Abstract
The ability of 2 combinations of oximes (HI-6 + trimedoxime and HI-6 + K203) to reactivate VX-inhibited acetylcholinesterase and reduce acute toxicity of VX was compared with the reactivating and therapeutic efficacy of antidotal treatment involving a single oxime (HI-6, trimedoxime, K203) in rats and mice. Our results showed that the reactivating efficacy of both combinations of oximes studied in rats is significantly higher than the reactivating efficacy of all individual oximes in diaphragm and roughly corresponds to the most effective individual oxime in blood and brain. Both combinations of oximes were found to be more effective in the reduction of acute lethal toxicity of VX in mice than the antidotal treatment involving the most efficacious individual oxime although the difference is not significant. Based on the obtained data, we can conclude that the antidotal treatment involving the chosen combinations of oximes brings benefit for the reactivation of VX-inhibited acetylcholinesterase in rats and for the antidotal treatment of VX-induced acute poisoning in mice.
Introduction
Highly toxic organophosphorus compounds called nerve agents are considered the most dangerous chemical warfare agents. The most important representatives of nerve agents are tabun, sarin, soman, cyclosarin, and VX. Their acute toxic effects are based on the irreversible inhibition of acetylcholinesterase ([AChE], EC 3.1.1.7) leading to overstimulation of postsynaptic cholinergic receptors due to the accumulation of the neurotransmitter acetylcholine (ACh) in synapses of the central and peripheral nervous systems.1,2 The current standard antidotal treatment of poisoning with nerve agents usually consists of a muscarinic cholinergic receptor antagonist to block the overstimulation of cholinergic receptors by ACh and an oxime to reactivate nerve agent-inhibited AChE. Generally, anticholinergics (mainly atropine) are used for relieving muscarinic signs and symptoms; whereas AChE reactivators (called oximes) are used for reactivation of nerve agent-inhibited AChE.1,3 In the last century, several oximes (pralidoxime, obidoxime, trimedoxime, and HI-6) were developed as antidotes against nerve agents, but, unfortunately, their potency to counteract the acute toxic effects of nerve agents differs depending on the type of nerve agent.4,5
The threat of misuse of different nerve agents in wartime or by terrorists6,7 emphasizes the necessity of the development of oximes with the ability to counteract acute toxicity of all nerve agents. Recently, a lot of new oximes have been developed and evaluated.8–12 However, published results confirm that there is no single, broad-spectrum oxime suitable for the antidotal treatment of poisonings with all nerve agents.5,13,14
One possible approach to broaden the spectrum of oximes to obtain sufficiently effective antidotes against all nerve agents regardless of their chemical structure is to combine chosen oximes in the antidotal treatment. In this article the combination of HI-6 with trimedoxime or newly developed oxime K203 was used (Figure 1). Both combinations of oximes were considered suitable for the antidotal treatment of acute tabun and soman poisonings,15,16 but it is necessary to know whether these combinations of oximes are also sufficiently effective against all other nerve agents.
