Abstract
The results of 18 months mouse and 24 months rat carcinogenicity studies with the oral direct thrombin inhibitor ximelagatran are presented. In the mouse, gavage doses of ximelagatran up to 180 μmol/kg per d produced no neoplastic changes in any of the tissues examined. In the rat, gavage doses up to 240 μmol/kg per d produced multiple macroscopically detectable nodules in the pancreas, which are seen to be focal/multifocal acinar cell hyperplasia and focal/multifocal acinar cell adenoma upon histological evaluation. There were no other treatment-related effects on tumor incidence or distribution in the rat. The studies show a clear species difference in pancreatic effects between the rat and the mouse to long-term treatment with ximelagatran.
Keywords
Introduction
Ximelagatran has been shown to be active as an anticoagulant in preclinical and clinical models. It is a prodrug that is rapidly converted in vivo to melagatran, an inhibitor of the serine protease thrombin (Ki = 2.0 nmol/L). Melagatran shows high selectivity for thrombin with Ki values for other serine proteases chymotrypsin and elastase greater than 100 nmol/L for each but is also capable of inhibiting trypsin (Ki = 3.6 nmol/L). 1
Trypsin inhibition has long been known to increase pancreatic exocrine secretion and to have pancreatic growth stimulatory effects in the rat. Short-term exposure (up to 3 weeks) to certain dietary raw protein sources, including soybean or cowpea, has been shown to produce rapid pancreatic enlargement in rats, and this effect has also been produced by isolated inhibitor concentrates from soy or potato. 2 –5 Additional observations include hypertrophic and hyperplastic changes, increased organ weight, and increased enzyme secretion in the pancreas. Long-term feeding with soybean trypsin inhibitors or raw soya flour (duration of 1 year or longer) in the rat produces sustained enlargement leading to preneoplastic changes and pancreatic neoplasia. 3,4,6 –8 Trypsin inhibitors, such as the Kunitz-type and Bowman-Birk inhibitors isolated and characterized from the soybean, are considered responsible for the effects. 9 –11 Similar observations in the rat have been made following short- and long-term treatment with the synthetic specific trypsin inhibitor camostat. 12 –16
The mechanism for the effects in the rat pancreas is thought to be the result of trypsin inhibition in the duodenum causing increased cholecystokinin (CCK) release, which produces stimulatory effects on the rat pancreas via CCK1 receptors. 12,17 –26 Sustained inhibition of trypsin produces continuous stimulation of the pancreas through a feedback loop, eventually resulting in nodules/tumors in the rat.
Although these phenomena are well documented for the rat, there are species differences in response to dietary trypsin inhibition. 2,27,28,29 As an example, the mouse is susceptible to the hypertrophic and hyperplastic effects of dietary trypsin inhibition mediated by raw soy flour 28 or camostat. 30 Yet the mouse and also the hamster have been shown to be resistant to the formation of pancreatic nodules and tumors. 24,28
We present here the results of the carcinogenicity studies in mice and rats with ximelagatran, a specific inhibitor to thrombin and trypsin, independent of any effects on other gastrointestinal serine proteases.
Materials and Methods
Ximelagatran (molecular weight: 473.6 g/mol) was supplied by AstraZeneca LP (Molndal, Sweden). Purity of the batches used in the studies ranged from 97.6% to 99.3% by high-pressure liquid chromatography (HPLC) analysis. The test compound was dissolved in purified water and the pH adjusted to 3.0 with hydrochloric acid. Tap water served as the control formulation. Formulations were stored at approximately −20°C.
Analyses for melagatran were performed by a validated liquid chromatography/mass spectrometry procedure.
Animals and Animal Husbandry
All animals were managed under the criteria for humane treatment as set forth by relevant national legislation (Sweden and the United Kingdom) and care was subjected to review of the Institutional Animal Care and Use Committee (IACUC) at AstraZeneca LP.
On arrival at the laboratory and before assignment to the study, animals were subjected to a visual inspection; any animal showing signs of disability was excluded. The air in the housing rooms was changed about 15 times per hour, and temperature and humidity were controlled within approximately 17°C to 23°C and 40% to 70%, respectively. The animal rooms were illuminated by artificial light from fluorescent tubes on a 12-hour light/dark cycle. The bedding material consisted of hardwood chips.
Animals were randomized to balance, as far as possible, the body weight distributions across dose groups. Animals were allowed free access to tap water and pelletized diet (mice: R&M No.1, Special Diet Services, Witham, Essex, UK; rats: RM1.E.SQC low-protein feed for rats and mice, Special Diets Services Ltd, England).
Mice
Male and female CD-1 (Crl:CD-1(ICR)BR) mice (Charles River UK, Ltd, Manston Road, Margate, Kent, UK), approximately 6 weeks of age at start of dosing were used. Body weight ranges at the start of dosing for the carcinogenicity study were from 24.6 to 36.4 g (males) and 20.6 to 30.1 g (females). The animals were acclimatized to laboratory conditions for at least 12 days prior to the start of dosing.
Male mice were singly housed and the female mice were kept 5 in a cage. Clean cages were provided at least once a week.
Rats
Male and female Wistar rats (Mol: WIST; M&B A/S, Denmark), approximately 2 months of age, at the start of dosing were used. Body weight ranges at the start of dosing for the carcinogenicity study were from 180 to 290 g (males) and from 120 to 210 g (females). The animals were acclimatized to laboratory conditions for at least 4 weeks prior to the start of dosing.
Male rats were housed 2 to a cage and the female rats were kept 3 or 4 to a cage. Cages were cleaned at least once a week.
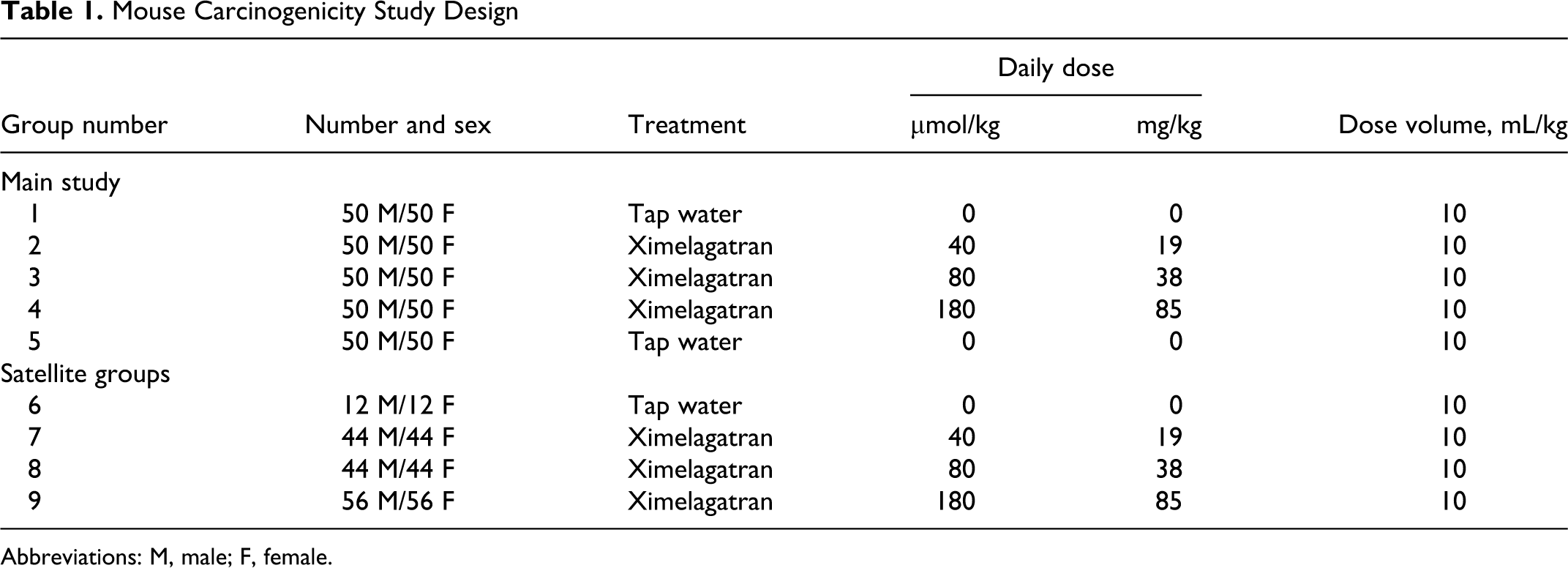
Mouse Carcinogenicity Study Design
The mouse study was conducted at AstraZeneca R&D, Charnwood, UK. The study design was presented to and agreed to by the Carcinogenicity Assessment Committee at the FDA prior to study initiation.
