Abstract
Nephrotoxicity is one of the serious dose-limiting complications of methotrexate (MTX) when used in the treatment of various malignancies and nononcological diseases. The aim of this study was to investigate the role of poly(adenosine diphosphate ribose) polymerase (PARP) activity in MTX-induced nephrotoxicity. Rats were divided into 4 groups as control, MTX treated (MTX, 7 mg/kg per d, intraperitoneally [ip], once daily for 3 consecutive days), MTX plus 1,5-isoquinelinediol (ISO, a PARP inhibitor, 3 mg/kg per d, i.p.) treated, or ISO treated. Histopathology of kidneys was evaluated by light microscopy. Terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end labeling assay was used to analyze apoptosis in kidney sections. Blood urea nitrogen (BUN), serum creatinine, and urinary N-acetyl-β-
Introduction
Methotrexate (MTX), a classical antifolate, is widely used as a cytotoxic chemotherapeutic agent in the treatment of a variety of malignancies. 1 –4 It is also an important component in the treatment regimes of nononcological diseases, including rheumatoid arthritis, psoriasis, and refractory inflammatory bowel disease. 5,6 However, the therapeutic benefits of MTX are often limited by the occurrence of acute toxicity which contributes to drawback of MTX therapy. Thus, minimizing the toxicity of MTX may enhance the therapeutic efficacy of the drug.
Nephrotoxicity is one of the major complications of MTX treatment. 7 Acute renal failure induced by MTX therapy occurs less frequently (in 1.4% of patients) than the minor toxic effects but may be life threatening. 8,9 Von Hoff et al reviewing the records of 498 patients treated with MTX noted 6% incidence of drug-related deaths. 10 Of these deaths, 20% were attributed to renal failure.
Tubular secretion, reabsorption, and glomerular filtration are all involved in the renal elimination of MTX. 11 As MTX is primarily cleared by renal excretion, MTX-induced renal dysfunction leads to delayed elimination of MTX, and the resulting sustained elevated plasma MTX concentration may lead to a marked enhancement of MTX’s other toxicities. 2,12 –15 It is essential to understand the mechanism of nephrotoxicity of MTX in order to diminish the adverse effects and hence maximize the benefits of chemotherapy. 7 The etiology of MTX-induced renal dysfunction is believed to be via a direct toxic effect of MTX on the renal tubules, while the mechanism of nephrotoxicity of MTX is not fully understood. 16 Numerous studies support the hypothesis that MTX-induced nephrotoxicity may be a consequence of oxidative stress. 7,17 –19 Previous studies clearly demonstrated that neutrophil infiltration and oxidative stress contribute to MTX-induced renal damage. 20 The MTX administration resulted in increased malondialdehyde levels and myeloperoxidase activity and decreased glutathione levels in the blood, liver, and kidney. 21
Poly(adenosine diphosphate-ribose) polymerase (PARP), also known as poly-(ADP-ribose) synthetase (PARS), is an abundant nuclear enzyme that has been implicated in the cellular response to DNA injury. 22,23 Under physiological conditions or limited DNA damage, PARP plays a protective role. 24 On the other hand, after DNA damage caused by oxidative stress, PARP activation plays a crucial role in cell death program. 25 –27 Activation of PARP is triggered by single-strand breaks in DNA and subsequently results in a rapid depletion of intracellular nicotinamide adenine dinucleotide (NAD+) and adenosine triphosphate (ATP) and forms a large body of PAR. This slows the rate of glycolysis and mitochondrial respiration and eventually leads to cellular dysfunction and death. 26,27 Activation of PARP represents an important mechanism of tissue damage in various pathological conditions related to oxidative stress. 28 –35 Since the previous studies suggest that oxidative stress may contribute to the development of MTX-induced nephrotoxicity, 7,17 –19 activation of the PARP pathway as a consequence of increased oxidative stress and subsequent DNA strand breaks can be hypothesized to explain renal damage in MTX-treated rats. While the association between reactive oxygen species (ROS) and MTX-induced renal damage is partly investigated, the downstream cellular mechanisms involved after MTX-induced ROS generation in renal tissues are still unknown. Taking the above into account, the aim of the present study was to investigate whether there is a relationship between MTX-induced nephrotoxicity and activation of PARP pathway. In previous studies, pharmacological inhibitors of PARP have been shown to have strong anticancer activity. 36,37 Therefore, we also investigated whether the pharmacological PARP inhibition might be a therapeutically viable strategy in alleviating the renal damage induced by MTX treatment.
Materials and Methods
Experimental Procedures
This study was registered by the Animal Ethics Committee of Akdeniz University Medical Faculty, Antalya, Turkey. Briefly, male Wistar rats, weighing 250 to 300 g, were used. Rats were randomly divided into 4 groups. The first group that served as control (C group, n = 6) received only intraperitoneally (ip) physiological saline of 1 mL/kg. The second group (MTX-treated group, n = 6) received 3 consecutive daily ip injections of MTX at the dose of 7 mg/kg per d. The third group (MTX plus ISO-treated group, n = 6) was treated with ISO (3 mg/kg per d, i.p.) 24 hours before the first administration of MTX and repeated at 24-hour intervals for 3 days. Finally, the fourth group (ISO-treated group) received only ISO for 3 days at the same dose. Doses and route of administration of MTX and ISO were determined from those described in the literature. 18,20,21,34,35
Determination of BUN, Serum Creatinine, and Urinary NAG Levels
After the last MTX injection, all animals were placed in metabolic cages to collect urine samples. After 24 hours, all rats were weighted and anesthetized with a cocktail of ketamine hydrochloride (90 mg/kg) and xylazine (10 mg/kg) administered intramuscularly before sacrificing the rats. Blood samples were obtained from the renal vein. Thereafter, serum was separated by centrifugation at 2000g for 5 minutes at 4°C for determination of blood urea nitrogen (BUN) and serum creatinine. The kidneys were removed and weighted.
