Abstract
Female F344 rats were fed diets containing 0.02%, 0.2%, or 2.0% white mineral oil for 90 days. There were no gross or microscopic differences in target organs at the 0.02% level. In the higher-dose groups, relative liver and mesenteric lymph node (MLN) weights were increased, and MLN inflammation was observed. At the 2% level, there was very limited evidence of microgranuloma formation in the liver but at a lower incidence and at lesser severity than has been reported in studies of C22 -C25 oils. Analysis of liver extracts from treated rats revealed that C15 -C20 constituents were underrepresented by comparison to their corresponding concentrations in the test oil. These results provide evidence that although hydrocarbons with carbon numbers <C20 are absorbed, they are not preferentially retained and do not contribute to inflammatory processes in liver.
Introduction
Highly refined mineral oils, often called white oils in the industry, are widely used in applications in which there is the potential for widespread human contact and ingestion including uses in food processing, cosmetics and personal care products, and pharmaceuticals. 1 These highly refined mineral oils are commonly manufactured from either paraffinic (P) or naphthenic (N) crude oils, by a process which involves distillation at atmospheric pressure followed by a second distillation under reduced pressure to create raw lubricating oil base stocks. The raw lubricating oil base stocks are then refined by solvent extraction to remove the condensed ring polycyclic aromatic constituents and then further refined by treatment, normally via strong acid, and/or hydrogenation. The severity of the refining processes for food grade products removes nearly all aromatics and other impurities (sulfur, oxygen, metals, etc).
Based on historical information that white oils did not produce toxicological effects when repeatedly administered to Long-Evans rats or beagle dogs 2,3 and the absence of human evidence to the contrary, these oils were considered to be safe for their various approved uses. However, questions arose in the late 1980s based on the evidence that these oils produced multifocal lipogranulomas in mesenteric lymph nodes (MLNs) and livers of Fischer 344 (F344) rats following dietary administration for 90 days at levels greater than 500 ppm (approximately 30-60 mg/kg per d). 4 Also noted by Baldwin et al 4 the effects were more pronounced in female than male F344 rats. Further study by Firriolo et al 5 both confirmed and extended the previous data. More specifically, repeated dietary exposure to a P white mineral oil (P15H) resulted in the induction of hepatic and MLN microgranulomas in F344 rats but did not produce effects in Sprague-Dawley (SD) rats treated under similar conditions.
These results raised 3 questions:
which oils (or constituents) are responsible for these effects;
what is the explanation for the species and strain specificity of the effects; and
what are the human health consequences of these findings?
The results of a systematic study to characterize the effects of highly refined mineral oils in F344 rats were reported by Smith et al 6 who tested 7 oils with average molecular weights ranging from 320 (average carbon number of 22) to 510 (average carbon number of 36). The most active oils were those with average molecular weights of 320 to 330 (average carbon numbers 22-23), whereas there were no effects in studies with oils having molecular weights greater than 420 (average carbon numbers >28). These results were then extended by Scotter et al 7 who examined the effects of 3 of these oils in F344 rats and also analyzed the mineral hydrocarbon content (MHC) of liver extracts from treated animals. They reported that the severity of the effects was correlated with the presence in the liver extracts of mineral oil constituents with carbon numbers in the range of C20 -C35 at concentrations greater than those found in the corresponding test substances. It was hypothesized that preferential accumulation of constituents in the C20 -C35 carbon number range by F344 rats led to the development of the characteristic effects of these substances. 7,8 Hydrocarbons with carbon numbers >C35 are not well absorbed 9,10 and are unlikely to make substantial contributions to the development of these effects. These studies as well as other studies of white mineral oils and low melting point waxes 11,12 provided evidence that constituents with carbon numbers <C20 were also not preferentially accumulated, but as these lower molecular weight constituents represented relatively small fractions of the tested oils, the relationship between the characteristic histological changes and low-molecular-weight hydrocarbons was less well defined.
The question of strain specificity has been recently investigated by a pharmacokinetic study 13,14 in which it was shown that F344 rats exhibited higher oral bioavailability of a P15H oil than did SD rats. Further, at dose levels associated with no adverse effects when administered repeatedly in subchronic oral toxicity studies (20 mg/kg in F344 rats and 1500 mg/kg in SD rats), the blood concentrations in both rat strains approximated 6 μg/mL. This study provides evidence that F344 rats have an absorption/distribution/metabolism/excretion (ADME) profile that differs from the one in SD rats; and further that in SD rats, it is not possible to achieve blood levels high enough to result in liver microgranuloma formation. These data are consistent with the hypothesis that increased white oil accumulation is responsible for the effects specific to F344 rats and allow the use of a target tissue dose metric rather than applied dose to reconcile apparent differences in effects between F344 and SD rats. Additionally this target tissue dose metric can be used to assess the relevance of rodent studies to humans. However, human relevance has been dealt with in great detail elsewhere 15 and will not be further discussed in this article.
To further address issues related to lower-molecular-weight hydrocarbons, a dietary administration study was conducted in F344 rats using a white mineral oil with an average molecular weight of 265 (average carbon number C20 ). The toxicological effects of this oil were investigated, and the hydrocarbon contents of liver extracts were characterized. The specific objectives were to test whether after dietary administration, hydrocarbons with relatively low-molecular-weight were preferentially accumulated and to assess the potential of these oils to produce the characteristic histological effects in liver and MLN associated with repeated treatment with mineral oils and waxes which are primarily composed of hydrocarbon components in the range of C20-C35. F344 rats were used specifically in this study because, as the most sensitive strain, the likelihood of observing a response was maximized.
Materials and Methods
Test Animals and Maintenance
Female F344 rats (CDF F-344/CrlBR), approximately 4 weeks of age, were obtained from Charles River Laboratories, Inc Raleigh, North Carolina, and acclimated for 14 days prior to study initiation. The rats were single housed in wire mesh cages with ad libitum access to feed and water. The animal rooms were maintained at a temperature range of 18°C to 22°C and a relative humidity range of 30% to 70%, with a 12:12 light–dark cycle. This study was conducted in accordance with Good Laboratory Practice Guidelines, 16 under US guidelines for the care and use of laboratory animals, 17 and in a laboratory accredited by the American Association for Accreditation of Laboratory Animal Care (AALAC).
