Abstract
Cyclomethicone (mixture) and the specific chain length cyclic siloxanes (n = 4-7) reviewed in this safety assessment are cyclic dimethyl polysiloxane compounds. These ingredients have the skin/hair conditioning agent function in common. Minimal percutaneous absorption was associated with these ingredients and the available data do not suggest skin irritation or sensitization potential. Also, it is not likely that dermal exposure to these ingredients from cosmetics would cause significant systemic exposure. The Cosmetic Ingredient Review Expert Panel concluded that these ingredients are safe in the present practices of use and concentration.
Keywords
Introduction
A safety assessment of cyclomethicone was published in 1991. 1 On the basis of the available data, the Cosmetic Ingredient Review (CIR) Expert Panel concluded that cyclomethicone was safe as a cosmetic ingredient in the (then) present practices of use. In that assessment, cyclomethicone was described as a mixture of cyclic dimethyl polysiloxane compounds. Because new studies are available which address the safety of the individual cyclomethicones from chain length 3 to 7, the CIR Expert Panel reopened consideration of cyclomethicone to consider these data and to include each of these individual chain length ingredients now identified as cosmetic ingredients. Accordingly, the following cyclic dimethyl polysiloxane compounds (cyclomethicones) are reviewed in this safety assessment: cyclomethicone, cyclotetrasiloxane (D4), cyclopentasiloxane (D5), cyclohexasiloxane (D6), and cycloheptasiloxane (D7). These ingredients function as anticaking agents, hair conditioning agents, skin conditioning agents—emollients, and solvents in cosmetic products. The definition of cyclomethicone has recently been revised to state that it is a mixture of individual chain length cyclic dimethyl polysiloxane compounds from D4 to D6. While there is no indication that cyclotrisiloxane (D3) is being used as a cosmetic ingredient in products marketed in the United States at this time, it is known to be an impurity of D4 (and possibly D556-D7) and has being detected in cosmetic products. D3 had been defined as an ingredient in the International Cosmetic Ingredient Dictionary and Handbook 2 ; however, the International Nomenclature Committee (INC) of the Personal Care Products Council recently approved the deletion of D3 as an entry in the future issues of the International Cosmetic Ingredient Dictionary and Handbook.
The ingredients reviewed in this safety assessment account for a minor portion of the composition of silicone gel-filled breast implants for humans, which consist predominantly of higher molecular weight cyclic dimethyl polysiloxane compounds. These higher molecular weight compounds are not cosmetic ingredients and present different exposure-related issues, when compared with their lower molecular weight counterparts in cosmetic products. The US Food and Drug Administration (FDA) has approved the safety of 1 silicone gel-filled breast implant with a small percentage of cyclomethicones of the sizes considered in this assessment.
Chemistry
Definition and Structure
Cyclomethicones
In the published CIR Final Report 1 , cyclomethicone (CAS No. 69430-24-6) was defined as a mixture of cyclic dimethyl polysiloxane compounds that conform to the formula in Figure 1, where n has an average value that ranges from 3 to 6. Also, according to this final report, the tetramer (n = 4) and pentamer (n = 5) of cyclomethicone are frequently the predominant polymers found in cosmetic formulations. The tetramer polymer is illustrated in Figure 2. 3
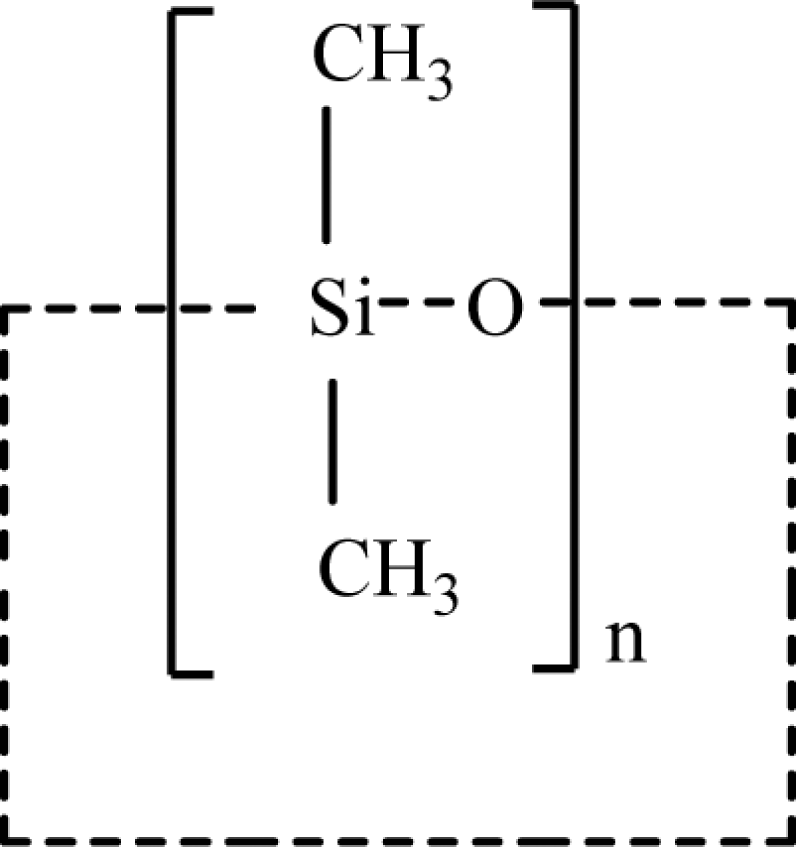
Structure of cyclomethicone. 1
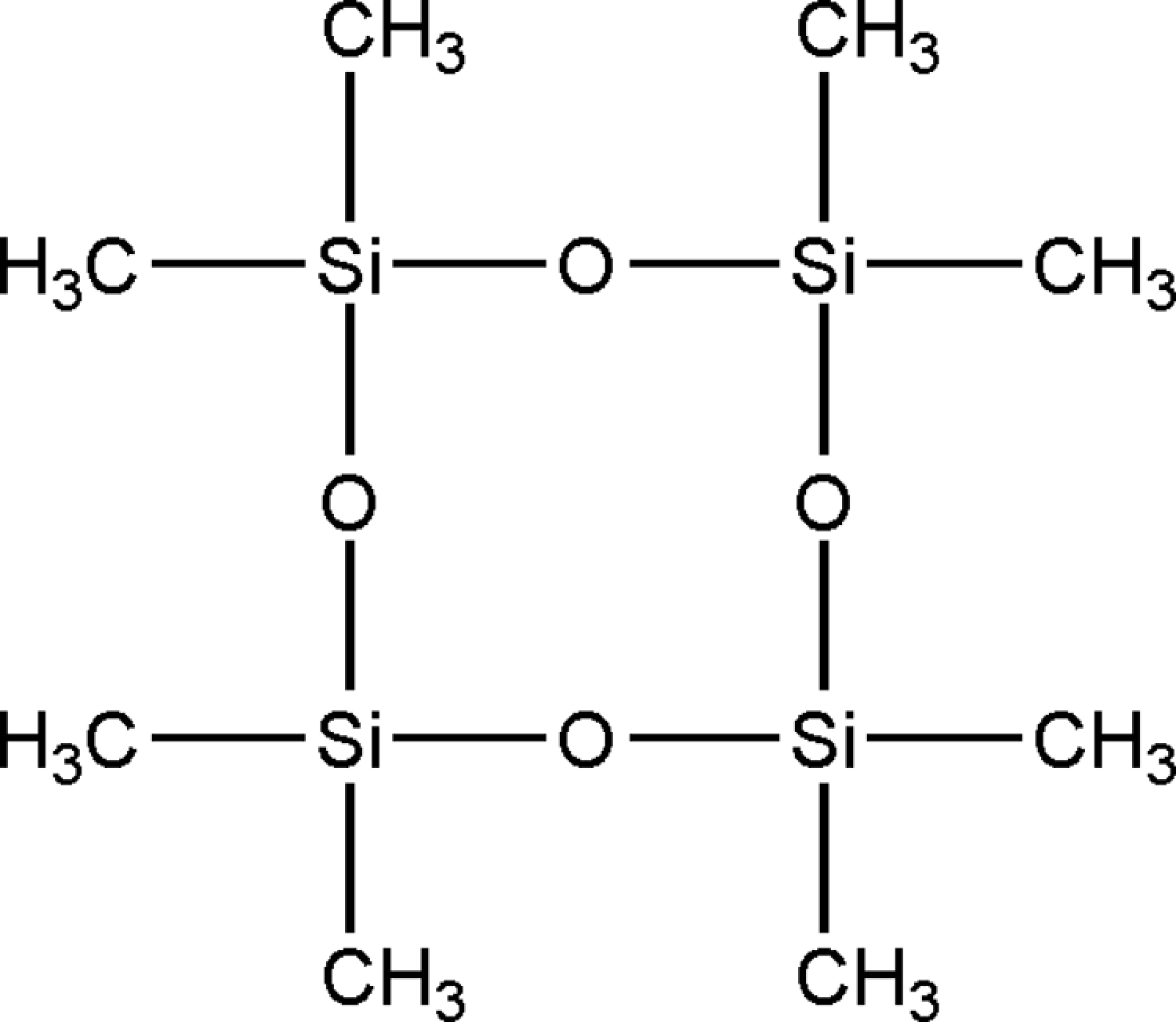
Structure of tetramer (n = 4). 3
The most recent information indicates that the definition of cyclomethicone has been revised. It had been given as a generic name for cyclic dimethyl polysiloxane compounds that conform to the formula included in Figure 1 in this safety assessment, where n has a value between 3 and 7, but now the range of values for n is between 4 and 6, because this more accurately reflects the current composition of cyclomethicone that is used in the personal care industry. 4 Other names for cyclomethicone include cyclic decamethyl cyclopentasiloxane/octamethyl cyclotetrasiloxane resin; cyclosiloxanes, Di-Me; and methylcyclopolysiloxane.
The INCI names for labeling specific cyclic dimethyl polysiloxane compounds are: cyclotetrasiloxane (n = 4), cyclopentasiloxane (n =5), cyclohexasiloxane (n = 6), and cycloheptasiloxane (n = 7). Cyclomethicone was listed as a technical name for each compound 2 , but, given the new definition of cyclomethicone, it is no longer considered a technical name for D3 or D7. Accordingly, cyclomethicone is not included among the technical names for cyclotrisiloxane and cycloheptasiloxane in chemical definitions under the following subheadings. In common usage and as given in the studies provided by the Silicones Environmental, Health and Safety Council (SEHSC), these individual chain length cyclic dimethyl polysiloxanes are also known as D3, D4, D5, D6, and D7, respectively, and are identified as such in the report text.
The following chemical definitions refer to the structure in Figure 1.
Cyclotrisiloxane
Cyclotrisiloxane (CAS No. 541-05-9) is the cyclic dimethyl polysiloxane that conforms to the generic structure of cyclic dimethyl polysiloxane compounds, where n = 3, and the other components of cyclomethicone (where n = 4, 5, 6, or 7) are present at the levels of less than 1%. Other names for cyclotrisiloxane include cyclotrisiloxane, hexamethyl- and hexamethylcyclotrisiloxane. 2 The most recent information indicates that the monograph for cyclotrisiloxane in the International Cosmetic Ingredient Dictionary and Handbook has been withdrawn by the INC of the Personal Care Products Council, 5 presumably because it is not being used as a cosmetic ingredient and the detection of D3 in cosmetic products is due to its presence as an impurity. In keeping with this decision, this committee also removed any listing of D3 as a potential component/impurity of D4 to D7 in these respective monographs. Accordingly, D3 is not listed as a potential component/impurity of cyclotetrasiloxane, cyclopentasiloxane, cyclohexasiloxane, or cycloheptasiloxane in chemical definitions under the following subheadings.
Cyclotetrasiloxane
Cyclotetrasiloxane (CAS Nos. 293-51-6 and 556-67-2) is the cyclic dimethyl polysiloxane that conforms to the generic structure of cyclic dimethyl polysiloxane compounds, where n = 4, and the other components of cyclomethicone (where n = 5, 6, or 7) are present at the levels of less than 1%. Other names for cyclotetrasiloxane include cyclomethicone; cyclotetrasiloxane, octamethyl-; and octamethylcyclotetrasiloxane. 2
Cyclopentasiloxane
Cyclopentasiloxane (CAS No. 541-02-6) is the cyclic dimethyl polysiloxane that conforms to the generic structure of cyclic dimethyl polysiloxane compounds, where n = 5, and the other components of cyclomethicone (where n = 4, 6, or 7) are present at the levels of less than 1%. Other names for cyclopentasiloxane include cyclomethicone; cyclopentasiloxane, decamethyl-; and decamethylcyclopentasiloxane. 2
Cyclohexasiloxane
Cyclohexasiloxane (CAS No. 540-97-6) is the cyclic dimethyl polysiloxane that conforms to the generic structure of cyclic dimethyl polysiloxane compounds, where n = 6, and the other components of cyclomethicone (where n = 4, 5, or 7) are present at the levels of less than 1%. Other names for cyclohexasiloxane include cyclomethicone; cyclohexasiloxane, dodecamethyl-; and dodecamethylcyclohexasiloxane. 2
Cycloheptasiloxane
Cycloheptasiloxane (no CAS No.) is the cyclic dimethyl polysiloxane that conforms to the generic structure of cyclic dimethyl polysiloxane compounds, where n = 7, and the other components of cyclomethicone (where n = 4, 5, or 6) are present at the levels of less than 1%. Other names for this chemical include cycloheptasiloxane, tetradecamethyl- and tetradecamethylcyclo-heptasiloxane. 2
Chemical and Physical Properties
Cyclomethicone
According to Todd and Byers, 6 cyclomethicone is a colorless, odorless, transparent, nongreasy, silicon fluid. It has a low viscosity and surface tension and a relatively high vapor pressure, which allows the majority of the silicone portion to evaporate from the surface to which it is applied. Variations in the volatility of cyclomethicone can be achieved through the blending of its different polymers. Cyclomethicone is soluble in ethanol (99%), isopropanol (99%), mineral oil, paraffin wax, stearyl alcohol, stearic acid, and aliphatic, chlorinated, and fluorinated solvents. It is highly insoluble in water, but hydrolytically stable as to be easily emulsified into most cosmetic preparations.
Since the chemical name cyclomethicone encompasses a number of different polymers, slight variations will exist in calculations of molecular weight, solubility, specific gravity, viscosity, and so on. Table 1 includes the physicochemical properties of 4 cyclomethicones (trade name mixtures). 3,7 Data on D4, D5, and D6 appear in Table 2.
Properties of Cyclomethicone Dow Corning Fluidsa (SEHSC, 2009) 3
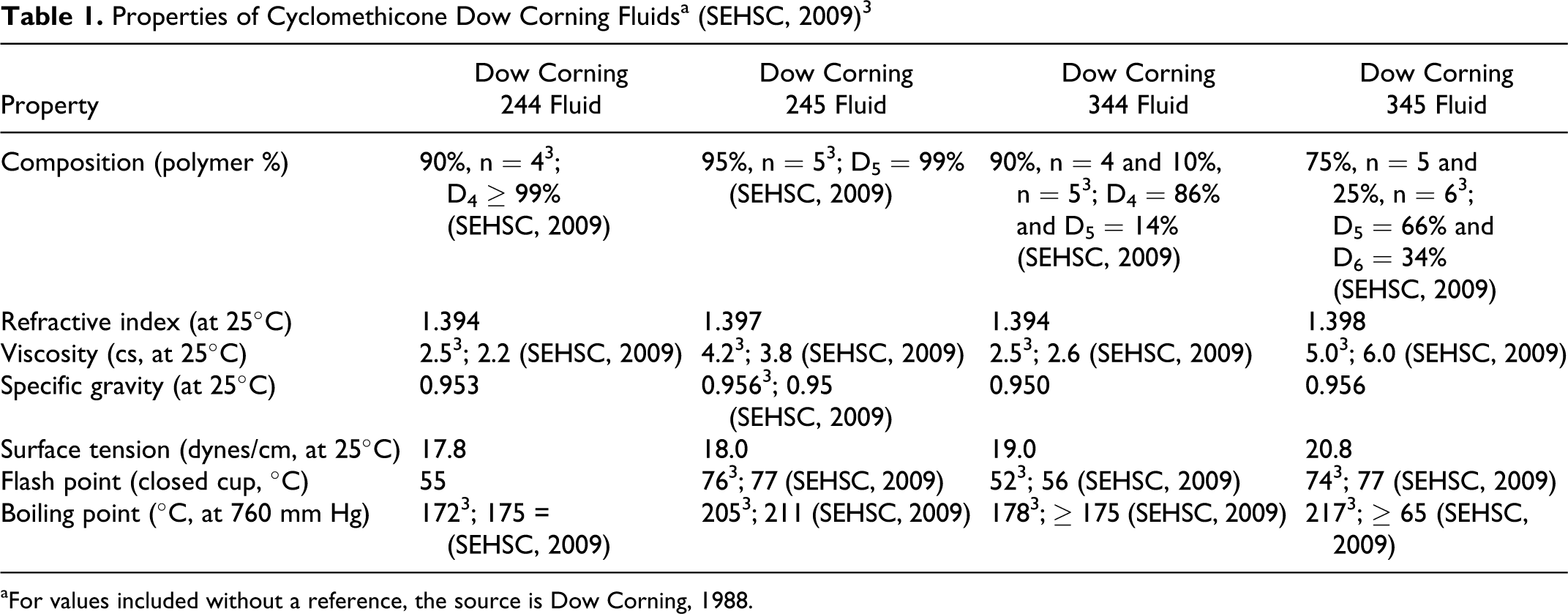
aFor values included without a reference, the source is Dow Corning, 1988.
Properties of Cyclomethicones

Analytical Methods
Cyclomethicone
A commonly used analytical method for the identification of cyclomethicone is the gas chromatography (GC). 8,9
Cyclotrisiloxane (D3),Cyclotetrasiloxane (D4), Cyclopentasiloxane (D5), and Cyclohexasiloxane (D6)
Ferninandi and Beattie 10 validated a high resolution GC method for the measurement of D4 in a nose-only inhalation chamber. A linear response over the range from 76.27 to 829.02 ppm was reported with a correlation coefficient of .9988.
In a study by Varaprath and Lehmann, 11 D4, D5, and D6 were analyzed using GC/mass spectrometry (MS). Samples of these siloxanes were obtained purely by distillation of the commercial Dow Corning polydimethylsiloxane (PDMS) fluids.
Varaprath et al 12 described a method for extracting D4 from tissues (using glass beads), so that the levels could be measured, as given in the Absorption, Distribution, Metabolism, and Excretion section (under In Vivo/In Vitro study subheading) later in this report. In in vitro experiments, heparinized blood samples freshly collected from rats were spiked with 14C-D4 (specific activity = 1.33 μCi/mL) at 3 different concentrations (21, 210, and 2100 ppm) and then subjected to extraction with tetrahydrofuran. The extraction efficiencies were determined to be ∼90% at the concentrations of 14C-D4 that were tested (21, 210, and 2100 ppm). The extraction efficiency was low (∼40%) when D4 was present in the blood at nanogram per milliliter levels (50-500 ppb D4).
In these instances, performing the extractions in the presence of glass beads greatly improved the extraction efficiency. In a single extraction, at concentrations as low as 60 ppb D4, the extraction efficiency was >80%. The glass beads, coated with blood, provided greater surface area for solvent interaction and greater agitation with the solvent, resulting in improved efficiency. Therefore, it was determined to be advantageous to use glass beads for extraction when the materials were present at low levels. Results indicated that the recoveries of D4 and its unbound metabolites from the plasma were essentially quantitative in 3 extractions. The recoveries were >90%, even in a single extraction. Extraction efficiencies were adipose follows: lung (98.2% ± 0.3%), liver (95.4% ± 0.4%), adipose tissue (99.4% ± 0.8%), urine (98.1% ± 0.2%), and feces (94.1% ± 0.6%). 12 In more recent studies, D4 was also analyzed using GC/MS 13 and the same was true for D3, D4, D5, and D6. 14
Impurities
Cyclotetrasiloxane (D4)
According to the Scientific Committee on Consumer Products (SCCP) 15 , the purity of D4 used in tests is described as unknown or >95% (maximum of 99.8%). Decamethylcyclopentasiloxane (D5; 5% maximum) and hexamethylcyclotrisiloxane (D3; 1% maximum) are listed as impurities/accompanying contaminants.
Reactivity
Cyclomethicone
According to Todd and Byers, 6 cyclomethicones are nondegradable, inert polymers. Under normal cosmetic conditions and in formulations, they are nonreactive. Cyclomethicone is compatible with cosmetic silicones as well as the following other cosmetic ingredients: beeswax, glycerine, isopropyl myristate, isopropyl palmitate, lanolin, mineral oil, paraffin, and stearic acid. Information from Dow Corning 3 states that the low viscosity of cyclomethicone, like other volatile carrier fluids, allows the silicone portion to evaporate without cooling the skin. By blending cyclomethicones (mixtures of the n value in the structural formula), the volatility of the compound can be adjusted to correspond to the amount of time that the silicone portion should remain in contact with the skin. At 22°C, the tetramer component (n = 4) evaporates nearly twice as slowly as water. The pentamer (n = 5) evaporates more slowly than the tetramer.
Cyclotrisiloxane (D3), Cyclotetrasiloxane (D4), and Cyclopentasiloxane (D5)
Almond et al 16 used matrix isolation infrared spectroscopy to study the vacuum pyrolysis of D3, D4, and D5. The results were interpreted in the context of various kinetic models. It was shown that the significant pyrolysis products (CH3,CH4,C2H2,C2H4,C2H6, and SiO) may be accounted for radical reactions involving dimethylsiloxane (D1). The authors noted that the evidence that D1 is formed from D4 is strong. Furthermore, the results of this study indicate that D3 is formed from both D4 and D5 and that the small molecules (CH3,CH4,C2H2,C2H4,C2H6, and SiO) are formed from D3, D4, and D5. Thus, it seems likely that the decomposition mechanisms of all 3 oligomers are similar.
The working hypothesis is that each of the oligomers decomposes by elimination of D1 and that the larger ones depolymerize. Therefore, it seems very likely that all of the smaller products result from the decomposition of D1, which is not stable at high temperatures. It was concluded that the experiments in this study have established that the 3 cyclic siloxanes (D3, D4, and D5) produce essentially the same hydrocarbon decomposition products when undergoing vacuum pyrolysis, indicating that a common precursor is involved in the process. 16
Finocchio et al 17 studied the decomposition of D3 (at room temperature and in the 473-673 K range) over the surface of basic (CaO and MgO) and acidic oxides (Al2O3 and SiO2). All results are based on D3 data. Results indicate that alumina can be used as an adsorbent for the hot cleaning of biogas from siloxanes. At 673 K, alumina reacts, producing the hydrolysis of the Si–C bond. Due to this reaction, the alumina surface is silicized and methane is released. Silica, which is an excellent adsorbent for siloxanes at room temperature, shows an adsorption capacity of 0.76 g of adsorbed siloxane per gram of silica and loses its adsorption ability at high temperatures. Basic oxides such as MgO and CaO have strong reactivity in decomposing siloxanes in the absence of CO2, but lose reactivity when in contact with carbon dioxide because of surface carbonation.
Use
Purpose in Cosmetics
According to the International Cosmetic Ingredient Dictionary and Handbook 2 , the following ingredients function as hair conditioning agents, skin conditioning agents—emollient, and solvents in cosmetics: cyclomethicone, cyclotetrasiloxane, cyclopentasiloxane, and cyclohexasiloxane. Cycloheptasiloxane functions as an anticaking agent, skin conditioning agent—emollient, and solvent, but not as a hair conditioning agent.
Scope and Extent of Use in Cosmetics
According to Klykken et al, 18 the second largest use of cyclotetrasiloxane is in personal care products, such as, antiperspirants, deodorants, skin creams, and shampoos. In these applications, it is commonly blended with cyclopentasiloxane and is referred to as cyclomethicone. Zareba et al 19 have stated that some commercially available roll-on antiperspirants have contained up to 60% cyclotetrasiloxane as a vehicle.
Data submitted to the FDA in 1984 by cosmetic firms participating in the Voluntary Cosmetic Registration Program (VCRP) indicate that cyclomethicone was being used in a total of 168 products at a concentrations ranging from #0.1% to >50%. These data were reported in the published CIR Final Report on cyclomethicone 1 and are also included as historical data in the current safety assessment in Table 3. Current VCRP data, 20 also given in Table 3 as a function of product category, indicate that cyclomethicone is being used in a total of 1499 products. For example, cyclomethicone is reportedly used in 29 of a total of 463 mascara products reported in the VCRP, suggesting that less than 10% of mascara products on the market contain cyclomethicone.
Historical and Current Cosmetic Product Uses and Concentrations for Cyclomethicones
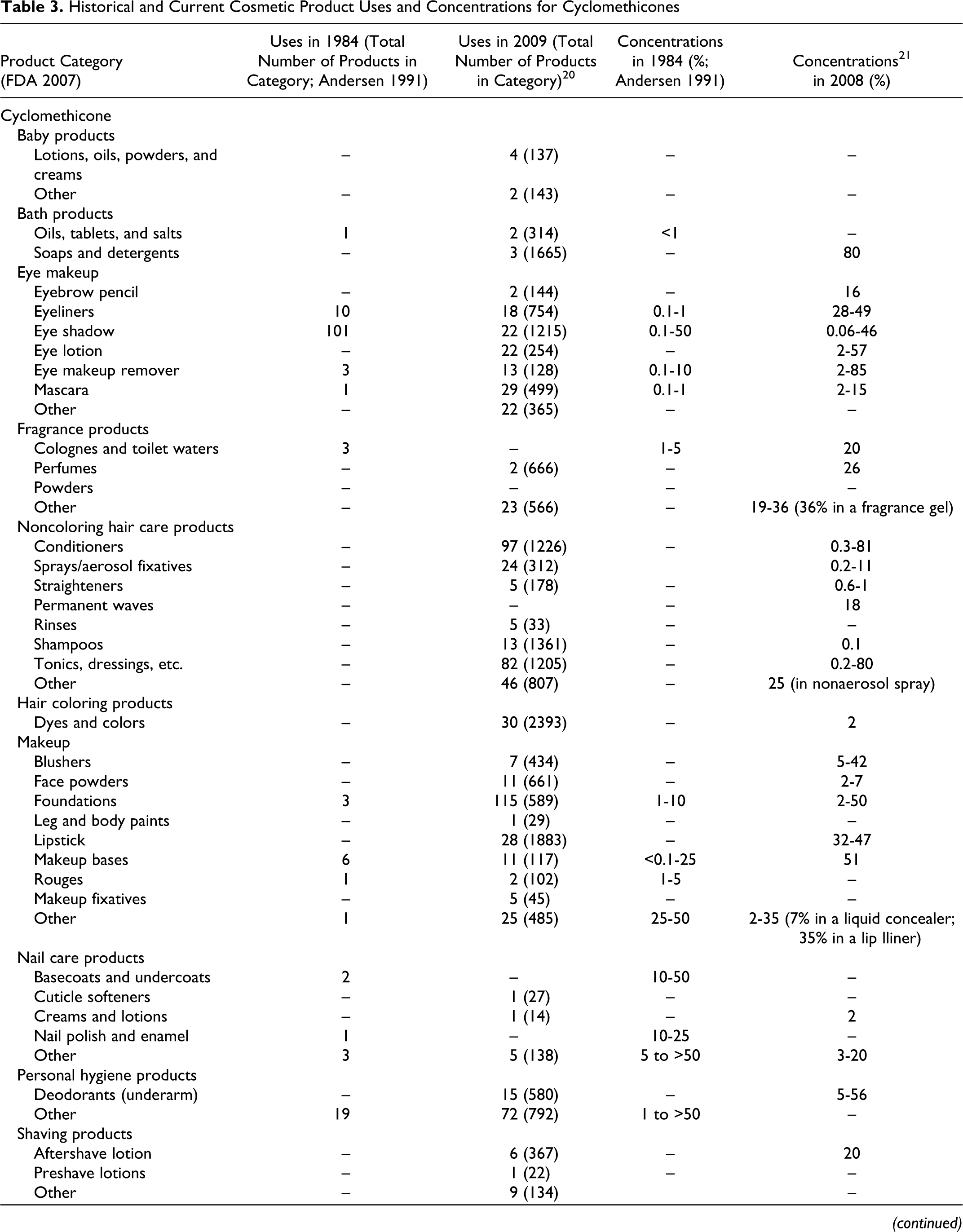
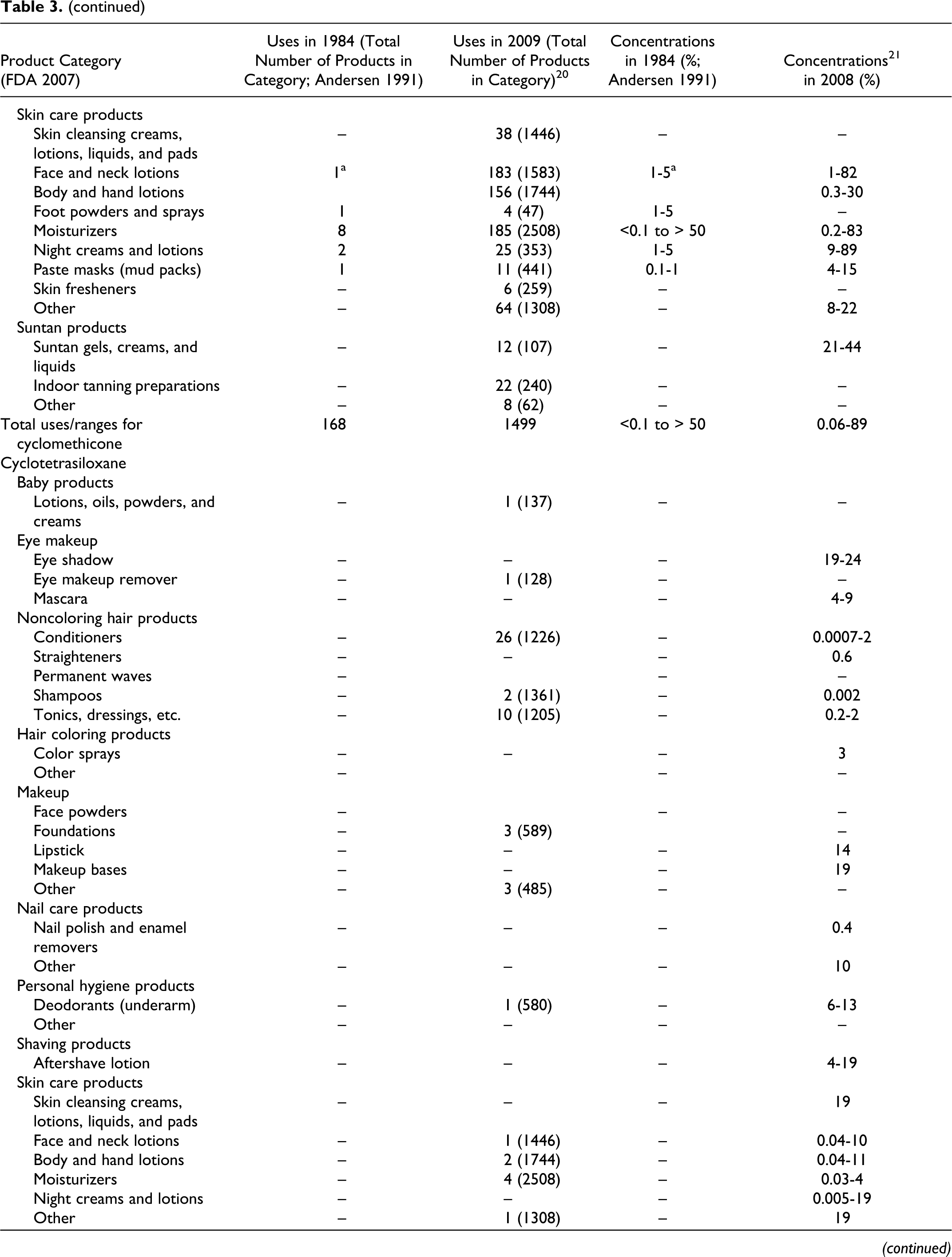
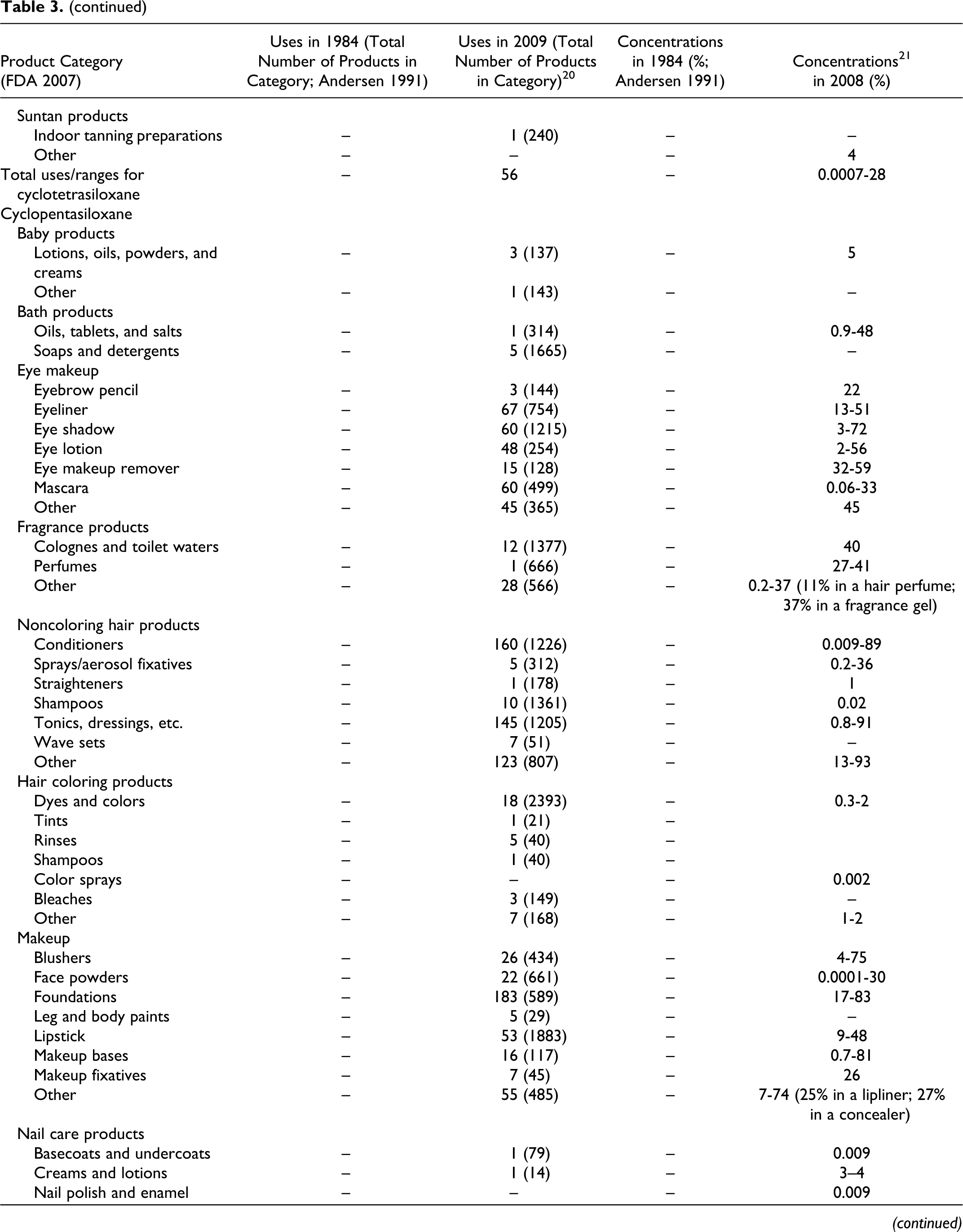
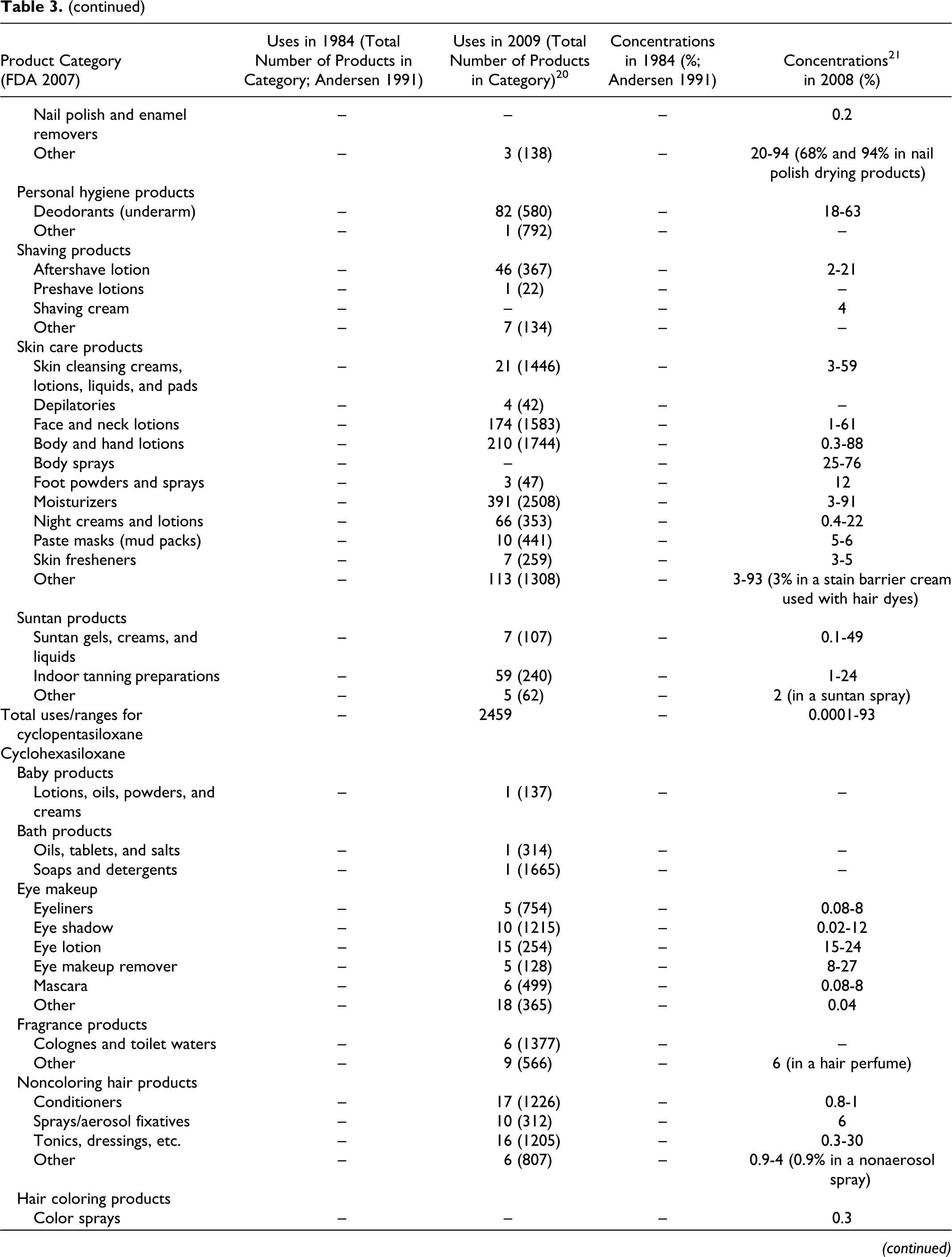
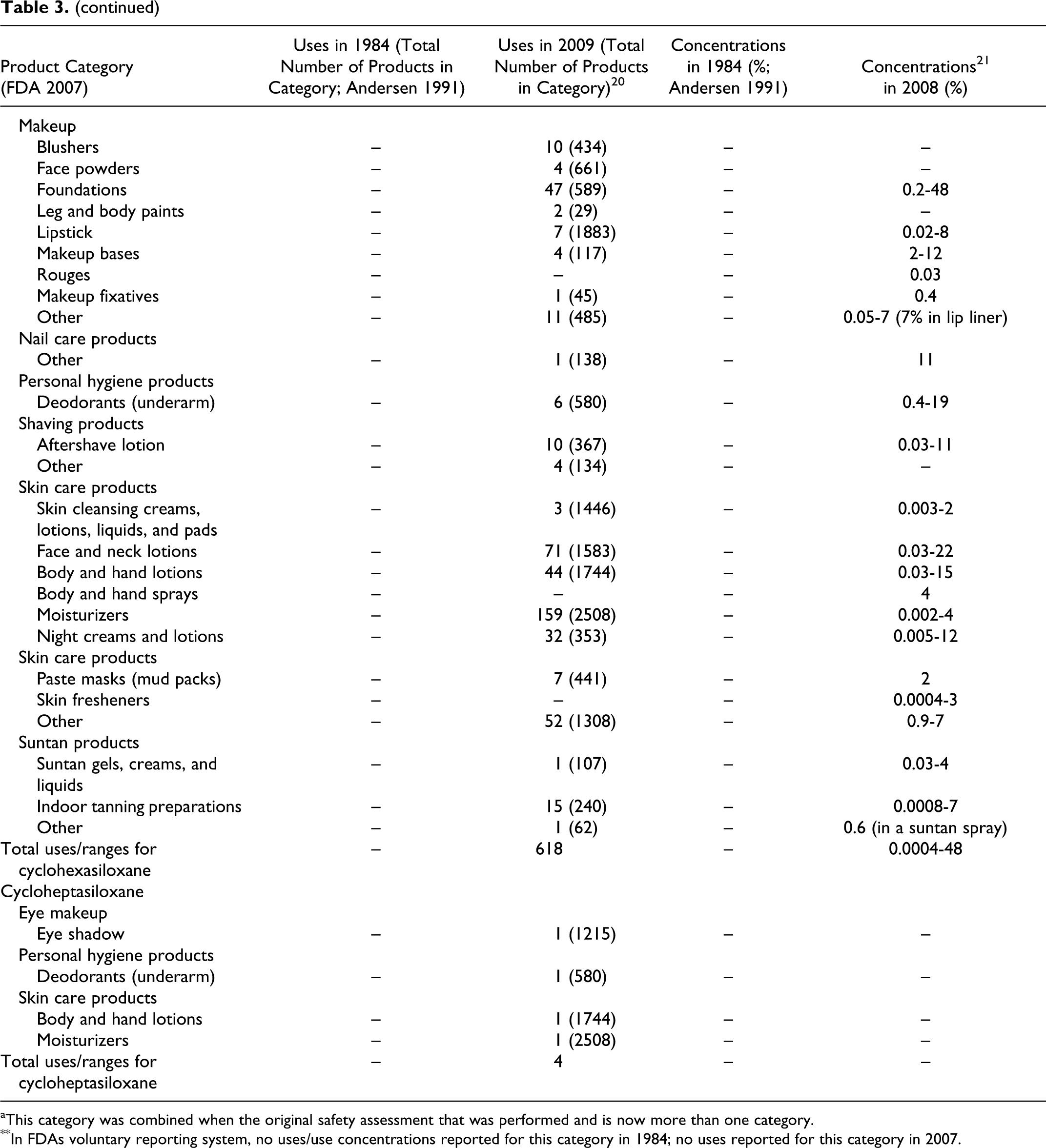
aThis category was combined when the original safety assessment that was performed and is now more than one category.
**In FDAs voluntary reporting system, no uses/use concentrations reported for this category in 1984; no uses reported for this category in 2007.
As shown in Table 3, the following other cyclic dimethyl polysiloxane compounds also are being used: cyclotetrasiloxane (56 products), cyclopentasiloxane (2459 products), cyclohexasiloxane (618 products), and cycloheptasiloxane (4 products). The 2009 VCRP data also included 6 uses of cyclotrisiloxane in cosmetics, but these uses were not substantiated by FDA and have been deleted from Table 3.
Of the many product categories reported, all of the ingredients are being used in personal hygiene products and that, except for Cycloheptasiloxane, all are being used in baby products. The results of a survey of current ingredient concentrations used conducted by the Personal Care Products Council in 2008 21 are as follows: cyclomethicone, 0.06% to 89%; cyclotetrasiloxane, 0.0007% to 28%; cyclopentasiloxane, 0.0001% to 93%; and cyclohexasiloxane, 0.0004% to 48%. In the example of the use of cyclomethicone in mascara products given earlier, the concentration used ranged from 2% to 15%.
Data of concentrations used for cyclotrisiloxane in cosmetic products were reported in this survey, 21 but these data were subsequently withdrawn based on the assertion that cyclotrisiloxane is not specifically added to cosmetic products.
Earlier data from Dow Corning, 22 indicated that, depending on the product type, the concentration of cyclotetrasiloxane in formulations has varied between 0.1% and 54%.
Using GC/MS, Horii and Kannan 23 determined concentrations of the following siloxanes (cyclic and linear) in a variety of consumer products, including personal care products: octamethylcyclotetrasiloxane (D4), decamethylcyclopentasiloxane (D5), dodecamethylcyclohexasiloxane (D6), tetradecamethylcycloheptasiloxane (D7), and linear siloxanes (L4-L14). Both personal care and household products were purchased from retail stores in Albany, New York, and in Tsukuba, Japan, during 2006. The data for personal care products and a few household products are summarized in Table 4. Data on household products are included in Table 4 because one of the product categories (identified as other) consisted of personal care and household products. According to Wang et al, 14 the results of a survey of 252 cosmetic and personal care products sold in Canada indicated the presence of D3 in 0.8% of the products. The 2 product types that were listed as containing D3 were a fragrance (D3 concentration = 0.12 mg/g wet weight) and a diaper cream (D3 concentration = 0.45 mg/g wet weight). D4, D5, and D6 were also detected in these products. Product samples were extracted with different organic solvents, depending on the nature of the products, and then subjected to GC–MS.
Concentrations (μg/g, Mean/Range) of Organosiloxanes in Personal care and Household Products 23
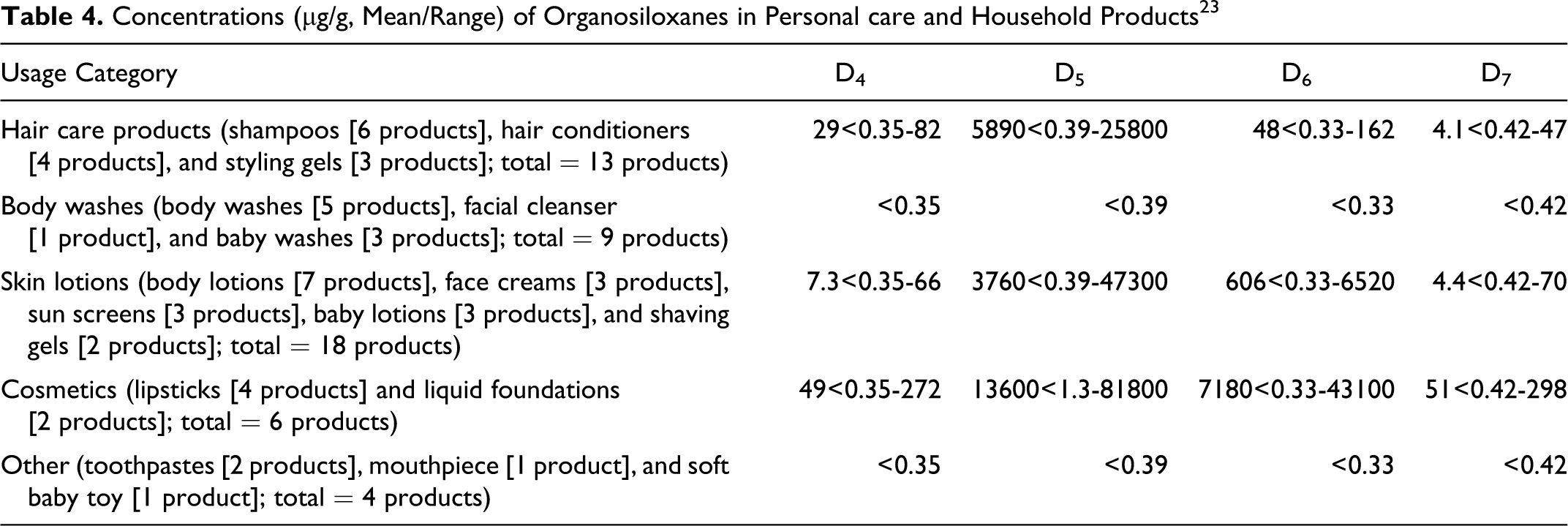
Cosmetic products containing cyclic dimethyl polysiloxane compounds are applied to most areas of the body and could come in contact with the oral, ocular, or nasal mucosa. These products may be used on a daily basis and could be applied frequently over a period of several years.
