Abstract
We newly synthesized organic selenium compounds (5-membered ring compounds) including 2-selenoxo-1,3-thiazolidin-4-ones (compounds A) and 3-alkoxy-4,5-dihydro-5-selenoxo-1H-1,2,4-triazole-1-carboxylates (compounds B). To address whether these compounds show antioxidative effects, we also examined their superoxide radical (O2
−)-scavenging effects. Moreover, we examined the effects of compound A
Keywords
Reactive oxygen species
1
including superoxide (O2
−), hydrogen peroxide (H2O2), and hydroxyl radicals (
Superoxide dismutases (SODs), catalase, glutathione peroxidases (GPXs), and some vitamins are representative antioxidants that protect against reactive oxygen species toxicity in cells. 4,5 Glutathione peroxidases are believed to be an important antioxidant enzyme and effectively reduce the toxicity of H2O2 in vitro and in vivo. 5,9 Furthermore, the active domain of GPX essentially contains selenium atoms. 9 Various studies have reported that selenoproteins, including GPX, reduce oxidative stress in cells. 10,11 Thus, various organic selenium compounds may be candidates for reactive oxygen species scavengers.
Ebselen is a 5-membered ring selenium-containing heterocyclic compound showing GPX-like activity 12 and is a synthetic antioxidant with reactive oxygen species scavenger effects. 13 The antioxidant effects of ebselen are due to its selective blockade of leukocyte infiltration and activation, leading to the elimination of H2O2. 13 This compound is a multifunctional antioxidant and a potential chemopreventive agent in inflammation-associated carcinogenesis. 14 As such, various types of organic selenium compounds may be applicable to the reduction of oxidative stress 15 –17 and may be used as antioxidative/anti-inflammatory substances for therapeutic drugs against inflammatory diseases (ie, atopic dermatitis) and stroke. 13 Morey et al demonstrated that some selenoproteins, such as selenoprotein P, regulate the redox potential in cells, resulting in the modulation of various phosphorylation pathways including Ras/mitogen-activated protein kinase (MAPK) signaling. 18 On the other hand, we demonstrated that ebselen also influenced the redox potential in rat pheochromocytoma cells (PC12 cells) and induced activation of MAPK/extracellular signal-regulated protein kinases ([ERK1/2], MAPK/ERK1/2) and neural differentiation via regulation of kinases or phosphatases involved in intracellular signaling. 19 Thus, such selenium compounds are thought to play roles not only as reactive oxygen species scavengers but also as modulators of intracellular signaling.
Against this research background, we sought to synthesize various organic selenium compounds, such as selenoamides,
17
thio- and selenoureas,
16
and selenocarbamates,
15
in order to evaluate their biological potential. It is already known that most of these compounds are also highly efficient in vitro O2
− scavengers.
15,16
Selenoureas in particular showed pronounced antiinflammatory effects with low toxicity in various human cell lines.
20
In this study, we newly synthesized 5-membered ring compounds such as 2-selenoxo-1,3-thiazolidin-4-one (compound A and its analogues: compounds A
Materials and Methods
Materials
Compounds A
Synthetic methods for the preparation of compounds Aa-d
A round-bottom flask equipped with a condenser and a magnetic stirrer was charged with an aryl isoselenocyanate (1.0 mmol) in 20 mL of a mixture of ethyl alcohol and water (2:1). An equimolar amount of the respective mercapto carboxylic acid was then added dropwise. The mixture was stirred at room temperature for several hours before being poured into 50 mL of cold water. After 2 hours of stirring, the precipitate was filtered and purified, if necessary, by recrystallization from ethyl alcohol.
3-(2,6-Dimethylphenyl)-2-selenoxo-1,3-thiazolidin-4-one (compound Aa)
Yield 56%. Orange powder. Mp: 123°C to 125°C. 1 H-NMR (300 MHz, CDCl3): δ 2.10 (s, 2 Me), 3.95 (s, CH2), 7.18–7.33 (m, 3 arom. H). 13C-NMR (75 MHz, CDCl3): δ 17.5, 37.3, 127.5, 127.9, 130.0, 136.1, 173.2, 200.2. CI-MS: 303 (60, [M+NH4]+), 286 (100, [M+H]+). Theoretically calculated values were for C11H11NOSSe: C, 46.48; H, 3.90; N, 4.93; S, 11.28. Found: C, 46.33; H, 4.12; N, 5.00; S, 11.32.
3-(4-Methylphenyl)-2-selenoxo-1,3-thiazolidin-4-one (compound Ab)
Yield 91%. Mp: 201°C to 203°C (ethyl alcohol). 1 H-NMR (300 MHz, CDCl3): δ 2.42 (s, Me), 3.90 (s, CH2), 7.10, 7.35 (AA′BB′, J = 8.2, 4 arom. H). 13C-NMR (75 MHz, CDCl3): δ 21.3, 37.4, 128.0, 130.3, 133.2, 140.0, 173.2, 203.3. CI-MS: 289 (31, [M+NH4]+), 272 (100, [M+H]+). Theoretically calculated values were for C10H9NOSSe: C, 44.45; H, 3.36; N, 5.18; S, 11.87. Found: C, 44.30; H, 3.21; N, 5.03; S, 11.78.