Test Material
The test material was a white mineral oil prepared from a P lubricant base oil by hydrogenation. It was a colorless liquid with the following characteristics: viscosity at 100°C = 2.16; carbon number range = C14 -C32, average carbon number = C20; average molecular weight = 265. Of particular importance for this study, approximately 48% of the hydrocarbon constituents had carbon numbers
The nomenclature that has been developed for white mineral oils starts with a single letter (P or N) that indicates whether the oil was derived from a P or N crude oil. This is followed by a number that represents the viscosity in centistokes (cSt) of the oil at 40°C. Finally, there is another letter to indicate whether the final refining step involved hydrogenation (H) or acid treatment (A). Accordingly, using this nomenclature, the test oil used in the present investigation was identified as P7H, a P white mineral oil with a viscosity of 7 cSt @ 40°C and with hydrogenation (H) as the final refining step.
Test Diets
The test material was mixed with feed (PMI Certified Rodent Diet Meal 5002, PMI Feeds, Richmond, Indiana) at concentrations of 0.02%, 0.2%, and 2% by weight. The feed was then thoroughly mixed to assure homogeneity. The stability of the test material in the feed was verified through analyses on days 4, 8, and 15, and the concentrations of test material in the feed were verified after weeks 1, 5, 9, and 13.
Study Design
Female F344 rats were randomly allocated by body weight to 4 groups, each containing 15 animals. The rats were given ad libitum access to control diet or diets containing one of the 3 experimental levels of white mineral oil. Ten rats in each group, used to assess the toxicological properties of the oil, were sacrificed and subjected to gross external and internal examination after 13 weeks of treatment. Selected tissues were removed, examined, and prepared for histological examination as described below. The remaining animals were also sacrificed after 13 weeks of treatment, and the livers were removed to characterize and quantify the hydrocarbon constituent contents.
Observations
In life
The rats were examined for viability twice daily on Monday through Friday and once daily during the weekend. Detailed clinical observations were made weekly for signs of toxicity; the onset, nature, severity and duration of these observations were recorded. Body weights were measured pretest for sorting purposes, prior to dose initiation, on day 0, and weekly thereafter until sacrifice. Feed consumption was measured weekly and used to calculate doses.
Necropsy
On study day 91, rats scheduled for pathological examination were weighed and then exsanguinated under methoxyflurane anesthesia. A gross necropsy including examination of the external surfaces, all orifices, and the cranial, thoracic, and abdominal cavities and their contents was conducted. On study day 96, rats scheduled for hydrocarbon analysis were exsanguinated under barbiturate anesthesia.
Hematology and clinical chemistry
On the day of sacrifice, blood was collected from the abdominal aorta of each rat scheduled for pathological examination. Hematological and serum chemistry analyses were conducted by LabCorp, Burlington, North Carolina. Hematological parameters measured included hematocrit, hemoglobin, erythrocyte count, leukocyte count, platelet count, reticulocyte count, mean corpuscular volume, and mean corpuscular hemoglobin. Serum chemistry parameters measured or calculated included albumin, urea nitrogen, calcium, creatinine, electrolytes (Na+, Cl−, and K+), glucose, phosphorus, gamma-glutamyl transpeptidase (GGT), serum alanine aminotransferase, serum alkaline phosphatase, total protein, total bilirubin, cholesterol, triglycerides, urea nitrogen/creatinine ratio, total globulin, albumin/globulin ratio, and amylase.
Organ weight and histopathology
After gross examination, the identified target organs liver, kidneys, heart, spleen, and MLN, as well as ovaries were removed from all animals scheduled for pathologic examination and weighed. These organs, as well as a number of other organs which were removed but not weighed were preserved in 10% neutral-buffered formalin for fixation. The organs which were preserved included adrenals, thoracic aorta, brain, esophagus, eyes, femoris muscle with sciatic nerve, Harderian gland, heart, kidneys, large intestine, liver, lungs, MLN, ovaries and oviducts, pancreas, pituitary, salivary glands, skin, small intestine, spinal cord, spleen, sternum with marrow, stomach, thymus, thyroid, trachea, urinary bladder, uterus, and all gross lesions.
The histopathological investigation focused on tissues from kidneys, liver, MLN, and spleen as well as other tissues with gross lesions. These specified tissues were processed, embedded in paraffin, sectioned, and stained with hematoxylin and eosin for microscopic examination. All of the selected tissues from female rats in all dose groups were examined.
Mineral Hydrocarbon Analyses
Dietary concentrations
Feed samples were extracted and analyzed at weeks 1, 5, 9, and 13 to confirm test material concentrations at each of the concentration levels. Three-gram feed samples from the control, 0.02% and 0.2% dietary levels were extracted with 10 ml of 95% hexanes/5% dichloromethane in ca 20 mL glass vials with septum lined caps. Samples were extracted using a combination of manual and automated shaking for 30 minutes. Sample extracts were permitted to settle and cleaned up by passing 7 mL of each extract through small columns containing 0.75 g activated silica gel and 0.5 g sodium sulfate. The columns were pre-rinsed with 2 mL 95% hexanes/5% dichloromethane prior to application of the feed extracts. The cleaned extracts were collected along with 3 mL of solvent which was used to rinse each column and adjusted to final volumes of 10 mL with hexane. Feed samples from the 2% dose level were similarly prepared except that 2 g of feed were extracted and the final volume was 25 mL.
Tissue levels
Samples of liver, kidneys, MLN, and spleen were taken from each of the 5 animals/dose group designated for analysis of MHC of selected organs. To prepare extracts, organ tissue samples were placed in glass bottles. Hydromatrix drying reagent (Varian, Inc, Walnut Creek, California) was then added to each of the bottles in an amount equal to approximately 50% of the sample weight. The sample and drying reagent were blended for initial drying and shredding. Octadecyl-derived silica (ODS) was added to each sample at approximately the sample weight and the entire mixture was blended for 1 minute or until the structure was completely disrupted. Samples were permitted to dry overnight at room temperature. Dried samples were extracted with 25 mL (kidney, MLN, and spleen) or 50 mL (liver) 95% hexanes/5% dichloromethane solvent similar to the feed samples. Five milliliter portions of each extract were passed through individual clean-up columns containing 0.5 g activated silica gel and 0.5 g sodium sulfate). The eluents were collected in 15 mL glass centrifuge tubes. An additional 3 mL volume of 95% hexanes/5% dichloromethane was used to rinse each column. If necessary, the final sample extract was diluted prior to analysis.
Gas chromatographic analysis
Both feed and tissue extracts were analyzed by gas chromatography with flame ionization detection (GC-FID) on a Perkin Elmer Autosystem XL (PerkinElmer, Sheldon, Connecticut) equipped with an autosampler and 15 m × 0.53 mm internal diameter, 0.15 μm HP-1 methyl silicone capillary column.