None of the ingredients included in this safety assessment is included on the list of ingredients that cosmetic products marketed in Japan must not contain or cosmetic ingredient lists with restrictions. 24 The same is true relative to the absence of these cosmetic ingredients from similar lists of ingredients for cosmetic products marketed in the European Union. 25
The SCCP 15 did issue the following opinion on D4:
On the basis of provided data, the SCCP is unable to assess the risk to consumers when octamethylcyclotetrasiloxane (D4) is used in cosmetic products.
Despite the size of the dossier submitted by the industry for evaluation, it is unfortunate that the dossier lacked meaningful information/data on actual consumer exposure to D4.
The following information is required before any further consideration:
adequate information on the use of D4 in cosmetics in particular in different cosmetic products,
relevant/appropriate percutaneous absorption studies at different concentrations used, and
information on the co-use, and hence consumer exposure, of related organosiloxanes, particularly decamethylcyclopentasiloxane (D5).
Noncosmetic Use
Cyclomethicone
According to Dow Corning, 3 noncosmetic applications of cyclomethicone include glass and specialty cleaners, lubricants, and penetrating oils. In other publications, 6,26,27 nonvolatile silicones are reported as possible barriers in the prevention or recovery of skin damage. Frant 26 reported that some siloxane oils are used widely for their lubricating properties and water repellency on walls, textiles, leather, in molds, and so on. Also, inhaled silicone oil vapor has acted as an antifoaming agent in the treatment of chronic bronchitis. According to Gabel et al, 28 PDMS, a specific silicone oil, also has been used in intraocular applications for the treatment of complicated retinal detachment.
According to Dow Corning 22 , cyclic siloxanes, including cyclomethicone, are used as precursors in the production of PDMSs, which are widely used in various industrial and consumer applications, topical pharmaceutical formulations, and as breast implants. Additionally, certain food products are processed using silicone antifoam containing cyclotetrasiloxane.
Cyclotrisiloxane (D3), Cyclotetrasiloxane (D4), Cyclopentasiloxane (D5), Cyclohexasiloxane (D6), and Cycloheptasiloxane (D7)
According to Varaprath et al, 12 approximately 80% of all the D4 that is produced is used as an intermediate in making PDMS polymers for applications such as building sealants, rubber products, and fabric coatings. The remaining 20% of the D4 that is produced is used in personal products. D4 is also used as a building block in the industrial synthesis of long chain silicone polymers. 18
The gel of silicone gel-filled breast implants consists of 1% to 2% low molecular weight silicones with structures identified mainly as cyclic compounds, such as D3, D4, D5, D6, and D7. 13 The results of a GC/MS analysis of breast implant distillate were as follows: D3, 18%; D4, 60%; D5, 20%; and D6, 2%. Low molecular weight linear siloxanes (<1%) and platinum (40 mg/kg distillate) were also detected. 29
The US FDA has approved the safety of one silicone gel-filled breast implant with a small percentage of cyclomethicones of the sizes considered in this assessment (D4-D7). 20 D5 is used in the dry cleaning process. 30
General Biology
Percutaneous Absorption
In Vivo Studies—Animal
Cyclomethicone
Data 31 obtained in skin irritation studies (species not stated) indicated that cyclomethicone (average n = 4) was not absorbed through the skin in toxic amounts.
Cyclotetrasiloxane (D4) and Cyclopentasiloxane (D5)
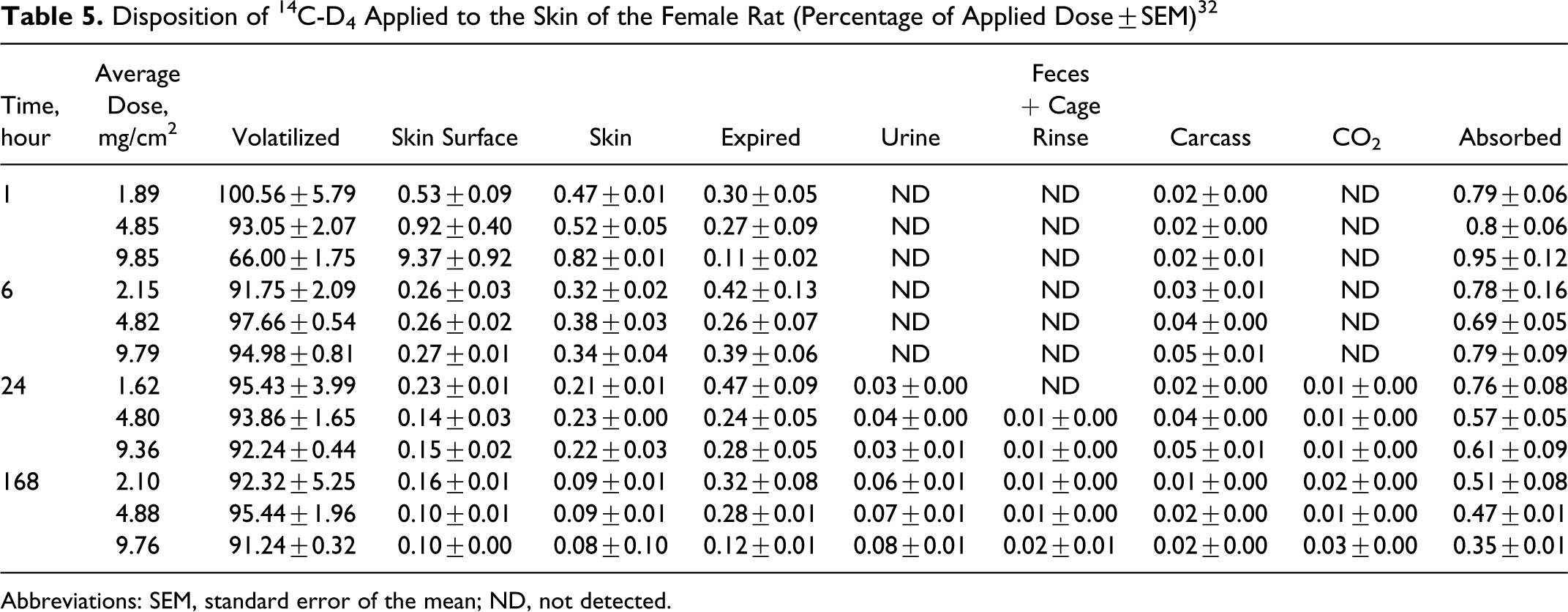
Jovanovic 32 conducted an in vivo percutaneous absorption study of D4 using female Fisher 344 rats (10-11 weeks old). [14C]D4 was applied at the doses of 2, 4, 8, and 10 mg/cm2 skin. Each of these levels was a separate experiment with 2 control animals. At 1, 6, or 24 hours after continuous exposure to radiolabeled D4 in metabolism cages, 4 animals per each time point were killed and blood levels of 14C-D4 were determined. There were 4 rats in each group and 2 animals served as controls for each dose level. Rats that had signs of abrasion or problems with attachment of the skin depot were replaced by a reserve animal. A skin depot apparatus was glued to shaved skin. After application of D4, a charcoal basket was inserted immediately into the dosing chamber and secured with an open aluminum cap. A hole previously made in the plastic cap of the charcoal basket allowed air to circulate, providing semiocclusive conditions. Following dosing, the animals were placed in metabolism cages that allowed collection of expired air, urine, feces, and CO2. At the appropriate time, the charcoal basket was removed, desorbed in toluene, and the extract was counted for radioactivity content. The surface area was cleaned and wiped with cotton swabs. Animals were killed and blood samples were obtained for analysis. Exposure sites were tape stripped and counted. The skin was then excised, digested with tetraethyl-ammonium hydroxide (TEAH), and counted. Animal carcasses were digested with TEAH and counted. After 24 hours of exposure, 1 group of animals at each exposure level was removed from the cages, dose sites were washed, charcoal baskets were replaced, and the animals were returned to metabolism cages for another 6 days; radioactivity in excreta was measured daily.
Six animals were exposed to 10 mg/cm2 D4 in normal cages and 2 control animals were not dosed. Blood (200-300 μL) was drawn at 0.5, 2, 4, and 10 hours. These animals were killed at 10th hour. The levels of radioactivity in all of the blood samples were below the level of quantification and were not tabulated.
The results of the metabolism studies are given in Table 5. The author concluded that less than 1% of applied radiolabel was absorbed, independent of the amount applied, and that the amount absorbed decreased overtime (percentage absorbed at 24 hours was statistically significantly less than at 1 hour), again independent of the amount applied. The authors suggested that the amount of D4 that remained in the skin after 24 hours may actually have migrated to the skin surface and evaporated over the ensuing 6-day-period over which the animals were monitored. 32
Disposition of 14C-D4 Applied to the Skin of the Female Rat (Percentage of Applied Dose±SEM) 32
Abbreviations: SEM, standard error of the mean; ND, not detected.
Zareba et al 19 studied the percutaneous absorption of D4 using the human skin/nude mouse model. Female BALB/C nude mice (weights = 25-30 g) were used, and human fetal forearm skin was obtained from aborted fetuses (estimated gestational range = 16-22 weeks). Human tissue grafts (10-20 mm in diameter) were transplanted subcutaneously (SC) on to mice following sodium pentobarbital anesthesia. The mice were maintained for 2-4 months to allow graft healing and growth to approximately 25 mm in diameter. Aluminum skin depot chambers were attached to the human skin grafts at 15 minutes prior to application of D4. The first experiment (4 mice) was performed using nonlabeled D4. At 15 minutes after chamber attachment, 15.7 mg of neat unlabeled D4 per cm2 (volume = 10 μL) were applied using a 50-μL syringe. After dose administration, a charcoal basket was placed in the dosing chamber above the application site. The animals were then placed in metabolism cages, and the experiment was terminated 24 hours after D4 application. The animals were killed by cervical dislocation while under pentobarbital anesthesia. The skin depot chamber was removed and the human skin graft, charcoal baskets, and expired volatile traps were collected. D4 distribution within whole skin layers was evaluated. D4 concentrations in the epidermis, dermis, and adipose tissue at the application site were expressed as nanograms of total D4 and as the percentage of the total measured in tissue at the application site.
To study the percutaneous absorption of D4, a second experiment was performed using 14C-D4. The dose that was applied to the skin was the same as that described for nonlabeled D4. After dosing, the mice were placed in metabolism cages equipped with charcoal tubes as expired volatile traps. After 24 hours, application sites were tape stripped to remove any remaining D4 that was present in the stratum corneum, and the animals were returned to their metabolism cages for an additional 48 hours. After 72 hours, the mice (under pentobarbital anesthesia) were killed by cervical dislocation, and the human skin graft, mouse skin, carcass, and cage wash were collected.
The purpose of the first experiment (nonlabeled D4, 4 mice) was to determine whether D4 undergoes accumulation in SC adipose tissue. After 24 hours, the total concentrations of D4 in the human epidermis, dermis, and adipose tissue at the application site were 470, 220, and 75 ng of D4, respectively. The mean distribution of the total D4 that was recovered in the skin was 61% in the epidermis, 29% in the dermis, and 10% in the adipose tissue.
The results of the in vivo percutaneous absorption study (7 mice) indicated that, after 24 hours of exposure to 14C-D4, a mean of 1.09% ± 0.46% of the applied dose was absorbed through human skin under semiocclusive conditions. Approximately 0.02% of the applied dose remained in the skin at 24 hours postapplication. Most of the applied dose (94.59% ± 12.28%) had evaporated from the application site. The volatile trap that captured D4 accounted for 42% of the radioactivity that was absorbed, while 49% was excreted in the urine and feces. 19
Plotzke and McMahon 33 studied the in vivo percutaneous absorption of D5 using young adult Sprague-Dawley CD male and female rats (weighing 314-332 g [12 males] and 212-239 g [12 females]). 14C-D5 was applied to the dorsal surface (clipped free of hair) of males and females over a 24-hour period, after which the metabolism cages and exposure site were washed. The animals were then rewrapped with a fresh nonocclusive bandage and returned to metabolism cages for the continued collection of samples. At 96 hours postinitial exposure, the animals were removed from the cages and killed and the exposure site was excised.
The application site, washed prior to excision at 96 hours, contained only 0.35% of the applied dose. Less than 1% of the 14C dose was recovered in the urine and carcass. Trace levels of 14C were found in the feces, CO2 traps, and tissues. The amount of 14C-D5 absorbed (ie, total activity in the excreta, carcass, and dose site) was 0.80% ± 0.62% (n = 11); the total recovery was ∼89%. Most (∼85%) of the 14C-D5 was volatilized from the skin surface (Plotzke and McMahon). 33
Jovanovic and Crofoot 34 studied the in vivo percutaneous absorption of D5 using 4 groups of 4 female Fischer 344 rats (10-11 weeks old, test groups) and untreated control rats. 14C-D5 was applied topically (10.9 mg/cm2 of skin). Of the 4 test groups, 1 group (nonrespiring rats euthanized prior to dose application) was included in order to differentiate expired air from 14C-D5 that had escaped from the skin depot. Another group (wash group) was included, such that the disposition of the residual D5 following a soap and water wash could be evaluated. During exposure, the animals were housed in Roth-style metabolism cages to allow the collection of urine, feces, and expired or escaped volatiles associated with D5. All of the animals were exposed in a semiocclusive manner, using an aluminum skin depot. One group of animals was exposed for 6 hours, and the remaining 3 groups were exposed for 24 hours. At the end of exposure at 24 hours or at 168 hours postexposure, the test animals were killed. The 2 control rats were killed at the 24 hours time point. The charcoal baskets were removed and extracted, and the skin was washed, tape stripped, excised, and solubilized; carcasses were also solubilized. The radioactivity content in each sample was measured by liquid scintillation counting (LSC). The percentage dose absorbed was defined as the amount of radioactivity in the following: carcasses, feces, urine, skin dosing sites, and cage rinses. Radioactivity found in expired volatile and CO2 traps was attributed to leakage of D5 from the dosing chamber and was not included in the total absorption.
The majority of the 14C-D5 that escaped from the dosing chamber was trapped in the charcoal tubes within 6 hours. It was noted that most of the 14C-D5 was evaporated from the skin surface and was trapped in an activated charcoal basket that had been placed above the exposure site. The absorption of 14C-D5 (± standard error [SE] of the mean) in the wash group after 168 hours (0.089% ± 0.0302% of applied dose) was significantly lower (P < .05) than that observed after 24 hours of exposure (0.243% ± 0.0259% of applied dose). The results of this study indicate that the portion of D5 that remained in the skin (without stratum corneum) after 24 hours of exposure (could be considered part of the absorbed dose) migrated to the skin surface and continued to evaporate, resulting in a significant decrease in the apparent absorption of D5 to 0.089% of the applied dose. 34
Jovanovic et al 35 studied in vivo the fate of D4 and D5 absorbed into the skin using rats. A single dose of 14C-D4 (10, 4.8, and 2 mg/cm2) and of 14C-D5 (10 mg/cm2) was applied topically inside of a dosing chamber that was attached to the dorsal area. The collection of urine, feces, and expired/escaped volatiles in metabolism cages occurred over a 24-hour period. The majority of applied D4 or D5 volatilized from the skin surface. Less than 1.0% of applied D4 and 0.2% of applied D5 was absorbed. Approximately 60% of absorbed D4 and 30% of absorbed D5 reached systemic compartments. The amount absorbed into the skin decreased with time, indicating that residual D4 and D5 diffused back into the skin surface and continued to evaporate. Overall, a low tendency to pass through the skin and into systemic compartments was demonstrated for both D4 (≤0.5% of the applied dose) and D5 (<0.1% of the applied dose). 35
In Vitro Studies—Human
Cyclotetrasiloxane (D4) and Cyclopentasiloxane (D5)
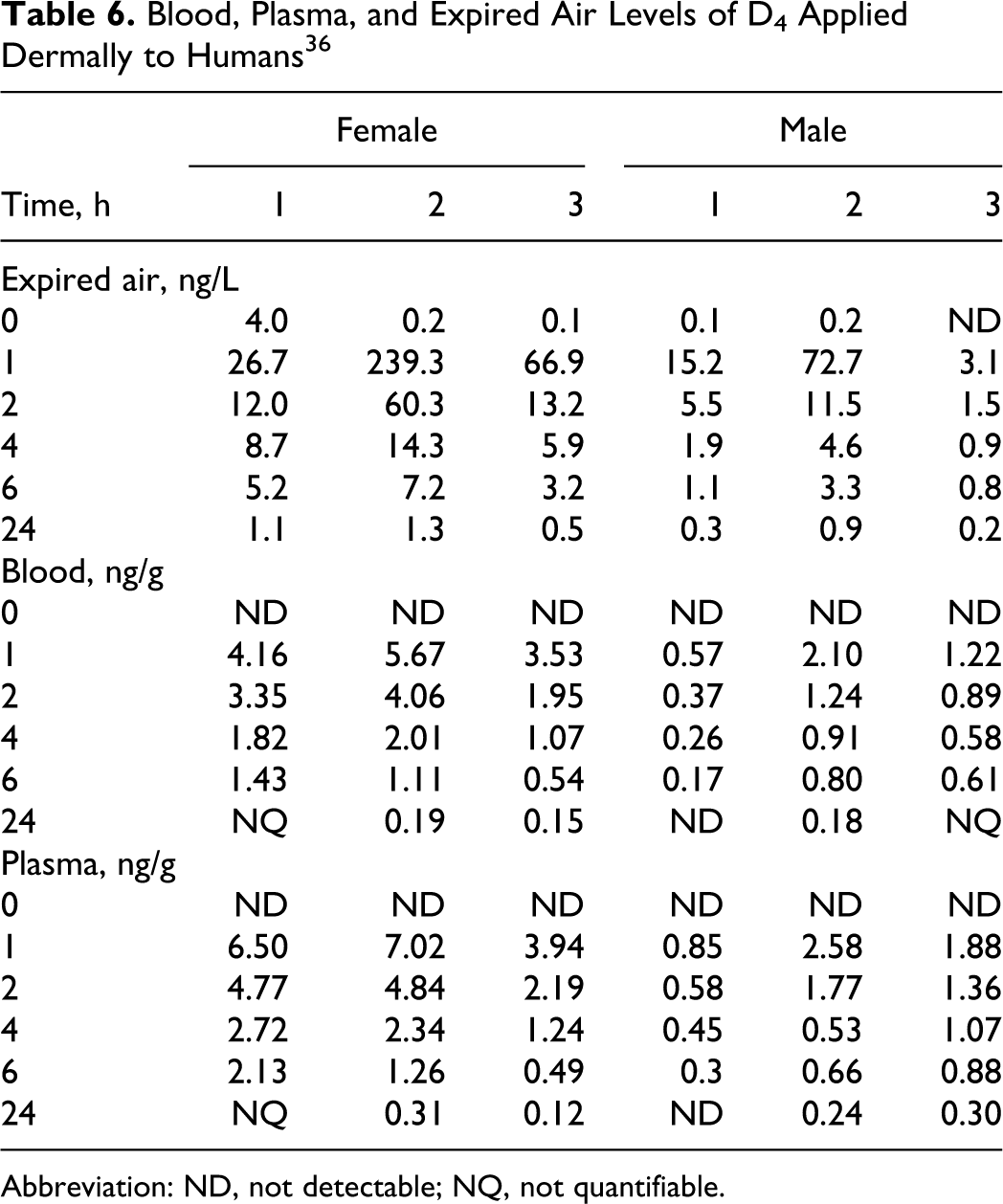
Looney et al 36 conducted a study using normal, healthy volunteers dermally exposed to either 1.4 (3 males) or 1 g (3 females) of C 13 -labeled D4 by applying the compound to the axilla. Study results are included in Table 6. The dose was split between axilla and applied once. Samples of blood and expired air were collected prior to exposure and at 1, 2, 4, 6, and 24 hours postexposure. Of the participants tested, there was considerable variation in the amount of D4 detected in expired air, but this was true to a lesser extent for blood measurements. Peak values were always the 1-hour determinations. Female volunteers were exposed to less D4 than males, nonetheless, they had higher D4 levels in expired air and blood.
Blood, Plasma, and Expired Air Levels of D4 Applied Dermally to Humans 36
Abbreviation: ND, not detectable; NQ, not quantifiable.
In a study by Reddy et al, 37 data for the human dermal absorption of D4 and D5 through axilla skin in vivo were interpreted using pharmacokinetic models of dermal absorption, by adding the dermal exposure route to inhalation physiologically based pharmacokinetic (PBPK) models that were developed previously. Both the D4 and D5 experiments involved 3 male and 3 female participants. Participants in the D5 study were asked not to shave the underarms several days prior to the study, and there was uncertainty as to whether these instructions were given to participants in the D4 study group. In 2 separate syringes, a total of 1.4 g 13C-D4 or 13C-D5 (men) and 1.0 g 13C-D4 or 13C-D5 (women) was weighed. Participants were positioned on their sides prior to administration of the applied dose to the axillae. The contents of 1 syringe were administered to the axilla and allowed to be absorbed and evaporate for approximately 5 minutes. The participant then changed sides, and the second half of the dose was administered to the other axilla. During both studies, the area of the application site was measured and recorded.
In volunteers exposed to either D4 or D5, the maximum concentration of chemical in exhaled air was reached at or prior to 1 hour postadministration of the test material. According to the model calculations, the percentage of applied dose of D4 that was absorbed into the systemic circulation of men and women was 0.12% and 0.309%, respectively. For D5, approximately 0.05% of the applied dose was absorbed in men and women. Model calculations for both D4 and D5 indicate that more than 83% of the chemical that reached the systemic circulation was eliminated via exhalation within 24 hours. 37
In Vitro Studies
Cyclotetrasiloxane (D4)
Jovanovic 38 conducted a study to evaluate the dermal absorption of 14C-D4 (formulated in 3 personal care applications) following application to swine skin. Skin membranes were prepared from fresh Yucatan miniature swine skin. Each personal care formulation was applied to skin samples prepared from 3 swines that were assessed in 6 replicates. The in vitro study was conducted under semioccluded conditions using the Bronaugh flow-through diffusion cell system. Each formulation (skin moisturizer, roll-on antiperspirant, and cuticle coat) was prepared using radiolabeled D4 at 2 different concentrations and analyzed for D4 content by GC with flame ionization detection. The skin moisturizer was formulated with 5.0% and 41.7% 14C-D4, and the roll-on antiperspirant was formulated with 10.6% and 62.2% 14C-D4. The cuticle coat was formulated with 51.6% and 95.8% 14C-D4. The epidermis, with the top layer of dermis, was separated from the remainder of the skin by dermatoming, and each formulation was applied at a targeted dose of 10 mg/cm2.
At the end of the 24-hour exposure, charcoal baskets were removed, the application site was gently blotted and the skin was tape-stripped and solubilized. The penetration of applied 14C-D4 through the skin was determined by analyzing the receptor fluid for radioactivity content. Percentage dose absorbed was calculated from the radioactivity recovered from the skin samples after washing and tape stripping, and receptor fluid samples were collected over a 24-hour period.
Study results indicated that, regardless of the D4 formulation (skin moisturizer, roll-on antiperspirant, or cuticle coat) and applied dose of 14C-D4 (between 0.6 and 10 mg D4 per cm2 swine skin), the majority of applied 14C-D4 volatilized from the skin surface (>90% of applied dose) and was captured in the charcoal baskets placed above the exposure site. The volatilized dose represents 99.5% of the total recovered dose from all formulations with 14C-D4 at all dose levels tested. A small amount of the applied dose (<0.5%) was detected on the skin surface following 24 hours of exposure. The total percentage of the dose absorbed in the skin and receptor fluid was estimated at #0.05% of the applied dose in all experiments. Only a small amount of applied 14C-D4 (< 0.01%) penetrated through the skin into the receptor fluid. The mean cumulative penetration of 14C-D4 over a 24-hour period was <0.60 μg equivalents D4 per cm2 of skin, regardless of the formulation and dose of 14C-D4 applied to the skin. The permeability coefficient was estimated at between 1 × 10−7 cm/h (skin moisturizer, 5% D4) and 1.9 × 10−9 cm/h (antiperspirant, 10.6% D4). 38
Cyclotetrasiloxane (D4) and Cyclopentasiloxane (D5)
Jovanovic et al 35 studied the in vitro percutaneous absorption of 14C-D4 and 14C-D5 through dermatomed human skin using flow-through diffusion cells. Single doses were applied neat and in antiperspirant formulations to the skin for 24 hours. Most of the D4 and D5 that was applied (∼90%) has volatilized before being absorbed. Only 0.5% of applied D4 was absorbed, and the absorption of D5 was one order of magnitude lower (0.04%). The greatest percentage of (>90%) absorbed D4 and D5 was found in the skin.
Cyclopentasiloxane (D5)
Plotzke and McMahon 33 studied the percutaneous absorption of D5 through excised split-thickness skin (381-629 μm) from young adult Sprague-Dawley rats (7 males and 4 females), using Franz diffusion cells. The male and female rats used were 57 and 65 days old, respectively. Following an initial screening to check for skin barrier integrity, 14C-D5 (6.4 mg/cm2) was applied to each skin sample. Over a 24-hour period, measurements of the 14C-D5 that could be rinsed from the skin, that was associated with the skin, or that penetrated through the skin into the receptor were made. The percentages of radioactivity detected in the skin were 0.67% and 1.19% for males and females, respectively. Values for the total amount of D5 absorbed (percentage of radioactivity in the skin and receptor fluid) were 1.08% and 1.54% in males and females, respectively. The majority of the radioactivity was said to have volatilized from the test system, having been trapped in either charcoal baskets or volatile expired air traps.
Jovanovic and Plotzke 39 conducted a study to evaluate the percutaneous absorption of 14C-D5, both neat and formulated in an antiperspirant formulation, when applied to human skin (dermatomed intact abdominal skin from cadavers; semiocclusive conditions) using the Bronaugh flow-through diffusion cell system. 40 The epidermis was separated from the dermis and skin discs from 6 donors were mounted in replicate in the flow-through chambers. Following an initial screening to check for barrier integrity, 2 experiments were performed. In the first experiment, dermatomed full thickness skin samples from 3 participants were placed in diffusion cells in duplicate and dosed with neat 14C-D5. Dermatomed skin samples from the remaining 3 participants were placed in diffusion cells in duplicate and dosed with 14C-D5 formulated into an antiperspirant formulation.
In the second experiment, the second piece of skin (not previously dosed) from the 3 participants dosed with neat 14C-D5 in the first experiment was dosed with the antiperspirant formulation containing 14C-D5. The second piece of skin (not previously dosed) from the 3 participants dosed with the antiperspirant formulation in the first experiment was dosed with neat 14C-D5. In both the experiments, the target application for D5 was 6 mg/cm2 of skin and the target radioactivity for each piece of skin was 6 μCi. The actual applications of D5 ranged from 3.28 to 12.97 mg/cm2 and radioactivity ranged from 2.24 to 8.97 μCi per piece of skin. The average dose of neat D5 was 6.18 mg/cm2, and formulated D5 delivered an average dose of 7.68 mg D5 per cm2 of skin. The percentage dose absorbed was defined as the amount of radioactivity in the receptor fluid (including the radioactivity associated with the Saran wrap that the skin was placed on prior to tape stripping) and the amount that remained on the skin after rinsing and tape stripping.
Based on a statistical analysis of the data, only 0.04% of the applied dose of neat D5 was absorbed at the end of the assay (SE of the mean = 0.007% of the applied dose). This value was not significantly different from that observed with formulated D5 (0.022% ± 0.005% of the applied dose). The percentage of the applied dose that was recovered from all the samples of neat D5 analyzed was 91.45% ± 1.60% and 98.80% for D5 formulated in a generic antiperspirant. The majority of the dose volatilized from the application site and was collected in the charcoal baskets. 39
Cyclohexasiloxane (D6)
Jovanovic and Crofoot 34 evaluated the percutaneous absorption of neat D6 radiolabeled with 14C following application to human skin in vitro. The test substance was applied under semiocclusive conditions in a Teflon flow-through diffusion cell system. Human epidermis was prepared from abdominal skin (6 donors). The epidermis with the top layer of dermis was separated from the rest of the skin by dermatoming, and the skin samples were mounted in replicate. Skin samples from 3 donors passed the barrier integrity test. A physiological receptor fluid was pumped beneath the skin samples. Skin samples from each of the 6 donors were dosed with neat 14C-cylohexasiloxane (14C-D6) at a target dose of 6 mg/cm2 during the 24-hour-exposure period.
At the conclusion of the assay, the majority of the applied dose was located on the skin surface (46.407% of applied dose) or volatilized from the dosing site and collected in charcoal traps (40.057% of applied dose). Practically no 14C-D6 penetrated through the skin and into the receptor fluid. The percentage of applied neat 14C-D6 recovered from all samples that were analyzed was 89.542% ± 4.154%, which included 3.075% of the applied neat 14C-D6 (SE of the mean = 0.852% of the applied dose) that was found in the skin. The results of an additional experiment indicated that, after the skin was washed at 24 hours, the portion of 14C-D6 observed in the skin did not penetrate through the skin, but continued to evaporate. Thus, it was concluded that, under the conditions of this assay, D6 was not percutaneously absorbed. 34
Effect on Corneal Permeability
Cyclotrisiloxane (D3), Cyclotetrasiloxane (D4), and Cyclopentasiloxane (D5)
Green et al 41 evaluated the effect of D3, D4, or D5 on corneal permeability using adult albino rabbits. Two different chemical sources, Petrarch Systems and Ohio Valley Specialty Chemicals, were reported. The eyes were perfused with a purified oil, to which either D3, D4, or D5 was added. In vitro endothelial permeabilities to inulin and dextran were determined after 1 week. D3, D4, and D5 induced a modest increase in permeability. However, another series from a different supplier had no effect.
Green et al 42 evaluated the effect of D3, D4, or D5 on corneal endothelial permeability using adult albino rabbits. Two different chemical sources, United Chemical Technology (source A) and Ohio Valley Specialty Chemicals (source B), were used. The eyes were perfused with a nontoxic oil to which either D3, D4, or D5 was added. Atfter 1 week, the in vitro permeability to inulin and dextran was determined. Dose-response relationships were generated.
The data showed that, where a response is evoked, there is a little effect of concentration (from 1 to 25 mg/mL) on induced permeability changes. D3 (from source A) slightly increased permeability, especially at a dose of 1 mg/mL; D3 (from source B) caused essentially no change. D4 (from both sources) increased dextran permeability in a consistent manner at all test concentrations and enhanced inulin permeability at a dose of 1 mg/mL. D5 (from source A) increased inulin/dextran permeability at doses of 10 and 25 mg/mL, increasing with higher concentrations. However, D5 (from source B) increased dextran permeability at all concentrations, although with a greater increase at 1 mg/mL; D5 increased inulin permeability at a dose of 1 mg/mL.
Generally speaking, a concentration effect was not observed, although lower concentrations tended to cause greater increases in inulin and dextran permeability. However, the exception was D5 (from source A), where a concentration-dependent permeability increase was noted. Dextran permeability, a measure of a smaller pathway across the endothelium (compared with inulin), tended to be influenced to a greater extent than inulin. 42
Absorption, Distribution, Metabolism, and Excretion
In Vivo Studies—Oral
Cyclotetrasiloxane (D4)
Plotzke 43 conducted a study to evaluate the effect of carrier on the absorption and disposition of D4 after oral administration to Fischer 344 rats. The carriers were corn oil, an over-the-counter antacid product (EmulphorTM emulsifying agent), 35 centistoke PDMS fluid, and a Simethicone fluid that was developed to be similar in composition to the PDMS fluid currently found in the commercially available antacid/antiflatulent products. D4 was also delivered neat.
Following a single oral dose (300 mg/kg) of [14C]D4 (neat or in one of the above carriers), animals were placed in glass metabolism cages for the collection of expired air, urine, and feces. Additional animals were euthanized at predetermined time points for the collection of blood. The absorption of radioactivity, expressed as the percentage (mean of 5 animals + SE) of recovered radioactivity in the urine, carcass, expired volatiles, and expired CO2, was studied using each carrier.
D4 absorption was 51.95% ± 4.96%, 12.11% ± 1.21%, and 28.14% ± 5.77% in corn oil, Simethicone, or neat, respectively. The area under the curve (AUC), generated from the blood data, also indicated that the test article was most readily absorbed when delivered in corn oil. The author concluded that the oral absorption of D4 is significantly influenced by the carrier. 43
Cyclopentasiloxane (D5)
Varaprath et al 44 conducted a study to identify urinary metabolites of D5 in the rat. 14C-D5, diluted with unlabeled D5 to a specific activity of 17.377 mCi/mmol, was administered orally to 2 female Fischer 344 rats (CDF(F-344)/CrlBR, −7 to 10 weeks old). The rats received 136.55 or 151.42 μCi of diluted 14C-D5. The animals were placed in a glass metabolism cage to facilitate the collection of urine samples over a 24-hour period. Dimethylsilanediol (Me2Si(OH)2) and methylsilanetriol (MeSi(OH)3) were the major metabolites. The minor metabolites were as follows: MeSi(OH)2-O-Si(OH)3, MeSi(OH)2-O-Si(OH)2 Me, MeSi(OH)2-O-Si(OH)Me2, Me2 Si(OH)-O-Si(OH)Me2, and Me2 Si(OH)-OSiMe2-OSi(OH)Me2. The formation of D4D'OH and MeSi(OH)3 clearly established some demethylation at the siliconmethyl bonds. Parent D5 was not present in the urine.
Cyclohexasiloxane (D6)
Jovanovic 45 evaluated the disposition of 14C-D6 using 10 groups of Fischer 344 rats (CDF(F-344)/CrlBR strain). The animals were 8 to 10 weeks old and body weight ranges were 163 to 219 g (males) and 133 to 155 g (females). A single oral dose of 14C-D6 (in corn oil, 1000 mg/kg body weight) was administered to a group of 4 males and 4 females; metabolism cages were used for the collection of urine, feces, and expired air. The animals were killed at 168 hours postdosing, and selected tissues and remaining carcasses collected and analyzed for radioactivity. Expired volatiles and feces were also analyzed for parent D6 concentration.
A separate group of rats (6 males and 6 females), cannulated via jugular vein, was used to determine radioactivity and parent D6 concentration in the blood at 15 minutes and at 1, 6, 12, 18, 24, 48, 72, 96, 120, 144, and 168 hours postdosing. Whole-body autoradiography (WBA) was used for qualitative in vivo assessment of tissue distribution of radioactivity in male and female rats after single oral administration of D6 (in corn oil). Animals in the WBA groups were killed at 1, 4, 24, 48, 96, and 168 hours postdosing.
In males and females, the majority of the administered dose was excreted in the feces. Based on the recovered radioactivity (urine, expired volatiles, expired CO2, tissues, and carcass), the absorption of D6 was 11.88% (males) and 11.83% (females) of the administered dose. For most of the recovered radioactivity, a similar pattern of distribution of the radioactivity was noted in males and females. However, considerable variability in the levels of radioactivity in expired volatiles was reported, which may have been due to off gassing from the fecal pellets that were not collected, as intended, but remained on the inside of the cage. The authors noted that this phenomenon could potentially produce some false high values for expired volatiles and absorption due to partitioning from the fecal matter into the air. All of the radioactivity in the expired volatiles was attributed to parent D6. Metabolic profile evaluation of the urine and feces indicated that all of the radioactivity in the urine consisted of polar metabolites, whereas, in the feces, the majority was parent D6, with a trace nonpolar metabolite.
Whole body autoradiography data supported mass balance data showing that the majority of administered D6 in corn oil stayed in the gastrointestinal (GI) tract and was excreted in the feces within 48 hours. Low levels of radioactivity were detected in organs and tissues, such as the liver, fat tissue, and bone marrow, indicating some absorption of D6. Statistical analysis of blood curves indicated the presence of small amounts of metabolites in the blood, based on the difference between radioactivity and parent AUCs (AUCmetabolites = AUCradioactivity − AUCparent). 45
In Vivo Studies—Animal Inhalation
Cyclotetrasiloxane (D4)
Crofoot and Plotzke 46 conducted a pilot study to determine the absorption and distribution in blood and tissue, and excretion of [14C]D4 in male Fischer 344 rats following a single nose-only vapor inhalation exposure to 700 ppm for 6 hours. A total of 18 animals were exposed for 6 hours and 3 animals served as controls. Immediately to postexposure, 1 group of 3 exposed animals and 1 control animal were killed and the carcasses were solubilized; radioactivity was counted to determine total body burden. Another 12 animals were used in the distribution portion of the study. At each of 4 given times (0, 24, 48, and 96 hours postexposure), 3 animals were killed, and blood, liver, lung, adrenal, kidney, and spleen tissues, and peritoneal fat tissue were collected and radioactivity was counted (a control animal was killed at 168 hours). The final group of 3 animals was maintained in a metabolism cage for 168 hours, during which time excretia were collected and counted (a control animal was included). At the end of this period, these animals were killed and examined as the 168-hour group for the distribution portion of the study.
The total radioactivity in the carcasses (body burden) after exposure was 6.53% of the exposure. Radioactivity in blood decreased exponentially as a function of time after exposure. Up to 24 hours, radioactivity in plasma paralleled that in blood, but then decreased exponentially at a much less steep slope up to 168 hours. Liver, adrenal, kidney, and spleen tissue followed the same pattern that was noted for the plasma. Radioactivity in lung tissue and fat tissue decreased much less rapidly. At 168 hours, only 7.43% ± 1.97% of the body burden was recovered in the carcasses. In the excretion portion of the study, urine contained 35.75% ± 1.09%, exhalation contained 33.72% ± 14.72%, and feces contained 29.68% ± 2.84% of radioactivity. Only 1.72% was recovered as expired CO2. The authors presented only empirical findings, other than to note that exhaled CO2 was not a major route of excretion. 46
Ferninandi and Beattie 10 developed methodology for measuring inhalation chamber levels of D4 and identified a liquid-trapping medium for capturing expired volatile chemicals in inhalation studies. They used these techniques in a preliminary study to determine blood, plasma, and tissue levels, and excretia and expired volatile chemicals of radioactivity after exposure of 42 male Fischer 344 rats to [14C]D4 in a nose-only inhalation exposure system.
Maximum blood levels were reached in 1 hour and maximum plasma levels in 3 hours after termination of a 6-hour exposure. Most tissues (nasal mucosa, larynx, trachea, lung, liver, kidneys, adrenal glands, pancreas, thymus, bone, and skin) had maximum levels after the 6-hour exposure (0 hours), except fat tissue, which reached maximum levels at 12 hours after the exposure, and the large intestine, which reached a maximum level at 6 hours after the exposure. Elimination from tissue followed the same pattern as that of the plasma (half-life [t ½ ] = 59 hours), except for fat tissue, which was slower (t ½ = 114 hours). Most elimination occurred in expired air (30.68% ± 2.26%) and urine (47.01% ± 2.49%. Fecal recovery was 12.33% ± 0.95%. While elimination of D4 in expired air was high, the amount of radioactivity as 14CO2 was only 1.83%. No unmetabolized D4 was detected in urine and only a small peak that may have been D4 was found in the feces. 10
Ferninandi and Beattie 47 measured the pharmacokinetics of D4 absorption in male and female Fischer 344 rats following single nose-only inhalation exposure to 3 concentrations (50 animals of each sex per group). Exposures (6 hours) were at chamber [14C]D4 concentrations of 716, 70.4, and 7.52 ppm. After exposure, 5 animals per sex were killed and analyzed. Another 5 animals per sex were placed in glass metabolism cages and samples were collected for 168 hours. Other groups of 4 animals per sex were killed as a function of time after exposure up to 120 hours.
As in the study above, maximum levels of radioactivity in blood, plasma, and tissue were achieved 0 to 3 hours postexposure, except for fat tissue, which remained high up to 48 hours postexposure. There were no gender differences in the levels of radioactivity in blood, plasma, or tissues (lung, nasal mucosa, fat tissue, eye, trachea, pancreas, and spleen), except for fat tissue and adrenal glands in females, which were somewhat higher than in males at each dose level. Except for fat tissue (and, to a lesser extent, the vagina and uterus), the increases in tissue radioactivity (AUC values) were generally proportional to the increase in dose level.
Elimination was determined to be biphasic, with a rapid initial decline in the first 24 hours postexposure followed by a long terminal phase, independent of gender. Except for fat tissue in both genders and in the testes in males, the elimination profile for tissues, blood, and plasma were similar. The half-life for the testes was calculated to be 273 hours, indicative of the long terminal elimination phase. Excretion of radioactivity was primarily in expired air and urine in both sexes. In a mass balance analysis, radioactivity remaining in animal carcasses at 168 hours postexposure and ranged from approximately 8% to 12% of the inhaled dose. 47
Varaprath and Beattie (1997) examined the effect of repeated nose-only inhalation exposure of Fischer 344 rats to D4 on absorption, distribution, and excretion. Male and female (50 each) Fischer 344 rats were exposed (nose-only), for 14 days (6 h/d), to D4 and, on day 15, to [14C]D4 at target exposure levels of 7 and 700 ppm. Because of sampling irregularities in the 700 ppm group, an additional group of 29 animals of each sex was exposed at 700 ppm and an additional group of 45 animals of each sex was exposed to 7 ppm. Background radioactivity levels were determined in 2 animals of each sex from each group that were not exposed to [14C]D4.
The calculated level of radioactivity exposure ranged from 35.11 to 40.53 μCi for males and from 27.35 to 33.00 μCi for females. A subset of animals was killed immediately after completion of the day 15 exposure and processed to determine total body burden. Another subset was transferred to glass metabolism cages for determination of radioactivity in urine, feces, expired volatiles and CO2, and cage rinses up to 168 hours postexposure, at which time these animals were killed and processed to determine radioactivity in the blood and tissues. Other subsets were housed in wire mesh cages after completion of exposure on day 15, killed as a function of time postexposure (0, 1, 3, 12, 24, 48, 72, 96, and 120 hours), and processed to determine radioactivity in the blood and tissues.
The total body retention of radioactivity was between 4.38% and 5.96% in males and between 4.50 and 6.14% in females in the 700 ppm groups, and was 5.23% for males and 5.75% for females in the 7 ppm group. At the end of exposure (0 hours), the highest tissue radioactivity levels were seen in the liver (3.6%-4.4%). Liver values were considerably higher than those for the lung (0.71%) or the testes (0.37%). The largest difference between the 2 exposure groups was seen in the radioactivity levels in the nasal mucosa, at 0.29% for the 7 ppm group and 0.06% for the 700 ppm group. Radioactivity in all other tissues was less than 0.2% at 0 hours. When the radioactivity concentrations measured in the adipose tissue samples were extrapolated to the total fat content in the animal, the total fat content of radioactivity was larger than in the liver. The extrapolated fat content values were based on an estimated 7.08% fat content, relative to body weight established in male Holtzmann white rats. The total fat content of radioactivity (extrapolated) was 8.57% ± 0.90% for males and 9.04% ± 1.41% for females at 0 hours. Levels of radioactivity deposited in the fat tissue of 700 ppm males was >8% and >9% in females. In the 7 ppm group, the fat tissue of males contained over 7.5% and the fat tissue of females contained over 12% of radioactivity.
Blood and plasma radioactivity levels peaked at 0 hours and decreased steadily thereafter. At 24 hours postexposure, radioactivity in the liver was reduced by almost an order of magnitude, mimicking the pattern seen with blood and plasma, whereas radioactivity in fat tissue remained constant. Excretion was primarily via the urine (37.4%-40.0%), and the remainder excreted as follows: feces (12.6%-19.1%), expired volatiles (25.9%-35.4%), CO2 (2.06%-4.54%), cage wash (1.31%-1.86%). There was no gender difference, but there was a dose-effect on the route of elimination. High exposure animals excreted more radioactivity in the expired volatiles and CO2 than did the low exposure animals and, correspondingly, high exposure animals had significantly lower excretion in urine and feces compared with low exposure animals. The total elimination, however, was similar in both exposure groups. Radioactivity remaining in the carcasses at 168 hours postexposure ranged from 6.53% to 8.50%, almost all of which was in fat tissue. 48
Tobin 49 reported the disposition of [14C]D4 in female Fischer 344 and Sprague-Dawley IGS rats following a single (nose-only) vapor inhalation exposure to 700 ppm. The protocol described by Crofoot and Plotzke 46 was used with the exception that certain animals were jugular-cannulated to ease blood collection.
At the end of the 6-hour-exposure period, female Fischer 344 rats had a greater body burden (8.3% ± 0.22%) compared with female Sprague-Dawley rats (5.9% ± 0.13%). Total excretion was similar in both the species (amounts in the urine, feces, and expired volatiles of ∼30%, ∼20%, and ∼25%, respectively), except that the excretion was greater in urine and lower in feces for Sprague-Dawley rats compared with Fischer 344 rats. The concentration of radioactivity in blood and lung was similar in the 2 species over the 168 hours postexposure time. Radioactivity levels in the fat tissue and liver were higher for Fischer 344 rats. These authors went on to separately analyze for [14C]D4 (parent compound) as a basis for determining how much radioactivity was associated with metabolites. Overall, the detection of parent compound was greater in Sprague-Dawley rats, although no parent compound was found in the urine of either strain. The metabolites found in urine included dimethylsilanediol and methylsilanetriol. The author concluded that the female Fischer 344 rats appear to metabolize D4 more readily than the female Sprague-Dawley rats. 49
In a study by Plotzke et al, 50 the retention, distribution, metabolism, and excretion of [14C]D4 were evaluated using young adult male and female Fischer 344 rats (weighing 125-210 g). The animals were exposed to D4 vapor in a nose-only inhalation chamber; single and multiple exposures to 7, 70, or 700 ppm [14C]D4 were conducted. There were 4 or 5 male or female animals per group per dose. In multiple exposure studies, the animals were subjected to fourteen 6-hour exposures to unlabeled D4, followed on day 15 by a 6-hour exposure to 14C-D4.
The retention of inhaled D4 was described as relatively low (5%-6% of inhaled D4). It was noted that radioactivity derived from inhaled D4 was distributed widely to tissues of the rat. Except for fat tissue, maximum concentrations of radioactivity in the plasma and tissues occurred at the end of exposure and up to 3 hours postexposure. In fat tissue, maximum concentrations of D4 occurred as late as 24 hours postexposure. Compared with the plasma and other tissues, the elimination of radioactivity from the fat tissue was much slower. The excretion of radioactivity was mainly via exhaled breath and the urine and, to a lesser extent, via the feces. The urinary metabolites included dimethylsilanediol and methylsilanetriol and 5 minor metabolites. The relative abundance of these metabolites was the same for each exposure group. In only female rats subjected to a single exposure, small dose-dependent shifts in elimination pathways were observed. Following multiple exposures, the elimination pathways were dose- and gender-independent. Although the gender difference and dose dependencies noted were statistically significant, they were relatively small in magnitude. Generally speaking, the rates and routes of elimination were similar in males and females (at high and low D4 concentrations). 50
In a study by Siddiqui, 51 male and female Sprague-Dawley rats, CD7 mice, Golden Syrian hamsters, New Zealand White rabbits, and Hartley guinea pigs were repeatedly exposed to D4 (10 or 700 ppm, whole-body exposure) via inhalation 5 d/week (6 h/d) for 5 weeks. Urine samples were collected on days 1, 3, 5, 12, 19, and 25. Urine samples were analyzed for the demethylation of D4 as an indication of liver enzyme metabolism of D4.