3-(4-Methoxyphenyl)-2-selenoxo-1,3-thiazolidin-4-one (compound Ac)
Yield 93%. Mp: 182°C to 184°C (ethyl alcohol). 1 H-NMR (300 MHz, CDCl3): δ 3.85 (s, MeO), 3.89 (s, CH2), 7.03, 7.14 (AA′BB′, J = 8.2, 4 arom. H). 13C-NMR (75 MHz, CDCl3): δ 37.3, 55.4, 114.9, 128.2 (1 arom. C), 129.4, 160.3, 173.3, 203.6. CI-MS: 305 (32, [M+NH4]+), 288 (100, [M+H]+). Theoretically calculated values were for C10H9NO2SSe: C, 41.96; H, 3.17; N, 4.89; S, 11.20. Found: C, 42.23; H, 3.33; N, 4.88; S, 11.12.
3-(4-Bromophenyl)-2-selenoxo-1,3-thiazolidin-4-one (compound Ad)
Yield 89%. Mp: 111°C to 113°C (ethyl alcohol). 1 H-NMR (300 MHz, CDCl3): δ 3.92 (s, CH2), 7.11, 7.67 (AA′BB′, J = 8.1, 4 arom. H). 13C-NMR (75 MHz, CDCl3): δ 37.4, 123.3, 130.0, 132.9, 138.5, 171.8, 203.8. CI-MS: 353 (35, [M+NH4]+), 336 (100, [M+H]+). Theoretically calculated values were for C9H6NOSSeBr: C, 32.26; H, 1.80; N, 4.18; S,9.57. Found: C, 32.41; H, 2.02; N, 3.98; S, 10.00.
Synthetic methods for the preparation of compounds Be-j
A 25-mL round-bottom flask equipped with magnetic stirrer and condenser was charged with a mixture of diethyl or bis(tert-butyl) azodicarboxylate (0.92 mL, 2.0 mmol) and Ph3P (524 mg, 2.0 mmol) in dichloromethane (20 mL). The mixture was stirred under N2 atmosphere at 0°C (ice bath) for 30 minutes. The appropriate amount of isoselenocyanate (2.0 mmol) was added in 1 portion. The mixture was stirred for 15 hours at room temperature and then evaporated to dryness under reduced pressure. The crude product was purified by column chromatography on SiO2 using hexane/ethyl acetate (100/0 to 50/50) as the eluent and recrystallized in ethyl acetate.
Ethyl 3-ethoxy-4,5-dihydro-4-phenyl-5-selenoxo-1H-1,2,4-triazole-1-carboxylate (compound Be)
Yield: 552 mg (81%). Yellowish crystals. Mp: 142°C to 144°C (ethyl acetate). IR (cm−1): 3422w (br), 2981w, 2931w, 1773s, 1619s, 1595w, 1501w, 1451m, 1386m, 1368m, 1328s, 1308s, 1220s, 1175w, 1155w, 1109w, 1088w, 1066w, 1028m, 1005m, 979w, 902w, 861w, 847w, 776w, 710w, 689w. 1 H-NMR (300 MHz, CDCl3): δ 1.36 (t, J = 7.1, CH3), 1.49 (t, J = 7.1, CH3), 4.49 to 4.62 (m, 2 CH2), 7.35 (d, J = 8.1, 2 arom. H), 7.52 to 7.59 (m, 3 arom. H). 13C-NMR (75 MHz, CDCl3): δ 14.0, 14.1, 65.0, 68.5, 128.1, 129.4, 130.0, 132.6, 148.2, 156.3, 167.8. ESI-MS: 360 (14), 361 (13), 362 (52), 363 (3), 364 (100, [M+Na]+), 365 (11), 366 (15). Theoretically calculated values were for C13H15N3O3Se: C, 45.89; H, 4.44; N, 11.86. Found: C, 45.58; H, 4.58; N, 12.64.
Ethyl 3-ethoxy-4,5-dihydro-4-(4-methylphenyl)-5-selenoxo-1H-1,2,4-triazole-1-carboxylate (compound Bf)
Yield: 554 mg (78%). Yellowish crystals. Mp 120°C to 122°C (ethyl acetate). IR (cm−1): 3442w (br), 2979w, 2956w, 1768s, 1619s, 1514m, 1477w, 1454m, 1389m, 1368m, 1327s, 1307s, 1294s, 1220s, 1173w, 1152w, 1109w, 1099w, 1064w, 1029m, 981w, 905w, 850w, 817w, 753w, 712w, 622w. 1 H-NMR (300 MHz, CDCl3): δ 1.37 (t, J = 7.1, CH3), 1.49 (t, J = 7.1, CH3), 2.43 (s, CH3), 4.48 to 4.60 (m, 2 CH2), 7.22, 7.34 (AA′BB′, J = 8, 4 arom. H). 13C-NMR (75 MHz, CDCl3): δ 14.0, 14.3, 21.3, 65.0, 68.4, 127.7, 130.0, 127.3, 140.2, 149.0, 156.0, 167.1. ESI-MS: 374 (12), 375 (14), 376 (48), 377 (4), 378 (100, [M+Na]+), 379 (11), 380 (13), 733 (3). Theoretically calculated values were for C14H17N3O3Se: C, 47.46; H, 4.84; N, 11.86. Found: C, 47.25; H, 4.85; N, 11.85.