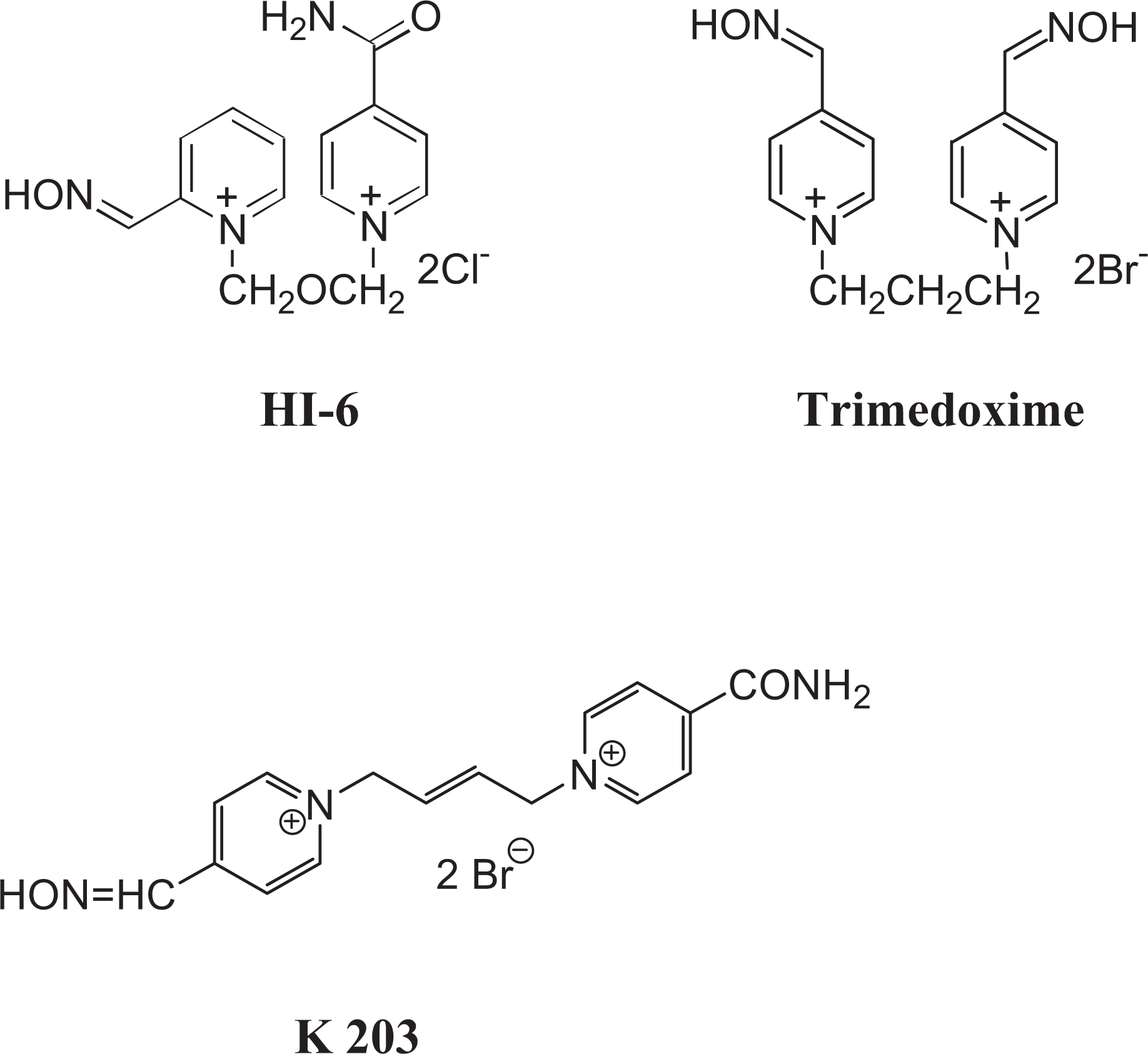
Chemical structure of oximes.
In this article, the reactivating and therapeutic efficacy of chosen mixtures of oximes in combination with atropine was evaluated against VX (O-ethyl S-(2-diisopropylaminoethyl) methyl phosphonothioate) to find how the combination of the oxime HI-6 with another oxime (trimedoxime, K203) can influence the reactivating and therapeutic effectiveness of antidotal treatment of acute VX poisoning. VX is a lipophilic liquid nerve agent with an extremely high toxicity. It can rapidly penetrate the skin because of its lipophilicity and produce toxic effects. 17 The main aim of this study was to compare the reactivating and therapeutic efficacy of 3 individual oximes (trimedoxime, the oxime HI-6, and K203) with 2 mixtures of oximes containing the oxime HI-6 and trimedoxime or the oxime K203 against VX in rats and mice.
