Abstract
The safety of isoparaffins as used in cosmetic products is reviewed in this safety assessment. These ingredients function mostly as solvents and also function as emollients in the 0001% to 90% concentration range. The Cosmetic Ingredient Review (CIR) Expert Panel has reviewed relevant animal and clinical data and concluded that these ingredients are safe in the present practices of use and concentration described in this safety assessment.
Keywords
Introduction
Isoparaffinic hydrocarbons (isoparaffins) are branched alkanes that function mostly as solvents in cosmetics. The safety of the following isoparaffins is reviewed:
C7-8 isoparaffin;
C8-9 isoparaffin;
C9-11 isoparaffin;
C9-12 isoparaffin;
C9-13 isoparaffin;
C9-14 isoparaffin;
C9-16 isoparaffin;
C10-11 isoparaffin;
C10-12 isoparaffin;
C10-13 isoparaffin;
C11-12 isoparaffin;
C11-13 isoparaffin;
C11-14 isoparaffin;
C12-14 isoparaffin;
C12-20 isoparaffin;
C13-14 isoparaffin;
C13-16 isoparaffin;
C18-70 isoparaffin;
C20-40 isoparaffin;
C15-35 isoparaffin/isoalkylcycloalkanes;
Isooctane;
Isohexadecane;
Isododecane;
Isoeicosane.
Related Cosmetic Ingredient Review (CIR) final safety assessments on fossil and synthetic waxes, including paraffin 1 and 2 other branched alkanes, isobutane and isopentane, 2 have been published. The CIR Expert Panel concluded that fossil and synthetic waxes, including paraffin, and isobutene and isopentane are safe in the present practices of use and concentration. These 2 conclusions were confirmed in 2005.3,4
Chemistry
Definition and Structure
The systematic name for the paraffins is alkanes (CnH2n+2); isoparaffins are branched alkanes. Definitions, other chemical names, and cosmetic ingredient functions for the isoparaffins reviewed in this safety assessment are included in Table 1. The naming convention for many of these ingredients, for example, C7-8 isoparaffin, connotes that the ingredient is a mixture of branched chain aliphatic hydrocarbons with 7 or 8 carbons in the alkyl chain. In the case of C18-70 isoparaffin, the mixture has a broad range of chain lengths.
Isoparaffinic Hydrocarbons. 20
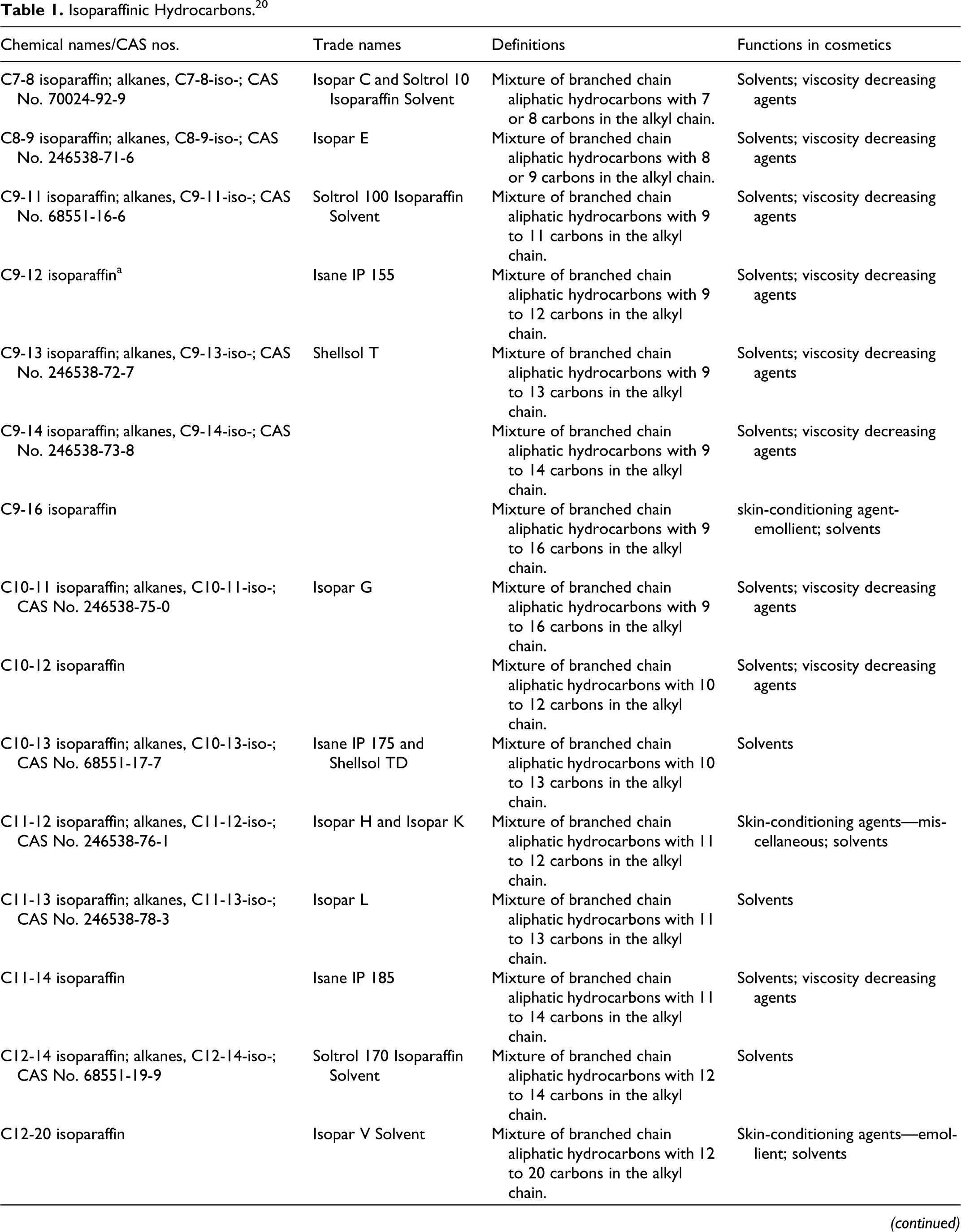
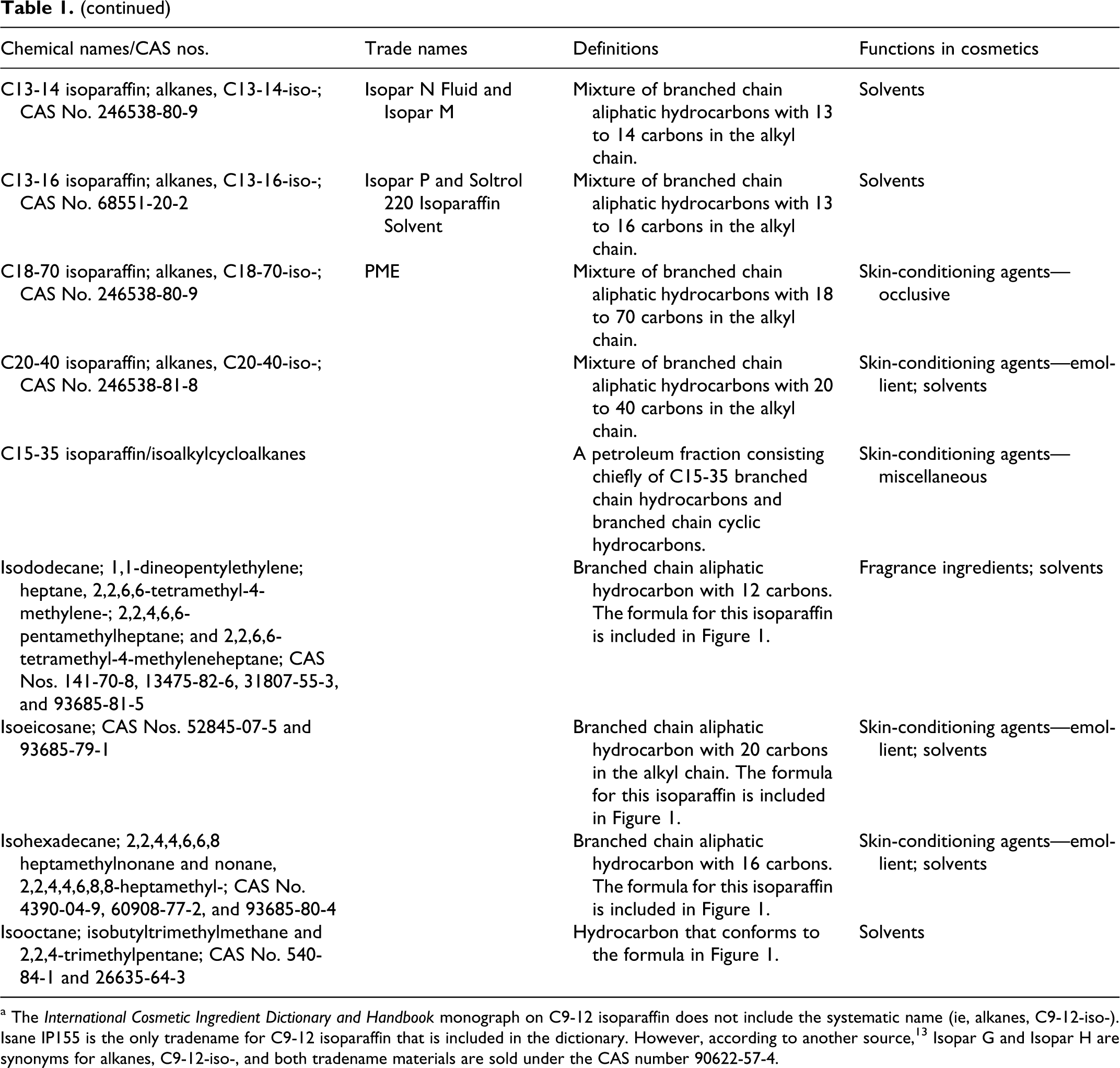
a The International Cosmetic Ingredient Dictionary and Handbook monograph on C9-12 isoparaffin does not include the systematic name (ie, alkanes, C9-12-iso-). Isane IP155 is the only tradename for C9-12 isoparaffin that is included in the dictionary. However, according to another source, 13 Isopar G and Isopar H are synonyms for alkanes, C9-12-iso-, and both tradename materials are sold under the CAS number 90622-57-4.
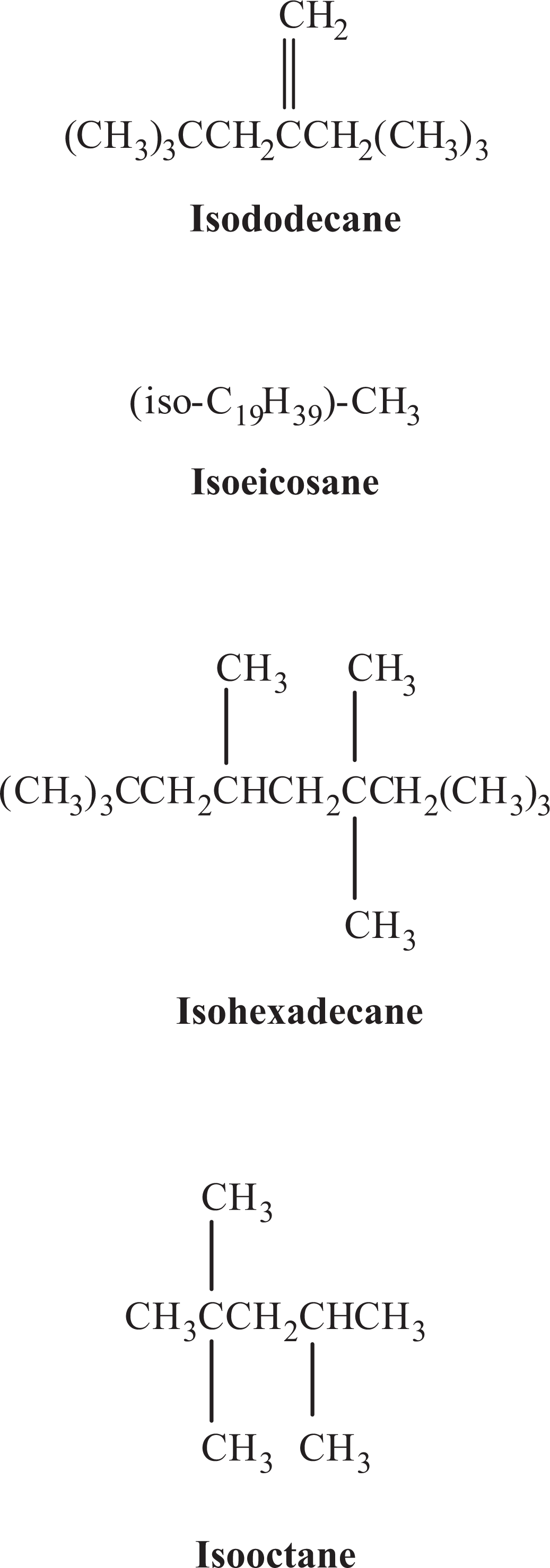
Isoparaffin formulas.
Chemical and Physical Properties
Isooctane (colorless liquid) is practically insoluble in water, somewhat soluble in absolute alcohol, and soluble in the following chemicals: benzene, toluene, xylene, chloroform, ether, carbon disulfide, and carbon tetrachloride. 5 Additional properties of isoparaffins are included in Tables 2 and 3.
