Abstract
Polyvinyl methyl ether/maleic acid (PVM/MA) copolymer, and its related salts and esters, are used in cosmetics, mainly as binders, film formers, and hair fixatives. Animal and human data relevant to the use of these ingredients in cosmetic products were reviewed by the CIR Expert Panel. The Panel concluded that these ingredients are safe for use in cosmetic products.
Introduction
A safety assessment of the ethyl ester and butyl ester of polyvinyl methyl ether/maleic anhydride or polyvinyl methyl ether/maleic acid (collectively referred to as PVM/MA) copolymers was published in 1993 with the conclusion from the Cosmetic Ingredient Review (CIR) Expert Panel that these chemicals “are safe in neutralized form as cosmetic ingredients in the present practice of use.” 1
The Expert Panel determined that the available data in the original safety assessment on ethyl ester of PVM/MA copolymer and butyl ester of PVM/MA copolymer are sufficient to support the safety of calcium/sodium PVM/MA copolymer, potassium ethyl ester of PVM/MA copolymer, sodium ethyl ester of PVM/MA copolymer, potassium butyl ester of PVM/MA copolymer, Sodium butyl ester of PVM/MA copolymer, isopropyl ester of PVM/MA copolymer, and PVM/MA copolymer. These ingredients consist of the parent copolymer, PVM/MA copolymer, and additional simple esters and salts of this ingredient.
Chemistry
Definition and Structure
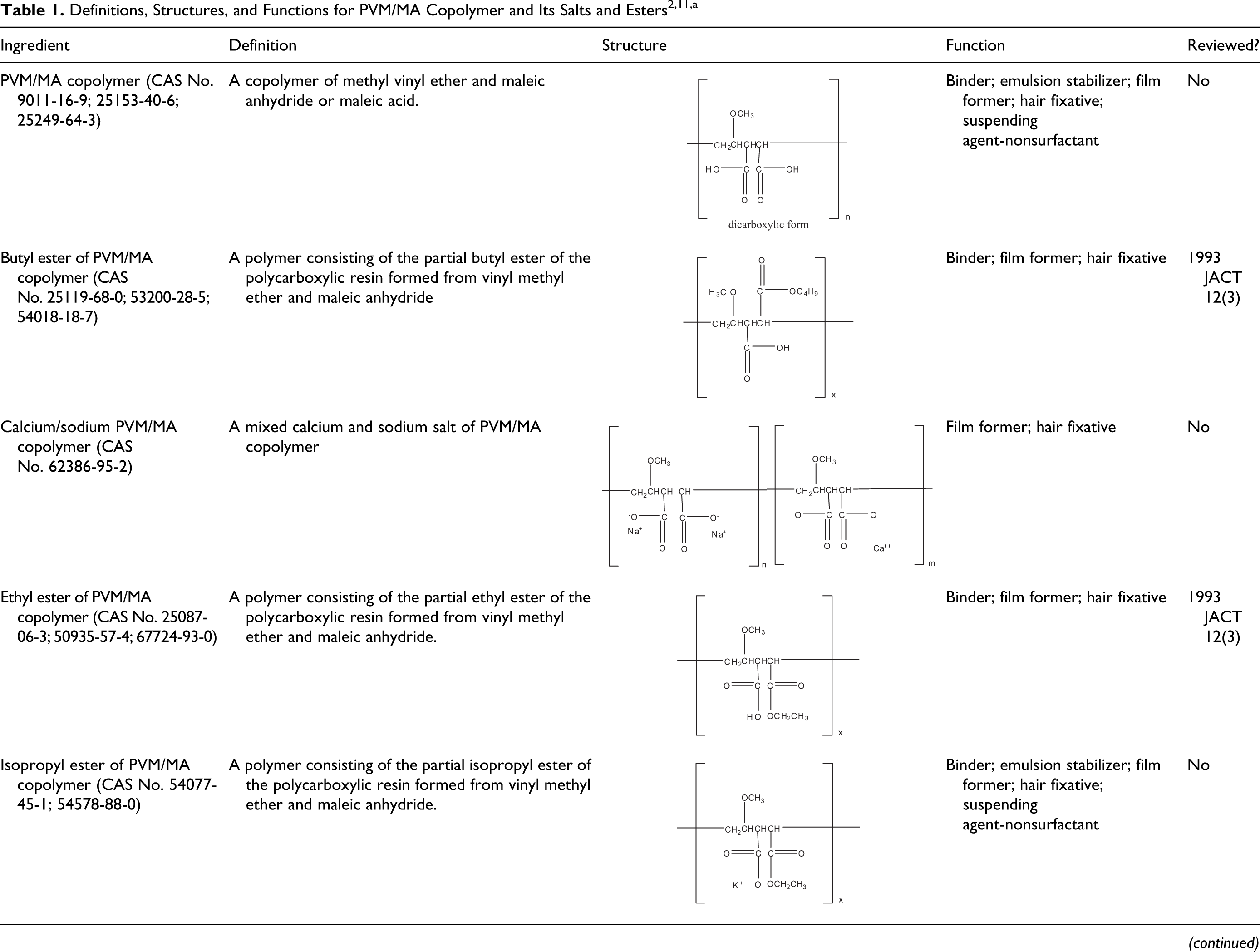
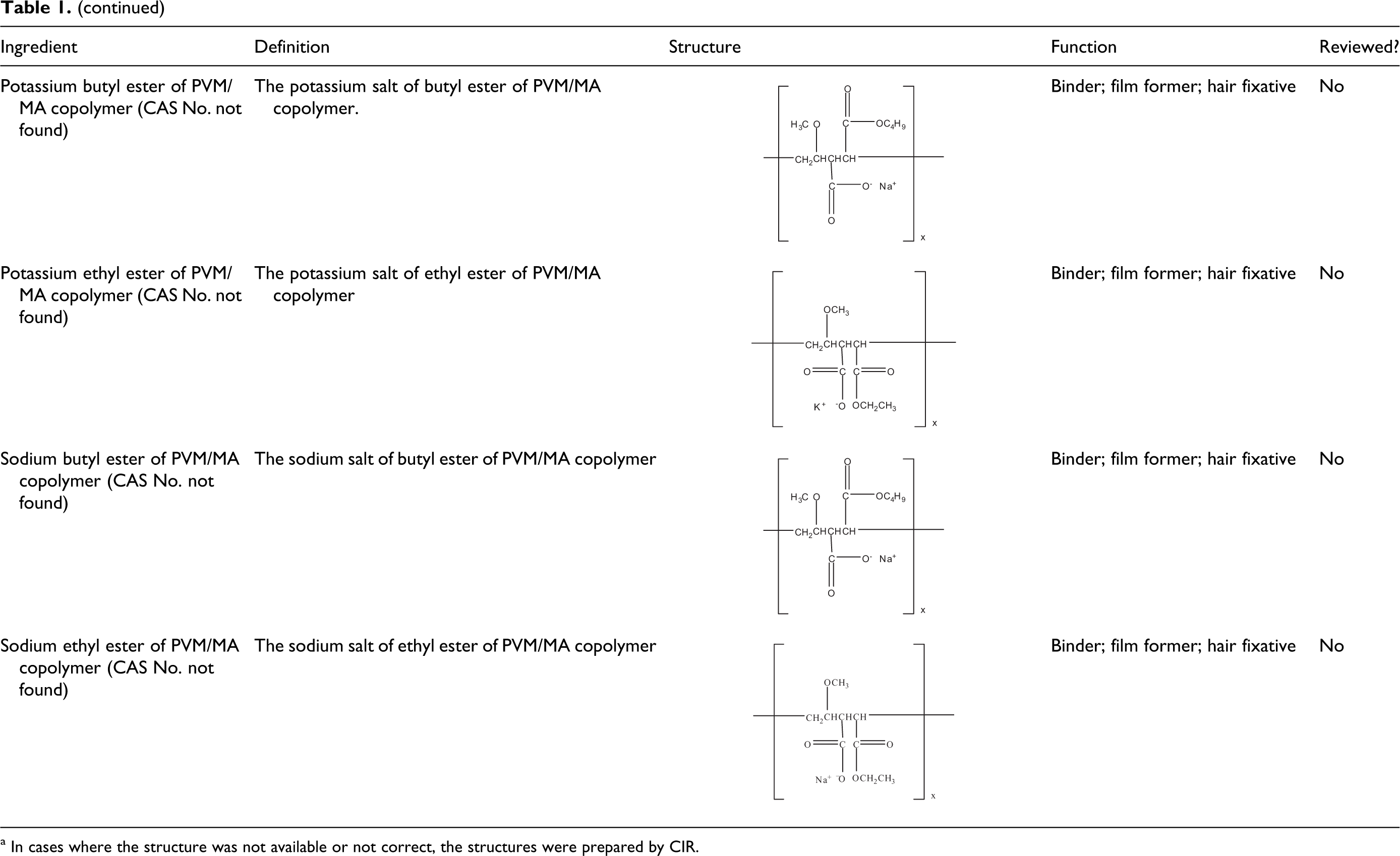
The definitions and structures of the salts and esters of the PVM/MA copolymer ingredients presented in this report are listed in Table 1. Technical and trade names 2 for these ingredients, according to Gottschalck and Bailey, are listed in Table 2.
a In cases where the structure was not available or not correct, the structures were prepared by CIR.
