Abstract
The correlation between 52 physicochemical parameters and mean residence time (MRT) for 27 drugs used in human and dog were investigated. The physicochemical parameter values calculated provided a basis for deriving a series of arithmetic expressions, which were used to build a mathematical model describing the relationship between them and the MRT values. From the entire set of analyzed parameters, a subset of 14 was identified that contributed to the derivation of an arithmetic expression:
the value of which is highly correlated with the MRT value in dogs (P < .001) and allowed prediction of the MRT predicted (MRT(pred)). In humans, no correlation was found that allowed the calculation of MRT(pred). These results indicate that predicting the pharmacokinetics of any specific drug for humans based on pharmacokinetic data obtained in the dog should be undertaken with knowledge of the inherent limitations.
Introduction
Mean residence time (MRT) and half-life in elimination phase (t1/2kel) are the key pharmacokinetic parameters describing elimination of a drug from the body. The elimination half-life in the in vivo studies is usually determined based on the last 3 to 4 points obtained on the concentration versus (vs) time curve created for the analyzed drug. 1 These values/points are qualified for the analysis by the person performing it. Based on these selected points, the elimination rate constant (kel) is calculated using least-squares analysis, and the value of t1/2kel is determined based on the kel. The determination of t1/2kel often bears a significant error. The reasons for that are numerous, for example: (a) the sampling period in the study is too short; (b) the lower limit of quantification (LLOQ) for the analytical method is too high; the LLOQ is higher than the concentration value obtained in the last sampling point; (c) sampling points in the elimination phase are distributed incorrectly; (d) the selection of points on the concentration versus time (C-T) curve for calculating t1/2kel is incorrect; (e) there are fluctuations on the C-T curve in the elimination phase, for example, due to enterohepatic circulation, and so on. 2
As to MRT, this parameter is calculated based on the values of area under the curve (AUC) and area under the first moment curve (AUMC). Therefore, the MRT calculation from data obtained in in vivo experiments is not related in any way to specific parts of the C-T curve but rather to all observations starting after the absorption phase until the end of drug elimination. The MRT calculation with the use of the equation
Drug elimination is a result of several different processes occurring simultaneously within the body. In many cases, it can be additionally modified by the processes of reabsorption caused by the circulation of the drug with lymph, bile, or saliva; metabolic induction; chiral inversion; resorption in renal tubules; reabsorption being a result of enzymatic decomposition conducted by intestinal flora and other metabolic processes. 5–8
Currently some of the pharmacokinetic parameters, processes, and phenomena observed in vivo are believed to be primarily determined by or associated with the molecular structure of the drug. The examples of such attempts include the analysis of the relationship between molecular structure of the drug and its enterohepatic circulation, 9 ,10 volume of distribution, 11,12 clearance 13–16 elimination half-life, 17 and/or absolute bioavailability. 18–21 Such studies are usually performed in relation to data from in vivo experiments conducted in a single species and/or with the use of data from a limited group of homologous drugs.
Therefore, the aim of this study was an attempt to find a correlation between physicochemical characteristics of a drug and its MRT determined in vivo both in human and in dog for the group of drugs of diverse molecular structure and pharmacological effects.
Materials and Methods
Tested Group of Drugs
The analysis was performed using a group of 27 drugs, for which the MRT (as statistical moment:
Values of Mean Residence Time (MRT) and Half-Life in Elimination Phase (t1/2kel) for the group of 27 Drugs Administered Intravenously in Human22 and dog
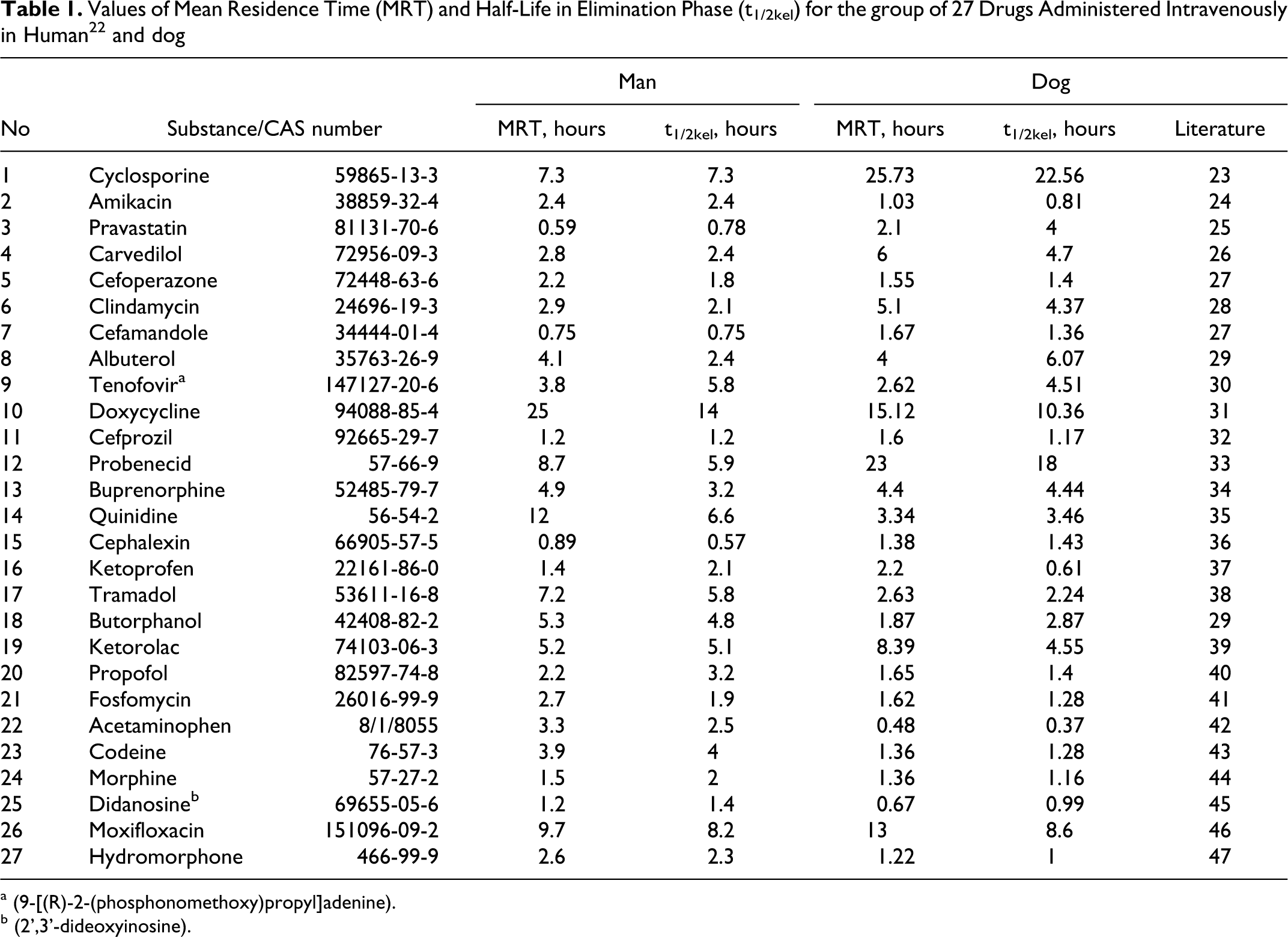
a (9-[(R)-2-(phosphonomethoxy)propyl]adenine).