The study design is summarized in Table 1. Groups of 50 male and 50 female mice were dosed with ximelagatran of 40, 80, or 160 μmol/kg per d in the main study. Two further control groups of 50 males and 50 females received the vehicle, purified water.
Mouse Carcinogenicity Study Design
Abbreviations: M, male; F, female.
The dose selection was based on a 3-week and 3-month oral toxicity studies utilizing up to 300 and 120 μmol/kg per d, respectively. Dosing for 3 months at 120 μmol/kg per d was well tolerated but produced changes in hematological parameters associated with the pharmacological properties of the compound on coagulating factors (thrombin time [TT], prothrombin time [PT], and activated partial thromboplastin time [APPT]) and bruising of limbs, ventral surface or head, mainly in the males. In the 3-week toxicity study, 2 weeks of dosing at ≥220 μmol/kg per d caused a low incidence of mortality and loss of body weight occurred, in addition to changes in hematological parameters and indications of bruising and bleeding.
Based on this information, the maximal tolerated dose (MTD) was judged to be 180 μmol/kg per d and was selected as the high dose for the study. The low dose of 40 μmol/kg per d was selected based on toxicokinetics (TK) information to approximate the exposure following therapeutic dosing in humans (3.0 μmol/h per mL) and 80 μmol/kg per d was selected as the intermediate dose.
The test formulations were given orally by gavage, once daily between 8:00
Mortality, clinical signs, body weight, and food and water consumption were monitored routinely throughout the study. Blood samples were collected from all the animals in the main study (animals killed before termination of the study and from all surviving animals at the end of the study), and hematocrit (Hct), erythrocytes (RBC), and white blood cell (WBC) counts were determined by Technicon H1 hematology analyzer.
At the end of the dosing period, mice were humanely killed by sodium pentobarbitone injection. Immediately thereafter, complete necropsies were performed.
Tissue samples for microscopic examination were taken from all main study animals and were preserved in neutral 4% formaldehyde solution, sectioned, and stained with hematoxylin and eosin. For details, see Table 3.
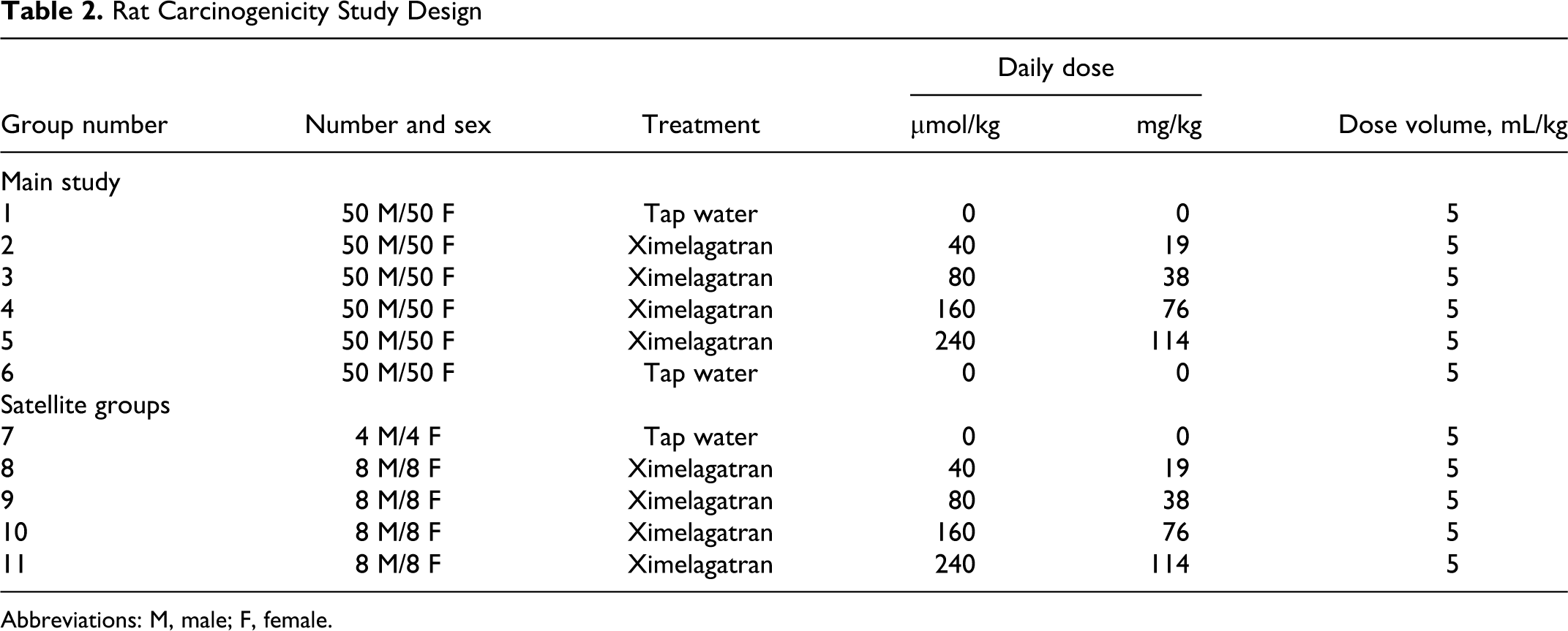
Rat Carcinogenicity Study Design
Abbreviations: M, male; F, female.
Protocol Tissues Sampled at Necropsy for Histopathological Examination

Rat Carcinogenicity Study Design
The rat carcinogencitiy study was conducted at AstraZeneca R&D (Södertälje, Sweden). The study design was presented to and agreed to by the Carcinogenicity Assessment Committee at the FDA prior to study initiation.
The study design for main study animals is summarized in Table 2. Groups of 50 male and 50 female rats were dosed with ximelagatran of 40, 80, 160, and 240 μmol/kg per d. Two further control groups of 50 males and 50 females received the vehicle, purified water. The dose selection was based on the results obtained in previously conducted studies for up to 6 months in rats with ximelagatran and melagatran, the major active metabolite (Fransson-Steen, 1999 & Frankenberg L, 1997, unpublished data). The comparisons of the peak plasma concentration (
Daily oral administration of ximelagatran (160 μmol/kg per d) for 6 months increased plasma coagulation times and produced signs of bleeding in some rats, including hematuria. A slight decrease in RBC, Hct, and hemoglobin was observed in females. Signs of hemorrhage in the abdominal/thoracic cavities or gastrointestinal tract were observed macroscopically in 1 preterminally killed animal after 35 days of dosing, in 1 preterminally dead animal after 130 days of dosing, and in 1 animal terminally killed after 6 months of dosing. The high dose of ximelagatran (240 μmol/kg per d) was included to ensure the achievement of a toxic end point in a lifetime study, since minimal effects were seen at 160 μmol/kg per d in previous studies. Low and medium-low doses of 40 and 80 μmol/kg per d, respectively, were chosen as the fractions of the medium-high and high doses, respectively. All dose levels have been shown to produce dose-dependent prolongations of the coagulation parameters TT, PT, and APTT in rats, as expected from the pharmacological activity of the compound.
The test formulations were given orally by gavage, once daily between 8:00
Mortality, clinical signs, body weight, and food consumption were monitored routinely throughout the study. Ophthalmoscopic examinations were conducted on rats before dosing is commenced, mid-term and after approximately 2 years of treatment routinely throughout the study. Blood samples were collected from all the animals in the main study (animals killed before termination of the study and from all surviving animals at the end of the study), and Hct, RBC, and WBC counts were determined by Cell-Dyn 3500, Abbott Diagnostics (USA), using standard methodology.
