Abstract
A paving asphalt and a vacuum residuum (derived from crude oil by atmospheric and subsequent vacuum distillation and used as a blend stock for asphalt) were tested in skin carcinogenesis assays in mice and in optimized Ames assays for mutagenic activity. In the skin cancer tests, each substance was applied twice weekly for 104 weeks to the clipped backs of groups of 50 male C3H mice. Neither the paving asphalt nor the vacuum residuum (30% weight/volume and 75% weight/weight in US Pharmacopeia mineral oil, respectively) produced any tumors. The positive control benzo[a]pyrene (0.05% w/v in toluene) induced tumors in 46 of 50 mice, demonstrating the effectiveness of the test method. Salmonella typhimurium tester strain TA98 was used in the optimized Ames assay to evaluate mutagenic potential. Dimethylsulfoxide (DMSO) extractions of the substances were not mutagenic when tested up to toxic limits. Thus, under the conditions of these studies, neither the paving asphalt nor the vacuum residuum was carcinogenic or mutagenic.
Keywords
Introduction
Asphalts are produced from the vacuum residuum obtained from atmospheric distillation followed by vacuum distillation of crude oil. These residua are complex substances containing hydrocarbon constituents with boiling points typically above 450°C, viscosities in the range of 300 cSt to 500 cSt at 100°C to 135°C and high molecular weights (500-5000 g/mol). 1 Both asphalt and vacuum residuum have similar physical and chemical properties, molecular weight ranges, and contain similar molecular structures, including asphaltenes, resins, and highly alkylated aromatic hydrocarbons. Components of particular concern are 4- to 6-ring polycyclic aromatic hydrocarbons (PAHs) due to their carcinogenic potential. However, carcinogenic PAH species are present in asphalts at very low concentrations due to the limited overlap between boiling points of PAHs and the lower distillation range of asphalts and vacuum residuum, for example, benzo(a)pyrene has a boiling point 2 of 496°C. In contrast, asphalt and similar materials typically contain much higher levels of highly alkylated polycylic aromatic compounds (PACs), which are a broader category of polycyclic aromatic substances containing sulfur, oxygen, and nitrogen.
A number of mouse dermal carcinogenesis studies have been conducted on whole asphalt. Simmers 3 reported that asphalt applied as a 90% solution in toluene to the backs of mice 3 times a week for 2 years induced tumors in 9 of 20 animals. Severe skin irritation was also observed in this study, likely due to the toluene exposure. Twice weekly application of roofing asphalt in toluene (50%) for 80 weeks did not induce tumors in a group of 50 mice. 4 When diluted in acetone, 3 samples of paving asphalt and 1 roofing asphalt applied twice weekly for 2 years did not produce evidence of dermal carcinogenesis (0%-2% tumor-bearing animals). 5 A fifth sample, a paving asphalt, exhibited a weak potential for carcinogenicity (4% tumor-bearing animals). Similar to the results with asphalt, application of vacuum residuum diluted in toluene for 130 weeks (twice weekly) produced tumors in 5 of 50 mice with one sample and 2 of 50 mice with a second sample, with a mean latency period of 113 and 120 weeks, respectively. 6 Tumor incidence in the treated animals was not statistically different from tumor incidence in control animals, in which toluene alone produced tumors in 4 of 50 animals, with a mean latency period of 111 weeks. Accordingly, the vacuum residua were not considered carcinogenic in this assay. The overall interpretation of these studies has been that asphalts have low potential to induce dermal carcinogenesis. However, interpretation of these historical skin-painting studies is confounded by methodological problems including nonstandard protocols, occurrence of intercurrent disease/early mortality or use of solvents that may have affected the bioavailability of the asphalts or compromised the integrity of the skin. One of the possible impacts of solvent use is chronic skin irritation. Chronic skin irritation has been shown to act as a tumor promoter or co-carcinogen, 7 as seen with petroleum middle distillates.8–10
In addition to animal bioassays, short-term predictive tests for genotoxicity, such as Ames assays in bacteria, have found that whole asphalts and vacuum residuum are nonmutagenic or weakly mutagenic only in the presence of metabolic activation. Dimethylsulfoxide (DMSO) extracts of paving tar were found to be weakly positive in Salmonella typhimurium assays with metabolic activation; however, this sample may have contained coal tar, which contains levels of 4- to 6-ring PAHs 4 to 5 orders of magnitude higher than asphalt.11,12 Three additional samples of paving asphalt and asphalt-based paints (60% asphalt diluted in mineral spirits), tested as DMSO extracts in S typhimurium, were inactive in both the presence and absence of metabolic activation.13,14 In a modified version of the Ames assay in which larger amounts of metabolizing enzymes were used (ASTM E 1687), DMSO extracts of both roofing and paving asphalts were found to be marginally positive. 15 Similarly, weakly positive results were found for DMSO-solubilized vacuum residuum tested in the mouse lymphoma cell gene mutation assay with metabolic activation.16,17
Due to some of the uncertainties in the interpretation of the historical skin carcinogenesis studies, an important grade of paving asphalt (asphalt cement 20 [AC-20]) was tested in the skin carcinogenesis model. Although actual occupational exposures are most likely to occur through contact with fumes and fume condensates, the current study demonstrates that asphalt and vacuum residuum are not carcinogenic in situ. Study conditions were established to minimize skin irritation and possible secondary co-carcinogenic or tumor-promoting activity. A mutagenicity test of the asphalt was also conducted.