Properties of Isoparaffins. 101

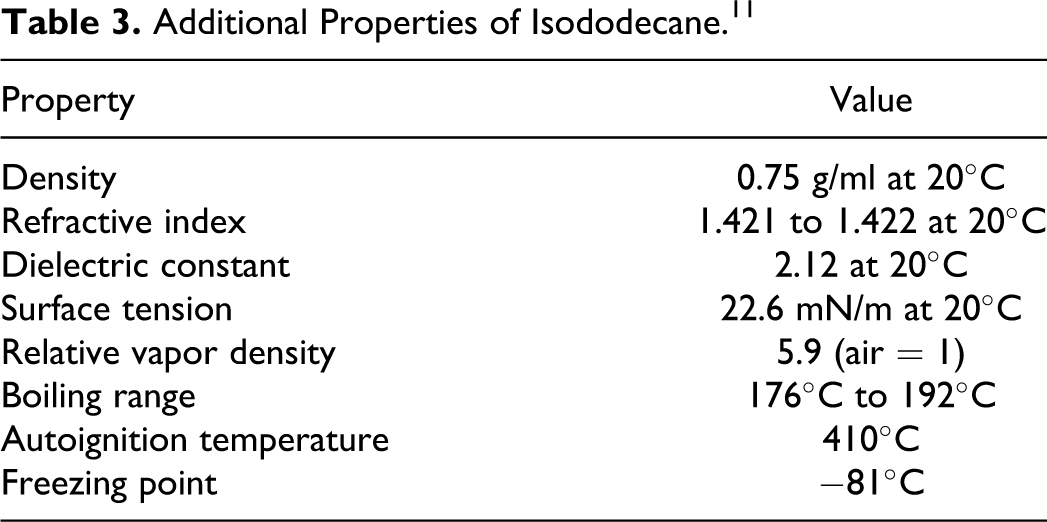
Additional Properties of Isododecane. 11
Properties of isoparaffinic hydrocarbon tradename materials produced by various companies are included in Tables 4 and 5. Branched aliphatic hydrocarbons that are predominantly C10-15 isoparaffinic hydrocarbons in Table 4 are colorless to water-white liquids with a faint petroleum odor. 6 Isopar C is one of the tradename materials included in Table 5. According to Exxon Company, Isopar C is approximately 85% isooctane. 7
Properties of Isoparaffinic Tradename Materials. 6
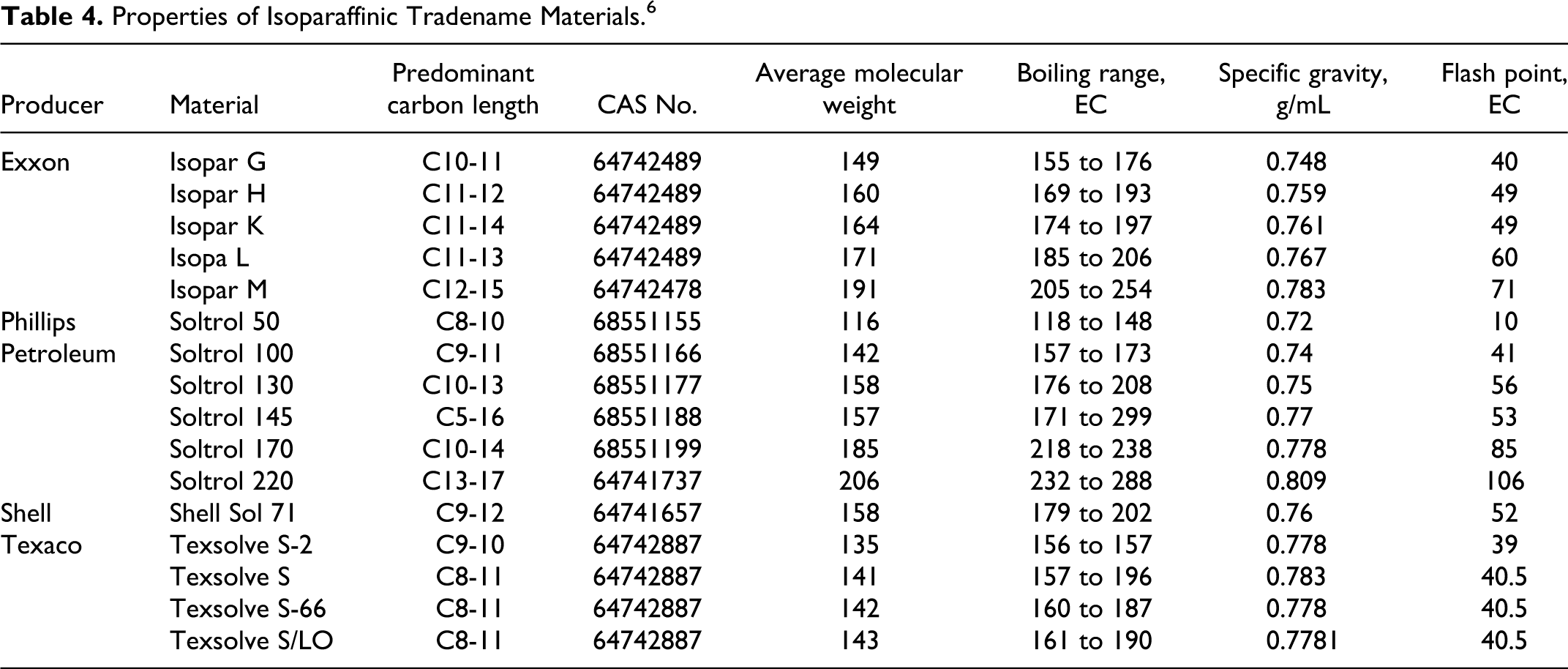
Properties of Isopar Isoparaffinic Hydrocarbon Solvents. 102
a All manufactured by Exxon Corporation.
Stability/Reactivity
Branched aliphatic hydrocarbons are predominantly C10-15 isoparaffinic hydrocarbons. These chemicals are quite stable and relatively unreactive, such that polymerization will not occur. However, they are incompatible with strong oxidants (eg, liquid chlorine, sodium hypochlorite, or concentrated O2). The incomplete combustion of these compounds may produce CO and aldehydes. 6
Isooctane, an example of this class of compounds, is considered stable under normal ambient and anticipated storage and handling conditions of temperature and pressure. It may react with oxygen and strong oxidizing agents, such as chlorates, nitrates, peroxides, and so on. Hazardous decomposition products include simple hydrocarbons and carbon oxides. Hazardous polymerization is not expected to occur. 8
Methods of Production
Alkylation is the chemical combination of 2 light hydrocarbon molecules to form a heavier one and involves the reaction of butenes in the presence of a strong acid catalyst, such as sulfuric or hydrofluoric acid. The product is a heavier multibranched isoparaffin. Propene and various pentenes may be used to produce C7 or C9 isoparaffins. Additionally, isomerization is a catalytic process that converts normal paraffins to isoparaffins. The feed is usually light virgin naphtha and, the catalyst, platinum on an alumina or zeolite base. 9 According to Ineos, their manufacturing process does not start with naptha; a pure C4 stream rich in isobutylene is used, leading to C8, C12, C16, and C20 isoparaffins. 10
According to Ineos oligomers, their manufacturing process of isoparaffins does not start with naptha. A pre C4 steam rich in isobutylene is used, leading to C8, C12, C16, and C20 isoparaffins.
The production of isododecane has been described as a patented process from a prepurified isobutene (branched C4) containing feedstock and, therefore, is totally sunthetic. The dimethyl branches (termed as germinal or gem dimethyls) in the isobutene monomer are retained in the final product. 11
2,2,4-Trimethylpentane (isooctane) is synthesized from the catalytic hydrogenation of trimethylpentene with a nickel catalyst. 12
Composition/Impurities
According to Ineos oligomers, isoparaffin substances are produced from well-defined feedstocks and are very pure. 10
Isododecane (a.k.a. hydrocarbons, C4, 1,3-butadiene-free, polymd., triisobutylene fraction, hydrogenated [CAS No. 93685-81-5]) is a mixture of highly branched C12 isoparaffins, mainly the 2,2,4,6,6-pentamethylheptane isomer (typically ∼85%). 10 The structure of this isomer is close to a fully permethylated hydrocarbon structure, containing the maximum number of methyl groups. Isododecane also contains ∼15% of other pentamethylheptanes that have properties that are similar to the main isomer. 11
Isoeicosane (hydrocarbons, C4, 1,3-butadiene-free, polymd., pentaisobutylene fraction, hydrogenated [CAS No. 93685-79-1]) is a mixture of highly branched C20 isoparaffins, with not more than 2% of C16 isoparaffins. Isohexadecane (hydrocarbons, C4, 1,3-butadiene-free, polymd., tetraisobutylene fraction, hydrogenated [CAS No. 93685-80-4]; minimum purity of 99%) is a mixture of highly branched C16 isoparaffins with trace amounts of C12 and C20 paraffins of similar structure. Isooctane (90% 2,2,4-trimethylpentane [CAS No. 540-84-1] is a mixture of C8 isoparaffins. 10
Isododecane is virtually free of aromatics, sulfur-containing molecules, and polar compounds and isooctane also contains virtually no aromatics or sulfur compounds. 10 The INEOS sales specification for isododecane is as follows 11 :
Sum of C12 hydrocarbons (% by weight): 98 min.
Sum of C8 and C16 hydrocarbons (% by weight): 2 max.
Aromatics (mg/kg): 1 max.
Carbonyls (mg/kg): 5 max.
Bromine index (mgBr2/100 g): 15 max.
Sulfur (mg/kg): 1 max.
Peroxides (mg/kg, calculated as H2O2): 1 max.
Water (mg/kg): 50 max.
Evaporation residue (mg/100 mL): 1 max.
Neutralization number (mg KOH/g): 0.01 max.
Reportedly, according to the hydrocarbon solvent producers, the actual composition of Isopar G and Isopar H (tradenames for C10-11 isoparaffin and C11-12 isoparaffin, respectively) may be different from batch to batch within a specific producer and may vary from producer to producer, depending upon the actual feedstock used to prepare the product. Data on average composition indicate that Isopar G contains mostly C10 (53% w/w) and that Isopar H contains mostly C12 (60% w/w). The average Isopar H branching is 3.25 (average number of branches/molecule), and estimates for the number of different branches are as follows: C (0.6), CH (2), CH2 (4.2), and CH3 (5.3). Additionally, the producers have provided information indicating that 5% to 15% w/w of the substance consists of cyclic alkanes, typically C6 with varying degrees of branching. 13
Both Exxon and Texaco have reported benzene (<0.1 ppm) as an impurity for all of their isoparaffinic products, and data from Ricoh Corporation in Japan established that benzene contamination was <0.2 ppm for Isopar G. These include, but do not appear to be limited to C9 to C15 chain lengths. 6 According to another publication, C9-C13 aliphatic solvents (≤ 2% aromatics) contain <1 ppm benzene. 14
As direct food additives, isoparaffinic petroleum hydrocarbons synthesized from petroleum gases consist of a mixture of liquid hydrocarbons that meet the following specifications (21 CFR 172.882). 15
Boiling point: 93°C to 260°C
Ultraviolet (UV) absorbance at 260 to 319 nm: 1.5 max.
UV absorbance at 320 to 329 nm: 0.08 max.
UV absorbance at 330 to 350 nm: 0.05 max.
Nonvolatile residual: 0.002 g/100 mL max.
Additionally, these direct food additives may contain antioxidants authorized for use in food, in an amount not to exceed that reasonably required to accomplish the intended technical effect nor to exceed any prescribed limitations. 15 The boiling point specification for isoparaffinic petroleum hydrocarbons as indirect food additives is 63°C to 260°C (21 CFR 178.3530), 16 and the preceding specifications relating to direct food additive use also apply.
Petroleum hydrocarbons classified as odorless and light (a mixture) is also an approved direct food additive. It is a mixture of liquid hydrocarbons derived from petroleum or synthesized from petroleum gases and is chiefly paraffinic, isoparaffinic, or naphthenic in nature. This food additive meets the following specifications (21 CFR 172.884) 17 :
Odor is faint and not kerosenic
Initial boiling point is 300°F min.
Final boiling point is 650°F max.
UV absorbance at 280 to 289 nm: 4.0 max.
UV absorbance at 290 to 299 nm: 3.3 max.
UV absorbance at 300 to 329 nm: 2.3 max.
UV absorbance at 330 to 360 nm: 0.8 max.
The preceding specifications for odorless light petroleum hydrocarbons as a direct food additive is also applicable to its use as an indirect food additive (21 CFR 178.3650). 18
Analytical Methods
Gas chromatography has been used to identify isoparaffins. 19 Specifically, isooctane has been identified using gas chromatography with simultaneous flame ionization detection and radiolabeling analytical techniques. 7
Use
Purpose in Cosmetics
Most of the isoparaffins reviewed in this safety assessment function as solvents in cosmetics, and these ingredients are also used as emollients. 20 Additional functions are included in Table 1.
Scope and Extent of Use in Cosmetics
According to information supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP) in 2010, the following ingredients were being used in cosmetics. C7-8 isoparaffin, C8-9 isoparaffin, C9-11 isoparaffin, C10-11 isoparaffin, C11-12 isoparaffin, C11-13 isoparaffin, C12-14 isoparaffin, C13-14 isoparaffin, C13-16 isoparaffin, C18-70 isoparaffin, isododecane, isoeicosane, and isohexadecane. 21 These data are summarized in Table 6. Isooctane is not included in the VCRP database; however, Isopar C (C7-8 Isoparaffin) contains approximately 85% isooctane. Independent of these data, the results of a survey of ingredient use concentrations that was conducted by the Personal Care Products Council in 2010, also shown in Table 6, indicate that the following ingredients were being used: C8-9 isoparaffin (5%-40%), C9-11 isoparaffin (1%-18%), C10-13 isoparaffin (0.08%-0.60%), C11-12 isoparaffin (1%-67%), C11-13 isoparaffin (1%-27%), C13-14 isoparaffin 0.0001%-75%), C13-16 isoparaffin (0.40%-18%), isododecane (0.008%-90%), isoeicosane (0.3%-37%), and isohexadecane (0.2%-42%). 22
Current Frequency and Concentration of Use According to Duration and Type of Exposure Provided in 2010.a
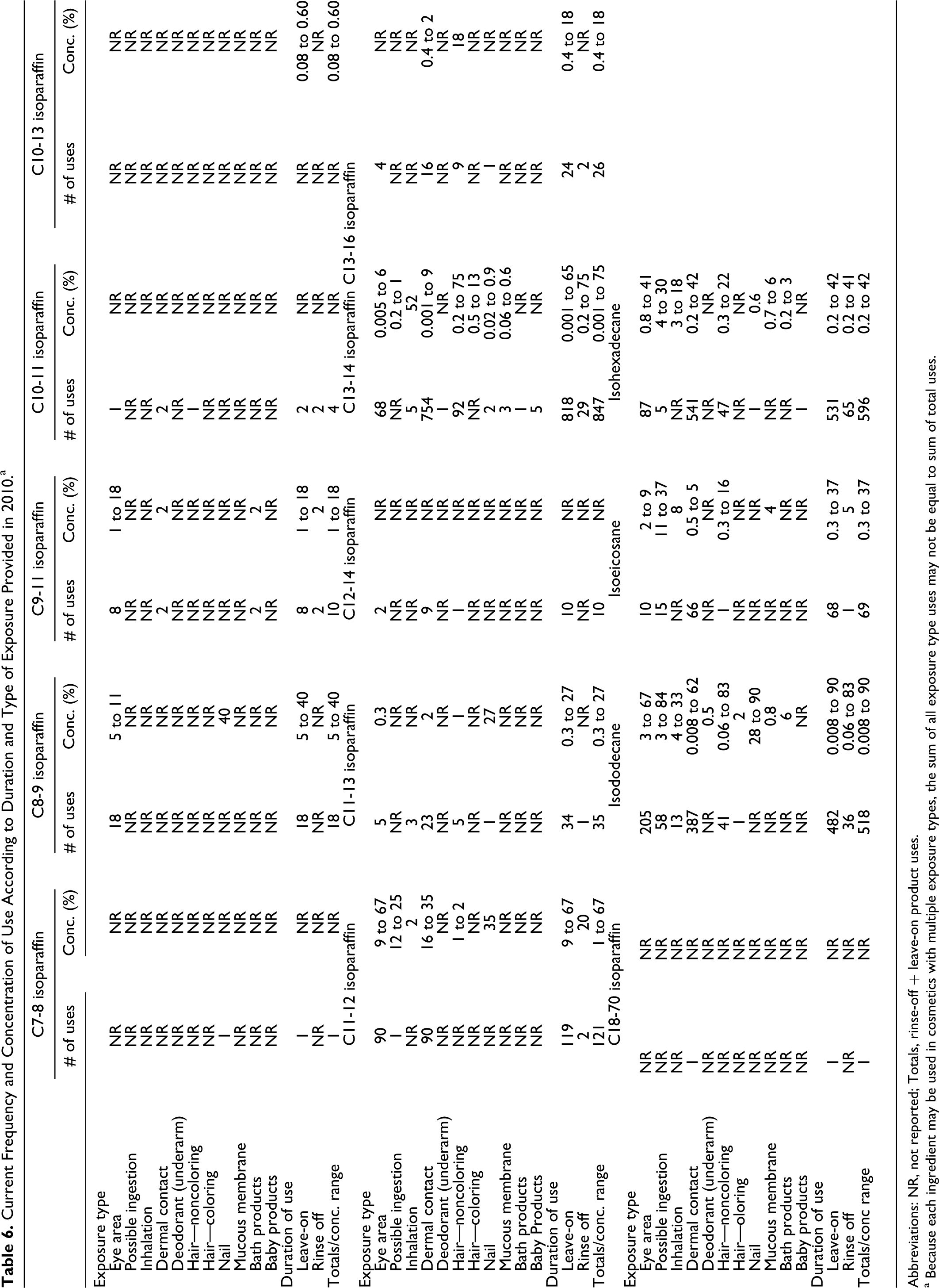
Abbreviations: NR, not reported; Totals, rinse-off + leave-on product uses.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not be equal to sum of total uses.