After approximately 6 months of dosing, normal and dark/black feces were collected from cages in the control group and in ximelagatran-dosed groups from the study in rats. The samples were stored frozen at approximately −20°C. Occult blood was determined in the collected fecal samples using Hemoccult II, (Kemila, Sweden). In addition, blood samples (approximately 0.5 mL) were taken during terminal necropsy (ie, from surviving animals) to enable the analysis of hormones related to pancreatic function.
At the end of the dosing period, rats were killed under enflurane (Efrane, Abbott Laboratories, USA) and N2O anesthesia. Immediately thereafter, complete necropsies were performed.
Tissue samples for microscopic examination were taken from all the main study animals and were preserved in neutral 4% formaldehyde solution, sectioned, and stained with hematoxylin and eosin. For details, see Table 3.
Statistical Analysis (Both Studies)
Statistical analysis of body weight for each week was performed using analysis of covariance, with the initial body weight as a covariate. The analysis of food consumption was performed for each assessment week as well as for the mean during the whole study, using 1-way analysis of variance (ANOVA). The method of Dunnett was used to adjust for the multiplicity of group comparisons. Hematology data were analyzed by Dunnett tests, and mortality data were analyzed by the log-rank test. Calculations were made using the SAS software v. 8.2. Statistical evaluation of RBC and Hct in preterminally killed animals was performed using 1-way ANOVA with the method of Dunnett to adjust for the multiplicity of group comparison. Calculation of group differences for neoplastic findings was performed using the mortality-adjusted method of Peto. Toxicokinetic data were subjected to ANOVA using the SAS software v. 8.01.
Results
Mouse carcinogenicity study
Survival
There were approximately 50% mice surviving the 18-month treatment period. There was no difference in survival rate between treated mice and controls.
Toxicokinetics
The AUC (0-5 hours) and maximum observed concentrations (
Mean AUC (0-τ) and

Abbreviations: AUC, areas under the mean plasma concentration; M, male; F, female.
aAUC0-0.5 hours.
bAUC0-5 hours.
Preterminal Death or Killing of Mice Treated With Ximelagatran

Abbreviations: M, male; F, female.
Clinical signs
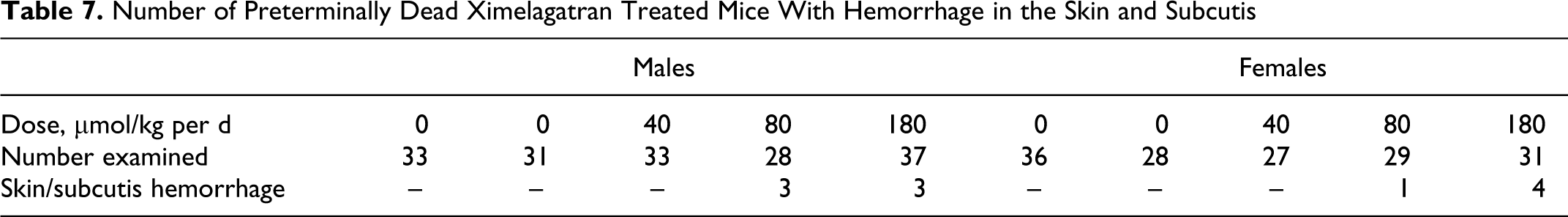
The most prominent clinical signs observed in the study were related to discoloration (bruising) to the body and limbs. There was generally a greater incidence in males (10%-20% incidence) than females (7% incidence) in both 80 and 180 μmol/kg per d dose groups. These findings were consistent with observed pathology findings as shown in Table 7. The bruising was interpreted to be caused by the treatment with ximelagatran, since similar findings observed in previous short-term studies in the mouse could be correlated with the pharmacological effect on coagulating factors (see Mouse Carcinogenicity Study Design section).
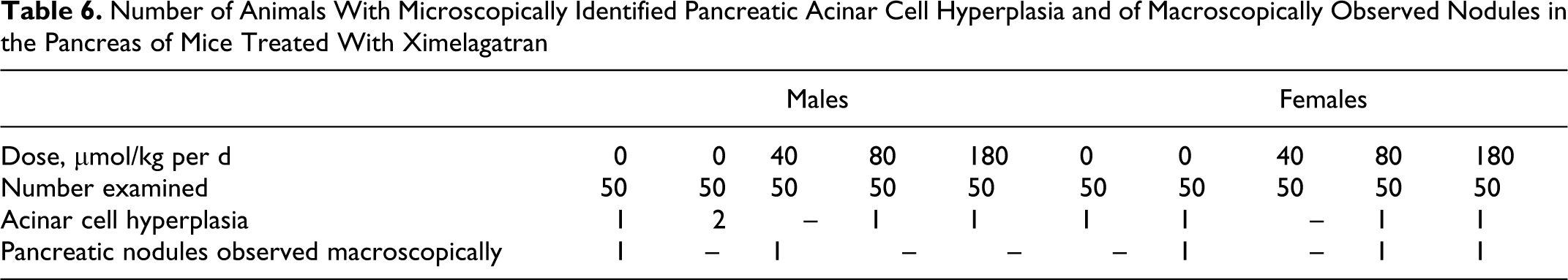
Number of Animals With Microscopically Identified Pancreatic Acinar Cell Hyperplasia and of Macroscopically Observed Nodules in the Pancreas of Mice Treated With Ximelagatran
Number of Preterminally Dead Ximelagatran Treated Mice With Hemorrhage in the Skin and Subcutis
Preterminal mortalities
There were no clear treatment-related effects on mortality in the treated mice (Table 5).
Body weights, food and water consumption, and hematology
Body weights are shown in Figures 1 and 2. There were no treatment-related effects on body weights, body weight gains, or food and water consumption compared with controls. There were no treatment-related effects on hematology parameters.
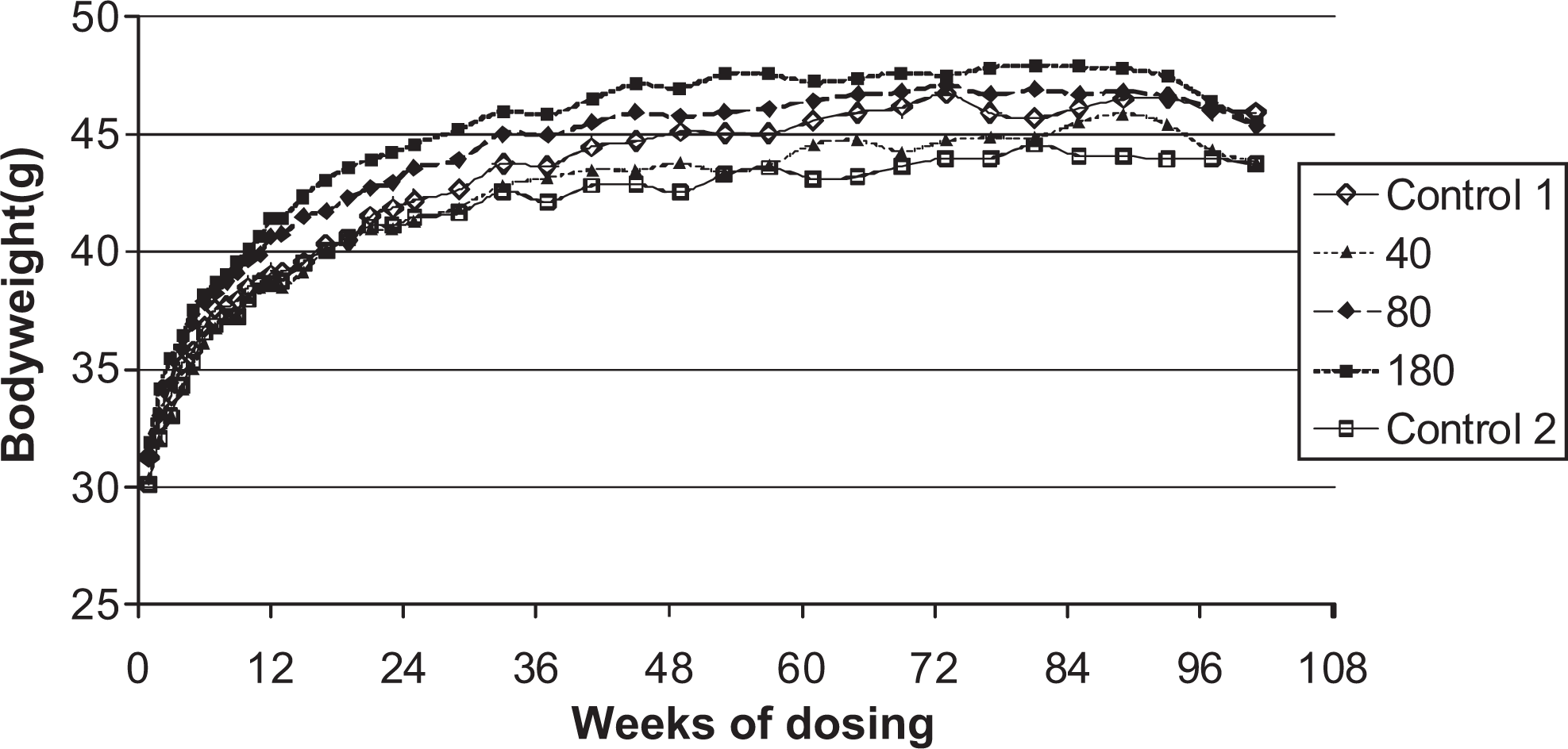
Body weights of male mice given daily oral doses (μmol/kg/day) of ximelagatran for 104 weeks.