Evaluation of Apoptosis by TUNEL Labeling
Apoptosis was detected by enzymatic labeling of DNA strand breaks using terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end labeling (TUNEL). The TUNEL labeling was conducted using a Cell Death Detection kit (Roche, Mannheim, Germany) and performed according to the manufacturer instructions. Cryo sections of 5-μm thickness from the renal tissues were collected on slides covered with poly-
Tissue Processing and Immunohistochemistry
Renal tissues obtained from all groups were fixed in 10% formalin and were processed routinely for paraffin embedding. Paraffin-embedded tissue samples were cut into 5-μm thick sections and mounted on SuperFrost Plus slides (Erie Scientific Company, Portsmouth, New Hampshire). For poly(ADP-Ribose [PAR]) and PARP-1 immunostaining, sections were deparaffinized in xylene and rehydrated in a graded series of alcohol. For antigen retrieval, slides were placed in 10 mmol/L citrate buffer (pH 6.0) and were microwaved twice for 5 minutes. Tissue sections were blocked for endogenous peroxidase activity with methanol containing 3% hydrogen peroxide (H2O2) for 10 minutes. After several washes with PBS, to eliminate nonspecific binding, sections were incubated with Ultra V Block (Labvision, Fremont, California) for 7 minutes at room temperature. Mouse monoclonal PAR (anti-PAR Mab [10H], Alexis, San Diego, California) and rabbit polyclonal PARP-1 (Ab 6079, Abcam, Cambridge, United Kingdom) primary antibodies were applied in a dilution of 0.6 μg of immunoglobulin (Ig)G3/mL (1:1500 dilution) and 6 μg of IgG/mL (1:300), respectively. Sections were washed in PBS and incubated with biotinylated horse anti-mouse IgG (Vector Laboratories, Burlingame, California) secondary antibody at 1:1000 dilution for PAR and anti-rabbit IgG (Vector Laboratories) secondary antibody at 1:500 dilution for PARP-1 for 1 hour at room temperature. After several PBS rinses, the antigen–antibody complex was detected using an avidin–biotin horseradish peroxidase complex with a Universal LSAB Kit (Dako, Glostrup, Denmark). Diaminobenzidine (3,3-diaminobenzidine tetrahydrochloride dihydrate; Sigma Chemical, St Louis, Missouri) was used as the chromogen. Sections were mounted with Permount (Fisher Chemicals, Pittsburgh, PA) on glass slides and then evaluated under light microscope. For controls, sections were incubated with nonimmune IgG3 (R&D Systems, Minneapolis, MN) and rabbit serum (Dako) at the same concentrations with the primary antibodies. Pictures were taken with Spot Advanced Imaging Software (Diagnostic Instruments, Sterling Heights, MI).
Sodium Dodecyl Sulfate–Polyacrylamide Gel Electrophoresis and Western Blotting
Kidney tissues were weighed and placed in homogenation buffer (10 mmol/L Tris-HCl, 1 mmol/L EDTA, 2.5% sodium dodecyl sulfate [SDS], 1 mmol/L phenylmethylsulfonylfluoride, and 1 mg/mL leupeptin) supplemented with CompleteR protease inhibitor cocktail (Boehringer, Mannheim, Germany). After homogenization and sonication, samples were centrifuged at 10 000g for 10 minutes. Supernatants were collected and protein concentrations were determined using a Standard BCA assay (Sigma Chemical) and 100 µg protein was applied per lane. Prior to electrophoresis, samples were heated for 5 minutes at 95°C. Samples were then subjected to SDS polyacrylamide gel electrophoresis under standard conditions and then transferred onto polyvinylidene fluoride membrane in a buffer containing 0.2 mol/L glycine, 25 mmol/L Tris, and 20% methanol, overnight. The membranes were blocked for 1 hour with 5% nonfat dry milk and were then incubated for 1 hour at room temperature with the same rabbit antisera against PARP-1 (0.5 µg/mL, Ab 6079) and mouse antisera against PAR (3 µg/mL, anti-PAR Mab [10H]) used for immunohistochemistry. After washing, the membranes were incubated with anti-rabbit and anti-mouse IgG horseradish peroxidase conjugate diluted 1:5000 for 1 hour at room temperature. Immunolabeling was visualized using the chemiluminescence-based SuperSignal CL HRP Substrate System and the membranes were exposed to hyperfilm. As an internal standard to confirm the equal loading of the proteins, β-actin was loaded to the gels.
Drugs
In this study, MTX was administered as the clinical formulation. Methotrexate was purchased from Sandoz, Turkey (Edicin 0.5 g) and prepared fresh daily during the experiments.
Statistical Analysis
All values are expressed as mean ± standard error of the mean. Statistical analysis of the results was performed by 1-way analysis of variance. Post hoc comparisons were done using Tukey multiple comparison test. P value <.05 was considered as significant.
Results
Although MTX administration caused a decrease in body weight of rats, there were no significant differences between the initial and final body weights among all groups (Table 1). However, kidney/body weight ratios significantly decreased after MTX administration when compared with control rats. Moreover, the daily water intake of rats decreased significantly after MTX administration, whereas urinary output values were similar in all groups (Table 2). In animals treated with ISO + MTX, kidney/body weight ratios and daily water intake did not change significantly as seen in Tables 1 and 2. In the ISO-treated rats, body weights and kidney/body weight ratios were similar to the controls, while it was significantly higher than that of the MTX-treated rats.
Body Weights and Kidney/Body Weight Ratios in Control, MTX, MTX + ISO, and ISO Groupsa
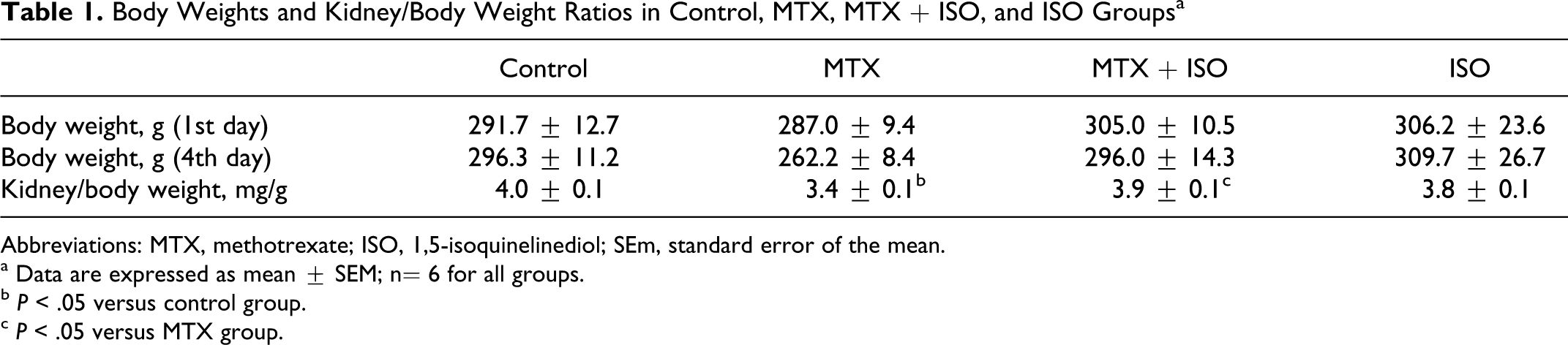
Abbreviations: MTX, methotrexate; ISO, 1,5-isoquinelinediol; SEm, standard error of the mean.
a Data are expressed as mean ± SEM; n= 6 for all groups.
b P < .05 versus control group.
c P < .05 versus MTX group.
