Abstract
The protective effects of red ginseng extract and ginseng wine against ethanol-induced male reproductive toxicity were evaluated in male mice using computer-assisted sperm analysis. Mice were divided into 4 groups of 10 and fed plain saline, 6 g/kg per d of ethanol in saline, red ginseng extract plus ethanol, or a fermented preparation of red ginseng extract daily for 5 weeks. We found that the average seminal vesicle weight was significantly lower in the ethanol-treated group compared to the control group, while those of the ginseng-treated groups tended to be higher than the ethanol-treated group. We found a significant decrease in sperm motility and progressiveness in mice treated with ethanol for 5 weeks, while administration of ethanol plus red ginseng extract appeared to minimize the negative effects of ethanol toxicity on male fertility. Serum testosterone, luteinizing hormone (LH), and follicle stimulating hormone (FSH) were insignificantly lower in the ethanol-treated group than in the control group.
Introduction
Ethanol has been shown to inhibit the male reproductive system in rodents, monkeys, and humans. 1 –3 Numerous studies have shown the effects of ethanol on the reproductive system at several target sites, 4 –7 including altered spermatogenesis, altered testicular and accessory gland morphology, reduction in reproductive organ weight, reduced caudal epididymal sperm motility and content, and impaired epididymal sperm maturation. 8 –10 Furthermore, ethanol abuse in males has been correlated with hypogonadism, feminization, decreased testosterone production, reduced libido, ejaculation problems, and impotence. 11 –13
Korean ginseng (the root of Panax ginseng C. A. Meyer) has been used in herbal medicine as a tonic for approximately 2000 years in Asian countries such as Korea, China, and Japan and remains one of the most valued herbal products in the world. 14 Ginseng saponins, commonly known as ginsenosides, have been studied to elucidate the pharmacological properties responsible for their medicinal effects. 15 Specifically, ginseng and its components have been reported to have anticancer, 16 antihypertensive, 17 antidiabetic, 18 and antistress 19 effects. Research has shown that ginseng improves male gonadal function 20 and protects against testicular toxicity. 21 In addition, a tissue-cultured root of wild P ginseng exhibited therapeutic effects on 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD)-induced spermatogenetic disorder. 22 Recently, ginsenosides have been shown to increase human sperm motility in vitro. 23,24 However, to our knowledge there is no literature regarding ginseng’s preventive or therapeutic potential against the toxic effect of ethanol on male reproductive function. In this study, the protective effects of red ginseng extract and ginseng wine on ethanol-induced reproductive toxicity was investigated in male mice using computer-assisted sperm analysis (CASA) to evaluate sperm quality. Changes in the weight of reproductive organs and in levels of sex hormones were recorded and evaluated.
Materials and Methods
Chemicals
Ethanol was purchased from Merck (Germany). The testosterone enzyme-linked immunosorbent assay (ELISA) kit was obtained from IBL International (Germany). Luteinizing hormone (LH) and follicle stimulating hormone (FSH) ELISA kits were purchased from Endocrine Technologies (California).
Red Ginseng Extract
We extracted 500 g of dried Korean red ginseng (supplied by the Ginseng Genetic Resource Bank) with 5 L of 50% ethanol at 80°C for 24 hours with stirring. The extraction procedure was repeated 2 times and the collected solution was filtered through a filter paper. The solution was concentrated in a vacuum evaporator and then pulverized after freeze-drying.
Alcoholic Fermentation for Red Ginseng Wine Preparation
The red ginseng extracts (100 g/L) were autoclaved, cooled to room temperature, then inoculated with a 3-day YPD broth culture of Saccharomyces bayanus to a final concentration of 1%. The yeast cultures were incubated at 30°C for 20 days with swirling at 150 rpm. The ethanol content of the red ginseng wine was analyzed by gas chromatography using a flame ionization detector and carbowax-fused silica capillary column (30 m, 0.25 mm id, 0.25 μm, Macherey-Nagel, Duren, Germany). The column temperature was held at 40°C for 1 minute, then increased to 230°C at a rate of 10°C/min, holding for 5 minutes. The temperature of the injector and detector was set at 260°C. Helium was used as carrier gas and the sample injection volume was 1.0 mL. The final ethanol content of the red ginseng wine was 15.0%.
High-Performance Liquid Chromatography Analysis of Ginsenosides
The ginsenosides of the red ginseng extract and ginseng wine were analyzed by high-performance liquid chromatography (HPLC) using an Alltech Prevail Carbohydrate ES column (4.6 × 250 mm, Alltech Associates, Illinois). The mobile phase was acetonitrile (solvent A) and water (solvent B). Gradient elutions were performed as follows: 15% (A) for 5 minutes, 21% (A) for 20 minutes, 38% (A) for 30 minutes, 52% (A) for 15 minutes, 70% (A) for 10 minutes, 90% (A) for 12 minutes, and 15% (A) for 18 minutes. The injection volume was 20 μL, the solvent flow rate was 1.6 mL/min, and the UV absorbance was monitored at 203 nm.
Animals
Six-week-old male C57BL/6J mice weighing 18 to 20 g (KOATECH, Animal Breeding Center, Korea) were housed in a temperature- (21°C ± 2°C) and humidity- (65% ± 5%) controlled room in which a 12-hour light/12-hour dark cycle was maintained. The animals were allowed free access to food pellets and water and were acclimatized for 1 week prior to usage. Animal experimentation was performed in accordance with the National Institutes of Health (NIH) Guidelines for Use of Laboratory Animals.