Ethyl 3-ethoxy-4-(4-fluorophenyl)-4,5-dihydro-5-selenoxo-1H-1,2,4-triazole-1-carboxylate (compound Bg )
Yield: 603 mg (84%). Yellowish crystals. Mp 151°C to 153°C (ethyl acetate). IR (cm−1): 3442w (br), 3085w, 2982w, 1769s, 1621s, 1512s, 1475w, 1454m, 1386m, 1367m, 1311s, 1290s, 1216s, 1171w, 1150w, 1112w, 1088w, 1063m, 1024m, 1004w, 977m, 905w, 853m, 821w, 759w, 725w, 711w, 635w, 622m. 1 H-NMR (300 MHz, CDCl3): δ 1.35 (t, J = 7.1, CH3), 1.49 (t, J = 7.1, CH3), 4.41 to 4.51 (m, 2 CH2), 6.95, 7.19 (AA′BB′, J = 8, 4 arom. H). 13C-NMR (75 MHz, CDCl3): δ 14.0, 14.3, 65.1, 68.6, 116.4, 130.1, 128.4, 148.9, 155.8, 162.3, 167.1. ESI-MS: 378 (15), 379 (12), 380 (55), 381 (4), 382 (100, [M+Na]+), 383 (15), 384 (17). Theoretically calculated values were for C13H14N3O3SeF: C, 43.59; H, 3.94; N, 11.73. Found: C, 43.37; H, 4.00; N, 11.88.
Ethyl 4-(4-chlorophenyl)-3-ethoxy-4,5-dihydro-5-selenoxo-1H-1,2,4-triazole-1-carboxylate (compound Bh )
Yield: 726 mg (97%). Yellowish crystals. Mp 142°C to 144°C (ethyl acetate). IR (cm−1): 3442w (br), 2982w, 1765s, 1625s, 1495m, 1469w, 1442w, 1373m, 1310s, 1289s, 1218s, 1173w, 1148w, 1089m, 1025m, 977m, 907w, 836w, 753w, 703w. 1 H-NMR (300 MHz, CDCl3): δ 1.31 (t, J = 7.1 Hz, CH3), 1.41 (t, J = 7.1, CH3), 4.39 to 4.51 (m, 2 CH2), 7.24, 7.43 (AA′BB′, J = 8, 2H, 4 arom. H). 13C-NMR (75 MHz, CDCl3): δ 14.1, 14.3, 65.1, 68.7, 124.6, 129.5, 129.7, 131.0, 148.9, 155.6, 166.9. ESI-MS: 394 (13), 395 (14), 396 (51), 397 (4), 398 (100, [M+Na]+), 399 (18), 400 (49). Theoretically calculated values were for C13H14N3O3SeCl: C, 41.67; H, 3.77; N, 11.21. Found: C, 41.63; H, 3.84; N, 11.34.
Ethyl 4-(4-bromophenyl)-3-ethoxy-4,5-dihydro-5-selenoxo-1H-1,2,4-triazole-1-carboxylate (compound Bi )
Yield: 686 mg (82%). Yellowish crystals. Mp 147°C to 149°C (ethyl acetate). IR (cm−1): 3444w (br), 2982w, 2935w, 1755s, 1718m, 1638s, 1591w, 1493m, 1451m, 1392m, 1366m, 1327s, 1303s, 1286s, 1215w, 1170w, 1154w, 1104w, 1068w, 1030m, 1011m, 988w, 909w, 850w, 754w, 711w. 1 H-NMR (300 MHz, CDCl3): δ 1.31 (t, J = 7.1, CH3), 1.41 (t, J = 7.1, CH3), 4.41 to 4.51 (m, 2 CH2), 7.20, 7.58 (AA′BB′, J = 8, 4 arom. H). 13C-NMR (75 MHz, CDCl3): δ 14.1, 14.3, 65.1, 68.7, 124.2, 129.8, 132.7, 131.5, 148.9, 155.6, 166.8. ESI-MS: 438 (15), 439 (12), 440 (55), 441 (4), 442 (100, [M+Na]+), 443 (14), 444 (12). Theoretically calculated values were for C13H14N3O3SeBr: C, 37.25; H, 3.37; N, 10.03. Found: C, 37.12; H, 3.23; N, 9.99.
Tert-Butyl-3-(tert-butoxy)-4-(4-bromophenyl)-4,5-dihydro-5-selenoxo-1H-1,2,4-triazole-1-carboxylate (compound Bj )
Yield: 855 mg (90%). Yellowish crystals. Mp 118°C to 120°C (ethyl acetate). IR (cm−1): 3443w (br), 2985w, 2931w, 1764s, 1701w, 1612s, 1488m, 1453w, 1408w, 1372m, 1337m, 1320s, 1297m, 1278s, 1223m, 1143s, 1065w, 1000m, 895w, 848m, 836m, 804w, 761w, 655w. 1 H-NMR (300 MHz, CDCl3): δ 1.53 (s, 3 CH3), 1.67 (s, 3 CH3), 7.21, 7.62 (AA′BB′, J = 8, 4 arom. H). 13C-NMR: δ 27.6, 27.8, 86.6, 88.1, 123.8, 129.8, 132.5, 132.1, 147.1, 153.5, 165.1. ESI-MS: 494 (17), 495 (15), 496 (55), 497 (25), 498 (100, [M+Na]+), 499 (18), 500 (75), 501 (12), 502 (10). Theoretically calculated values were for C17H22N3O3SeBr: C, 42.96; H, 4.67; N, 8.83. Found: C, 43.22; H, 4.95; N, 9.15.