Water Intake and Urinary Output Values in Control, MTX, MTX + ISO, and ISO Groupsa
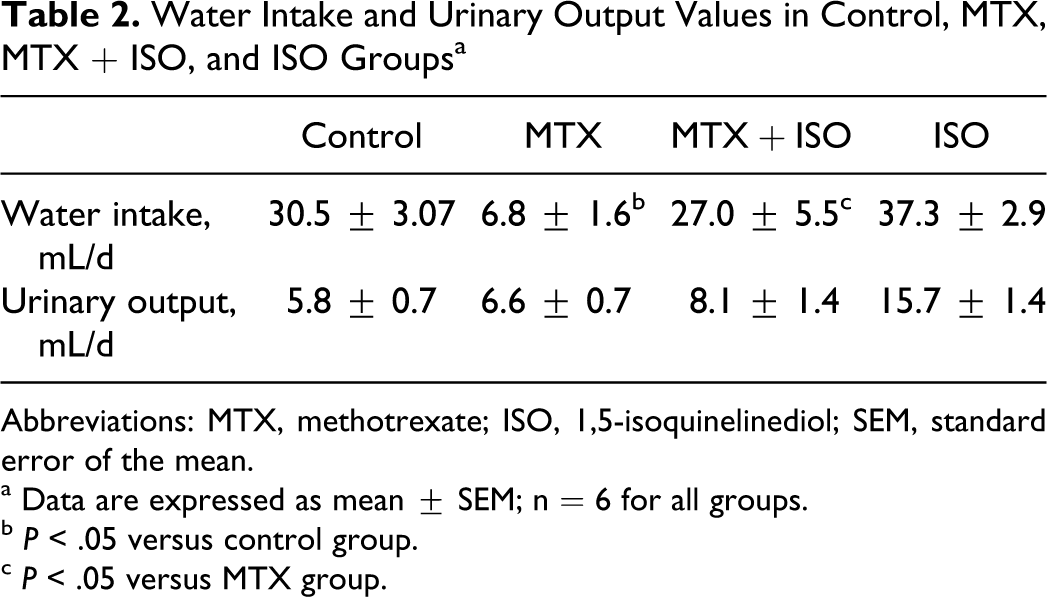
Abbreviations: MTX, methotrexate; ISO, 1,5-isoquinelinediol; SEM, standard error of the mean.
a Data are expressed as mean ± SEM; n = 6 for all groups.
b P < .05 versus control group.
c P < .05 versus MTX group.
The BUN, Serum Creatinine, and Urinary NAG Levels
As shown in Figure 1A, BUN values significantly increased with MTX treatment when compared to control (by 1.46-fold). Treatment with ISO significantly reduced BUN levels when compared to MTX-treated rats. Furthermore, serum creatinine levels in MTX-treated rats were higher than that of the control group and it decreased by ISO treatment (Figure 1B). Serum creatinine levels were 0.24 ± 0.01, 0.31 ± 0.01, 0.24 ± 0.02, and 0.24 ± 0.01 mg/dL for control, MTX, MTX plus ISO, and ISO groups, respectively. These findings were accompanied by significant increases in urinary N-acetyl-β-
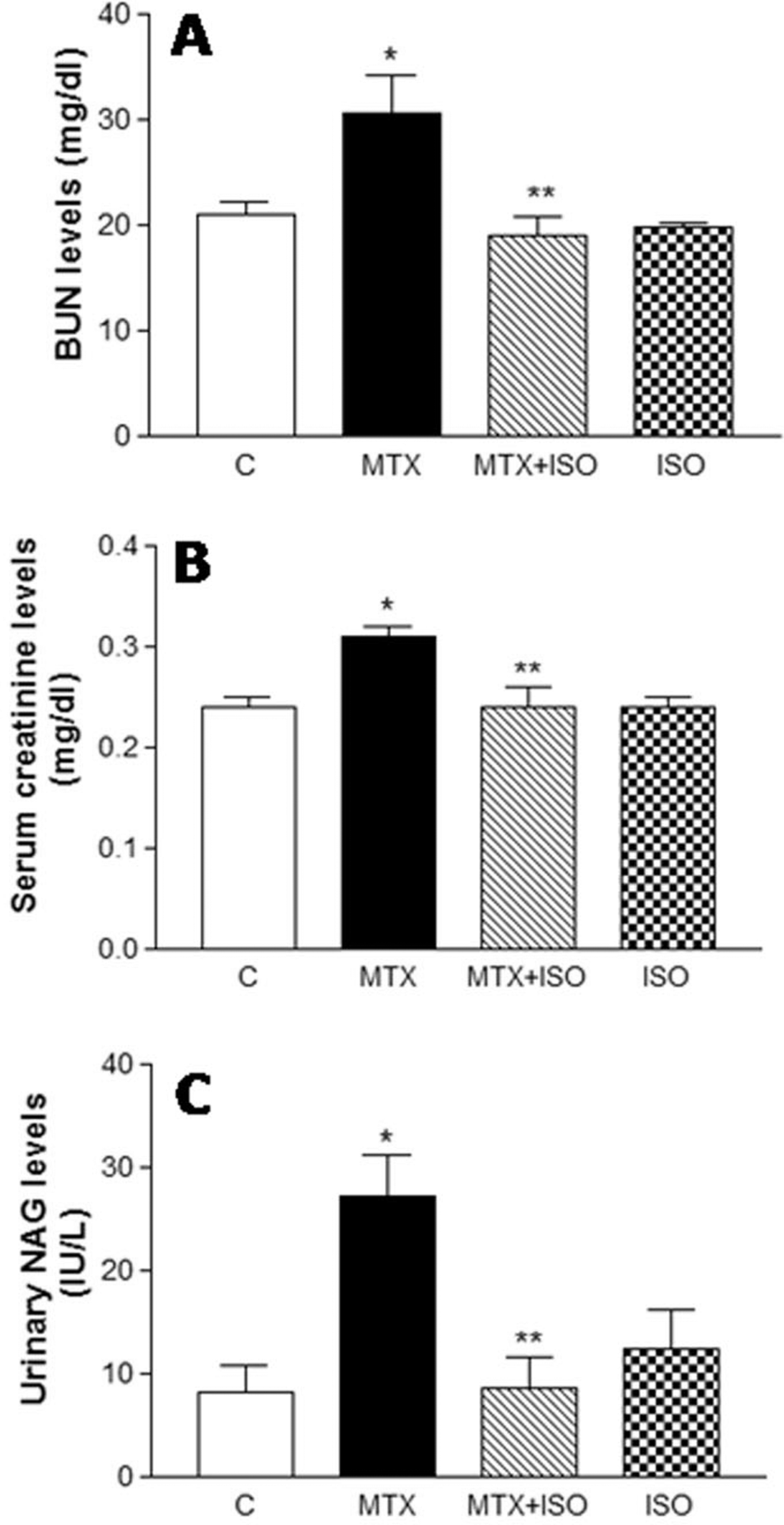
Effect of ISO treatment on BUN (A), serum creatinine (B), and urinary NAG (C) levels after MTX administration. All values are expressed as mean ± SEM, n = 6 for all groups. BUN indicates blood urea nitrogen; ISO, 1,5-isoquinelinediol; MTX, methotrexate; NAG, N-acetyl-β-
Renal Histology
Kidneys from rats treated with MTX showed some tubular damage when compared with controls (Figure 2). The occurrence of desquamation and vacuolization of renal tubule epithelium and interstitial tissue inflammation due to the accumulation of inflammatory cells were seen in the MTX-treated rats. Treatment with ISO markedly diminished the tissue damage induced by MTX administration (Figure 2C).