Fifteen microliter large volume injections were made with the inlet temperature ballistically programmed from 50°C to 320°C. The GC oven was temperature programmed from 45°C to 300°C at 45°C/minute after an initial hold of 6 minutes.
The FID was operated at 300°C, and the helium column flow was programmed from 3to 15 mL/min. Data were collected and processed using Perkin Elmer Turbochrom software. Feed and tissue sample extracts were quantified against standards of the same white mineral oil test article used for dosing and analyzed under the same conditions. The total chromatographic area was used for quantification.
Data Quantitation
As a means of comparing the severity of the MLN effects across a range of studies, a microgranuloma severity index was developed in which a mean microgranuloma score was calculated by multiplying the incidence and severity scores in each group divided by the number of animals/group and then multiplied by 100. The severity scale was minimal = 1, mild = 2, moderate = 3, and marked = 4. This method of quantifying incidence/severity of MLN lesions is as described in Smith et al 6 and was used to compare the data from the present study to those from previous publications.
Statistical Analysis
Statistical treatment of the results was conducted where appropriate. Statistical evaluation of equality of means was by 1-way analysis of variance (ANOVA) and a test for ordered response. First, Bartlett test 18 was performed to determine whether the dose groups had equal variance. If the variances were equal, testing was done using parametric methods, otherwise nonparametric methods were used.
For the parametric procedures, a standard 1-way ANOVA 18 using the F distribution to assess significance was used. If significant differences among the means were indicated, Dunnett test 19 was used to determine which treatment groups differed significantly from control. In addition to the ANOVA, a standard regression analysis for linear response 18 in the dose groups was performed. The regression also tested for linear lack of fit in the model.
For the nonparametric procedures, the test of equality of means was performed using the Kruskal-Wallis Test. 20 If significant differences among the means were indicated, Dunn Summed Rank Test 20 was used to determine which treatment groups differed significantly from control. In addition to the Kruskal-Wallis Test, Jonckheere Test 20 was used to test for monotonic trend in the dose response. Bartlett test for equal variance was conducted at the 1% level of significance. All other tests were conducted at the 5% and 1% level of significance.
Results
In Life Observations
All rats survived to scheduled termination, and there were no clinical signs which were judged to have been related to treatment. All animals gained weight over the treatment period, and there were no significant differences in terminal body weights between groups. The rats consumed similar amounts of feed on a daily basis, over the lifetime of the study. Thus, as body weights increased, the daily doses, expressed in terms of body weight, decreased. Average weekly doses are shown in Table 1.
Mean (±SD) Weekly Doses of P7H(H) Oil Consumed by Female F344 Rats During the 13-Week Dietary Administration Study a
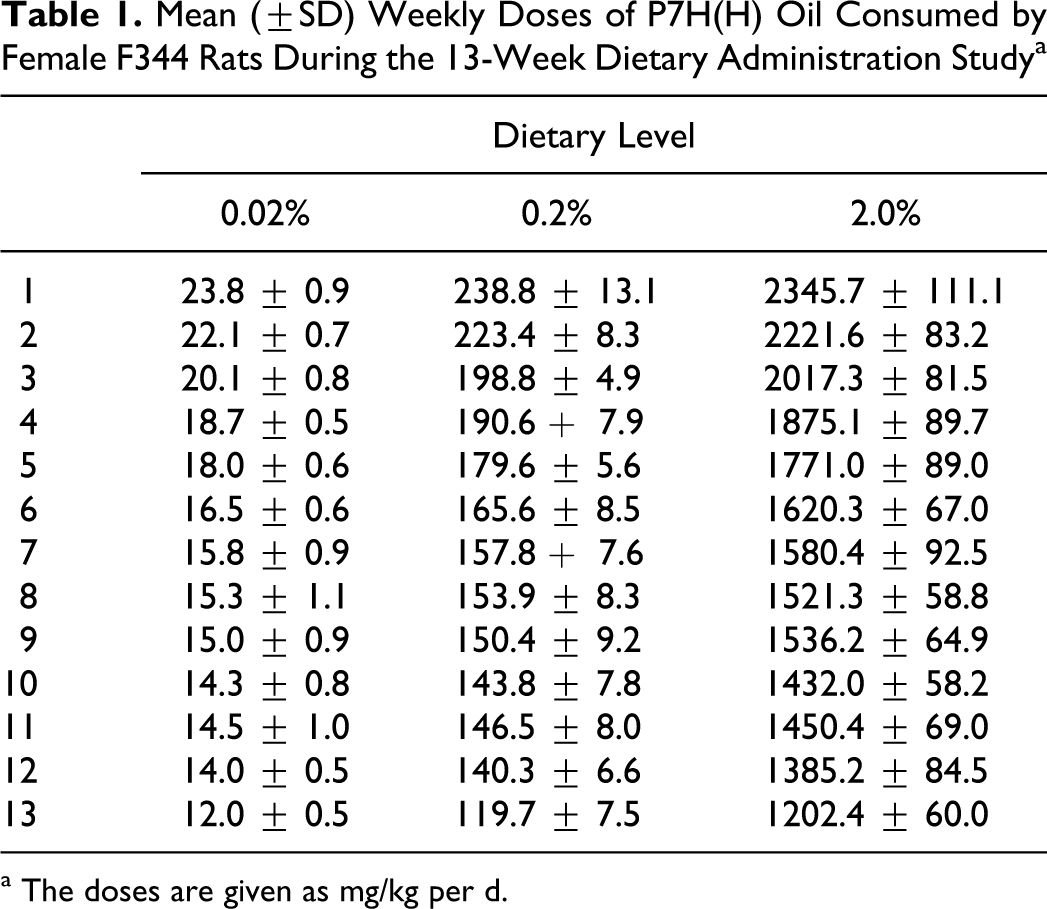
a The doses are given as mg/kg per d.
Hematology and Clinical Chemistry
Among the hematological parameters there was a small but statistically significant increase in numbers of neutrophils in the 2% dose group and a small but statistically significant decrease in the percentage of lymphocytes, also in the 2% dose group. All other hematological parameters were either similar across dose groups or when elevated did not show clear evidence of dose response (Table 2). The increase in neutrophil values was considered treatment related. The decrease in percentage of lymphocytes was most likely a consequence of the increase in neutrophils as the absolute number of lymphocytes did not change.
