Abstract
Zingiber officinale Roscoe, ginger, is a major spice extensively used in traditional medicine. The toxicity profile of ginger oil was studied by subchronic oral administration for 13 weeks at doses of 100, 250, and 500 mg/kg per day to 6 groups of Wistar rats (5/sex per dose). Separate groups of rats (5/sex per group) received either paraffin oil (vehicle) or were untreated and served as comparative control groups. There was no mortality and no decrease in body weight or food consumption as well as selective organ weights during the study period. Administration of ginger oil to rats did not produce any treatment-related changes in hematological parameters, hepatic, renal functions, serum electrolytes, or in histopathology of selected organs. The major component of ginger oil was found to be zingiberene (31.08%), and initial studies indicated the presence of zingiberene in the serum after oral dosing. These results confirmed that ginger oil is not toxic to male and female rats following subchronic oral administrations of up to 500 mg/kg per day (no observed adverse effect level [NOAEL]).
Introduction
Ginger, rhizome of Zingiber officinale Roscoe, is one of the most widely used spices, belonging to the family Zingiberaceae. It is a common condiment in various foods and beverages and mainly used to impart aroma and flavor. 1 Ginger oil is obtained by steam distillation of the rhizome of Z officinale. This product possesses the aroma and flavor of the spice but lacks the pungency.
Until recently, essential oils have been studied mostly for their flavor and fragrance. Essential oils and their components are now gaining worldwide interest because of their potential multipurpose functional use. 2 Antimicrobial, antifungal, and radical-scavenging properties of spices and their essential oils have been reported. 3,4 Essential oils and some of their components are also used as food preservers and for nutraceutical applications.
A number of studies have been reported on the pharmacological properties of ginger oil. Ginger oil has been used in the prevention and therapeutic management of nausea in general anesthesia patients. 5 It is also shown to have antiplatelet activity in vitro. 6 It is also reported to suppress chronic adjuvant arthritis induced in both the paw and knee of Sprague-Dawley rats. 7 The antinociceptive and immunomodulatory activities of ginger oil have also been studied. 8,9
Acute (single dosage) and subacute (30 days) oral toxicity studies have shown the essential oil of ginger in Swiss albino mice and Wistar rats to be nontoxic and nonadverse at doses up to 600 mg/kg per day. 10 In the present study, we conducted a subchronic toxicity study of the essential oil of ginger isolated from Z officinale Roscoe in rats after oral administration at concentrations of 100, 250, and 500 mg/kg per day once daily for 13 consecutive weeks to assess the oral safety of ginger oil. We have also analyzed the chemical composition of ginger oil (test article) and separately for the presence of the ginger oil components in the serum after oral dosing.
Materials and Methods
Ginger Oil
The essential oil isolated by steam distillation from the rhizome of ginger was supplied by Kancore Ingredients Limited, Angamali, Kerala, India (www.kancor.in). Ginger oil (Sample No. GIO-91204) was supplied as a pale yellow to light amber liquid. Ginger oil is stable up to 2 years. We used the same lot in all the toxicity studies. Liquid paraffin oil (CAS No-8012-95-1), which was used as the vehicle, was purchased from Merck Specialities Private Limited, Mumbai, India.
Chemical Composition of Ginger Essential Oil
The composition of ginger oil prepared from fresh ginger rhizomes was determined by gas chromatography (GC) and GC-mass spectrophotometry (MS) techniques using a Hewlett–Packard gas chromatograph (Model 6890) coupled with a quadruple mass spectrometer (Model HP 5973) and a HP-5MS capillary column (5% phenylmethylsiloxane; 30 m × 320U m × 0.25U m) supplied by Hewlett-Packard, CA, USA. The interphase, ion source, and selective mass detector temperatures were maintained at 243°C, 230°C, and 150°C, respectively. Helium was used as a carrier gas at a flow rate of 1.4 mL/min For the essential oil, the oven temperature was programmed linearly at 60°C; then increased from 60°C to 243°C at a rate of 3°C/min.
Identification of Components
The components are identified using the National Institute of Standards Technology (NIST) library search facility provided with the data analysis software supplied along with GC-MS system.
Animals
All animal experiments were conducted after getting prior permission from Institutional Animal Ethics Committee (IAEC) and per the instructions prescribed by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forest, Government of India, under the supervision of a certified veterinarian.
Young adult male and female Wistar rats weighing 140 to 220 g and aged 7 to 8 weeks old were purchased from Small Animal Breeding Station, Kerala Agricultural University, Mannuthy, Kerala, India. They were housed in the animal facility of Amala Cancer Research Centre, in well-ventilated polypropylene cages. Experiments in rats were started after acclimatization for 1 week. The animals were divided into 5 groups (randomized by initial body weight) with each cage containing 5 rats of the same sex. The housing environment was maintained at a controlled temperature of 22°C ± 2°C and relative humidity 60% ± 10% and provided 12-hour light/dark cycles. Rats were fed with normal pelleted rat chow (Sai Durga Feeds and Foods, Bangalore, India) and supplied with purified water ad libitum. The composition of the feed administered is as follows: Crude protein: 20% to 21%, ether extract: 4% to 5%, crude fiber: 4%, ash: 8%, calcium: 1.2%, phosphorous: 0.6%, nitrogen-free extract: 54%, mean energy (kcal/kg): 3600, pellet size: 12 mm.
Analysis of Ginger Oil Components in Serum
In a separate experiment, male Wistar rats aged 8 to 10 weeks and weighing 180 to 190 g were used for the determination of serum concentrations of ginger oil components. The animals were fasted overnight with free access to water. Animals were administered orally by gavage a single dose of 500 mg/kg body weight of ginger oil dissolved in paraffin oil. Animals without any treatment and those treated with paraffin oil served as untreated and vehicle controls, respectively. Blood samples were collected at 1 hour and 2 hours post dosing.
The blood samples were centrifuged at 4000g for 10 minutes at room temperature. Serum was collected and 1 mL of serum was extracted with 2 mL ethyl acetate. The ethyl acetate extract was analyzed by gas chromatography analysis and checked for ginger oil components as given below.
A fused silica capillary column with a stationary phase equivalent to Carbowax 20 mol/L with a diameter 30 m × 0.32 mm and 1.0 µm film thickness was used for GC/MS analysis. The sample volume used was 0.5 µL. Nitrogen was used as carrier gas with linear flow velocity 25 cm/s. Temperature of the column was initially programmed at 150°C for 1 minute, and then increased at the rate of 10/min to a final temperature of 280°C hold for 30 minutes. Flame ionization detector was used to detect the peaks. zingiberene was used as the reference standard.
Subchronic Toxicity Study
Experimental design and conduct
A total of 50 healthy rats, 25 male and 25 female, were acclimatized to laboratory conditions and divided into 5 groups: each group consisting of 5 males and 5 females. The animals were divided as follows:
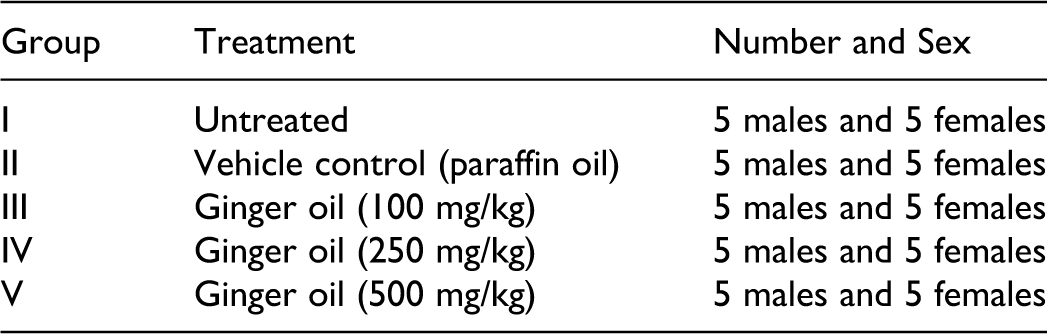
Ginger oil and paraffin oil was administered once daily in the morning by oral gavage and continued for 13 weeks (6 d/week). Ginger oil was dissolved in paraffin oil (500 mg/mL) and administered to the rats at doses of 100, 250, and 500 mg/kg/d. The highest dose of 500 mg/kg/d was selected based on the therapeutic dose of ginger oil. 10 The vehicle control animals were given paraffin oil (5 mL/kg); the untreated group did not receive any treatment.
