Abstract
Cationic amphiphilic drugs and aminoglycoside antibiotics can induce phospholipidosis (PLD), an abnormal accumulation of phospholipids in lysosome-derived vesicles, in preclinical studies. The incidence of PLD in patients and its clinical relevance are difficult to assess without noninvasive biomarkers. Di-docosahexaenoyl bis(monoacylglycerol)phosphate (di-22:6-BMP) is a phospholipid that is enriched in lysosomal membranes and a proposed urinary biomarker of drug-induced PLD. The specificity of di-22:6-BMP for PLD was compared to other phospholipid species that can increase in urine with nephrotoxicity. Using liquid chromatography coupled to mass spectrometry, 12 phospholipids were assayed in the urine of rats treated with drugs that induced PLD or caused renal or skeletal muscle injury. In receiver operating curve analyses, urinary di-22:6-BMP was a significantly better predictor of PLD and the least predictive of tissue injury of the phospholipids assayed. The data provide evidence supporting the use of di-22:6-BMP as a urinary biomarker of PLD in rats.
Keywords
Introduction
Drug-induced phospholipidosis (PLD) is a lipid storage disorder marked by the excessive cellular accumulation of drug and phospholipids in lysosome-derived vesicles. 1 Drugs that induce PLD typically have a cationic amphiphilic structure or are aminoglycoside antibiotics. Cationic amphiphilic drugs (CADs) can diffuse across membranes at neutral pH, become trapped in the low pH environment of secondary endosomes and lysosomes, and inhibit the degradation of phospholipids, leading to PLD. There is evidence that CADs interfere with phospholipid breakdown by direct inhibition of lysosomal phospholipases or by forming complexes with phospholipids that protect them from degradation. 1 Gentamicin and other aminoglycoside antibiotics that induce PLD in the kidney enter renal proximal tubular epithelial cells (PTECs) using the cellular process for reabsorption of plasma proteins from the glomerular filtrate. 2,3 These antibiotics bind to the membrane receptors megalin and cubulin, which deliver ligands to the lysosomes of PTEC through receptor-mediated endocytosis.
Although PLD is not thought to cause toxicity directly and is usually reversible, concerns have been raised about certain morphologic similarities to genetic lysosomal storage disorders. 1 The incidence of PLD in medicated populations is largely unknown, although it has been observed in biopsies from patients that incurred hepatotoxicity from 4, 4′-diethylaminoethoxyhexestrol dihydrochloride (DEAEH) treatment, 4 nephropathy and myopathy associated with long-term chloroquine therapy, 5,6 renal insufficiency associated with high dosages of amiodarone, 7 or that were administered therapeutic doses of gentamicin treatment prior to nephrectomy. 8 The occurrence, severity, number of organs affected, and time for manifestation of PLD differ among drugs and species.
The ability to monitor PLD noninvasively would help in evaluating this condition in nonclinical safety testing and, if translatable to the clinical setting, identifying the presence and risk associated with PLD in patients. Currently, the definitive diagnosis of PLD is based on the morphologic appearance of lamellar or myeloid bodies in tissues or cells by transmission electron microscopy (TEM). At the light microscopic level, PLD manifests as distended cytoplasmic vacuoles that can be distinguished from lipidoses by immunohistochemical staining for lysosomal-associated membrane protein 2 (LAMP-2), a membrane protein specific to lysosomal vesicles. 9 Levels of the phospholipid bis(monoacylglycerol)phosphate (BMP), also known as lysobisphosphatidic acid (LBPA), are elevated in tissues with drug-induced PLD. 10 Bis(monoacylglycerol)phosphate is a minor component (1%-2%) of total cellular phospholipids but is highly enriched in the membranes of late endosomal vesicles and in the multilamellar membranes of myeloid bodies. 11 Bis(monoacylglycerol)phosphate with 18:1 acyl chains is found in high levels in plasma from patients with lysosomal storage disorders, especially Niemann-Pick disease. 12 In several studies, increased levels of the di-docosahexaenoyl conjugate of BMP (di-22:6-BMP) have been observed in urine samples from animals treated with drugs that induce TEM-confirmed PLD. 13 Therefore, there is interest in di-22:6-BMP as a potential noninvasive assay for PLD.
Increased levels of urinary phospholipids are associated with the administration of nephrotoxicants like gentamicin and HgCl2 that primarily target renal proximal tubules 14 –17 and with agents that cause renal papillary necrosis like nonsteroidal anti-inflammatory drugs and 2-bromoethanamine. 18,19 Although some of these studies measured levels of individual phospholipids as well as total phospholipid, none specifically assayed urinary levels of BMP. To better characterize the potential response of di-22:6-BMP to tissue injury, we assayed phospholipid levels in urine from studies with drug treatments that induced PLD, skeletal muscle injury, and/or nephrotoxicity. The drugs DEAEH and azithromycin were administered to induce PLD, simvastatin was used to induce skeletal muscle injury, and nephrotoxicity was produced in rats by treatment with gentamicin and cisplatin. 4, 4′-Diethylaminoethoxyhexestrol dihydrochloride was one of the first pharmaceuticals shown to cause generalized PLD in the clinic. 4 Patients administered DEAEH for over 6 months had markedly increased levels of phospholipids, including BMP, in liver, spleen, muscle, lymph node, and urinary sediment. 20,21 Similar changes were observed in follow-up studies in several animal models (dogs, rabbits, rats, and mice) that were administered a daily oral dose of 58.5 or 117 mg/kg DEAEH for 5 to 7 days. 22
The specificity of di-22:6-BMP and other phospholipids as urinary biomarkers of tissue PLD or injury was assessed in receiver operating characteristic (ROC) plots by combining data from studies of drugs that induce the target indication (PLD) with studies of drugs that induce toxicities often seen in preclinical studies. This analysis produced a more complete description of the diagnostic accuracy of di-22:6-BMP for PLD by providing a direct comparison to other phospholipids.
Materials and Methods
Chemicals
4, 4′-Diethylaminoethoxyhexestrol dihydrochloride (CAS No. 69-14-7) was synthesized to 99.8% purity using a modification of a published procedure. 23 Upon performing scale-up, the reference approach was generally followed except excess sodium hydride was used to drive the reaction to completion, and the order of addition of reagents was altered. The product was recrystallized twice from a 1:1 mixture of ethanol and ethyl acetate. Cisplatin and gentamicin were obtained from Sigma Aldrich (St Louis, Missouri). Simvastatin was obtained from Sequoia Research Products (Pangbourne, UK). Azithromycin was purchased from LKT Laboratories, Inc (St Paul, Minnesota).
Animals
All procedures involving animals were approved by the Institutional Animal Care and Use Committee at the US Food and Drug Administration (FDA) and complied with the guidelines for the Care and Use of Laboratory Animals (NIH Publication 85-23). Male Fisher rats (F344/NHsd), 6 weeks in age, and male or female Sprague Dawley rats, 8 to 12 weeks in age, were purchased from Harlan Laboratories (Indianapolis, Indiana). Animals were housed individually, given rodent chow (Lab Diet 5002), and water ad libitum.