Assay of superoxide anion-scavenging activities
The superoxide anion-scavenging activities of compounds A
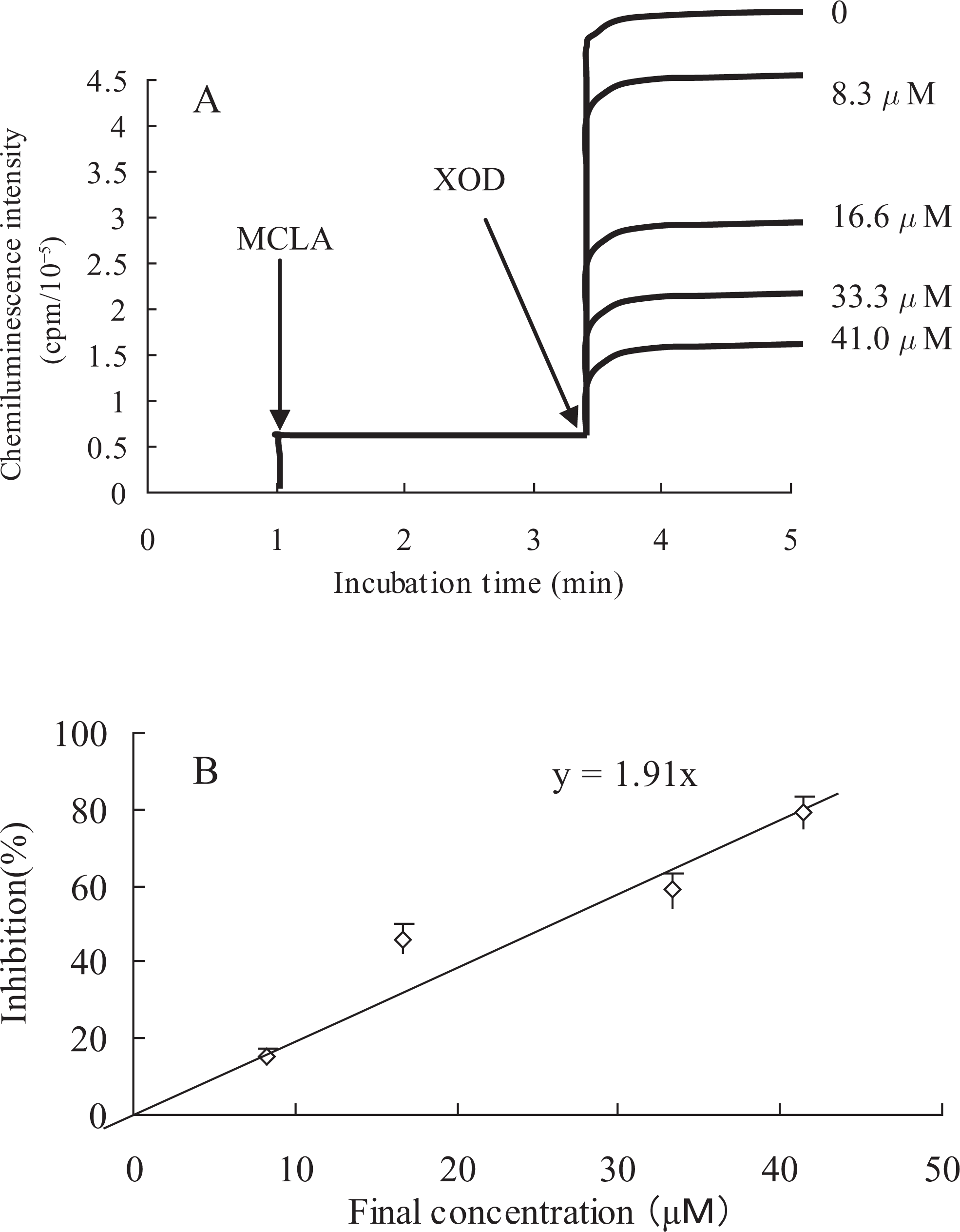
Effect of compound A
Measurement of Uric Acid Generation in Hypoxanthine–Xanthine Oxidase System
The amounts of uric acid were measured using a uric acid measurement kit (L type WAKO UA M; Wako Pure Chemical Industries, Ltd).
Cell Culture
In the present study, we used rat PC12 cells because previous reports suggested that this cell line is a useful tool as various models of neurological dysfunctions. 24 Pheochromocytoma cells were cultured as described previously. 25 In brief, the cells were maintained in Dulbecco modified Eagle medium (DMEM; Sigma) supplemented with 10% heat-inactivated horse serum ([HS] Gibco BRL, Grand Island, New York) and 5% heat-inactivated fetal bovine serum ([FBS] Sanko Junyaku, Co, Ltd, Tokyo, Japan; serum-containing medium) or in DMEM supplemented with 1% bovine serum albumin ([BSA] serum-free medium).
Detection of Phosphorylated Proteins
Each compound was suspended in the serum-free medium and sonicated until fully emulsified. Pheochromocytoma cells were seeded at 2 × 106 cells per well onto collagen-coated 6-well plates (Corning Incorporated Life Sciences, Lowell, Massachusetts) in the serum-containing medium and precultured for 2 days at 37°C in an atmosphere of 95% air/5% CO2. The cells were then washed with phosphate-buffered saline (PBS) and incubated with the above-mentioned culture medium containing the various agents shown in Figure 2 for 10 minutes at 37°C. The culture plates were then placed on ice and each well was washed with 3 mL of 2 mmol/L Tris-HCl buffer (pH 8.0) containing 0.33 mol/L NaF and 6.25 mol/L Na3VO4 and subsequently lysed with 150 μL of 20 mmol/L Tris-HCl buffer (pH 8.0) containing 150 mmol/L NaCl, 2 mmol/L EDTA, 1% Nonidet P-40 (w/v), 1% sodium deoxycholate (w/v), 0.1% sodium dodecyl sulfate (SDS; w/v), 50 mmol/L NaF, 0.1% aprotinin (w/v), 0.1% leupeptin (w/v), 1 mmol/L Na3VO4, and 1 mmol/L phenylmethylsulfonylfluoride (PMSF). Cell lysates were collected using a cell scraper and centrifuged at 15 000g for 30 minutes at 4°C. The supernatant was collected, and the overall protein concentration was determined by a BCA Protein Assay Reagent Kit (Pierce, Rockford, Illinois) with BSA as the standard.