b (2',3'-dideoxyinosine).
In Silico Calculations
The analysis of the selected physiochemical parameters of the proposed structures was preceded by the transformation of a traditional chemical structure record to sdf files. The properties and descriptors of analyzed structures were predicted using QikProp 3.1 from Schrödinger package (v 31207). QikProp was run in normal mode. The three-dimensional structures of compounds were prepared in LigPrep 2.2 using settings recommended in the QikProp's user manual. In the case of compounds that are chiral or undergo tautomerization up to 32 stereoisomers were generated for each compound or tautomer. The software allowed the calculation of the following 51 parameters: stars—the number of property or descriptor values that fall outside the 95% range of similar values for known drugs; AMINE—number of nonconjugated amine groups; amidine—number of amidine and guanidine groups; ACID—number of carboxylic acid groups; AMIDE—number of nonconjugated amide groups; rotor—number of nontrivial (not CX3), nonhindered (not alkene, amide, small ring) rotatable bonds; FG—number of reactive functional groups; CNSa—predicted central nervous system activity on a −2 (inactive) to +2 (active) scale; MW—molecular weight of the molecule; p—computed dipole moment of the molecule; SASA—total solvent accessible surface area in square angstroms using a probe with a 1.4 Å radius; FOSA—hydrophobic component of the SASA (saturated carbon and attached hydrogen); FISA—hydrophilic component of the SASA (SASA on N, O, and H on heteroatoms); PISA—π (carbon and attached hydrogen) component of the SASA; WPSA—weakly polar component of the SASA (halogens, P, and S); volume—total solvent-accessible volume in cubic angstroms using a probe with a 1.4 Å radius; HBD—estimated number of hydrogen bonds that would be donated by the solute to water molecules in an aqueous solution.; HBA—estimated number of hydrogen bonds that would be accepted by the solute from water molecules in an aqueous solution; dip2/V—square of the dipole moment divided by the molecular volume; ACxDN5/SA—index of cohesive interaction in solids; Glob—globularity descriptor; α—predicted polarizability in cubic angstroms; QPlogPC16—predicted hexadecane/gas partition coefficient; QPlogPoct‡—predicted octanol/gas partition coefficient; QPlogPw—predicted water/gas partition coefficient; QPlogPo/w—predicted octanol/water partition coefficient; QPlogS—predicted aqueous solubility; CIQPlogS—conformation-independent predicted aqueous solubility; QPlogHERG—predicted IC50 value for blockage of HERG K+ channels; Caco2—predicted apparent Caco2 cell permeability in nm/sec; QPlogBB—predicted brain/blood partition coefficient; QPPMDCK—predicted apparent MDCK cell permeability in nm/sec; LogKp—predicted skin permeability; IP—calculated ionization potential; EA—calculated electron affinity; #metab—number of likely metabolic reactions; QPlogKhsa—prediction of binding to human serum albumin; HumanOralAbsorption—predicted qualitative human oral absorption; PercentHuman-OralAbsorption—predicted human oral absorption on 0% to 100% scale; SAF—solvent-accessible surface area of fluorine atoms; SAamideO—solvent-accessible surface area of amide oxygen atoms; PSA—Van der Waals surface area of polar nitrogen and oxygen atoms; NandO—number of nitrogen and oxygen atoms; RuleOfFive—number of violations of Lipinski’s rule of 5; Rule Of Three—number of violations of Jorgensen’s rule of 3; ringatoms—number of atoms in a ring; in34—number of atoms in 3- or 4-membered rings; in56—number of atoms in 5- or 6-membered rings; noncon—number of ring atoms not able to form conjugated aromatic systems; #nonHatm—number of heavy atoms (nonhydrogen atoms); and Jm—the predicted maximum transdermal transport rate. The values of the partition coefficient octanol/water (XLogP) for the analyzed group of drugs were drawn from PubMed database. 48
Creation of Expressions
While searching for a correlation between the grouped in silico parameters and the MRT value, a number of arithmetic expressions were created. Overall, several forms of different expressions were analyzed. Table 2 presents an example of designing such expressions. By successively incorporating various physicochemical parameter values a series of arithmetic expressions was derived. The arithmetic expressions were constructed but not based on the values of individual parameters but rather by combining several of the parameters together (Figure 1 ). The equation derived served as a means to calculate predicted MRT values (MRT(pred)).