Material and Methods
Animals
Male albino Wistar rats weighing 180 to 220 g and NMRI male mice weighing between 20 and 25 g were purchased from VELAZ (Prague, Czech Republic). They were housed in propylene cages (56 × 36 × 19 cm3, 6 rats or 10 mice per cage) in acclimate- and access-controlled rooms (22°C ± 2°C and 50% ± 10% relative humidity). The day/night cycle was 12/12 hours schedule. Food and tap water were available ad libitum. The rats and mice were divided into groups of 8 animals (n = 8). Handling of the experimental animals was done in accordance with the Ethics Committee of the Faculty of Military Health Sciences in Hradec Kralove (Czech Republic).
Chemicals
VX was obtained from the Technical Institute in Brno (Czech Republic) and was 97% pure. Its purity was assayed by acidimetric titration. All oximes (trimedoxime, the oxime HI-6, and the oxime K203) were synthesized at our Department of Toxicology of the Faculty of Military Health Sciences (Czech Republic) and they were more than 98% pure.18,19 Their purity was analyzed using high-performance liquid chromatography (HPLC) technique. 20 All other drugs and chemicals of analytical grade were obtained commercially and used without further purification. All substances were administered intramuscularly (im) at a volume of 1 mL/kg body weight (bw) in rats and 10 mL/kg bw in mice.
In Vivo Experiments
To evaluate the reactivating efficacy of the oximes, either atropine sulfate monohydrate (21 mg/kg) alone or atropine sulfate monohydrate (21 mg/kg) in combination with one of the studied oximes (trimedoxime, HI-6, and K203) or with the mixture of HI-6 with trimedoxime or K203 were administered im in rats. The oximes were administered in equitoxic doses (5% LD50) regardless of the type of antidotal treatment (single oxime, combination of oximes). The LD50 values of all oximes studied in rats were previously published. 16 The oximes in combination with atropine were administered 1 minute15,16 after the rats had received VX im at a dose of 10 μg/kg corresponding to its LD50 value that was assessed using probit-logarithmical analysis of death occurring within 24 hours after im administration of VX at 5 different doses between 9 and 12 µg/kg with 6 rats per dose. 21 The rats were decapitated and exsanguinated to obtain the blood 60 minutes after VX poisoning.15,16 The blood was hemolyzed in Tris-HCl buffer (0.02 mol/L, pH 7.6, 1:20). Tissues (diaphragm and brain) were collected and immediately homogenized in Tris-HCl buffer (0.02 mol/L, pH 7.6, 1:10) to determine AChE activity by a standard spectrophotometric method. 