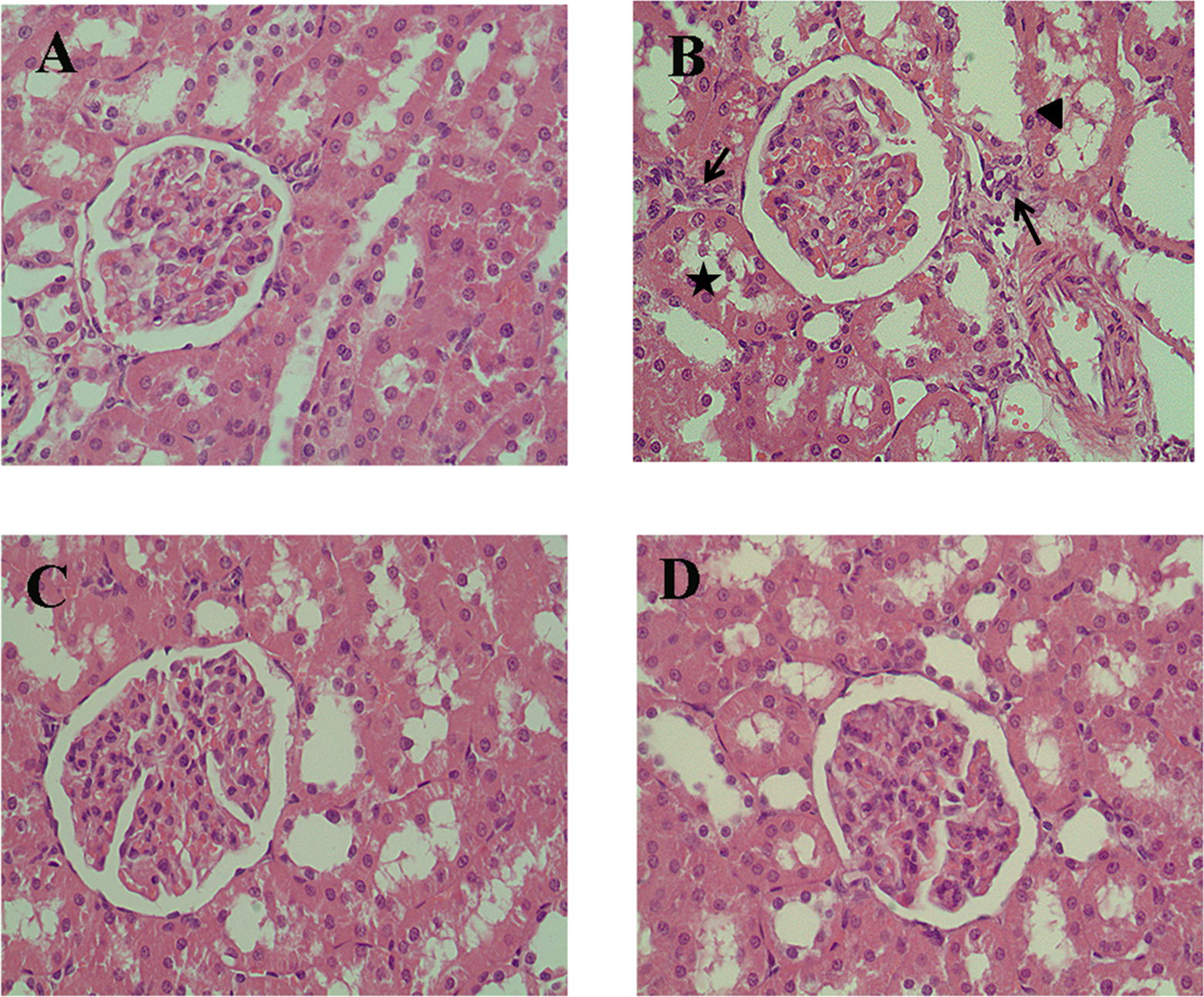
Representative photomicrographs from control (A), MTX-treated (B), MTX plus ISO-treated (C), and ISO-treated (D) rat renal cortex sections stained with hematoxylin and eosin. (A) and (D) shows normal renal histology. In MTX-treated rat kidneys (B), epithelial cell vacuolization (arrowhead) and desquamation (star) and interstitial tissue inflammation indicated by inflammatory cell infiltration (arrows) were seen. The MTX plus ISO-treated group (C) showed marked reduction in renal cortex tubulointerstitial damage. ISO indicates 1,5-isoquinelinediol; MTX, methotrexate.
Evaluation of Apoptosis in Kidney Tissues
Using TUNEL assay, we found profound DNA damage in the renal tubular cells in MTX-treated rats as reflected by the quantitative assessment of TUNEL-positive cells. Kidneys from rats treated with MTX showed marked apoptosis when compared with the controls (Figures 3 and 4). On the other hand, the number of TUNEL-positive apoptotic cells in MTX plus ISO-treated rats was similar to the control groups (Figures 3 and 4).
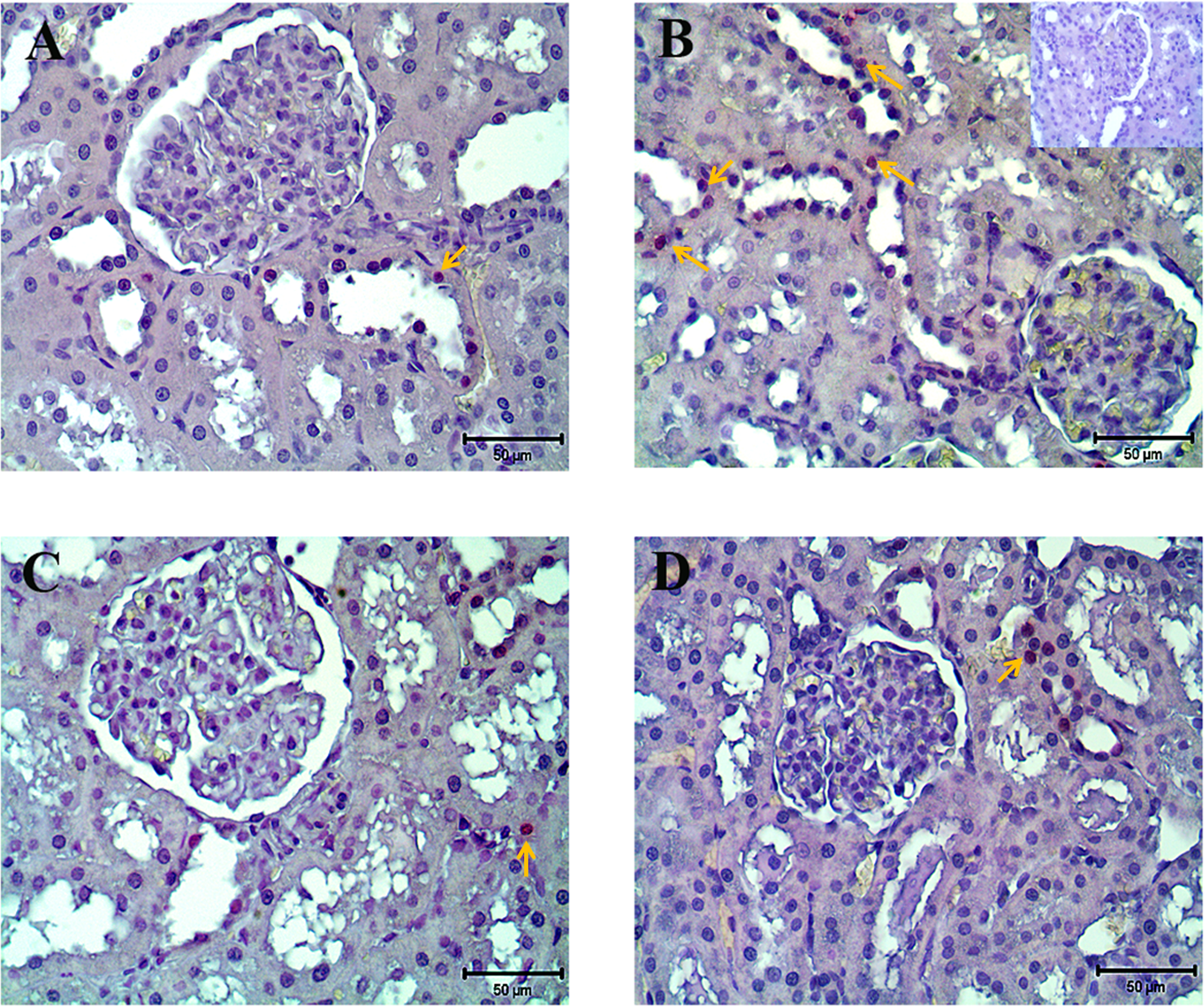
Representative photomicrographs of kidneys for terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end-labeling (TUNEL) in control (A), MTX-treated (B), MTX plus ISO-treated (C), and ISO-treated (D) rats. Apoptotic cell death increased in renal tubular cells treated with MTX (arrows), whereas apoptosis decreased in tubular cells treated with MTX plus ISO. Inset in B represents the negative control section for TUNEL method. ISO indicates 1,5-isoquinelinediol; MTX, methotrexate.