The amount of demethylated D4 was less in animals in the 10 ppm exposure group when compared with the 700 ppm group, in all the species at days 3 and 25. Demethylated D4 did not increase uniformly in all the species or sexes between days 3 and 25. The amount of demethylated D4 in the urine of animals of various species roughly follows the order, hamster and mouse > rat > rabbit and guinea pig, with no striking sex differences. The authors suggested that the relatively low levels of demethylated D4 at 25 days were evidence that D4 was not being accumulated in the liver. The authors also postulated that the ratio of demethylated D4 to D4 would be a better measure of liver enzyme metabolic activity. This ratio was highest for hamsters and mice and was significantly higher than those reported for rats, rabbits, and guinea pigs. 51
Lee 52 evaluated the potential chronic toxicity and oncogenicity of D4 in Fischer 344 rats (7-8 weeks old) following whole-body inhalation exposure for 24 months (6 hours + T90 /d, 5 d/week), and details relating to the results of this study are included in the Chronic Inhalation Toxicity and Carcinogenicity sections reported later in the text. Five groups of rats (96 males and 96 females per group) were used and the exposure concentrations were as follows: 0, 10, 30, 150, and 700 ppm D4, respectively. The animals were subdivided into 4 subgroups: A (scheduled for necropsy after 6 months of exposure—tissue level study); B (scheduled for necropsy after 12 months of exposure—chronic toxicity study); C (scheduled for necropsy at 24 months on study, after 12 months of exposure, and a 12-month recovery period—chronic recovery study); and D (scheduled for necropsy after 24 months of exposure—oncogenicity study). Following 6 months of exposure (subgroup A), D4 concentrations in the plasma, liver, and fat tissues increased with increasing D4 exposure concentrations. When compared with male rats, female rats had consistently higher D4 concentrations in these tissues, except for the 700 ppm exposure group.
Cyclopentasiloxane (D5)
Mast 53 conducted a study to evaluate the absorption, distribution, and excretion of radioactivity in female Fischer 344 rats (48 rats; mean weight = 186 – 2.3 g) after a single, nose-only inhalation exposure to 14C-D5. The rats were exposed to 165 – 4.6 ppm 14C-D5 vapor (specific activity = 1.08 – 0.01 mCi/g) for 6 hours. A limited number of male rats was included in the study for the purpose of collecting minute volume data during exposure. Three rats were used as controls in order to establish background radioactivity values. The exposed rats consisted of the following 4 subsets: (1) a body burden group, (2) a distribution group, (3) a distribution and elimination (mass balance) group, and (4) a minute volume group. In the body burden group, 3 of 6 rats were solubilized in toto and the remaining 3 of 6 rats were pelted. The pelt and carcass were solubilized and counted separately to evaluate the amount of deposition on the fur. Rats in the distribution subset were killed at specific times during the 6-hour-exposure period (1.5, 3, and 4.5 hours) or after exposure (0, 1, 3, 12, 24, 48, 72, and 120 hours). Only whole blood and plasma were collected from rats killed during exposure. For rats killed after exposure, whole blood, plasma, and selected tissues were collected.
Rats in the mass balance group were placed in glass metabolism cages for 168 hours and the expired air, urine, and feces were collected at specified intervals. Data from the body burden group were used to determine the estimated dose, and these data were also compared with the data from the mass balance group in order to determine a percent recovery for the study. Data from the mass balance and distribution subsets were used to model the rate of tissue distribution and excretion of 14C-D5. Minute volume measurements were obtained from 6 rats (3 males and 3 females) during the 6-hour-exposure period.
The maximum concentration in each tissue (C max) and time of maximum concentration (T max, determined in hours from the end of exposure) were determined by inspection. The AUC from time 0 to the last measurable concentration was calculated using the trapezoid rule. The estimated terminal-phase t ½ was also calculated.
Mean minute volume measurements were 150 ± 13 and 98 ± 26 mL/min for male and female rats, respectively. The mean achieved dose of 14C-D5 was 88 ± 2 μCi and the mean body burden dose was 2 ± 0.6 μCi (∼3% of the achieved dose). A mean of 97% ± 26% of the body burden dose was recovered from the mass balance group. The following plasma toxicokinetic values were calculated: t ½ = 58.9 hours; AUC = 77 μg ≅ h/g; T max = 0 hour postexposure; and C max = 3.39 μg/mL. 53
Mast 54 conducted a study to evaluate the absorption, distribution, metabolism, and excretion of radioactivity in male and female Fischer 344 rats after a single inhalation exposure (nose-only) to 14C-D5 at 2 dose levels. The first group (69 males [weights: 217.0 ± 7.3 g] and 70 females [weights: 144.6 ± 3.8 g]) was exposed to a nominal concentration of 7 ppm (actual, 6.9 ± 0.2 ppm) 14C-D5 vapor (specific activity = 18.9 ± 0.8 mCi/g), and the second group (70 males [weights: 198.7 ± 5.8 g and 70 females [weights: 128.2 ± 3.2 g]) was exposed to a nominal concentration of 160 ppm (actual, 167.3 ± 3.7 ppm) 14C-D5 vapor (specific activity = 0.919 ± 0.012 mCi/g). Each group was exposed (single exposure) to the test substance for 6 hours. The treated rats consisted of the following 3 subsets: (1) a body burden group, (2) a distribution group, and (3) a distribution and elimination (mass balance) group.
In the body burden group, 4 of 8 rats were solubilized in toto and the remaining 4 of 8 rats were pelted; the pelt and carcass were solubilized and counted separately. Restraining tube rinses containing feces and urine were also analyzed (both groups). Rats in the distribution subset were killed at specified times (at 3 hours) during the 6-hour-exposure or after exposure (0, 1, 3, 12, 24, 48, 72, 96, 120, and 168 hours). Only blood and plasma were collected from rats that were killed during exposure. For rats killed after the exposure period, whole blood, plasma, and selected tissues were collected. Rats in the mass balance group were placed in glass metabolism cages for 168 hours. Data from the body burden group were used to determine the estimated dose, and these data were also compared with data from the mass balance group in order to determine a percent recovery for the study. Data from the mass balance and distribution subsets were used to model the rate of tissue distribution and excretion of 14C-D5.
Approximately 2% of inhaled 14C-D5 was retained in males or females, regardless of the exposure concentration. Mean percentage recovery of the body burden dose for the 7 ppm exposure group was approximately 83% and 72% for males and females, respectively; for the 160 ppm exposure group, these values were 110% and 80% for males and females, respectively.
The highest concentrations of radioactivity (>1 μg equiv/g) immediately following exposure (0 hours) in male and female rats exposed to 7 ppm were detected in the following organs: small and large intestines, stomach, thyroid gland (male only), lungs, and adrenal glands. The highest concentrations of radioactivity (>30 μg equiv/g) immediately following exposure in male and female rats exposed to 160 ppm were detected in the following organs: small and large intestines, stomach, lungs, adrenal glands, and liver. The distribution of radioactivity among the tissues and overtime was approximately the same for males and females. However, the percentage of radioactivity cleared as expired volatiles was significantly greater in males than in females for both exposure concentrations (P < 0.01).
Radioactivity was excreted in approximately equal amounts (in urine and feces) in all groups, with the exception of males exposed to 160 ppm. The excretion of radioactivity was greater in the feces than in the urine in this group. The results of a metabolite profile analysis using high performance liquid chromatrography (HPLC) indicated that the major peak in the feces was parent D5; but, the major peak in the urine did not correspond to 14C-D5. Data analyses showed that, for most tissues, C max occurred at 0 hours postexposure; the predominant exceptions were the thyroid gland (160 ppm males and females) at 120 hours and the perirenal fat tissue (males and females), where C max varied from 3 to 168 hours postexposure. 54
Tobin et al 55 evaluated the disposition of D5 in young adult male and female Fischer 344 rats (weighing 100-220 g) after single or repeated inhalation exposures. Single-exposure groups were subjected to a single 6-hour nose-only exposure to 7 or 160 ppm 14C-D5. Repeated exposure groups were subjected to fourteen 6-hour nose-only exposures to unlabeled D5. The 14th exposure was followed by a 6-hour exposure to 14C-D5 on day 15. Subgroups of exposed animals were used to evaluate body burden, distribution, elimination, and deposition on the fur. The retention of radioactivity after both single and repeated exposures was relatively low (∼1-2% of inhaled D5). Radioactivity and parent D5 were widely distributed to tissues of both male and female rats; the maximum concentration of radioactivity was observed in most tissues by 3 hours postexposure. Adipose tissue was a depot for D5; elimination of D5 was much slower compared with the plasma and other tissues.
The primary route for the elimination of radioactivity was through expired air in all groups. Analyses for parent D5 indicated that essentially all of the radioactivity in the expired volatiles was unchanged D5. Compared with a single exposure, repeated exposure gave rise to higher levels of parent D5 in the lung and fat tissue of males and females and in the liver of females. In fat tissue, immediately after the animals were killed, approximately 50% of the radioactivity was attributed to parent D5. Five polar metabolites of D5 were identified in the urine; parent D5 was not detected. The 2 primay metabolites identified in the urine were methylsilanetriol and dimethylsilanediol. Radiochromatograms demonstrated 2 peaks in the feces. One of the peaks corresponded to the retention time for D5 and the second has been putatively identified as hydroxylated D5. 55
In Vivo Studies—Human Inhalation
Cyclotetrasiloxane (D4)
Utell et al 56 conducted a study, using participants in the age ranging from 25 to 49 years, to examine the respiratory intake and uptake (absorption), blood levels, and elimination patterns of D4 following inhalation exposure to 10 ppm D4. The product of the mean minute ventilation and the inspired D4 concentration constitutes the intake (amount of D4 taken into the lungs), and the product of the mean intake and the respective deposition fraction (fractional uptake of D4) yields the mean uptake. A total of 12 volunteers (8 males and 4 females) were exposed via a mouthpiece-exposure system (two 1-hour exposures) and 8 volunteers were exposed via a nasal device (two 16-hour exposures). D4 blood concentrations (whole blood, 7 volunteers) were measured before, during, immediately after, and at 1, 6, and 24 hours postexposure. Plasma D4 concentrations were measured before and after exposure in 17 participants.
When the exposure data (mouthpiece exposure, 12 participants) were pooled, the mean inspired concentration was equal to 123 ± 7.4 μg/mL, with an overall mean intake of 137 ± 25 mg and a mean uptake of 11 ± 3 mg. The mean D4 elimination in expired air was 2.7 mg, which is equivalent to 24% of the mean uptake. For nasal exposure (8 participants), the D4 exposure concentration averaged 123 ± 4.6 μg/mL. When mouthpiece and nasal breathing were compared, the average total intake was 11.5 versus 14.8 mg and the estimated uptake was 1.1 versus 2.0 mg, respectively. Plasma measurements (17 participants) indicated a mean peak value of 78 ± 6 ng/g and a nonlinear blood clearance. When D4 levels in whole blood were measured (7 volunteers), the values were similar to those reported for plasma, indicating little or no D4 in blood cells.
Symptoms were self-reported, including cough, sputum production, shortness of breath, chest pain, throat irritation, nasal congestion, headache, fatigue, nausea, sneezing, chest tightness, and eye irritation. Ratings were scored on a 1 (minimal/not noticeable unless asked) to 5 (incapacitating) scale. Clinical symptoms were minimal and not different between air and air plus D4. All respiratory function measurements were within the normal range for mouthpiece and nasal exposures. Blood levels of aspartate aminotransferase (AST) were lower following D4 exposures, but the results were not statistically significant. No other blood chemistry was different when air and D4 exposures were compared. 56
Utell et al 57 conducted a pharmacokinetic study of D4 using 12 healthy volunteers (8 males and 4 females; 25-49 years old). The participants inhaled 10 ppm D4 (122 μg/L, via a mouthpiece) or air (control) for 1 hour in a double-blind, randomized fashion. Individual D4 uptakes were measured (under steady-state conditions) during 3 rest periods. These rest periods were 10, 20, and 10 minutes in duration respectively, alternating with two 10-minute-exercise periods. The mean D4 intake (137 – 25 mg) and the mean deposition efficiently were equivalent to 0.74/(1 + 0.45 VE; where VE is minute ventilation).
Changes in lung function were not induced by D4 vapor. Plasma measurements of D4 revealed a mean peak value of 79 – 5 ng/g and a nonlinear blood clearance. A model was developed using lung volume and respiratory surface area estimates based on functional residual capacity. Based on this model, the effective mass transfer coefficient for D4 was determined to be 5.7 × 10−5 cm/s from lung air to blood.
Using an additional 8 participants (6 males and 2 females), D4 deposition with mouthpiece and nasal breathing at resting ventilations was compared. For these participants, deposition was similar for the 2 exposure protocols, averaging 12% after correction for exposure system losses. 57
Utell et al 58 studied the pharmacokinetics of D4 using 6 healthy volunteers (ages: 24-52 years). The participants inhaled 10 ppm labeled D4 (122 μg/L) for 1 hour. The 1-hour-exposure period included two 10-minute exercise periods at a workload that tripled VE. Based on continuously measured inspiratory and expiratory D4 concentrations, it was determined that the mean D4 intake was 154 mg. The expired air washout rates indicated a tetraphasic elimination, amounting to −25% of the D4 uptake. Plasma measurements immediately postexposure revealed a mean peak value of 115 ± 50 ng/g for D4 and 161 ± 53 ng/g in 14C activity equivalents, respectively, and indicated a rapid, nonlinear clearance from the plasma. Similar relationships were found in the blood, with a mean peak value of 82 ± 35 ng/g for D4 in and 115 ± 37 ng/g in 14C activity equivalents.
Urine samples were collected until up to 8 days postexposure. D4 was not identified in the urine; however, 14C-labeled metabolites that were equivalent to 30% of the D4 uptake were recovered. During the first 24 hours, a shift in the elimination pattern of complex to simpler metabolites occurred. Chromatographic peaks at 30, 32, and 37 minutes were noted in all of the zero time spot urine samples. Substances at these retention times have been tentatively identified as trimethyldisiloxane-1,1,3,3-tetrol, tetramethyldisiloxane-1,3-diol, and hexamethyltrisiloxane-1,5-diol. These larger fragments were not detected at times greater than 16 hours postexposure. At postexposure times greater than 16 hours, the metabolites detected in the urine were methylsilanetriol, dimethyldisiloxane-1,3,3,3,-tetrol, and dimethylsilanediol. 58
Reddy et al 59 used data from the preceding study, that is, D4 inspiratory and expiratory concentrations and metabolite concentrations in blood and urine 58 to develop a PBPK model for D4 in humans. The authors noted that D4 has the unusual combination of low blood:air and high fat:blood partitioning, giving rise to preferential storage in lipid compartments in the body, including some sequestration in blood, and rapid elimination from all tissues (other than fat tissue) following cessation of exposure.
In Vivo Studies—Intravenous
Cyclotetrasiloxane (D4)
Huntington Research Centre 60 conducted an intravenous (IV) injection study of the pharmacokinetics of [14C]D4 at 7 and 70 mg/kg, doses designed to mimic those in available inhalation studies. Male and female Sprague-Dawley rats (195-225 g, 7-8 weeks of age) were used in a series of 6 experimental protocols.
In experiment 1 (single low dose—blood and tissue analysis), 20 animals (10 of each sex) were administered [14C]D4 intravenously via the lateral tail vein (dose volume of 1.1 mL) at a nominal dose level of 7 mg/kg. After dosing, the animals were divided randomly into 2 groups of 5 animals per sex. Blood samples (0.3 mL, from tail vein) were placed in heparinized tubes at 10 and 40 minutes, and 2, 6, 24, and 30 hours postdosing for 1 group, and at 20 minutes, 1, 4, 12, and 48 hours postdose for the second group. Animals were killed at the end of blood sampling and the liver, kidneys, lungs, and samples of fat tissue were obtained from each animal. An additional 2 rats (1 of each sex) were dosed in the same fashion, but killed at 0.5 hours postdosing. Whole blood (for plasma analysis) and tissues (liver, lungs, and kidneys) and samples of fat tissue and the tail were taken from each of these animals for analysis.
In experiment 2 (single high dose—blood and tissue analysis), 20 rats (10 of each sex) were administered [14C]D4 at a dose of 70 mg/kg, using the dosing procedure from experiment 1. These animals were divided randomly into 2 groups and blood and tissue samples were taken as described for experiment 1, with the exception that the lungs were not sampled after the animals were killed.
In experiment 3 (single low dose—excretion and tissue analysis), 10 rats (5 of each sex) were administered [14C]D4 at a dose of 7 mg/kg using the dosing procedure from experiment 1. Animals were immediately placed in separate glass metabolism cages for the separate collection of urine, feces, and expired air. Urine was collected separately from each animal at the 0 to 6 hours, 6 to 12 hours, and subsequent 24 hours intervals for up to 5 days. Feces were collected at 24 hours intervals for 5 days. Expired air was monitored for 0 to 6 hours, 6 to 12 hours, 12 to 24 hours, and subsequent 24 hours intervals up to 5 days. The animals were killed at the end of the day 5 and the liver, kidneys, lungs, samples of fat tissue, the GI tract, and remaining carcasses obtained for measurement of radioactivity. The cages were washed and radioactivity was determined in the wash.
In experiment 4 (repeated low dose—blood and tissue analysis), 20 rats (10 per sex) were administered [14C]D4 intravenously (volume 1.1 mL) via the lateral tail vein for 14 consecutive days at a dose of 7 mg/kg per d. Animals were divided randomly into 2 groups (5 per sex). For group 1, blood samples (0.5 mL, from tail vein) were placed in heparinized tubes at 24 hours after days 3 and 9 and at 10 and 40 minutes and 2, 6, 24, and 30 hours after day 14. For group 2, blood samples were taken 24 hours after days 6 and 13 and at 20 minutes and 1, 4, 12 and 48 hours after dosing on day 14. The animals were killed on completion of the sampling schedule and the liver, kidneys, and samples of fat tissue were taken from each animal.
In experiment 5 (single low dose—whole body autoradiography), 6 animals (3 per sex) were treated with [14C]D4 at a dose of 7 mg/kg, as in experiment 1. Two animals (1 of each sex) were killed at 6, 24, and 48 hours postdosing and the carcasses were autoradiographed.
In experiment 6 (repeated low dose—whole body autoradiography), 6 animals (3 per sex) were administered at 14 consecutive daily doses, as in experiment 4. Two animals (1 of each sex) were killed at 6, 24, and 48 hours postdosing and the carcasses were autoradiographed.
Results from experiments 1 and 2 indicated that radioactivity in the plasma was highest at 10 minutes after dosing, having decreased rapidly over the first 6 hours, and, then, more slowly over the remainder of the sampling duration (up to 40 hours), independent of the dose. The ratios of AUC during a dosing interval after multiple doses (experiment 4) to AUC to infinite time following a single dose (experiment 1) were 1.25:1 and 1.16:1 in male and female rats, respectively. This suggested to the authors that there was no plasma accumulation during repeated daily dosing with 7 mg/kg 14C-D4 in rats.
From experiment 3, the routes and rates of excretion of radioactivity varied between male and female animals. For males, excretion was via the urine (48.1%) and feces (10.4%) when compared with females (urine: 28.5%; feces: 7.9%). Correspondingly, the expired air of female animals contained more radioactivity (35.2%) than male animals (22.4%). More radioactivity was found in male CO2 (6.5%) when compared with females (3.2%). The authors suggested that the higher levels of radioactive CO2 could mean higher D4 metabolism in males when compared with females. Tissue levels at 5 days following the single low doses were different between males and females. Overall, levels were higher in females, especially in fat tissue. Table 7 compares the levels of radioactivity in different tissues in males and females.
Concentration of Radioactivity (μg equivalent D4 /g tissue)a in the Tissues of rats Killed 120 hours after a single iv dose of 14C-D4 at 7 mg/kg. 60
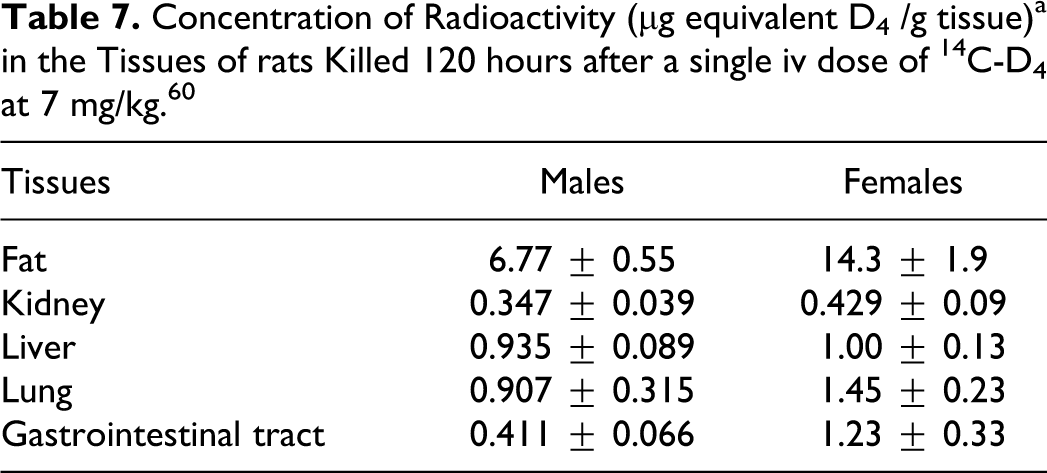
A comparison of the tissue levels from experiments 2 and 4 (single high and low dose, and repeated low doses) was made. Comparing tissue levels after the single low dose with levels after repeated low doses indicates an accumulation of D4 in the fat tissue, liver, and kidney tissue. Tissue levels were higher when measured at 30 or 48 hours after the single high dose when compared with measured values at 30 or 48 hours after the end of the 14th repeated low dose. Table 8 shows the radioactivity levels (without standard deviations) for fat tissue, liver, and kidney tissue.
Mean Concentrations of Radioactivitya in Tissues of Male and Female Animals Killed at Various Times After a Single IV Dose of 70 mg 14C-D4 /kg, a Single IV Dose of 7 mg 14C-D4 /kg, or After Completion of Repeated (14 Days) IV Doses of 7 mg 14C-D4 /kg/d. 60
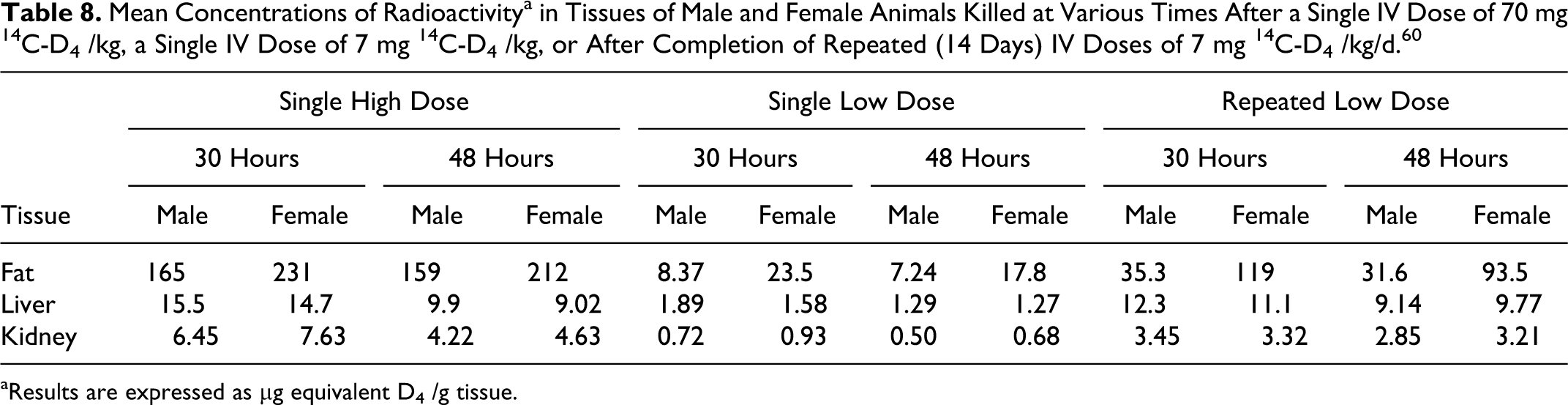
aResults are expressed as μg equivalent D4 /g tissue.
The WBA of animals killed at various times after a single dose at 7 mg/kg (experiment 5) confirmed that radioactivity was well-distributed throughout the animals. The main areas of concentration of radioactivity were the fat tissue, liver, and kidneys of males and females. The autoradiographs from the repeat dose experiment (experiment 6) showed that substantial accumulation of radioactivity in tissues occurs after repeated IV exposure. 60
In a study by Varaprath et al, 61 D4 metabolites in urine samples from 2 separate studies were investigated. In the first study, [14C]D4 was administered intravenously (dose = 70 mg/kg) as an emulsion to a total of 8 male and female Fischer-344 rats (CDF(F-344)/CrlBR) rats, 7 to 10 weeks old. The 12-hour samples from all of the animals were pooled and centrifuged prior to being analyzed. In the second study, 4 female F-344 rats (jugular vein cannulated) were induced with phenobarbital (PB) once a day for 4 consecutive days, and then administered an emulsion (300 μL) of 14C-D4 (IV dose = 70 mg/kg). The 12-hour urine samples from all the animals were pooled and centrifuged in order to obtain a clear fluid prior to analysis.
According to the HPLC radiochromatogram, 2 major metabolites and at least five minor metabolites were identified. The 2 major metabolites (75%-85% of the total radioactivity) were identified as dimethylsilanediol [Me2 Si(OH)2 ] and methylsilanetriol [MeSi(OH)3]. The authors noted that the formation of MeSi(OH)3 clearly established demethylation at the silicon–methyl bonds of D4. No parent D4 was present in the urine. The minor metabolites were as follows: tetramethyldisiloxane-1,3-diol [MeSi(OH)-O-Si(OH)Me2], hexamethyltrisiloxane-1,5-diol [Me2 Si(OH)-OSiMe2-OSi-(OH)Me2], trimethyldisiloxane-1,3,3-triol [MeSi(OH)2-O-Si(OH)Me2], dimethyldisiloxane-1,1,3,3-tetrol [MeSi(OH)2-O-Si(OH)2 Me], and dimethyldisiloxane-1,1,1,3,3-pentol [Si(OH)3-O-Si(OH)2 Me]. 61
In Vivo Studies—Subcutaneous
Cyclotrisiloxane (D3), Cyclotetrasiloxane (D4), Cyclopentasiloxane (D5), Cyclohexasiloxane (D6), and Cycloheptasiloxane (D7)
Kala et al 62 studied the distribution of low molecular weight cyclosiloxanes in the organs of female CD-1 mice (ages 8-10 weeks old; weights: 25-30 g), which were SC injected with breast implant distillate (low molecular weight cyclosiloxane mixture) consisting primarily of D3, D4, D5, D6, and D7. Mice in the test group received a single SC injection of breast implant distillate (dose = 250 mg) and mice in the corresponding control group were each injected with soy oil (dose = 250 mg). Groups of 6 to 8 control and test mice were killed at 3, 6, 9, or 52 weeks postexposure. In groups killed at 3, 6, and 9 weeks, the following organs were dissected: brain, heart, kidney, liver, lung, mesenteric lymph nodes, ovaries, spleen, skeletal muscle, and uterus. The adrenal glands, abdominal fat tissue, and perirenal fat tissue were collected from mice killed at 52 weeks. Tissue extracts were analyzed using GC/MS.
Of the individual cyclosiloxanes that were measured in the organs, D7 was the only one that was not detectable. At 3, 6, and 9 weeks, cyclosiloxanes were detected in every organ that was examined. Of the organs that were studied, the mesenteric lymph nodes, ovaries, and uterus contained the highest levels of cyclosiloxanes. Over a period of 3 to 6 weeks, the levels of total cyclosiloxanes increased in the following organs: heart, kidney, lungs, mesenteric lymph nodes, ovaries, and uterus; at 9 weeks, a slight decline in these levels was noted. It was noted that the relative distribution (from organ to organ) of the cyclosiloxanes varied from mouse to mouse. [In an entirely independent experiment, the 3-week and 6-week cyclosiloxane protocols were completed. Nine mice injected with 250 mg of breast implant distillate and 5 mice injected with 250 mg of soy oil were used for each time point. In the test group, similar levels of total and individual cyclosiloxanes in different organs were found at both time points, indicating that results from the first experiment are reproducible.]
D4, D5, and D6 were detected in all of the organs. The organs from mice killed at 3 weeks, contained proportions of D4, D5, and D6 that were similar to those found in the breast implant that was injected. The levels of D4 at 6 weeks were similar to those noted at 3 weeks. This was not the case for D5 and D6, where, compared with the 3-week values, the levels of these 2 were increased. According to the authors, these data are suggestive of a selective retention of D5 and D6, relative to D4.
In light of the significant retention of cyclosiloxanes that was noted in all organs over a 9-week period, another group of mice was killed 1 year after injection. It was noted that, even after a year, most of the organs had measurable levels of the cyclosiloxanes. The highest levels were detected in the mesenteric lymph nodes, abdominal fat tissue, and in the ovaries. Cyclosiloxane levels in the mesenteric lymph nodes at 1 year were similar to those noted at 9 weeks. However, in the ovaries and uterus, cyclosiloxane levels approached values that were 50% of those noted at 9 weeks. It was also noted that the levels of D5 and D6 were relatively higher than those of D4.
The results of this study indicate that low molecular weight cyclosiloxanes persist in the organs of mice for at least 1 year after a single SC injection and that D5 and D6 appear to persist longer in the organs when compared with D4. 62
In Vivo Studies—PBPK Modeling
Cyclotetrasiloxane (D4)
Andersen et al 63 developed a PBPK model to quantitatively characterize the retention, distribution, and elimination of parent D4 and its hydrolysis and oxidation products from the body following controlled inhalation exposures in the rat. Both single-exposure and multiple-exposure inhalation studies were conducted, where male and female F344 rats were exposed to D4 at the concentrations of 7, 70, and 700 ppm. 50 In the single exposure studies, the animals were exposed to 14C-D4 vapor for 6 hours in a nose-only inhalation chamber. Exposure was initiated following a 4-day conditioning period. Multiple-exposure studies were initiated following a similar conditioning period. In multiple exposure studies, the animals were exposed to unlabeled D4 6 h/d for 14 consecutive days. The animals were then exposed to 14C-D4 vapor for 6 hours on day 15. Immediately after the last exposure, rats were placed in glass metabolism cages for the collection of excreta (urine and feces) and expired air.
Following inhalation exposure, radioactivity was distributed widely throughout rat tissues; the highest concentrations were reported for the lung, liver, and fat tissue. Within 1 hour postexposure, the peak concentration of D4 in most of the solid tissues and plasma was noted. The radioactivity time course in these tissues displayed multiphasic behavior, that is, a relatively rapid initial decline, up to 24 hours postexposure, followed by a much slower terminal phase. Radioactivity persisted longer in the fat tissue, with peak concentrations noted after several hours (3-24 hours) postexposure. The decline in the concentration of D4 in the fat tissue was nonexponential. The excretion of radioactivity following exposure to radiolabeled D4 was mainly via exhaled breath and urine, with a lesser extent in the feces. The radioactivity in the expired volatiles was almost all parent D4; however, the radioactivity that was detected in the urine was entirely due to metabolized D4. In male and female rats, the radioactivity time courses in the tissues were similar. Also, the disposition of radioactivity in the multiple exposure experiments was similar to that observed in the single exposure studies.
A basic PBPK model was applied to characterize the biological determinants of D4 kinetics. Blood:air partition coefficients were small (range: 1.9-6.9, 6 samples); perirenal fat:air partition coefficients were large (range: 1400-2500). The average fat:blood partition coefficient was determined to be 485. The authors noted that the basic PBPK model failed to describe D4 tissue kinetics in the postexposure period, which necessitated expansion by adding deep-tissue compartments in the liver, and lung, a mobile chylomicron-like lipid transport pool in the blood, and a second fat tissue compartment. Model parameters for the refined model were optimized using single-exposure data for male and female rats exposed to 7, 70, and 700 ppm D4. When the induction of D4 metabolism at 700 ppm (3-fold in males and 1-fold in females) was included, the parameter that was set from the single exposures successfully predicted PBPK results from the 14-day multiple exposures at 7 and 700 ppm.
Although D4 is highly lipophilic, it does not have prolonged retention because of the high hepatic and exhalation clearance. The low blood:air partitioning and high fat:blood partitioning led to the incorporation of D4 into lipid storage compartments that were not equilibrated with free circulating D4. Despite the unusual transport and storage behavior in lipid compartments, the high pulmonary and hepatic clearance, coupled with the induction of metabolizing enzymes at high exposure concentrations, rapidly remove free D4 from the body and ensure that there is no accumulation on multiple exposures. 63
Luu and Hutter 64 developed a PBPK model to predict the target organ doses of D4 following IV, inhalation, or implantation exposures. For these modes of exposure, the plasma radioactivity profiles of 14C-D4 showed nonlinear kinetics. In the IV rat studies, the highest radioactivity accumulated in fat tissue, followed by richly perfused tissues (ie, lungs and brain), blood, liver, and kidneys. Approximately 60% to 80% of the absorbed D4 dose was exhaled and excreted in the urine and feces. The animals were placed in metabolism cages and urine, feces, and expired air were collected at 0 to 6 hours, 6 to 12 hours, and subsequent 24 hours intervals for up to 5 days.
In the PBPK model, 14C-D4 IV disposition data in rats were used to estimate tissue distribution coefficients, metabolism, and excretion parameters. The model was validated by comparing the predicted blood and tissue concentrations of D4 after inhalation exposure to experimental results in both rats and humans. The model was then used to simulate D4 kinetics following single and/or repeated D4 exposures in rats and humans. This model predicted the bioaccumulation of D4 in fatty tissues (eg, breast), especially in women. Due to its high lipid solubility, (log P oct/water = 5.1), D4 persisted in fat tissue, with a t ½ of 11.1 days after inhalation and 18.2 days after breast implant exposure. With repeated exposures, larger doses and/or different routes of exposure, both metabolism and excretion remained constant. The authors noted that the accumulation of D4 in fatty tissues should play an important role in the risk assessment of D4, especially in women exposed daily to multiple personal care products and silicone breast implants. 64
Meeks 65 commented on the bioavailability of D4 following inhalation and implantation exposure. He noted that Luu and Hutter 64 had developed a PBPK model for exposure to D4 via the following 2 routes: (1) inhalation in association with daily use of multiple personal care products and (2) migration of small amounts of silicone fluid from silicone gel breast implants. This PBPK model was based on data generated by IV administration of D4 as a microemulsion 63 that was then modeled for intra-arterial dosing. In this model, it was assumed that all radioactivity was parent D4, although there is significant conversion of D4 to hydrophilic metabolites.
Meeks 65 noted that a complete PBPK model was developed from an extensive inhalation data set on D4 50 , including an evaluation of D4 metabolism. Meeks concluded that this more comprehensive model and the data from a 6-month inhalation study indicate that there are only modest increases in the concentration of D4 in fat tissue after repeated exposures to D4, compared with the concentrations achieved following single exposures. Meeks 65 also noted that Luu and Hutter 64 also overestimated the contributions to daily intake that resulted from the migration of D4 from a breast implant. However, according to Meeks, 65 this overestimation of the daily intake by Luu and Hutter 64 did not significantly change the margin of exposure (MOE) for D4, and the conservative MOE of >500 indicates that current usage practices with D4 have adequate safety margins. The MOE of >500 is based on the following: (1) conservative estimate of daily D4 intake from personal care products = 78 μg/kg per d, based on use of D4 in roll-on antiperspirants; (2) addition of estimated daily intake or exposure by Luu and Hutter 64 yields an estimated total daily intake of 85 μg/kg per d for D4; (3) the exposure of rats to 300 ppm (v/v) of D4 for 6 h/d equates to an inhaled dose of 45 000 μg/kg per d, using an absorption value of 5%, as determined in the inhalation pharmacokinetic studies 50 ; and (4) the preceding values yield a margin of safety (MOS or MOE), by dividing the no-observed-adverse effect level (NOAEL) by the estimated daily intake; thus, MOE ≥ 500.
Andersen et al 66 also commented on the publication by Luu and Hutter. 64 Using human modeling, Andersen et al 66 found that the structure of the rat PBPK model for D4 (blood:air partition coefficient = 1) was entirely adequate for describing all of the available data from human volunteers. The authors expressed concern over the inaccurate attribution of conclusions of the modeling efforts by Luu and Hutter. 64 Andersen et al 66 emphasized that the kinetics are well described, with a blood:air partition coefficient of 1 in both rats and humans, when sequestration in blood lipids is included in the model structure. It was noted that because of the high rate of metabolism and exhalation of poorly soluble D4 from the blood, there should be little tendency for D4 to bioaccumulate in any tissues upon repeated exposures.
Taking into consideration that the inhalation of D4 induces cytochrome P450 (CYP) 2B1/2 protein and causes liver enlargement, Sarangapani et al 67 developed a pharmacodynamic (PD) extension to a PBPK model to characterize these dose-response behaviors in Fischer 344 rats. It was noted that the PD model simulates interactions of D4 with a putative receptor, leading to increased production of CYP2B1/2. Both 1-compartment and 5-compartment liver models were evaluated. The authors noted that the PBPK model provided excellent simulations of tissue D4 and hepatic CYP2B1/2 protein concentrations following 5-day (6 h/d) inhalation exposures to 0, 1, 7, 30, 70, 150, 300, 500, 700, or 900 ppm D4.
Sarangapani et al 68 developed a PBPK model to describe the tissue dosimetry, plasma concentration, and clearance in the F344 rat, after inhalation, dermal, oral, and IV exposure to D4. It was determined that a refined model describing the delivery of D4 from the GI tract to the nonexchangeable/deep blood compartment provided the best fits to the observed plasma D4, exhaled D4, and D4 metabolites excreted in the urine after oral exposure. The authors noted that pharmacokinetics following IV administration was best described by the delivery of D4 directly into the deep blood compartment, possibly reflecting a kinetically identifiable characteristic of the administration of D4 as an emulsion for the IV route of exposure. The results of this particular model-based analysis indicate that the pharmacokinetics of D4 delivered by inhalation or dermal routes is similar, and these are different from the IV or oral delivery routes. 68
Dobrev et al 69 conducted a study with the following objectives: (1) to evaluate rate constants for saturable metabolism in the body, (2) to estimate possible presystemic D4 clearance by respiratory tract tissues, and (3) to assess rate constants for uptake of D4 after oral dosing. Kinetic parameters for oral uptake and metabolism were evaluated using sensitive closed and open chamber inhalation techniques. Closed chamber exhalation analysis following oral gavage provided an opportunity to verify oral uptake rate constants inferred from blood time-course results at constant concentration inhalation and the estimates of metabolic rate inferred from total amount metabolized in these same studies. The kinetics of oral absorption of D4 was studied using male Fischer 344 rats that had been dosed by oral gavage (doses of 10, 50, 100, 200, or 300 mg/kg D4 in corn oil). Immediately after dosing, the rats were transferred to either an open or a closed chamber system. The concentration time course of exhaled D4 in the chamber air was recorded for up to 10 hours. Gas uptake studies were used to independently assess the whole body metabolism of D4, including the possibility of presystemic clearance by respiratory tissue before reaching the pulmonary blood. These experiments were conducted using male Fischer 344 rats in an all-glass closed chamber system. Chamber concentrations of D4 ranged from 19 to 1020 ppm. The elimination of D4 from the chamber atmosphere was monitored for up to 6.7 hours.
These experiments provided the opportunity to refine current PBPK models for D4. The PBPK model could only be fitted to gas uptake results when metabolic capacity was included in the respiratory tract epithelium. The model simulations were highly sensitive to the parameter for total percent of whole-body metabolism allocated to the respiratory tract, with optimal fits observed with this value equal to 5%. Conclusions from the oral uptake studies were also verified by comparisons with independent data sets for blood concentrations of D4 after oral dosing. The pharmacokinetic analysis of uptake from the gut and release from the blood into chamber air results for oral doses ranging from 10 to 300 mg D4/kg body weight were consistent with a combination of prolonged and slow uptake of D4 from the GI tract and of reduced absorption at higher doses as well as the extrahepatic clearance of D4 in pulmonary tissues. The authors stated that these closed chamber gas uptake studies provide a valuable confirmation of the conclusions reached in other pharmacokinetic studies and have uncovered a situation whereby closed chamber loss is highly sensitive to respiratory tract clearance. This sensitivity arises largely from the unusual characteristics of D4, high-affinity metabolic clearance, and low blood:air partitioning.
The authors noted that, in this study, the examination of an independent data set for establishing kinetic parameters for modeling D4 metabolism and oral uptake has provided fairly good confirmation of the values utilized in previous multidose modeling studies by Saranapani et al, 68 as summarized above. 69
Cyclopentasiloxane (D5)
Reddy et al 70 developed an inhalation PBPK description for D5 (using D4 model structure as a starting point), with the objective of understanding factors that regulate free blood and tissue concentrations of this highly lipophilic vapor after inhalation in rats and humans. The authors noted that the inhalation pharmacokinetics of cyclotetrasiloxane (D4) have been extensively investigated and successfully modeled with a multispecies PBPK model. Compared with D4, the more lipophilic D5 required deep compartments in the lung, liver, and plasma to account for slow release from tissues after the exposure period. Simulations of the kinetics of a stable D5 metabolite, HO-D5, required diffusion-limited uptake in fat tissue, a deep tissue store in the lung, and its elimination by fecal excretion and metabolism to linear silanols.
The combined D5/HO-D5 model described blood and tissue concentrations of parent D5 and the elimination of total radioactivity in single and repeat exposures in male and female rats at 7 and 160 ppm. In humans, D5 kinetic data are more sparse and the model structure, although much simplified, still required free and bound blood D5 to simulate exhaled air and blood time courses from 1 hour inhalation exposures at 10 ppm in 5 human volunteers. This multispecies PBPK model for D5 highlights complications in interpreting kinetic studies where chemical in the blood and tissues represents various pools with only a portion free. The ability to simulate free concentrations is essential for dosimetry-based risk assessments for volatile cyclic methyl siloxanes. 70
Andersen et al 71 evaluated the extent to which highly cleared, lipophilic vapors are expected to accumulate in blood and tisues. Pharmacokinetic analysis was conducted using both a generic PBPK model for inhalation of volatile compounds and a more detailed PBPK model specifically developed for a highly lipophilic volatile (D5). The generic PBPK model for inhalation of volatile compounds in humans showed that highly metabolized, lipophilic compounds with a low blood:air partition coefficient do not accumulate in blood or systemic tissues with repeat exposures, although a period of days to weeks may be required for fat tissue to reach periodic steady state. Volatile compounds with higher blood:air partition coefficients (in the hundreds) and lower hepatic extraction accumulate in the blood on repeat exposures. The more detailed PBPK model for D5 also showed that the lipophilic volatile compound does not accumulate in blood and the predictions of the increase in D5 in fat tissue with repeat exposures in rats agreed with experiments. The authors noted that the term bioaccumulation should be used to refer to cases where repeat exposures lead to increases in the blood (or central compartment) volatile compound concentration. Based on this definition, highly cleared volatile compounds, such as D5, would not be considered to bioaccumulate on repeat exposures.
In Vivo/In Vitro Study
Cyclotetrasiloxane (D4)
Varaprath et al 12 obtained biological samples from several pharmacokinetic studies that were being conducted to develop an efficient method for extracting both D4 and its metabolites. Male and female Fischer-344 rats (CDF(F-344)/CrlBR strain; ∼7 to 10 weeks old) were used. The studies involved the administration of 14C-D4 at different dosage levels and collecting samples at specified time points. This means that the test samples contained varying levels of parent D4 and metabolites. In the pharmacokinetic investigation of the disposition and metabolism of D4, an efficient extraction methodology was used to recover the parent D4 and its metabolites from the following biological matrices: blood, plasma, urine, feces, liver, lung, and fat tissue. There were no qualitative differences in the distribution of radioactivity or metabolite profiles between male and female rats in either of the pharmacokinetic studies. Therefore, the choice of samples from male and female rats was based on availability.
In one of the in vivo studies, male F-344 rats were exposed (nose-only) to 14C-D4 vapor (mean chamber concentration of 716 ppm) for 6 hours. Groups of 3 rats were killed and tissues were collected at selected time points. Feces samples were obtained from a subset of 3 rats and placed in all-glass metabolism chambers.
In another in vivo study, emulsified 14C-D4 was administered IV to 4 F-344 male rats at a dose 130 mg in order to achieve a 20-mg/kg (20 μCi) dose of D4. Blood samples were obtained from male rats at 1, 8, 24, and 48 hours. Male rats from another group were dosed IV with 245 μL of 14C-D4 emulsion (26.1 μCi; D4 dose = 70 mg/kg). Plasma samples were separated from blood samples obtained at 24 hours. Female F-344 rats each received an IV dose of ∼400 mg of 14C-D4 emulsion and urine samples were collected (at 24 hours) from female F-344 rats that each received an IV dose.
Following inhalation or IV injection, only metabolites of D4 were present in the urine; parent D4 was completely absent. For lung tissues collected at 24 hours after exposure to a single dose (tail vein) of 14C-D4, the parent D4 silicone moiety predominated. In all the other matrices, varying proportions of D4 and its metabolites were present.
Varaprath et al 12 described an in vitro method for extracting D4 from tissues (using glass beads), so that levels could be measured in the in vivo studies. This method and extraction efficiencies data are included in Analytical Methods section reported earlier in the text.
In Vitro Study
Cyclotetrasiloxane (D4)
Usuki 72 conducted a study to determine the role of human liver microsomal enzymes in the in vitro metabolism of [14C]-D4. [14C]-D4 was incubated with pooled liver microsomes obtained from humans, saline-treated (control) rats, or PB-treated rats to assess species differences and proportionality of metabolite formation with respect to incubation time and protein concentration (determination of the initial rate conditions). Other experiments were conducted to determine the CYP enzymes that are involved in the metabolism of [14C]-D4 by human liver microsomes. A combination of the following 2 experimental approaches was used: (1) metabolism of [14C]-D4 by individual recombinant human CYP enzymes and (2) evaluation of the effects of specific antibodies against selected CYP enzymes on the metabolism of [14C]-D4 by human liver microsomes. Study results are included below.
[14C]-D4 was metabolized by human liver microsomes in vitro to a single metabolite that was named M8. Although the conversion of D4 to M8 did not exceed 10%, M8 formation was not proportional to protein concentration or the incubation time. The results of the experiment to assess [14C]-D4 binding to human liver microsomes suggest that the loss of radioactivity in the incubation sample was not due to the binding of [14C]-D4 and its metabolite(s) to the microsomal protein. Considering that D4 is a volatile chemical, it is possible that one or more of its metabolites is also volatile, making its quantification difficult. The volatility of metabolites is one possible explanation for the lack of proportionality of metabolite formation with incubation time and protein concentration.
With human microsomes (and, possibly, liver microsomes from saline-treated rats), the results (lack of proportionality with incubation time and protein concentration) appear to suggest that only a portion of added D4 is available for metabolism, which might occur if D4 is bound to plastic, formed micelles, or formed a film on the surface of the incubation medium. The observation of extensive metabolism of D4 with microsomes from PB-treated rats was not considered an assay artifact, but possibly suggests that microsomal metabolism of D4 in the uninduced system is a complex blend of enzyme action and inhibition.
Based on the results of experiments with recombinant human CYP enzymes and polyclonal antibodies, it was concluded that [14C]-D4 is primarily metabolized to M8 in vitro, and that CYP2B6 and CYP3A4 are largely responsible for the formation of M8. 72
Modification of Metabolism
Cyclotetrasiloxane (D4)
Plotzke and Salyers 73 reported a pilot study to determine whether classical inducing agents alter the metabolic profile of a single IV dose of [14C]D4 in rats. The authors noted that D4 has been demonstrated to induce hepatic CYP enzymes, but that it was not clear whether CYP is involved in the metabolism of D4. The authors postulated that pretreatment with PB would increase the endogenous levels of CYP and potentiate the metabolism of D4. To examine if other P450 subfamilies may be involved, pretreatment with 3-methylcholanthrene was performed.
Female Fischer 344 rats were pretreated for 4 days with either PB (80 mg/kg per d, 4 animals intraperitoneally [IP]) in saline or with 3-methylcholanthrene (30 mg/kg per d, 4 animals IP) in corn oil or saline (3 animals) or corn oil (2 animals). Each of these pretreated animals was given a single IV dose of [14C]D4 (70 mg/kg). Another group of 5 animals received a single oral dose [14C]D4 (70 mg/kg) only, with no pretreatment of any kind. Urine, feces, and expired air were collected over a 72-hour period, at which time the animals were killed and the carcasses were solubilized and analyzed for total radioactivity.
