Abstract
Polybrominated diphenyl ethers (PBDEs) are a class of flame retardants whose levels have increased in the environment and in human tissues in the past decades. Exposure to PBDEs has been associated with developmental neurotoxicity, endocrine dysfunction, and reproductive disorders. In spite of their widespread distribution and potential adverse health effects, only few studies have addressed the potential neurotoxicity of PBDEs. In the present study, we evaluated the cyto- and genotoxicity of 2,2′,4,4′-tetrabromodiphenyl ether (BDE-47) and decabrominated diphenyl ether (BDE-209) in human neuroblastoma cells (SK-N-MC). The DNA damage was measured using the alkaline version of the Comet assay, while specific oxidative-generated DNA damage was evaluated by a modified version of the Comet assay with the repair enzyme formamidopyrimidine glycosylase (FPG). The results show that BDE-47 and BDE-209 (5-20 μmol/L) are able to induce DNA damage in human SK-N-MC cells. Pretreatment with the antioxidant melatonin significantly reduced the DNA damage induced by both congeners. The Comet assay carried out in the presence of FPG suggests that both congeners increase purine oxidation. In all cases, BDE-47 was more potent than BDE-209. The results indicate that 2 environmentally relevant PBDEs cause DNA damage which is primarily mediated by the induction of oxidative stress and may contribute to adverse health effects.
Keywords
Introduction
An important class of flame retardant chemicals is represented by polybrominated diphenyl ethers (PBDEs), a group of 209 congeners of brominated hydrocarbons with a fundamental structure formed by 2 phenyl rings linked by an oxygen atom (Figure 1). These compounds were added at concentrations between 5% and 30% by weight to several commercial products, such as polyurethane foams, textiles, and a variety of plastic materials, because of their ability to slow ignition and rate of fire. 1,2 Since PBDEs do not bind chemically to the product, they continuously leach out into the environment, and contamination with PBDEs, especially lower brominated congeners, has significantly increased in recent years, 3 –7 in contrast to dioxins, dibenzofurans, and polychlorinated biphenyls, whose levels have been declining. 8 The PBDE residues have been detected in house dust and foods 9 and also in human adipose tissue, blood, breast milk, and cord blood. 10 –13 The PBDEs can cross the placenta, and various studies suggest that fetuses may be particularly sensitive to PBDEs’ effect. 2,14 –16 Body burden of PBDEs is quite high in infant and in children due to higher exposure (approximately 306 ng/kg per d of PBDEs vs 1 ng/kg per d for adults) through breast milk and house dust. 17
Chemical structures of 2′,4,4′-tetrabromodiphenyl ether (BDE-47) and 2,2′,3,3′,4,4′,5,5′-decabromodiphenyl ether (BDE-209).
Several animal studies have indicated that PBDEs may cause neurodevelopmental toxicity, particularly in the domains of motor activity and cognitive behavior, 17 –20 though the exact mechanism/mechanisms of these effects remain elusive. Some studies have reported that exposure to various doses of PBDEs during pre- or postnatal development decreases the levels of thyroid hormones. 21,22 The PBDEs can also disrupt signal transduction mechanisms, including protein kinase C and calcium homeostasis. 23,24 Furthermore, several studies indicate that PBDEs may cause oxidative stress in neurons leading to apoptotic cell death. 1,23,25 –29 DNA is a cellular target of endogenous oxidation processes of various reactive oxygen species (ROS), and modified DNA bases constitute one of the major classes of oxidative DNA damage. It has been suggested that some PBDEs may cause DNA damage through generation of ROS, as they do not appear to induce gene mutations. 28 –30
2,2′,4,4′-Tetrabromodiphenyl ether (BDE-47) and decabromodiphenyl ether (BDE-209) are the major source of exposure to PBDEs in the diet. 2 The BDE-47 was present in commercial pentaBDE mixtures and has been detected at high levels in human tissues and in wildlife. 12 Through epidemiological studies of exposure to BDE-47 and occurrence of cancer in humans are not available, 31 BDE-47 may be a mutagen in mammalian cells, showing a weakly recombinogenic activity in vitro. 31,32 In neuroblastoma cells and in rat hippocampal neurons in vitro, BDE-47 was shown to increase the ROS levels and to cause DNA damage, micronuclei and nucleoplasmatic bridges increases. 28,29 In addition, 2 BDE-47 metabolites were reported to inhibit proliferation, alter cell cycle, and cause apoptosis, DNA damage, and micronucleus induction in the human hepatoma cell line HepG2 and in L02 cells. 33,34