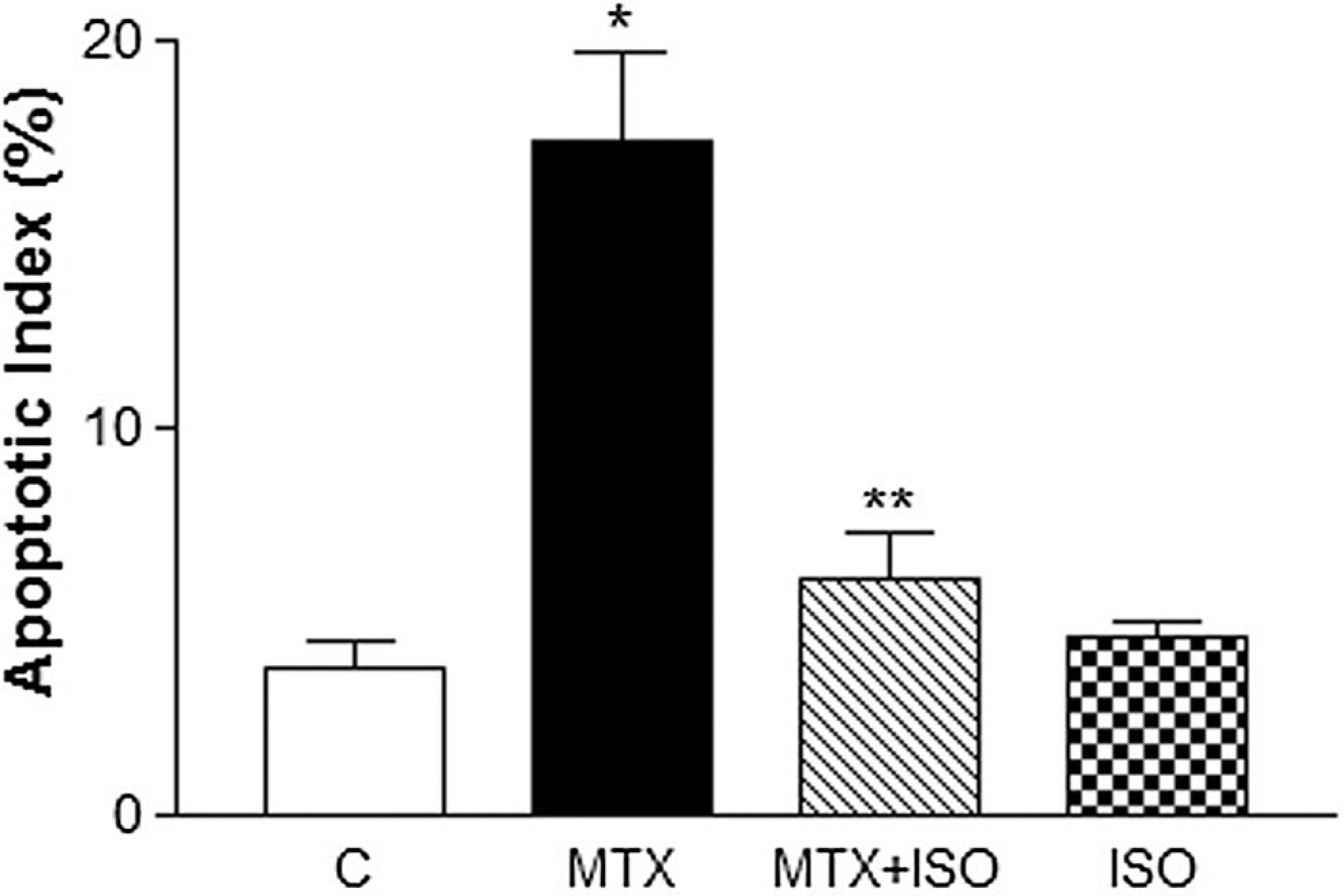
Graph shows the apoptotic index of terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end-labeling (TUNEL)-positive cells in control and experimental groups. The percentage of apoptotic cells were significantly higher in MTX group when compared to controls. n = 6 for all groups. ISO indicates 1,5-isoquinelinediol; MTX, methotrexate. *P < .05 as compared with control and **P < .05 as compared with MTX.
Expressions of PARP-1 and PAR in Kidney Tissues
For the localization of cellular expression of PARP-1 and PAR, renal tissue sections were immunohistochemically stained with anti-PARP-1 and anti-PAR antibodies. There was weak PARP-1 and PAR expression in the cortex of control kidneys (Figures 5A and 6A). After the administration of MTX, strong expression of both PARP-1 and PAR in renal cortex was observed (Figures 5 and 6). Immunostaining intensity of PAR increased significantly in the epithelium of proximal and distal renal tubules and collecting ducts. All epithelial cells of the renal tubules showed strong PAR immunostaining (Figure 5B). Although the nuclear immunoreactivity of PAR was seen mainly in epithelial cells of renal tubules, some cells of the glomeruli also showed PAR expression. Strong PARP-1 immunostaining was present in renal tubule epithelium of MTX-administrated rats, whereas mild PARP-1 expression was seen in glomerulus (Figure 6B). In the MTX plus ISO-treated rats, the intensity of PARP-1 and PAR expressions was less than that of the MTX-treated rats (Figures 5C and 6C).