Observations of clinical signs were made at 10, 30, and 60 minutes after dosing during the first day and once daily thereafter for 91 days. The animals were monitored for clinical and behavioral symptoms such as watery stool, immobility, neuromuscular problems, mortality, and any adverse reactions. The body weight of each rat was recorded on day 0 and at 5-day intervals throughout the course of the study and mean body weights were calculated. The quantity of food and water consumed by groups consisting of 5 rats were recorded every 5 days and the food consumption per rat was calculated for all the groups. After 13 weeks, the animals were sacrificed under light ether anesthesia.
Blood was collected by direct heart puncture method. A part of the blood was collected in heparinized tubes and used for the determination of hematological parameters and the other part was collected in nonheparinized tubes and used for serum chemistry analysis. Necropsy was performed in all animals in the presence of a veterinary pathologist and observations were recorded. The organ weights were recorded as absolute values and their relative values (ie, percentage of the body weight) were calculated.
Parameters Assessed
Blood samples were analyzed for hematological parameters (total erythrocyte count [RBC], hemoglobin, platelet, and total and differential leukocyte counts). Total white blood cells were measured after diluting the blood in Turk fluid and counted using a hemocytometer. 11 For differential leukocyte count, blood smears were prepared on a clean glass slide, stained with Leishman stain and various types of cells were counted manually with a microscope. 12 Platelet count was determined by diluting the blood with Rees Ecker diluting fluid and counted using a hemocytometer. 12 Total RBC count was measured by diluting the blood with Dacie fluid and counted using a counting chamber. 12 Hemoglobin content (Hb) was measured by cyanmethemoglobin (Drabkin) method using the kit from Agappe Diagnostics, Thane, India.
Blood collected in nonheparinized tubes were centrifuged at 5000 rpm for 10 minutes. The clear serum obtained was used for the following investigations: total bilirubin was determined by Jendrassik-Diazotized sulfanilic acid method 13 ; albumin was determined based on its reaction with bromocresol green (binding method); and total protein concentration was determined by biuret method. 14 Kidney function markers such as creatinine and blood urea nitrogen were estimated by Jaffe-Kinetic and urease-colorimetric method, respectively. 15 Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were estimated by the IFCC (International Federation of Clinical Chemistry) kinetic method 16 and alkaline phosphatase (ALP) by PNNP (p- nitrophenyl phosphate) hydrolysis method 17 with kits supplied by Raichem Lifesciences Pvt. Ltd., Mumbai, India, using a Merck Microlab300 Analyzer from Systronics (India) Limited, Ahmedabad, India. Cholesterol and low-density lipoprotein (LDL) was estimated using CHOD-PAP enzymatic method, 18 and high-density lipoprotein (HDL) and triglycerides by phosphotungistic precipitation and GPO-PAP method, respectively. 19 The kidney, liver, and lipid profile markers were estimated using commercially available kits supplied by Piramal Healthcare Limited, Navi Mumbai, India. Serum sodium, potassium, and bicarbonate were estimated using Flame photometer 129 ion selective electrolyte analyzer supplied by Piramal Healthcare Limited, Navi, Mumbai, India. Chloride was estimated by mercurous thiocyanate method using a kit supplied by Raichem Lifesciences Pvt. Ltd., Mumbai, India.
Histopathological Examinations
A portion of selected tissues such as liver, kidney, spleen, stomach, intestine, as well as cerebral cortex and cerebellum from untreated, vehicle control, and animals treated with 500 mg/kg per day of ginger oil were fixed in 10% neutral buffered formalin. Tissue sections were taken and stained with hematoxylin-eosin and observed under high power (×40).
Statistical Analysis
Mean ± standard deviation (SD) was calculated. Two separate statistical analyses were conducted for evaluating data on relative organ weights, lipid profile, hematology, and clinical chemistry parameters that is, the vehicle control group (paraffin oil treated) was compared with the untreated group and the ginger oil-treated groups were compared with the vehicle control (paraffin oil) group for both male and female rats separately. One-way analysis of variance (ANOVA) followed by appropriate post hoc test (Dunnett multiple comparison test) using Graph pad in Stat software were used. Levels achieving differences of P < .05 and P < .01 were considered significant.
Results
Composition of Ginger Oil
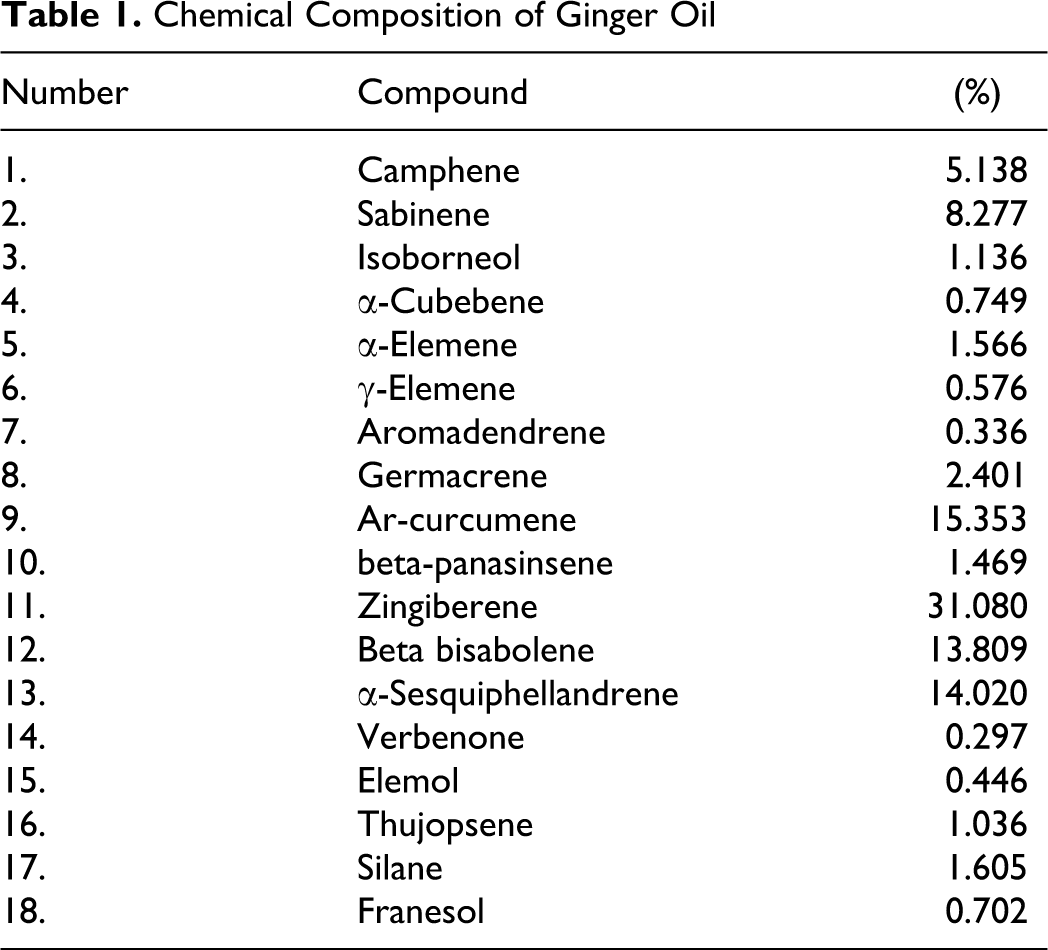
A number of compounds were identified by GC/MS analysis of ginger essential oil and those components identified are given in Table 1 . The structure of the main sesquiterpene hydrocarbon (zingiberene) identified is shown in Figure 1 .