Technical and Trade Names for PVM/MA Copolymers and Its Salts and Esters 2

Physical and Chemical Properties
Butyl ester and ethyl ester of PVM/MA copolymer consist of the partial butyl ester or ethyl ester, respectively, of the polycarboxylic resin formed from vinyl methyl ether and maleic anhydride. 3 The base polymer is a true interpolymer, with a 1:1 molar ratio of vinyl methyl ether to maleic anhydride and an alternating sequence of these 2 monomer units. 4 Butyl ester and ethyl ester of PVM/MA copolymer have free carboxyl groups with an equal number of adjacent ester groups along the chain. 5
Monoesters of PVM/MA copolymer contain a hydrophobic ester group that decreases the penetration of water into the polymer and a solubilizing carboxylic group. 6
Ethyl ester of PVM/MA copolymer and butyl ester of PVM/MA copolymer are supplied as
ethanol solutions that are clear viscous liquids with concentrations of 50%
The physical and chemical properties of PVM/MA copolymer and its salts and esters are summarized in Table 3.
Physical and Chemical Properties of PVM/MA Copolymer and Its Salts and Esters


Impurities
The amount of residual methyl vinyl ether in ethyl and butyl esters of PVM/MA copolymer has not been determined. 7 Methyl vinyl ether is a gas at room temperature (boiling point, 6°C-7°C), and it is therefore expected that very little remains in the polymers. See Table 3 for additional impurity information on specific copolymers.
Manufacture and Production
PVM/MA copolymers are all derived from similar chemistry. 8 Methyl vinyl ether and maleic anhydride are reacted to form the copolymer of methyl vinyl ether and maleic anhydride. This copolymer can then be hydrolyzed to create the copolymer of methyl vinyl ether and maleic acid (the dicarboxylic form).