No uses of the following isoparaffins were reported in the 2010 VCRP database or in the Personal Care Products Council survey conducted in 2010: C9-12 isoparaffin, C9-13 isoparaffin, C9-14 isoparaffin, C9-16 isoparaffin, C10-12 isoparaffin, C11-14 isoparaffin, C12-20 isoparaffin, C20-40 isoparaffin, and C15-35 isoparaffin/isoalkylcycloalkanes. The results of a subsequent 2010 use concentration survey by the Personal Care Products Council confirmed that C15-35 isoparaffin/isoalkylcycloalkanes is not being used in cosmetic products. 23
Cosmetics containing the ingredients reported as being used may be applied to the skin, nails, or hair, or, incidentally, may come in contact with eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin, nails, or hair for variable periods following application. Daily or occasional use may extend over many years.
C11-13 isoparaffin, C13-14 isoparaffin, and isododecane are used in hair sprays, and effects on the lungs that may be induced by aerosolized products containing these ingredients are of concern.
The aerosol properties that determine deposition in the respiratory system are particle size and density. The parameter most closely associated with deposition is the aerodynamic diameter, da, defined as the diameter of a sphere of unit density possessing the same terminal settling velocity as the particle in question. In humans, particles with an aerodynamic diameter of ≤10 µm are respirable. Particles with a da from 0.1 to 10 µm settle in the upper respiratory tract and particles with a da < 0.1 µm settle in the lower respiratory tract.24,25
Particle diameters of 60 to 80 µm and ≥80 µm have been reported for anhydrous hair sprays and pump hairsprays, respectively. 26 In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range and the mean particle diameter in a typical aerosol spray has been reported as ∼38 µm. 27 Therefore, most aerosol particles are deposited in the nasopharyngeal region and are not respirable.
Noncosmetic Use
Isopar E is a mixture of predominantly C8-C9 isoparaffinic hydrocarbons that has been used as a solvent in industrial and consumer products, including, but not limited to, typewriter correction fluids. 28
Liquid gasoline is a complex mixture of petroleum chemicals. In addition to other components, this complex mixture consists of approximately 60% to 75% alkanes (paraffins) that comprise straight-chain hydrocarbons (C4-C12) and isoparaffins (branched-chain hydrocarbons) with approximately the same range of chain lengths. 29 2,2,4-Trimethylpentane (isooctane) is used primarily in the alkylation step to derive high-octane gasoline fuels. 12 California phase-2 reformulated gasoline contains isooctane at a concentration of 32.1%. 30
According to the US Pharmacopoeia, use of ACS reagent grade isooctane is the reagent specification for prescription and over-the-counter drug products. 31
Synthetic isoparaffinic petroleum hydrocarbons are used as direct and indirect food additives. Specifications for these classes of direct and indirect food additives are included in the earlier section on Composition/Impurities (21 CFR 172.882; 178.3530).15,16
Odorless light petroleum hydrocarbons, a mixture that is chiefly paraffinic, isoparaffinic, or naphthenic in nature, is used as a direct and indirect food additive. Specifications for these classes of direct and indirect food additive mixture are included in the earlier section on Composition/Impurities (21 CFR 172.884; 178.3650).17,18
According to Ineos, isododecane is used in the polymerization process, where it is used as either a solvent or a dispersing agent for high-reactivity organic peroxide catalysts. 10 High-activity peroxide catalysts used in the production of polyvinyl chloride and in cross-linked unsaturated polyester are frequently dissolved in isododecane. 11 Isoeicosane has been used as a heat transfer medium for heating and cooling circuits used in chemical processes, particularly in conjunction with isohexadecane. Other applications of isoeicosane include hydraulic fluids and as an extender in silicone-based dental impression materials. 10
Toxicokinetics
Isooctane—Inhalation Exposure
Groups of 15 male F344 rats were exposed for 2 hours, nose-only, to 14 C-labeled isooctane vapor at concentrations of ∼1 and 350 ppm. Absorbed 14 C-isooctane equivalents were eliminated almost exclusively via the kidneys, and excretion was protracted over the entire 70 hours postexposure observation period. Of all, 1% to 2% of the 14 C-isooctane equivalents inhaled at either exposure concentration (∼1 or 350 ppm) remained in the carcass 70 hours after inhalation exposure. 32
Results from another inhalation study identified the following 8 principal urinary metabolites of isooctane in the urine of male Fischer 344 rats: 2,2,4-trimethyl-1-pentanol; 2,4,4-trimethyl-1-pentanol; 2,4,4-trimethyl-2-pentanol; 2,2,4-trimethyl-l-pentanoic acid; 2,4,4-trimethyl-l-pentanoic acid; 2,4,4-trimethyl-2-hydroxy-1-pentanoic acid; 2,2,4-trimethyl-5-hydroxy-1-pentanoic acid; and 2,4,4-trimethyl-5-hydroxy-1-pentanoic acid. 33
Isooctane—Oral Dosing
The metabolism of 14 C-isooctane was studied using 8 groups of male Fischer-344 rats (3 per group). 7 The animals were dosed by oral gavage at a dose level of ∼1 mL/kg (2.9 × 107 DPM/kg).
Most of the radioactivity was recovered in the urine and feces after 72 hours, with much of this excretion occurring between 24 and 72 hours. More than one-third of the dose was detected (and identified as unmetabolized isooctane) in exhaled air after 24 hours; essentially no additional lung excretion occurred between 24 and 72 hours. Approximately 2.5% of the dose remained in the tissues after 72 hours. Meaningful blood levels of isooctane were not found; however, plasma radioactivity indicated the presence of circulating metabolites. Tissue concentrations of radioactivity indicated initial accumulation in the liver over the first 8 hours, followed by a decline in liver radioactivity. A high concentration of radioactivity was detected in the kidney, surpassing the liver by 12 hours. A maximum was reached at 24 hours, and levels declined slowly thereafter. The authors suggested that tissue and plasma levels were consistent with liver formation of metabolites, followed by transport to the kidney in blood plasma and concentration or accumulations in the kidney. 7
Sexually mature male and female Fischer 344 rats were dosed by gavage (5 mL/kg) with a single dose of 3 H-isooctane, in corn oil, at 4.4 mmol/kg and 230 µCi/kg. The kidneys of male rats retained more radiolabeled material than those of female rats. Subcellular fractionation of the kidneys of male rats at 24 hours postdosing showed that approximately 60% of the radiolabeled material was localized in the 116 000 g supernatant. Gas chromatography–mass spectrometry of an ethyl acetate extract of the α2u-globulin-containing fractions of the 116 000 g supernatant identified 2,4,4-trimethyl-2-pentanol as the only metabolite bound to α2u-globulin. 34 The role of this protein in nephrotoxicity is mentioned under that subheading.
Adult Fischer 344 rats of both sexes were given 5 µCi of [ 14 C-5]-isooctane (0.5 g/kg oral dose). 35 Whole body and kidney autoradiography were performed using [ 14 C-5]-isooctane (50 µ Ci/rat) at the same dose. At an oral dose of 0.5 g/kg, exhaled organics (presumably the parent compound) accounted for 45% to 50% of the dose in both males and females. There were no differences between the sexes in percentages of the administered dose excreted in the urine, feces, or as expired CO2. A small percentage of the radioactivity was associated with exhaled CO2, indicating that minimal terminal carbon oxidation on the isooctane molecule had occurred. Approximately 1% of the radiolabel remained in the carcasses and tissues of both male and female rats.
Relatively little radioactivity was detected in the tissues at 72 hours, However, kidney levels of radioactive isooctane indicated a pronounced sex difference; male rats retained approximately 10-fold greater amounts of radioactivity than female rats. Less than 0.02% of the dose was detected in the brain, heart, spleen, fat, lung, and liver, with no apparent sex differences. Analysis of whole body autoradiography of isooctane at 72 hours confirmed that the majority of the radioactivity in the male rat was localized in the kidney, with minor amounts in the peritoneal fat. Autoradiography results indicated that the radioactivity detected in the kidney was associated with the renal cortex. 35
In another study, male and female Fischer 344 rats received a single oral dose of 14 C-isooctane (4.4 mmol/kg; 2 pCi/mmol), and radiolabeled material in the kidney, liver, and plasma was determined at 4, 8, 12, 24, and 48 hours after dosing. The maximum concentration of isooctane-derived radioactivity in the kidney, liver, and plasma of male rats was found after 12 hours (1252, 1000, and 403 nmol eq/g, respectively); maximum concentrations in females were found after 8 hours (577, 1163, and 3 17 nmol eq/g, respectively). A selective retention of the isooctane-derived radiolabel in the kidneys of male rats was noted when peak tissue concentration was expressed as a percentage of administered dose. Kidney concentrations of isooctane-derived radiolabel increased in a nonlinear, but dose-dependent, manner. 2,4,4-Trimethyl-2-pentanol was the major metabolite detected in the male rat kidney but was absent from the female rat kidney. However, compared to males, female rats excreted more conjugates of 2,4,4-trimethyl-2-pentanol in the urine. 36
C9 to C14 Isoalkanes/Isododecane
Under the provisions of the Registration, Evaluation, Authorization, and Restriction of Chemical Substances (REACH) regulation in Europe, metabolism data on C9 to C14 isoalkanes were summarized in a chemical safety report on isododecane. 37 According to this report, C9 to C14 isoalkanes are taken up into the blood, distributed to the internal organs, and rapidly eliminated following exposure. The concentration of isoalkanes in the blood, brain, liver, and fat increases with increasing carbon chain lengths. These findings are consistent with other metabolism data on isoparaffins included in this section.
Animal Toxicology
Single-Dose Toxicity
Inhalation
The lethal concentration was greater than the highest concentration generated in each acute inhalation study on a number of isoparaffins as summarized in Table 7.
Acute Inhalation Toxicity Studies on Isoparaffins and Isooctane.
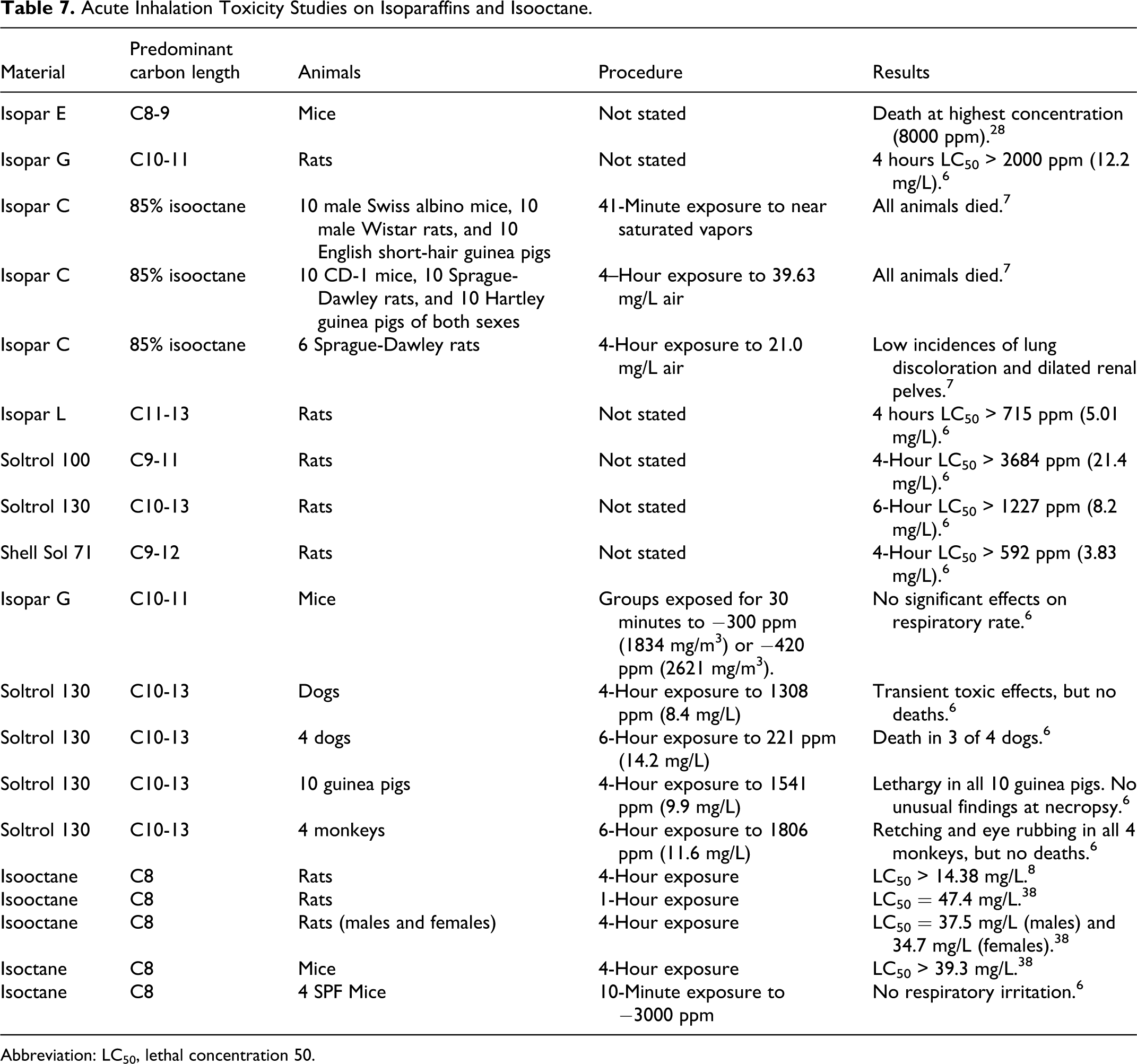
Abbreviation: LC50, lethal concentration 50.