Mean (± SD) Quantitative Hematology Values in Female F344 Rats Exposed to P7(H) Oil by Dietary Administration
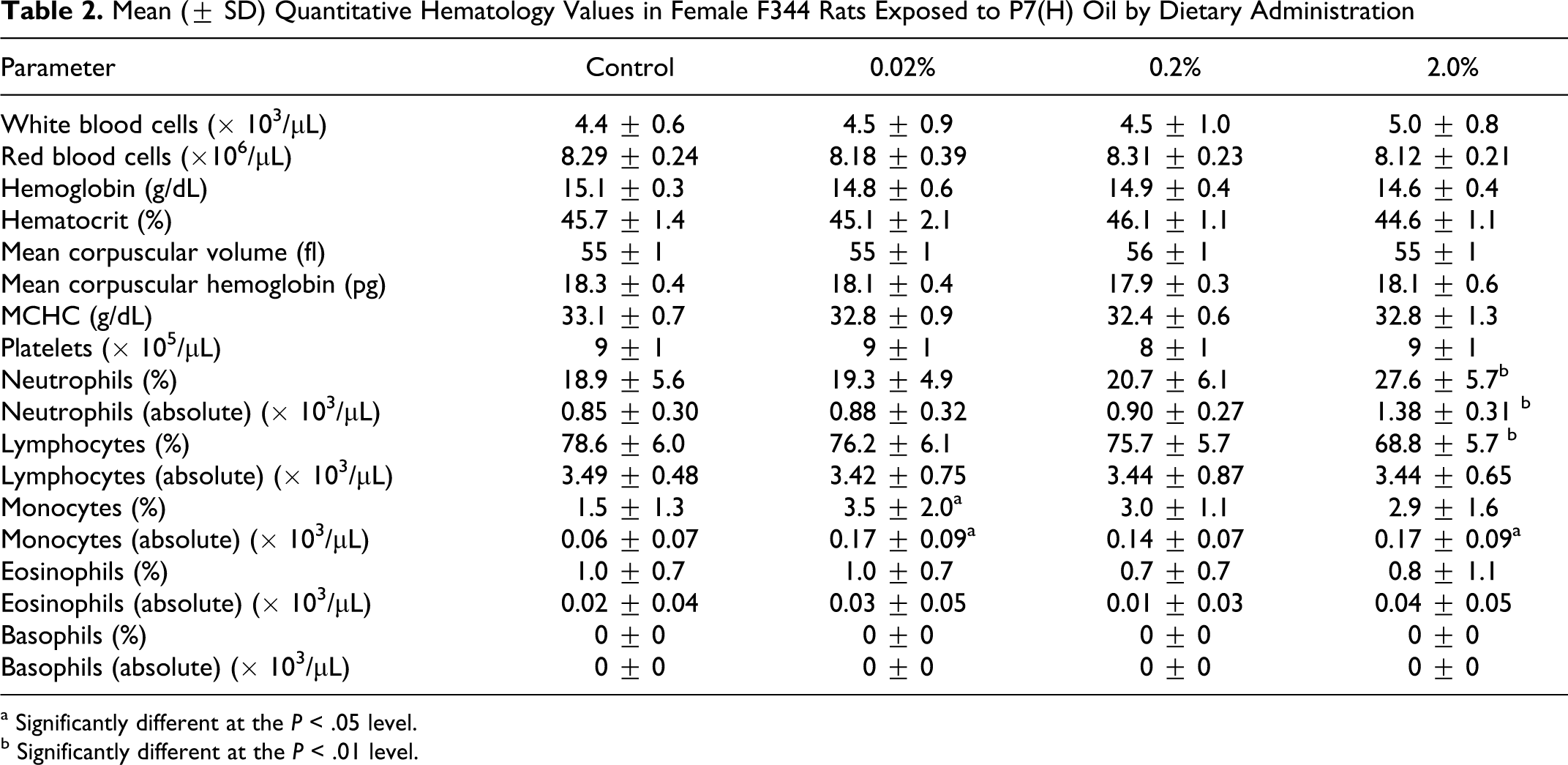
a Significantly different at the P < .05 level.
b Significantly different at the P < .01 level.
Among clinical chemistry parameters there was a statistically significant decrease in alkaline phosphatase and statistically significant increases in total protein, albumin, globulin, GGT, and amylase (Table 3). Most of the differences were small and were within normal physiological ranges. However, in some cases, similar changes were reported in previous studies and may have had biological significance. The increase in amylase values was relatively large but as it was not correlated with gross changes in the pancreas or histological changes in the kidneys, the toxicological relevance was not clear.
Mean (
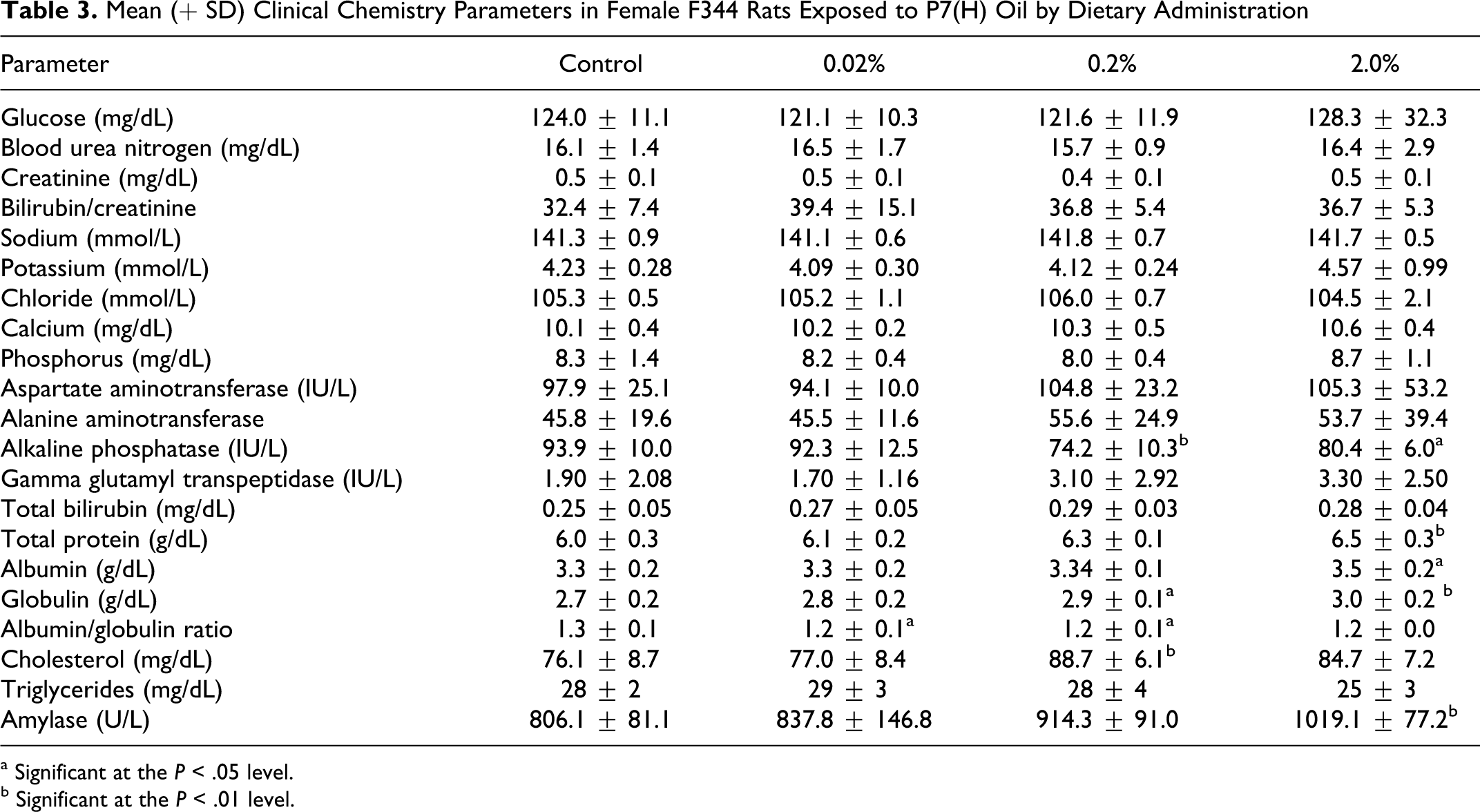
a Significant at the P < .05 level.