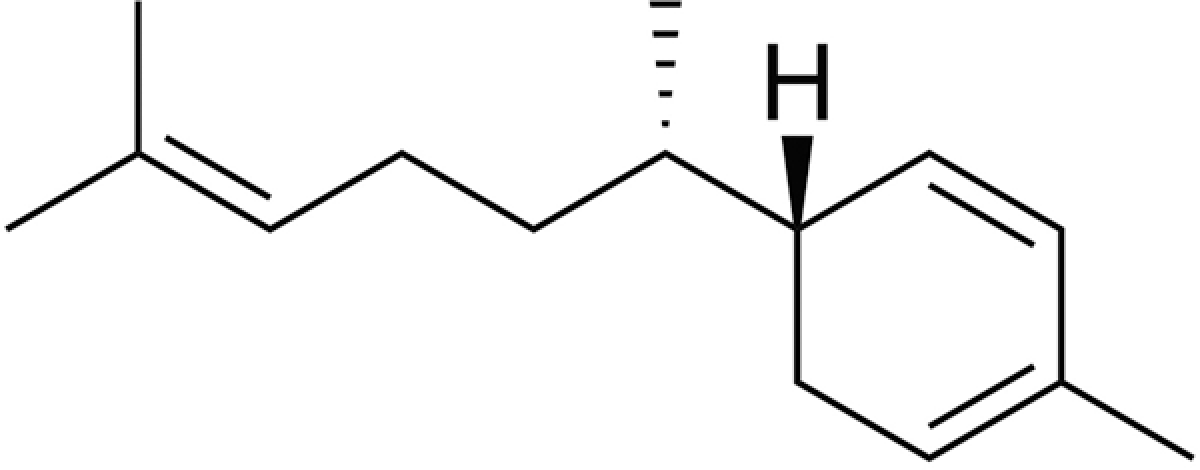
Structure of zingiberene, the major component present in ginger oil.
Chemical Composition of Ginger Oil
Analysis of Ginger Oil in Serum
Ethyl acetate extract of the serum after dosing with ginger oil showed the presence of several peaks at 12.8 to 13.7 minutes in the 2-hour post dosing sample (Figure 2 ), which were not seen in the serum of normal rats or serum of rats treated with paraffin oil alone, indicating that ginger oil was absorbed and detected in blood. The highest peak observed, peak 4, which appeared at 12.8-minute retention time, was identified as zingiberene.
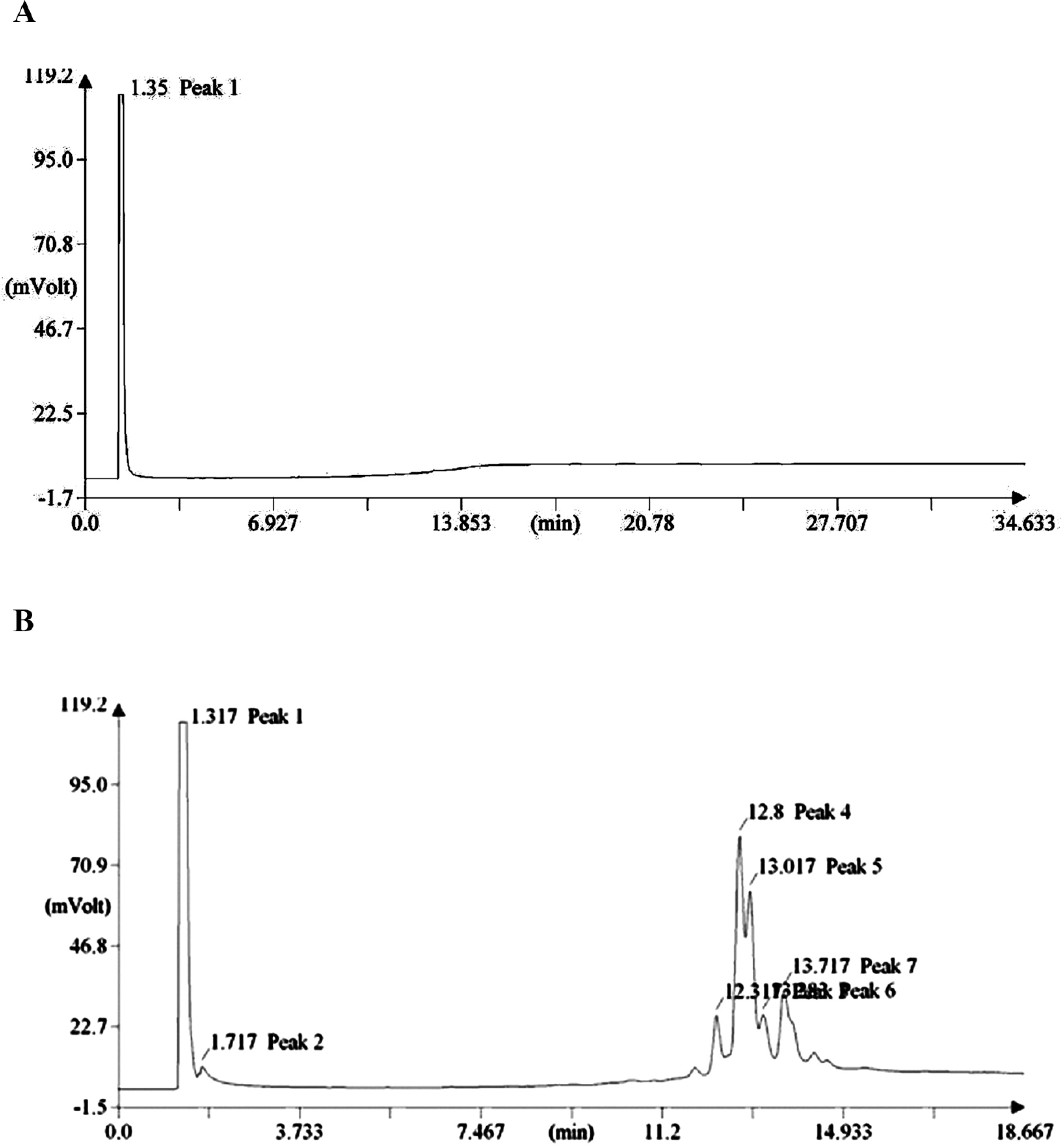
A, Gas chromatographic analysis of serum from control (paraffin treated) animals. B, Gas chromatographic analysis of serum from ginger oil–treated animals at 2 hours. Serum was extracted with ethyl acetate successively and all the washings were pooled; 0.5 µL was injected into the column. The retention time obtained was 12.8 to 13.7 minutes, which was same for the standard zingiberene.