Oral
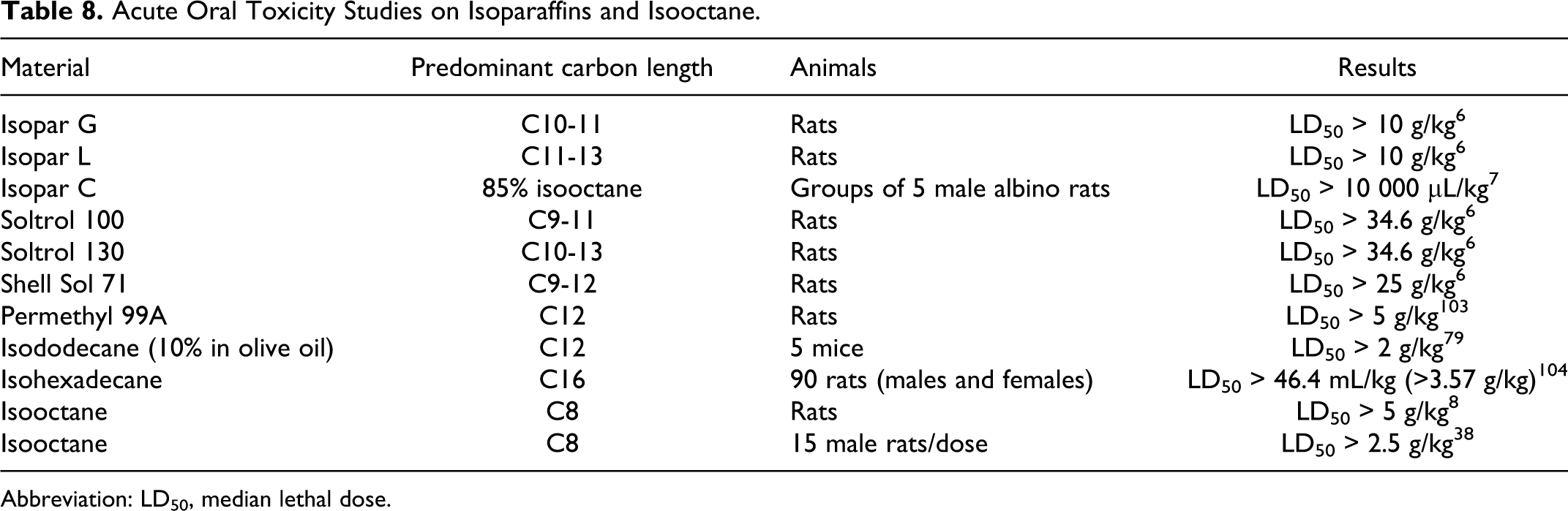
Isododecane had an oral median lethal dose (LD50) of >2 g/kg in mice and other isoparaffin mixtures had oral LD50 values >10 g/kg in rats as summarized in Table 8.
Acute Oral Toxicity Studies on Isoparaffins and Isooctane.
Abbreviation: LD50, median lethal dose.
Dermal
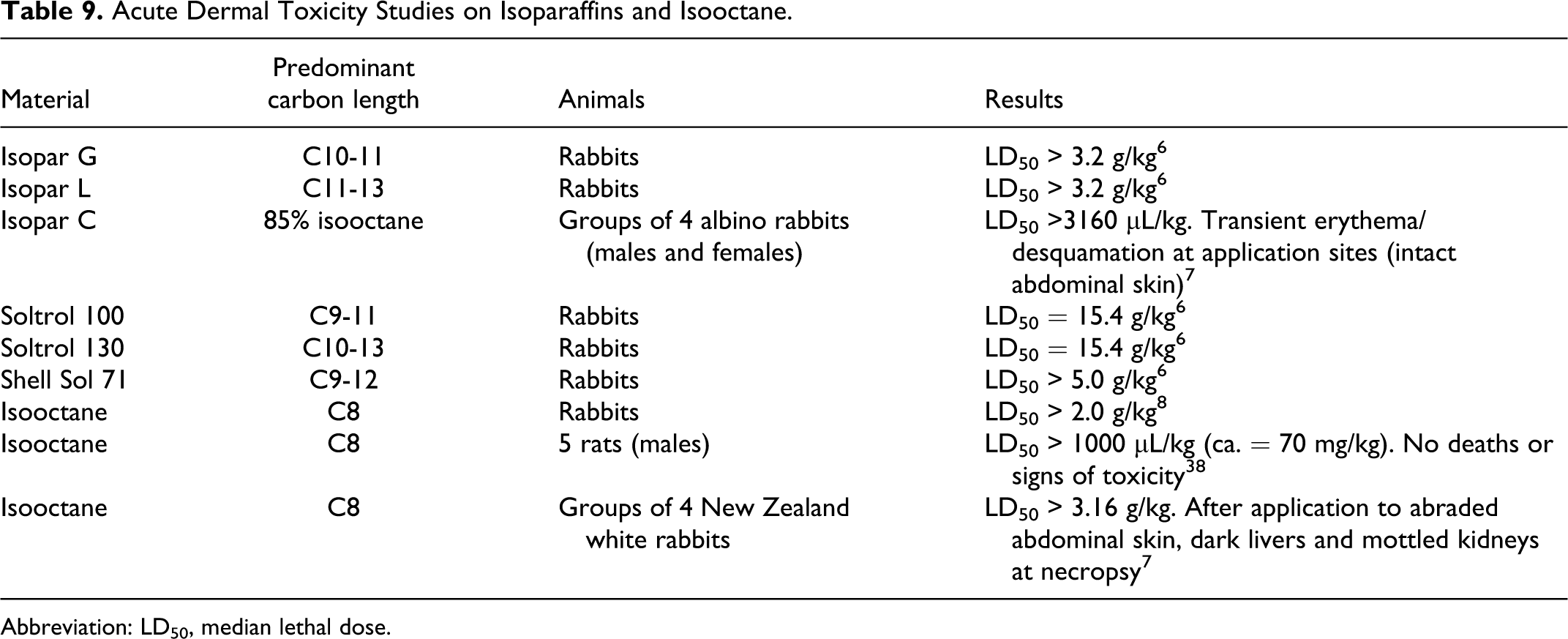
Isoparaffin mixtures of various chain lengths resulted in dermal LD50 values of >2 g/kg in rats as summarized in Table 9.
Acute Dermal Toxicity Studies on Isoparaffins and Isooctane.
Abbreviation: LD50, median lethal dose.
Intraperitoneal
In an acute intraperitoneal (ip) toxicity study, rats (15/dose) were dosed with isooctane (in vegetable oil), followed by a 14-day observation period. An LD50 of 2375 mg/kg was reported. 38
Repeated-Dose Toxicity
Inhalation
C10-11 isoparaffin
Groups of 50 male and 50 female Fischer 344 rats were exposed to 0, 300 (1.83 g/m3), or 900 ppm (5.48 g/m3) C10-11 isoparaffin 5 days per week (6 h/d) for a total of 8 weeks. 39 The ability of males to concentrate urine was decreased following 4 and 8 weeks of exposure; evidence of recovery was observed 4 weeks after the cessation of exposure. Additionally, compared to controls, the urinary excretion of glucose, protein, and epithelial cells in male rats was increased following 4 and 8 weeks of exposure. Decreased creatinine clearance was observed after 8 weeks. All of these changes, considered mild, returned to normal after 4 weeks of recovery. Microscopic changes in the kidneys of exposed males (both groups) included an increased incidence of regenerative tubular epithelia and tubules dilated at the corticomedullary junction, with proteinaceous debris in the tubules; structural recovery was not complete at the end of the 4-week recovery period. Neither functional nor microscopic renal changes were observed in female rats.
In another study, 3 groups of 15 Fischer 344 rats/sex were exposed (inhalation) to the same concentrations of C10-11 isoparaffin according to a similar 8-week test procedure. 40 An increased incidence of protein droplets was found in the cytoplasm of renal tubular cells of male rats. Other renal changes (both exposures) included foci of regenerative epithelium and tubular dilatation, with intratubular protein occurring between the inner and outer stripe of the medulla. After 20 and 40 days of exposure, focal loss of the brush border, with degeneration and sloughing of necrotic cells, was evident. Reversal of the exposure-related tubular changes was noted at the end of the 4-week recovery period. Renal changes were not observed in female rats.
C10-13 isoparaffin (Soltrol 130)
In a short-term study, 4 rhesus monkeys were exposed to Soltrol 130 (mean exposure concentration = 654 ppm [4.2 mg/L]) 3 days per week (6 h/d) for a total of 13 exposures. 6 None of the animals died. Clinical chemistry, urinalysis, and gross and microscopic findings were unremarkable. However, slight lymphocytopenias and neutrophilia were observed in the differential leukocyte count at both the midpoint and end of the study. There were also no remarkable changes in behavioral patterns, body weight, or food consumption.
C5-8 isoparaffin
Groups of Sprague-Dawley rats (12/sex per group) were exposed to light alkylate naphtha (concentrations up to 6646 ppm) 5 days per week (6 h/d) for 13 weeks. 41 Light alkylate naphtha is a substance described as containing >95% isoparaffins with carbon numbers predominantly in the C5-C8 range. Other than alpha 2u-globulin–mediated effects on the male rat kidney, the only effect reported was increased liver weight (males and females) in the 6646 ppm exposure group. There were no pathological changes in the liver, and the NOEL for subchronic toxicity was 2220 ppm. A neurological examination was also performed, and the NOEL for neurotoxicity was ≥6646 ppm.
C8-9 isoparaffin (Isopar E)
A study was conducted to evaluate the behavioral effects of Isopar E in groups of 8 adult male CFW albino mice. 28 Static exposure chambers were used for mice tested in the functional observational battery, one of the behavioral test methods used. Vapor exposures were conducted in 29 L cylindrical jars, and all vapor exposures were 30 minutes in duration. Three groups of mice were exposed to concentrations of 2000, 4000, and 6000 ppm, respectively. A fourth group was exposed to air only. Isopar E produced few effects over the range of concentrations tested, with most effects being observed at the highest test concentration (6000 ppm). Results of the functional observational battery conducted during the last 2 minutes of solvent exposure indicated that Isopar E induced decreases in central nervous system activity (ie, arousal), muscle tone/equilibrium (ie, gait, mobility, and landing foot splay), and sensorimotor reactivity (ie, approach response). Exposure to a higher concentration of Isopar E (8000 ppm) caused death due to convulsions.
C10-11 isoparaffin (Isopar G)
The subchronic inhalation toxicity of Isopar G was evaluated using groups of 35 male and 35 female Sprague-Dawley rats. 42 The groups were exposed (inhalation) to 0, 300 (1.91 g/m3), or 900 ppm (5.62 g/m3) Isopar G 5 days per week (6 h/d) for a total of 12 weeks. Decreased body weight was noted in male rats exposed to 300 and 900 ppm. A concentration-related increase in absolute and/or relative kidney weight was observed in male rats from 300 and 900 ppm exposure groups and in female rats exposed to 900 ppm.
Kidney tubule damage (male rats only) was described as mild, but a concentration- and duration-related increase in severity was noted. Also damage to the kidneys of male rats was essentially comparable at 8 and 12 weeks. An increase in both absolute and relative liver weight was observed in male and female rats exposed to 900 ppm; however, there was no microscopic evidence of liver damage. Furthermore, this change was considered representative of a nonspecific metabolic and/or physiological response to the uptake of hydrocarbon. Study results indicated that subchronic exposure to Isopar G at doses up to 900 ppm did not induce significant toxic effects, other than mild structural changes in the kidneys of male rats. 42
C10-12 isoparaffin (Shell Sol TD)
In another subchronic study, male and female rats were exposed (inhalation) to Shell Sol TD at the following concentrations: 359 (2.53), 737 (5.20), or 1444 ppm (10.19 g/m3). 6 A nonexposed control group was also included. Groups were exposed 5 days per week (6 h/d) for a total of 13 weeks. Neither deaths nor clinical signs occurred in both the low and intermediate dose groups. Rats in the high exposure group became slightly lethargic. Aspartate aminotransferase and alanine aminotransferase levels were decreased in females of all exposure groups, whereas alkaline phosphatase, potassium, chloride, and albumin were increased only in males of the high-exposure group. Changes in the levels of these enzymes, ions, and so on were considered minor and their toxicological significance remains unknown. A low-grade anemia, characterized by slight reductions in hemoglobin, packed cell volume, and total erythrocyte counts, was noted in males of all exposure groups.
Increased spleen weights were observed in male rats of the high-exposure group. Liver weights were increased in males of the high-exposure group and in males and females of the intermediate- and high-exposure groups. However, in the absence of histological changes, these changes in organ weight were regarded as adaptation rather than as a toxic effect. Increased kidney weights were observed in males of all exposure groups, and these weight changes were accompanied by hyaline intracytoplasmic inclusions, an increased incidence of tubular degeneration and/or dilatation in the cortical tubules. Increased kidney weights were also observed in females of the high-exposure group, in the absence of any exposure-related kidney lesions. The low-grade anemia and mild renal degenerative changes were considered related to Shell Sol TD exposure and biologically significant. 6
Isooctane
Two groups of 70 Sprague-Dawley rats were exposed, by inhalation, to Isopar C (85% isooctane) at cumulative mean exposure concentrations of 385 and 1180 ppm, respectively. 7 A third group served as the untreated control and received sham air exposures. Each group consisted of 35 males and 35 females. The animals were exposed to the test substance 5 days per week (6 h/d) for 12 weeks. No treatment-related mortalities occurred in the study. Body weights for control and test animals were comparable and unremarkable. Some of the hematocrit, hemoglobin, and red cell values were significantly depressed, compared to control values; however, all values were within the biological limits. Of the clinical chemistry parameters evaluated, an elevated blood urea nitrogen level in the 385 ppm exposure group was the only finding that may have indicated a slight treatment-related response.
