats tended more toward prostration. Two rats and 7 mice died during exposure. All surviving animals recovered shortly after termination of the exposure. Some animals had irritation of the eyes, nose, throat, and respiratory passages.
Dodecyl alcohol has an oral LD50 of 12 800 mg/kg for rats. 66 The dermal LD50 of dodecyl alcohol was reported to be 3560 mg/kg in rabbits. 67 Tridecyl alcohol has an oral LD50 of 17 200 mg/kg for rats. 66 The dermal LD50 of tridecyl alcohol was reported to be 5600 mg/kg in rabbits. 67 Ssc:CF-1 mice (n = 10) were exposed to tert-pentadecyl alcohol (2800-14 000 ppm), with and without tracheal cannulation, after anesthesia for 30 minutes. 70 Sensory irritation of the upper respiratory tract was measured by timing the pauses before exhalation compared with those of untreated controls (n = 37). Stimulation of pulmonary receptors by airborne irritants was measured by the decrease in respiratory rate caused by a pause between the end of expiration and the beginning following inspiration, resulting in a net decrease in respiratory rate.
The characteristic sensory irritation pattern was observed in the mice immediately after the onset of the exposure. The pattern was most evident within the first minute and was followed by rapid fading of the responses. The pattern was occasionally seen during the entire exposure period. After a decrease in respiratory rate in the first minute of exposure, the rate partly increased in the next minutes followed by a new slowly progressing decrease. A concentration-dependent recovery of the respiratory rate was seen after cessation of the exposure. In cannulated mice, no sensory irritation pattern was observed. The authors stated that this was due to bypass of the trigeminal nerves. The pattern indicated that pulmonary irritation was present.
Myristyl alcohol has an oral LD50 33 000 mg/kg for rats. 66 Oral LD50 s for ethylhexyl alcohol in rats range from 2049 to 7100 mg/kg body weight and 2380 to >5000 mg/kg for rabbits. 9 Rats, mice, and guinea pigs (n = 10 per species) exposed (whole body) to air bubbled through ethylhexyl alcohol for 6 hours exhibited signs of irritation of the eyes nose, throat, and respiratory passages, including blinking, lacrimation, nasal discharge, salivation, gasping, and chewing movements, but none died.9,69 None of the 6 rats inhaling concentrated ethylhexyl alcohol for up to 8 hours died. 71 The oral LD50 of hexyldecyl alcohol for Sprague-Dawley rats was reported to be >8.42 g/kg. 69 The dermal LD50 of hexadecyl alcohol for albino rabbits was reported to be >2.6 g/kg. 69 There were signs of CNS depression. Recovery occurred within 4 to 48 hours. Swiss mice, Wistar rats, and English short hair guinea pigs (n = 10) were exposed to aerosolized hexyldecyl alcohol (1060 ppm; 9.6 mg/m3) for 6 hours and observed for 14 hours. 69 The alcohol was a slight irritant and no systemic effects were observed.
Repeated-Dose Toxicity
Benzoic acid and sodium benzoate
In multiple-dose oral/feed toxicity studies on rats and mice, decreased feed consumption, depressed growth, and toxic effects were observed at concentrations >1% benzoic acid or sodium benzoate (Table 4). 1
Repeated Dose Oral/Feed Toxicity Studies on Benzoic Acid and Sodium Benzoate.a

a Reviewing the studies, the GRAS report (Informatics Inc., 1972) concluded, ". at a level of approximately 1% [in food], the benzoates are at maximum non-toxic level; higher than this, they result in decreased food intake, depressed growth, and toxic effects on test animals."
Crossbred white mice (n = 100) were orally administered benzoic acid at 80 mg/kg/d for 3 months. 72 The treated group had decreased weight gain and increased mortality (68% vs 60% in the control group).
Sodium benzoate (0, 1%, or 2%; 735 or 880 mg/kg/d) was incorporated into the feed of Fischer 344 rats (n = 102; 50 males, 52 females) for 18 to 24 months. 73 There were no differences in mortality between groups. Necropsies were unremarkable.
Alcohols
Amyl alcohol (0, 50, 150, 100 mg/kg/d) was administered orally to ASH/CSE rats (n = 30; 15/sex) for 13 weeks. 74 The rats were killed and necropsied 24 hours after the last treatment. No adverse effects were observed at any dose.
The no observed adverse effect level (NOAEL) and lowest observed adverse effect level (LOAEL) for the inhalation of isopropyl alcohol was 1230 mg/m3 and 3690 mg/m3, respectively, in Fischer 344 rats and CD-1 mice (n = 10/sex) exposed (0, 1230, 3690, or 12 300 mg/m3) 6 h/d, 5 d/week for 13 weeks. 75 Adverse effects observed at the LOAEL included narcosis in rats and mice, hematological changes in rats, and increased liver weights in mice.
An NOAEL of 1230 mg/m3 for rats and mice was reported (based on kidney and testicular effects) in a study in which Fischer 344 rats (n = 65/sex) and CD-1 mice (n = 55/sex) were exposed by inhalation to isopropyl alcohol (0, 1230, 6150, or 12 300 mg/m3) for 6 h/d, 5 d/week for 104 weeks in rats and 78 weeks in mice. 76
All of the rats (n = 10) exposed to isobutyl alcohol (0, 770, 3100, or 7700 mg/m3) by inhalation for 6 h/d, 5 d/week for 14 weeks exhibited a slight reduction in responsiveness to external stimuli, which was reversed by terminating the exposures. 77
Male Wistar rats (n = 10) were exposed to aerosolized n-pentadecyl alcohol (0, 100, 300 or 600 ppm) 6 h/day, 5 d/week for 7 or 14 weeks. 78 The rats were then killed and necropsied. There were no mortalities and there was no effect on body weights. No valeraldehyde, the primary metabolite of n-pentanol, was found in the blood, while the n-pentadecyl alcohol concentration in blood was linearly correlated with the dose. The brain n-pentadecyl alcohol was related to the blood alcohol at 7 weeks. At 14 weeks, this relationship changed because the brain n-pentadecyl alcohol concentration decreased. Valeraldehyde was measured in the brain only at the high-dose level.
The liver n-pentadecyl alcohol dehydrogenase activity did not change at all. The microsomal cytochrome P-450 contents and 7-ethoxycoumarin-O-deethylase activities in the liver remained unaffected, while the kidney deethylase activity was enhanced in a dose-dependent manner at 7 weeks. This effect lessened after 14 weeks. The kidney n-pentadecyl alcohol dehydrogenase activity was slightly deceased at the mid and high doses after 7 weeks. The brain acetylcholinesterase activity was greater than the control range at all doses at 7 weeks. Similar effects were noted in the muscles at the mid and high doses. The authors suggested that moderate pentadecyl alcohol vapor exposure may cause metabolic and functional adaptation in its target organs.
Rats (n = 10) dermally treated daily for 17 days with ethylhexyl alcohol (100%; ∼1600 mg/kg/d) administered to shaved backs exhibited decreased thymus weights and spermatogenesis, liver granulomas, bronchiectasis, renal tubular epithelial necrosis, edema in heart and testes, and increased lipid levels in the adrenal glands. 79
Fischer 344 rats (n = 20; 10/sex) were topically administered ethylhexyl alcohol (500 or 1000 mg /kg/d) for 5 days under occlusion, followed by 2 days untreated, and then 4 days treatment with S9.80,81 Both doses produced exfoliation (minimal severity), and the high dose caused transient erythema of the treated skin. The female rats exhibited elevated serum triglycerides at both doses and decreased peripheral blood lymphocytes and spleen weights at the high dose.
NOAELs ranged from 100 to 150 mg/kg body weight/day in several studies in which mice or rats were exposed orally for 9 to 11 days to ethylhexyl alcohol by gavage, in drinking water, or in feed.80–84 Doses ≥330 mg/kg body weight/day produced CNS depression, lacrimation, and decreased food consumption and body weights.
The NOAEL was 125 mg/kg/d for male and female F344 rats and B6C3Fl mice treated daily with ethylhexyl alcohol (0, 25, 125, 250, or 500 mg/kg/d) by gavage for 13 weeks. 85
Multiple in vitro and in vivo short-term repeated dose studies showed that ethylhexyl alcohol is a peroxisome proliferator and liver enzyme inducer in mice and rats, and that doses ≥60 mg/kg body weight/day can cause these effects and alpha-2u-nephropathy in male rats. 86
No local or systemic effects were found in male and female Wistar rats (n = 20; 10/sex) exposed 6 h/d, 5 days/week, for 90 days to aerosolized 2-ethyl-l-hexanol (15, 40, or 120 mL/m3; purity 99.9%). 87
The oral NOAEL for noncancer systemic toxicity end points were 200 and 50 mg/kg/d in mice and rats, respectively, exposed chronically to ethylhexyl alcohol. 88
Ocular/Mucosal Irritation
Methyl benzoate
Methyl benzoate (100%) was administered to the center of the cornea while the lids were retracted in rabbits (n = 5). 53 The eye lids were released after ∼1 minute. The eyes were scored after 18 to 24 hours in daylight and with staining. Methyl benzoate was given a grade 1 (iritis, slight internal congestion).