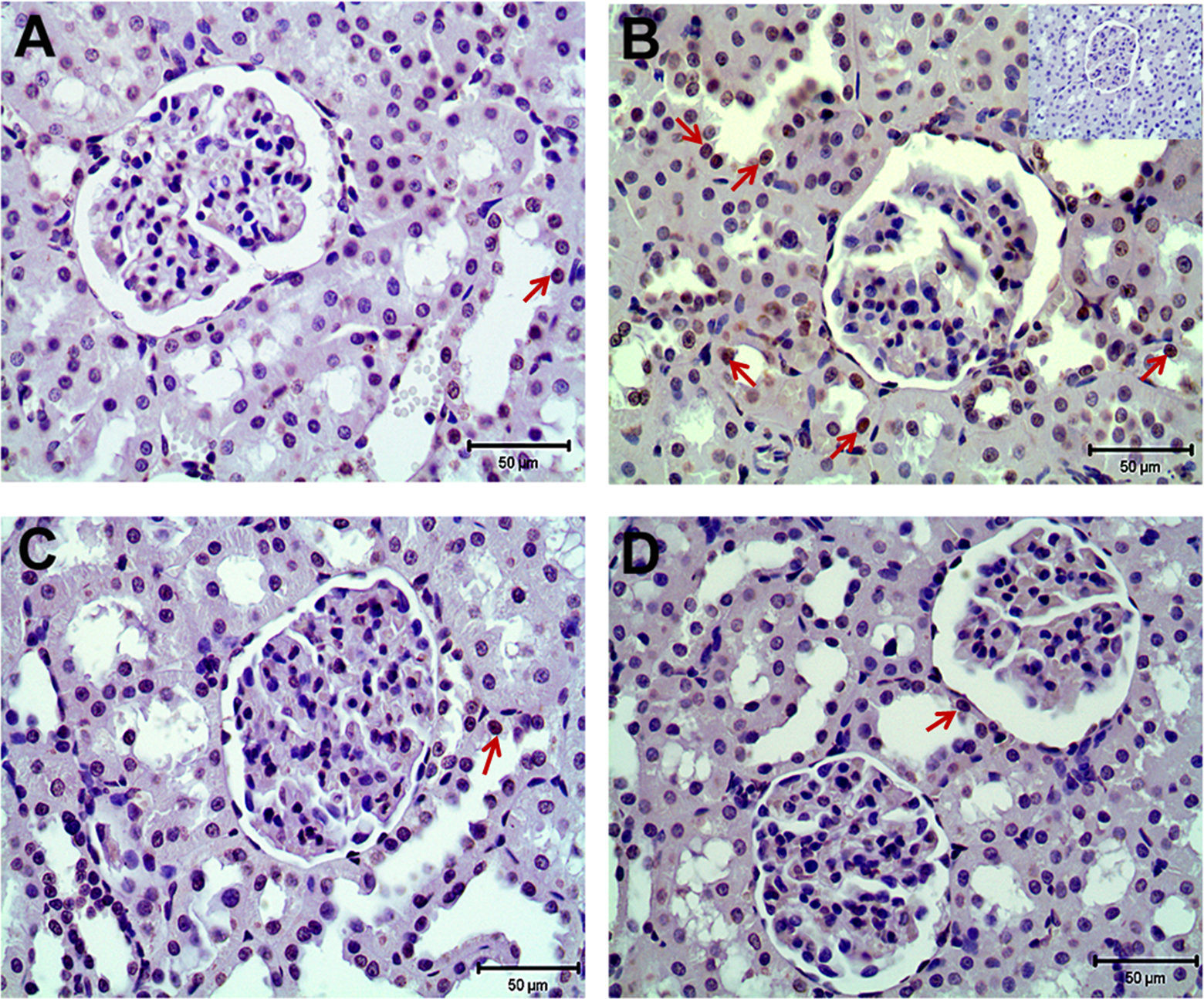
Representative photomicrographs for PAR (A-D) immunohistochemistry in control (A), MTX-treated (B), MTX plus ISO-treated (C), and ISO-treated (D) kidneys. Arrows indicate expression and localization of PAR in renal cortex. Strong immunostainings of PAR (B) were observed in tubular cells of MTX-treated groups. In MTX plus ISO-treated rats, the expression of PAR (C) in tubular cells were similar to controls. Inset in B represents the negative control immunostainings of PAR. ISO indicates 1,5-isoquinelinediol; MTX, methotrexate; PAR, poly(ADP-Ribose).
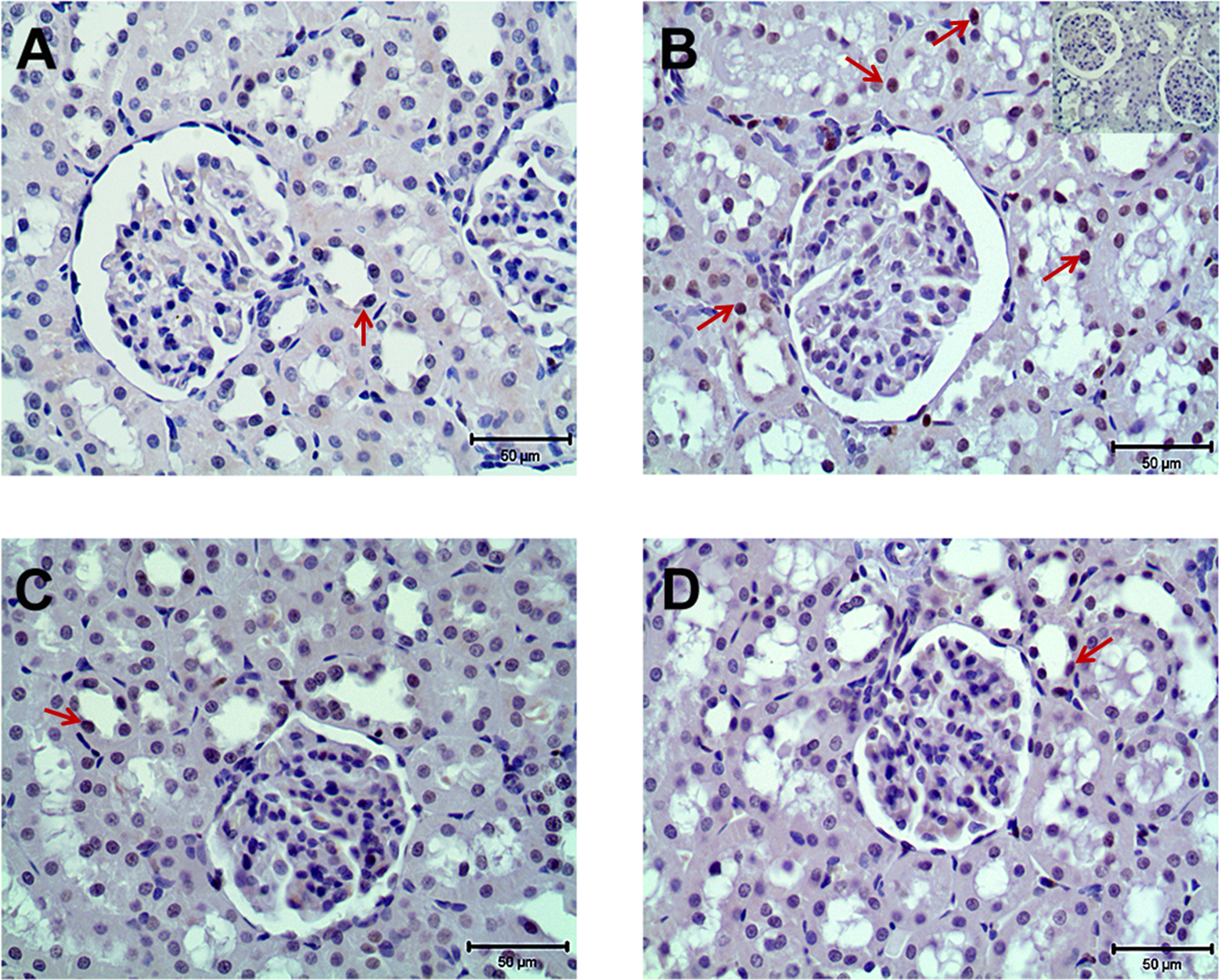
Representative photomicrographs for PARP-1 (A-D) immunohistochemistry in control (A), MTX-treated (B), MTX plus ISO-treated (C), and ISO-treated (D) kidneys. Arrows indicate expression and localization of PARP-1 in renal cortex. Strong immunostainings of PARP-1 (B) were observed in tubular cells of MTX-treated groups. In MTX plus ISO-treated rats, the expression of PARP-1 (C) in tubular cells was similar to the controls. Inset in B represents the negative control immunostainings of PARP-1. ISO indicates 1,5-isoquinelinediol; MTX, methotrexate; PARP-1, poly(adenosine diphosphate ribose) polymerase.