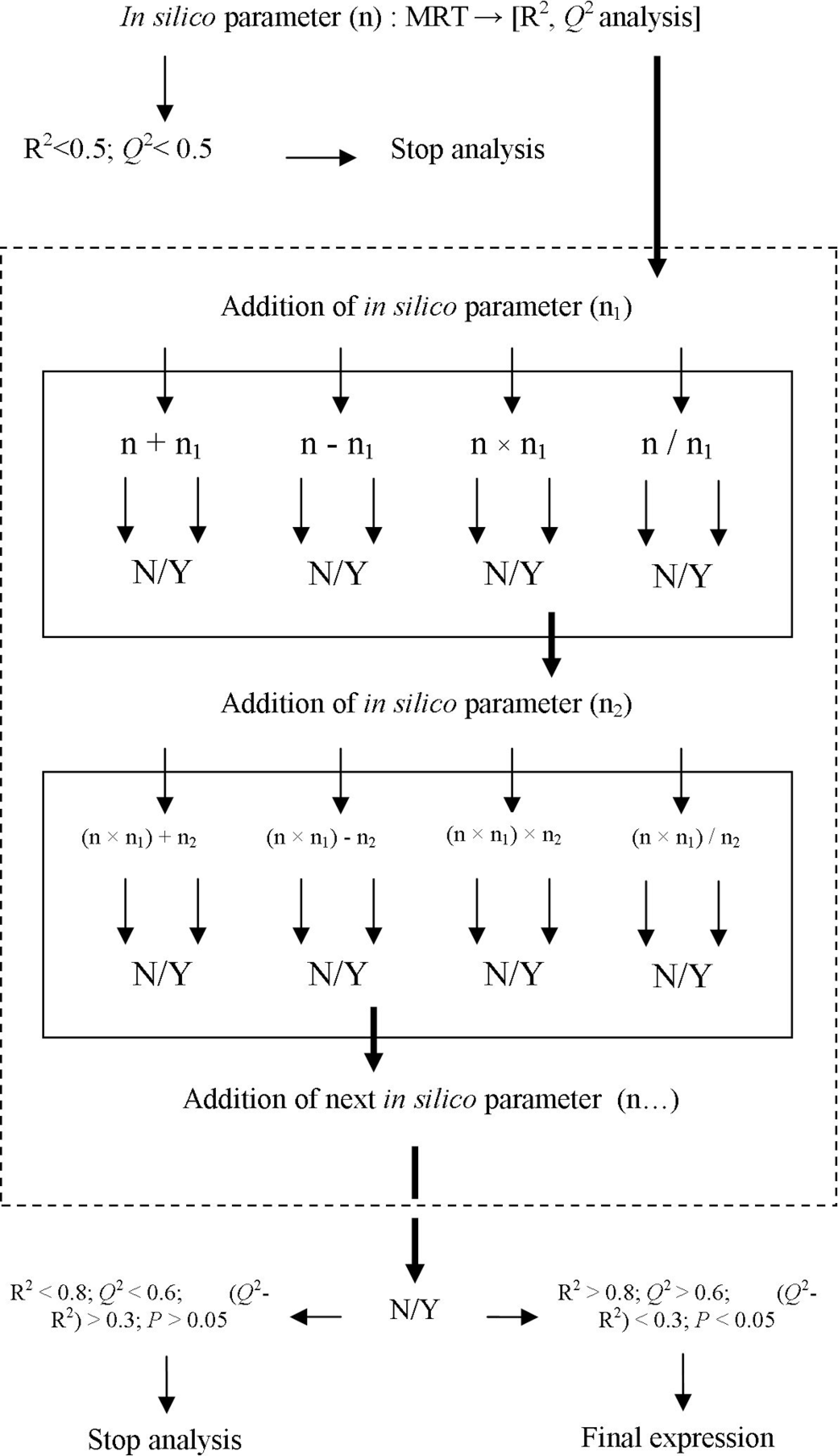
Examples of Arithmetic Expressions Created for Selected Physiochemical Parameters in the Group of 27 Drugs Administered Intravenously and Their Correlation With Mean Residence Time in Human and Dog

Abbreviations: WPSA, weakly polar component of the SASA (halogens, P, and S); PSA, Van der Waals surface area of polar nitrogen and oxygen atoms; XLogP, partition coefficient octanol/water; LogKp, predicted skin permeability; Caco2, predicted apparent Caco2 cell permeability; EA, calculated electron affinity; AMINE, number of nonconjugated AMINE groups; AMIDE, number of nonconjugated amide groups; IP, calculated ionization potential; ACID, number of carboxylic acid groups; FG, number of reactive functional groups; SAF, solvent-accessible surface area of fluorine atoms; PISA, π (carbon and attached hydrogen) component of the SASA; MW, molecular weight of the molecule.
Statistical Analysis and Validation
A statistical analysis was performed using Microsoft Office Excel software. The polynomial correlation and regression analysis functions were used for the determination of a relationship between the mathematical model value and MRT. All correlations were confirmed by the Fischer test (confidence interval 95%) and the standard estimation error (SEE) was calculated (differences with P < .05 were regarded as statistically significant). Goodness of fit was evaluated based on MRT and MRT(pred) values. The coefficient of determination (R
2
) of the observed versus predicted data was analyzed. The leave-one-out (LOO) method was used for model cross-validation. Squared cross-validated correlation coefficient (Q
2
) parameter and difference between Q
2
and R
2
was calculated as measure of the internal performance and model predictive ability. Q
2
was calculated according to the formula:
Results
For the tested group of drugs, no significant correlation was demonstrated between MRT values in human and dog; the R 2 for linear fit of logarithmically transformed MRT values for the 2 species was 0.4194 (Figure 2 ). The relationship between MRT and t1/2kel in both human and dog was found to be linear for the tested group of drugs (Figure 3 ).