Ethyl benzoate
The above experiment was repeated with ethyl benzoate (100%). 53 Ethyl benzoate was given a grade 1.
Butyl benzoate
The above experiment was repeated butyl benzoate (100%). 53 Butyl benzoate was given a grade 1 (iritis, slight internal congestion).
C12-15 Alkyl benzoate
In an ocular irritation test of C12-15 alkyl benzoate (1.8%-2.4%; 0.1 mL) using rabbits, the test material was administered to the eye and washed after 24 hours. 58 The eyes were observed for 7 days. There was no ocular irritation in rabbits under these test conditions.
C12-15 alkyl benzoate (100%) administered to the eyes of albino New Zealand rabbits caused diffuse crimson coloration, slight swelling, and some discharge. 89 The reactions were resolved in <6 days.
In an EpiOcular tissue model toxicity testing system, human-derived epidermal keratinocytes were incubated in culture medium to which C12-15 alkyl benzoate (2% or 20%; 10 µL), corn oil (negative control), and (0.3%) Triton X-100 (positive control) were added. 90 Using the instructions of the test kit, it was extrapolated that C12-15 alkyl benzoate was nonirritating at 2% and 20%.
Isopropyl benzoate
Isopropyl benzoate (100%) was administered to the center of the cornea while the lids were retracted in rabbits (n = 5). 62 The eye lids were released after ∼1 minute. The eyes were scored after 18 to 24 hours in daylight and with staining. Isopropyl benzoate was given a grade 1.
Isostearyl benzoate
In an ocular irritation assessment of a body lotion containing isostearyl benzoate (0.95%) using neutral red release (NRR) assay, the hen’s egg test on the chorioallantoic membrane (HET-CAM) assay, and the reconstituted human epithelial culture (REC) assay, the authors rated the lotion as slightly irritating. 91
Alcohols
In an NRR assay using human keratinocytes and fibroblasts from natal foreskins to assess ocular irritancy, methyl alcohol was rated a mild irritant. 92
Amyl alcohol (100%, 0.1 mL) was rated as a severe ocular irritant in the Draize test and ocular cell count assay. 93 Severe swelling of conjunctival tissue interfered with accurate assessment of Draize scores and cell washes at l hour after instillation. Amyl alcohol (100%) was rated a severe ocular irritant when applied to rabbits. 69
Dodecyl alcohol was reported to have a maximum average score (MAS) or 24.2 in an ocular Draize test. 94
Isopropyl alcohol has been described to be a severe ocular irritant based on tests in rabbits. 69
Several in vitro tests to investigate the eye irritation potential showed an irritating effect of ethylhexyl alcohol.95–101
Undiluted ethylhexyl alcohol was moderately to severely irritating to the eyes in rabbits.53,68,71,79,102–104 Effects ranged from conjunctival redness and swelling, lacrimation, and discharge, which did not clear within 96 hours after treatment, to persistent corneal dullness and vascularization. Hexyldecyl alcohol (100%) was rated a slight ocular irritant when applied to rabbits. 69
Dermal Irritation
Methyl benzoate
Methyl benzoate (100%) was applied to both the clipped dorsum (0.5 mL) and external surface of the outer ear (0.2 mL) of male New Zealand albino rabbits (n = 14) daily for 6 days. 105 On the dorsum, there were marked cellular reactions and dermal edema beginning on day 2 followed by dermal hemorrhages, desquamated crust, and thickening of the malpighian stratum beginning on day 5. On the inner ear, there was slight hyperkeratosis at day 6.
Methyl benzoate (100%; 0.01 mL) was applied to the clipped skin of albino rabbits (n = 5) and observed within 24 hours. 53 Irritation was rated as grade 3 in a 1 to 10 system (grade 1 = least visible capillary injection from the undiluted material; 6 = necrosis with the undiluted material; and 10 = necrosis from a 0.01% solution).
Methyl benzoate (100%) was administered to the clipped and depilated skin of guinea pigs (n = 3 – 4) using filter paper soaked in the test substance for up to 2 minutes. 106 Before and after treatment, the guinea pigs were administered Evans Blue dye iv The permeability was measured by exuded dye at the treated sites. There was a minimal response observed.
Ethyl benzoate
Ethyl benzoate was administered to the clipped dorsum (0.5 mL) and external surface of the outer ear (0.2 mL) to male New Zealand albino rabbits (n = 14) daily for 6 days. 105 On the dorsum, there were marked cellular changes, edema, desquamated crusts, and thickening of the malpighian stratum beginning on day 1. On the inner ear, there were slight cellular reaction, no edema or hemorrhages, no necrosis, slight to marked desquamated crusts, marked thickening of malpighian stratum, hyperkeratosis, and slight hyperplasia of sebaceous glands beginning day 1.
Ethyl benzoate (100%; 0.01 mL in water, propylene glycol, or kerosene) was administered to the clipped skin of rabbits (n = 5) and scored after 24 hours. 53 Irritation (grade 4 out of 10) was observed.
Propyl benzoate
Propyl benzoate was applied to the clipped dorsum (0.5 mL) and external surface of the outer ear (0.2 mL) to male New Zealand albino rabbits (n = 14) daily for 6 days. 105 On the dorsum, there were marked cellular reactions, necrosis, thickening of the malpighian stratum beginning on day 1 followed by dermal hemorrhages, desquamated crusts beginning on days 3 or 4. On the inner ear, there were slight cellular reactions, necrosis, and moderate thickening of the malpighian stratum beginning on day 1 or 3.
Butyl benzoate
Butyl benzoate was applied to the clipped dorsum (0.5 mL) and external surface of the outer ear (0.2 mL) to male New Zealand albino rabbits (n = 14) daily for 6 days. 105 On the dorsum, there were marked cellular reaction, necrosis, and slight detachment of the dermoepidermis beginning on day 1 followed by desquamated crusts and thickening of the malpighian stratum beginning on day 3. On the inner ear, there were slight cellular reactions, necrosis beginning on day 1, and moderate desquamated crusts and hyperplasia of the sebaceous glands on day 2 or 3.
Butyl benzoate (100%; 0.5 mL) was applied to the clipped skin of female New Zealand white rabbits (n = 4) under occlusion for 4 hours. 57 Observations were made at 1, 24, 48, and 72 hours. There were no effects at 1 hour, well-defined erythema and slight edema at 24 h, and very slight erythema and edema at 72 hours.
In a Draize test, butyl benzoate (5 g/kg) had slight to no irritation effects after 24 hours. 57 The treated skin was scaly at necropsy.
C12-15 Alkyl benzoate
In a primary dermal irritation test of C12-15 alkyl benzoate (100%; 0.5 mL), the test material was applied to the intact and abraded clipped skin of albino New Zealand rabbits (n = 6). 58 The primary irritation index was 0.08. C12-15 alkyl benzoate was not a primary irritant to rabbits.
In a repeat 14-day irritation study, C12-15 alkyl benzoate (62% and 100% in corn oil; 0.5 mL) was administered to the clipped dorsal skin of New Zealand white rabbits (n = 10; 5/sex). 107 Mineral oil and isopropyl myristate were used as controls. The average combined erythema and edema score/animal/day was 4.64 and 4.11 for the high and low dose of C12-15 alkyl benzoate, respectively. The scores were 2.68 and 5.40 for mineral oil and isopropyl myristate, respectively.
C12-15 alkyl benzoate (100%) administered to the skin of male albino New Zealand rabbits (n = 3) caused slight erythema and edema at 1 hour, which resolved at 24 hours. 60
Human derived epidermal keratinocytes (NHEK) were incubated with C12-15 alkyl benzoates (10% and 100% in corn oil; 100 µL) and Triton X-100 (1%; positive control) for 3 hours in an EpiDerm in vitro toxicity testing system. 108 The test material was found to be nonirritating at both concentrations.