Urinary excretion was the primary route of elimination of 14C-D4 (injected IV) derived radiolabel in rats pretreated with PB. Within 72 hours, approximately 55% of the administered radiolabel was excreted in the urine of PB-treated rats, while control and 3-methylcholanthrene-pretreated rats excreted approximately 24% to 27% in the urine over the same period. The major route of elimination of radiolabel in control and 3-methylcholanthrene-pretreated rats was via expired volatiles. Following a single IV dose of 14C-D4, approximately 38% of the dose was excreted as expired volatiles in control rats, while 3-methylcholanthrene-pretreated rats excreted approximately 29% and PB-pretreated rats excreted 9% over the same period.
After a single oral dose of 14C-D4, 22% of the dose was excreted as expired volatiles over a 72-hour period. Control rats receiving a single IV dose excreted 38% of the administered dose. Urinary excretion was similar for the 2 routes of D4 administration (IV or oral). Control rats dosed IV with 14C-D4 excreted 24% of the administered dose in the urine over a 72-hour period, while control rats dosed orally excreted 31% of the dose.
The elimination of 14CO2 appeared to have been independent of the route of administration. Rats dosed orally or IV with 14C-D4 expired 3% of the administered dose as 14CO2 over the 72-hour period. Following IV dosing, the elimination of 14C-D4-derived radioactivity in the feces (<8%) was a minor route of excretion in control rats. However, after oral dosing with 14C-D4, fecal elimination accounted for 29% of the administered dose. At 72 hours postdosing, 18% of the radioactivity remained in the carcass of rats dosed orally with D4; 29% of the radioactivity remained in the carcass of control rats dosed IV with D4.
An HPLC analysis of urine did not indicate any qualitative change in metabolites in rats pretreated with PB or 3-methylcholanthrene, when compared with controls. A similar profile of at least 6 D4 metabolites and absence of the parent compound was associated with all samples, including the rats dosed orally with 14C-D4. The results of this study indicated that PB pretreatment increased both the amount and rate of urinary excretion of radioactivity after a single IV dose of 14C-D4, and that there were differences in the major route of excretion following different routes of administration. 73
Enzyme Effects
Cyclotetrasiloxane (D4) and Cyclopentasiloxane (D5)
McKim et al 74 determined the time course for changes in hepatic microsomal CYP enzyme expression after repeated inhalation exposure to D4. Male (weighing 175-200 g) and female (weighing 130-145 g) Fischer 344 rats were used. Groups of 3 to 6 rats were exposed to 0, 70, or 700 ppm D4 5 days per week (6 h/d) for 4 weeks. The animals were killed on exposure days 3, 7, 14, 21, and 28. Microsomal fractions were prepared and enzyme activity as well as immunoreactive protein levels of several (CYP) enzymes, epoxide hydrolase (EH), and UDP-glucuronosyltransferase (UDPGT) were evaluated. The time course for enzyme induction was monitored by measuring 7-ethoxyresorufin-O-deethylase (EROD) and 7-pentoxyresorufin-O-depentylase (PROD) activities on days 3, 7, 14, 21, and 28. The detection of immunoreactive proteins was accomplished with polyclonal antibodies raised against rat liver microsomal CYP1A1/2, CYP2B1/2, CYP3A1/2, CYP4A, and EH.
Results indicated that CYP1A1/2 activity (determined by EROD activity) increased approximately 2- to 3-fold during the exposure period. In consideration of this finding, it was noted that an examination of immunoreactive protein revealed no induction of CYP1A1 and suppression of CYP1A2 in the 700 ppm D4 exposure group. CYP2B1/2 enzyme activity (determined by PROD activity) was significantly increased, as early as on day 3, in the 70 and 700 ppm exposure groups (male and female rats). On day 28, PROD activity was induced by more than 10-fold in the 70 ppm groups and by more than 20-fold in the 700 ppm exposure groups. A comparable increase in CYP2B1/2 immunoreactive protein paralleled the increase in PROD activity. As determined by 6β-hydroxylation of testosterone and Western blot analysis, a modest (1.5- to 4-fold) increase in CYP3A1/2 activity and immunoreactive protein was noted. There was no effect on CYP4A activity or protein level.
By day 3 for 700 ppm and day 7 for 70 ppm, the expression of CYP enzymes was at or near maximum and remained relatively constant throughout the exposure period. On day 28, EH activity and immunoreactive protein were induced (2- to 3-fold) in a dose-dependent manner. The slight changes in the expression and activity of UDPT detected were not found to be dose-related.
It was concluded that repeated inhalation exposure to D4 induces CYP enzymes and EH in a manner that is similar to that observed for PB. D4 was described as a PB-like inducer of hepatic microsomal enzymes in the Fischer 344 rat. 74
Madan 75 evaluated the ability of D4 to inhibit the following major P450 enzymes in human liver microsomes: CYP1A2, CYP2A6, CYP2B6, CYP2C9, CYP2C19, CYP2D6, CYP2E1, CYP3A4/5, and CYP4A9/11. The first experiment was performed to evaluate D4 as a direct-acting (metabolism-independent) reversible inhibitor of P450 activity. In this experiment, pooled human liver microsomes from 7 participants were incubated with marker substrates at the concentrations Km/2, Km, and 4Km, with or without D4 at the concentrations ranging from 0.032 to 2.9 μmol/L.
In the second experiment, D4 was evaluated for its ability to function as a metabolism-dependent (mechanism-based) reversible inhibitor of P450 activity. Pooled human liver microsomes from 7 participants were incubated for 0 or 15 minutes with D4 and NADPH prior to addition of the marker substrate at a final concentration Km. The highest concentration of D4 that resulted in less than 30% inhibition as a reversible inhibitor was the target concentration of D4 at which the preincubations were carried out, provided that the D4 concentration was not greater than 3.3 μmol/L.
A third experiment was performed to evaluate D4 as an irreversible metabolism-dependent inhibitor of P450 activity. Pooled human liver microsomes from 7 participants were preincubated with D4 and NADPH for 15 minutes, to allow the generation of metabolites that could inhibit CYP noncompetitively. Following the preincubation period, an aliquot of microsomes was removed and added to an incubation mixture containing the marker substrate. Another incubation was carried out to measure the residual P450 activity. The highest concentration of D4 that resulted in less than 30% inhibition as a reversible inhibitor was multiplied by the dilution factor to give the target concentration of D4 at which the preincubations were carried out, provided that the D4 concentration was not greater than 3.7 μmol/L. In addition to the use of human liver microsomes, D4 was evaluated for its ability to inhibit CYP1A1/2 and CYP2B1/2 in liver microsomes from rats treated with 3-methylcholanthrene and PB, respectively.
Study results are summarized as follows: (1) D4 appears to be a noncompetitive inhibitor of rat CYP2B1/2, with an estimated Ki value of 0.11 μmol/L. (2) D4 appears to be a noncompetitive inhibitor of human CYP2B6, with an estimated Ki value of 3.6 μmol/L. (3) D4 appears to be a competitive inhibitor of human CYP1A2, with an estimated Ki value of 12 μmol/L. (4) D4 appears to be a noncompetitive inhibitor of human CYP2D6 and CYP3A4/5, with estimated Ki values of 14 and 11 μmol/L, respectively. (5) D4 appears to be either a competitive or noncompetitive inhibitor of CYP2C19, with estimated Ki values of 6.4 or 11 μmol/L, respectively. (6) D4 has little or no capacity to inhibit rat CYP1A2 and human CYP2A6, CYP2C9, and CYP4A9/11 activity. (7) The Ki value for D4 as an inhibitor of human CYP2E1 was not estimated because D4 is an activator, not an inhibitor, of human CYP2E1. (8) D4 has little or no capacity to function as a metabolism-dependent (reversible or irreversible) inhibitor of any of the P450 enzymes examined, with the possible exception of rat CYP1A1/2 and human CYP3A4/5, which were weakly inhibited by D4 in a reversible metabolism-dependent manner. 75
In an inhalation toxicity study by Siddiqui, 51 summarized later in the report text (Short-Term Inhalation Toxicity section), the enzymes assayed in male and female rats and guinea pigs were glutathione S-transferase, EH, and ECOD. Animals were exposed to either 10 or 700 ppm D4 for 6 h/d for 5 weeks. Microsomal and cytosolic fractions were obtained from all livers following standard procedures, and respective enzyme assays were conducted.
All 3 enzymes were significantly activated in treated male rats, compared with controls, but only EH and ECOD were activated in female rats. Activation of these liver enzymes was found in treated guinea pigs. 51
McKim et al 76 investigated the effects of D5 on the expression and activity of selected rat hepatic phase I and phase II metabolizing enzymes. Female Fischer-344 rats (weighing 128-138 g) were exposed to 160 ppm D5 vapors 7 days per week (6 h/d) for 28 days. The animals were exposed via whole body inhalation. Changes in the activity and relative abundance of hepatic microsomal CYPs (CYP1A, CYP2B, CYP3A, and CYP4A), EH, and UDPGT were measured. By day 28, the repeated inhalation exposure of rats to D5 increased liver size by 16% relative to controls. During a 14-day postexposure period, significant recovery from the increased liver size was noted. D5 exposure did not change total hepatic P450, but increased the activity of hepatic NADPH-cytochrome c reductase by 1.4-fold. An evaluation of CYP enzymes in hepatic microsomes prepared from D5-exposed rats indicated a slight (1.9-fold) increase in EROD activity, but no change in immunoreactive CYP2B1/2 protein was observed.
Testosterone 6β-hydroxylase activity was also increased (2.4-fold), as was CYP3A1/2 immunoreactive protein. Although a small increase in 11- and 12-hydroxylation of lauric acid was detected, no change in immunoreactive CYP4A levels was measured. Liver microsomal EH activity and immunoreactive protein increased 1.7- and 1.4-fold, respectively, in the D5-exposed group. UDPGT activity toward chloramphenicol was induced 1.8-fold, while no change in UDPGT activity toward 4-nitrophenol was observed. Study results suggest that the profile for enzyme induction following inhalation exposure of female Fischer-344 rats to D5 vapors is similar to that reported for PB; therefore, D5 may be described as a weak PB-like inducer. 76
Zhang et al 77 conducted a study to characterize the ability of D4 and D5 to induce drug metabolizing enzymes in male and female Sprague-Dawley rats (weights = 200-230 g). D4 or D5 (in corn oil) was administered at doses of 1, 5, 20, or 100 mg/kg daily by gavage for 4 days. Significant increases in the liver-to-body-weight ratio were observed in female rats dosed with either D4 or D5 at doses ≥ 20 mg/kg. In male rats, increases in liver-to-body weight were noted at doses ≥100 mg/kg D5, but were not associated with D4 exposure. With increasing doses of D4 and D5, there were relatively large increases in CYP2B1/2 enzymatic activity and immunoreactive protein.
D4 at doses of ≥5 mg/kg induced significant increases in PROD activity in male and female rats. Dosing with D5 induced an increase in PROD activity in male rats (at doses ≥ 20 mg/kg) and in female rats (at doses ≥ 5 mg/kg). In both male and female rats dosed with ≥ 20 mg/kg D4 or ≥ 5 mg/kg D5, EROD activity was increased. There was no indication of changes in CYP1A1/2 immunoreactive protein levels in male or female rats. Significant increases in CYP3A1/2 immunoreactive protein were noted only in male rats dosed with either D4 or D5 at a dose of 100 mg/kg. In female rats, significant increases in CYP3A1/2 immunoreactive protein were observed at doses of ≥ 20 mg/kg (D4) and ≥ 5 mg/kg (D5).
The induction of NADPH cytochrome P-450 reductase immunoreactive protein was observed after female rats were dosed with D4 and after male and female rats were dosed with D5. The induction of CYP2B/1/2, CYP3A1/2, and NADPH cytochrome P-450 reductase was observed in rats injected IP with 50 mg/kg PB.
The authors noted that both D4 and D5 induced CYP2B1/2 in the adult rat liver in a manner that was similar to that observed following dosing with PB. However, differences between D4 and D5 in the ability to induce CYP3A1/2 and NADPH cytochrome P-450 reductase were observed. Additionally, when compared with male rats, female rats were more sensitive to the inductive properties of low doses of both D4 and D5; male rats were more responsive to PB induction. 77
Madan 78 evaluated the ability of D5 to inhibit the following major P450 enzymes in human liver microsomes in 3 experiments: CYP1A2, CYP2A6, CYP2B6, CYP2C9, CYP2C19, CYP2D6, CYP2E1, CYP3A4/5, and CYP4A9/11. The first experiment was performed to evaluate D5 as a direct-acting (metabolism-independent) reversible inhibitor of P450 activity. In this experiment, pooled human liver microsomes from 7 participants were incubated with marker substrates at concentrations Km/2, Km, and 4Km, with or without D5, at concentrations ranging from 0.040 to 3.5 μmol/L.
In the second experiment, D5 was evaluated for its ability to function as a metabolism-dependent (mechanism-based) reversible inhibitor of P450 activity. Pooled human liver microsomes from 7 participants were incubated for 0 or 15 minutes with D5 and NADPH prior to addition of the marker substrate at a final concentration Km. The highest concentration of D5 that resulted in less than 30% inhibition as a reversible inhibitor was the target concentration of D5 at which the preincubations were carried out.
A third experiment was performed to evaluate D5 as an irreversible metabolism-dependent inhibitor of P450 activity. Pooled human liver microsomes from 7 participants were preincubated with D5 and NADPH for 15 minutes, to allow the generation of metabolites that could inhibit CYP noncompetitively. Following the preincubation period, an aliquot of microsomes was removed and added to an incubation mixture containing the marker substrate. Another incubation was carried out to measure the residual P450 activity. The highest concentration of D5 that resulted in less than 20% inhibition as a reversible inhibitor was multiplied by the dilution factor to give the target concentration of D5, at which the preincubations were carried out.
In addition to the use of human liver microsomes, D5 was evaluated for its ability to inhibit CYP1A1/2 and CYP2B1/2 in liver microsomes from rats treated with 3-methylcholanthrene and PB, respectively.
Study results are summarized as follows: (1) D5 appears to inhibit 7-pentoxyresorufin-O-dealkylation (marker reaction of CYP2B1/2 activity) catalyzed by liver microsomes from rats treated with PB in a manner that does not conform to prototypical types of inhibition (ie, competitive, noncompetitive, uncompetitive, or mixed competitive–noncompetitive); this inhibition increases significantly with an increase in marker substrate concentration. (2) D5 did not inhibit another marker substrate reaction for CYP2B1/2, namely testosterone 16β-hydroxylation, suggesting that the observed inhibition of 7-pentoresorufin was an experimental artifact. This was attributed to the direct interaction of D5 with the marker substrate, 7-pentoxyresorufin. (3) D5 did not directly inhibit rat CYP1A1/2 activity; however, preincubation of D5 in the presence of microsomes and a NADPH-generating system produced metabolites that appeared to reversibly inhibit CYP1A1/2. (4) D5 appears to be a weak competitive inhibitor of human CYP3A4/5, with an estimated Ki value of 6.9 μmol/L. (5) D5 appears to be a strong reversible (and weak irreversible) metabolism-dependent inhibitor of human CYP3A4/5. (6) D5 has little or no capacity to inhibit rat CYP1A2 and human CYP1A2, CYP2A6, CYP2B6, CYP2C9, CYP2C19, CYP2D6, CYP2E1, and CYP4A9/11 activities in a reversible metabolism-independent manner. (7) D5 has little or no capacity to function as a metabolism-dependent (reversible or irreversible) inhibitor of rat CYP2B1/2 activity or human lCYP1A2, CYP2A6, CYP2B6, CYP2C9, CYP2C19, CYP2D6, CYP2E1, and CYP4A9/11 activities. 78
Hormonal Effects
Cyclotetrasiloxane (D4)
McKim et al 79 examined the potential estrogenic and antiestrogenic activities of D4 using Sprague-Dawley (Cr: CD7 VAF/Plus7 strain, 18 days old; weight = 40 g; 12 pups per group) and Fischer-344 rats (COBS7CDF7 (F-344/CrlBR) strain, 21 days old; weight = 30 g; 12 pups per group). These 2 strains were used to address potential differences in sensitivity. Estrogenicity was determined by measuring absolute and relative uterine weights in immature rats and by monitoring uterine epithelial cell height. To place the data obtained for D4 in perspective, with respect to strong and weak estrogenic compounds, the response produced by D4 at 0, 10, 50, 100, 250, 500, and 1000 mg/kg per d was compared with responses produced by ethinyl estradiol (EE, 1, 3, 10, or 30 μg/kg per d), diethylstilbestetrol dipropionate (DES-DP, 0.5, 1.5, 5, and 15 μg/kg per d), and coumestrol (CE, 10, 35, 75, and 150 mg/kg per d). Antiestrogenic effects were studied by coadministering D4 (500 mg/kg per d) with EE.
D4 and the other substances tested were administered (in sesame oil, dose volume = 5 mL/kg) by oral gavage. Beginning on postnatal day (PND) 18 (Sprague-Dawley rats) or 21 (F-344 rats), each pup (12 per group) was given a single oral dose once per day for 4 consecutive days. The pups were killed on the morning after the last dose; uteri were removed and processed for microscopic examination.
In the 1000 mg/kg per d dose group (Sprague-Dawley rats), D4 caused a statistically significant decrease in absolute body weight on day 21, but not on day 22. In F-344 rats dosed with D4 (1000 mg/kg per d), a statistically significant decrease in absolute body weight was noted on days 23, 24, and 25.
Both EE and DES-DP induced a significant dose-dependent increase in absolute and relative uterine weights and uterine cell height. The weak phytoestrogen, CE, also induced an increase in both absolute and relative uterine weight as well as epithelial cell height; however, the response was noted over a much higher range of doses.
At doses >100 mg/kg per d, both absolute and relative uterine weights and uterine epithelial cell height were increased in both Sprague-Dawley and F344 rats; these changes were classified as statistically significant. Regarding uterine weight, D4 was approximately 0.6 million times less potent than EE or DES-DP in Sprague-Dawley pups and 3.8 million times less potent than EE or DES-DP in F-344 pups. Compared with controls, the maximal increase in uterine weight induced by D4 was approximately 160% in Sprague-Dawley rats; the maximum increase induced by D4 in F-344 rats was 86%. It was concluded that D4-induced weak estrogenic and antiestrogenic activity that was several orders of magnitude less potent than EE, and many times less potent than the weak phytoestrogen CE. 79 It is important to note that a similar uterotrophic effect of D4 was reported in a study by Quinn et al 80 summarized in Receptor Agonist/Binding Activity section later in the report text.
Plotzke 81 conducted a study to evaluate the potential estrogenic properties of D4. An in vitro estrogen responsive reporter gene system was used and D4 as well as 17β-estradiol (E2), diethylstilbesterol, and bisphenol A were evaluated using the MCF-7 human cell line (7 experiments). In experiments 1 and 2, the cells were exposed to D4 (10.0 μmol/L) or E2 (0.3 nmol/L) for 24 or 48 hours. An approximate 3- to 6-fold induction of the pS2 reporter gene mRNA, with significant induction during the 24-hour-exposure period, was reported for D4 and E2. The results of experiment 3 indicated that, for D4, the increase in pS2 is a dose-dependent effect in the range of 0.1 to 10 μmol/L. E2 (0.03-0.3 μmol/L), diethylstilbesterol (0.01-1.0 μmol/L), and bisphenol A (1.0-10.0 μmol/L) also induced a dose-dependent increase in pS2 expression.
The effect of shorter exposure times on pS2 induction was evaluated in experiment 5. Increased pS2 expression was observed following exposure to D4 (10 μmol/L) and E2 (0.3 nmol/L) at exposure periods ranging from 15 minutes to 24 hours. Maximal expression was observed between 6 and 24 hours; shorter exposures (15 minutes to 6 hours) resulted in increasing levels of mRNA. A rapid loss of D4 from the medium was noted, reaching a plateau at −12 hours (<10% D4 remaining).
In experiment 6, the potential for D4 to interfere with the action of E2 was evaluated by exposing MCF-7 cells to a combination of E2 and D4 and then measuring reporter gene activation. When E2 (0.3 nmol/L) in combination with a range of D4 doses (25nmol/L to 10 μmol/L) was added, a reduction in E2-induced pS2 expression was absent, with the exception of a slight decrease in one dose group (1 μmol/L). The purpose of experiment 7 was to compare the E2 dose response curve, with and without coincubation with 1.0 μmol/L D4. The regression analysis resulted in parallel slopes with statistically indistinguishable y-intercepts, indicating that D4 was not reducing the potency of E2 under these conditions.
The results of this study indicate that, using the in vitro reporter gene system, D4 appears to have estrogenic potential at an applied dose ranging from 0.1 to 10 μmol/L. The combination of a wide range of D4 concentrations in combination with E2 did not cause a significant reduction in E2 (0.3 nmol/L)-induced pS2 expression, except at a concentration of 1.0 μmol/L; however, D4 did not alter the regression analysis of the E2 dose response curve. These data suggest that D4 can elicit an estrogenic effect that is dose-dependent, with no significant antiestrogenic activity. 81
Stump 82 evaluated the potential of D4 to affect the preovulatory luteinizing hormone (LH) surge using 5 groups of ovariectomized (OVEX) female Sprague-Dawley Crl:CD (SD) IGS BR rats. Each of the 3 groups (groups 1-3) contained −50 rats and the remaining 2 groups (groups 4 and 5) each contained 5 rats. The target test article concentrations were 700 ppm (group 2) and 900 ppm (groups 3 and 5); the animals were exposed to D4 for 6 hours on day 0. The 2 control groups (groups 1 and 4) were exposed to clean, filtered air according to the same procedure. On day 0, the mean body weights were as follows: 0 ppm (group 1 [49 rats] and group 4 [4 rats]: 267 ± 13.0 g and 269 ± 3.3 g, respectively), 700 ppm (group 2 [49 rats]: 265 ± 14.4 g), and 900 ppm (group 3 [50 rats] and group 5 [4 rats]: 268 ± 12.9 g and 267 ± 3.3 g). In order to synchronize the estrous cycles, the females were OVEX and SC implanted with E2.
The animals were killed at appropriate intervals following the 6-hour-exposure period, and blood was collected and analyzed for prolactin, LH, E2, and/or estrone (E1). Rats from groups 1, 2, and 3 were killed by decapitation and blood was collected during this procedure. Blood from anesthetized rats of groups 4 and 5 was collected from the vena cava, after which the animals were killed via exsanguination. There were no significant, exposure-related clinical observations at the conclusion of exposure. In blood samples (from 700 and 900 ppm groups) collected via decapitation, there were no statistically significant effects on LH levels. Statistically significant decreases of prolactin in blood samples collected via decapitation were noted in females exposed to 700 or 900 ppm D4. Estradiol blood levels were decreased at the end of exposure, but subsequently returned to control levels.
The level of E1 in blood samples collected from the vena cavae of rats in the 900 ppm exposure group (4 rats per group) was similar to that of control rats. The E2:E1 ratio in these samples from the 900 ppm exposure group was decreased (not a statistically significant reduction) when compared with control values. It was concluded that group mean LH levels in OVEX female rats treated with E2 (SC implant) were similar to the control group mean after a single 6 hours exposure to 700 or 900 ppm D4. 82
Quinn et al 83 studied the potential for D4 to suppress the pre-ovulatory LH surge, to block or delay ovulation, and evaluated potential effects on reproductive hormones in female Sprague-Dawley Crl:CD7 (SD) IGS BR rats. The exposure protocol was divided into 2 phases. In phase 1, groups of noncannulated rats were exposed (whole-body inhalation) to 0 (filtered air), 700, or 900 ppm D4 (groups of 24, 22, and 27 rats, respectively). The exposure duration was 6 hours per day on diestrus days 1 and 2 and a 2.5-hour exposure on the day of proestrus. Trunk blood obtained at proestrus was evaluated for levels of the following hormones: follicle stimulating hormone (FSH), E2, E1, and progesterone (P4).
In phase 2 of the experiment, groups of cannulated animals were exposed to 0 (filtered air), 700, or 900 ppm D4. The duration of exposure was 6 hours per day for 3 days (day 3 of diestrous through proestrus). Serial blood samples were collected via a cannula at 2, 4, 6, 8, and 10
During proestrus, significant increases in the levels of E1 in rats of the 700 and 900 ppm exposure groups and in the levels of P4 in the 900 ppm exposure group were noted. During estrus, significant increases in E1, E2, and the E1/E2 ratio, and decreases in FSH were observed in the 700 and 900 ppm exposure groups. Regardless of the treatment, there was a correlation between suppression of the LH surge and the blockage of ovulation. The percentages of rats that ovulated were 42% (at 700 ppm) and 31% (at 900 ppm) compared with a control value of 79%. The results of this study indicate that high exposures to D4 attenuated the preovulatory LH surge and significantly decreased the proportion of female rats that ovulated. 83
Receptor Agonist/Binding Activity
Cyclotrisiloxane (D3), Cyclotetrasiloxane (D4), and Cyclopentasiloxane (D5)
He et al 84 conducted a study to characterize the estrogenic potential of D3, D4, and D5 using female estrogen receptor-α knockout (αERKO) mice. Wild-type control mice of the same genetic background (129/J/C57BL/6j) as the αERKO mice and female B6C3F1 mice were also used. The mice received a total of seven 1000 mg/kg doses. Serum E2 concentrations decreased in a dose-dependent manner following exposure to oral doses of D4 ranging from 100 to 1000 mg/kg. The results of studies involving adrenalectomized animals indicated that the decreased serum E2 levels were not caused by elevated levels of serum cortisone. In OVEX mice, uterine wet weights were significantly increased in a dose-dependent manner following exposure to oral doses of D4 ranging from 250 to 1000 mg/kg. However, this was not observed following exposure to D3 or D5. Similarly, uterine peroxidase activity (marker for estrogenic activity) was significantly increased in mice dosed orally with D4, but not in mice dosed orally with D3 or D5.
Pretreatment with ICI 182,780 (estrogen receptor [ER] agonist) resulted in complete blockage of the increase in uterine weight that was induced by D4. No increases in uterine weight were observed after OVEX ER-α knockout mice were dosed orally with D4 or E2. The results of an in vitro ER-binding assay indicated significant competition between D4 and 3H-E2 relative to binding to the ER-α, but not ER-β. The data presented in this study indicate that D4 has weak estrogenic activity and that these effects are mediated through ER-α. 84
Quinn 85 conducted a study to measure the ability of D5 to competitively displace tritiated E2 from the ER, subtype alpha. Initial experiments measured the ability of radiolabeled 14C-D5 vapor (160 ppm) to partition into the aqueous mixture, monitored over a 4-hour incubation period. There was a rapid increase in radiolabeled material in the aqueous mixture that initially reached equilibrium after 3 hours. The concentration of D5 was measured in the 4-hour incubation mixture by LSC and by GC/MS. The calculated 14C-D5 concentration was 0.29 μmol/L by LSC. The calculated GC/MS D5 concentration was 0.36 μmol/L.
Once the optimal incubation time and reaction conditions were established, saturation binding experiments and competition experiments were carried out. The saturation binding experiments were used as an internal control evaluating the state of the receptor and the binding conditions with the natural ligand. In each case, the calculated Kd of the E2 was within the expected range (0.5-5 nmol/L), indicating that the receptor binding assay conditions were appropriate.
The measurement of D5 binding to the ER-α was carried out using a relative binding affinity (RBA) method. D5 (160 ppm) was incubated with a limited amount of the alpha receptor in the presence of physiological levels of radiolabeled E2 (3.4 nmol/L). The D5, delivered as a 160-ppm vapor in the headspace of the aqueous reaction, was unable to displace any of the radiolabeled E2, indicating that, in this in vitro system, D5 is not a direct competitor for the receptor. 85
Quinn 86 conducted a study to measure the ability of D5 to competitively displace tritiated E2 from the subtype beta, ER. To evaluate the binding potential of D5 with this receptor system, both saturation binding experiments and competition experiments were carried out. The saturation binding experiments were used as an internal control to evaluating the state of the receptor by E2. In each case, the binding curves indicated that the specific binding of the receptor by E2 was consistent and that the receptor binding assay conditions were appropriate.
The measurement of D5 binding to the ER-β was carried out using an RBA method. A 160 ppm D5 vapor was introduced into the 1-mL headspace of 2 mL sealed vials. The test article was incubated for 3 hours at 37°C, with gentle inversion in the presence of a limited amount of receptor beta and physiological levels of radiolabeled E2 (3.4 mmol/L). D5 was unable to displace any of the radiolabeled E2, indicating that, in this in vitro system, D5 is not a direct competitor for the subtype beta, ER. 86
You 87 conducted a study to test the hypothesis that the in vivo induction of CYP2B1/2 expression by D4 and D5 suggests the possibility that D4 and D5 are activators of the constitutive androstane receptor (CAR) in the rat. CAR is a nuclear receptor that has been shown to be a key regulator in PB-induced, upregulation of CYP2VB1/2 gene expression. This study evaluated the potential for D4 and D5 to activate CARs representative of rat and human, utilizing reporter gene assays.
The test system comprised HepG2 cells transiently infected with CAR plasmid (coding for rat or human CAR), a luciferase reporter plasmid containing a 1.8-kb human CYP2B6 promoter, and a transfection control plasmid (Renilla luciferase). The transfected cells were exposed to D4 and D5 at nominal concentrations of 0, 1, 5, 15, 31, 62.5, 125, 250, 500, and 1000 μmol/L for 24 hours. The siloxane/media dose solutions were replaced after the initial 12 hours incubation, in anticipation of decreasing media concentrations related to evaporation and adherence of the cyclic siloxane to the plastic culture plates. Reporter gene expression was determined by the level of luminescence after the second 12 hours incubation.
The evaluation of D4 in the rat CAR assay demonstrated statistically significant induction of reporter gene expression of up to 5-fold. In contrast, the evaluation of D5 in this assay over the same range of nominal concentrations gave rise to a maximum induction of less than 2-fold. The increased response with increased dose represented a statistically significant trend; however, individual dose responses were not statistically different from the control. Both D4 and D5 demonstrated statistically significant induction of reporter gene expression in the human CAR assay, with induction as high as 11-fold and 6-fold, respectively. Maximum induction was observed at the nominal dose level of 62 μmol/L, with the exception of D5, in the rat CAR assay. In this case, maximal induction occurred at 250 μmol/L.
The inclusion of positive and negative assay controls demonstrated that the assay was performed as designed. The compound 1,4-bis[2-(3,5- dichloropyridyloxy)] benzene, a known rodent CAR activator, was used at a concentration of 250 nmol/L. The compound 6-(4-chlorophenyl)imidazo{2,1-b][1,3]thiazole-5-carbaldehyde O-(3,4-dichlorobenzyl)oxime (CITCO) was used as an activator of human CAR at a concentration of 1 μmol/L. Transfection with an empty vector plasmid, one not containing the CAR gene rather than the CAR expression plasmid, served as the negative control.
Considering that CAR activity can be repressed by androstenol and that repression can represent CAR activation, D4 and D5 were assessed in the presence of an inhibitory concentration of androstenol. D4 induced reporter gene expression in the rat CAR assay by approximately 6-fold in the presence of androstenol. In contrast, the most effective concentrations of D5 were only able to reverse the effects of androstenol, returning reporter gene expression to the levels present in the constitutive control. Androstenol did not repress reporter gene expression in the human CAR assay, a finding that is consistent with known species differences.
The induction of reporter gene expression by D4 in the human CAR assay was not affected by androstenol. Conversely, the presence of androstenol gave rise to a generalized increase in response to CITCO (positive control) and to D5. There was an apparent extension of the dose range for D5, with maximal induction occurring at 250 μmol/L in the presence of androstenol, versus 62 μmol/L without androstenol. This unusual response remains unexplained. The stabilization and/or enhancement of effective media concentrations are postulated contributors to the noted effect.
It was concluded that the results of this in vitro study identify D4 and D5 as functional activators of rat and human CAR. The authors noted that this finding is consistent with in vivo hepatic enzyme induction studies in rats.{You, 2005 178 /id}
Jean 88 investigated the potential for D4 and D5 to act as pituitary dopamine receptor agonists using an in vivo rat model. Groups of female Fischer-344 rats (6-10 rats/ group, 8 in most groups; weight minimum = 90 g) were used. The model was explained as follows: Female Fischer-344 rats were pretreated with reserpine (depletes dopamine in the brain). Dopamine is a key regulator of serum prolactin levels, because dopamine released from the hypothalamus activates dopamine D2-receptors on the pituitary gland to inhibit the secretion of prolactin. Thus, reserpine-induced dopamine depletion results in a persistent and marked increase in serum prolactin levels. The administration of bromocriptine (dopamine receptor agonist) under these conditions results in a marked decrease in serum prolactin. However, if the rat is treated with sulpiride (dopamine receptor antagonist) prior to treatment with bromocriptine, the decrease in serum prolactin may be partially or completely blocked. This outcome serves to demonstrate that the actions of each agent are related to their interaction with/competition for the dopamine receptor.
Rats pretreated with reserpine were exposed (nose-only, vapor inhalation) to 700 ppm D4 or 160 ppm D5 for 6 hours. OVEX rats that were not pretreated with reserpine served as controls. There was also a reserpine-treated control group. Trunk blood was obtained immediately after exposure for prolactin analysis. Two separate experiments were performed. A sulpiride (dopamine receptor antagonist) pretreatment group was added to the second experiment to determine whether D4 acts at the level of the receptor. In each experiment, exposure to 700 ppm D4 reduced serum prolactin levels to those of control rats (non-reserpine-treated, OVEX rats). This reduction amounted to more than 80% relative to the reserpine-treated control group. Sulpiride administration prior to D4 exposure (experiment 2) blocked the prolactin-lowering activity of D4, indicating that D4 is a dopamine D2-receptor agonist.
The results for D5 were similar to those reported for D4. The exposure of reserpine-treated rats to 160 ppm D5 (nose-only vapor inhalation) for 6 hours produced a 34% decrease (average of 2 experiments) in serum prolactin. The administration of sulpiride prior to D5 exposure blocked the prolactin-lowering activity of D5, indicating that D5 acted as a dopamine D2-receptor agonist. It was concluded that serum prolactin levels were significantly decreased in reserpine-pretreated rats after a single 6-hour vapor inhalation exposure to 700 ppm D4 and 160 ppm D5 (each independently). This finding was said to be consistent with D4 and D5 acting as dopamine D2-receptor agonists. 88
Jean 89 conducted a study to screen for the potential of cyclic siloxanes (D4 and D5) as pituitary dopamine D2-receptor agonists, using an in vitro cell line (MMQ cells) derived from rat pituitary tumor. This cell line was selected as the test system because it produces and secretes prolactin and expresses functional dopamine D2 receptors. The model system used requires an inducer, such as maitotoxin, to elevate prolactin secretion. Maitotoxin-induced prolactin secretion is dopamine D2-receptor agonist sensitive, allowing for assessment of test materials as dopamine D2-receptor agonists.
MMQ cells propagated in the laboratory produced and secreted prolactin, with and without induction by maitotoxin. Maitotoxin (1.5 and 3 ng/mL) induced less than a 2-fold increase in prolactin secretion over the course of a 30-minute incubation. Pretreatment with 1 μmol/L dopamine reduced (>70%) the maitotoxin-induced increase in prolactin secretion.
Both D4 and D5 completely inhibited maitotoxin-induced prolactin secretion at a nominal concentration of 10 μmol/L. Whether or not this action was receptor mediated has not been confirmed in this system. However, the demonstrated activity in this screening assay provided additional evidence in support of a proposed study designed to assess the potential of D4 and D5 to activate pituitary dopamine D2-receptors and downregulate pituitary prolactin secretion in vivo.
The results of this screening study provide data supporting the hypothesis that D4 and D5 are dopamine D2-receptor agonists. 89
Jean 90 evaluated the potential of D4 and D5 as progesterone receptor (PR) ligands using the following receptor-binding assays: those utilizing cell-free, classical radioligand binding assays and a state-of-the-art nonradioligand-binding assay. Additionally, the potential for D4 and D5 to act as functional PR ligands (agonists) was evaluated using an in vitro cell-based reporter gene assay. The receptor-binding assays utilized calf uterine PR and recombinant human PR-α and -β. The activation of recombinant human PR-β was evaluated in the reporter gene assay.
Receptor binding was not apparent in any of the receptor-binding experiments involving either D4 or D5. This finding suggests that D4 and D5 are not PR ligands. Both D4 and D5 did not induce reporter gene expression in the cell-based reporter gene assay, suggesting that they are not functionally active PR ligands. No evidence of PR binding/activation was apparent for D4 or D5 in the series of in vitro studies presented in this report. 90
Jean 91 investigated the potential for D4 and D5 to act as functional pregnane X receptor (PXR) ligands using the in vitro PXR reporter gene assay. The assay utilized HepG2 cells stably transfected with human PXR and a PXR-luciferase reporter gene construct. Ethanol was used as the carrier and a wide range of concentrations was evaluated (5, 15, 31, 62, 125, 250, 500, and 1000 μmol/L D4 and D5).
Statistically significant increases in reporter gene expression were observed for both D4 and D5 at nominal concentrations of ≥ 31 and ≥ 5 μmol/L, respectively. Maximal induction of gene expression was observed at concentrations of 125 μmol/L (D4) and 250 μmol/L (D5). The response to nominal concentrations below the concentration that yielded maximal expression was generally dose related.
The results of this study indicate that D4 and D5 are functional ligands for human PXR. The authors noted that this finding is consistent with reports in the literature relating to the induction of CYP3A1/2 by D4 and D5 in the rat. 91
Quinn et al 80 conducted a study to determine the potential estrogenic, androgenic, and progestagenic activity of D4 and D5. Receptor-binding experiments and a luciferase reporter gene assay were used to determine whether D4 and D5 can bind and activate the ERs or PRs, α or β. Both the in vivo rat uterotrophic assay for estrogenic activity and the Hershberger assay for androgenic activity were also performed. In the receptor-binding experiments, the binding of D4 to ER-α, but not ER-β, was demonstrated; D5 did not bind to either of the 2 receptors. In the luciferase reporter gene assay, D4 (at 10 μmol/L) activated the receptor gene; results for D5 were negative. Neither D4 nor D5 was a ligand for the PRs.
The uterotrophic assay was conducted using OVEX adult Sprague-Dawley and Fischer-344 rats. Groups of 10 rats were exposed to D4 (700 ppm) or D5 (160 ppm) via whole-body inhalation exposure for 3 days (16 h/d). For comparative purposes (estrogenic activity), additional groups of rats were injected SC with EE (at doses of 0.3, 1.0, and 3.0 μg/kg per d, groups of 6 rats) and genistein (10, 25, and 50 mg/kg per d), followed by control inhalation exposure of filtered air.
In the Hershberger assay, castrated male Fischer-344 rats were exposed (groups of 10, whole-body inhalation) to 700 ppm D4 or 160 ppm D5 for 10 consecutive days (16 h/d). For comparative purposes (androgenic activity), a dose response for testosterone propionate (0.1 to 1.6 mg/kg per d) was utilized.
In the uterotrophic assay, D4 induced a small but significant (P = .0002) increase in both wet and blotted uterine weight, as well as increases in both luminal and glandular epithelial cell height in both Sprague-Dawley and Fischer-344 rats. Results for D5 were negative in both strains, which means that D5 does not have estrogenic activity. Significant antiestrogenic activity was not associated with D4 or D5. Results were negative for D4 and D5 in the Hershberger assay, which means that no significant androgenic activity was associated with D4 or D5. The results of this study indicate that D4 has a low affinity for ER-α in vitro, and a weakly estrogenic response in vivo. 80 It is important to note that a similar uterotrophic effect of D4 was reported in a study by McKim et al, 79 summarized in Hormonal Effects reported earlier in the report text.
Immunological Effects
Cyclotetrasiloxane (D4)
Munson 92 conducted studies to determine the potential effects of D4 on the immune system, using male and female Fischer-344 rats (6-8 weeks old; weights: 120-150 g). The vehicle and experimental groups consisted of 10 rats and the positive control groups consisted of 6 rats. Groups of 10 rats were dosed orally (by gavage) with 10, 30, 100, and 300 mg/kg D4 (in corn oil), respectively, daily for 28 days. The negative control group was dosed with corn oil (0.05 mL/10 g body weight). The positive control group was dosed with cyclophosphamide (25 mg/kg, IP) on days 25 through 28. Another positive control (Anti Asialo GM1 [rabbit] antibody), for the natural killer (NK) cell assay, was injected IV at a dose of 0.05 mL/10 g body weight. Maleic vinyl ether served as the positive control for modulation of the functional activity of the reticuloendothelial system; it was injected IV at a dose of 0.05 mL/10 g body weight. Blood for hematological studies and serum for antibody titers were collected by cardiac puncture.
The rats dosed with D4 had a normal increase in body weight, with body weight change being equivalent to the vehicle control group. A slight, but dose-dependent decrease in the erythroid elements was noted in male and female rats. Increased liver weights and decreased thymus weights, both as a function of the D4 dose, were reported. These changes in organ weights were more pronounced in females.
Humoral immunity (measured by immunoglobulin M [IgM] responses to the T-dependent antigen sheep erythrocytes [sRBC]) was not altered by D4 exposure in male rats (measured by the hemolytic antibody plaque assay, also known as the splenic antibody-forming cell [AFC] assay, and serum antibody titers). In female rats, a dose-dependent increase in the IgM response to sRBC was observed when assessed by the AFC response but not with the antibody titers.
No changes in cell-mediated immunity, as measured by the 1-way mixed lymphocyte response, were noted. Additionally, there were no changes in NK cell activity. Slight D4-associated changes in macrophage function were noted. Macrophage function was measured by the vascular clearance and phagocytic uptake of 51Cr sRBC into the liver, spleen, and lungs. These changes are considered related to liver size and, thus, liver blood volume associated with D4. The phenotyping of lymphocytes derived from the blood and spleen indicated no biologically significant alterations that were associated with D4 exposure.
In separate studies, D4 was administered in 4 different vehicles. Liver weight served as an indicator of systemic exposure. The greatest increase in liver weight was induced by D4 administered in corn oil, followed by 10% emulphor and Maalox. An increase in liver weight was not observed when D4 was administered in a PDMS vehicle.
Overall, the results of these studies indicate that D4, when administered in corn oil at doses between 10 and 300 mg/kg, did not induce immune suppression in male or female Fischer-344 rats. 92
Klykken et al 18 conducted a short-term inhalation toxicity study using groups of 25 Fischer-344 rats per sex, and the results are summarized in the Short-Term Inhalation Toxicity section reported later in the report text. Both test and control rats were exposed for 4 weeks (6 h/d, weekends excluded) to target concentrations of 0, 7, 20, 60, 180, and 540 ppm D4. In this study, 10 rats/sex/group were used for the immunotoxicology analyses. These animals were also exposed each of the 4 days to sRBC immunization and spleen harvest. Because a weekend was involved, these animals received 2 additional D4 exposures. The animals were immunized with sRBC 96 hours before the animals were killed. Four additional rats of each sex were assigned to the cyclophosphamide immunosuppressive control group for the AFC assay and enzyme-linked immunosorbent assay (ELISA) determinations. Injections (IP, 5 mL) of 5 mg/mL cyclophosphamide per kg body weight were administered on days 25 to 28, in addition to room air exposure and sRBC immunization.
When the data were analyzed as AFC/spleen or when normalized as AFC/106 splenocytes, there were no differences in immune competence between the D4 exposed animals and room air controls. ELISA determinations were consistent with these in vitro splenocyte data, in that they were also unremarkable. No alterations in circulating anti-sRBC IgM antibodies were noted between the control and test groups. It was noted that the room air control animals were within historical control values for these 2 determinations and that CYP induced marked immunosuppression in both the ELISA (88%) and AFC (99%) assays.
It was concluded that, under the experimental conditions of this study, D4 did not cause alterations in immune system function up to the highest level of exposure, 540 ppm. 18
Cyclotetrasiloxane (D4) and Cyclopentasiloxane (D5)
Looney et al 93 studied the immunological effects of D4 and D5 in vitro using cultured human peripheral blood mononuclear cells (PBMCs). In a serum-free medium, D4 and D5 inhibited phytohemagluttinin A (PHA)-induced proliferation of PBMCs at concentrations greater than 10 μmol/L. Also, in cultures with serum-free medium, D4 and D5 inhibited the proliferation of PBMCs that was induced by tetanus toxoid or alloantigens. The inhibitory effect of both D4 and D5 was completely reversed by the addition of small amounts of serum or plasma to the serum-free medium. The components of serum that were responsible for reversing the effects were present in the lipoprotein fractions of the serum, namely, very-low-density lipoprotein, low-density lipoprotein, and high-density lipoprotein. The inhibition of proliferation in serum-free medium seems to be due to a toxic effect of D4 and D5, in that the viability of PBMCs assessed by trypan blue exclusion was less than 20% when cultured with D4 or D5 (10 μmol/L) for 24 hours in a serum-free medium. Culturing PBMCs and D4 or D5 with or without serum was not associated with the production of tumor necrosis factor-α (TNF-α).
These findings suggest that the exposure of cells to high levels of D4 or D5 may be deleterious under conditions in which other lipophyllic substances are not present. However, the toxic effects of D4 and D5 described are unlikely to be relevant systematically, in that high levels of lipids in the plasma and tissues would neutralize these potential effects. 93
Cyclopentasiloxane (D5)
Burns-Naas et al 94 assessed the potential toxicity and effect on humoral immunity following short-term (28 days, 6h/d), whole-body inhalation exposure to D5 in male and female Fischer-344 rats (groups of 25 males and females; 4-5 weeks old). Results are summarized in Short-Term Inhalation Toxicity section later in the report text. In this study, immunotoxicology-designated rats (10 rats/sex/group) were exposed daily (28 days) to D5 concentrations of 0 (control - room air), 10, 25, 75, or 160 ppm. On day 25 of exposure, the rats were immunized IV with sRBC. Cyclophosphamide was injected IP into positive controls on days 24 through 28. The anti-sRBC AFC response (humoral immune response) was evaluated on day 29 using a standard hemolytic plaque assay. Blood samples were also collected for evaluation in the anti-sRBC ELISA.
The AFC responses of the D5-exposed rats and room air controls were essentially the same. These data were not different when expressed as AFC/spleen, in that there were no significant exposure-related changes in spleen size or cellularity. It is also important to note that the antibody response to sRBC, as measured in the spleen, was consistent with the results obtained from sera using an anti-sRBC ELISA. It was concluded that D5 did not modulate humoral immunity. However, cyclophosphamide (positive control) induced the expected suppression of the AFC response. 94
Cytotoxicity
Cyclotetrasiloxane (D4)
Felix et al 95 compared the cytotoxic effects of the following 3 siloxanes in vitro: Cyclotetrasiloxane (D4), decamethyltetrasiloxane (M10TS), and 1,3,5,7-tetravinyltetramethyl–cyclotetrasiloxane (TVTM-D4). D4 and M10TS are methyl substituted siloxanes, and TVTM-D4 is a vinyl substituted siloxane. Because the siloxanes tested are very hydrophobic, inclusion complexes of γ-cyclodextrin with each were prepared. The action of the 3 siloxanes on B-cell lymphoma lines, peritoneal macrophages, and P388D1 cells were investigated. XRPC24 and 2-11 plasmacytoma, CH12.LX lymphoma, and P388D1 macrophage-like cells were used as indicator cell lines.
The MTT proliferation test, a colorimetric method to determine the number of viable cells, was used to measure toxicity in XRPC24 and CH12.LX cells. This test is based on the reduction of a tetrazolium salt to a formazan product by active mitochondrial enzymes. The 50% minimal lethal doses (CD50) for the siloxane compounds were dependent on the cell line that was used and ranged from 30 to 50 μmol/L. D4 was less toxic than M10TS and produced a similar dose–response curve for all 3 cell lines; TVTM-D4 was much more potent than either of the 2.
During treatment with the siloxanes, the P388D1 cells were observed under the microscope, and changes in the membrane structure and shape of these cells were observed at 4 hours after exposure to the highest siloxane concentration. The P388D1 cells were said to have been approximately 3 times less sensitive to the 3 test compounds when compared to the B-lymphocyte-derived tumor cells. Sublethal doses (≤15 μmol/L) of the test compounds resulted in a striking loss of lactate dehydrogenase (LDH) from XRPC24 cells and a slight decrease in intracellular glutathione (GSH) from the cytosolic compartment of CH12.LX, 2-11, and XRPC24 cells, indicating cytotoxicity. Lactate dehydrogenase, a cytosolic enzyme, can only be released after permeabilization or disruption of the cell membrane. Cytoplasmic depletion of GSH results in the shift of the oxidative balance of the cell to the prooxidative state, which, in turn, is followed by cellular damage in the form of lipid peroxidation.