Compared to controls, an analysis of absolute and relative organ/body weight ratios indicated an increase in both absolute and relative mean kidney weights at 1180 ppm (males, week 8) and at both 385 and 1180 ppm (males, week 12). These changes in organ weight were considered treatment related. At microscopic examination, mild tubular injury at 8 and 12 weeks was observed in some of the male rats exposed to 1180 ppm. All other necropsy findings were considered unremarkable. 7
Wistar rats (number not given) were exposed to isooctane vapor (10.3 or 24.2 mg/L; 4 h/d) for 5 days. 38 None of the animals died during the 14-day observation period (LD50 > 24.2 mg/L). Signs of toxicity included sedation and impeded breathing. Rats (10 males, 10 females) were exposed to isooctane vapor (9.66 mg/L, 6 h/d) 5 days per week for 12 weeks. 38 An untreated control group was also included in this study protocol. There were no clinical signs or hematological/macroscopic findings. Increased relative kidney weights were noted; however, the results of liver and kidney function tests were unremarkable. Decreased body weight gain was noted in male rats only.
Isododecane
In a subchronic inhalation toxicity study, groups of rats (20 males, 20 females/group) were exposed to atmospheres containing 0, 200 (1.4 air), 600 (4.2), or 1800 ppm (123.6 g/m3) 5 days per week (6 h/d) for 13 weeks. 43 Growth retardation and a transient reduction in red blood cell counts were noted in males and females exposed to 1800 ppm isododecane. When compared to control rats, both the degree and incidence of inflammatory reactions in the respiratory tract appeared to have been lower for males in the 1800 ppm exposure group and for females in the 600 and 1800 ppm exposure groups. Increased plasma alkaline phosphatase activity was reported only for female rats at this level of exposure.
Relative kidney weights were increased in rats of both sexes exposed to 1800 ppm and in male rats exposed to 600 ppm. At necropsy, there appeared to have been an increased incidence of green kidneys following exposure to 1800 ppm. Microscopic examination confirmed a dose-related increase in the incidence of tubular nephrosis in male rats. Data relating to effects on the reproductive system are included in the Reproductive and Developmental Toxicity sectionlater in the report text. It was concluded that the no-adverse-effect level of isododecane in rats was <200 ppm. 43
Oral
Isooctane
According to the US Environmental Protection Agency (EPA), an oral reference dose (RfD) is an estimate of a daily oral exposure to the human population that is likely to be without appreciable risk of deleterious effects during a lifetime. 44
The EPA noted that a number of acute and short-term studies have been identified in the literature, but that these studies are limited, in that they were designed to only investigate end points specific to α2u-globulin–associated nephropathy in male rats. Detailed studies on isooctane (2,2,4-trimethylpentane) that identify sufficient dose–response and duration information for other end points are lacking. The available studies provided evidence that the kidney toxicity induced by isooctane in male rats is related to α2u-globulin accumulation in the proximal tubules (a response that is specific to male rats). EPA concluded that this end point is not appropriate for determining noncarcinogenic hazard. No other studies were considered suitable for the derivation of an RfD, and, therefore, an oral RfD for chronic oral exposure was not derived. 44
Nephrotoxicity
Isooctane
Renal function changes were evaluated in a study involving Fisher 344 rats (sex not stated). 12 Groups of 4 rats were dosed orally with isooctane (in corn oil) at a dose of 0.1 or 0.5 g/kg per d for 4 weeks (5 times/week). Groups of 3 rats served as controls (corn oil). A statistically significant decrease in the inulin clearance (marker for glomerular filtration rate [GFR]) was observed after 2 and 4 weeks of gavage with 0.5 g/kg per d. The decrease in GFR was more profound at 4 weeks than at 2 weeks. A significant increase in the urinary enzyme, N-acetyl-beta-glucosaminidase (at 2 and 4 weeks) was associated with this reduction in the GFR.
The nephrotoxicity of isooctane was evaluated using groups of 8 male Fischer-344 rats. 7 The undiluted test substance was administered by oral gavage (dose volume = 1.0 mL/kg) to the 5 test groups on consecutive days as follows: group 1 (1 day), group 2 (3 days), group 3 (7 days), group 4 (14 days), and group 5 (21 days). Similarly, control groups were dosed with distilled water on consecutive days. A 90-min pulse label (ip) with 3 H-thymidine, followed by extraction of DNA from the liver and kidney was used to determine new DNA synthesis. Compared to controls, kidney-to-body weight ratios were significantly greater in treatment groups at 3, 14, and 21 days of dosing. By day 21, kidney weight ratios were 29% greater in treatment groups. Liver-to-body-weight ratios were significantly higher at 3, 7, 14, and 21 days; ratios were 61% higher by day 21. Both the liver and kidney showed an increase in relative organ weight and a concomitant decrease in DNA content, with the liver showing the greater effect. It was noted that much of the decreased DNA content could be accounted for by the relative increase in organ weight.
Daily dosing with isooctane resulted in stimulation of DNA turnover in the kidney. This effect was maximal ranging from 7 to 14 days of dosing, when 3 H-thymidine incorporation into DNA was 4-fold greater when compared to controls. On the other hand, the liver only showed stimulation of DNA turnover following 1 day of dosing, and subsequent dosing did not produce a significant effect. It was noted that these results are consistent with the findings that the kidney is the target organ for isooctane and compounds in this class. 7
Groups of 5 male Fischer 344 rats received oral doses of isooctane (50-500 mg/kg) by gavage for 21 days. 45 The animals were injected ip with [CH3- 3 H]-thymidine on day 22, and sites of renal cell proliferation induced by isooctane were localized and quantitated using histoautoradiographic analysis. Light microscopic lesions in the proximal convoluted tubule consisted of protein droplet and crystalloid body accumulation, degeneration, and necrosis. These renal lesions were not dose related, but a finding of tubular dilation of the thin segments with granular cell debris was dose related.
Isooctane induced a nondose-related, 5- to 6-fold increase in the labeling index of the same proximal convoluted tubule portions (P2 segment) that contained severe crystalloid body accumulation, degeneration, and necrosis. Less pronounced, but statistically significant (P ≤ .05), increases in cell proliferation were also observed in other nephron segments, indicative of a generalized regenerative response of the kidney to isooctane. It was noted that the cytotoxic and regenerative effects of oral dosing with isooctane suggest that similar mechanisms may be involved in the induction of kidney tumors in male rats exposed (chronic inhalation) to unleaded gasoline. 45 The observed isooctane-induced increase in cell proliferation in nephron segments is also mentioned briefly in the Carcinogenicity section later in the report text.
The EPA has made an effort to derive a reference concentration for isooctane chronic inhalation exposure (RfC). 44 The RfC (mg/m3), analogous to the RfD, takes into consideration toxic effects for both the respiratory system (portal of entry) and the effects peripheral to the respiratory system (extrarespiratory effects). The EPA noted that α2u-globulin–associated nephropathy was observed in a repeated-dose inhalation study on isooctane, providing evidence that the kidney toxicity induced by this chemical is related to α2u-globulin accumulation in the proximal tubules. The EPA concluded that this end point is not appropriate for determining noncarcinogenic hazard. No other studies were considered suitable for the derivation of an RfD, and, therefore an inhalation RfC was not derived.
Nephrotoxicity/Hepatotoxicity
Isooctane
The potential for isooctane-induced nephrotoxicity or hepatotoxicity was evaluated using groups of 6 male Wistar albino rats. 46 The test substance (in corn oil [2:1]) was administered by gavage at a single daily dose of 2 mL/kg. Control rats received a similar volume of corn oil alone. After 2 days of treatment, all test rats had signs of toxicity and had lost a considerable amount of weight. Thus, 6 control animals and all treated animals were killed by the third day. Macroscopic examination of the kidneys revealed no visible lesions; however, white patches (slightly raised) on the liver were found in 2 rats.
Centrilobular and confluent necrosis, hydropic degeneration, and vaculolation of hepatocytes were noted at microscopic examination. Analysis of plasma alkaline phosphatase and aspartate transaminase activity revealed increases that are consistent with liver damage. Microscopic examination of the kidneys revealed eosinophilic hyaline droplet accumulation in cells of the tubules and tubular dilation. Analysis of urinary N-acetyl-B-glucosaminidase and alkaline phosphatase activity showed increases that are consistent with renal toxicity. Based on the results of this study, the authors noted that it would appear that isooctane possess hepatotoxic as well as nephrotoxic properties. 46
Nephrotoxicity Mode of Action in Rats
While male rat nephrotoxicity observed after exposure to isoparaffins has been attributed to reversible binding of the hydrocarbon to α2u-globulin, this mechanism of action is not relevant in humans.
Subchronic or chronic inhalation exposure to C10-12 isoparaffin at a concentration of 6.5 mg/L caused both functional and morphological renal changes in normal male Sprague-Dawley rats, but not female or castrated male rats of the same strain. 47 Male rat nephrotoxicity has been attributed to reversible binding of hydrocarbon to α2u-globulin, which is not synthesized in humans. 48 Based on the measurement of several biochemical indicators of nephrotoxicity, isooctane (single oral dose, 12 or 24 mmol/kg) in corn oil was not found to impair renal proximal tubular function in male-specific pathogen free rats of the Alderley park strain (Alpk/AP). There was a strong association between the presence of renal hyaline droplets and the occurrence of α2u-globulin at these doses; however, the toxicological significance of increases in renal hyaline droplet formation was not established. 49 Histopathological changes in the kidney were not observed, and neither hyaline droplets nor α2u-globulin were detected in a study involving NCI-Black-Reiter male rats receiving isooctane (in corn oil) at daily oral doses of 500 mg/kg. The NCI-Black-Reiter rat is the only strain of male rat that is known not to synthesize the hepatic form of the low-molecular-weight protein α2u-globulin. In the absence of this protein, isooctane did not induce kidney injury, and these data provide further support for the role of α2u-globulin in nephrotoxicity. 50
Ocular Irritation
Isooctane
In a chemical safety report that was prepared in accordance with the REACH regulation, isooctane was not irritating to the eyes of rabbits. 51 This finding is consistent with ocular irritation study results for other isoparaffins included below.
Isohexadecane and isododecane
The EpiOcular human cell construct (EOT) was used to assess the ocular irritation potential of a tan-enhancing spray containing 42% isohexadecane. The 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazoliumbromide (MTT) conversion assay was used to assess cellular metabolism by EOT following product exposure. This assay measures the nicotinamide adenine dinucleotide phosphate-oxidase–dependent microsomal enzyme reduction of MTT, and, to a lesser extent, the succinate dehydrogenase reduction of MTT, to a blue formazan precipitate. The duration of exposure resulting in a 50% decrease in MTT conversion (ET50) in product-treated EOTs, relative to controls, was determined. An ET50 of 698.25 min (no/minimal irritation) was reported for the tan-enhancing spray containing 42% isohexadecane. 52
In the in vitro hens’s egg test, EXP-SR5 (contains 55.5% isohexadecane, 35% isododecane), 53 permethyl 216C (contains 40% isohexadecane), 54 permethyl 284C (contains 20% isododecane), 55 and permethyl 296C (contains 50% isododecane), 56 were classified as negative for ocular irritation potential. Results for permethyl 222C (material containing 40% isoeicosane) indicated practically no irritation to slight ocular irritation. 57 In this assay, the chorioallantoic membrane of the chick embryo responds to injury with a complete inflammatory reaction, comparable to that induced in the rabbit eye test.
In a chemical safety report that was prepared in accordance with the REACH regulation, isohexadecane was nonirritating to the eyes of rabbits. 58 This finding is consistent with ocular irritation study results for other isoparaffins included in this section. Data on hydrocarbons, C10-C12, isoalkanes, <2% aromatics in another chemical safety report were among the data used to evaluate the ocular irritation potential of isododecane, and the results were negative in rabbits. 37
Isoeicosane
In a chemical safety report that was prepared in accordance with the REACH regulation, data on hydrocarbons, C14-C18, n-alkanes, isoalkanes, cyclics, and ≤2% aromatics were among the data used to evaluate the ocular irritation potential of isoeicosane, and results were negative in rabbits. 59
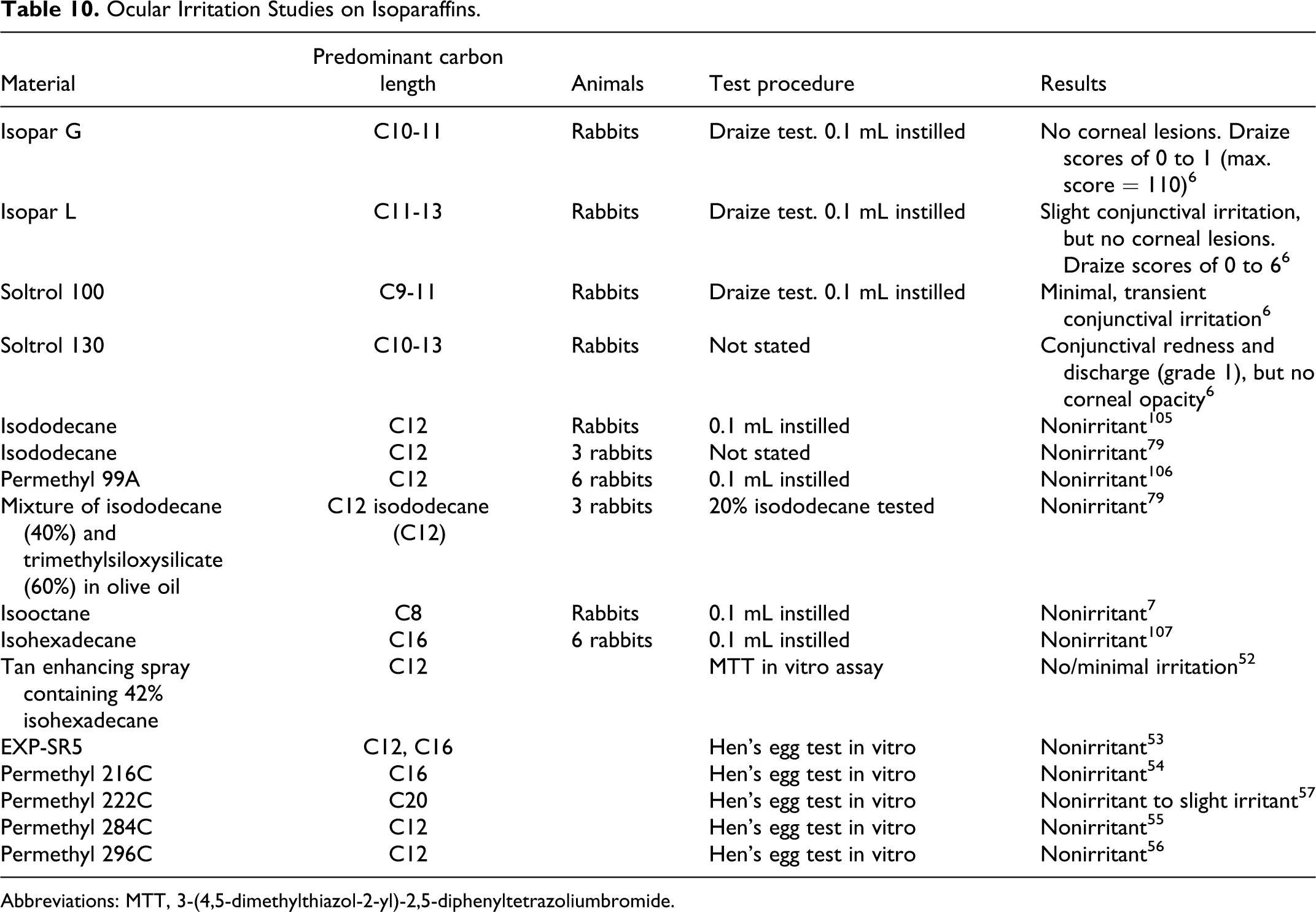
Ocular irritation study results are also summarized in Table 10.