22 Acetylthiocholine was used as a substrate (Tris-HCl buffer, 0.01 mol/L, pH 7.6). Helios Alpha, the spectrophotometer, was used for the determination of absorbancy at 436 nm. The AChE activity was derived from absorbance values with the help of the calibration curve with cystein and expressed as μkat/kg or L (μmol substrate hydrolyzed/kg wet tissue or L blood within 1 second). The percentage of reactivation was calculated using the AChE activity values: {1 − [((saline) − (oxime + atropine))/((saline) − (atropine control))]} × 100 (slightly modified equation from Clement et al). 23
The ability of oximes (single oxime or mixture of oximes) in combination with atropine to reduce acute toxicity of VX in mice was determined as follows. The LD50 value of VX and its 95% confidence limit in nontreated mice was assessed using probit-logarithmical analysis of death occurring 24 hours after im administration of VX agent at 5 different doses with 8 mice per dose. 21 To determine the LD50 of VX without antidotal treatment, we used the doses of VX between 0.5 and 3 µg/kg bw. Then, VX agent-poisoned mice were treated im with atropine alone or with atropine in combination with one of tested oximes or with the mixture of oximes. The oximes were administered at equitoxic doses (5% LD50) in combination with atropine sulfate monohydrate (21 mg/kg) 1 minute after im challenge of VX agent. The LD50 values of all oximes studied in mice were previously published. 16 The LD50 values of VX agent and their 95% confidence limits in the treated VX agent-poisoned mice were assessed by the same method. To determine the LD50 of VX treated with atropine alone, we used the doses of VX between 2 and 10 µg/kg bw. To determine the LD50 of VX treated with atropine and the oxime K203, we used the doses of VX between 5 and 20 µg/kg bw. To determine the LD50 of VX treated with atropine and trimedoxime, we used the doses of VX between 15 and 30 µg/kg bw. To determine the LD50 of VX treated with atropine and the oxime HI-6 alone or in combination with trimedoxime or K203, we used the doses of VX between 40 and 70 µg/kg bw. The efficacy of tested antidotes was expressed as protective ratio (LD50 value of VX in protected mice/ LD50 value of VX in unprotected mice). Statistical significance was determined by the use of 1-way analysis of variance (ANOVA) test with Scheffe’s post hoc test and differences were considered significant when P < .05. Statistical evaluation was determined with the relevant computer programs. 21
Results