Materials and Methods
Test Materials
An AC-20 (see note 1) asphalt (CASRN 8052-42-4) was derived from a mixture of naphthenic crude oils consisting of 60.0% Alaska North Slope, 35.8% Maya, and 4.2% Nigerian Light and was produced by a blend that included vacuum residuum. Physical parameters used to characterize the substance include viscosity, measured as 392 cSt at 135°C, and penetration depth, which is typically reported as tenths of a millimeter (decimillimeter, or dmm) and was measured as 69 dmm at 25°C. Coastal residuum ([CR] CASRN 64741-56-6) is a vacuum residuum also produced from naphthenic crude. In the dermal carcinogenesis study, benzo[a]pyrene ([BaP] Aldrich Chemical Co, Milwaukee, Wisconsin) was used as the positive control, and toluene (Fischer Scientific, Fair Lawn, New Jersey) and US Pharmacopeia (USP) mineral oil ([MO] ExxonMobil Lubricants and Specialties Company, Fairfax, VA) were used as vehicle controls. In the optimized Ames assay, both DMSO and cyclohexane were obtained from Aldrich Chemical Co, and the positive control material used was a heavy catalytically cracked gas oil (CASRN 64741-62-4; ExxonMobil Refining and Supply Company, Fairfax, VA), a carcinogenic substance that contains significant levels of PAH.
Dermal Carcinogenicity Study
Male C3H/HeNCrlBR mice, approximately 6 to 8 weeks of age, were obtained from Charles River Breeding Laboratories (Montreal, Quebec, Canada) and acclimated for 14 days prior to study initiation. Animals were randomly distributed into 5 test groups of 50 mice each. Mice were housed individually in suspended stainless steel cages and were allowed access to Purina-Certified Rodent Chow Pellets (#5002; Ralston Purina Company, St. Louis, Missouri) and water ad libitum. A 12-h light–dark lighting was maintained, and the room temperature and humidity were controlled within a range of 68°C to 76°C and 40% to 70%, respectively.
Test materials were applied as 37.5 μL doses to the clipped backs of each mouse twice weekly, in order to achieve a total weekly dose of 75 μL. Both AC-20 and CR are too viscous to be applied in an undiluted form. Accordingly, MO was used as a diluent. Mineral oil has been found to be noncarcinogenic and nonirritating to the skin. 8 Asphalt cement 20 and CR were diluted and applied at the limits of solubility in MO, 30% (weight/volume [w/v]) and 75% (weight/weight [w/w]), respectively, and BaP was applied at a 0.05% (w/v) dilution in toluene. Mice were examined twice daily for viability, and clinical observations were conducted at study initiation and weekly thereafter, specifically noting the presence of dermal growths and irritation. Body weights were recorded the week prior to dosing, during the first study week, and every 4 weeks thereafter until study termination. At study termination, gross necropsies were conducted on each animal, consisting of an examination of the external surface, all orifices, the thoracic, abdominal and cranial cavities and their viscera, the cervical tissues, organs, and the carcass.
After 24 months of treatment, the animals were euthanized by asphyxiation using carbon dioxide gas. Animals considered either moribund or exhibiting a dermal carcinoma were euthanized prior to 24 months. Treated skin, untreated skin, and any grossly observable masses were preserved in 10% neutral buffered formalin for histopathological evaluation and confirmation of gross observations.