Subchronic Toxicity Study
Subchronic administration of ginger oil at doses 100, 250, and 500 mg/kg per day did not produce any mortality. The animals were healthy and no clinical signs of toxicity were noted. There were no unusual changes in behavior or in locomotor activity observed during the period of study. The 13-week administration of ginger oil did not produce any decrease in the body weight of male and female rats (Figure 3A and B).
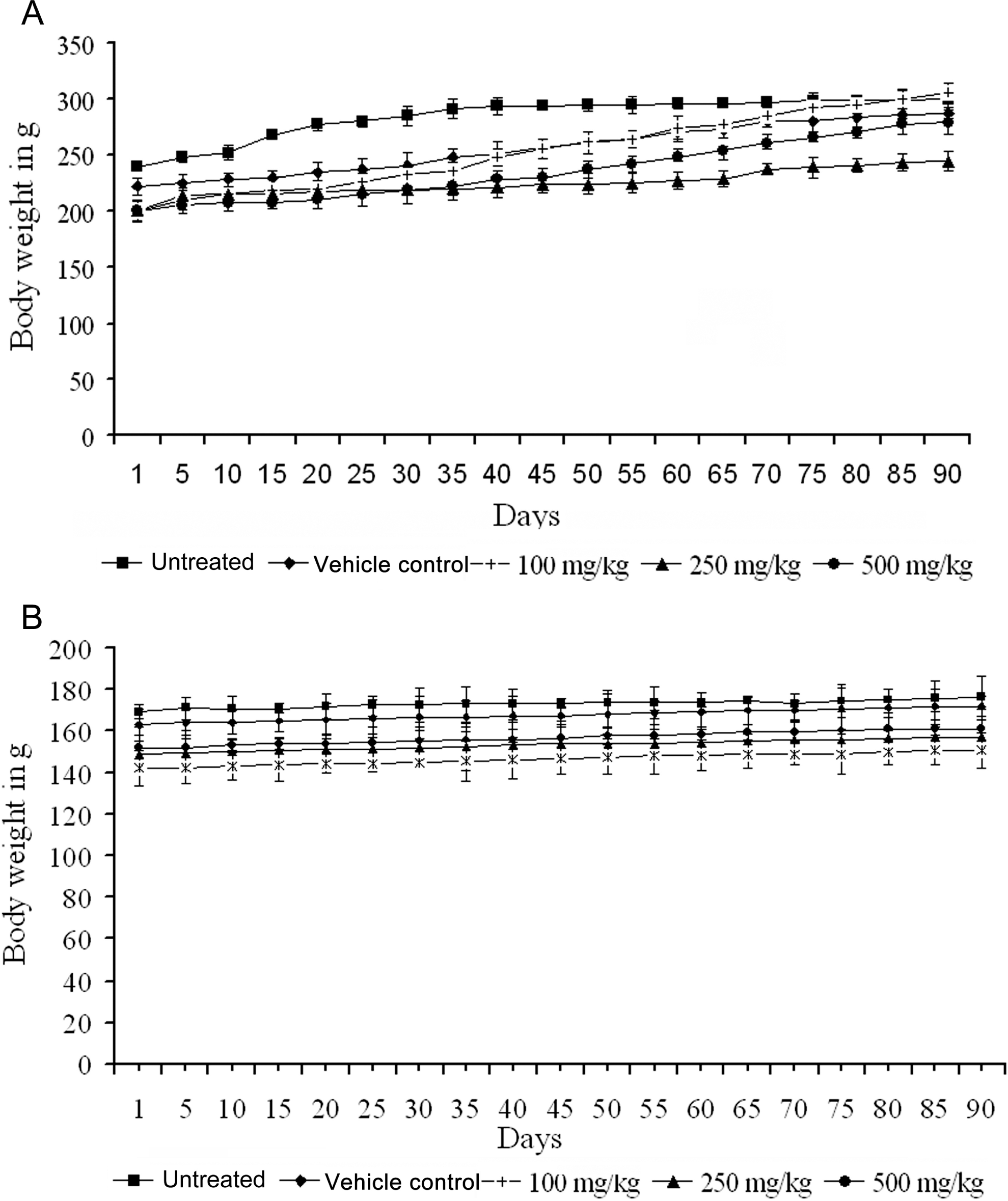
A, Effect of subchronic administration of ginger oil on body weight of male rats. B, Effect of subchronic administration of ginger oil on body weight of female rats.
Administration of ginger oil did not produce any difference in the food consumption of male and female rats when compared with vehicle control (Table 2 ). Similarly, the water consumption pattern was also not altered in treated groups when compared to vehicle control (data not shown). Watery stool was observed in some animals in treated groups including vehicle control, for the first 2 days and may be attributed to the administration of paraffin oil, with resolution beginning on day 3. The average food intake was nearly 11 g/d for male rats and 9 g/d for female rats.
Mean Food Consumption at Week 1 and at Weeks 3, 6, 9, and 12 in Rats During Subchronic Study
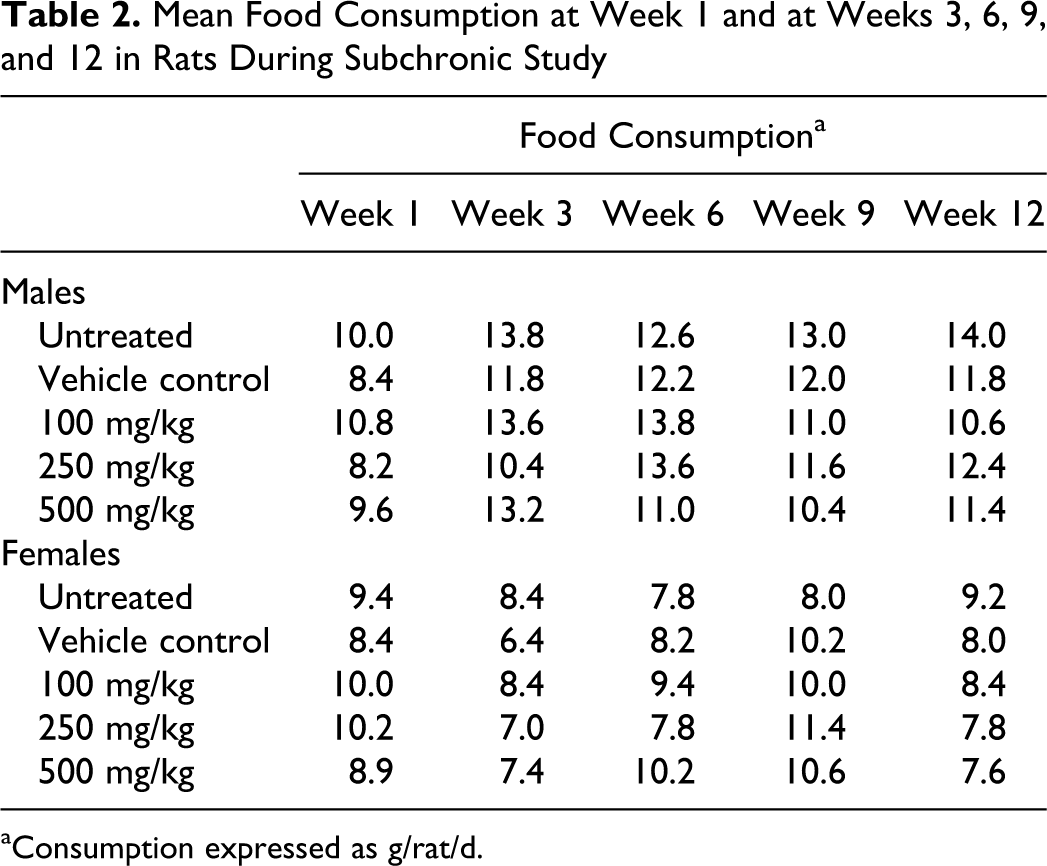
aConsumption expressed as g/rat/d.