Body weights of female mice given daily oral doses (μmol/kg/day) of ximelagatran for 104 weeks.
Pathology
The incidence and distribution of neoplastic changes were not affected by the treatment with ximelagatran. No increases in the incidence of pancreatic nodules or acinar cell hyperplasia were seen (Table 6). There was a low incidence of hemorrhage in the skin and subcutis in animals in the 80 and 180 μmol/kg per d dose groups (Table 7) indicating a dose-related effect.
Rat Carcinogenicity Study
Survival
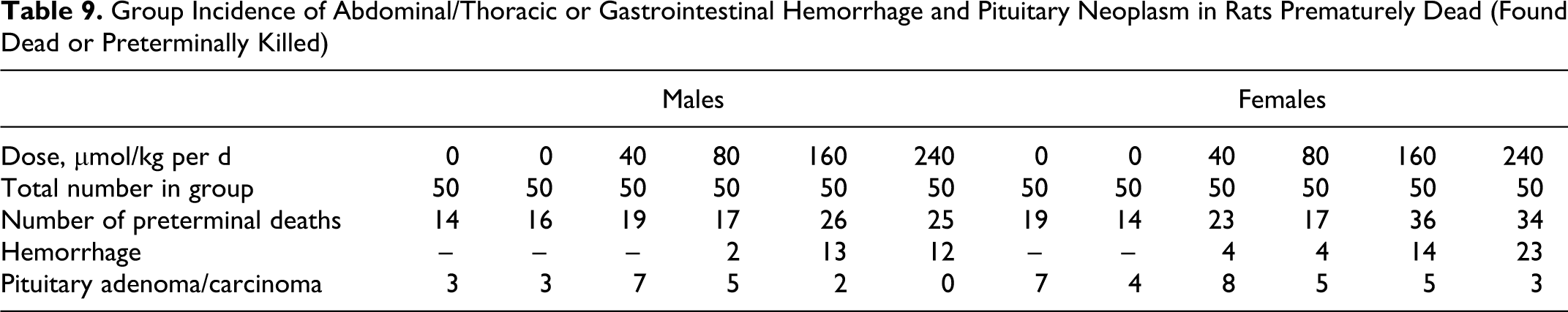
During the 2-year treatment period, there were 44% (males 39% and females 48%) preterminal deaths in the study. In both the sexes, the preterminal mortality increased over time in the 2 highest dose groups with females (68%-75% perterminal deaths) somewhat more affected than males (50%-52% perterminal deaths). In the control groups, the preterminal deaths varied between 28% and 32% for males and 28 and 38% for females.
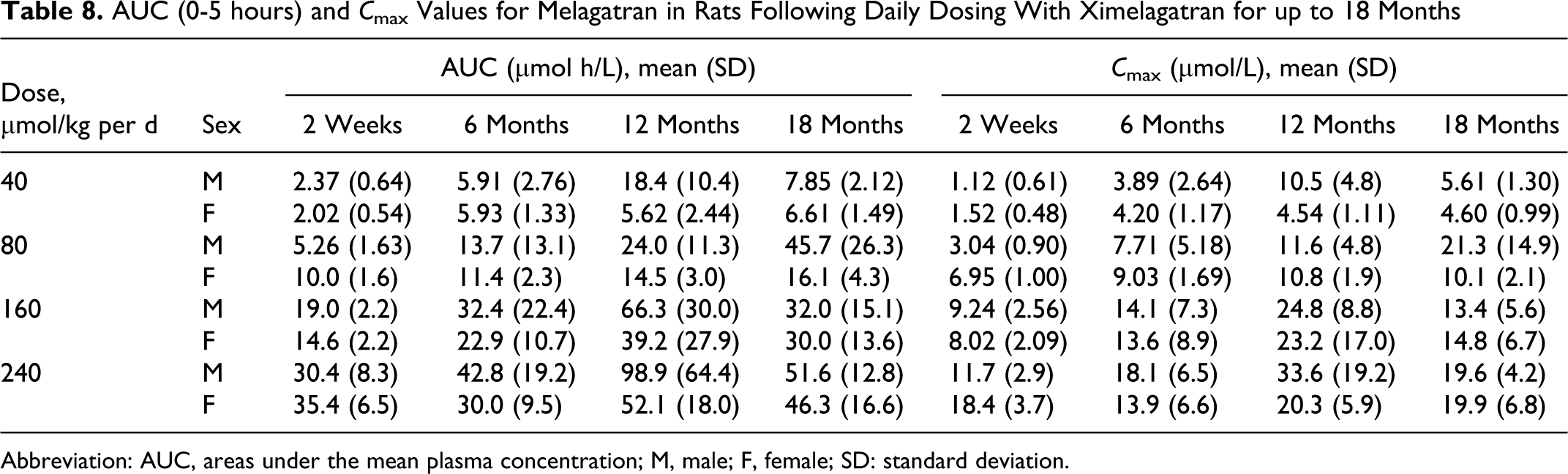
Toxicokinetics
The AUC (0-5 hours) and maximum observed concentrations (
AUC (0-5 hours) and
Abbreviation: AUC, areas under the mean plasma concentration; M, male; F, female; SD: standard deviation.
Clinical signs
The most prominent clinical signs observed in the study were related to an increased tendency to bleeding, such as paleness and/or cages containing dark/black feces. These signs were observed in both the sexes in all the dose groups in a dose- and time-dependent manner. In addition, during the course of study, overt bleeding (eg, from vagina, urethra, skin lesions, or ruptured tumors) was observed in few animals, predominantly in animals in the high-dose groups. Traces of blood were occasionally observed in the bedding material of animals exposed to the test compound.
Preterminal mortalities
In rats, hemorrhage in the abdominal/thoracic cavities or gastrointestinal tract was the most common cause or primary contributor to preterminal death in animals treated with ximelagatran and occurred in a dose-related manner (Table 9). Preterminal death caused by hemorrhage was more common in females than in males. Another common cause of death was tumors in the pituitary gland (adenoma and less common carcinoma). This finding occurred in all the groups, including the controls and is a common tumor in rats with and incidence increasing with age.