Ethyl ester of PVM/MA copolymer, butyl Ester of PVM/MA copolymer, and isopropyl ester of PVM/MA copolymer are manufactured by the catalytic polymerization of a solution of maleic anhydride with methyl vinyl ether followed by hydrolysis of the anhydride using ethanol, n-butanol, or isopropyl alcohol to yield monoethyl, monobutyl, or monoisopropyl ester, respectively, of the PVM/MA copolymer. 5,8 For cosmetic use, the free carboxyl groups are neutralized with a choice of bases, although 2-amino-2-methylpropanol is common. 5
Calcium/sodium PVM/MA copolymer is formed by adding a mixture of calcium and sodium bases to the dicarboxylic form of the PVM/MA copolymer. 8
Analytical Methods
Both ethyl ester and butyl ester of PVM/MA copolymer can be identified by adding newly purified resorcin and sulfuric acid to the ingredient, heating it, cooling the solution, and then adding water to dissolve the residue. Sodium hydroxide TS (1M) is added to the solution, which is then observed under ultraviolet (UV) light at 366 nm. The solution generates a green-blue fluorescence. 9
Use
Cosmetic
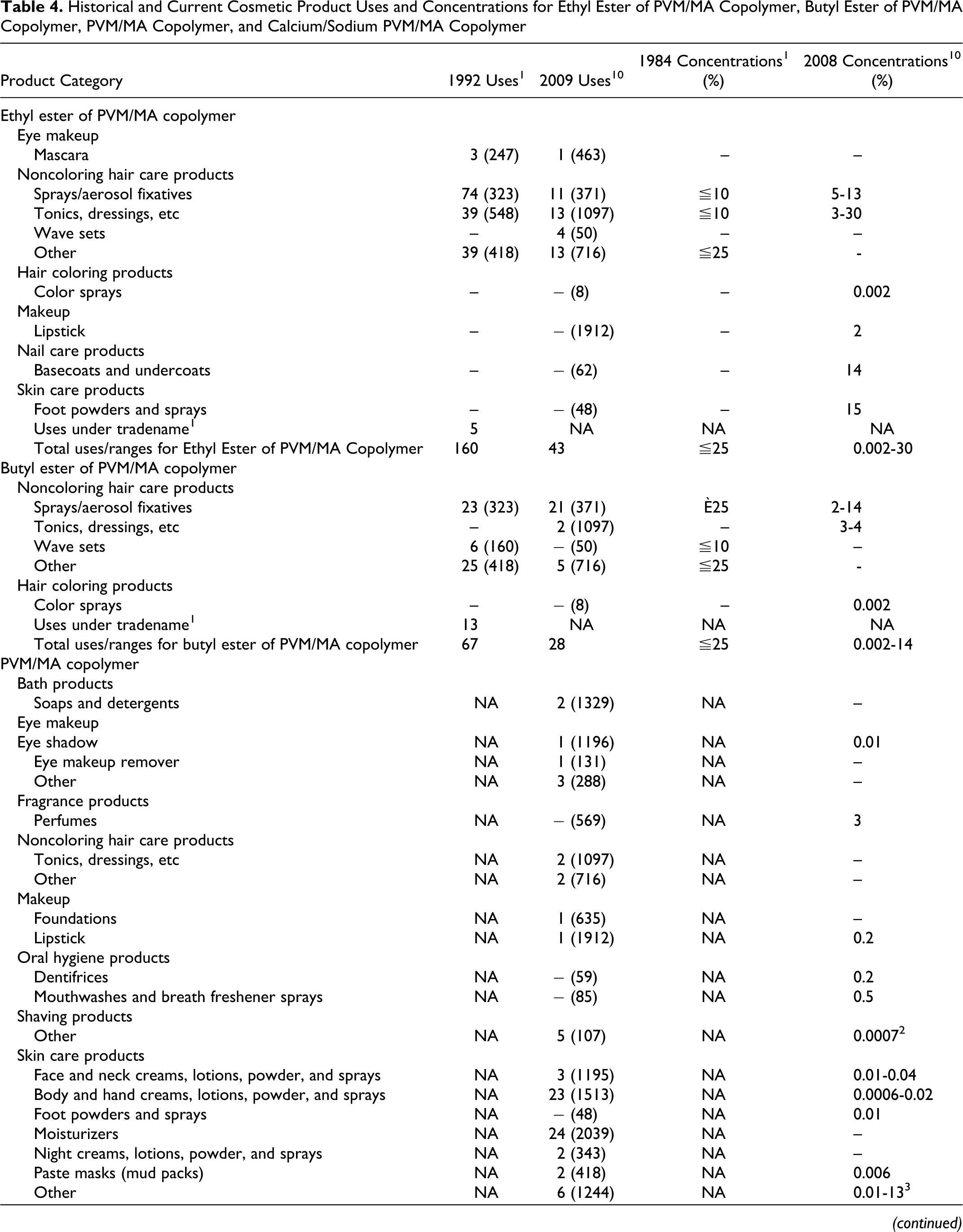
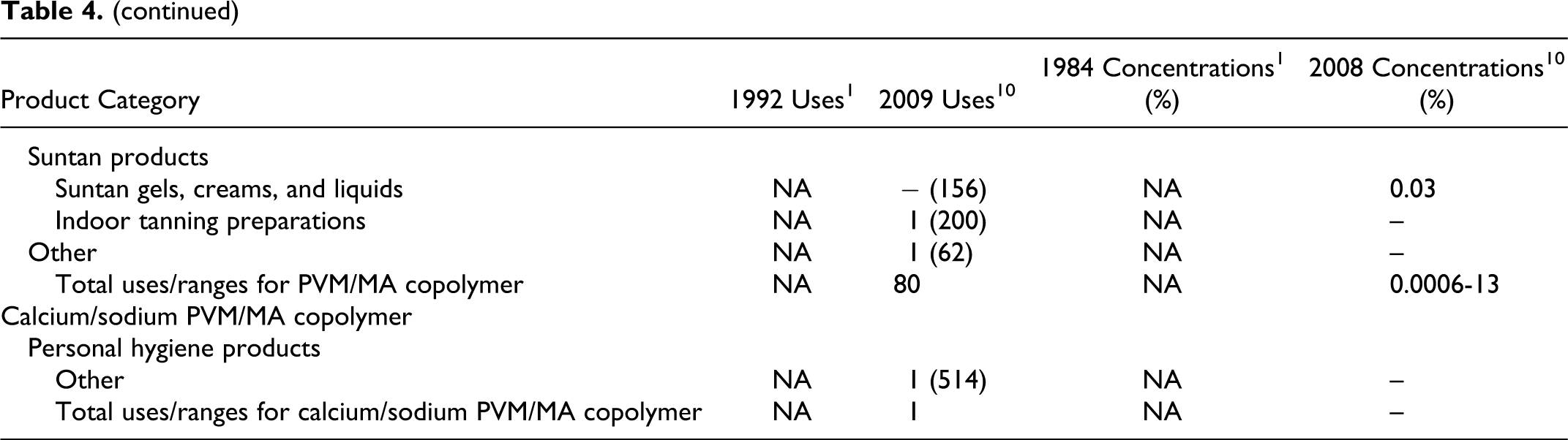
The available product formulation data for the PVM/MA copolymer ingredients are presented in Table 4. At the time of the original safety assessment (1993), ethyl ester of PVM/MA copolymer and butyl ester of PVM/MA copolymer were used in a total of 160 and 67 cosmetic products, respectively, at use concentrations of ≦25% for both ingredients. 1 Current Voluntary Cosmetic Registration Program (VCRP) data indicate that ethyl ester of PVM/MA copolymer and butyl ester of PVM/MA copolymer are used in 42 and 28 cosmetic products, respectively. 11 A survey of current use concentrations conducted by the Personal Care Products Council reported a range from 0.002% to 30% for ethyl ester of PVM/MA copolymer and 0.002% to 14% for butyl ester of PVM/MA copolymer. 12 The total uses for the related ingredients were reported for PVM/MA copolymer (80 uses) and calcium/sodium PVM/MA copolymer (1 use). 11 The Council reported use concentration ranges from 0.0006% to 13% for PVM/MA copolymer. Use concentration data were not available for calcium/sodium PVM/MA copolymer. Use frequency and concentration data were not available for the remaining salts and esters.
Historical and Current Cosmetic Product Uses and Concentrations for Ethyl Ester of PVM/MA Copolymer, Butyl Ester of PVM/MA Copolymer, PVM/MA Copolymer, and Calcium/Sodium PVM/MA Copolymer
PVM/MA copolymer and its salts and esters are used in hair sprays, and effects on the lungs that may be induced by aerosolized products containing this ingredient are of concern.