The ability of oximes to reactivate VX-inhibited AChE in rat blood, diaphragm, and brain in rats is shown in Table 1. The reactivating efficacy of the oxime K203 and trimedoxime is similar, while the oxime HI-6 seems to be a better reactivator of VX-inhibited AChE among oximes studied in blood and especially in the brain where K203 and trimedoxime were ineffective. When the combination of HI-6 with trimedoxime or K203 was used, the reactivating efficacy of antidotal treatment corresponded to the reactivating efficacy of the most effective single oxime (HI-6) in blood, and it was markedly higher than the reactivating efficacy of all single oximes in diaphragm (P < .05). On the other hand, the reactivating efficacy of both tested combinations of oximes in the brain was slightly lower than the most effective oxime (HI-6) but the difference was not significant.
Percentage of Reactivation of VX-Inhibited AChE by Oximes in Rat Blood, Diaphragm, and Brain After im Administratiom of VX at a Dose of 10 µg/kg bw Corresponding to its LD50 Value
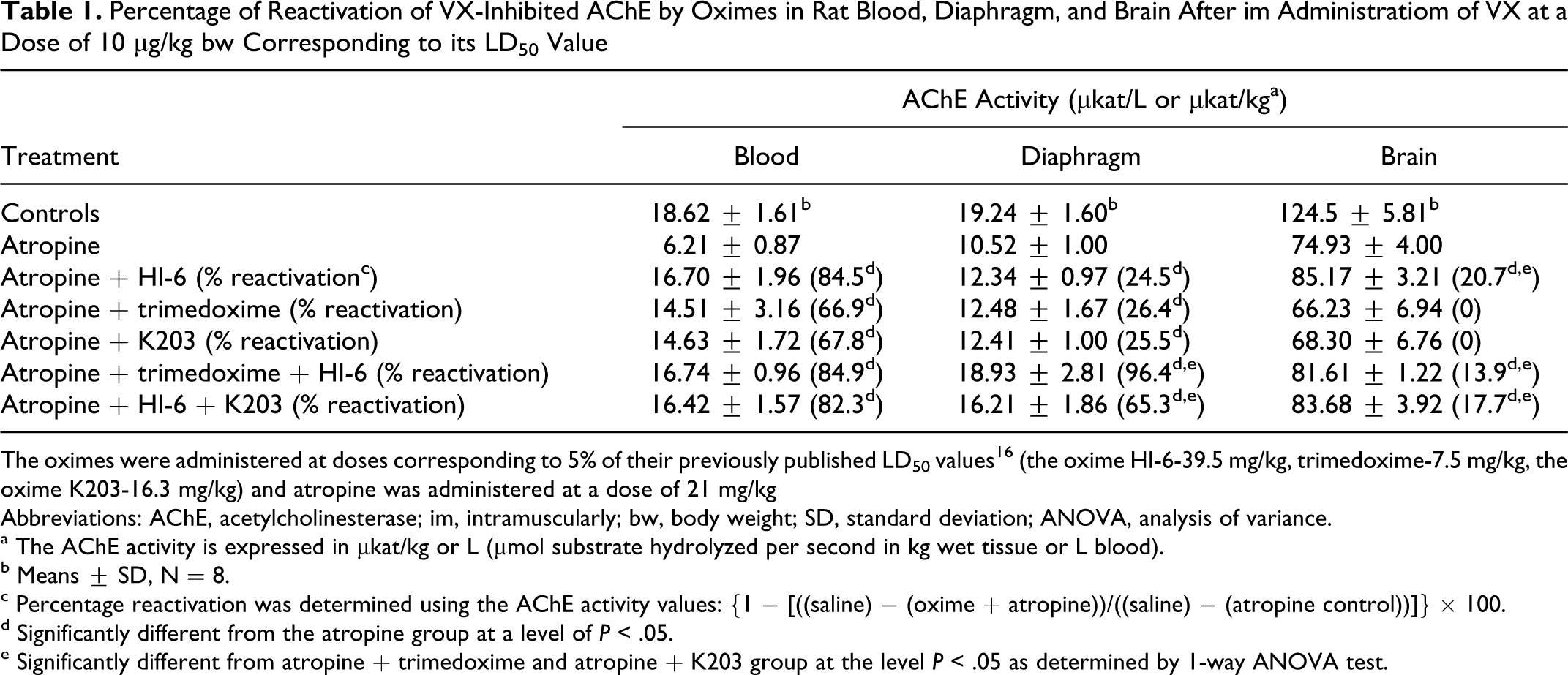
The oximes were administered at doses corresponding to 5% of their previously published LD50 values16 (the oxime HI-6-39.5 mg/kg, trimedoxime-7.5 mg/kg, the oxime K203-16.3 mg/kg) and atropine was administered at a dose of 21 mg/kg Abbreviations: AChE, acetylcholinesterase; im, intramuscularly; bw, body weight; SD, standard deviation; ANOVA, analysis of variance.
a The AChE activity is expressed in µkat/kg or L (µmol substrate hydrolyzed per second in kg wet tissue or L blood).
b Means ± SD, N = 8.
c Percentage reactivation was determined using the AChE activity values: {1 − [((saline) − (oxime + atropine))/((saline) − (atropine control))]} × 100.
d Significantly different from the atropine group at a level of P < .05.
e Significantly different from atropine + trimedoxime and atropine + K203 group at the level P < .05 as determined by 1-way ANOVA test.
Above mentioned results roughly correlate with the therapeutic efficacy of antidotal treatment involving a single oxime or a combination of oximes in mice (Table 2). VX-poisoned mice showed peripheral muscarinic (salivation) and niconitic (tonic–clonic convulsions) signs within a few minutes. Some of them died within 40 to 60 minutes after poisoning when VX was administered at a dose corresponding to its LD50 value. In the case of administration of a single oxime, the oxime K203 was less effective in reducing the acute toxicity of VX than trimedoxime, and both of them were markedly less effective than the oxime HI-6 that was able to reduce acute toxicity of VX more than 36 times. In the case of antidotal treatment of VX-poisoned mice with atropine and combination of HI-6 with trimedoxime or K203, the therapeutic efficacy of antidotes was slightly higher than the therapeutic efficacy of the oxime HI-6 alone (Table 2).