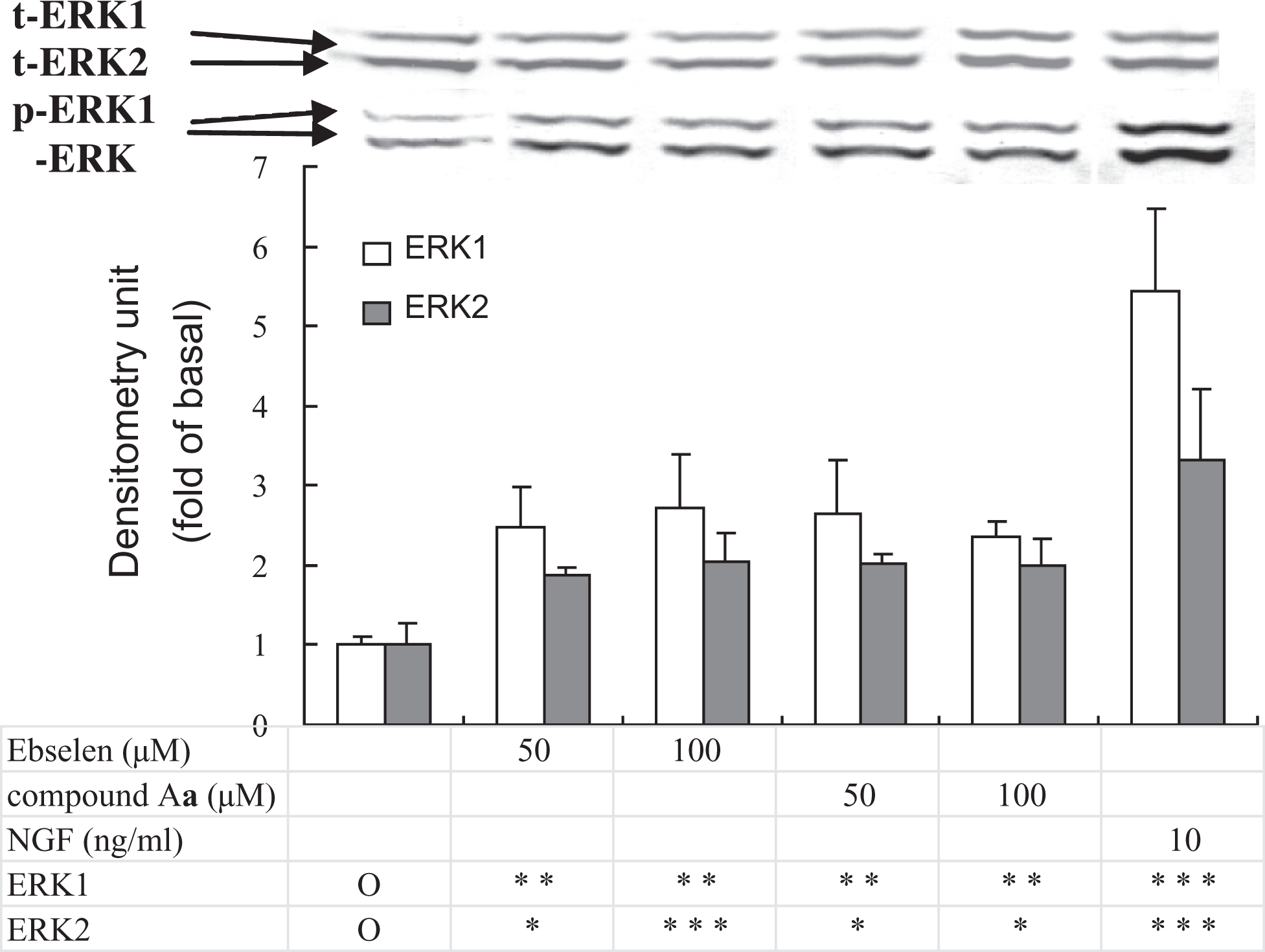
Effects of compound A
Supernatant fluids containing proteins (20 μg) were mixed with lithium dodecyl sulfate (LDS) sample buffer (Invitrogen Corp, Carlsbad, California) and incubated for 5 minutes at 80°C. Proteins in samples were separated on SDS-polyacrylamide gel electrophoresis, and the proteins in gels were electroblotted onto polyvinylidene fluoride (PVDF) filters (Fluorotrans membrane W, 0.2 μm; Nihon Genetics, Tokyo, Japan). Immunoblotting analysis was performed using monoclonal antibodies against p44/42 ERK or phospho p44/42 ERK (Cell Signaling Technology, Lake Placid, New York) as primary antibodies, followed by reaction with horseradish peroxidase-conjugated anti-rabbit or anti-mouse immunoglobulin G (IgG) antibodies from Promega Co (Fitchburg, Wisconsin) as the secondary antibody. The blots were developed by the enhanced chemiluminescence method (Hyperfilm-ECL plus; Amersham Biosciences Corp, Piscataway, New Jersey).