Optimized Ames Assay
Mutagenic potential of the test materials was evaluated in an optimized Ames assay (often referred to as the modified Ames test) as detailed by Blackburn et al.18,19 Briefly, this method evaluates the ability of a test material to cause reverse mutations at the histidine locus in S typhimurium to overcome nutritional histidine dependence when histidine is withheld from the media. Optimized parameters include DMSO extraction to concentrate the PAC-containing fraction; use of metabolizing enzymes from hamster liver 8 times the standard concentration; and use of a single strain, TA98, shown to be the most responsive to PAC.
Test materials were first diluted in cyclohexane and extracted with DMSO, followed by brief vortexing every 5 minutes for 30 minutes. After separation, the DMSO fraction was removed and stored at room temperature overnight. Appropriate dilutions in DMSO were prepared from these stock solutions. Coastal residuum was tested in an initial assay at concentrations of 1, 5, 10, 25, and 50 μL/plate and in a second assay at 1, 5, 7.5, 10, and 15 μL/plate, due to significant toxicity at the 2 highest doses in the initial assay. Asphalt cement 20 was completely toxic at the doses used in an initial assay starting at 1 μL/plate, thus, AC-20 was tested in duplicate assays at concentrations of 0.005, 0.01, 0.05, 0.1, 0.5, and 1 μL/plate. The positive control material, a heavy catalytically cracked gas oil, was tested at 1 μL/plate. Vehicle dose levels were kept below the concentration known to be toxic to the tester organism. The preincubation method was followed, such that the test material dilutions were added to a sterile glass tube followed by hamster metabolic enzymes and then bacteria. After a 20-minute preincubation at 37°C, top agar was added, and the mixtures were vortexed and immediately plated. Once cooled, the plates were inverted and incubated for 2 days at 37°C, following which the plates were evaluated for toxicity and the colonies were counted.
Statistics
For the dermal carcinogenesis study, an analysis of survivorship was based on the Weibull estimate of median survivorship with comparisons made on the basis of 95% confidence intervals, as well as by the Kruskal-Wallis analysis and Cox test.20–22 Mean body weights were evaluated at each time period using the MO control group as the control. First, Bartlett test was performed to determine whether the dose groups had equal variance. If the variances were equal, a standard 1-way analysis of variance using the F distribution to assess the significance was used. If significant differences among the means were indicated, Dunnett test was used to determine which treatment groups differed significantly from the control.
For the mutagenicity assay, the mean plate count and standard deviation for each dose were determined according to published procedures. 23 An assay was considered positive if a reproducible, dose-related increase was observed in the number of revertant colonies over at least 3 concentrations of test material including at least 1 positive dose. A positive dose is considered to be one at which the mean colony count on the test plates is greater than twice the mean number of spontaneous revertants on the vehicle plates.
Results
Skin-Painting Bioassay
At the end of the skin-painting bioassay, no tumors were observed in animals treated with AC-20, CR, or the vehicle controls (toluene and MO; Table 1). The positive control, BaP, produced histopathologically confirmed tumors in 92% of the animals. Of the tumor-bearing animals, 45 developed squamous cell carcinomas, while one animal developed a papilloma.
Tumor Incidence in the Skin-Painting Bioassay
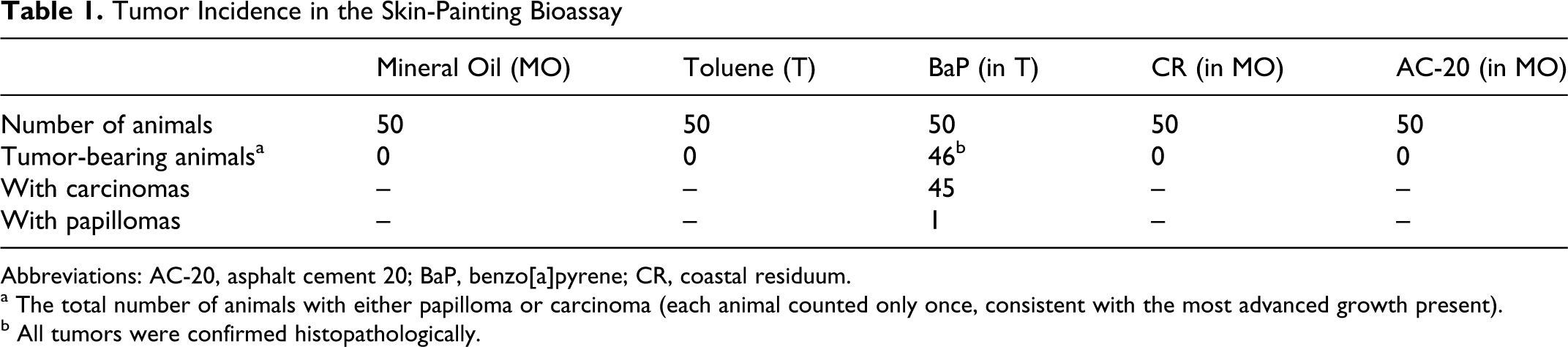
Abbreviations: AC-20, asphalt cement 20; BaP, benzo[a]pyrene; CR, coastal residuum.