In order to elucidate the expression levels of PARP-1 and PAR, we performed Western blot analysis of these proteins from whole kidneys of control, MTX-treated, MTX plus ISO-treated, and ISO-treated rats (Figure 7). In control and ISO groups, weak PARP-1, and PAR, the expressions were detected. However, the expressions of PARP-1 and PAR increased in MTX-treated rats. Additionally, the level of cleaved PARP-1 expression as a marker for apoptotic cell death increased after MTX administration (Figure 7). The ISO administration significantly reduced the expression levels of PARP-1, cleaved PARP-1, and PAR in the kidney of MTX-treated rats. The ISO treatment alone did not cause a significant change in PARP-1, cleaved PARP-1, and PAR expression.
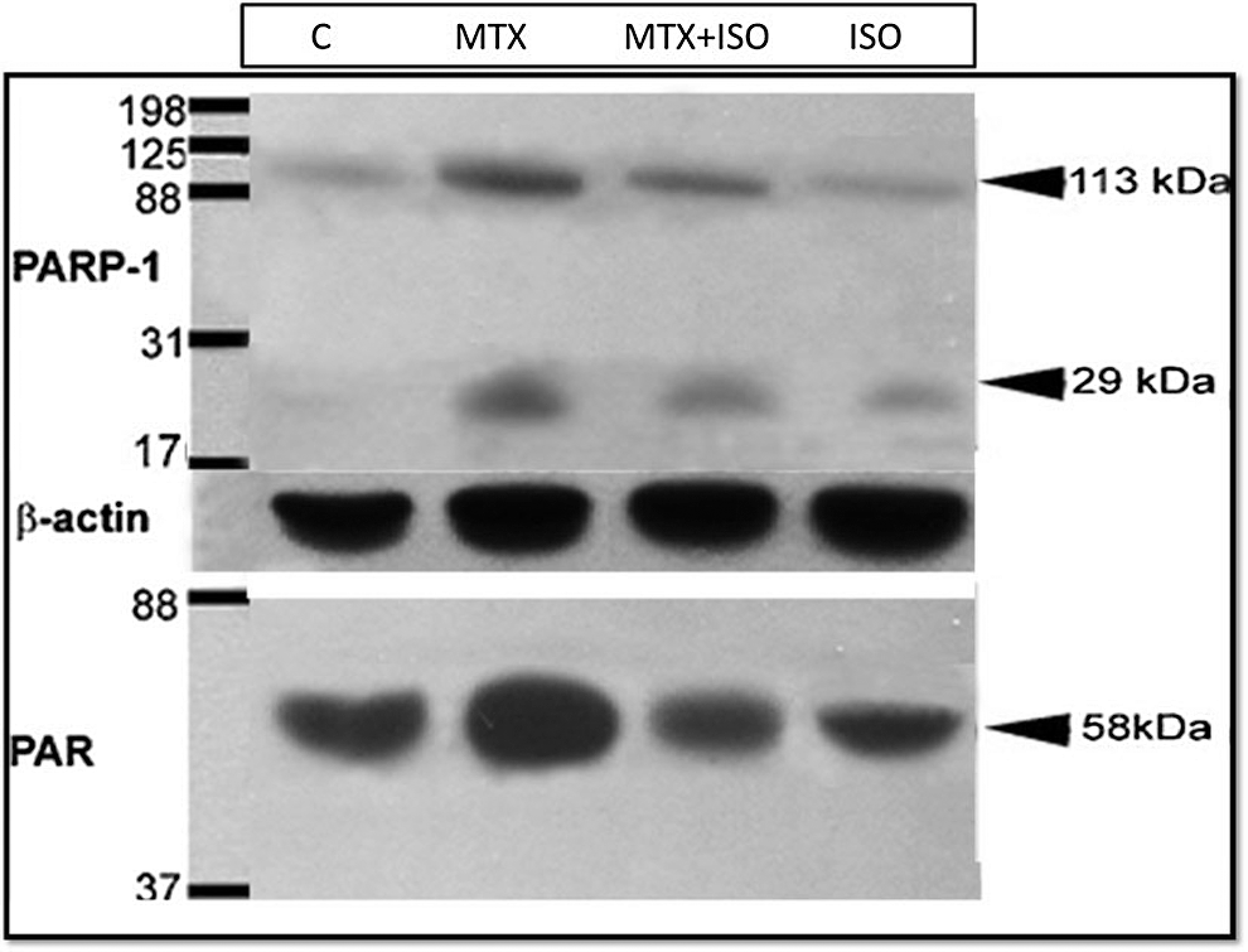
Evaluation of PARP-1 and PAR protein expression of renal tissues by Western blot analysis in control, MTX-treated, MTX plus ISO-treated, and ISO-treated rats. C indicates control rats; ISO, 1,5-isoquinelinediol–treated rats; MTX, methotrexate-treated rats; MTX + ISO, MTX plus ISO-treated rats; PAR, poly(ADP-Ribose); PARP-1, poly(adenosine diphosphate ribose) polymerase.
Discussion
Many potential therapeutic approaches for the prevention of MTX-induced renal injury have been studied. To date, several factors have been proposed to explain MTX-induced nephrotoxicity. 7 ,17 –20 The present study was carried out to investigate the effect of PARP inhibition on MTX-induced acute renal injury. Our study is the first to demonstrate the role of PARP activation as a possible mechanism of MTX-induced nephrotoxicity and renal tubular apoptosis.