The Measurement of Cytotoxicity by 5-Dimethyl-2-Thiazolyl)-2,5-Diphenyl-2-Htetrazolium Bromide Assay
Pheochromocytoma cells were cultured onto collagen-coated 96-well plates (2 × 106 cells per well) in serum-containing medium for 2 days at 37°C in an atmosphere of 95% air/5% CO2. Culture medium was replaced with 50 μL of the serum-free medium containing each test agent after washing with PBS, and the cells were cultured with each test agent for a range of days. The cytotoxicity was determined by the 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) reduction assay. 26 The cells were incubated with 0.25 ng of MTT/mL (final concentration) for 2 hours, and the reaction was stopped by adding 50 μL of 50% (volume/volume [v/v]) dimethylformamide (DMFA) containing 20% (w/v) SDS. The amount of MTT formazan product was determined photometrically using a microplate reader (Model Ultrospec Visible Plate Reader II of Amersham Biosciences, Tokyo, Japan) by measuring the absorbance at 562 nm with a reference wavelength of 630 nm. The relationships between absorbance and cell numbers were clarified, and the number of cells in each well was calculated.
Measurement of H2O2 Concentrations
The concentration of H2O2 was determined by the horseradish peroxidase-catalyzed oxidation of fluorescent scopoletin method with minor modifications.
27
Briefly, compound A
Statistical Analysis
The results were expressed as means ± standard deviation (SD). The significant differences between the groups compared were determined using analysis of variance (ANOVA) followed by Dunnett test.
Results
Superoxide Anion-Scavenging Activities of Compounds A and B and Ebselen
The chemical structures, superoxide anion-scavenging activities, and IC50 values of compounds A
Chemical Structures, Superoxide Anion-Scavenging Activities, and IC50 Values of 2-Selenoxo-1,3-Thiazolidin-4-Ones 1, 3-Alkoxy-4,5-Dihydro-5-Selenoxo-1H-1,2,4-Triazole-1-Carboxylates and Ebselen a
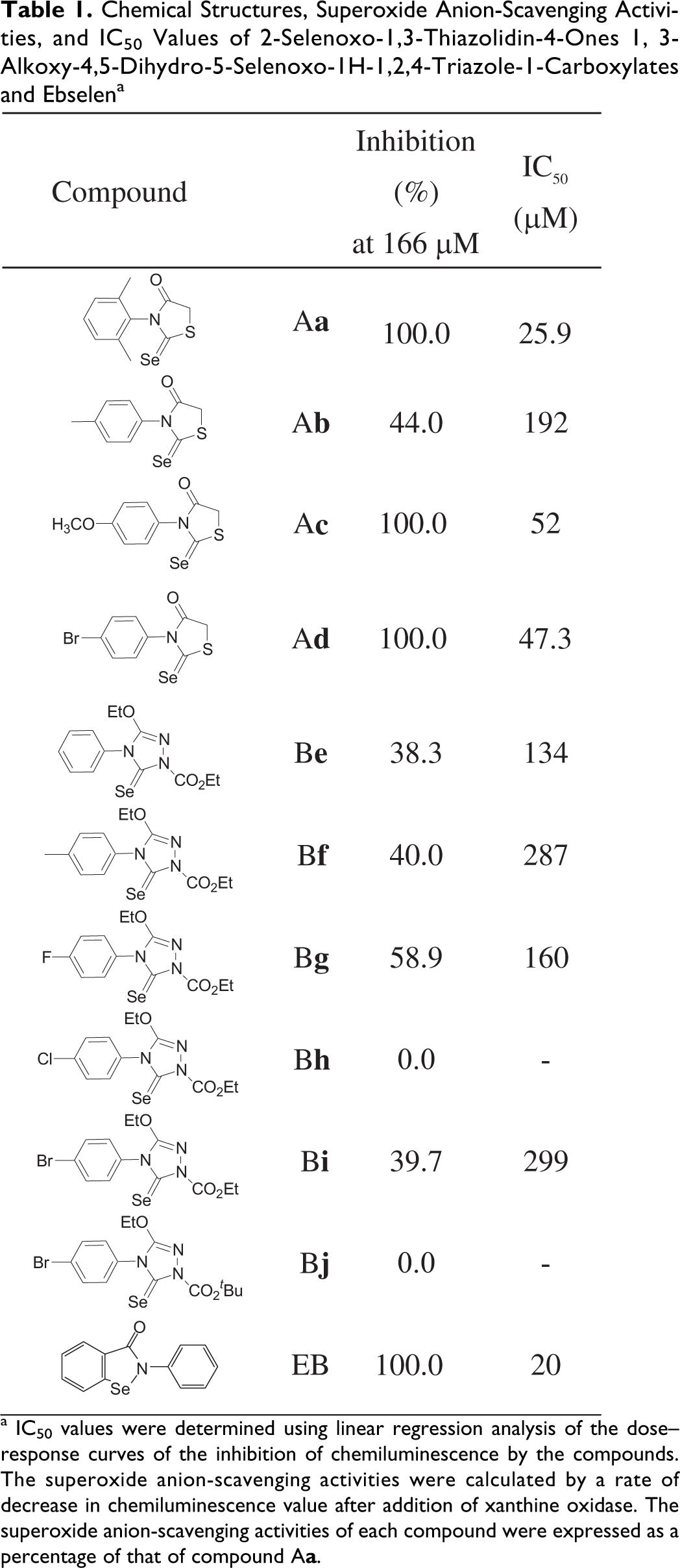
a IC50 values were determined using linear regression analysis of the dose–response curves of the inhibition of chemiluminescence by the compounds. The superoxide anion-scavenging activities were calculated by a rate of decrease in chemiluminescence value after addition of xanthine oxidase. The superoxide anion-scavenging activities of each compound were expressed as a percentage of that of compound A
We investigated whether compounds A
Effects of Compound Aa , Ebselen, or Nerve Growth Factor on the Activation of ERK1/2
The activation of ERK1/2 is one of the checkpoints to assess the activation of the classical Ras/MAPK cascade,
28
which is triggered by an engaged tyrosine kinase receptor or G-protein-coupled receptor and results in proliferation and/or differentiation. We previously confirmed that ebselen is an activator of the Ras/MAPK cascade and an initiator of neuronal differentiation.