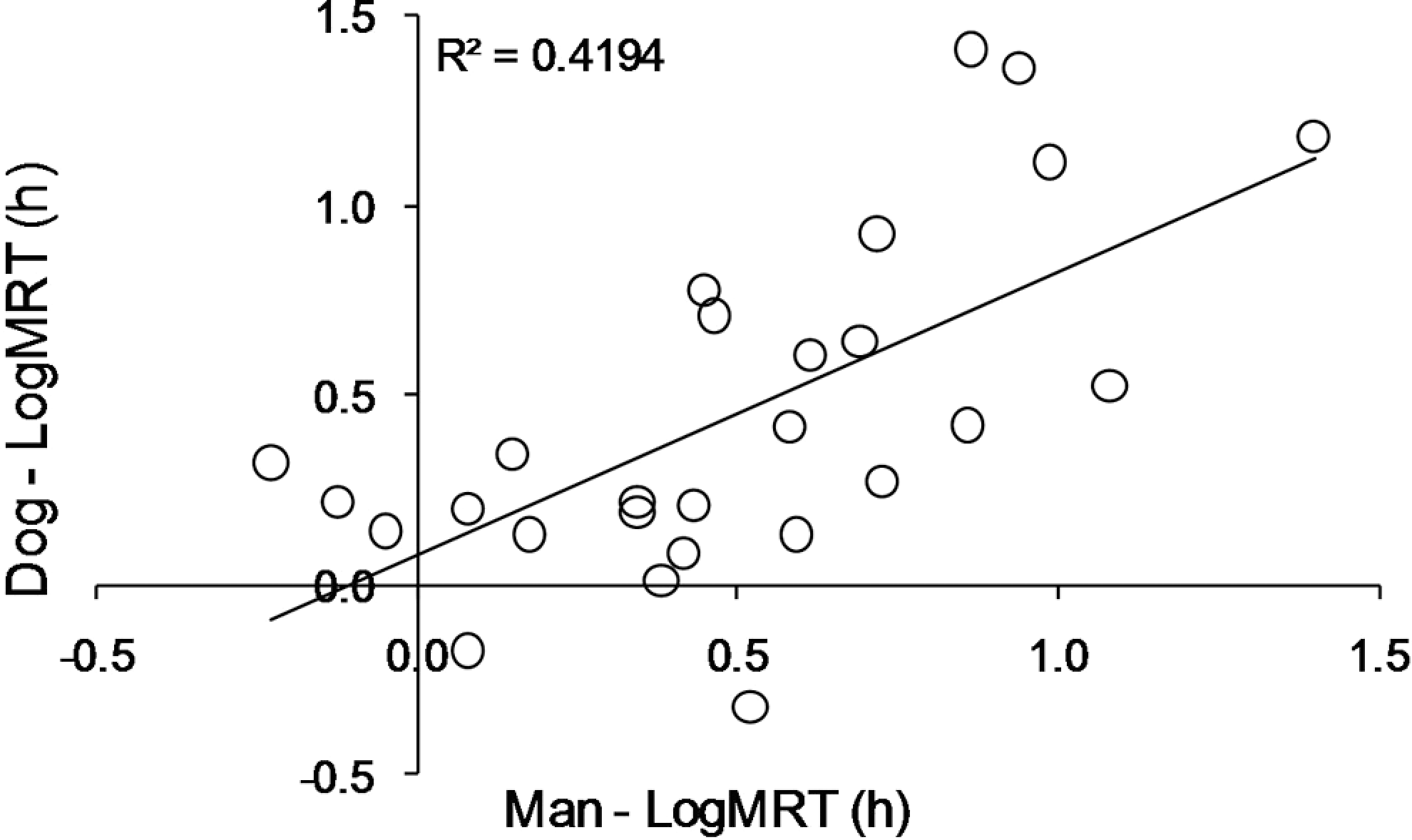
Linear correlation between logarithmically transformed values of mean residence time (LogMRT) in the tested group of 27 drugs administered intravenously in man and dog.
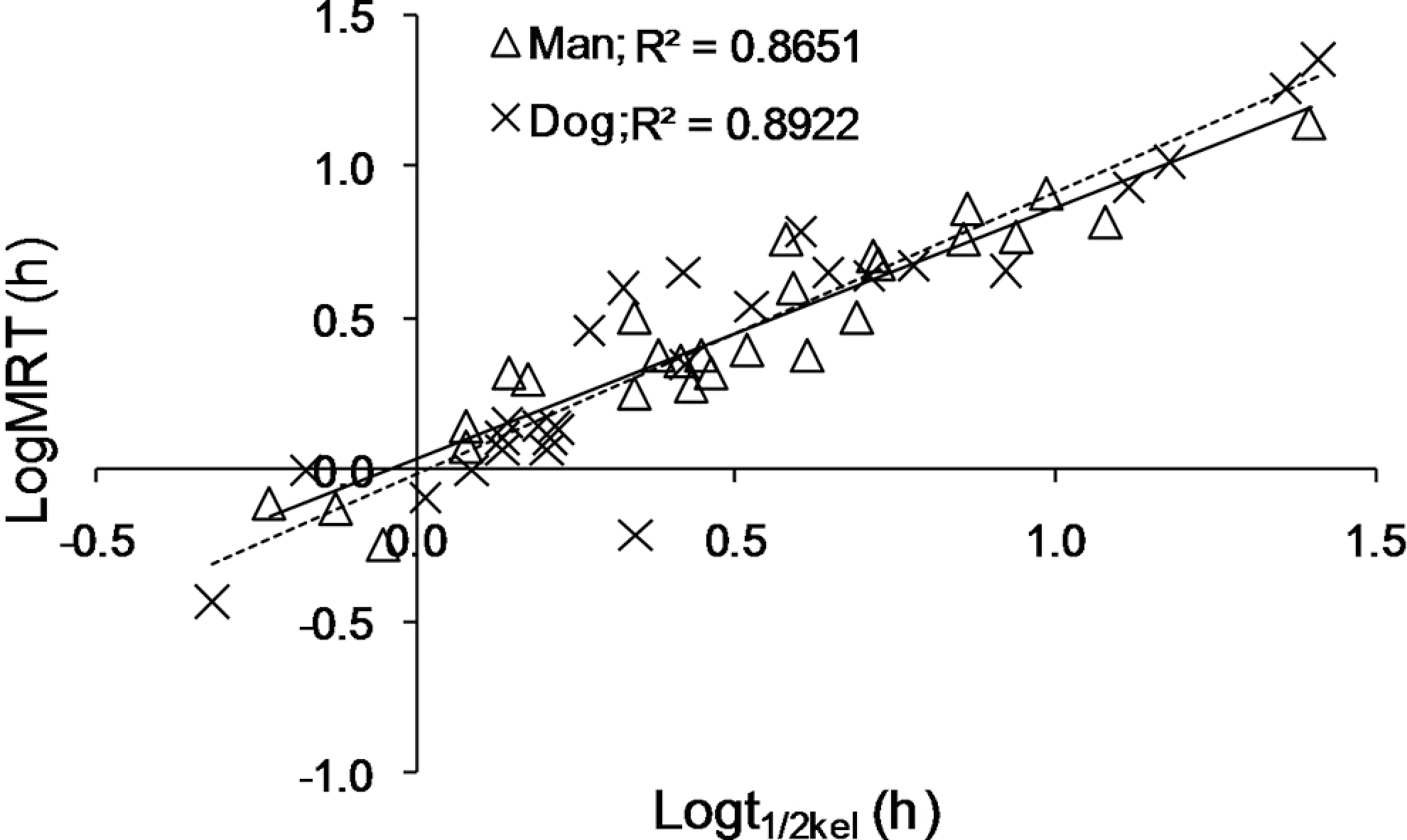
Linear correlation between logarithmically transformed values of mean residence time (LogMRT) and logarithmically transformed values of the half-life in the elimination phase (Log t1/2kel) in the tested group of 27 drugs administered intravenously in human (solid line) and dog (dashed line).
In either of the tested species, no significant direct dependence between the MRT value and physicochemical parameters analyzed was detected for the tested group of drugs. The highest R 2 values obtained for the relationship between single physicochemical parameter and MRT were 0.3180 (AMIDE) and 0.1114 (FG) for the dog and human, respectively; however, they were also determined for different parameters in each of the species (Table 3 ).