A panel of 20 women was used to determine the amount of respirable ethyl ester of PVM/MA
copolymer resulting from the use of 2 hair spray formulations, one in an aerosol can and
the other in a pump spray; this study was not an inhalation study.
13
The panelists were fitted with a wig, safety glasses, filter mask, and laboratory
coat and instructed to use the first hair spray in a normal manner. Using open-face
filters and a cyclone sampler, a total of 5 different test air samples were taken over 15
minutes. The open-face filters sampled all airborne material, regardless of particle size,
while the cyclone sampler only collected particles
The potential adverse effects of inhaled aerosols depend on the specific chemical species, the concentration, the duration of the exposure, and the site of deposition within the respiratory system. 14 In general, the smaller the particle, the further into the respiratory tree the particle will deposit and the greater the impact on the respiratory system. 15
Anhydrous hair spray particle diameters of 60 to 80 μm have been reported, and pump hair sprays have particle diameters of ≥80 μm. 16 The mean particle diameter is around 38 μm in a typical aerosol spray. 17 In practice, aerosols should have at least 99% of particle diameters in the 10 to 110 μm range. This means that most aerosol particles are deposited in the nasopharyngeal region and are not respirable.
Jachowicz and Yao studied the properties of hair spray resins, including ethyl ester of PVM/MA copolymer and butyl ester of PVM/MA copolymer, on several hair types. 18 Properties of hair treated with a fixative are affected by the concentration and molecular weight of a polymer, with stiffness ratios increasing with the increase of both these parameters. Higher degrees of neutralization usually increase the moisture sensitivity of a polymer, with decreased stiffness retention at high humidity.
PVM/MA copolymer and its salts and esters are not included among the substances listed as prohibited, restricted, or provisionally allowed in the use of cosmetic products marketed in Japan. 19,20 In addition, these ingredients are not restricted from use in any way under the rules governing cosmetic products in the European Union. 21
Noncosmetic
According to Hawley’s Condensed Chemical Dictionary, PVM/MA copolymer is used as a protective colloid, dispersing agent, thickener, binder, adhesive and “size in coatings,” detergents, paper, textiles, leather, latex, rust preventative, and foam stabilizer. 22
Isopropyl ester of PVM/MA copolymer may have potential use in ophthalmic matrices for drug release in place of eye drops. 6,23 These matrices are put in contact with the conjunctiva for noncorneal drug absorption.