The treatment of murine macrophages with siloxanes resulted in a higher production of interleukin 6 (IL-6) than was exhibited by untreated macrophages. The B9 cell bioassay of the treated cells indicated as much as a 10-fold higher production (500 U/mL) of IL-6, compared to untreated cells. The extent of the increase was dependent upon the test substance and test concentration. Over the range of concentrations tested (6-90 μmol/L D4), the amounts of IL-6 secreted ranged from 28 to 246 B9 units/mL; that is, a dose response was observed.
The results of this study indicate that siloxanes–gamma-cyclodextrin complexes in the 25 to 50 μmol/L range are toxic to B-lymphoma and plasmacytoma cells such as CH12.LX, XRPC24, and 2-11, as well as macrophage-like P388D1 cells. 95
Effect on Cell Proliferation
Cyclotetrasiloxane (D4)
Siddiqui 51 performed cell replication assays using male and female rats (10 rats/sex/group) exposed to 700 ppm D4 for 3 or 5 days for 6 h/d. An additional group of rats was similarly exposed for 5 days and then allowed to recover for 14 days prior to being killed. All animals were administered 5-bromo-2′-deoxyuridine (BrdU) via an osmotic pump. BrdU-labeled cells, mitotic cell, and apoptotic cell counts were performed (2000 cells counted). Cell proliferation (measured by BrdU labeling) or the number of mitotic or apoptotic cells in female rats was increased, compared to controls, but the effect disappeared when exposure was discontinued for 14 days. In male rats, D4 exposure increased hepatic cell proliferation, as measured using BrdU labeling but not with mitotic or apoptotic cell counts.
Animal Toxicology Data on Cyclomethicone
Acute Inhalation Toxicity
Dow Corning 96 performed an acute inhalation toxicity study using albino rats. The 10 rats (5 males and 5 females) were exposed to a 90-mg/L aerosol burst of 10% cyclomethicone (average n = 4) every 0.5 hours for 7.5 hours. Physiological and pathological examinations were performed and no adverse effects were found.
In an acute inhalation toxicity study by Dow Corning, 31 rats were exposed to 2 atmospheric saturations of cyclomethicone (average n = 4) for 7 hours. In the 3 rats exposed to an approximate atmospheric saturation of 200 ppm (from a 23°C test material bath temperature), very slight initial weight loss and moderate liver pathology were noted. Three rats exposed to an approximate atomospheric saturation of 1000 ppm (from a 100°C bath temperature) also had slight initial weight loss. Fog was generated in the 1000 ppm setup. The authors concluded that the material should present no problem from a single exposure to vapor at room temperature or when heated (100°C).
Acute Oral Toxicity
In an acute oral toxicity study involving rats, 31 10% cyclomethicone (average n = 4) in corn oil was fed in 1.0 and 2.0 g/kg doses. In both cases, each group of 2 rats had very slight initial weight loss and slight-to-moderate liver pathology. No deaths occurred.
Skin Irritation
In a study by Dow Corning, 31 undiluted cyclomethicone (average n = 4) was applied to intact skin of the ear and abdomen of the rabbit. Little or no response was observed following 10 applications. Three applications of the ingredient to abraded skin of the abdomen produced slight, transitory irritation.
Genotoxicity
Dow Corning 97 evaluated the mutagenicity of cyclomethicone (n = 4; n = 5) in Ames assays, both with and without metabolic activation, using the following Salmonella typhimurium strains: TA1535, TA1537, TA1538, TA98, and TA100. Cyclomethicone was tested at concentrations of 0.5 to 500 μg/plate. Negative results for all 5 strains were reported, and cyclomethicone was considered nonmutagenic under the conditions of this test.
Clinical Assessment of Safety
Skin Irritation
Nichols et al 98 conducted a study to examine the effects of different ingredients that are present in sunscreens on facial cutaneous irritancy in patients with rosacea. Four different sunscreen preparations were tested. The patients (number not stated) were instructed not to wear any makeup or skin products on the day of testing. Each patient received 2 vials of sunscreen and was instructed to apply 1 on each side of the face. The patients were asked to score stinging at 1 minute postapplication according to the following scale: 0 (no symptoms) to 4 (intolerable discomfort, requiring removal of the lotion from the face). Study results indicated that the removal of dimethicone and cyclomethicone from the sunscreen lotion resulted in a significantly more irritating preparation. The authors noted that sunscreens that contain protective ingredients such as dimethicone and cyclomethicone may prevent irritation in patients with inflammatory conditions such as rosacea.
Case Report
In a case report by Schianchi et al, 99 an itchy skin eruption was observed on the face, neck, and hand of a 17-year-old patient at 3 days after using a lipophilic gel. Patch test results for the gel were positive (++) on days 2 and 3. However, patch test results were negative for the individual ingredients (1% in petrolatum) of the gel, including cyclomethicone.
Animal Toxicology Data on Cyclotrisiloxane (D3)
Short-Term Oral Toxicity
Crofoot et al 100 performed a 28-day oral exposure study on D3 using groups of 12 Sprague-Dawley rats (6 males and 6 females per group). Mean body weights for test animals were 279 g (males) and 226 g (females). The 2 control groups (males: 282 g; females: 226 g and males: 277 g; females: 229 g) received distilled water and sesame oil, respectively. The animals (test and controls) received doses of 1000 or 1500 mg/kg body weight per day for 28 days and were observed for signs of local or systemic toxicity, general appearance, behavioral abnormalities, and mortality. No treatment-related deaths, overt signs of toxicity, or changes in behavior were observed in any of the groups.
A statistical comparison of the mean body weight and feed consumption data showed no treatment-related effects between test and control groups, with the exception of D3-treated males with significant decreases in these parameters. A statistical comparison of organ weight data between test and control groups indicated a significant increase in the relative and absolute liver weights of male and female rats dosed with D3. No gross pathological changes were observed in any of the organs or tissues of male and female rats in test or control groups. It was concluded that oral administration of D3 may produce statistically significant increases in liver weight. 100
Reproductive and Developmental Toxicity
The following summary of a reproductive/developmental toxicity study on D3 was provided by SEHSC. 101 A combined repeated-exposure toxicity study of D3 that included reproductive/developmental toxicity screening in Sprague-Dawley rats by whole-body vapor inhalation was conducted at exposure levels of 0 (control), 100, 500, and 2500 ppm (approximately 0, 0.91, 4.5, and 22.7 mg/L). Rats were exposed to the test atmosphere for 6 hours per day for up to 39 consecutive days. Females in each exposure level were divided into a toxicity group (10 animals per group) and a reproductive group (10 animals per group). A single group of males (10 animals per group) was used for both the toxicity and reproductive phases of the study. Males and toxicity group females were treated for 28 and 29 days, respectively. Reproductive group females were treated for 14 days prior to the mating period, during the mating period and then up to and including gestation day (GD) 19.
No adverse effects attributable to the treatment were noted in female rats for the repeat dose portion of the study; therefore, the NOAEL for systemic toxicity in female rats was 2500 ppm (22.7 mg/L). At 500 and 2500 ppm (4.5 and 22.7 mg/L), there was an increased incidence of protein droplet nephropathy was observed in the male rats, which was considered to be specific to the male rat and not associated with a human health risk. In male rats exposed to 2500 ppm (22.7 mg/L), there was a statistically significant decrease in the absolute and relative weight of the seminal vesicles. There was also slight atrophy of the seminal vesicles in 4 of the 10 animals in the 2500 ppm (22.7 mg/L) exposure group. The NOAEL for systemic toxicity in the male rats was 500 ppm (4.5 mg/L) based on decreased seminal vesicle weight and 100 ppm (0.91 mg/L) based on the protein droplet nephropathy. Within the reproductive/developmental toxicity screening test portion of this study, there was a decrease in litter size and the number of implantation sites at 2500 ppm (22.7 mg/L); therefore the NOAEL for reproductive/developmental was considered 500 ppm (4.5 mg/L). 101
Genotoxicity
Isquith et al 102 studied the genotoxicity of D3 in the in vivo cytogenetic tests using bone marrow cells from male Sprague-Dawley rats (groups of 5). The test substance was injected IP at doses of 125, 225, 300, 400, and 515 mg/kg (1 dose per group). The positive and negative controls for each experiment were cyclophosphamide (22 mg/kg) and an equivalent volume of the vehicle, paraffin oil, respectively. Colchicine was injected (1.5 mg/kg) approximately 2 hours before the animals were killed, and bone marrow samples were collected at 6, 24, and 48 hours. A minimum of 100 metaphases from each animal per group was scored for the incidence of aberrations.
D3 did not induce a significant, treatment-related increase in chromosome damage. More specifically, the test substance did not induce statistically significant increases in the proportion of cells with chromosome damage. The predominant forms of chromosome damage were simple chromatid gaps and breaks, and the incidences of aberrations were within the expected ranges of variation for rat bone marrow cells noted in previous tests performed at the testing laboratory used in this study. It was concluded that D3 lacks significant clastogenic potential. 102
Isquith et al 102 studied the in vitro genotoxicity of D3 using the following assays: plate incorporation assay by Ames et al (1975; S typhimurium strains TA1535, TA 1537, TA1538, TA98, and TA100), mitotic gene conversion assay (Saccharomyces cerevisiae strain D4) by Brusick and Andrews, 103 , the Escherichia coli pol A-repair assay for DNA damage (E coli strain W3110 (pol A+) by Slater et al 104 ), and the mouse lymphoma assays (L5178Y mouse lymphoma cells), according to the method of Clive and Spector 105 ). The following were evaluated in the mouse lymphoma tissue culture assays: forward gene mutation, sister chromatid exchange (SCE), DNA damage, and chromosome aberration potential. All of the assays were performed with and without metabolic activation.
In the plate incorporation, mitotic gene conversion, and DNA damage assays, D3 was tested at doses of 0.1 to 500 μg/plate. In each of the 3 assays, there was no evidence of increases in the incidence of reverse mutation, mitotic recombination, or DNA damage, respectively, either with or without metabolic activation. Ethanol and hydrochloric acid (control agents; potential organosilane hydrolysis products), also produced no significant end point increases in the respective microbial assays.
The following doses of D3 were tested in the mouse lymphoma assays (L5178Y mouse lymphoma cells): forward mutation assay (0.067-1.060 mg/mL); SCE assay (0.0975-3.129 μg/mL), and chromosome aberrations assay (0.0975-3.120 μg/mL). In the forward gene mutation assay, D3 did not produce a positive response, as measured by a dose dependent, reproducible highest relative mutation frequency (HRMF) of 2.5 or greater. Absolute ethanol (solvent control, 10 μL/mL) did not induce a significant increase in mutant colonies, compared to values for tissue culture medium that was used as the negative control. Hydrochloric acid also did not induce any significant mutagenic activity at the maximum dose level of 5 μL/mL. The positive control chemicals, ethylmethane sulfonate (EMS) and dimethylnitrosamine, were mutagenic.
In the SCE assay, D3 was considered genotoxic, and a dose response was noted with and without metabolic activation. In the chromosome aberrations assay, aberrations in concurrent solvent controls (10 μL/mL ethanol) were noted in 0% to 4% of the cells, with 0% to 2% having 2 or more aberrations. Chemicals that caused aberrations in 8% or more of the cells were classified as having clastogenic (chromosome damaging) potential. Ethylmethane sulfonate (positive control) produced a range of 22% to 40% aberrant cells; 0% to 6% of the cells had 2 or more aberrations. Dimethylnitrosamine (positive control) produced 13% to 38% aberrant cells; 0% to 20% of the cells had 2 or more aberrations. D3 was genotoxic (aberrations in 8% or more of the cells) with, but not without, metabolic activation in the chromosome aberrations assay. D3 did produce a positive response in the DNA/alkaline elution assay; however, the positive control compounds, methylmethane sulfonate and benzo[a]pyrene, induced significant damage (Isquith et al). 102
Animal Toxicology Data on Cyclotetrasiloxane (D4)
Acute Inhalation Toxicity
An acute inhalation toxicity study was conducted by Research and Consulting Company Ltd (RCC) 106 using F-344 rats. Fifteen male and 15 female animals were acclimatized over 5 days, during which feed consumption was measured once to establish a baseline of feed consumption. The feed was a standard rat maintenance diet, provided ad libitum, as was water.
Animals were randomly assigned (5 males and 5 females) to each of 3 exposure groups. The composition of the test material was given as 96% D4, 4% cyclopentasiloxane (D5), and 0.1% cyclotrisiloxane (D3). Exposure concentrations were 20.12, 30.03, and 54.37 mg/L of air for the low-, medium-, and high-exposure groups, respectively, for 4 hours in a nose-only, flow-past exposure system. The particle size of the test material was not determined. Food consumption was measured, in those animals that survived, during 4 postexposure intervals: 1 to 6 days; 6 to 9 days, 9 to 12 days, and 12 to 15 days.
Feed consumption, compared with baseline data, was decreased in all surviving animals (all exposure groups) at 1 to 6 days; feed consumption/body-weight ratios were also decreased. Feed consumption increased in all animals at postexposure times longer than 6 days, as did feed consumption/body-weight ratios, which returned to baseline levels.
None of the animals in the low-exposure group died. One male and 2 females in the medium-exposure group died, and 4 males and all females in the high-exposure group died. Clinical signs of restlessness/excitement were noted in all animals that died. Hunched posture, stiff gait, and ruffled fur were noted postexposure in all groups, but all these signs disappeared after 6 days. Organ weights were determined, but there was no control group with which to compare the findings. The lungs of all the animals that died had a dark red discoloration. The authors estimated the LC50 exposure to be 36 mg/L. 106
Acute Intraperitoneal Toxicity
In a study by Lieberman et al, 29 the acute IP toxicity of D4 was evaluated using female CD-1 mice (5-12 mice; weights = 25-30 g). The IP doses of D4 ranged from 3.5 to 35 g/kg. Control mice were dosed with 1 mL of soy oil (dose = 32 g/kg). The mice were observed for 14 days; moribund and surviving animals were killed. For histopathologic and biochemical studies, separate groups of mice (5-6 per group) were injected with D4 and killed at day 4 or day 14. Concentrations of the following enzymes were determined in blood samples: AST, alanine aminotransferase (ALT), and LDH. For the studies involving hydroxyl radical measurement (as a marker of free radical activity in the liver and lung), separate groups of mice (4 per group) were injected with D4 (8.75 g/kg) or soy oil. The animals were killed at day 1 or day 4 postinjection to determine the extent of 2,3-dihydroxybenzoate formation in the liver and lung tissues.
None of the mice receiving the 3.5 g/kg dose died; however, all 6 mice dosed with 35 g/kg died. An LD50 of 6 to 7 g/kg was reported. Histopathologic examination, at day 14, of mice that survived (3.5 and 8.75 g/kg dose groups) revealed lung, liver, and peritoneal changes. An evaluation of acute tissue injury and serum enzyme levels following D4 injection in a separate experiment revealed severe pulmonary and hepatic lesions at day 4. The severity of the lesions observed was dose related. Large increases in serum enzyme activities were also reported. At day 4 postdosing with 8.75 g/kg D4, results were as follows: ALT (15-fold 8), AST (6-fold 8), and LDH (4-fold 8). Doses as low as 3.5 g/kg caused statistically significant increases in serum enzyme activities. The IP injection of soy oil resulted in acute inflammation (mild, focal) on the peritoneal surface of the liver and adjacent superficial parenchyma.
As a marker of free radical activity, hydroxyl radical formation in liver and lung tissues of control (soy oil-injected) mice and mice injected IP with D4 (dose = 8.75 g/kg) was measured. By day 1, there was no increase in hydroxyl radical formation in the liver or lung. A dramatic increase in hydroxyl radical formation in the liver (20-fold increase) and lung (7-fold increase) was observed by day 4.
Study results indicate that D4 produced significant injury to tissues, as documented by histopathologic changes in the liver and lung and increased serum enzyme activities. Dramatic increases in hydroxyl radical formation in the liver and lungs of mice injected with D4 were also noted. 29
Short-Term Inhalation Toxicity
Kolesar and Hobbs 107 performed a 14-day range-finding vapor inhalation toxicity study with D4 in the rat. Male and female Sprague-Dawley rats (∼175 to 250 g) were placed into 1 of 3 treatment groups or a control group (5 males and 5 females in each group). Animals were exposed to 0, 100, 200, and 400 ppm D4 for 7 days/week (6 h/d) in inhalation chambers. Body weights and feed consumption were measured every 4 days. Each animal was observed during treatment each day for any signs of toxicity.
No signs of toxicity were observed during the study. There were no differences in body weights between control and treatment groups. During the first week of the study, females in the 400 ppm exposure group had significantly reduced food consumption; loss of this effect was noted during week 2. At the end of the study, there was no statistically significant decrease in feed consumption in this group. 107
Kolesar et al 108 conducted a 14-day inhalation toxicity study of D4 using rats. Adult and young Sprague-Dawley rats (5 males and 5 females per group) were exposed to D4 at 854 ppm or to room air (control) in inhalation chambers using the same schedule described above. The authors noted that this exposure approaches the saturation limit for D4, which is 1000 ppm at 20°C and 76 mm Hg. Body weights and feed consumption were determined at the onset of the study and on every fourth day during the study and for a week after exposures ended.
Young male rats exposed to D4 had significantly decreased body weights (compared to controls) at days 4, 8, and 12 during treatment and at day 16 during the posttreatment period but not at day 20. Adult female rats exposed to D4 had significantly decreased body weights from day 4 through day 20. No other significantly body weight differences were reported.
Young male rats exposed to D4 also consumed significantly less feed (compared to controls) in the 0- to 4-day, 4- to 8-day, and 8- to 12-day ranges but not in the 12- to 16-day or 16- to 20-day ranges. Young and adult female rats exposed to D4 consumed significantly less feed (compared to controls) in the 0- to 40day, 4- to 8-day, and 8- to 12-day ranges but not in the 12- to 16-day or 16- to 20-day ranges. No other differences between treatment and control animals were found. The authors stated that the biological significance of the findings could not be determined based on these findings. 108
Siddiqui 109 performed a 28-day inhalation toxicity study of D4 using hamsters, guinea pigs, rabbits, and mice. Adult male and female Golden Syrian hamsters (10/sex/ group), Hartley guinea pigs (10/sex/group), New Zealand White rabbits (5/sex/group), and CD-1 Swiss mice (10/sex/group) were exposed to 0 or 697 mg/L D4 7 d/week (6 h/d) for 28 days. Body weights and feed consumption were measured at the onset, every 4 days thereafter, and before necropsy, except that rabbit feed consumption was measured every 2 days during treatment.
No mortality was observed in either control or treatment groups in any species. No clinical signs, including distress, were noted. Female rabbits had a statistically significant reduction in weight gain in the treatment group, compared to controls. The authors noted a general depression of male weight gain across species in the treatment groups, but this was not significant. While there were some feed consumption values that were lower in the treatment group, compared to control, statistical significance was not consistently observed.
Relative liver weights in treated female hamsters were significantly increased. Male and female mice exhibited the same finding. No difference in organ weights in guinea pigs or rabbits was found. The authors attributed the effect on liver weights in hamsters and mice to the effects of D4. 109
Research and Consulting Company Ltd 110 conducted a 1-month repeated dose inhalation toxicity study of D4 using rats. Male (weights: 162-198 g) and female (weights: 144-176 g) Fisher-344 albino rats (10 males and 10 females per group) were assigned as either controls (room air) or were exposed to D4 at target exposures of 2.5 (actual: 2.78 ± 0.52), 5 (actual: 5.13 ± 0.64), and 9 (actual: 8.62 ± 1.66) mg/L for 20 days (males) or 21 days (females). All exposures were carried out 5 d/week (6 h/d) using a flow-past, nose-only exposure system.
An additional group of 20 animals was exposed to D4 at a target exposure of 16 mg/L (actual: 14.21) for days 1 to 5, followed by 12 mg/L (actual: 13.25 ± 2.12) for days 6 to 29. The authors noted that the D4 vapor concentration at saturation is approximately 13 mg/L, suggesting the presence of D4 in liquid aerosol form in the last exposure group. The composition of the test material was given as 96% D4, 4% D5, and 0.1% D3.
Animals were observed for morbidity and mortality twice daily, except once per day for 2 days. An ophthalmoscopic examination was done prior to exposure and after exposure for all surviving animals. Body weights were determined before exposure (2×), daily during the first week, and 3× per week for the remainder of the study. Feed consumption was determined for a 4-day period twice before exposure, daily during the first exposure week, then for 2-day or 3-day periods for the remainder of the study. Animals were fasted for 18 hours (water, ad libitum) and blood and urine were collected prior to necropsy.
The odd dosing pattern for the high-dose group resulted from the deaths of 3 females in this group after the first and fifth days of treatment. Thereafter, the dose was decreased, as described above. One male in the 9 mg/L group and 1 female in the control group were found dead on the day of necropsy; the authors offered no explanation for these 2 deaths, but suggested that treatment was unlikely the cause because the death was an isolated finding. No ophthalmoscopic findings were reported.
Body weights and weight gains were generally decreased in the high-dose group, compared to lower-dose groups and controls, often reaching statistical significance when compared to controls. Feed consumption and consumption/body-weight ratios were decreased on days 1 to 9 in the high-dose group, compared to controls, but the males increased their feed intake to a level comparable to that of the lower-dose groups and the control, and female rats increased their food intake beyond these levels on days 16 to 23.
Liver weights increased in treated animals in a statistically significant, dose-dependent manner. Thymus and adrenal gland weights were significantly increased in males and females in the high-dose group and in females in the 9 mg/L group. No other significant organ weight differences were found.
Histological examination of the lungs revealed changes that were consistent with respiratory tract irritation. In the 2 highest dose groups, slight hepatocellular hypertrophy and an increase in adrenal cortex vacuolation were noted. In the high-dose group, moderate thymic atrophy and vaginal mucification were noted. A decrease in the mean corpora lutea (CL) score was noted in all treated females.
Examination of liver tissue using electron microscopy revealed a dose-dependent decrease in hepatocyte mitochondria. Other liver findings were not consistent across dose groups.
Abnormal posture, gait, and ruffled fur were noted in all animals in the high-dose group, in most of the females exposed to 9 mg/L, and in 2 females exposed to 5 mg/L during weeks 1 to 3. Hematologic evaluation revealed several statistically significant changes, such as higher mean corpuscular volume (MCV) in treated animals, but the authors suggested that there was little or no toxicologic significance for these findings. Changes in blood chemistry, such as lower serum alkaline phosphatase (SAP) activity in treated animals, were considered stress related and not an effect of D4 directly. Urinalysis revealed no changes that were of toxicological significance.
The authors considered that treatment at all levels resulted in some treatment-related effect. While most organ changes were not considered of toxicological significance or were directly related to stress, the vaginal effects and reduced ovarian CL scores suggested a possible effect on the estrous cycle. 110
Klykken et al 18 conducted a study to evaluate the dose–response relationship for liver enlargement observed following inhalation exposure, and ascertain whether or not there is evidence of immunosuppression or immunopotentiation that is associated with this exposure paradigm. The study was conducted using groups of 25 Fischer-344 rats per sex. Both test and control rats were exposed for 4 weeks (6 h/d, weekends excluded) to target concentrations of 0, 7, 20, 60, 180, and 540 ppm D4. Ten rats/sex/group were killed for the toxicological assessment on day 29 after the initial exposure. An additional 5 rats/sex/group served as recovery animals and were killed 14 days after the final exposure for a similar toxicological assessment. The rats were evaluated daily, during weekdays, for signs of exposure-related effects, and a complete gross pathological examination was performed on all of the general toxicology animals. Tissues were prepared for microscopic examination. Hematological evaluations and urinalyses were also performed. Results relating to immunosuppression or immunopotentiation are included in the Immunological Effects section reported earlier in the text.
Of the Fischer-344 rats included in this study, 20% had ocular opacities; this was consistent with the genetic predisposition of Fischer rats for ocular opacities. Other clinical signs included porphyrin staining around the nose and urine staining of the fur. These were random observations and were not concentrated within any 1 exposure group. No signs of neurotoxicity, such as, stereotypical behavioral changes, changes in respiration, ruffled fur, or modifications in gait were observed in rats treated with D4. Male and female rats exposed by whole-body inhalation to D4 exhibited normal weight gain over the course of the study. Additionally, there were no differences in feed consumption between test or control groups throughout the study.
At necropsy (terminal or recovery sacrifice), there was no evidence of microscopic lesions in any of the exposure groups. Except for the liver, there were no statistically significant exposure-related changes in organ weights. For male and female exposure groups, both liver weights and liver-to-body-weight ratios were significantly increased over the respective room air control groups. Female rats were more sensitive to the exposure-induced increase in liver weight, with significant increases at 20 to 540 ppm (7.4%-30%) at terminal sacrifice. Changes in liver weight in male rats became statistically significant at exposure levels of 180 ppm and 540 ppm (16% increase). These changes in male and female rats were fully reversible, in that they were not observed in the 14-day recovery animals. In male or female rats, there were no histopathological findings that were considered treatment-related, either at the end of the 28-day exposure period or at the end of the 14-day recovery period.
Regarding serum chemistry parameters in male rats, the only statistically significant change was a 22% decrease in triglycerides. This effect appeared to have been exposure related but was not observed in recovery animals. The following significant changes were reported for female rats: SAP (14% decrease), ALT (17% increase), blood urea nitrogen (BUN, 12% decrease), globulin (14% increase), and cholesterol (29% increase) at the high-dose level. The changes in SAP, ALT, globulin, and cholesterol appeared to have been exposure related. After the 14-day nonexposure recovery period, significant decreases in SAP, triglycerides, and glucose were noted.
No differences in urinalysis parameters or in urinary sediment microscopic analysis were observed between D4-exposed and room air control animals. The authors noted that there was little indication that D4 exposure resulted in any biologically significant changes in hematological parameters. Statistical trends toward an exposure-related effect of D4 on the percentage of neutrophils and lymphocytes, the MCV, and the total number of platelets were noted. However, statistically significant data were observed, only at the highest concentration, for both the lymphocyte percentage (8% decrease) and MCV (2% increase). There were no exposure-related hematological trends, although a statistically significant decrease in mean corpuscular hemoglobin content (MCHC) was noted for the following exposure groups: 7 ppm (2% decrease), 180 ppm (2% decrease), and 540 ppm (2% decrease). In recovery animals, the only significant hematological finding was a 7% decrease in the total number of red blood cells in female rats. All of the other hematological parameters in male and female rats were equivalent to room air controls.
The results of this study indicate that D4 exposure did not cause adverse effects on body weight, feed consumption, or urinalysis parameters, and that there were no exposure-related histopathological alterations in either of the exposure groups. Additionally, a statistically significant increase in liver weights and in the liver-to-body-weight ratios was observed in both male (180-540 ppm exposures) and female (20-540 ppm exposures) rats; however, these statistically significant findings were not associated with the 14-day recovery group animals. 18
Plotzke et al studied the effects of repeated whole body inhalation exposure to D4 vapor on hepatic microsomal CYP2B1/2 induction using 11 groups of 10 female Fischer-344 rats (8-10 weeks old; weights:130-141 g). Ten groups were exposed to 0, 1, 7, 30, 70, 150, 300, 500, 700, and 900 ppm D4 vapors 6 h/d for 5 days. The positive control group received PB (0.05% w/v). The animals in 1 of the groups (group 1) were killed on the morning after the last exposure and changes in liver size and hepatic microsomal enzyme induction were evaluated. Animals from a second group (group 2) were killed immediately after the last exposure; blood, fat tissue, and livers were collected for the analysis of total D4 content.
For group 1 rats at each exposure concentration, liver-to-body-weight ratios, CYP2B1/2 activity (as determined by 7-pentoxyresorufin-O-dealkylation [PROD]), and immunoreactive CYP2B1/2 protein were measured. Liver size (expressed as a liver-to-body-weight ratio relative to controls) increased with increasing exposure concentrations; a maximum response of 24% over the control value was noted at 900 ppm D4, with no apparent change in slope (plateau). A dose-related increase in the induction of PROD activity (maximum response between 300 and 500 ppm) was also noted. Changes in the absolute amount of CYPB2B1/2 protein conformed to a pattern that was similar to that observed for PROD activity. These data demonstrate that the observed increase in PROD activity was associated with a concomitant increase in CYP2B1/2 protein levels.
Though the maximum increase in PROD activity (1355.9 pmol/min per mg) occurred between 300 and 500 ppm D4, a slight, but statistically significant, dose-related decrease in PROD activity (between 500 and 900 ppm D4) was noted. These data suggest that maximum induction of CYP2B1/2 by D4 was achieved and that hepatic concentrations of D4 may have reached levels that were capable of saturating CYP2B1/2 activity.
Total D4 in the plasma, fat tissue, and liver increased with increasing exposure concentration. Overall, the increase in D4 concentration in each tissue increased with increasing exposure concentrations, with no indication of a plateau under test conditions. At the highest exposure concentration (900 ppm), D4 content was greatest in fat tissue (1115.0 μg/g), followed by the liver (179.9 μg/g) and plasma (10.9 μg/g). The lowest concentration of D4 that caused significant liver enlargement and CYP2B1/2 induction were 150 ppm and 7 ppm, respectively. These data demonstrate that the induction of hepatic CYP2B1/2 enzymes is an early and sensitive biochemical response to D4 exposure in the rat and that a maximum response can be achieved following 5 days of repeated inhalation exposure to 500 ppm D4.
Whether or not exposure to D4 induces regional expression of CYP2B enzymes was also investigated in this study. CYP2B expression was evaluated immunohistochemically. Liver sections from the control group, all D4 exposure groups, and the PB-positive control group were stained for CYP2B protein, randomized, and qualitatively evaluated using light microscopy. Very little staining in the centrilobular region was noted in livers from negative control rats. With an increase in the exposure concentrations of D4, there was an increase in the intensity of CYP2B staining. At an exposure concentration of 70 ppm D4, the expression intensity was described as moderate, with centrilobular to midzonal expression. Extensive CYP2B staining with expression across all regions was noted in 700 and 900 ppm exposure groups. These data indicate that the expression of CYP2B following exposure to low levels of D4 is confined to the centrilobular regions and expands across the hepatic lobule as exposure concentrations increase. The findings in this study provide evidence that D4 is a PB-like inducer of rat hepatic CYP enzymes (Plotze et al 1999).
McKim et al 111 evaluated the effects of repeated inhalation exposure to D4 vapors on hepatic and thyroid function and hypertrophy (in relation to exposure time and concentration) using groups of female Fischer-344 rats (weight = 145-160 g). In the first experiment, a group of 10 rats was exposed (whole-body inhalation) to 700 ppm (6 h/d; 5 d/week) over a 4-week period, and the control group received control air only. The incorporation of BrdU and the abundance of proliferating cell nuclear antigen were used as indicators of cell proliferation. The positive control group of 10 rats received 0.05% PB in drinking water over the 4-week period. Phenobarbital produces liver enlargement characteristics that are characterized by transient hyperplasia and sustained hypertrophy. Designated animals from each treatment group were killed on days 6, 13, and 27.
In the second experiment (groups of 10 rats), the effect of exposure to the following concentrations of D4 on hepatic cell proliferation was evaluated: 0, 7, 30, 70, 150, 300, or 700 ppm. Again, the positive control group received 0.05% PB in drinking water over a 4-week period. In the 700 ppm exposure group, liver-to-body-weight ratios increased by 18%, 20%, and 22% on days 6, 13, and 27, respectively, when compared to controls. Animals treated with PB had increases in liver-to-body-weight ratios of 33%, 27%, and 27% on days 6, 13, and 27, respectively. The hepatic incorporation of BrdU after exposure to D4 was highest on day 6 (labeling index = 15%-22%) and was either at or below control values by day 27. The authors noted that this pattern of hyperplasia was observed in all hepatic lobes that were examined and was similar to the pattern that was observed after treatment with PB. 111
Siddiqui 51 noted that previous studies have demonstrated liver weight changes in male and female rats and mice and female hamsters, but not in rabbits or guinea pigs, and conducted a study to further characterize the species differences in liver response to D4 treatment by studying the following: urinary metabolites in each species, induction of selected liver enzymes in rats and guinea pigs, and cell replication in rats. Groups of male and female Sprague-Dawley rats, C-1 mice, hamsters, guinea pigs, and rabbits were exposed to D4 (at 10 or 700 ppm, whole-body exposure) 5 d/week (6 h/d) for 5 weeks.
No mortality or overt signs of toxicity were observed in any of the control and treated animals. The author confirmed a statistically significant increase in liver weights in male and female hamsters, mice, and rats exposed to 700 ppm D4. No statistically significant increase in liver weights was observed in rabbits and guinea pigs exposed to 700 ppm. 51
Short-Term Oral Toxicity
Siddiqui and Stanton 112 used microencapsulated D4 in a solid diet to conduct a 28-day feeding study using rats. Young and adult male and female rats were divided into control and treatment groups, each containing 5 male and 5 female animals. The rats were fed with either control diet or diet containing 2.1% D4 for 4 weeks. Water was provided ad libitum. Body weights and feed consumption were determined at the beginning of the study and every 4 days thereafter. Gross necropsies were performed at the end of the study.
Both young and adult male and female animals that ate the diet containing D4 experienced stress, rough fur, and emaciation, but none of the animals died; they consumed significantly less food than controls. Necropsy demonstrated severely depleted fat tissue reserves in all animals consuming the diet containing D4; most had watery cecal contents. The testicular changes observed in a few male rats were attributed, possibly, to the overall weight loss. 112
In a study by Mudgett, 113 the short-term oral toxicity of D4 was evaluated using groups of 10 male Sprague-Dawley rats (weights = 250-325 g). Groups 1 and 2 received 0.5 weight percentage methylcellulose in deionized water. Groups 3 and 4 received D4 (in 0.5% methylcellulose) at doses of 1600 mg/kg per d. The animals were dosed daily (by gavage) for at least 14 days, and the dosage volume for each animal was 4.10 mL/kg body weight. During the dosing period, the animals were observed for the following: signs of toxicity, general appearance, behavioral abnormality, signs of local and systemic toxicity, and mortality. All of the animals in groups 1 and 3 were killed following 14 days of dosing. For groups 2 and 4, 2 or 3 animals were killed each day until completion of the life phase of the study.
Adverse clinical signs were not observed in any of the animals during the dosing period. There were no apparent differences in body weights between control and test animals. Terminal sacrifices, performed in order to obtain livers from all of the animals, morphometrics, and DNA assays were performed. It was noted that the results of morphometric and DNA assays would be documented in a separate report. 113 These and other results are summarized below.
Zimmer and Bejarano 114 performed morphometric and electron microscopic evaluation of hepatic changes in rats dosed orally (gavage) with D4 in the preceding study. The purpose of morphometric analysis of the liver was to determine whether increases in liver weight were due primarily to hyperplasia, hypertrophy, or a combination of the 2. The procedure involved determining the relative volume of hepatocytes in a given liver volume (volume fraction), the number of hepatocytes in a given area (NA), and using these values to calculate the absolute number of hepatocytes per given volume of liver (Nv). This value and the liver volume were used to calculate the total number of hepatocytes per liver. For electron microscopy, 1 liver lobe (left lateral) per rat was sampled. At least 5 blocks from each area (centrilobular or peripheral) were prepared.
All of the animals survived to termination of the study. One rat died during anesthesia and was not examined. There was no evidence of gross lesions in livers. The few histopathologic changes observed were not treatment related but were considered normal findings in rats of this age and strain. Compared to controls, a statistically significant increase in liver weights was noted. The comparison of Nv supported a finding of hepatocellular hyperplasia.
There were no significant differences in the number of cells per given volume of liver between treated and control rats. The treated livers had a much greater volume; however, a statistically significant increase in the total number of hepatocytes in the livers of treated rats was reported. The results of these 2 analyses indicate that D4 causes hepatocellular hyperplasia. The mean hepatocyte profile diameter in each of the 3 lobular zones in treated rats was not significantly different from that of control rats. This finding indicates that the hepatomegaly observed was due to hyperplasia. Within group cell size variances did not support a finding of hypertrophy.
The findings in this study indicate that the hepatomegaly observed in rats exposed to D4 results from hepatocellular hyperplasia, and there was no evidence to support the occurrence of hepatocellular hypertrophy. 114
Zimmer and Bejarano 115 performed a quantitative analysis of liver nuclear DNA content. The livers examined were from rats exposed to D4 in the short-term oral toxicity study by Mudgett 113 described earlier in this section. A sample from each liver was homogenized and the nuclei were isolated via gradient centrifugation. A fraction of the sample was then mixed with diphenylamine and incubated. The DNA concentration was determined spectrophotometrically. The results indicated no significant difference in DNA values between treated and control groups. This finding was said to support the conclusion that hepatomegaly due to D4 administration resulted from hepatocellular hyperplasia.
Stanton et al 116 studied the short-term oral toxicity of D4 (suspended in 0.5% methocel in distilled water) using groups of 6 nonpregnant New Zealand White rabbits (weights = between 2.26 and 2.69 kg). The test substance was administered (oral gavage) at doses of 500 and 1000 mg/kg per d (dose volume = 3 mL/kg, 7 d/week) for 14 days. Control rabbits were dosed with 0.5% methocel according to the same procedure. Gross necropsies were performed on all major organs and tissues at the end of the 14-day dosing period; the brain and liver were collected and weighed.
There were no treatment-related deaths, overt signs of toxicity, or changes in behavior during the study. Compared to controls, dosing with D4 resulted in a marked, significant reduction in body weight and feed consumption. At gross examination, a decrease in the size of the thymus gland was noted in most animals. Also, in some cases, the spleen and lymph nodes appeared smaller. Liver weights relative to body weight were significantly increased in both treatment groups. This finding was not considered treatment related, in that the absolute liver weight and the liver-to-brain-weight ratios were not significantly different from controls. Changes in the liver included an accentuated lobular pattern and areas of pallor and increased fragility in animals receiving 1000 mg/kg doses. It was concluded that the oral administration of D4 resulted in a marked decrease in feed consumption and body weights in treated rabbits. 116
Subchronic Inhalation Toxicity
Siddiqui et al 117 conducted a 90-day inhalation toxicity study of D4 using Sprague-Dawley rats. Groups of 10 male and 10 female rats were exposed to 0, 51, 301, and 700 ppm D4 vapors 7 days per week (6 h/d) for 13 weeks. An additional control group and 703 ppm exposure group were also exposed for 13 weeks and then held for an additional 28 days. Animals were observed for toxicity and mortality during exposure; feed consumption and animal weights were measured weekly throughout the study. At the end of the study, animals were killed, organs weighed, and gross and histopathological examinations were performed. Hematology, clinical chemistry, and urinalysis were also performed.
No mortality or obvious signs of toxicity were reported. A slight (not statistically significant) reduction in weight gain in females of the high-exposure group was reported through 13 weeks, but this effect was no longer observed after the 28-day recovery period. Feed consumption was significantly decreased in the high-exposure female group in the first week of the study, but the average feed consumption over the course of the study was not different for control and high-exposure females. Statistically significant increases in red blood cell counts, hemoglobin, and hematocrit in high-exposure females were seen at 13 weeks, but these decreased significantly in the high-exposure females after 28 days of recovery. Potassium and phosphorus levels were increased in high-exposure males, but these results were attributed to hemolysis. Overall, the authors stated that no differences in toxicological significance in hematology, clinical chemistry, urinalysis, gross pathology, or histopathology were found.
Significant increases in mean absolute and relative liver weights were observed in both sexes in all but the lowest exposure group of females. In high-dose males, this effect disappeared after the 28-day recovery period but was still evident in high-dose female rats after 28 days of recovery. The authors suggested that these liver weight changes were related to exposure, even though no gross pathology or histopathology changes in the liver were reported. Ovarian weights were significantly decreased in high-dose females, and the authors suggested that this may be related to the overall trend in weight gain in these animals. 117
The International Research and Development Corporation (IRDC) 118 conducted a 13-week inhalation toxicity study on D4 in rats. Male and female rats were exposed to 0, 5, 10, or 300 ppm D4 5 d/week (6 h/d) for 13 weeks; necropsy (10 males and 10 females per group) was performed at the end of the study. An additional 10 males and 10 females in each of the control and high-exposure groups were maintained for 4 weeks postexposure and then necropsied. An additional 20 males exposed to room air (controls), and 20 to 300 D4, were scheduled for necropsy at 4 weeks. Another 10 males were scheduled for necropsy at 13 weeks, but initial findings in 10 animals necropsied at 4 weeks indicated no changes in liver weight, and this arm of the study was abandoned. Thus, all animals were killed at 13 weeks and discarded.
Animals were observed for signs of toxicity during exposures. Body weights and feed consumption were measured weekly during exposure the postexposure period. Ophthalmoscopic examinations were done at the onset of the study, at 13 weeks, and after the recovery period. At necropsy, organ weights were determined, tissues evaluated, and blood and urine were analyzed.
No statistically significant differences in body weights were reported. Feed consumption was generally increased in the treatment groups, with some increases in specific time ranges statistically significant, but there was no pattern to these findings. Liver weights were increased significantly over controls in the 300 ppm group. Otherwise, no significant differences in liver weights were found. No changes in blood or urine were reported as a function of D4 exposure at any level. No pathology was reported in the liver tissue of the female rats in the 300 ppm group, or in any of the exposure groups. No differences between exposed and control animals were found during the ophthalmoscopic examinations. 118
Research and Consulting Company Ltd 119 conducted a 3-month repeated dose inhalation toxicity study on D4 using rats; a 1-month recovery period was observed. Male (weights: 173-209 g) and female (weights: 135-155 g) Fisher-344 albino rats (30 males and 30 females per group) were assigned as either controls (room air), or exposed to D4 at a target exposure of 12 (actual 10.87 ± 0.75) mg/L. Groups of 20 males and 20 females were exposed to D4 at 0.3 (actual: 0.42 ± 0.08), 1.2 (actual: 1.48 ± 0.27), and 5 (actual: 5.91 ± 1.05) mg/L. All exposures were carried out 5 d/week (6 h/d) for 13 weeks using a flow-past, nose-only exposure system. In the control and the high-exposure group, 10 animals of each sex were retained for an additional 29 days in room air.
Animals were observed for morbidity and mortality twice daily on weekdays, and mortality was determined twice daily on weekends and holidays, where feasible. An ophthalmic examination was performed prior to exposure and, on all surviving animals, after exposure. Body weights were determined before exposure, daily during the first week and 3 times per week for the remainder of the study. Feed consumption was determined for a 3- or 4-day period twice before exposure, twice weekly for the first 3 exposure weeks, and, then, once weekly for the remainder of the study. Animals were fasted for 18 hours (water, ad libitum) and blood and urine were collected prior to necropsy.
A total of 5 females in the high-exposure group died. No other mortality was reported. Clinical signs in the high-exposure group included hunched posture and a single incidence of stiff gait, but these observations were not consistently seen. The ophthalmoscopic examinations uncovered only 1 abnormal finding, unilateral anophthalmus, in a female in the high-exposure group. No other ophthalmoscopic findings were reported. Body weights and weight gains were generally decreased in the high-exposure group compared to lower-exposure groups and to controls, often reaching statistical significance when compared to controls. During the recovery period, weight gains were comparable to controls. Compared to controls, a decrease in feed consumption was reported for most data points on days 1 to 10 in the high-dose group, followed by an increase that extended into the recovery period.
Liver weights increased in treated females exposed to 1.2, 5, and 12 mg/L and in males exposed to 12 mg/L. Increased lung weights were reported for both sexes in the high-exposure group, but this effect was absent after recovery. Increased adrenal gland and decreased thymus weights were reported for females in the 5 and 12 mg/L groups, but these effects were absent after recovery in the 12 mg/L group. A decrease in ovarian weights was reported for females in the high-exposure group, but this effect was absent after the recovery period. No other significant organ weight differences were found.
Histological examination of tissues (adrenal glands, brain, heart, kidneys, liver, lungs, ovaries, spleen, testes, thymus, and vagina) obtained during necropsy revealed changes that are consistent with respiratory tract irritation. Ovarian atrophy was observed in 1 rat in the 0.3 mg/L group, 10 rats in the 12 mg/L group, and in 2 rats after the recovery period. Vaginal mucification was reported in 17 rats in the 12 mg/L group and in 1 rat after the recovery period. In the testes, minimal to massive tubular atrophy and oligospermia (epididymides) were reported only in rats exposed to 0.3 or 12 mg/L. The testes findings were also observed in negative control rats, and, thus, were considered unrelated to test substance administration.
Hematologic evaluations revealed several statistically significant changes, such as, slightly decreased erythrocyte counts and hemoglobin concentration in male animals and a higher MCV in male and female animals in the 5 and 12 mg/L dose groups. A decreased MCHC was reported for males and females in the 12 mg/L group. During the recovery phase, these effects mostly disappeared, except for a slightly decreased erythrocyte count in both sexes and a slight increase in MCV in males (recovery examined only in the high-exposure group).
Changes in blood chemistry parameters were reported, including decreased bilirubin and increased cholesterol in females exposed to 1.2, 5, or 12 mg/L; decreased triglycerides in males and females exposed to 1.2, 5, or 12 mg/L; increased ALT activity in males and females exposed to 12 mg/L; decreased creatine kinase (CK) activity in males exposed to 0.3, 5, or 12 mg/L, but not in the 1.2 mg/L group; increased γ-glutamyl transferase activity in females in the 5 mg/L group and in males and females in the 12 mg/L group; decreased calcium in females in the 12 mg/L group; increased albumin in males in the 12 mg/L group; and increased total protein in both sexes exposed to 5 or 12 mg/L. On recovery, only the increase in ALT activity in males remained. Urinalysis showed no changes that were of toxicological significance.
The authors considered that the effects of D4 were restricted to the 2 highest exposure levels. They offered the opinion that the blood chemistry data suggested hepatocellular alterations, that the respiratory tract findings were consistent with irritation, that the ovary and vaginal findings suggested possible reproductive organ effects, but that the effects on the testes likely were related to stress 119 .
Burns-Naas et al 120 studied the subchronic inhalation toxicity of D4 using groups of Fischer-344 rats (9 weeks old). The following groups of rats were exposed (nose-only to D4 vapor for 3 months (6 h/d, 5 d/week) or to control air: 35 ppm (20/sex/group), 122 ppm (20/sex/group), 488 ppm (20/sex/group), 898 ppm (30/sex/group), and control air (30/sex/group). Exposures were conducted according to the methods of Berstein et al 121 and Cannon et al 122 The system (nose-only inhalation) was designed to ensure a uniform test article distribution, provided a constant stream of fresh test article to each animal and precluded rebreathing from exhaled air. Hematology and serum chemistry evaluations, urinalyses, and histopathological evaluations were performed. Results relating to effects on the reproductive system are included in Reproductive and Developmental Toxicity section reported later in the text. Five female rats in the highest exposure group (898 ppm) died. At gross necropsy, the cause of death was not determined.
In the 898 ppm exposure group, the body weights of male and female rats were slightly less (4%-6%) than those of controls throughout the course of the study. This finding may have been due to decreased feed consumption during the first 3 days of exposure in male (20% decrease) and female (28% decrease) rats. For the remainder of the study, feed consumption and body weight gain were generally not significantly different. At the end of the 3-month exposure period, minor alterations in the weight of certain organs were reported for male rats. These changes in organ weight were not statistically significant and were not considered exposure related. At the end of the 1-month recovery period, a statistically significant decrease in absolute lung weight and in both absolute and relative kidney weight was observed in male rats exposed to D4 (898 ppm). The significance of these changes was described as unknown, considering that they were not present at the end of the 3-month-exposure period.
Compared to the control group, female rats of the 488 ppm and 898 ppm exposure groups had a concentration-dependent, statistically significant increase (20%-25%) in mean absolute liver weight. In female rats exposed to 488 ppm and 898 ppm D4, exposure-related, statistically significant increases in absolute and relative adrenal weights were noted. Increases in absolute adrenal gland weight ranged from 17% to 29% in the 488 and 898 ppm exposure groups, respectively. In female rats exposed to 898 ppm D4, ovarian weight (absolute and relative) was significantly decreased (38%). In both 488 ppm and 898 ppm exposure groups, a small, but statistically significant, decrease in mean absolute and relative thymus weights was observed. Female organ weights and organ-to-brain-weight ratios were no longer statistically significantly different when compared to the control group after a 1-month recovery period.