Ocular Irritation Studies on Isoparaffins.
Abbreviations: MTT, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide.
Skin Irritation
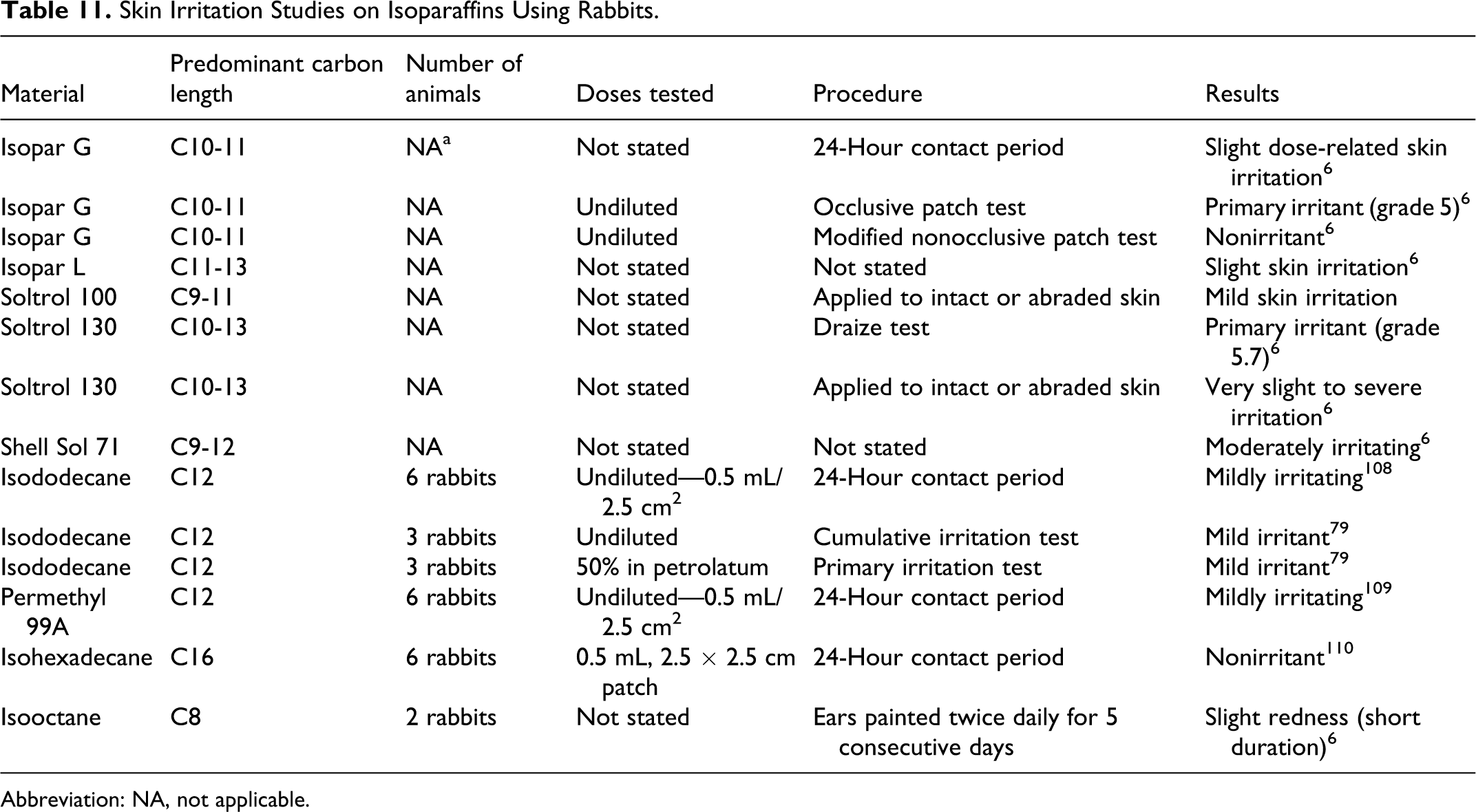
Isoparaffins are generally slightly to moderately irritating to skin. Summaries of skin irritation studies are included in Table 11. In a chemical safety report that was prepared in accordance with the REACH regulation, isooctane was irritating to the skin of rabbits. 51 This finding is consistent with skin irritation test results for other isoparaffins included in this section. In a chemical safety report that was prepared in accordance with the REACH regulation, data on hydrocarbons, C16-C20, aliphatics, and ≤2% aromatics were among the data used to evaluate the skin irritation potential of isoeicosane, and results were negative in rabbits. 59 Similarly data on hydrocarbons, C10-C12, isoalkanes, and <2% aromatics in another chemical safety report were among the data used to evaluate the skin irritation potential of isododecane, and results were negative in rabbits. 37
Skin Irritation Studies on Isoparaffins Using Rabbits.
Abbreviation: NA, not applicable.
Comedogenicity
Isododecane
Isododecane was applied undiluted to the ear of each of 3 New Zealand White rabbits for 3 consecutive weeks (5 days/week). The right ear served as the untreated control. At microscopic examination, hyperkeratosis was observed on the treated and control ear of one rabbit and on the treated ear of a second rabbit. There was no evidence of comedone formation on treated or control ears of rabbits, and isododecane was considered noncomedogenic. 60 Permethyl 99A (isododecane) was also classified as noncomedogenic when tested according to the same procedure. 61
Isohexadecane
Permethyl 101A (isohexadecane) was classified as noncomedogenic in rabbits when evaluated according to the preceding test procedure. 62
Skin Sensitization
C11-13 isoparaffin (Isopar L)
Reportedly, in a guinea pig sensitization test, Isopar L was classified as a nonsensitizer. 6 These data were referenced as unpublished data from Exxon Cooporation and study details were not included.
Isooctane
In a chemical safety report that was prepared in accordance with the REACH regulation, data on hydrocarbons, C7-C9, n-alkanes, isoalkanes, and cyclic were used to evaluate the skin sensitization potential of isooctane, and results were negative. 51
Isododecane
In another chemical safety report that was prepared in accordance with the REACH regulation, data on hydrocarbons, C10-C12, isoalkanes, <2% aromatics and hydrocarbons, C10-C13, isoalkanes, and <2% aromatics were among the data used to evaluate the skin sensitization potential of isododecane, and results were negative in guinea pigs. 37
Isoeicosane
Similarly, in a chemical safety report that was prepared in accordance with the REACH regulation, guinea pig maximization test data on C10-C12 isoalkanes, <2% aromatics were among the data used to evaluate the skin sensitization potential of isoeicosane, and results were negative in guinea pigs. 59
Reproductive and Developmental Toxicity
Isopar G (C10-11 Isoparaffin)
Reportedly, in a reproductive toxicity study, mated Sprague Dawley rats were exposed (inhalation, 6 h/d) to 0, 300, or 900 ppm Isopar G on days 6 to 15 of gestation. 6 The dams were killed on day 21 of gestation, and fetuses were examined for external, visceral, and skeletal malformations. Compared to controls, there were no changes in the following parameters: resorptions, fetal size, sex distribution, and fetal alterations. Isopar G was neither fetotoxic nor teratogenic to rats at concentrations up to 900 ppm. These data were referenced as unpublished data from Exxon Company, and the number of animals per dose group was not included.
Isooctane
The embryotoxic and/or teratogenic potential of Isopar C (85% isooctane) was evaluated using groups of 20 mated Sprague-Dawley rats. 7 Two groups were exposed to the test substance at concentrations of 400 and 1200 ppm, respectively, on days 6 to 15 of gestation. A negative control (air exposed) group and a positive control (acetylsalicylic acid orally, 400 mg/kg per d) group were included. Female rats were killed on day 21 of gestation, and fetuses were evaluated for external, soft-tissue, and skeletal malformations.
Compared to controls, rats exposed to 400 or 1200 ppm Isopar C had a significantly higher implantation efficiency. However, this finding was not indicative of a treatment-related adverse effect. Also, in these 2 groups, there were no treatment-related effects on the following: uterine implantation data, fetal size or sex distribution data, or fetal external, soft-tissue, or skeletal examination data. The incidence of fetuses with ossification variations was significantly increased in the 1200 ppm exposure group. However, the types and incidences of ossification variations were generally comparable to observations in the control group. It was concluded that Isopar C was neither embryotoxic nor teratogenic in Sprague-Dawley rats exposed at concentrations of 400 and 1200 ppm. The incidence of fetal malformations was increased in the positive control group. 7
According to a chemical safety report that was prepared in accordance with the REACH regulation, no reproductive effects are expected after exposure to isooctane. 51 This expectation is based on negative results for hexane, a structurally related substance, in reproductive and developmental toxicity studies.
Isododecane
Results from a subchronic inhalation toxicity study on isododecane, summarized earlier in the report text, indicated increased relative weights of the gonads in male and female rats exposed (inhalation) to 1800 ppm isododecane for 13 weeks. 43 The test protocol and other study results are included in the Subchronic Inhalation Toxicity section.
In a chemical safety report that was prepared in accordance with the REACH regulation, data on hydrocarbons, C9-C12, n-alkanes, isoalkanes, cyclic, and aromatics (2 to 25%) were among the data used to evaluate the reproductive toxicity of isododecane in rats and data on hydrocarbons, C9-C11, isoalkanes, cyclic, and <2% aromatics were among the data used to evaluate the developmental toxicity of isododecane in rats. 37 Neither reproductive nor developmental toxic effects were observed.
Isohexadecane and Isoeicosane
Similarly, in chemical safety reports that were prepared in accordance with the REACH regulation, data on hydrocarbons, C16-C20, n-alkanes, isoalkanes, cyclic, and <2% aromatics were among the data used to evaluate the developmental toxicity of isoeicosane.58,59 The NOAEL was >1000 mg/kg per d, and there were no signs of maternal toxicity or treatment-related adverse effects on fetal development in rats.
Genotoxicity
The following text includes additional details from the mutagenicity study on Isopar C (85% isooctane) summarized in Table 12. In this study, the mutagenicity of Isopar C following inhalation exposure was evaluated in the dominant lethal test using groups of 10 male Sprague-Dawley rats, which were subsequently mated with females. 7 Two groups were exposed (inhalation) to the test substance at concentrations of 400 and 1200 ppm, respectively. Male rats were exposed 5 days per week (6 h/d) for 8 consecutive weeks. Treatment was followed by a 2-week mating period. A negative control group (air exposed) and a positive control group (triethylenemelamine [TEM], 0.5 mg/kg ip) were also included in the study. Mean body weights were comparable between negative control and test groups.
Genotoxicity of Isoparaffins and Isooctane.
Abbreviation: UDS, unscheduled DNA synthesis.
The mating of females with TEM-positive control males resulted in fewer implants and lower implantation efficiency values (indicative of preimplantation loss), compared to females mated with negative control males. There were no treatment-related effects on mortality, in-life physical observations, or necropsy observations following exposure to 400 or 1200 ppm. Pregnancy rates, implantation data, and implantation efficiency values and fetal death data for females mated to males exposed to Isopar C were comparable to data for females mated to negative control males. Microscopic evaluation of the following tissues from 5 randomly selected males did not reveal any treatment-related effects: testes, seminal vesicles, epididymides, and prostate. It was concluded that Isopar C was not mutagenic in the dominant-lethal test at doses of 400 and 1200 ppm. 7
Isododecane
In a chemical safety report that was prepared in accordance with the REACH regulation, mammalian genotoxicity data on isododecane and bacterial and mammalian genotoxicity data on hydrocarbons, C10-C12 isoalkanes, and < 2% aromatics were used to evaluate the genotoxicity potential of isododecane, and results were negative. 37
Carcinogenicity
Isooctane
Petrolatum (15% in isooctane [concentration not stated]) was applied to the skin in groups of 30 male and 30 female Swiss mice. 63 Applications (3 drops, ∼ 60 µL/application) were made to dorsal skin twice weekly during lifetime treatment. Survival of the mice was good and no significant tumor incidence was found.
Findings relating to isooctane-induced increases in cell proliferation in rat nephron segments are included in the section on Nephrotoxicity/Cell Proliferation earlier in the report text. 45
A study was performed to better characterize the pathogenesis of α2u-globulin nephropathy. 64 Groups of 3 F344 rats per sex were exposed (inhalation) to 10, 70, or 300 ppm unleaded gasoline or 50 ppm isooctane from 3 to 50 weeks (6 h/d, 5 days/week). Cell proliferation was quantitated within 3 proximal tubule segments of the kidney (P1, P2, and P3). Immunohistochemical staining of α2u-globulin was performed on kidney sections. To determine whether accumulated α2u-globulin was concentration related, the ranking of slides based on the severity and extent of accumulation of crystalloid α2u-globulin droplets and single-cell necrosis of the affected P2 tubule was performed. Results indicated significant increases in rank above age-matched controls in kidneys from male rats exposed to 300 ppm unleaded gasoline or 50 ppm isooctane at each exposure interval. Mild but detectable increases in α2u-globulin staining were observed in groups exposed to 10 or 70 ppm unleaded gasoline.