The BDE-209 is the dominant compound in the deca-BDE commercial mixture. Deca-BDE is classified as a “possible human carcinogen” (group C), according to Environmental Protection Agency cancer guidelines because of “suggestive evidence of carcinogenic potential” based on induction of neoplastic nodules in male and female rats and hepatocellular adenomas and carcinomas (combined) in male mice. 35 However, International Agency for Research on Cancer has classified Deca-BDE in group 3 (not classifiable as to its carcinogenicity to humans). 2,36 The BDE-209 induces DNA damage, but no irreversible effects on DNA hemocytes in Dreissena polymorpha. 37 In vivo cytogenetic studies on bone marrow cells from rat femur showed no induction of chromosomal aberrations. 35 Deca-BDE does not appear to induce gene mutations in Salmonella typhimurium strains or in mouse L5178Y lymphoma cells. 35 Moreover, this compound does not induce sister-chromatid exchanges and chromosomal aberrations in the presence or absence of metabolic activation systems, in Chinese hamster ovary cells. 35
The aim of the present work was to determine whether these 2 common PBDE congeners (BDE-47 and BDE-209) would induce DNA damage in SK-N-MC cells. To assess DNA damage, the single-cell gel electrophoresis assay (Comet assay) was utilized, as this assay is being used in environmental genotoxicology, due a high sensitivity for DNA damage at low levels. 38 Both congeners had been previously shown to induce oxidative stress, though BDE-209 to a minor extent, most likely because of its lower ability to enter cells due to its bulky configuration. 26
Materials and Methods
Materials
Cell culture medium, fetal calf serum, penicillin–streptomycin solution, reagents for the Comet assay, reagents for electrophoresis, melatonin, normal melting point and low melting point agarose, dimethyl sulfoxide (DMSO), ethidium bromide, buffers, and general laboratory chemicals were purchased from Sigma-Aldrich (Milan, Italy). Hank balanced salt solution was purchased from Invitrogen. Both BDE-209 (purity 98%; CAS: 1163-19-5) and BDE-47 (purity 98,5%; CAS: 5436-43-1) were purchased from Chem Service Inc (West Chester). Formamidopyrimidine glycosylase (FPG) was kindly provided by Prof Andrew R. Collins (University of Oslo, Norway).
Polybrominated Diphenyl Ether solutions
Both PBDEs were diluted in DMSO to obtain 2 stock solutions: 20 mmol/L for BDE-47 and 1.25 mmol/L for BDE-209. To achieve the concentrations used in the assays, stock solutions were diluted using the culture medium (Dulbecco modified eagle medium [DMEM]). Final concentration of DMSO never exceeded 0.1%, a concentration which did not alter the cell viability.
Cell Culture
The SK-N-MC cells, a human neuroblastoma cell line, were obtained from American Type Culture Collection (Rockville, Maryland). The SK-N-MC cells were cultured in DMEM medium (Euroclone, Milano, Italy), supplemented with 10% (vol/vol) fetal bovine serum, 100 U/mL penicillin, 100 μg/mL streptomycin, and 2 mmol/L
Assessment of Cytotoxicity
Cell viability was assessed by the MTS assay and by Trypan blue exclusion. The CellTiter 96 AQueous Solution Reagent kit (Promega, Madison, Wisconsin) was utilized for the MTS assay. The reagent is composed of a tetrazolium compound [3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt; MTS] and an electron coupling reagent. Cells were cultured at a density of 5 × 103 cells per well in a 96-well plate in DMEM supplemented with 1% glutamine, 0·5% penicillin/streptomycin, and 5% fetal bovine serum. After seeding, SK-N-MC cells were treated with different concentrations of either PBDE (0, 5, 10, and 20 μmol/L) and incubated for 24 hours. The MTS is reduced by cells into a formazan product that is soluble in tissue culture medium. The quantity of formazan product measured after 4 hours at 450 nm (in a 96-well plate reader MULTISKAN EX; Thermo Electron Corporation, Vantaa, Finland) is directly proportional to the number of living cells in culture. The percentage of live cells was assessed by Trypan blue exclusion. After treatment with either PBDE (0, 5, 10, and 20 μmol/L, 24 hours), SK-N-MC cells were resuspended in DMEM, and Trypan blue was added. A minimum of 300 cells for each concentration were counted manually using a hemocytometer.