Experimental Protocol
Animals were randomly divided into 4 groups, each containing 10 animals. Group 1 served as the control and received saline (0.9% NaCl w/v) by oral gavage daily for 5 weeks. Group 2 received ethanol (6 g/kg body weight and 15% v/v in saline) daily orally by gavage for 5 weeks. Group 3 received ethanol as in group 2 but were also coadministered with red ginseng extract (2 g/kg body weight). Group 4 received red ginseng wine (6 g/kg body weight), which was fermented by S bayanus (ethanol content 15.0%).
Body and Organ Weights
Body weight and food and water consumption were measured daily. At the end of the 5-week treatment period, all animals were anesthetized with ether and blood was collected for hormone analysis. The mice were then sacrificed by cervical vertebral dislocation and the testes, epididymis, seminal vesicles, brain, liver, kidneys, and thymus were removed, freed from adherent tissues, and weighed.
Sperm Analysis
Epididymal sperm motility was evaluated using methods previously described. 25 Briefly, sperm motility was assessed by recovering sperm from excised caudal epididymis and allowing the sample to capacitate for 5 minutes at 37°C in M199 media containing 0.5% bovine serum albumin. The suspension was placed by micropipette on a 2X-CEL slide glass (depth: 80 μm, thickness: 0.15 mm, Hamilton Thorne, Massachusetts) warmed to 37°C. Sperm motility was measured using a CASA with IVOS-TOX Sperm Analyzer software (Hanilton Thorne Biosciences, Massachusetts).
Hormone Analysis
Blood for sex hormone analysis was collected from the abdominal aorta on the next day after the last injection. Serum was separated by centrifugation at 3500 rpm for 15 minutes at room temperature and stored at −20°C. Serum testosterone, LH, and FSH were measured by ELISA at 450 nm with a microwell reader (Promega, Madison, Wisconsin).
Statistical Analysis
All values are expressed as mean ± standard error of the mean (SEM). Data were analyzed using 1-way analysis of variance (ANOVA) with Dunnett correction for pairwise comparisons. Statistical significance was assigned if P < .05.
Results
Ginsenoside Content of Red Ginseng Extract
Ginsenosides are a class of triterpene saponins and consist of a dammarane skeleton with various sugar moieties attached to the C-3 and C-20 position. More than 30 ginsenosides have been identified and split into 2 groups: (1) the 20(S)-protopanaxadiol ([PPD]; Rb1, Rb2, Rb3, Rc, Rd, Rg3, Rh2, and Rs1) and (2) the 20(S)-protopanaxatriol ([PPT]; Re, Rf, Rg1, Rg2, and Rh1). 26,27 Ginsenosides are named as “Rx,” where “R” signifies the root and “x” stands for the chromatographic polarity in an alphabetical order. 28
The ginsenoside standards Rb1, Rb2, Rc, Rd, Rf, Rg1, Rg3, Rh2, and compound K were purchased from Ginseng Genetic Resource Bank (Yongin, Gyeonggi, Korea). To create calibration curves, each ginsenoside standard was dissolved individually in HPLC-grade methanol and serially diluted to the appropriate concentration. Analytical data were shown in Table 1. The contents of the 4 major ginsenosides in the red ginseng extract were Rb1 (1.63 mg/g), Rb2 (1.09 mg/g), Rc (1.60 mg/g), and Rd (0.43 mg/g; Table 1). In the red ginseng wine, the contents of ginsenoside Rg3 and Rf were 1.91 mg/g and 0.58 mg/g, respectively; while the contents of ginsenoside Rb1, Rb2, and Rc were decreased to 0.67 mg/g, 0.76 mg/g, and 1.34 mg/g, respectively (Table 1).