Hematological data were reported for male and female rats. In male rats, a slight decrease in red blood cells (5%) and hemoglobin (3%-4%) was reported. In both the 488 ppm and 898 ppm exposure groups (male rats), an increase in MCV (2%-8%) was noted. A decrease in MCHC (2%) was also observed in male rats of the 898 ppm exposure group. In female rats (898 ppm group only), the following results were reported: 6% increase in hematocrit (Hct), 4% increase in MCV, and 5% decrease in MCHC. With limited exception (noted by statistical significance), these minor alterations were resolved at the end of the 4-week recovery period. Compared to the control group, red blood cells in males decreased by 3% and the MCV and MCHC were slightly elevated (1.7% and 1.9%, respectively) at the end of the recovery period. It was noted that these hematological changes were within the normal limits for rats of this age and strain and are not thought to be related to D4 exposure.
In the serum chemistry data, the exposure-related findings that were common to males and females included increases in ALT and γ-glutamyltransferase (γ-GT) activity, and total protein, and decreases in triglycerides. The most striking increase in γ-GT activity was said to have occurred in female rats exposed to 488 ppm (330% increase) and in males (168% increase) and females (975% increase) exposed to 898 ppm. In male rats, the increase in ALT activity appeared concentration-related at all dose levels (10%-26% increase). This was not true for female rats; however, the increase in ALT activity attained statistical significance (15% increase) in females from the 898 ppm exposure group. In males, CK activity was significantly decreased at all exposure levels.
Decreased activities of CK were noted in female rats from 488 and 898 ppm exposure groups; however, compared to controls, the decrease was not statistically significant. Small, but statistically significant, decreases in bilirubin (5%-11%) were noted in female rats exposed to D4 at concentrations ranging from 122 to 898 ppm. Decreases in bilirubin were not observed in male rats. The results of urinalyses indicated no changes that were of toxicological significance, either at the termination of exposure or at the end of the recovery period.
Except for the lung, the incidence and types of gross observations did not appear to be related to D4 exposure. Gross lesions in the lungs of dosed rats consisted of multiple gray–white foci (1 mm in diameter). These foci generally corresponded to foci of macrophage accumulation and/or interstitial inflammation. It was noted that these observations were generally correlated with the presence of aerosol droplets in the mixed aerosol/vapor test atmospheres of the high-dose group (898 ppm). In both male and female rats exposed to 898 ppm D4 for 3 months, foci of alveolar macrophage accumulation increased in incidence and severity.
The incidence of these foci was also increased in females of the 122 and 488 ppm groups and severity was slightly increased in 1 female rat in the 488 ppm exposure group. It was noted that interstitial inflammation increased in both incidence and severity in male rats of the 898 ppm exposure group and in female rats exposed to D4 at concentrations ranging from 122 to 898 ppm. The severity of interstitial inflammation also increased slightly in 1 female rat of the 488 ppm exposure group. The incidence of leukocyte infiltration (ie, eosinophilic influx) increased in male and female rats exposed to 898 ppm. In female rats exposed to 898 ppm, the incidence and severity of eosinophilic infiltration increased. In male and female rats, the incidence of alveolar macrophage accumulation and interstitial inflammation was decreased at the end of the 1-month recovery period but was still slightly higher than the background incidence for these changes. Eosinophilic infiltrates were not present at the end of the recovery period.
Increased liver weights occurred in male rats (898 ppm group; 10% increase) and in female rats (488-898 ppm groups; 20%-25% increase) at the termination of exposure. However, there were no histopathological changes in the livers that could be attributed to D4 exposure. The authors noted that there were also no histological changes that support the alterations in γ-GT and ALT activities that were observed. The infiltrates of mononuclear cells were described as foci of inflammation comprised primarily of macrophages and lymphocytes randomly distributed throughout the liver lobule of most rats in both control and D4-exposed groups and were considered incidental findings.
Although increases in the incidence and severity of macrophage accumulation, interstitial inflammation, and eosinophil infiltration were observed in the lungs of male and female rats exposed to D4, there is uncertainty relating to the toxicological significance of these findings. 120
Chronic Inhalation Toxicity
Lee 52 evaluated the potential chronic toxicity and oncogenicity of D4 in Fischer-344 rats (7-8 weeks old) following whole-body inhalation exposure for 24 months (6 hours + T90 /d, 5 d/week). Results relating to carcinogenicity are included in the Carcinogenicity section reported later in the text. Five groups of rats (96/sex/group) were used and the exposure concentrations were as follows: 0, 10, 30, 150, and 700 ppm D4, respectively. Mean body weights prior to exposure were as follows: controls (males: 157.0 ± 8.4 g; females: 112.7 ± 6.3 g); 10 ppm (males: 157 ± 8.2 g; females: 112.2 ± 6.2 g); 30 ppm (males: 156.0 – 8.2 g; females: 11.9 ± 6.1 g); 150 ppm (155.8 ± 8.1 g; females: 111.9 ± 6.0 g); and 700 ppm (males: 154.8 ± 7.6 g; females: 111.4 ± 6.2 g). The animals were subdivided into 4 subgroups: A (scheduled for necropsy after 6 months of exposure—tissue level study); B (scheduled for necropsy after 12 months of exposure—chronic toxicity study); C (scheduled for necropsy at 24 months on study, after 12 months of exposure, and a 12-month recovery period—chronic recovery study); and D (scheduled for necropsy after 24 months of exposure—oncogenicity study). Clinical pathology parameters were measured at 3, 6, 9, and 12 months on study.
With the exception of subgroup A, a complete necropsy was performed on all rats, including those that died or were killed due to moribund condition. Selected organs were collected and weighed at the scheduled necropsies. Complete histopathology was performed on specified tissues from all early removal rats (except subgroup A), and for all rats in the control and 700 ppm exposure groups.
Overall, the effects of D4 exposure on in life parameters (survival, clinical signs, and body weights) were minimal. One exception was that the male rats in subgroup D exposed to 700 ppm had a significantly decreased 2-year survival and terminal body weight when compared to concurrent controls. The decreases in survival and body weight observed for male rats exposed to 700 ppm were apparent mainly during the last few months of the study. The in life parameters of other D4-exposed subgroup D male and female rats were comparable to control rats. When the survival of subgroup C was assessed after 12 months of recovery, there was no significant difference between the exposed and control groups of either sex. There was no incidence of early death in either subgroup A or subgroup B prior to the scheduled terminations. Additionally, there were no adverse clinical signs that were clearly associated with D4 exposure.
Regarding clinical pathology parameters measured, overall erythrocyte and urinalysis parameters of either sex were not affected by D4 exposure. Leukocytosis was consistently observed in male and female rats exposed to 700 ppm D4 at all time points; this finding resulted from increased lymphocytes. At histopathological examination, the primary target organs that were affected by D4 exposure included the uterus, respiratory tract, kidney, and liver.
Mean liver weights of D4-exposed rats killed after 6 months of exposure (subgroup A rats) were increased, particularly in males exposed to 700 ppm D4. The absolute liver weights tended to increase with increasing D4 exposure concentrations. Relative to concurrent controls, the difference was statistically significant at ∃30 ppm and at 700 ppm for males and females, respectively. At 12 months, the absolute liver weights in males and females of subgroup B were significantly increased, compared to controls, following exposure to 150 and 700 ppm D4. Subgroup B relative liver weights (normalized either to body or to brain weight) generally increased with increasing exposure concentrations. It was noted that this increase in liver weights may be associated with the centrilobular hypertrophy of hepatocytes that was diagnosed in subgroup B male rats, but not females, exposed to 700 ppm D4. These animals were necropsied after 12 months of exposure to 700 ppm D4.
The absolute and/or relative kidney weights increased in some of the male and female rats (subgroup B) at 12 months, but the differences were statistically significant, compared to controls, only for the 700 ppm exposure group. There were no significant changes in urinalysis results that may be indicative of renal pathology.
For subgroup D (exposure for 24 months), the absolute and/or relative liver and kidney weights were significantly increased, compared to controls, in male and female rats exposed to 700 ppm D4. It was noted that increases in kidney weights may reflect increases in the severity of chronic nephropathy in these groups. In male rats, increased liver weights were associated with increased hypertrophy of centrilobular hepatocytes at an exposure concentration of 700 ppm D4. However, this liver lesion did not accompany the weight increases that were observed in subgroup D females.
Compared to concurrent controls, exposure to D4 for up to 24 months appeared to increase the severity of chronic nephropathy in male and female rats of subgroup D exposed to 700 ppm D4. A statistical analysis supported a significant increase in the severity of chronic nephropathy in male and female rats exposed to 700 ppm D4. Though the results of the statistical analysis supported significant increases, compared to controls, in recovery subgroup C males exposed to 30, 150, or 700 ppm D4 and females exposed to 150 ppm D4, these differences were not considered toxicologically significant because of the relatively small difference in the mean severity of nephropathy and the lack of evidence of a dose-response relationship. Therefore, the only treatment-related renal effect appeared to have been an increase in the severity of chronic nephropathy observed in male and female rats following 24 months of exposure to 700 ppm D4.
Compared to controls, exposure to 700 ppm D4 for 24 months (subgroup D) resulted in a substantial increase in both absolute and relative uterine weights. It was noted that this increase in uterine weights may be related to the endometrial epithelial hyperplasia that was observed microscopically. Also, following exposure to 700 ppm D4 for 24 months, the body weight-normalized testes weight and the body weight-normalized heart weight in female rats increased, compared to controls. However, there were no associated concurrent histopathologic lesions in males or females in these groups.
In subgroup C, increased weights of the liver, kidney, and/or uterus were noted in a few animals exposed to D4. However, compared to controls, the weight differences were less than the weight differences in subgroup D. This was likely due to 12 months of recovery.
A statistically significant increase, compared to controls, in minimal or mild suppurative rhinitis was observed in subgroup B male rats exposed to 700 ppm D4 for 12 months. A statistical analysis of the incidence of suppurative rhinitis in male and female rats exposed to D4 indicated a positive trend that is related to D4 exposure. In subgroup D, the incidence of suppurative rhinitis following 24 months of exposure was much lower. This incidence was statistically significant only in females and there was no evidence of a dose–response relationship. The incidence of suppurative rhinitis was also lower in recovery subgroup C; no statistically significant differences were noted. These results suggest that suppurative rhinitis is related to exposure to 700 ppm D4 for 12 months but is not related to exposure for 24 months or recovery in subgroup C.
Eosionphilic globules noted in the nasal epithelium were not considered related to D4 exposure in subgroup C, despite statistical indications of differences in incidence. The diagnosis of eosinophilic globules in subgroup B male rats was treatment related, but was questionable in female rats. The incidence was also treatment-related in subgroup D male and female rats. The presence of a dose-related increase and statistical results indicating a significant increase in eosinophilic globules in male and female rats exposed to 700 ppm D4 and female rats exposed to 150-ppm D4 for 24 months provide good evidence that the eosinophilic globules in these groups are related to D4 exposure for 24 months (subgroup D).
The centrilobular hypertrophy of hepatocytes that was observed in male rats exposed to 700 ppm D4 for 12 or 24 months was considered related to D4 exposure; a statistical analysis confirmed the significance of this finding. The increased incidence of hematopoietic proliferation in spleens from females exposed to 700 ppm D4 for 24 months was confirmed by statistical analysis. The increase in hematopoietic proliferation in males exposed to 30 ppm, although not statistically significantly different, was considered related to D4 exposure.
An exposure-related decrease in the following enzyme activities was noted at 3, 6, 9, and 12 months of exposure in male and female rats exposed to D4: AST, ALT, CK, and LDH. Particularly at the 6- and 9-month time points, these decreases in enzyme activity were dose related. Relative to histopathology findings, there was no clear toxicologic significance of these decreases in serum enzymes.
Exposure to D4 for up to 24 months induced the following effects in male and female rats in this chronic study: (1) reduced 2-year survival and terminal body weight of male rats exposed to 700 ppm D4; (2) lymphocytic leukocytosis in male and female rats exposed to 700 ppm D4; (3) increases in absolute and/or relative weight of the liver, kidney, and uterus in rats exposed to D4, especially at an exposure concentration of 700 ppm; (4) increased incidence of suppurative rhinitis in male rats exposed to 700 ppm D4 for 12 months; (5) increased incidence and severity of intracytoplasmic eosinophilic globules in the nasal epithelium of male and female rats exposed to 700 ppm D4 for 24 months; (6) increased severity of chronic nephropathy in male and female rats exposed to 700 ppm D4 for 24 months; and (7) increased incidence of centrilobular hypertrophy of hepatocytes in male rats exposed to 700 ppm D4 for 12 or 24 months. 52
Ocular Toxicity/Irritation
Nakamura et al 123 evaluated the ocular toxicity of D4 using pigmented rabbits. When D4 (0.1-0.2 mL) was injected into the rabbit anterior chamber, severe corneal edema and opacification were elicited within 1 day; symptoms worsened as time progressed. Marked fibrin formation around the globule of D4 was observed, and vascularization of the peripheral cornea was prominently developed in eyes injected with D4.
Lee 52 evaluated the potential chronic toxicity and oncogenicity of D4 in Fischer-344 rats (7-8 weeks old) following whole-body inhalation exposure for 24 months (6 hours + T90 /d, 5 d/week), and details relating to the results of this study are included in the Chronic Toxicity and Carcinogenicity section of this report. Five groups of rats (96/sex/group) were used and the exposure concentrations were as follows: 0, 10, 30, 150, and 700 ppm D4, respectively. The animals were subdivided into 4 subgroups: A (scheduled for necropsy after 6 months of exposure—tissue level study); B (scheduled for necropsy after 12 months of exposure—chronic toxicity study; C (scheduled for necropsy at 24 months on study, after 12 months of exposure, and a 12-month recovery period—chronic recovery study); and D (scheduled for necropsy after 24 months of exposure—oncogenicity study). Ocular examination conducted 2 weeks prior to the scheduled sacrifices for subgroups B and D did not reveal eye lesions that were clearly associated with D4 exposure.
Reproductive and Developmental Toxicity
Inhalation Studies
IRDC 124 reported the results of a range-finding inhalation developmental toxicity study on cyclotetrasiloxane (D4) using rats. These and other reproductive/developmental toxicity study results are summarized in Table 9. Mated Charles River Crl:CD7 VAF/Plus7 female rats (6 per group) were exposed to 0, 10, 101, 301, and 700 ppm D4 vapors for 6 hours on each of the GDs 6 to 15. Dams were observed for signs of toxicity during the exposure period. Maternal body weights were determined on GDs 0, 6, 9, 12, 16, and 20. Feed consumption was determined at intervals up to GD 20. At day 20, the animals were killed and the uterus was excised. Gravid uterine weights were recorded and the location of viable and nonviable fetuses, early and late resorptions, and the numbers of total implantations and CL were recorded.
Reproductive and Developmental Toxicity Studies on Cyclotetrasiloxane (D4 )

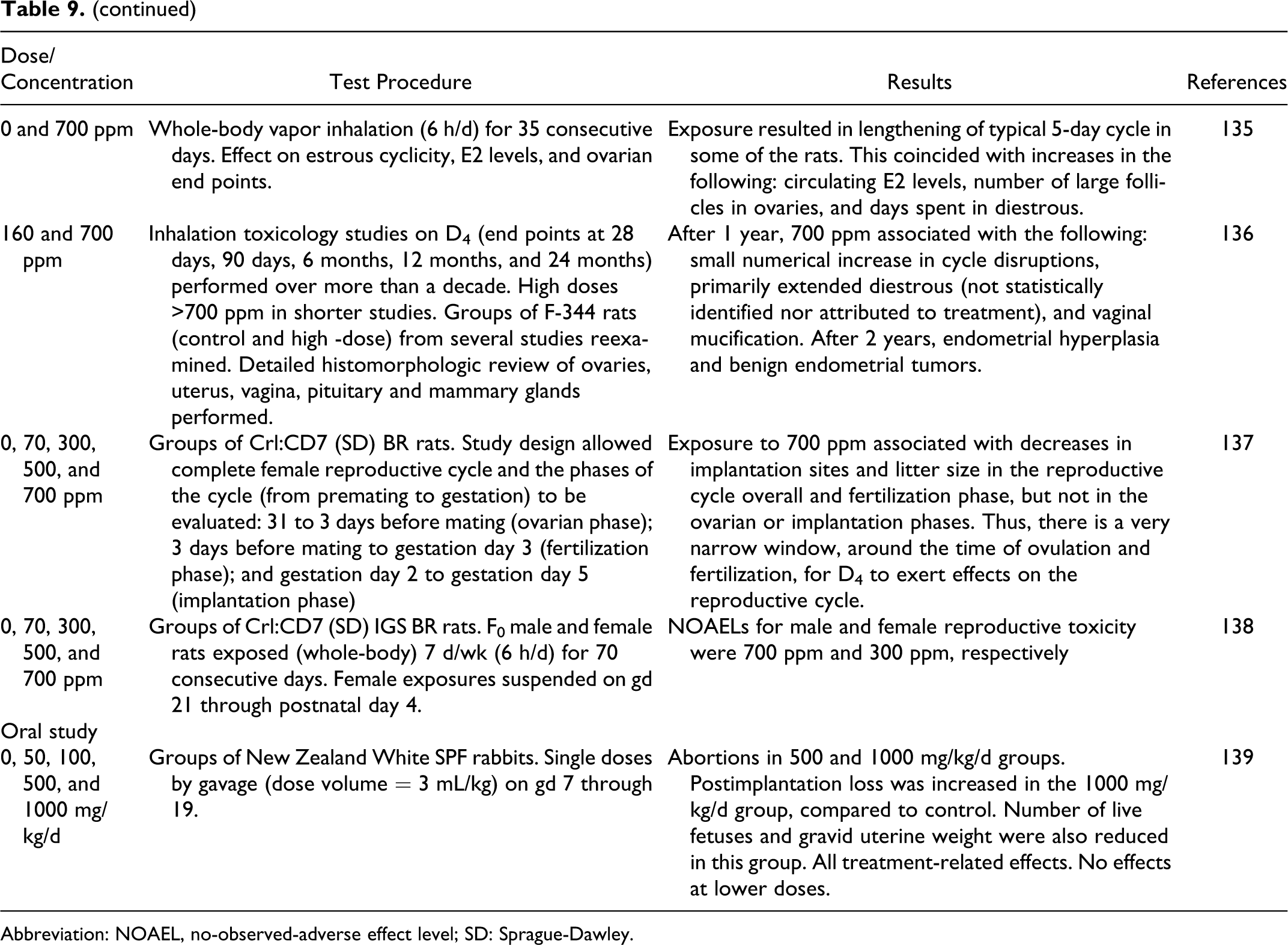
Abbreviation: NOAEL, no-observed-adverse effect level; SD: Sprague-Dawley.
No signs of maternal toxicity were reported. Significant decreases in body weight gains in dams were reported for the high-exposure group and considered treatment related by the authors. Significant decreases in feed consumption in the high-exposure group generally followed the pattern of decreased body weight gain in dams. No differences, between control and exposed animals, in numbers of the following were reported: pregnant animals, viable fetuses, animals with resorptions, CL, implantation sites, or postimplantation loss. 124
International Research and Development Corporation 125 reported the results of a D4 inhalation developmental toxicity study using rats. Mated female Charles River Crl:CD7 VAF/Plus7 rats (30 per group) were exposed to D4 vapor for 6 hours per day at 0, 100, 300, and 700 ppm on GDs 6 to 15. Dams were observed for signs of toxicity throughout the study. Maternal body weights were determined on GDs 0, 6, 9, 12, 16, and 20. Feed consumption was measured on GDs 0, 6, 9, 12, 16, and 20, and for the intervals 12 to 16 and 6 to 16. Animals were killed on GD 20; gravid uterine weights were recorded and the location of viable and nonviable fetuses, early and late resorptions and the number of total implantations and CL were recorded. The thoracic cavities and organs of females were examined for grossly evident morphological changes. Individual fetuses were weighed, sexed, tagged, and examined for external malformations and variations. Fetuses were dissected, internally sexed, and examined for visceral malformations and variations. For skeletal malformations, fetuses were fixed in alcohol, macerated with potassium hydroxide, stained with Alizarin Red S and cleared with glycerin.
No treatment-related signs of toxicity in the dams were reported. Body weight gains, however, were decreased in dams in the high-exposure group, as was feed consumption. The percentage of pregnant animals was not different between control and treated animals.
Fetal death (pre- and postimplantation loss), litter sizes, sex distribution, and fetal body weights were not different between treatment and control groups. A small number of skeletal variations was reported in the control and 100 and 300 ppm exposure groups, but none was reported for the 700 ppm group, suggesting to the authors that there were no treatment-related malformations. Decreased ossification of cervical vertebrae and ischia and bent ribs occurred only in fetuses of exposed animals, but their low frequency, absence of a dose response, and the normal spontaneous occurrence in historical controls suggested to the authors that these findings were not treatment related. 125
International Research and Development Corporation 126 reported the results of a range-finding inhalation developmental toxicity study using female rabbits. New Zealand White SPF rabbits (6 per group) were exposed to 0, 10, 101, 301, and 700 ppm D4 vapor for 6 hours on each of GDs 6 to 18. Animals were observed for signs of toxicity during exposure. Maternal body weights were determined on GDs 0, 6, 9, 12, 15, 19, 24, and 29. Feed consumption was determined for at intervals up to GD 29. The animals were killed on GD 29 and the uterus was excised, weighed, and examined, as described above in the preceding study involving rats.
Decreased defecation, soft stools, and/or anogenital staining were observed in the 0 (control), 101, 301, and 700 ppm groups. Decreased body weight gains were reported for the high-exposure group compared to controls. Animals in this group had increased weight gain (compared to controls) on GDs 19 to 29, suggesting to the authors that the inhibition of body weight gain was treatment related. Decreased feed consumption paralleled the decreased body weight gains in the high exposure group. There were no treatment-related effects on uterine weight, or in number of: pregnant animals, viable fetuses, with resorptions, CL, implantation sites, or postimplantation loss. 126
International Research and Development Corporation 127 conducted a D4 inhalation developmental toxicity study using rabbits. Artificially inseminated female New Zealand White rabbits (20 per group) were exposed to 0, 100, 300, and 501 ppm D4, 6 h/d, from GDs 6 to 18. Dams were observed for signs of toxicity throughout the study and killed on GD 29. Dams were weighed on GDs 0, 6, 9, 12, 15, 19, 24, and 29. Feed consumption was determined at intervals up to GD 29. Fetuses were examined as described above in the preceding study.
No significant differences in maternal weight gains between control and treated animals were observed. Statistically significant decreases in feed consumption were observed in the high-exposure group on GDs 6 to 9 and 9 to 12. Overall, feed consumption in the 501-ppm group was slightly less than in control animals. The authors considered the effects on feed consumption to be treatment related. No difference in either fetal malformations or developmental toxicity were reported between treated and control animals. 127
Holson and Stump 128 reported the results of a range-finding inhalation reproductive toxicity study of D4 using rats. Three groups (20 F0 males and 20 F0 females each) of Sprague-Dawley Crl:CD7BR rats were exposed to 0, 70, and 700 ppm D4, 6 h/d, for at least 28 days prior to mating through the end of the study, with the exception that F0 females were not exposed from GD 21 through lactation day 4. During mating, females and males were returned to the male’s cage after daily exposure until evidence of mating, at which point the females were again separately housed. Animals were observed 2× daily for signs of toxicity. Body weights were recorded weekly, and, for dams, on GDs 0, 7, 10, 14, and 20 and lactation days 0, 4, 7,14, and 21. Feed consumption was measured at intervals prior to mating, and during gestation and lactation. F0 males were killed during week 6 and examined. Pregnant animals were allowed to deliver and rear the pups. F0 females were killed and examined on lactation day 21.
At the same time, F1 pups (1 of each sex) were selected from each litter in each exposure group, with additional selections of up to 20 animals of each sex per group. Animals not selected were killed and examined. The exposure of selected F1 offspring continued up to PND 28, at which time all remaining animals were killed.
One female in the control group and 2 in the high-exposure group were killed early because they did not deliver litters. All other animals survived until the scheduled necropsy. Dried red material around the nose and dried clear material around the eyes was noted in the 700 ppm group, more so in females than males. Ejaculatory plugs found at the bottom of the cage were statistically significantly increased in the 700 ppm group, compared to controls.
Reproductive toxicity was not observed. Fertility indices for males were 95%, 100%, and 90% for the 0, 70, and 700 ppm groups, respectively. Fertility indices for females were 95%, 100%, and 95% for the 0, 70, and 700 ppm groups, respectively. Male mating indices were 95%, 100%, and 95%, and female mating indices were 95%, 100%, and 100%, for the 0, 70, and 700 ppm groups, respectively. The mean number of days between pairing and coitus was 2.3, 2.5, and 3.5 days for the 0, 70, and 700 ppm groups, with the historical control mean of 3.3 and a range from 2.3 to 6.1 days.
In the premating period, male body weights were significantly reduced in weeks 3 to 6 and female body weights were significantly decreased in weeks 1 to 3 in the 700 ppm group, compared to control animals. No differences between the 70 ppm and control groups were observed. The mean body weight in females was significantly decreased over the gestational period in the 700 ppm group, compared to controls. No differences between the 70 ppm and control groups were observed. Additionally, no differences in weight gain or mean body weights during lactation were reported for any treatment group, compared to controls. Feed consumption patterns were consistent with body weight data. The mean gestation period was not different between treatment and control groups; 21.6, 21.8, and 22 days for the 0, 70, and 700 ppm groups, respectively, with an historical control value of 21.9 days.
The number of dead pups at lactation day 0 was increased in the treatment groups, compared to the control group; but, because 1 litter in the 70 ppm group (5 of 8) and 1 litter in the 700 ppm group (5 of 8) accounted for most of the dead pups, this was not considered a treatment-related effect. Pup survival remained within the historical control values for both treatment groups. No differences in the sex ratio of F1 animals as a function of treatment were seen. Seven, 14, and 12 pups were found dead and 0, 4, and 4 pups were missing in the 0, 70, and 700 ppm groups, respectively. The mean live litter size was reduced in the 700 ppm group, compared to the control. The only difference in pup body weight was a slight increase on lactation days 1 and 4 in the 700 ppm group, compared to controls. No clinical signs of toxicity were noted in any group.
Necropsy of F0 males and F0 females revealed no findings that were related to exposure, except that the number of implantation sites was reduced in the 700 ppm group, compared to 0 and 70 ppm groups. The authors suggested that this was more related to the abnormally high number of implantation sites in the 0 and 70 ppm groups than to treatment with 700 ppm. Necropsy of pups revealed only isolated cases of dark red lungs.
The authors concluded that (1) there were signs of maternal toxicity in the 700 ppm group (decreased weight gain and feed consumption) but not at 70 ppm; (2) possible reproductive toxicity was seen in the 700 ppm group (decreased mean litter size and implantation sites); and (3) no postnatal toxicity was evident. 128
Holson and Stump 129 repeated the above study using 22 F0 male and 22 F0 female Sprague-Dawley Crl:CD7BR rats in each of 2 control groups and one 700 ppm D4 exposure group. The design was similar to that of the preceding study described above, except that all exposures of F0 females ended at GD 20, there were no postnatal exposures of pups, and all animals were killed at lactation day 4.
Maternal toxicity, in the form of decreased body weights and feed consumption, was seen in the 700 ppm group, compared to controls. As in the previous study, reproductive parameters of fertility, mating, and days between pairing and coitus were not affected by exposure to 700 ppm D4. The authors reported a decrease in the mean live litter size and a reduction in pup viability at PND 1 and 4 (where this study was terminated). Exposed females also had a decreased number of implantation sites and CL, an increase in preimplantation loss, and a difference in the number of implantation sites and the number of offspring. The authors concluded that the findings of the previous study were confirmed. 129
Holson and Stump 130 conducted an inhalation range-finding reproductive toxicity study of D4 using female rats. Groups of female Sprague-Dawley Crl:CD7BR rats (22 per group) were exposed to 0, 70, 300, 500, or 700 ppm D4 for 6 hours per day for at least 70 consecutive days prior to mating with unexposed males. The exposure of females was continued through mating up to necropsy, except during GD 21 to lactation day 4. During mating, females were exposed for 6 hours and then returned to the male’s cage. After mating was confirmed, the females were returned to their cages after daily exposure; the males were killed and discarded. All females were allowed to deliver and rear their pups until lactation day 21. Females were observed for clinical signs throughout the study. Body weights and feed consumption were determined on a regular basis. Necropsies were performed on 10 pups/sex/exposure group at PND 28, and on all pups that died between PND 5 and 28. Microscopic examination of tissue and ovarian follicle counts were performed on control and high-exposure females. The mean measured D4 exposure concentrations were 72, 302, 498, and 700 ppm. All animals survived until their scheduled necropsy.
Dried, red material was seen around the nose and mouth of females in the 300, 500, and 700 ppm exposure groups at 1 hour following exposure. Dried, red material was observed around the eyes of females in the high-exposure group, as well as wet, red material around the nose at 1 hour following exposure.
Mean body weights were decreased in the first week of exposure in females in the 500 and 700 ppm groups. Mean body weight gains were reduced in high-exposure females during GDs 10 to 14 and 14 to 20. Mean body weights were decreased in this group during GDs 14 to 20 and remained decreased through lactation day 14. Feed consumption was not different, however, between exposed and control animals. No gross morphologic differences were noted between control and exposed females at necropsy. In the females in the high-exposure group that delivered, the mean number of implantation sites was decreased and the difference between the number of implantation sites and the number of offspring was increased. Mean liver weights were increased in females in the 300, 500, and 700 ppm groups.
The mean number of pups born and the mean litter size were decreased in the high-exposure group. No differences in pup body weight, percentage of males per litter, gross morphology at pup necropsy, and pup organ weights between control and exposed animals were found. The authors concluded that exposure to D4 was associated with maternal toxicity at levels of 300 ppm and greater based on clinical signs and liver weight increases, and body weight decreases at 500 and 700 ppm. Reproductive toxicity was in the form of decreased litter size and number of pups born, and the decreased numbers of implantation sites and the increased difference between the implantation sites and the number of offspring in the 700 ppm group. 130
Holson and Kaufman 131 conducted a range-finding inhalation reproductive toxicity study of D4 (at 70, 300, 500, and 700 ppm). Groups of male Sprague-Dawley Crl:CD7(SD) BR rats (22 F0 males) were exposed 6 hours per day for at least 70 days prior to mating with 22 F0 female rats of the same strain that had been exposed to filtered air for 21 days prior to 1:1 mating. Males continued to be removed from their cages and were exposed to D4 6 hours each day during mating and females were removed and exposed to filtered air throughout mating. Females were returned to the male’s home cage after each exposure for mating. Following evidence of mating (GD 0), females were caged separately. Exposure of males ended and 10 male rats were necropsied at that point. Females were exposed to filtered air daily for 6 hours through GD 20. Pregnant females were allowed to deliver and rear their pups until lactation day 4 (pup necropsies performed). Surviving F0 dams were necropsied (weeks 15-16) after completion of F1 pup necropsies, and surviving F0 males were necropsied at week 12 or just prior to necropsy of F0 females. Body weights, feed consumption, and clinical signs for all animals were recorded weekly prior to mating, then on GDs 0, 7, 10, 14, and 20, and on lactation days 1 and 4 (or the equivalent time line for males).
One male in the high-exposure group died of septicemia on day 97, but the authors did not relate the death to exposure to D4. One moribund female, nominally in the high-exposure group, was killed on lactation day 1. Because females were not exposed to D4 at any point, the authors did not relate the death to D4 exposure.
Clinical signs included red material around the nose at 1 hour following exposure in the high-exposure group males and increased numbers of ejaculatory plugs produced by males in all exposure groups, primarily during the daily 6-hour exposure. Both effects were considered treatment related. Mean body weights, body weight gains, and feed consumption were not different between exposed and control F0 animals. Reproductive performance, including days between pairing and coitus, mating and fertility indices, and duration of gestation and parturition, were not different between exposed and control animals. At necropsy, macroscopic findings, microscopic examination of tissues, and sperm motility and sperm morphology in males were not different between exposed and control animals. Mean absolute and relative liver, kidney, and thyroid gland weights were increased in the high exposure males, and mean absolute and relative liver weights were also increased in males in the 500 ppm group at the first necropsy point (10 animals, as described above). This effect was not seen in the 12 animals necropsied after 1 month without exposure, which the authors suggested was evidence that the effects were reversible.
Decreases in pup survival were reported in the high-exposure group at PND 0 and in the birth to PND 4 interval, but the differences were not statistically significant and complicated by the occurrence of pup deaths primarily in 2 litters. Mean pup body weights in the high-exposure group were significantly decreased, compared to controls, but the authors stated that this finding was uncertain because the weights were within the range of historical controls. No clinical signs were reported in pups from birth to PND 4 nor were any findings reported, as a result of pup necropsy, that were different between exposed and control animals.
The authors interpreted the clinical observations in males in the high-exposure group as evidence of paternal toxicity and suggested that there was slight paternal toxicity at 500 ppm, as evidenced by the reversible increases in liver weights. Because of the differences between the concurrent and historical pup body weight data, the authors did not offer a conclusion for effects on the F1 animals. 131
Stump and Stump 132 reported the results of a range-finding inhalation reproductive toxicity study of D4 (at 500 and 700 ppm) using rats. Groups of 40 male Sprague-Dawley Crl:CD7(SD) IGS BR rats (F0 males) were exposed 6 h/d for at least 70 days prior to mating with F0 female rats of the same strain that had been exposed to filtered air only (no D4 exposure) for 21 days prior to 1:1 mating in each male’s home cage. Males continued to be removed from their cages and were exposed to D4 for 6 h/d during mating, and females were exposed to filtered air throughout the mating for the same 6-hour period. Following evidence of mating (GD 0), females were caged separately. Females were not exposed to filtered air from GD 21 through lactation day 4. Male animals continued to be exposed to D4 for 6 h/d through study day 113, followed by a 5-week recovery (nonexposed) period. The 700 ppm group exposure was determined to be 693 ppm.
Body weights, feed consumption, and clinical signs were recorded throughout the study. Pregnant females were allowed to deliver. F1 pups were observed daily until PND 21, when they were killed and necropsied. F0 females were then killed and necropsied. At the end of the 5-week recovery period, males were killed and necropsied.
One male in the 700 ppm group died during week 15. All other F0 animals survived to the scheduled necropsy. Clinical signs (dried red material around the nose) were observed in the 700 ppm group at 1 hour after exposure began. Mean body weight gain and feed consumption both were reduced in F0 males in the first week of exposure to 700 ppm but not 500 ppm D4. Overall mean body weights were not different between the treatment group and controls.
A dose-related increase in male-produced ejaculatory plugs was observed during the exposure period. The incidence of ejaculatory plugs decreased during the 5-week recovery period. Reproductive parameters of fertility, mating, days between pairing and coitus, and parturition were not affected by exposure.
No adverse effects were seen in F0 males at necropsy in either exposed group, and organ weights were unaffected by exposure to D4. The mean live litter size, number of pups born, sex ratio, pup survival, and pup body weights were the same in all treated and control animals. No treatment-related effects were found in pups at necropsy. The authors concluded that exposure to D4 at 700 ppm did have an effect on F0 males, based on the clinical signs and on the reduced weight gain and feed consumption in week 1. 132
Stump 133 conducted a follow-up inhalation reproductive toxicity study of 700 ppm D4 using female rats given multiple and single-day exposures, from premating day 4 through GD 3. The 700 ppm D4 exposure groups (premating) were as follows: group 2 (25 females) exposed for 6 hours on premating day 1 only; group 3 (25 females) exposed on premating day 2 only; group 4 (50 females) exposed on premating day 3 only; group 5 (25 females) exposed on premating day 4; group 7 (70 females) exposed from premating day 3 to GD 3; and group 6 (125 females) exposed from premating day 3 to premating day 1. Postmating group exposures were group P2 (25 females) on GD 0; group P3 (25 females) on GD 1; and group P4 (25 females) on GD 2. The final postmating group P5 (25 females) was exposed from GDs 0 to 2. All males were unexposed.
The pregnancy rate was decreased for the group exposed on premating day 1 but not for any of the other premating days or for any of the postmating days. Mean body weights in group 6 were reduced on GD 0 but increased during GDs 0 to 4 and 0 to 8. In group 7, mean body weights decreased from GDs 0 to 4 and increased from GDs 4 to 8, although feed consumption was reduced from GDs 0 to 4 to 0 to 8. In group P5, reduced mean body weights were seen during GDs 0 to 4 and feed consumption was reduced from GDs 0 to 4 to GDs 0 to 8.
Decreased CL and implantation sites were seen in group 7 but not in groups 2 to 6. Thus, the study failed to narrow the period of reproductive toxicity sensitivity. No preimplantation loss was seen in any premating groups, although an increase in small implantation sites (<3.6 mm) was seen in group 7. All of these parameters were unaffected in postmating groups P2, P3, P4, or P5. Mean uterine weights were decreased in group 7, but mean ovarian weights were unaffected. Mean uterine and ovarian weights were unaffected by any other exposure regimen. 133
Stump 134 conducted a 2-generation reproductive toxicity and developmental neurotoxicity study using Crl:CD@ (SD) IGS BR rats (165 males and 165 females). Whole-body vapor inhalation exposure to D4 (minimum of 70 days, 6 hours daily) for F0 animals (30/sex/group) were at 70, 300, 500, and 700 ppm (actual observed mean exposure concentrations were 71 (±1.5), 298 (±7.5), 502 (±6.8), and 700 (±7.2) ppm. Exposures for F1 animals (minimum of 70 days, 6 hours daily) were at the same target concentrations (actual measured mean exposure concentrations were 71 (±1.9), 301 (±6.4), 502 (±8.8), and 702 (±12.2) ppm). A control group of identical design was exposed to clean, filtered air on a comparable regimen. Pregnant animals were not exposed from GD 21 to lactation day 4; exposure was reinitiated on lactation day 5.
End points were the reproductive capabilities of the F0 and F1 generations, neonatal survival, growth and development, and functional and morphological changes to the nervous system of the developing F2 rats. The F0 parental animals were mated once to produce the F1 generation. F1 animals were mated to produce the F2a litters. F1 parental animals were mated a second time to produce the F2b litters after a minimum of 31 days following weaning of the F2a litters. Following completion of the second mating of the F1 parental animals, F1 males were paired with sexually mature, unexposed females from the same strain to produce F2c litters.
All animals were observed twice daily for appearance and behavior. Clinical observations, body weights, and feed consumption were recorded prior to mating and during gestation and lactation. Neurological tests (motor activity, auditory startle test, and Biel maze swimming trials) and functional observational battery (FOB) observations were performed for all F1 females on GD 10 and lactation day 20, following the first mating. All F0 and Fl females were allowed to deliver and rear their pups until weaning on lactation day 21. Thirty pups/sex/group from the F2a generation were selected for developmental landmarks, neurobehavioral testing, neuropathology, brain weights, and/or brain dimension measurements. Surplus F1 and F2a pups were necropsied on PND 21 or 28, and selected organs were weighed. Spermatogenic evaluations were performed on all F0 and F1 males, and ovarian primordial follicle and CL counts were recorded for F0 and F1 females in the control and high-exposure groups, and CL counts were performed on F1 females in the 70, 300, and 500 ppm groups
Evaluations of clinical observations and survival of F0 animals showed that during the first week of exposure, statistically significant reductions in mean body weight gain were observed in males and females in the 700 ppm group and in females in the 500 ppm group only.
Mean body weight gain was decreased (statistically significantly) during gestation in the 700 ppm group in both the F0 and F1 parental animals. Organ weight changes (liver and kidney increases) in both the F0 and F1 animals were consistent with the previous study discussed above. Ejaculatory plugs were noted in an exposure-related manner in most of the males throughout the exposure, but, as in the previous study, the significance of this finding was not known to the authors. No other significant clinical signs were noted at any test article concentration. Overall, no developmental neurotoxicity was observed.
Effects of the test article on reproduction were noted in F0 and F1 generations. Extended parturition and/or dystocia were observed in 2 and 3 F0 females in the 500 and 700 ppm groups, respectively, and in 3 F1 dams, 1 each in the 300, 500, and 700 ppm groups. Two of the 3 F0 700 ppm group dams and 1 F1 500 ppm group female died as a result of the dystocia. Statistically significant decreases in mean live litter sizes and mean number of pups born were observed in the 500 and 700 ppm groups for F0 animals. Statistically significant decreases (for 1st mating period) in the F1 animals related to mean live litter size in the 500 and 700 ppm groups and the mean number of pups born in the 700 ppm group were observed. Other nonstatistically significant, nondose-response–related effects were noted.
When F1 males were paired with unexposed females, no effects on reproductive performance were observed. In the F1 generation, mating indices were decreased in the 700 ppm group for the first and second matings (statistically significant for the females in both matings and for males in the second mating). Fertility indices were statistically significantly reduced in the 700 ppm group for the first F1 mating period. In the second F1 mating period, male and female fertility indices were statistically significantly reduced in the 500 and 700 ppm groups. The authors stated that microscopic evaluation of the ovaries, uterus, vagina, mammary gland, and pituitary gland from the 0, 70, 300, 500, and 700 ppm F1 females revealed a nondose-related effect that suggested perturbation of the estrous cycle and accelerated reproductive senescence in F1 (but not F0) females at 70, 300, and 500 ppm, with a more obvious effect at 700 ppm.
No adverse effects, at any exposure level, on anogenital distance, vaginal patency, and preputial separation were observed. The same was true for male functional reproductive parameters, male spermatogenic end points, microscopic evaluation of male reproductive tissue, or when the D4-exposed F1 males were mated with the unexposed, nulliparous females. These observations suggested that the reproductive toxicity observed was due to the exposure of females to D4. 134
Burns-Naas et al 120 studied the subchronic inhalation toxicity of D4 using groups of 20 male and female Fischer-344 rats (9 weeks old). The following groups of rats were exposed to D4 vapor for 3 months (6 h/d, 5 d/week) or to control air: 35 ppm (20/sex/group), 22 ppm (20/sex/group), 488 ppm (20/sex/group), 898 ppm (30/sex/group), and control air (30/sex/group). Exposures were conducted according to the methods of Berstein et al 121 and Cannon et al. 122 Results relating to effects other than those on the reproductive system are included in Subchronic Inhalation Toxicity section reported earlier in the text.
Compared to controls and intermediate exposure groups, more female rats from the 898 ppm group appeared to be in the diestrous stage of the rat estrus cycle. At the end of the 1-month recovery phase, the estrous cycles for control and 898 ppm exposure groups were similar; this comparison was based on the vaginal changes that were observed. Exposure-related histopathological findings were limited to the ovary and vagina, and these changes were observed only in the 898 ppm exposure group. Regarding mucification of the vaginal mucosa, the incidence was increased and the thickness of the mucinous cell layer was more prominent in the 898 ppm exposure group. By the end of the 3-month study, the incidence of ovarian hypoactivity in female rats exposed to 898 ppm had increased. A diagnosis of ovarian hypoactivity was made when histological sections of the ovaries contained no CL or only 1 or 2 small, atretic CL. The increased incidence of ovarian hypoactivity in the 898 ppm exposure group was not present by the end of the 1-month recovery phase.
In summary, nose-only inhalation of a high concentration of D4 (898 ppm) resulted in reversible histopathological changes in the female rat reproductive tract. However, lower exposure concentrations (35 ppm, 122 ppm, and 488 ppm) did not elicit these same effects. 120
Quinn 135 conducted a study to evaluate the effect of whole body vapor inhalation of D4 on estrous cyclicity, E2 levels, and ovarian end points in female Fischer-344 rats (13 weeks old; 145-165 g). The animals were exposed to the test substance (concentration = 700 ppm; 20 rats) via whole body inhalation for 35 consecutive days (6 h/d). Another group of animals (20 rats, exposed to filtered air) served as the control group. Food and water were withheld during the 6-hour exposure period. For 20 days prior to the initiation of exposure and throughout the exposure period, vaginal lavage was used to monitor the effect of D4 on estrous cyclicity. The animals were necropsied based on their estrous cycle. If possible, the animals were necropsied in estrus so that the number of ovulated eggs in the oviducts could be counted. Also, trunk blood was collected and E2 levels were measured using radioimmunoassay.
At necropsy, all of the control animals were in estrus (n = 20) and had ova in the oviducts (mean = 9.4 eggs/animal). Treated animals were divided into the following 2 groups: animals necropsied in estrus (n = 17) and animals that were not necropsied in estrus (n = 3), due to cycle disruption. A mean value of 10.3 eggs/animal (mean number of ova) was reported for the 17 animals necropsied in estrus. Ova were not present in the oviducts of the 3 remaining animals. When control and treated animals that ovulated were compared, there was no significant difference in the number of eggs ovulated.
For the following end points, there were no statistically significant differences between the control and 700 ppm exposure group at the end of exposure: body weights, ovarian weights, uterine weights, brain weights, uterine/brain-weight ratios, uterine/body-weight ratios, ovarian/brain weight, ovarian/body weight, day 1 CL or atretic antral follicles. There was a statistically significant increase in terminal body weights (P < .05) in the 700 ppm D4 exposure group, as well as an increase in the levels of E2 (P < .01). This significant increase in E2 levels coincided with a significant increase in the number of large follicles in the treatment group (P < .05).
Prior to the initiation of exposure, the mean cycle length was 4.98 days, with no statistically significant difference in cycle length between treated and control groups. After the initiation of exposure, treated animals began showing deviations in the typical 5-day cycle. Specifically, an increase in the number of days spent in diestrous II was noted. The control animals continued to cycle normally, with no difference in the cycle length over the entire time course. The individual cycle length during exposure was also compared. The control animals had a mean cycle length of 4.98, which was identical to that reported prior to exposure. D4-treated animals had a statistically significant increase in mean cycle length of 5.70 days during the exposure period.
Study results indicated that D4 exposure resulted in a lengthening of the typical 5-day estrous cycle in some of the Fischer-344 rats. This coincided with an increase in the circulating E2 levels on the morning of estrus, an increase in the number of large follicles in the ovaries of treated animals, and an increase in the days spent in diestrous. 135
Crissman 136 performed a detailed histomorphologic review of the ovaries, uterus, vagina, pituitary and mammary glands from D4-high-dose exposed and control rats to discover any previously unappreciated evidence for test article-related disruption of the estrous cycle or pituitary–gonadal axis. The basis for this study is stated as follows: in the ultimate 2-year oncogenicity study on D4, there were exposure-related increases in proliferative lesions of the uterus, including endometrial epithelial neoplasms. While there were significant differences in the incidence and apparent progression of lesions associated with the test material, the similarities suggested that a uniform and detailed expert histopathologic review of reproductive tissues from the entire series of studies with both materials was warranted.
Two series of 3 inhalation toxicology studies on D4 (F-344 rats used; end points at 28 days, 90 days, 6 months, 12 months, and 24 months) were performed over more than a decade. (The same is true for D5 —See Reproductive and Developmental Toxicity subheading in section on Animal Toxicology Data on Cyclopentasiloxane (D5) later in report text.). Female reproductive effects were reevaluated from control and high-dose groups in the following 3 inhalation studies. The original hematoxylin and eosin stained slides of the ovary, uterus, vagina, pituitary, and mammary gland from the high-dose and control groups were reexamined. The highest targeted exposure concentration, the exposure method (whole body or nose only), and the targeted number of animals in each study group (N) are stated below.
D4 studies:
Study number DC 8256: 1-Month Repeated Dose Inhalation Toxicity Study with Octamethylcyclotetrasiloxane (Cyclotetrasiloxane) in Rats (initial 16 mg/L [1330 ppm], lowered to 12 mg/L [997 ppm)]after 3 exposures because of mortalities; nose only, N = 10) 110 ;
study number DC 8257: 3-Month Repeated Dose Inhalation Toxicity Study with Octamethylcyclotetrasiloxane (Cyclotetrasiloxane) in Rats With 1 Month Recovery Period (12 mg/L [997 ppm]; nose only, N = 20) 119 ;
Study number DC 9106: 24-month Combined Chronic Toxicity and Oncogenicity Whole Body Vapor Inhalation Study of Octamethylcyclotetrasiloxane (Cyclotetrasiloxane; [D4] in Fischer-344 Rats (700 ppm, whole body; 6 month interim, N = 6; 12 month interim, N = 10; 24 month terminal, N = 60) 52
The nose-only exposure method and very-high-dose levels, theoretically yielding mixed vapor and aerosol exposures, as well as spontaneous deaths and other indicators of significant stress confound assigning effects as test article-related in the 28- and 90-day studies. After a year, 700 ppm D4 exposure was associated with a small numerical increase in cycle disruptions, primarily extended diestrous (not statistically identified nor attributed to treatment), and vaginal mucification. After 2 years, the ultimate findings of endometrial hyperplasia and benign endometrial tumors with D4 were confirmed and are consistent with the original reports. 136
Meeks et al 137 conducted a study to evaluate the phase of the female rat reproductive cycle that was affected by D4, using a study design that allowed the complete female reproductive cycle, as well as phases of the cycle (from premating to gestation) to be evaluated. The study was conducted using groups of 24 virgin male and female Crl:CD7 (SD) BR rats (age of female rats: 59-71 days old). In the overall phase, 4 groups were exposed to target concentrations of 70 ppm, 300 ppm, 500 ppm, or 700 ppm D4 for 28 days prior to mating and through the mating period and gestation, until GD19.