a The total number of animals with either papilloma or carcinoma (each animal counted only once, consistent with the most advanced growth present).
b All tumors were confirmed histopathologically.
Asphalt cement 20- and CR-treated animals, as well as MO-treated animals, were generally free of dermal irritation during the course of the study and upon histopathological examination. Animals treated with BaP and its vehicle, toluene, exhibited slight-to-moderate dermal irritation throughout the study, characterized macroscopically by desquamation, exfoliation, atonia, eschar, erythema, and/or edema. Dermal irritation was confirmed after microscopic evaluation by the presence of acanthosis and subepidermal inflammatory infiltrate. No other treatment-related clinical observations were reported for any of the treatment groups.
Analysis of the mean body weights revealed a significant difference among the means on days 86 and 226. However, pairwise comparisons between treated and control groups yielded no statistically significant comparisons (data not shown). As such, it is unlikely that any of these differences were treatment related.
Survivorship in AC-20- and CR-treated animals and the vehicle controls was indistinguishable, as measured by the Weibull estimate of median survivorship, where mean survivorship in days ranged from 537 days (AC-20) to 563 days (toluene vehicle control; Figure 1). Survivorship in positive-control animals was distinctly lower (ie, 335 days); however, this is in part a reflection of the fact that the mice were terminated from the study once the tumors developed were grossly diagnosed as squamous cell carcinomas. Similar results were obtained when the data were analyzed by the generalized Kruskal-Wallis and Cox tests for time-corrected survivorship. By this method, animals treated with test materials and vehicle controls had a median survivorship ranging from 523 days (MO vehicle control) to 554 days (CR), respectively; while animals treated with BaP had a median survivorship of 324 days.
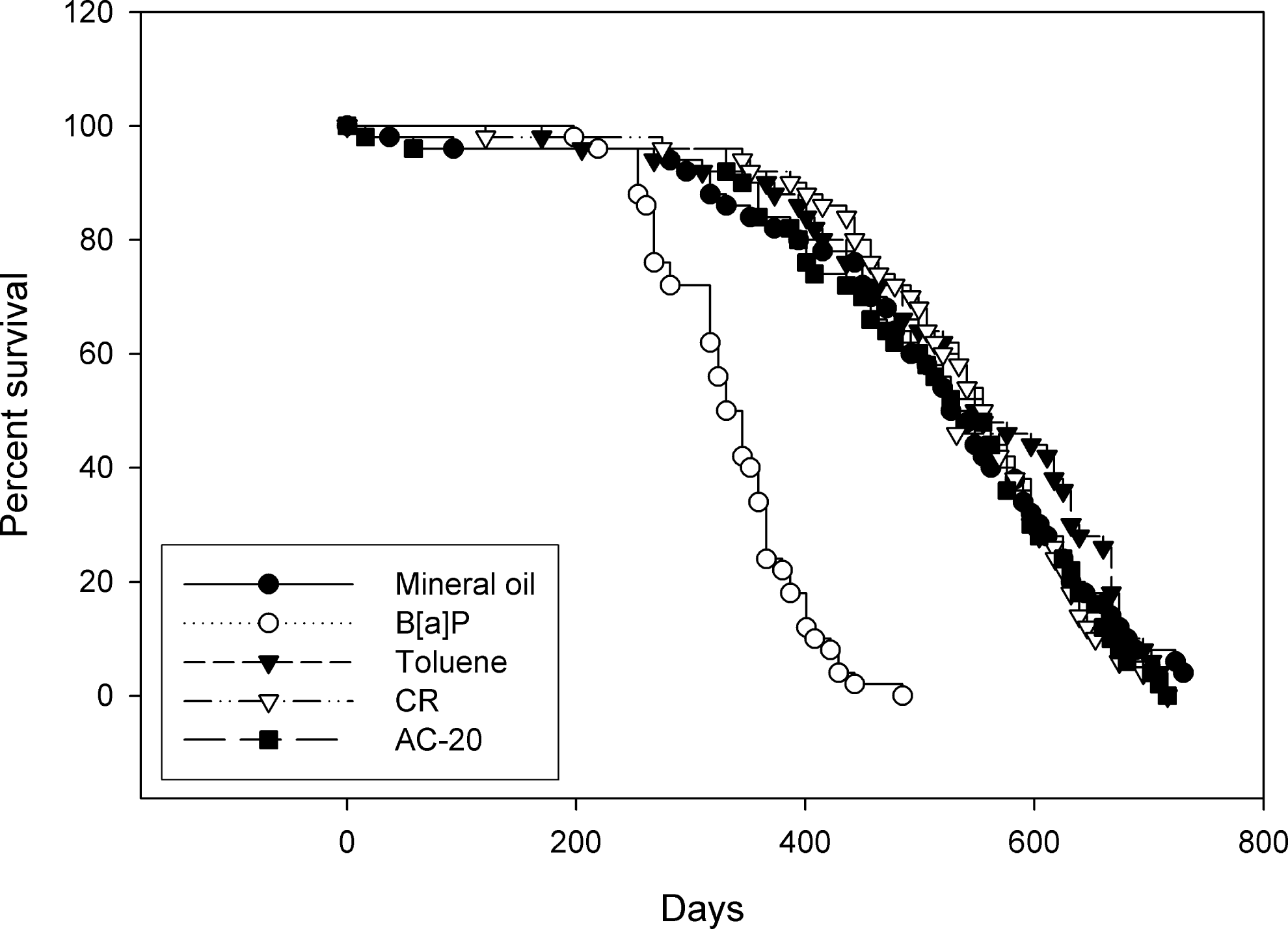
Survivorship in animals treated with asphalt cement 20 (AC-20), coastal residuum (CR), and vehicle controls was generally equivalent and was distinctly lower in positive control animals. Animals were administered test material twice weekly at doses of 37.5 μL for 24 months or until an animal exhibited a carcinoma. Survivorship was based on the Weibull estimate of median.