No abnormal changes were observed in the relative organ weights of liver, kidney, spleen, lungs, brain, and stomach with respect to body weight in ginger oil-treated animals when compared to vehicle control animals (Table 3 ). Variations observed between untreated and vehicle control groups are within the normal variation in rats. 20 At terminal sacrifice, necropsy observations were unremarkable.
Effect of Subchronic Administration of Ginger Oil on Relative Organ Weight a
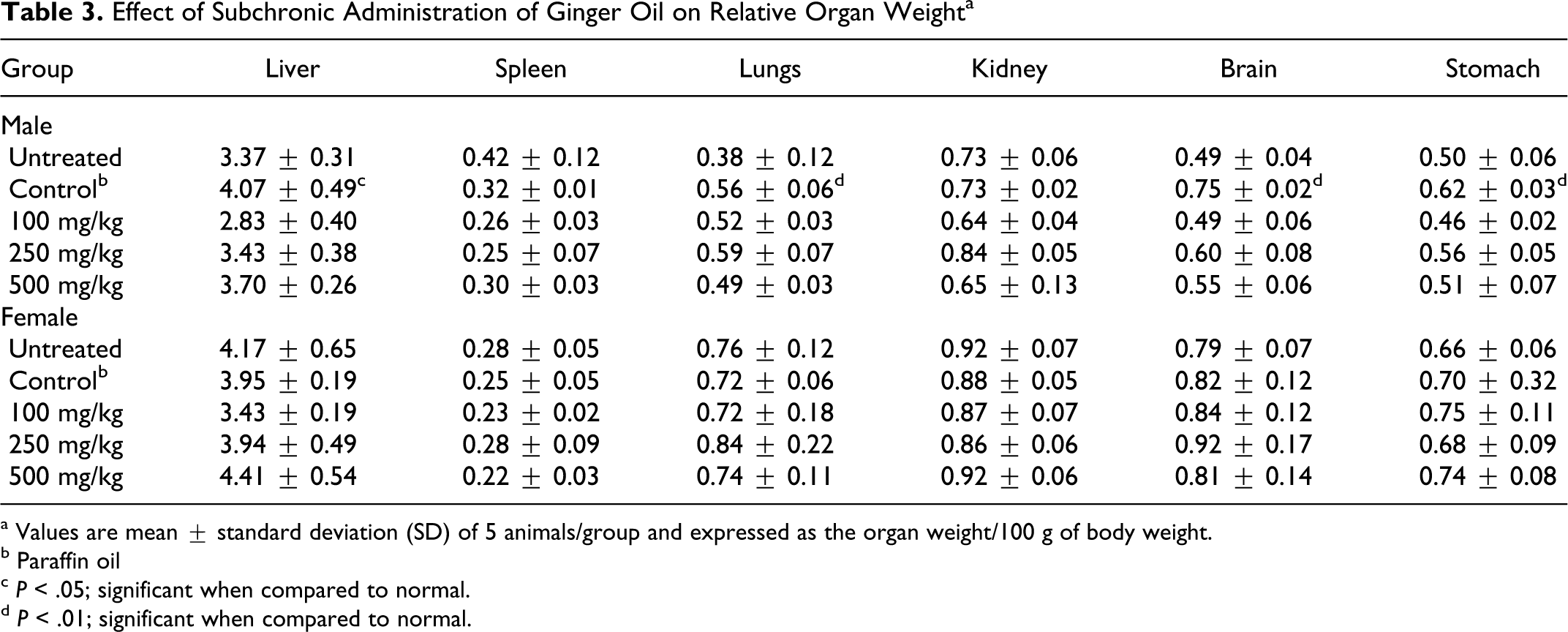
a Values are mean ± standard deviation (SD) of 5 animals/group and expressed as the organ weight/100 g of body weight.
b Paraffin oil
c P < .05; significant when compared to normal.
d P < .01; significant when compared to normal.
Administration of ginger oil did not produce any change in the hematological parameters (hemoglobin, RBC counts, platelet counts, and total and differential leukocyte counts) in untreated, vehicle control, and ginger oil-treated rats (Table 4 ).
Effect of Subchronic Administration of Ginger Oil on Hematological Parameters a
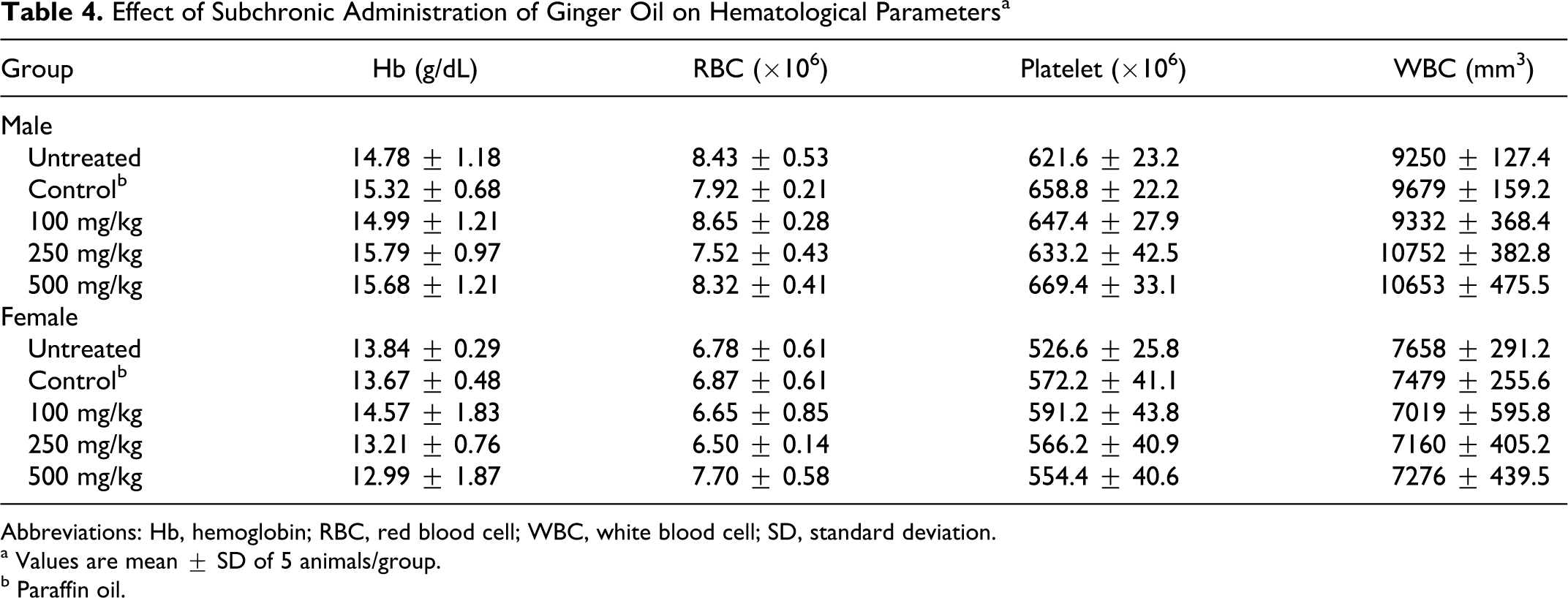
Abbreviations: Hb, hemoglobin; RBC, red blood cell; WBC, white blood cell; SD, standard deviation.
a Values are mean ± SD of 5 animals/group.
b Paraffin oil.