Assessment of DNA Damage by the Comet Assay
The SK-N-MC cells treated with different concentrations of each PBDE (0, 5, 10, and 20 μmol/L) were maintained in a humidified incubator at 37°C with 5% CO2; after 4 or 24 hours, cells were washed and resuspended in DMEM medium. In some experiments, 20 hours after seeding, SK-N-MC cells were pretreated with 100 μmol/L of melatonin at 37°C in a humidified atmosphere with 5% CO2 for 4 hours, 39,40 and then exposed to PBDEs. Styrene oxide (100 μmol/L), a known DNA damaging compound, was used as positive control. The Comet assay was carried out only when cell viability was ≥70%, as indicated by Singh et al. 41 Tail intensity (TI%, percentage of DNA in the tail region of the comet) was chosen to represent the genotoxic effect. Cells were seeded at a concentration of 1 × 105 cells/mL in 1 mL wells 24 hours before treatment with PBDEs. After 4 or 24 hours of treatment at 37°C, cells were washed in Hank balance salt solution, mixed with 90 μL of 0.7% low-melting agarose (LMA), and then transferred onto microscope slides previously dipped in 1% normal melting agarose. After 15 minutes at 4°C, a final layer of LMA (0.7%) was added. Cell lysis was carried out at 4°C overnight in a buffer containing 2.5 mol/L NaCl, 10 mmol/L Na2EDTA, 10 mmol/L Tris–HCl, 1% Triton X-100, and 10% DMSO, pH 10. DNA unwinding (20 minutes) was performed in an alkaline buffer (1 mmol/L Na2EDTA, 300 mmol/L NaOH, pH > 13), and electrophoresis was carried out for 20 minutes (0.78 V cm−1, 300 mA). After treatment with neutralizing solution (0.4 mol/L Tris–HCl, pH 7.5), slides were stained with 75 μL of ethidium bromide (10 μL/mL). Samples were examined at ×400 magnification under a Leica DMLS fluorescence microscope (excitation filter BP 515–560 nm, barrier filter LP 580 nm), using an automatic image analysis system (Comet Assay III—Perceptive Instruments Ltd).
Modified Comet Assay for Detection of Oxidative DNA Damage
After cell lysis, the slides, prepared as described above, were washed 3 times with the enzyme buffer (0.1 mol/L KCl, 0.5 mmol/L Na2EDTA, 40 mmol/L HEPES [4-[2-hydroxyethyl]-1-piperazineethanesulfonic acid], 0.2 mg/mL bovine serum albumin, pH 8 with KOH], and incubated at 37°C for 30 minutes with FPG. In the Comet assay, the use of lesion-specific endonucleases allows the identification of oxidative DNA alterations. This damage can be detected using lesion-specific endonucleases to create breaks at the sites of damage, as is the case with FPG which recognizes the common oxidized purine 8-oxoG, as well as ring-opened purines, or formamidopyrimidines (Fapy). 38,42 The DNA unwinding migration (40 minutes) and electrophoretic migration (30 minutes) were performed in a buffer at pH 12.1 (0.78 V cm−1, 300 mA), as described by Collins et al. 43 The use of different unwinding/electrophoresis time and pHs allows the identification of different pattern of DNA damage. For example, unwinding and electrophoresis at pH 12.1 to 12.4 facilitates the detection of single- and double-strand breaks and incomplete excision repair sites, whereas unwinding and electrophoresis at a pH > 13 express alkali labile sites, in addition to all types of lesions described above. Slides were washed with a neutralization solution, stained and analyzed with an automatic image analysis system (Comet Assay III; Perceptive Instruments Limited, Haverhill, Suffolk, UK). Hydrogen peroxide (100 μmol/L) was used as a positive control.
Statistical Analysis
Samples were coded and evaluated blindly, and experiments were repeated at least 3 times. One hundred cells per sample, selected at random, were analyzed, and the TI% average values were calculated. The SPSS 18 (SPSS Inc, Chicago, Illinois) statistical package was used to determine the statistical differences between samples. Variance analysis (analysis of variance) was done by one or multiple pairwise comparisons and the data obtained were also subjected to Student t test (Bonferroni version).