Isopropyl benzoate
Isopropyl benzoate (100%) administered to the skin of rabbits (n = 5) in a Draize test had an irritation score of 3 (strong capillary injection). 53
Isobutyl benzoate
In an acute toxicity test (see above), isobutyl benzoate (100%) administered under occlusion for 24 hours to the intact and abraded clipped backs of rabbits (n = 4) produced no effects at 5 mL/kg. 64
Isobutyl benzoate (100%) administered to the skin of rabbits (n = 5) in a Draize test had an irritation score of 5. 53
Alcohols
Methyl alcohol (10 and 35 mg in water; 35 mg in paraffin, and 10 mg in oil) was injected intracutaneously into the dorsal skin of shaved rabbits (n = 4). The sizes of the wheals at 24 hours were 9, 0, 3, and 1 mm2, respectively. 109
Amyl alcohol was rated a severe irritant at 3.2 g/kg when applied to the abraded abdominal skin of albino rabbits. 69
Amyl alcohol (10 and 35 mg in water; 35 mg in paraffin and 10 mg in oil) was injected intracutaneously into the dorsal skin of shaved rabbits (n = 4). The sizes of the wheals at 24 hours were 74, 19, 53, and 40 mm2, respectively. 109
When lauryl alcohol was applied to the skin of CD(SD) hrBI hairless rats, moderate erythema was observed. 110
Lauryl alcohol was reported to have a primary irritative index (PII) of 0.96 in a dermal Draize test. 94
A single dermal administration of ethylhexyl alcohol (5000 mg/kg) caused slight or moderate skin irritation in studies using clipped rabbits (n = 10). 111
Slight redness and scabbing was reported in rabbits (n = 10) after 10 daily dermal administrations of ethylhexyl alcohol (100%; 2 mL/kg/d). 79
Occlusive exposures to ethylhexyl alcohol (100%; 3.16 mg/kg/d) administered to the clipped intact skin of rabbits (n = 4) for 7 days caused moderate dermal irritation, erythema, edema, desquamation, necrosis, and eschar formation. 69
Hexydecyl alcohol was rated a slight irritant at 2.6 g/kg when applied to the abraded abdominal skin of albino rabbits. 69 One rabbit showed transient CNS depression and labored respiration.
Dermal Sensitization
Methyl benzoate
In a modified Freund complete adjuvant test using guinea pigs (n not provided), methyl benzoate (10%; 30 mg) was not sensitizing. 112
A guinea pig open epicutaneous test (OET; n = 6-8) of methyl benzoate (up to 4%; vehicle not provided; 0.1 mL) was performed. 113 The test material was applied daily for 3 weeks onto the shaved skin. Controls (n = 10) were untreated or treated with the vehicle. Challenge was conducted on days 21 and 35 on the opposite flank. Observations were made at 24, 48, and 72 hours. Methyl benzoate at 4% was not sensitizing.
Ethyl benzoate
A guinea pig OET (n = 6-8) of ethyl benzoate (up to 8%; 0.1 mL; vehicle not provided) was performed. 114 Controls (n = 10) were untreated or treated with the vehicle. The test material was applied daily for 3 weeks onto the clipped skin (8 cm2). Challenge was conducted on days 21 and 35 on the opposite flank to the test and control animals. Observations were made at 24, 48, and 72 hours. Ethyl benzoate at 8% was not sensitizing.
Amyl benzoate
A guinea pig open OET (n = 6-8) of amyl benzoate (up to 6%; vehicle not provided; 0.1 mL) was performed. 114 The test material was applied daily for 3 weeks onto the clipped skin. Challenge was conducted on days 21 and 35 on the opposite flank. Amyl benzoate at 6% was not sensitizing.
C12-15 Alkyl benzoate
In a guinea pig sensitization test, C12-15 alkyl benzoate (10%; 0.5 mL) was administered to the clipped backs and flanks of white male guinea pigs (n = 12) for 6 h/d under occlusion, 3 times/week for 3 weeks. 61 Two challenges were performed 14 days after the last application. There were no topical or systemic reactions observed.
Isobutyl benzoate
A guinea pig OET (n = 6-8) of isobutyl benzoate (up to 2%; vehicle not provided; 0.1 mL) was conducted. 114 Water or the vehicle were administered to the controls (n = 10). The test material was applied daily for 3 weeks onto shaved skin (8 cm2). Challenge was conducted on days 21 and 35 and read at 24, 48, and 72 hours; the opposite flank was treated with the minimal irritation concentration and lower concentrations (not provided). Isobutyl benzoate at 2% was not sensitizing.
Comedogenicity
Alcohols
Alcohols were tested for comedogenicity by repeated application to the inner ear of rabbits (5 days/week for 2 weeks). 115 Cetyl alcohol (100%), stearyl alcohol (100%), benzyl alcohol (100%), and propyl alcohol (100%) had no comedogenic activity. Lauryl alcohol (50% in mineral oil) and myristyl alcohol (50% in mineral oil) had slight comedogenicity. Octyl alcohol (100%) had strong comedogenicity.
Reproductive and Developmental Toxicity
No data were available for reproductive and developmental toxicity of the alkyl benzoates used in cosmetics. One study on the related compound isononyl benzoate is described below, as are the available data on benzoic acid, sodium benzoate, and alcohol metabolites of alkyl benzoates.
Isononyl Benzoate
Isononyl benzoate (100, 300, 1000 mg/kg/d) was orally administered to male and female Sprague-Dawley rats before mating through 3 days postpartum. 116 The males began treatment 4 weeks before mating and the females 2 weeks before mating. Body weights, body weight gains, and feed consumption were not affected by the test substance. There were no differences between treatment groups and controls with regard to fertility index, precoital interval, and copulatory index compared to controls. There was a slight decrease in mean pup weights and consequently in litter weights in high-dose females at birth and on day 4 postpartum. Histopathological examination of the ovaries and testes (including the stage in the spermatogenic cycle) did not reveal any differences between the treated and control animals. The authors concluded that 1000 mg/kg/d was the NOAEL.
Benzoic Acid and Sodium Benzoate
Sodium benzoate (0, 1%, 2%, 4%, or 8%; 0, 667, 1333, 1600, or 710 [sic] mg/kg/d) was administered in the feed of female Wistar rats (n = 27-30) during gestation (number of days not provided). 117 On day 20, 20 to 25 rats of each group were killed and necropsied. The rest were allowed to live through pregnancy and nurse for 3 or 8 weeks and then were killed. Half of the pups were then killed at each of these times and necropsied. The 2 highest dose groups had an increase in the number of dead fetuses and resorbed embryos. The body weights of the viable pups were decreased and mild systemic edema was observed. The number of fetal abnormalities increased in a dose-dependent manner. The number of pups born decreased, the number of perinatal deaths increased to 100%, lactation rate decreased, and survival rate decreased to 0 in the 2 highest dose groups. The effects on the fetus occurred only at maternally toxic concentrations of ≥4% sodium benzoate.
Sodium benzoate (up to 5 mg/egg) was injected twice into the air sac of fertilized chicken eggs at 0 and 96 hours and incubated to hatching. 118 Surviving chicks were killed and necropsied. There were no teratogenic effects reported. The LD50 was 4.74 mg/egg.
Female Wistar rats (n = 20) were orally administered sodium benzoate (0, 1.75, 8.0, or 175 mg/kg/d) during days 6 to 15 of gestation. 119 On day 20 of gestation, the pups were delivered by Caesarean section. There were no differences in the types or incidences of abnormalities observed in any of the treatment groups compared to the control. The fetal and maternal NOAEL was 175 mg/kg.
The study above was repeated using mice (n = 20), hamsters (n = 21-22; gestation days 6-10), and rabbits (n = 10). Similar results were reported.
In oral teratogenicity studies, benzoic acid administered on gestation days 6 to 10 increased the number of resorptions at ≥30 mg/kg/d and increased the number of fetal malformations at >600 mg/kg/d in hamsters. Results for benzoic acid were negative in 2 oral rat studies up to 500 mg/kg/d. 120
Crossbred white mice (n = 50; 25/sex) were orally administered benzoic acid (40 mg/kg/d) for 8 months before breeding. 72 This was continued for 5 generations. The parental and F1 cohorts had increased mortality compared to controls after a 5-day 100% food restriction test. Otherwise, there were no effects on reproduction.
A neurobiological study on the effects of sodium benzoate (0.1%, 0.5%, and 1.0%) on the offspring of rats and mice was negative. 121 The dams (n = 8) were administered feed incorporated with sodium benzoate (0, 0.1%, 0.5%, or 1.0%) from gestation day 5 until weaning. Locomotor activity and brain chemistry of the pups were not affected.
Alcohols
Sprague-Dawley rats were exposed to aerosolized methyl alcohol (5000, 10 000, or 20 000 ppm) for 6 h/d on days 1 to 19 of gestation. 122 There were no maternal effects observed at any concentration. The offspring had reduced weights in the mid and high-dose groups. The high-dose group also had increased incidences of external malformations, skeletal malformations, and visceral malformations compared to controls.