Ginsenoside Content in the Red Ginseng Extract and Ginseng Wine Determined by HPLC (mg/g) a
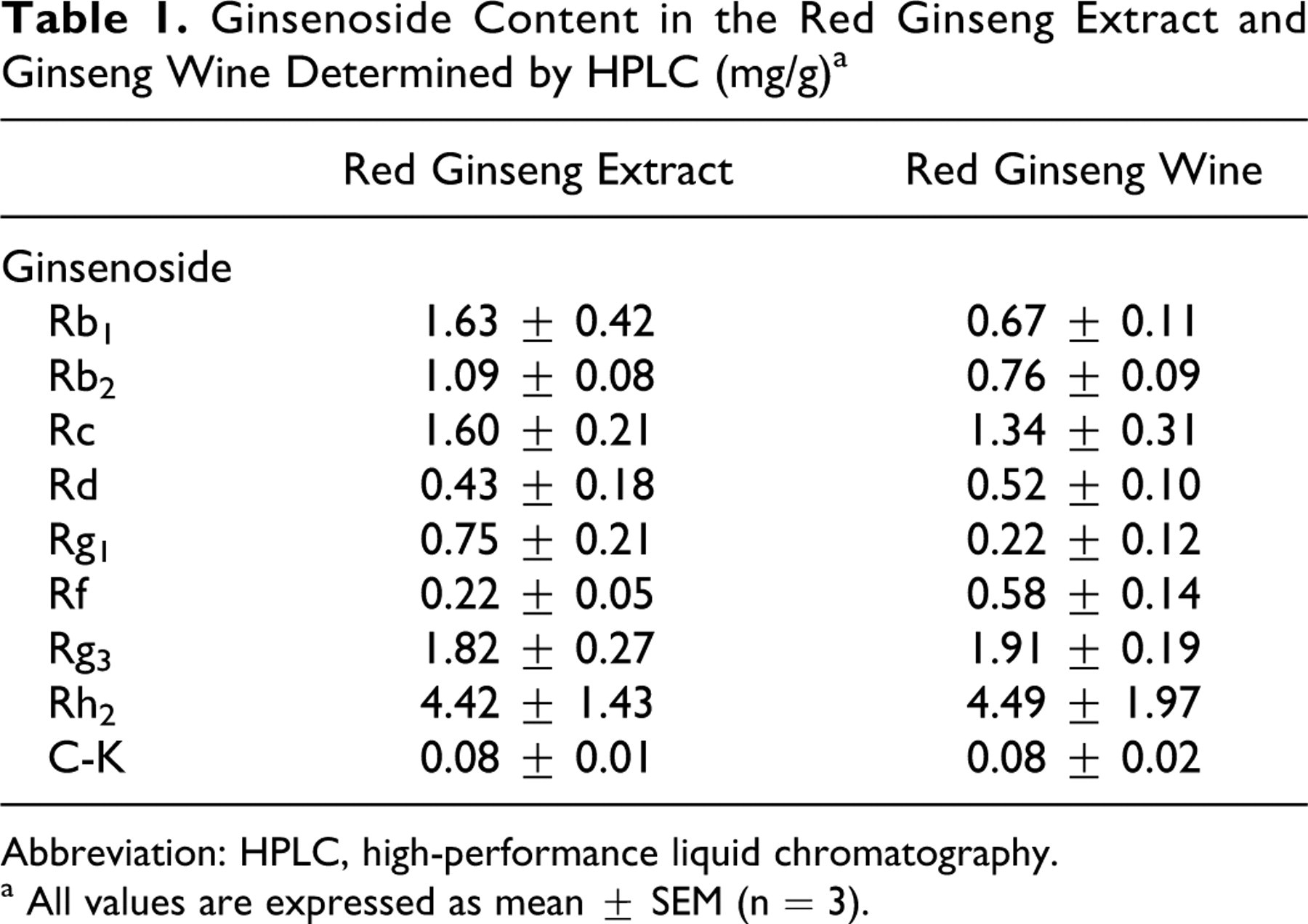
Abbreviation: HPLC, high-performance liquid chromatography.
a All values are expressed as mean ± SEM (n = 3).
Body and Reproductive Organ Weights
There were no significant changes observed in body weight or differences in food and water intake during the experiment (Table 2).
Average Body Weight and Average Daily Food and Water Consumption a
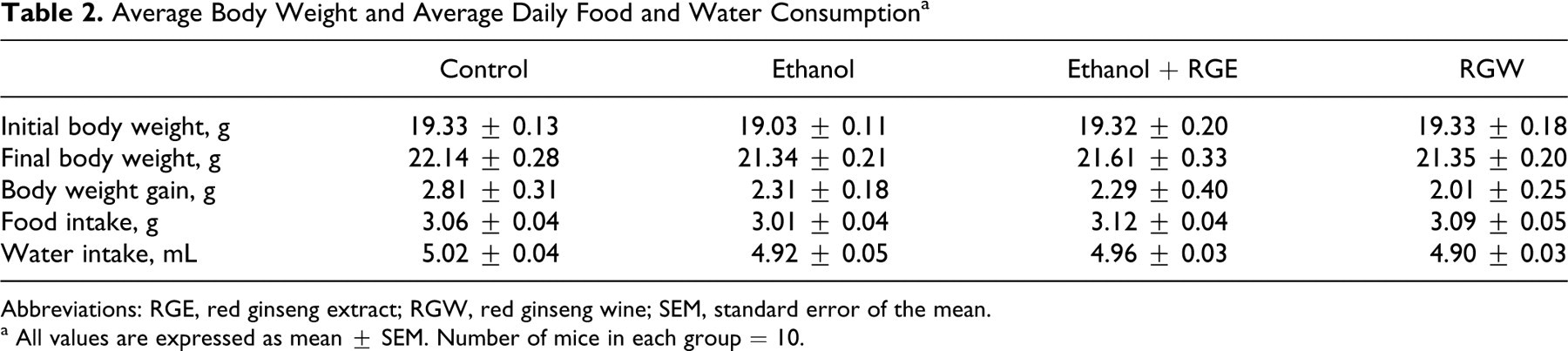
Abbreviations: RGE, red ginseng extract; RGW, red ginseng wine; SEM, standard error of the mean.
a All values are expressed as mean ± SEM. Number of mice in each group = 10.
Although not statistically significant, the absolute and relative weights of the testis and epididymis of ethanol administered mice were lower than those of control animals. There was a statistically significant decrease in seminal vesicle and thymus weights (P < .05) in the ethanol group compared to control and ginseng-treated mice (Table 3). There was no significant difference in the absolute or relative weights of the testis and epididymis between control and ginseng-treated mice (P < .05; Table 3).