Correlations of 14 Physiochemical Parameters Calculated In Silico With Mean Residence Time (MRT) Value for the Group of 27 Drugs Administered Intravenously in Human and Dog
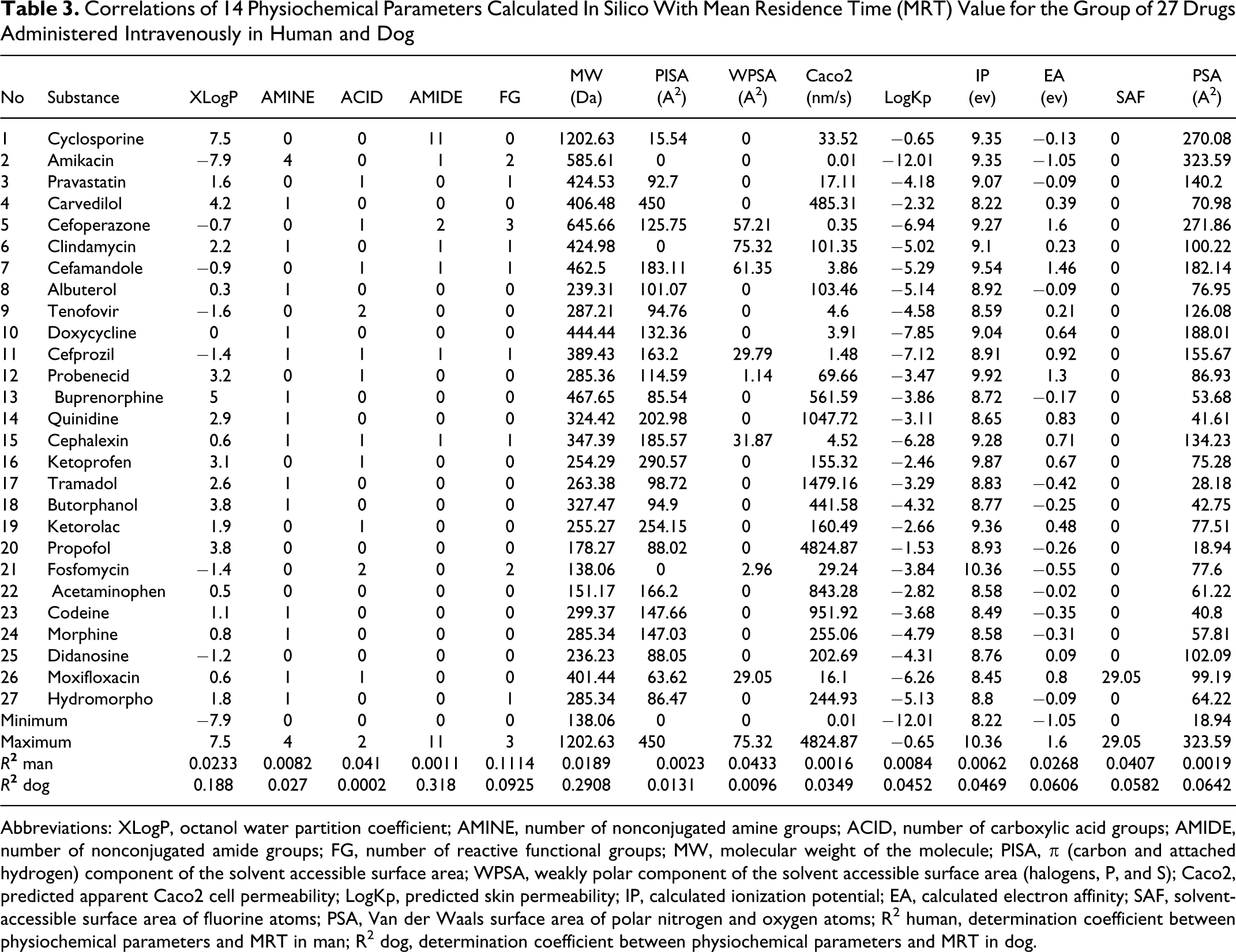
Abbreviations: XLogP, octanol water partition coefficient; AMINE, number of nonconjugated amine groups; ACID, number of carboxylic acid groups; AMIDE, number of nonconjugated amide groups; FG, number of reactive functional groups; MW, molecular weight of the molecule; PISA, π (carbon and attached hydrogen) component of the solvent accessible surface area; WPSA, weakly polar component of the solvent accessible surface area (halogens, P, and S); Caco2, predicted apparent Caco2 cell permeability; LogKp, predicted skin permeability; IP, calculated ionization potential; EA, calculated electron affinity; SAF, solvent-accessible surface area of fluorine atoms; PSA, Van der Waals surface area of polar nitrogen and oxygen atoms; R 2 human, determination coefficient between physiochemical parameters and MRT in man; R 2 dog, determination coefficient between physiochemical parameters and MRT in dog.