Results
Cytotoxicity of BDE-47 and BDE-209 in SK-N-MC human neuroblastoma cells was assessed by MTS assay and by Trypan blue exclusion. Cells were exposed for 24 hours to different concentrations of either PBDE (0, 5, 10, and 20 μmol/L), chosen on the basis of experiments reported in the literature. 1,26,44,45 Results obtained with both assays indicate that either PBDE can induce a concentration-dependent decrease of cell viability (Table 1). However, the effects were small and not statistically significant. Since the cell viability was always greater than 70%, DNA damage was assessed by the Comet assay. 41
Effect of BDE-47 or BDE-209 on Cell Viability a
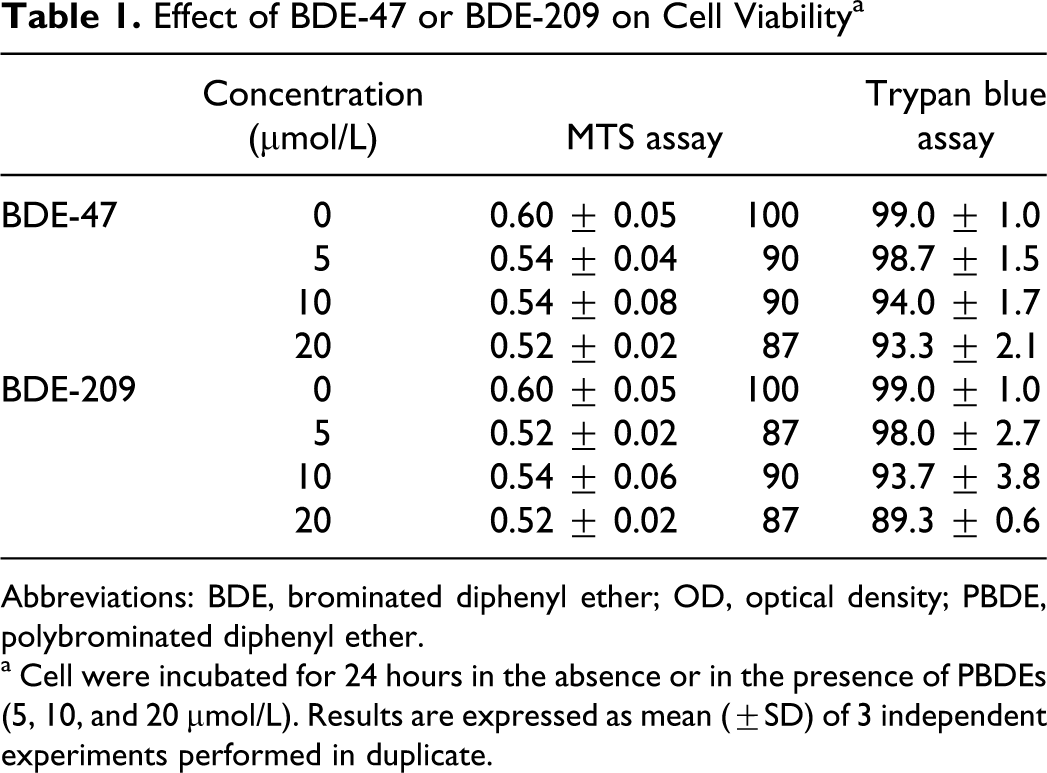
Abbreviations: BDE, brominated diphenyl ether; OD, optical density; PBDE, polybrominated diphenyl ether.
a Cell were incubated for 24 hours in the absence or in the presence of PBDEs (5, 10, and 20 μmol/L). Results are expressed as mean (±SD) of 3 independent experiments performed in duplicate.
The SK-N-MC cells, treated for 4 or 24 hours at 37°C with different concentrations of BDE-47 or BDE-209, were analyzed by the alkaline Comet assay (pH > 13) for the evaluation of DNA damage. This protocol allows the detection of DNA single- and double-strand breaks, of alkali-labile sites, and of single-strand breaks associated with incomplete excision repair sites. 38 Figure 2 shows that both PBDEs induce a concentration-dependent DNA damage, with BDE-47 being more potent (by about 2-fold) than BDE-209. Significant DNA migration was observed in cells treated for 4 hours, and this was significantly decreased after 24 hours, suggesting that neuroblastoma cells may activate DNA repair pathways following PBDE insult (Figure 2A and B). An exception was the results obtained with the highest concentration of BDE-209 (20 μmol/L), where no repair was observed (Figure 2B).