Mating success was not affected in male (n = 18) and female (n = 15) Sprague-Dawley rats exposed to propyl alcohol (8.61 mg/L) via inhalation 7 h/d and 7d/week for 62 days. 123 The decreased mating success of the male rats exposed to a higher dose of propyl alcohol (17.2 mg/L) in this study was reversed 15 weeks after exposure. Changes in activity measures were observed in the offspring of the 8.61 mg/L propyl alcohol maternally exposed group, and crooked tails were found in 2 to 3 offspring.
The NOAELs for the maternal and developmental toxicity of isopropyl alcohol were 400 mg/kg in rats (maternal and developmental) and 240 mg/kg (maternal) and 480 mg/kg (developmental) in rabbits exposed by gavage during gestation (rats: 0, 400, 800, or 1200 mg/kg/day on gestation days 6 through 15; rabbits: 0, 120, 240, or 480 mg/kg/day on gestation day 6 through 18). 124
No effects on rat reproductive cells or organs (liver, lungs, spleen, lymph nodes, kidneys) were observed in several in vitro studies of ethylhexyl alcohol (200 µmol/L) for up to 48 hours or in oral in vivo studies using rats (up to 500 mg/kg/d) for up to 90 days.85,87,124–130
In Wistar rats administered a single dose of 1666 mg/kg ethylhexyl alcohol by gavage on day 12 of pregnancy, 22.2% of the surviving fetuses had malformations (compared to 2% and 0% for 833 mg/kg and controls, respectively), and average fetal weight was decreased. 125 No maternal toxicity or effects on implantation index or numbers of dead and resorbed fetuses were found in this study.
The embryos of pregnant Sprague-Dawley rats (n = 6) administered a single dose of ethylhexyl alcohol (1625 mg/kg) by gavage exhibited decreased 64 Zn content, although the percentage of resorptions was not affected.131,132
No treatment-related increases in the incidences of malformations or variations were found in F344 rats (n = 25) dermally exposed (clipped dorsal skin) to 2-ethyl-1-1hexanol (0, 252, 420, 840, 1680 or 2520 mg/kg/d, 6 h/d, occlusive) on gestation days 6 to 15.41,124,133 Maternal effects at ≥840 mg/kg/d included persistent exfoliation, crusting, and erythema at the site of application, and doses ≥1680 mg/kg/d were associated with decreased maternal weight gain.
Maternal and developmental NOAEL was found to be 130 mg/kg/d in Wistar rats (n = 10) orally exposed to ethylhexyl alcohol (0, 130, 650, and 1300 mg/kg/d) on days 6 to 19 of gestation. 134
The pups of Charles River CD-l mice (n = 50) exposed to 2-ethyl-1-1hexanol (1,525 mg/kg/d in corn oil) on days 7 to 14 of pregnancy exhibited reduced viability and body weights on day 3 of lactation. 135 Maternal effects included decreased fertility and pregnancy indexes, body weights, and other signs of toxicity.
No maternal, reproductive, or developmental toxicities were found in CD-1 Swiss mice (n = 28) ingesting microencapsulated ethylhexyl alcohol (0.13, 43, and 129 mg/kg/d; >99% pure) in the diet on days 0 to 17 of pregnancy.136,137
The NOAEL for maternal and developmental toxicity was 850 mg/m3 (160 mL/m3) aerosolized ethylhexyl alcohol in Sprague-Dawley rats (n = 15) exposed (whole body) 7 h/d on gestation days 0 to 19. 123
Genotoxicity
Methyl Benzoate
In an Ames test using Salmonella typhimurium (TA97, TA98, TA100, TA1535, and TA1537), methyl benzoate (6666 µg/plate) was not mutagenic with or without metabolic activation. 138 Methyl benzoate (dose not reported) was not found to be mutagenic in Escherichia coli (Sd-4-73). 139
Ethyl Benzoate
In an Ames test using S typhimurium (TA98, TA100, TA102, TA1535, and TA1537), ethyl benzoate (15-5000 µg/plate without metabolic activation and 5 to 5000 µg/plate with metabolic activation) was not mutagenic with or without metabolic activation. 55
C12-15 Alkyl Benzoate
In an Ames test using S typhimurium (TA98, TA100, TA1535, TA1537, and TA1538), C12-15 alkyl benzoate (100%; 0.1 mL) was not mutagenic to any strain tested, with or without metabolic activation. 140 Saline was the negative control and Dexon and 2-aminofluorene were the positive control.
Isononyl Benzoate—Related Compound
In an oral in vivo micronucleus test of benzoic acid isonylester (500, 1000, 2000 mg/kg) using Sprague-Dawley SD rats (n = 5/sex), no remarkable adverse reaction was observed after treatment. 141 A slight depression of bone marrow erythropoietic cell division was observed at the high- and intermediate-dose levels of treatment for female animals from the 24-hour sampling time. A slight depression of bone marrow erythropoietic cell division was also observed at the 48-hour sampling time for both male and female animals from the high-dose group. The authors concluded that isononyl benzoate administered orally at these dose levels to male and female rats does not induce micronuclei in the polychromatic erythrocytes.
Benzoic Acid and Sodium Benzoate
Benzoic acid and sodium benzoate had both positive and negative results in genotoxicity assays (Table 5). Representative studies are presented below.
Genotoxicity Tests of Sodium Benzoate and Benzoic Acid.
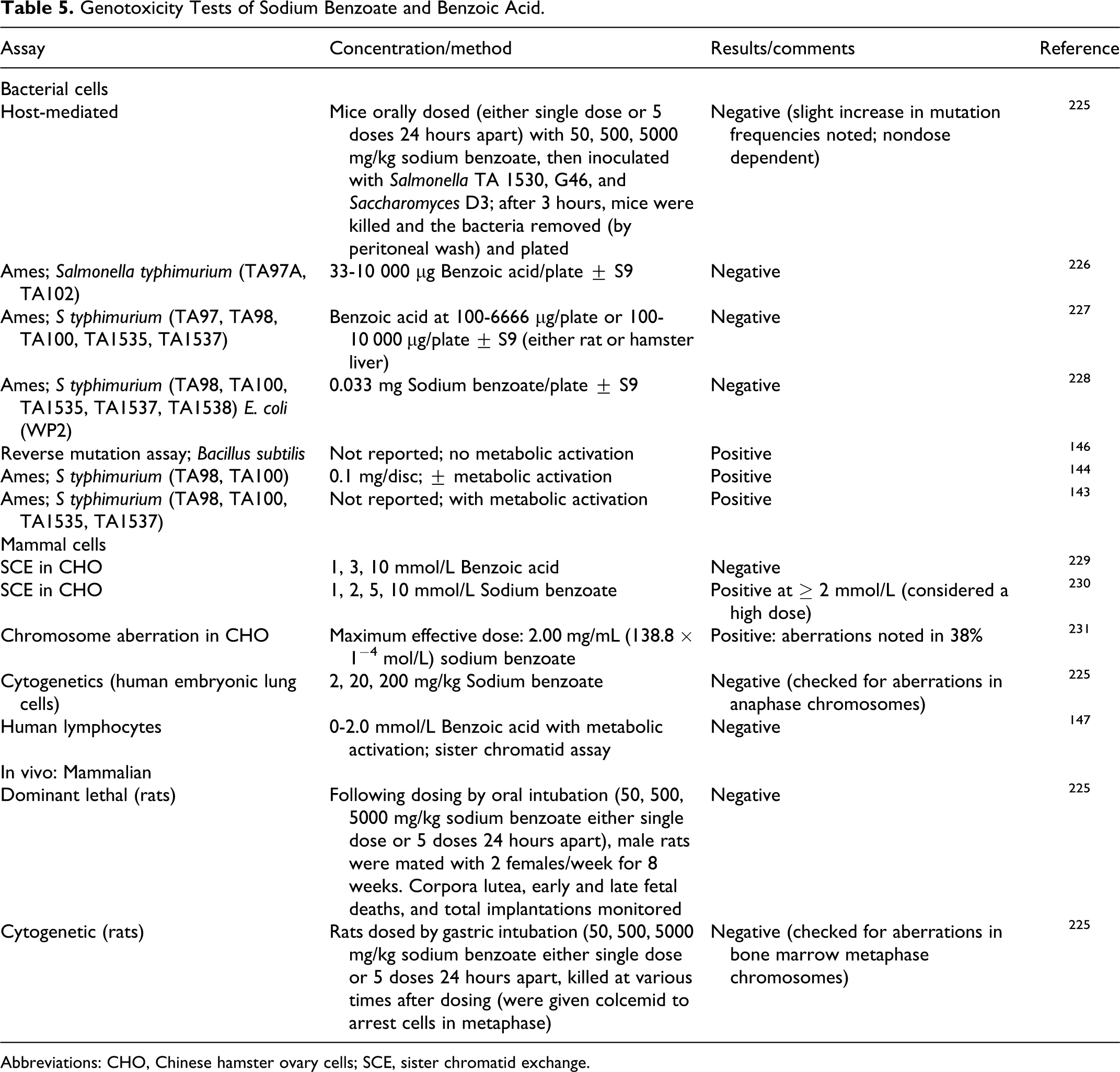
Abbreviations: CHO, Chinese hamster ovary cells; SCE, sister chromatid exchange.