Group Incidence of Abdominal/Thoracic or Gastrointestinal Hemorrhage and Pituitary Neoplasm in Rats Prematurely Dead (Found Dead or Preterminally Killed)
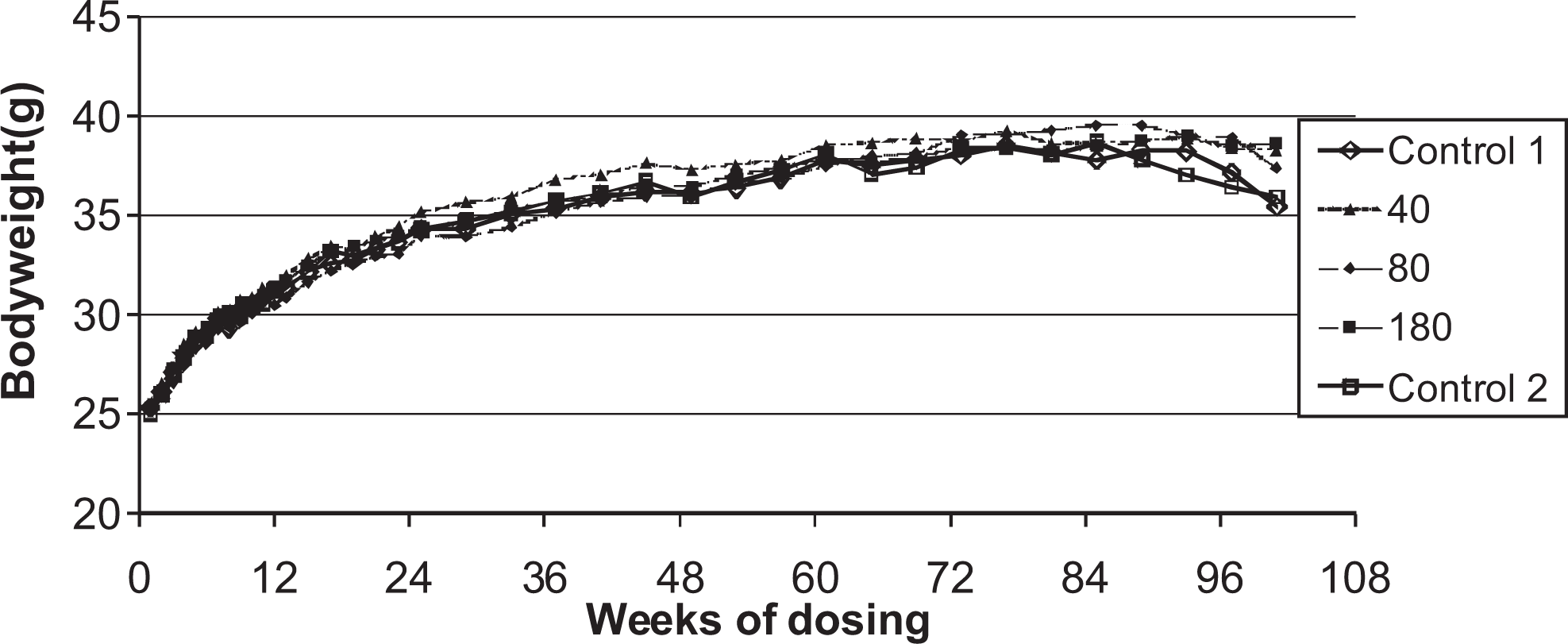
Body weights
Body weights are shown in Figures 3 and 4. In males, the overall body weight gains were lower in all dose groups relative to the control animals. In the high dose males (240 μmol/kg per d), lower mean body weights were observed particularly during the second half of the study. Statistically significant decreased mean body weights were also observed in the low-medium–dose group (80 μmol/kg per d) from week 58 (
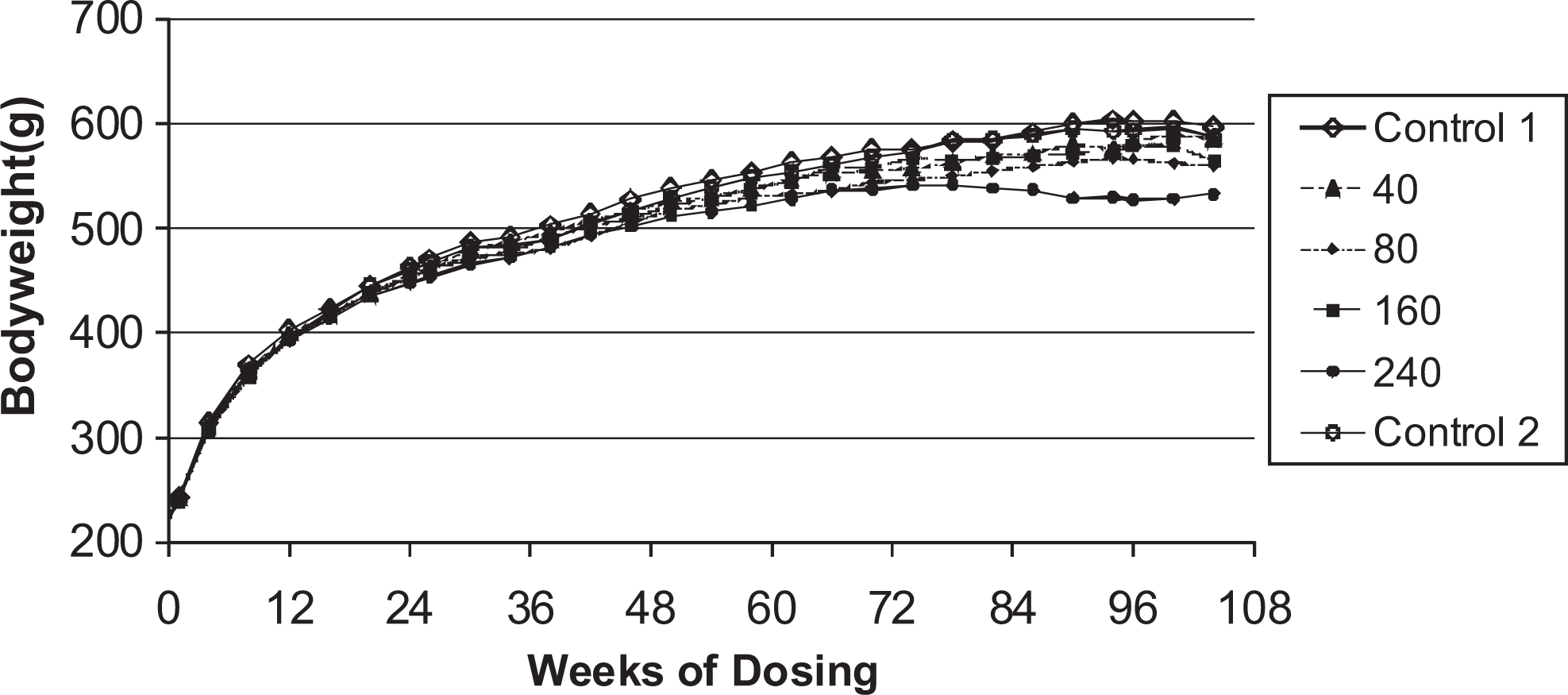
Body weights for male rats given daily oral doses (μmol/kg/day) of ximelagatran for 104 weeks.