Toxicity Studies in Rats
4, 4′-Diethylaminoethoxyhexestrol dihydrochloride was prepared in saline and administered at daily doses of 0, 25, 50, or 100 mg/kg by oral gavage for 1 or 2 weeks to groups of 4 to 6 male Fisher rats. The rats were placed in metabolism cages without access to food for overnight (15 hour) urine collection prior to study termination. Overnight urine was collected at −20°C in 2 aliquots. Samples were thawed, mixed, and stored in aliquots at −80°C. Cisplatin was administered to male Sprague Dawley rats in a single intraperitoneal (ip) injection of 0, 2, or 3 mg/kg, animals were placed in metabolism cages with access to food, and urine was collected during the last 16 hour of the 72 hour study. Simvastatin was prepared in 0.5% methylcellulose and administered to 9- to 10-week-old female Sprague Dawley rats in daily oral doses of 80 mg/kg for up to 14 days. Overnight urines were collected prestudy and at study days 7 and 14. Azithromycin was prepared in 0.13 mol/L sodium citrate pH 4 and administered to male Sprague Dawley rats by oral gavage at 150 mg/kg/d for 14 days. Overnight urines were collected immediately prior to study termination in nonfasted rats. The gentamicin study has been described elsewhere. 24
In all studies, rats were exsanguinated under isoflurane anesthesia. Terminal serum clinical chemistry analysis was performed on the VetScan model #200-1000 using the Comprehensive Diagnostic and Large Animal rotors (Abaxis, Inc, Union City, California). Tissue sections were fixed in 10% neutral buffered formalin embedded in paraffin and stained with hematoxylin-eosin. For the DEAEH study, sections of the lungs, liver, spleen, and kidney were fixed for 48 hour in a solution of 2% glutaraldehyde, 4% formaldehyde, and 0.1 mol/L sodium cacodylate pH 7.4 and stored in 0.1 mol/L sodium cacodylate pH 7.4 until samples were embedded for TEM. Sample preparation and imaging using TEM was performed at the Biomedical Instrumentation Center of the Uniformed Services University of the Health Sciences (Bethesda, Maryland).
The frequency and severity of treatment induced microscopic changes in collected tissues was assessed using standardized toxicological pathology criteria. Changes in severity of findings were recorded in increasing order of minimal, mild, moderate, and marked. Semiquantitative scores were assigned as follows: no significant findings = 0, minimal = 1, mild = 2, moderate = 3, and marked = 4. Histopathology reports for the DEAEH and azithromycin studies were prepared by Applied Veterinary Pathobiology LLC (Bainbridge Island, Washington). Histopathological evaluation of tissue sections from the cisplatin, gentamicin, and simvastatin studies was performed by pathologists in CDER’s Division of Drug Safety Research.
Protein Biomarker Assays
Serum levels of skeletal muscle troponin I (sTnI) were measured as part of the rat Muscle Injury Panel 3 kit (Meso Scale Discovery, Gaithersburg, Maryland). Five samples from the DEAEH study with evidence of hemolysis were excluded from the statistical analysis. Urinary levels of kidney injury molecule 1 (Kim-1), osteopontin (Opn), lipocalin 2 (Lpn2), and albumin (Alb) were measured with the rat Kidney Injury Panel 1 Assay Kit (Meso Scale Discovery). Lipocalin 2 was below the lower limit of detection in 10-fold diluted urine from the DEAEH study. Kidney injury biomarker levels were normalized to urinary creatinine concentration, which was measured by colorimetric assay (Arbor Assays, Ann Arbor, Michigan).
Di-22:6-BMP and Phospholipid Analysis
Phospholipid analysis was performed at Nextcea (Woburn, Massachusetts). A liquid–liquid phase extraction method was used to extract di-22:6-BMP and species of phosphatidylinositol (PI), phosphatidylcholine (PC), and phosphatidylethanolamine (PE) phospholipids from 200 μL of each urine sample. Samples were vortexed twice during extraction and then centrifuged at 12 000 rpm for 10 minutes. The top liquid layer was removed and concentrated to dryness under a stream of nitrogen. The dry urine extracts were reconstituted to 50 μL with 0.05% formic acid in acetonitrile/methanol (2:1 by volume). The molecular profile of each sample was acquired using liquid chromatography coupled to mass spectrometry (LC/MS), as previously described, 13 with the following modifications. The mobile phase A was 0.25% ammonium hydroxide, 0.05% formic acid in 88:12 methanol/water (v/v). Mobile phase B was 0.25% ammonium hydroxide, 0.05% formic acid in 80:20 methanol/hexane (v/v). The flow rate was 0.25 mL/min. Initial conditions were 90% A and 10% B. The percentage B was increased to 40% over a 14-minute period. The total running time per sample, including column wash and re-equilibration, was 25 minutes. Phospholipid levels were monitored using a quadrupole time-of-flight (Q-TOF) MS as previously described. 13 Data were acquired in full-scan TOF MS mode (m/z 200-1200) with negative electrospray ionization (ESI). The Turbo IonSpray interface was set at 400°C and maintained at an ion spray voltage of −4.5 kV with a declustering potential of −50 V. Ionization was assisted with a nebulizer and ionspray gas (nitrogen) set at 45 and 45 (arbitrary units), respectively. The identities of di-22:6-BMP and PI, PC, and PE phospholipids in the rat urine samples were confirmed by LC-MS/MS as described. 13 The intensity levels for each phospholipid were determined based on manual integration of the extracted ion chromatogram peak areas using Analyst QS software (Applied Biosystems, Foster City, California). 14:0,16:0-PC was present at detectable levels in urine from gentamicin treatment groups but not DEAEH treatment groups. Phospholipid intensity levels were normalized to urinary creatinine concentration.
Immunohistochemistry
Unstained slides were prepared for tissue collected in the DEAEH study and immunostained for LAMP-2 according to published methods. 9 Lysosomal-associated membrane protein 2 immunostaining was performed on 5-micron tissue sections using a Discovery ST Autostainer (Ventana Medical Systems, Tucson, Arizona). The slides were analyzed using a ScanScope CS Digital Tissue Scanner and ImageScope software (Aperio Technologies, Inc, Vista, California).