b Significant at the P < .01 level.
Gross PostMortem Examination and Organ Weights
Postmortem findings were limited to increased organ weights, enlarged MLN, and histopathological changes described below. Liver weights were increased in rats from the 2% dose groups. Kidney weights were also increased in the 2% dose group. The organ weight assessment provided evidence of statistically significant increases in weights of MLN in rats from the 0.2% and 2% dose groups, consistent with observations of MLN enlargement in both dose groups (0.2%, 1 of 15 rats; 2%, 7 of 15 rats). Weights of heart, spleen, and ovary were similar to control values (Table 4).
Mean (± SD) Organ Weights in Female F344 Rats Exposed by Dietary Administration to P7(H) Oil
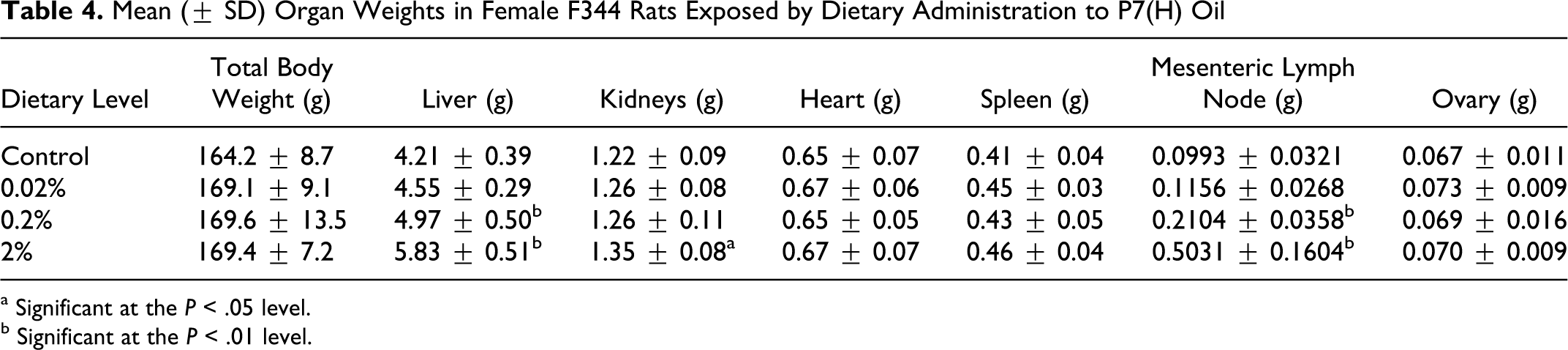
a Significant at the P < .05 level.
b Significant at the P < .01 level.
Histopathology
The histological findings were limited to the evidence of microgranuloma formation in the liver and MLN (Table 5). In the liver, minimal evidence of microgranuloma formation was apparent in only the highest dose group. With respect to the MLN, both the number of animals affected and the severity of response increased with dose in the 0.2% and 2.0% dose groups. The response in the 0.02% group was not significantly different from control.