PVM/MA copolymer is included in the Code of Federal Regulations (CFR) at 21 CFR §175.105 and 21 CFR §175.125 for its approved use in indirect food contact applications as adhesives and pressure-sensitive adhesives. 24,25 It is also included in 21 CFR §872.3500 for its use in dental devices. 26 Calcium/sodium PVM/MA copolymer is included in 21 CFR §872.3490 for its use in dental devices (dental adhesives). 27 PVM/MA copolymer and its butyl ester are listed on the Food and Drug Administration (FDA) inactive database for uses in a dental paste and a topical solution at 30%, respectively. 28
Absorption, Distribution, Metabolism, and Excretion
No studies concerning the absorption, distribution, metabolism, or excretion of PVM/MA copolymer or its salts or esters were found in the published literature.
Animal Toxicology
Acute Oral Toxicity
Butyl and ethyl esters of PVM/MA copolymer
Five groups of 5 albino Sherman-Wistar rats, sex not specified, were used to determine the oral LD50 of ethyl ester of PVM/MA copolymer and butyl ester of PVM/MA copolymer. 29,30 The test articles, evaporated to dryness and suspended in corn oil, were administered to fasted rats using a syringe and stomach tube. The dosages tested ranged from 1.6 to 25.6 g/kg. All groups were observed for 2 weeks after dosing. The oral LD50 of both ethyl ester and butyl ester of PVM/MA copolymer was >25.6 g/kg
Butyl ester of PVM/MA copolymer had an oral LD50 of >10 g/kg in rats (gender and strain not specified) and >2 g/kg in dogs (gender and strain not specified). 31
Short-Term Oral Toxicity
Butyl ester of PVM/MA copolymer
A group of 6 female Holtzman rats was fed a diet containing 25% butyl ester of PVM/MA copolymer daily for 10 days. 32 A group of 6 female Holtzman rats were fed a similar diet, with lactose replacing butyl ester of PVM/MA copolymer and used as a control group. Physical observations were made and the animals were weighed prior to receiving each dose. All animals were killed on day 10 and their livers were removed. There were no significant differences in either the wet or dry liver weights or liver-to-body-weight ratios between the treated and control groups. No gross physical changes were observed in the test animals.
Subchronic Inhalation Toxicity
Ethyl ester of pvm/ma copolymer
Four groups of Sprague-Dawley rats, 15 males and 15 females per group, were used in an inhalation study to assess the toxicity of ethyl ester of PVM/MA copolymer. 33 The rats were placed in inhalation chambers for 4 hours plus equilibration, if necessary, 5 days/week for 13 weeks, for a total of 65 exposures.
The mean chamber concentrations of solids based on 65 determinations as measured by
gravimetric analysis over the entire study were 0.017
Determination of the particle size distribution of the aerosol reported the geometric mean (50% size) to be approximately 20, 3.2, 3.4, and 1.6 μm for the control, low-, mid-, and high-dosage groups, respectively. The difference in particle size for the high-dosage group compared to the low- and mid- dosage groups was probably due to the mechanics of delivery of the aerosol to the chamber and was acceptable. The amount of the aerosol <10 μm (considered respirable) was approximately 38%, 71%, 73%, and 88% for the control, low-, mid-, and high-dose groups, respectively. The control group values were not considered meaningful since the background values were so low.
Body weights and 24-hour feed consumption were determined immediately prior to dose initiation; weekly measurements were made during the study. All rats were observed before and after dosing for signs of toxicity or moribundity, and all animals were observed on nontreatment days. Blood and urine samples were taken from 8 nontest rats/gender at study initiation and from 8 randomly selected rats/gender/group during week 6 of the study and at study termination. After 13 weeks of dosing, all animals were killed for necropsy, and a microscopic examination was performed on selected tissues.
There were no significant differences in mean body weights between the treated and control groups, neither during the study nor at study termination. At various times during the study, feed consumption was significantly decreased for all test groups, both males and females, when compared to control values. This did not appear to be dose related.
With the exception of 2 mid-dosage females, pharmacotoxic observations were similar for all test and control animals. The 2 mid-dosage animals had alopecia starting at week 7 or 11 and continuing until study termination. This lesion was not observed in the other dosage groups.
At week 6, females of all test groups had significantly increased erythrocyte counts, females of the low-dosage group had a significantly increased total leukocyte count, and females of the low- and high-dosage groups had decreased blood glucose values compared to the controls. No significant differences in either these or other values were observed at study termination.
At necropsy, the absolute heart weights of females of the low- and mid-dosage groups and the absolute kidney weights of females of the low-dosage group were decreased compared to the controls. However, there were no significant differences observed in any of the organ-to-body-weight ratios for either these groups or any other group. Upon microscopic examination, the lungs of animals of the mid- and high-dosage groups had “scattered small foci of foamy cells” (alveolar macrophages). Ethyl ester of PVM/MA copolymer did not produce any obvious signs of toxicity.
Butyl ester of PVM/MA copolymer
Groups of Sprague-Dawley rats were used in an inhalation study to assess the toxicity of butyl ester of PVM/MA copolymer. 34 The control, low-dosage, and mid-dosage groups consisted of 15 males and 15 females per group and the high-dosage group consisted of 30 males and 30 females. The study was conducted in the same manner as for the ethyl ester of PVM/MA copolymer described above.
The actual mean chamber concentrations of solids based on 65 determinations as measured
by gravimetric analysis over the entire study were 0.027
The particle size distribution of the aerosol reported as the geometric mean (50% size) was 7.0, 3.4, 2.2, and 1.4 μm for the control, low-, mid-, and high-dose groups, respectively. The amount of the aerosol <10 μm (considered respirable) was 66%, 72%, 73%, and 82% for the control, low-, mid-, and high-dose groups, respectively. The control group values were not considered meaningful since the background values were so low.