Benzoic acid was negative in several Ames tests using S typhimurium (including TA98, TA100, TA1535, TA1537, and TA 1538) with and without metabolic activation.1,142–145
In one Ames test using S typhimurium (TA98 and TA100), benzoic acid (0.1 mg/plate) and sodium benzoate (0.1 mg/plate) were genotoxic with activation. 144 In a reverse mutation test, benzoic acid (5 mg/disc) was positive for genotoxicity.
In a sister chromatid exchange assay using human lymphocytes, benzoic acid (0 to 2.0 mmol/L) was not genotoxic with metabolic activation. 145
Sodium benzoate was positive without metabolic activation and negative with metabolic activation in a reverse mutation assay using Bacillus subtilis. 142 Benzoic acid (1.5 mg/mL) was positive in a chromosomal aberration test without metabolic activation. In a sister chromatid exchange assay using hamster lung fibroblasts, sodium benzoate was not clastogenic without metabolic activation. 146
Alcohols
In an Ames test, methyl alcohol (5-5000 µg/plate) was not mutagenic to Salmonella typhimuium (strains TA98, TA100, TA1535, TA1537, and TA1538) and Escherichia coli (WP2uvrA). 147 Genotoxicity tests of ethylhexyl alcohol were generally negative, including in vitro tests for chromosome aberrations,123,148–150 unscheduled DNA synthesis, 151 mutagenicity (Ames, TK+/− mouse lymphoma, and HPRT assays),152–154 and cell transformation.40,155,156 The exceptions include one positive result in one of 2 rec-assays157,158 and another in a test of mutagenicity in S typhimurium TA-l00 (mutation resistance to 8-azaguanine), 159 both of which were questionable. 9
Urine samples from Sprague-Dawley rats exposed to ethylhexyl alcohol (1000 mg /kg/d) by gavage for 15 days tested negative for mutagenicity in S typhimurium with and without rat liver microsomes or beta-glucuronidase/arylsulfatase.160,161
In vivo tests of ethylhexyl alcohol genotoxicity were also negative, including assays for covalent binding to liver DNA,162,163 dominant lethal mutations, 164 and bone marrow micronuclei.165,166
Carcinogenicity
Benzoic Acid
Crossbred white mice (n = 100) were orally administered benzoic acid (40 mg/kg/d in a paste). 72 After 8 months, the mice were bred and also administered the benzoic acid paste. This was repeated for 5 generations. Eight of 100 mice in the first generation and 1 of 100 in the third generation were found to have malignant tumors. No tumors were found in the control group. In a follow-up tumor transplantation test, benzoic acid fed to mice for 3 months did not increase tumor growth.
A nonoxidative hair dye containing benzoic acid (0.016%) and benzyl alcohol (2.0%) was negative for carcinogenicity when dermally applied to mice (n = 60) 3 times per week for 20 months. 73 In a feeding study of rats and mice (n = 102), feed containing sodium benzoate (1% or 2%; 102-151 or 202-280 mg/d) was not carcinogenic after 6 weeks. 167
Alcohols
Orally administered methyl alcohol, amyl alcohol, and lauryl alcohol at one-fifth of the lethal dose caused increases in polyploidy cells, cells with gaps, and cells with aberrations in the bone marrow of rats. 168
Male rats exhibited a concentration-dependent increase in the incidence of interstitial (Leydig) cell adenomas of the testes at all doses in a study in which Fischer 344 rats (n = 65/sex) and CD-1 mice (55/sex) were exposed by inhalation to isopropyl alcohol (0, 1230, 6150, or 12 300 mg/m3) for 6 h/d, 5 d/week for 104 weeks in rats and 78 weeks in mice. 169 No other tumors or neoplastic lesions were found in the rats or mice. International Agency for Research on Cancer (IARC) 170 has determined that isopropyl alcohol is not classifiable as to its carcinogenicity to humans (group 3).
Male and female rats and mice were exposed chronically to ethylhexyl alcohol by gavage 5 times a week (rats: 0, 50, 150, 500 mg/kg/d for 24 months; mice: 0, 50, 200, 750 mg/kg/d for 18 months). 88 The results of this study suggested that ethylhexyl alcohol was a weak liver tumor promoter in female mice. Mechanistic studies suggest that tumor promotion in mice is attributable to the induction of peroxisome proliferation by ethylhexyl alcohol, which has questionable relevance for human exposures. Belsito et al 9 concluded that “while this mechanism cannot be completely discounted, it is reasonable to assume that humans are less sensitive than rodents.”
Clinical Assessment of Safety
Toxicity
Benzoic acid
In clinical studies, toxic symptoms (including discomfort, malaise, nausea, headache, weakness, esophageal burning, irritation, hunger, indigestion, vomiting, itching, perspiration) were observed following oral doses far exceeding the acceptable daily intake (ADI; 0-5 mg/kg) 1 established by JECFA. 171 The Registry of Toxic Effects of Chemical Substances (RTECS) cited the human low lethal oral dose of benzoic acid to be 500 mg/kg. 172
Ocular/Mucosal Irritation
Ethylhexyl benzoate
A sunscreen liquid containing ethylhexyl benzoate (3.5%) was randomly administered to the eyes of participants (n = 30; 10 male, 20 female) with an eye swab. 173 The reactions were scored at 5 minutes then the eyes were washed. Scoring was repeated at 15 and 60 minutes. The control was a baby shampoo (10%) with no ethylhexyl benzoate. The test material and the control exhibited no differences at all scoring times, and all reactions were cleared at 1 hour. The test material caused no tearing.
Alcohols
Self-reported nasal and eye irritation and perceived odor intensity were increased in a concentration-related manner in male volunteers exposed to ethylhexyl alcohol (≥10 mL/m3) for 4 hours in an exposure chamber. 174
Dermal Irritation
Methyl benzoate
Occluded patch tests were conducted on methyl benzoate (0.05%-0.5% in a perfumed base cream, a nonperfumed base cream, or 99% ethanol) in multiple studies (total n = 4737; 2341 Japanese men and 2396 Japanese women). 175 There were no visible reactions to the test substance observed.
Ethyl benzoate
Occluded patch tests were conducted on ethyl benzoate (0.05%-0.5% in a perfumed base cream, a nonperfumed base cream, or 99% ethanol) in multiple studies (total n = 4737; 2341 Japanese men and 2396 Japanese women). 55 There were no visible reactions to the test substance observed. In a 48-hour closed patch test (n = 5 males), ethyl benzoate (8% in petrolatum) produced no effects. 176 In a human maximization test (n = 25), ethyl benzoate (8% in petrolatum) was not sensitizing. 175
Butyl benzoate
In a 48-hour closed patch test (n = 5 males), butyl benzoate (8% in petrolatum) produced no effects. 176
C12-15 Alkyl benzoate
In an irritation study, C12-15 alkyl benzoate (0, 3%, 10%, 30%, and 100% in vegetable oil) was applied to the backs of participants (n = 21) under occlusion for 48 hours. 177 No signs of irritation were observed at 48 and 72 hours.
Isobutyl benzoate
Isobutyl benzoate (2%) administered in a 24 h patch test (n = 5 males) produced no effects. 64
Ethylhexyl benzoate
In a 14-day cumulative irritation test of a sunscreen liquid containing ethylhexyl benzoate (3.5%), the test material was applied to the skin of participants (n = 28). 178 The positive control was sodium lauryl sulfate (SLS; 0.25%) and the negative control was saline. There was a total dermal irritation score of 5.0 out of 1120. The authors concluded that there was no potential for eliciting cumulative dermal irritation.
A sunscreen lotion spray containing ethylhexyl benzoate (3.5%) was administered to the skin on the arms and legs of participants (n = 35; male and female; 7 months to 8 years old) daily for 4 weeks. 179 There were no increases in erythema, edema, or dryness of the arms and no increase in erythema and edema of the legs. One participant exhibited mild dryness of the legs following the 4-week use period.