Results of the Histopathological Investigation of Tissues From Female F344 Rats Following Dietary Exposure to P(7)H Oil for 90 Days
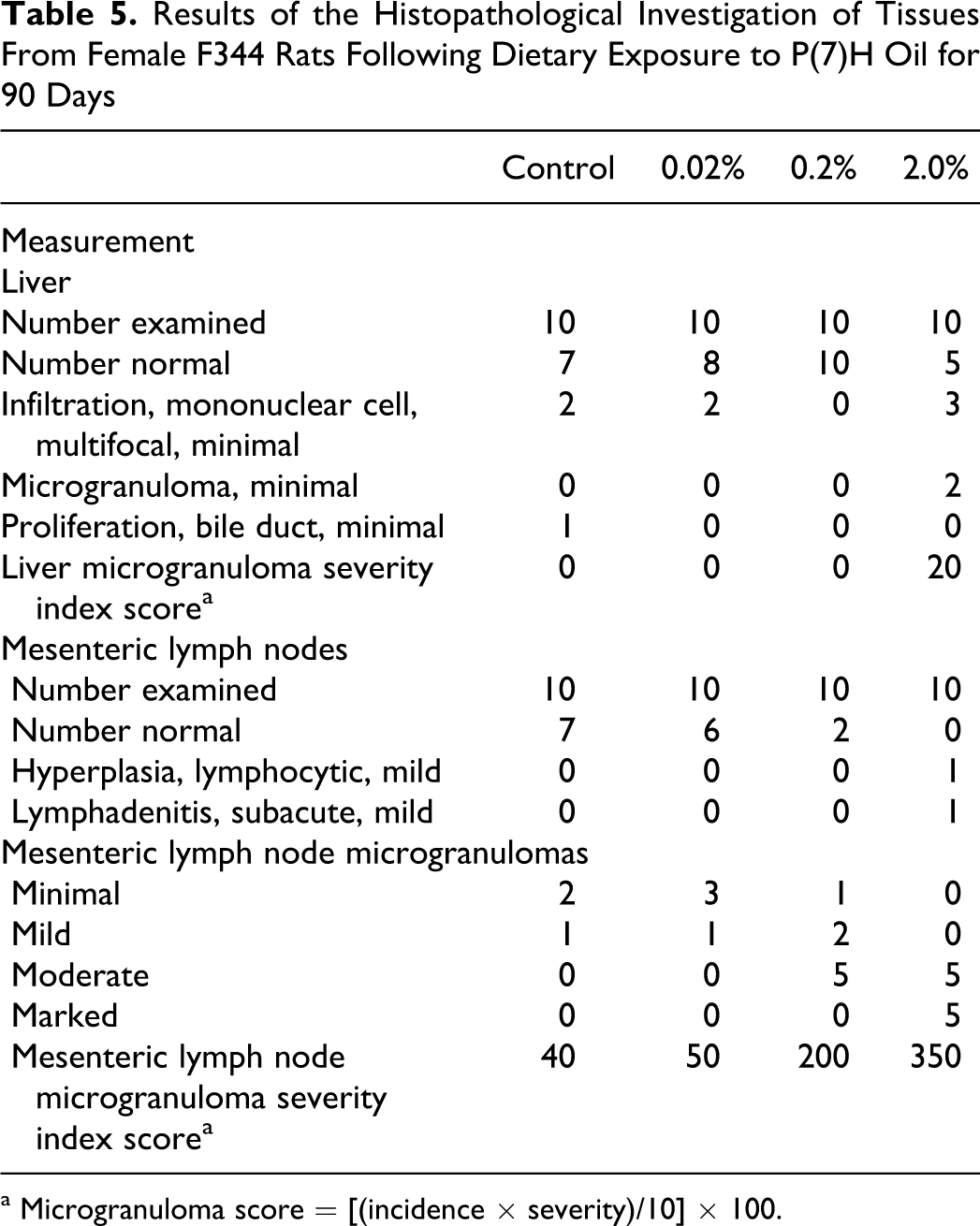
a Microgranuloma score = [(incidence × severity)/10] × 100.
Mineral Hydrocarbons in Feed and Tissues
Feed analysis indicated measured dietary levels in the 0.02% feed ranged from 0.0198% to 0.0231% with an overall average of 0.0213%. In the 0.2% feed, the levels ranged from 0.194% to 0.203% with an overall average of 0.196%. In the 2.0% feed, the levels ranged from 1.94% to 2.03%, with an overall average of 1.97%. The test article blended in feed was demonstrated to be stable at room temperature. Mineral hydrocarbon content in feed at the 0.02% level decreased by 8.5% after 15 days and by 7.1% at the 2% level. Batches of blended feed were also shown to be homogeneous. Analysis of replicate feed samples taken in triplicate from the top, middle, and bottom of batches of the 0.02% and 0.2% diets yielded relative standard deviations (n = 9) of 7.5% and 5.2%, respectively.
Mineral hydrocarbon content was not detected in spleen samples and only at low and variable concentrations in kidneys from rats in the 2% dose group (Table 6). The greatest MHC concentrations were measured in liver and MLN. The concentrations of MHC in livers were approximately 30% greater in the livers from rats of the 2% dose group by comparison to those of the 0.2% dose group. Mineral hydrocarbon content concentrations were lower in MLNs from rats of the 2% dose group by comparison with those in the 0.2% dose group. However, these differences are at least partially a reflection of the increased size of the organs in the 2% dose group. The liver weights increased by about 17%, but the MLN weights in the 2% group were approximately 2.5 times of those in the 0.2% group. The absolute amounts of MHC were 50% to 60% greater in the livers and MLNs of rats fed 2% P7H oil in diet by comparisons to those from the 0.2% dose group. However, MHC concentrations in the MLNs were lower in the 2% dose group than in the 0.2% dose group. As indicated above, this may have been at least partially a dilution effect as the MLNs in the 2.0% group were approximately 2.5 times the size of those in the 0.2% group.
Mean (

Discussion
When mineral hydrocarbons and waxes are administered to F344 rats, a characteristic response is observed including increased weights of liver and MLN, histiocytotic responses including microgranulomas in liver and MLN, and in F344 rats exposed to some waxes, mitral valve inflammation. 4 –7,12 These changes were not observed when these substances were given to rats of other strains or to dogs. 2,3,5 The effects appear to be associated with selective retention of aliphatic hydrocarbons with carbon numbers ranging from C20 to C35 in liver and MLN, 7,8,11,21 and the strain and species differences seem to be a consequence of a higher rate of absorption and lower rate of metabolism in F344 rats by comparison to other strains. 8 This imbalance between uptake and metabolism results in accumulation of these hydrocarbons in liver and MLN, leading to inflammatory processes, 12 and ultimately to the formation of microgranulomas. While the formation of microgranulomas in the liver are correlated with MHC accumulation in tissue, microgranuloma formation in MLN does not follow a similar dose-related response. 5,22 Due to the lack of a dose-related response, microgranuloma formation is considered a nonspecific clearance response to the presence of foreign material. 23,24 Accordingly, the use of microgranuloma formation as a starting point for human health risk assessment is questionable.
Hydrocarbons with carbon numbers greater than C35 are not well absorbed 9,10 and are unlikely to contribute significantly to microgranuloma formation. In analyses of liver extracts from F344 rats, molecules with carbon numbers ranging from C20 to C30 are found at levels greater than those in the corresponding oils, suggesting selective accumulation as described above. Molecules with carbon numbers less than C20 are found at levels below those in the corresponding mineral oils, 7,11 and, as hydrocarbons with carbon numbers in this range are relatively well absorbed, are presumed to be relatively rapidly excreted. However, molecules with carbon numbers <C20 have been less well studied in this respect as research has focused on mineral oils and waxes with constituents primarily in the C20 to C30 range. The current study investigated the pathologic and tissue distribution properties of a lighter oil (P7H) in which approximately half of the constituents had carbon numbers less than C20. The expectation was that, as the hydrocarbons tested were of lower molecular weight than those present in previously tested white oils and waxes, they would not be accumulated to the same extent as the constituents of higher molecular weight oils and would not be as effective in producing the characteristic pathologic changes in the liver.