At various times during the study, mean body weights of males of the mid-dosage group were significantly decreased compared to control values. At various times throughout the study, feed consumption was significantly decreased in males and females of the mid- and high-dosage groups or significantly increased in males of the high-dosage group and in females of all dosage groups when compared to control values.
Pharmacotoxic observations were similar for all test and control animals. The results of clinical chemistry, hematology, and urinalysis were similar for treated and control animals. The only difference observed was a significantly increased urine albumin value for females of the high-dosage group at week 6.
After 13 weeks of dosing, absolute lung weights were significantly decreased in males of the mid-dosage group and the lung-to-body-weight ratios were significantly decreased in females in all 3 dosage groups compared to control values. No significant microscopic findings were noted. There were no significant observations for animals of the high-dosage recovery group. No toxicological effects were produced by butyl ester of PVM/MA copolymer.
Chronic Toxicity
No chronic toxicity studies of PVM/MA copolymer, its salts, or esters were found.
Ocular Irritation
Butyl and ethyl esters of PVM/MA copolymer
Two groups of albino rabbits, 6 per group, were used to determine the ocular irritation index of ethyl ester of PVM/MA copolymer and butyl ester of PVM/MA copolymer. 35,36 Both ingredients were tested as a 5% weight/volume (w/v) solution in distilled water; the pH was adjusted to 6.1 or 6.7 using aminoethyl propanediol (AMPD) for ethyl ester and butyl ester of PVM/MA copolymer, respectively. A volume of 0.1 mL of test solution was applied to the conjunctival sac of the right eye of each rabbit; the eyes were not rinsed. The left eye served as the control. The eyes were examined for ocular lesions 24, 48, and 72 hours after application of the test material and scored according to the Draize method. Neither ethyl ester of PVM/MA copolymer nor butyl ester of PVM/MA copolymer was an ocular irritant.
Isopropyl ester of PVM/MA copolymer
Finne et al investigated the use of ophthalmic matrices for drug release.
23
The matrices consisted of isopropyl ester of PVM/MA copolymer (50% isopropanol
solution of the polymer), 0.12 mmol timolol maleate, and 2.11 mmol of sodium acetate and
were 5 mm in diameter and 0.59
Dermal Irritation
Ethyl and butyl esters of PVM/MA copolymer
Two groups of albino rabbits, 6 per group, were used to determine the primary dermal irritation of ethyl ester of PVM/MA copolymer and butyl ester of PVM/MA copolymer. 37,38 Both ingredients were tested as a 5% w/v solution in distilled water; the pH was adjusted to 6.1 or 6.7 using AMPD for ethyl ester and butyl ester of PVM/MA copolymer, respectively. Ethyl and butyl esters of PVM/MA copolymer were not primary irritants as defined by the Federal Hazardous Substances Labeling Act.
Reproductive and Developmental Effects
No studies concerning the reproductive and development effect of PVM/MA copolymer, its salts or esters were found in the existing published literature.
Genotoxicity
Butyl ester of PVM/MA copolymer
A mouse lymphoma mutation assay was performed on butyl ester of PVM/MA copolymer to determine the ability of the test material to induce forward mutations in the L5178Y TK+/- mouse lymphoma cell line. 39 Dimethylsulfoxide (DMSO) was used as the solvent. The test material was soluble in the culture medium at concentrations of ≦125 nL/mL; precipitate formed at concentrations 2250 nL/mL. In a preliminary cytotoxicity assay, 24-hour cell growth was only slightly reduced by a concentration of 1000 nL/mL butyl ester of PVM/MA copolymer.
Butyl ester of PVM/MA copolymer was assayed at concentrations of 15.6 to 2000 nL/mL with metabolic activation and at concentrations of 500 to 2000 nL/mL without metabolic activation. Three trials were performed with and 1 without metabolic activation. Negative controls, solvent and untreated media, and positive controls, dimethylnitrosamine (DMN) with activation and ethylmethane sulfate (EMS) without activation, were used.
Without metabolic activation, mutant frequency remained comparable to the negative control values at all concentrations. A range of toxicities from weak to moderate was observed; however, the degree of toxicity did not increase as a function of increased concentration. At a concentration of 2000 nL/mL, relative growth was decreased to 35.4%.
In the first acceptable trial with metabolic activation, mutant frequency was not increased as compared to the controls for all dosages except the 2000 nL/mL dosage. For the concentration range of 15.6 to 2000 nL/mL, weak-to-moderate toxicity was observed.
The second trial used duplicate treatments of 2000 nL/mL to evaluate the mutant frequency results obtained in the first trial. In one experiment, the concentration was too toxic and mutant analysis could not be performed. In the second experiment with 2000 nL/mL, moderate-to-high toxicity was observed, but mutant frequency did not vary significantly from the negative control values. (Therefore, the increase observed in trial 1 could not be confirmed.) At the other concentrations used, the mutant frequency was similar to that observed for the negative controls.
Butyl ester of PVM/MA copolymer was not mutagenic with or without metabolic activation in the mouse lymphoma forward mutation assay.
An in vitro transformation of Balb/3T3 cells assay was performed using butyl ester of PVM/MA copolymer in order to evaluate its carcinogenic potential. 40 Butyl ester of PVM/MA copolymer was completely insoluble in culture medium at a concentration of 1.0 mg/mL; therefore, DMSO was used as the solvent.
A preliminary cytotoxicity test using concentrations of 0.06 μg/mL to 1.0 mg/mL resulted in survival rates of 71% to 85%. A second preliminary cytotoxicity test used concentrations of 0.313 to 10.0 mg/mL. The survival rates were 46.5% to 90.5% for the concentrations of 0.313 to 2.5 mg/mL; no survivors were observed at 5.0 or 10.0 mg/mL.