The largest increases in labeling indices (above controls) occurred in the P2 segment, that is, 6- to 11-fold increases in labeling indices at 3, 10, and 22 weeks of exposure to 300 ppm unleaded gasoline or 50 ppm isooctane. These changes were indicative of dose-related increases in cell turnover, and this proliferative response closely paralleled the extent and severity of detectable α2u-globulin in the P2 segment. Neither α2u-globulin nor cytotoxicity was evident in cells of the P1 or P3 segment; however, in the P3 segment, cell proliferation was increased (up to 8-fold) for up to 22 weeks of exposure. Compared to controls, increased numbers of proximal tubules affected by chronic progressive nephrosis were found in males exposed to unleaded gasoline or isooctane for 22 or 48 weeks. These lesions contained epithelial cells that were highly proliferative. Neither α2u-globulin nephropathy nor increases in P2 or P3 cell turnover were observed in control or treated female rats. The authors noted that the results of this study suggest that chronic cell proliferation associated with α2u-globulin nephropathy and chronic progressive nephrosis in male rats exposed to unleaded gasoline or its isoparaffinic components, such as isooctane, may be responsible for nephrocarcinogenic effects of unleaded gasoline in male rats. 64
The majority of the reported studies contribute information specifically related to the histopathological sequence of α2u-globulin-associated nephrotoxicity. Thus, these studies do not examine any other tissue/organ except the kidney. In comparing the tumor-promoting capability between isooctane and unleaded gasoline (UG, a mixture), Short et al 65 showed that both agents had promoting potential in male, but not female rats. However, the results were not sufficiently descriptive to ascribe the portion of the promoting potential of UG that could be attributable to isooctane. The few studies available on its genotoxic potential were negative, as isooctane does not increase mutations at the TK locus in a study by Richardson et al, 66 induce DNA double-strand breaks in a study by McLaren et al, 67 or stimulate unscheduled DNA synthesis in a study by Loury et al. 68
The US EPA concluded that there are no available chronic bioassays or epidemiological studies in humans that assess the carcinogenicity of isooctane, and that this overall lack of information represented a data gap and did not allow for a quantitative assessment of the carcinogenicity of isooctane.44,69
Isododecane
In a chemical safety report that was prepared in accordance with the REACH regulation, data on the following chemicals were among the data used to evaluate the carcinogenicity of isododecane 37 : Inhalation exposure to hydrocarbons, C10-C12 isoalkanes, and <2% aromatics (Stoddard solvent IIC) caused neoplastic effects in male rats and female mice. Dermal exposure to Stoddard solvent and hydrocarbons, C10-C13, n-alkanes, and <2% aromatics caused neoplastic effects in male mice. Regarding the inhalation study, it was noted that there was no evidence of carcinogenic activity of Stoddard solvent IIC in female F344/N rats or in B6C3F1 male mice exposed to 2200 mg/m3, and that the National Toxicology Program (NTP) concluded that there was equivocal evidence of carcinogenic activity of Stoddard solvent IIC in female B6C3F1 mice based on increased incidences of hepatocellular adenoma. It was also noted that the incidences of benign pheocoromocytoma in 550 and 1100 mg/m3 male rats and benign or malignant pheochromocytoma exceeded the historical chamber control ranges, suggesting that exposure to Stoddard solvent IIC caused the increased incidences of these adrenal medulla neoplasms. The incidence of malignant pheochromocytoma was described as one malignant tumor in control animals and 2 malignant tumors in 1100 mg/m3 male rats. However, it was stated that the relevance of pheochromocytoma in humans is equivocal at best. These carcinogenicity data could be considered along with other available data in evaluating the carcinogenicity of isoparaffins.
Isohexadecane and Isoeicosane
According to chemical safety reports on isohexadecane and isoeicosane that were prepared in accordance with the REACH regulation, hydrocarbons, C14-C20 aliphatic, and <2% aromatics are metabolized and excreted rapidly, are not genotoxic, and there is no evidence from the repeat-dose studies that they are able to induce hyperplasia or preneoplastic lesions.58,59 Thus, it is highly unlikely that these chemicals would be carcinogenic. These data were used to evaluate the carcinogenicity of isohexadecane and isoeicosane.
Tumor Promotion
Isooctane
An initiation-promotion study was performed using 30 Fischer 344 rats/sex per experiment. Rats were given N-ethyl-N-hydroxyethylnitrosamine (170 ppm) in drinking water for 2 weeks, followed by a 2-week nontreatment period. 65 The rats were then exposed to isooctane via inhalation (50 ppm [∼ 2.4 mg/L],6 h/d) 5 days per week for 24 or 59 to 60 weeks. Decreased kidney weights were noted in male rats only, and incidences of atypical cell foci (ACF) and renal cell tumors (RCTs) were 79% and 14%, respectively, in these animals. Incidences in control rats were 35% (ACF) and 4% (RCT), and the differences between test and control rats were not found to be statistically significant. Female rats had normal kidney weights, and there were no increases in ACF or RCT.
Cell Proliferation
HeLa cell (S3 cell line) suspensions were exposed to isooctane at concentrations ranging from 0.1% to 7.5% and examined for morphological changes associated with toxicity. 70 Cultures were incubated for 2 to 3 days at a temperature of 37°C. The overall physiologic state of the cells after exposure was quantified, in terms of the intracellular adenosine triphosphate (ATP) concentration, using a chemiluminescence ATP assay. There was no obvious effect on cell proliferation, for example, the absence of mitotic figures was not noted after day 1 of exposure. Additionally, there was no evidence of differences in cell shape, granularity around the nucleus, or visible damage to the cell membrane. Other results indicated that exposure to isooctane produced little change in the intracellular ATP concentration.
Clinical Assessment of Safety
C8-12 Isoparaffin (Shell Sol TD)
A total of 7 participants were exposed (inhalation) to Shell Sol TD at a concentration of 100 ppm for 5 days (6 h/d). The mean concentration in the fat was 41.1 mg/kg (measured value), and the estimated mean half-life in fat was 7 days. At steady state, the maximum brain isoparaffin concentration was estimated to be 11 mg/kg, with an estimated maximum half-life of 18 to 19 hours. 71 Following exposure of 8 participants to Shell Sol TD at a concentration of 100 ppm (0.6 g/m3) for 3 hours, the maximum steady-state concentration was calculated (using mathematical modeling) to be 55 mg/kg for fat and 5 mg/kg for the brain. 72
C8-12 Isoparaffin (Shell Sol TS)
None of the following symptoms associated with solvent exposure was observed in 12 human participants, following a single, 6 hours of inhalation exposure to 100 ppm Shell Sol TS: headache, dizziness, feeling of inebriation, visual disturbances, tremor, muscular weakness, impairment of coordination, or paresthesia. A mean blood concentration of 2.3 mg/L (14 nmol/L) was reported at the end of exposure. There were no changes in blood chemistry, and results of urinalyses indicated that exposure had no effect on the 2 urine variables albumin and β2-microglobulin. 73
A total of 7 participants were exposed (inhalation) to Shell Sol TS at a concentration of 103 ppm (0.61 g/m3) for 5 days (6 h/d), and 5 participants served as nonexposed controls. Compared to controls, significant differences in creatine kinase and follicle-stimulating hormone were noted following exposure. It was noted that there was marked intraindividual and interindividual variability in the serum concentrations of these parameters. There was no evidence of changes in plasma immunoglobulin or orosomucoid. 74
Ocular Irritation
C11-12 isoparaffin
The ocular acceptability of a mascara containing 48.28% C11-12 isoparaffin was evaluated using 48 female participants. The product was applied to the eyelashes twice daily (morning and early afternoon) for 4 weeks and was considered well tolerated. Nine participants presented with the following subjective signs, described as being of slight intensity and of very short to long (1 day) duration: general sensation of irritation, ocular stinging, palpebral stinging, sensation of foreign body, discomfort, and sensation of dryness and tightness). One participant presented with subjective signs (ocular stinging) that were of slight intensity and very short duration. These signs were frequent and said to have been probably due to mascara. 75
Isododecane
In another ocular acceptability study, a mascara containing 63.7% isododecane was evaluated using 10 female participants. The product was applied to the eye lashes once daily for 5 days and there was no evidence of ocular irritation. 76 When the test procedure was repeated in another study (same product, 50 females) over a 4-week period, an ocular irritation rate of 0.04% was reported. 77
Skin Irritation
C10-11 isoparaffin (Isopar G);
C11-13 isoparaffin (Isopar L);
C12-15 isoparaffin (Isopar M).
Under closed or semiocclusive conditions where evaporation cannot freely occur, C10-11, C11-13, and C12-15 isoparaffins can produce defatting of the skin and irritation. 6
C11-12 isoparaffin
The skin irritation potential of a hair shine containing 41.25% C11-12 isoparaffin (as supplied) was evaluated using 20 participants. The test substance was applied (3 repeated applications) under a semiocclusive patch for 24 hours. The application area was not stated. It was concluded that the product had very good skin compatibility. Whether or not the hair shine product caused skin irritation was not stated in the English translation (summary) of this study. 78
Isododecane
Results were negative for isododecane (contained ∼85% 2,2,4,6,6-pentamethylheptane) in an open patch test involving 20 participants. However, skin irritation was observed in a study in which 20 participants were patch tested (closed patches) with the following concentrations of isododecane (contains >98% 2,2,4,6,6-pentamethylheptane) in petrolatum: 10% (2 participants), 20% (3 participants), and 50% (6 participants). A mixture containing 40% isododecane and 60% trimethylsiloxysilicate caused skin irritation in 2 of 19 participants patch tested (closed patches) at a concentration of 40% in petrolatum (effective isododecane concentration ∼16%). 79
Isohexadecane
The in-use safety of a tan-enhancing spray containing 42% isohexadecane, following 2 consecutive weeks of use (once daily), was evaluated using 30 volunteers (males and females). Safety was determined by clinically evaluating changes in dermatological data (dryness and redness) and changes in opthalmological data (eg, eyelids and margins, conjunctivae, and corneas). Product-related adverse reactions (4 participants total) included mild itching of forehead (1 participant), burning sensation in left eye (1 participant), mild tingling and itching of arms, chest, shoulders, and face (1 participant), and mild tingling of cheeks. The changes observed in participants tested were reported to be not clinically significant. 80
In a chemical safety report that was prepared in accordance with the REACH regulation, the results of a skin irritation test on isohexadecane involving 15 volunteers were negative. 58 This finding is consistent with the results of skin irritation tests on other isoparaffins included in this section.
Isoeicosane
In a chemical safety report that was prepared in accordance with the REACH regulation, the results of a skin irritation test on isoeicosane involving 15 volunteers were negative. 59 This finding is consistent with the results of skin irritation tests on other isoparaffins included in this section.
Predictive Skin Irritation and Sensitization
In most of the studies summarized in this section, an RIPT procedure involving 24-hour patch applications (induction and challenge phases) to the back was used. The single application procedure in the in-use safety test also involved a 24-hour application period (challenge site not stated). If provided, the amount of test material applied is expressed in grams or milliliters, and, in most cases, patch dimensions (in inches or centimeters) are included.
C11-12 isoparaffin
A mascara containing 48.28% C11-12 isoparaffin was applied (0.2 g under 2 cm × 2 cm semiocclusive patches) to a total of 107 male and female participants. Transient erythema and edema (slight reactions) were observed in 1 participant during induction, and the product was classified as a nonirritant (mean irritation index <0.25) and nonsensitizer. 81
Isododecane
The in-use safety of a hair oil mist spray containing 90.3% isododecane, following 6 consecutive weeks of use, was evaluated using 69 volunteers (males and females). The ability of the product to induce contact allergy was determined by conducting a challenge procedure (2 cm × 2 cm site) approximately 10 to 14 days after the use of the product was discontinued. A total of 69 participants completed the in-use phase and 65 participants completed the challenge phase. Safety was determined by evaluating dryness, redness, and stinging of the scalp and challenge phase data. The product was classified as having very good tolerance, that is, no clinically meaningful changes in redness and dryness on the scalp. The skin reactivity observed during the challenge procedure was considered as neither evidence of allergenicity nor clinically meaningful irritation. 82
The skin irritation and sensitization potential of a lip primer containing 80.74% isododecane was evaluated using 108 healthy male and female participants. The lip primer did not demonstrate a potential for eliciting skin irritation or sensitization. 83 Results for an eye shadow containing 47.64% isododecane were also negative for skin irritation and sensitization in a study involving 104 healthy male and female participants, 84 and the same was true for an eyeliner containing 40.16% isododecane that was tested (2 cm × 2 cm patch) on 108 participants during induction and 100 participants from the same group during the challenge phase. 85 In another study, the skin irritation and sensitization potential of a mascara containing 63.7% isododecane was evaluated using 204 healthy participants (males and females; 2 cm × 2 cm patches). It was concluded that the mascara was nonirritant and nonsensitizer. 86
Patch applications of permethyl 296C (50% isododecane; 1′′ × 1′′ patches) were made to 52 healthy male and female participants. Results were not indicative of skin irritation or sensitization potential. 87 In another study, permethyl 284C (20% isododecane; 1′′ × 1′′ patches) was applied to 52 healthy male and female participants. Two participants had a moderate and barely perceptible reaction postchallenge. The barely perceptible reaction was considered clinically insignificant and the moderate reaction was associated with a reactive participant who should be prohibited from future patch testing. Results for permethyl 284C were not indicative of skin irritation or sensitization potential. 88
In a chemical safety report that was prepared in accordance with the REACH regulation, data on hydrocarbons, C10-C12, iasoalkanes, and <2% aromatics were among the data used to evaluate the skin irritation and sensitization potential of isododecane in human participants. 37 Results were positive for skin irritation and negative for skin sensitization.