Absolute and Relative Organ Weights of Mice a
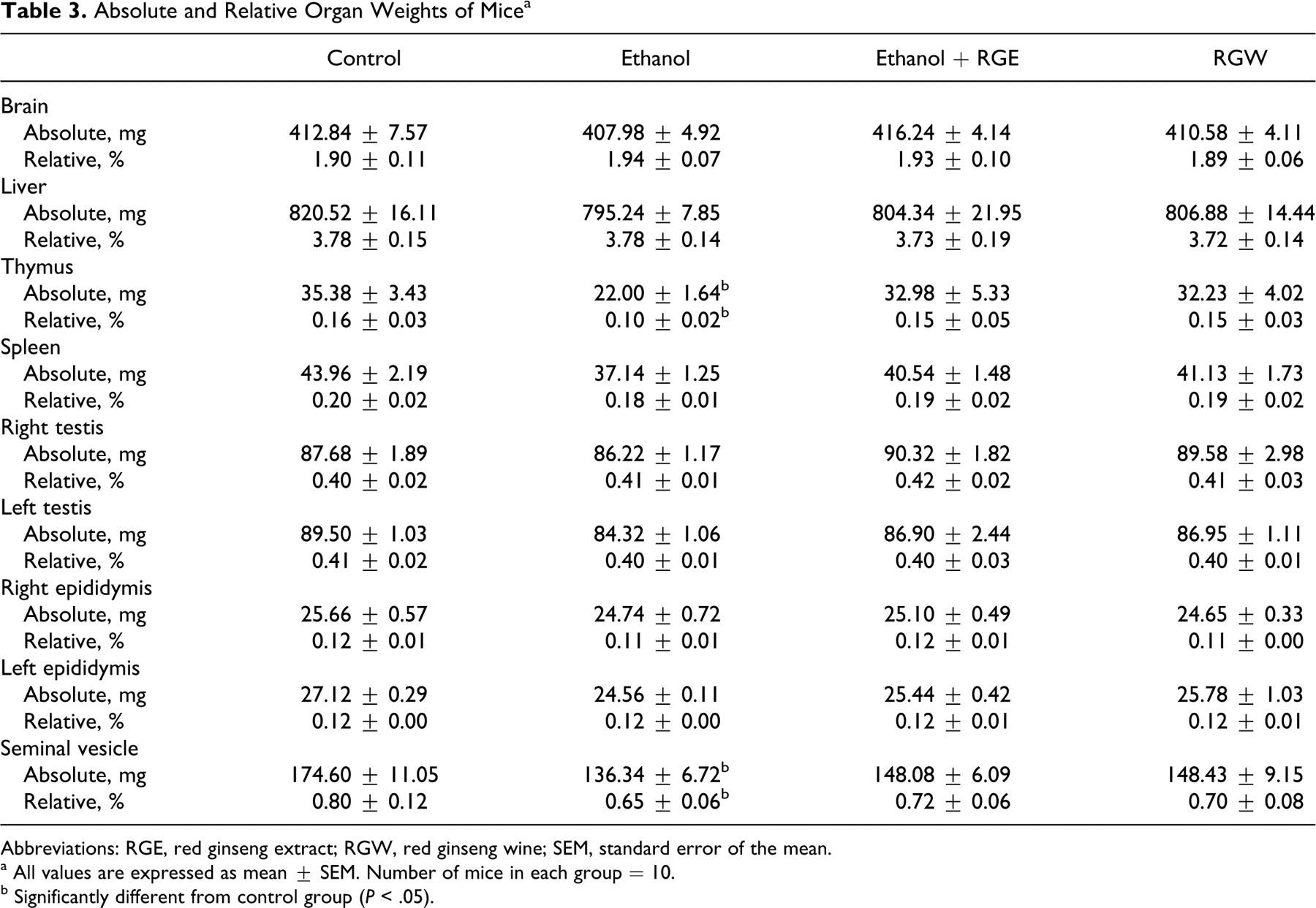
Abbreviations: RGE, red ginseng extract; RGW, red ginseng wine; SEM, standard error of the mean.
a All values are expressed as mean ± SEM. Number of mice in each group = 10.
b Significantly different from control group (P < .05).
Effect on Sperm Motility
As shown in Figure 1, the sperm motility ratio in the ethanol, ethanol plus red ginseng extract, and red ginseng wine groups were 59.49% ± 0.87%, 66.40% ± 3.71%, and 67.80% ± 3.17%, respectively. Sperm motility was significantly decreased in the ethanol group compared to the control group (P < .05). There was no difference in sperm motility between ginseng-treated mice and controls. Interestingly, the decreased sperm motility observed in ethanol-treated mice was absent in mice treated with either red ginseng extract or ginseng wine (Figure 1).