Of the 52 parameters analyzed, only 14 (XLogP, AMINE, ACID, AMIDE, FG, MW, PISA, WPSA, Caco2, LogKp, IP, EA, SAF, PSA; Table 3) contributed value to the creation of the arithmetic expression, the value of which highly correlated with MRT. This expression was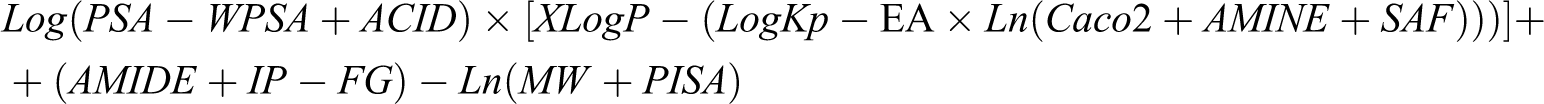
For the polynomial fit, a correlation of statistical significance (P < .001; R
2 = .8227; Figure 4
) was demonstrated between the arithmetic expression and MRT value in the dog. This correlation has been illustrated by the following equation:
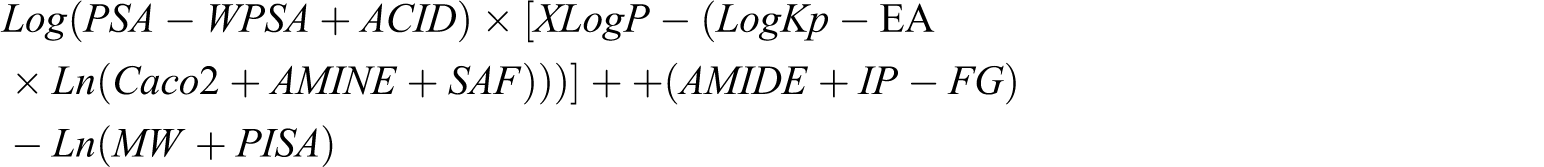
Polynomial correlation between values of mean residence time (MRT) and values of arithmetic expression
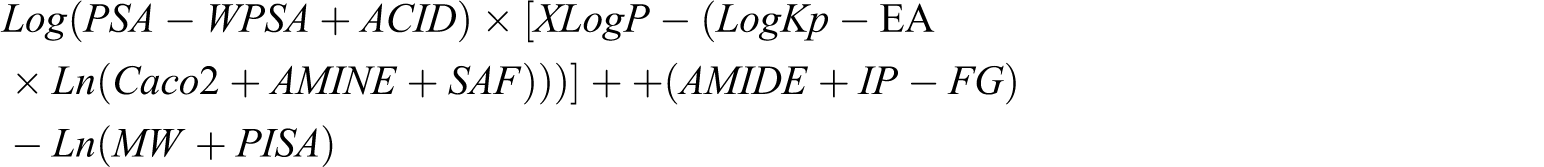
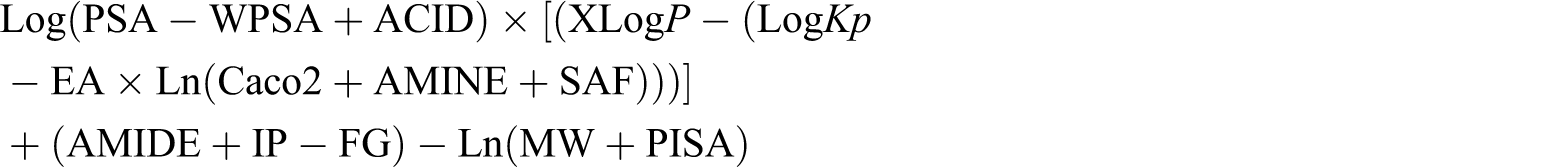
Polynomial correlation between value of mean residence time (MRT) and value of arithmetic expression
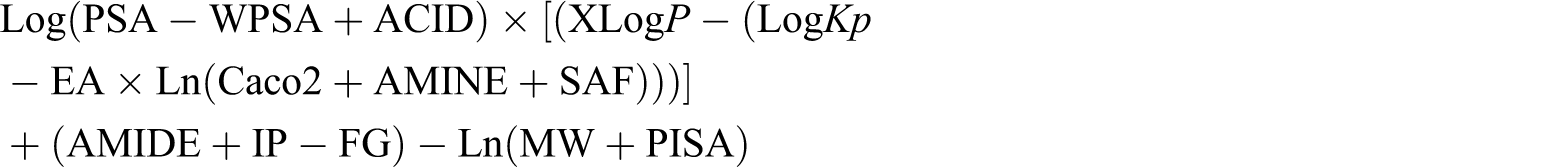
Values of the Arithmetic Expression
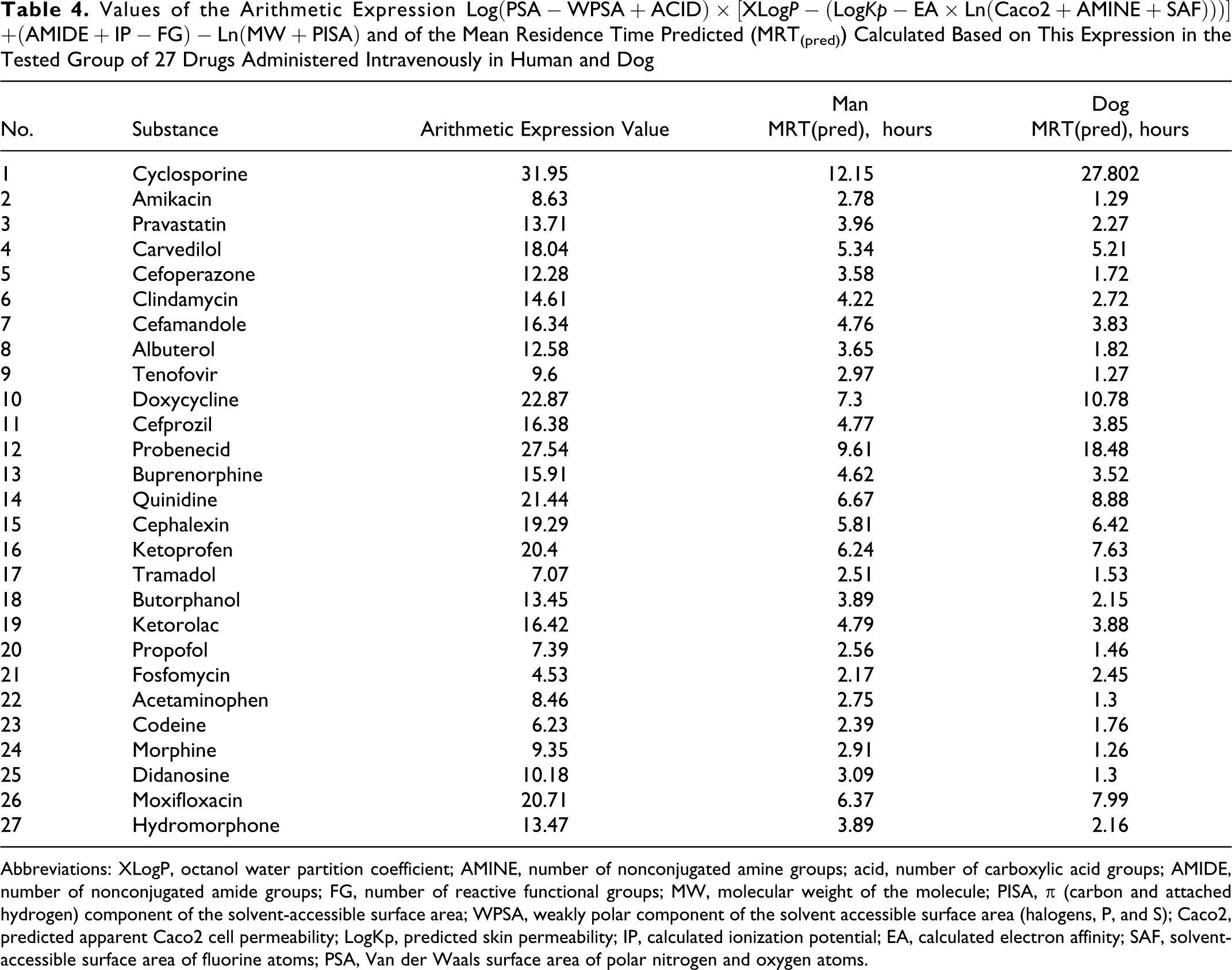
Abbreviations: XLogP, octanol water partition coefficient; AMINE, number of nonconjugated amine groups; acid, number of carboxylic acid groups; AMIDE, number of nonconjugated amide groups; FG, number of reactive functional groups; MW, molecular weight of the molecule; PISA, π (carbon and attached hydrogen) component of the solvent-accessible surface area; WPSA, weakly polar component of the solvent accessible surface area (halogens, P, and S); Caco2, predicted apparent Caco2 cell permeability; LogKp, predicted skin permeability; IP, calculated ionization potential; EA, calculated electron affinity; SAF, solvent-accessible surface area of fluorine atoms; PSA, Van der Waals surface area of polar nitrogen and oxygen atoms.
The linear correlation between MRT and MRT(pred) in dog and human is shown in Figure 6. The dependence between MRT and MRT(pred) values was found significant only in the case of the dog (F = 116.03; SEE = 2.84; P < .001; Q 2 = 0.666). In dog, differences between R 2 and q 2 did not exceed 0.3, which confirms that the model was not overfitted and did not have irrelevant values or outliers in the data. Statistically significant dependence was not found for human (F = 6.80; SEE = 2.09; P > .05; Q 2 = 0.525); Moreover, the difference between R 2 and Q 2 (<.3) demonstrated that the model based on human data was overfitted. The result of the F test (MRT\MRT(pred)) was 17 times higher in case of data obtained for the dog than for the data obtained for human.