Isohexadecane
The skin irritation and sensitization potential of permethyl 216C (40% isohexadecane; 0.1 mL on 1′′ × 1′′ patch) was evaluated using 52 healthy male and female participants, and results were negative. 89 There was also no evidence for skin irritation or sensitization in 100 male and female participants tested with an indoor tanning product containing 42% isohexadecane (0.1 mL/patch) 90 or in 102 healthy male and female participants tested with an eye makeup remover containing 20% isohexadecane according to the same procedure. 91 In another study, a skin cleanser containing 15% isohexadecane (under 48 hour patch [0.5 in 2 ]) did not induce skin irritation, skin fatiguing, or allergic eczematous contact dermatitis in 600 healthy male and female participants. 92
In a chemical safety report that was prepared in accordance with the REACH regulation, human skin sensitization test data on hydrocarbons, C14-C18, n-alkanes, isoalkanes, cyclic, and <2% aromatics were among the data used to evaluate the skin sensitization potential of isohexadecane, and results were negative. 58
Isododecane and isohexadecane
The skin irritation and sensitization potential of 20% EXP SR5 (contains 55.5% isohexadecane and 35% isododecane; 0.2 g on 1′′ × 1′′ patch) in petrolatum was evaluated using 54 healthy male and female participants. The effective concentrations tested were ∼11.1% isohexadecane and ∼7% isododecane. Neither skin irritation nor sensitization was observed. 93
Isoeicosane
Patch applications of permethyl 222C (40% isoeicosane; 1′′ × 1′′ patches) were made to 52 healthy male and female participants. Results were not indicative of skin irritation or sensitization potential. 94 The same conclusion was stated in another study in which 106 healthy male and female participants were tested with a lip balm containing 27.15% isoeicosane. 95
In a chemical safety report that was prepared in accordance with the REACH regulation, data on hydrocarbons, C14-C18, n-alkanes, isoalkanes, cyclic, and <2% aromatics were among the data used to evaluate the skin sensitization potential of isoeicosane in human participants, and results were negative. 59
Provocative Skin Sensitization
Isohexadecane
The skin sensitization potential of isohexadecane was evaluated using a classical repetitive open application patch test. Patch applications were made to the outer upper arm of participants with dermatitis and to the anterior forearm and upper back of healthy participants (controls). 96 Patch test reactions to isohexadecane (undiluted) were positive in 2 of 26 dermatitis patients and in 11 of 55 control participants; however, the difference between these 2 groups was not statistically significant (P > .05). Isohexadecane (10% in petrolatum) did not induce positive reactions in 19 dermatitis patients or in 56 control participants. The authors stated that, compared to control participants, the pattern of reactivity of isohexadecane in participants with dermatitis suggested that it acts unspecifically as an irritant when undiluted.
Skin Sensitization, Irritation, and Photosensitization
C10-11 isoparaffin (Isopar G)
C11-13 isoparaffin (Isopar L)
C12-15 isoparaffin (Isopar M)
Skin sensitization, phototoxicity, and photosensitization tests on Isopar G, Isopar L, and Isopar M were conducted using panels consisting of more than 100 participants. Each test material was patch tested (semiocclusive patches) at a concentration of 50% in petrolatum, and there was no evidence of skin sensitization, phototoxicity, or photosensitization. 6
Isododecane. In a chemical safety report that was prepared in accordance with the REACH regulation, data on hydrocarbons, C10-C12, iasoalkanes, and <2% aromatics were among the data used to evaluate the skin irritation and phototoxicity potential of isododecane in human participants, and results were negative. 37
Isohexadecane and isoeicosane. Similarly, in chemical safety reports that were prepared in accordance with the REACH regulation, data on hydrocarbons, C14-C19, iasoalkanes, cyclic, and <2% aromatics were among the data used to evaluate the skin irritation and phototoxicity potential of isohexadecane and isoeicosane in human participants, and the results were negative.58,59
Case Report
Isohexadecane
A 64-year-old female presented with a history of an eczematous rash after application of a commercially available sunscreen that contained isohexadecane. Patch test reactions to 10% isohexadecane were positive (+ reaction) at days 2 and 4. Negative results were reported for 20 control participants patch tested with isohexadecane. 97
Occupational Exposure
The use of solvent mixtures containing isoparaffins in the workplace has produced a low incidence of hypersensitization. 6
Reportedly, C10-11 and C11-13 isoparaffins have been used by Versatec (a Xerox company) and Xerox Medical Systems, collectively, for over 16 years. Out of more than 2000 employees, there were only 2 cases of health-related incidents (skin rash and hives) following skin contact. 6
Of the 74 male employees of a manufacturing facility, 63 developed dermatitis after exposure to the following 2 metalworking fluids: lubricant containing >80% C9-12 isoparaffin (lubricant 1) or >99.8% C10-14 isoparaffin (lubricant 2). 98 Irritation test (procedure not stated) results indicated that 22 of the 63 cases were due to lubricant 1 (PII = 2.1) and 32 were due to lubricant 2 (PII = 1.1); both were classified as irritants. Neither cumulative irritation nor sensitization tests were performed.
The American Conference of Governmental Industrial Hygienists set the occupational exposure limit for inhaled isooctane at 300 ppm, as time-weighted average. 99
Epidemiology
Aliphatic and other hydrocarbons
A study was designed to provide an estimate of the occupational risk of contracting cancers of the urinary tract. 100 The 25-year incidence of bladder cancer (BC) and renal cell carcinoma (RCC) in the entire Finnish workforce was compared in relation to occupation and occupational exposure to solvents and gasoline. The study cohort consisted of 1.6 million Finns (born in years 1906-1945) who participated in the national population census on December 31, 1970. All cancers, diagnosed from 1971 through 1995, in individuals born in years 1906 to 1945 were extracted from the nationwide Finnish Cancer Registry and sent to Statistics Finland and compared with the 1970 census files. Cancer risk estimates were adjusted for smoking and obesity.
Overall, there appeared to be a tendency for an elevated risk of BC in women exposed to solvents, but not among men. The relative risk estimates were above 1.2 in nearly all exposure categories studied. However, a statistically significant elevated risk of BC in women was associated with the following: a low level of aliphatic hydrocarbon solvents (1.40; 95% confidence interval (CI) = 0.85-2.32), a low level of chlorinated hydrocarbon solvents (1.43; 95% CI = 1.0-2.03), and a middle level of chlorinated hydrocarbon solvents (1.68; 95% CI = 1.15-2.45). The relative risks of BC in men and women occupationally exposed to gasoline (low level exposure) were men 1.0 (95% CI = 0.89-1.13) and women 1.55 (95% CI = 0.50-4.86). There was no significantly elevated risk of RCC in any exposure category for any solvent in either gender. It was concluded that these findings suggest that occupational exposure to certain solvents may have an impact on BC risk, but the risk of RCC does not appear to be altered by exposure to hydrocarbon solvents or gasoline. 100
Summary
The 24 isoparaffins reviewed in this safety assessment function mostly as solvents in cosmetics, and the following 15 are being used: C7-8 isoparaffin, C8-9 isoparaffin, C9-11 isoparaffin, C10-11 isoparaffin, C10-13 isoparaffin, C11-12 isoparaffin, C11-13 isoparaffin, C12-14 isoparaffin, C13-14 isoparaffin, C13-16 isoparaffin, C18-70 isoparaffin, isododecane, isoeicosane, isohexadecane, and isooctane. The results of a personal care products industry survey indicate that ingredient use concentrations have ranged from 0.0001% (C13-14 isoparaffin) to 90% (isododecane).
As a frame of reference, liquid gasoline is a complex mixture of petroleum chemicals that includes approximately 60% to 75% alkanes (paraffins) that comprise straight-chain hydrocarbons (C4-C12), and isoparaffins (branched-chain hydrocarbons) in approximately the same range of chain lengths. While some of the isoparaffins described as cosmetic ingredients are longer in chain length, most are in this range. 2,2,4-Trimethylpentane (isooctane) is used primarily in the alkylation step to derive high-octane gasoline fuels.
Following inhalation in rats, 14 C-isooctane was eliminated almost exclusively by the kidneys, and various urinary metabolites, 2,4,4-trimethyl-2-pentanol included, have been identified. Following oral dosing of 14 C-isooctane in rats, more than half of the administered dose was recovered in the urine and feces. Accumulation in the liver and kidneys was observed, with males retaining substantially greater amounts of radioactivity in the kidneys, compared to females. 2,4,4-Trimethyl-2-pentanol was the major metabolite detected in the male rat kidney but was absent from female rat kidney. However, female rats excreted more conjugates of this metabolite, compared to males. Following human inhalation exposure, the maximum steady-state concentration of isooctane in the brain and fat was calculated using mathematical modeling.
Following exposure of 8 participants to Shell Sol TD at a concentration of 100 ppm (0.6 g/m3) for 3 hours, the maximum steady-state concentration was calculated (using mathematical modeling) to be 55 mg/kg for fat and 5 mg/kg for the brain.
Following lifetime dermal application of petrolatum (15% in isooctane) to mice, no significant tumor incidence was found. A 2007 toxicological review on 2,2,4-trimethylpentane (isooctane), published by the EPA is available. The EPA has determined that there is inadequate information to assess carcinogenic potential of 2,2,4-trimethylpentane, having noted that no epidemiological studies in humans and no chronic bioassay studies are available that assess the carcinogenic effects of 2,2,4-trimethylpentane.
In many of the animals studies, involving mostly male rats, either oral or inhalation exposure to isooctane resulted in some degree of nephrotoxicity. According to some investigators, study results suggest that chronic cell proliferation associated with α2u-globulin nephropathy and chronic progressive nephrosis in male rats exposed to unleaded gasoline or its isoparaffinic components, such as isooctane, may be responsible for nephrocarcinogenic effects of unleaded gasoline. α2u-Globulin nephropathy associated with exposure to these chemicals is not relevant to man due to the absence of this protein. The isoparaffins were not found to be genotoxic in in vitro or in vivo assays and were neither embryotoxic/fetotoxic nor teratogenic in rats. The results of an initiation-promotion study involving rats were negative for isooctane.
The isoparaffins have produced slight ocular irritation and mild-to-severe skin irritation but were not comedogenic, in rabbits. Furthermore, skin sensitization was not induced in guinea pigs. Eye area cosmetic products containing isoparaffins were classified as well tolerated, in terms of ocular irritation potential, following application to the eye lashes of female participants. Also, these chemicals, alone or in product formulations, were not classified as irritants, sensitizers, phototoxic, or photosensitizers in human patch tests. However, it should be noted that isohexadecane (undiluted) induced a low incidence of skin irritation in patients, but this incidence was not significantly different from that of healthy controls. Isohexadecane (10% in petrolatum) did not induce any reactions in patients or healthy controls. Also, a low incidence of skin irritation was reported in a closed patch test, but results were negative in an open patch test involving human participants tested with isododecane.
Occupational exposure to isoparaffins has produced irritation and a low incidence of sensitization in the workplace. In an epidemiology study (occupational exposure), a statistically significant elevated risk of BC in women was associated with a low level of exposure to aliphatic hydrocarbon solvents. However, a significantly elevated risk of RCC was not associated with these exposures.
Discussion
The CIR Expert Panel noted that most of the available data related to oral or inhalation exposure to isoparaffins, but the dermal and ocular exposure data that were available, suggested mild ocular irritation, mild-to-severe irritation, no sensitization or photosensitization, and no phototoxicity.
No significant toxicity was identified in oral or inhalation exposure studies of the following end points: genotoxicity, reproductive and developmental toxicity, or carcinogenicity. Nephrotoxicity, however, was a concern. The Expert Panel noted the involvement of α2u-globulin in the mechanism for isoparaffin-induced nephrotoxicity/renal tubule cell proliferation in male rats of various strains in oral and inhalation exposure studies, but noted that nephrotoxic effects were not observed in one strain of rats, NCI-Black-Reiter, that does not have the α2u-globulin protein. Humans also lack this protein and, thus, the Panel agreed that findings associated with the α2u-globulin protein in male rats were not relevant to humans. This view was consistent with the US EPA position that it was not possible for the agency to derive an oral RfD for chronic oral exposure or a reference concentration for chronic inhalation exposure to isooctane because the available studies were limited, in that they were designed to only investigate the end points specific to α2u-globulin-associated nephropathy. The EPA also concluded that there was inadequate evidence to assess the carcinogenic potential of isooctane, based on the absence of human epidemiological studies and chronic bioassays on this compound. However, the CIR Expert Panel noted that no significant tumor incidence was found following lifetime dermal application of petrolatum (15% in isooctane) to mice and also found no evidence of any concern regarding carcinogenic potential from exposure to isoparaffins as used in cosmetics.
The Expert Panel also reviewed data in chemical safety reports on isooctane, isohexadecane, isododecane, and isoeicosane that were submitted in accordance with the REACH regulation. Very minimal data on either chemical are included in these reports, and the majority of the data are on chemical mixtures, comprising n-alkanes, isoalkanes, and aromatic compounds that were used to evaluate the safety of the 4 isoparaffins. Except for Stoddard solvent IIC (CAS No. 64742-88-7), these mixtures were not identified by common names or CAS numbers in the safety reports, and percentage composition data were not included for any of the mixtures evaluated. Therefore, the Expert Panel did not agree that data on these mixtures should be used to evaluate the safety of isoparaffins in this safety assessment, even though, except for the NTP carcinogenicity data on Stoddard solvent IIC, most of the data would not raise any safety concerns and the findings are consistent with the results of similar studies on isoparaffins that support a safe conclusion for these ingredients in cosmetic products.
The NTP concluded that inhalation exposure to Stoddard solvent IIC caused cancer of the adrenal gland in male F344/N rats, and that an increase in liver tumors in female B6C3F1 mice may have been related to exposure to Stoddard solvent IIC. There was no evidence that Stoddard solvent IIC increased tumor rates in female rats or male mice. The Expert Panel noted that male F344/N rats are uniquely susceptible to the development of adrenal medulla neoplasms under experimental conditions. These neoplasms are commonly observed in rats, both unexposed and exposed to numerous, diverse, usually nonmutagenic chemicals in animal studies, but rarely in humans. Currently, there is no indication that substances inducing adrenal medulla neoplasms in animal experiments can induce these tumors in humans. Thus, the findings are generally considered not relevant to a human safety assessment. Similarly, it was agreed that the liver tumors observed in B6C3F1 female mice are not relevant, because this strain of mice is uniquely susceptible to hepatocellular adenomas, which are common in this strain.
The potential adverse effects of inhaled aerosols depend on the specific chemical species, the concentration and the duration of the exposure and their site of deposition within the respiratory system. In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range and the mean particle diameter in a typical aerosol spray has been reported as ∼38 µm. Particles with an aerodynamic diameter of ≤10 µm are respirable. After reviewing the positive acute and subchronic inhalation toxicity data considered in this safety assessment, the Expert Panel determined that isoparaffins can be used safely in hair sprays, because the product particle size is not respirable.
Conclusion
The CIR Expert Panel concluded that the following ingredients are safe in the present practices of use and concentration described in this safety assessment:
C7-8 isoparaffin;
C8-9 isoparaffin;
C9-11 isoparaffin;
C9-12 isoparaffin*;
C9-13 isoparaffin*;
C9-14 isoparaffin*;
C9-16 isoparaffin*;
C10-11 isoparaffin;
C10-12 isoparaffin*;
C10-13 isoparaffin;
C11-12 isoparaffin;
C11-13 isoparaffin;
C11-14 isoparaffin*;
C12-14 isoparaffin;
C12-20 isoparaffin*;
C13-14 isoparaffin;
C13-16 isoparaffin;
C18-70 isoparaffin;
C20-40 isoparaffin*;
C15-35 isoparaffin/isoalkylcycloalkanes*;
Isooctane;
Isohexadecane;
Isododecane;
Isoeicosane.
Asterick (*) indicates the ingredients are not in current use. Were these ingredients to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in the group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