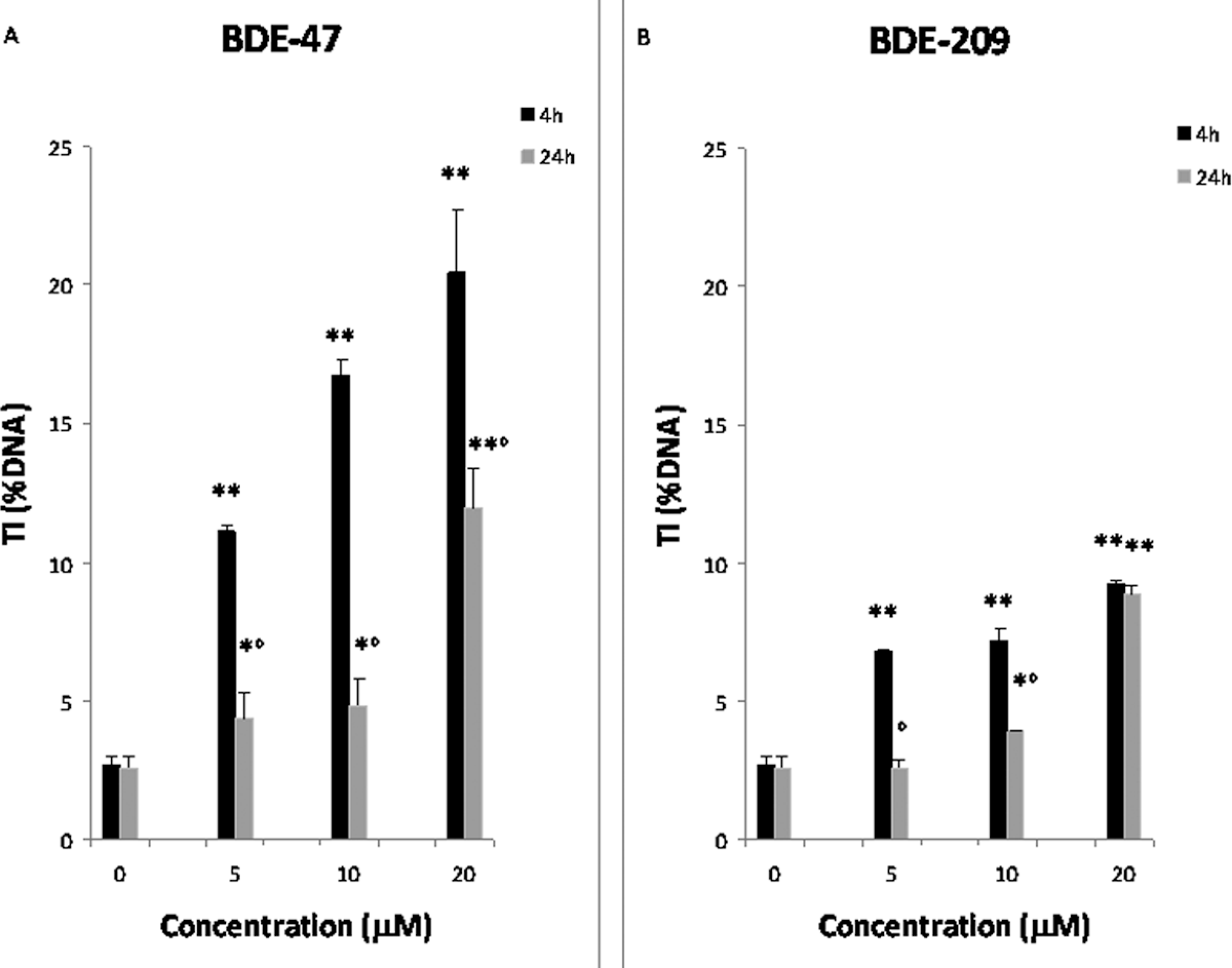
DNA damage detected by the Comet assay (pH > 13) in SK-N-MC cells treated with 5, 10, and 20 μmol/L BDE-47 (A) or BDE-209 (B) for 4 or 24 hours. DNA damage is expressed as percentage of DNA in the tail region of the Comet (TI%). Results represent the mean (±SD) of 3 independent experiments. Significantly different control *P < .05; **P < .01. Significantly different from 4 hours, °P < .05. BDE indicates brominated diphenyl ether; SD, standard deviation; SK-N-MC cells, human neuroblastoma cells; TI, tail intensity.
Various studies have shown that PBDEs can cause oxidative stress, 25 –29 which is thought to mediate some of the observed effects of these compounds on DNA. 2 To determine whether DNA damage detected by the Comet assay was a consequence of oxidative stress, SK-N-MC cells were pretreated with the antioxidant melatonin (100 μmol/L for 4 hours 39 ) before exposure to BDE-47 or BDE-209 (20 μmol/L for 4 hours). The results show that melatonin significantly inhibits DNA damage induced by both BDE-47 (98%) and BDE-209 ([70%] Figure 3).