Statistical Analysis
The statistical significance between 2 groups was determined using a Mann-Whitney t test or, for the gentamicin dose and time course series, applying a Tukey-Kramer multiple comparison test post hoc to a 1-way analysis of variance in GraphPad Prizm (GraphPad Software Inc, La Jolla, California). The area under the curve (AUC) in ROC plots was calculated using format 5 (continuous rating scale format) in a Web-based calculator for ROC curves (JROCFIT). 25 Statistical analysis of differences between AUCs was performed using the method described by Hanley and McNeil. 26
Results
Animal Model of Drug-Induced PLD
4, 4′-Diethylaminoethoxyhexestrol dihydrochloride was used to create an animal model of drug-induced PLD based on its well-documented effect in patients and laboratory species. 20 –22 4, 4′-Diethylaminoethoxyhexestrol dihydrochloride was administered by gavage to male Fisher rats at daily doses of 0, 25, 50, or 100 mg/kg for 7 or 14 days. No early mortality or clinical signs were observed in any treatment group. Groups receiving the high dose of DEAEH had significantly lower weights than vehicle controls at days 7 and 14 (P < .05). At day 14, the average body weight (± SD) was 143.7 ± 8.4 g in the high-dose group and 193.4 ± 18.4 g in the vehicle control group. No significant effect of treatment on liver or kidney weight was observed.
4, 4′-Diethylaminoethoxyhexestrol dihydrochloride induced a dose and time dependent increase in vacuolation in multiple tissues (Table 1 ). A treatment-related increase in vacuolated macrophages was observed in the lung, spleen, and mesenteric lymph node (MLN). 4, 4′-Diethylaminoethoxyhexestrol dihydrochloride induced a dose-related increase in microvesicular vacuolation in hepatocytes at the 14 day time point. 4, 4′-Diethylaminoethoxyhexestrol dihydrochloride administration also induced myofiber vacuolation in gastrocnemius. Vacuolation in the liver, lung, spleen, and MLN but not skeletal muscle was associated with increased levels of LAMP-2 immunostaining. An example of vacuole-associated LAMP-2 staining resulting from DEAEH treatment is shown in Figure 1 for MLN. No treatment associated vacuolation or LAMP-2 immunostaining was observed in the heart or kidney. For confirmation of PLD, spleen, lung, liver, and kidney samples from rats in the 14 day control and high-dose groups were examined by TEM. Significant ultrastructural alterations of autolysosomes including typical myelin figures that display as multilamellar bodies were seen within the cytoplasm of hepatocytes, in pulmonary type II alveolar macrophages of the lung, macrophages of the spleen, and tubular epithelial cells of the kidney (figures not shown).
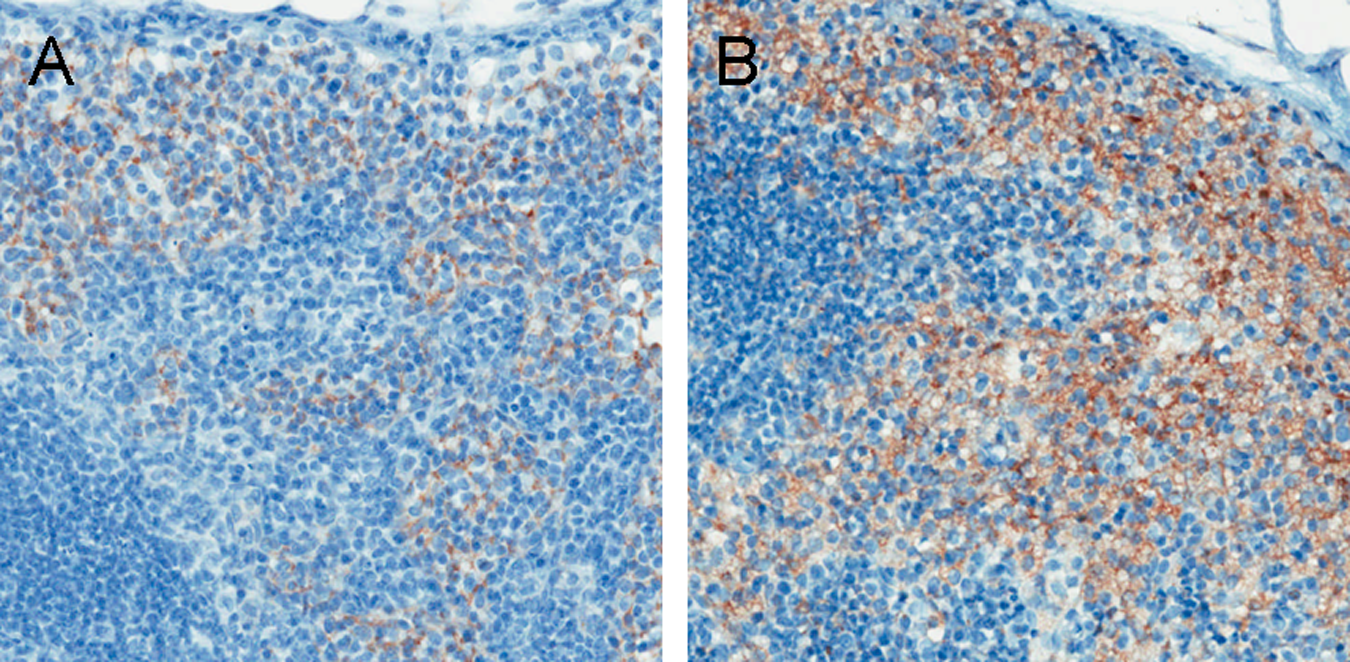
LAMP-2 immunohistochemistry of mesenteric lymph node from rats treated with DEAEH. A, Control group at day 14. Vacuolation score of 0 (no finding). B, 100 mg/kg/d DEAEH group at day 14. Vacuolation score of 3 (moderate incidence). ×20 magnification. DEAEH indicates 4, 4′-diethylaminoethoxyhexestrol dihydrochloride; LAMP-2, lysosomal-associated membrane protein 2.