Benzoic acid
Benzoic acid (0.2%) was not irritating to participants (n = 12) after 3 occlusive patches were applied over 1 week. 180 Benzoic acid (0.2%) caused mild, transient irritation when applied daily in a liquid foundation product at least twice/day for 45 days. 181
Alcohols
Propyl alcohol produced no dermal irritation or skin sensitization in several clinical studies in which it was used as a vehicle and control.182–187 These studies include a cumulative irritation study (n = 20 males) in which Al-test patches containing propyl alcohol were applied daily for 10 days to the interscapular area of each participant, each application remaining in place for 24 hours. 185
In a patch test lauryl alcohol (C12), participants (n = 20) had scores of ∼0.02, 0, and 0.05 for irritation for 2, 1, and 0.5 mg in petrolatum, respectively. In a nitrocellulose-replica test for skin sensitization, the scores were ∼0.35, 0.2, and 0.1, respectively. 188
One participant of 80 males (21 to 52 years old) exposed to cetyl alcohol (11.5%) in a cream base 5 times daily (every 3 hours) for 10 days developed erythema, folliculitis, and pustules (forearm site).5,185 Mild cumulative irritation (total score 418 for 21 applications) was reported in 12 female participants (18-60 years old) exposed to cetyl alcohol (6.0%) using the same protocol.
No irritation was found in female participants (n = 110) exposed to cetyl alcohol (8.4%), 10 patch application sites per participant, followed 14 days later by a challenge patch.5,185
No reactions were observed in healthy individuals (n = 12; 8 males, 4 females; 18-64 years old) exposed to isopropyl alcohol (Finn chambers, occlusive patches) on the flexor side of the right and left forearm for 24 hours. 189
Isostearyl alcohol (25.0% in petrolatum and 25.0%, 27.0%, and 28.0% in lipstick) did not induce skin irritation in participants (n = 19; 18-65 years old).5,185
No skin irritation was found in 29 healthy male volunteers in an occlusive patch test with 4% ethylhexyl alcohol in petrolatum. 190
Dermal Sensitization
The results of several human insult patch tests (HRIPTs) of products containing various benzoate esters are summarized in Table 6. None were irritating or sensitizing.
HRIPTs of Products Containing Benzoate Esters.a
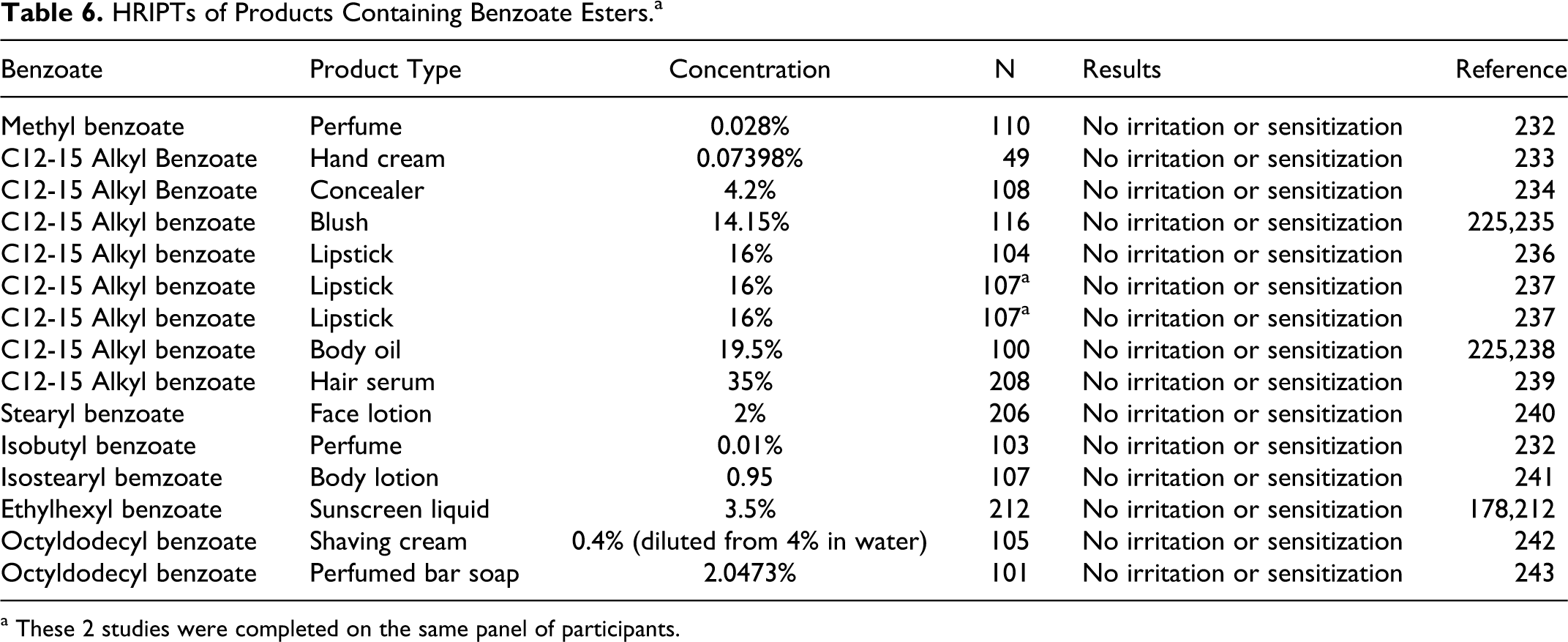
a These 2 studies were completed on the same panel of participants.
Methyl benzoate
A human maximization test (n = 25), methyl benzoate (4% in petrolatum) was not sensitizing. 175
Ethyl benzoate
In a human maximization test (n = 25), ethyl benzoate (8% in petrolatum) was not sensitizing. 175
C12-15 Alkyl Benzoate
In an HRIPT (n = 101) was conducted on C12-15 alkyl benzoate (100%), 173 no visible reactions to the test substance were observed.
An HRIPT (n = 48) was conducted on C12-15 alkyl benzoate (20% in corn oil). 184 Induction consisted of 10 applications under occlusion over 3.5 weeks. The challenge was applied ∼14 days after last application on a naive site. There were no signs of irritation or sensitization.
Isobutyl benzoate
In a human maximization test, isobutyl benzoate (in petrolatum) was applied to the volar surface of male participants (n = 25) on 5 alternate days. 64 The test surfaces were pretreated with 5% aqueous SLS under occlusion for 24 hours. After 10 days, fresh sites were treated with 10% SLS for 1 hour then isobutyl alcohol was applied. Test sites were read at removal and at 24 hours. There was no sensitization at 2% isobutyl benzoate.
Isostearyl benzoate
An HRIPT (n = 107) was conducted on a body lotion product containing isostearyl benzoate (0.95%) under semiocclusion. 193 Except for 1 participant, who also reacted to several other test substances on the shared panel, there were no visible reactions to the product containing isostearyl benzoate at 0.95%.
Octyldodecyl benzoate
An HRIPT (n = 105) was conducted on a shaving cream product containing octyldodecyl benzoate (4%) under semiocclusion. 194 The product was diluted to a 10% aqueous solution. There were no visible reactions to the product containing octyldodecyl benzoate at 0.4%.
Benzoic acid
In 4 clinical studies, tests for the sensitization of benzoic acid were negative. 1 A liquid/powder foundation containing benzoic acid (0.2%) produced no reactions at induction or challenge (n = 75). 195 Benzoic acid (2.0%) in petrolatum produced no reactions at induction or challenge (n = 25).196,197 Benzoic acid (5% in petrolatum) produced no reaction at induction or challenge (n = 10). 198 In a cosmetic intolerance assay, a reaction to benzoic acid (concentration not provided) was observed in 34 of 5202 participants; a reaction was observed in 1 of 155 participants described as having a cosmetic allergy. 199
Alcohols
No primary sensitization was found in female participants (n = 110) exposed to cetyl alcohol (8.4%), 10 patch application sites per participant, followed 14 days later by a challenge patch. 5
An isopropyl alcohol (80.74%) spray concentrate did not exhibit any potential for dermal sensitization in human participants (n = 9). 200
An HRIPT study on test participants (n = 9) showed that a hair dye base formulation of isopropyl alcohol (2.85%) and a isopropyl acetate (1.95%) caused no dermal sensitization in humans. 201
Three of 12 male participants (21-60 years old) exposed to isostearyl alcohol (25% v/v in 95.0% isopropyl alcohol) exhibited erythema during induction.5,185 However, 12 of 148 male and female participants exhibited signs of sensitization after exposure to a pump spray antiperspirant containing isostearyl alcohol (5.0%) using a occlusive patch applied to the upper arm for 24 hours, 3 times/week for 3 weeks. Of the 10 participants, 6 had reactions during the rechallenge 2 months later, and all 4 of the 6 participants rechallenged with isostearyl alcohol (5.0%) in ethanol tested positive 6 weeks after the first rechallenge. In a second study, 5 of 60 male and female participants had positive responses after the first challenge with the same product and test protocol; one of which was later rechallenged with isostearyl alcohol (5.0%) and again tested positive.