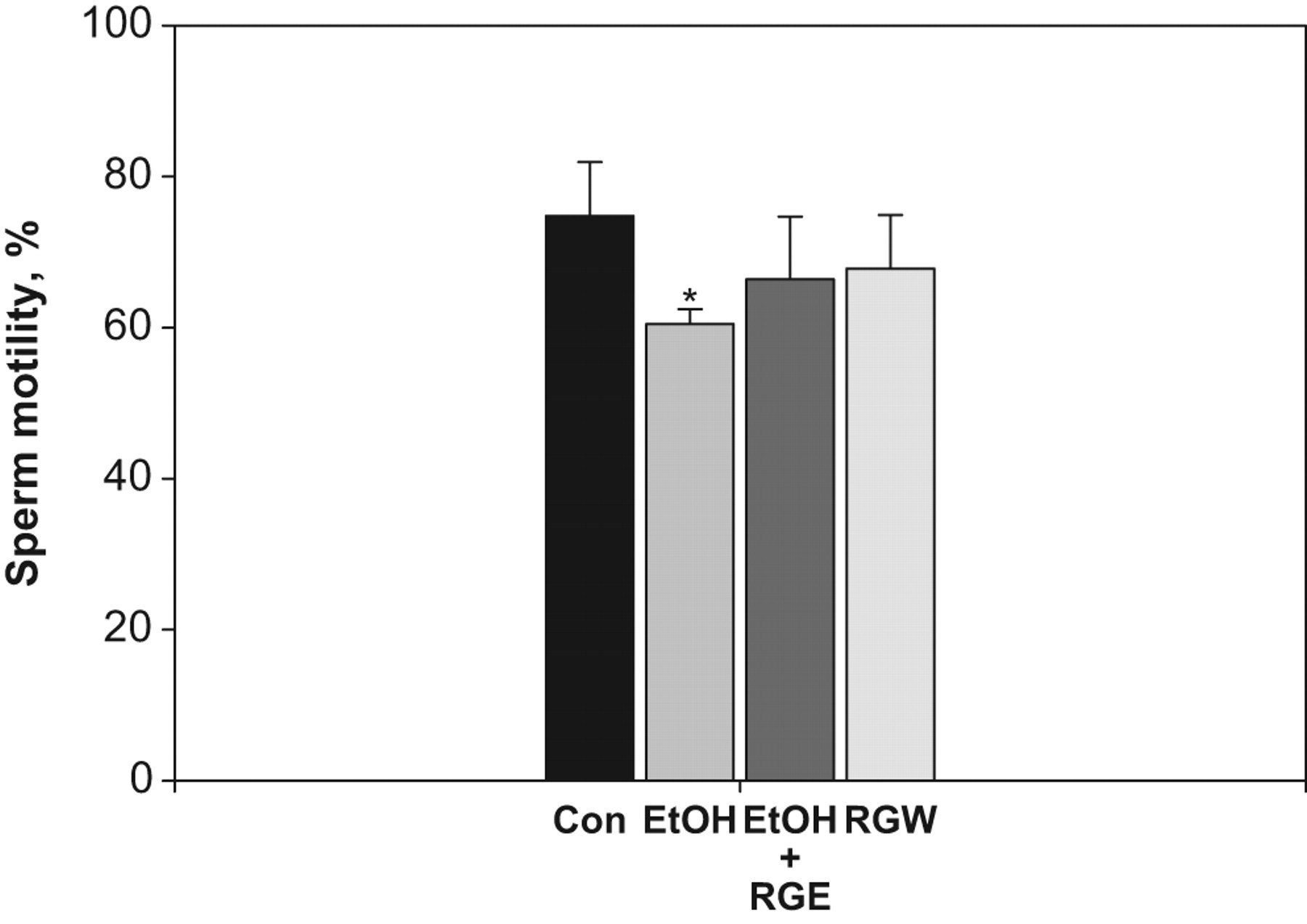
Sperm motility in control and treatment groups. RGE indicates red ginseng extract; RGW, red ginseng wine. All values are expressed as mean ± SEM. *Significantly different from control group (P < .05).
In evaluating motility with most species, sperms are divided into nonmotile, progressively motile or nonprogressively motile. A progressively motile sperm swims forward in an essentially straight line, whereas a nonprogressively motile sperm swims but with an abnormal path, such as in tight circles. The ratios of sperm progressiveness were 11.40% ± 0.51%, 15.40% ± 0.51%, and 15.80% ± 2.13% in ethanol, ethanol plus red ginseng extract, and ginseng wine groups, respectively (Figure 2). The effects of ethanol treatment on sperm progressiveness followed a similar pattern. Ethanol-treated mice had lower sperm progressiveness than controls, but mice treated with ethanol plus red ginseng extract showed no reduction in sperm progressiveness (Figure 2).