The Influence of the Type of Oxime and Combinations of Oximes on the Ability of Antidotal Treatment to Reduce Acute Lethal Effects of VX a in Mice
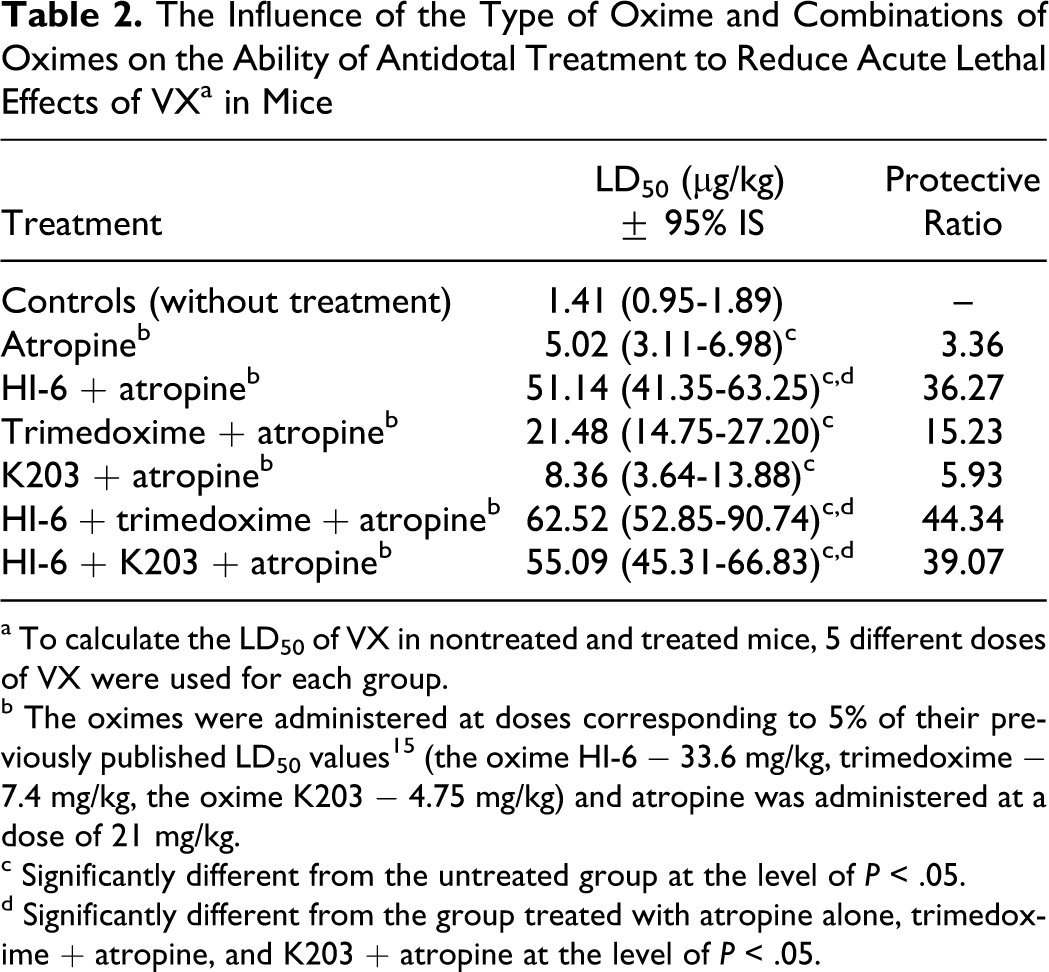
a To calculate the LD50 of VX in nontreated and treated mice, 5 different doses of VX were used for each group.
b The oximes were administered at doses corresponding to 5% of their previously published LD50 values 15 (the oxime HI-6 − 33.6 mg/kg, trimedoxime − 7.4 mg/kg, the oxime K203 − 4.75 mg/kg) and atropine was administered at a dose of 21 mg/kg.
c Significantly different from the untreated group at the level of P < .05.
d Significantly different from the group treated with atropine alone, trimedoxime + atropine, and K203 + atropine at the level of P < .05.