Tissue Vacuolation and Necrosis Scores in DEAEH Treatment Groups
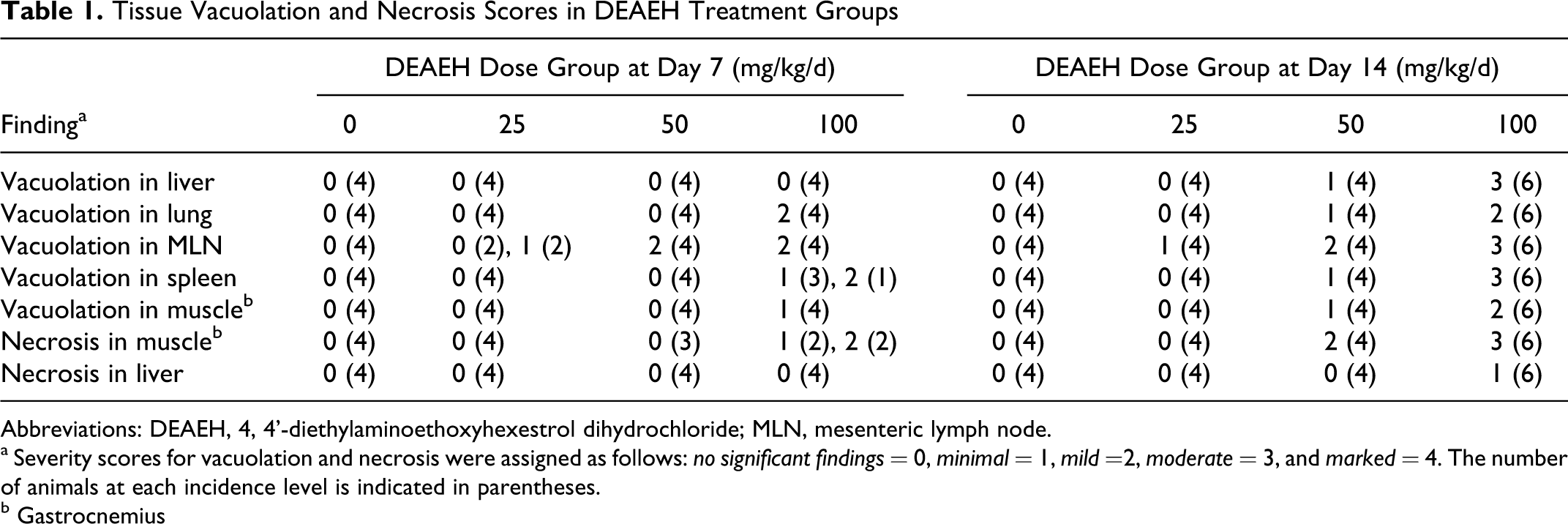
Abbreviations: DEAEH, 4, 4'-diethylaminoethoxyhexestrol dihydrochloride; MLN, mesenteric lymph node.
a Severity scores for vacuolation and necrosis were assigned as follows: no significant findings = 0, minimal = 1, mild =2, moderate = 3, and marked = 4. The number of animals at each incidence level is indicated in parentheses.
b Gastrocnemius
4, 4′-Diethylaminoethoxyhexestrol dihydrochloride treatment induced minimal-to-mild levels of degeneration and necrosis in skeletal muscle myofibers by day 7 and mild-to-moderate levels by day 14 of the study (Table 1). 4, 4′-Diethylaminoethoxyhexestrol dihydrochloride treatment also induced a minimal level of multifocal degeneration and necrosis of hepatocytes in the high-dose group at 2 weeks. Small, statistically significant increases in serum ALT and in the ratio of albumin to globulin in serum were also observed in the DEAEH high-dose group at 2 weeks (P < .05). 4, 4′-Diethylaminoethoxyhexestrol dihydrochloride treatment had no effect on renal histopathology or on urinary levels of Kim-1 or albumin. A small but statistically significant increase in urinary levels of Opn was observed in the high-dose group at day 7 and in the middle- and high-dose groups at day 14 (P < .05; Table 2 ).
Mean Urinary Levels of Biomarkers in DEAEH Treatment Groups
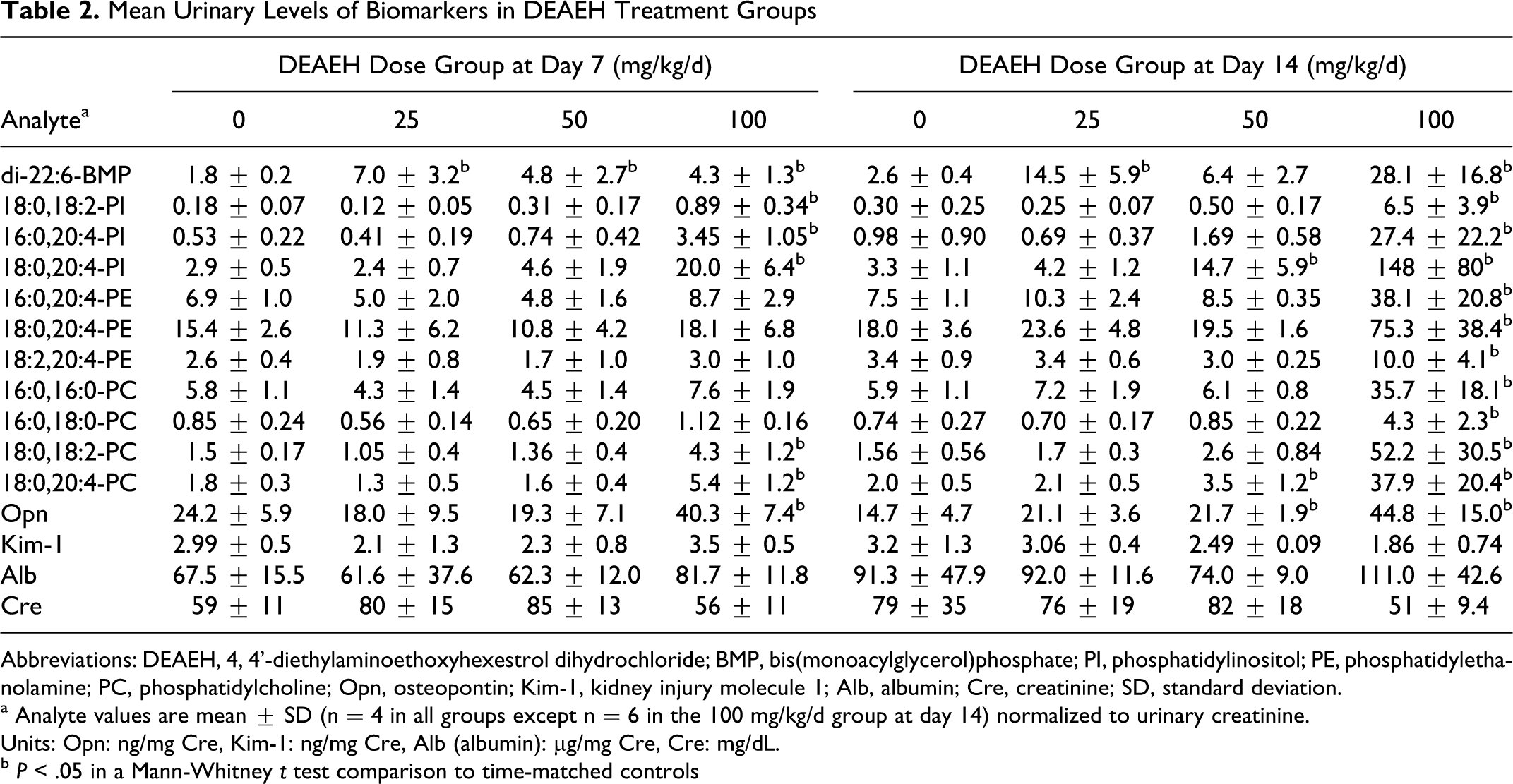
Abbreviations: DEAEH, 4, 4'-diethylaminoethoxyhexestrol dihydrochloride; BMP, bis(monoacylglycerol)phosphate; PI, phosphatidylinositol; PE, phosphatidylethanolamine; PC, phosphatidylcholine; Opn, osteopontin; Kim-1, kidney injury molecule 1; Alb, albumin; Cre, creatinine; SD, standard deviation.
a Analyte values are mean ± SD (n = 4 in all groups except n = 6 in the 100 mg/kg/d group at day 14) normalized to urinary creatinine.