Subchronic administration of ginger oil did not produce any significant changes in hepatic function parameters such as alkaline phosphatase, total protein, albumin, and globulin content. A slight increase in total bilirubin was observed in female rats treated with ginger oil and a decrease in AST and ALT levels (Table 5 ). The variations observed between untreated and vehicle control groups even though significant were within the normal range of serum values for rats. 20
Effect of Subchronic Administration of Ginger Oil on Liver Function Test a
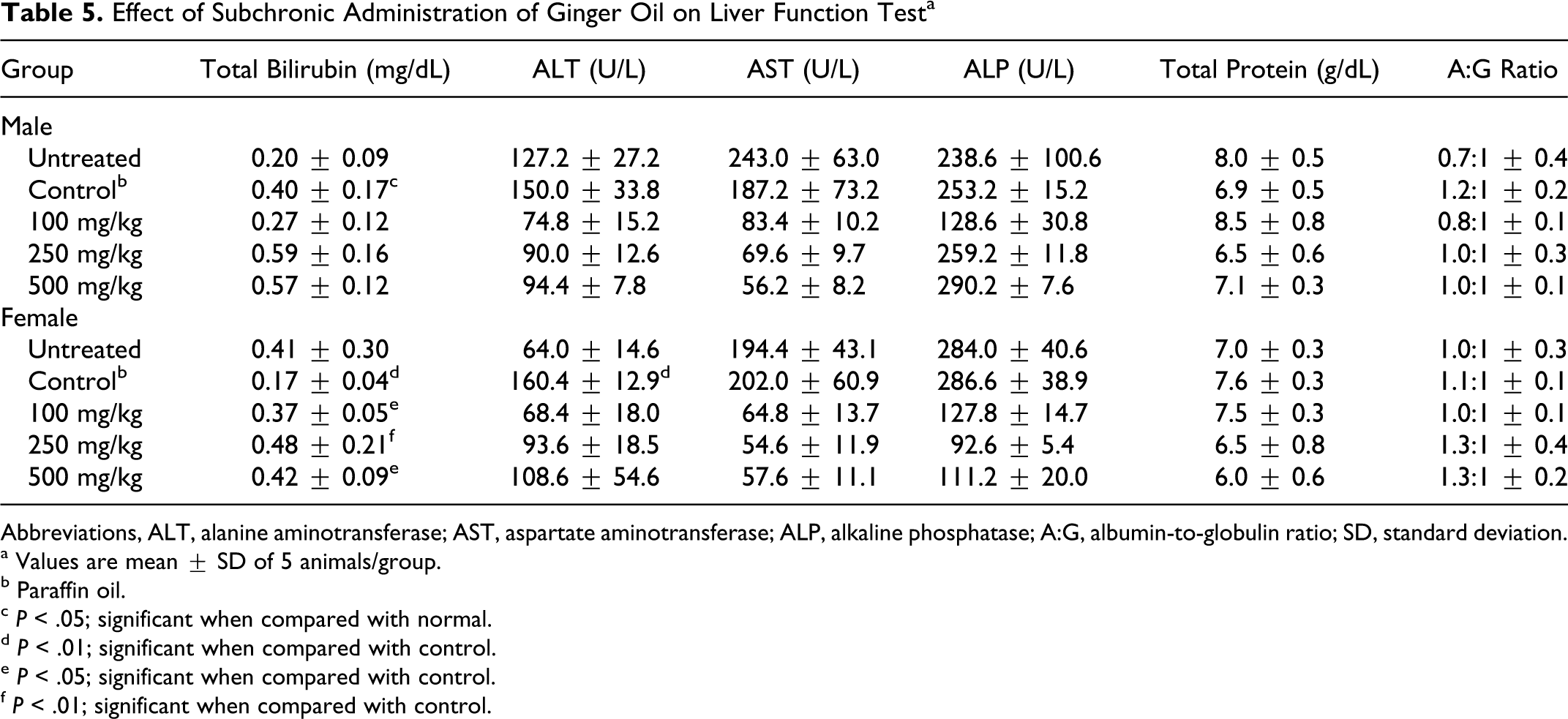
Abbreviations, ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; A:G, albumin-to-globulin ratio; SD, standard deviation.
a Values are mean ± SD of 5 animals/group.
b Paraffin oil.
c P < .05; significant when compared with normal.
d P < .01; significant when compared with control.
e P < .05; significant when compared with control.
f P < .01; significant when compared with control.
The renal function markers, blood urea and serum creatinine, did not show any variation in treated animals compared to vehicle control (Table 6 ). There was no change in the levels of serum electrolytes chloride, potassium, and bicarbonate. An increase in the levels of sodium was observed in male rats treated with 500 mg/kg per day ginger oil (Table 7 ). In the absence of changes in chloride levels or similar changes in females, this change was not considered biologically significant.
Effect of Subchronic Administration of Ginger Oil on Renal Function Test a
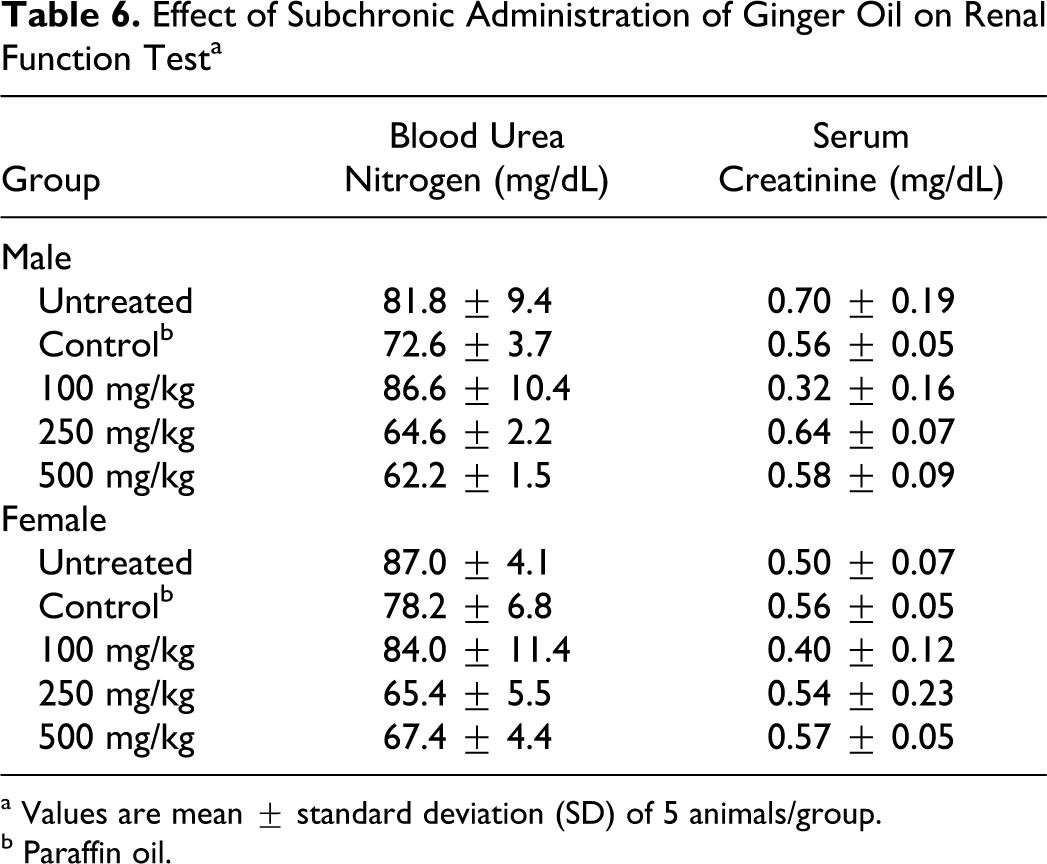
a Values are mean ± standard deviation (SD) of 5 animals/group.
b Paraffin oil.