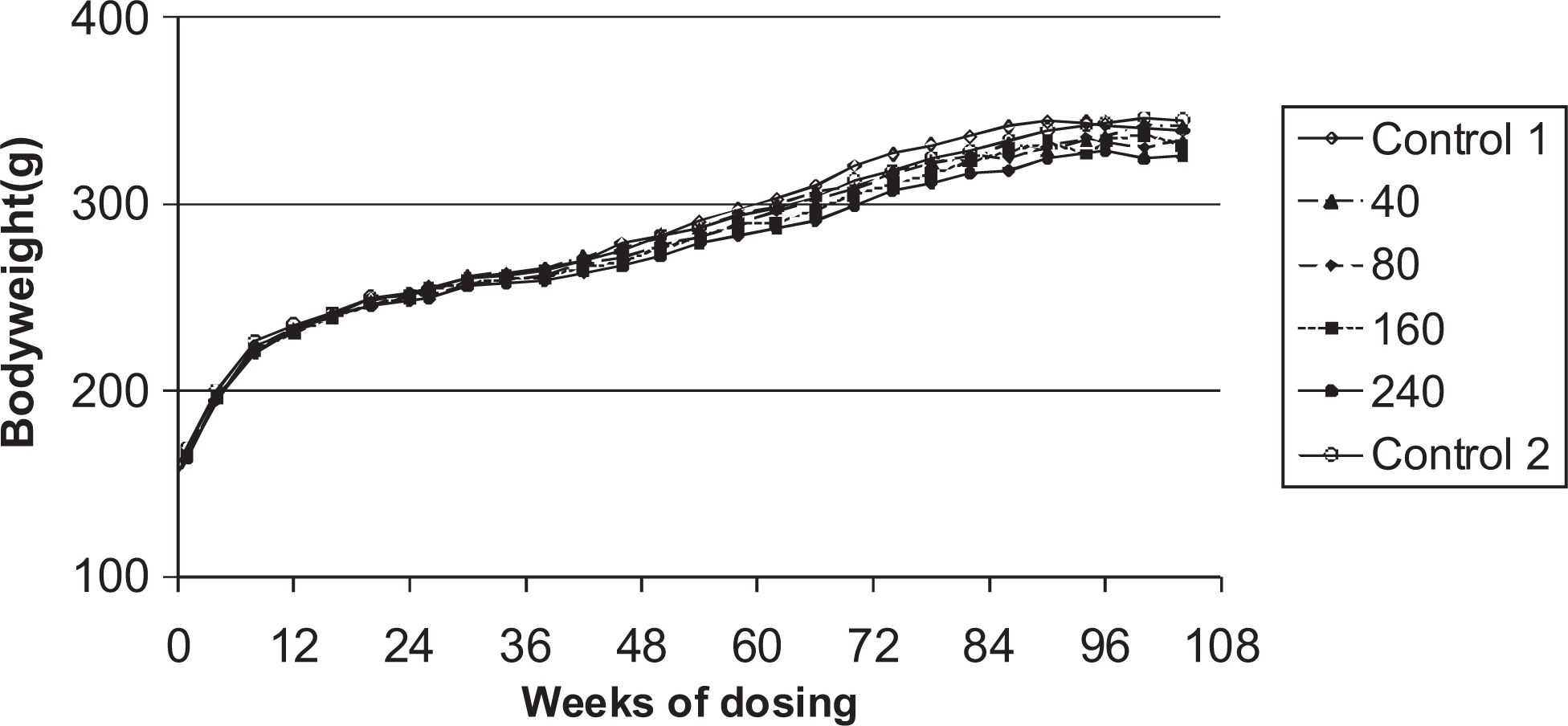
Body weights of female rats given daily oral doses (μmol/kg/day) of ximelagatran for 104 weeks.
Food consumption and ophthalmoscopy
There were no consistent differences in food consumption and no qualitatively different findings in the ophthalmoscopic examinations in treated animals compared with the control animals during the study.
Hematology
The RBC and the Hct values were significantly lower in the preterminally killed animals given with 240 μmol/kg per d of ximelagatran than in the preterminally killed animals in the control groups. The RBC values for control males/females were 7.4/6.7 and for high-dose animals 4.4/4.7, respectively. The Hct values for control males/females were 0.40/0.38 and for 240 μmol/kg per d animals were 0.23/0.28, respectively. Coagulation factors were not investigated as a part of the carcinogenicity bioassay due to the limitations of blood sampling and because the effects on coagulation factors were well established in rats (See Rat Carcinogenicity Study Design section).
Gross pathology
At necropsy, increased incidence of multiple nodules (size ranging from approximately 3-15 mm in terminal animals) was observed in the pancreas (Figure 5). The size of the nodules increased with the dose and was higher in males than in females. The incidence of nodules was treatment related. The macroscopic findings are summarized in Table 10. There were no other treatment-related findings observed at necropsy.
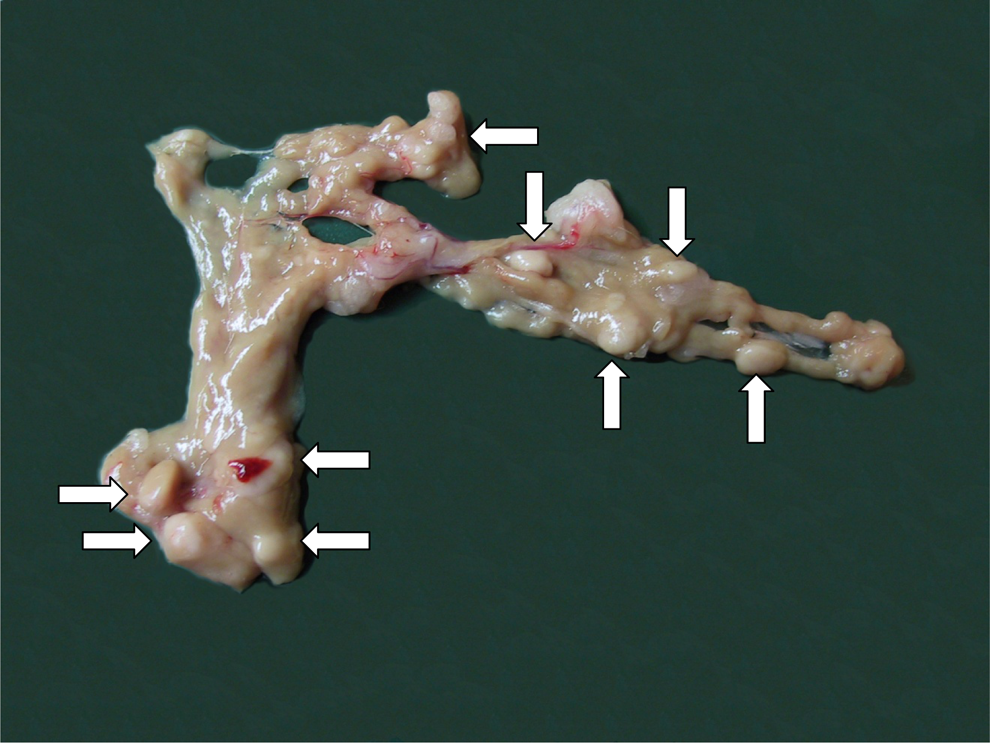
Rat pancreas from animal in the high dose group (240 μmol/kg/dayof ximelagatran). Arrows indicate a few of multiple nodules distributed in the pancreas.
Incidence and Mean Size of Macroscopically Identified Nodules in the Pancreas of Rats Treated With Ximelagatran
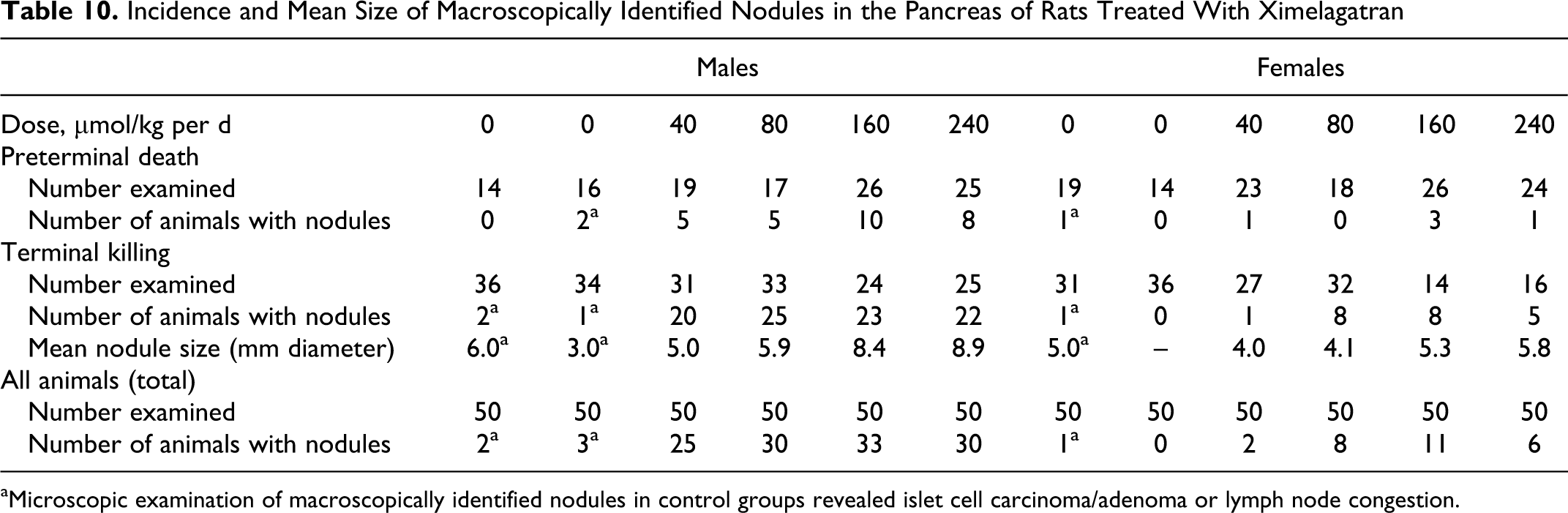
aMicroscopic examination of macroscopically identified nodules in control groups revealed islet cell carcinoma/adenoma or lymph node congestion.