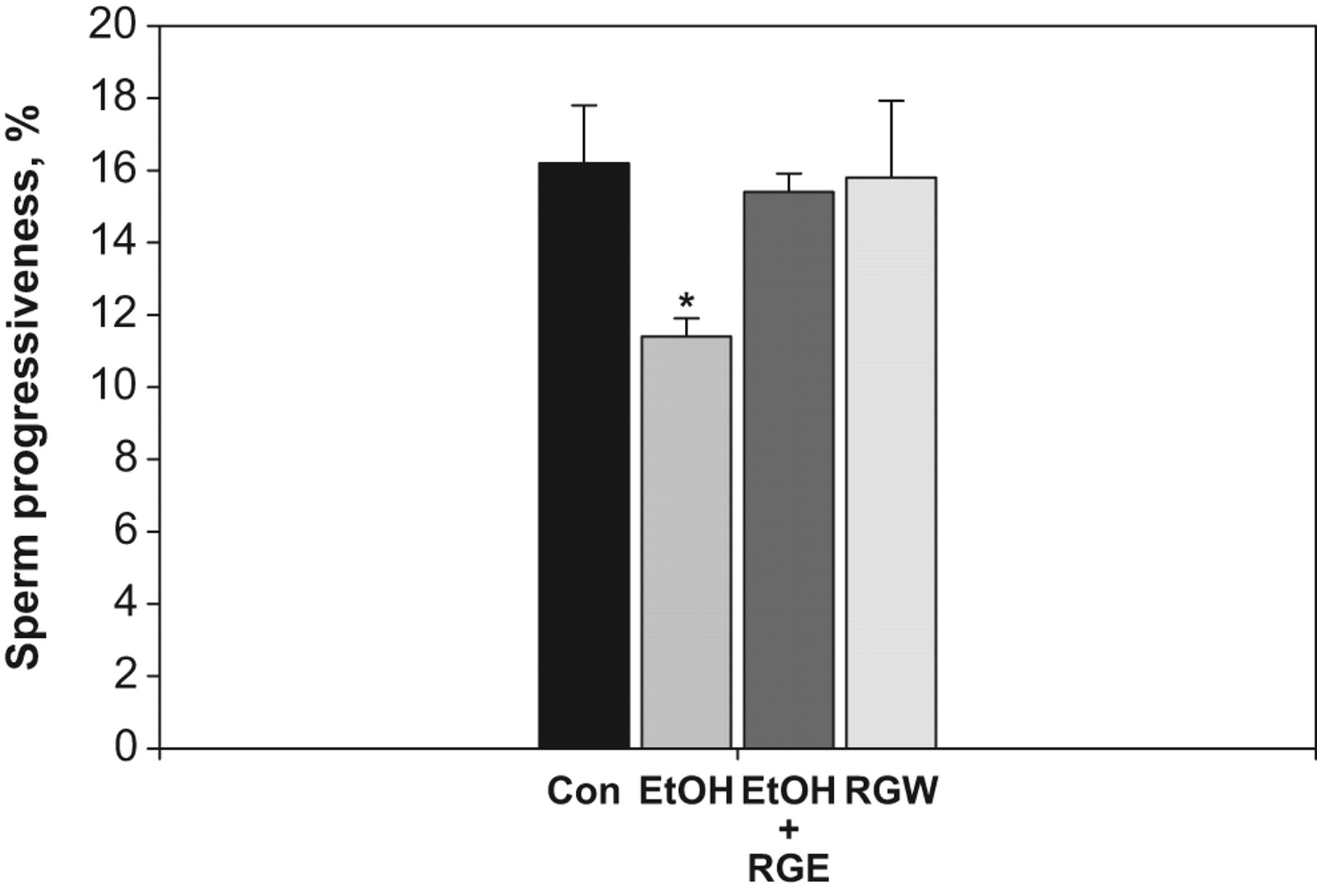
Sperm progressiveness in control and treatment groups. RGE indicates red ginseng extract; RGW, red ginseng wine; FSH, follicle-stimulating hormone; LH, luteinizing hormone; SEM, standard error of the mean. All values are expressed as mean ± SEM. *Significantly different from control group (P < .05). A, Testosterone. B,LH. and C, FSH.
We measured the following sperm motility end points: path velocity (VAP), progressive velocity (VSL), track speed (VCL), amplitude of lateral head displacement (ALH), straightness (STR), and linearity (LIN). Path velocity equals the total distance along the smoothed average position of sperm divided by the time elapsed. Progressive velocity equals the straight-line distance from the beginning to the end of the sperm track divided by the time elapsed. 29 As a whole, sperm quality decreased in ethanol-treated mice. Average path velocity and straight-line velocity (VSA) were significantly lower in the ethanol group compared to the control and ginseng-treated groups (P < .05; Table 4). Thus, parameters associated with sperm motility seemed to improve significantly by the administration of red ginseng extract (P < .05). There was no significant difference in sperm motility-related parameters between red ginseng extract and ginseng wine groups (Table 4).
Mean Sperm Motility Parameters of Mice a
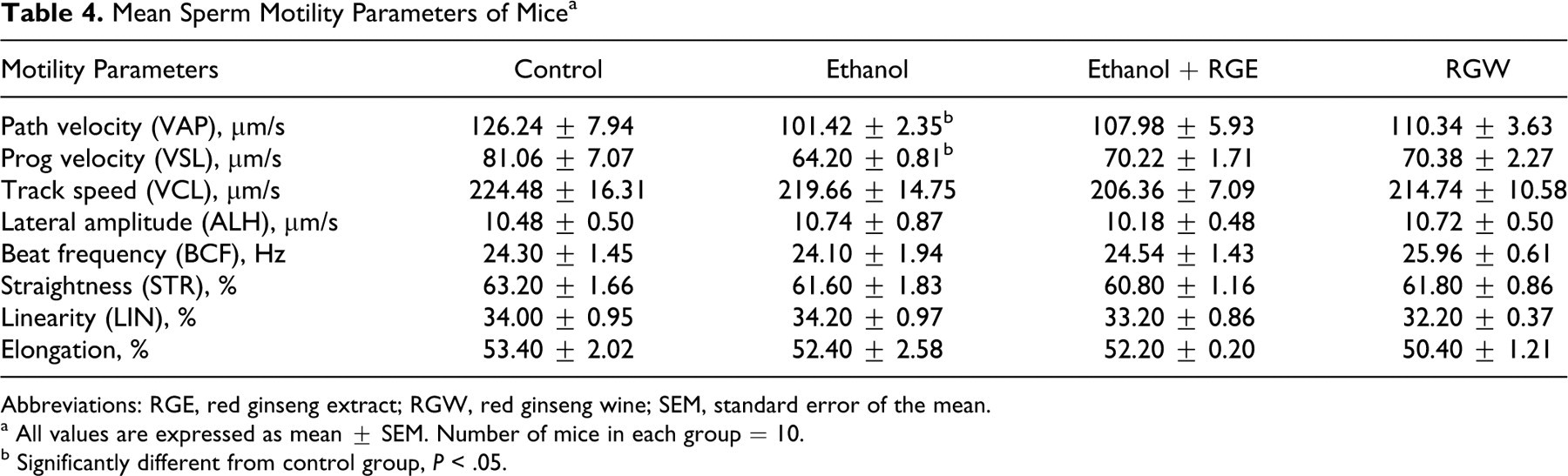
Abbreviations: RGE, red ginseng extract; RGW, red ginseng wine; SEM, standard error of the mean.