Discussion
The purpose of this study was to compare the reactivating and therapeutic efficacy of 3 individual oximes (trimedoxime, the oxime HI-6, and the oxime K203) with 2 mixtures of oximes containing the oxime HI-6 and trimedoxime or the oxime K203 against VX using rats and mice. Due to the threat of misuse of different nerve agents for military as well as terrorist purposes, the broad-spectrum oxime, sufficiently effective against nerve agents regardless of their chemical structure, is necessary to reach the satisfactorily effective antidotal treatment of nerve agent exposures. As no broad-spectrum oxime has been developed till now,5,13,24 the combination of 2 oximes with their different spectrum of reactivating and therapeutic efficacy seems to be the possible approach of how to increase the efficacy of antidotal treatment of acute poisonings with nerve agents regardless of their chemical structure.25–28
To combine the oximes for the antidotal treatment of acute nerve agent poisonings, the oxime HI-6 should be chosen for oxime combination because it is considered the most promising antidote against many nerve agents.14,29–31 However, its potency to counteract the acute toxicity of tabun is rather low.5,29 Therefore, the second oxime for the antidotal treatment of nerve agent poisonings should be the oxime which is sufficiently effective against tabun. Among currently available oximes, trimedoxime is considered one of the most effective against tabun.5,32,33 In addition, 3 years ago, the oxime K203 [1-(4-carbamoylpyridinium)-4-(4-hydroxyiminomethylpyridinium)-but-2-ene dibromide] (Figure 1) was synthesized at our department of toxicology 18 and evaluated as a promising oxime against tabun.34,35
In vitro evaluation of the ability of the combination of 2 oximes (HI-6 and obidoxime) to reactivate nerve agents-inhibited AChE showed that combining HI-6 and obidoxime had no negative effects on the reactivation of sarin-, cyclosarin-, VX- and tabun-inhibited human AChE and, in addition, it had beneficial effects by broadening the spectrum of the individual oximes. 36 The results published by Worek and his coworkers correspond to ours as well as literature in vivo data from mice, rats, and guinea pigs. Surprisingly, only a few studies are available in literature investigating the effects of combination of oximes against nerve agents in animal models.25–28 They described the beneficial effects of various combinations of oximes to counteract nerve agent-induced acute intoxication. Our results showed that a combination of HI-6 with trimedoxime or K203 was markedly more effective than single oxime treatment related to the reactivation of VX agent-inhibited AChE in diaphragm in rats. On the other hand, the reactivating efficacy of both combinations of oximes did not prevail the ability of the oxime HI-6 to reactivate VX-inhibited AChE in rat blood and brain was demonstrated. The reactivation of VX-inhibited AChE in the brain is generally markedly lower compared to the peripheral compartment because the penetration of oximes across the blood–brain barrier (BBB) is limited due to the presence of positively charged quaternary nitrogens in their molecule. Nevertheless, their presence in the brain in at least small amounts is detected by autoradiography and by HPLC.37,38 The presence of oximes in the brain was also demonstrated indirectly as a result of their ability to reactivate nerve agent-inhibited AChE at the central nervous system (CNS) sites during protection studies.39,40 Our pharmacokinetic studies demonstrated that the level of bispyridinium oximes in the brain tissue corresponds to approximately 1% of their administered doses. 41 Based on these results, we suppose that oximes penetrate BBB in a sufficient concentration to produce a biochemical and physiological action against nerve agent poisoning. We also suppose that the usage of combination of oximes could bring an advantage due to higher concentration of oximes in the brain. Based on our results, both combinations of oximes studied were also slightly more effective than HI-6 alone in the reduction of acute toxicity of VX in mice. The beneficial effect of the same oxime mixtures compared to single oximes was also demonstrated for the antidotal treatment of acute poisoning with tabun. 15 On the other hand, the beneficial effect of oxime mixtures for the antidotal treatment of acute poisoning with soman was not found, probably due to the rapid aging of soman-inhibited AChE. 16 The beneficial effects of combinations of oximes compared to individual oxime treatment could be explained by an elevated plasma oxime level and synergetic effects of both oximes. 36
Conclusion
The above described data demonstrates that antidotal treatment involving a combination of 2 oximes and atropine represents not only a promising treatment of acute poisoning with tabun 15 but also with VX. This nerve agent preferably acts in the peripheral nervous system and the beneficial effect of both oxime combinations is also pronounced in the peripheral compartment (diaphragm). Thus, the combination of 2 oximes seems to be the suitable approach of how to broaden the effectiveness of antidotal treatment of poisonings with nerve agents regardless of their chemical structure.
Footnotes
Acknowledgments
The authors wish to thank to Mrs Jana Uhlirova for her skilful assistance and Dr Blaha for statistical data evaluation.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: grant of Ministry of Defence, No. MO0FVZ0000501