Microscopic pathology
The histological examination of the pancreas revealed a treatment-related increase in the incidence of focal/multifocal acinar cell hyperplasia and focal/multifocal acinar cell adenoma. These findings could be correlated with nodules found at necropsy. The incidence and severity of these findings were higher in males than in females. Toxocokinetic data show that the higher exposure to ximelagatran in male rats relative to females (Table 8) may be the reason for the higher incidence of hyperplasia and adenomas in males. Acinar cell adenomas are histologically similar to acinar cell hyperplasia, except that the lesions with a diameter greater than 3 mm are designated as acinar cell adenomas (Figure 6). 31,32 Acinar cell carcinoma was diagnosed in 4 terminally killed male rats. Table 11 presents a summary of the findings.
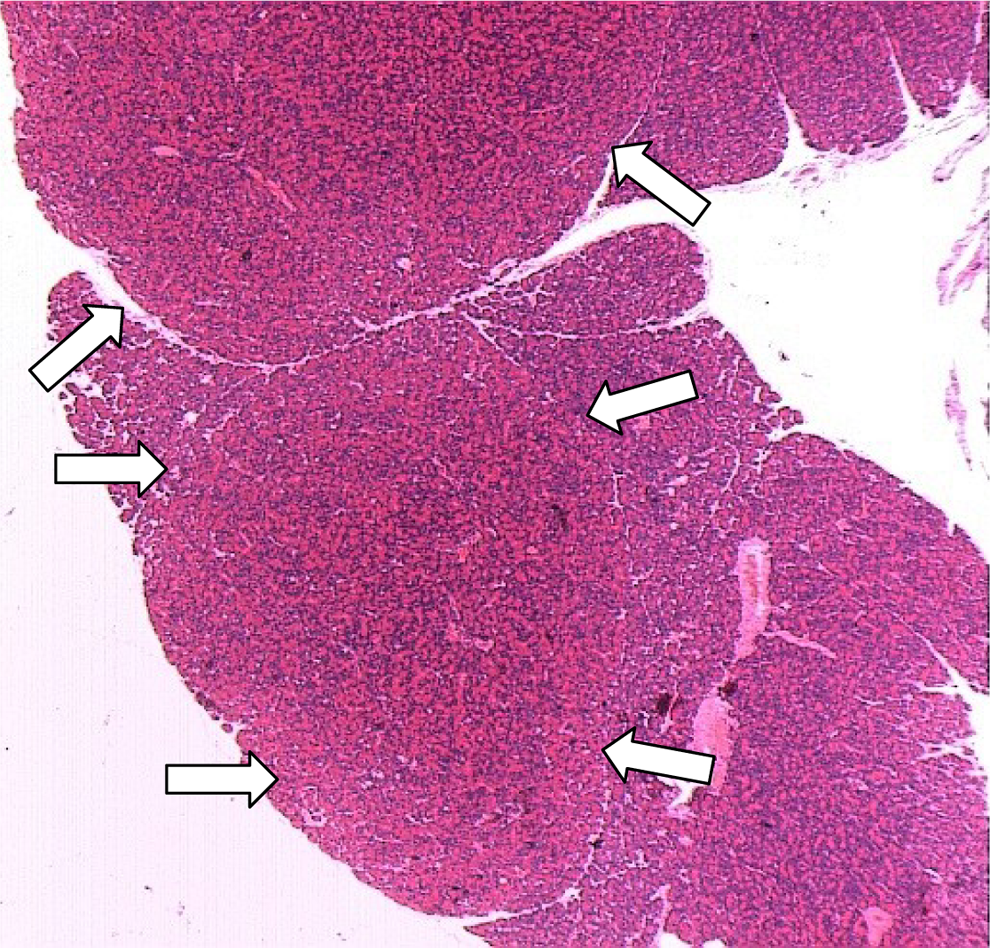
Rat pancreas from an animal in the high dose group (240 μmol/kg/day of ximelagatran). Arrows indicate 2 adenomas of the exocrine pancreas
Treatment-Related Neoplastic/Hyperplastic Findings in Exocrine Pancreas in Rats a
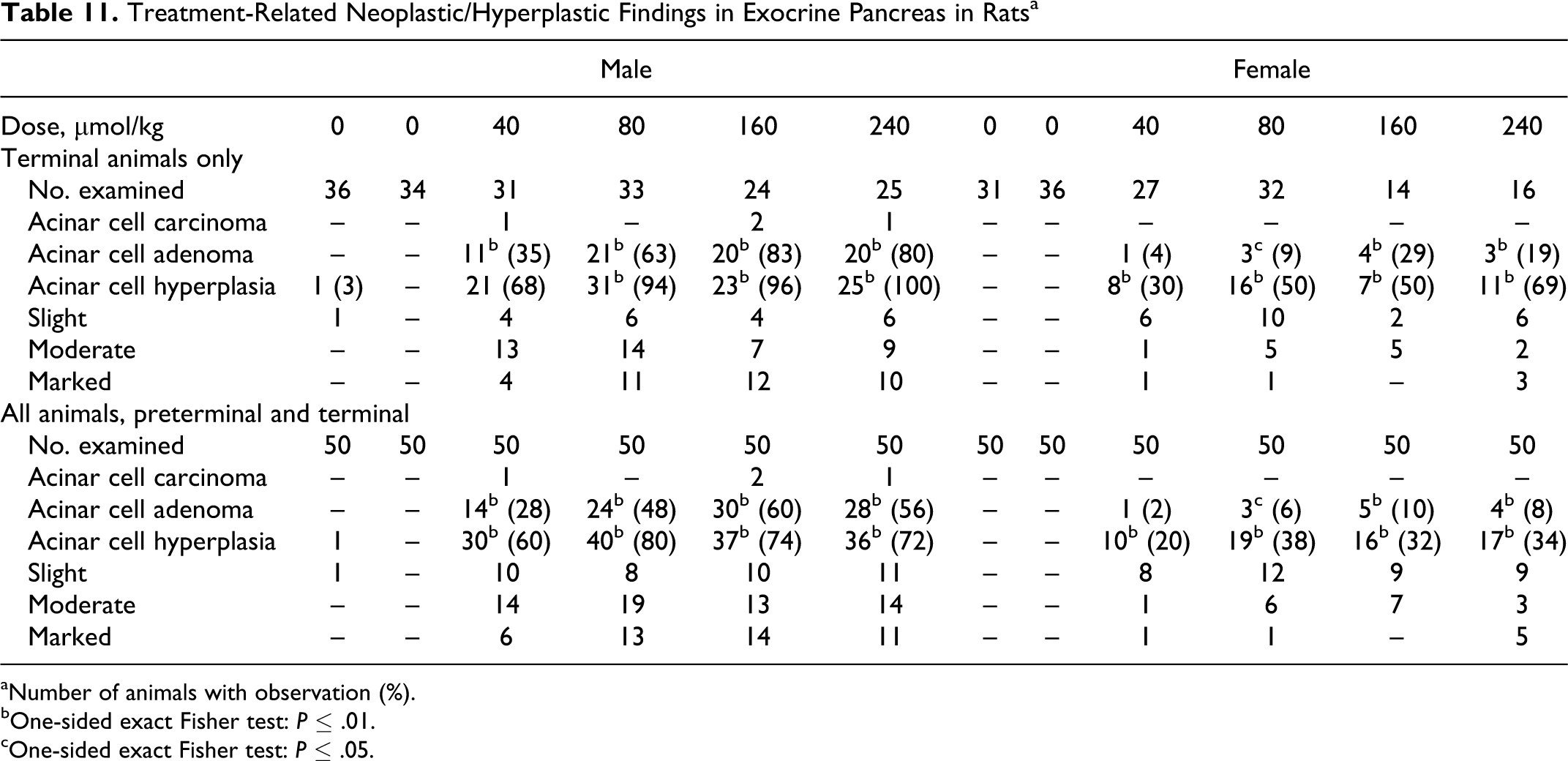
aNumber of animals with observation (%).
bOne-sided exact Fisher test:
cOne-sided exact Fisher test:
In order to evaluate the exposure time to ximelagatran needed for the development of acinar cell hyperplasia/neoplasia incidence, tables on 60-day intervals from the start of the study were created on preterminal animals (Table 12). The results show that the proliferative/neoplastic changes in pancreas occur in the latter part of the study, mainly during the second year, beginning in the higher dose groups of males. The earliest observation of exocrine pancreatic hyperplasia was observed in a male preterminally killed on day 342 of dosing. The earliest identification of pancreatic acinar cell adenoma was observed in a male killed prematurely on day 557 of dosing. These results indicate a time-dependent progression from acinar cell hyperplasia to adenoma and in a few cases to carcinoma.