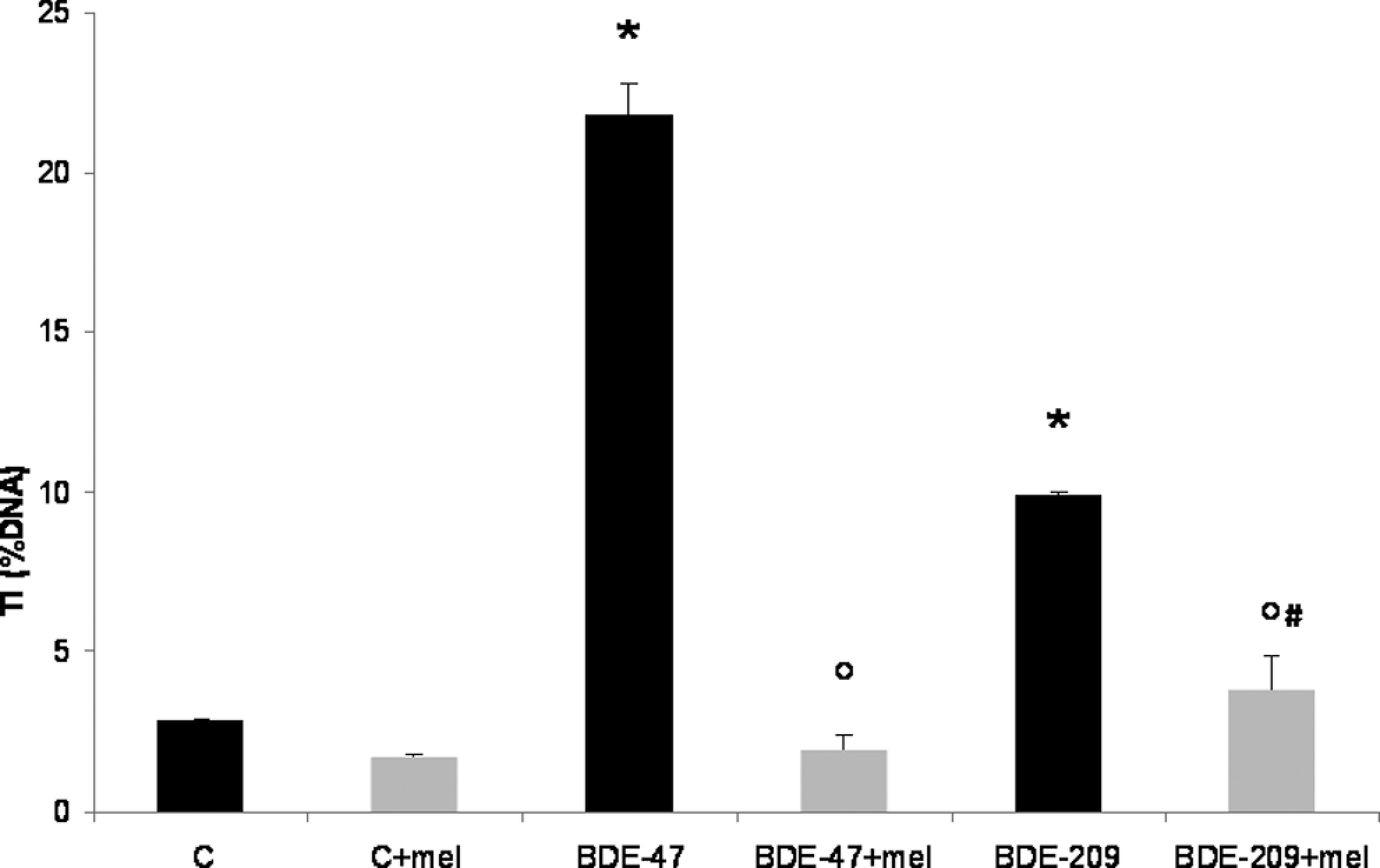
DNA damage detected by the Comet assay performed at pH > 13 in SK-N-MC cells pretreated (4 hours) with melatonin (100 μmol/L), and then exposed to BDE-47 or BDE-209 (20 μmol/L) for 4 hours. DNA damage is expressed as percentage of DNA in the tail region of the Comet (TI%). Results are the mean (±SD) of 3 independent experiment. Significantly different control *P < .05. Significantly different from either PBDEs alone °P< .05. Significantly different from melatonin alone # P < .05. BDE indicates brominated diphenyl ether; PBDE, polybrominated diphenyl ether; SD, standard deviation; SK-N-MC cells, human neuroblastoma cells; TI, tail intensity.
As further evidence that DNA damage induced by PBDEs was secondary to induction of oxidative stress, we measured the presence of oxidized DNA bases utilizing a modified protocol of the Comet assay. 43 This variant of the method includes an extradigestion phase of the DNA nucleoids with bacterial repair endonuclease (FPG), an enzyme that recognizes a particular kind of oxidative damage along the nucleotide chain where it creates a break. 42,43 As specified in the method by Collins et al, 43 to prevent a nonspecific alkali-labile sites occurrence, the Comet assay with the endonuclease was carried out at pH 12.1, to detect only the DNA breaks introduced by FPG. Results of these experiments show that BDE-47 and BDE-209 induce purine oxidation, revealed by FPG, particularly after a short-term treatment (4 hours), with a decrease after long-term exposure (24 hours; Figure 4). As found previously (Figure 2), BDE-47 was more potent than BDE-209 in inducing purine oxidation (Figure 4). Furthermore, repair at 24 hours was much less pronounced in case of BDE-209 (Figure 4).