a All values are expressed as mean ± SEM. Number of mice in each group = 10.
b Significantly different from control group, P < .05.
Hormone Analysis
Serum levels of testosterone, LH, and FSH are listed in Figure 3. No statistically significant differences in sex hormone levels were observed in any group (Figure 3). Although not statistically significant, the average serum testosterone tended to be lower in the ethanol group compared to the control group and higher in the ethanol plus ginseng group compared to controls (P < .05;Figure 3A). In addition, the average serum LH tended to be lower in the ethanol group compared to the control group and higher in the ethanol plus ginseng group compared to controls, but no statistically significant difference was found (P < .05; Figure 3B). Although not statistically significant, the average serum FSH tended to be lower in the ethanol group compared to the control group (P < .05; Figure 3C).
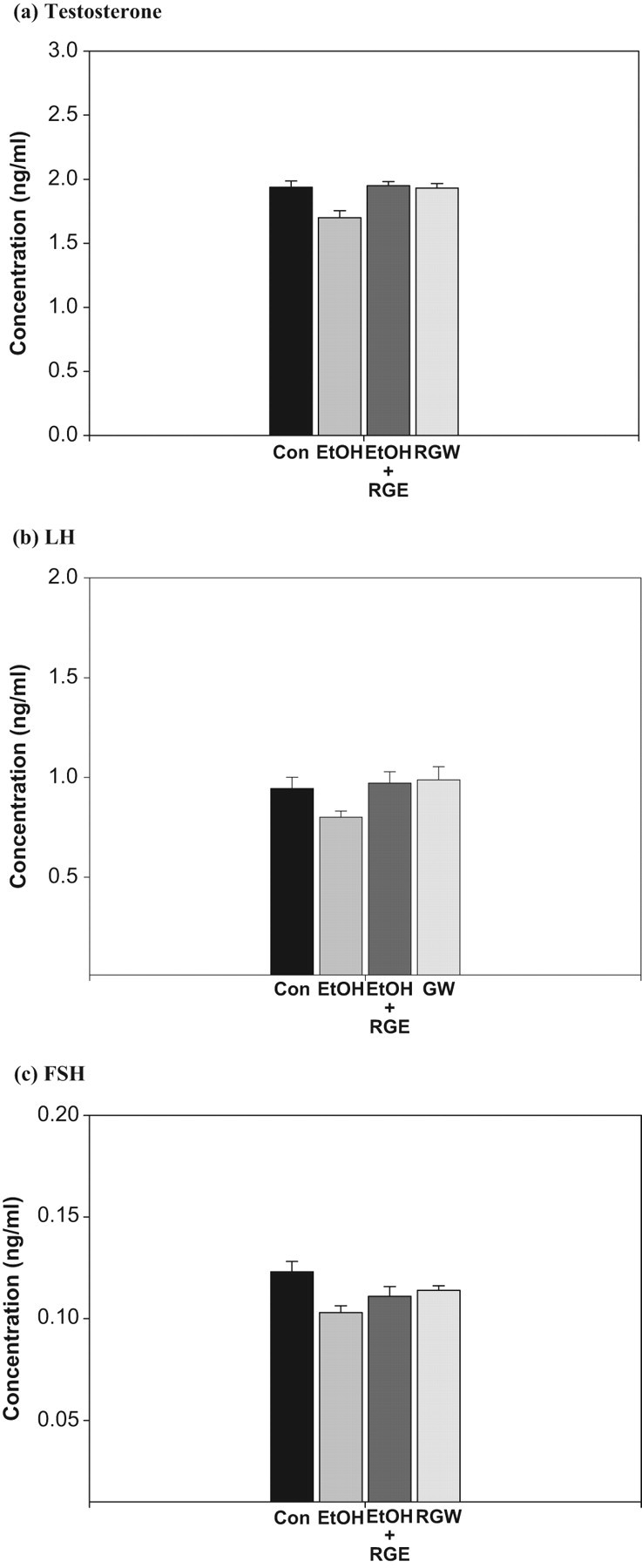
Mean serum testosterone, luteinizing hormone (LH), and follicle stimulating hormone (FSH). RGE indicates red ginseng extract; RGW, red ginseng wine; SEM, standard error of the mean. All values are expressed as mean ± SEM. *Significantly different from control group (P < .05).