Optimized Ames Assay
The AC-20 asphalt and CR were both inactive in the Salmonella mutagenicity assay when tested to the limits of toxicity (Tables 2 and 3). Asphalt cement 20 did not produce a dose-dependent increase in the number of revertant colonies in either the initial or repeat assay. Coastal residuum was also inactive in this mutagenicity assay. The positive control, a heavy catalytically cracked gas oil, produced significant increases in revertants, indicating that the test system was responding appropriately.
Revertants Per Plate After Treatment With AC-20
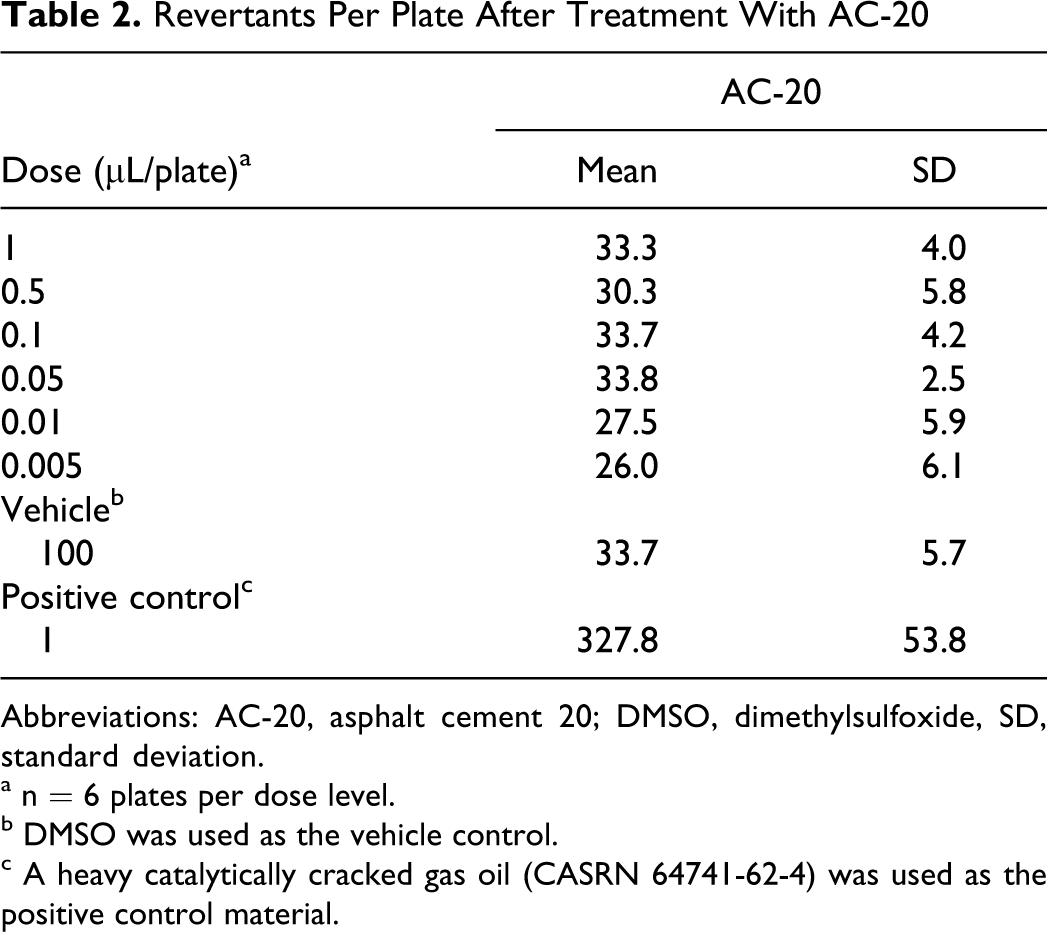
Abbreviations: AC-20, asphalt cement 20; DMSO, dimethylsulfoxide, SD, standard deviation.
a n = 6 plates per dose level.
b DMSO was used as the vehicle control.
c A heavy catalytically cracked gas oil (CASRN 64741-62-4) was used as the positive control material.
Revertants Per Plate After Treatment With Coastal Residuum
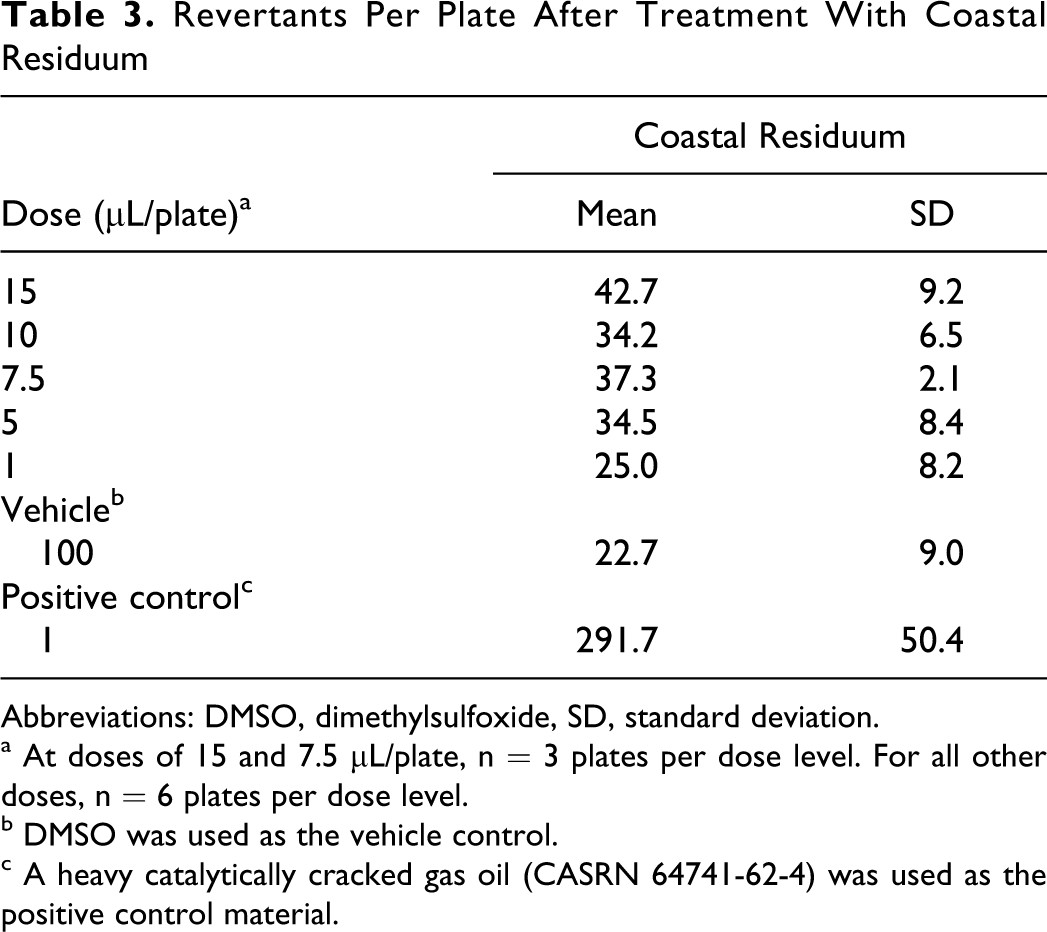
Abbreviations: DMSO, dimethylsulfoxide, SD, standard deviation.