Units: Opn: ng/mg Cre, Kim-1: ng/mg Cre, Alb (albumin): µg/mg Cre, Cre: mg/dL.
b P < .05 in a Mann-Whitney t test comparison to time-matched controls
Levels of Urinary Phospholipids in DEAEH Treatment Groups
Levels of di-22:6-BMP and 10 other phospholipids were measured in urine samples from the 34 animals in control and DEAEH treatment groups. Di-22:6-BMP was significantly elevated in 5 of the 6 DEAEH treatment groups relative to time-matched controls (P < .05; Table 2). Di-22:6-BMP was the only phospholipid of the 11 assayed that was significantly elevated in urine from the low-dose group at day 7 or 14 (P < .05). Five phospholipids in addition to di-22:6-BMP were significantly elevated in urine from the high-dose group at day 7 and all 11 phospholipids were significantly elevated in the high-dose group at day 14 (P < .05).
Diagnostic Accuracy of Urinary Phospholipids for PLD or Skeletal Muscle Injury
Drug-induced tissue injury could be a contributing factor to the elevation seen in levels of most of phospholipids assayed in urine from the high-dose groups. Skeletal muscle was the predominant site of injury induced by DEAEH in this study. Multifocal degeneration, necrosis, and vacuolation of myofibers were observed in the gastrocnemius of rats administered DEAEH at 100 mg/kg/d for 7 days, or 50 or 100 mg/kg/d for 14 days (Table 1) and a significant elevation in sTnI was seen in groups administered DEAEH at 100 mg/kg/d for 7 or 14 days compared to time-matched controls (P < .05).
To compare the relative sensitivity and specificity of urinary phospholipids for PLD versus myopathy, a combined analysis was performed of urine samples from the DEAEH study and from a study of simvastatin, which induces type II fiber selective muscle necrosis at high doses. 27 Phospholipid profiles were measured in 10 urine samples from a 10-day study of simvastatin in female Sprague Dawley rats. Overnight urine was collected pre-study and at days 7 and 10 from 1 control and 2 treated rats, and at day 10 for an additional control. By day 10, simvastatin-treated rats had incurred mild levels of necrosis in type II skeletal muscle, moderate levels of liver necrosis, and highly elevated serum levels of type II sTnI (A.K., manuscript in preparation). Relative to pre-study values, all assayed phospholipids except di-22:6-BMP and 18:0,18:2-PI were increased 2- to 4-fold in urine at day 10 of simvastatin treatment.