Discussion
In several studies, ethanol has been used to produce reproductive disturbances. 30,31 The effects of ethanol on the reproductive system include reduction of reproductive organ weight and reduced caudal epididymal sperm content and sperm motility. 8 –10 However, ginseng saponins, commonly known as ginsenosides, have been shown to increase human sperm motility in vitro. 23,24 In the present study, the protective effects of ginseng saponins on the reproductive system in male mice were evaluated. Body weight measurement provided is a good indicator for the overall animal health status and may be relevant to the interpretation of reproductive effects. According to measurements of body weight and food and water intake during the experiment, we concluded that ethanol, red ginseng extract, and ginseng wine had no effect on the appetites of mice in this study.
It has been strongly suggested that ethanol affects both absolute and relative organ weights (organ-weight-to-body-weight ratio). 29 In the present study, ethanol treatment caused no significant reduction in the absolute or relative weights of the testis, epididymis, or seminal vesicle compared to controls. There was a trend toward increased absolute and relative weights of these reproductive organs in mice treated with ethanol plus ginseng extract or ginseng wine, albeit this difference did not reach statistical significance. There was a trend toward a decrease in the weights of both the left and right epididymis in the ethanol group compared to controls. Epididymal and caudal epididymal weights are important in reproductive studies in which sperm parameters are evaluated, as reductions in sperm production can alter the epididymal weight. 32 The seminal vesicle is androgen dependent, thus weight changes may reflect alterations in endocrine status or testicular function. 32 Any decrease in reproductive organ weight may be due to decreased testosterone from ethanol’s oxidative damage to the brain, testis, and liver. Ingested ethanol is distributed widely to these organs and metabolized by the liver, producing toxic effects. 33 Panax ginseng C. A. Meyer saponins normalize the weight of the testis, epididymis, and seminal vesicle and are positively correlated with increased serum testosterone. 34 Increased serum testosterone may be due to a direct effect on the central nervous system and gonadal tissues or an indirect effect via the hypothalamus–pituitary–testis axis. 35
To evaluate sperm quality, we assessed the motility of sperm recovered from the excised caudal epididymis and found that sperm motility was significantly lower in the ethanol group than the control group. Previous studies have reported a similar decrease in average sperm velocity in ethanol-treated mice. Ethanol may alter epididymal secretory products or directly affect sperm motility and morphology. 30 In ethanol-treated rats, impaired sperm motility is indicative of a defect in the acquisition or maintenance of motility. 36 In alcoholic humans, impaired forward motility of spermatozoa has been observed. 37 In our study, sperm motility was higher in the ethanol plus red ginseng extract and red ginseng wine groups than the ethanol group (P < .05), suggesting that red ginseng extract prevents ethanol-induced reduction in sperm motility. In another study, treatment of Sprague-Dawley rats with oral Korean P ginseng extract for 28 days led to improved reproductive function, including improved sperm motility and morphology. 38 It has also been reported that ginseng enhanced sperm motility and progression in vitro. 23 Sperm motility is a reproductive end point and an integral part of certain reproductive toxicity tests. In addition, sperm motility is related to fertility in humans. 39
In our study, VAP and VSL were significantly lower in the ethanol group compared to the control group (P < .05), while VAP and VSL were higher in the ethanol plus ginseng and ginseng wine groups compared to the ethanol group. There were no significant differences in VCL, ALH, STR, or LIN between groups. Korean red ginseng has traditionally been used as a tonic to improve physical strength in China, Japan, and Korea. Modern scientific research indicates that Korean red ginseng plays beneficial roles in sexual, endocrine, immune, and reproductive dysfunction. 40 –42
In this study, although there were no significant differences in sex hormone levels between groups, serum testosterone was lower in the ethanol group than the control group and higher in the ethanol plus ginseng group. Testosterone is essential to male accessory sex gland function and maintains their structure. The absence of testosterone impedes spermatogenesis. 43 We found that serum LH tended to be lower in the ethanol group compared to the control group, while red ginseng extract appeared to prevent the ethanol-induced decrease in LH and testosterone levels. Also, serum FSH levels were insignificantly lower in the ethanol group than the control group. Ethanol administration has shown to reduce the LH content of the anterior pituitary, 13 suggesting that the decrease in serum LH after chronic ethanol administration is a result of decreased synthesis. 44 Although the reduction of LH synthesis and secretion may be due to a direct effect on gonadotropins, several reports suggest that ethanol acts at the hypothalamic level rather than on the pituitary or the gonad to suppress male reproductive activity. 45 In humans, consumption of P ginseng C. A. Meyer extract has been associated with increased sperm count, motility, and testosterone level in patients with fertility problems. 34
In conclusion, our study found that ethanol significantly impaired sperm motility but did not significantly reduce reproductive organ weight or serum testosterone, LH, or FSH. Administration of red ginseng extract with ethanol appeared to partially prevent the toxic effect of ethanol on male reproductive function. It is traditionally believed that red ginseng improves male reproductive function and can prevent or treat male infertility. Additional studies are needed to explore the mechanism of red ginseng extract on spermatogenesis and clarify the biological effects of P ginseng and its myriad molecular compounds, each of which may have unique effects on male reproductive function.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by KGCMVP (Korean Ginseng Center for Most Valuable Products & Ginseng) for Technology Development Program of Agriculture and Forestry, Ministry for Food, Agriculture, Forestry and Fisheries, Republic Korea. [grant number 607002-5].