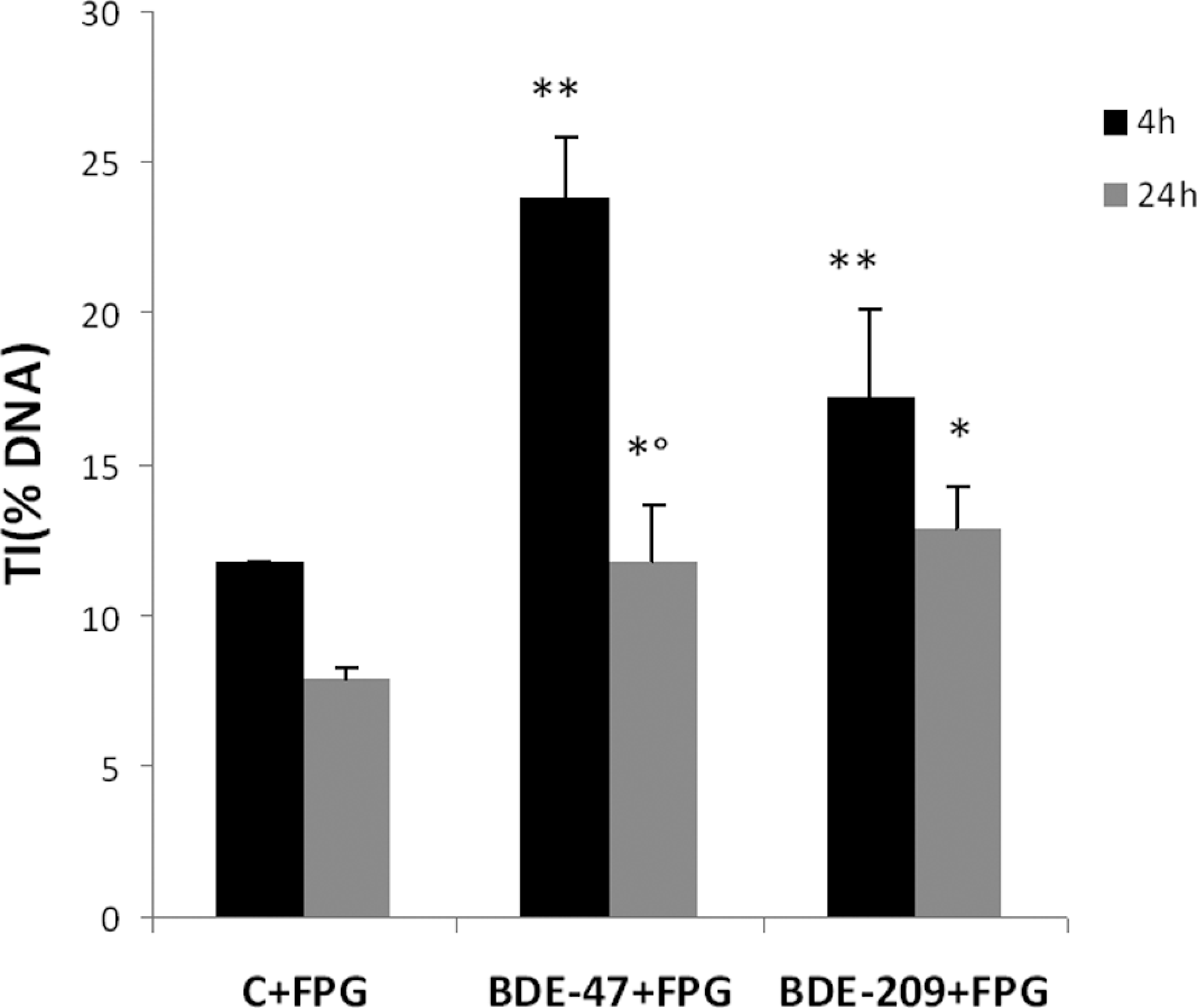
DNA damage assessed by the modified Comet assay (pH = 12.1) for detection of oxidized bases with the use of the bacterial repair endonuclease FPG, in SK-N-MC cells treated with BDE-47 or BDE-209 (20 μmol/L) for 4 or 24 hours. DNA damage is expressed as percentage of DNA in the tail region of the Comet (TI%). Results are mean (±SD) of 3 independent experiments. Significantly different from control *P < .05 and **P < .01. Significantly different from 4 hours, °P < .05. BDE indicates brominated diphenyl ether; PBDE, polybrominated diphenyl ether; SD, standard deviation; SK-N-MC cells, human neuroblastoma cells; TI, tail intensity.