The diagnostic accuracy of urinary phospholipids for the end points of PLD or tissue necrosis was assessed in ROC plot analyses using the lowest treatment-associated incidence as the decision threshold. The earliest evidence of drug-induced PLD in tissues by light microscopy was a minimal level of vacuolation associated with enhanced LAMP-2 immunostaining that was seen in MLN in 2 of 4 rats administered 25 mg/kg/d DEAEH for 7 days (Table 1). Other studies of drug-induced PLD have also found lymph node to be a more sensitive indicator of this side effect than lung or liver. 28 For this analysis, a minimal or greater incidence of tissue vacuolation associated with LAMP-2 staining was considered a positive finding of PLD and the decision threshold for the end point of skeletal muscle necrosis was set at a mild or greater incidence of necrosis in type II skeletal muscle.
Of the 11 phospholipids measured at detectable levels in urine, di-22:6-BMP had the highest diagnostic accuracy for PLD (Figure 2 ) and the lowest for skeletal muscle necrosis (Figure 3 ). An AUC of 0.931 for detection of PLD by di-22:6-BMP was significantly higher than AUCs calculated for 9 of the 10 other phospholipids in this analysis (P < .01; Table 3 ). For the end point of skeletal muscle injury, all of the other phospholipids tested were significantly better predictors of skeletal muscle necrosis than di-22:6-BMP, based on AUC comparisons (P < .01; Table 3). At a specificity of 95%, di-22:6-BMP had a sensitivity of 66% for detecting PLD, which corresponded to a 4-fold change threshold, and a sensitivity of 30% for detecting skeletal muscle necrosis in this analysis. The phospholipids that showed the best combination of sensitivity and specificity for detection of skeletal muscle necrosis (ie, an AUC > 0.9) were 2 species of PI (18:0,20:4-PI and 16:0,20:4-PI) and 2 species of PC (18:0,20:4-PC and 18:0,18:2-PC). Of the 4 phospholipids, 18:0,20:4-PI had the highest sensitivity (66%) at 95% specificity for this end point, which corresponded to a decision threshold of 9-fold above control levels.
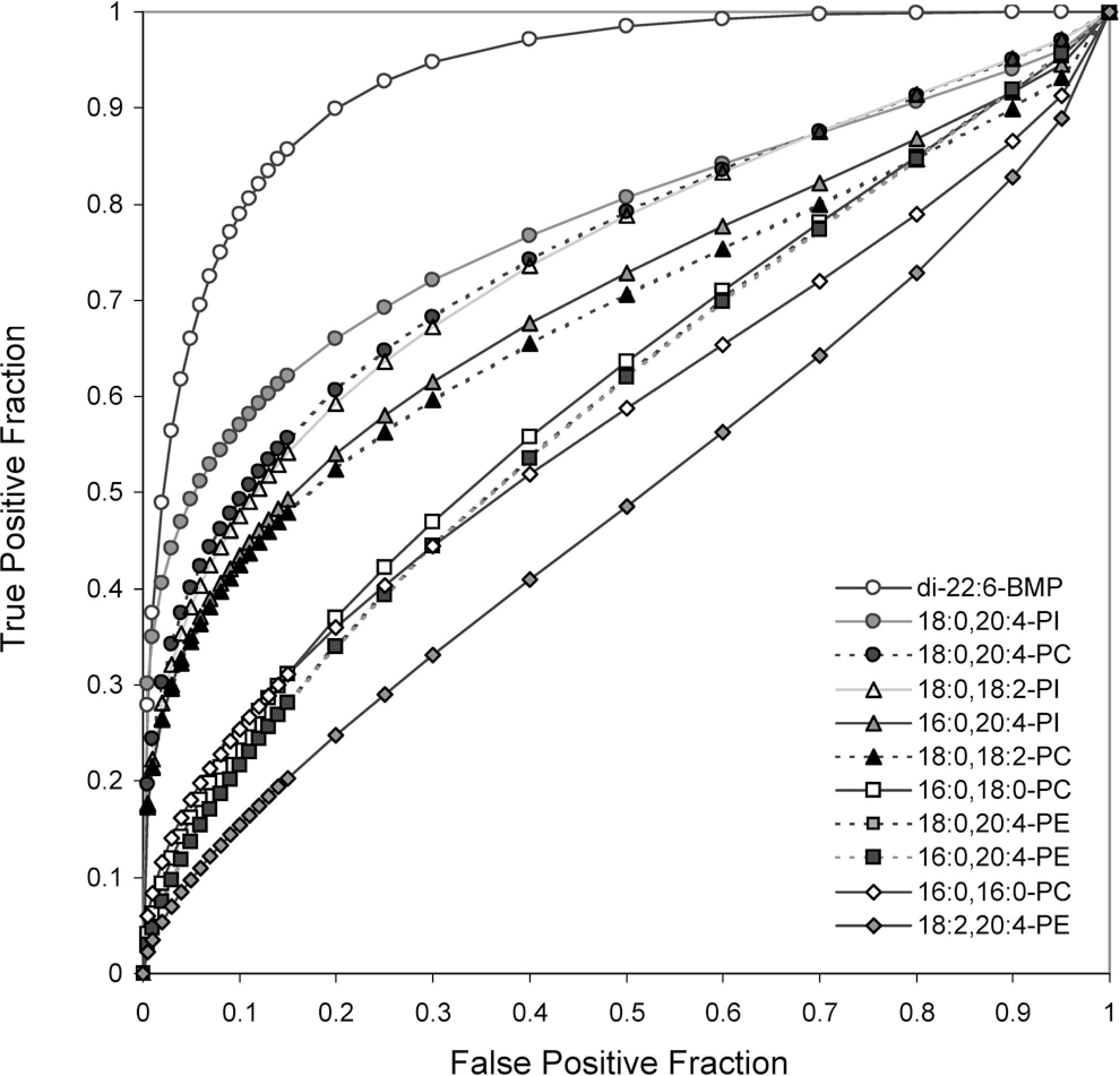
Receiver operating characteristic (ROC) plot analysis of diagnostic accuracy of individual urinary phospholipids for phospholipidosis. Minimal or higher levels of mesenteric lymph node vacuolation were scored as phospholipidosis (PLD) positive. Phospholipid levels were normalized to urinary creatinine level and transformed by dividing by the average time-matched control value. True positives = 24; true negatives = 20.
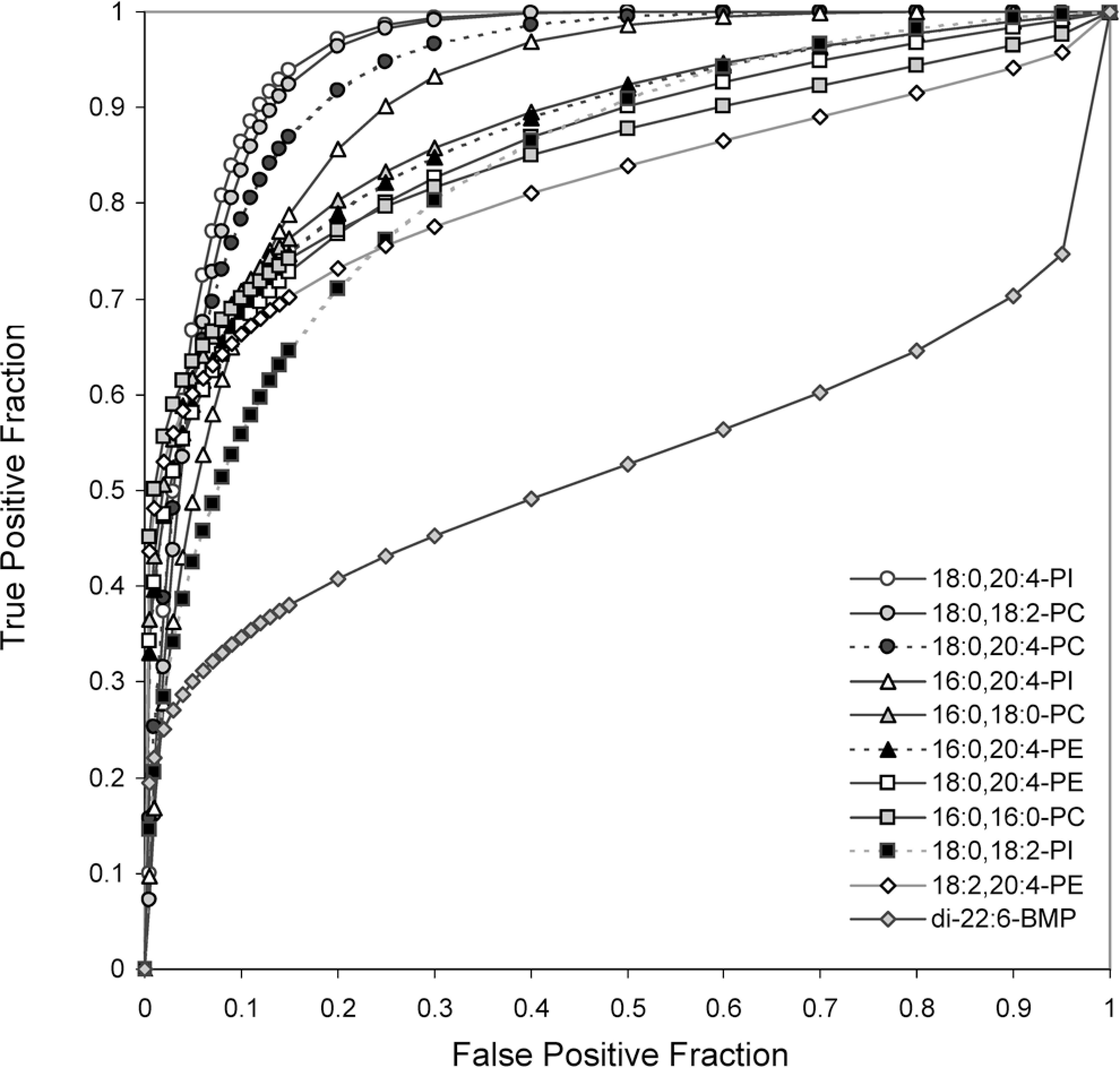
Receiver operating characteristic (ROC) plot analysis of diagnostic accuracy of individual urinary phospholipids for skeletal muscle necrosis. Samples with mild or greater levels of necrosis in type II skeletal muscle were scored positive. Phospholipid levels were normalized to urinary creatinine levels and transformed by dividing by the average time-matched control value. True positives = 16; true negatives = 28.