The kidney is especially vulnerable to toxic insults by several drugs and xenobiotics, because it receives nearly one quarter of the cardiac output, and it transports, metabolizes, and concentrates various toxic substances within its parenchyma. 38 In this study, the dosage regimen chosen for MTX produced reproducible kidney damage, in accordance with previous observations. 20,39 The present investigation reveals that MTX treatment leads to a decrease in body weight and kidney/body weight ratios of rats. The reduction in body weight following MTX administration may possibly be related to damaged renal tubules and the subsequent loss of the tubular cells to reabsorb water, indicating some degree of dehydratation, thus loss of body weight. This hypothesis was supported by a decrease in water intake/urinary output (water turnover rate) ratio in MTX-treated rats. The results of present study indicated that MTX-administrated rats demonstrated a defect in the kidney’s ability to produce concentrated urine during dehydration. The urinary concentrating process depends on the coordinated function of the loop of Henle and the collecting ducts. 40 Water excretion is dependent on a balance between the glomerular filtration rate and the net rate of water absorption along the renal tubule. 41 One of important factors associated with this decrease in tubular water reabsorption is osmotic diuresis that may accompany the elimination of urea and creatinine that accumulated in the blood due to the renal failure. 42 Another important factor is a decrease in the abundance of water channels that are largely responsible for enabling water to penetrate the epithelium of collecting duct and returning it to the bloodstream in response to vasopressin. 43,44 The histopathological examination of kidneys of MTX-treated rats revealed increased number of apoptotic cells in collecting tubules to some extent. Thus failure to concentrate urine may be a consequence of injury in collecting tubules of MTX-administrated rats. Although the histology in the provided photomicrographs indicates minimal cellular damage in kidneys of MTX-treated rats, our data show increased TUNEL-positive cells in renal tubules of MTX-treated rats. This finding suggests tubular cell death in the early stages of acute tubular injury by apoptosis. Besides histopathological changes, MTX administration led to a marked increase in serum creatinine and BUN levels. It is generally accepted that a marked decrease in water consumption and a failure to concentrate urine during dehydration results in prerenal azotemia. The most common cause of elevation in BUN and creatinine is prerenal azotemia as well as progressive loss of functional nephrons. Due to the observed decrease in water consumption and failure to concentrate urine, it is expected that a mild increase in BUN and creatinine would occur. Hence, prerenal azotemia may contribute to the increased BUN and creatinine levels in MTX-treated rats. In the present study, successful induction of acute renal failure was confirmed also by an increase in urinary NAG excretion, an early and more sensitive marker of changes in proximal tubular function than BUN and serum creatinine. 45 –47 Urinary NAG excretion in MTX-treated rats increased significantly when compared with control rats. These observations are in agreement with the reported early alterations observed in serum and urine profiles following MTX administration. 20,39
The nephrotoxic mechanism/mechanisms of MTX is not yet been fully established. There is evidence in the literature about the possible role of oxidative stress in the pathophysiology of MTX-induced acute renal injury. 7,17,18,20 It is well known that oxidative stress accompanied by increased formation of superoxide anions (O2 −), H2O2, and peroxynitrite (ONOO−) are endogenous inducers of DNA single-strand breakage and is the obligatory of PARP activation. 48 –50 The ROS-DNA injury-PARP pathway has recently been established as a major downstream intracellular pathway of oxidative stress. When massive DNA damage occurs, the associated extensive activation of PARP is involved in the repair process of DNA but uses NAD+ as a substrate, which is the donor of the ADP-Ribose group. Subsequently, activation of NAD+ recycling functions to restore its levels but as cellular stores of ATP are depleted, the cell cannot support the NAD+ salvage, leading to further energy failure and cell death. 51 –53 It is also speculated that poly(ADP ribosyl)ation may represent a quantity of DNA damage and some intracellular signal mechanisms for cell death may exist after_poly(ADP-ribosyl)ation. 54,55 The importance of PARP pathway is clearly documented in ischemic acute renal failure. 56,57 Also, Shevalye et al have shown that PARP-1 gene deficiency alleviates diabetic kidney diseases. 58 Recently, we reported the protective effect of PARP inhibition in alleviating vancomycin-induced renal injury. 34 Our study shows that ISO, a potent PARP inhibitor, protects tubular cells against MTX-induced renal injury. Our results clearly indicate that MTX treatment markedly increased serum creatinine, BUN, and urinary NAG levels. Treatment with ISO for 3 consecutive days, starting 24 hours before MTX administration, efficiently reduced acute nephrotoxicity by reducing the increase in BUN, plasma creatinine, and urinary NAG levels. These results support the role of PARP activation in MTX-induced renal impairment. Treatment with ISO restored the kidney weights similar to that of controls. Moreover, MTX administration leads to apoptotic cell death as indicated by TUNEL assay. Importantly, we have shown that administration of ISO seems to protect against apoptosis and nephrotoxicity induced by MTX.
In addition, immunohistochemical studies showed increased expression of both PARP-1 and PAR in renal tubular cells after MTX administration. The expression patterns of PARP-1 and PAR were similar to each other. In accordance with these results, Western blot analysis confirmed increased PAR expression in MTX-administrated rat kidneys. The ISO treatment reduced PAR expression to control levels. Inhibition of PARP-1 and PAR immunoreactivities also paralleled with the reduction in renal dysfunction. Taken together, the results of this study indicate that MTX may lead to tubular apoptosis and renal failure by overactivation of PARP pathway. The detailed amelioration mechanism of MTX-induced nephrotoxicity by PARP inhibition is unclear. The literature has suggested that either depletion of NAD+ or activation of PARP-1 with subsequent PAR polymer formation are critical steps in the induction of cell death. 52,59 When activated by DNA single-strand breaks, PARP initiates an energy consuming cycle by transferring ADP-Ribose units from NAD+ to nuclear proteins resulting in rapid depletion of the intracellular NAD+ and ATP pools, slowing the rate of glycolysis and mitochondrial respiration eventually leading to cellular dysfunction and death. 52,53,60 Hence, it is most likely that exposure to MTX may lead to ATP failure and renal damage as a result of increased PARP enzyme activity. Thus, marked renal dysfunction after MTX treatment may be related with low levels of ATP content as a consequence of increased enzymatic activity of PARP. By the means of our results, it may suggest that MTX triggers PARP activation, resulting in cellular energetic impairment in the tubular cells, which subsequently induces renal damage. Protective effect of ISO treatment may be related with the inhibition of PARP activity within the proximal tubules with an improved cellular energy status. The PARP activation is robust in the MTX-induced rat kidneys producing variable lengths of PAR polymers as by-products of PARP activation. The PAR polymers may be directly toxic by triggering mitochondrial apoptosis–inducing factor release independently of NAD+ depletion. 61 It has been reported that excessive PAR polymer formation mediates DNA damage and cell death. 62,63 Poly(ADP-Ribose) has been linked to the p53 and caspases, which play an important role in apoptosis. 64 The results of the present study showed that PAR expression in tubular cells markedly increased after MTX treatment which may be responsible from apoptosis of tubular cells in the kidney. Additionally, cleaved PARP (an apoptotic form of PARP) expression increased in MTX-treated group which indicates tubular cell apoptosis.
In conclusion, our study demonstrates that PARP is activated in the renal tubular cells after MTX administration, and this activation may result in MTX-induced apoptosis and nephrotoxicity. Thus, inhibition of PARP pathway may represent a novel therapeutic approach for the treatment of MTX-induced renal injury probably by improving cellular energy status and reducing tubular apoptosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Akdeniz University Research Foundation.