Discussion
The PBDEs are a group of chemicals used as flame retardants in a variety of polymer resins and plastics, which persist in the environment where they undergo bioconcentration and biomagnification. 2,12 Several studies suggest that exposure to these chemicals may result in adverse health effects, especially in infants, as PBDEs may exhibit potential endocrine disrupting effects and developmental neurotoxicity. 2,3,12,20 One of the mechanisms by which PBDEs exert their neurotoxicity has been suggested as being their ability to induce oxidative stress. 25,27,29,46
In the present study, we investigated whether oxidative stress produced by BDE-47 and BDE-209, 2 of the congeners most present in the environment, in human tissues, and in foods, may induce DNA damage in human neuroblastoma cells. The 2 congeners induced only limited cytotoxicity in SK-N-MC cells at the concentrations tested, with a maximal decrease of cell viability of 13% after 24 hours of exposure (Table 1).
Both PBDEs induced a significant DNA migration, which was dependent upon their concentration and the duration of exposure (Figure 2A and B). The results with BDE-47 confirm previous studies, 28,29,47 which observed DNA damage with this congener. Our novel results with BDE-209 further indicate that this congener can cause a similar damage. The BDE-209 was less potent than BDE-47 in inducing DNA damage, in agreement with a recent study which reported a general lower biological activity for BDE-209. 26 For both congeners, the DNA migration observed after a 4-hour treatment was significantly higher than that observed after 24 hours, suggesting a repair of DNA damage. However, minimal repair was observed at 24 hours after the highest concentration of BDE-209 (20 μmol/L). This may be due to kinetic differences in the accumulation of BDE-209 in cells 26 or by still unexplored effects of BDE-209 on enzymes involved in DNA repair.
To determine whether the observed DNA damage was due to the induction of oxidative stress, SK-N-MC cells were pretreated with the antioxidant melatonin before the addition of PBDEs. As shown in Figure 3, melatonin was able to protect SK-N-MC cells from oxidative DNA damage induced both by BDE-47 and BDE-209. The results with BDE-47 confirm those obtained by Gao et al, 47 who reported that the antioxidant N-acetyl cysteine was able to prevent DNA damage caused by this congener. Previous studies had suggested that PBDEs’ cytotoxicity and apoptosis were mediated by oxidative stress. 25,28,29 Huang et al 26 showed that of 5 PBDE congeners, BDE-47 was one of the most potent in inducing ROS formation, while BDE-209 appeared to be the least potent. Furthermore, in case of BDE-209 protection by melatonin was only partial (Figure 3), suggesting that this compound may also induce DNA damage through as yet unidentified different mechanisms.
Additional evidence for an involvement of oxidative stress in DNA damage induced by PBDEs was provided by experiments with FPG. The Comet assay performed with the bacterial repair enzyme FPG shows that both compounds act through the induction of oxidized DNA bases; BDE-47 and BDE-209 oxidize purines, and BDE-47 appears to be more potent than BDE-209, in agreement with our previous observations. Furthermore, oxidative DNA damage was partially repaired 24 hours after exposure to both congeners, but repair after BDE-209 (20 μmol/L) was less, confirming the findings shown in Figure 2. Oxidative stress-induced DNA damage, in particular guanine oxidation (8-oxoguanine [8-oxoG]), may affect the development of embryo and fetus and alter neurodevelopmental processes through several mechanisms, such as alterations in gene transcription. 48,49 In particular, 8-oxoG is a miscoding lesion that induces G:C to T:A transversion mutations, which may induce loss of genomic integrity and cellular regulation, leading, for example, to increased susceptibility to cancer and neurodegenerative diseases. 49 Oxidative damage has been suggested in other cases to play a role in developmental neurotoxicity. 50 –52 However, whether oxidative stress and DNA damage are involved in the developmental neurotoxicity of PBDEs remains to be further investigated. Indeed, PBDEs have been shown to cause endocrine disruption and to interfere with cell-signaling pathways, all of which can contribute to their developmental neurotoxic effects. 2,3,12
In summary, the present study shows that the 2 major PBDE congeners induce oxidative DNA damage in human neuroblastoma cells. While the results with BDE-47 confirm and expand those previously obtained by He et al, 28,29 a novel finding is the determination of DNA damage caused by BDE-209. This compound is less potent than BDE-47, most likely due to its lower ability to accumulate in cells, 26 but appeared to induce a hard-to repair damage at higher concentrations, and may possibly damage DNA by more than one mechanism. Although BDE-47 has been banned in most countries, and production of BDE-209 will be discontinued in the United States in 2013, they will undoubtedly remain long-term environmental pollutants. In this regard, their ability to damage DNA should be further investigated in the context of developmental neurotoxicity and of other toxicity end points.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