No skin sensitization was found in healthy male participants (n = 29) in an occlusive patch test with ethylhexyl alcohol (4% in petrolatum). 202 The saturated alcohol 2-ethyl-1-hexanol has little skin-sensitizing potential. 9
Phototoxicity
Methyl benzoate
Human erythrocytes in suspension (0.4 mL) in methyl benzoate (0.1 mL) were exposed to UVA and UVB for 1 hour. 203 Photohemolysis was not induced.
Ethylhexyl benzoate
A sunscreen liquid containing ethylhexyl benzoate (3.5%; 0.2 mL) was administered to a 2-to 4-cm2 area of the backs of participants (n = 21) that had fair skin. 204 Patches were removed and the test area cleaned 24 hours later. After scoring, UVA was applied to one of the test sites and to a naive site. Sites were graded at 24, 48, and 72 hours. There were no signs of photoxicity observed.
Benzoic acid
Phototoxicity and photosensitivity tests of benzoic acid were negative for a matte eye shadow (0.1%; n = 77) and a liquid/powder foundation (0.2%; n = 10 and 30).205–207
Alcohols
No photosensitization reactions were found in participants (n = 52) exposed to cetyl alcohol (40%) in a lipstick product or to cetyl alcohol (1.0%) in participants (n = 407) tested (product and experimental procedure not stated). 5
Case Reports
Alcohols
Over 19 months, 33 cases of acute allergic contact dermatitis from epilating waxes and/or accompanying tissue were presented. 208 Patch tests of 26 of the patients resulted in 9 positive tests for lauryl alcohol (10% in petrolatum) varying from minor to severe.
Patients (n = 34) with allergic reactions to fatty alcohols had no positive reactions to lauryl alcohol (5% in petrolatum). 209
A 37-year-old man presented with severe genital swelling and inflammation that was not responding to treatment. 210 Prolonged oral prednisolone and antihistamines relieved the symptoms. Patch testing revealed a persistent 3+ reaction to octydodecyl alcohol (13.5% in liquid paraffin) at 48 and 96 hours.
A 62-year-old man had a 5-year history of eczamatous eruption that he treated with a topical corticosteroids, emollients, and an itch reliever. 211 Patch test revealed a + reaction to octyldodecyl alcohol (3% in petrolatum).
Summary
Alkyl benzoates function in cosmetics as skin-conditioning agents, preservatives, solvents, and plasticizers. In general, the alkyl benzoates can be produced industrially via esterification of benzoic acid. The manufacturing processes of the benzoic esters are typically high yielding (
The toxicity of the metabolites (benzoic acid and the parent alcohols) was taken into consideration in this safety assessment.
Alkyl Benzoates
The total number of uses of C12-15 alkyl benzoate was 971 (858 leave-on and 113 rinse-off products) at concentrations up to 59% and 50% in leave-on and rinse-off products, respectively. The highest concentrations of use for C16-17 alkyl benzoates, stearyl benzoate, behenyl benzoate, ethyl benzoate, isobutyl benzoate, isostearyl benzoate, methyl benzoate, and octyldodecyl benzoate were reported to be from 0.01% to 4%. No uses or concentrations of use were reported for propyl benzoate, butyl benzoate, amyl benzoate, lauryl/myristyl benzoate, isopropyl benzoate, ethylhexyl benzoate, butyloctyl benzoate, and hexyldecyl benzoate.
Benzoate esters are metabolized into benzoic acid and the corresponding alcohols. Benzoic acid is further metabolized to benzoyl glucuronide and benzoyl CoA. The benzoyl CoA is metabolized to hippuric acid, the principal metabolite excreted in the urine.
Methyl benzoate, ethyl benzoate, propyl benzoate, and butyl benzoate did penetrate the skin. C12-15 alkyl benzoate did not detectably penetrate through the skin but did penetrate into the skin.
Methyl benzoate was cytotoxic to HeLa cells at 683.30 mmol/L, A flavus at 2.5 mg/mL, A parasiticus at 5.0 mg/mL, and lung fibroblasts at 25 mmol/L. Ethyl benzoate was cytotoxic to Hep-2 cells and lung fibroblasts at 289 mg/L. Propyl benzoate and butyl benzoate were cytotoxic to Hep-2 cells at 122 mg/L and 61 mg/L, respectively.
The oral LD50 of methyl benzoate was 2170 mg/kg for rabbits, 4100 mg/kg for guinea pigs, 1350 to 3500 mg/kg for rats, and 3000 to 3330 mg/kg for mice. The oral LD50 of ethyl benzoate was 2630 mg/kg for rabbits and 2100 to 6480 mg/kg for rats. The oral LD50 of butyl benzoate was 5140 mg/kg for female rats. Orally administered C12-15 alkyl benzoate was not toxic to rats at 5000 g/kg. The oral LD50 for isopropyl benzoate was 3730 mg/kg and 3685 mg/kg for isobutyl benzoate in rats.
The dermal LD50 of methyl benzoate was >2000 mg/kg for rabbits. Dermally administered ethyl benzoate at 10% caused no effects to mice and calves; at 100% it was lethal to cats. Dermally administered butyl benzoate caused diarrhea in rabbits at 5000 g/kg. C12-15 alkyl benzoate, at 100% or 2 mg/kg, was not dermally toxic to rabbits. The dermal LD50 of isopropyl benzoate was 20 mg/kg for rabbits. Isobutyl benzoate was not toxic to rabbits when administered dermally.
Methyl benzoate, ethyl benzoate, butyl benzoate, and isopropyl benzoate were rated as grade 1 ocular irritants at 100% when using rabbits. In in vivo tests, C12-15 alkyl benzoate was a mild ocular irritant at 100%. Isopropyl benzoate was rated as nonirritant to mild ocular irritant at 100% as was isostearyl benzoate at 0.95% in in vitro tests.
Methyl benzoate, ethyl benzoate, propyl benzoate, and butyl benzoate at 100% were dermally irritating to rabbits. C12-15 alkyl benzoate was a mild dermal irritant to rabbits at 100% and was found to be irritating in an in vitro test.
In guinea pigs, methyl benzoate up to 10%, ethyl benzoate up to 8%, amyl benzoate up to 6%, C12-15 alkyl benzoate up to 10%, and isobutyl benzoate up to 2% were nonsensitizing to guinea pigs.
One oral study of the related compound isononyl benzoate resulted in a slight decrease in mean pup weight and consequently in litter weight in high-dose females at birth and on day 4 postpartum. The NOAEL was 1000 mg/kg/d.
In Ames tests, methyl benzoate, ethyl benzoate, and C12-15 alkyl benzoate were not genotoxic. Isononyl benzoate administered orally at 2000 mg/kg to rats did not induce micronuclei in the polychromatic erythrocytes. Ethylhexyl benzoate was not an ocular irritant to humans in a sunscreen at 3.5%.
Methyl benzoate was not dermally irritating to humans at 4%. Ethyl benzoate and butyl benzoate were not dermally irritating to humans at 8%. C12-15 Alkyl benzoate was not irritating at 100%. Isobutyl benzoate was nonirritating at 2% as was ethylhexyl benzoate at 3.5%.
In HIRPTs and maximization studies, methyl benzoate and ethyl benzoate at 0.5%, C12-15 alkyl benzoate at 100%, stearyl benzoate at 2%, isobutyl benzoate at 2%, isostearyl benzoate at 0.95%, ethylhexyl benzoate at 3.5%, and octyldodecyl benzoate at 0.4% were not sensitizing. Methyl benzoate at 0.1% and ethylhexyl benzoate at 3.5% were not phototoxic to humans.
Benzoic Acid and Sodium Benzoate
Dermally applied benzoic acid is excreted in the urine within 24 hours.
The oral LD50 of benzoic acid was reported to be 1996 mg/kg in mice and 2000 to 2500 mg/kg in rats. The oral LD100 was reported to be 1520 to 2000 mg/kg for rabbits, and 2000 mg/kg for cats and dogs. The oral LD50 for sodium benzoate was 2100 to 4070 mg/kg for rats and 2000 mg/kg for rabbits and dogs.
Benzoic acid and sodium benzoate were orally toxic to rats and mice in short-term feeding studies at concentrations >1% in short-term, subchronic, and chronic studies. In subchronic studies, benzoic acid was toxic to mice at oral doses of 80 mg/kg/d. Sodium benzoate at 880 mg/kg/d incorporated into the feed of rats for 18 to 24 months was not toxic.