The ovarian phase, fertilization phase, and implantation phase each consisted of one D4 exposure group (60, 60, and 24 females, respectively), with a target concentration of 700 ppm. In the ovarian phase, female rats were exposed from 31 days prior to the start of the mating interval until 3 days prior to the start of the mating interval (total duration of exposure = 28 days). In the fertilization phase, female rats were exposed from 3 days prior to the start of the mating interval and through the mating interval until GD 3. In the implantation phase, female rats were exposed on GDs 2 through 5. Concurrent female control groups of similar design in each phase were exposed to filtered air on comparable regimens.
A second experiment (protocol similar to experiment 1) focused on the fertilization and implantation phases to further define the temporal responsiveness of the effects that were observed after D4 exposure during these phases. The duration of all exposures (0 or 700 ppm D4) was 6 h/d. In the premating phase, the rats were exposed to D4 according to the following protocols: as a single 6-hour exposure at 1, 2, 3, or 4 days prior to mating (days 1-4 groups, respectively); or daily for 3 days prior to mating through 1 day prior to mating (days 3-1); or daily, beginning 3 days prior to mating and continuing through GD 3 (day 3 through GD 3). In the postmating phase, groups were exposed to D4 as a single 6-hour exposure on GDs 0, 1, or 2 (GD 0, GD 1, or GD 2 groups, respectively). The last group was exposed from GDs 0 to 2. After exposure, females in the premating phase were mated with unexposed male rats.
In the first experiment, the analyzed mean exposure concentrations for the overall phase were 72, 301, 503, and 698 ppm. The analyzed mean exposure concentrations for the 700 ppm target concentration in the ovarian phase, fertilization phase, and implantation phase were 702, 696, and 702 ppm, respectively. The following results relate to the overall phase. In the 700 ppm exposure group, 1 female delivered 7 pups with no apparent external malformations during exposure on GD 19. This unscheduled delivery was not attributed to D4 exposure. No treatment-related clinical signs were observed in either of the exposure groups. Mean absolute maternal adrenal gland weight was significantly increased in the 700 ppm group (by 16.8%, relative to the control group).
Regarding other maternal organ weights (brain, ovary, and thyroid), all were unaffected by D4 exposure. Days between pairing and coitus and mating and fertility indices were not adversely affected by exposure to D4. Mean numbers of CL were reduced in the 3 highest exposure groups (300, 500, and 700 ppm). However, statistical significance was achieved only in the 300 and 500 ppm exposure groups. A significant reduction in the mean number of viable fetuses was observed in the 500 and 700 ppm exposure groups. Mean gravid uterine weight was also reduced in both exposure groups, but the decrease was statistically significant only in the 700 ppm exposure group. In the 70 and 300 ppm exposure groups, intrauterine survival (viable fetuses and pre-implantation and postimplantation losses) was unaffected by D4 exposure.
In the ovarian phase, no exposure-related clinical signs or internal findings were observed. Mean maternal organ weights (brain, ovary, adrenal, and thyroid) in the 700 ppm exposure group were comparable to controls. Days between pairing and coitus, mating indices, and fertility indices were not adversely affected by exposure to D4. The number of CL and intrauterine survival were not affected by D4 exposure.
No exposure-related clinical signs or internal findings were noted folllowing exposure during the fertilization phase. Mean maternal organ weights (brain, adrenal, and thyroid) in the 700 ppm exposure group were comparable to controls. The mean number of CL in this exposure group was significantly lower when compared to controls, and the mean number of implantation sites was significantly reduced. Due to the lower number of implantation sites and significant increase in the percentage of early resorptions, the mean number of viable fetuses and gravid uterine weight were significantly reduced following exposure to 700 ppm D4.
In the implantation phase, the number of CL and intrauterine survival were unaffected by D4 (700 ppm) exposure, and the same was true for mean maternal organ weights (brain, ovary, adrenal, and thyroid) when compared to controls.
In experiment 2, the analyzed mean exposure concentration for both the premating phase and the postmating phase was 700 ppm. In an effort to further define the sensitive period for D4 exposure, a single 6-hour exposure to D4 on the day prior to mating resulted in a significant decrease in fertility.
Study results indicated that D4 exposure was associated with decreases in implantation sites and litter size in the overall and fertilization phases but not in the ovarian or implantation phases. The authors noted that these data indicate that there is a very narrow window, around the time of ovulation and fertilization, for D4 to exert effects on the reproductive cycle of the female rat. 137
In a study by Siddiqui et al, 138 the reproductive toxicity of D4 was evaluated using virgin male and female Crl:CD7 (SD) IGS BR rats (Sprague-Dawley rats, 165 per sex; 29-30 days old). F0 male and female rats (30/sex/group) were exposed (whole-body vapor inhalation) to D4 vapor at concentrations of 0 (filtered air), 70, 300, 500, or 700 ppm 7 d/week (6 h/d) for at least 70 consecutive days prior to mating. Exposure continued through weaning of the pups on PND 21. Female exposures were suspended from GD 21 through PND 4 to prevent parturition from occurring in the inhalation chamber and to avoid separating the dams from their offspring during early neonatal life.
Beginning on PND 22, F1 weanlings were exposed to D4 according to the same procedure that was described for the F0 generation; F2 pups were not exposed to D4 directly. F0 rats were mated once to produce the F1 generation, and F1 parental rats were mated twice in order to produce 2 F2 litters. Additionally, the F1 males were mated with unexposed females. In the F1 generation exposed to D4 during the first and second matings, prolonged estrous cycles and decreased mating and fertility indices were noted. In the 500 and 700 ppm exposure groups (F0 and F1 generations), significant decreases in the mean number of pups born and mean live litter size were observed. Following exposure to 700 ppm D4 (F0 and F1 generations), implantation sites were also reduced. For all exposure groups, there were no adverse effects on anogenital distance, vaginal patency, and preputial separation.
In male rats, there were no adverse effects on the following: functional reproductive parameters, spermatogenic end points, and microscopic evaluation of reproductive tissue. Additionally, no adverse effects were observed when D4-exposed F1 males were mated with unexposed females; this demonstrated that the reproductive toxicity observed was due to female rats exposed to D4. The NOAEL for male reproductive toxicity was determined to be 700 ppm, and this was based on the absence of an effect on reproduction when D4-exposed male rats were mated with naive females. Based on the statistically significant effects on fertility (P < .05) and litter size (P < .01), the NOAEL for female reproductive toxicity was determined to be 300 ppm.
The authors noted that the findings in this study are consistent with suppression of or delaying the LH surge, as well as acceleration of the onset of female reproductive senescence in the rat. It was also noted that though analogous pathways control ovulation in both rats and humans, there are significant differences in the mechanism for the timing and release of LH and resulting changes in the control of ovulation and mating behavior between the 2 species. The authors pointed out that if D4 delays rather than causing prolonged suppression or ablation of the LH surge, the reproductive mode of action (MoA) of D4 would not likely be relevant for humans. 138
Oral Studies
International Research and Development Corporation 139 conducted a range-finding developmental toxicity study using rabbits. Inseminated New Zealand White SPF female rabbits (6 per group) received 0, 50, 100, 500 or 1,000 mg/kg per d D4 by gavage (3.0 mL/kg) as a single daily dose on GDs 7 through 19. The control group received the Methocel7 vehicle only. The animals were observed for signs of toxicity during the exposure. Maternal body weights were determined on GDs 0, 7 to 19 (daily), 23, 26, and 29. Feed consumption was determined for GDs 0 to 7, daily between GDs 7 and 19, and for GDs 19 to 23, 23 to 26, 26 to 29, 7 to 19 and 0 to 29. Animals were killed on GD 29 and the uterus was excised, weighed, and examined. Gravid uterine weights, the number and location of viable and nonviable fetuses, early and late resorptions, and the number of total implantations and CL were recorded.
A single death was observed at the 500 mg/kg per d level on GD 26. Prior to death, decreased defecation, body surface staining, reduced activity, and moribundity were observed and, at necropsy, thick, dark green material was observed in the intestines, and ulcerated areas were observed in the stomach. No mortalities occurred in the other groups.
Statistically significant decreases in body weights were observed in the 100, 500, and 1000 mg/kg per d groups, and a trend to decreased weight was observed in the 50 mg/kg per d group. These results generally followed a similar pattern of decreased feed consumption. Many animals consumed less than 20 g feed/d.
Abortions were seen in 5 animals in the 500 mg/kg per d group and 4 animals in the 1000 mg/kg per d group. The authors considered these abortions to be treatment related and suggested that they correlated with decreased feed consumption. With only 2 animals to evaluate at the high-dose level, the authors expressed concern about the small numbers. Postimplantation loss was increased at the 1000 mg/kg per d level when compared to the control group and the number of live fetuses and, correspondingly, gravid uterine weight was reduced; these differences were considered treatment related. No effects were reported for the 50 or 100 mg/kg per d groups, compared to controls. 139
Genotoxicity
In a study by Dow Corning, 140 D4 was evaluated in an extended rodent dominant lethal assay. Male rats received D4 doses of 100, 500, and 1000 mg/kg per d (by gavage) 5 d/week for 8 weeks. Rats given an oral dose of triethylenemelamine (0.05 mg/kg per d), a known clastogen served as the positive control group; the negative control group received tap water. Fifteen male rats were used in each of the dose and control groups. A 2-week mating period (2 females per male/week) followed, to allow for the expression of any germinal cell damage in pregnant females.
The numbers of CL and live and dead implantations were counted in females killed 14 days after the midweek of mating. Male body weights were also measured over the dosing period. No statistically significant differences in body weight (males) were found among the D4-dosed rats and the positive and negative control rats. Compared to the negative control, no significant decrease in fertility or prevalence of dead implants in D4-dosed rats was found. In the positive control group, statistically significant differences in fertility over the 2-week test period and in the number of dead implants were noted. 140
Isquith et al 102 studied the in vitro genotoxcity of D4 using the following assays: plate incorporation assay (S typhimurium strains TA1535, TA 1537, TA1538, TA98, and TA100), mitotic gene conversion assay (Saccharomyces cerevisiae strain D4), the E coli pol A-repair assay for DNA damage (E coli strain W3110 (pol A+), and mouse lymphoma assays (L5178Y mouse lymphoma cells). The following were evaluated in the mouse lymphoma tissue culture assays: forward gene mutation, SCE, DNA damage, and chromosome aberration potential. All of the assays were performed with and without metabolic activation. Details relating to the conduct of these assays and results for the positive/negative controls are included under the Genotoxicity subheading in an earlier section of this safety assessment that is entitled Animal Toxicology Data on Cyclotrisiloxane (D3).
In the plate incorporation, mitotic gene conversion, and DNA damage assays, D4 was tested at doses of 0.001 to 5 μL/plate. In each of the 3 assays, there was no evidence of increases in the incidence of reverse mutation, mitotic recombination, or DNA damage, respectively, either with or without metabolic activation.
The following concentrations of D4 were tested in the mouse lymphoma assays: forward mutation assay (0.0032-0.05 μL/mL); SCE assay (0.0032-0.050 [without metabolic activation] and 0.0032-0.10 μg/mL [with metabolic activation]); and chromosome aberrations assay (0.0032-0.050 [without metabolic activation] and 0.0032-0.10 μg/mL [with metabolic activation]). In the forward gene mutation assay, D4 did not produce a positive response, as measured by a dose-dependent, reproducible HRMF of 2.5 or greater. In the SCE assay, D4 induced a statistically significant response at one dose level (0.0032 μg/mL, with metabolic activation); a dose–response relationship was not observed. In the chromosome aberrations assay, D4 was genotoxic with, but not without, metabolic activation. D4 did not produce a positive response in the DNA/alkaline elution assay. 102
The mutagenicity of D4 was evaluated in a battery of tests. 141 In the preincubation test, the mutagenicity of D4 was evaluated at doses up to 5 mg/plate using the following S typhimurium strains: TA98, TA 100, TA 1535, TA 1537, and TA 1538. D4 was not mutagenic with or without metabolic activation in either of the S typhimurium strains over the range of doses tested.
In the chromosome aberrations test, the treatment of cultured Chinese hamster ovary (CHO) cells was limited by cytotoxicity at D4 concentrations >0.003 mg/mL without metabolic activation and >0.03 mg/mL with metabolic activation. No significant dose-related increases in chromosomal aberration frequencies were observed in CHO cells treated with doses up to 0.003 mg/mL (without metabolic activation) and up to 0.03 mg/mL (with metabolic activation). Additionally, no significant dose-related increases in SCEs were noted in CHO cells tested with D4 at concentrations up to 0.003 mg/mL (without metabolic activation) and up to 0.03 mg/mL (with metabolic activation). Thus, the results for D4 were negative in the SCE assay.
A test for in vivo clastogenic potential was conducted using 7-week-old rats. The weight range for male rats was 213 to 242 g, and 135 to 163 g, for female rats. The rats were exposed to 700 ppm D4 by whole-body vapor inhalation for 5 days (6 h/d). Negative control rats were exposed to filtered air only and positive control rats were exposed to filtered air and, then, cyclophosphamide (30 mg/kg, IP) 24 hours before exposure was terminated. Animals exposed to D4 were killed at 6 hours and 24 hours after the final exposure. Positive and negative control animals were killed at 24 hours after the last exposure. The induction of chromosome aberrations in rat bone marrow in vivo was evaluated. There were no significant, treatment-related increases in chromosomal aberrations.
The results of the preceding in vitro tests indicate that D4 does not possess significant in vitro genotoxic potential. Additionally, no adverse genetic findings were noted in the in vivo screen for chromosomal aberrations. 141
Carcinogenicity
Lee 52 evaluated the potential chronic toxicity and oncogenicity of D4 in Fischer-344 rats (7-8 weeks old) following whole-body inhalation exposure for 24 months (6 hours + T90 /d, 5 d/week). Five groups of rats (96/sex/group) were used and the exposure concentrations were as follows: 0, 10, 30, 150, and 700 ppm D4, respectively. Mean body weights prior to exposure were as follows: controls (males: 157.0 ± 8.4 g; females: 112.7 ± 6.3 g); 10 ppm (males: 157 ± 8.2 g; females: 112.2 ± 6.2 g); 30 ppm (males: 156.0 ± 8.2 g; females: 11.9 ± 6.1 g); 150 ppm (males: 155.8 ± 8.1 g; females: 111.9 ± 6.0 g); and 700 ppm (males: 154.8 ± 7.6 g; females: 111.4 ± 6.2 g). The animals were subdivided into 4 subgroups: A (scheduled for necropsy after 6 months of exposure—tissue level study); B (scheduled for necropsy after 12 months of exposure—chronic toxicity study); C (scheduled for necropsy at 24 months on study, after 12 months of exposure, and a 12-month recovery period—chronic recovery study); and D (scheduled for necropsy after 24 months of exposure—oncogenicity study). Clinical pathology parameters were measured at 3, 6, 9, and 12 months on study. Results relating to ocular irritation are included in the Ocular Toxicity/Irritation section and results relating to effects on enzyme activity are included in the section on Enzyme Effects earlier in the report text. Results relating to chronic toxicity are included in the section on Chronic Inhalation Toxicity earlier in the report text.
With the exception of subgroup A, a complete necropsy was performed on all rats, including those that died or were killed due to moribund condition. Plasma, fat tissue, and liver samples were obtained from subgroup A for the determination of D4 content. [The distribution of D4 in these tissues is reported in the section on Absorption, Distribution, Metabolism, and Excretion earlier in the report text.] Histopathology was performed on specified tissues from all early removal rats (except subgroup A), and for all rats in the control and 700 ppm exposure groups.
At histopathological examination, the primary target organs that were affected by D4 exposure included the uterus, respiratory tract, kidney, and liver. Endometrial adenomas were present in sections of the uterus from 4 of 60 (7%) female rats exposed to 700 ppm D4 for 24 months. The incidence of this lesion in the concurrent control group was 0 of 59. A statistical analysis of these data indicated a significant (P < .05) positive trend. The authors noted that the incidence of this lesion in historical control F344 females is 2/1109 or 0.2%. The historical control data related to the tumor incidence for rats on a vehicle control National Toxicology Program 2000 diet.
A clear increase in the incidence and mean severity of endometrial epithelial hyperplasia was reported for female rats exposed to 700 ppm D4 for up to 24 months, compared to concurrent controls. Statistical analysis of the incidence of endometrial epithelial hyperplasia in all exposure groups indicated a significant increase (P < .01) in female rats exposed to 700 ppm D4 and a significant positive trend. This provides further evidence for a proliferative effect of 700 ppm D4 on the endometrial epithelium. These data are interpreted as evidence for induction of endometrial epithelial hyperplasia and adenomas as a result of exposure to 700 ppm D4 for 24 months.
One endometrial adenoma was diagnosed in a rat from subgroup C that was necropsied after 12 months of exposure to 30 ppm D4 and 12 months of recovery. One endometrial adenoma was diagnosed in a rat from the same subgroup that was exposed to 150 ppm D4. Compared to concurrent controls, the incidence of these tumors was not statistically significant. The incidence of endometrial epithelial hyperplasia was increased in all exposure groups, compared to controls in subgroup C. However, this increase was not dose related and was not statistically significant, except for the group that was exposed to 10 ppm D4. Therefore, the tumors and hyperplasia of the endometrial epithelium in subgroup C are not considered related to D4 exposure.
The low incidence of hyperplasia of the squamous epithelium lining the cervical lumen observed in subgroup D rats exposed for up to 24 months and in the subgroup C recovery group exposed to 700 ppm D4 was not considered related to D4 exposure. Compared to controls, the incidence of endometrial stromal polyps was increased in subgroup C rats exposed to D4; however, the increase was not dose related or considered related to D4 exposure. This increase was not observed in subgroup D rats.
Inhalation exposure to D4 for 12 or 24 months increased the incidence of goblet cell hyperplasia in the nasal mucosa of male and female rats exposed to 700 ppm D4. However, the increased incidence in subgroup C rats exposed to D4 was small, indicating that goblet cell hyperplasia was related to D4 exposure. The exposed tissues returned to levels similar to those of controls when exposure was stopped for 12 months. Inhalation exposure to 700 ppm D4 for 12 months induced minimal hyperplasia of the squamous epithelium in the nasal vestibule of male and female rats. This lesion was present at a much lower incidence in D4 rats necropsied after 24 months of exposure, suggesting that this lesion returns to normal with continued exposure.
Inhalation exposure to D4 for up to 24 months induced the following effects in male and female rats: increased incidence of endometrial adenomas and endometrial epithelial hyperplasia in the uteri of rats exposed to 700 ppm D4 for 24 months; minimal-to-mild goblet cell hyperplasia in the nasal mucosa of rats exposed to 700 ppm D4 for 12 or 24 months; and minimal hyperplasia of squamous epithelium in the nasal vestibule of rats exposed to 700 ppm D4 for 12 months. 52
Clinical Assessment of Safety
Immunological Effects
In a study by Utell et al 56 in the section on Absorption, Distribution, Metabolism, and Excretion (In Vivo Studies—Human) earlier in the report text, immunological evaluations performed on blood samples at the same times that pulmonary function measurements were performed included the following: erythrocyte sedimentation rate, C-reactive protein, IL-6 levels, lymphocyte (CD3, CD4, CD8, CD56/16, and CD19) counts, lymphocyte proliferation and NK cell cytotoxicity in PMBC, and IL-2, γ-interferon, and TNF-α production. No immunologic findings were different between air and D4 exposures. The immunologic screening done at 3 months after the initial study also failed to identify any sensitization that might have occurred with the initial study. 56
In a study by Looney et al (1998), immunological effects of D4 were investigated following acute inhalation exposure. Twelve human volunteers (12 participants; 25-49 years old) were exposed to 10 ppm D4 for 1 hour via a mouthpiece apparatus. Crossover exposures were separated by at least 1 week, and the order of exposure to D4 or air was randomized. The assessment of immunotoxicity included enumeration of peripheral lymphocyte subsets and functional assays using PMBC. Because a direct test for the adjuvant effect of respiratory exposure in humans does not exist, proinflammatory cytokines and acute phase reactants in peripheral blood and markers for a systemic inflammatory response, as surrogate markers for adjuvancy, were analyzed. The test protocol (both air and D4 exposures) was repeated after approximately 3 months to determine whether re-exposure modified immunological end points. Blood was obtained prior to exposure, immediately postexposure, and at 6 and 24 hours postexposure. No immunotoxic or proinflammatory effects of respiratory exposure to D4 were found.
Looney et al (1998) studied the immunological effects of D4 using 12 normal human volunteers (7 females, 5 males; 20-52 years old). The participants ingested either 12 mg D4 (in corn oil, 1 mL) per day or corn oil alone (placebo) in a double-blind, placebo-controlled crossover study design. Doses were administered for 14 days, after which a 2-week nondosing period was observed. The nondosing period was followed by another 14-day dosing period. The first 14-day dosing period involved D4 and, the second, corn oil or vice versa. Blood was drawn prior to exposure and after 7 and 14 days of exposure. A variety of parameters were followed after oral exposure, including routine laboratory tests to assess toxicity (complete blood count and differential and routine chemical survey), immunological tests, clinical chemistries, and samples of blood and urine for D4 measurements. Assays for immunotoxicity included enumeration of peripheral lymphocytes subsets and functional assays using PMBC.
Regarding blood chemistry (BUN, creatinine, calcium, phosphorus, total protein, albumin, uric acid, billirubin, AST, SAP, LDH, and cholesterol), there were no statistically significant differences between D4 exposure and the placebo. There was also no difference in the total white blood cell count or in the percentage of lymphocytes (determined by complete blood count and differential count) between the D4-exposed samples and placebo-exposed samples. Additionally, there was no treatment-related effect on PHA- and alloantigen-induced proliferation, nor NK cell cytotoxicity.
The production of IL-2, γ-interferon, and TNF-α were measured using the ELISA, where supernatant from human PBMCs was stimulated by PHA for 48 hours in vitro. There was no significant difference between the placebo and D4 in these assays, and the same was true for TNF-α production by diluted whole blood and the levels of IL-6 or serum amyloid A. There were also no significant differences in helper T cells, supressor T cells, B cells, nor NK cells between the D4 and placebo groups.
It was concluded that, no immunotoxic or proinflammatory/adjuvant effect of D4 ingestion was observed in this study (Looney et al 1998).
Animal Toxicology Data on Cyclopentasiloxane (D5)
Acute Inhalation Toxicity
In a study by RCC, 106 the acute inhalation toxicity of D5 was evaluated using 4 groups (5/sex/group) of F344 albino rats. The rats were subjected to a single 4-hour, continuous (nose-only) exposure. The achieved test atmosphere concentrations (sum of the liquid and vapor phase) were as follows: 4.62 mg/L air (group 1), 6.73 mg/L air (group 2), 9.82 mg/L air (group 3), and 15.37 mg/L air (group 4). Exposure was followed by a 15-day observation period. All of the animals were killed and necropsied, and macroscopic abnormalities were recorded. The following organs were weighed and organ-to-body-weight ratios determined: lungs, liver, spleen, and thymus.
Four males and 4 females in the 9.82 mg/L air and all animals in the 15.37 mg/L air exposure groups died. There were no mortalities in the 4.62 or 6.73 mg/L air exposure groups. No intergroup differences in organ weights or organ-to-body-weight ratios were found. Treatment-related macroscopic observations included gray–white coloration (focal or general) in the eyes (15.37 mg/L group) and incompletely collapsed, reddish/dark red lungs (9.82 and 15.37 mg/L groups). An LC50 of 8.67 mg/L air (both sexes) was reported (RCC 106 ).
Short-Term Inhalation Toxicity
In a study by Siddiqui et al, 142 the subchronic inhalation toxicity of D5 was evaluated using groups of Sprague-Dawley rats (weights = 125-175g), and the results of this study are included in the section on Subchronic Inhalation Toxicity later in the report text. In the short-term inhalation toxicity portion of this study, 2 groups of 10 rats were exposed to D5 at concentrations of 0 and 120 ppm, respectively, for 28 days (6 h/d, 7 d/week). None of the animals died. There were no statistically significant differences in body weight or feed consumption between test and control animals. Treatment-related effects were not observed at necropsy, and the few clinopathologic changes that were of statistical significance were said to have been due to the biological variation that is typical of rats of this age and strain. The results of urinalyses were comparable with control values.
No statistically significant differences in absolute or relative liver weights were observed. The few histopathological changes observed were considered spontaneous changes that were typical of rats of this age and strain and unrelated to treatment. 142
Thevanez et al 143 conducted a 1-month, repeated dose inhalation toxicity study to assess the toxicological profile of D5 in Fischer 344 albino rats (body weights: 176-201 g [males] and 117-142 g [females]). Four groups of rats (10/sex/group) were exposed (nose-only exposure chamber) to D5 during a continuous exposure period of 6 h/d for 20 days (males) and 21 days (females). The control group (group 1) was exposed to filtered, clean air and the following groups were exposed to D5: 0.44 mg/L air (group 2), 0.65 mg/L air (group 3), 1.50 mg/L air (group 4), and 2.27/3.06 mg/L air (group 5, exposure days 1-6/7-termination, respectively).
Treatment-related effects that were presumed to be of possible toxicological relevance were generally of low incidence and minor severity. These effects were primarily restricted to rats in the 3.06 mg/L air exposure group, and their severity and incidence were more pronounced in female rats. Furthermore, these effects were characterized mainly by various clinical signs and hematological changes that were possibly indicative of a mildly stimulated immunological response.
Morphological alterations were observed in the nasal cavity, lungs, and liver of rats exposed to 2.27/3.06 g mg/L air (group 5). In group 5 rats, an increase in the incidence and severity of goblet cell proliferation in the nasal cavity, regarded as an adaptive response to an irritant, was observed. Minimum-to-moderate degrees of interstitial inflammation (evidence of minor organ toxicity) were observed in the lungs of group 5 rats. Changes in the liver were described as a slight degree of hepatocellular hypertrophy, also observed in group 5.
It was concluded that inhalation exposure to D5 at concentrations up to 2.27/3.06 mg/L air was not associated with any treatment-related effects that were considered to be of toxicological relevance. 143
Burns-Naas et al 94 assessed the potential toxicity and effect on humoral immunity following inhalation exposure to D5 in male and female Fischer 344 rats (groups of 25 males and females; 4-5 weeks old). Results relating to humoral immunity were included in the section on Immunological Effects earlier in the report text. The animals were exposed to D5 concentrations (whole-body inhalation exposure) of 0, 10, 25, 75, or 160 ppm 7 d/week (6 h/d) for 28 days. On the day after the final exposure, animals were killed (10 rats/sex/group) and complete necropsies were performed (terminal necropsy). After a 14-day nonexposure period, the remaining animals (5/sex/group) were killed and necropsies were performed (recovery necropsy). Tissue samples were collected for microscopic examination.
D5 exposure did not cause adverse effects on body weight, feed consumption, or urinalysis parameters. A statistically significant decrease in SAP in female rats was noted at terminal necropsy (12% decrease, at 160 ppm) and at recovery necropsy. A statistically significant increase in liver weight (15% increase, female rats) was observed in the 160 ppm exposure group, and appeared to have been exposure related. A statistically significant increase in the liver-to-body-weight ratios at the end of exposure (13% increase, at 160 ppm) was also noted for female rats; this was not noted for recovery animals from the same exposure group.
In male rats, a statistically significant increase in lung weights (15% increase, at 160 ppm) was reported at the end of the exposure period. A slight (4%) increase in liver weight was also noted in this exposure group. When this increase in liver weight was expressed as a percentage of the final body weight, it was considered statistically significant. A statistically significant increase in the thymus-to-body-weight ratio was also noted in the 160 ppm exposure group. The increases in liver-to-body-weight and thymus-to-body-weight ratios were noted at terminal necropsy but not at recovery. At recovery, only a significant increase in the spleen-to-body-weight ratios (14% and 17% at 25 ppm and 160 ppm, respectively) was reported.
At microscopic examination, an increased incidence and severity of nasal (level 1) goblet cell proliferation was noted in rats of both sexes exposed to 160 ppm, compared to controls. Only an increased incidence of this finding was associated with the 75 and 10 ppm exposures. An increased incidence of focal macrophage accumulation in the lung, at 160 ppm, was also associated with both sexes. Microscopic examination of the liver did not reveal any signs of treatment-related pathology, in light of the increase in liver weights.
As noted above, exposure-related increases in liver weights were observed at terminal necropsy. The results of serum chemistry analysis, organ weight measurements, and microscopic examination suggest that the slight increase in liver weight observed may not be toxicologically or biologically significant. However, based primarily on the changes in liver weights that were observed, the NOAEL for systemic toxicity was determined to be 75 ppm. The results of microscopic examination indicate that the respiratory tract is the primary target organ for D5 inhalation exposure, where minimal inflammatory cell infiltration, interstitial inflammation, and goblet cell proliferation in the anterior nasal passages were observed. It should also be noted that, at 14 days after the end of exposure, morphological alterations (excluding goblet cell proliferation) were found to be reversible, and the same was true for the alterations in organ weights.
Overall, these data suggest that high concentration D5 vapor inhalation provokes a minor reversible pulmonary inflammatory response and did not alter humoral immunity. 94
Short-Term Oral Toxicity
Crofoot et al 100 performed a 28-day oral exposure study on D5 using groups of 12 Sprague-Dawley rats (6/sex/group). Mean body weights for test animals were 276 g (males) and 233 g (females). The 2 control groups (males: 282 g; females: 226 g and males: 277 g; females: 229 g) received distilled water and sesame oil, respectively. The animals (test and controls) received doses of 1000 or 1500 mg/kg body weight per day for 28 days and were observed for signs of local or systemic toxicity, general appearance, behavioral abnormalities, and mortality. No treatment-related deaths, overt signs of toxicity, or changes in behavior were observed in any of the groups.
A statistical comparison of the mean body weights and feed consumption data showed no treatment-related effects between test and control groups. A statistically significant increase in absolute liver weights was observed in female rats dosed with D5. No gross pathological changes were observed in any of the organs or tissues of male and female rats in test or control groups. It was concluded that oral administration of D5 may produce statistically significant increases in liver weight. 100
Crofoot et al 144 studied the short-term oral toxicity of D5 using 5 groups of 16 (8/sex/group) Sprague-Dawley rats. Mean body weights for the 5 groups (control included) ranged from 331.12 to 341.16 g. Four groups received oral doses (via gavage) of 25, 100, 400, and 1600 mg/kg body weight, respectively. The fifth group served as the negative control (distilled water). The animals were dosed daily (5 d/week) for 2 consecutive weeks. No treatment-related deaths, overt signs of toxicity, or changes in behavior were observed in any of the groups.
A statistical comparison of liver weight data between control and test groups indicated significant increases in both the absolute and relative liver weights of female rats in 100, 400, and 1600 mg/kg dose groups. Based on statistics and an interpretation of the biological effect, it was noted that treatment-related increases in liver weights were observed at doses as low as 100 mg/kg in female rats. The effect at 25 mg/kg was said to have been unclear, and it was not possible to make a decision as to the biologic and toxicologic significance of the liver weight increase in female rats. A no-observed-effect-level (NOEL; for liver weight) of 100 mg/kg was reported for male rats. No significant changes were observed at gross pathological examination. Liver lesions, not treatment-related, were observed in 6 rats. These lesions were scattered between treatment groups and were considered typical and spontaneous for rats of this age and strain.
The results of this study indicate that oral administration of D5 can produce increases in liver weights at doses of 100, 400, and 1600 mg/kg in female rats, and that the NOEL for D5 in male rats is 100 mg/kg during a 14-day exposure period. 144
Short-Term Dermal Toxicity
Stanton et al 145 evaluated the dermal toxicity of D5 in a 28-day study using the following groups of Sprague-Dawley rats (10/sex/group). The weight ranges were 259 to 286 g (males) and 215 to 263 g (females). The test substance was applied to dorsal skin (clipped free of hair), and the area was then covered with plastic wrap and a cloth bandage. The groups received D5 doses of 0, 200, 800, and 1600 mg/kg, respectively, 7 days per week for 28 days. Additionally, a control and test group (5 males, 5 females per group) were treated with 0 and 1600 mg/kg body weight, respectively, and both groups were observed for reversibility, persistence, and delayed effects for a duration of 14 days after the treatment period. The animals were observed for 14 days after the cessation of daily application for reversibility, persistence, or the delayed occurrence of toxic effects. After the 28-day application period, blood and urine samples were collected and major organs were collected at necropsy for microscopic examination.
No mortality, overt signs of toxicity, or behavioral changes were observed in any of the dose groups. A statistical comparison of mean body weights and feed consumption between control and test groups indicated no treatment-related effects. There were also no statistically or biologically significant differences in hematology between control and test groups at either the terminal or recovery sacrifices. The few statistically significant differences in clinical chemistry parameters that were observed were considered within the range of normal biological variability. Treatment-related effects were not noted at histopathological examination (terminal and recovery necropsies). The presence of D (Me2SiO2) and T (MeSiO3) silicone species in the urine indicate that D5 was absorbed and metabolized when applied dermally to rats.
It was concluded that the dermal application of D5 at doses up to 1600 mg/kg for a period of 28 consecutive days does not produce significant toxicological effects in rats. 145
Subchronic Inhalation Toxicity
In a study by Siddiqui et al, 142 the subchronic inhalation toxicity of D5 was evaluated using groups of Sprague-Dawley rats (weights = 125-175g). The results of a short-term inhalation toxicity experiment from this study are included in the section on Short-Term Inhalation Toxicity earlier in the report text. Four groups of rats were exposed to 0, 60,120, and 200 ppm D5 7 days per week (6 h/d) for 13 weeks. With the exception of satellite group (120 ppm recovery group) animals, all rats were necropsied. Complete gross pathological examination was performed, and the following organs were perfused and fixed for possible electron microscopy: liver, brain, heart, kidneys, adrenal glands, testes, ovaries, lungs, and spleen.
None of the animals died, and clinical observations did not indicate any changes that were attributable to D5 exposure. There were no statistically significant differences in mean body weights or feed consumption between test and control groups, and clinical pathology data were not indicative of treatment-related effects. The results of urinalyses and hematological evaluations revealed no treatment-related effects. Compared to controls, an increase in relative liver weights was noted in female rats (120 ppm exposure) killed at the end of the study. However, by the end of the recovery period, liver weights had decreased to normal values. Neither gross nor microscopic changes considered treatment related were observed in any of the tissues/organs of test or control rats. The NOEL for female rats was 60 ppm, based on the increased liver weight data. Under test conditions, D5 did not appear to have either a biological or toxicologic effect in male rats. 142
Burns-Naas et al 146 studied the subchronic inhalation toxicity of D5 using groups of Fischer 344 rats (9 weeks old). The rats were exposed (nose-only inhalation) to the following D5 target concentrations 5 d/week (6 h/d) for 13 weeks: 0 (control air exposure, 30/sex/group), 26 ppm (20/sex/group), 46 ppm (20/sex/group), 86 ppm (20/sex/group), and 224 ppm (30/sex/group). The recovery groups (0 and 224 ppm) consisted of 10 rats/sex/group. For pathological examination, 20 rats/sex/group were killed at the end of the 13-week exposure period. Ten rats/sex/group were killed at the end of the recovery period. There were no clinical observations that were attributed to D5 exposure; the animals were observed for clinical signs daily.
Of the biochemistry parameters studied, an increase in γ-GT activity at 224 ppm in both sexes was most notable. This finding was dose related (46-224 ppm) in female rats and the cessation of exposure did not lead to recovery. It is important to note that other serum chemistry parameters that also reflect the status of the liver (AST and ALT activities) were not adversely affected by D5 exposure. A decrease in serum LDH activity was also observed in female rats of 86 ppm and 224 ppm exposure groups; cessation of exposure did not lead to recovery.
An increase in absolute and/or relative liver weights was noted in rats of both sexes. Also, in both sexes, exposure-related increases in absolute and relative lung weights were noted at terminal necropsy. These changes in lung weight were observed in females, but not in males, during the recovery phase. The results of microscopic examination indicated that the lung is the primary target organ in rats following D5 inhalation exposure. An increase in focal macrophage accumulation and interstitial inflammation were observed in the lungs of male and female rats exposed to 224 ppm. These lung changes had not resolved by the end of a 1-month period of nonexposure. The incidence of lung focal macrophage accumulation and interstitial inflammation was also slightly increased in male and female rats exposed to 86 ppm. Though liver weight was increased in both sexes, there were no microscopic findings indicating that the liver is a target organ in this study.
It was concluded that D5 inhalation exposure (nose-only) induces minimal changes in the lung. These changes were similar in both incidence and severity to the spontaneously occurring changes that were observed in control animals. There appears to be a sex-related difference in observed end points, considering that females appeared more sensitive to several exposure-related alterations in toxicological parameters. 146
Chronic Inhalation Toxicity
A chronic toxicity/carcinogenicity study on cyclopentasiloxane (D5) was performed by Smith et al. 147 Four groups of 96 male and 96 female Fischer 344 rats (SPF quality, CDF(F-344)/CrlBR) each, yielding a total of 768 rats, were used. Results relating to carcinogenicity are included in the Carcinogenicity section later in the report text. The rats were treated with cyclopentasiloxane (D5) via whole-body exposure. The target exposure levels were: 0 ppm (control group 1), 10 ppm (0.15 mg/L, group 2), 40 ppm (0.6 mg/L, group 3), and 160 ppm (2.42 mg/L, group 4). The rats were exposed for 5 d/week (6 h/d). The subgroup A animals (6 males, 6 females per group) were killed after 6 months and subgroup B animals (10/sex/group) were killed following 1 year of exposure. Subgroup C animals (20/sex/group) were killed after 1 year of exposure and 1 year of recovery. Subgroup D animals (60/sex/group) were killed following 2 years of exposure.
At all levels of exposure (10, 40, and 160 ppm), there were no test substance-related mortalities, clinical signs, or palpable masses. The results of ophthalmoscopic examinations performed during this study on D5 are included in the section on Ocular Toxicity/Irritation later in the report text.
Body weights for D5-treated groups were higher (statistically significant) when compared to controls. These differences occurred in group 3 (40 ppm) and group 4 (160 ppm) females of subgroups C (1 year of exposure/1 year of recovery) and subgroup D (2 years of exposure) and in all D5-treated male rats of subgroup D (2 years of exposure). However, this finding is considered unlikely to be related to treatment because of the following: (1) no evidence of a dose relationship, (2) the numerical differences were minimal, and (3) there was little evidence of progression and it was not evident in males of subgroup C (1 year of exposure/1 year recovery). Furthermore, the differences remained evident in group 4 (160 ppm) females of subgroup C when exposures had ceased during the second year. In group 3 (40 ppm) and group 4 (160 ppm) females of subgroup D (2 years of exposure), the differences were no longer evident, and were only occasionally evident in males of subgroup D (2 years of exposure), despite continuous exposures in this subgroup.
The only possible effect on the hematological parameters was confined to a decrease in the red blood cell count, with associated changes in the mean cell volume and mean cell hemoglobin values in males at 40 ppm and 160 ppm. However, these changes were temporary and minimal, and, thus, considered to be of no toxicological importance. Furthermore, no evidence of adverse effects on hematopoietic activity was recorded.
The decrease in urea concentration and increase in cholesterol/triglycerides, proteins, and gamma glutamyl transferase in females were possibly related to treatment with D5. These findings are suggestive of metabolic adaptive changes, primarily related to the liver. The only other treatment-related change was an increased serum calcium level in males and females receiving the 160 ppm dose. No toxicological relevance is associated with these findings, considering that the changes were minimal and not accompanied by any pathological findings.
The urinalysis changes, especially in males after 3 months, were likely treatment related. However, these changes were small, not present at subsequent measurements, and had no pathological correlates. Thus, they were considered to have been of no toxicological importance.
The determination of D5 levels in the plasma, fat tissue, and liver after 6 months of exposure indicated dose-related increases, with slightly higher values in the fat tissue and liver of females, compared to males.
The only organ weight changes that were considered possibly related to treatment with D5 were increased liver weights in females after 6 and 12 months of exposure, and in males after 2 years of exposure. However, this finding was not present in males after 6 and 12 months or in females after 2 years. This finding also was not dose related (not apparent in females dosed with 40 ppm), and there were no correlated findings at microscopic examination. Therefore, it was noted that this finding could be the consequence of a transient metabolic adaptation without any toxicological relevance.
Microscopic observation identified the nasal cavities, and, possibly, the uterus, as target organs. The statistically significant increased incidence of hyaline inclusions in the nasal respiratory/olfactory epithelium was noted in male and/or female rats of group 4 (160 ppm) killed after 6, 12, and 24 months; this finding was considered a nonspecific exposure-related effect. An increased incidence of hyaline inclusions was also observed in males receiving high dose (160 ppm), following the recovery period. It was not clear from this study whether or not this increase was related to dosing with D5. Considering that there were no other changes that were associated with a response to an irritant, such as inflammatory cell infiltration or degenerative changes in the epithelium, this finding was considered nonspecific and of low toxicological importance.
No intercurrent deaths, considered exposure-related, occurred in this 2-year inhalation toxicity study with cyclopentasiloxane in Fischer-344 rats. The gross findings observed in the study were considered unrelated to test substance exposure. The increased incidence of hyaline inclusions (statistically significant) in the nasal respiratory/olfactory epithelium of male and/or female rats of group 4 (160 ppm) killed after 6, 12, and 24 months represented a nonspecific treatment-related effect. It is not clear from this study whether or not the increased incidence of hyaline inclusions observed in rats receiving mid dose (40 ppm) and high dose (160 ppm), after the recovery period is related to test substance exposure. 147
Ocular Toxicity/Irritation
Nakamura et al 123 evaluated the ocular toxicity of D5 using pigmented rabbits. After D5 (0.1 to 0.2 mL) was injected into the rabbit anterior chamber, severe corneal edema and opacification were elicited within 1 day. These symptoms worsened with time.
A combined toxicity/oncogenicity study 147 comprised 4 groups of 96 male and 96 female Fischer-344 rats (SPF quality, CDF(F-344)/CrlBR) each yielding a total of 768 rats. The rats were treated with cyclopentasiloxane (D5) via whole body exposure. The target exposure levels were 0 ppm (control group 1), 10 ppm (0.15 mg/L, group 2), 40 ppm (0.6 mg/L, group 3), and 160 ppm (2.42 mg/L, group 4). The rats were exposed for 5 d/week (6 h/d) over a 1- to 2-year period. No test substance-related abnormal findings were observed at ophthalmoscopic examination. Additional results from this study on D5 are summarized in the Carcinogenicity section later in the report text.
Reproductive and Developmental Toxicity
Inhalation Studies
Holson and Stump 148 conducted an inhalation range-finding reproductive toxicity study of D5 in Sprague-Dawley Crl:CD7BR rats (weight ranges: 290-390 g [∼71-day-old males] and 233-302 g [108-day-old females]). The purpose of this study was to determine the exposure levels that would be appropriate for studying the potential adverse effects of D5 on male and female reproduction in rats. D5 was administered via whole body inhalation to 2 groups of 22 F0 male and 22 F0 female rats at exposure concentrations of 26 ppm and 132 ppm, respectively, 6 h/d for 28 days. The F0 animals were exposed prior to mating and through the day of necropsy, except for F0 females, where exposure was suspended from GD 21 through lactation day 4. The control group was exposed to clean, filtered air.
All of the F0 females were allowed to deliver and rear their pups to weaning on PND 21. The offspring were potentially exposed in utero (placental transfer), through suckling and/or dermal contact during lactation, and via 6-hour exposures after weaning until the animals were killed on PND 28. Surviving F0 dams were necropsied on lactation day 21 and the F0 males were necropsied after the breeding period. Reproductive parameters (fertility, mating, days between pairing and coitus, gestation, and parturition) were not adversely affected by D5 exposure at concentrations of 26 and 132 ppm. For F0 rats that survived to the scheduled necropsies, there were no exposure-related clinical signs. Mean body weights, body weight gains, and feed consumption in F0 males and females (during pre-breeding, gestation, and lactation periods) were unaffected by exposure to D5 at concentrations of 26 and 132 ppm. Also, no internal findings related to exposure to either concentration were noted at necropsy of F0 males or necropsy of F0 females on postmating day 25 (following total litter loss on lactation day 21). At the scheduled necropsy of F0 females on lactation day 21, the mean numbers of implantation sites and sites unaccounted for (ie, implantation sites not producing a pup) in exposed groups were similar to control values.
Mean live litter size was unaffected by D5 exposure (26 or 132 ppm). Except for total litter loss in 2 litters from the 132 ppm exposure group, there were no exposure-related effects on the number of dead pups on PND 0 or on pup viability throughout lactation. There were also no exposure-related signs in pups from the 26 ppm or 132 ppm exposure group. Also, at both exposure concentrations, F1 pup sex ratios and mean pup weights were unaffected by D5 exposure. No exposure-related findings were noted at necropsy of F1 pups on PND 21 and 28. 148
Crissman 136 performed a detailed histomorphologic review of the ovaries, uterus, vagina, pituitary gland, and mammary glands from D5-high-dose exposed and control rats to discover any previously unappreciated evidence for test article-related disruption of the estrous cycle or pituitary–gonadal axis. The basis for this study is stated as follows: In the ultimate 2-year oncogenicity study on D5, there were exposure-related increases in proliferative lesions of the uterus, including endometrial epithelial neoplasms. While there were significant differences in the incidence and apparent progression of lesions associated with the 2 test materials, the similarities suggested that a uniform and detailed expert histopathologic review of reproductive tissues from the entire series of studies with both materials was warranted.
Two series of 3 inhalation toxicology studies on D5 (F-344 rats used; end points at 28 days, 90 days, 6 months, 12 months, and 24 months) were performed over more than a decade. Female reproductive effects were reevaluated from control and high-dose groups in the following 3 inhalation studies. The original (except recuts on D5 uterine tumor slides) hematoxylin and eosin stained slides of the ovary, uterus, vagina, pituitary, and mammary gland from the high-dose and control groups were reexamined. The highest targeted exposure concentration, the exposure method (whole body or nose only), and the targeted number of animals in each study group (N) are stated below.
D5 Studies
Number DC 8258: One-Month Repeated Dose Inhalation Toxicity Study With Decamethylcyclopentasiloxane (Cyclopentasiloxane) in Rats (initial 2.2 mg/L [145 ppm], increased to 3.4 mg/L [224 ppm] after 6 exposures; nose only) The high dose was raised because of the lack of apparent toxic effects. N = 10143
Study Number DC 8259: 3-Month Repeated Dose Inhalation Toxicity Study With Decamethylcyclopentasiloxane (Cyclopentasiloxane) in Rats with a 1-Month Recovery Period. (3.4 mg/L [224 ppm]; nose only; regular study N = 20, recovery group, N = 10) 146
Study number: DC 9346: Decamethylcyclopentasiloxane (D5; Cyclopentasiloxane): A 24-Month Combined Chronic Toxicity and Oncogenicity Whole Body Vapor Inhalation Study in Fischer-344 Rats (160 ppm [2.42 mg/L], whole-body, 6-month interim, N = 6; 12 -month interim, N = 10; 24-month terminal, N = 60; recovery group, N = 20) 147
The nose-only exposure method and very-high-dose levels, theoretically yielding mixed vapor and aerosol exposures, as well as spontaneous deaths and other indicators of significant stress confound assigning effects as test article related in the 28- and 90-day studies. One year of exposure to 160 ppm D5 resulted in equal percentages of control and treated groups exhibiting abnormal estrous cycle (60%), with 5 of 6 of the animals in the controls in an estrogenic phase at necropsy as contrasted with 5 of 6 of the animals in the high-exposure group in a progestagenic phase. After 2 years, the ultimate finding of malignant endometrial tumors, without any preceding hyperplasia, with D5 was confirmed and is consistent with the original reports. 136
Genotoxicity
Isquith et al 102 studied the in vitro genotoxcity of D5 using the following assays: plate incorporation assay (S typhimurium strains TA1535, TA 1537, TA1538, TA98, and TA100), mitotic gene conversion assay (Saccharomyces cerevisiae strain D4), the E coli pol A-repair assay for DNA damage (E coli strain W3110 (pol A+)), and the mouse lymphoma assays (L5178Y mouse lymphoma cells). The following were evaluated in the mouse lymphoma tissue culture assays: forward gene mutation, SCE, DNA damage, and chromosome aberration potential. All of the assays were performed with and without metabolic activation. Details relating to the conduct of these assays and the results for positive/negative controls are included under the Genotoxicity subheading in an earlier section of this safety assessment that is entitled Animal Toxicology Data on Cyclotrisiloxane (D3).