25
Therefore, we examined whether compounds A and B could activate ERK1/2 and found that ERK1/2 was activated only by compound A
The effect of ebselen, compound A
Effects of Compound Aa or Ebselen on Survival of PC12 Cells Exposed to H2O2
The oxidative stress-induced cytotoxicity was evaluated by determining the viability of PC12 cells exposed to H2O2 (Figure 3 ). First, we studied the relationship between cell death and H2O2 concentration. Two hours after the addition of 200, 400, 1000, or 2000 μmol/L H2O2, the cell viability was 100%, 36%, 8%, or 0%, respectively. At 400 μmol/L of H2O2, a significant decrease in cell viability was observed, suggesting the potent oxidative stress-induced cytotoxicity of H2O2.
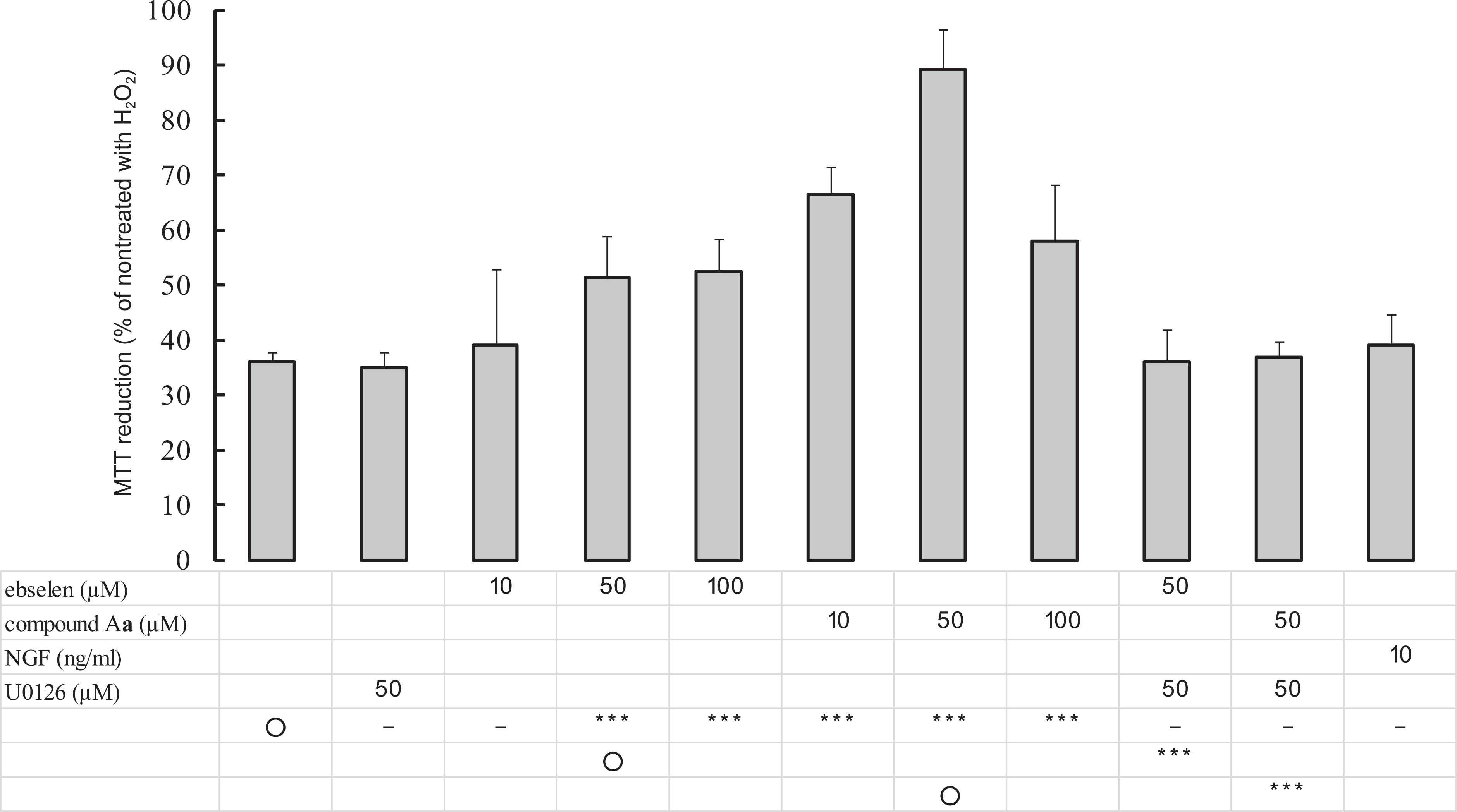
Effects of compound A
Pretreatment of PC12 cells with ebselen or compound A
Effects of Cell Viability of Compound Aa , Ebselen, or NGF on PC12 Cells
Ebselen prevents ischemia- or lead-induced cell death,
1,2
so we compared the effects of compound A
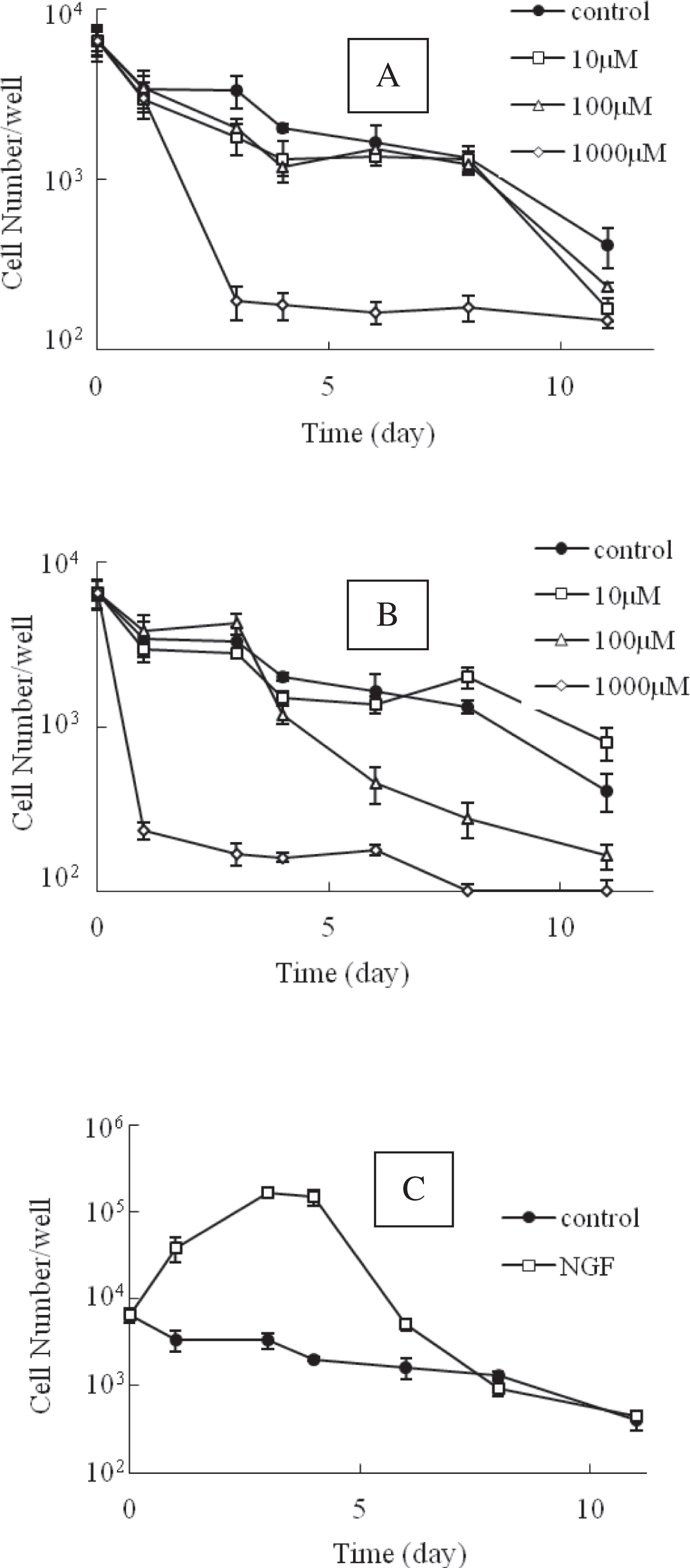
Effects of cell viability of compound A
Discussion
In the present study, we have systematically synthesized various organic selenium compounds including selenocarbamates, selenoureas, thioureas, tertiary selenoamides, and 2-amino-1, 3-selenazoles and bis-(2-amino-5-selenazoyl) ketones and found that these compounds have superoxide anion-scavenging activities, and their IC50 values
15
–17,30,31
range approximately from 0.1 to 100 μmol/L. From the results shown in Table 1, it was confirmed that the superoxide anion-scavenging activities of these newly synthesized compounds, 2-selenoxo-1,3-thiazolidin-4-ones compounds A
A low concentration of O2 − in the human body generally plays a beneficial role in biological defenses and intercellular signal transduction. 5 On the other hand, excessive O2 − production is involved in the pathogenesis of a number of disorders, including inflammation, rheumatoid arthritis, and asthma. 