Studies on sodium benzoate and benzoic acid did not show reproductive or developmental toxicity in rats. Where effects of the fetus were noted, they occurred at maternally toxic concentrations (>4% sodium benzoate in rats).
Benzoic acid and sodium benzoate produced both positive and negative results in several genotoxic assays.
Benzoic acid (40 mg/kg/d) orally administered to mice increased the number of tumors compared to controls. Benzoic acid was negative for carcinogenicity when dermally applied to mice at 0.016% in a nonoxidative hair dye.
Benzoic acid at 0.2% was not irritating to participants. Benzoic acid at 0.2% caused mild, transient irritation when applied daily in a liquid foundation product at least twice/day for 45 days. Benzoic acid was negative for sensitization up to 2%.
Alcohols
Methyl alcohol and ethylhexyl alcohol permeated the skin or nail plates. Aerosolized lauryl alcohol caused mild dyspnea and scattered hemorrhagic areas in the lungs of rats.
Methyl alcohol was cytotoxic to yeast cells at 0.05% but not Chinese hamster cells at 10%. Amyl alcohol was cytotoxic to yeast cells and Chinese hamster cells at 0.5%. Dodecyl alcohol was cytotoxoic to human erythrocytes 15 µmol/L.
Methyl alcohol has an oral LD50 of 5628 mg/kg for rats and 7300 mg/kg for mice. The oral LD50 of amyl alcohol for rats was reported to be 2.69 g/kg. Dodecyl alcohol has an oral LD50 of 12 800 mg/kg for rats. Tridecyl alcohol has an oral LD50 of 17 200 mg/kg for rats. Tetradecyl alcohol has an oral LD50 33 000 mg/kg for rats. The oral LD50 for ethylhexyl alcohol in rats ranged from 2049 to 7100 mg/kg and in rabbits from 2380 to >5000 mg/kg. The oral LD50 of hexyldecyl alcohol for rats was reported to be >8.42 g/kg. The dermal LD50 of methyl alcohol was reported to be 15 800 mg/kg in rabbits.
The dermal LD50 of amyl alcohol for rabbits was reported to be >3.2 g/kg. The dermal LD50 of dodecyl alcohol was reported to be 3560 mg/kg in rabbits. The dermal LD50 of tridecyl alcohol was reported to be 5600 mg/kg in rabbits. The dermal LD50 of hexadecyl alcohol for rabbits was reported to be >2.6 g/kg. Aerosolized amyl alcohol at near saturation caused irritation of the eyes, nose, throat, and respiratory passages of mice and guinea pigs. Rats exposed to aerosolized ethylhexyl alcohol exhibited signs of irritation of the eyes nose, throat, and respiratory passages, including blinking, lacrimation, nasal discharge, salivation, gasping, and chewing movements. No rats died from exposure for up to 8 hours. Hexyldecyl alcohol was a slight irritant with no systemic effect to mice, rats, and guinea pigs at 9.6 mg/m3.
Thirteen-week oral exposure to amyl alcohol at 100% was not toxic to rats. Oral NOAELs ranged from 100 to 150 mg/kg in several studies using mice or rats exposed to ethylhexyl alcohol. Dermal exposure to ethylhexyl alcohol caused physiological changes in rats at 500 mg/kg. Inhalation of isobutyl alcohol at 770 mg/m3 caused reversible inhibition of responsiveness in rats. Adverse effects of isopropyl alcohol at the LOAEL included clinical signs in rat and mice, hematological changes in rats, and increased liver weights in mice; higher doses caused kidney and testicular effects. Aerosolized n-pentadecyl alcohol was not toxic to rats.
Methyl alcohol was a mild ocular irritant at 100% in in vitro tests. Amyl alcohol, dodecyl alcohol, isopropyl alcohol, and ethylhexyl alcohol were determined to be severe ocular irritants to rabbits at 100%. Hexyldecyl alcohol was a slight ocular irritant at 100% to rabbits. Ethylhexyl alcohol, and hexydecyl alcohol were dermal irritants at 100%.
Ethylhexyl alcohol ≥10 mL/m3 increased nasal and eye irritation and perceived odor intensity at ≥10 mL/m3. Little or no dermal irritation was observed in tests of propyl alcohol, lauryl alcohol, cetyl alcohol, cetyl alcohol, isostearyl alcohol, and ethylhexyl alcohol on humans. Dodecyl alcohol, cetyl alcohol, isopropyl alcohol, isopropyl alcohol, isostearyl alcohol, and ethylhexyl alcohol were not sensitizing. A product containing isostearyl alcohol at 5.0% was sensitizing. A product containing cetyl alcohol was not phototoxic in humans. Cetyl alcohol, stearyl alcohol, benzyl alcohol, and propyl alcohol had no comedogenic activity. Lauryl alcohol and myristyl alcohol had slight comedogenity. Octyl alcohol had strong comedogenicity.
Aerosolized methyl alcohol caused no maternal effects in rats but caused decreased weights and increased malformations in offspring. Male mating success was reversibly decreased. The oral NOAELs for the maternal and developmental toxicity of isopropyl alcohol were 400 mg/kg in rats (maternal and developmental) and 240 mg/kg (maternal) and 480 mg/kg (developmental) in rabbits.
Methyl alcohol and ethylhexyl alcohol were not genotoxic is various assays.
Orally administered methyl alcohol, amyl alcohol, lauryl alcohol, and dodecyl alcohol caused increases in polyploidy cells, cells with gaps, and cells with aberrations in the bone marrow of rats. Ethylhexyl alcohol was a weak liver tumor promoter in female mice.
Discussion
The CIR Expert Panel noted the shorter chain length alkyl benzoates penetrated the skin and resulting in systemic exposure. Less clear was the dermal penetration of long chain length alkyl benzoates. Alkyl benzoates, which have octanol/water partition coefficient values above 8, are not expected to leave the stratum corneum and reach the epidermis. However, 5% to 8% of C12-15 alkylbenzoate applied in 3 different product formulations was reported to penetrate the epidermis of pig skin in vitro.
The Panel reasoned that the data indicating moderate penetration of alkyl benzoates into the epidermis suggested the need for a conservative approach that would consider the potential for systemic exposure to long chain length alkyl benzoates. Alkyl benzoates are largely nontoxic, but, given that these ingredients may penetrate the skin, they may be cleaved and result in systemic exposure to the component alcohol and benzoic acid. Component alcohols may be toxic at high levels, for example, so the Panel considered available data on the parent compounds and on benzoic acid and component alcohols. While data were not available on reproductive/developmental toxicity of alkyl benzoates used in cosmetics, one study on the structurally similar isononyl benzoate demonstrated an absence of reproductive/developmental toxicity.
The Panel noted that carcinogenicity data were not available for alkyl benzoates, but that available data indicated that these alkyl benzoate cosmetic ingredients are not genotoxic and are not dermal sensitizers. In addition, available data demonstrate that benzoic acid and tested component alcohols are not reproductive or developmental toxicants, are not genotoxic in almost all assays, and are not carcinogenic. The Panel considered, therefore, that the low levels at which most alkyl benzoates are used would not result in any significant systemic toxicity for their metabolites; C12-15 alkyl benzoates, which can be used at a higher concentration is poorly absorbed through the skin and therefore would not likely cause systemic toxicity.
Several of the shorter chain length alkyl benzoates were cytotoxic at high doses and dermal irritants in animal tests but were not significant irritants in clinical tests. Due to the lack of irritation by the shorter chain length alkyl benzoates at use concentrations when used in cosmetic formulations, alkyl benzoates are not expected to result in any cytotoxicity.
While plants are the source of some components in the ingredients of this report, they result from significant processing, and as such are not expected to contain residual pesticides or heavy metals.
Certain of the alkyl benzoates are used in cosmetic products that may be inhaled during their use. In practice, however, the particle sizes produced by cosmetic aerosols are not respirable.
Conclusion
The CIR Expert Panel concluded that the following ingredients are safe in the present practices of use and concentration described in this safety assessment (ingredients not in current use identified with an *):
methyl benzoate,
ethyl benzoate,
propyl benzoate*,
butyl benzoate*,
amyl benzoate*,
lauryl/myristyl benzoate*,
C12-15 alkyl benzoate,
C16-17 alkyl benzoate,
stearyl benzoate,
behenyl benzoate*,
isopropyl benzoate*,
isobutyl benzoate*,
isostearyl benzoate,
ethylhexyl benzoate,
butyloctyl benzoate*,
hexyldecyl benzoate*, and
octyldodecyl benzoate.
Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.