Comparison of AUCs for Individual Phospholipids in Urine for Detection of PLD or Tissue Injury

Abbreviations: AUC, area under curve; DEAEH, 4,4’-Diethylaminoethoxyhexesterol in male Fisher rats; SV, simvastatin in female SD rats; GM, gentamicin in male SD rats; CP, cisplatin in male SD rats; AZ, azithromycin in male SD rats; PLD, phospholipidosis; TP, true positives; TN, true negatives; nd, not detected in at least one study in a data set; Kim-1, kidney injury molecule 1; BMP, bis(monoacylglycerol)phosphate; PI, phosphatidylinositol; PE, phosphatidylethanolamine; PC, phosphatidylcholine.
a Decision threshold is a minimal or greater incidence of vacuolation in mesenteric lymph node.
b Decision threshold is a mild or greater incidence of necrosis in type II skeletal muscle at necropsy.
c Decision threshold is a 4.5-fold or greater increase in urinary Kim-1 levels normalized to urinary creatinine, relative to the time-matched control average.
d Significantly different from the AUC for BMP for detection of the same end point (P < .01).
Diagnostic Accuracy of Urinary Phospholipids for Renal Injury
Phospholipiduria has been observed in the clinic and in nonclinical studies in association with drug-induced kidney injury. 14 –19 To compare the diagnostic accuracy of individual urinary phospholipids for detecting kidney injury, we assayed a set of 88 urine samples from studies of drugs that induce nephrotoxicity (gentamicin and cisplatin) or PLD in the kidney (gentamicin and azithromycin) in nonfasted male Sprague Dawley rats. In one study, groups of rats received intramuscular (im) injections of gentamicin for 3 consecutive days at doses of 0, 75, or 150 mg/kg/d. 24 Urine collected on days 1 to 15 was analyzed for phospholipid content. In a second study, rats received a single ip dose of 0, 2, or 3 mg/kg cisplatin and urine was collected during the last 18 hours of the 72-hour study. Phospholipid levels were analyzed in urine samples from 5 cisplatin-treated animals that showed a 10-fold or greater increase in Kim-1 and from 6 controls. In a third study, azithromycin was administered by gavage at a dose of 0 or 150 mg/kg/d for 17 days and urine collected during the final 18 hours of the study. A significant increase in LAMP-2 immunohistochemical staining was observed in the renal cortex of rats treated with azithromycin compared to controls (data not shown). No evidence of kidney toxicity was seen with azithromycin treatment.
Gentamicin treatment induced a dose- and time-related increase in renal tubular epithelial cell necrosis and apoptosis that peaked in incidence on study day 7 (day 4 of recovery). 24 Urinary Kim-1 levels were also observed to be significantly increased at days 7 and 10 in the 150 mg/kg dose group (Table 4 ). Urinary levels of di-22:6-BMP were significantly elevated on the third consecutive day of dosing with 75 or 150 mg/kg gentamicin but not after dosing cessation (Table 4). The most extensive change in urinary phospholipid levels occurred at day 7 in the 150 mg/kg gentamicin dose group when significant elevations were seen in 8 of the 12 phospholipids assayed in urine (P < .05). Cisplatin treatment induced a significant increase in urinary levels of 3 species of PC, 18:0,20:4-PI, and 18:0,20:4-PE but had no effect on di-22:6-BMP. Azithromycin treatment induced a significant increase in di-22:6-BMP, 18:0,20:4-PI, and 16:0,16:0-PC levels in urine (P < .05) but had no effect on 5 other phospholipids or on urinary Kim-1 levels.
Fold Change in Mean Urinary Levels of Biomarkers in Gentamicin Treatment Groups
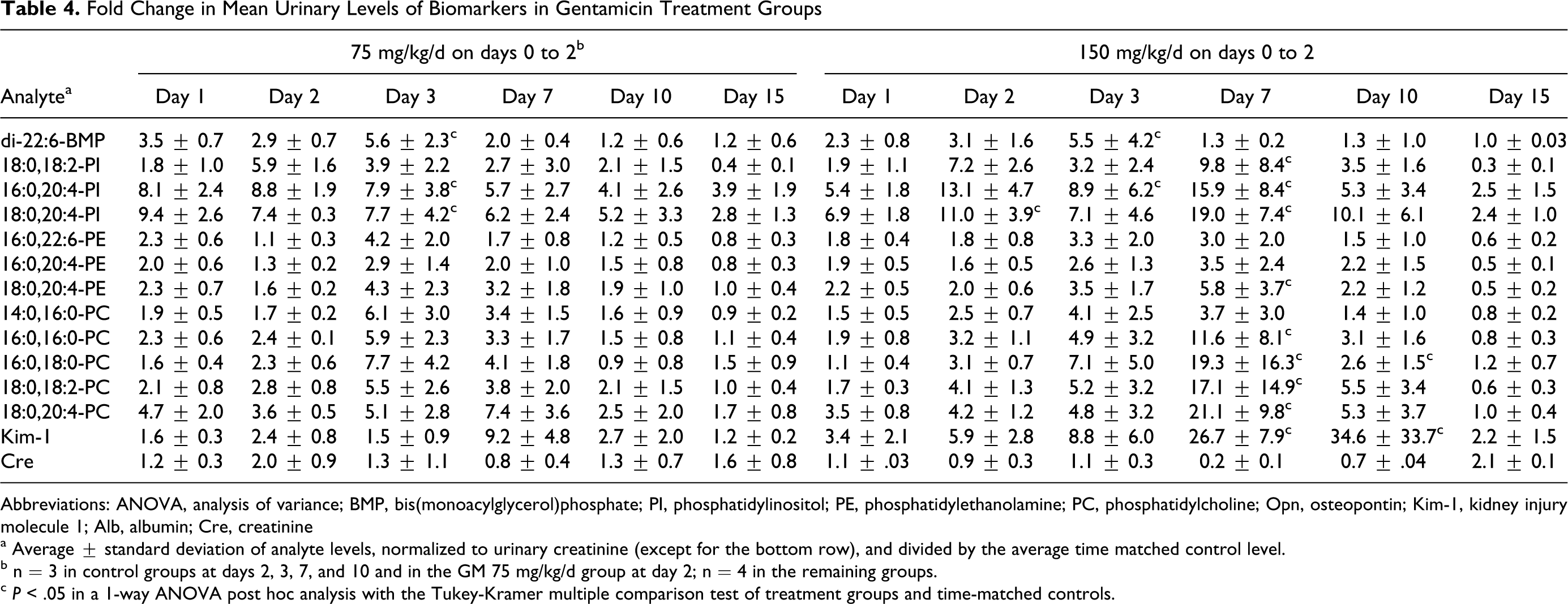
Abbreviations: ANOVA, analysis of variance; BMP, bis(monoacylglycerol)phosphate; PI, phosphatidylinositol; PE, phosphatidylethanolamine; PC, phosphatidylcholine; Opn, osteopontin; Kim-1, kidney injury molecule 1; Alb, albumin; Cre, creatinine
a Average ± standard deviation of analyte levels, normalized to urinary creatinine (except for the bottom row), and divided by the average time matched control level.
b n = 3 in control groups at days 2, 3, 7, and 10 and in the GM 75 mg/kg/d group at day 2; n = 4 in the remaining groups.
c P < .05 in a 1-way ANOVA post hoc analysis with the Tukey-Kramer multiple comparison test of treatment groups and time-matched controls.