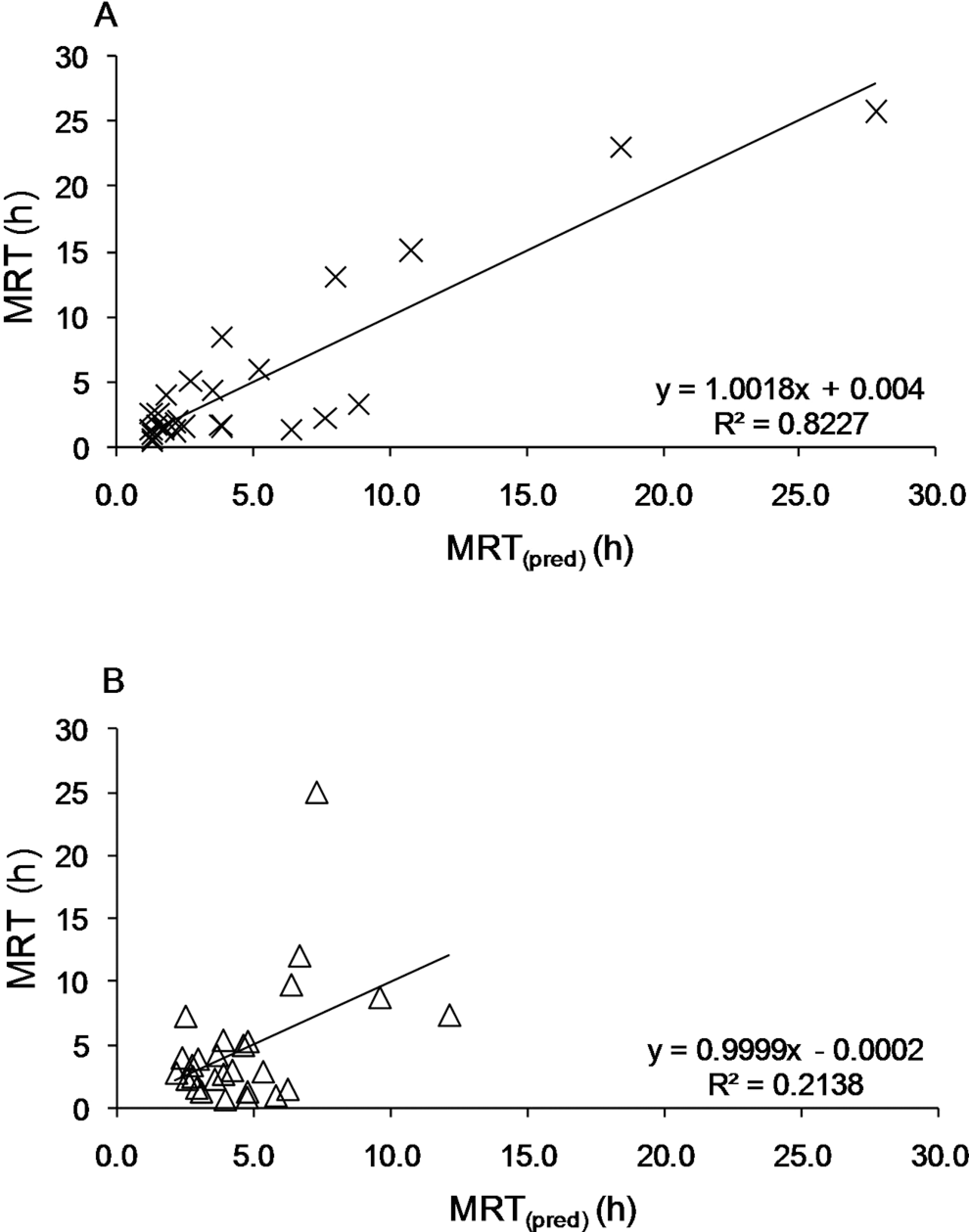
Linear correlation between values of mean residence time (MRT) and values of mean residence time predicted (MRT(pred)) in the tested group of 27 drugs administered intravenously both in dog (A) and human (B).
Discussion
The analysis of a relationship between the molecular structure of the drug (in relation to single physicochemical parameters) and MRT value did not render any significant correlation neither in humans nor in the dog. However, such correlation has been observed for the arithmetic expression created based on several physicochemical characteristics for the group of drugs used in the dog. The existence of a correlation between a mathematical model derived from multiple measured parameters (illustrating a complicated chemical composition of a molecule) and not for single physicochemical parameters of drugs reflects in a way a complicated nature of processes described by the MRT. On the other hand, it must be pointed out that for the group of drugs analyzed, no significant correlation between MRT values for human and dog was detected. The lack of such correlation is consistent with allometrical scaling analysis performed by Ward and Smith. 51,52 in which the authors showed that the dog is not a good general model for human MRT analysis. The above findings indicate that pharmacokinetic processes, to which drugs are submitted within the body, vary significantly between the species. Studies which are conducted to analyze the relationship between physicochemical parameters of drugs and pharmacokinetic parameters determined in vivo are usually performed on a limited amount of data. This is in particular the case of studies, in which a search for a relationship between the chemical composition of a drug and parameters related to drug elimination such as t1/2kel and clearance is conducted. There are some studies in which a correlation has been investigated for renal and nonrenal clearance versus octanol/water partition coefficient. These were performed on a small group of drugs with similar chemical composition and pharmacodynamic properties. Calculations conducted by Hinderling et al 14 were based on 12 homologs and the coefficient of correlation for the polynomial equation was below 0.9. In other studies, the analysis has been conducted for 6 different homologs and the correlation of octanol/water partition coefficient and renal clearance was determined to be 0.96. 15 However, it should be taken into account that it is very difficult at best extrapolate an analysis of this type to bigger groups of drugs with differentiated chemical structure and pharmacokinetic as well as pharmacodynamic properties.
A trend to use complex equations and mathematical models, as in the present study, has been established in scientific research and used at earlier stages for other pharmacokinetic parameters, for example, volume of distribution. 11 A correlation with an mathematical model based on several physicochemical parameters has also been demonstrated for the NOEL (no observed effect level) parameter value for the group of 135 drugs. 53 – 55
In conclusion, it can be stated that the present study constitutes an attempt to find a correlation between the value of MRT and MRT(pred), a value derived from an optimal set of multiple physicochemical parameters determined in silico and combined in one arithmetic expression. Although such correlation was demonstrated and validated for the dog, analogical calculations performed for the data obtained in human could not be successfully predicted. The results of the present in silico analysis demonstrate that the extrapolation of pharmacokinetic data from the dog to human, particularly in relation to MRT, has significant limitations.
Footnotes
Acknowledgements
The authors thank Professor Witold GumuŁka for the critical reading of the manuscript.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