The transformation assay used concentrations of 0.313 to 5.0 mg/mL butyl ester of PVM/MA copolymer. A negative control, solvent, a historical negative control, and a positive control, 3-methylcholanthrene (MCA), were used. The results obtained with the negative control differed significantly from the historical control value; therefore, experimental results were evaluated independently.
Butyl ester of PVM/MA copolymer produced absolute and dosage-related increases in the number of transformed foci at all concentrations; these increases were statistically significant at concentrations of 2.5 and 5.0 mg/mL. Butyl ester of PVM/MA copolymer was weakly active in the in vitro transformation of Balb/3T3 cells assay.
An unscheduled DNA synthesis (UDS) assay using primary rat hepatocytes was performed on butyl ester of PVM/MA copolymer. 41 Dimethylsulfoxide was used as the solvent. Butyl ester of PVM/MA copolymer was assayed at concentrations of 9.77 to 5000 μg/mL. A negative control, solvent, and a positive control, 2-acetyl aminofluorene (2-AFF) were used. Freshly isolated cells were exposed to [ 3 H]thymidine in addition to the test material to improve the results.
An exposure of 625 to 5000 μg/mL butyl ester of PVM/MA copolymer produced rounded and loosely attached cells. Although these cells were still viable, they were not considered surviving cells. There were no survivors at concentrations of 2500 and 5000 μg/mL and a very low number of survivors at concentrations of 625 and 1250 μg/mL were used. As concentrations decreased, survival rate increased. An analysis of nuclear labeling was performed at all concentrations. Butyl ester of PVM/MA copolymer did not produce a significant amount of UDS in primary rat hepatocytes and was inactive in the assay.
Carcinogenicity
No carcinogenicity studies of PVM/MA copolymer, its salts or esters were found in the existing published literature.
Clinical Assessment of Safety
Irritation/Sensitization
Ethyl and butyl esters of PVM/MA copolymer
Ethyl ester and butyl ester of PVM/MA copolymer were applied to participants
simultaneously to evaluate the human irritation and sensitization potentials of the test articles.
42
A repeated insult patch test (RIPT) was performed using the Draize-Shelanski
method. The test materials were applied as supplied (50%
Patches were applied to the scapular area of the back 3 times a week for 3 weeks for a total of 9 induction applications. The sites were evaluated for irritation 24 hours after application. After the ninth patch, there was a nontreatment period of at least 10 days. A challenge patch was then applied to a site adjacent to the induction site for 24 hours. Both sites were graded upon patch removal and 48 and 72 hours after the challenge application.
During the induction phase of the study, 0.21% of the applications induced skin irritation reactions that were regarded as “singular, transient occurrences of a slight nature.” Ethyl ester and butyl ester of PVM/MA copolymer were neither irritants nor sensitizers.
Photoallergenicity and Contact Allergenicity
Ethyl ester of PVM/MA copolymer
The photoallergenic and contact allergenic potentials of ethyl ester of PVM/MA
copolymer were evaluated.
43
The test material was applied as supplied (50%
Patches were applied to the inner aspect of the forearm on Mondays, Wednesdays, and Thursdays until 10 doses were received. For half of the participants, the inner aspect of the right arm was irradiated; for the other half, it was the inner aspect of the left arm. The opposite arm served as the control (nonirradiated) site. The irradiated sites were exposed to ultraviolet A (UVA) radiation for 15 minutes at a distance of 10 cm from the source, resulting in a dosage of 4400 μW/cm2. To avoid irradiation of a test site on 2 consecutive days, Thursdays’ patches were applied to an adjacent site, which was irradiated on Friday; the first site was considered the original site at challenge. The application sites that were not irradiated were covered with black adhesive tape to prevent UV exposure.
After a 10- to 13-day nontreatment period, a challenge patch was applied to an untreated site adjacent to the original site. After 24 hours, the patches were removed and the test sites were graded for irritation. The challenge site was then irradiated and examined 24 and 48 hours after irradiation.
Only slight transient reactions were observed during the study. Ethyl ester of PVM/MA copolymer did not induce either a photoallergenic or a contact allergenic response in human participants.
Oral Health
Numerous clinical studies on the efficacy of dentifrices containing PVM/MA copolymer for several dental health parameters were discovered in the published literature. No adverse effects from exposure to PVM/MA copolymer were reported in any of these studies. Table 5 contains a brief overview of these studies.
Summary of Clinical Studies of Dentifrices Containing PVM/MA Copolymer.


Summary
PVM/MA copolymer is the polymer of vinyl methyl ether and either maleic anhydride or maleic acid. Butyl ester of PVM/MA copolymer, ethyl ester of PVM/MA copolymer, and isopropyl ester of PVM/MA copolymer are polymers consisting of the partial butyl ester, ethyl ester, or isopropyl ester, respectively, of the polycarboxylic resin formed from vinyl methyl ether and maleic anhydride. These esters both have free carboxyl groups with an equal number of adjacent ester groups along the chain. The free carboxyl groups are neutralized for cosmetic use.The salts of these polymers are formed by adding the salt bases to the dicarboxylic form of the PVM/MA copolymer or the esters of PVM/MA copolymer.
The butyl and ethyl esters are supplied as clear viscous ethanol solutions with
concentrations of 50%
No uses or concentrations were reported for the remaining salts and esters of PVM/MA copolymer discussed in this report. The mean particle size for 2 formulations ranges from 36 to 42 μm, which is greater than the size that is considered respirable.