As described in the results section, dietary administration of P7H oil to female F344 rats caused significant increases in liver weights at the 2% dietary level and in weights of MLN at the 0.2% and 2% levels (Table 4). There was a small but statistically significant increase in kidney weights, but weights of other organs were not different from control values. There was a small but statistically significant increase in absolute numbers of neutrophils (Table 2) and an increase in GGT levels which was dose responsive although the difference was not statistically significant (Table 3). Previous investigators have reported increased neutrophil and GGT levels following mineral oil treatment and attributed it to a mild inflammatory response. 5,12 The increases found in rats from the 2% dietary level in the present study are similar to those found at the 0.2% dietary level in studies in which rats were given higher-molecular-weight mineral oils. 5,12 The histological evaluation (Table 5) provided at most very limited evidence for microgranuloma formation in liver at the 2% level, and no evidence for liver effects at lower treatment levels. In the MLN, there was no significant evidence for microgranuloma formation at the 0.02% dietary level, but there was evidence of microgranuloma formation at increasing frequency and severity at the higher dietary levels. As noted above, microgranuloma formation in MLN is probably a normal physiological response to the presence of foreign material, in contrast to microgranuloma formation in the liver which may be an adverse effect. 24
When the results of the present study were compared to previous results with mineral oils of higher molecular weight (Table 7, data for the P7H oil are from the present investigation, data on other oils are from Smith et al 6 ), it was apparent that dietary treatment with the P7H oil resulted in liver effects which were much more limited than those associated with exposure to N10A, N15H, and P15H oils with carbon numbers ranging from C22 to C25. The MLN data are somewhat more complicated. It is apparent (Table 7) that the no effect level for the P7H oil was a dietary level of 0.02%; whereas the no effect levels for the N10A, N15H, and P15H oils were dietary levels of 0.002%. The effects of the P7H, N10A, N15H, and P15H oils were similar at the 0.2% dietary level, and, if anything, there was a greater response at the 2.0% dietary level with the P7H oil than with the other 3 oils. This is consistent with a dose–response relationship. Approximately 50% of the content of the P7H oil is comprised of constituents >C20 , so it would be reasonable to expect that the P7H oil would produce MLN changes similar to those associated with the higher carbon number oils but only at higher treatment levels.
Comparative Microgranuloma Scores Among Mineral Oils of Differing Molecular Weights and Carbon Numbers a
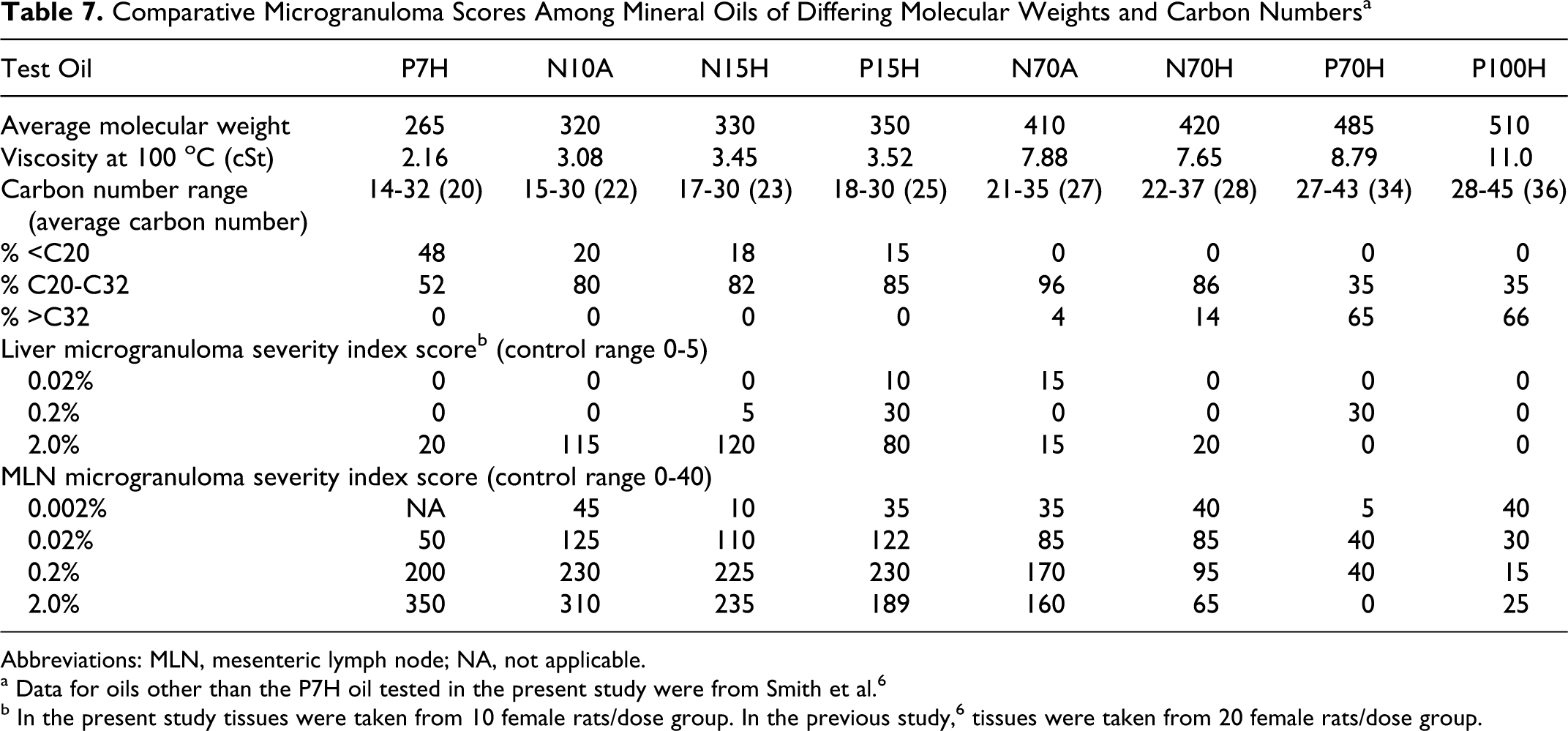
Abbreviations: MLN, mesenteric lymph node; NA, not applicable.
a Data for oils other than the P7H oil tested in the present study were from Smith et al. 6
b In the present study tissues were taken from 10 female rats/dose group. In the previous study, 6 tissues were taken from 20 female rats/dose group.
Smith et al 6 reported that previously unpublished investigations had shown that there were no differences between oils refined by acid treatment (A) versus hydrogenation (H) or from oils derived from N versus P crude oils. This leaves carbon number range as the only distinguishing feature among these oils which could explain the differences in response levels.