The diagnostic accuracy of 8 individual urinary phospholipids for detecting kidney injury was assessed in an ROC plot analysis using a decision threshold of a 4.5-fold or greater increase in urinary Kim-1 levels relative to the average time-matched control value. This level of change in urinary Kim-1 was the 95% specificity threshold for detection of renal histopathology (at a mild level of incidence) on days 1 to 15 of a study on the reversibility of gentamicin-induced renal injury and renal injury biomarkers. 24 At this threshold, all samples from animals treated with cisplatin and 19 of the 47 urine samples from animals in gentamicin dose groups were positive for renal injury.
Five of the 7 urinary phospholipids assayed in this set were significantly better predictors of proximal tubule injury than di-22:6-BMP, based on AUC comparisons (P < .01; Table 3). For all 8 phospholipids, the AUCs calculated for detection of proximal tubule injury were not significantly different from the AUCs calculated for detection of skeletal muscle necrosis in an independent sample set (P > .01). However, there was a significant difference between the AUC for detection of renal injury and for detection of PLD for 4 of the 8 phospholipids assayed in both sample sets (P < .01).
Discussion
In this study, we assessed the specificity of urinary di-22:6-BMP for PLD in order to differentiate this response from phospholipiduria. Phospholipiduria has been observed during treatment with aminoglycoside antibiotics and by nephrotoxicants that are not associated with PLD, such as mercuric chloride and nonsteroidal anti-inflammatory drugs. 18,19 Urinary phospholipids may originate from different cellular sites, depending on the mechanism of toxicity. In a comparative study of HgCl2 and gentamicin, using doses that induced renal proximal tubule injury and increased total urinary phospholipid levels by about 10-fold, HgCl2 treatment was associated with a much greater increase in urinary levels of brush border enzyme activity and of sphingomyelin, a phospholipid enriched in plasma membranes, than was gentamicin treatment. 17 The phospholipid composition of urine and the appearance of multilamellar bodies in urinary sediment was consistent with myeloid bodies being the principal source of phospholipids in gentamicin-induced phospholipiduria. Although this study by Josepovitz et al 17 did not assay di-22:6-BMP, it suggests that necrotic injury that induces phospholipiduria through the shedding of brush border membranes may not significantly increase levels of this endosome-enriched phospholipid in urine. Using the phospholipidotic drug DEAEH, we found that urinary di-22:6-BMP maintains high specificity for PLD when assayed in samples with a background of drug-induced renal or skeletal muscle injury.
Of the 5 drugs in this study, only DEAEH and gentamicin caused a significant increase in urinary di-22:6-BMP levels and phospholipiduria. With gentamicin, the peak elevation in urinary di-22:6-BMP levels appeared on the third and final day of consecutive daily dosing, while the peak in phospholipiduria occurred 4 days post-dosing on study day 7. The results with di-22:6-BMP are in agreement with evidence that drug accumulation in tissues is a key characteristic of PLD, along with reversibility upon termination of drug administration. 1 Gentamicin nephrotoxicity occurs as a result of cytosolic redistribution of drug that has accumulated in renal tubules, which compromises the integrity and function of the endoplasmic reticulum, mitochondria, and lysosomes. 2 The largest increase in PC, PE, and PI species in urine occurred at the gentamicin dose and time point with the highest incidence of renal tubule necrosis and highest elevation in urinary Kim-1 level (Table 4). Phospholipiduria has been shown to correlate better with renal injury than PLD in a comparative study of 4 aminoglycoside antibiotics. 29 In that study, the rank order for induction of phospholipiduria was the same as the rank order for kidney injury, based on serum creatinine levels, and was inverse to the rank order for induction of phospholipid levels in renal cortex.
Although significant increases in urinary di-22:6-BMP levels were seen in most DEAEH treatment groups, the increase was not dose dependent at day 7. In addition, urinary di-22:6-BMP levels in the 50 mg/kg DEAEH dose group at day 14 were not significantly elevated compared to controls and were lower than levels in the 25 mg/kg group. These results do not appear to be due to dosing errors, since serum levels of di-22:6-BMP and tissue levels of DEAEH and its metabolites in liver both increased with dose (K.L.T., manuscript in preparation). The observed lack of a dose response in DEAEH-induced urinary levels of di-22:6-BMP at day 7 may be due to dose-dependent differences in the time of maximal excretion of di-22:6-BMP into urine. Other investigators have observed that peak elevations in urinary phospholipids including BMP can occur early during chronic dosing with phospholipidotic compounds. 30 It is also possible that renal clearance of DEAEH had not reached steady state by day 7 due to the drug’s lipophilicity or pharmacologic action.
The relationship between the tissue site/sites of PLD and di-22:6-BMP levels in biofluids is currently not well understood. The kidney is the probable source of urinary di-22:6-BMP that was increased with DEAEH administration since ultrastructural alterations of lysosomes were observed in the PTECs of the kidney at the high dose level at 14 days. However, we did observe a good correspondence between elevations in urinary di-22:6-BMP and incidence of PLD in the lymph node. Di-22:6-BMP may be excreted from the lymph node or other PLD tissues into the blood. Di-22:6-BMP may resist hydrolysis by phospholipases and may be removed from circulation by the kidney and excreted in urine.
In this study, phospholipiduria correlated well with skeletal muscle injury in several DEAEH treatment groups and with simvastatin treatment. Neither drug induced necrotic injury in the kidney or had a significant effect on urinary Kim-1 levels, blood urea nitrogen, or serum creatinine. The selective effect of the 100 mg/kg/d dose of DEAEH on urinary Opn levels may be an indicator of cryptic renal injury in distal tubules or of drug-induced hepatotoxicity or myopathy. Osteopontin is a serum marker of hepatic injury mediated by inflammation as well as a urinary marker of kidney injury. 31,32 Simvastatin treatment was associated with greater than 5-fold elevations in urinary renal papillary antigen 1 and albumin levels but less or no change was seen in other renal injury biomarkers (α-glutathione S-transferase, Kim-1, Opn, Lpn2, and clusterin). The difference in response that is seen in site-specific biomarkers of proximal tubular segments and of collecting ducts may be indicative of renal papillary impairment, followed by collecting duct dysfunction that occurs as a result of simvastatin-induced myopathy.
In a combined analysis of 132 urine samples from preclinical studies of drugs that induce PLD and/or tissue injury, we have shown that di-22:6-BMP has a distinctly different profile from other, more common species of phospholipids. While many PC, PE, and PI species can be elevated in urine as a result of direct or indirect renal injury, di-22:6-BMP maintained specificity for PLD in our study. The data provide evidence supporting the use of di-22:6-BMP as a urinary biomarker of PLD in rats.
Footnotes
The authors declare no conflicting interests. The findings and conclusions in this paper have not been formally disseminated by the US Food and Drug Administration and should not be construed to represent any Agency determination or policy.
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This project received funding from the FDA Critical Path Initiative.