In the plate incorporation, mitotic gene conversion, and DNA damage assays, D5 was tested at doses of 0.001 to 5 μL/plate. In each of the 3 assays, there was no evidence of increases in the incidence of reverse mutation, mitotic recombination, or DNA damage, respectively, either with or without metabolic activation. Ethanol and hydrochloric acid (control agents; potential organosilane hydrolysis products) also produced no significant end point increases in the respective microbial assays.
The following doses of D5 were tested in the mouse lymphoma assays: forward mutation assay (0.8-12.5 μL/mL [without metabolic activation] and 0.8-6.4 μL/mL [with metabolic activation]); SCE assay (0.8-25.0 μg/mL), and chromosome aberrations assay (0.8-25.0 μg/mL). In the forward gene mutation assay, D5 did not produce a positive response, as measured by a dose-dependent, reproducible HRMF of 2.5 or greater. In the SCE and chromosome aberrations assays, D5 was not genotoxic. D5 also did not produce a positive response in the DNA/alkaline elution assay 102 .
In the chromosome aberrations assay (L5178Y mouse lymphoma cells), aberrations in concurrent solvent controls (10 μL/mL ethanol) were noted in 0% to 4% of the cells, with 0% to 2% having 2 or more aberrations. Chemicals that caused aberrations in 8% or more of the cells were classified as having clastogenic (chromosome damaging) potential. Ethylmethane sulfonate (positive control) produced a range of 22% to 40% aberrant cells; 0% to 6% of the cells had 2 or more aberrations. Dimethylnitrosamine (positive control) produced 13% to 38% aberrant cells; 0% to 20% of the cells had 2 or more aberrations. D5 was not genotoxic.
Sokolowski 149 conducted a study to investigate the potential of D5 (in ethanol) to induce gene mutations in the plate incorporation test (experiment 1) and in the preincubation test (experiment 2) using S typhimurium strains TA1535, TA1537, TA98, and TA100, and E coli strain WP2 uvrA. The assay was performed with and without metabolic activation at the following test concentrations of D5: 33, 100, 333, 1000, 2500, and 5000 μg/plate. Sodium azide, 4-nitro-o-phenylenediamine, and methyl methane sulfonate served as positive controls without metabolic activation, and 2-aminoanthracene served as the positive control with metabolic activation. Ethanol served as the solvent control. All strains incubated with D5 had normal background growth up to 5000 μg/plate, with and without metabolic activation.
There was no evidence of toxic effects, that is, reduction in the number of revertants, in any of the strains tested either with or without metabolic activation. No substantial increase in revertant colony numbers was observed in any of the strains over the range of concentrations tested; this was true both with and without metabolic activation. Additionally, there was no tendency toward higher mutation rates with increasing test concentrations in the range below the generally acknowledged border of biological relevance. The positive controls induced a distinct increase in revertant colonies. D5 did not induce gene mutations by base pair changes or frameshifts in the genome of all of the strains tested, and, thus, was considered nonmutagenic in this assay. 149
Schulz 150 evaluated the genotoxicity of D5 in Chinese hamster V79 cells using the chromosome aberrations test in vitro. Two independent experiments were conducted (with and without metabolic activation), and each involved 2 parallel cultures. D5 was tested at concentrations up to 5 μL/mL. Ethanol served as the solvent control. Ethylmethane sulfonate and cyclophosphamide served as positive controls without and with metabolic activation, respectively. In experiments 1 and 2 without metabolic activation, toxic effects were observed. These toxic effects were indicated by reduced cell numbers and/or mitotic indices of below 50%, compared to the control. In experiments 1 and 2 with metabolic activation, no toxic effects were observed. Compared to the controls, there was no increase in the frequencies of polyploid metaphases. In the cytogenetic experiments, in the absence and presence of metabolic activation, no statistically significant and biologically relevant increase in the number of cells carrying structural chromosome aberrations was observed. The positive controls induced statistically significant increases (P < .05) in the number of cells with structural chromosome aberrations. It was concluded that D5 did not induce structural chromosome aberrations, and, thus, was considered nonclastogenic both with and without metabolic activation when tested up to the highest recommended concentrations and up to cytotoxic concentrations, respectively. Toxic effects were observed in the test without activation.
Honarvar 151 analyzed the genotoxic potential of D5 in Fischer-344 rats using the in vivo unscheduled DNA synthesis (UDS) and micronucleus assays. Groups of 12 Fischer-344 rats (6 males and 6 females per group) of the SPF quality (CDF(F-344)/CrlBR) were used. Male rats were 8 weeks old and female rats were 9 to 12 weeks old. Test animals were exposed to 160 ppm D5 (whole body vapor inhalation exposure, 6 h/d) for 7 days. Filtered air was used for the negative control groups. The positive control for the UDS assay was 2-acetylaminofluorene (2-AAF) and the positive control for the micronucleus assay was cyclophosphamide. Positive control animals were treated in a manner identical to that of the air control groups. However, after the last exposure, positive control animals were also dosed orally (by gavage) with the positive control substances.
For the analysis of DNA repair (UDS), the respective animals were killed at 5 hours or 6 hours and at 16 hours after the final treatment. Primary hepatocytes were obtained by liver perfusion and hepatocyte cultures were established and exposed for 4 hours to methyl-3H-thymidine (3HTdR), which is incorporated if UDS occurs. For each experimental group, controls included, hepatocytes from 6 treated animals per sex were assessed for the occurrence of UDS. The viability of the hepatocytes was not substantially affected due to in vivo treatment with D5. Compared to the current air controls, D5 did not induce UDS induction. Treatment with the positive control (2-AAF) revealed distinct increases in the number of nuclear and net grain counts.
For the micronucleus assay, bone marrow cells of the respective animals were collected for micronuclei analysis at 24 hours after the last treatment. For each experimental group, controls included, bone marrow cells from 6 treated animals per sex were assessed for the occurrence of micronuclei. Following treatment with D5, the number of polychromatic erythrocytes (PCEs) was not substantially decreased, as compared to the mean value for PCEs in the vehicle control group. This means that D5 did not exert cytotoxic effects in the bone marrow. Compared to the air controls, there was no biologically relevant or statistically significant enhancement of the frequency of the detected micronuclei following D5 administration. Treatment with the positive control (CPA) resulted in a substantial increase in the induced micronucleus frequency.
It was concluded that D5 induced neither DNA damage leading to increased repair synthesis in the hepatocytes of treated rats nor micronuclei. 151
Carcinogenicity
A combined chronic toxicity/oncogenicity study 147 comprised 4 groups of 96 male and 96 female Fischer-344 rats (SPF quality, CDF(F-344)/CrlBR) each, yielding a total of 768 rats. Test results other than those relating to carcinogenicity are included in sections on Chronic Inhalation Toxicity and Ocular Toxicity/Irritation earlier in the report text. The rats were treated with cyclopentasiloxane (D5) via whole-body exposure. The target exposure levels were 0 ppm (control group 1), 10 ppm (0.15 mg/L, group 2), 40 ppm (0.6 mg/L, group 3), and 160 ppm (2.42 mg/L, group 4). The rats were exposed for 5 d/week (6 h/d). The subgroup A animals (6 males, 6 females per group) were killed after 6 months and subgroup B animals (10/sex/group) were killed following 1 year of exposure. Subgroup C animals (20/sex/group) were killed after 1 year of exposure and 1 year of recovery. Subgroup D animals (60/sex/group) were killed following 2 years of exposure.
The authors noted that the increased incidence (statistically significant) of endometrial adenocarcinomas of the uterus of high-dose (160 ppm) rats following 2 years of exposure (subgroup D) may have been exposure related, considering that no such neoplasms were diagnosed in control rats of this subgroup. However, it was noted that these neoplasms also occur occasionally in control rats. Therefore, the relationship to exposure is unclear. All of the other findings diagnosed in this study were considered incidental findings that are commonly observed in rats of this age and strain (Smith et al 2005).
According to the EPA, 30 the final results of the 2-year chronic toxicity and carcinogenicity study on D5 (in rats) were received from Dow Corning in July 2005. 147 These results confirmed the significant increase in uterine tumors, following inhalation exposure to 160 ppm D5 (highest concentration tested), that was reported in preliminary study results. A significant increase in uterine tumors was not observed at lower doses of D5 (10 or 40 ppm). In addition to the 2-year chronic toxicity/carcinogenicity study, EPA received a series of studies that were conducted by Dow Corning to determine the specific MoA for D5-induced uterine tumors in rats.
The MoA studies conducted by Dow Corning, received from the SEHSC of North America, are included in this CIR safety assessment. 80,85 –91,102,135,136,149,151,152 Furthermore, these and other studies were taken into consideration in the safety assessment of D5 that was completed by Environ International Corporation in 2006 (included in following section on Clinical Assessment of Safety).
Clinical Assessment of Safety
Skin Irritation and Sensitization
A study was conducted 153 to evaluate the skin irritation/sensitization potential of a heat protection hair spray (leave-on) product containing 90.37% D5 (undiluted). In the repeated insult patch test (RIPT), 24-hour semiocclusive induction patches (test substance, 0.2 mL) were applied to the back (same site) 3 times per week for 3 consecutive weeks. The last induction patch application was followed by a 10- to 17-day nontreatment period. Challenge patches (24 hours application) were then applied and reactions were scored at 24 and 72 hours postapplication. One hundred ten participants (ages and sex not stated) completed the study. During the induction/challenge phase, reactions classified as barely perceptible (0.5-level) to mild (1-level) were observed in 22 participants. The authors noted that the skin reactivity observed was considered neither clinically meaningful irritation nor allergic in nature.
Another study 154 was conducted to evaluate the skin irritation/sensitization potential of a heat protection hair spray (leave-on) product containing 89.75% D5 (undiluted). The RIPT was conducted according to the procedure in the preceding study. One hundred ten participants (ages and sex not stated) completed the study. During the induction or challenge phase, barely perceptible reactions (0.5-level) were observed in 15 participants. The authors noted that the skin reactivity observed was considered neither evidence of clinically meaningful irritation nor allergy.
TKL Research 155 conducted an RIPT (occlusive patches) to evaluate the skin sensitization potential of a deodorant containing 56.3% D5 (undiluted). Patches were applied to the infrascapular area of the back. The 106 participants (males and females) who completed the study ranged in age from 18 to 66 years. More than 96% of the participants had self-assessed sensitive skin. The test procedure was similar to that for the preceding 2 studies, with the exception that a 10- to 15-day nontreatment period was observed after the induction phase and challenge reactions were scored at 48 and 72 hours postapplication. Reactions were scored according to the following scale: 0 (no visible erythema) to 3 (severe erythema [very intense redness]). None of the participants had reactions during induction or the challenge phase. Thus, there was no evidence of sensitization reactions to the product.
TKL Research 156 conducted an RIPT (occlusive patches) to evaluate the skin sensitization potential of an antiperspirant containing 55.76% D5 (undiluted) according to the procedure in the preceding study. The 105 participants (males and females) who completed the study ranged in age from 18 to 70 years. Reactions were scored according to the following scale: − (no reaction) to +++ (definite erythema, definite edema, and vesiculation). None of the participants had reactions during induction or the challenge phase. Thus, there was no evidence of sensitization reactions to the product.
Safety Assessment
Environ International Corporation 157 conducted a safety assessment to evaluate the potential hazard of D5 to various populations by defining a level at which no effects would be expected, and then comparing that to the amount of D5 to which workers, consumers, or the general public may be exposed. Persons who may be exposed to D5 include workers in the manufacture of D5 or personal care products containing D5; workers in dry cleaning establishments that use D5 as a replacement for other cleaning solvents; consumers who use personal care products containing D5, including antiperspirants/deodorants and hair care/skin care products; and the general public living in the vicinity of a plant that produces or processes these materials.
The safety assessment involved a critical review of the available toxicity and oncogenicity studies, as well as supporting information, including toxicokinetic data, mutagenicity and genotoxicity, reproductive/developmental toxicity, and immunotoxicity studies. These data were discussed as part of the hazard assessment. The review of these and other supporting data, in particular those studies designed to elucidate the MoA for observed effects in laboratory animals, was considered as part of a weight-of-evidence evaluation.
Following the hazard assessment, a dose–response assessment was performed to identify the dose associated with any observed effects that were relevant for extrapolation across species and for quantifying the dose–response relationship in order to define the point of departure (POD), as defined by the EPA. The relevance of the MoA of the key findings was a significant determinant of the approach for the dose–response assessment. The dose–response assessment was completed using a PBPK model to convert the experimental concentrations in the bioassay to human equivalent exposures, defined as the internal dose metric, AUC, in blood.
An exposure assessment was conducted using this PBPK model with human parameter values (for both physiological parameters, such as ventilation rate or cardiac output, and for D5-specific parameters, such as partition coefficients) to develop estimated internal dose metrics that were unique to the receptor, route of exposure, and exposure pattern. Characterization of exposure scenarios and estimation of D5 intake for the selected receptors and modes of exposure (ie, worker, consumer, and general public) were conducted.
Finally, MOS were developed, which compared the AUC for the POD to the AUC for the estimated internal dose metric estimated for each receptor and exposure scenario. The relative magnitude of the MOS estimated for selected receptors exposed by different routes of exposure was evaluated. Assumptions or parameter values (ie, variables, pathways, or parameter values) contributing most to estimates of risk or to the uncertainty in this assessment were identified, and, where possible, the impact on these assessments was quantified.
Numerical estimates were put into context, and an interpretation of those estimates was made, and conclusions as to the safety to the following populations were drawn: (1) persons who work in the production of D5, in the formulation of this material into personal care products, in the dry cleaning industry, or in the use of these products in professional settings; (2) consumers who use these personal care products, including antiperspirant/deodorants and hair care/skin care products; and (3) the general public living in the vicinity of a plant that produces or processes these materials and who may be exposed to ambient levels of D5 released to the environment during manufacturing activities.
The internal dose metric, AUC, based on the LED10 (lower bound on the dose corresponding to a 10% increase in risk), or the NOAEL were compared with the internal dose metric based on the various exposure scenarios to determine whether typical exposure to D5 might represent a hazard to human health. Exposure for all 3 populations was considered to occur via dermal and/or inhalation exposure. As with the derivation of the AUC for the LED10 or the NOAEL, a PBPK model was used to estimate the internal dose metric associated with the dermal or inhalation exposures for each population. These dose metrics were then compared to that derived for the LED10 or the NOAEL in an MOS. An MOS is the ratio of the internal dose metric or AUC associated with the POD to the internal dose metric estimated for each relevant exposure scenario.
Regardless of the POD considered for conducting a safety assessment, the MOS estimated for each exposure scenario was not greater than values that would be deemed acceptable by the appropriate regulatory agency (ie, Occupational Safety and Health Administration for occupational exposure). Therefore, it could be concluded that typical exposure to D5, whether occupationally or through the use of D5 containing consumer products or to the general public, would not result in a significant human health hazard. 157
Animal Toxicology Data on Cyclohexasiloxane (D6)
Acute Oral Toxicity
Pels Rijcken 158 studied the acute oral toxicity of D6 using the 6-week-old, fasted Wistar rats (Crl: (WI) BR strain [outbred, SPF-Quality]; 3 males and 3 females). Mean body weights for males and females were 194 and 147 g, respectively. A single oral dose of 2000 mg/kg (2.22 mL/kg) was administered, by gavage, to each animal. The animals were observed daily and macroscopic examination was perfomed after the terminal sacrifice (on day 15). None of the animals died and no clinical signs of toxicity were observed. Additionally, no abnormalities were noted at macroscopic postmortem examination. Body weight gain during the study was considered normal. The acute oral LD50 was >2000 mg/kg.
Acute Dermal Toxicity
Pels Rijcken 159 evaluated the acute dermal toxicity of D6 using 9-month-old Wistar rats (Crl: (WI) BR strain [outbred, SPF-Quality]; 5 males and 5 females). Mean body weights for males and females were 356 and 241 g, respectively. A single dose of the test substance (2000 mg/kg [2.22 mL/kg]) was applied to an area of skin that was 10% of the total body surface (25 cm2 in males; 18 cm2 in females). The test substance was maintained in contact with the skin with a dressing consisting of a surgical gauze patch that was covered with aluminum foil and Coban flexible bandage. For females, micropor tape was also used for fixation of the bandages. Dressings were removed after a 24-hour contact period. The animals were observed for clinical signs at periodic intervals up until day 15, at which time the animals were killed and subjected to necropsy.
None of the animals died and clinical signs were not noted in any of the animals tested. Additionally, no abnormalities were observed at macroscopic postmortem examination. The mean body weight gain during the observation period was within the range that is expected for rats that are used in this type of study. The acute dermal LD50 for D6 was >2000 mg/kg. 159
Short-Term Oral Toxicity
Crofoot et al 100 performed a 28-day oral exposure study using groups of 12 Sprague-Dawley rats (6/sex/group). Mean body weights for test animals were 278 (males) and 229 g (females). The 2 control groups (males: 282 g; females: 226 g and males: 277 g; females: 229 g) received distilled water and sesame oil, respectively. The animals (test and controls) received doses of 1000 or 1500 mg/kg body weight per day for 28 days and were observed for signs of local or systemic toxicity, general appearance, behavioral abnormalities, and mortality. No treatment-related deaths, overt signs of toxicity, or changes in behavior were observed in any of the groups.
A statistical comparison of the mean body weight and feed consumption data showed no treatment-related effects between test and control groups. A statistical comparison of organ weight data between test and control groups did not indicate a significant increase in the relative and absolute liver weights of male and female rats dosed with D6. No gross pathological changes were observed in any of the organs or tissues of male and female rats in test or control groups. 100
Ocular Toxicity/Irritation
Pels Rijcken 160 studied the ocular irritation potential of D6 using male New Zealand albino rabbits (SPF-Quality; 8 weeks old). The test substance (0.1 mL) was instilled into 1 eye of each of the 3 rabbits, and reactions were scored at 1, 2, 48, and 72 hours postinstillation. None of the animals died and there were no signs of systemic toxicity. Instillation of the test substance induced conjunctival irritation (redness) in all 3 rabbits, and all reactions had cleared within 24 hours. Neither iridial irritation nor corneal opacity was observed. Following treatment of the eyes with 2% fluorescein at 24 hours postinstillation, there was no evidence of corneal epithelial damage in any of the animals. The test substance did not induce staining of the periocular tissues.
Skin Irritation
Pels Rijcken 161 performed a study to evaluate the skin irritation or corrosion potential of a single dose of D6, administered to intact skin of 3 male New Zealand albino rabbits (SPF quality; 6 weeks old; body weights <35 kg). The test substance (0.5 mL) was applied to clipped skin of the flank of each animal using a Metalline patch that was mounted on Micropore tape. The tape was wrapped around the abdomen and secured with Coban elastic bandage. The dressing was removed at 4 hours postapplication. Irritation reactions were scored at approximately 1, 24, 48, and 72 hours postremoval according to the following scales: 0 (no erythema) to 4 (severe erythema [beet redness]) and 0 (no edema) to 4 (severe edema [raised more than 1 mm and extending beyond area of exposure]). Skin histopathology was not performed. None of the animals died and no signs of systemic toxicity were noted. The 4-hour dermal application of D6 did not induce irritation, corrosion, or staining in any of the animals tested.
Skin Sensitization
Pels Rijcken 162 studied the skin sensitization potential of D6 using the maximization test. Ten female albino guinea pigs (Himalayan strain, 4 weeks old, SPF-quality) comprised the test group, and 5 female guinea pigs served as controls (corn oil = control). The test animals were intradermally injected and epidermally exposed to undiluted D6, and the 5 control guinea pigs were treated similarly with corn oil. Approximately 24 hours prior to epidermal induction exposure, all of the animals were treated with 10% sodium dodecyl sulfate. At 2 weeks after epidermal application, all of the animals were challenged with undiluted D6 and the vehicle. One week later, the animals were subjected to a second challenge with 50% D6 and 20% D6.
Following the first challenge, grade 1 skin reactions were observed among 3 test animals and 1 control animal in response to undiluted D6. After the second challenge, no reactions to 50% D6 or 20% D6 were observed. Based on the responses to undiluted D6 during the first challenge phase, it was not possible to arrive at a conclusion regarding the sensitization rate. Considering that comparable reactions were observed in test and control guinea pigs, these reactions were described as signs of primary irritation. Based on the results of the second challenge phase, it was concluded that there was no evidence that D6 had caused skin hypersensitivity in guinea pigs. This is based on the observation that no responses to 50% D6 or 20% D6 were observed in test animals. These results indicate a sensitization rate of 0%. 162
Reproductive and Developmental Toxicity
Oral Studies
Siddiqui 163 conducted a study to evaluate the potential toxicity of cyclohexasiloxane (D6) in a combined repeated oral gavage toxicity study that included reproductive/developmental toxicity screening in groups of Sprague-Dawley albino rats(Crl:CD7 (SD) IGS BR VaF/Plus7 strain, 10 rats/sex/group). Weights ranged from 172.6 to 254.3 g (females) and from 284.9 to 386.2 g (males). The test substance was administered in corn oil daily (7 d/week) at doses of 0, 100, 330, or 1000 mg/kg per d for up to 45 consecutive days. Females were divided into toxicity and reproductive effects groups. The same males were used for both the toxicity and reproductive phases of the study. All males and toxicity group females were treated for 28 and 29 days, respectively. Females of the reproductive effects group were treated for 14 days prior to mating, during mating, during gestation, and up to and including postpartum day 3.
The animals were observed twice daily for mortality, morbidity, and moribundity. Clinical examinations were performed daily after dosing. Complete necropsy was performed on all males and toxicity group females. Microscopic examination was performed on protocol-specified tissues from the control and 1000 mg/kg dose males and toxicity group females. The thyroid glands and lungs of both sexes and the livers from females were also examined (100 and 330 mg/kg per d groups).
Mating was initiated after 2 weeks of dosing and continued for up to a maximum of 14 days. Both dams and pups were killed on post partum day 4 and examined for external gross lesions. The number of CL and the number of uterine implantation sites were determined for all reproductive females. The reproductive and developmental parameters evaluated were as follows: evidence of mating, pregnancy, duration of gestation, mean litter size, mean litter weight, mean ratio of live births/litter size, and sex ratio.
The oral gavage dosing of male and female Sprague-Dawley rats with D6 (in corn oil) at doses up to 1000 mg/kg per d for 28 to 46 consecutive days was generally well tolerated. There were no effects on clinical signs, body weights, or feed consumption. No treatment-related changes were observed in any FOB or motor activity parameters. There were no changes in hematology; particularly, there were no changes in serum chemistry that were clearly attributable to treatment. In males, prothrombin time was prolonged at the 2 highest dose levels; however, there were no clinical indications of clotting abnormalities. Absolute and/or relative organ weight increases were observed in the liver and kidneys (both sexes) and in the adrenal glands (females only). Except for the liver (in females), dose level effects on organ weight were not apparent. The authors noted that historical Dow Corning laboratory control data cast further doubt on the significance of apparent liver and kidney weight increases.
Histopathological findings included an increase in periportal lipidosis in the liver of females at all doses but without a dose-related effect on incidence or severity. This finding for the liver was considered of minimal toxicologic significance. Follicular cell hypertrophy of the thyroid gland (both sexes) was also reported, and there was no dose-related effect on the incidence in males and no effect on the severity in either sex. This finding for the thyroid gland was considered to be a secondary and adaptive effect. Granulomatous inflammation of the lungs was observed in 1 to 4 treated rats per group but not in controls; there was no dose-related effect on incidence or severity. The finding for the lungs was considered an artifact of the route of administration. Though an increase (not statistically significant) in the number of sperm-positive, nongravid females was observed in the high-dose group, no other effects were identified in any of the reproductive or developmental parameters that were evaluated.
Based on study results for the administration of D6 by repeated dose oral gavage, the NOAEL was 1000 mg/kg per d for systemic toxicity, reproductive performance, and developmental end points. 163
Genotoxicity
Verspeek-Rip 164 evaluated the mutagenicity of D6 in the S typhimurium reverse mutation assay using the following S typhimurium strains: TA 98, TA100, TA1535, and TA1537. The mutagenicity of D6 was also evaluated in the E coli reverse mutation assay using E coli strain WP2 uvrA. In both assays, D6 was tested at concentrations up to 1000 μg/plate both with and without metabolic activation. The bacterial background lawn was not reduced at all concentrations tested and there was no increase in the number of revertants.
D6 did not induce a dose-related increase in the number of revertant (His+) colonies in any of the 4 S typhimurium strains either with or without metabolic activation. D6 also did not induce a dose-related increase in the number of revertant (Trp+) colonies in E coli strain WP2 uvrA either with or without metabolic activation. The results of this E coli assay were confirmed in an independently repeated experiment. Based on the results of this study, it was concluded that D6 is not mutagenic in the S typhimurium reverse mutation assay or in the E coli reverse mutation assay. 164
Summary
Cyclomethicone
According to the most recent published version of the International Cosmetic Ingredient Dictionary and Handbook, cyclomethicone is a generic name for cyclic dimethyl polysiloxane compounds that conform to the formula included in Figure 1 in this safety assessment, where n has a value between 3 and 7. The INC intends to revise the INCI monograph definition of cyclomethicone to state that n has a value between 4 and 6, which more accurately reflects the current composition of cyclomethicone that is used in the personal care industry. This assessment includes cyclotrisiloxane (D3) because it is being detected in cosmetic products and because it has been identified as a contaminant of D4.
Cyclomethicone functions as a hair conditioning agent, skin conditioning agent—emollient, and solvent in cosmetic products. Frequency of use data provided by FDA in 2009 indicate that this ingredient is being used in 1499 cosmetic products. Data provided by the Personal Care Products Council in 2008 indicate that cyclomethicone has been used at concentrations up to 89%.
Reportedly, the gel of silicone gel-filled breast implants consists of 1% to 2% low-molecular-weight silicones with structures identified mainly as cyclic compounds such as the following: D3, D4, D5,D6, and D7. The results of a GC/MS analysis of breast implant distillate were as follows: D3 (18%), D4 (60%), D5 (20%), and D6 (2%). Low-molecular-weight linear siloxanes (<1%) and platinum (40 mg/kg distillate) were also detected.
After CD-1 mice were injected SC with breast implant distillate consisting primarily of low-molecular-weight cyclosiloxanes (D3-D7), D7 was the only cyclosiloxane that was not detectable in organs (heart, kidney, and lungs included). Furthermore, study results indicated that low-molecular-weight cyclosiloxanes persist in the organs of mice for at least 1 year after a single SC injection, and that D5 and D6 appear to persist longer in the organs when compared to D4.
The results of 2 acute inhalation toxicity studies indicated no adverse effects (10% cyclomethicone [average n = 4] aerosol burst) and moderate liver pathology (200 ppm cyclomethicone [average n = 4]), respectively, in rats.
In a study involving patients with Rosacea, the removal of cyclomethicone from a sunscreen lotion resulted in a significantly more irritating preparation.
Cyclomethicone (n = 4; n =5) was not mutagenic to S typhimurium strains in the Ames test. Single oral doses of Antifoam A (containing 10% cyclic oligomers) and Antifoam M (containing <0.022% of lower-molecular-weight, cyclic components) were administered to human participants. Study results indicated that the lower-molecular weight cyclosiloxanes could be absorbed to some extent. In RIPTs, cyclomethicone (average n = 4) was neither an irritant nor a sensitizer in human participants.
Cyclotrisiloxane (D3)
Cyclotrisiloxane once was listed in the International Cosmetic Ingredient Dictionary and Handbook but is no longer considered to be a cosmetic ingredient. D3 has been detected in cosmetic products but the expectation is that it is present as an impurity.
In OVEX αERKO mice, uterine wet weights were not significantly increased in a dose-dependent manner following exposure to oral doses of D3. Similarly, uterine peroxidase activity (marker for estrogenic activity) was not significantly increased following oral exposure.
No treatment-related deaths or overt signs of toxicity were observed in groups of Sprague-Dawley rats dosed orally with D3 in a short-term oral toxicity study. However, a statistically significant increase in liver weight was noted.
The results of a repeated dose, inhalation reproductive/developmental toxicity study on D3 indicated an NOAEL for systemic toxicity of 2500 ppm in female rats, based on no adverse treatment-related effects at the highest dose tested. The NOAEL for systemic toxicity in male rats was 500 ppm, based on decreased seminal vesicle weight, and 100 ppm, based on protein droplet nephropathy. Because a decrease in both litter size and the number of implantation sites was noted following dosing with 2500 ppm D3, the NOAEL for reproductive/developmental toxicity was considered to be 500 ppm.
D3 lacked significant clastogenic activity in the in vivo cytogenetic tests involving rat bone marrow cells. In the plate incorporation (Ames test) and mitotic gene conversion and DNA damage assays, positive results for D3 were not observed with or without metabolic activation. In the forward gene mutation assay (L5178Y mouse lymphoma cells), D3 did not produce a positive response.
In the SCE assay (L5178Y mouse lymphoma cells), D3 was genotoxic and a dose response was observed with and without metabolic activation. In the chromosome aberrations assay (L5178Y mouse lymphoma cells), D3 was genotoxic with, but not without, metabolic activation. D3 did not produce positive results in the DNA/alkaline elution assay.
Cyclotetrasiloxane (D4)
Cyclotetrasiloxane is the cyclic dimethyl polysiloxane that conforms to the generic structure of cyclic dimethyl polysiloxane compounds, where n = 4, and the other components of cyclomethicone (where n = 5, 6, or 7) are present at levels of less than 1%. Cyclotetrasiloxane functions as a hair conditioning agent, skin conditioning agent—emollient, and solvent in cosmetic products. Frequency of use data provided by FDA in 2009 indicate that this ingredient is being used in 56 cosmetic products. Data provided by the Personal Care Products Council in 2008 indicate that cyclotetrasiloxane has been used at concentrations up to 28%.
Both D3 and D5 have been listed as impurities/accompanying contaminants of D4. The results of in vivo and in vitro studies indicate that D4 is percutaneously absorbed to a very small extent. Other results showed that the absorption of D4 in animals after oral dosing was significanly influenced by the carrier; D4 in corn oil was readily absorbed. In inhalation studies involving Fischer 344 rats of both sexes, 14C-D4 was excreted in the expired air, urine, and feces; the expired air and urine appeared to have been the primary routes of excretion in most of the studies. High-exposure animals excreted more radioactivity in the expired volatiles and CO2 than did low-exposure animals in 1 study, and female Fischer-344 rats appeared to have metabolized D4 more readily than did female Sprague-Dawley rats in another study.
Following repeated IV dosing with 14C-D4, there was substantial accumulation in tissues; the main areas of concentration of radioactivity were the fat tissue, liver, and kidneys. In another IV dosing study, administered 14C-D4 was excreted in the urine primarily as dimethylsilanediol and methylsilanetriol.
The PBPK models that characterize the pharmacokinetics of D4 following various routes of exposure have been developed.14C-D4 was metabolized by human liver microsomes in vitro to a single metabolite, named M8. Though the conversion of D4 to M8 did not exceed 10%, M8 formation was not proportional to protein concentration or the incubation time.
D4 induced weak estrogenic and antiestrogenic activity that was several orders of magnitude less potent than EE, and many times less potent than the weak phytoestrogen CE in a study involving Sprague-Dawley rats and Fischer-344 rats. In OVEX αERKO mice, uterine wet weights were significantly increased in a dose-dependent manner following exposure to oral doses of D4. Similarly, uterine peroxidase activity (marker for estrogenic activity) was significantly increased following oral exposure. Inhalation exposure to D4 at high concentrations attenuated the preovulatory surge and significantly decreased the proportion of female rats that ovulated. D4 was also found to act as a pituitary dopamine receptor agonist (D2 receptor) using an in vivo Fischer-344 rat model.
Sprague-Dawley rats injected with D4 did not show signs of arthritis; however, the incidence of arthritis in rats tested with 1% in silicone oil was 1 in 10 rats.
A study was conducted to determine whether the components of the silicone breast implant (silicone oil, silicone gel, and D4) potentiate the antibody response to BSA in A/J mice. Results indicated that both D4 and silicone gel potentiated the production of antibodies to BSA. When data from an inhalation study were analyzed as AFCs/spleen or when normalized as AFC/106 splenocytes, there were no differences in immune competence between D4-exposed Fischer-344 rats and room air controls; therefore, D4 did not cause alterations in immune system function. In an in vitro study, D4 and D5 (in a serum-free medium) inhibited PHA-induced proliferation of human peripheral blood mononuclear cells. This inhibitory effect was completely reversed by the addition of small amounts of serum or plasma to the serum-free medium.
In an in vitro study, D4 (in a serum-free medium) inhibited PHA-induced proliferation of human peripheral blood mononuclear cells. This inhibitory effect was completely reversed by the addition of small amounts of serum or plasma to the serum-free medium.
In an acute inhalation toxicity study involving F-344 rats, the LC50 was estimated to be 36 mg/L. In an acute IP toxicity study involving CD-1 mice, significant injury to tissues (ie, histopathologic changes in the liver and lungs, and elevated serum enzymes) was noted.
In a short-term inhalation toxicity study, a statistically significant increase in relative liver weight (treatment related) was reported for female hamsters and male and female mice, but not guinea pigs or rabbits exposed to D4. In another short-term inhalation study, liver weights (F-344 rats) increased in a statistically significant, dose-related manner; thymus and adrenal gland weights were significantly increased in the highest and second highest dose groups. The results of short-term oral toxicity studies did not reveal any treatment-related adverse effects in rats or rabbits.
In subchronic inhalation toxicity studies, a statistically significant increase in liver weight was observed. This finding was reported for male and female rats, mice, and hamsters, but not for rabbits or guinea pigs, exposed to 700 ppm D4. Additionally, this finding was reported in a chronic inhalation toxicity study involving Fischer-344 rats. The primary target organs affected by chronic D4 exposure included the uterus, respiratory tract, kidney, and liver.
Following the injection of D4 into the anterior chamber in rabbits, severe corneal edema and opacification were elicited within 1 day and the symptoms worsened with time. In a 24-month chronic toxicity study, ocular examinations conducted 2 weeks before the rats were killed did not reveal exposure-related ocular lesions.
In reproductive and developmental toxicity studies, disruption of the female rat reproductive cycle, and a reduction in fertility and litter sizes were noted following inhalation exposure to a high concentration of D4 (700 ppm) in Sprague-Dawley rats. High oral doses of D4 (1000 mg/kg per d) in New Zealand White rabbits also resulted in increased postimplantation loss. However, it is important to note that there were no treatment-related malformations at these levels of inhalation/oral exposure. In rats, the prolonged estrous cycles and decreased mating and fertility indices observed following high levels of D4 inhalation exposure were consistent with the suppression of or delaying the LH surge, as well as acceleration of the onset of female reproductive senescence.
In the plate incorporation (Ames test), mitotic gene conversion, and DNA damage assays, positive results for D4 were not observed with or without metabolic activation. Forward gene mutation assay (L5178Y mouse lymphoma cells) results for D4 were negative.
In the SCE assay (L5178Y mouse lymphoma cells), D4 induced a statistically significant response at one dose level, but a dose–response relationship was not obtained. In the chromosome aberrations assay (L5178Y mouse lymphoma cells), D4 was genotoxic, with, but not without, metabolic activation. D4 did not produce positive results in the DNA/alkaline elution assay.
Inhalation exposure to D4 for up to 24 months induced the following effects in male and female rats: increased incidence of endometrial adenomas and endometrial epithelial hyperplasia in the uteri of rats exposed to 700 ppm D4 for 24 months; minimal-to-mild goblet cell hyperplasia in the nasal mucosa of rats exposed to 700 ppm D4 for 12 or 24 months; and minimal hyperplasia of squamous epithelium in the nasal vestibule of rats exposed to 700 ppm D4 for 12 months.
According to model calculations, the percentage of the applied dose of D4 that was absorbed into the systemic circulation of male and female participants following application to the axillae was 0.12% and 0.309%, respectively. More than 83% of the chemical that reached the systemic circulation was eliminated via exhalation within 24 hours. Following inhalation exposure to 14C-D4, 14C-labeled metabolites equivalent to 30% of the D4 uptake, but not D4, were recovered from the urine. The following urinary metabolites were identified: trimethyldisiloxane-1,1,3,3-tetrol; tetramethyldisiloxane-1,3-diol; hexamethyltrisiloxane-1,5-diol; methylsilanetriol; dimethyldisiloxane-1,3,3,3,-teriol; and dimethylsilanediol.
Inhalation exposure to or oral dosing (in corn oil) with D4 did not cause immunotoxic or proinflammatory effects in human participants.
Cyclopentasiloxane (D5)
Cyclopentasiloxane is the cyclic dimethyl polysiloxane that conforms to the generic structure of cyclic dimethyl polysiloxane compounds, where n = 5, and the other components of cyclomethicone (where n = 4, 6, or 7) are present at levels of less than 1%. Cyclopentasiloxane functions as a hair conditioning agent, skin conditioning agent—emollient, and solvent in cosmetic products. Frequency of use data provided by FDA in 2009 indicate that this ingredient is being used in 2459 cosmetic products. Data provided by the Personal Care Products Council in 2008 indicate that cyclopentasiloxane has been used at concentrations up to 93%.
The results of in vivo and in vitro studies indicate that D5 is percutaneously absorbed to a very small extent. Following oral administration to rats, the major D5 urinary metabolites were dimethylsilanediol and methylsilanetriol; parent D5 was not excreted in the urine. In an inhalation study, 14C-D5, was excreted in approximately equal amounts in the urine and feces of male and female Fischer-344 rats, except for male rats in the higher-exposure group, where excretion was greater in the feces than in the urine. In another study, expired air was the primary route of excretion of 14C-D5; methylsilanetriol and dimethylsilanediol were the 2 primary urinary metabolites. The PBPK models that characterize the pharmacokinetics of D5 following inhalation exposure have been developed.
In OVEX αERKO mice, uterine wet weights were not significantly increased in a dose-dependent manner following exposure to oral doses of D5. Similarly, uterine peroxidase activity (marker for estrogenic activity) was not significantly increased following oral exposure. D5 was found to act as a pituitary dopamine receptor agonist (D2 receptor) using an in vivo Fischer-344 rat model.
In an in vitro study, D5 (in a serum-free medium) inhibited PHA-induced proliferation of human peripheral blood mononuclear cells. This inhibitory effect was completely reversed by the addition of small amounts of serum or plasma to the serum-free medium.
In an acute inhalation toxicity study of D5 involving F-344 rats, and LC50 of 8.67 mg/L air was reported. In a short-term inhalation study involving rats at concentrations up to 2.27/3.06 mg/L air, there were no treatment-related effects that were considered to be of toxicological relevance. Short-term inhalation exposure to 120 ppm D5 did not result in treatment-related gross or microscopic changes in rats. Based on microscopic findings from a short-term inhalation study (rats, concentrations up to 160 ppm), the respiratory tract is the primary target organ for D5 inhalation exposure. The NOAEL for systemic toxicity was 75 pm in this study.
No treatment-related deaths or overt signs of toxicity were observed in groups of Sprague-Dawley rats dosed orally with D5 in a short-term oral toxicity study. However, a statistically significant increase in liver weight was noted. In a short-term dermal toxicity study on D5 involving Sprague-Dawley rats, no significant toxicological effects were observed. Neither gross nor microscopic treatment-related changes were observed in the tissues/organs of Sprague-Dawley rats following subchronic inhalation exposure to D5.
Following the injection of D5 into the anterior chamber in rabbits, severe corneal edema and opacification were elicited within 1 day and the symptoms worsened with time. In a 24-month chronic toxicity study, ocular examinations conducted 2 weeks before the rats were killed did not reveal exposure-related ocular lesions.
No exposure-related findings were noted at necropsy of F1 pups on PNDs 21 and 28 in a reproductive toxicity study on D5 involving Sprague-Dawley Crl:CD7BR rats.
In the plate incorporation (Ames test), mitotic gene conversion, and DNA damage assays, positive results for D5 were not observed with or without metabolic activation. Forward gene mutation assay (L5178Y mouse lymphoma cells) results for D5 were negative.
In the SCE and chromosome aberrations assays (L5178Y mouse lymphoma cells), D5 was not genotoxic and did not produce positive results in the DNA/alkaline elution assay. D5 induced neither DNA damage leading to increased repair synthesis in the hepatocytes of treated rats nor induced micronuclei.
The inhalation carcinogenicity of D5 was evaluated using Fischer-344 rats. The increased incidence (statistically significant) of endometrial adeocarcinomas of the uterus in high-dose rats following 2 years of exposure may have been exposure related, in that no such neoplasms were diagnosed in control rats. However, it was noted that these neoplasms also occur occasionally in control rats, making the relationship to exposure unclear.
According to model calculations, the percentage of the applied dose of D5 that was absorbed into the systemic circulation of male and female participants following application to the axillae was approximately 0.05%. More than 83% of the chemical that reached the systemic circulation was eliminated via exhalation within 24 hours.
The following products dd not induce skin irritation nor sensitization in RIPTs (semiocclusive/occlusive patches) involving human participants: undiluted heat protection hair spray (leave-on) product containing 90.37% D5; undiluted heat protection hair spray (leave-on) product containing 89.75% D5; undiluted deodorant containing 56.3% D5; and an undiluted antiperspirant containing 55.76% D5.
A safety assessment was conducted to evaluate the potential hazard of D5 to various populations. This assessment was based on exposures to D5 via inhalation and dermal routes. It was concluded that typical exposure to D5, whether occupationally, or through the use of D5-containing consumer products, or to the general public, would not result in a significant human health hazard.
Cyclohexasiloxane (D6)
Cyclohexasiloxane is the cyclic dimethyl polysiloxane that conforms to the generic structure of cyclic dimethyl polysiloxane compounds, where n = 6, and the other components of cyclomethicone (where n = 4, 5, or 7) are present at levels of less than 1%. Cyclohexasiloxane functions as a hair conditioning agent, skin conditioning agent—emollient, and solvent in cosmetic products. Frequency of use data provided by FDA in 2009 indicate that this ingredient is being used in 618 cosmetic products. Data provided by the Personal Care Products Council in 2008 indicate that cyclohexasiloxane has been used at concentrations up to 48%.
The results of an in vitro study indicate that D6 was not percutaneously absorbed. Following oral administration to rats, most of the administered dose was excreted in the feces.
In both acute oral and acute dermal toxicity studies involving Wistar rats, the acute oral LD50 was >2000 mg/kg.
No treatment-related deaths or overt signs of toxicity were observed in groups of Sprague-Dawley rats dosed orally with D6 in a short-term oral toxicity study.
Neither iridial irritation nor corneal opacity was observed following the instillation of D6 into the eyes of albino rabbits. A 4-hour dermal application of D6 to albino rabbits did not induce irritation, corrosion, or staining. In the maximization test, D6 was not classified as a sensitizer in guinea pigs challenged with undiluted D6, 50% D6, or 20% D6.
In a combined repeated oral gavage toxicity study that included reproductive/developmental toxicity screening, the NOAEL for D6 was 1000 mg/kg per d.
D6 was not mutagenic in the Salmonella typhimurium reverse mutation assay or the E coli reverse mutation assay.
Cycloheptasiloxane (D7)
Cycloheptasiloxane is the cyclic dimethyl polysiloxane that conforms to the generic structure of cyclic dimethyl polysiloxane compounds, where n = 7, and the other components of Cyclomethicone (where n = 4, 5, or 6) are present at levels of less than 1%. Cycloheptasiloxane functions as an anticaking agent, skin conditioning agent—emollient, and solvent in cosmetic products. Frequency of use data provided by FDA in 2009 indicate that this ingredient is being used in 4 cosmetic products. Data provided by the Personal Care Products Council in 2008 did not include a use concentration for cycloheptasiloxane.
After CD-1 mice were injected SC with breast implant distillate consisting primarily of low-molecular-weight cyclosiloxanes, D7 was the only cyclosiloxane that was not detectable in organs (heart, kidney, and lungs included).
Discussion
While the biological activity of individual chain length cyclic dimethyl siloxanes appears to be related to chain length, the CIR Expert Panel recognized that there are data for cyclomethicone,a mixture of cyclic dimethyl siloxanes, used as an ingredient in cosmetic formulations. Consequently, studies on cyclomethicone have been included in this review. The Expert Panel considers that the safety test data on the individual chain length siloxanes (D4-D6) can be used to support the safety of that particular chain length cyclic dimethyl siloxane as well as cyclomethicone, currently defined as a mixture of D4-D6 siloxanes.
The Expert Panel noted that cycloheptasiloxane (D7) was no longer included in the definition of cyclomethicone but was still listed as a separate cosmetic ingredient in the International Cosmetic Ingredient Dictionary and Handbook. The Expert Panel recognizes that cyclotrisiloxane (D3) is not a cosmetic ingredient. However, the available data indicate that D3 may be of a low level impurity of other cyclic dimethyl siloxanes. For these reasons, the Expert Panel determined that safety test data on D3 should be included in this safety assessment. At the level of this impurity that was found, the Panel determined that there was no safety risk.
The Expert Panel is aware of the voluminous data on rheumatologic effects resulting from the injection/insertion of silicone into the body. However, the Panel noted that these data would not be relevant to the current cosmetic uses of cyclomethicones. The Expert Panel also noted that the results of a single study on silicone gel showed that cyclotetrasiloxane (D4, 1% in silicone oil), but not D4 alone, induced arthritis in rats.
The Expert Panel also noted that the ingredients reviewed in this safety assessment are lipophilic compounds with a low blood:air partition coefficient, and, therefore, that it is not likely that they would accumulate in blood or tissues.
The reproductive toxicity (nondose-related) that was noted in rats after inhalation exposure to a high concentration of D4 was attributed to perturbation of the estrous cycle, due to a delay in the LH surge, which is not relevant to humans. Reproductive and developmental toxic effects were observed in rats after inhalation exposure to D3 at a high concentration. The Expert Panel noted that it would not be expected that these effects would result from exposure to cyclomethicone, D4, cyclopentasiloxane (D5), cyclohexasiloxane (D6), or cycloheptasiloxane (D7) in personal care products. Furthermore, dermal exposure to these ingredients during product use would not likely result in any significant systemic exposure.
The Expert Panel is also aware of an inhalation study in which Fischer-344 rats exposed to 160 ppm D5 over a 2-year period had a statistically significant increase in endometrial adenocarcinomas of the uterus. However, it was noted that these neoplasms occur occasionally in control rats, making the relationship to exposure unclear. The Expert Panel noted the potential ingestion of D5 from a lipstick. Oral toxicity data were not available, but exposure to D5 from lipstick would not likely be higher when compared to inhalation and dermal routes. The available inhalation and dermal toxicity data are interpreted to provide a high MOS when compared to possible exposures from cosmetic products containing D5.
In addition to the available inhalation toxicity data, the Panel determined that cyclomethicone and D6 can be used safely in hair sprays, because the ingredient particle size is not respirable. The Panel reasoned that the particle size of aerosol hair sprays (38 μm) and pump hair sprays (>80 μm) is large, compared to respirable particulate sizes (<10 μm).
Conclusion
The CIR Expert Panel concluded that cyclomethicone, cyclotetrasiloxane, cyclopentasiloxane, cyclohexasiloxane, and cycloheptasiloxane are safe as cosmetic ingredients in the practices of use and concentration as described in this safety assessment.
Footnotes
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St., Suite 412, Washington, DC 20036, USA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