Effect of Subchronic Administration of Ginger Oil on Serum Electrolytes a
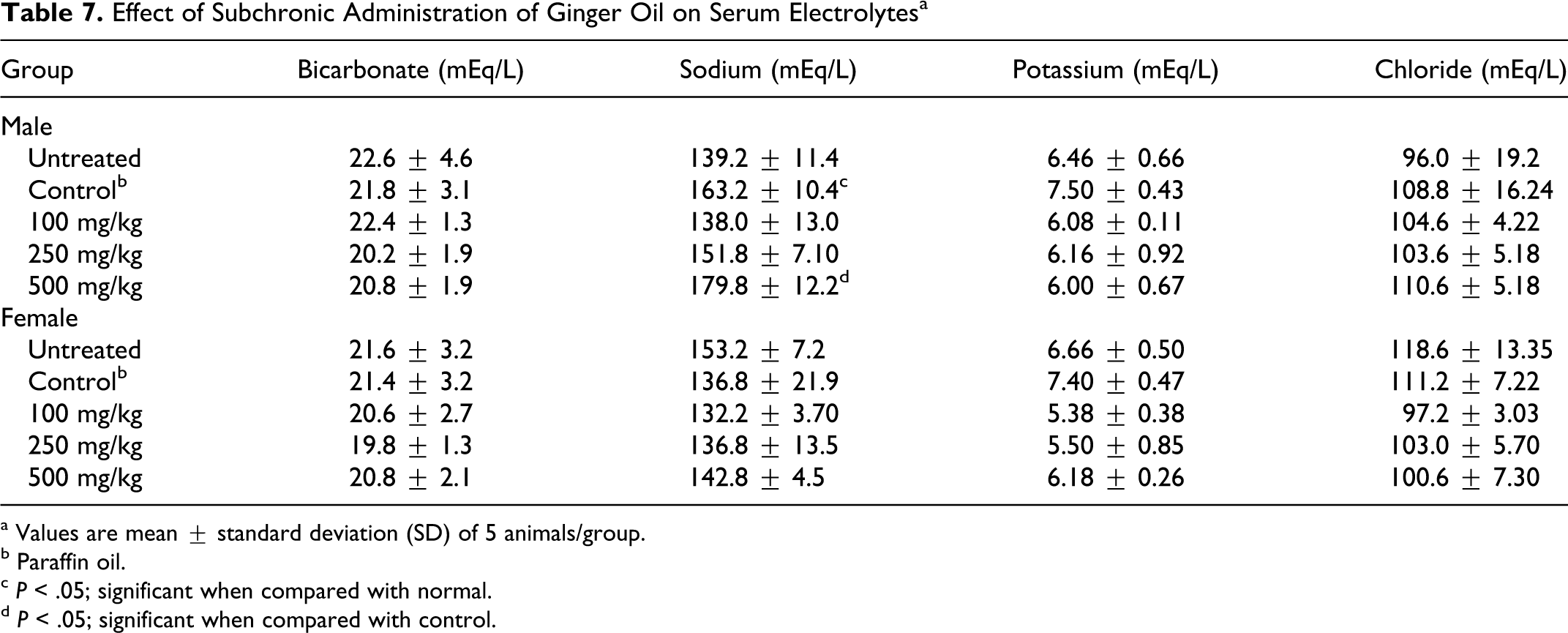
a Values are mean ± standard deviation (SD) of 5 animals/group.
b Paraffin oil.
c P < .05; significant when compared with normal.
d P < .05; significant when compared with control.
A slight decrease (not significant) was observed in cholesterol and triglycerides levels, but there was no change in HDL, LDL, and very-LDL (VLDL) cholesterol, indicating that administration of ginger oil for 13 weeks did not produce any change in lipid profile (Table 8 ). These results indicated that ginger oil did not adversely affect hepatobiliary function involved in fat metabolism.
Effect of Subchronic Administration of Ginger Oil on Lipid Profile a
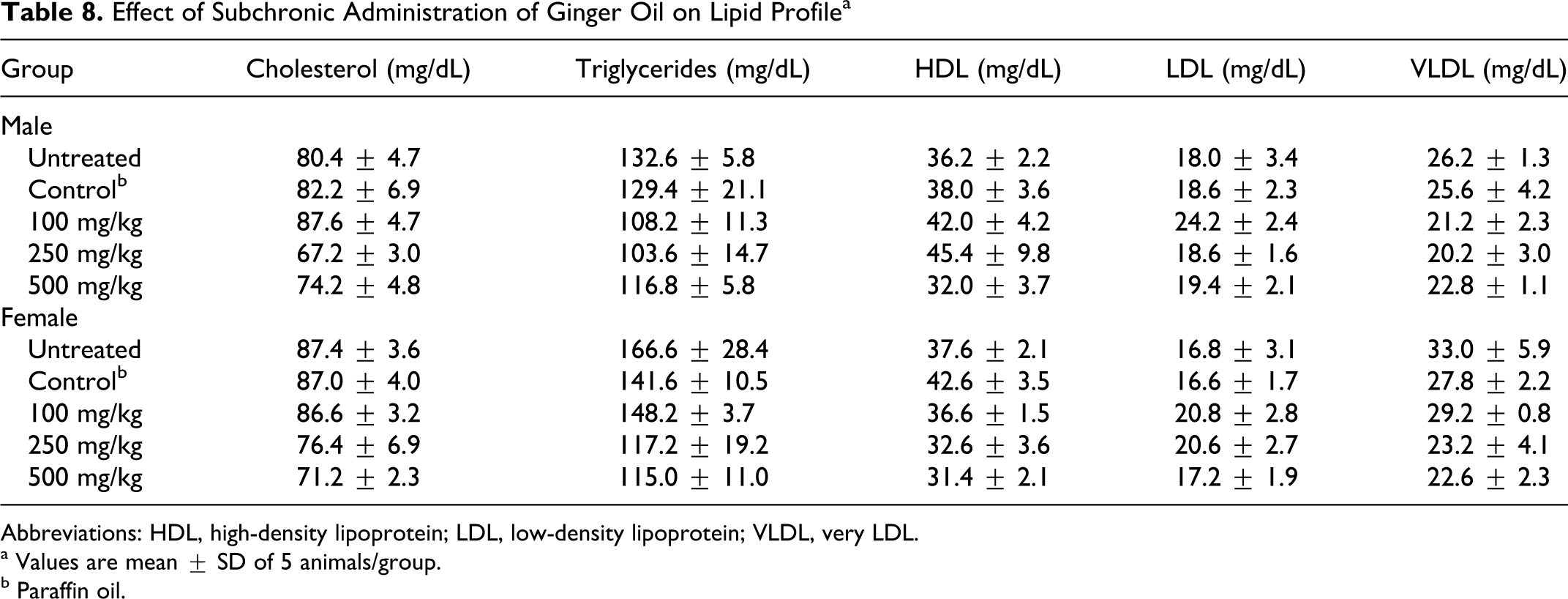
Abbreviations: HDL, high-density lipoprotein; LDL, low-density lipoprotein; VLDL, very LDL.
a Values are mean ± SD of 5 animals/group.
b Paraffin oil.