a At doses of 15 and 7.5 μL/plate, n = 3 plates per dose level. For all other doses, n = 6 plates per dose level.
b DMSO was used as the vehicle control.
c A heavy catalytically cracked gas oil (CASRN 64741-62-4) was used as the positive control material.
Discussion
Neither AC-20 nor CR was carcinogenic to mouse skin. Both were tested at the limit of their solubility in MO. This vehicle was selected because it is nonirritating and noncarcinogenic and, as such, minimizes possible confounding conditions identified from earlier studies. When skin contact is a reasonable route of exposure, it is important to consider the physical characteristics that could impact the bioavailability of the substance. In the case of highly viscous materials such as asphalt, skin penetration of PAHs is expected to be minimal, as the bioavailability has been reported to be inversely proportional to viscosity. 24 When radiolabeled BaP was applied to mouse skin in bitumens (expected to have viscosities similar to AC-20), absorption decreased more than 5-fold compared to those when applied in oils with viscosity comparable to the MO control. Thus, use of MO as a diluent in this study would be expected to enhance the bioavailability of the presumptive toxic components of the AC-20 and thereby increase the sensitivity of the mouse bioassay as a hazard-identification test. Although no tumor response was observed, the dose used in this study was consistent with the standard experimental procedure for assessing the dermal carcinogenic potential of petroleum oils.25,26 In petroleum oils with high concentrations of 4- to 6-ring PAHs, such as untreated vacuum distillates, a clear tumor response occurs at comparable doses (25-75 μL weekly).25,27
Because the presumptive toxic components in paving asphalts, PAHs, are expected to be mutagenic, 28 bacterial mutagenicity assays were conducted to evaluate the mutagenic potential of AC-20 and CR. When tested at the limits of solubility, neither test material exhibited a mutagenic potential, consistent with the lack of a tumor response in the mouse bioassay. Although it is difficult to make a comparison between doses in the in vitro mutagenicity test and the in vivo bioassay, Blackburn et al19,29 demonstrated a correlation between the optimized version of the Ames test and the mouse bioassay, indicating that both tests are useful as hazard-identification tools.
Recent industry research has focused on whether asphalt fumes may be carcinogenic. This is important because in typical uses, asphalts must be heated to soften them sufficiently such that they can be applied to roads or other surfaces and, during heating, fumes are produced. Methods have been developed to collect fumes from storage tanks such that the samples are compositionally similar to those found in the workplace.30,31 Kriech et al 30 report that approximately 70% of asphalt fume is made up of alkanes, monocycloparaffins, alkylbenzenes, and branched chain aliphatics, while the remaining 30% is a mixture of PACs, with alkylated 2- and 3-ring compounds representing over 90% of the detected PACs. Such “field-matched” fumes from paving asphalts were noncarcinogenic for both the skin 32 and the inhalation 33 routes of exposure. Thus, paving asphalts do not present a carcinogenic hazard either as manufactured or under typical use conditions.
Conclusion
Paving asphalt AC-20 and CR were neither carcinogenic to mouse skin nor mutagenic as measured in a bacterial mutagenicity assay.
Footnotes
The authors are currently or were previously employed by ExxonMobil Biomedical Sciences, Inc., a separately incorporated but wholly owned affiliate of Exxon Mobil Corporation.
The author(s) received financial support from Exxon Mobil Corporation for the research, authorship, and/or publication of this article.