32,33 Oxidative stress may be defined as an imbalance between the cellular production of reactive oxygen species, a key component of inflammation and inflammatory disorders, and antioxidant defense mechanisms. 5 The processes associated with inflammatory responses are complex and often involve the production of reactive oxygen species including O2 −. In this study, our newly synthesized compounds acted as effective O2 − scavengers in vitro. Thus, these compounds may eliminate excessive O2 − and lead to suppression of reactive oxygen species overproduction.
Compound A
It is known that serum deprivation induces apoptosis in PC12 cells.
25
Although NGF increased cell viability under serum-free cultivation, compound A
It was suggested that H2O2 can induce cell death by apoptosis in PC12 cells.
38
In the present study, vesicles were formed and nucleus fragmentation was observed by the addition of Hoechst 33342 solution in the dead cells; therefore, H2O2 (400 μmol/L)-induced cell death in the cells may be apoptosis. On the other hand, serum deprivation induced apoptosis in PC12 cells.
25
Because H2O2-induced cell death was suppressed by A
We examined whether A
Oxidative stress is one of the common causal factors of neuronal cell death.
39
H2O2-induced cytotoxicity was clearly suppressed by compound A
Antioxidants such as ebselen regulate intercellular signaling via regulation of kinase and phosphatase.
19
In this study, we found that compound A
On the other hand, NGF did not suppress H2O2-induced cell death, although ERK1/2 proteins in the PC12 cells were phosphorylated, and serum deprivation-induced apoptosis was suppressed. Thus, it was assumed that compound A
In this study, we found that 3-(2,6-dimethylphenyl)-2-selenoxo-1,3-thiazolidin-4-one, compound A
Footnotes
Abbreviations used: NMR, nuclear magnetic resonance; CI, chemical ionization; Ph3P, triphenylphosphane; IR, infrared spectroscopy; ESI, electro spray ionization.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