Histopathological Examinations
Histopathological examinations of the brain, kidney, spleen, liver, stomach, and intestine did not show any pathological lesions in the organs of animals treated with ginger oil (Figure 4 ). Histopathological examination of the organs in the untreated, vehicle control, and ginger oil-treated groups did not show any differences.
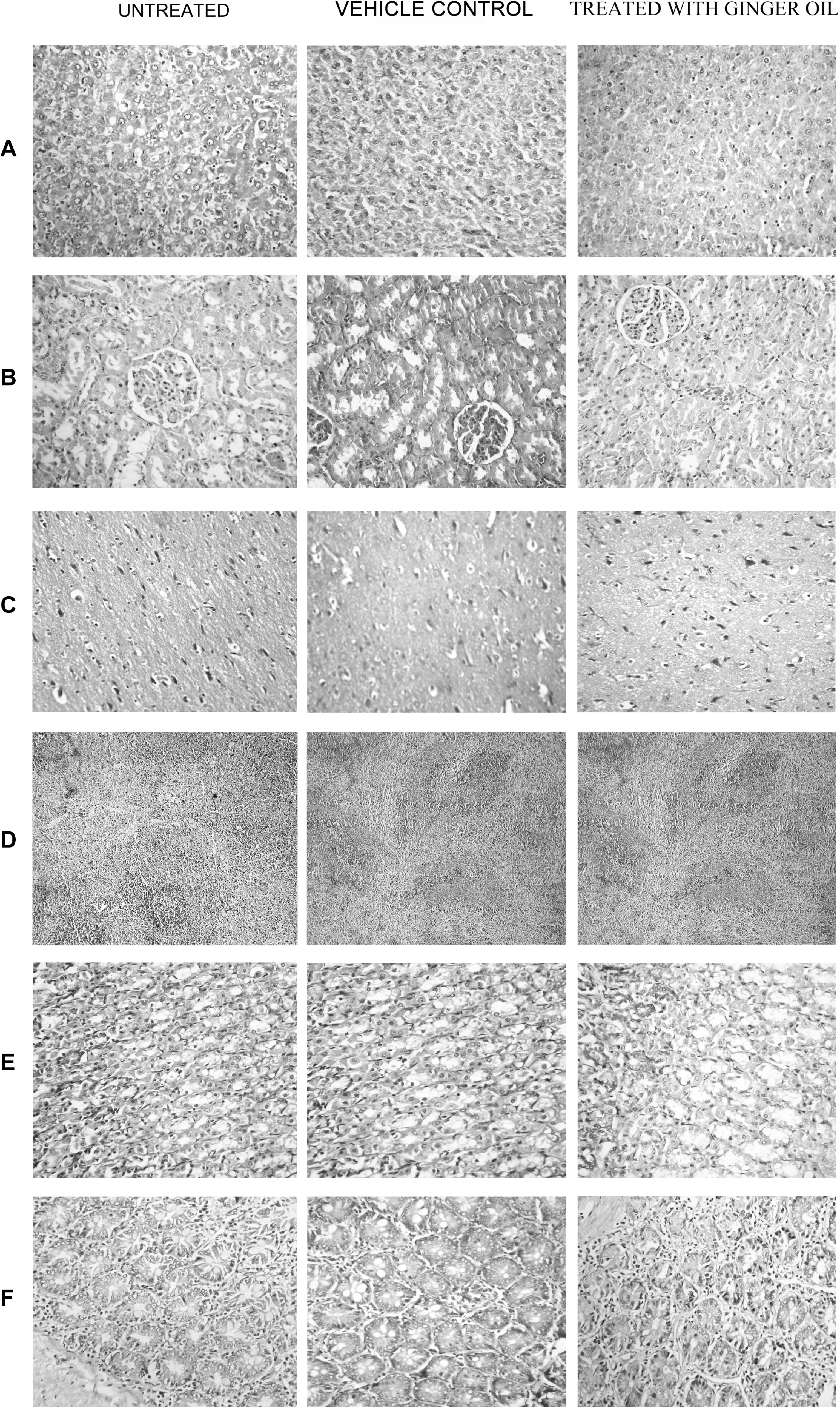
Histopathological analysis of organs from normal, vehicle control (paraffin oil treated) and ginger oil (500 mg/kg body weight). A, Liver. B, kidney. C, cerebral cortex. D, spleen. E, stomach. F, intestine.
Discussion
The main usage of essential oils lies in the flavoring of beverages, confectionery, perfumery, and fragrance industries. 21 However, essential oils have been reported to possess pharmacological properties and are being used for nutraceutical and medicinal applications. The use of essential oils is also gaining more consumer interest.
There are very few reports on the toxicity of ginger oil. A 35-day safety evaluation of ginger powder and that of a patented ginger extract EV.EXT 33 has been reported. 22,23 The acute and subacute (30 days) toxicity profile of ginger oil has also been reported. 10 The current investigation describes a detailed study on the subchronic (13 weeks) toxicity of ginger oil using toxicological and pathological analysis.
The major component of ginger oil used in this study was found to be zingiberene (31.08%). Our initial data on Wistar rats after single oral (gavage) dose showed that oral gavage administration of ginger oil yielded ginger oil components especially zingiberene in serum 2 hours post dosing.
Our study showed that ginger oil did not cause toxicity in rats after oral doses of up to 500 mg/kg per day. No mortality was observed during the period of study. Ginger oil also did not retard growth or affect food consumption, indicating normal metabolism of the animals. No changes were observed in the absolute or relative organ weights of liver, kidney, spleen, stomach, and brain in ginger oil-treated rats compared with vehicle control. Differences observed between the untreated and vehicle control groups may be attributed to animal variation in the groups which may not be treatment related. The variations, even though significant in some cases, were found to be within the normal range reported in Wistar rats. 20
There was also no significant change in hematological parameters between the vehicle control and treated groups. Administration of ginger oil at the concentration studied did not produce any elevation of ALP, AST, and ALT levels when compared to the vehicle control. However, when untreated animals were compared with vehicle control, AST and ALT were found to be slightly higher which was not significant in males but significant in females. Reasons for the increased activity in these animals are not known at present. Similarly, the renal markers, serum creatinine and urea levels, were also not altered compared with the vehicle control. An increase was observed in serum sodium electrolyte levels in males, but there were no toxic effects on kidney as illustrated by other renal parameters and supported by the histopathology of kidney. Reasons for the increase in serum sodium may warrant further investigation. The decrease in the levels of total cholesterol and triglycerides may indicate its possible hypolipidemic activity which needs further investigation. Histopathology of liver, kidney, brain, stomach, spleen, lungs, and intestine also showed normal architecture.
Ginger powder at a very high dose of 2 g/kg was shown to slightly reduce the relative weight of the testes (11.5%). 22 But there were no changes in the relative weight of testes with subacute (35 days) administration of ginger powder up to 1000 mg/kg per day. 22 In the present study, testes were not further evaluated as there were no obvious changes observed at necropsy. Ginger powder did not change any other biochemical and hematological parameters. Our findings in the current study are also consistent with earlier results obtained in the 30-day subacute toxicity study of ginger oil. 10 These results confirmed that ginger oil is not toxic to male and female rats following subchronic oral administrations of up to 500 mg/kg per day (no observed adverse effect level [NOAEL]).
Footnotes
Acknowledgment
We are thankful to Mr Shankar Iyer, New Udaya Pharmacy and Laboratory, Cochin, Kerala, India, for doing gas chromatographic analysis.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: funded by Spices Board, Cochin, Kerala, India [NO.MD/M & H/01/08-09].