Time to Observation of Neoplastic/Hyperplastic Findings in Exocrine Pancreas in Rats Prematurely Killed or Found Dead a
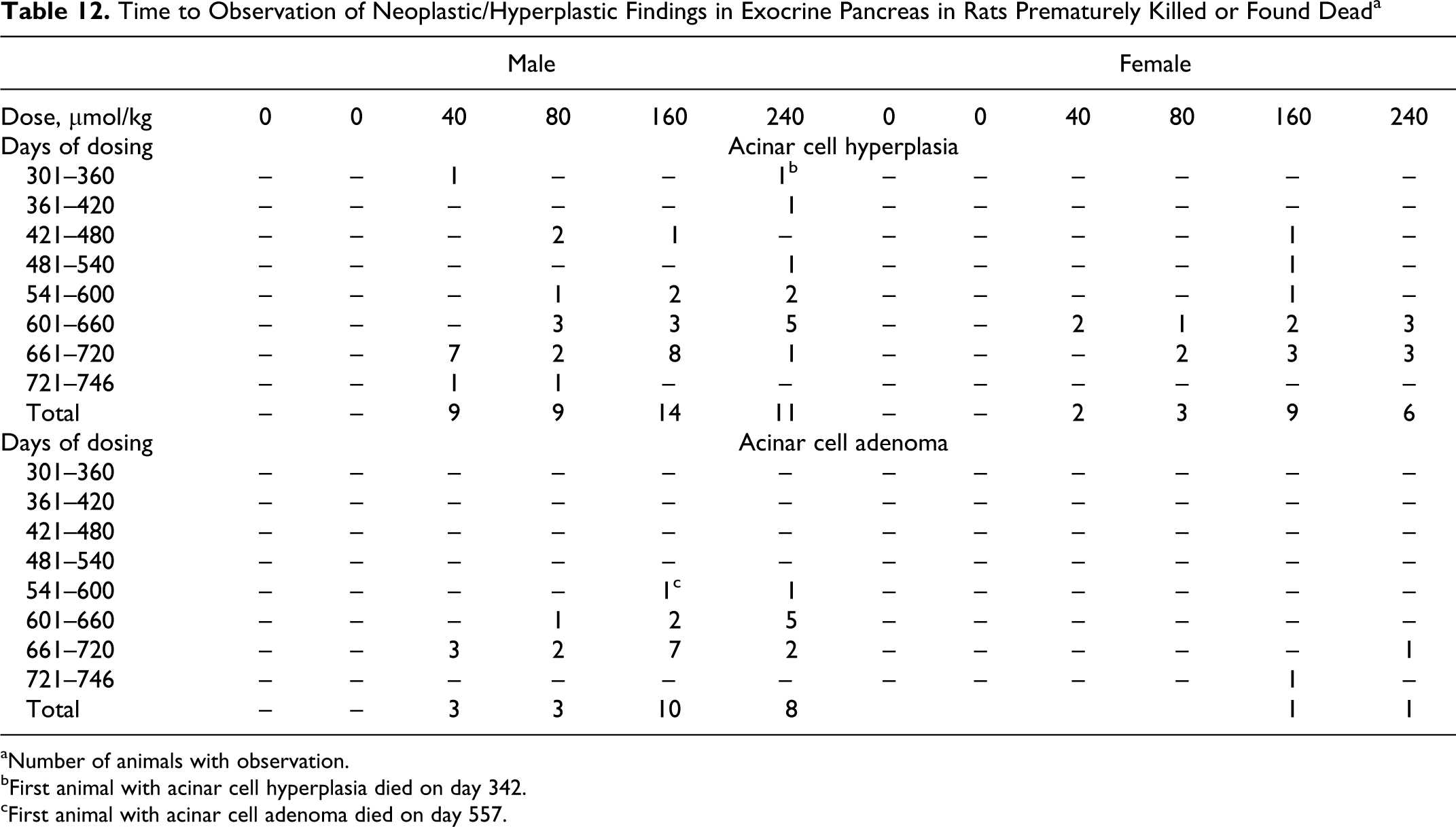
aNumber of animals with observation.
bFirst animal with acinar cell hyperplasia died on day 342.
cFirst animal with acinar cell adenoma died on day 557.
A low incidence of foci of cellular alteration and focal atrophy was observed in the exocrine pancreas of males and females. There was a low incidence of islet cell adenoma, islet cell carcinoma, and islet cell hyperplasia. The incidence of all these findings decreased with the dose. A low incidence of other findings in the pancreas (ductal cell hyperplasia, arteritis/periarteritis, inflammatory cell infiltration/inflammation, peritonis, and vascular mineralization) was not considered related to the treatment.
The incidence and distribution of neoplastic changes in other tissues examined were not considered to be related to treatment with ximelagatran.
Discussion
The results of the 2 carcinogenicity studies with ximelagatran demonstrate that both the studies were adequate for evaluating its carcinogenic potential. Survival was acceptable in both the studies with over 50% of the mice and between 28% and 76% of the rats surviving to term. Mice tolerated all dose levels, the only major observation being bruising in high-dose males, which is the expected pharmacological effect of the compound. Attainment of the MTD in mice was not obvious except for the bruising seen in the high-dose groups and for the mortality seen in the dose range-finding studies at 220 μmol/kg per d. Although not reflected in the mortality rates, lower body weights and increased incidence of bleeding in the higher dose groups support that the MTD had been reached in the rat study. The rats tended to be sensitive to fatal bleeding into abdominal cavities, while the mouse only showed nonfatal subcutaneous bleedings of low incidence. The species difference in this aspect remains unclear.
Except for the bleedings at higher doses, interpreted to be effects on coagulation related to the inhibition of thrombin, and the pancreatic effects seen in the rat related to the inhibition of trypsin, no effects on other organ systems were seen in either study. The pancreatic acinar cell hyperplasia and acinar cell adenoma seen in the rat is consistent with those documented following feeding with serine protease inhibitors such as soy flour or camostat. The occurrence of adenocarcinomas is interpreted to be a progression from adenoma to its malignant counterpart. It is of interest that selective trypsin inhibition with ximelagatran produces similar pancreatic effects, with comparable incidence and timing of appearance, as those seen following feeding with the more broad spectrum serine protease inhibitors. Soy flour and camostat inhibit most serine proteases, 27 while ximelagatran has been shown to be selective for thrombin and trypsin over other members of the class, including chymotrypsin, elastase, and kallikrein. 1
The appearance of pancreatic tumors seen in the rat, but not in the mouse, is also consistent with other published reports. 28,24 However, feeding of broad spectrum serine protease inhibitors has been shown to produce pancreatic hyperplastic responses in both the species, the main difference has been the progression to neoplasia in the rat, but not in the mouse. In these studies with ximelagatran, no treatment-related hyperplasia was evident in the mouse. One possibility for this observation could be related to the selectivity of ximelagatran, in that the hyperplastic response in the mouse pancreas may be dependent on the inhibition of more than one luminal protease.
In any case, the similarities of the observations in these studies with ximelagatran to those in the literature for broad spectrum serine protease inhibitors support the consensus that the progression to pancreatic neoplasia is unique to the rat.
Based on these studies, we conclude that ximelagatran does possess significant carcinogenic potential in the rat, but not in the mouse.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: all the authors were AstraZeneca employees at the time of conduct of the studies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by AstraZeneca Pharmaceuticals, LLC.