The comparative results shown in Table 7 indicate that the microgranuloma scores were higher in oils with average molecular weights of 330 to 350 but were lower in oils with average molecular weights of 410 and greater and that the oils with carbon numbers greater than C30 were essentially inactive. Early studies indicated that these high-molecular-weight oils were not well absorbed, 9,10 and initial studies of liver extracts revealed that higher-molecular-weight hydrocarbon constituents were underrepresented in comparison to their concentrations in the corresponding test materials. 11 These results were further extended by Scotter et al, 7 leading to the view that the MHC constituents most likely to be selectively retained and ultimately to cause these effects were predominantly in the range of C20 to C35. However, as these studies had focused on higher-molecular-weight substances, the properties of oils with carbon numbers <C20 were less well defined. The present study afforded the opportunity to better characterize the absorptive properties of lower-molecular-weight MHC.
The results of the liver extract examination (Figure 1) indicate that, as expected, the MHC with molecular weights <C20 were underrepresented by comparison to the carbon number distribution in the P7H oil. As hydrocarbons with average carbon numbers <C20 are well absorbed, 9,10 the most likely explanation for this underrepresentation is that these lower-molecular-weight hydrocarbon constituents were metabolized at a rate sufficient to prevent selective retention.
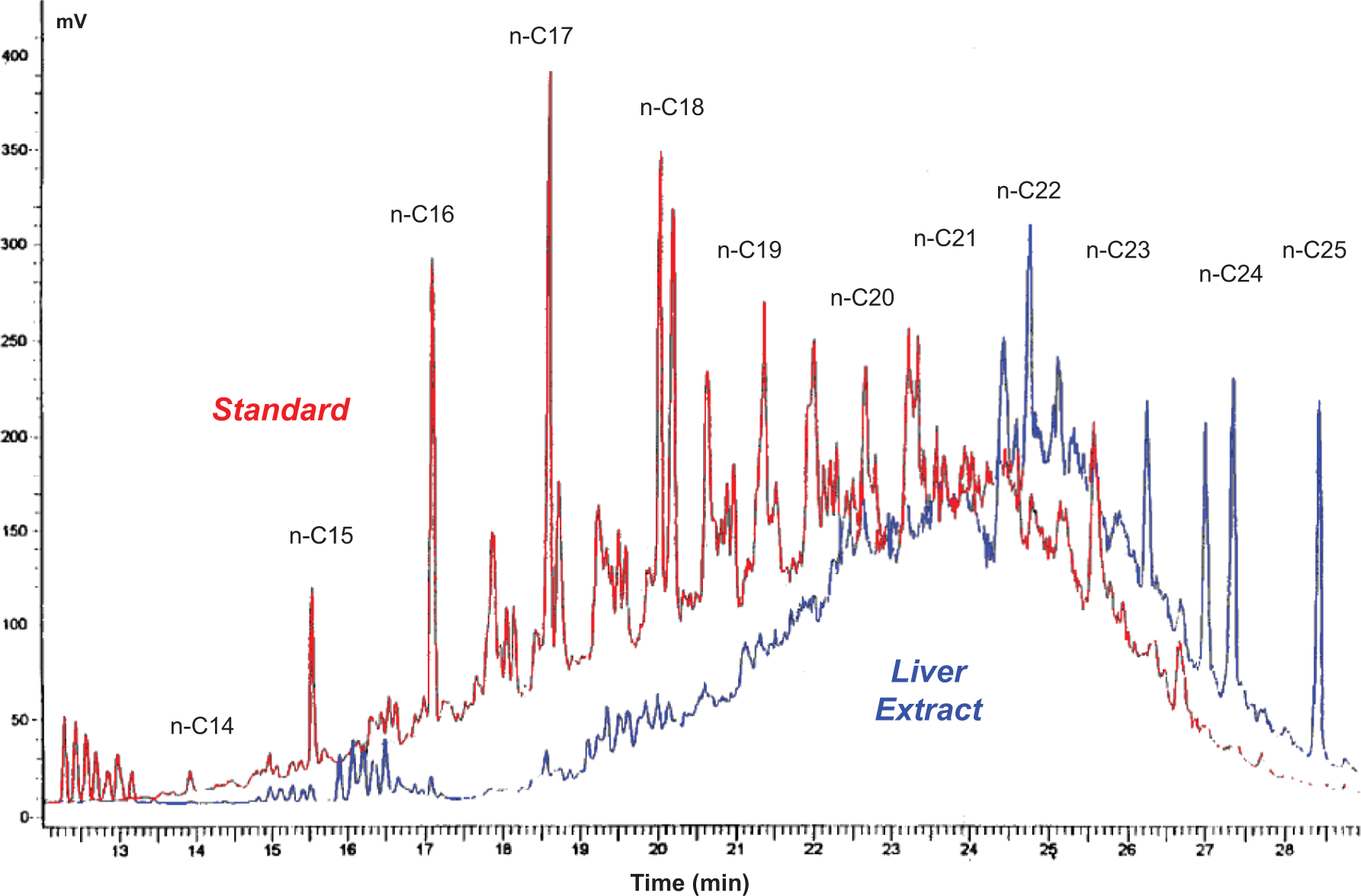
Gas chromatography with flame ionization detection (GC-FID) chromatograms of P7H oil versus rat liver extract.
In summary, the present study was conducted to further explore the potential for MHC with carbon numbers less than C20 to produce microgranulomas in liver and MLN of female F344 rats. The test substance, a P7H oil, had an average carbon number of C20 and approximately half of the constituents had carbon numbers <C20. The histological examination revealed that dietary exposure resulted in microgranuloma formation in the liver at a severity which was well below that found with MHC with constituents predominantly in the range of C20 to C30 , and microgranuloma formation in MLN at comparable severity but at higher treatment levels. Hydrocarbon analysis of liver extracts revealed that MHC with carbon numbers greater than C20 were in excess by comparison to the P7H oil whereas those with carbon numbers <C20 were underrepresented. Thus, these data supported the hypothesis that MHC with carbon numbers <C20 were unlikely to cause microgranuloma formation in the livers and MLN of this strain of rat. As MHC with carbon numbers <C20 are well absorbed, the most likely explanation is that they are also metabolized at a rate sufficient to prevent them from being selectively retained.
Footnotes
Acknowledgments
The authors would like to thank Ryan Manning for constructing the figure, Lynn Bennett for assistance in manuscript preparation and Robert Barter, Katy Goyak, and Serge Riffard for critical review and comment.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are currently or have been employed by a company which manufactures mineral oils and waxes.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The costs of the research, authorship and publication were paid by the employer, Exxon Mobil. The authors, all of whom were Exxon Mobil employees when they were involved in this project, wrote the publication as part of their normal employment.