No studies concerning the absorption, distribution, metabolism, elimination, or other general biology of PVM/MA copolymer or its salts or esters were found in the existing published literature.
Ethyl ester and butyl ester of PVM/MA copolymer were relatively harmless to rats according to the terminology of Hodge and Sterner, with both compounds having an oral LD50 for rats of >25.6 g/kg. 44 For dogs, butyl ester of PVM/MA copolymer had an LD50 of >2 g/kg. Butyl ester of PVM/MA copolymer was not toxic in a short-term oral toxicity study. Ethyl ester and butyl ester of PVM/MA copolymer were not toxic in subchronic inhalation studies. Ethyl and butyl esters of PVM/MA copolymer were neither ocular nor primary dermal irritants.
Butyl ester of PVM/MA copolymer was not mutagenic in a mouse forward mutation assay with or without metabolic activation, was weakly active in the in vitro transformation of Balb/3T3 cells assay, and was inactive in a UDS assay.
Ethyl and butyl esters of PVM/MA copolymer were neither irritants nor sensitizers in an RIPT using human participants. Ethyl ester of PVM/MA copolymer did not produce a photoallergenic or contact allergenic response in humans. PVM/MA copolymer did not produce any adverse effects in clinical studies of the ingredient in dentifrices.
Discussion
While very few toxicity studies were identified specifically in the published literature for the additional salts and esters that were added to this safety assessment, there is no reason to expect the parent copolymer or its salts and esters to differ in toxicity from butyl ester of PVM/MA copolymer and ethyl ester PVM/MA copolymer. The salts and esters of the expanded group of PVM/MA copolymer ingredients are expected to have similar toxicological profiles as the butyl and ethyl esters. In solution, the salts are expected to dissociate in any product formulation independent of whether the salt is calcium, sodium, or potassium. The esters likely will break down into their component parts, none of which present any safety issues. The PVM/MA copolymer-derived esters that have been added to this safety assessment, that is isopropyl ester of PVM/MA copolymer, do not raise any significant toxicity concerns. Accordingly, the available data for butyl ester of PVM/MA copolymer and ethyl ester of PVM/MA copolymer are considered supportive of the safety of the expanded group of derivatives as used in cosmetics. Therefore, the Expert Panel determined that the toxicity data on butyl ester of PVM/MA copolymer and ethyl ester of PVM/MA copolymer could be extrapolated to include calcium/sodium PVM/MA copolymer, potassium ethyl ester of PVM/MA copolymer, sodium ethyl ester of PVM/MA copolymer, potassium butyl ester of PVM/MA copolymer, sodium butyl ester of PVM/MA copolymer, isopropyl ester of PVM/MA copolymer, and PVM/MA copolymer.
The Expert Panel recognizes that use concentration data are not available for all ingredients in this group and that some ingredients in this group are not in current use. The Panel considers that the use concentrations for the ingredients that are in use are not likely to be different from the use concentration for butyl ester of PVM/MA copolymer and ethyl ester of PVM/MA copolymer.
The Expert Panel noted the absence of reproductive and developmental toxicity and carcinogenicity data. Because these ingredients are very large molecular weight structures, the Panel considered that they would not be easily absorbed into the skin. While hydrolysis products may form as a result of any oral intake, the resulting compounds separately are considered safe. In addition, there are no structural alerts for toxic activity of these compounds.
The Expert Panel recognizes that the amount of the ingredient that can be respired is an important factor and, therefore, examined this issue. Data submitted to the Panel indicate that the average particle size of ethyl and butyl esters of PVM/MA copolymer is larger than the size that is considered respirable (ie, ≦10 μm). In the subchronic inhalation animal studies presented in this report, the particle size of the test material was smaller than the particle size normally used in cosmetic formulation; therefore, a greater amount of material was respired by the animals as compared to usual human use. In the absence of inhalation toxicity data for the remaining PVM/MA copolymer ingredients, the Panel determined that PVM/MA copolymer and its salts and esters can be used safely in hair sprays, because the particle sizes of the ingredients are not respirable. The Panel reasoned that the particle size of aerosol hair sprays (−38 μm) and pump hair sprays (>80 μm) is large compared to respirable particulate sizes (≦10 μm).
Amended Conclusion
The CIR Expert Panel concludes that PVM/MA copolymer, butyl ester of PVM/MA copolymer, calcium/sodium PVM/MA copolymer, ethyl ester of PVM/MA copolymer, isopropyl ester of PVM/MA copolymer, potassium butyl ester of PVM/MA copolymer, potassium ethyl ester of PVM/MA copolymer, sodium butyl ester of PVM/MA copolymer, and sodium ethyl ester of PVM/MA copolymer are safe as cosmetic ingredients in the practices of use and concentration as described in this safety assessment (note 1).
Footnotes
Acknowledgment
The Cosmetic Ingredient Review Program is financially supported by the Personal Care Products Council. No potential conflict of interest relevant to this article was reported. F. Alan Andersen, PhD, and Christina L. Burnett are employed by Cosmetic Ingredient Review.
The 2009 Cosmetic Ingredient Review Expert Panel members are Chairman, Wilma F. Bergfeld, MD, FACP; Donald V. Belsito, MD; Ronald A. Hill, PhD; Curtis D. Klaassen, PhD; Daniel C. Liebler, PhD; James G. Marks Jr, MD, Ronald C. Shank, PhD; Thomas J. Slaga, PhD; and Paul W. Snyder, DVM, PhD. The CIR Director is F. Alan Andersen, PhD. This report was prepared by Christina L. Burnett, CIR Scientific Analyst/Writer. The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.